Abstract
Articular cartilage, vital to the health and functioning of joints, remains challenging to regenerate. The pericellular matrix (PCM) is critical for transducing biophysical stimuli to the articular chondrocytes (ACs) that it envelops. Given the mechanobiological sensitivity of ACs, it is pivotal in maintaining the chondrogenic phenotype and the production of extracellular matrix (ECM) during articular cartilage tissue engineering. While the maintenance of the native PCM significantly improves the quality of neocartilage, current isolation methods are limited. A solution to this challenge is facilitating ACs to regenerate their PCM. However, the regulation of PCM synthesis remains poorly understood, hindering the development of effective tissue engineering strategies. This narrative review aims to provide a comprehensive analysis of the complex interplay between extracellular cues and intracellular pathways regulating PCM synthesis during articular cartilage tissue engineering. Our analysis reveals that mechanical cues, such as material stiffness and mechanical stimulation, are the primary regulators of PCM synthesis. Additionally, the use of scaffold-free techniques potentially affects the structuring of newly created PCM. Tuning these stimuli can significantly impact the quality of the formed PCM, ultimately influencing neocartilage quality. Furthermore, we highlight intracellular mechanisms involved in the transduction of these extracellular cues, including actin polymerization, yes-associated protein and transcriptional coactivator with PDZ-binding motif localization, and transforming growth factor beta-induced Smad signaling. Although the current literature suggests the involvement of these signaling pathways in regulating the synthesis of PCM components, we found that studies investigating these processes in ACs are lacking. Elucidating the relationships between extracellular stimuli, intracellular signaling, and the expression of PCM components could provide a framework for designing new cartilage tissue engineering approaches that facilitate proper PCM synthesis. Ultimately, this can improve ECM quality and advance articular cartilage regeneration.
Impact Statement
Matrix synthesis during articular cartilage tissue engineering is highly dependent on scaffold material properties, mechanical stimulation, and the availability of growth factors. The presence of a PCM plays a critical role in mediating cellular responses to extracellular cues, directly affecting matrix synthesis. However, the process of PCM synthesis remains poorly understood. This review aims to bridge the knowledge gap by examining how extracellular cues impact PCM formation, as well as the intracellular mechanisms that transduce these signals. This is key to optimize articular cartilage tissue engineering strategies and ultimately improve outcomes for patients suffering from articular cartilage defects.
Keywords
Introduction
Articular cartilage is a highly specialized connective tissue covering the ends of the long bones. Its unique biochemical composition and structure ensure almost frictionless and pain-free movement of joints and the transmission of biomechanical loads. 1 However, due to its avascular nature, it has a limited regenerative capacity, and the tissue is highly susceptible to degeneration. This often leads to the development of osteoarthritis, but the lesion itself can have similar debilitating effects. 2 The field of tissue engineering offers a promising approach to repair and regenerate damaged articular cartilage.
A key aspect of successful articular cartilage tissue engineering is the cell microenvironment. In the native tissue, articular chondrocytes (ACs) are surrounded by a thin layer of highly specialized pericellular matrix (PCM), together called the chondron. 3 The PCM is key for the transduction of biophysical stimuli to the cell, and the use of chondrons in articular cartilage tissue engineering improves the quality of the newly synthesized tissue. Chondrons produce significantly more collagen and proteoglycan compared with chondrocytes in vitro,4–7 also when isolated from damaged tissue. 8 When cocultured with allogenic mesenchymal stromal cells (MSCs), chondrons had superior outcomes in vitro and in vivo compared with chondrocytes and microfracture, respectively.9,10
Unlike the extracellular matrix (ECM), which mainly consists of type-II collagen and glycosaminoglycans (GAGs), mostly aggrecan, type-VI collagen and perlecan are matrix components exclusively found in the PCM. 11 Type-VI collagen and perlecan are essential for the mechanical stiffness of the PCM and the transduction of mechanical stimuli.12,13 Small proteoglycans such as decorin and biglycan support the type-VI collagen network and the linking of the PCM to the ECM. 14 Hyaluronic acid (HA) facilitates important cell–matrix interactions and retains other matrix components.15–17 Together, these proteins give the PCM its unique structure and function.
Although the use of chondrons for articular cartilage tissue engineering seems promising, its use as an autologous cell source is limited. Digestion of articular cartilage results in a heterogeneous mixture of ACs and chondrons, resulting from enzymatic isolation and the limited yield due to the low cell content of articular cartilage.18,19 Fortunately, ACs are able to reestablish their PCM when embedded in biomaterials such as hydrogels, possibly recapturing its function.6,20,21 However, while the structure and function, 11 the effects on cellular behavior, 22 and the importance of a proper cell microenvironment for cartilage tissue engineering 23 have been thoroughly investigated, the process of PCM synthesis and the influence of in vitro culture conditions remain to be understood.
This review aims to explore the complex interplay of extracellular and intracellular cues that regulate PCM synthesis during articular cartilage tissue engineering. We will review the current literature investigating PCM synthesis to establish the effects of scaffold materials and culture conditions. In addition, we explore intracellular mechanisms that could transduce these extracellular stimuli and affect PCM production.
Extracellular cues regulating PCM formation
To investigate the effect of external cues (material properties, mechanical stimulation, biochemical stimulation, and cell–cell interactions) on PCM synthesis, only articles specifically investigating PCM-specific components were considered. An overview of the collected literature can be found in Table 1. From these studies, we have identified material properties (stiffness, permeability), mechanical stimulation, growth factor stimulation, cell–matrix interaction, and cell–cell interactions as the most significant extracellular signals regulating PCM formation.
Overview of Literature Investigating Pericellular Matrix Synthesis
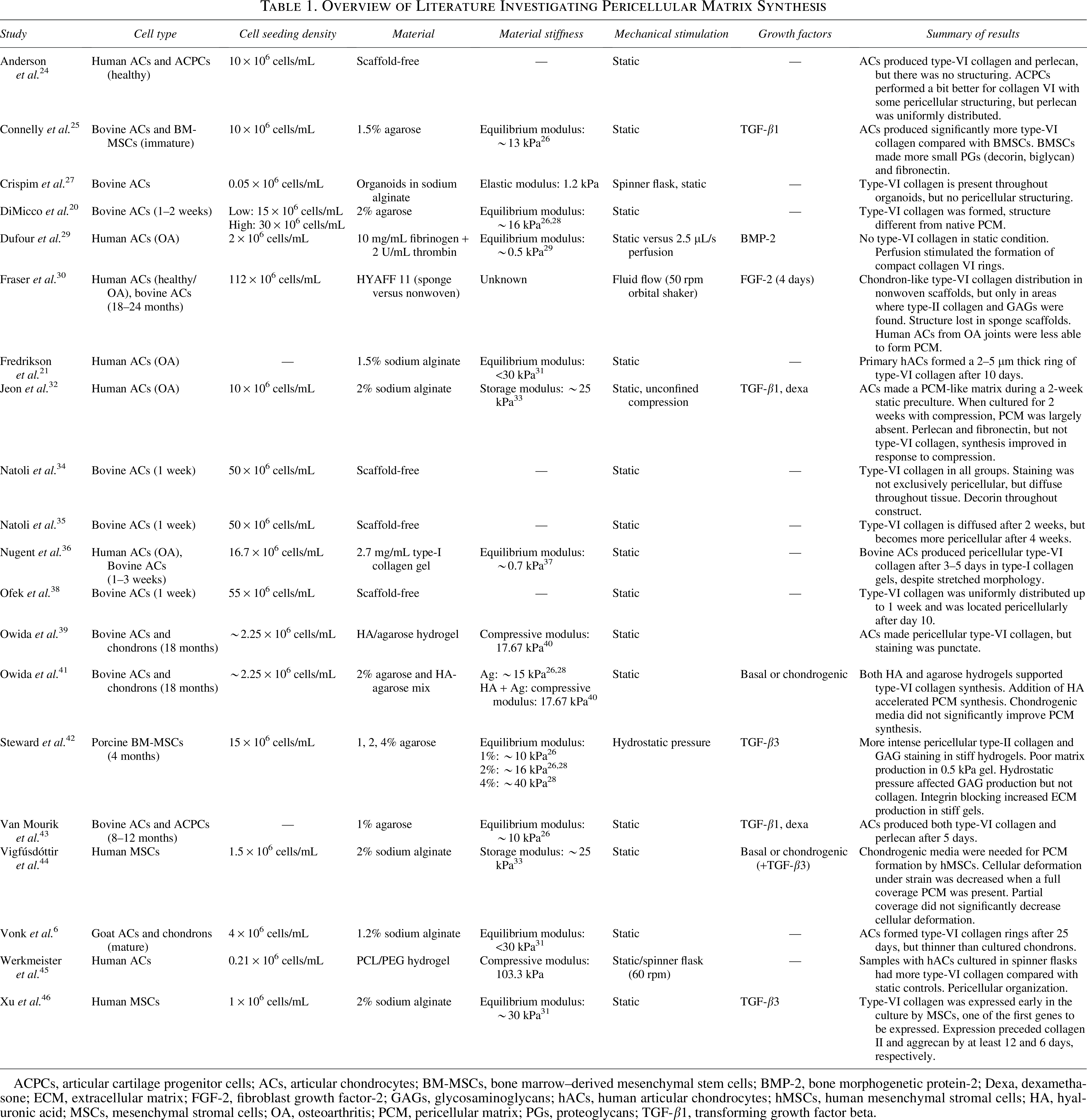
ACPCs, articular cartilage progenitor cells; ACs, articular chondrocytes; BM-MSCs, bone marrow–derived mesenchymal stem cells; BMP-2, bone morphogenetic protein-2; Dexa, dexamethasone; ECM, extracellular matrix; FGF-2, fibroblast growth factor-2; GAGs, glycosaminoglycans; hACs, human articular chondrocytes; hMSCs, human mesenchymal stromal cells; HA, hyaluronic acid; MSCs, mesenchymal stromal cells; OA, osteoarthritis; PCM, pericellular matrix; PGs, proteoglycans; TGF-β1, transforming growth factor beta.
Material properties and composition
Hydrogels are considered a favorable material for cartilage tissue engineering, since articular cartilage is a proteoglycan-rich tissue with high fluid content.47,48 Hydrogels from natural and synthetic origins have been used for articular cartilage tissue engineering, attempting to recapitulate the properties of the native tissue. 48 In this endeavor, however, the main focus is on the bulk properties of the tissue, while the demands of the AC microenvironment are different. 23 Nevertheless, frequently used hydrogels such as agarose, 20 alginate,6,21,49 and type-I collagen gel 36 allow for PCM synthesis.
Material stiffness is an important factor in AC mechanosensing that largely governs the function of the native PCM and determines the cell phenotype during cartilage tissue engineering. Since articular cartilage and hydrogels are poroelastic materials, the material stiffness is often described by the aggregate or equilibrium modulus, which is the stiffness of a material at equilibrium when the fluid has flowed out. 50 The current literature suggests that a relatively stiff material (>10 kPa) is most favorable for the production of PCM components.6,20,21,25,32,39,42,43,45,46 MSCs make a denser and stiffer PCM when the stiffness of agarose gels is increased. 42 However, increasing hydrogel stiffness is commonly achieved by changing the solid volume fraction, decreasing the material porosity, and affecting the diffusion of matrix proteins.51,52 Other studies investigating PCM formation in agarose hydrogels found that type-VI collagen was produced in 1.5–2% agarose.20,25,41 In our recent study, we found that bovine ACs produced both type-VI collagen and perlecan in 1% agarose as quickly as in 5 days. 43 These results indicate that agarose and alginate with an equilibrium modulus ranging from ∼10 to 30 kPa (Table 1) are suitable for PCM production.
In contrast, softer hydrogels appear less favorable for PCM formation. ACs from human osteoarthritic knees in soft fibrin gels 29 (0.5 kPa 53 ) and type-I collagen gels 36 (0.7 kPa 37 ) were not able to produce a PCM. Additionally, fibrin and collagen gels have cell-binding sites, promoting an elongated cell morphology, which is unfavorable to the chondrogenic phenotype. 54 Adding cell-binding sites to soft agarose (3.7 kPa) also significantly decreases GAG synthesis by ACs, while having no notable effect in stiffer agarose (53.2 kPa). 55 This suggests that cell-binding sites, especially in soft hydrogels, negatively influence PCM synthesis. However, since the studies investigating PCM production in stiff hydrogels lack cell-binding sites, drawing a firm conclusion remains challenging.
Aside from the mechanical properties different constitutions of hydrogels provide, the direct interactions of the cells with the matrix and other cells can play a role. Besides the availability of hydrogel cell-binding sites, the availability of matrix components such as HA can also alter PCM production. The addition of HA, also present in the native PCM, 11 can accelerate PCM formation, 41 suggesting that such additions could have beneficial effects. Furthermore, the cell seeding densities have been investigated as high cell seeding densities in scaffolds were observed to have a stimulatory effect on the chondrogenic phenotype, increasing type-II collagen production and GAG content in the ECM.56–58 However, in the current literature, no evidence was found that cell density in hydrogels has a significant effect on PCM formation. However, the wide variety of materials and cell seeding densities used in contemporary literature makes this comparison difficult. As high cell seeding density can have a significant positive effect on neocartilage production, the effect on PCM formation remains a research area of interest.
Mechanical stimulation
Mechanical stimulation is known to have a great impact on cartilage development, homeostasis, and degeneration.59,60 Although mechanical stimulation is such an important factor for in situ tissue engineering of articular cartilage, the majority of the studies investigating PCM formation are performed under static conditions. However, there is some evidence on the effects of mechanical loading.
Shear loading through fluid flow was the most commonly used mechanical stimulus in the literature on PCM synthesis. Crispim et al. 27 grew cartilage organoids in spinner flasks by combining bovine ACs with notochordal cell-derived matrix. This process allowed the formation of organoids rich in cells and ECM and PCM components, although no static control was used to study the effect of flow. Similarly, Fraser et al. 30 observed good PCM formation in HYAFF 11, a HA-based material, when cultured on an orbital shaker, but no static controls were added in the study. Marsano et al. cultured human ACs in HYAFF 11 under static conditions. Compared with the results of Fraser et al., the type-VI collagen formed under static conditions was more diffusely stained. 61 This suggests that fluid flow, most likely inducing shear, influences PCM structure.
Dufour et al. 29 used soft fibrin gels to investigate cartilage deposition by human ACs. As stated earlier, no type-VI collagen was synthesized by the cells under static conditions. However, when perfusion was added to the culture, the cells produced compact, pericellular rings of type-VI collagen. Additionally, the synthesis of type-II collagen and aggrecan greatly improved (Fig. 1A). Likewise, Werkmeister et al. 45 reported that a spinner flask culture of polyethylene glycol-polycaprolactone (PCL–PEG) constructs improved type-VI collagen deposition.
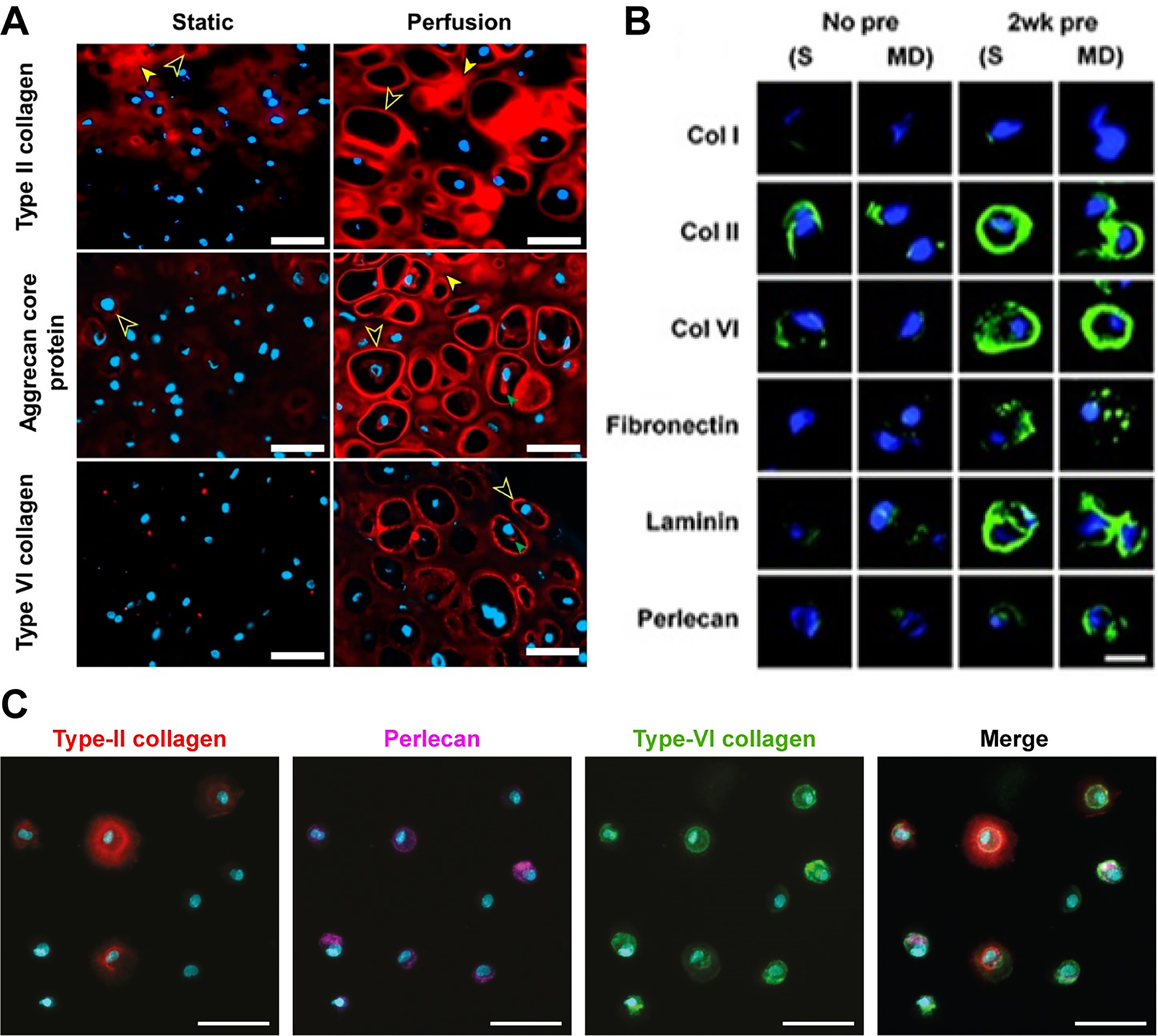
The differences in histological staining between the extracellular matrix (ECM) and intracellular matrix and good versus bad pericellular matrix (PCM) formation.
The addition of fluid flow and/or perfusion creates a cofounded effect of both shear loading and increased solute transport. ACs are known to be sensitive to fluid shear stress, exhibiting both anabolic and catabolic behavior depending on the magnitude of the shear loading. 62 Spinner flask cultures appear to stimulate ACs to form organoids, with Crispim et al. reporting a decrease in chondrogenic marker expression when fluid shear stress is removed. 27 However, these culture conditions only stimulate the cells directly in contact with the fluid, while perfusion culture will ensure that cells in central regions of the construct are stimulated. 29 An increase in solute transport is expected as the culture media circulate when fluid flow is introduced. However, this is expected to only have a significant effect on perfused tissues, as the mass transfer in the center of the tissue is improved. While Dufour et al. report that this likely affects PCM production, its effects cannot be decoupled from the shear stress stimulation, as both require fluid flow. Regardless, adding fluid flow to in vitro cultures appears to have a stimulatory effect on PCM synthesis.
While compressive loading has a significant effect on cartilage metabolism, only one study investigated its effects on PCM production. Jeon et al. 33 found that compression hampers PCM formation by human ACs in alginate in the early stages of culture, as type-VI collagen, perlecan, fibronectin, and laminin stainings were lost. However, 2 weeks of static preculture significantly increased the presence of all PCM markers when compared with no preculture and the static control group (Fig. 1B). An explanation for these results could be provided by Vigfúsdottír et al., 44 who found that cell deformation of MSCs only decreased compared with no PCM when the cells were fully covered with a PCM. Partial PCMs did not significantly decrease cell deformation. These results suggest that, while the application of compressive loading can improve PCM formation, the timing might be crucial.
Besides compression, hydrostatic pressure also influences chondrogenesis. Steward et al. 42 observed an increase in GAG production and SOX9 and COL2A1 gene expression in MSCs in 4% agarose under hydrostatic pressure compared with free swelling gels. Interestingly, this effect was not observed in 1% agarose gels. This effect was diminished when integrin binding to the formed PCM was blocked, indicating that the transduction of hydrostatic pressure is dependent on material stiffness and cell binding to the formed PCM.
Nevertheless, the fact that a PCM is produced without any mechanical loading is an interesting concept since ACs are very mechanoresponsive. In native articular cartilage, one of the main functions of the PCM is strain amplification in low-deformation areas.63–66 It could be hypothesized that the lack of strain on cells in free swelling hydrogels under static conditions initiates the cells to produce a PCM to amplify strains. The observation of PCM formation in materials lacking cell-binding sites supports this hypothesis, as the cells lack attachments to their surroundings and may rely on the PCM to interact with their environment. This hypothesis is, however, purely speculative and requires further investigation.
Growth factor stimulation
Cell stimulation using exogenous growth factors can be used to stimulate chondrogenic differentiation, cell proliferation, and matrix deposition. The most effective exogenous growth factors used in cartilage tissue engineering are transforming growth factor beta (TGF-β) and bone morphogenetic proteins (BMPs).67,68 Additionally, other media supplements such as ascorbic acid and dexamethasone are often added to enhance the effects.69,70
While the use of exogenous growth factors such as TGF-β is known to significantly enhance the synthesis of ECM proteins such as type-II collagen and aggrecan, there is evidence that exogenous growth factor stimulation is not necessary for PCM formation. The majority of the studies investigating PCM formation were executed without exogenous stimulation of the cells.6,20,21,41,45 Owida et al. 41 even showed that the addition of chondrogenic media, supplemented with dexamethasone and TGF-β1, has no significant effect on PCM formation by bovine ACs and chondrons. It should be noted that the basal media in this study contained serum, which can hold a wide range of growth factors that affect cell behavior. 71 Additionally, this does not conclude that TGF-β has no effect on PCM synthesis since ACs are known to produce TGF-β, which is incorporated into the matrix. 72 Due to its central role in chondrogenesis, it is, therefore, highly likely that TGF-β does influence PCM synthesis.
Although the addition of exogenous TGF-β might not be needed, using other cell sources besides ACs could make the use of exogenous growth factors a necessity. Many researchers advocate for the use of alternatives to ACs, since it is a limited autologous cell source, 73 and the chondrogenic phenotype is lost during passaging. 54 Some of the studies that investigated PCM formation used bone marrow–derived mesenchymal stem cells25,42,44,46 or articular cartilage progenitor cells (ACPCs).24,43 To differentiate these cells toward the chondrogenic lineage, exogenous growth factors are still needed. For example, Vigfúsdottír et al. 44 showed that human MSCs needed exogenous TGF-β3 to produce a PCM. Taken together, the current literature implies that exogenous growth factors may not be needed to promote PCM synthesis by ACs but are necessary when using other cell sources.
Scaffold-free approaches
Besides cartilage tissue engineering approaches utilizing biomaterials, many studies have used scaffold-free methods to regenerate articular cartilage tissue. Different techniques can be used to facilitate cell condensation for cartilage growth. Cells can be seeded in a nonadherent environment to form cell aggregates,24,34,35,38 although an adherent surface is needed if a particular geometry is desired. 24 These approaches typically involve very high cell densities (Table 1). Alternatively, ACs can self-assemble into organoids when cultured in a spinner flask. 27
While the methodology to form scaffold-free neocartilage might be different, the analysis of PCM formation shows similar results. All studies analyzing PCM formation in scaffold-free approaches report diffuse type-VI collagen staining throughout the neocartilage tissue.24,27,34,35,38 These observations are similar to the distribution of matrix proteins during postnatal development of articular cartilage as type-VI collagen was not condensed around the cells but distributed throughout the matrix.74,75
However, during the maturation of articular cartilage, the matrix is remodeled, and typical chondron structures are formed.74,75 Such tissue maturation is reported by Ofek et al., 38 who observed pericellular type-VI collagen staining after 4 weeks while staining was diffuse after 2 weeks of culture. Anderson et al. 24 reported similar tissue maturation, but only by articular cartilage progenitor cells (ACPCs). The human primary ACs did not display this fast rate of tissue remodeling. ACPCs are known to produce matrix much faster than primary ACs, 76 possibly explaining the earlier tissue maturation. Additionally, Ofek et al. used ACs from skeletally immature calves, which are likely to remodel their matrix faster compared with the adult human ACs used by Anderson et al. 30 Taken together, scaffold-free approaches to regenerate articular cartilage produce inferior PCMs in early stages of development but can mature fairly quickly. However, the speed is highly dependent on the cell source and donor.
Intracellular pathways regulating PCM formation
Smad signaling
Signal transduction from growth factors is a complex interplay between intracellular pathways. TGF-β plays a key role in all phases of chondrogenesis, which is used to differentiate precursor cells such as MSCs and chondroprogenitors, stimulate cell proliferation, and accelerate matrix deposition.67,76,77 TGF-β signaling is largely Smad-dependent. 78 Specifically, the Smad2/3 complex is known to stimulate chondrogenesis after differentiation through stimulation of the SRY-box transcription factor 9 (Sox9) and inhibit hypertrophy through the runt-related transcription factor 2 (Runx2). 77 The regulation of these genes requires nuclear translocation of phosphorylated Smad2/3. 78 Phosphorylation is mainly mediated by activin receptor–like kinase-5 (ALK5).79,80
Alternatively, BMPs mainly activate the Smad1/5/8 complex through ALK1, instead of ALK5, stimulating the activity of Runx2. While Runx2 is essential for early chondrogenesis, 80 it stimulates terminal differentiation in mature ACs. 81 The ratio ALK1/ALK5 changes during aging, potentially contributing to the pathological phenotype of ACs in osteoarthritic tissue. 82 Although no research directly links this to PCM synthesis, this decreased sensitivity to endogenous TGF-β of aged ACs may reduce the rates of PCM synthesis by ACs from osteoarthritic tissue.24,30,36
The role of Smad signaling in PCM synthesis remains unclear. While its involvement in ECM synthesis is established, studies investigating the regulation of PCM components are lacking. Since the addition of exogenous TGF-β seems to have little effect on PCM production, 41 it is possible that other mechanisms besides Smad signaling play a key regulatory role during PCM synthesis.
Actin-related mechanotransduction
The actin cytoskeleton is key for chondrogenic differentiation and phenotypic stability. In response to mechanical stimuli such as substrate stiffness, mechanical loading, and dimensionality, it induces changes in cell shape and cellular forces.54,83 Integrins are heavily involved in the transduction of these signals, 84 and deficiencies have been related to pathological cell behavior.85–87 ACs bind to collagens primarily through integrin α2β1, which has downstream effects on cell shape, proliferation, and matrix matelloproteinase (MMP) transcription. 88 Integrin α5β1 is considered essential for cell binding to fibronectin. 89 Integrins can be incorporated with focal adhesions, through which the actin cytoskeleton is connected with the matrix. 90 In native tissue, ACs have a distinct cortical actin distribution, giving the cells a round morphology. 91 Changes to the cell morphology and an increased polymerization of actin are directly associated with a significant decrease in Sox9, type-II collagen, and aggrecan expression and an increase in type-I collagen.54,92,93 On the contrary, decreasing actin polymerization can result in the reestablishment of the chondrogenic phenotype,54,94–96 underscoring the importance of this highly dynamic system. Actin dynamics could hold the key to explaining the differences in PCM synthesis observed in the literature.
Actin polymerization, regulated by RhoA, forms filaments that can bundle into stress fibers, determining cell contractility. 97 RhoA transduces extracellular mechanical stimuli through integrins and is responsive to material stiffness and mechanical stimulation such as fluid flow and compression.98,99 RhoA activity and actin polymerization directly affect Sox9 gene expression, indicating its central role in the expression of the chondrogenic phenotype. 100
Yes-associated protein and transcriptional coactivator with PDZ-binding motif (YAP/TAZ) is a key effector of Rho and is regulated by actin dynamics.101–105 In ACs, YAP/TAZ has strong cross talk with Smad2/3,106–108 intertwining TGF-β-induced signaling with mechanotransduction pathways. YAP/TAZ activity is dependent on its translocation within the cell, influenced by substrate stiffness, cell shape, and culture dimensionality. 104 Studies showed that nuclear YAP/TAZ increases with substrate stiffness in ACs,109,110 chondroprogenitor cells, 105 MSCs,106,111 and other cell types unrelated to chondrogenesis.112–116 While YAP/TAZ activation is important for early chondrogenic differentiation and inhibits terminal differentiation through direct intranuclear interactions with Runx2,117,118 YAP/TAZ activity does hamper the expression of chondrogenic markers, including PCM components, as simultaneous knockdown of both YAP and TAZ significantly upregulates Sox9 and type-VI collagen protein expression. 105
YAP/TAZ has interactions with many downstream proteins and signaling cascades. In the context of chondrogenesis, especially the cross talk with the Smad2/3 complex is interesting. In fibroblasts 107 and embryonic stem cells, 108 YAP/TAZ activity directly controls Smad2/3 localization, but not TGF-β-induced phosphorylation of Smad2/3.
The majority of these results are, however, based on 2D cultures, while dimensionality greatly affects the relationship between substrate stiffness and YAP/TAZ activation. 119 When cultured in 3D, the effect of substrate stiffness on YAP/TAZ localization is inverted compared with that in a 2D environment. 120 Unlike 2D cultures, where stiff substrates cause cell elongation, high-stiffness materials restrict the spreading and contractility of embedded cells. 121 In the context of cartilage tissue engineering, only 3D cultures are considered relevant. Therefore, the implications of studies conducted in 2D should be taken with care.
However, Aprile et al. 111 found that a soft 3D environment allowing cell spreading improved chondrogenesis by MSCs, decreasing YAP expression when cells could spread compared with when they were confined to a round morphology. This is contradictory to the suggestion that YAP/TAZ activity is increased in soft materials, allowing cell elongation. Cui et al. 109 suggest that YAP/TAZ can be phosphorylated at multiple locations on the protein, depending on the use of anabolic or catabolic stimuli on murine chondrocytes. Phosphorylation through catabolic stimuli resulted in cytosolic retention, while anabolic stimuli promoted nuclear translocation and subsequent Sox9 expression. These contradictory findings highlight the need for further study of YAP/TAZ response to biomaterials.
When comparing the possible influence of material stiffness, cell elongation, and YAP/TAZ activity to the experimental conditions used in the reviewed literature, there is an interesting pattern. As stated earlier, high-stiffness hydrogels6,20,21,25,32,39,42,43,45,46 seemed to result in better PCM synthesis compared with low-stiffness materials.29,36 Interestingly, when comparing other material properties besides stiffness, it is observed that cellular attachment to the material could play a role. While the high-stiffness materials, agarose20,25,41,43 and alginate,6,21,32,46 have no cell-binding sites, the low-stiffness fibrin 29 and type-I collagen 36 hydrogels do allow cell attachment. When adding arginylglycylaspartic acid (RGD) sequences to soft agarose hydrogels, chondrocytes start expressing a more dedifferentiated phenotype compared with unfunctionalized soft agarose or stiff agarose with RGD. 55 When considering both the outcomes of the applicational studies and the related intracellular mechanisms, we suggest that PCM synthesis is hampered by soft materials allowing cellular attachment.
Calcium signaling
Joint loading alters tissue osmolarity, directly affecting the phenotype of chondrocytes. Osmolarity is mainly transduced through intracellular calcium (Ca2+) signaling, which is associated with regulating gene and protein expression.122,123 Ca2+ signaling is mediated by transient receptor potential vanilloid 4 (TRPV4) ion channels, which are activated by mechanical stimulation and are key to regulating chondrogenic genes. 123 Activation of TRPV4 happens through changes in surface tension of the membrane as a result of cellular deformation. 124 Additionally, Ca2+ signaling through TRPV4 could be dependent on integrin α1β1, independent of adhesion to the ECM. 125 Modulation through integrins becomes apparent in the study by Zelenski et al., 13 who showed that deletion of type-VI collagen from the PCM influenced mechanotransduction through TRPV4.
Alternatively, Ca2+ signaling can be mediated by Piezo1/2 ion channels. Generally, Piezo channels respond faster to mechanical stimulation than TRPV4. 124 Unlike TRPV4, Piezo channels transduce mainly hyperphysiological forces 126 and may induce cartilage degradation and inflammation. 127 However, the exact functions of Piezo channels and potential overlapping mechanisms with TRPV4 in chondrocytes are still unknown and require further investigation. 128
Downstream, Ca2+ mediates a wide range of cellular functions. Hypo-osmotic stress can initiate actin depolymerization through gelsolin.129,130 Additionally, Ca2+ promotes chondrogenesis by stimulating Sox9 through Ca2+/calmodulin signaling and interactions with the TGF-β pathway.131,132 Although Ca2+-mediated signaling plays a role in cartilage anabolism, its specific role in PCM synthesis remains unclear.
CD44 signaling
CD44 is a transmembrane glycoprotein that plays a crucial role in chondrogenesis. It is the main receptor for HA, and it mediates important cell–matrix interactions.15,16 The interaction between HA and CD44 stimulates the chondrogenic differentiation of MSCs. 133 Besides its role in chondrogenic differentiation, 134 it plays an important role in the assembly of newly synthesized matrix. Multiple studies suggest that interactions between CD44 and HA are key to the retention and structuring of PCM components.64,135,136 These findings agree with the results of the study by Owida et al. 41 who observed accelerated PCM formation when HA was added to agarose hydrogels. This suggests that the presence of HA and its interactions with CD44 could be key to PCM formation during cartilage tissue engineering.
Recommendations and future perspectives
The presence of a PCM can improve the outcomes of articular cartilage tissue engineering. Since the use of isolated chondrons is limited, PCM formation during tissue engineering is a relevant topic. This review examined the capacity of cells to produce PCM during tissue engineering. Key factors influencing PCM production include scaffold material properties, mechanical stimulation, and the availability of growth factors. The transduction of these extracellular cues and their potential downstream effects are summarized in Figure 2.
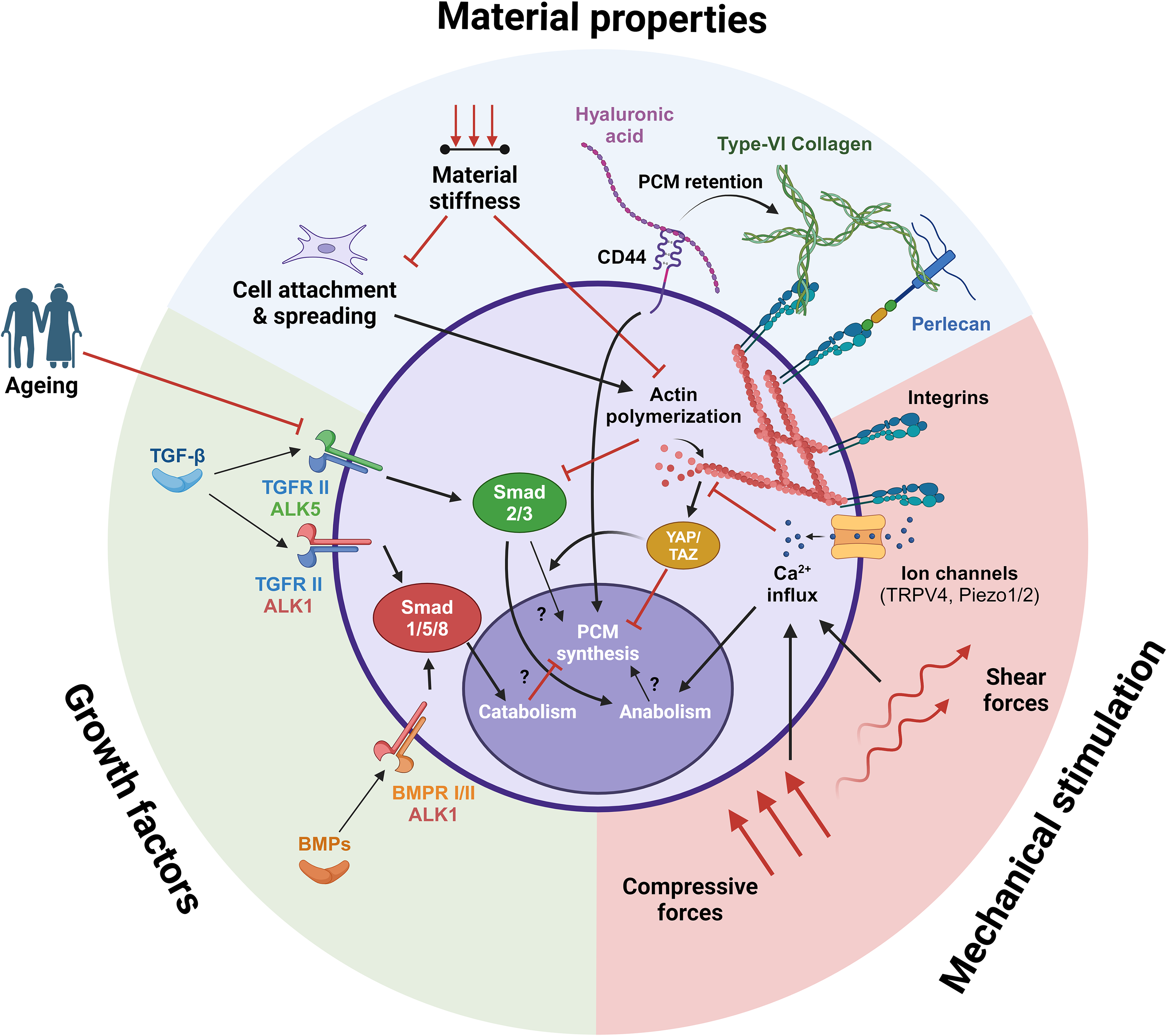
Transduction of material properties, mechanical stimulation, and growth factors in a 3D environment and their potential downstream effects on general chondrogenic anabolism and catabolism and PCM synthesis. Although application-focused research shows that these extracellular stimuli affect PCM synthesis, most intracellular mechanisms are unknown. Created in BioRender. Van Mourik, M. (2024) BioRender.com/z69u812.
Material stiffness plays a crucial role in PCM production. Hydrogels such as agarose and alginate, with equilibrium moduli of 10–30 kPa, have been shown to facilitate PCM production, whereas soft hydrogels (<1 kPa) hinder this process. It should be noted that the soft hydrogels allowed cell binding, while the stiffer hydrogels did not. It is likely that the actin cytoskeleton determines the difference in cellular response, as a round morphology is maintained in the stiff hydrogels, and the soft hydrogels promote actin polymerization and YAP/TAZ signaling. The need for cell-binding sites in stiffer materials is still uncertain. Although cell–matrix interactions are important for transducing mechanical signals, chondrocytes can produce a PCM in 3–7 days, suggesting that binding sites in the material may not be needed as they are produced by the cell on a short term. Nevertheless, adding components such as HA seems to promote PCM production. Fine-tuning these material properties could improve the quality and quantity of matrix production.
Increasing material stiffness is generally accompanied by decreased permeability. While lower permeability seems to be favorable to PCM formation, it also hampers the diffusion of ECM proteins. Early accumulation of matrix can limit diffusion in later stages of culture, which affects the quality of the neocartilage. 137 Additionally, material permeability affects fluid flow and transient Ca2+ signaling, which can influence matrix synthesis. Nevertheless, compact PCMs resembling native chondrons are formed in stiff hydrogels. Altogether, the effects of material permeability on PCM and subsequent ECM production require further investigation.
Mechanical stimulation also affects PCM production. Compressive loading during the early stages of regeneration can inhibit PCM production, but it promotes PCM maturation after the cells are surrounded by a PCM. Partial PCMs lack protective function, 32 possibly explaining this phenomenon and underscoring the importance of the timing of mechanical stimulation.
Growth factors, such as TGF-β, have varying effects on PCM production depending on the chondrogenic differentiation stage of the cells used. Exogenous TGF-β seems to have minimal influence on PCM production by mature ACs but seems important when using progenitor cell types such as MSCs and ACPCs. The effect and necessity of growth factors on PCM synthesis should be considered with care. Additionally, the cell type can have an influence on the quality of the PCM, as the structure and composition of PCMs produced by MSCs and ACPCs can differ from mature ACs. Also, donor age and potential related senescence can affect cell behavior and their receptiveness to extracellular stimuli, potentially making autologous cell sources less effective for older patients. Although the current literature indicates that cell type and source can influence PCM production, the effects on neocartilage formation remain to be elucidated.
This review highlights a gap between tissue engineering applications and fundamental biological research. Much fundamental research on PCM synthesis is performed in a 2D environment, while the dimensionality greatly affects cellular responses. Studying cellular mechanisms in 3D, which is more relevant to tissue engineering, is, however, challenging as the chosen environment will directly affect the process. Additionally, the fundamental mechanisms governing PCM synthesis remain unclear. While some connections between type-VI collagen and perlecan synthesis and YAP/TAZ 105 and Smad2/3 activity53,138 were found, these studies were primarily performed in 2D and/or with nonchondrogenic cell types. No direct link between Sox9, a key transcription factor for chondrogenesis, and PCM synthesis has been established, and one study suggests that type-VI collagen production is regulated posttranscriptionally rather than by gene expression. 44 Together, this shows that combining fundamental and tissue engineering research is essential to accelerate the development of functional cartilage tissue engineering strategies.
To conclude, this review highlights the effect of different tissue engineering strategies on PCM formation. Material stiffness, cell-binding sites, mechanical stimulation, and growth factors can significantly impact PCM production. However, a lot of questions remain to be answered. Notably, numerous cartilage tissue engineering studies do not take PCM synthesis into account, despite the PCM’s recognized importance in cartilage function. The findings of this review highlight the need to consider PCM formation in the design of tissue engineering scaffolds. Additionally, the underlying mechanisms controlling PCM formation remain to be elucidated. Future research should focus on uncovering these mechanisms, using a culture environment relevant to the context of articular cartilage tissue engineering. Incorporating PCM formation in the design of tissue engineering scaffolds and understanding the underlying mechanisms can greatly improve the outcomes of cartilage tissue engineering strategies.
Authors’ Contributions
M.v.M.: Conceptualization, investigation, visualization, writing—original draft, and writing—review and editing. F.A.: Conceptualization and writing—review and editing. K.I.: Conceptualization, funding acquisition, and writing—review and editing.
Footnotes
Funding Information
This research was financially supported by the Gravitation Program “Materials Driven Regeneration,” funded by the Netherlands Organization for Scientific Research (024.003.013).
Disclosure Statement
The authors declare that there is no conflict of interest
