Abstract
Objective:
Cerium nitrate (CeN) plus silver sulfadiazine (SSD) cream has been used for 40-plus years to manage burns. CeN produces a hardened eschar believed to resist bacterial colonization/infection. To evaluate this potential mechanism, we treated in vitro skin models or Pseudomonas aeruginosa with CeN and measured mechanical properties of the models and bacterial virulence, respectively.
Approach:
We treated three-dimensional-collagen matrix and ex-vivo–burned porcine skin with CeN and evaluated stiffness and P. aeruginosa penetration. In addition, we treated P. aeruginosa with CeN and evaluated the bacteria's motility, skin model penetration, susceptibility to be phagocytized by the human monocytic cell line THP-1, and ability to stimulate this cell line to produce cytokines.
Results:
CeN treatment of skin models stiffened them and made them resistant to P. aeruginosa penetration. Inversely, CeN treatment of P. aeruginosa reduced their motility, penetration through skin models (ex-vivo–burned porcine skin), and ability to stimulate cytokine production (tumor necrosis factor-α [TNF-α] and interleukin 8 [IL-8]) by THP-1 cells. In addition, CeN-treated Pseudomonas was more readily phagocytized by THP-1 cells. Finally, P. aeruginosa inoculated on CeN-treated ex-vivo–burned porcine skin was more susceptible to killing by a silver dressing.
Innovation:
In vitro skin models offer a platform for screening drugs that interfere with bacterial penetration into wounded tissue.
Conclusion:
CeN treatment reduced P. aeruginosa virulence, altered the mechanical properties of ex-vivo–burned porcine skin and collagen matrix, retarded penetration of P. aeruginosa through the skin models, and resulted in increased vulnerability of P. aeruginosa to killing by antimicrobial wound dressings. These data support the use of CeN in burn management.
Kai P. Leung, PhD

INTRODUCTION
Severe burn injuries pose a significant problem worldwide, disproportionately affecting military populations (combat or noncombat operations). 1 Burns disrupt the skin barrier, local blood supply, 2 and immune function, 3 making them susceptible to colonization and infection by endogenous normal skin flora and exogenous bacteria. Infections of burn wounds can delay healing and contribute to hypertrophic scarring, nonhealing wounds, sepsis, multiorgan failure, and mortality. 4
Infection is the leading cause of mortality responsible for up to 50% of burn related deaths, 5 and ∼75% of deaths in patients with 40% total body surface area burn. 6 A frequently encountered pathogen infecting burn wounds is Pseudomonas aeruginosa, a highly invasive species. 7,8 Tribble et al (2016) showed that P. aeruginosa is the most common gram-negative bacteria isolated from the combat-related injuries in wounded military personnel in the Afghanistan Operation Theater. 9
To reduce the risk of burn infection, the standard care includes topical antimicrobials (mafenide acetate, silver sulfadiazine [SSD], chlorhexidine, and other combinatorial drugs) and early excision of the burn eschar and skin autografting. However, eschar excision and grafting requires a sterile operation room, highly skilled surgeons, equipment, and available donor skin or other coverage material and is only practiced under specialized hospital settings 10,11 that are not available on the battlefield or in resource-limited regions of the world. 12,13 For such environments, wound care products are needed that can temporize burn wounds, such as by stabilizing the eschar, reducing its toxicity, and mitigating microbial colonization and infection. In hospital settings, one product that has been used for these purposes for 40-plus years is the Flammacerium® cream containing cerium nitrate (CeN, 2.2 wt %) and SSD (1.0 wt %).
CeN has broad spectrum antibacterial activity. Burkes and McCleskey performed a systematic investigation of the bacteriostatic properties of salts of cerium, lanthanum, and thallium against a panel of 39 bacterial species across 16 genera, including gram-positive Staphylococcus aureus and gram-negative P. aeruginosa. Using radial streak inoculations (eight cultures to each plate) on petri plates of solidified agar containing the specified amount of the salts and basal media, they found that CeN was an effective bacteriostatic agent against the whole spectrum of bacteria. Pseudomonas species were the most susceptible genera, with inhibition of growth at concentrations 0.1–0.4 mM of CeN followed by Escherichia and Salmonella species at 0.5 mM. However, almost twice that concentration is effective on S. aureus. 14
Marone et al evaluated the antimicrobial activity of SSD, alone and in combination with CeN, gentamicin, and amikacin against 130 clinical isolates, including multidrug resistant bacteria such as P. aeruginosa or methicillin-resistant S. aureus (MRSA). The results showed that the combination of SSD and CeN was as effective as SSD alone. 15,16
In these early studies CeN has demonstrated broad but greater antimicrobial activity against gram-negative bacteria at concentrations of 10–40 mM, which led to the use of CeN combined with SSD to complement SSD's gram-positive antimicrobial activity. 17,18 However, subsequent testing of the complementarity of these agents in vitro produced conflicting results, 19 possibly due to cerium precipitating as cerium phosphate or cerium binding to proteins, as well as SSD having poor solubility giving erroneous results in agar disc diffusion tests or broth assays. 17,20,21 Nonetheless, topical application of CeN-SSD on patient's burns has been associated with decreased bacterial contamination and P. aeruginosa infection. 18,19,22 –24
Alongside CeN's inherent antimicrobial activity, in treating human burn wounds CeN in CeN-SSD produces an eschar that adheres well for longer periods to the underlying tissue and may resist bacterial colonization and invasion. 20,22,25 Thus, CeN's effects both on the eschar and bacteria may mitigate bacterial colonization and infection of burn wounds. 26,27 However, little experimental evidence for these mechanisms is available. In this study, we independently treated P. aeruginosa and tissue models (three-dimensional [3D]-collagen matrix and ex-vivo–burned porcine skin).
After treating the in vitro models with CeN, we evaluated their stiffness of ex-vivo–burned porcine skin. In addition, after treating the bacteria, we evaluated its survival, motility, and ability to be phagocytized by the human monocytic cell line THP-1 and to stimulate this cell line to produce pro-inflammatory cytokines. Furthermore, the ex-vivo–burned porcine skin treated with CeN was tested for P. aeruginosa penetration and the sensitivity of bacteria inoculated on it to be killed by a silver dressing. Our results show that CeN acted on P. aeruginosa and burn tissue by stiffening the skin models and by affecting the motility of P. aeruginosa, resulting in lesser penetration through the skin models (Fig. 1). Furthermore, CeN increased the susceptibility of P. aeruginosa to phagocytosis by the THP-1 cells and to the killing by antimicrobial agents such as SSD or Ag.
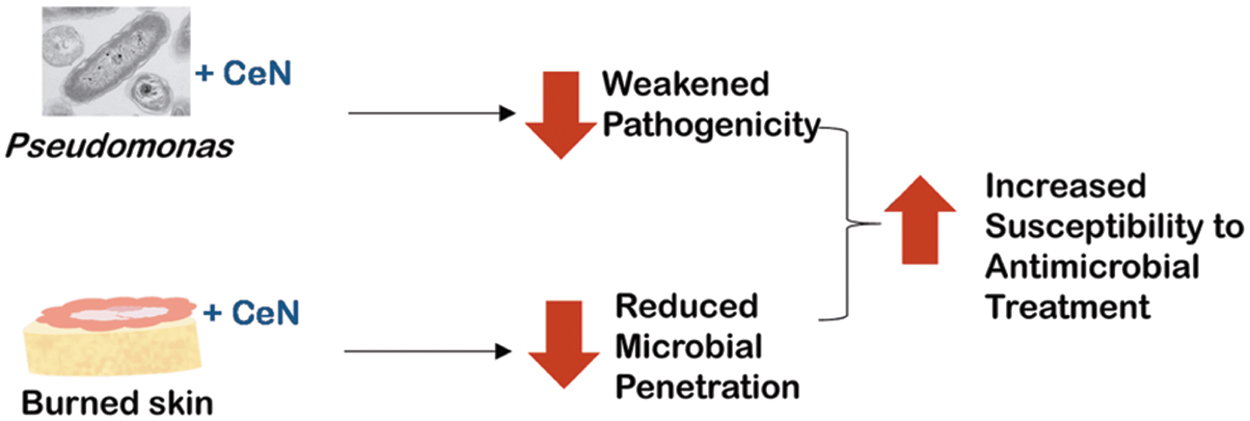
Diagram: Summarizing potential effects of CeN on Pseudomonas aeruginosa and on burned skin model leading to reduced virulence, reduced skin penetration, and increased vulnerability to antimicrobial treatment. CeN, cerium nitrate.
CLINICAL PROBLEM ADDRESSED
CeN-SSD treatment enhances the adherence of the eschar to underlying tissue while producing a dried wound that resists infection and facilitates postponement of surgical eschar excision and grafting. Additional benefits reported for CeN-SSD treatment are fewer dressing changes, less soak-through of exudate, less labor-intensive care, and greater comfort for patients. 20,24,28,29 In this study, in in vitro skin models we show that CeN stiffens burn tissue and makes it more resistant to Pseudomonas penetration. In addition, CeN treated-P. aeruginosa was less virulent and more susceptible to antimicrobials. This evidence supports the use of CeN in the management of burn wounds.
MATERIALS AND METHODS
Bacterial strains, cell line, and culture conditions
Three P. aeruginosa strains were used in this study: strain PAO1 (ATCC, Manassas, VA), PAO1 transformed with a red-fluorescent expression plasmid pMT RFP (provided by Dr. Seok Jong Hong, Northwestern Medical School), and a clinical strain 1244. 30 All strains were grown in tryptic soy broth (TSB) and grown overnight or to mid-log growth phase (OD600nm = ∼0.1–0.4).
For phagocytosis and inflammatory response assays, THP-1 cell suspensions (ATCC) were used. The cells were propagated for four generations before storing in a −150°C freezer. For each experiment, THP-1 cells were freshly thawed, washed once with Hank's balanced salt solution (with phenol red but without Ca2+ and Mg2+; Hank's balanced salt solution [HBSS]; Gibco-Life Technologies, Carlsbad, CA), and propagated in media containing 45% RPMI 1640 (ATCC), 45% HBSS, and 10% fetal bovine serum (FBS; Gibco-Life Technologies) for a maximum five passages in TC- Flask T75 Standard, Vented Cap (Ref. 83.3911.002; SARSTEDT). The THP-1 cells were grown to a density of ∼106 per mL at 5% CO2 and 37°C, and the treatment assays were performed in the 5 mL polypropylene round-bottom tubes (Ref. 352063; Corning Sciences). CeN [Cerium(III) nitrate hexahydrate, 99.5%, CAS No. 10294-41-4] was obtained from Arcos.
Preparation of 3D-collagen gels and measurements of mechanical/physical changes
Collagen gels were prepared as per the manufacturer's instructions with the pregelation procedure carried out on ice with cold-incubated pipette tips. Briefly, collagen (Collagen I heavy chain [HC], rat tail −0.02 N in Acetic Acid; BD Biosciences, Bedford, MA) was mixed with HBSS (10% v/v without Ca++ or Mg++), neutralized with sodium bicarbonate, and equilibrated to 5 mg/mL collagen in HBSS. Gelation was carried out at room temperature for 30 min, and the gels were further incubated at 37°C for a minimum of 1 h.
For measuring the physical property change, 280 μL collagen gels were incubated with 280 μL of 0 or 1 mM CeN for 30 min. After incubation, CeN solution was removed, and the gels were blot dried and incubated for 2 days at 37°C before the measurements. Oscillatory amplitude sweep with 1 to 100 Pa at an angular frequency of 1 Hz, as described by Evani et al, 31 was used to calculate viscoelastic properties of the gels using the HAAKE MARS modular advanced rheometer (ThermoFisher Scientific, Inc.) equipped with parallel plate-PP20. Storage modulus in linear viscoelastic range was measured as an indicator stiffness (Fig. 2).
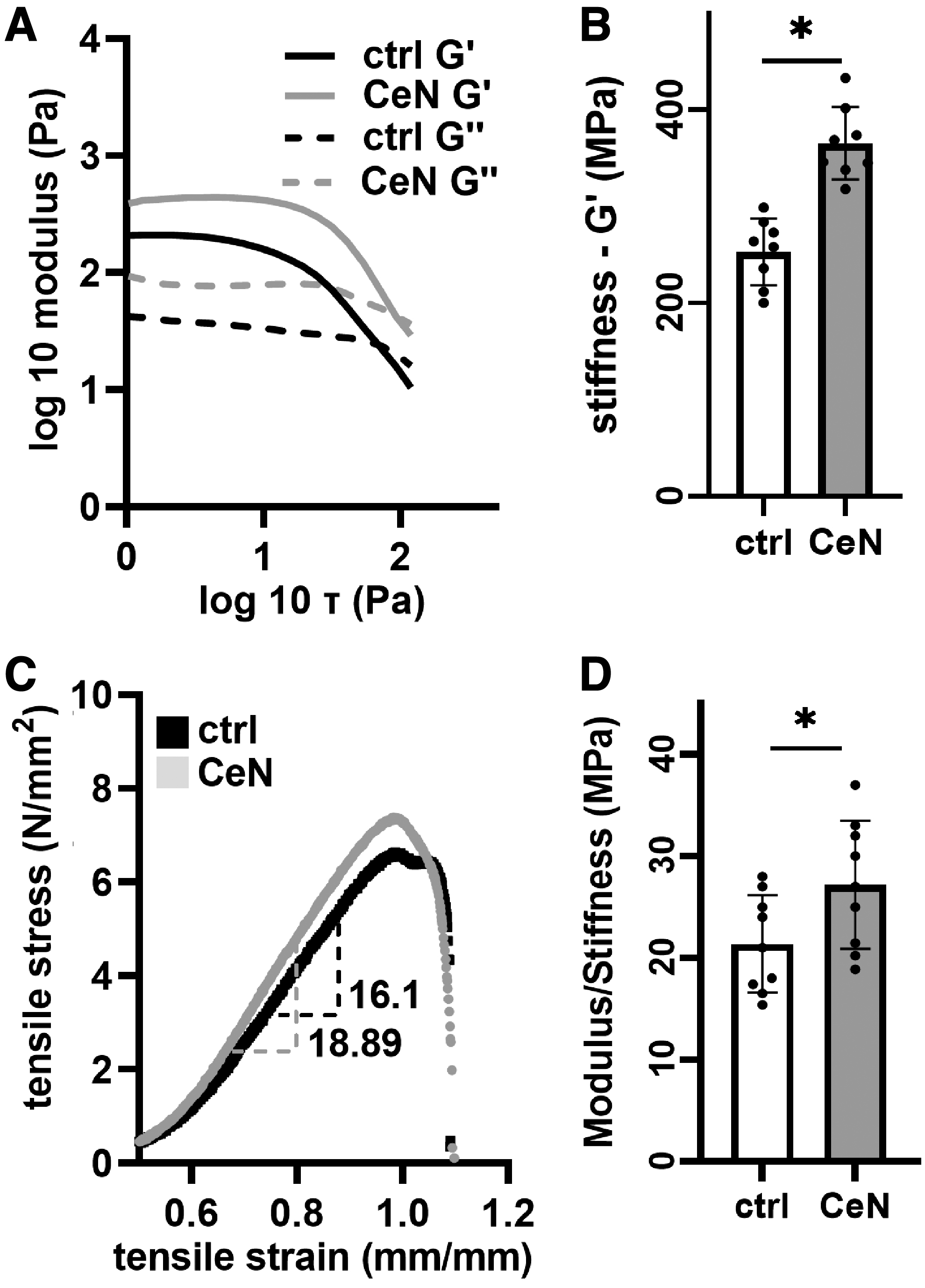
CeN altered mechanical properties of 3D-collagen gel and ex-vivo–burned porcine skin: Collagen gels were treated with 1 mM CeN or water control (ctrl) for 30 min followed by a 48-h incubation at 37°C without treatment. Viscoelastic properties were measured by performing amplitude sweep at 1 Hz using a rheometer.
P. aeruginosa PAO1 penetration of 3D-collagen gel
To visualize P. aeruginosa PAO1 penetration, green fluorescent-labeled collagen (Collagen Type I-FITC Conjugate from bovine skin, ∼1 mg/mL solution, Cat. No. C4361; Sigma, St. Louis, MO) was used. Collagen gels were prepared as described above. Briefly, green fluorescent-labeled collagen was added at 10% v/v before neutralization. Next, 120 μL of this collagen was dispensed into the wells of eight-well cover-glass chamber slides (LabTek-1; ThermoFisher Scientific Inc.). The preformed gels were then incubated with 120 μL of CeN solution (0 or 1 mM) for 30 min. The gels were removed from the CeN solution and blot dried before incubation for 2 days at 37°C in humidified petri dishes.
Red-fluorescent-tagged PAO1 (mid-log-phase, ∼107 colony-forming unit [CFU] in 10 μL) was added to the top of the gel and their penetration into it was measured after 2 and 16 h by taking Z-stack images (Zeiss 710-live imaging confocal microscope, Thornwood, NY) at a magnification of 10 × and at the same laser power, gain, and digital offset (Fig. 3A). The images were analyzed over time for changes in red fluorescence intensity from the top to the bottom of the Z stack (Carl-Zeiss Zen software). The fluorescence at 16 h as a ratio of the 2 h fluorescence was plotted as a measure of bacterial penetration.
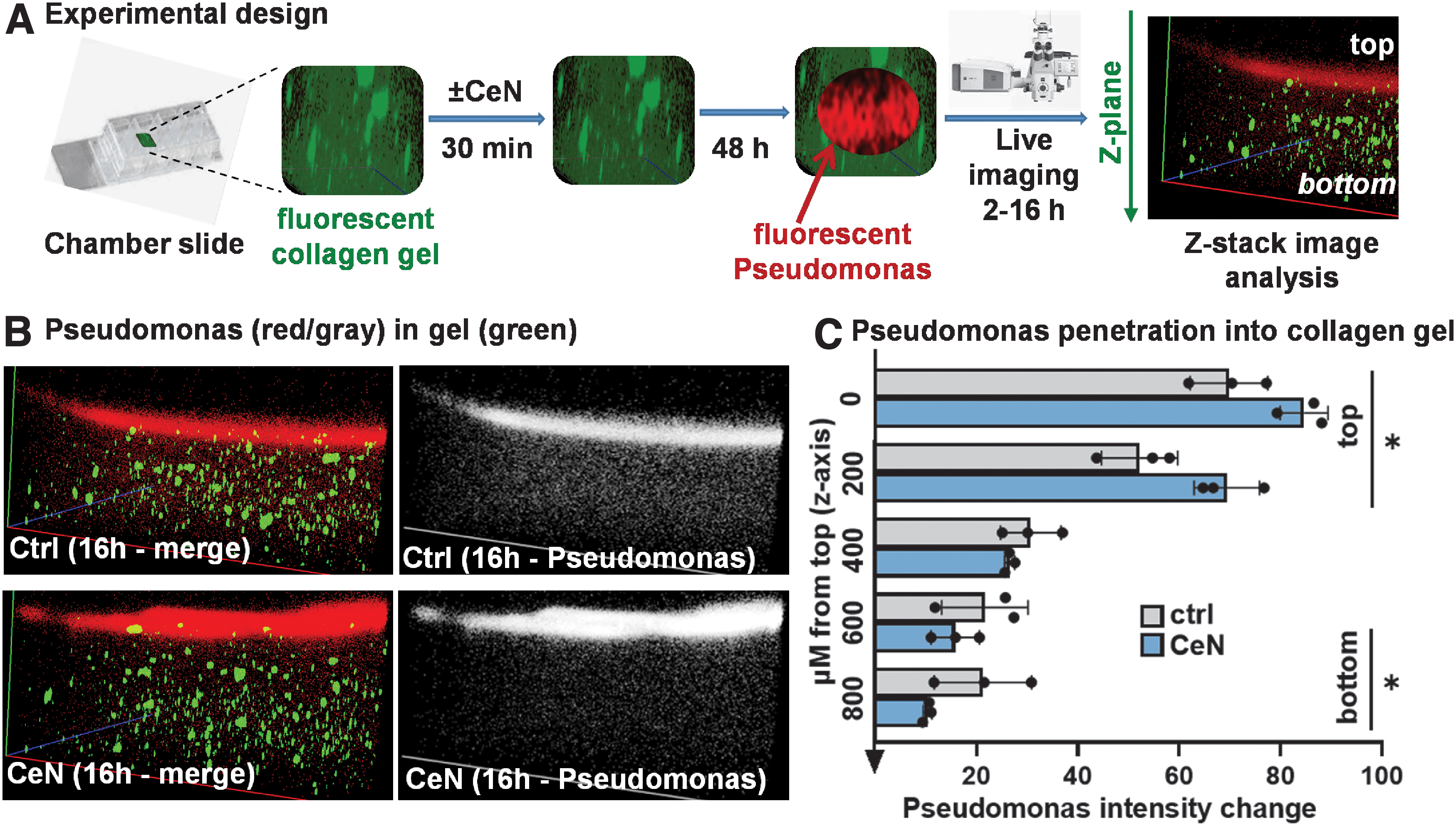
CeN attenuated Pseudomonas aeruginosa PAO1 penetration of 3D-collagen gels:
Mechanical properties of ex-vivo–burned porcine skin
Porcine skin (Description 59490-0 PG Skin; Pel-Freez Biologicals, Rogers, AR) stored at −80°C was thawed to room temperature and cut into dumbbell-shaped pieces. The skin pieces were placed in a 50 mL conical tube containing betadine solution (NDC 67618-150-09; Purdue Products, Stamford, CT) and were slowly mixed at 20 RPM (revolutions per minute) on a tube rotator for 10 min. After treatment with betadine, the skin pieces were washed 3 × in phosphate-buffered saline (PBS, 1 × ) by mixing at 80 RPM for 10 min. These skin pieces were pat dried with sterile gauze and then burned for 13 s at ∼100°C using an in-house burn device.
Burned skin pieces were incubated with either CeN (0 or 40 mM) or CeN (40 mM) + SSD (30 mM) in 50 mL tubes and incubated with slow rotation (20 RPM) for 30 min at room temperature on a tube rotator. The skin pieces were removed from either CeN or CeN+SSD solutions, blot dried, and incubated for 2 days at 37°C in humidified petri dishes (described in P. aeruginosa 1244 Penetration of Ex-Vivo–Burned Porcine Skin section), before performing the uniaxial tensile strength test (ElectroPuls™ E3000 instrument employing Dynacell™ load cell; Instron, Norwood, MA). Stiffness of the skin was assessed as the ratio of tensile stress to tensile strain from the test outputs from the instrument software.
P. aeruginosa 1244 penetration of ex-vivo–burned porcine skin
Viable counts from filters
Porcine skin was cut into discs (1.1 cm diameter) and processed as described above (Mechanical Properties of Ex-Vivo–Burned Porcine Skin section). These skin discs were then placed on top of 0.2 μM filters (Whatman; Sigma-Aldrich). Mid-log-phase P. aeruginosa-1244 (∼106 CFU in 10 μL) was pipetted onto the surface of each burned skin. After 2 or 3 days of incubation at 37°C in humidified petri dishes (sterile water in a small container to maintain moisture inside the petri dish, Fig. 4A), the 0.2 μM filters were separated from the replicate skin discs and homogenized in 1 mL PBS in MagNA Lyser Beads tubes (Roche) using FastPrep®-24 Tissue Homogenizer (MP Biomedicals, LLC. Santa Ana, CA).
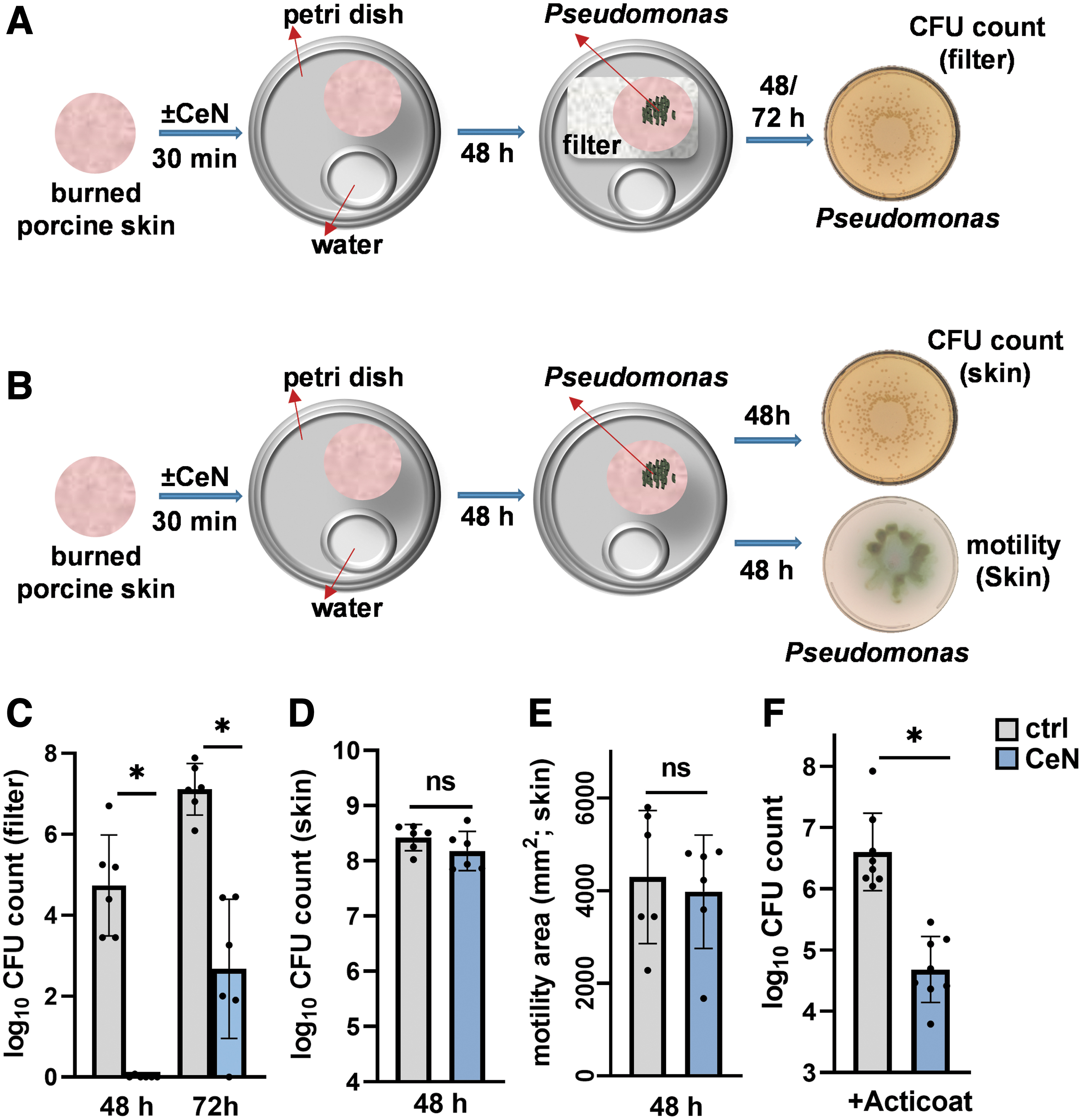
CeN attenuated Pseudomonas aeruginosa penetration through burn porcine skin samples
The homogenized samples were plated on P. aeruginosa isolation agar (Cat. No. P0144; Teknova, Hollister, CA) and incubated at 37°C for ∼18 h. The viable counts (CFUs) of the 0.2 μM filters were determined using an automated colony counter (ProtoCOL 3 instrument form SYNBIOSIS, Frederick, MD).
Viable counts from skin
Porcine skin was cut into discs (1.1 cm diameter) and processed as described above (Mechanical Properties of Ex-Vivo–Burned Porcine Skin section). Mid-log-phase P. aeruginosa-1244 (∼106 CFU in 10 μL) was pipetted onto the surface of each burned skin. After 2 days of incubation at 37°C in humidified petri dishes (described above), the replicate skin discs were removed from the petri dishes and were separately homogenized in 1 mL PBS in MagNA Lyser Beads tubes (Roche) using a FastPrep-24 Tissue Homogenizer (MP Biomedicals, LLC). The homogenized samples were plated on P. aeruginosa isolation agar and incubated at 37°C for 18 h. The viable counts of skin discs were determined using an automated colony counter as described above.
Antimicrobial activity of Acticoat against P. aeruginosa 1244 inoculated on ex-vivo–burned porcine skin ± CeN treatment
As described above, the skins were cut into discs, betadine treated, washed with PBS, tap dried with sterile gauze, burned, treated with 40 mM CeN, and incubated for 2 days at 37°C. The burned-skin discs were then inoculated with ∼106 CFU of P. aeruginosa-1244 per skin disc and 4 h later were overlaid with Acticoat dressing (Smith & Nephew) that was wetted with sterile H2O. After incubation for 2 days at 37°C, Acticoat was removed and the burned-skin discs were homogenized in 1 mL PBS in MagNA Lyser Beads tubes (Roche) using a FastPrep-24 Tissue Homogenizer (MP Biomedicals, LLC). The homogenized samples were plated on P. aeruginosa isolation agar, incubated at 37°C for 18 h, and the viable CFU of skin discs was determined using an automated colony (as described in above section).
P. aeruginosa PAO1 viable counts, motility (swarming and twitching), inflammatory-stimulation activity, and susceptibility to phagocytosis by THP-1 cells
Viable counts
Overnight P. aeruginosa PAO1 culture was serially diluted and adjusted 106 CFU in 10 μL with the sterile saline solution (0.9% w/v, pH7.0) and was mixed with 990 μL of saline solution containing different concentrations of CeN (0, 0.1, 1, and 10 mM). After incubation at 25°C for 24 h with shaking, these solutions were serially diluted and plated on P. aeruginosa isolation agar, incubated at 37°C for 18 h. The viable cells (CFU) recovered from the CeN treatment were determined using an automated colony counter (as described in above P. aeruginosa 1244 Penetration of Ex-Vivo–Burned Porcine Skin section).
Swarming motility
Assays were performed on plates containing 0.5% w/v Bacto agar (Difco), 8 g of nutrient broth (Oxoid) l-1, and 0.5% w/v D-glucose as previously described. 32,33 Swarming motility was performed on P. aeruginosa PAO1 cells that were treated with different CeN concentrations (or) bacteria recovered from the CeN treated and untreated skins and normalized to ∼106 CFU/mL. Five microliters of culture was spotted in four replicates. Swarming was observed after 18 h of incubation at 37°C. The zone of the swarming motility area was determined by measuring the mean diameter of the motility circle.
Twitching motility
Assays were performed on plates containing 10 g/L tryptone, 5 g/L yeast extract, and 0.5% w/v D-glucose solidified with 1.0% (w/v) Difco granulated agar as previously described. 34 P. aeruginosa PAO1 cells were treated with different concentrations of CeN and normalized to 106 CFU/mL. The cells were pelleted and resuspended in 50 μL of the LB media. A sharp toothpick, which has been immersed in the above bacterial suspension, was used to stab through the agar to the bottom of the 1-day-old plates. The plates were then incubated at 25°C for 24 h, and the zone of twitching motility area at the agar/petri dish interface was determined by measuring the mean diameter of the motility circle.
Inflammatory-stimulation activity
P. aeruginosa PAO1 cells were treated with different concentrations of CeN, spun down, and heat inactivated (65°C for 1 h). Dead bacteria (confirmed by plating) were added (106) to THP-1 cells (5:1 ratio bacteria to THP-1 cells) in 500 μL and incubated for 1 h at 37°C. Supernatants were then assayed for tumor necrosis factor-α (TNF-α) and interleukin 8 (IL-8; enzyme-linked immunosorbent assay [ELISA] kit; R&D Systems, Minneapolis, MN) on a DSX automated ELISA machine (Dynex Technologies) as described previously. 35
In a separate experiment, for the mock group, THP-1 cells (106 THP-1 cells; 500 μL total volume) were incubated with 0 mM CeN (control [ctrl]) or 0.1 mM CeN. For the experimental group, the THP-1 cells (106 THP-1 cells; 500 μL total volume) were incubated with lipopolysaccharide (LPS; Cat. No. L9143, P. aeruginosa; Sigma), either untreated or pretreated with 0.1 mM CeN for 30 min. After incubation for 1 h at 37°C, supernatants were collected and then quantified for TNF-α and IL-8 as described above.
Phagocytosis assay
P. aeruginosa PAO1 cells were treated with CeN (or saline control) and were stained with live stain (green; live/dead bacterial staining kit; Invitrogen). In another experiment, the CeN-treated bacteria were heat inactivated at 65°C for 45 min and then stained with dead cell stain (red; live/dead bacterial staining kit; Invitrogen). The stained bacteria were added to 106 THP-1 cells (5:1 ratio of normalized live or heat killed bacteria to THP-1 cells in a total volume of 500 μL) for 1 h incubation at 37°C. Before the THP-1 cells were spun down, they were stained with corresponding Cytotox-red (or) green stain (Incucyte, Cat. No. 4632 (red); 4633 (green); Essen BioScience, Inc., Ann Arbor, MI) and assessed for fluorescence from bacterial phagocytosis.
Bacterial phagocytosis was assayed as the percentage of cells containing green fluorescent (live) or red fluorescent (dead) bacteria and the mean fluorescence intensity (MFI) of the phagocytized bacteria using the Attune NxT flow cytometer (Invitrogen-Life Technologies, Carlsbad, CA). Viable THP-1 cells were gated on forward scatter and negative events from corresponding Cytotox-fluorescence (dead THP-1 cells). Unstained and compensation controls were used to gate the events positive for phagocytosis.
Statistical analyses
Experiments were performed in duplicate or triplicate, and each experiment was repeated a minimum of three times on different days. All statistical analyses were performed in GraphPad Prism 9 (GraphPad Software, San Diego, CA). Values are presented as mean with standard deviation, unless otherwise stated. Significant differences between two groups were established by t-test or two-tailed Mann–Whitney test for nonparametric data. For testing significant differences among multiple groups, one-way analysis of variance with Holm-Šídák's multiple comparisons test was used.
Electronic laboratory notebook was not used.
RESULTS
Stiffness of both 3D collagen and ex-vivo–burned porcine skin was increased by CeN
CeN treatment (1 mM) of collagen gel increased stiffness detected as storage modulus (G′, kPa) (Fig. 2A, B). Similarly, burned porcine skins treated with 40 mM CeN demonstrated increased modulus stiffness (Fig. 2C, representative tensile stress vs. strain curves) and stiffness (Fig. 2D and Supplementary Fig. S1). Burned porcine skin either treated with CeN (40 mM) alone or combination of CeN (40 mM) and SSD (30 mM) showed comparable increases of stiffness (Supplementary Fig. S2).
Penetration of P. aeruginosa through CeN-treated 3D-collagen gel or ex-vivo–burned porcine skin was retarded
As shown in the experimental design (Fig. 3A), red-fluorescent-labeled P. aeruginosa PAO1 was pipetted on top of collagen gels that had been treated with or without 1 mM CeN, and 2–16 h later the red fluorescence intensity of P. aeruginosa in layers of the gel was measured. In the gel that was pretreated with CeN, P. aeruginosa PAO1 was increased over time in upper layers and decreased in lower layers (Fig. 3B, C).
Similarly, we assessed penetration of P. aeruginosa strain 1244 through CeN-treated ex-vivo–burned porcine skin. P. aeruginosa 1244 was pipetted onto the surface of the CeN-treated skin (schematic in Fig. 4A, B). After 2 or 3 days of incubation at 37°C, the number of P. aeruginosa recovered from filters underneath the skin was assessed (Fig. 4A). After 48 h, ∼106 bacteria were recovered under the untreated skin while under the CeN-treated skin no bacteria were recovered (Fig. 4C). After 72 h postinoculation, ∼102 CFU was recovered from the filters under the CeN-treated skin, while ∼107 CFU was recovered from the filters under the untreated skin.
P. aeruginosa 1244 recovered from ex-vivo–burned porcine skin discs treated with or without CeN had similar viable counts and motility
P. aeruginosa 1244 was pipetted onto the surface of the CeN-treated skin. Bacteria were recovered after 2 days of incubation at 37°C for determination of viable counts (Fig. 4B). The number of viable bacteria recovered from the treated and untreated skin discs was not significantly different (Fig. 4D).
For swarming motility assay, bacteria recovered from the CeN-treated and untreated skins as described above were plated on soft agar. After 18 h incubation at 37°C, the area of bacterial spread around the spot of inoculation was measured. There was no significant difference in the area of the bacteria recovered from the treated and untreated skin discs (Fig. 4E).
Greater killing by silver dressing of P. aeruginosa inoculated on ex-vivo–burned porcine skin treated with CeN
Ex-vivo–burned porcine skin discs were untreated or treated with CeN (40 mM), inoculated with P. aeruginosa 1244, and 4 h later was overlaid with silver dressing. The skins were then homogenized to extract bacteria for determining viable counts. Viability of the bacteria was significantly lower (∼2-logs) in the CeN-treated porcine skin overlaid with silver dressing than the untreated porcine skin overlaid with silver dressing (Fig. 4F).
Viable counts, motility, and penetration through skin models were reduced by treatment of P. aeruginosa with CeN
In the studies above, the collagen gel and the porcine skin were treated with CeN. In this study, P. aeruginosa PAO1 was treated directly with different concentrations of CeN for 24 h at 25°C. As shown in Fig. 5A, the viability reduction was concentration dependent, with 10 mM CeN reducing viability by 1.5 logs, while 0.1 mM CeN was not bactericidal compared to untreated control. In addition, CeN dose dependently killed a variety of microbes known to colonize wounds, including P. aeruginosa, Acinetobacter baumannii, Klebsiella pneumoniae, Enterococcus faecalis, S. aureus, and MRSA (Supplementary Fig. S3).
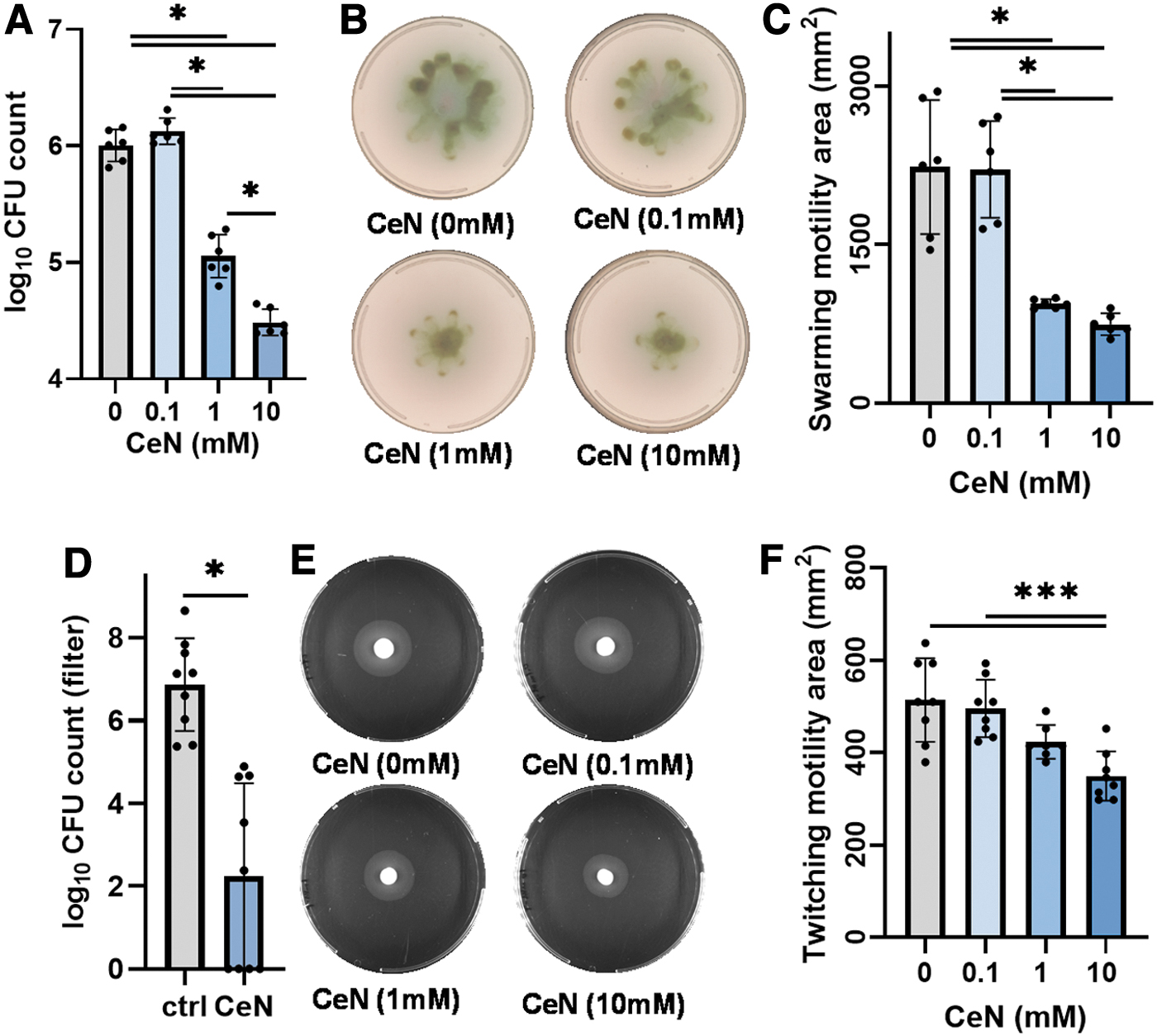
CeN reduced Pseudomonas aeruginosa viable counts, motility (twitching and swarming), and penetration: P. aeruginosa PAO1 was treated with 0, 0.1, 1, and 10 mM CeN for 24 h and then assayed for viability and motility.
In addition, we measured the swarming motility of CeN-treated P. aeruginosa PAO1. The bacteria treated with a range of CeN concentrations were plated on soft agar, and after 18 h incubation at 37°C the area of spread of the bacteria around the spot of inoculation was measured. One to 10 mM but not 0.1 mM CeN reduced swarming motility (Fig. 5B, C). The twitching motility was also measured for P. aeruginosa PAO1 treated with series of CeN concentrations. The bacteria were stabbed into the agar, and after 24 h incubation at 37°C, the area of spread of the bacteria around the stab was measured. Compared to 0 and 1 mM CeN, 10 mM CeN showed a significant reduced twitching motility (Fig. 5E, F).
Furthermore, we assessed the ability of CeN-treated P. aeruginosa 1244 to penetrate ex-vivo–burned porcine skin by determining the number of viable Pseudomonas recovered from filters underneath the skin discs. Recovery of the bacteria treated with 10 mM from the filters was reduced by 1.5 logs (Fig. 5D).
CeN-treated P. aeruginosa was more susceptible to phagocytosis by THP-1 cells
To assess CeN treatment of viable P. aeruginosa PAO1 on phagocytosis, live CeN-treated P. aeruginosa (stained green) was added to THP-1 cells. After a 1-h incubation at 37°C, the percentage of viable THP-1 cells containing live green-fluorescent P. aeruginosa was determined by flow cytometry. The bacteria treated with 1 and 10 mM CeN were phagocytized to a significantly greater extent than the untreated bacteria (Fig. 6A–C). In addition, the P. aeruginosa treated with 1 and 10 mM CeN had a greater mean green-fluorescent intensity per THP-1 cell.
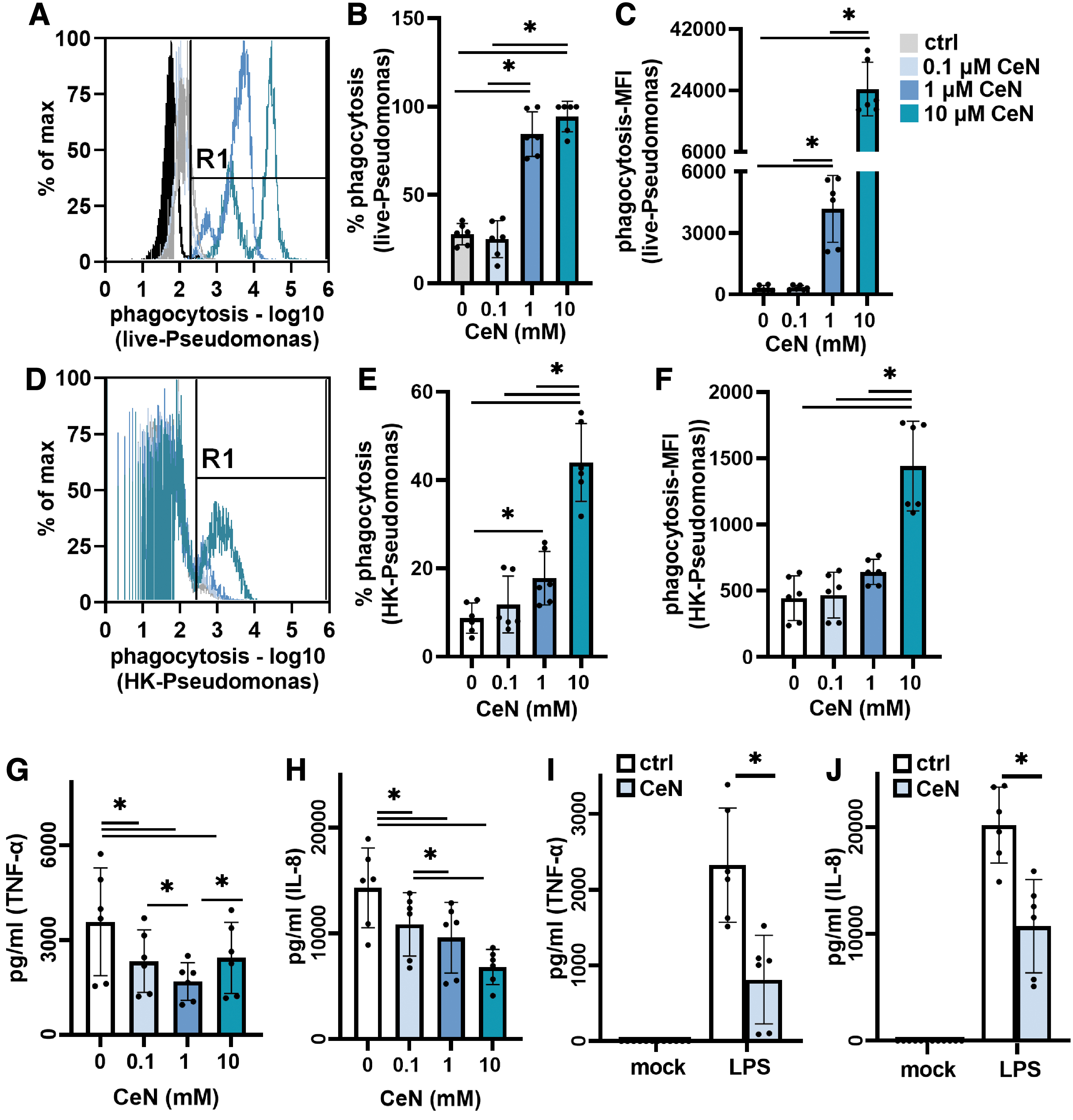
Pseudomonas aeruginosa PAO1 treated with CeN was phagocytized more by THP-1 cells and less stimulatory to THP-1 cytokine production:
To assess CeN treatment of P. aeruginosa PAO1 on phagocytosis of nonviable P. aeruginosa, CeN-treated P. aeruginosa was heat killed (stained-red) before incubation with THP-1 cells. After a 1-h incubation at 37°C, the percentage of viable THP-1 cells containing dead red-fluorescent P. aeruginosa was determined by flow cytometry. The bacteria treated with 1 and 10 mM CeN that were heat killed were phagocytized to a significantly greater extent than the untreated bacteria (Fig. 6D–F). In addition, the P. aeruginosa treated with 10 mM CeN had a greater mean red-fluorescent intensity per THP-1 cell. In addition, in our experimental conditions, the THP-1 cells treated with CeN showed no significant changes in phagocytosis (Supplementary Fig. S4).
Inflammatory stimulatory activity of P. aeruginosa or LPS treated with CeN was reduced
Inflammatory activity of CeN-treated P. aeruginosa
P. aeruginosa PAO1 was treated with CeN and then heat-killed because the live bacteria induced significant THP-1 cell death. The bacteria treated with CeN (0.1, 1, and 10 mM) elicited significantly less TNF-α and IL-8 from THP-1 cells (Fig. 6G, H).
Inflammatory activity of CeN-treated LPS from P. aeruginosa
The LPS of P. aeruginosa when treated with CeN was also less stimulatory to THP-1 cells: compared to the untreated LPS, LPS treated with 0.1 mM CeN elicited significantly less TNF-α and IL-8 released from THP-1 cells (Fig. 6I, J). Treatment of THP-1 cells with CeN alone or PBS did not elicit any inflammatory response (Fig. 6I, J).
DISCUSSION
Compared to SSD cream (e.g., Silvadene®), CeN-SSD topical burn cream (e.g., Flammacerium) produces a firm and dry eschar described as leather like or shell like, which is thought to resist bacterial colonization/infection, and is used, in addition to the general management of burn wounds, to facilitate postponement of eschar excision and autografting. To mimic CeN’ treatment of wound tissue and investigate its physical effects, we used two in vitro skin models: 3D-collagen matrix and ex-vivo–burned porcine skin. These models were treated with CeN solutions for 30 min to mimic the 30-min bathing in 40 mM CeN solution of severely burned patients that were associated with reduced mortality. 36
We also studied CeN treatment of P. aeruginosa for effects on viability and in vitro virulence. Although the ex-vivo–burned porcine skin model used in this study was adapted from the previously developed studies, 37 –39 the current study has expanded the model to understand the mechanical properties of CeN-treated skin models, as well as P. aeruginosa penetration, which together add some novelty to the model.
CeN effects on mechanical properties of skin models
CeN increased the stiffness of both skin models. In addition, the ex-vivo–burned porcine skin treated with CeN was less penetrable by P. aeruginosa, and P. aeruginosa inoculated on this CeN-treated skin model was more susceptible to killing by a silver dressing. Penetration of P. aeruginosa into ex-vivo–burned porcine skin pretreated with CeN was absent for up to 2 days for the highly invasive P. aeruginosa strain 1244. 30
Penetration of P. aeruginosa into 3D-collagen matrix was also inhibited, such that more bacteria were confined to the superficial layers of the matrix, which may explain why bacteria inoculated on ex-vivo–burned porcine skin were more effectively killed by the silver dressing. This interpretation may also explain our previous in vivo finding that silver dressings supplemented with CeN were more effective than dressings alone at inhibiting the regrowth of normal flora in rat full-thickness burn wounds. 40
CeN may change the properties of the skin models by cross-linking collagen in the 3D-collagen matrix. Similarly, in ex-vivo–burned porcine skin, CeN may cross-link native and burn-denatured macromolecules. Cerium ions exist in solution as Ce3+ or Ce4+ ions and have an ionic radius like calcium but with greater charge that contributes to high-affinity binding to Ca2+-binding sites on biological molecules. 41 In our preliminary experiments (not shown), monomeric G-actin (rabbit skeletal muscle), high mobility group box-1 (HMGB1), and rat tail collagen were aggregated by CeN. Thus, cross-linking of macromolecules within burned skin may contribute to the observed changes in elasticity and tensile strength and the resistance to P. aeruginosa penetration.
An alternate possibility is that cerium ions available in the collagen gel and porcine skin reduce P. aeruginosa viability and motility. However, P. aeruginosa recovered from ex-vivo–burned porcine skin treated with or without CeN had similar viable counts and swarming motility. In addition, there was no detectable free cerium ions in the supernatants extracted from the CeN-treated ex-vivo–burned porcine skin (not shown). These results suggest that CeN in the skin models was not freely available at sufficient concentrations to affect viability or swarming motility of P. aeruginosa and that the action of CeN on the skin models was likely responsible for impeding P. aeruginosa penetration.
The burn eschar—dead tissue—produces toxins, including denatured macromolecules, which contribute to inflammation (local and systemic), immunosuppression, hypermetabolism, and at the extreme, multiorgan dysfunction. 42 –44 Due to the loss of skin barrier function, eschar is also a nidus for microbial infections that aggravate burn-induced inflammation. 17,43 –45 The findings that CeN stiffens ex-vivo–burned porcine skin may have some clinical implications. Increased stiffness may hamper bacterial penetration and delay the pathological progression of infection. In addition, stiffer skin, as a result of CeN ions cross-linking with the denatured macromolecules within the burned skin, some of which could be pro-inflammatory molecules such as damage-associated molecular patterns (DAMPs), may decrease systemic dissemination of these molecules and the resultant inflammation, as well as wound progression. 26,46 –48
CeN effects on P. aeruginosa viability and virulence
Treatment of P. aeruginosa with CeN at 10–40 mM concentrations reduced their survival by ∼1.5–4 logs. The antimicrobial activity of CeN against gram-negative bacteria, including P. aeruginosa, likely results from positively charged cerium ions (Ce3+ and Ce4+) binding to negatively charged LPS. In an earlier study, CeN at ∼2 mM in saline (24 h treatment in suspension without shaking) caused Escherichia coli flocculation, outer membrane separation, and a nearly 1-log reduction in viable counts, suggesting that cerium ions interacting with LPS permeabilize the outer membrane. 49 In addition, a CeN concentration as low as ∼0.02 mM produced membrane perturbations (ruffles) in E. coli. 49
In our study, at sublethal 1 mM concentration, CeN reduced the swarming and twitching motility and other virulence properties of P. aeruginosa. Such an effect of CeN on LPS and outer membrane in E. coli 49 may also explain the reduced swarming and twitching motility of P. aeruginosa as observed in our study. Alternatively, the reduction in swarming and twitching motility may be due to cerium ions binding to flagellar and pili proteins and interfering with their motor function. Reductions in P. aeruginosa swarming ability by specific mutations or treatment with a short cationic peptide (peptide 1018) in a murine model of skin abscess have been shown to reduce colonization and dissemination. 50 The reduced swarming and twitching motility of P. aeruginosa after CeN treatment may contribute to the reduced penetration of P. aeruginosa through the skin models seen in this study.
Furthermore, CeN treatment of P. aeruginosa reduced their stimulation of the monocyte cell line THP-1 to produce inflammatory cytokines (TNF-α and IL-8). To explore why CeN-treated P. aeruginosa was less stimulatory to THP-1 cells, we tested LPS from P. aeruginosa. Treatment of P. aeruginosa LPS with CeN reduced its stimulation of cytokine production by THP-1 cells. Finally, CeN treatment of P. aeruginosa increased their phagocytosis by THP-1 cells. After CeN treatment, live and heat-killed P. aeruginosa was significantly more susceptible to phagocytosis. Similar to above, a possible explanation for these observations (reduced inflammatory stimulation and increased phagocytosis) is that Ce3+ or Ce4+ ions neutralize negative charges on LPS. Treatment of LPS with other positively charged molecules has shown to block the endotoxic effects of LPS. 51
Limitations
The in vitro and ex vivo models used herein are artificial systems that do not completely recapitulate the complex burn wound environment that includes systemic circulation, immune functions, and healing. Our studies also do not address the hypothesis that cerium ions can replace Ca++ from proteins, altering their structures and/or activities, 20 the hypothesis that cerium ions bind tissue pyrophosphate, releasing calcium and pyrophosphate's inhibition on the formation of calcium phosphate crystals, as within cancellous or cortical bone, 52,53 or the hypothesis that positively charged cerium ions interact with negatively charged bacterial membranes resulting in structural changes and bacterial growth inhibition. These mechanistic hypotheses warrant further exploration using in vitro and ex vivo models.
Summary
Our data suggest multiple ways that CeN may reduce colonization and infection by acting on both the bacteria and the burn eschar: increasing phagocytosis of bacteria, decreasing pro-inflammatory cytokine production (possibly inhibiting wound progression), inhibiting swarming and twitching motility, and increasing the resistance of burn tissue to bacterial penetration. By inhibiting penetration, CeN treatment may confine P. aeruginosa to the superficial skin where vulnerability to killing by silver dressings may be increased. In addition, our studies demonstrate that ex vivo skin models may be an effective platform for screening treatments that interfere with bacterial penetration of wounded tissue.
INNOVATION
Infection is the leading cause of mortality among burn patients and responsible for up to 50% of burn-related deaths. Multiple topical products have been developed to control burn infections to improve the healing. Conventional drug susceptibility testing focuses on identifying new drugs that exhibit bactericidal efficacy to a wide range of microorganisms. Our in vitro skin models offer a platform for screening drugs that change the property of burn tissues and result in preventing/retarding bacterial penetration/infection into wounded tissues.
KEY FINDINGS
CeN treatment of in vitro skin models:
o Increased stiffness of 3D collagen matrix and ex-vivo–burned porcine skin
o Reduced penetration of P. aeruginosa into 3D collagen matrix
o Increased vulnerability of P. aeruginosa inoculated on ex-vivo–burned porcine skin to killing by silver dressing.
CeN treatment of P. aeruginosa:
o Reduced viability
o Reduced swarming and twitching motility
o Reduced ability to penetrate 3D-collagen matrix and ex-vivo–burned porcine skin
o Reduced stimulation of cytokine production by THP-1 cells
o Increased vulnerability to phagocytosis by THP-1 cells.
Footnotes
ACKNOWLEDGMENTS AND FUNDING SOURCES
The views expressed in this article are those of the authors and do not reflect the official policy or position of the U.S. Army Medical Department, Department of the Army, DoD, or the U.S. Government. The authors acknowledge the support of Dr. Wen Lien and Dr. Tao You for their assistance with the tensile strength test. This work was supported, in part, through the Congressionally Directed Medical Research Programs, U.S. Army Medical Research and Development Command (MRDC) W81XWH-20-2-0004, Intramural Program (CO210007_USAISR), Combat Casualty Care Research Directorate, MRDC, and the Naval Medical Research Center's Naval Advanced Medical Development Program (MIPR N3239820MHX040).
AUTHOR DISCLOSURE AND GHOSTWRITING
No competing financial interests exist. The content of this article was expressly written by the authors listed. No ghostwriters were used to write this article.
ABOUT THE AUTHORS
His research expertise focuses on improving health within the U.S Military.
SUPPLEMENTARY MATERIAL
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Abbreviations and Acronyms
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
