Abstract
Background:
Rearranged during transfection (RET) alterations are targetable oncogenic drivers in thyroid cancer. Primary data from the open-label, phase 1/2 ARROW study demonstrated clinical activity and manageable safety with pralsetinib, a selective RET inhibitor, in patients with advanced/metastatic RET-altered thyroid cancer. We present an updated analysis with more patients and longer follow-up.
Methods:
Adult patients with advanced/metastatic RET-mutant medullary thyroid cancer (MTC) or RET fusion-positive thyroid cancer who initiated oral pralsetinib at 400 mg once daily were included. Primary endpoints were overall response rate (ORR) by blinded independent central review (per RECIST v1.1) and safety. Secondary endpoints included duration of response (DoR), progression-free survival (PFS), and overall survival. Responses were assessed in three cohorts of patients with baseline measurable disease: patients with RET-mutant MTC who had received prior cabozantinib and/or vandetanib (C/V), treatment-naïve patients with RET-mutant MTC, and patients with previously treated RET fusion-positive thyroid cancer. Patient-reported outcomes (PROs) were an exploratory endpoint.
Results:
As of October 18, 2021, the measurable disease population comprised of 61 patients with RET-mutant MTC and prior C/V, 62 treatment-naïve patients with RET-mutant MTC, and 22 patients with RET fusion-positive thyroid cancer who had received prior systemic therapy, including radioactive iodine. The ORR was 55.7% [confidence interval; 95% CI: 42.4–68.5] in patients with RET-mutant MTC and prior C/V, 77.4% [95% CI: 65.0–87.1] in treatment-naïve patients with RET-mutant MTC, and 90.9% [95% CI: 70.8–98.9] in patients with previously treated RET fusion-positive thyroid cancer. Median DoR and median PFS were both 25.8 months in patients with RET-mutant MTC and prior C/V, not reached in treatment-naïve patients with RET-mutant MTC, and 23.6 and 25.4 months, respectively, in patients with previously treated RET fusion-positive thyroid cancer. In the RET-altered thyroid cancer safety population (N = 175), 97.1% of patients reported a treatment-related adverse event (TRAE); these led to discontinuation in 5.7% and dose reduction in 52.6% of patients. There was one death (0.6%) due to a TRAE. PROs improved or remained stable after pralsetinib treatment.
Conclusions:
In this updated analysis of the ARROW study, pralsetinib continued to show deep and durable clinical activity and a manageable safety profile in patients with advanced/metastatic RET-altered thyroid cancer. Clinical Trial Registration: NCT03037385
Introduction
Activating alterations in the rearranged during transfection (RET) proto-oncogene are known oncogenic drivers in medullary thyroid cancer (MTC) and differentiated thyroid cancer. 1,2 MTC accounts for ∼1–4% of all thyroid cancer cases and can be either sporadic (∼75%) or hereditary (∼25%). 3 –5 RET mutations are found in ∼50% of sporadic MTC (potentially more frequently in advanced/metastatic disease), and nearly all cases of hereditary MTC. 6,7 Despite being rare, MTC accounts for >10% of thyroid cancer-related deaths. 8 Papillary thyroid cancer is the most common type of differentiated thyroid cancer (80–85%) 9 and an estimated 10–20% of these patients harbor a RET fusion. 7 RET fusions are less common (<10%) in other types of thyroid cancer such as follicular and anaplastic thyroid cancer. 10
Multikinase inhibitors (MKIs) are the standard of care for both advanced MTC (cabozantinib, vandetanib) and radioactive iodine-refractory differentiated thyroid cancer (lenvatinib, sorafenib, and cabozantinib). 11 However, these MKIs are associated with significant adverse events (AEs) that result in a high incidence of dose reductions and treatment discontinuations, 12 –15 and potentially affect patient health-related quality of life (HRQoL).
Two selective RET kinase inhibitors, selpercatinib and pralsetinib, have been approved in the United States for the treatment of advanced/metastatic RET-altered thyroid cancer. 16,17 Pralsetinib (formerly BLU-667, Blueprint Medicines) is a highly potent, oral, once-daily (QD), selective RET inhibitor. 7,16 We have previously published data from the phase 1/2 ARROW trial (data cutoff: May 22, 2020), 18 showing that pralsetinib 400 mg QD has clinical activity and manageable safety in patients with RET-altered thyroid cancer. Overall response rates (ORRs) by blinded independent central review (BICR) were 60% in patients with RET-mutant MTC who had previously received cabozantinib and/or vandetanib (C/V), 71% in patients with treatment-naïve RET-mutant MTC, and 89% in patients with previously treated RET fusion-positive thyroid cancer.
Here, we present updated efficacy and safety data with a larger number of patients and longer follow-up, along with the first report of patient-reported outcomes (PROs) from this study.
Materials and Methods
Study design and patients
ARROW is a global, open-label, first in-human phase 1/2 study of pralsetinib; the study design was described previously (protocol available online). 18 The enrollment cutoff for this analysis was February 18, 2021, and the data cutoff was October 18, 2021. The study population included patients with RET-mutant MTC with/without prior systemic treatment and patients with previously treated RET fusion-positive thyroid cancer.
This study was conducted in accordance with the ethical principles of Good Clinical Practice and the Declaration of Helsinki and based on the International Council for Harmonisation E6 requirements. The study adhered to the legal requirements of the countries where it was performed. The protocol was approved by the institutional review boards at all sites, and all patients provided signed informed consent. Safety was initially monitored by a safety review committee consisting of investigators and sponsor representatives. An independent data monitoring committee was established during the phase 2 part of the study (following protocol amendment 9; July 3, 2019).
Treatment and assessments
In the phase 2 dose-expansion part of the ARROW study, patients were initiated with pralsetinib treatment at 400 mg QD until disease progression (PD), intolerance, consent withdrawal, or investigator decision. Dose reductions for study drug-related toxicities were permitted in 100 mg decrements. Treatment was discontinued if a patient required a dose reduction to <100 mg QD; doses could be interrupted for study drug-related toxicities for a maximum of 28 days. Specific guidelines on dose modification are provided in the published protocol. 18
Tumor assessments were performed by BICR per RECIST v1.1; the reviewers were blinded to treatment cohort (phase 1), the patient's treatment status, and the results from other reviewers. RET alterations were identified by local testing: DNA or RNA sequencing using tumor tissue or blood, or fluorescent in situ hybridization in tumor tissue. Assessment of known sites of disease by computed tomography scan or magnetic resonance imaging was performed at screening, then approximately every 8 weeks (2 cycles) until Cycle 17, followed by every 16 weeks (4 cycles) thereafter. For patients with MTC, serum calcitonin and carcinoembryonic antigen (CEA) concentrations were followed longitudinally (by central laboratory assessment).
European Organization for Research and Treatment of Cancer (Quality of Life of Cancer patients; EORTC QLQ-C30) questionnaires were completed at baseline (before treatment initiation) and on day 1 of each cycle (until Cycle 12). EORTC QLQ-C30 is a 30-item questionnaire comprising scales for global health status (GHS/QoL), physical, cognitive, role, emotional, and social functioning, and disease and/or treatment-related symptoms (e.g., diarrhea). 19 Scores range from 0 to 100; higher scores for GHS/QoL or functioning scales denote better HRQoL; higher scores for symptom scales indicate poorer HRQoL. Patients who did not complete the questionnaire at baseline were not asked to complete it subsequently.
AEs were graded according to the US National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.03). Safety clinical laboratory evaluations were carried out at local laboratories, following the protocol-specified schedules.
Outcomes
The phase 2 primary endpoints were ORR (proportion of patients with a complete or partial response [CR/PR]) in the measurable disease population (MDP) and safety. Key secondary endpoints included duration of response (DoR), clinical benefit rate (proportion of patients with confirmed CR, PR, or stable disease [SD] of ≥16 weeks), disease control rate (proportion of patients with CR, PR, or SD), progression-free survival (PFS), and overall survival (OS). Efficacy endpoints (with the exception of OS) were assessed by BICR, per RECIST v1.1.
Biochemical response rates for serum calcitonin and CEA were reported in patients with RET-mutant MTC. These were defined as the proportion of patients with a biochemical confirmed CR or PR for ≥2 assessments ≥28 days apart (and no PD in between) before PD and/or other anticancer therapy. Biochemical responses were defined as follows: biochemical CR, within the normal range after treatment and confirmed ≥4 weeks later; biochemical PR, ≥50% decrease maintained for ≥4 weeks; biochemical SD, between −50% and +50% change maintained for ≥4 weeks; biochemical PD, ≥50% increase maintained for ≥4 weeks. Patients with normal serum levels at baseline were not included in these analyses.
PROs were an exploratory endpoint (introduced in protocol v4.1); score changes >10 points from baseline were considered clinically meaningful. 20
Statistical analysis
Three patient populations were used in this analysis: RET-altered thyroid cancer safety population, which included all patients with RET-altered thyroid cancer who initiated pralsetinib at 400 mg once daily by the data cutoff (October 18, 2021); the intention-to-treat (ITT; efficacy) population, comprising all patients with RET-mutant MTC or RET fusion-positive thyroid cancer dosed by the enrollment cutoff (February 18, 2021); and the MDP population, comprising all patients in the ITT population who had measurable baseline disease (RECIST v1.1; by BICR). Safety data are also reported in the overall safety population, which included all patients from ARROW (any tumor type) initially dosed at 400 mg QD.
Tumor response was evaluated in both the MDP (primary population) and the ITT population; PFS, OS, and biochemical response were assessed in the ITT population. PROs were assessed in the ITT population for patients who consented and had their first treatment on or after Protocol Amendment 4.1.
Two-sided 95% confidence intervals [CIs] were based on exact binomial distributions through the Clopper–Pearson method. The Kaplan–Meier method was used to estimate time-to-event endpoints. Estimates of duration of follow-up were based on the inverse Kaplan–Meier method, with 95% CIs based on the Greenwood formula. All statistical analyses were performed using SAS version 9.4.
Results
Patients
At the data cutoff, the ITT population included 145 patients with RET-mutant MTC with/without prior systemic treatment and 25 patients with RET fusion-positive thyroid cancer who had received prior systemic therapy, including radioactive iodine. Of the patients with RET-mutant MTC, 67 had received prior C/V, 11 had received other prior systemic therapy, and 67 were systemic-treatment-naïve (Fig. 1). Median age ranged between 55 and 60 years, and most patients had an Eastern Cooperative Oncology Group performance status of 0 or 1. The most prevalent mutation was M918T and the most prevalent RET fusion partner was CCDC6 (Table 1).
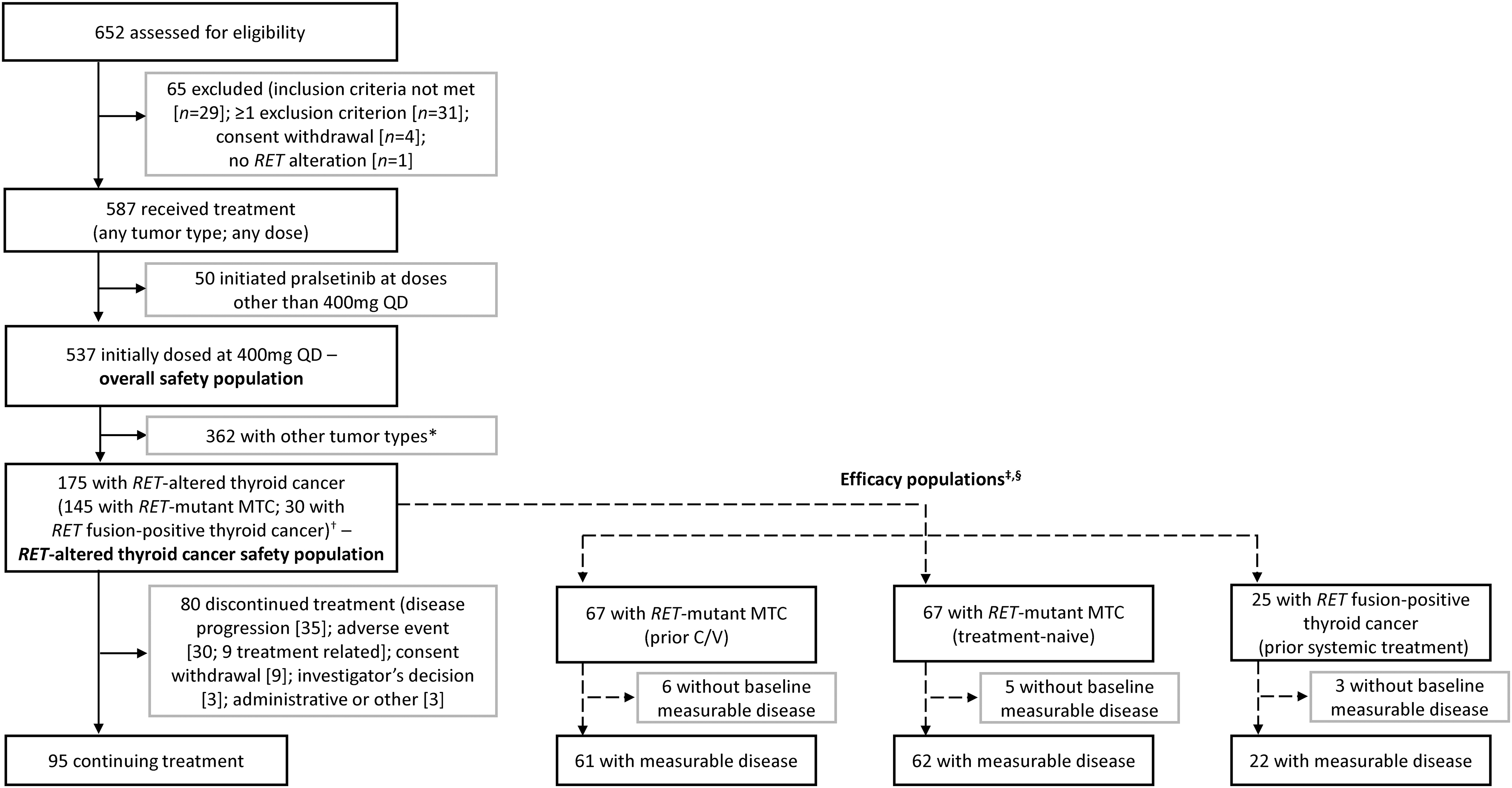
Patient flow diagram. *Includes patients with RET fusion-positive NSCLC or other solid tumors outside of thyroid cancer, patients with MTC with no documented RET mutation, patients with RET-mutant solid tumors outside of thyroid, and patients who had previous treatment with a selective RET inhibitor (selpercatinib); †all patients with RET-altered thyroid cancer who initiated pralsetinib at 400 mg QD by October 18, 2021; ‡patients who enrolled by February 18, 2021; §11 patients with MTC had a prior systemic treatment other than C/V; three patients with RET fusion-positive thyroid cancer were enrolled after February 18, 2021, and therefore were not included in the efficacy population; two patients with RET fusion-positive thyroid cancer had had no prior systemic treatment. C/V, cabozantinib and/or vandetanib; MTC, medullary thyroid cancer; NSCLC, nonsmall cell lung cancer; QD, once daily; RET, rearranged during transfection.
Demographics and Baseline Characteristics of Patients with Rearranged During Transfection-Altered Thyroid Cancer Who Initiated Pralsetinib at 400 mg QD (Intention-To-Treat Population)
Of the 25 patients with RET fusion-positive thyroid cancer with prior systemic treatment, 23 had papillary thyroid cancer, 1 had anaplastic thyroid cancer, and 1 had poorly differentiated thyroid cancer. Patients who received cabozantinib and/or vandetanib may have been treated with prior cabozantinib only, vandetanib only, or both cabozantinib and vandetanib; similarly, patients who received lenvatinib and/or sorafenib may have been treated with prior lenvatinib only, sorafenib only, or both lenvatinib and sorafenib.
Italics were used to indicate that these treatments were sub-categories of MKI.
An ECOG PS of 2 was permitted before a protocol amendment.
Three patients classed as having M918T as the primary mutation also had a V804L/M mutation.
Includes patients with single-nucleotide variants of or short indels that include but are not limited to C609, C611, C618, C620, C630, and/or C634 of RET.
Includes D898_E901del, L790F, A883F, K666E, and R844W.
Includes ACBD5, DLG5, and SNRNP70.
CNS, central nervous system; C/V, cabozantinib and/or vandetanib; ECOG PS, Eastern Cooperative Oncology Group performance status; ITT, intention to treat; MKI, multikinase inhibitor; MTC, medullary thyroid cancer; QD, once daily; RET, rearranged during transfection; TNM, tumor/node/metastasis.
Overall efficacy
In patients with RET-mutant MTC who had received prior C/V, the median duration of treatment was 25.4 months (range: <1 to 45.0). In the MDP, ORR was 55.7% (n = 34/61 [95% CI: 42.4–68.5]); one patient (1.6%) had a CR and 33 (54.1%) had a PR (Table 2); median time to response was 3.8 months (range: 1.8–20.2) (Supplementary Fig. S1A), and median DoR was 25.8 months [95% CI: 18.0–not estimable; NE]. Median PFS (ITT population; n = 67) was 25.8 months [95% CI: 19.7–35.0] (Table 2; Fig. 2A).
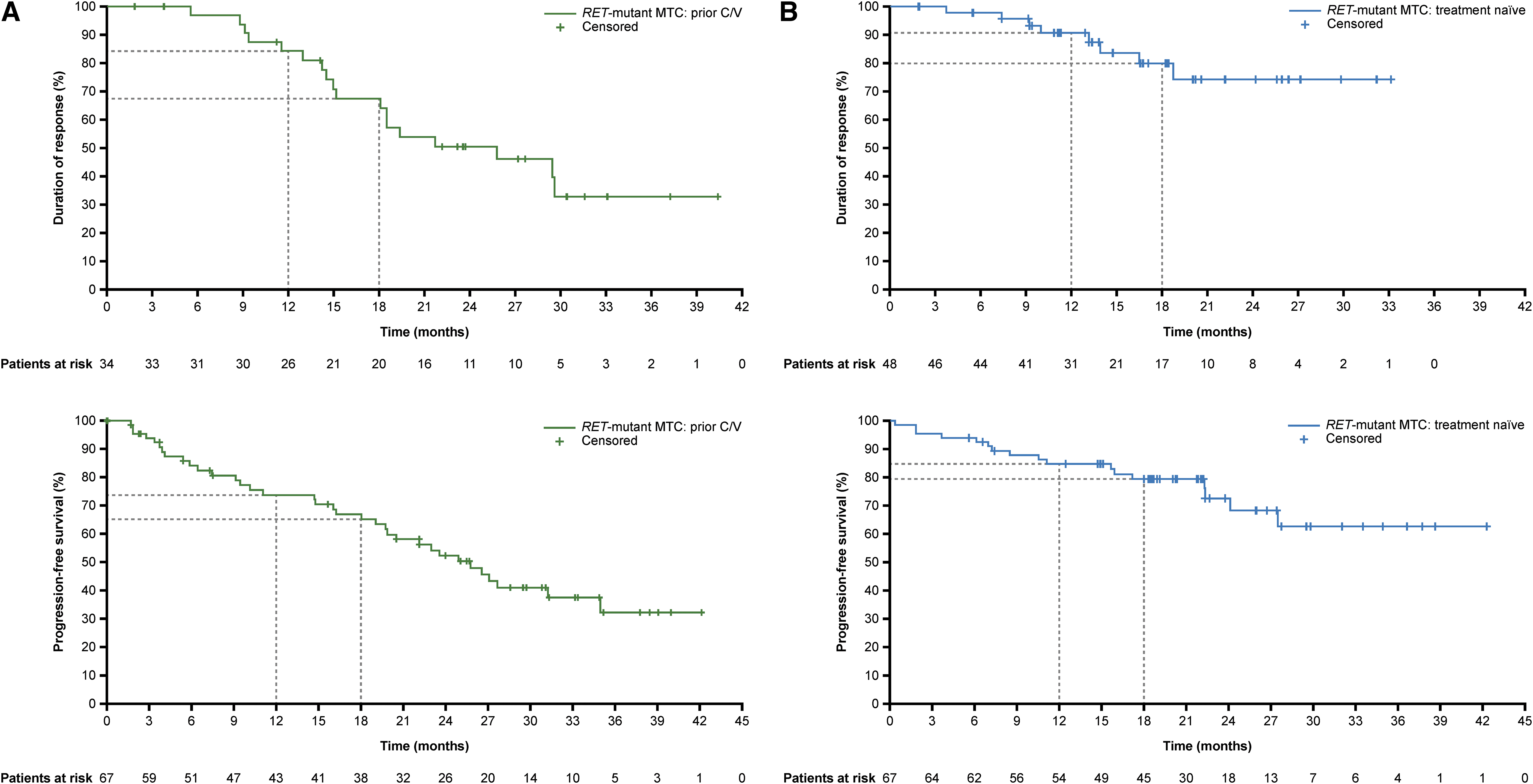
DoR (MDP) and PFS (ITT population) for (
Overall Efficacy with Pralsetinib in Patients with Rearranged During Transfection-Mutant Medullary Thyroid Cancer and Rearranged During Transfection Fusion-Positive Thyroid Cancer Who Initiated Pralsetinib at 400 mg QD
Response endpoints were assessed in the MDP and the ITT population; survival endpoints were only assessed in the ITT population. DoR and PFS results per EMA censoring rules.
Assessed by BICR using RECIST v1.1.
The proportion of patients with the best overall response of confirmed complete or partial response.
The proportion of patients with the best overall response of confirmed complete response, partial response, or stable disease lasting ≥16 weeks.
The proportion of patients with the best overall response of complete response, partial response, or stable disease.
BICR, blinded independent central review; CI, confidence interval; DoR, duration of response; EMA, European Medicines Agency; MDP, measurable disease population; NE, not estimable; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; QD, once daily; RECIST, Response Evaluation Criteria in Solid Tumors; RET, rearranged during transfection.
In patients with treatment-naïve RET-mutant MTC, the median duration of treatment was 22.1 months (range: <1 to 45.1). In the MDP, the ORR was 77.4% (n = 48/62 [95% CI: 65.0–87.1]), including four patients (6.5%) with CR and 44 (71.0%) with PR (Table 2); median time to response was 5.6 months (range: 1.8–18.6) (Supplementary Fig. S1B). Median DoR was not reached (NR [CI: NE–NE]) and median PFS (ITT population; n = 67) was also NR [95% CI: 27.5–NE] (Table 2; Fig. 2B); both DoR and PFS data remain immature at the data cutoff (Table 2).
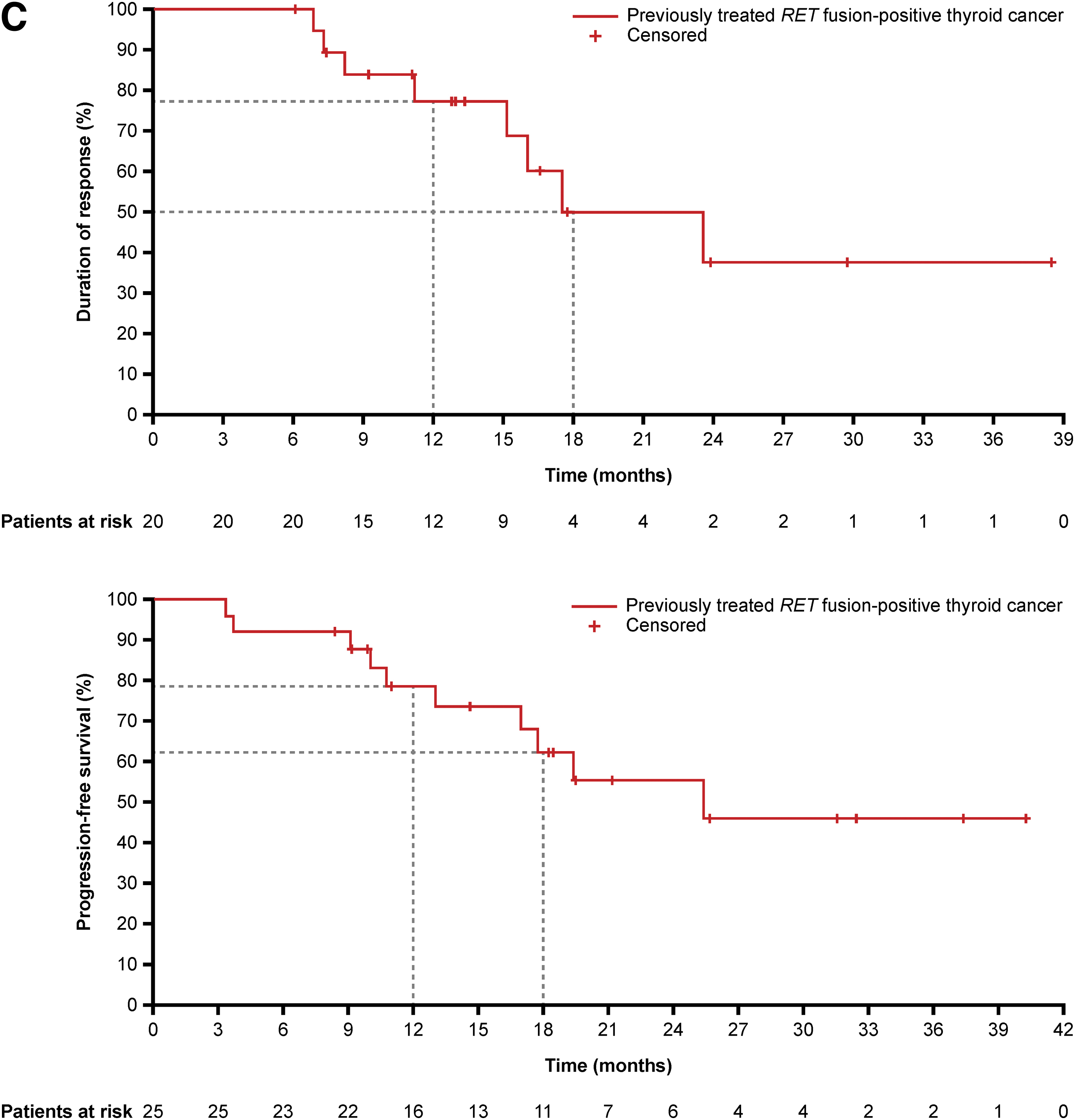
In patients with previously treated RET fusion-positive thyroid cancer, the median duration of treatment was 15.7 months (range: 3.3–43.5). In the MDP, the ORR was 90.9% (n = 20/22 [95% CI: 70.8–98.9]); three patients (13.6%) had a CR and 17 (77.3%) had a PR (Table 2); median time to response was 1.8 months (range: 1.7–7.9) (Supplementary Fig. S1C). Median DoR was 23.6 months [95% CI: 15.1–NE] and median PFS (ITT population; n = 25) was 25.4 months [95% CI: 17.0–NE] (Table 2; Fig. 2C); both DoR and PFS data remain immature at the data cutoff (Table 2).
Responses were seen across the three cohorts regardless of RET mutation genotype or fusion partner (Fig. 3).
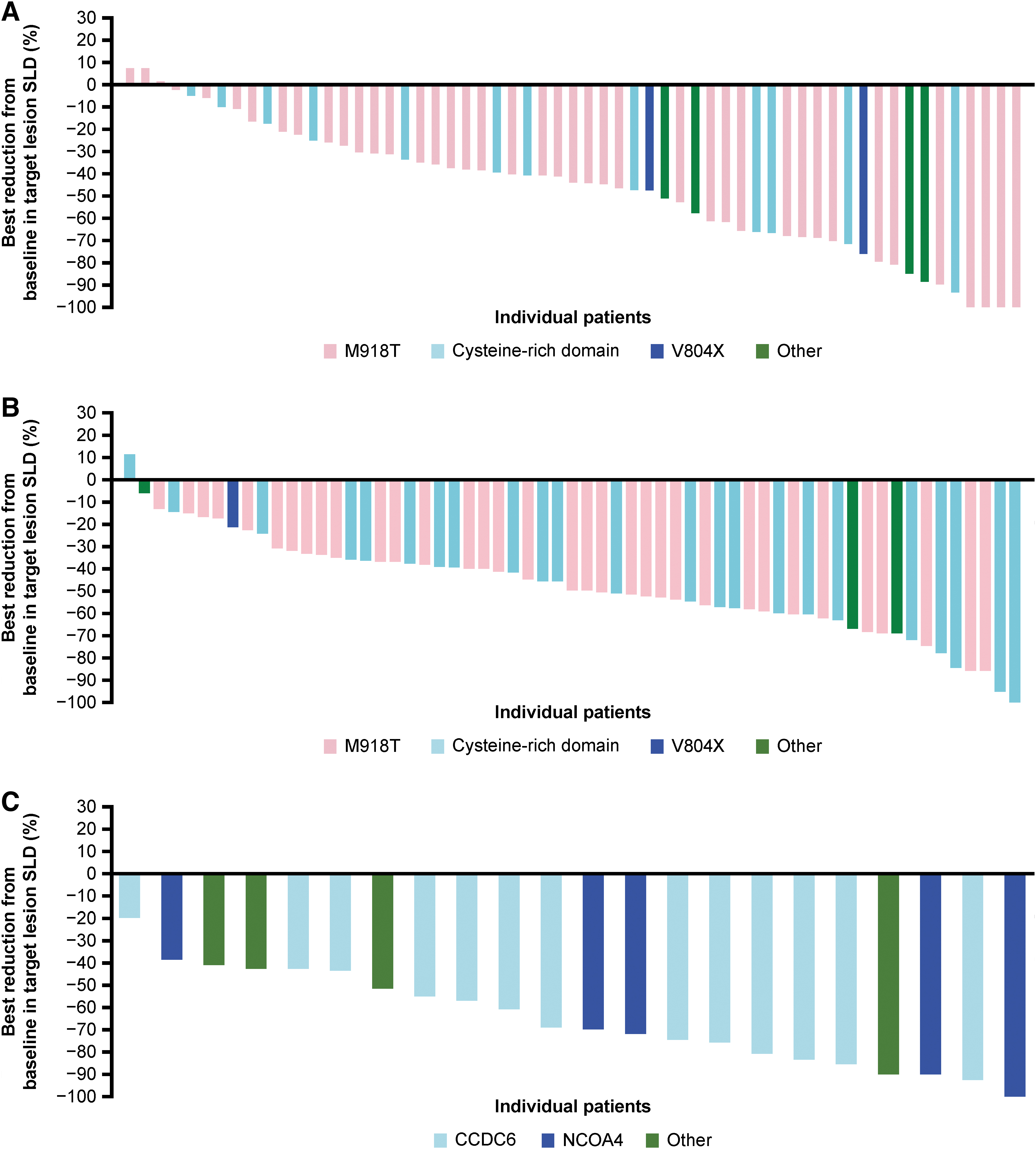
Waterfall plot of maximum percentage change from baseline in target lesion diameter (central radiology assessment per RECIST v1.1) in (
With a median follow-up of 22–31 months in the three cohorts, OS data remain immature and median OS was NR for all cohorts (Table 2; Supplementary Fig. S2A–C). Estimated 12-month OS rates were 88.7% in patients with RET-mutant MTC who had received prior C/V, 94.0% in patients with treatment-naïve RET-mutant MTC, and 87.1% in patients with previously treated RET fusion-positive thyroid cancer (Table 2).
Biochemical responses
In the RET-mutant MTC cohort, detectable baseline concentrations of calcitonin and CEA were reported in 142 and 130 patients, respectively. Concentrations of both markers decreased over time (Supplementary Fig. S3A, B), with biochemical response rates of 85.9% (n = 122/142 [95% CI: 79.1–91.2]) to calcitonin and 63.8% (n = 83/130 [95% CI: 55.0–72.1]) to CEA (Table 3).
Calcitonin and Carcinoembryonic Antigen Responses in Patients with Rearranged During Transfection-Mutant Medullary Thyroid Cancer Who Initiated Pralsetinib at 400 mg QD
Biochemical complete response was defined as a normalization of calcitonin or carcinoembryonic antigen serum levels (i.e., having serum values within the normal range) after treatment, confirmed ≥4 weeks later; partial response was defined as ≥50% decrease from baseline serum levels maintained over ≥4 weeks; stable disease was defined as between −50% and +50% change from baseline in serum levels maintained over ≥4 weeks; progressive disease was defined as ≥50% increase from baseline serum levels maintained over ≥4 weeks. Patients with normal serum levels at baseline were not included in biochemical response rate analyses.
CEA, carcinoembryonic antigen.
Patient-reported outcomes
Baseline PRO assessments were completed by 98/100 RET-mutant MTC patients who consented and had their first treatment on or after Protocol Amendment 4.1. In patients with RET-mutant MTC who had received prior C/V (n = 39), baseline mean GHS/QoL score was 59.0/100 and functioning scores were >68/100 (Table 4). Clinically meaningful improvements in mean scores from baseline were observed during weeks 8–32 for GHS/QoL (Fig. 4A), weeks 24–32 for physical functioning, and weeks 12–36 and 44 for role functioning (Supplementary Fig. S4A). Several disease-related symptoms including diarrhea (mean change from baseline: −20.6; Fig. 4A), fatigue (−14.8), appetite loss (−12.7), and insomnia (−12.7) were improved at week 44 (cutoff applied due to ≤50% of patients remaining in the study at this timepoint; Table 4).
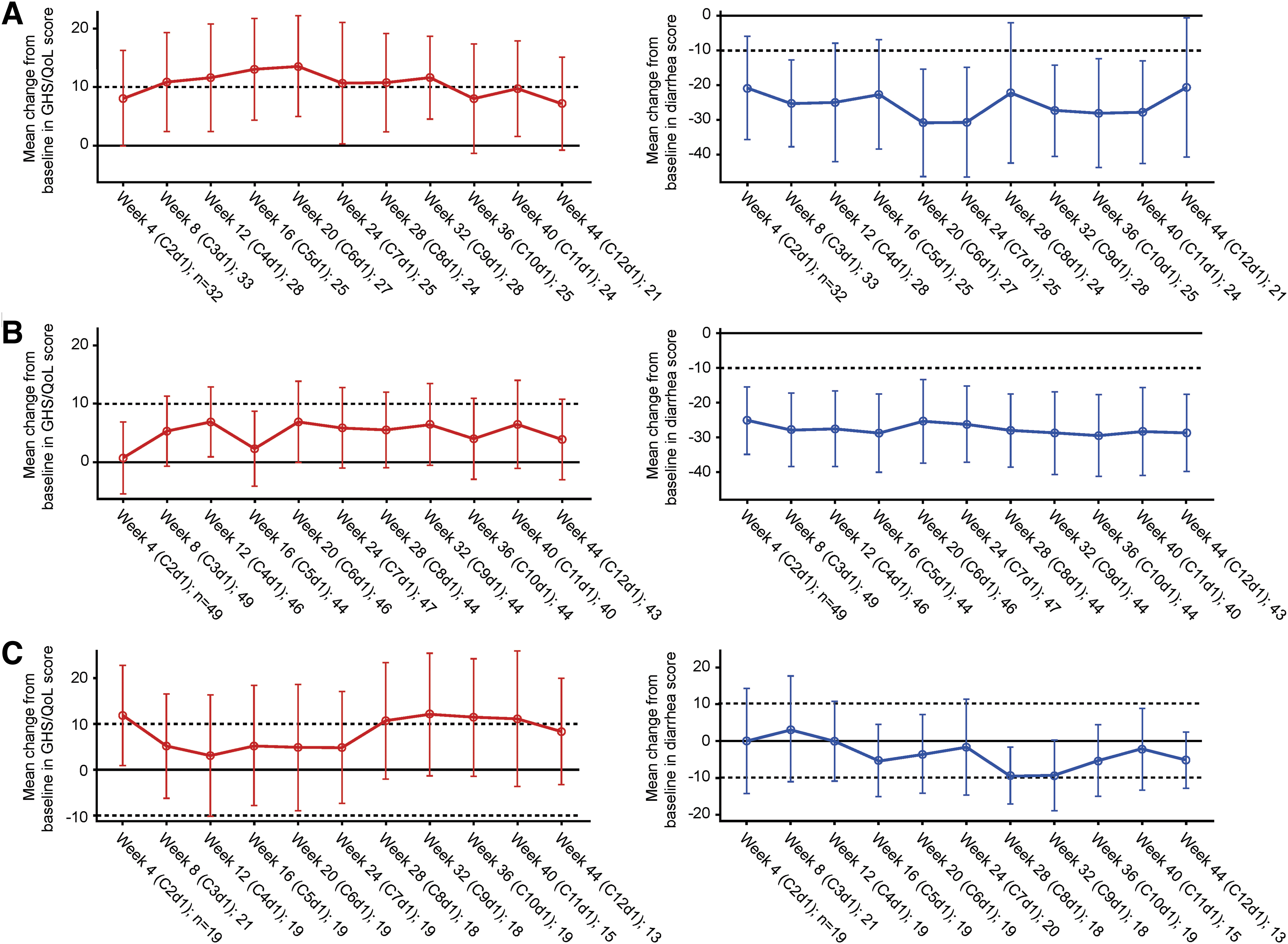
Mean changes from baseline in GHS/QoL and diarrhea scores in (
Health-Related Quality-of-Life Mean Scores at Baseline and Mean Change from Baseline to Week 44, in Patients with Rearranged During Transfection-Mutant Medullary Thyroid Cancer and Previously Treated Rearranged During Transfection Fusion-Positive Thyroid Cancer Who Initiated Pralsetinib at 400 mg QD
Patients who consented and had their first treatment on or after protocol v4.1 (100 efficacy-evaluable patients with RET-mutant MTC and 22 patients with previously treated RET fusion-positive thyroid cancer).
Week 44 cutoff applied due to ≤50% of patients remaining in the study at this timepoint.
EORTC QLQ-C30, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; GHS, global health status; QoL, quality of life.
Treatment-naïve patients with RET-mutant MTC (n = 54) had higher GHS/QoL (mean score: 71.5/100) and functioning scores (mean scores: >82/100) at baseline than those who had received prior C/V (Table 4; Fig. 4B; Supplementary Fig. S4B). PROs in this cohort remained stable after pralsetinib treatment, except for diarrhea, which improved (mean change from baseline at week 44: −28.7; Table 4; Fig. 4B).
Patients with previously treated RET fusion-positive thyroid cancer (n = 22) had moderate GHS/QoL (mean score: 58.7/100) and moderate-to-high functioning scores (mean scores: >68/100) at baseline (Table 4). Clinically meaningful improvements were seen during weeks 28–40 for GHS/QoL and weeks 4 and 40 for role functioning; symptom burden remained low throughout (Fig. 4C; Supplementary Fig. S4C).
Safety
Safety analyses are reported for the RET-altered thyroid cancer safety population (N = 175) and the overall safety population (N = 537). Most patients experienced ≥1 treatment-related AE (treatment-related adverse event [TRAE]; RET-altered thyroid cancer safety population: 97.1%; overall safety population: 93.9%; Table 5). The most frequent all-grade TRAEs were increased aspartate aminotransferase (AST; 40.0%), anemia (36.6%), and hypertension (33.1%) in the RET-altered thyroid cancer safety population and increased AST (41.7%), anemia (38.2%), and increased alanine aminotransferase (31.5%) in the overall safety population.
All-Grade Treatment-Related Adverse Events (by Preferred Term) for the Rearranged During Transfection-Altered Thyroid Cancer Safety Population and the Overall Safety Population (400 mg QD Dose) from the ARROW Study
MedDRA, Medical Dictionary for Regulatory Activities (version 19.1); QD, once daily; RET, rearranged during transfection; TRAE, treatment-related adverse event.
Grade 3–5 TRAEs occurred in 61.1% of patients with RET-altered thyroid cancer and 60.7% of patients overall; the most common TRAEs are summarized in Supplementary Table S1. Treatment-related serious AEs (Supplementary Table S2) occurred in 16.6% of patients with RET-altered thyroid cancer (most common: pneumonitis [2.9%; all resolved]; increased blood creatine phosphokinase, pneumonia, and anemia [all 1.7%]) and 23.3% of patients overall (most common: pneumonitis [4.3%]; anemia [3.4%]; and pneumonia [3.0%]). In the RET-altered thyroid cancer safety population, most patients with treatment-related serious pneumonitis received steroids and/or interrupted treatment until symptoms improved (treatment then resumed at a lower dose).
Deaths due to an AE were reported in 13.1% and 17.1% of patients in the RET-altered thyroid cancer and overall safety populations, respectively; deaths due to a TRAE were reported in one (0.6%) patient in the RET-altered thyroid cancer population and seven (1.3%; pneumonia [n = 2], death [n = 2], and rhabdomyolysis, pneumonitis, and interstitial lung disease [n = 1 each]) patients, in the overall safety population. Dose reductions and discontinuations due to a TRAE occurred in 52.6% and 5.7% of patients, respectively, in the RET-altered thyroid cancer safety population, and in 48.4% and 8.4% of patients, respectively, in the overall safety population. The most common TRAEs leading to discontinuation included anemia (2.3%) in the RET-altered thyroid cancer safety population, and pneumonitis (2.0%) and anemia (1.1%) in the overall safety population.
Median dose intensity over the full treatment duration was 340.9 mg/day (range: 86–401) in patients with RET-mutant MTC and 323.8 mg/day (range: 116–400) in patients with RET fusion-positive thyroid cancer.
Discussion
Results from this updated analysis of the phase 1/2 ARROW study, with more patients and a longer follow-up, confirm that pralsetinib 400 mg QD provides durable efficacy in patients with advanced/metastatic RET-altered thyroid cancer. Treatment with pralsetinib demonstrated ORR between 56% and 91%, median DoR of >20 months, and median PFS of >25 months across the different patient cohorts (including NR in treatment-naïve patients with RET-mutant MTC). These data are consistent with the primary analysis of this study, 18 as well as with data reported for selpercatinib, 21 and support the use of RET inhibitors for the treatment of patients with RET-altered thyroid cancer.
As discussed previously, 18 our study suggests that pralsetinib may provide higher ORR in treatment-naïve patients with RET-mutant MTC versus those reported with the first-line MKIs cabozantinib (ORR 32%; actively progressing disease) 15 and vandetanib (ORR 46%; with/without actively progressing disease). 14 Similarly, in patients with RET fusion-positive thyroid cancer, responses with pralsetinib were favorable compared with those with lenvatinib (ORR 65%) 13 and sorafenib (ORR 12%; progressive disease). 12
In this patient population, the ORR with pralsetinib (91%) was comparable to that of selpercatinib (79%). 21 Comparisons of data from independently conducted clinical trials should be made with caution due to differences in study designs and patient populations; however, a PFS benefit with selpercatinib versus cabozantinib or vandetanib was recently reported from an ongoing head-to-head trial in patients with advanced RET-mutant MTC. 22
Responses with pralsetinib were observed regardless of RET mutation genotype or RET fusion partner, including in patients with the gatekeeper mutation V804L/M. Although the time-to-event endpoint data remain mostly immature, the DoR and PFS results from our study suggest that pralsetinib provides durable clinical activity.
Median time to response was longest in patients with RET-mutant MTC, particularly those who were treatment naïve (∼6 months), and shortest in patients with previously treated RET fusion-positive thyroid cancer. An explanation for this observation is unclear at this time.
The safety profile of pralsetinib was tolerable, with TRAEs generally managed by dose adjustments. The rate of dose reductions due to TRAEs in our study was lower compared with studies of MKIs, 12,13,15 suggesting that selective RET inhibitors may be more tolerable than MKIs in patients with RET-altered thyroid cancer. The improved GHS/QoL scores and reduced symptom scores compared with baseline, particularly in patients with RET-mutant MTC who had received prior C/V, suggest that pralsetinib also confers HRQoL benefits. Although this was an exploratory analysis, the data are consistent with those from a study of selpercatinib, 23 suggesting a possible class effect. The improvement in diarrhea with pralsetinib is important and of particular clinical interest, given that diarrhea is a common symptom of MTC 24 as well as a side effect of cabozantinib and vandetanib, 14,15 and can significantly impact HRQoL.
Experience from other tumor types (e.g., nonsmall cell lung cancer) suggests that targeted therapies tend to become the preferred treatment option if they demonstrate superior efficacy and/or tolerability versus the established standards of care. A major limitation of MKIs is that their established toxicity profiles, which include off-target side effects, may lead to dose reductions or drug discontinuations. 25,26 Based on the current data from studies of RET-specific inhibitors, treatment guidelines have been updated to reflect the availability of these targeted therapies, 11 with regulatory constraints posing the main obstacle to use in earlier lines.
Moreover, the efficacy of MKIs as second- or third-line treatments, following progression on a RET-specific inhibitor, is unknown. As such, when choosing the first-line therapy for their patients, physicians should also consider what options might be available to the patient in the second or third line if they were to progress.
A decrease in calcitonin and CEA concentration was also observed in our analysis (response rates: 86% for calcitonin; 64% for CEA) and may explain in part the improvement in symptomatic diarrhea for some patients. RET inhibition will lead to lower levels of calcitonin as a result of decreased RET signaling and reduced tumor burden. Future analyses could look at whether there is a correlation between tumor regression and the degree of calcitonin/CEA regression, as this may help inform clinical decisions.
One strength of our study is that tumor assessments were carried out by BICR and therefore remain unbiased. One key limitation is the lack of a comparator arm. The molecular testing to determine the presence of a RET alteration was done locally rather than centrally, which may introduce inconsistencies; however, patients had to provide a tissue sample for confirmation of their RET status, which may address this limitation. The strength of the data and their consistency with those for selpercatinib support the use of RET inhibitors for the treatment of patients with RET-altered thyroid cancer.
In conclusion, pralsetinib continues to show robust and durable clinical activity, with a manageable safety profile, and improved or stable PROs in patients with RET-altered thyroid cancer. This study highlights the importance of molecular testing in thyroid cancer to identify patients who may benefit from targeted therapies, including pralsetinib. The implementation of wider molecular testing, with identification of more targetable oncogenic drivers, could transform the treatment of thyroid cancer.
Footnotes
Acknowledgments
Hui Zhang of Blueprint Medicines Corporation, Cambridge, Massachusetts, USA, provided additional support with the statistical analysis.
Authors' Contributions
V.S. and M.I.H. assisted with investigation, writing—original draft, reviewing, and editing. A.S.M., M.H.T., M.S., V.W.Z., J.H., G.C., L.W., J.F.G., G.A., D.A., Y.G., M.-J.A., P.A.C., B.C.C., C.-C.L., and M.S.B. contributed to investigation, writing—review and editing. A.Z. and A.S. guided with writing—review and editing. T.B. and P.T. assisted with software, formal analysis, data curation, and writing—original draft, review, and editing. W.B. and S.H. contributed to writing—original draft, review, and editing.
Data Sharing
For eligible studies, qualified researchers may request access to individual patient-level clinical data through a data request platform. At the time of writing, this request platform is Vivli.
Author Disclosure Statement
V.S. reports research funding/grant from Blueprint Medicines, and Roche/Genentech for clinical trials research support; support for clinical trials from AbbVie, Agensys, Inc., Alfasigma, Altum, Amgen, Bayer, BERG Health, Blueprint Medicines Corporation, Boston Biomedical, Inc., Boston Pharmaceuticals, Celgene Corporation, D3 Bio, Inc., Dragonfly Therapeutics, Inc., Exelixis, Fujifilm, GlaxoSmithKline, Idera Pharmaceuticals, Inc., Incyte Corporation, Inhibrx, Loxo Oncology, MedImmune, MultiVir, Inc., NanoCarrier, Co., National Comprehensive Cancer Network, NCI-CTEP, Northwest Biotherapeutics, Novartis, PharmaMar, Pfizer, Relay Therapeutics, Roche/Genentech, Takeda, Turning Point Therapeutics, UT MD Anderson Cancer Center, and Vegenics Pty Ltd; travel support from ASCO, ESMO, Helsinn Healthcare, Incyte Corporation, Novartis, and PharmaMar; consultancy/advisory board participation for Helsinn Healthcare, Jazz Pharmaceuticals, Incyte Corporation, Loxo Oncology/Eli Lilly, MedImmune, Novartis, QED Therapeutics, Relay Therapeutics, Daiichi Sankyo, R-Pharm US, and Jazz Pharmaceuticals; and other relationships with Peerview and Medscape.
M.I.H. reports participation in advisory board for Eli Lilly & Co, participation in clinical trial steering committee (nonfinancial) for Eli Lilly & Co; research support from Eli Lilly, and participation in ATA guidelines committee for update on systemic therapies for MTC.
A.S.M. is a nonremunerated member of the Mesothelioma Applied Research Foundation Board of Directors and Friends of Patan Hospital; reports direct research funding from the National Cancer Institute, Department of Defense, Mark Foundation, Novartis, and Verily; institution honoraria for participation in advisory boards for AbbVie, BeiGene, Bristol Myers Squibb, Genentech, and Janssen; and travel support from Shanghai Roche.
M.H.T. reports honoraria for participation in speaker's bureaus for BMS, Eisai, Merck, and Blueprint Medicines; honoraria for participation in advisory boards for Bayer, Blueprint Medicines, Bristol Myers Squibb, Cascade Prodrug, Eisai Inc., Exelixis, Immuneonc, Loxo Oncology, Merck, Novartis, Pfizer, Regeneron, and Sanofi/Genzyme.
M.S. reports consulting fees from Amgen, AstraZeneca, Blueprint Medicines, Boehringer Ingelheim, Bristol Myers Squibb, GlaxoSmithKline, Janssen, Merck Serono, Novartis, Roche, Sanofi, and Takeda; honoraria for CME presentations from Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Janssen, MSD, Novartis, Roche, and Sanofi; his institution received research funding from AstraZeneca and Bristol Myers Squibb.
V.W.Z. reports honoraria from AstraZeneca, BeiGene, Blueprint Medicines, Roche-Foundation Medicine, Roche/Genentech, Takeda, and Xcovery; had stock ownership of TP Therapeutics until May 2020; and is currently an employee of Nuvalent with stock ownership.
J.H. reports honoraria for participation in advisory boards for Ipsen, Eli Lilly, Roche, Pharma Mar, and AAA; research support from Novartis.
G.C. reports honoraria from AstraZeneca, BMS, Celcuity, Daiichi Sankyo, Exact Sciences, Gilead, Eli Lilly, Novartis, Pfizer, Roche, and Seagen; consulting fees from AstraZeneca, BMS, Celcuity, Daiichi Sankyo, Exact Sciences, Gilead, Eli Lilly, Novartis, Pfizer, Roche, and Seagen; honoraria for participation in speaker's bureaus from AstraZeneca, Daiichi Sankyo, Exact Sciences, Eli Lilly, Novartis, Pfizer, Roche, and Seagen; research funding from Merck; compensation for travel/accommodation/expenses from Pfizer and Roche.
L.W. reports honoraria received for advisory roles from Bayer HealthCare Pharmaceuticals, Coherus, Curie Therapeutics, Eli Lilly, Eisai, Exelixis, Genentech USA, Merck, Morphic Therapeutics, Tome Biosciences; honoraria received for serving on a data safety monitoring board for PDS Biotechnology Corporation.
J.F.G. reports advisory board fees from AI Protein, AstraZeneca, BeiGene, Bristol Myers Squibb, Roche/Genentech, GlydeBio, iTeos, Karyopharm, Eli Lilly, Merck, Mirati, Moderna, Novartis, Pfizer, Silverback Therapeutics, and Takeda; personal and institutional support (with financial interest) as a coordinating principal investigator from Novartis; institutional support (no financial interest) as a coordinating principal investigator from Alexo, AstraZeneca, Bristol Myers Squibb, Genentech, Jounce, Merck, and Moderna; institutional support (no financial interest) as a local principal investigator from Palleon and Scholar Rock; owns stocks/shares in Ironwood Pharmaceuticals.
G.A. has no conflicts of interest to disclose.
D.A. reports research funding as site principal investigator from Pfizer, Eli Lilly, Merck, Celgene/BMS, AstraZeneca, Blueprint Medicines, Kura Oncology, Exelixis, Aduro Biotech, Cue Biopharma, Cofactor Genomics, Shanghai Denovo, Hookipa, Debio, Adlai Nortye USA, BeiGene, Epizyme, Gilead, ISA, Roche, Rubius, Immutep, Tizona, Vaccinex, Genmab, Tacti, Calliditas, Natco Pharma, BioAtla, Erasca, Epizyme (an Ipsen Company), Boehringer Ingelheim, and Kymab; is a consultant for Merck, Cue Biopharma, Coherus, Eisai, Exelixis, Genmab, Jazz Pharmaceuticals, Pfizer, Natco Pharma, Calliditas, Immunitas, Seagen, Kura Oncology, Targimmune Therapeutics, TwoXAR, Vaccinex, Xilio Therapeutics, and Boehringer Ingelheim.
Y.G. reports honoraria for participation in advisory boards for Ipsen, Eli Lilly, Roche, Bayer, EISAI, and AstraZeneca.
M.-J.A. reports honoraria as an invited speaker for Amgen, AstraZeneca, Daiichi Sankyo, Eli Lilly, Merck MSD, Pfizer, Roche, Takeda, and Yuhan; honoraria for participation in advisory boards for Alpha Pharmaceuticals, Amgen, ARCUS, AstraZeneca, Daiichi Sankyo, Eli Lilly, Merck, MSD, Pfizer, Roche, Takeda, and Yuhan.
P.A.C. reports honoraria from Amgen, ITeos Therapeutics, and Janssen; consulting fees from OSE Immunotherapeutics; research funding to institution from AbbVie, AstraZeneca, Bayer, Blueprint Medicines, Bristol Myers Squibb, Celgene, GlaxoSmithKline, Innate Pharma, Janssen, Eli Lilly, Loxo Oncology, Merck Serono, Merck Sharp & Dohme, Novartis, Plexxikon, Roche/Genentech, Taiho Pharmaceutical, Toray Industries, and Transgene; compensation for travel/accommodation/expenses from Amgen, AstraZeneca/MedImmune, Merck Serono, Merck Sharpe & Dohme, Netris Pharma, and Roche.
B.C.C. reports research funding from AbbVie, AstraZeneca, Bayer, Champions Oncology, Blueprint Medicines, Dizal Pharma, Dong-A ST, Eli Lilly, Janssen, Interpark Bio Convergence Corp., Medpacto, GI Innovation, MOGAM Institute, MSD, Novartis, Ono, and Yuhan; consulting fees from AstraZeneca, Blueprint Medicines, BMS, Boehringer Ingelheim, Eli Lilly, Janssen, Medpacto, GI Innovation, MSD, Novartis, Ono, Pfizer, Roche, Takeda, and Yuhan; stock ownership in Bridge Biotherapeutics, Cyrus Therapeutics, Gencurix Inc., Interpark Bio Convergence Corp., Kanaph Therapeutics, Inc., and TheraCanVac Inc.; honoraria for participation in advisory boards for Bridge Biotherapeutics, Cyrus Therapeutics, Guardant Health, Joseah BIO, and Kanaph Therapeutics, Inc.; is on the board of directors of Gencurix Inc., and Interpark Bio Convergence Corp.; receives royalties from Champions Oncology, and is a founder of DAAN Biotherapeutics.
C.-C.L. reports honoraria from Daiichi Sankyo, Eli Lilly, Novartis, and Roche; consulting fees from AbbVie, Bayer, Blueprint Medicines, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Novartis, and PharmaEngine; compensation for travel/accommodation/expenses from BeiGene, Daiichi Sankyo, and Eli Lilly.
A.Z. is an employee of Blueprint Medicines and holds stocks/shares in the company.
T.B. is an employee of F. Hoffmann-La Roche Ltd.
P.T. is an employee of Genentech, Inc., and holds stocks/shares in F. Hoffmann-La Roche Ltd.
A.S. is an employee of Roche Products Ltd and holds stocks/shares in F. Hoffmann-La Roche Ltd.
W.B. and S.H. are employees of F. Hoffmann-La Roche Ltd. and hold stocks/shares in the company.
M.S.B. reports participation in advisory board for Eli Lilly & Co; participation in clinical trial steering committee (nonfinancial) for Eli Lilly & Co; research support from Eli Lilly; honoraria for participation in advisory boards for Blueprint Medicines, Eisai, Loxo Oncology, Eli Lilly, and Bayer; and research grants to institution from Blueprint Medicines, Eisai, Loxo Oncology, Eli Lilly, and Bayer.
Funding Information
V.S. is an Andrew Sabin Family Foundation fellow at the University of Texas MD Anderson Cancer Center. V.S. acknowledges the support of the Jacquelyn A. Brady Fund and is also supported by U.S. National Institutes of Health (NIH) (Grant Nos. R01CA242845 and R01CA273168). The MD Anderson Cancer Center Department of Investigational Cancer Therapeutics is supported by the Cancer Prevention and Research Institute of Texas (No. RP1100584), the Sheikh Khalifa Bin Zayed Al Nahyan Institute for Personalized Cancer Therapy (No. 1U01 CA180964), a National Center for Advancing Translational Sciences (NCATS; Center for Clinical and Translational Sciences) (Grant No. UL1 TR000371), and an MD Anderson Cancer Center Support (Grant No. P30 CA016672). Third-party medical writing assistance, under the direction of the authors, was provided by Claire White, PhD, and Lietta Nicolaides, PhD, of Ashfield MedComms, an Inizio company, and funded by F. Hoffmann-La Roche Ltd. The study was designed by the funder in collaboration with the investigators. The funder had a role in data collection, data analysis, and data interpretation in conjunction with the authors, who all had access to the data.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
