Abstract
This study aimed to evaluate the effects of therapeutic hypothermia (TH) and the warming phase on cardiovascular hemodynamics among neonates suffering from perinatal asphyxia. The reviewed literature on hemodynamic changes among neonates undergoing TH was obtained from the following databases: PubMed, Embase, POPLINE, Cochrane Reference Libraries, Google Scholar, Cochrane Central Register of Controlled Trials, and the Cochrane Database of Systematic Reviews. Search strategies included keywords, combinations, medical subject headings, and snowball searches of related articles. The following search terms were used: brain injury, hypoxic–ischemic encephalopathy, left ventricular (LV) dysfunction, right ventricular (RV) dysfunction, and TH. We selected publications evaluating RV and LV heart function and cerebral, renal, and visceral circulation function for analysis. There were 12 prospective studies, with a total of 361 patients in the study groups and 149 patients in the control groups. There were seven retrospective studies, with a total of 1637 patients. One study was a randomized controlled trial, one was a systematic review, and one was a Cochrane review. The limitations of the review are that most of the studies are observational, making it difficult to precisely assess the causes of the observed changes, whether they are related to asphyxia, hypothermia, or other pathology. The results of the observational studies were not consistent with those of the randomized trials for ethical reasons. The hemodynamic characteristics of the cardiovascular system during TH and the rewarming phase are significantly complex. Therefore, an in-depth understanding of the pathophysiological attributes associated with these aspects is essential to provide individualized therapeutic approaches for optimizing cerebral perfusion pressure and reducing secondary injuries.
Keywords
Introduction
Perinatal asphyxia (PA) has a high perinatal mortality rate and severe long-term consequences. The incidence of PA is reportedly as high as 10 cases per 1000 newborns in less-developed countries. In developed countries, severe PA occurs in 1 per 1000 newborns (Endrich et al., 2017; Lawn et al., 2005; Lawn et al., 2014; Lawn et al., 2011). PA damages neural tissue and cardiac muscles. Myocardial development remains incomplete at birth, making the heart inadequately responsive to aggressive cardiac diseases. Impaired myocardial perfusion results from primary hypoxic–ischemic encephalopathy (HIE), as well as the abnormal redistribution of blood to an injured brain. Multiple organ failure, particularly cardiovascular failure, plays a significant role in the development of secondary HIE, a major cause of mortality (Thoresen and Whitelaw, 2000; Hankins et al., 2002).
Therapeutic hypothermia (TH) is the gold standard for treating HIE; however, the fragile balance between therapeutic gain and cardiovascular instability must be considered. Among neonates undergoing TH, cardiovascular failure occurs in 25–81% of those suffering from severe PA (Rakesh et al., 2018; Ambalavanan et al., 2012; La Rosa et al., 2017; Balushi et al., 2018). Therapeutic goals should focus on maintaining cardiovascular stability and counteracting the consequences of cardiovascular damage among neonates suffering from HIE.
However, the cardiovascular effects of the rewarming phase on neonates are yet to be fully studied. This phase impacts the cardiovascular status and hemodynamic stability of neonates, decreases pulmonary vascular resistance (PVR) and systemic vascular resistance (SVR), affects the redistribution of blood to organs, and increases the heart rate (HR) and cardiac output (CO). However, the mean blood pressure may decrease or remain unchanged during this period due to a decrease in diastolic blood pressure. Although TH has changed the outcomes for children suffering from moderate-to-severe HIE, not all children benefit from it.
The effects of TH on the cardiovascular system may be more pronounced among neonates suffering from other concomitant pathologies, such as sepsis and meconium aspiration syndrome. In addition to studies indicating improved myocardial function, some have reported the transient adverse effects of TH (Thoresen and Whitelaw, 2000; Rakesh et al., 2018). Among them are sinus bradycardia, a drop in blood pressure, and increased oxygen demand (Ambalavanan et al., 2012; Jacobs et al., 2013). Cardiac dysfunction, including impaired contractility with subsequent low stroke volume, bradycardia, and pulmonary hypertension, complicates the already impaired circulatory status of newborns with PA (Mathew and Lakshminrusimha, 2017). However, these effects are transient and can disappear with the rewarming procedure.
Data published on changes in hemodynamics during the rewarming phase among neonates are limited. A reduction in SVR may result in a concomitant reduction in diastolic pressure and subsequent reperfusion of cerebral circulation. During the rewarming period, the active adjustment of cardiovascular medications may be necessary to avoid iatrogenic hypertension and excessively unregulated cerebral blood flow (CBF). However, cardiovascular hemodynamics during rewarming requires further research.
There are many evaluation and measurement methods based on different physical bases, each with its own advantages and disadvantages. New echocardiography and Doppler techniques can clarify the pathomorphology of cardiovascular failure in hypothermic neonates. They can also assess the effects of the hypothermia and warming phases on the blood flow and perfusion of the heart and other organs. To assess the changes associated with hemodynamics in newborns with PA, new methods such as near-infrared spectroscopy (NIRS) and magnetic resonance imaging arterial spin labeling perfusion imaging (MRI [ASL-PI]) are used.
Methods
The reviewed literature on hemodynamic changes in neonates undergoing TH was obtained from the following databases: PubMed, Embase, POPLINE, Cochrane Reference Libraries, Google Scholar, Cochrane Central Register of Controlled Trials, and the Cochrane Database of Systematic Reviews published until July 4, 2024.
The studies were meticulously assessed using the Newcastle–Ottawa Scale (NOS) by two independent writers (N.B. and V.B.).
Disagreements were discussed with the third author (R.B.) until a common consensus was achieved among all reviewers (Stang, 2010). The NOS employs a star rating system, allowing a maximum of 9 points for prospective and cross-sectional research and 10 points for case–control studies. The assessment has eight questions across three domains: selection, comparability, and exposure. Each question is assigned a maximum score of one point, except for comparability, which permits a score of two points. Studies that received a score of nine points or higher were deemed to be of good quality.
The systematic review and meta-analysis were performed in alignment with the Cochrane Collaboration’s recommendations and adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses standards (Page et al., 2021) and are present in Supplementary Data and Figure 1. Table 1 provides a detailed description of the inclusion and exclusion criteria of the search strategy.
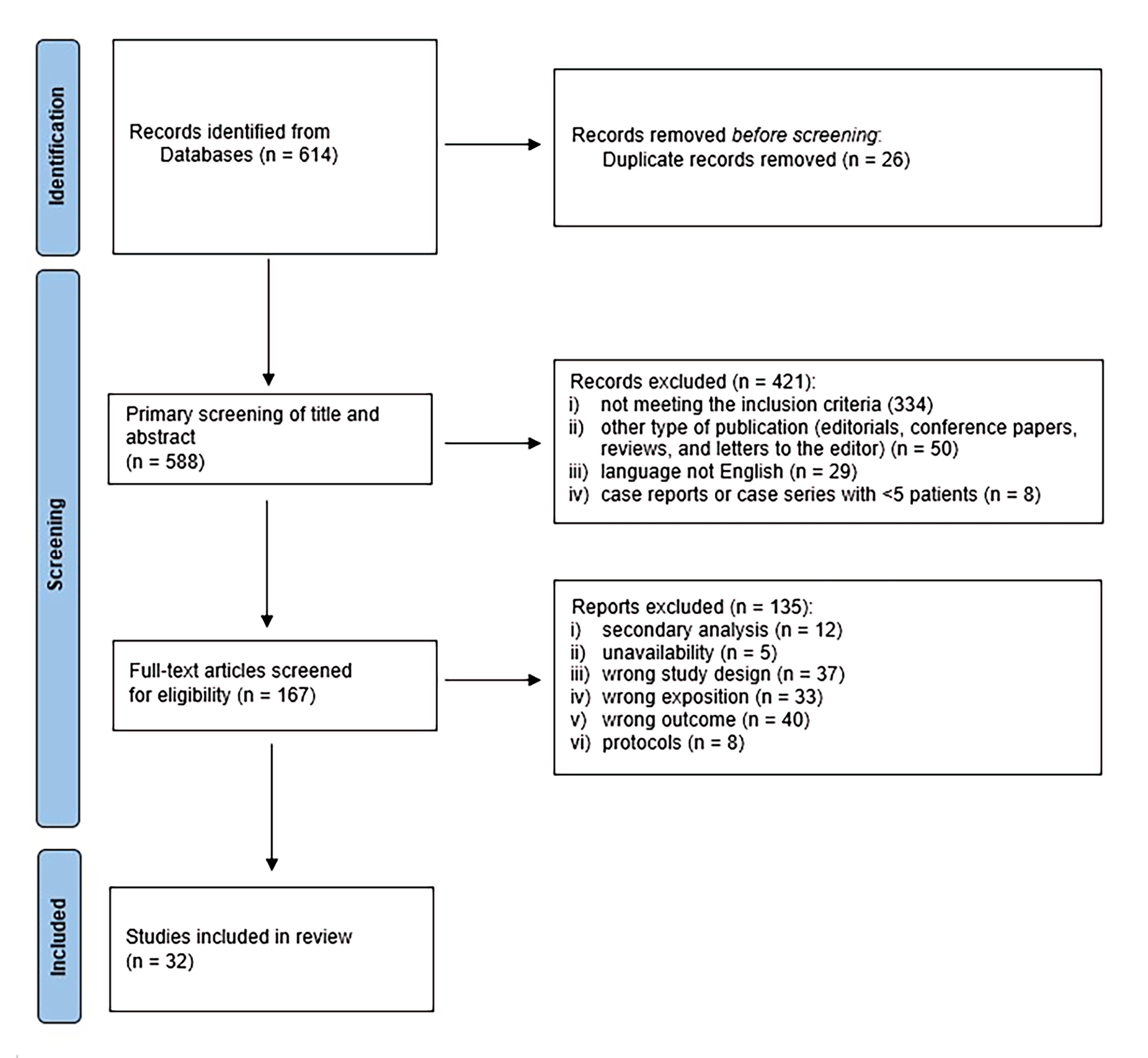
PRISMA flowchart. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Inclusion and Exclusion Criteria in the Search Strategy


The selected publications are restricted to research conducted within the past 15 years. When restricted to hemodynamics, 167 articles were obtained. After excluding duplicates and previous studies, 32 publications were selected. Of the accessed articles, 10 publications did not include studies involving patients. The exact divisions are listed in Table 2 and illustrated in Figure 1.
Division of Publications
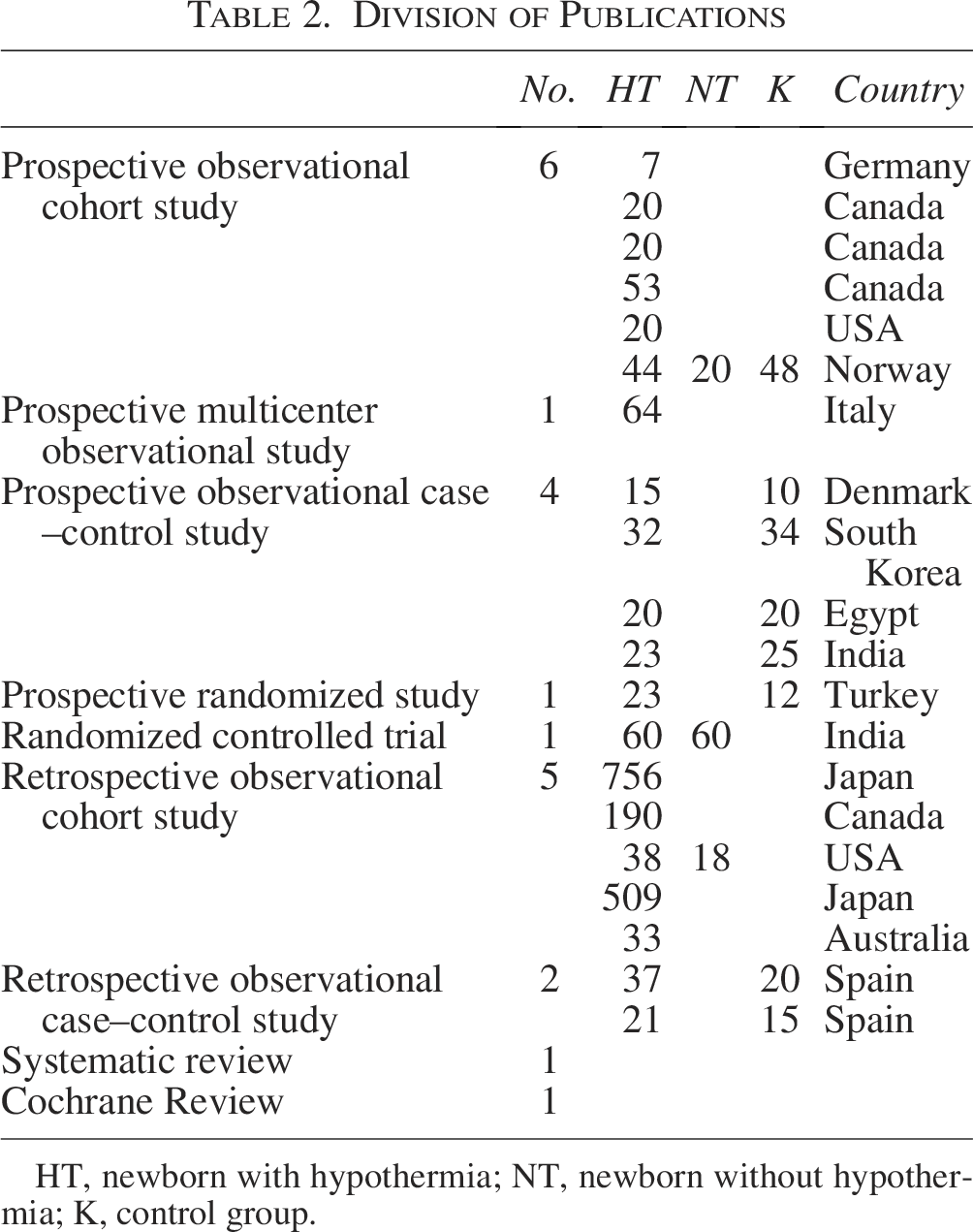
HT, newborn with hypothermia; NT, newborn without hypothermia; K, control group.
Clinical Effect of TH and the Rewarming Phase
HR and CO
The normal HR range in a healthy neonate during the transition to pulmonary respiration over the first few minutes after birth is not well agreed upon or established (Fleming et al., 2011). The changes in the neonate’s cardiovascular system during the first day of life (DOL) are dictated by a gradual stabilization in cardiac function; in healthy neonates, HR declines during the first DOL (Fleming et al., 2011). In healthy-term infants
A decrease in body temperature to below 34°C results progressive slowing of the HR. The direct effect of cooling on the HR, as manifested by sinus bradycardia, is mainly due to a reduction in sinus node repolarization from decreasing intracellular calcium release and the reduced influence of the autonomic nervous system (Nestaas and Brian, 2020; Gambassi et al., 1994). Decreased ion pump activity for sodium, potassium, and calcium and reduced cell membrane conductance for Na+, K+, and Ca2+ ions have also been observed during cooling. In an animal study, during deep hypothermia, coronary blood flow was significantly reduced (up to 40% of baseline flow) but was proportionally greater than in the circulation of other organs, where it reached values of <20% relative to baseline values (Tveita et al., 1994).
However, bradycardia is not associated with changes in amplitude on integrated electroencephalogram recordings, indicating no apparent effect on cerebral perfusion (Thoresen and Whitelaw, 2000). As confirmed in a Cochrane meta-analysis, there is an increased incidence of sinus bradycardia but no significant cooling-related arrhythmias (Jacobs et al., 2013), no increase in the incidence of hypotension among neonates undergoing TH, and no increase in the incidence of hypotension requiring inotropic support. Despite the reduced CO, the stable blood pressure observed during cooling could be explained by an increase in SVR (Jacobs et al., 2013; Giesinger et al., 2017).
Gebauer et al. found that LVCO was 67% of the output value after the warming phase. This reduction was mainly due to a decrease in HR but also a reduction in stroke volume (Gebauer et al., 2006; Hochwald et al., 2014). LVCO does not seem to have prognostic significance; similar values have been reported in neonates with normal development, as well as in those with short- and long-term side effects (Hochwald et al., 2014; Montaldo et al., 2018).
These changes are generally well tolerated and, although the CO is lower, it likely meets the metabolic needs of the neonate, particularly in the context of TH, where metabolic activity is further reduced. In a study involving 15 neonates with HIE + TH and 10 healthy control neonates, Eriksen et al. identified reduced metabolism rather than myocardial dysfunction as the cause of decreased CO during hypothermia (Eriksen et al., 2019).
This was confirmed in another study, where after rewarming, myocardial function returned to normal more rapidly than in neonates without TH (Nestaas et al., 2014). Some studies have attributed this increase to an increase in HR rather than an increase in contractile force (Sehgal et al., 2019).
Relative bradycardia may result in an overall reduction in myocardial substrate demand and, therefore, protect against ischemia, whereas the use of chronotropic drugs may increase metabolic demand and the risk of cell death. In contrast, a normal HR, despite low body temperature, may reflect subclinical systemic hypoperfusion and contribute to ongoing brain damage.
In two retrospective studies conducted by Kennosuke Tsuda, poorer psychomotor development and death were associated with higher Thompson scores on admission, a higher HR after 3–72 hours of cooling, lower base excess, and a higher body temperature after 24 hours of cooling (Tsuda et al., 2019; Tsuda et al., 2022). Another study reported that TH-eligible infants with abnormal development had higher HRs (Elstad et al., 2016). It was concluded that severe PA may be a common cause of faster HRs before and during TH; however, the causal relationship between slightly higher temperatures during cooling and adverse outcomes needs to be clarified in future studies.
During the rewarming phase, intracellular calcium release gradually normalizes, and the influence of the autonomic nervous system increases (Thoresen, 2008). This normalizes sinus node repolarization and causes normocardia. Yoon et al. found that LVCO and the HR increased after the rewarming phase, but lower values were recorded in the LVCO group, with a slightly reduced LV stroke volume (LVSV) compared with the control group (Yoon et al., 2018). After recovery from hypothermia, the HR quickly returns to normal, but hypoxia and myocardial ischemia may persist; therefore, LVSV and LVCO may remain below normal. This finding is supported by previous observations in animal studies showing that hypothermia reduces HR, but artificially increasing the HR via pacing reduces SV (Espinoza et al., 2013). Attention should be paid to vasopressor and inotrope doses during the warming phase; active adjustment of cardiovascular drugs may be necessary to avoid iatrogenic tachycardia and decrease CO.
In addition, during the rewarming phase, there is sometimes a sudden increase in HR and a reduction in SVR (Rajakumar et al., 2009), as confirmed in a Cochrane meta-analysis (Jacobs et al., 2013). These changes, especially rapid ones, may contribute to reperfusion injury in the brain and other organs (Mathew and Lakshminrusimha, 2017) and may be more pronounced in neonates who require cardiac support.
In acute ischemic conditions, the affected myocardial tissue does not demonstrate shortening or lengthening activity, but it can reflect a reduced systolic strain rate (SR) or strain.
Sehgal et al. conducted a case–control study involving 24 infants with severe PA. Three were not cooled and excluded, and speckle tracking echocardiography was used to assess the asphyxiated infants (Sehgal et al, 2013). These infants were already severely hemodynamically compromised, as reflected in the lower conventional parameters of fractional shortening (FS), stroke volume, and LV output (LVO). Mean LV global longitudinal strain (GLS) was significantly impaired, and CO was notably lower in the asphyxiated infants. Segmental strain and SR were both significantly lower in the asphyxiated infants; however, no significant segmental gradient was noted. In the asphyxiated infants, GLS correlated positively with CO and negatively with serum troponin levels. Infants who died had a lower global strain value than survivors. This could be a good marker of the overall disease process severity, so additional research is required (Sehgal et al., 2013).
As a practical assessment of HR, during TH, it is normal to have bradycardia. However, normal HR may be falsely reassuring. It is essential to pay attention to FS and LV filling (Z score). If LV filling is insufficient, an erroneous diagnosis of myocardial hypertrophy may be made. The fluid supply is usually 40–60 mL/kg/day, and to maintain blood pressure and adequate cerebral perfusion, the volume should be supplemented, and inotropic agents should be used. However, systemic hypertension and volume overload can worsen cerebral edema and should be avoided.
Pulmonary circulation
Fetuses are in a state of elevated PVR, enabling the preferential flow of oxygenated blood across the placenta into the low-resistance fetal systemic circulation. This elevated PVR is maintained by the compression of fluid-filled alveoli on the pulmonary vasculature and the constriction of the vascular lumen by the cuboidal arrangement of endothelial cells. Importantly, this results from hypoxic vasoconstriction associated with low alveolar oxygen content.
The change from high- to low-resistance pulmonary circulation occurs shortly after birth. At that time, there is a flow reversal in favor of left–right flow through the ductus arteriosus (DA) and the foramen ovale (FO). In neonates born at term, the increased oxygen partial pressure in the arterial blood after the onset of respiration causes a sharp decrease in PVR.
The adaptation of pulmonary circulation involves a complex balance between mediators such as endothelin 1, leukotrienes, and nitric oxide (NO) in the vascular endothelium. Hypoxia induces an imbalance between endothelin and NO in favor of the former, leading to vasoconstriction through endothelial dysfunction and reduced NO production (Lapointe and Barrington, 2011; Armstrong et al., 2012).
Persistent pulmonary hypertension (PPHN) in neonates undergoing TH has garnered attention since Eicher et al. reported an increased risk of pulmonary hypertension requiring treatment with inhaled NO (iNO) during TH (Eicher et al., 2005; Bhagat and Sarkar, 2019).
Further research has aimed to confirm or refute this hypothesis. In neonates born under hypoxic conditions, there is a progressive increase in PVR, directly resulting from hypoxemia and reduced pulmonary vascular oxygen sensitivity. In a study by Liu et al., indicators of pulmonary hypertension, such as pulmonary artery diastolic pressure, pulmonary arterial resistance, and the ratio of pulmonary arterial resistance to systemic resistance, were significantly elevated in 40 newborns with HIE compared with 40 healthy newborns on day one of life (Liu and Feng, 2009).
PPHN develops as a result of a vicious cycle of increased PVR, decreased pulmonary blood flow, decreased oxygenation, and a shift in right–left DA flow secondary to pulmonary hypertension. Reduced pulmonary perfusion diminishes pulmonary venous return to the left atrium, thereby reducing LV filling and performance (Nestaas and Brian, 2020). Increased RV afterload can result in RV dilatation and failure, further impeding pulmonary blood flow and reducing systemic circulation oxygenation (Giesinger et al., 2017; Habib et al., 2020). Hypothermia may modify PVR via the peripheral pathway vasoconstriction of pulmonary vessels, catecholamine release, or increased blood viscosity (Poulos and Mollitt, 1991). In studies of neonatal lambs during cooling, HR dropped by a 29% change; mean pulmonary arterial pressure increased from 29 to 40 mm/Hg. Metabolic acidosis and hypoxia developed during the cooling (Toubas et al., 1978). The sensitivity of pulmonary vessels in newborn rats to temperature resulted in a reduction in resistance of about 1% for every 1°C increase in temperature. Pulmonary vascular compliance increased, probably because of a direct effect of temperature on vessel wall stiffness (Rubini, 2005).
Although TH studies have not shown a higher overall incidence of clinical pulmonary hypertension, observational studies have reported increased FiO2 requirements in some neonates with PA and echocardiographic evidence of elevated PVR during TH, which reverses after the rewarming phase (Thoresen and Whitelaw, 2000; Sehgal et al., 2019; Thoresen, 2011). The discrepancy is likely multifactorial, with some studies suggesting that PPHN risk is related to the cause of PA or a response to ongoing postnatal hypoxia rather than cooling (Eicher et al., 2005). Moreover, PA is often complicated by meconium aspiration and sepsis/pneumonia (Lapointe and Barrington, 2011). Aggarwal’s study involving 56 neonates with HIE (38 with TH and 18 without due to a lack of hypothermia machine availability or late qualifications) and a control group of 35 healthy neonates confirmed this, with 54% of the HIE neonates being eligible for NO therapy, regardless of whether HT therapy was administered (Aggarwal and Natarajan, 2017). More et al. confirmed that neonates with concomitant pulmonary hypertension and HIE were more likely to have abnormal MRI findings despite TH (More et al., 2018; More et al., 2014).
In two systematic reviews, the risk of PPHN and the need for iNO or extracorporeal membrane oxygenation did not differ between newborns with and without TH (Jacobs et al., 2013; Shah et al., 2007). TH is not contraindicated in newborns with HIE already exhibiting clinical signs of PPHN (Thoresen, 2008). During the warming phase, there is a gradual (and sometimes sudden) reduction in pulmonary resistance, clinically manifested as reduced oxygen demand and normalized echocardiographic parameters (Thoresen and Whitelaw, 2000; Sehgal et al., 2019; Thoresen, 2011). The use of fast-acting pulmonary vasodilators such as iNO leads to increased pulmonary venous return and CO, potentially exacerbating reperfusion injury. These changes may further intensify during rewarming.
The use of oxygen to correct hypoxia and minimize hypoxic pulmonary vasoconstriction is an important clinical goal in treating neonates with PPHN; however, maintaining higher-than-normal blood oxygen content has not been scientifically proven to have additional benefits and could be harmful. Prior exposure to hyperoxia leads to excessive pulmonary vasoconstriction after hypoxic injury and impairs the vasodilatory effects of iNO. Treatment with recombinant superoxide dismutase reverses this effect, suggesting a role for oxygen-free radicals. In addition, oxygen-free radicals have been shown to interact with iNO to produce peroxynitrite, a metabolite of NO that is involved in mediating pulmonary vasoconstriction and RV dysfunction (Jain and McNamara, 2015). Simultaneous exposure to high oxygen concentrations and exogenous NO promotes reactive oxygen species production, potentially causing additional brain damage.
Practical assessment of pulmonary hypertension (echocardiography findings): confirm normal structure of heart; evaluate RV by considering hypertrophy and/or dilatation; assess RV and LV functions (see the RV and LV systolic function and RV and LV diastolic function sections); measure RV systolic pressure; evaluate shunts in cross-DA and FO; and evaluate time to peak velocity/RV ejection time (
RV and LV systolic function
Given that both the RV and LV eject blood into a singular vascular bed in the fetus, the systolic pressure of both ventricles is the same, but the ejection volume of each is different. The right ventricle has a greater mass in the fetus and is thus the dominant ventricle; it ejects 55% of the ejection volume of the heart. After birth, the situation is completely reversed. Both the RV and LV eject the same volume of blood, but the left ventricle generates higher systolic pressure; because of this, the left ventricle’s mass is much greater than that of the right. After birth, due to an increase in oxygen demand, the ejection volume of the LV increases 3-fold.
Myocytes survive hypoxia during fetal life due to their glycolytic metabolism. However, prolonged or sustained hypoxia delays the switch from anaerobic to aerobic energy production, making energy production less efficient. This lack of metabolic switch reduces adenosine triphosphate production, resulting in less efficient myocyte contraction and borderline myocyte survival with impaired function (Joynt and Cheung, 2018; Vrancken et al., 2018; Popescu et al., 2020).
Myocardial dysfunction with impaired cerebral autoregulation may delay cerebral reoxygenation after cerebral hypoxia and ischemia, exacerbating injuries and affecting 24–31% of patients (Rajakumar et al., 2009; Greisen, 2014). In a case–control study, Bhasin et al. noted that among 23 neonates, 5 developed grade I HIE, 10 developed grade II HIE, and 8 developed grade III HIE (TH was not utilized despite indications due to a lack of availability; Bhasin and Kohli, 2019). With a significantly reduced LV ejection fraction (LVEF) and RV ejection fraction in all neonates in the study group, eight patients (35%) died.
In a retrospective study by Aggarwal involving 58 newborns with HIE, significantly elevated myocardial performance indices (MPIs) for both ventricles and lower right ventricular output (RVO) and LVO were observed in children with HIE compared with healthy newborns (Aggarwal and Natarajan, 2017). The only significant difference between the newborns with HIE who underwent TH and the newborns with HIE + normothermia was a lower MPI LV in the TH group. Although most studies have focused on the LV, some suggest that perinatal hypoxia primarily affects the RV and that its effects are more long-lasting (Giesinger et al., 2019). RV systolic function impairment may stem from a cardiogenic origin, increased PVR, or both, disturbing the ventilation–perfusion ratio, reducing LV load, and affecting LV function, thereby enhancing the nonischemic–hypoxic effect by reducing systemic blood flow, including cerebral flow.
In a study involving 53 neonates, RV failure was associated with adverse outcomes among neonates with HIE eligible for hypothermia (Giesinger et al., 2019). RV systolic performance at 24 hours of life was significantly lower, correlating with high cerebral oxygen saturation and a low middle cerebral artery resistance index (RI-MCA). Abnormal RV systolic performance, including tricuspid annular plane systolic excursion, fractional area change, and RVO, was observed in neonates with adverse outcomes, whereas LV systolic performance measurements were similar between groups, although neonates with unfavorable outcomes trended toward smaller LVO values. Compared with LV function, RV function deterioration is more dramatically associated with an increased risk of death or abnormal MRI findings (Giesinger et al., 2019; Jain et al., 2018).
It is essential to assess global myocardial systolic and diastolic functions using both pulsed Doppler (PD) and tissue Doppler imaging (TDI) to determine the MPI. In a study by Karpuz involving 23 neonates with HIE and 12 neonates in a control group, the myocardial tissue index measured using PD for both ventricles was higher in the HIE group, but the difference was not significant (Karpuz et al., 2017). The results of two other studies indicate reduced LV systolic function in neonates with PA. They found that TDI was a more sensitive indicator of LV systolic function than LVEF, FS, or SV on M-mode echocardiography (Wei et al., 2009; Sehgal et al., 2012).
Moreover, TDI holds greater diagnostic value in assessing the cardiac function of neonates during TH and the rewarming phase. Nestaas et al. monitored myocardial function using TDI during TH (days 1 and 3) and after rewarming (day 4) in 44 newborns with HIE undergoing TH, 20 newborns with HIE not undergoing TH, and 48 healthy newborns (Nestaas et al., 2014). On days 1 and 3, all indices (peak systolic strain, peak systolic SR, early diastolic SR, and SR in atrial systole) in both HIE groups in both chambers were lower than those in healthy newborns. Both HIE groups had similar rates, except for peak systolic SR (S′) on days 1 and 3 and SR in the late diastole caused by atrial contraction (A′) on day 1. All echocardiographic indices improved after the rewarming phase in the TH group and reached values similar to those of healthy newborns on day 3. The indices were higher in the TH group after the rewarming phase than in the non-TH group with HIE on day 3, except for the early diastolic SR (E′), indicating that ventricular diastolic function was impaired for the longest period.
The Sehgal study also reported a reduction in peak systolic strain in the left ventricle during hypothermia (Sehgal et al., 2013). The neonatal myocardium was presumed to operate at maximal contractility, reflecting the value of the S′ wave (Johnson et al., 1979). Differences between the TH group and the non-TH group may reflect disparities in maximal cardiac capacity. The increase in parameters in the TH group, along with no difference between the TH group on day 4 and the control group on day 3, suggests that TH preserved the cardiac capacity to operate at a high contractile level.
Practical assessment of cardiac function (echocardiography findings): If the MPI is increasing and LVCO is decreasing with adequate filling and right renal output, inotropic therapy should be considered. Normal left and right MPIs in infants are 0.33 ± 0.08 and 0.29 ± 0.09, respectively (Siassi et al., 2019). When an abnormal MPI is found, other methods must be utilized to determine whether systolic pressure, diastolic pressure, or both are abnormal. Infants should be fluid-restricted to avoid fluid overload and cerebral edema.
RV and LV diastolic function
While most studies have focused on perinatal hypoxia–ischemia's effects on LV systolic function, recent research has highlighted the association between RV dysfunction and adverse neurological outcomes (Giesinger et al., 2019; Rodriguez et al., 2020; Mauritz et al., 2011). However, there has been less emphasis on diastolic cardiac function studies in neonates with HIE, with only marginal mentions in some studies. Diastolic dysfunction often arises from myocardial ischemia and increases mortality, as myocardial relaxation and ventricular filling are crucial for normal systolic performance.
In high-risk neonates, diastolic dysfunction may precede systolic dysfunction, and latent RV dysfunction may occur before LV failure onset. Neonates post-HIE may be particularly vulnerable to diastolic dysfunction due to the neonatal myocardium’s relatively increased collagen concentration, shorter filling time, and greater dependence on atrial contraction for diastolic function. Delayed relaxation significantly contributes to heart failure with preserved EF since 50–60% of diastoles occur during isovolumetric relaxation time (IVRT), necessitating an intact myocardium for normal early diastolic function.
One of the initial studies assessing diastolic function using TDI was conducted by Rodriguez et al. (2022), involving 21 patients with HIE + TH and 15 controls. This retrospective study demonstrated significant diastolic dysfunction in neonates with HIE during TH, gradually improving during treatment and normalizing after the rewarming phase. TDI assessment revealed dysfunction in the early diastolic phase during TH, whereas the late phase remained normal. Increased IVRT in both ventricles and the interventricular septum was noted, even after correcting TH-induced bradycardia, which gradually improved and normalized after the rewarming phase.
Some studies have suggested that the prolonged isovolumic time observed in patients with pulmonary hypertension could be attributed to a prolonged RV contraction phase after pulmonary valve closure due to increased afterload, rather than delayed isovolumic relaxation, which may not reflect diastolic dysfunction in this context (Mauritz et al., 2011). In Rodriguez’s study, this mechanism could partly explain the prolongation of IVRT, given the significantly increased PVR in the HIE + TH group (Rodriguez et al., 2022). However, the lower RV e’ waveform indicated diastolic dysfunction. Sobeih et al. conducted another study assessing diastolic dysfunction (Sobeih et al., 2021), where the study groups were evaluated using conventional and TDI. Significantly higher MPI values for both chambers were observed in the study group. Newborns with HIE + TH exhibited significantly impaired LV diastolic dysfunction compared with the control group, as assessed using PD and TDI. However, these changes were not observed in the right ventricle, possibly due to the physiological adaptation of pulmonary circulation and RV strain in the early neonatal period. Conversely, Matter et al. found no significant differences in mitral and tricuspid inflow velocity patterns measured using PD between neonates with HIE and a control group (Matter et al., 2010). Tissue Doppler detected more neonates with LV diastolic dysfunction than conventional PD. Assessing diastolic function in neonates using conventional echocardiography is challenging. TDI is relatively new and less influenced by loading conditions and cardiac structure geometry than conventional echocardiographic measurements. The MPI is one of the most consistent echocardiographic parameters for HIE and reflects changes in the combination of systolic and diastolic function.
Practical assessment of cardiac diastolic function: IVRT and E/A ratio at the mitral and tricuspid valve.
Cerebral flow redistribution
During the last trimester of pregnancy, resistance in the arterial vessels of the fetal brain decreases as blood is physiologically redistributed in favor of the brain in preparation for labor. During labor, pressure on the fetal head and subsequent decompression can affect CBF. Uterine contraction activity during the first period of labor also increases PI and RI flow rates in fetal cerebral vessels, which can reduce cerebral flow. Defending against a decrease in cerebral perfusion, the cerebral circulation before labor lowers its resistance, thereby increasing CBF.
Cerebral arteries are characterized by low-resistance flow. The blood flow velocity in the anterior cerebral artery (ACA) is about 30% lower than in the MCA on average, and the ratio of blood flow velocity in the MCA–ACA in term newborns is 1:0.86. The described differences in the RI value for the ACA and MCA indicate local changes in the resistance of the cerebral arteries. This suggests a physiological phenomenon related to uneven perfusion inside the brain. This is characteristic of more mature brains and does not occur in premature infants. The MCA is a phylogenetically younger vessel than the ACA. The MCA supplies more central brain structures concerning the cortical layer of the brain; therefore, it has greater autoregulation capacity than the ACA. The lower resistance in the ACA allows for increased blood flow to the cortical layer, particularly supplied by this vessel.
In sick newborns, changes in RI and PI blood flow rates occur more quickly in the ACA than in the MCA. Postnatal cerebral hemodynamic changes in selected neonatal pathological conditions should be evaluated primarily in the ACA. Animal studies have shown that reduced CBF reduces reperfusion-induced brain damage (Niatsetskaya et al., 2012) and that the brain’s reperfusion phase is an important mechanism of neuronal cell death in neonates with HIE. Moreover, clinical data suggest that neonates with severe brain injury have a preferential redistribution of cerebral blood circulation during whole-body TH (Hochwald et al., 2014). Whether preferential redistribution of cerebral blood circulation is a predictor of long-term neurological impairment requires further investigation. A decrease in body temperature is accompanied by a decrease in cerebral flow of 6–10% for every 1°C (Polderman, 2009). Increased vascular muscle tone and decreased CO are the primary effects of reduced body temperature on the cardiovascular system (Rios et al., 2021). At the same time, the excitability of cortical centers is reduced. Bradycardia with associated lower CO (which the literature suggests occurs in up to 62% of infants) and lower metabolic demand may be contributing factors to neuronal protection or may simply be responses to lower whole-body metabolism (Thomsen et al., 2016).
Montaldo et al. conducted one of the initial studies highlighting the correlation between CBF and long-term neurological impairment in HIE-affected children (Montaldo et al., 2018). This prospective, multicenter study involved 64 neonates with HIE eligible for TH, undergoing psychomotor development assessments at 18–24 months of age. Superior vena cava (SVC) flow and LVCO were evaluated during and post-TH. Abnormal outcomes, defined as death or moderate/severe disability, were associated with higher SVC flow during TH. Following the rewarming phase, LVCO and SV notably increased among children with abnormal neurological outcomes, whereas SVC flow remained constant. These findings may underscore the injured brain’s inability to respond to autonomic nervous system changes and cerebral circulation autoregulation (Gupta and Singh, 2019; Hochwald et al., 2014; Montaldo et al., 2018).
Yoon et al. conducted a study analyzing 32 neonates undergoing TH for HIE, assessing echocardiographic hemodynamics during and post-TH (Yoon et al., 2018). Compared with a control group of 34 healthy neonates, 50% of those with hypoxic–ischemic lesions on MRI exhibited a significantly elevated upper body blood flow–LVCO ratio during TH, indicating preferential cerebral LVCO redistribution and relative systemic organ hypoperfusion. This suggested impaired cerebral autoregulation in neonates with brain damage, a finding supported by Hochwald et al. (2014). Additionally, Rodriguez et al. noted a robust association between RV function and CBF size in TH-treated neonates with HIE (Rodriguez et al., 2020). RV dysfunction correlated with decreased CBF size, possibly reflecting a loss of cerebral autoregulation postoxygenation, particularly reliant on RV compliance. They also observed a strong correlation between cerebral oxygenation and RV function parameters. Giesinger et al. similarly found significant correlations between RV function parameters and RI in the MCA (Giesinger et al., 2019).
Compared with other methods used to measure brain perfusion, including Doppler sonography, NIRS, positron emission tomography, and xenon-enhanced computed tomography, only ASL-PI enables the direct assessment of CBF values in different brain areas without contrast injection MRI (ASL-PI). One study revealed a sequence of events during TH involving initial cerebral circulation hypoperfusion followed by hyperperfusion due to brain damage (Wintermark et al., 2011). Compared with control patients and asphyxiated neonates who were treated with hypothermia but did not develop brain injury, CBF values were markedly lower on the first DOL in the brain regions that showed injury in neonates treated with hypothermia and who developed MRI evidence of HI brain injury. CBF values increased in the injured areas at the second DOL. While CBF increases during heating to meet heightened metabolic demands, most increased CO during this period flows through the kidneys, suggesting vasoconstriction in other organs to maintain cerebral perfusion (Wu et al., 2018). Rewarming likely represents a particularly sensitive period for the central nervous system; it is thus important to avoid large fluctuations in CBF during this period (Al Yazidi et al., 2015).
Practical assessment: Active adjustment of cardiovascular medications throughout the rewarming period may be required to avoid iatrogenic hypertension and excessively unregulated CBF (Wintermark et al., 2011). Features of centralization of the circulation: decreased vascular resistance; increased Vmin; increased blood flow; and decreased RI. Features of cerebral edema: increased vascular resistance; decreased Vmin or reversed flow; and increased RI. An RI MCA of 0.5 or less is associated with impaired psychomotor development (Dobrzańska and Bokiniec, 2019).
Visceral circulation
Blood velocity and volume in the superior mesenteric artery (SMA) increase linearly with gestational age and body weight. In the first 2 hours of a newborn’s life, the average blood flow velocity in the SMA is very low, close to zero, reflecting the high resistance of the vessel. Visceral vascular resistance depends not only on the centralization of the circulation but also on the presence of DA. Introducing the first enteral feeding to a newborn infant increases flow in both the visceral trunk and the SMA. Under oxygen-deficit conditions, reduced blood flow can be observed in the intestines and kidneys, in which case, the precapillary vascular sphincter activity of the intestinal wall depends on the activity of the intestinal wall’s precapillary vasculature to bring oxygen to the cells of the intestinal wall. In less perfused areas of the intestine, the tension of the sphincters is reduced, equalizing the pO2 gradient between the capillary vessel and the endothelial cell (Nowicki and Nankervis, 1994). HIE often leads to impaired visceral blood flow velocity, yet the impact of TH on gastrointestinal blood flow in affected newborns remains uncertain (Sakhuja et al., 2019). While brain injury is the primary complication of HIE, multiorgan dysfunction, including renal, pulmonary, cardiac, and gastrointestinal issues, is also prevalent (Rodriguez et al., 2020; Elstad et al., 2016). Neonates with PA episodes are particularly susceptible to feeding intolerance and necrotic enterocolitis (Lu et al., 2017). In a prospective study by Sakhuja et al., flow in the celiac arteries, SMA, and LVO remained low during hypothermia but significantly increased post-rewarming, coinciding with elevated LVO (Sakhuja et al., 2019). This suggests that TH has a protective effect on the gastrointestinal tract, possibly mitigating reperfusion injury.
Practical assessment: SMA flow in ultrasound. Starting early, minimal enteral feeding (10–20 mL/kg/day) during hypothermia within the first few days of life is safe and feasible in neonates with HIE (Thyagarajan et al., 2015). Whole-body hypothermia may even have beneficial effects on gastrointestinal morbidity and feeding tolerance (Thornton et al., 2014). However, more than minimal feeding is less safe because intestinal perfusion may be reduced during cooling.
Renal circulation
As in SMA, flow in the renal artery (RA) increases with gestational age and weight; however, this occurs due to an increase in vessel diameter rather than in flow. This increase can be observed up to the 37th week of pregnancy (Visser et al., 1992). The kidneys are the most abundantly circulated organ of the body. Approximately 20% of the CO flows through the kidneys, which account for only 0.4% of body weight. With an open DA, only 6.6% of the heart’s minute volume flows through the kidneys (Zibolen, 1994; Cleary et al., 1996). The main resistance vessels in the kidney are the glomerular supply arteries. Along their course, blood pressure decreases from 90 to 50 mm Hg. The diameter of the DA and its resistance change depending on the arterial pressure. This mechanism (the Bayliss phenomenon) allows the kidneys to maintain a constant blood flow, regardless of fluctuations in systemic pressure. The average blood flow in a newborn in the right and left renal arteries is 21 mL/min/kg ± 5 (Cleary et al., 1996; van de Bor, 1995).
During the first 3 days of a newborn’s life, resistance in the renal vessels decreases, and renal blood flow increases, increasing diuresis. Particularly early on, vascular resistance decreases in the superficial areas of the renal cortex. Zibolen evaluated neonatal blood flows at different levels of the renal vessels (Zibolen, 1994). He found statistically significant differences between the RI values of the RA (RI: 0.82) and interstitial arteries (RI: 0.76), indicating that blood flow in the RA does not always reflect the total renal circulation. In hypothermic conditions, renal tubular reabsorption, especially of sodium ions, decreases proportionally with temperature reduction (Sosnowski et al., 2015). Research examining descending aorta blood flow (DABF) in neonates with HIE compared with healthy controls has highlighted TH’s impact on peripheral vascular resistance and lower-body organ perfusion (Yoon et al., 2018). The DABF pattern is a potential marker for systemic perfusion in lower-body organs, akin to observations in patients with DA (Evans and Iyer, 1994). During TH, DABF and the DABF–LVCO ratio reduce markedly, indicating increased vascular resistance and decreased perfusion to systemic organs. Consequently, cautious use of inotropic agents in HIE neonates during TH is advisable, given their potential adverse effects on systemic circulation and LV function.
While TH’s short-term effects on renal and myocardial function in HIE neonates have been investigated, there is a gap in our understanding of its long-term impact on renal function post-PA (van Wincoop et al., 2021). Meta-analysis results from nine studies suggest that TH reduces the incidence of acute kidney injury (AKI) after PA. A meta-analysis showed a significant difference in the incidence of AKI between TH-treated neonates (33.3%) and a control group (42.3%), potentially mitigating the risk of chronic kidney injury and offering renoprotective effects.
Practical assessment: RA flow on ultrasound. Serial measurements of SCr and electrolytes and continuous close monitoring of urine excretion are required to assess and track the effects of asphyxia on renal function. If urine excretion drops below 1 mL/kg/h, drug therapy should be considered.
Ductus arteriosus
During hypothermia, several effects can be seen in the DA, including the maintenance of its patency and an increased RI. Bidirectional or right–left flow through the DA can also be observed, indicating increased resistance in the pulmonary circulation (see the Pulmonary Circulation section). This was confirmed by Yoon et al. (2018), who measured the Doppler resistance index of the DA and DABF in newborns with HIE and healthy controls to assess the effect of TH on peripheral vascular resistance and lower-body organ perfusion. The pattern of DABF as a potential marker of lower-body systemic perfusion was studied in neonates with persistent DA. The results showed that the patients had significantly reduced lower-body organ perfusion along with increased vascular resistance during TH, as indicated by a decreased DABF and DABF–LVCO ratio and an increased DA resistance index. The authors investigated the change in peripheral vascular resistance of the lower body by measuring the DA resistance index. The results suggest that increased peripheral vascular resistance plays an additional role in the preferential cerebral redistribution of LVCO during TH. Half of the patients had HIE lesions on a brain MRI and showed more preferential cerebral LVCO redistribution during TH, similar to RI DA. A correlated finding was described by Hochwald et al. (2014).
The Potential Effects of Medications
PA and TH change the pharmacokinetics and pharmacodynamics of drugs. The volume of distribution and drug clearance decreases during cooling, increasing the risk of high drug concentrations and side effects (Zanelli et al., 2011). Drug distribution and clearance increase during the rewarming period, which is associated with a risk of low drug concentrations. As part of multiorgan dysfunction, renal and hepatic failure also affects drug elimination. Interactions between concurrently administered drugs and the cardiodepressant side effects of anticonvulsants and tranquilizers also add to this complexity (Davies and Lefkowitz, 1984; Kirupakaran et al., 2017).
A detailed description of the effects of cardiotonic drugs is beyond the scope of this review; general effects are presented by Nestaas (Table 3; Nestaas and Brian, 2020; Zanelli et al., 2011; Davies and Lefkowitz, 1984; Kirupakaran et al., 2017; Dempsey and Rabe, 2019; Noori and Seri, 2012; Giesinger and McNamara, 2016; Bischoff et al., 2021). However, these effects are not well studied in newborns, making the use of cardiotonic drugs less evidence-based. In addition, the effects of hypothermia on the potency, receptor response, distribution, and metabolism of cardiotonic drugs have not been studied in neonates. Animal studies have shown that temperature can affect the inotropic effects of these agents, which decrease as body temperature decreases.
Expected Effects of Fluid Bolus and Cardiotonic Drugs b

↔ no effect; ↓ decrease; ↑ increase.
Consider the benefits and risks of milrinone during TH. A significant reduction in DAP results in an escalation of cardiovascular support [Nestaas and Brian, 2020].
Nestaas and Brian, 2020; Zanelli et al., 2011; Davies and Lefkowitz, 1984; Kirupakaran et al., 2017; Dempsey and Rabe, 2019; Noori and Seri, 2012; Giesinger and McNamara, 2016; Bischoff et al., 2021.
LV, left ventricular; RV, right ventricular; TH, therapeutic hypothermia.
Conclusion
While TH is the cornerstone of HIE treatment, careful navigation of the delicate equilibrium between therapeutic benefits and potential cardiovascular instability is required. The intricacies of cardiovascular hemodynamics during both TH and the subsequent warming phase underscore the need for a comprehensive understanding. Understanding pathophysiological shifts is crucial for tailoring individualized therapeutic strategies aimed at optimizing cerebral perfusion while mitigating secondary injury risks. Our findings emphasized the importance of an approach that addresses the theoretical perils of isolated hyper- or hypoperfusion across various organs, as well as the risks associated with transitioning between these states. However, uncertainties persist regarding the optimal arterial pressure and CO values that can minimize aberrant fluctuations in cerebral perfusion, underscoring the necessity for further prospective scientific inquiry. Tables 4 and 5 present practical management strategies for cardiovascular alterations during hypothermia and rewarming.
Practical Management Strategies for Cardiovascular Alterations

BP, blood pressure; CBF, cerebral blood flow; HR, heart rate; LV, left ventricular; LVCO, left ventricular cardiac output; MPI, myocardial performance index; RI, resistance index.
Assessment of Systemic Hemodynamics and Suggested Therapy

DAP, diastolic arterial pressure; LV, left ventricle; PPHN, persistent pulmonary hypertension of the newborn; PVR, pulmonary vascular resistance; RV, right ventricle; r–l, right to left; SAP, systolic arterial pressure; SVR, systemic vascular.
Authors’ Contributions
N.B. conceptualized the study, conducted the investigation and data curation, drafted the initial article, and approved the final article as submitted. V.B. collected materials. R.B. completed the methodology and validation of the study, designed the study, reviewed and revised the article, and approved the final article as submitted. R.B. and N.B. contributed to the conception of the review. All authors read and approved the final version.
Footnotes
Acknowledgments
The authors would like to thank Editage for assistance in English language editing.
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Funding Information
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
