Abstract
Abstract
Introduction:
Unresectable differentiated thyroid cancer (DTC) at the time of diagnosis is a therapeutic challenge given that the first-line interventions, that is, surgery and radioactive iodine (RAI), are associated with high morbidity. Chemotherapy and radiotherapy provide poor disease control with significant toxicity. Fortunately, the advent of molecular profiling and targeted therapy has shown promising results for the treatment of advanced thyroid cancer. The use of targeted therapy or immune checkpoint inhibitors (ICIs) in unresectable DTC to facilitate surgery and RAI therapy remains mostly unexplored.
Case Presentation:
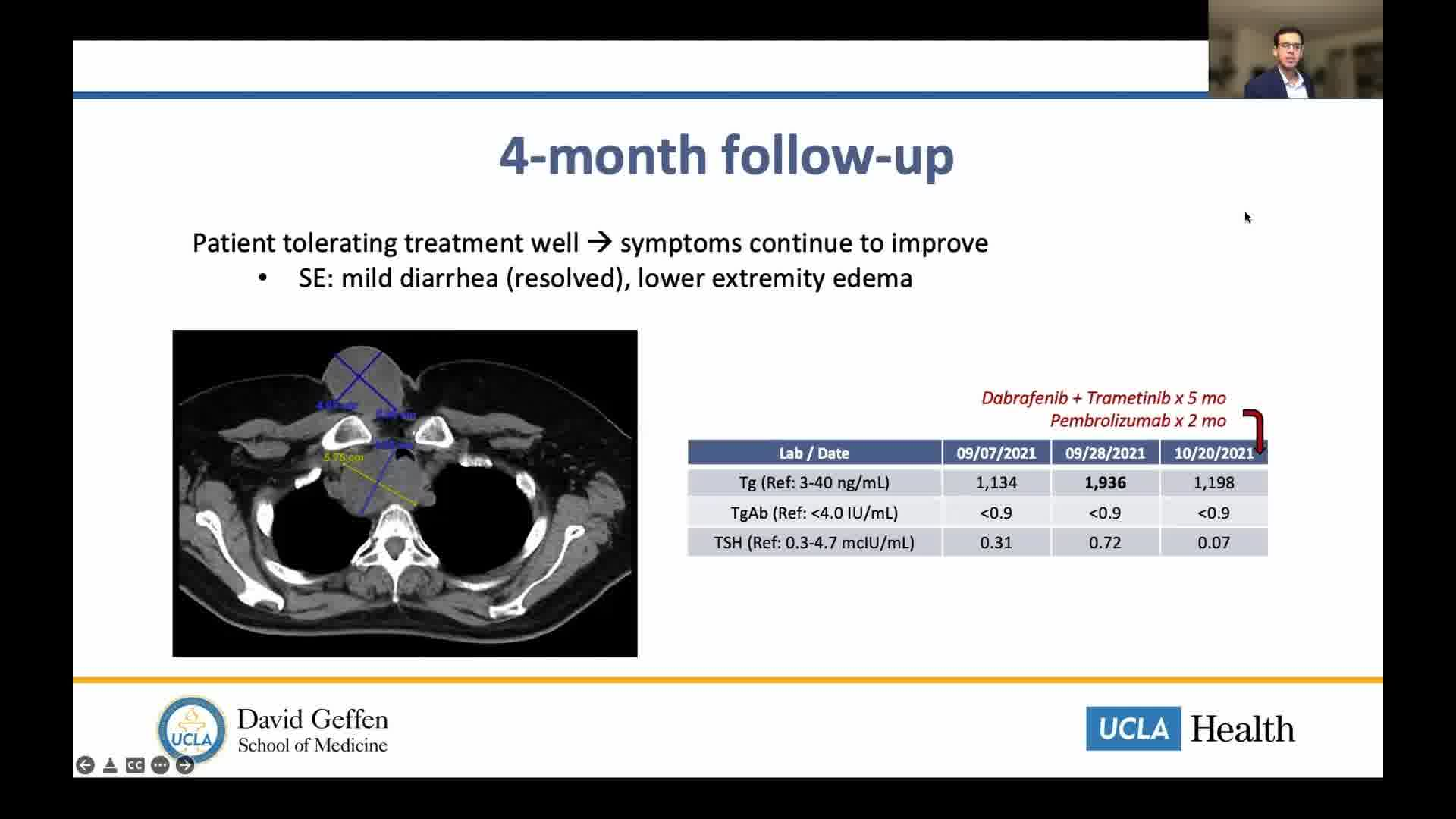
A 59-year-old woman was found to have papillary thyroid cancer (PTC) in 2014. She did not receive treatment at that time because of limited access to health care. She was lost to follow-up and presented in April 2021 with stridor and hemoptysis. CT of the neck showed an 8.5 × 6.1 cm right thyroid mass with infiltration of the larynx, proximal trachea, and cervical esophagus, as well as prominent bilateral level IV cervical lymph nodes. In addition, CT of the chest showed multiple subcentimeter pulmonary nodules concerning for metastases. She was evaluated by otolaryngology, endocrine surgery, and thoracic surgery; however, risks and morbidity of tumor resection were thought to outweigh its benefits because of extensive tracheal and esophageal invasion. Biopsy of the mass showed PTC without anaplastic features, and molecular testing was positive for BRAF-V600E mutation; immunohistochemistry for programmed death-ligand 1 was also positive (combined positive score >20). Given patient's poor candidacy for surgery, RAI, and radiotherapy, targeted therapy with dabrafenib and trametinib was started. Laboratories before starting therapy showed thyroid stimulating hormone 1.4 mcIU/mL (reference range [RR]: 0.3–4.7), thyroglobulin (Tg) 3851 ng/mL (RR: 3–40), and thyroglobulin antibody <0.9 IU/mL (RR: <4.0). At 1-month follow-up, the patient had decreased neck pressure and improved stridor, along with a significant drop in Tg to 659 ng/mL. Two months later, Tg level had plateaued in the 600 to 700 range. Immunotherapy with pembrolizumab was added. Patient was tolerating treatment well, with only side effects being mild diarrhea and lower extremity edema. CT of neck/chest 4 months later shows interval decrease in size of mass and tracheal luminal narrowing. Plan is to continue combined therapy until surgical resection becomes feasible; if not the case, placement of tracheal stent followed by radiotherapy is an alternative.
Discussion:
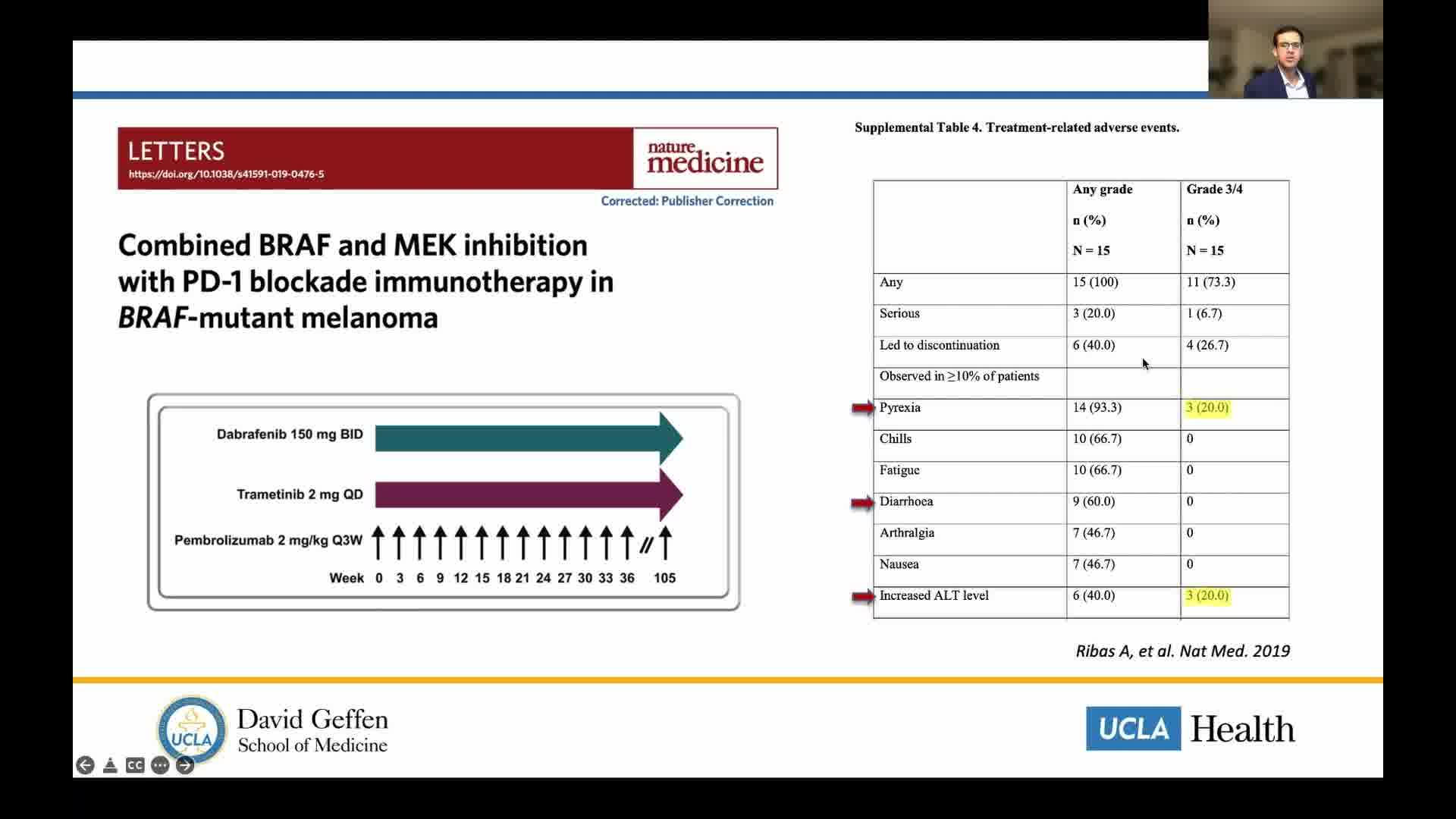
This case illustrates the potential utility of neoadjuvant targeted therapy using combined BRAF/MEK inhibition in BRAF-V600E-positive locoregionally advanced unresectable DTC. Previously, this approach has shown efficacy in BRAF-mutated anaplastic thyroid cancer (ATC). In contrast, use of ICI in conjunction with multikinase inhibitors (such as sorafenib and lenvatinib) has shown to potentiate response in advanced and refractory tumors compared with either therapy alone. However, combination of ICI and targeted molecular therapy has not been studied in DTC, and less so in the neoadjuvant setting. Despite differences in microenvironment and cell differentiation between ATC and DTC, we anticipate that targeting shared oncogenic drivers, such as BRAF, could be an effective therapeutic strategy in both entities.
No competing financial interests exist
.
Patient Consent:
Authors have received and archived patient consent for video recording/publication in advance of video recording of procedure.
Runtime of video: 19 mins 18 secs

Get full access to this article
View all access options for this article.
