Abstract
Perinatal asphyxia is a critical condition frequently associated with hemodynamic instability, often necessitating vasoactive support. Despite the established benefits of therapeutic hypothermia, significant mortality and long-term morbidity persist, particularly in severe cases. The objective of this narrative review is to critically evaluate the available evidence on vasoactive therapies in asphyxiated neonates, compare the hemodynamic effects of commonly used agents, and propose a physiology-based, phenotype-driven approach to cardiovascular management during therapeutic hypothermia. Available evidence remains limited and heterogeneous, with few high-quality studies specifically addressing this population. While commonly used vasoactive agents exert distinct physiologic effects, no single agent has demonstrated clear superiority in improving clinical outcomes. Current evidence supports an individualized, physiology-based approach to vasoactive therapy guided by the underlying hemodynamic phenotype and informed by multimodal monitoring. Further prospective studies are needed to define optimal therapeutic strategies and improve outcomes in this high-risk population.
Keywords
Introduction
Perinatal asphyxia is a critical event, with an estimated incidence of 3.0 per 1000 live births around the world.1,2 Perinatal asphyxia has a complex pathophysiology, often leading to multiorgan dysfunction of varying severity.3–5 Hypoxic ischemic encephalopathy (HIE) remains a major cause of neonatal mortality.6,7 Despite the evidence-based benefits of therapeutic hypothermia (TH) in moderate to severe HIE,8,9 the mortality remains high in severe HIE, 10 and approximately 30% of survivors develop major neurodevelopmental disabilities.11,12
In this review, perinatal asphyxia refers to the acute hypoxic-ischemic insult occurring around the time of birth, reflecting a pathophysiologic process of impaired gas exchange and reduced tissue oxygen delivery. This differs from the broader World Health Organization (WHO) definition of birth asphyxia, which describes a newborn who fails to initiate and sustain breathing at birth. In contrast, HIE denotes the neurological clinical syndrome resulting from perinatal asphyxia, characterized by neurological dysfunction and often accompanied by multisystem involvement.
A prior review by Joynt et al. provided an important overview of cardiovascular supportive therapies in neonates with asphyxia, summarizing both pre-clinical and clinical evidence. 13 Since then, additional studies have emerged, and there has been increasing recognition of the heterogeneity of hemodynamic disturbances in infants with HIE. The objective of this review is to critically evaluate the available evidence on vasoactive support in neonates with perinatal asphyxia, compare the hemodynamic effects of commonly used agents, and propose a physiology-based, phenotype-driven approach to cardiovascular management, particularly in the context of TH. This updated perspective aims to better align physiologic understanding with contemporary clinical practice and support practical bedside decision-making in this high-risk population.
Methods and literature search
This narrative review was based on a structured literature search of PubMed/MEDLINE, Embase, and Cochrane Library databases up to December 31, 2025. Search terms included combinations of “perinatal asphyxia,” “hypoxic-ischemic encephalopathy,” “neonate,” “vasoactive agents,” “inotropes,” “dopamine,” “dobutamine,” “epinephrine,” “norepinephrine,” “vasopressin,” “milrinone,” and “hydrocortisone.” Additional relevant articles were identified through manual review of reference lists. We included clinical studies, observational cohorts, randomized trials, and relevant experimental studies focused on neonatal populations or translational models of asphyxia. Given the limited availability of high-quality neonatal data, selected animal studies were included in order to support physiologic interpretation.
Perinatal asphxia and circulatory changes
Perinatal asphyxia occurs due to a failure of placental gas exchange or the lack of blood flow around the time of delivery (e.g., umbilical cord prolapse or placental abruption). Under normal fetal conditions, approximately 40% of the combined ventricular output is directed to the placenta via the umbilical circulation. The remaining fetal cardiac output is distributed among systemic organs, with a preferential proportion directed to vital organs such as the brain, heart, and adrenal glands. 14 Following an asphyxia event, a centralization of blood occurs with redistribution of cardiac output, preferentially, towards the three vital organs, the brain, heart and the adrenal glands. 15 This is considered a protective mechanism, known as the “diving reflex,” to preserve perfusion to such critical tissues.16,17 Diving reflex is primarily mediated by a catecholamine surge, which induces vasoconstriction in both pulmonary and peripheral vascular beds, thereby diverting blood away from less vital regions.16,17 As a result, there is a significant reduction in the fetal pulmonary blood flow (normally approximately 10% of right ventricular output). 18
While advantageous in utero, these adaptations may predispose the neonate to circulatory instability and delayed relaxation of pulmonary vascular tone after birth. Postnatally, elevated pulmonary vascular resistance promotes right-to-left ductal and atrial shunting, resulting in the delivery of poorly oxygenated blood to the postductal systemic circulation. In contrast, preductal circulations—including the coronary and cerebral circulations—may remain relatively better oxygenated, depending on the degree and pattern of shunting. 18 The PVR is further aggravated by TH, commonly used following asphyxia. 19 Other hemodynamic changes following the asphyxia events include systemic hypotension, cardiac compromise, and acute pulmonary hypertension. 8
Multiple factors contribute to hemodynamic instability following the asphyxia. The neonatal myocardium is susceptible to the effect of hypoxemia and ischemia, and direct myocardial injury following asphyxia is well recognized.
20
Such injury could be detected with a 12-lead electrocardiography,
21
elevated serum troponin T measurement,
22
or echocardiographic assessment.
23
Additionally, several other factors commonly observed include impaired autoregulatory function,
24
reperfusion injury,25–27 systemic hypotension,
28
adrenal hemorrhage, sedation-induced vasoplegia, and hypovolemia18,29 collectively contribute to ongoing cerebral hypoxic injury (Figure 1). Complexity of hemodynamics following perinatal asphyxia (adapted from Surak A et al., Hemodynamics in infants with hypoxic ischemic encephalopathy: pathophysiology and beyond. J Perinatol. 2025 Dec 1).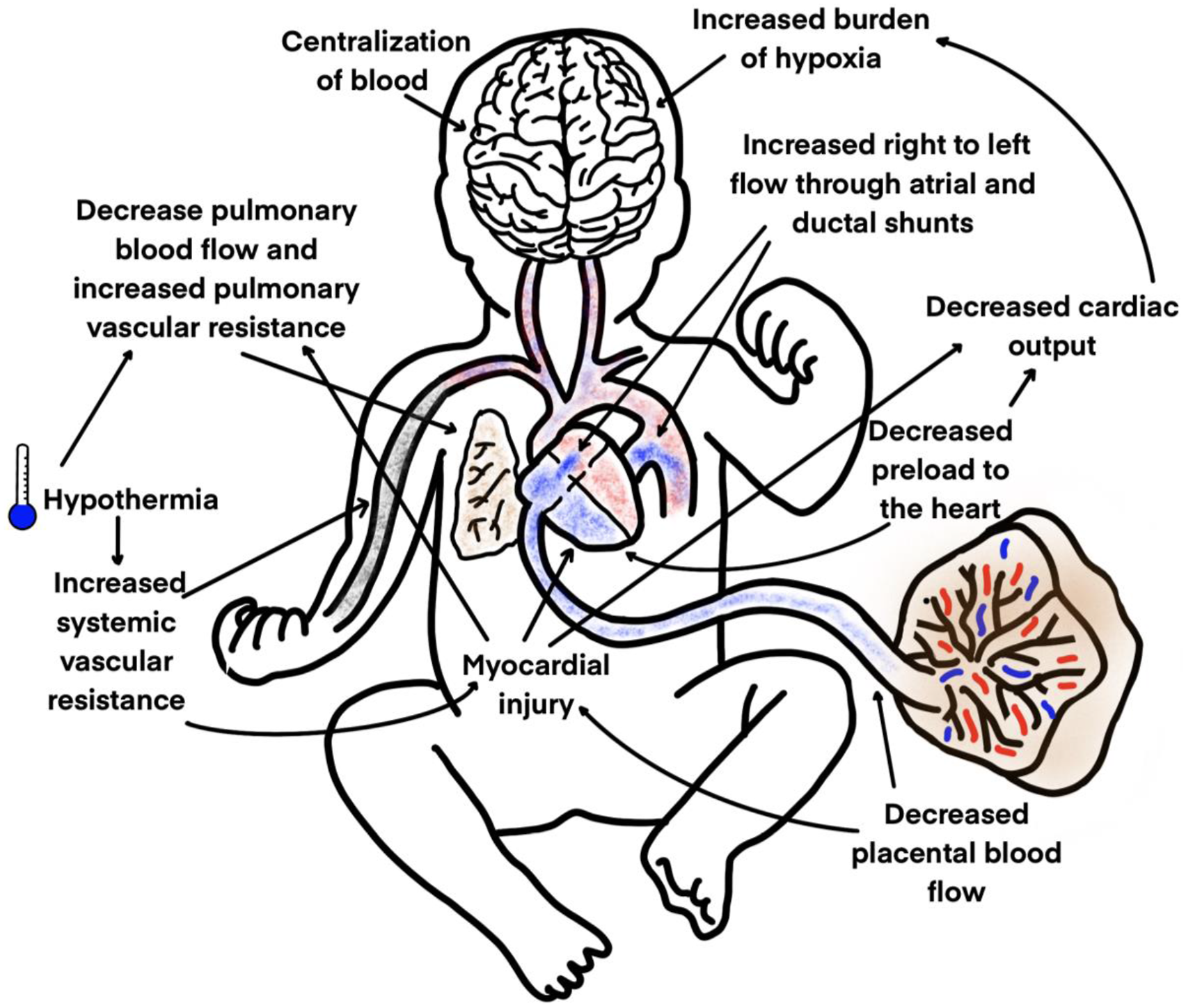
As a result, infants with HIE are commonly exposed to vasoactive agents for stabilization and reversal of shock. There is a paucity of literature regarding the impact of inotropic support in asphyxiated neonates and how different inotropic approaches may affect outcomes. In this review, we summarize the existing literature on inotropic use in neonates with HIE following perinatal asphyxia and discuss how different agents target distinct hemodynamic phenotypes in this population.
Effects of vasoactive agents following asphyxia.
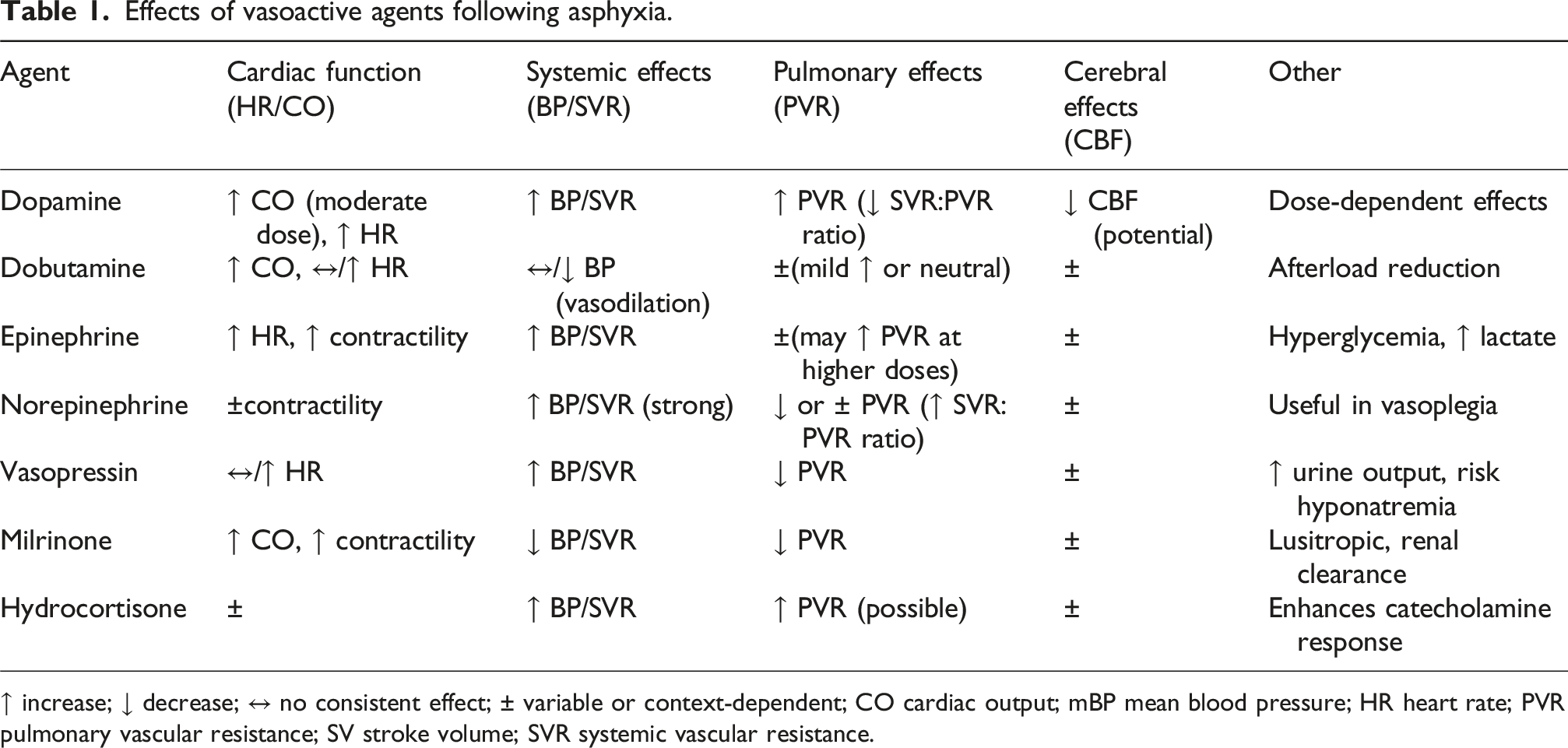
↑ increase; ↓ decrease; ↔ no consistent effect; ± variable or context-dependent; CO cardiac output; mBP mean blood pressure; HR heart rate; PVR pulmonary vascular resistance; SV stroke volume; SVR systemic vascular resistance.
Hemodynamic approaches and current use of vasoactive agents
Circulatory compromise and hemodynamic instability after asphyxia is typically treated using vasoactive agents, with limited evidence to support one agent over another.30–32 The choice of these agents varies across centers; a large survey of 71 tertiary care centers in North American reported a lack of consensus on how cardiovascular parameters (e.g., hypotension) are defined and managed, as well as inconsistency and variability of use of vasoactive agents. 33 Here we summarize the available clinical evidence for the most common vasoactive agents used in this context.
TH introduces additional complexity to vasoactive management by altering drug pharmacokinetics and pharmacodynamics. Cooling is associated with reduced hepatic enzyme activity, impaired renal clearance, and changes in drug distribution, which may prolong the half-life of vasoactive agents.34,35 A decrease in drug clearance—particularly for renally eliminated compounds—has been consistently demonstrated during hypothermia, increasing the risk of drug accumulation.34,36 Furthermore, both hypothermia and perinatal asphyxia contribute to alterations in drug absorption, metabolism, and elimination, leading to variability in drug exposure. 37 In addition, hypothermia may influence adrenergic receptor responsiveness and myocardial performance, potentially modifying the expected hemodynamic effects of vasoactive agents. These factors, together with dynamic changes during the rewarming phase, underscore the importance of careful titration and ongoing hemodynamic reassessment in this population. 35
Dopamine
Dopamine, an endogenous catecholamine and precursor of norepinephrine is the most commonly used vasoactive agent in the NICU due to its neuroendocrine and sympathetic effects.33,38 Dopamine has a dose-dependent effect, which is mediated through stimulation of adrenergic and dopaminergic receptors; the cardiovascular effects of dopamine are attributed to the release of norepinephrine from the peripheral sympathetic nerve ending. 39 With prolonged use, tachyphylactic downregulation of catecholamine receptors might occur. 40
Dopamine is commonly used as first-line vasoactive support to increase blood pressure (via adrenergic effects) and may improve cardiac performance in some infants.41,42 Walther et al. observed higher cardiac output, stroke volume, and heart rate with escalating dopamine doses (4-10 mcg/kg/min) in a cohort of 22 infants with myocardial dysfunction, 11 of whom were asphyxiated. 43 In a piglet model of hypoxia and reoxygenation, dopamine increased the pulmonary-to-systemic pressure ratio, thereby potentially worsening right-to-left shunting and further exacerbating hypoxemia.44–46
Although, dopamine is commonly used to reverse shock, the observed increase in blood pressure is not different, when compared to epinephrine (RR of increasing blood pressure = 0.806 (95% Confidence Interval 0.602, p = 1.080). 47 Although comparative data from neonatal shock populations suggest similar effects on blood pressure between dopamine and epinephrine, the applicability of these findings to infants with HIE remains uncertain.
A systematic review by Hunt et al. compared low-dose dopamine at 2.5 mcg/kg/min with placebo, found no difference in mortality or long-term neurodevelopmental outcome, concluding insufficient evidence from randomized trials that dopamine improves mortality or long-term neurodevelopmental outcomes in asphyxiated neonates. 41 It is worth mentioning that this review included only one randomized trial 48 by DiSessa et al. who reported no difference in mortality between the dopamine (0/7) and placebo (2/7). 48 This reflects the lack of robust interventional trials in this patient population.
Further, in a randomized trial by Pazandak et al.; authors demonstrated that dopamine was not associated with an increased risk of death or abnormal MRI findings in 18 infants with HIE, compared to 30 controls. 49
Despite its widespread use, robust comparative trials in neonates with HIE remain sparse, and current practice is largely guided by physiologic rationale.13,33 While dopamine reliably increases blood pressure, its effects on systemic blood flow and cerebral perfusion are variable, and outcome benefits remain unproven.33,50
Dobutamine
Dobutamine is a synthetic catecholamine that primarily acts through direct stimulation of β1-adrenoreceptors, enhancing inotropy and chronotropy, as well as β2-adrenoreceptors, which could promote peripheral vasodilation. 51 In neonatal piglets, dobutamine has been associated with mild peripheral vasodilation and subsequent afterload reduction. 52 Similar to dopamine, prolonged administration may result in tachyphylaxis due to receptor downregulation of catecholamine. 40
Clinically, dobutamine has some favorable hemodynamic effects in asphyxiated infants. In this setting, dobutamine is often chosen when the dominant problem is low cardiac output due to impaired myocardial performance, rather than pure vasodilatory hypotension. In an observation study of 7 asphyxiated neonates, dobutamine infusion was associated with increase in cardiac output, which has been observed during both TH and the rewarming phase. 53 Similarly, Martinez et al. reported improved cardiac output without a significant effect on the blood pressure in a cohort of 13 critically ill neonates. 54
There is limited evidence from small studies/physiology-driven evidence, suggesting dobutamine’s ability to augment cardiac output may be blunted at cooling temperatures and more effective during rewarming.13,33
Overall, evidence remains limited, but available data suggest dobutamine may improve cardiac output with variable effects on blood pressure, making it potentially useful in low cardiac output states; however, its impact on end-organ perfusion and long-term outcomes in infants with HIE remains uncertain.
Epinephrine
Epinephrine is an endogenous catecholamine, which has a dose-dependent mechanism on adrenergic receptors (α1, α2, β1 and β2). At infusion dose between 0.02 and 0.1 mcg/kg/min, the predominant effects are mediated through β1- and β2-adrenoreceptors, thereby enhancing cardiac contractility and chronotropy; higher doses (>0.1 mcg/kg/min) will stimulate the α1-adrenoreceptors, leading to peripheral vasoconstriction and increased blood pressure; while rarely used in the neonatal population, a very high doses (>0.5 mcg/kg/min) can cause coronary vasoconstriction potentially impairing coronary perfusion.55–57
In hypoxia-reoxygenation piglet model study, Manoucheheri et al. monitored the hemodynamics following infusion of either high dose dopamine or a combination infusion of both dopamine and epinephrine. 46 Authors demonstrated similar improvement in cardiac output in both groups (both groups vs control; p < 0.05), similar increase in the BP, and improved mesenteric and carotid blood flow. The commination infusion regimen resulted in decreased PVR/SVR ratio. 46 No significant effect on renal perfusion was observed. Other animal studies also have reported that epinephrine may augment the systemic-to-pulmonary vascular resistance (SVR:PVR) ratio, supporting its potential role in acute pulmonary hypertension.58,59
Clinical evidence of using epinephrine in human neonates is limited and studies are often small or observational. A pediatric pharmacotherapy review, reports that continuous low-dose epinephrine (starting around 0.03 mcg/kg/min in some series) was safe/effective for shock refractory to other measures in neonates including those with birth asphyxia, but high-quality asphyxia-specific RCT outcome data are lacking. 60 While epinephrine provides both inotropic and vasopressor support, its impact on cerebral perfusion and long-term outcomes in asphyxiated neonates remains uncertain.
Norepinephrine
Norepinephrine is an endogenous catecholamine. Its primary action is mediated through α1-adrenoreceptor stimulation, with less consistent β1- and β2-adrenoreceptors effects. 61 As a result, norepinephrine predominantly induces peripheral vasoconstriction, producing variable inotropic effects and thereby improving blood pressure. At higher doses or with prolonged infusion, severe vasoconstriction may impair myocardial contractility and increase myocardial oxygen consumption.
Using a newborn piglet model of hypoxia and reoxygenation, Cheung et al. demonstrated dose-related (0.05-0.2 mcg/kg/min) inotropic effects (e.g., blood pressure) and increased carotid blood flow. 62 These effects were comparable to epinephrine, without hyperlactatemia.
Clinically, the most common application of norepinephrine in asphyxiated newborns, is when the dominant physiology is vasoplegia/low peripheral vascular tone or refractory hypotensive states, particularly sepsis-related. 63 When HIE is complicated by acute pulmonary hypertension/right ventricular strain, most reviews note potential benefits from improving systemic/coronary pressures supporting RV perfusion; additionally, norepinephrine has a favorable impact when hypotension coexists with pulmonary hypertension physiology. 64 Norepinephrine influence on PVR remains unclear, however, it appears to increase the SVR:PVR ratio, which is a considered favorable effect in the setting of severe acute pulmonary hypertension, which often accompanies perinatal asphyxia. 65
Cerebral perfusion considerations: Because HIE care prioritizes brain protection, the effect of raising MAP with vasopressors on cerebral perfusion is a key concern; neonatal physiology studies discuss that NE can increase MAP and may influence cerebral blood flow measures depending on autoregulation and monitoring modality.
Regarding cerebral perfusion, which should be prioritized for brain protection, it appears that low-dose norepinephrine, despite a homogeneous and significant increase in arterial blood pressure, has little effects on cerebral perfusion and oxygenation during shock. 66
Overall, despite encouraging experimental findings, the benefit-risk profile of norepinephrine in asphyxiated neonates remains unclear. Clinically, norepinephrine may be beneficial in vasoplegic states and in the presence of pulmonary hypertension, although data on its effects on cerebral perfusion and neurodevelopmental outcomes are limited.
Vasopressin
Vasopressin is an endogenous hormone with potent systemic vasoconstrictive effects mediated primarily through the V1 receptors on vascular smooth muscle. 67 In contrast, within the pulmonary circulation it promotes nitric oxide release, resulting in selective vasodilation.68,69 Beyond its vascular effects, vasopressin also exerts antidiuretic actions via V2 receptors stimulation in the renal tubules, thereby regulating water reabsorption.70,71 Vasopressin has been increasingly used, particularly for refractory conditions such as septic shock, catecholamine- and corticosteroid-resistant shock, hypotension due to ventricular outflow tract obstruction, post-cardiopulmonary bypass instability after congenital cardiac surgery, and acute pulmonary hypertension.72,73 More recently, case series and observational studies have reported improved blood pressure,71,74 reversal of acidosis, better oxygenation indices, 75 increased urine output, reversal of shock,74,76–78 and improvement in hypoxemic respiratory failure states. 79
In experimental model of asphyxiated newborn piglets, vasopressin had a dose-dependent effect on the blood pressure, while preserving cardiac function, cerebral and mesenteric hemodynamics. 80
Direct human evidence of vasopressin role following asphyxia is limited, and it is mostly extrapolated from neonatal shock, acute pulmonary hypertension based on physiology-guided reasoning). However, pre-clinical studies suggest a favorable hemodynamic profile of vasopressin, characterized by pulmonary vasodilation,81,82 improved systemic blood pressure, and preserved cardiac output. 83 However, prolonged administration should be avoided due to adverse effects including hyponatremia, thrombocytopenia, and liver necrosis.76,84 Vasopressin may offer favorable effects on pulmonary and systemic hemodynamics; however, evidence regarding its impact on cerebral perfusion and long-term outcomes remains sparse.
Milrinone
Milrinone, a derivate of the bipyridine amrinone via methylation and carboxylation, is approximately 15 times more potent than its parent compound.85,86 It is a phosphodiesterase-3 (PDE3) inhibitor that increases intracellular cyclic adenosine monophosphate (cAMP), leading to vascular smooth muscle relaxation and vasodilation.87,88 In cardiac myocytes, milrinone enhances calcium handling, thereby improving contractility. 89
Milrinone has been used in select cases of neonatal cardiac dysfunction, particularly in the presence of pulmonary hypertension and impaired myocardial performance; however, evidence supporting its use in infants with HIE following perinatal asphyxia remains limited, and its use in this population should be approached with caution given the risk of systemic hypotension and limited safety data.90–94
Both animal and clinical studies have demonstrated its impact on pulmonary vasodilation properties across pediatric and adult population.95,96 Clinically, this translates into improved oxygenation requirements without compromising systemic hemodynamics.97–99 Evidence also suggests milrinone may reduce the incidence of low cardiac output state and improve survival.90,100 Beyond its inotropic effects, milrinone exerts lusitropic benefits by facilitating calcium dissociation from troponin C during diastole, thereby promoting myocardial relaxation.101,102
The combination of inotropic, lusitropic, and pulmonary vasodilatory properties makes milrinone theoretically advantageous in asphyxiated neonates with myocardial dysfunction and pulmonary hypertension. Additionally, PDE3 inhibition may promote ductal patency, 103 potentially contributing further benefit in this context. In a newborn piglet model of asphyxia, milrinone infusion improved blood pressure, enhanced pulmonary hemodynamics, and increases in carotid and intestinal blood flow. 104
However, there is paucity of clinical evidence in human neonates with asphyxia, and safety concerns have been raised in this particular population. In a retrospective cohort of 56 infants with HIE, Bischoff et al. reported significant reductions in diastolic blood pressure in those treated with milrinone, possibly due to drug accumulation with acute kidney injury. 105 This effect may be explained by milrinone’s relatively long half-life of milrinone (∼4 h) and its reliance on renal clearance, which is frequently impaired after asphyxia. 106
Overall, milrinone may be considered in carefully selected cases of myocardial dysfunction and/or pulmonary hypertension; however, its use in infants with HIE remains limited by safety concerns—particularly systemic hypotension and potential drug accumulation during therapeutic hypothermia—warranting cautious, individualized use with close hemodynamic monitoring.
Hydrocortisone
Hydrocortisone is frequently used alongside inotropic agents in critically ill neonates. 107 Hydrocortisone plays an important role enhancing the effects of catecholamines by inhibiting their degradation, increasing smooth muscle calcium availability, improving vascular tone, and reducing capillary leak.108,109
Adrenal insufficiency and inadequate response to ACTH stimulation, are associated with worse morbidity and mortality, particular is septic shock.110–112 This entity is not uncommon in asphyxiated neonates113,114; hence recognition and treatment of adrenal insufficiency is an important consideration for optimizing cardiovascular support. 115 Additionally, side effects of hydrocortisone should be addressed, including hyperglycemia, gastrointestinal bleeding, and increased pulmonary vascular tone. 116
The evidence of using hydrocortisone in infants with HIE is highlighted by Kovacs et al. In a randomized controlled trial of volume resistant hypotensive HIE infants, administering hydrocortisone 0.5 mg/kg/6 h was associated with increased BP compared to placebo group (94% vs 58%, P = 0.02). 117 Also, there was shorter duration of cardiovascular support (P = 0.001), as well as cumulative (P < 0.001) and peak inotrope dosage (P < 0.001) were lower in the hydrocortisone group. 117
A long term follow up study at a median of 20 months corrected age, death or severe neurodevelopmental impairment occurred in 40% in the hydrocortisone group compared to 18% in the placebo group (P = 0.13). 118 Furthermore, multiple logistic regression revealed that for every 1 mg/kg increase in cumulative hydrocortisone dose, the odds of adverse cognitive outcome increased by 16% (95% CI 1.01-1.37; P = 0.04). 118 Currently, there is ongoing pharmacokinetic study of hydrocortisone therapy during TH treatment in asphyxiated newborns, aiming to establish a personalized and safe care to infants with perinatal asphyxia and hemodynamic instability (UniCort trial, ClinicalTrials.gov ID # NCT05836610). While these data on the neurodevelopmental outcomes raise concerns, further trials are needed to identify a potential safe pharmacological window during perinatal asphyxia. Although hydrocortisone may improve blood pressure and reduce vasoactive requirements, concerns regarding potential adverse neurodevelopmental outcomes highlight the need for cautious use and further study to define a safe therapeutic window.
Synthesis of evidence and clinical implications
Overall, the evidence supporting vasoactive therapy in asphyxiated neonates remains limited and heterogeneous, with most studies being small, observational, or derived from mixed neonatal populations rather than infants with HIE undergoing therapeutic hypothermia. While experimental models demonstrate relatively consistent physiologic effects, translation into meaningful clinical outcomes remains uncertain, and findings across clinical studies are often inconsistent.
Importantly, the current literature is further limited by a lack of adequately powered randomized controlled trials, reliance on short-term physiologic endpoints, and a paucity of long-term neurodevelopmental outcome data. As a result, definitive conclusions regarding optimal agent selection cannot be made, and clinical practice continues to rely largely on physiologic rationale and individualized assessment.
When considered collectively, available evidence suggests that no single vasoactive agent is universally superior; rather, each agent exerts distinct hemodynamic effects that may be advantageous in specific clinical contexts. Dopamine and epinephrine are effective in increasing blood pressure but may not reliably improve systemic blood flow and, in some cases, may adversely affect pulmonary or cerebral hemodynamics. Dobutamine and milrinone may be more beneficial in states characterized by myocardial dysfunction and low cardiac output, although their impact on blood pressure is variable and may be attenuated during TH. Norepinephrine and vasopressin may be particularly useful in vasoplegic states or when pulmonary hypertension coexists, given their effects on systemic vascular resistance and the SVR:PVR ratio.
Taken together, these findings support a shift away from a uniform, blood pressure–driven approach toward a physiology-based strategy that targets the underlying hemodynamic disturbance. This reinforces the importance of multimodal hemodynamic assessment and individualized therapy when selecting vasoactive support in asphyxiated neonates. Importantly, across all agents, evidence linking short-term hemodynamic improvements to meaningful clinical outcomes—particularly cerebral perfusion and long-term neurodevelopment—is limited.
Current hemodynamic approach and future directions
The use of vasoactive agents in asphyxiated neonates remains highly variable, with limited evidence supporting one therapeutic approach over another.30–32 In a retrospective comparative cohort of 176 term infants from 2 countries across two centers in Canada and Hungary, Kovacs et al., examined contrasting management strategies. 32 Center A employed a permissive approach to hypotension, guided primarily by systolic and diastolic blood pressure measurements with echocardiography whenever feasible. In contrast, Center B followed a more aggressive strategy, treating mean blood pressure below gestational age with vasopressors and using echocardiography less frequently. 32 The study found that nearly half of the infants in Center A received cardiovascular agent compared to 97% of infants in Center B, despite similar incidence of documented hypotension. 32 Importantly, while mortality and the overall incidence of abnormal brain MRI findings were comparable, the pattern of brain injury differed. Infants in Center B were more likely to show basal ganglia injury, consistent with hemodynamic compromise, whereas those in Center A, exposed to less aggressive inotrope therapy, more often demonstrated milder forms of injury. 32
From a hemodynamic perspective, infants admitted with HIE following perinatal asphyxia are currently monitored clinically for signs of circulatory compromise, but there is no standardized framework for comprehensive cardiovascular assessment. The above-mentioned survey illustrates this challenge, showing marked inconsistency across centers where only 60% reported routine use of echocardiography, even when infants were receiving cardiotropic medications; cardiac output was assessed in just 20% of centers; and right ventricular function was evaluated in only in 57% of the cases. 33
In clinical practice, evaluating systemic circulation in this population is inherently complex, as it involves multiple interdependent components. 119 Common bedside parameters such as heart rate, blood pressure, or lactate are widely used but each carries significant limitations in accurately reflecting adequacy of perfusion.120–122
Emerging evidence supports the adoption of a comprehensive approach led by neonatologist. Such strategies have been associated with improved outcomes. Including a 50% increased survival free of intraventricular hemorrhage, 123 a reduced burden morbidity, 124 and significant reduction in mortality. 125 The challenges outlined above underscore the need for a comprehensive, standardized, and objective approach to the management of for infants with HIE. This is critical to establish the dominant hemodynamic phenotype likely to contribute to the clinical compromise. 126 Such approach offers the potential to optimize cardiovascular support and improve neurodevelopmental outcomes following neonatal asphyxia. Further research is essential to deepen our understanding of the underlying disease mechanisms, while the optimal pharmacotherapeutic strategy remains to be defined.
Contemporary hemodynamic management in infants with hypoxic-ischemic encephalopathy increasingly incorporates advanced monitoring modalities to better characterize circulatory status. Targeted neonatal echocardiography has emerged as a key tool to assess cardiac function, estimate systemic blood flow, evaluate pulmonary pressures, and identify the dominant hemodynamic phenotype. In addition, multimodal perfusion monitoring—including near-infrared spectroscopy (NIRS), lactate trends, and other biochemical markers—provides complementary information on end-organ oxygen delivery and utilization.
These approaches allow clinicians to move beyond reliance on isolated blood pressure measurements and instead adopt a more comprehensive assessment of cardiovascular performance. Integration of functional echocardiography with continuous or serial perfusion monitoring may facilitate earlier identification of evolving hemodynamic compromise and support more targeted selection and titration of vasoactive therapies. However, variability in access, expertise, and standardization of these modalities remains a limitation, highlighting the need for further research and consensus in this area.
Physiology-based framework for vasoactive selection
In asphyxiated neonates, circulatory compromise is heterogeneous and is best understood in terms of distinct hemodynamic phenotypes rather than isolated hypotension. Accordingly, the selection of vasoactive therapy should be guided by the dominant underlying disturbance rather than blood pressure alone.
Infants with myocardial dysfunction and low cardiac output may benefit from selective contractility agents such as dobutamine, which enhance contractility and augment systemic blood flow. In contrast, those with vasoplegia and reduced systemic vascular resistance are more likely to respond to vasoconstrictors such as norepinephrine or vasopressin, which restore vascular tone and support perfusion pressure.
When pulmonary hypertension coexists—particularly in the presence of right ventricular dysfunction—agents that increase systemic vascular resistance while minimizing or reducing pulmonary vascular resistance may be advantageous, thereby improving the systemic-to-pulmonary vascular resistance ratio. In cases of isolated hypotension without clear evidence of low cardiac output, dopamine or epinephrine may increase blood pressure; however, their effects on systemic blood flow and end-organ perfusion remain variable.
Importantly, these hemodynamic phenotypes often overlap and evolve over time, particularly during therapeutic hypothermia and rewarming. Therefore, integration of multimodal monitoring—including echocardiography, perfusion markers such as NIRS, and biochemical indices—is essential to guide individualized therapy. This phenotype-driven framework supports a shift away from a uniform, blood pressure–based approach toward targeted, physiology-guided hemodynamic management.
Figure 2 illustrates a practical, physiology-based framework for vasoactive selection in asphyxiated neonates, linking common hemodynamic phenotypes to targeted therapeutic strategies. Physiology-based framework for vasoactive selection in asphyxiated neonates. Vasoactive therapy should be guided by the dominant hemodynamic phenotype. Continuous reassessment using echocardiography and perfusion markers is essential, as physiology evolves during THand rewarming. CO, cardiac output; SVR, systemic vascular resistance; TH, therapeutic hypothermia; PVR, pulmonary vascular resistance; RV, right ventricle; HIE, hypoxic-ischemic encephalopathy.
Conclusion
In neonates with HIE following perinatal asphyxia, the choice of vasoactive support should be guided by the underlying hemodynamic phenotype rather than a uniform approach to hypotension. While commonly used agents such as dopamine, dobutamine, epinephrine, norepinephrine, vasopressin, and milrinone exert distinct physiological effects, high-quality comparative outcome data remain limited. Current practice is therefore largely driven by physiologic reasoning and bedside assessment. A targeted, multimodal hemodynamic approach—incorporating functional echocardiography and individualized therapy—holds promise for optimizing cardiovascular support and improving outcomes, but further prospective studies are needed to define optimal strategies.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
