Abstract
Objectives:
The longevity of homografts is determined by the activation of the recipients' immune system resulting from allogenic antigen exposition. Fresh decellularized pulmonary homografts (DPH) have shown promising early results in pulmonary valve replacement in children and young adults and could potentially avoid significant activation of the immune system, as more than 99% of the donor DNA is removed during the decellularization process. While the humoral immune response to decellularized allografts has been studied, detailed information on the more significant cellular immune response is currently lacking.
Methods and Results:
Peripheral blood samples were obtained from patients undergoing pulmonary valve replacement with DPH before, after, and for approximately 3 years after implantation. Absolute counts and percentages of mature T- (CD3+), B- (CD19+), and natural killer- (CD16+/CD56+) cells, as well as T helper- (CD4+) and cytotoxic T-cell- (CD8+) subsets, were determined by fluorescence-activated cell sorting (FACS). Between May 2009 and September 2013, 199 blood samples taken from 47 patients with a mean age at DPH implantation of 16.6±10.8 years were analyzed. The hemodynamic performance of DPH was excellent in all but one patient, and no valve-related deaths or conduit explantations were observed. The short-term follow up revealed a significant postoperative decrease in cell counts of most subtypes with reconstitution after 3 months. Continued assessment did not show any significant deviations in cell counts from their baseline values.
Conclusion:
The absence of cellular immune response in patients receiving DPH supports the concept that decellularization can provide a basis for autologous regeneration.
Introduction
S
Since the implantation of a mechanical valve carries a higher thromboembolic risk and requires lifelong anticoagulation therapy with potentially inherent complications, 3 biological valves of xenogenic origin or human cryopreserved allografts are preferred especially for younger patients or in the pediatric population. 4 However, both types have a limited durability due to calcification, 5 which is mainly driven by immunological-inflammatory processes that are triggered by viable preserved “foreign” donor cells in the xenograft/allograft valvular tissue. 6 Due to the fact that infants and children show stronger immunologic reactions to these grafts, re-interventions are needed at an earlier stage than in adult patients.7,8
Detergent decellularization, a process enabling an almost complete removal of all cells and cellular debris (>99%) without affecting the extracellular matrix (ECM), provides a promising approach. 9 It results in a theoretically less antigenic valvular matrix, which has been shown to be repopulated by viable autologous interstitial and endothelial cells after implantation in vivo, thus displaying potential for enhanced durability.10–14
This approach has been applied to allogenic as well as the more readily available xenogenic valves. Unfortunately, implanted decellularized xenogenic valves degenerated rapidly in a number of pediatric patients, who subsequently suffered from dramatic graft failure. A potential reason for the immunogenicity may lie in the xenogenic matrix itself. Accordingly, explanted grafts showed a severe foreign body reaction that is associated with a severe inflammatory reaction, exaggerated fibrotic scarring, and neo-intima formation.15,16
Recently, we reported early clinical results on the implantation of fresh decellularized pulmonary homografts (DPH) compared with glutaraldehyde-fixed bovine jugular veins and cryopreserved homografts in children and young adults. During the follow-up period of up to 5 years, DPH showed improved freedom from explantation, low transvalvular gradients, and adaptive growth.11,17,18 Since decellularized human valve allografts consist of ECM with a negligible amount of remaining donor cells, they should not trigger an immune response to the allograft. In contrast to several existing studies addressing the question of a humoral immune response to cryopreserved decellularized allografts,19–21 data on the cellular immune response are lacking to date. The cell-mediated immune response, however, is one leading factor for rapid structural implant deterioration.22,23 Therefore, the aim of our study was to evaluate whether the implantation of a fresh DPH elicits a cellular immune response in children and young adults.
Materials and Methods
Study cohort
Sixty-nine pediatric and adult patients have so far received a DPH for pulmonary valve replacement at Hannover Medical School, Germany, and in the State Medical and Pharmaceutical University Hospital, Chisinau, Moldova. Details of the decellularization protocol and the implantation procedure have been provided elsewhere. 11
From this cohort, 47 consecutive patients, operated on between May 2009 and July 2012 at Hannover Medical School, underwent blood sampling in addition to their regular follow-up investigations, which included a clinical examination, echocardiography, and cardiac magnetic resonance imaging.
Peripheral blood samples were taken before and at hospital discharge after pulmonary valve replacement, at the 3 and 6 months follow up, and, subsequently, every 6–12 months. All patients or parents provided written informed consent, and the study was approved by the local institutional ethics committee (No. 528/2009).
Cellular immune response
The absolute numbers of leukocytes and lymphocytes, as well as the absolute numbers and percentages of T- (CD3+), T helper- (CD4+), cytotoxic T- (CD8+), B- (CD19+), and natural killer (NK)- cells (CD16+CD56+) were determined by multi-color flow cytometry using a fluorescence-activated cell sorting system (FACS Canto II; BD Biosciences, San Jose, CA). According to the manufacturer's instructions, fresh whole blood samples were stained with monoclonal antibodies (mAbs) that were directly conjugated with different fluorochromes (BD Multitest TBNK reagent; BD Biosciences Pharmingen, San Jose, CA). The following anti-human mAbs were used: FITC-labeled CD3, PE-labeled CD16 and CD56, PerCp-Cy 5.5-labeled CD45, PE-Cy7-labeled CD4, APC-labeled CD19, and APC-Cy7-labeled CD8.
Statistical analyses
An intraindividual assessment for changes in the number of total white blood cells, lymphocytes, as well as the number and percentage of lymphocyte subsets was performed using a paired t-test. Two sided p-values of<0.05 were considered statistically significant. Potential time-dependent effects were evaluated by analyzing measurements across the whole study cohort at every given time point. Furthermore, a linear regression model was fitted to evaluate potential links between valvular gradients and cell numbers.
Statistical analyses were performed using R-Project (software version 2.10.1 for Linux). DPH performance is shown as a fraction of degenerated conduits below the Kaplan–Meier curve for freedom from explantation (IBM SPSS Statistics 20, Ehningen, Germany).
Results
Patients
To date, 69 pediatric and adult patients received fresh, non-cell seeded decellularized homografts for pulmonary valve replacement according to the Hannover protocol, starting in 2005. From this cohort, 47 consecutive patients with a mean age of 16.6±10.8 years at implantation were analyzed for cellular immune response. In total, 199 blood samples were analyzed between May 2009 and September 2013.
For this group, the mean follow-up period was 2.5±1.0 years and the total cumulative observation time was 81.75 years (981 months). Most of the patients (n=28, 60%) had undergone a previous heart operation, while 17 patients (36%) had undergone two or more surgical interventions. Ten patients (21%) had pulmonary valve replacement using a cryopreserved homograft before DPH. Twenty-seven patients (57%) received a DPH with a diameter between 24 and 30 mm. No patient received immunosuppressive medication.
For 22 patients, no immunological data were available, as they received a DPH before the start of the immunological monitoring in 2009 or because parental consent was not given. Detailed information on the study cohort, including underlying cardiac malformation, the type and number of previous procedures, and implanted homograft diameters, is given in Table 1.
DORV, double-outlet right ventricle; PA/IVS, pulmonary atresia with intact ventricular septum; PA/VSD, pulmonary atresia with ventricular septal defect; PI/PS, pulmonary stenosis and/or insufficiency; TAC, truncus arteriosus communis; TGA, transposition of the great arteries; TOF, tetralogy of Fallot; and RVOT, right ventricular outflow tract; BT, Blalock-Taussig.
Clinical course of DPH
During a total observation time of more than 224 years, no patient developed a neoplasma, neither the incidence of viral or bacterial infections was increased.
At discharge, echocardiography showed low-pressure transvalvular gradients in all patients.
After 3 years of follow up, freedom from any signs of valve degeneration (defined as peak gradient >49 mmHg over any part of the conduit and/or at least moderate valve insufficiency) was 92% for the study cohort under immunological monitoring.
During the 8-year follow up of all patients, no DPH valve had to be explanted. There were no valve-related deaths. Two patients died during the follow up due to non-valve-related reasons: one in Moldava as previously reported 11 and one 6-week-old infant who died in Hannover 9 weeks postoperatively due to fulminant sepsis in conjunction with persistent severe chylothorax. Figure 1 gives detailed information on the hemodynamic performance of DPH valves in patients both with and without immunological monitoring at different time points of follow up.
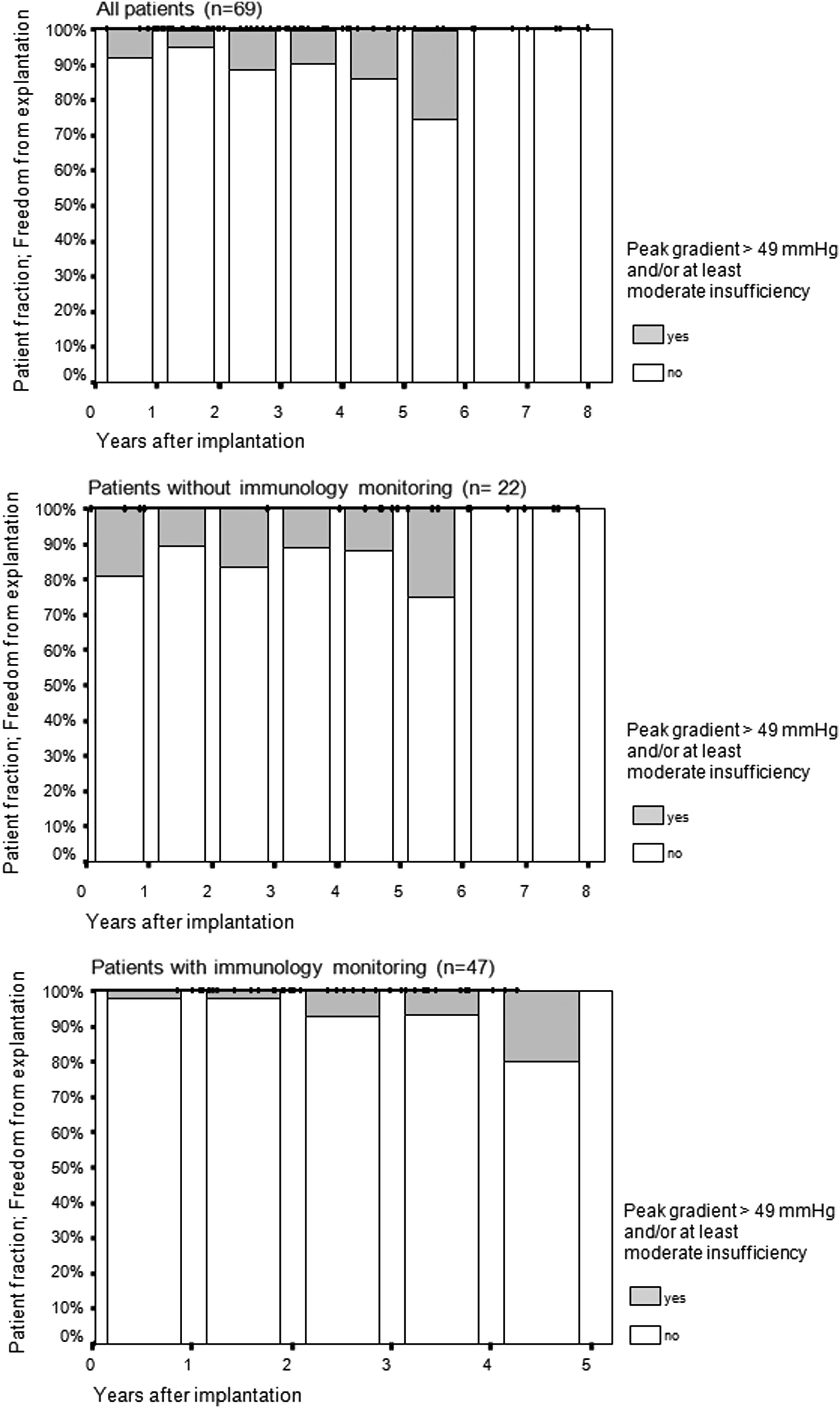
Freedom from explantation and hemodynamic performance of fresh decellularized pulmonary homografts (DPH). The y-axis shows the percentage of patients free from conduit explantation. Small black bars at the top of the respective columns represent each patient examined at the latest follow-up year indicated at the x-axis. The top graphic shows the hemodynamic data of all 69 patients who received a DPH from 2005 to 2012, the central graphic outlines the results of the 22 patients without immunological data, and the bottom graphic indicates the results of the 47 patients with immunological monitoring. Gray areas within the respective year column indicate impaired valve function defined by a transvalvular peak gradient >49 mmHg and/or at least moderate insufficiency.
One patient developed moderate non-calcific conduit degeneration with a maximum valvular gradient of 54 mmHg (mean gradient 27 mmHg) at 18 months after DPH implantation. He was successfully treated by balloon dilation, which reduced the peak gradient to 40 mmHg (mean 23 mmHg). During a further 24 months of follow up, the gradient remained stable and no significant regurgitation was observed.
Cellular immune response to DPH valves
The absolute numbers of white blood cells and lymphocytes, as well as the absolute numbers and percentages of T- (CD3+), T helper- (CD4+), cytotoxic T- (CD8+), B- (CD19+), and NK-cells (CD16+CD56+) determined by FACS analysis, are shown in Figures 2–4. The mean and median values of the pooled data of all patients are displayed to illustrate cell count variability and to facilitate a comparison.
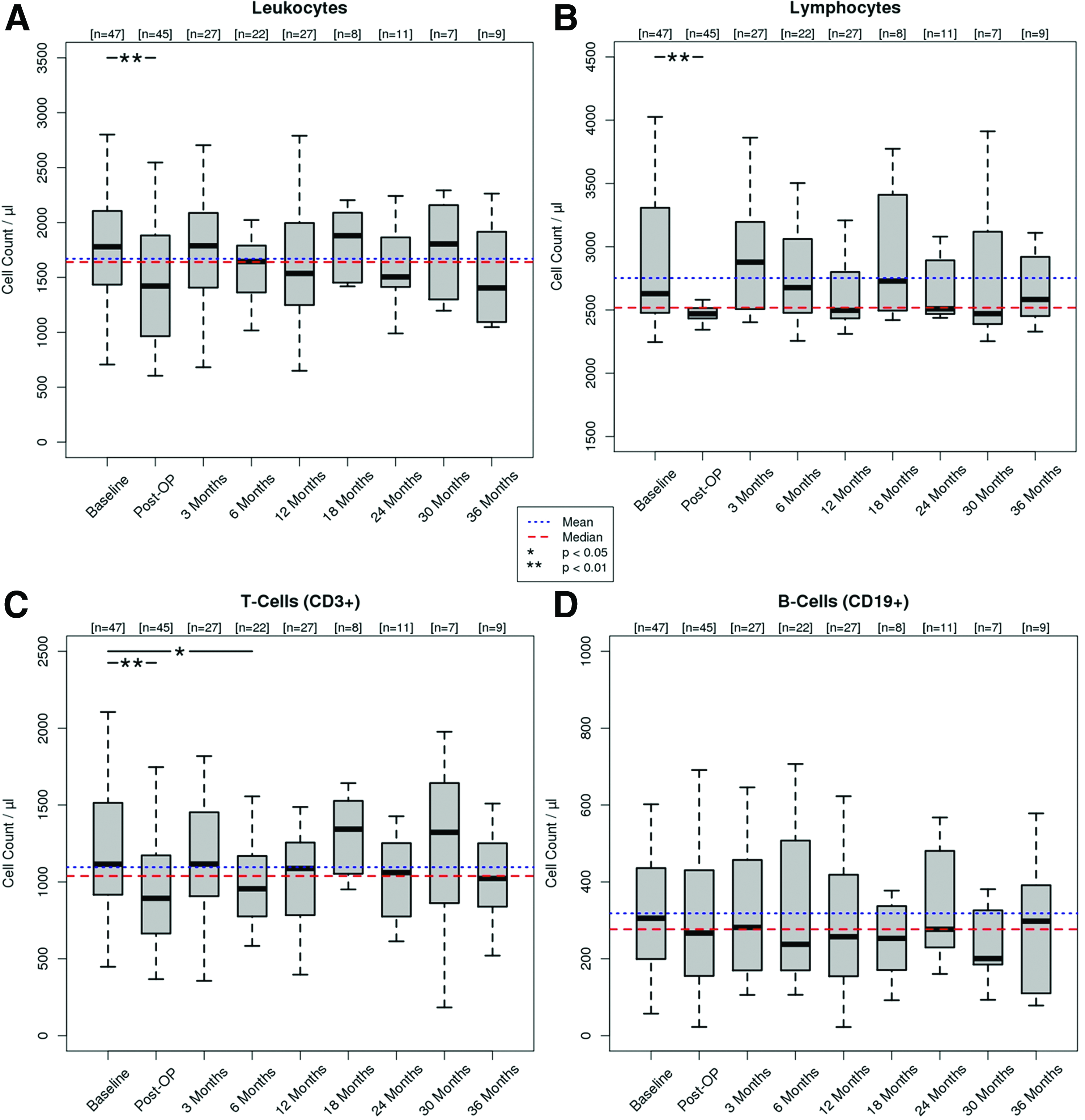
Systemic cellular immune response during follow up—Part I. Absolute counts of leukocytes
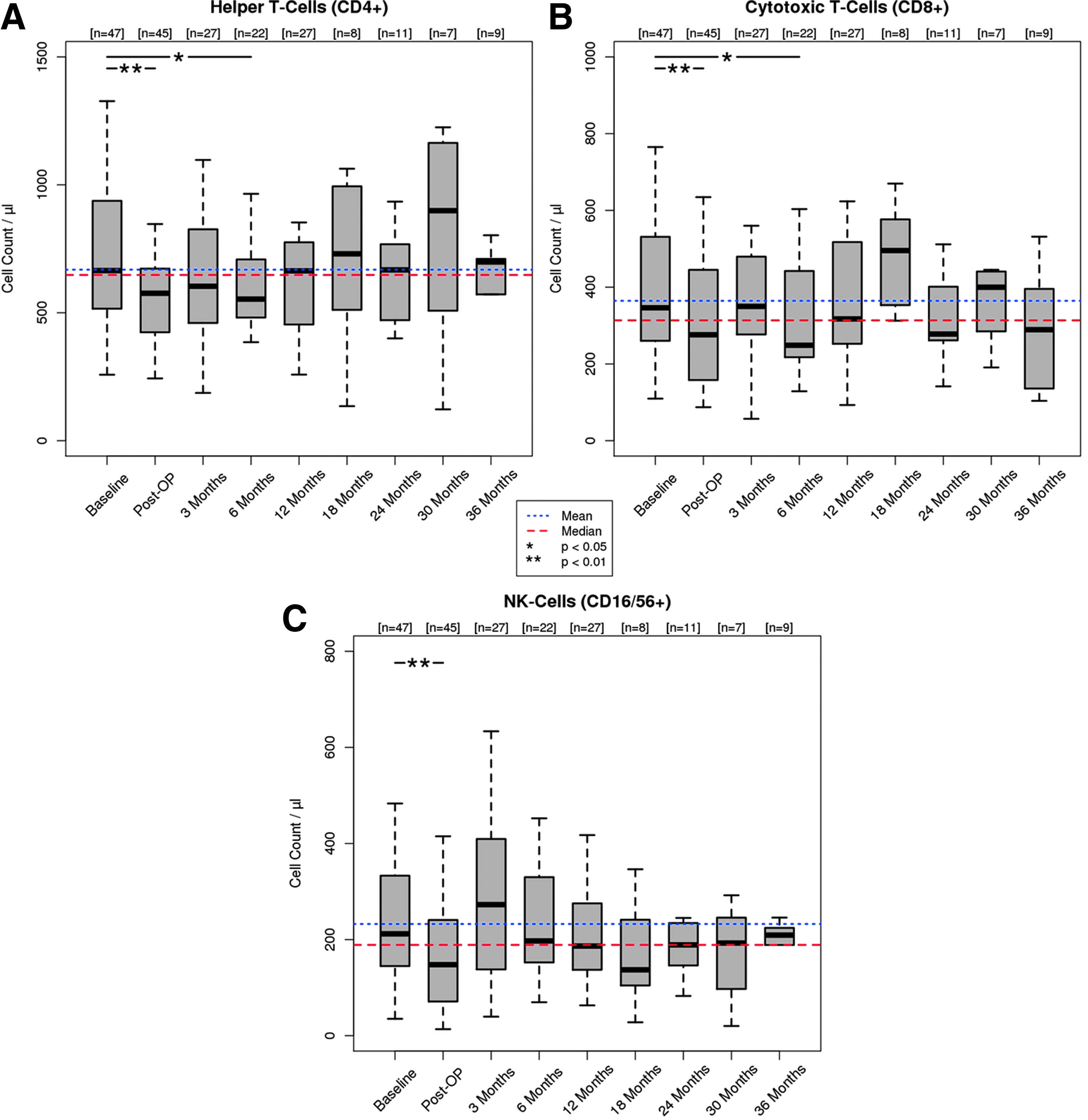
Systemic cellular immune response during follow up—Part II. Absolute counts of CD4+ Helper T-cells
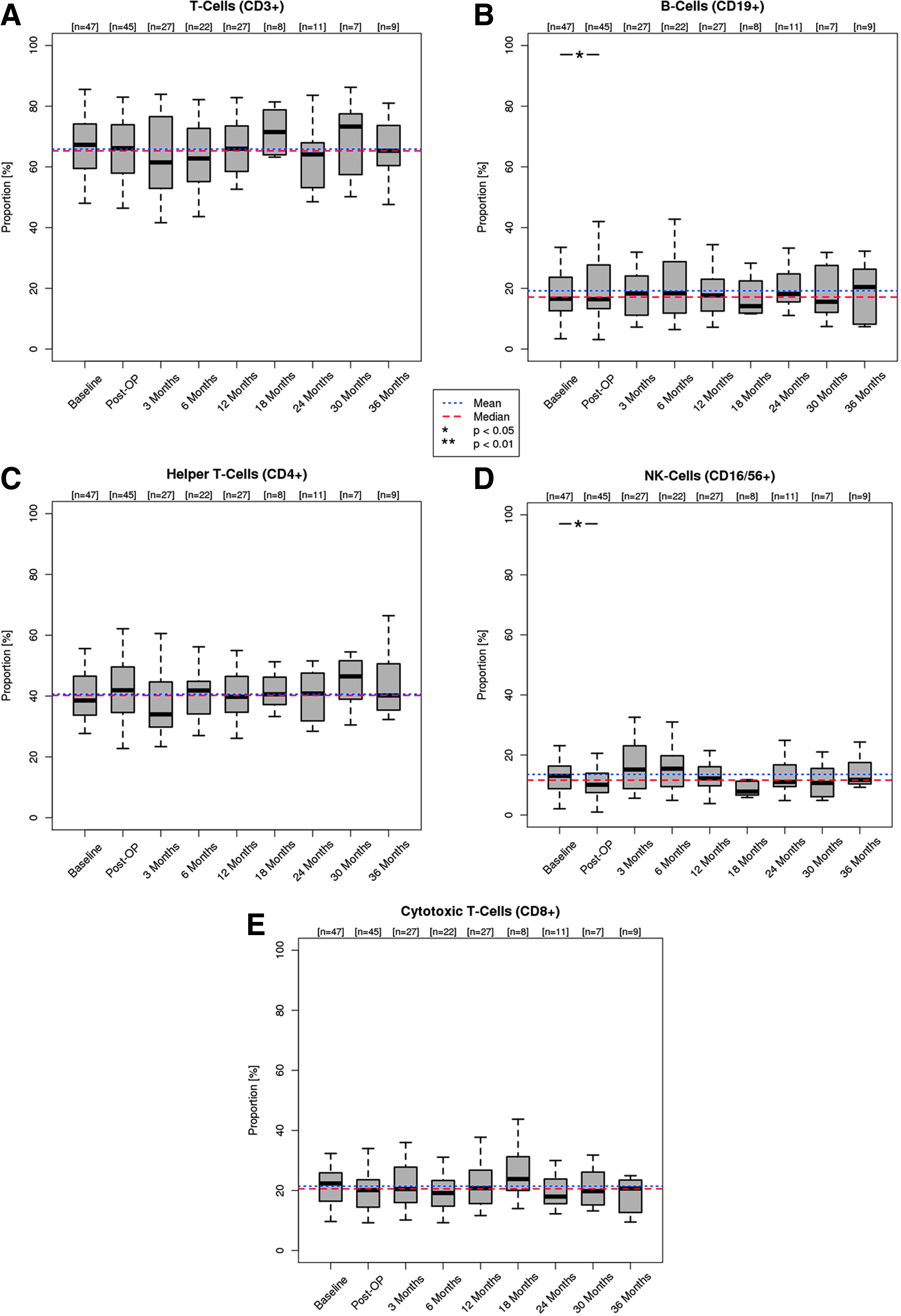
Systemic cellular immune response during follow up—Part III. Relative counts of CD3+ T-cells
Immediately after valve implantation, the numbers of white blood cells (p<0.01), lymphocytes (p<0.001), and lymphocyte subsets such as T- (p<0.01), T-helper (p<0.01), T- cytotoxic (p<0.01) and NK-cells (p<0.01) decreased significantly, while the number of B-cells (p=0.19) remained stable. Three months after DPH implantation, an inverse effect was observed and cell numbers normalized for all cell subsets. At 6 months, a temporary, small, nevertheless statistically significant (p<0.05) decrease of CD3+/CD4+/CD8+ T cells was seen with normal values for NK-cells and B-cells. Throughout the further study period of up to 36 months, all cell subsets showed no statistically significant alterations. Relative cell counts showed even less alteration within the study period (Fig. 4).
Linear regression models showed no correlation between valvular gradients and cell subsets. However, one patient with a moderate valvular stenosis displayed lymphocytosis and increased NK-cell counts. For this patient, a B- or T-cell proliferative disorder was excluded by analysis of kappa- and lambda-light chains as well as the T-cell receptor rearrangement by polymerase-chain reaction.
Discussion
Cryopreservation is the current standard preservation method for allografts.24,25 However, in recent years, there has been a growing body of evidence that cryopreservation may cause serious damage to the endothelial cells, resulting in reduced cell viability. 26 Moreover, it seems that the process of cryopreservation can cause serious damage to the ECM structures of the valve leaflets, which may be linked to delayed graft failure.27,28
The process of cryopreservation itself is complex and difficult to control, as there are several influencing factors, for example, harvesting (warm ischemia), sterilization with antibiotics, freezing which causes fluid shifts and extensive ice crystal formation that damage the ECM,29,30 extended storage times, and variability during thawing. In contrast to earlier reports, 31 cryopreservation does not seem to prevent the activation of the immune system. It is associated with an early infiltration of mononuclear cells and allo-reactive T-cells into valve leaflets. 32 These chronic inflammatory processes predispose cryopreserved valvular allografts to calcification with structural deterioration and dysfunction, resulting in increased re-intervention propensity. With the use of a fresh decellularized human allograft valve, the difficulties discussed earlier as being associated with the process of cryopreservation may be overcome.
Some studies have shown alterations of the mechanical behavior of decellularized pulmonary or aortic valve leaflets, especially an increase in leaflet extensibility and areal strain, reduced stiffness, and decreased flexural modulus.33,34 In contrast to other decellularization protocols being tested, the extracellular matrix (ECM) of sodium dodecyl sulfate-treated leaflets resembled that of a native valve with complete maintenance of the basement membrane.9,33 Of note, allografts used in these in vitro studies were cryopreserved before undergoing the decellularization procedure. Hemodynamic behavior of fresh decellularized, non-cryopreserved DPH in vivo was favorable in 8 years of follow up within this study, suggesting sufficient mechanical stability.
Using decellularization protocols, which allow an almost complete removal of cells and cellular debris from the graft without affecting the ECM, graft immunogenicity should be substantially reduced. Consequently, autologous cells should be able to reseed the graft and stimulate matrix remodeling. 35
A weakness of our study is the length of follow up, which has so far been limited to 36 months of implantation and this restricted statistical analysis for cell count alterations in the few patients with a longer follow up. Statistical analysis was furthermore hindered by the limited availability of reference data for cell subsets in children and the well-known variability in peripheral cell counts. We compensated for this by analyzing effects across the whole study cohort at every given time point pre- and postoperatively and compared all data for variance with mean and median cell counts of the entire study population.
A temporary decrease in cell numbers shortly after implantation of DPH appears to be a consequence of cardio-pulmonary bypass and has been described in this setting. 36 The initial decrease was followed by normalization to baseline values within 3 months. At 6 months, a temporary decrease of CD3+/CD4+/CD8+ T cells was seen with normal values for NK-cells and B-cells. Throughout the further study period, all cell subsets showed no statistical significant alterations, which stands in contrast to the results of Wojtalik et al. 37 In 19 children who received glutaraldehyde fixed xenogeneic valves (Contegra®) for pulmonary valve replacement, they found a significant rise of B-cell percentages between the 3rd and 6th month postoperatively and higher numbers of activated T-cells at 1 year after implantation.
The results obtained in this study support the concept that decellularization can provide a basis for autologous regeneration, as no significant activation of the cellular immune system was observed during short-term follow up, that is, during the period of autologous colonization of the valves. Since early graft failure is predominately a cell-mediated process, the observed lack of early systemic cellular immune response is an important and encouraging finding. This is further augmented by the excellent clinical performance of DPH, which in our view is clearly superior to any available heart valve prosthesis for pulmonary valve replacement in children.
One patient with a half-Asian ethnic background showed signs of conduit degeneration by developing moderate valvular graft stenosis with a peak gradient of 54 mmHg (mean 27 mmHg) within 18 months and an elevated number of T-, B-, and NK-cell subsets. Although the decellularization procedure has been proved to eliminate ≥99% of donor cells, in rare cases, sporadic cells might survive this process; for example, endothelial cells which carry HLA class I and II molecules that are capable of triggering an immune response.38–40 Hawkins et al. observed a transient antibody response to HLA-DR in a few patients who had received the Synergraft® valve, a decellularized cryopreserved allograft. They hypothesised that these antibodies were directed against cells that are highly immunogenic, such as HLA class II-expressing dendritic cells which may be more resistant to the decellularization process. 19
One may further speculate whether the conduit degeneration in this case may be related to ethnic background as the patient received a DPH of Caucasian origin. It has been shown that there are interethnic cellular immune system differences, for example, in B-cell and NK-cell responses, which may also apply to allogenic (collagen) matrices.41,42 Unfortunately, no baseline NK cell count was available in this patient. Regression analysis did not show a relationship between gradient course and cell subset counts, thereby rendering a correlation unlikely in this case.
Limitations
Our analysis was limited to the amount of cell subsets, for example, regulatory cells were not studied and did not include a further functional analysis of cell sub-cohorts. However, we observed no clinical events, suggesting a regular function of the immune system in our patients.
Our results may not be extrapolated to immune reactions to decellularized aortic allografts, as these allografts are significantly thicker, which may interfere with the decellularization protocols and antigenicity of the ECM. The time to recellularization is expected to be longer in aortic grafts, which may also influence cellular immune reactions.
The observed results in decelluarized fresh allografts may also not be extrapolated to decellularized and, subsequently, cryopreserved allografts, as cryopreservation may interfere with the ECM, thus causing a separate immune response to these valves. 43
Conclusions
In our young study cohort of recipients of decellularized fresh pulmonary allografts in the orthotopic position, no significant early activation of the cellular immune response was detected, which corresponded with the excellent mid-term clinical and hemodynamic outcomes observed.
Footnotes
Disclosure Statement
The authors declare that there are no conflicts of interest. A. Haverich holds shares in corlife oHG, a company for the future processing of decellularized allografts, equivalent to those used in this study.
