Abstract
Objective:
In recent years, reticulated open-cell foam-based closed-incision negative pressure therapy (ROCF-ciNPT) has shown effectiveness in management of various postoperative incisions. These dressings consist of a skin interface layer that absorbs fluid from the skin surface and reduces the potential for microbial colonization within the dressing by means of ionic silver. This study examines the ability of silver to reduce the bioburden within the dressing as well as the localized effect due to potential silver mobility.
Approach:
Ability of silver to reduce bioburden within the ROCF-ciNPT dressing was assessed using Staphylococcus aureus, Pseudomonas aeruginosa, and Candida spp. Furthermore, silver mobility was assessed using an in vitro skin model to study the zone of inhibition along with released silver quantification. Using a porcine model, diffusion of silver into blood and tissue was studied using emission spectrometry and histology.
Results:
Microbial growth in the ROCF-ciNPT dressing was significantly reduced (∼2.7–4.9 log reduction) compared to a silver-free negative control. No zone of inhibition was observed for microbial colonies for up to 7 days with minimal localized silver release (<5.5 ppm release). In vivo studies demonstrated no measurable concentration (<0.2 μg/g) of silver in the blood, urine, feces, kidney, and liver tissue biopsy.
Innovation:
This study provides an important insight into silver concentration and mobility within the ROCF-ciNPT dressing, given emerging concerns associated with potential silver cytotoxicity.
Conclusion:
These results indicate the concentration of silver (0.019% silver by weight) in the ROCF-ciNPT dressings has been adequate to reduce bioburden within the skin interface layer, while severely limiting the amount of silver leaching out.


Introduction
Postoperative complications, including surgical site infections (SSIs), formation of hematoma, seroma, tissue necrosis, and wound dehiscence, result in morbidity, mortality, and significant monetary cost. 1,2 Comorbidities such as obesity, vascular disease, and diabetes can increase the risk of occurrences of such complications. 3 Among these complications, SSIs account for ∼21.8% of all health care-associated infections and each incident is estimated to increase the cost by up to ∼$26,000 per patient, besides prolonging the mean length of hospital stay by ∼14 days when compared to noninfected patients. 4 –6 In addition, such complications negatively impact the mental and physical health of the patient and quality of life. Increased direct and indirect costs due to postoperative complications pose a substantial challenge for both clinicians and patients.
The current standard of care for management of closed surgical incision involves the use of dry, sterile gauze for absorbing wound exudate and a secondary dressing that acts as a physical barrier for external contaminants. Saturated gauze-based dressing, however, can present a conducive microenvironment for microbes that may lead to SSIs and hence, many progressive facilities have adopted the use of advanced wound dressings. Toward the management of closed surgical incisions, recently a reticulated open-cell foam-based closed-incision negative pressure therapy (abbreviated as ROCF-ciNPT, PREVENA™ Therapy; KCI, an Acelity Company, San Antonio, TX) has been used in which a hermetically sealed dressing is connected to a therapy unit delivering negative pressure over a closed surgical incision.
Several studies have shown reductions in SSIs in patients with hip and lower extremity fractures, laparotomy, abdominal wall reconstruction, cesarean, sternotomy, and vascular surgical sites using the ROCF-ciNPT. 7 –10 For example, Kwon et al. recently reported the effectiveness of the ROCF-ciNPT in a randomized prospective study of 119 closed incisions that were identified as high risk for wound complication based on parameters such as body mass index, pannus, reoperation, poor nutrition, and immunosuppression. 11 The use of the ROCF-ciNPT compared to the standard of care resulted in a significant reduction (11.9% vs. 26.7%, p < 0.001) in major wound complications such as dehiscence, infection, and hematoma.
Clinical Problem Addressed
The ROCF-ciNPT consists of a textile-based skin interface layer that absorbs fluid from the skin surface and reduces the potential for microbial colonization within the textile layer by means of ionic silver (0.019% silver by weight in the form of silver hydrogen zirconium phosphate) as shown in Fig. 1. The presence of silver is not intended for the prevention and/or management of infection within the incision. Given the effectiveness of silver against a wide range of microbes, including Gram-positive and Gram-negative bacteria, yeast, and fungi, there has been a vast increase in the use of silver over the last few decades. 12 –14 However, concerns regarding potential cytotoxicity of silver in humans, including argyria, argyrosis, and absorption in soft tissues (brain, liver, kidney, and spleen), still exist, highlighting the importance of responsible use of silver in wound dressings. 15,16
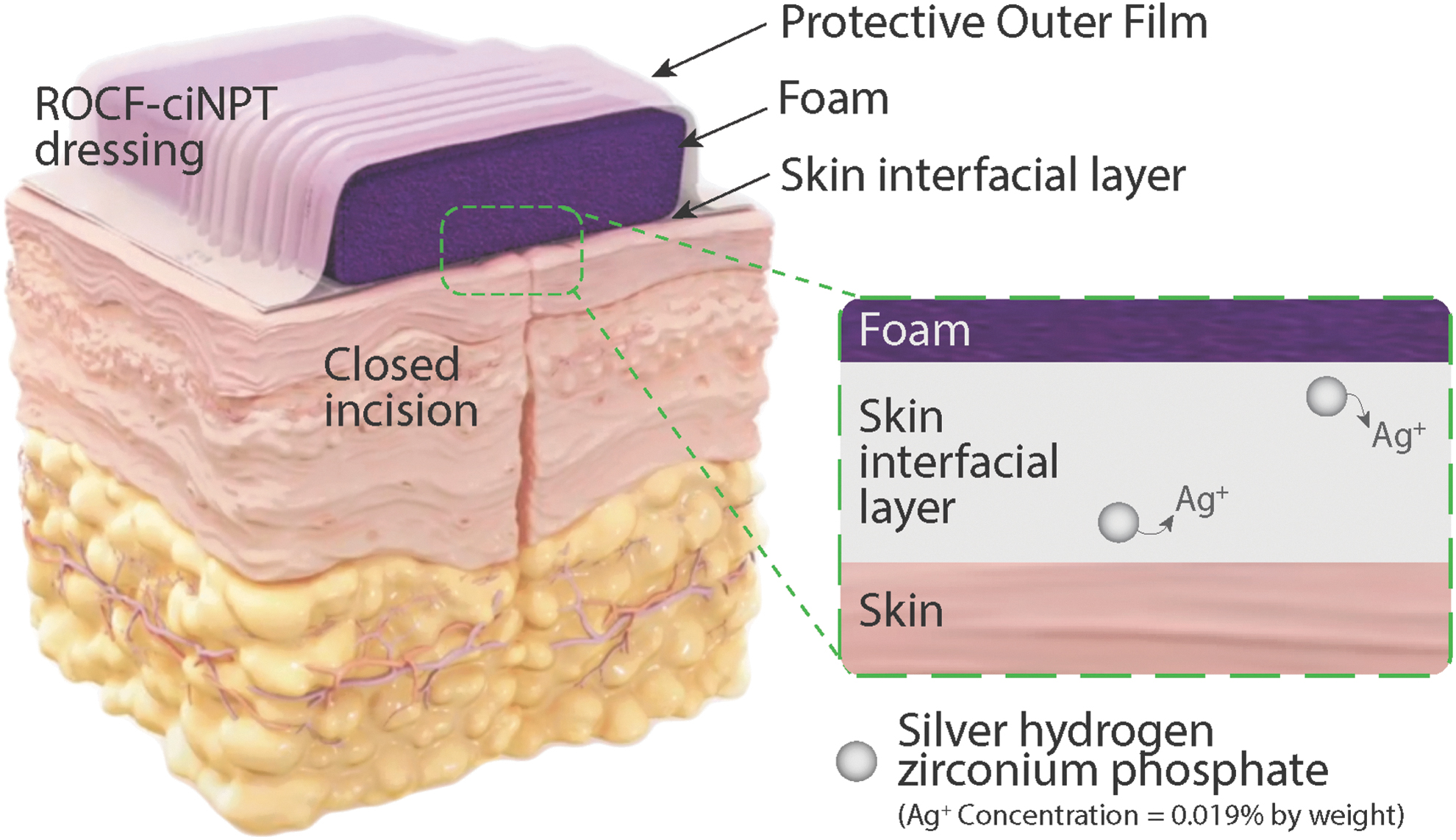
ROCF-ciNPT dressing design. The ROCF-ciNPT that is used for management of closed incisions consists of an outer protective polyurethane-based film barrier, a foam manifold, and a skin interfacial layer. The dressing is adhered to the incision site by means of a pressure-sensitive adhesive that is coated on the bottom side of the skin interfacial layer. The skin interfacial layer as shown in the inset (not to scale) contains silver hydrogen zirconium phosphate (0.019% by weight) that releases silver ion to prevent microbial growth within the ROCF-ciNPT dressing. ROCF-ciNPT, reticulated open-cell foam-based closed-incision negative pressure therapy.
In the work presented herein, we hypothesized that the amount of silver present in the ROCF-ciNPT skin interfacial layer is sufficient to hinder microbial growth within the dressing and it does not have significant mobility to have a localized effect on microbial colonies adjacent to or in proximal contact with the dressing. First, we investigated the ability of silver to hinder the microbial growth within the dressing over a 7-day period. Next, silver mobility and its subsequent effect on microbial colonies (i.e., zone of inhibition), as well as the amount of silver that leached out of the dressing, were determined in an in vitro agar-based skin model. Finally, we assessed silver levels in feces, urine, blood, kidney, and liver for potential accumulation of silver during actual use in an in vivo porcine wound model. In light of emerging concerns associated with use of silver in wound dressings, this study provides an important insight into silver concentration and mobility within the ROCF-ciNPT dressing.
Materials and Methods
Materials
The ROCF-ciNPT dressing, a silver containing commercially available dressing (abbreviated as AC, Acticoat® 7; Smith & Nephew, Inc., London, UK), and a dressing without silver (abbreviated as ME, Mepore®, Mölnlycke Health Care US, LLC) were obtained from commercial sources. All dressings were stored as recommended by the manufacturer's instructions and used within their respective expiration dates.
In vitro antimicrobial activity
Using aseptic technique, the sterile skin interface fabric component of the ROCF-ciNPT dressing containing silver was delaminated from the foam bolster and polyurethane film. The adhesive border on the perimeter of the fabric was also removed. Subsequently, the fabric samples were cut into predefined shapes with fixed dimensions for each time point and each microbe as described below.
Microbial growth within the skin interface layer
Toward studying the effect of silver on the microbial growth within the skin interface layer, microbial subculture for Staphylococcus aureus subsp. aureus Rosenbach (ATCC 6538), Pseudomonas aeruginosa (ATCC 9027), and Candida albicans (ATCC 10231) was prepared as recommended by the manufacturer (American Type Culture Collection (ATCC), Manassas, VA). Working cultures were grown using standard microbiological methods (∼24 h at 32°C) and suspended in a maximum recovery diluent (MRD; 0.1% peptone, 145 mM sodium chloride) to ∼108 colony-forming units per milliliter (CFU/mL). An inoculum suspension was prepared in a solution of 96% MRD broth supplemented with 5% bovine serum. For each time point (day 0 and 7), a negative control consisting of sterile gauze dressings (3–5 ply; Covidien Curity®, Dublin, Ireland) and ROCF-ciNPT dressing's skin interface layer containing silver (2.5 × 2.5 cm) were inoculated in triplicate to yield ∼105 CFU/mL of Candida spp. and S. aureus and ∼106 CFU/mL of P. aeruginosa.
At time 0 (inoculum) and after 7 days at 30°C, inoculated fabrics were removed from the Petri dishes and placed in Difco Dey/Engley Neutralizing Broth (Becton, Dickinson and Company, Franklin Lakes, NJ) to deactivate the residual silver. After vortexing, aliquots of this broth were plated onto Petrifilm (3M, St. Paul, MN) plates or equivalent solid media. After incubation for 24–72 h at 30–35°C for S. aureus and P. aeruginosa, or 20–25°C for Candida spp., CFU per plate were counted visually to determine mean log10 CFUs per sample.
Silver mobility and zone of inhibition
In examining potential silver mobility and its subsequent effect on the microbial growth, isolates of bacterial strain (i.e., S. aureus [ATCC 6538, ATCC 13358, and ATCC 19095] and P. aeruginosa [ATCC 27853, ATCC 12055, and ATCC 9027]) and yeast (Candida spp. [ATCC 10231, 14053, and ATCC 90028]) were used. Equal volumes of each bacterial and yeast strain (∼9.0 log10 CFU/mL) were combined and diluted to make a three-strain cocktail with a final concentration of ∼6.0 log10 CFU/mL. This suspension served as the inocula and was plated (100 μL) onto plates containing Tryptic Soy Agar (TSA; Becton, Dickinson and Company for the bacterial strain) or Sabouraud dextrose agar (SDA; Becton, Dickinson, and Company for the yeast strain). The TSA and SDA media components both included 1.5% agar solids. The spread plating was performed using a sterile loop until the surfaces of the plates were dry and all inoculum had been absorbed onto the agar.
Once the inoculum had been applied to the TSA or SDA plates, the skin interface fabric containing silver (2 × 1 cm) was aseptically placed directly onto the center of the inoculated TSA or SDA plates. A commercially available silver mesh containing silver nanocrystals applied onto a rayon and polyester polymer (AC), which is known to maintain a sustained silver release over 7 days, served as a positive control, whereas spread inoculation without any test article served as a negative control. 17 After the dressing material had been placed onto each plate, the TSA plates with S. aureus or P. aeruginosa and the SDA plates with Candida spp. were incubated for up to 7 days at 35 + 2°C and 25 + 2°C, respectively.
During incubation, all TSA and SDA plates were held in a polyethylene bag to minimize the moisture loss. Subsequently, at each time point (3 h and 1, 3, 5, and 7 days), the plates were removed (n = 3 for each treatment group) and were observed for any inhibition of growth. The growth or lack thereof in the area immediately surrounding the test article was observed and growth present in the area immediately surrounding the textile dressing material was considered to indicate that the levels of the silver ions migrating out of the material were insufficient to have an antimicrobial effect in the area surrounding the textile dressing material. The area of inhibition was measured using a graduated ruler and pictures were taken of each plate at each time point.
In vitro silver release quantification
For in vitro silver release studies, the ROCF-ciNPT skin interface layer was gently placed onto a TSA agar plate with a sterile tongue depressor and incubated for up to 7 days at 35 + 2°C. In addition, AC was gently placed onto a separate TSA agar plate as a positive control and a plate without any test article served as a negative control. The testing time points for the ROCF-ciNPT skin interface layer samples included analyses at 3 h following preparation as well as at 1, 3, 5, and 7 days (n = 3). The positive control samples and the negative control samples were subjected to analysis at 3 h and 7 days.
At each of the time points, the TSA agar directly beneath the textile was extracted from the Petri dish container and the amount of Ag was quantified using inductively coupled plasma mass spectrometry (ICP-MS, Agilent Model 7700; Agilent Technologies, Sanata Clara, CA). For the control samples, an equivalent area from the control agar plates was extracted and subjected to ICP-MS Ag analysis. Three replicates per time point were analyzed.
In vivo wound model creation and dressing application
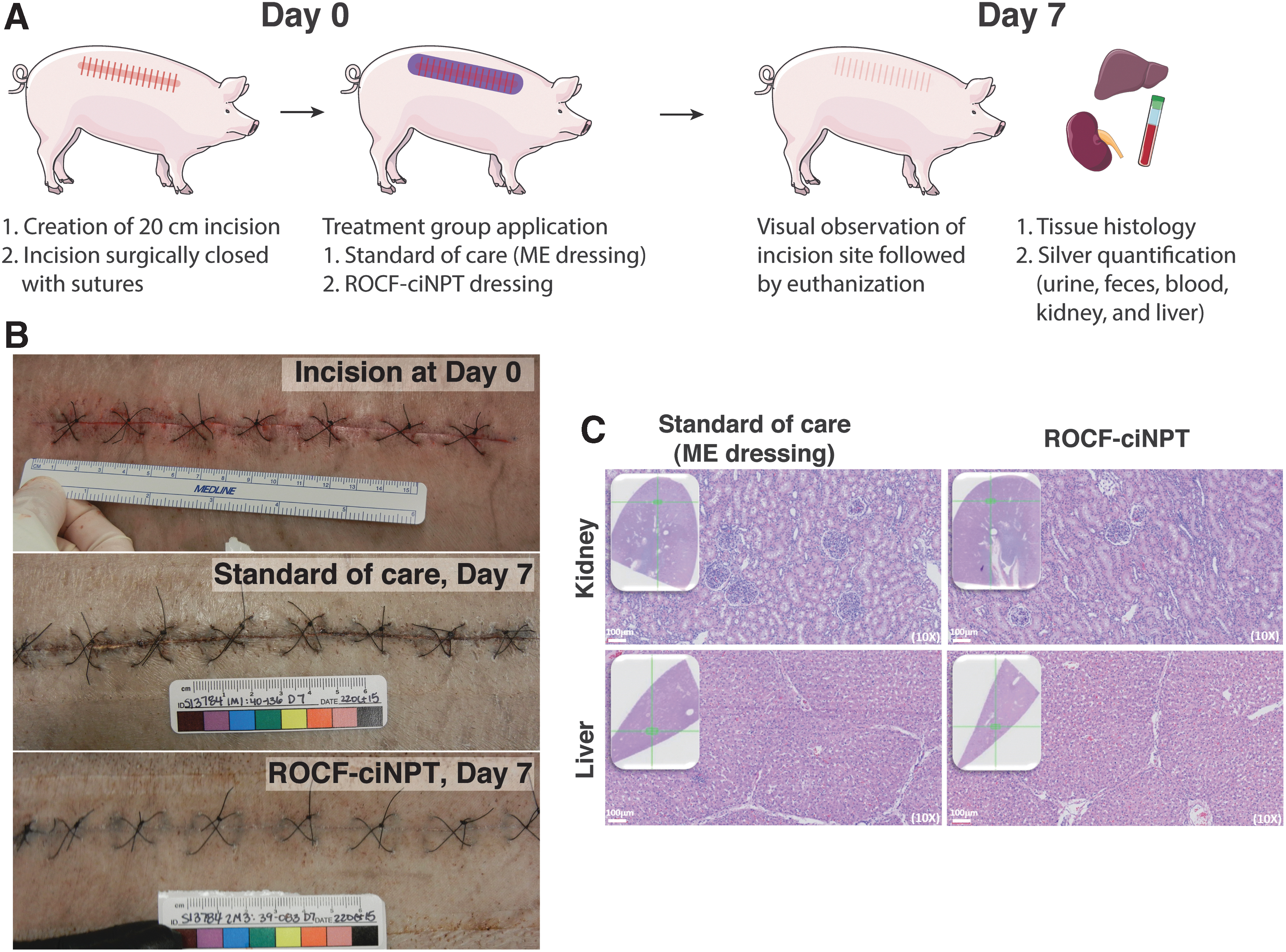
All in vivo experiments were conducted at the test facility (Sinclair Research, Auxvasse, MO) under a protocol approved by the Institutional Animal Care and Use Committee. Eight male Sus scrofa Yucatan miniature swine ∼5–6 months of age were included in this study. Animals were anesthetized using an intramuscular injection containing Telazol and Xylazine and subsequently maintained with isoflurane. Buprenorphine and tramadol were administered for analgesia. Animals were cleaned with disinfecting scrubs and isopropyl rinses. The entire dorsolateral back area of each animal was closely shaved and a single 20 cm linear paraspinal incision was made on one side of each animal between the crest of the shoulder and the coccygeal tuberosity. This full dermal thickness incision was created at the designated site using a sterile scalpel and then closed with a two-layer closure using sutures.
Photographs of the individual incisions were captured, and the control (ME) or test article (ROCF-ciNPT) was applied (n = 4 per test article) to the closed incision site and covered with a tear-resistant mesh and a cotton jacket. After 7 days, all animals were sedated, dressings were removed, and photographs of each individual incision were captured. Subsequently, animals were euthanized by an intravenous administration of sodium in conformance with American Veterinary Medical Association Panel on Euthanasia.
Histology
Liver and kidney tissue samples obtained after the in vivo study were preserved in 10% neutral buffered formalin, embedded in paraffin, sectioned to ∼5 μm thickness (three sections per tissue), and stained with hematoxylin and eosin. Slides were evaluated by light microscopy by a board-certified histopathologist (Histology & Pathology Research Lab, Mt. Jackson, VA) and gross and microscopic observations were recorded.
In vivo silver quantification
Sample portions (feces, whole blood, liver, and kidney tissue) weighing ∼0.5–1 g and 500 μL of urine samples were digested in nitric acid and then heated to 110°C for 1 h. After cooling to room temperature, 1 mL of 30% hydrogen peroxide was added and samples were heated for an additional 30 min. Samples were cooled before adding internal standard solutions diluted with high-purity water to a final mass of 100 g for feces, liver, and kidney, and 20 g for whole blood and urine. Silver content was measured by inductively coupled plasma atomic emission spectroscopy (ICP-OES) using a Thermo iCap 6000 spectrometer (ThermoFisher, Waltham, MA) at 328 nm. The content of silver was determined using a curve established with certified ICP grade standards (CPI International, Palo Alto, CA). Measured levels of silver ion in milligrams were normalized to background noise levels and reported in μg/g (n = 3).
Statistical analysis
Results are expressed as mean ± standard error of mean unless otherwise specified. Specific numbers of replicates (n ≥ 3) are noted for each experiment within the Results and Discussion section. Statistical significance was analyzed by performing a Student's t-test or one-way analysis of variance test, where p < 0.05 was considered significant. SigmaPlot and Microsoft Excel were utilized for data processing and statistical analysis.
Results and Discussion
Silver reduces the bioburden within the dressing
Studying the rate of microbial growth within the dressing material is of interest in this work, given our hypothesis that the presence of ionic silver can inhibit microbial growth within the dressing material. Pathogens commonly present in infected wound bed and surgical site such as S. aureus (Gram-positive bacteria), P. aeruginosa (Gram-negative bacteria), and Candida spp. (yeast) were inoculated in the ROCF-ciNPT skin interface layer. 18 The in vitro microbial growth was monitored over 7 days, consistent with the maximum ROCF-ciNPT application period, and was compared against current standard of care consisting of silver-free, sterile gauze (negative control). The mean log values for CFU per sample were plotted as a function of time as shown in Fig. 2. As indicated in the figure, no significant differences were observed between the negative control and ROCF-ciNPT skin interface layer at time 0. However, on day 7, the population of S. aureus, P. aeruginosa, and Candida spp. increased by ∼1.3, ∼2.4, and ∼0.6 log units, respectively, in the negative control compared to day 0. In contrast, the population of S. aureus, P. aeruginosa, and Candida spp. significantly decreased by ∼4.9, ∼3.8, and ∼2.7 log units, respectively, in the ciNPT skin interface layer compared to day 0. The ciNPT skin interface layer contains an inorganic silver carrier in the form of silver hydrogen zirconium phosphate, which releases Ag+ ions during the ionic equilibrium exchange process. 19 The reduction in microbial growth in the presence of such Ag+ is consistent with known antimicrobial properties of the silver. 12,20,21 Overall, these results indicate the ability of ionic silver, which is present within the ROCF-ciNPT skin interface layer, to reduce the bioburden within the dressing, which may arise during the negative pressure treatment.
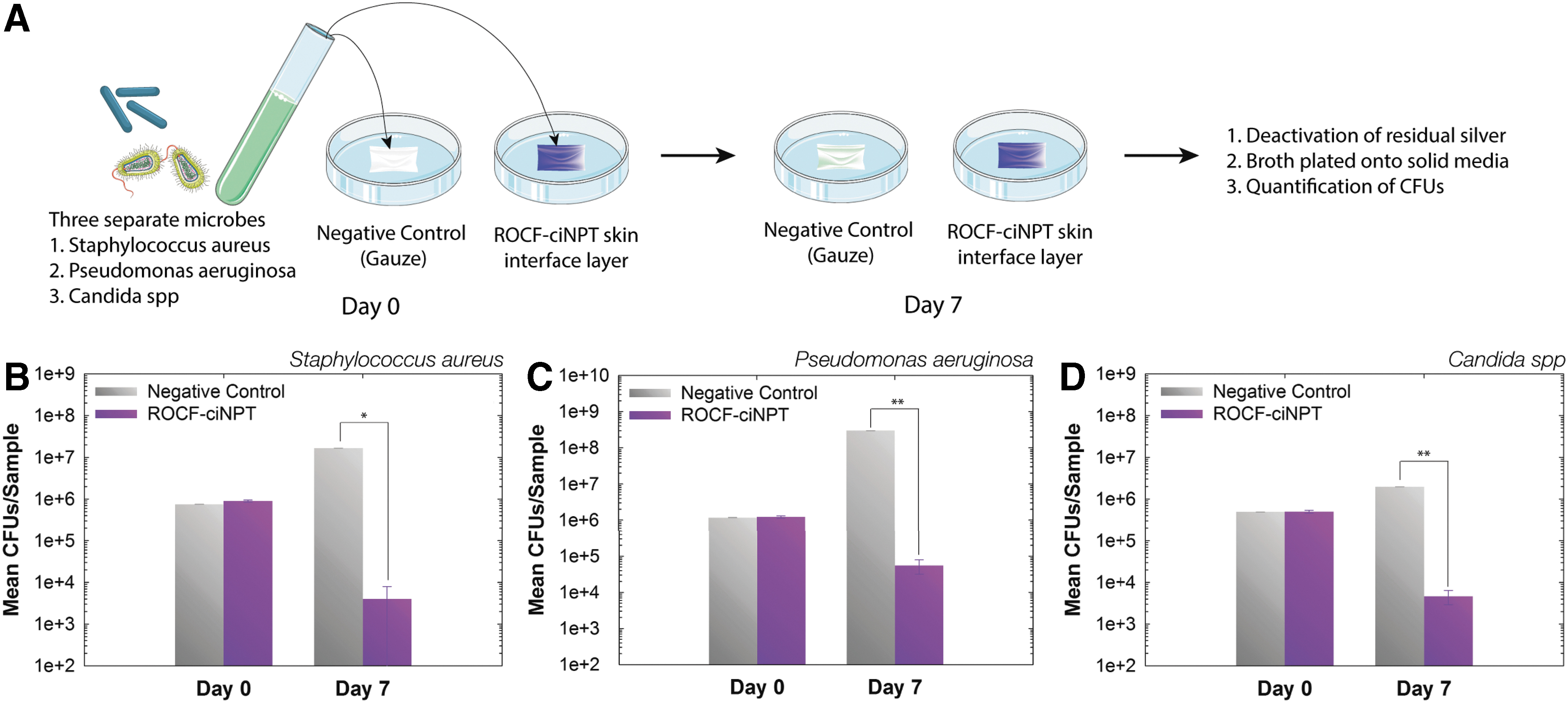
Reduction in bioburden within the ROCF-ciNPT dressing.
Limited silver mobility and subsequent zone of inhibition
While antimicrobial properties of silver are beneficial for the management of infected wounds and surgical sites, the potential risks associated with silver need to be carefully considered. For example, it has been reported that at a higher concentration of silver nanoparticles, undesirable side effects, including argyria and limited proliferation and metabolic activity of keratinocytes, were observed. 22 –24 As indicated earlier, the motivation behind inclusion of silver in the ROCF-ciNPT skin interface layer is to minimize the microbial growth within the dressing itself, and not to induce a localized antimicrobial effect (i.e., at the wound site or incision site) to prevent and/or manage the infection at the surgical site. Thus, we set to determine the effect of silver present in the ROCF-ciNPT skin interface layer on microbial growth when it is placed in direct contact with a microbial colony. Agar substrate containing 98.5% water by weight was used as a skin model. 25
To design the worst-case scenario from a clinical perspective (i.e., when negative pressure is disrupted with no directional fluid flow away from the incision site to the canister), a skin model was employed with (1) higher water content when compared to human epidermis where the water content is typically as high as ∼70%, given that ionic aqueous environment is the driving factor for silver ion release from the silver hydrogen zirconium phosphate in the ROCF-ciNPT skin interface layer, and (2) the ROCF-ciNPT skin interface layer was placed under ambient pressure condition (i.e., no negative pressure). 26,27
Briefly, S. aureus (Gram-positive bacteria), P. aeruginosa (Gram-negative bacteria), and C. albicans (yeast) were independently inoculated on the agar substrate and the ROCF-ciNPT skin interface layer was placed gently on top of it. A commercially available dressing (AC) that is known to have antimicrobial effects with sustained silver release up to 7 days was used as a positive control and inoculation without any test article served as a negative control. 17 The area where no microbial growth occurred immediately surrounding the textile dressing material was then measured at 3 h and on day 1, 3, 5, and 7 to determine if bacterial and yeast growth were inhibited.
The zone of inhibition (ZOI) values for each microbial colony were measured and are shown in Table 1 (additional time points available in Supplementary Data, Supplementary Table S1). Representative images for the ZOI studies for the ROCF-ciNPT skin interface layer are shown in Fig. 3 and representative images for the experimental controls are shown in Supplementary Fig. S1. For S. aureus and P. aeruginosa, growth was observed in the area immediately surrounding the ROCF-ciNPT skin interface layer throughout the experimental duration. In contrast, the positive control exhibited ∼0.5–2 mm ZOI through day 3, while limited ZOI (<0.5 mm) was observed on day 5 and 7 for S. aureus and ∼0.5–2 mm ZOI through day 5 and limited ZOI (<0.5 mm) on day 7 for P. aeruginosa. For the Candida spp., growth was observed in the area immediately surrounding the ROCF-ciNPT skin interface layer sample with no ZOI by day 3 until day 7. A small ZOI (0.5–1.0 mm) on day 1 was observed, which may be attributed to the slower rate of growth for yeast. In comparison, the positive control exhibited a ZOI of 2–4 mm on day 1, 1–3 mm on day 3, 1–2 mm on day 5, and 1–2 mm on day 7. No ZOI was observed for the negative control for S. aureus, P. aeruginosa, and Candida spp. throughout the experiment duration.
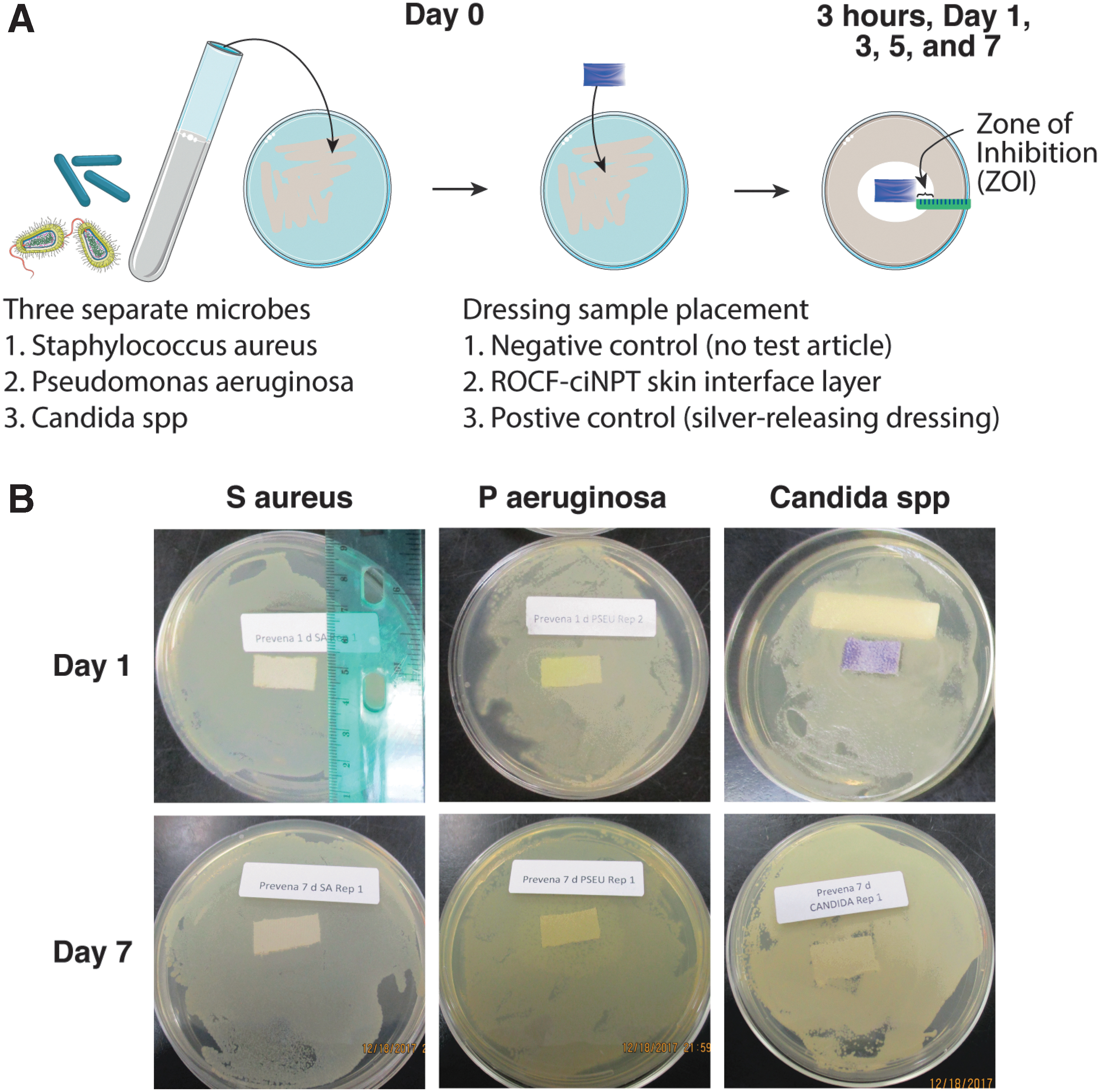
Limited silver mobility results in lack of zone of inhibition outside of dressing.
Zone of inhibition
ROCF-ciNPT, reticulated open-cell foam-based closed-incision negative pressure therapy.
Overall, these results suggest that the amount of silver ions that were transferred from the ROCF-ciNPT skin interface layer to the surface of the agar-based skin model directly underneath, as well as to the surrounding areas does not inhibit microbial growth. Since silver is known to antagonize microorganism growth, the presence of bacterial or fungal growth on areas adjacent to the ROCF-ciNPT skin interface layer is suggestive of low silver concentration and limited mobility. 28 Taken together, these results demonstrate that the loading concentration of silver in the skin interface layer is sufficient to reduce the bioburden significantly within the dressing, yet not to have any inhibitory effect on external microbial growth, thus minimizing any potential silver-associated cytotoxicity at the open wound, incision, or intact skin that is in direct contact with the ROCF-ciNPT dressing.
Minimal in vitro silver release
The amount of silver released when in direct contact with the agar-based skin model was investigated by means of inductively coupled plasma mass spectrometry. The amount of silver collected on the agar-based skin model was plotted as a function of time from day 0 to day 7 for the ROCF-ciNPT skin interface layer and compared against the positive control (i.e., AC dressing) and negative control (i.e., no dressing). The data for these experiments are shown in Fig. 4. As indicated in the figure, ∼5.5 ppm silver was released from the ROCF-ciNPT, which was significantly less than the AC dressing in which ∼281.4 ppm silver was released by day 7 at ∼37°C. No significant difference was observed between the ROCF-ciNPT and negative control that lacked any dressing substrate and thus indicating the baseline silver values in the agar substrate of ∼0.14 ppm silver. The concentration of Ag+ that was released from the ROCF-ciNPT skin interface layer is ∼50 × lower compared to positive control (AC), which is known to act locally to induce the antimicrobial effect.
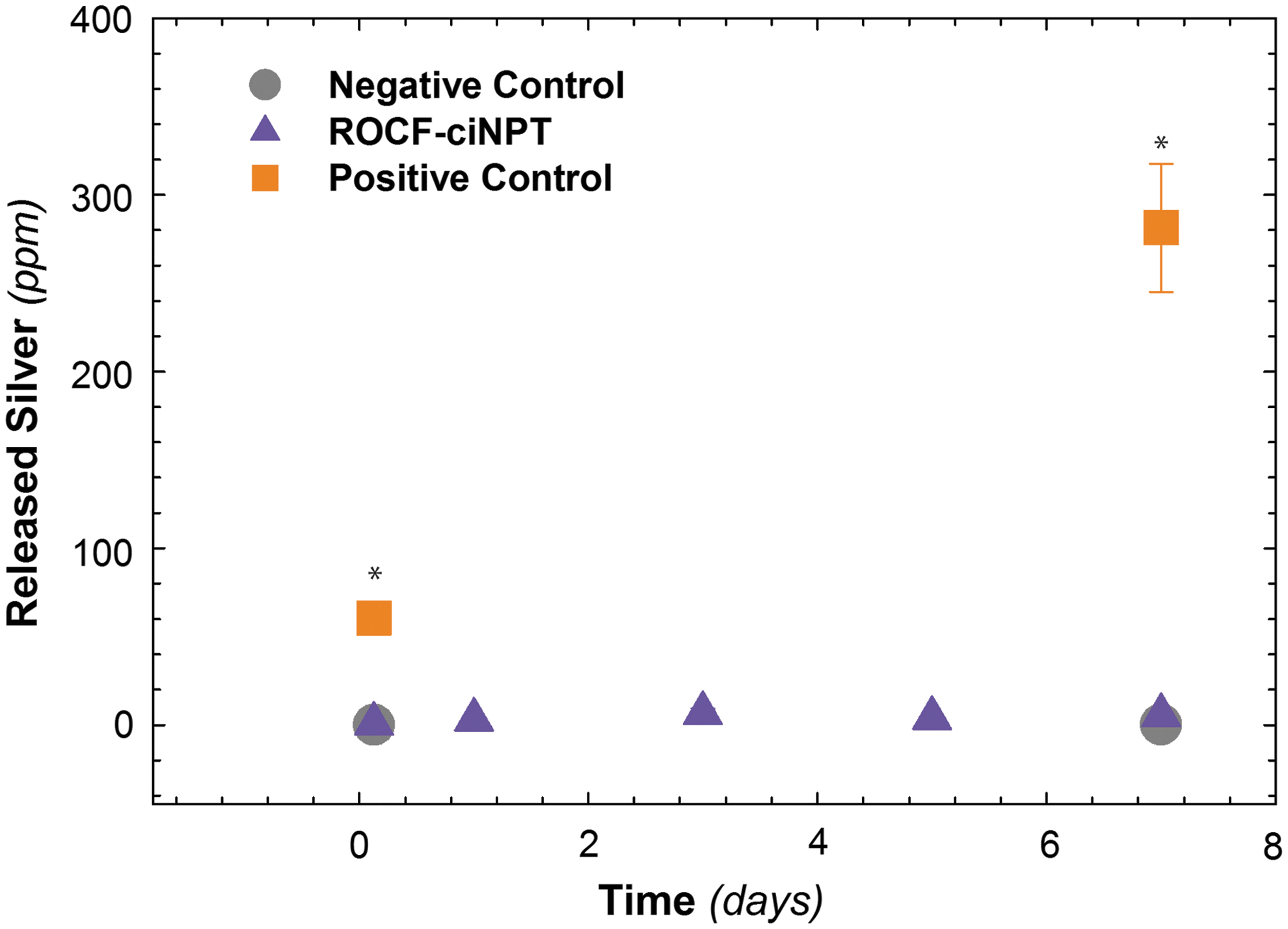
In vitro silver release. Amount of silver released from negative control, ROCF-ciNPT skin interface layer, and positive control (AC dressing) onto the agar-based skin model as a function of time. The data shown illustrate the mean (n = 3) with error bars showing the standard error. *p < 0.05.
Typical commercially available antimicrobial incision and wound dressings contain as high as ∼110 to ∼550 mg/100 cm2 of silver, whereas the ROCF-ciNPT dressing contains ∼0.3 mg/100 cm2 of silver, which is lower by ∼3–4 orders of magnitude. 29 Furthermore, while there are no clear guidelines from published case studies or regulatory agencies indicating a potential correlation between the amount of silver that induces argyria, preliminary studies suggest that ∼3.8–6 g of total body silver concentration can lead to argyria. These values are at least five orders of magnitude higher than the amount of silver present in the ROCF-ciNPT dressing. 15,30 Overall, these results highlight the significantly lower concentration of silver present in the ROCF-ciNPT dressing compared to commercially available dressings that are liable to act locally to induce the antimicrobial effect.
In vivo studies demonstrate negligible silver levels
Next, we sought to study the in vivo silver release under actual use conditions in ROCF-ciNPT dressings using a porcine wound model and compared this to the current standard of care (ME dressing). Briefly, a 20 cm linear paraspinal incision was created, subsequently closed using sutures, and then a ROCF-ciNPT dressing or ME dressing was applied for 7 days (Fig. 5A). As shown in the representative images of incisions in Fig. 5B, the incisions treated with the ROCF-ciNPT dressing appeared completely closed at the termination of treatment based on visual observations. In contrast, the incisions treated with the ME dressing appear nearly closed, although a few small gaps in the wound were observed. These visual observations are consistent with prior studies studying the closure by not only visual observations but also by means of mechanical properties. 31,32 Furthermore, the incisions treated with the ROCF-ciNPT dressing did not show visual signs of silver deposition or staining at the incision site on day 7.
In vivo silver release.
The distribution of silver in the body is dependent on the quantity and route of exposure. Once absorbed, studies have shown that a large amount (>90%) of the absorbed dose is excreted in the feces and urine and the remaining silver in the body that is not excreted tends to deposit in various tissues, including the liver, kidneys, skin, adrenal glands, lungs, pancreas, and spleen. 33 –35 Hence, to assess silver excretion and deposition, the levels of silver in the feces, urine, blood, kidney, and liver were measured for potential exposure and accumulation of silver during and after application of the ROCF-ciNPT dressing compared to ME dressing containing no silver.
Analysis for silver using acid digestion and subsequent ICP-OES revealed that under the conditions of this study, no measurable concentrations (i.e., detection limit <0.2 ppm or lower) of silver were detected in the blood, urine, feces, kidney, or liver tissue at any time point during or at the end of the 7-day treatment for both ROCF-ciNPT dressing and ME dressing containing no silver (Table 2). In contrast, results of spiked samples indicated excellent recovery of silver (≥90%) with the digestion and analysis methods, despite low detection limits (lower detection limit ranging from 0.04 to 0.2 ppm). Furthermore, as shown in Fig. 5C, gross or microscopic test article-related findings were absent in either the kidney or liver tissue for the ROCF-ciNPT dressing as well as the ME dressing. These results are not surprising as Barillo et al. in a recent study using a porcine wound model observed less than 0.05 μg/g of silver in blood, liver, spleen, and kidney using an antimicrobial wound dressing that contained ∼1,600 times higher silver compared to the ROCF-ciNPT dressing. 36
In vivo silver intake over 7-day study duration for ME dressing (negative control) and ROCF-ciNPT dressing
Overall, the negative findings of silver in the in vivo fluids and tissues tested are consistent with our hypothesis that the ROCF-ciNPT dressing contains an adequate amount of silver concentration to reduce the bioburden within the dressing, without any potential localized in vivo cytotoxic effect associated with ionic silver.
Innovation
Considering growing concerns associated with silver cytotoxicity, in this work, we investigated the effect of silver present in the ROCF-ciNPT dressing in an in vitro as well as in vivo wound model. Overall, the results of our study indicate that the silver present in the ROCF-ciNPT dressing is sufficient to control bioburden levels within the dressing throughout a 7-day therapy without any potential localized cytotoxicity and/or overloading of excessive silver within the in vivo wound or incision microenvironment.
Key Findings
The ability of silver to reduce the bioburden within the dressing itself demonstrated effectiveness of silver in inhibiting microbial growth by ∼2.7–4.9 log reduction for microbes commonly found in infected wounds and incisions.
However, given the low loading concentration of silver in the dressing (0.019%), the inhibitory effect of silver was not observed outside of the dressing when the dressing was placed in direct vicinity of microbial colonies grown on an agar-based skin model. Specifically, no zone of inhibition was observed for S. aureus, P. aeruginosa, and Candida spp. for up to 7 days.
Analytical measurements of silver released on the agar-based skin model indicated <5.5 ppm release, which is at least 50 times lower compared to a typical antimicrobial wound dressing.
Furthermore, in vivo studies showed no measurable concentration (i.e., <0.2 μg/g) of silver in the blood, urine, feces, kidney, or liver tissue after biopsy.
Overall our data indicate stability and low mobility of silver ions in the ROCF-ciNPT dressing.
Footnotes
Acknowledgment and Funding Sources
The authors would like to thank Rick Gaykowski, Peter Parsonson, Anona Goebel, Yasi Slavonic, and Tyler Simmons for their inputs toward study design. In addition, the authors would like to thank Marisa Schmidt for assistance with preliminary experiments and animal study, Brenda Marchand, Diwi Allen, and Milliken Healthcare Products R&D team for feedback on earlier version of this article, and Chris Krebs for assistance with figure preparation. The authors also thank the Servier Medical Art for use of a vector image template. This work was funded by KCI, an Acelity Company.
Author Disclosure and Ghostwriting
P.M.K., S.N.O., S.L.S., and K.M.K. are employees of KCI USA, Inc. (San Antonio, TX). A.P. is a former employee of FSNS, Inc. (San Antonio, TX). K.M.W. is a former employee of Milliken Healthcare (Spartanburg, SC).
About the Authors
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
Abbreviations and Acronyms
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
