Abstract
Introduction:
Laparoscopic retroperitoneal lymph node dissection (LRPLND) has become an accepted staging method for clinical stage I nonseminomatous germ cell testicular tumor. 1,2 As a result of gained experience, it is used for both diagnostic and therapeutic purposes in patients with postchemotherapy residual tumor masses. First published series have reported high number of intraoperative and postoperative morbidities with high conversion rates. 3,4 Herein we present our technique, indications, and results in postchemotherapy LRPLND.
Materials and Methods:
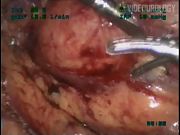
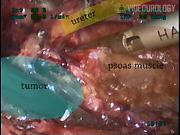
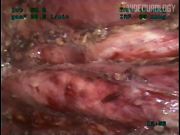
Between January 2005 and July 2009, 19 patients underwent postchemotherapy LRPLND. Patients presenting with unilateral residual tumor masses <6 cm in diameter after chemotherapy, normalization of tumor markers, and having received ≤5 cisplatin-based chemotherapy cycles were considered for the procedure. All patients were operated using modified unilateral template since they had a low volume residual masses. 5 The procedure is described in detail in the video edited from three selected cases illustrating variations in intraoperative findings. Outcomes were analyzed with regard to intraoperative and postoperative complications, hospital stay, operative variables, pathological outcome, and oncological follow-up.
Results:
All operations were completed laparoscopically. Mean tumor diameter after chemotherapy was 3.1 cm. In two patients minor intraoperative vascular injuries occurred, which were successfully controlled by laparoscopic techniques. Postoperatively, one patient had prolonged lymphorrhea in 10 days, which was managed by low-fat dietary measures. Antegrade ejaculation was preserved in all patients. Mean postoperative hospital stay was 4 days. Pathohistological examination revealed mature teratoma in 10 patients and necrosis in 9 patients. All patients were without in-field or distal recurrence during a mean follow-up of 30 months.
Conclusions:
Postchemotherapy LRPLND is a challenging procedure, mostly because of desmoplastic effect of chemotherapy. However, in selected patients it is a safe, oncologically effective, low morbidity procedure with all the benefits of minimally invasive surgery.
All authors state that no competing financial interests exist.
Runtime of video: 9 min 43 sec
Keywords
Get full access to this article
View all access options for this article.
