Abstract
Moderate and severe traumatic brain injury (TBI) can lead to harmful consequences for trauma victims. S100B is a widely studied biomarker to predict the outcomes of this injury, but gaps in knowledge about this protein make its clinical application still uncertain. We investigated the potential prognostic value of S100B for functional capacity and vital status up to 12 months after moderate and severe TBI and compared its performance when samples of this biomarker are collected in the early (≤6 h) and late (>6 h) phases following the injury. The systematic review followed the recommendations of Cochrane Methods Prognosis and Preferred Reporting Items for Systematic Reviews and Meta-Analyses. The protocol was registered with The International Prospective Register of Systematic Reviews (PROSPERO). The searches were carried out in the Embase, Psycinfo, Medline, and Scopus databases. The meta-analysis was carried out in R® version 4.2.3. Thirty-seven studies were included. The majority indicated higher concentrations of S100B in patients with unfavorable outcomes. In the meta-analysis, S100B showed good discriminative capacity for functional capacity at 3 and 6 months (pooled area under the curve [AUC]: 0.803 and 0.841), but unsatisfactory at 12 months (pooled AUC: 0.592) and for vital status, the pooled AUC was 0.818 for hospital discharge and 6 months, with worse results for 3 and 12 months (AUC: 0.687 and 0.731, respectively). Late samples showed better prognostic performance for functional capacity and vital status. The review indicated that S100B is a promising tool for estimating prognosis up to 6 months after moderate and severe TBI, especially when late samples are analyzed.
Introduction
Traumatic brain injury (TBI) is a form of acquired injury caused by an external mechanical force that can lead to damage to structures of the skull, scalp, meninges, and brain and can permanently or temporarily compromise an individual’s cognitive, physical, and psychosocial functions. 1 Severe and moderate TBI represent, respectively, 12–15.6% and 2–4% of cases of victims; however, it is known that 30–60% of critically ill patients die, and mortality rates for moderate TBI range from 14% to 15%.2,3
The total damage resulting from TBI is caused by the primary, secondary, and late injuries. Secondary injuries are treatable and preventable when diagnosed early. Strategies contributing to the identification of injuries after trauma are important, as failure to detect them early can contribute to unfavorable outcomes, such as death or severe disability.1,4–7
These negative consequences, observed more specifically in victims of severe and moderate TBI, make knowledge of these patients’ prognosis of special interest, as this information helps in early decisions regarding more invasive and immediate interventions, and highlights victims who do not present the expected progression.8–10
In recent decades, biomarkers found in biofluids following brain tissue damage have been considered promising tools to provide objective diagnostic measures and prediction of TBI outcomes.11–14 Among the biomarkers related to brain injury, S100 calcium-binding protein (S100B) is the most investigated to date in the context of TBI in different severities. Studies regarding the use of S100B began with the collection of cerebrospinal fluid (CSF) through ventriculostomy. However, as these biomarkers can permeate the blood–brain barrier (BBB), it has been analyzed in less invasive biological matrices, such as blood,15–17 and more recently, we have some research with saliva.18–23
S100B is synthesized by astrocytes and Schwann cells, melanocytes, adipocytes, and chondrocytes and the main functions of this protein are the control of phosphorylation and protein degradation, the stimulation of cell growth and differentiation processes, and the control of calcium levels. Extracellularly, it promotes neurogenesis and neuronal plasticity.24–27
S100B has been identified as a reliable biomarker for TBI victims, as increased concentration is directly related to brain damage.24–27 Studies evaluating the kinetic profile of this biomarker demonstrate that the release of S100B occurs soon after the injury, the peak concentration is in the early phase of the trauma (up to the 6th h) and, progressively, there is a reduction in concentration in subsequent assessments (samples after 6 h). Interestingly, studies have shown that high levels of S100B assessed in the early phase (up to the 6th h) and in late samples (collected after the 6th h) are associated with unfavorable outcomes (death and impaired functional capacity) in TBI victims. Despite these findings, the literature does not show clearly at which point in the evaluation of S100B the biofluid presents the best prognostic capacity in relation to the outcomes of functional capacity and vital status outcomes.
A recent systematic review with meta-analysis was published in 2024. 28 In this study, the diagnostic and prognostic capabilities of S100B were evaluated in patients with mild, moderate, and severe TBI. It is notorious that the diagnostic capability of this biomarker is relevant, particularly in cases of mild TBI due to the limitations of cranial computerized tomography and Glasgow Coma Scale scoring.29–31 Nevertheless the evaluation of the prognostic capacity of biomarkers is extremely important in cases of moderate and severe TBI, as dependence and mortality are more frequent in this population.32,33 Furthermore, it is known that S100B concentration is influenced by the time elapsed since the traumatic event and is associated with the severity of TBI, potentially affected by secondary injuries, which are especially present in moderate and severe TBI cases.34,35 In studies that assessed sequential samples, different concentrations and performances were observed.11,36–39 Thus, it is equally important to evaluate the prognostic capacity of S100B according to the timing of sample collection (early and late samples) in patients with moderate and severe TBI, as addressed in the present study.
Given the above, the objectives of this review were: (1) to compare the S100B concentrations of victims who presented favorable and unfavorable progression after moderate and severe blunt TBI; (2) to evaluate the prognostic capacity of S100B in relation to functional capacity and/or vital status measured until hospital discharge and/or at 3, 6, and 12 months after moderate and severe TBI; and (3) to compare the prognostic capacity of S100B samples collected in the early phase (≤6 h after the traumatic event) and in the late phase (>6 h after the traumatic event) in relation to functional capacity and/or vital status measured until hospital discharge and/or at 3, 6, and 12 months after moderate and severe TBI.
Method
Design of study
A systematic prognosis review is reported in accordance with Cochrane Methods Prognosis 202040,41 and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. 42 The protocol for this review was registered with PROSPERO (The International Prospective Register of Systematic Reviews); ID: CRD42021293391.
Selection criteria
To define the inclusion criteria, the Critical Appraisal and Data Extraction for Systematic Reviews of Prediction Modeling Studies was followed: The CHARMS Checklist. 43
The inclusion criteria were defined based on the PICOTS element: Population—adults, victims of moderate and severe TBI, regardless of the criteria used for classification, intervention (model/factor)-S100B protein assessed by blood samples collected during hospital stay, Comparator—not applicable, Outcomes—functional capacity and vital status, using Glasgow Outcome Scale (GOS) and GOS extended (GOSE). Timing-until hospital discharge and/or at 3, 6, and 12 months after the trauma, and Study design—prospective and retrospective observational studies (cohort and cross-sectional).
Therefore, articles that evaluated only mild TBI and/or injuries sustained in sports practice, studies that presented the results of mild TBI together with those of moderate or severe TBI, studies conducted on pediatric, neonatal, or geriatric samples, other populations not involving TBI, postmortem studies, studies conducted on animal models, studies evaluating other subunits of S100 other than the beta subunit, studies where concentration was assessed only in CSF, studies where S100B concentration was evaluated after the application of interventions, investigations focused on different techniques of biological sample preparation and collection, other study categories (literature review, event abstracts, letters to the editor, expert comments, book chapters, and clinical case discussions), and studies not published in Portuguese, Spanish, or English were excluded.
Search and data extraction strategy
The searches were performed in the electronic databases: Excerpta Medica Database (Embase), Psycinfo (produced by the American Psychological Association), Retrieval System Online (Medline) via United States National Library of Medicine (Pubmed), Scopus, and Virtual Health Library, since their inception until December of 2023. The details of the established search strategies are described in Table 1.
Databases, Search Terms, Filters, and Results
Two reviewers (L.C.F. and D.A.S.) independently extracted all relevant data and in case of disagreement, a third author was consulted. The information sought was related to author and year, type of study, sample size and scope, criteria for establishing the severity of the TBI, inclusion criteria, exclusion criteria, biomarkers analyzed (matrix used), time after the trauma of the samples collected, outcomes, and functional capacity assessment instruments used. The findings of this search were organized in the Rayyan software. 44
Assessment of risk of bias
To assess the risk of bias, the quality in prognosis studies (QUIPS) tool was used. 45 This instrument contains the following domains: study participation, study attrition, prognostic factor measurement, outcome measurement, study confounding, and statistical analysis and reporting; it also classifies investigations into low risk of bias, medium risk of bias, or high risk of bias according to established criteria. To evaluate the overall risk of bias, the domain with the highest risk of bias was observed.
Independently, two authors evaluated all selected studies in the domains indicated in the tool, and when there was disagreement, a third member was consulted. Prior to this procedure, the evaluators were trained to use QUIPS, and a pilot with the evaluation of six studies was carried out, to calibrate the application of the evaluation parameters.
Statistical analysis–meta-analysis
The software used for meta-analysis and the graphs was R® version 4.3.1 and the package was Metafor version 4.4. This analysis was conducted when two or more studies presented area under the curve (AUC) and standard error or confidence interval. When the study only provided the confidence interval, the formula proposed by Cochrane was applied to calculate the standard error. 24
First, meta-analysis was conducted to evaluate the prognostic ability of S100B in relation to outcomes. For this analysis, all AUC and standard error were entered, regardless of the S100B collection time. Subsequently, to compare the prognostic performance of early and late samples, meta-analyses were conducted, where, separately, the AUC and standard error for samples collected early (≤6 h) and later (>6 h) were entered. It is emphasized that only blood samples were considered for the meta-analysis.
The random effects model was used in meta-analyses. The heterogeneity (I2) of the studies was evaluated with Cochran (Q test) and high heterogeneity was defined when p < 0.10 or I2 > 75%. For all analyses, a significance level of 5% was adopted.
Results
Inclusion of studies
Of the 701 records identified in the databases consulted, duplicate studies (n = 359) were removed, with 342 studies being left for title and abstract analysis and, of these, 293 did not meet the eligibility criteria. Of the 49 studies evaluated in full, 37 met the purposes of this review. Figure 1 presents the process of identification, selection, and assessment/inclusion of studies.

PRISMA flow diagram. PRISMA, preferred reporting items for systematic reviews and meta-analyses.
General characteristics of the studies
The data from Table 2 show that articles on the topic of this review were published more frequently in 201316,36,61,62 and 2016.11,57–59 The first study that analyzed S100B in patients with moderate/severe TBI and its TBI outcomes of interest was conducted in 2001. 74 Just one study 39 was retrospective. All investigations were carried out in a single institution, the mean number of participants in the studies was 69.43 (standard deviation = 50.71), the median was 60, the minimum was 10, and the maximum was 305.
Description of Studies according to Author, Publication Year, Design Of Study, Sample Size/Scope, Criteria for Establishing the Severity of Traumatic Brain Injury, Inclusion Criteria, Exclusion Criteria, Biomarkers (Biofluid and Matrix), Time after Trauma for Sample Collection, Outcome and Assessment Instruments and Results
Database: SCOPUS, Pubmed, Psycinfo, and Embase.
Biomarkers: AB, peptide beta-amyloid; BDNF, brain-derived neurotrophic factor; GFAP, glial fibrillary acidic protein; HFABP, heart-type fatty acid binding protein; ICAM-5, intercellular adhesion molecule; IL, interleukin; MBP, myelin basic protein; MCP-1, monocyte chemoattractant protein-1; MMP, metalloproteinases; NFL, neurofilament light polypeptide; PNF-H, neurofilament subunit H; UCH-L1, ubiquitin carboxy-terminal hydrolase L1; SICAM, soluble intercellular adhesion molecule; SNE, serum neuron-specific enolase; S100B, S100 calcium-binding protein B; T-Tau, total tau-protein; TNF-α, tumor necrosis factor-alpha. Units: L, liter; mL, milliliter; mg, milligram; mmHg, millimeter of mercury; ng, nanogram 10–6 g; pg, picogram 10–9 g; μg, microgram 10–3 g. Scales: AIS, Abbreviated Injury Scale; DRS, Disability Rating Scale; FIM, Functional Independence Measure; GCS, Glasgow Coma Scale; GOS, Glasgow Outcome Scale; GOSE, Glasgow Outcome Scale Extended; 3MS, The Modified Mini-Mental State. Others: AUC, area under the curve; CI, confidence interval; CSF, cerebrospinal fluid; CT, computerized tomography; ICU, intensive care unit; IQR, interquartile range; MV, mechanical ventilation; NS, non-specification; OR, odds ratio; OTI, orotracheal intubation; ROC, receiver operating characteristic; SD, standard deviation; TBI, traumatic brain injury.
The inclusion only of victims of severe TBI was more common in investigations.9,11,16,36–39,47–49,51–56,58–69,71–74 Fifteen studies16,36–38,47,48,51,54,57,61,66,68,69,72,73 analyzed only the S100B protein; the others included two or more biomarkers. Blood was investigated in all studies of this review; some studies jointly analyzed CSF16,56–58,74 and one examined the urine. 38 No studies of S100B in the saliva of victims of moderate and severe TBI were found. Collections were isolated only in 10 studies47,49,50,52,53,56,66,70,71,73 and, in the others, they were sequential.
When evaluating the predictive capacity, 22 studies presented the AUC value9,11,16,33,36–39,48,49,52–54,57,60,62,63,65,66,69,71,72; however, only 12 studies included the standard error or confidence interval.9,11,33,36–39,52,53,60,63,65
Bias assessment
In the evaluation of the 37 studies using QUIPS (Table 3), it was found that 30 studies presented a high risk of bias,9,11,16,33,38,39,47–52,54–67,69,70,72,74 six presented a moderate risk,37,46,53,68,71,73 and only one was categorized as low. 36 The domains with the highest frequency of high risk of bias were study attrition and study confounding and those with the highest frequency of low risk were the outcome measurement and prognostic measurement domains.
Results of the Risk of Bias Evaluated by the Quality in Prognosis Studies Tool (Quality in Prognostic Studies)
S100B and functional capacity
Twenty-seven studies analyzing outcomes after TBI were found. They included functional capacity,9,16,33,37,46–48,50,51,53–60,62–67,70,71,73,74 and the majority (13) included assessments over a 6-month period (Table 2).
Regarding the instruments applied to assess functional capacity, all studies used versions of the GOS, 21 studies used the original GOS16,37,48,50,51,53–60,63,65–67,70,71,73,74 and six the GOSE.9,33,46,47,62,64 In addition to the GOS measurements, two studies used the Functional Independence Measure,48,59 one used the Modified Mini-Mental State Examination (3MS), 48 and two used the Disability Rating Scale.16,58
In the analyses of functional capacity, the GOS results were dichotomized into favorable (good recovery; moderate disability) and unfavorable (severe disability; vegetative state, and death). Similarly, the GOSE categories were pooled into favorable (good recovery lower; good recovery upper; moderate disability lower; moderate disability upper) and unfavorable (severe disability lower; severe disability upper; vegetative state and death).
Comparison of S100B concentration in dependent and independent patients after moderate and severe blunt TBI
The studies33,59 assessing functional capacity on the fourth day of hospitalization or at hospital discharge found higher concentrations among dependent patients. In relation to the functional capacity assessed at 3 months, only one 65 in a total of seven studies9,50,55,56,65,66,74 did not show this difference.
In the 13 studies that included assessment at 6 months,16,33,48,50,53,54,56–60,62,71 7 investigations carried out comparison tests between S100B concentration values within dependent and independent individuals16,48,50,56,59,60,71 and 6 studies16,47,48,50,56,71 showed a greater concentration among dependents.
In the 12-month assessments of functional capacity, higher values in the concentration of S100B in dependent patients when compared with independent ones were observed in four studies; however, in three other studies,58,59,65 this difference was not observed.
Regardless of the time elapsed after TBI for the functional capacity to be assessed, most investigations9,16,33,37,47,48,50,55,56,60,63,64,66,67,71 showed that dependent individuals had higher values of the S100B protein.
Prognostic capacity of S100B in relation to functional capacity at 3, 6, and 12 months after moderate and severe TBI
In studies analyzing functional capacity at 3 months, the AUC ranged from 0.585 to 0.9419,55,65,66 (Table 4), three of these studies55,65,66 were included in the meta-analysis; the pooled AUC was 0.803 (95% confidence interval [CI] 0.700–0.906); I2 = 73% (p < 0.01) (Fig. 2a).
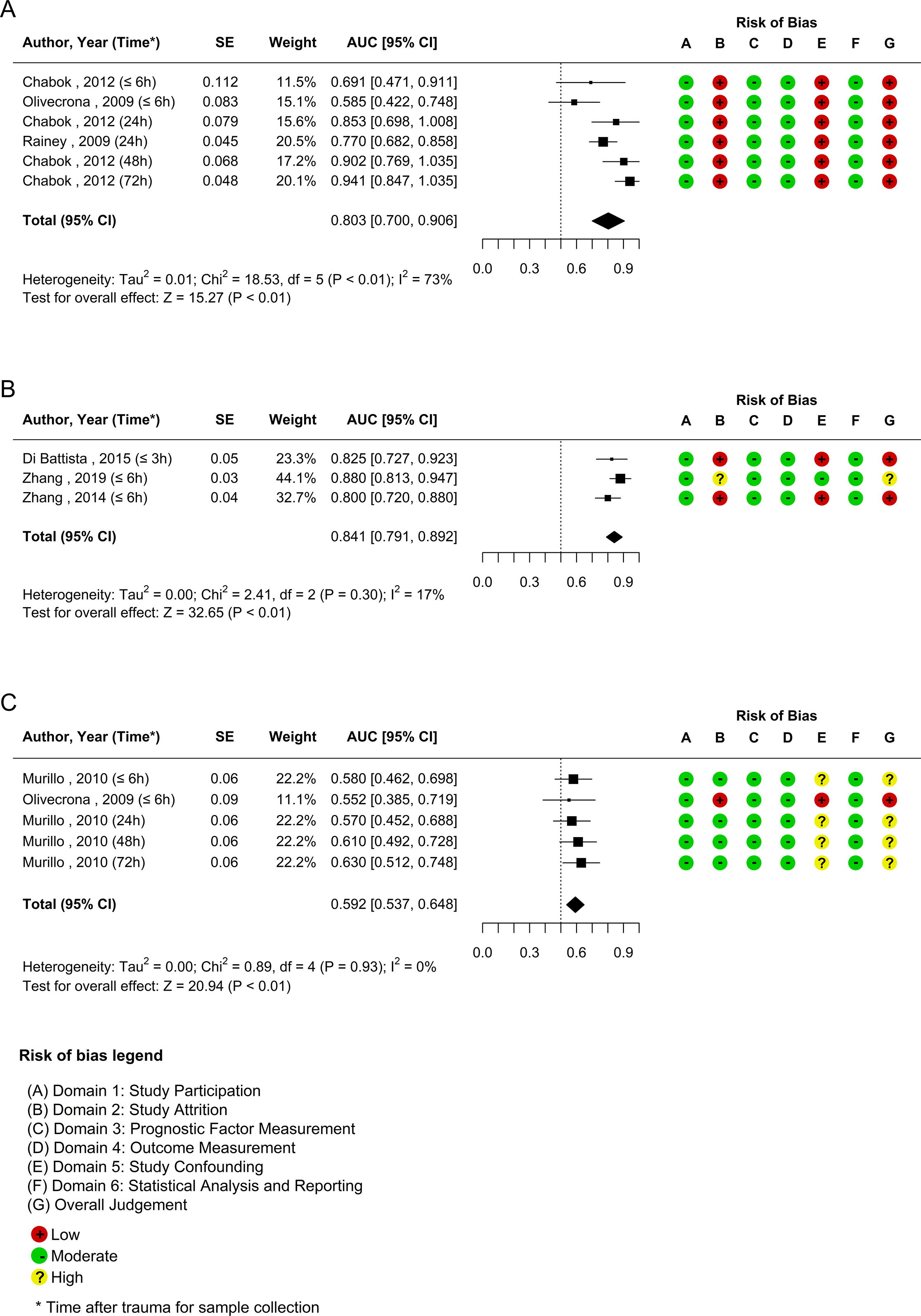
Meta-analysis of the prognostic capacity of S100B concerning functional capacity and risk of bias measured by QUIPS. Images grouped by (
Description of the Studies Evaluating the Relationship Between S100B Concentration and the Outcomes of Interest Through AUC
CI, confidence interval; SE, standard error; –: unavailable data.
When evaluating the prognostic capacity of S100B 6 months after trauma, an AUC of 0.758–0.8919 was found33,48,53,57,60 (Table 4); in the studies included in the meta-analysis, values ranged from 0.800 to 0.880; pooled AUC 0.841 (95% CI = 0.791–0.892); I2 = 17% (p = 0.30); however, only investigations with samples collected early, ≤6 h after trauma, were included in these analyses (Fig. 2b).
The studies evaluating the AUC after 12 months of trauma met the requirements for meta-analysis. An AUC of 0.552–0.630 was observed.37,65 The pooled AUC was 0.592 (95% CI = 0.537–0.648), I2 = 0% (p = 0.93) (Fig. 2c).
Comparison of the prognostic capacity of S100B in samples collected in the early and late phases in relation to functional capacity
The two studies assessing functional capacity at 3 months after trauma with early samples and that were included in the meta-analysis55,65 had pooled AUC of 0.623 (95% CI = 0.492–0.753); I2 = 0% (p = 0.45) (Fig. 3a); in relation to the late phase, the pooled AUC was 0.864 (95% CI = 0.776–0.952); I2 =59% (p = 0.06) (Fig. 3b).

Meta-analysis of the comparison of the prognostic capacity of S100B in samples collected in the early and late phases concerning functional capacity and risk of bias measured by QUIPS. Images grouped by (
As previously mentioned, all studies evaluating functional capacity after 6 months of TBI and that met the requirements for meta-analysis were carried out with samples from the early phase after this injury; for these studies, the pooled AUC was 0.841 (95% CI = 0.791–0.892); I2 = 17% (p = 0.30) (Fig. 2b). For late samples, AUC was presented in two studies, without other information. Nevertheless, in these samples, the values were 0.758 48 and 0.891, 57 close to that observed in early phase, between 0.800 and 0.88033,47,53 (Table 4).
Regarding the evaluation after 12 months of the traumatic event, the samples related to the early phase had an AUC of 0.552 65 and 0.580. 37 In the meta-analysis for these samples, the pooled AUC was 0.571 (95% CI = 0.475–0.667); I2 = 0% (p = 0.79) (Fig. 3c). Just one study 37 evaluated late samples and indicated, in collections of 24, 48, and 72 h, AUC of 0.570–0.630 (Table 4). For these late samples, meta-analysis was not performed as the values were from only one study. According to the AUC, late and early samples had insufficient discriminatory capacity for dependence at 12 months (AUC <0.70).
S100B and vital status
The vital status outcome was the basis of 22 investigations.11,16,33,36–39,48,49,52–54,58,60–62,65,66,68,69,71,72 For this outcome, in some studies GOS score 1 was considered an indication of death after TBI.
Comparison of S100B concentration in dead and surviving individuals after moderate and severe blunt TBI
Of the eight studies11,16,33,38,39,52,68,69 carrying out tests to compare mean S100B concentrations among patients who died and survivors until hospital discharge after TBI, only one study did not show a higher concentration in patients who died. 52
Two studies evaluated the relationship between S100B concentration and the outcome of death65,66 3 months after the trauma. In both cases, a higher concentration was found among patients who died when compared with those who did not die.
Seven investigations described the vital status of patients with TBI 6 months after injury,11,48,53,58,60,62,71 six carried out comparison tests between S100B concentration values among patients who died and survivors. In five studies,11,48,60,62,71 death cases had higher concentrations; however, in one study 58 the concentration was similar.
Three studies analyzed vital status at 12 months after trauma37,58,65 and in only one study the concentration of S100B was similar among patients who did or died or did not die. 58 Whenever vital status was analyzed after trauma, most studies showed higher concentrations of S100B among individuals who died.
Prognostic capacity of S100B for vital status until hospital discharge and/or at 3, 6, and 12 months after moderate and severe TBI
The AUC range for vital status at discharge was 0.67–0.9611,33,36,38,39,49,52 (Table 4); for the studies included in the meta-analysis, values ranged from 0.710 to 0.96011,33,36,39,52; the pooled AUC was 0.818 (95% CI = 0.757–0.879); I2 = 74%, p < 0.01) (Fig. 4a).

Meta-analysis of the prognostic capacity of S100B concerning vital status and risk of bias measured by QUIPS. Images grouped by (
Two studies evaluated the prognostic capacity of S100B for vital status 3 months after the traumatic event through AUC and the values were 0.68 65 and 0.69 66 (Table 4). In the meta-analysis, pooled AUC was 0.687 (95% CI = 0.607–0.766) (I2 = 0%, p = 0.91) (Fig. 4b).
The evaluation of the discriminative capacity of S100B for vital status at 6 months showed an AUC of 0.719–0.85511,47,48,53 (Table 4). In the three studies11,47,53 included for meta-analysis, the variation was similar (Table 4 and Fig. 9), and pooled AUC was 0.818 (95% CI = 0.784–0.852), I2 = 11% (p = 0.35) (Fig. 4c).
In the assessment of vital status at 12 months, AUC was highlighted in two studies37,65 and the values were 0.647–0.840 (Table 1). In the meta-analysis, the pooled AUC was 0.731 (95% CI = 0.661–0.801), I2 = 13% (p = 0.33) (Fig. 4d).
Comparison of the prognostic capacity of S100B in samples collected in the early and late phases in relation to vital status
In the meta-analysis of studies that analyzed the prognostic capacity of S100B for vital status until hospital discharge11,36,39 with early samples, the AUC range was 0.710–0.865 and the pooled AUC was 0.778 (95% CI = 0.703–0.853), I2 = 32% (p = 0.22) (Fig. 5a). In late samples, the AUC range was 0.776–0.960 and the pooled one was 0.842 (95% CI = 0.770–0.914), I2 = 74% (p < 0.01) (Fig. 5b).

Meta-analysis of the comparison of the prognostic capacity of S100B in samples collected in the early and late phases concerning vital status and risk of bias measured by QUIPS. Images grouped by (
It could be observed that the S100B performance of samples collected in the late phase was superior when compared with early samples in relation to the vital status assessed until hospital discharge; however, the heterogeneity of results in late samples was statistically significant.
Vital status analyses after 3 months of trauma included an investigation with early samples and another with late samples. For these studies, AUC values were similar, 0.680 and 0.690 for collection in the early and late phases, respectively (Table 4).
Regarding vital status 6 months after trauma, three studies11,47,53 evaluated the early phase and the AUC variation among these studies was 0.719–0.841. In the meta-analysis, pooled AUC was 0.808 (95% CI =0.739–0.877), I2 = 58% (p = 0.09) (Fig. 5c). Two studies evaluated the performance of late samples using AUC11,48 and values of 0.735 and 0.855 were observed (Table 4). However, they did not present additional information for meta-analysis.
Regarding vital status after 12 months of trauma, two studies evaluating early samples37,65 were found and the values were 0.647 65 and 0.690 37 (Table 4). In the meta-analysis, pooled AUC was 0.675 (95% CI = 0.548–0.802), I2 = 0%, p = 0.75) (Fig. 5d). Regarding late samples, only one study 37 brought AUC with values that varied from 0.670 to 0.840 (Table 4).
Discussion
The complex and heterogeneous nature of brain injury makes prognosis prediction challenging, especially in cases of moderate and severe TBI. This challenge falls on health professionals, who need to establish behaviors to provide the best outcomes, guide the family, and give information about the patient’s recovery process. Based on this provocative situation, possible tools have been discussed to equip the health team’s decision-making behavior, 34 the S100B protein being one of them.
S100B, the focus of this study, has stood out in recent years as a biomarker related to the diagnosis and prognosis of brain injury. The first evidence on this biomarker in the TBI setting was in 1995 75 and in 2001 there was research evaluating the relationship between S100B concentration and functional capacity and/or vital status outcomes in patients with moderate and severe blunt TBI.
The findings of this review endorsed the presence of a relationship between the concentration of this biomarker and unfavorable outcomes of TBI. In ∼80% of the investigations that tested the association among these variables, differences in S100B concentrations and higher values were found among individuals who died or became dependent after injury when compared with survivors and independent ones.9,11,16,33,37–39,47,48,50,55,56,60,62–69
Furthermore, the prognostic capacity of the biomarker observed in the AUC indicated satisfactory (AUC ≥0.70)11,36,37,39,66 or good (AUC ≥0.80)11,33,37,52,53,60,63 performance in most analyses of S100B. However, all investigations analyzing functional capacity at 12 months indicated unsatisfactory performance (AUC 0.552–0.630).37,65 The same was found regarding vital status at 3 months (AUC 0.68 and 0.69)65,66; however, for this period, only two studies were carried out (Table 4).
When the patient suffers a TBI, S100B is released into the blood immediately after injury, with the mechanism by which this happens not being completely clear. Studies indicate that this protein passes into the blood due to the breakdown in the BBB; others indicate that the level of the protein in the injured brain increases regardless of the integrity of this structure76,77 S100B probably initially rises in the brain extracellular space before being transported to the CSF, when passive diffusion occurs into the blood. Recently, the glymphatic system pathway, a system that unites blood vessels, interstitial fluid, CSF, and glial cells was discovered. This system can cause the release of S100B into blood and other biofluids as this is a low molecular weight protein. 78
Studies show that there is a correlation between the release of the protein and the volumes of brain contusion,10,79 justifying the diagnostic value of the protein for TBI severity and therefore for prognostic capacity since unfavorable outcomes are related to this severity. 10
S100B has a peak concentration in the third hour after TBI with reduced levels in subsequent samples. Prolonged elevation is associated with continuous influx of the biomarker. 80 Although the concentration of S100B commonly declines over time, it is described that patients with TBI who progress with dependence or death show a less significant drop between sequential samples; therefore, in this group, the average values and peak concentration are higher than those patients with TBI that do not present this outcome 16 and this kind of protein level behavior adds prognostic value to the consequences of this injury.
Similarly, the discriminative capacity of S100B can be reinforced by its different functions according to its concentration.27,81 At low levels, S100B promotes neurogenesis24,82 and neuronal plasticity.25,26 When there are high concentrations of this protein, there is stimulation of astrocytes, microglial cells and inflammatory cytokines, causing an inflammatory response, production of nitric oxide, oxidative stress and, consequently, neuronal dysfunction or cell death.83,84
Regarding the prognostic capacity for functionality in different periods after injury, results of the AUC meta-analysis indicated that S100B helps with prognosis 3 and 6 months after injury (pooled AUC: 0.803 and 0.841, respectively); however, for 6 months, better results were observed in heterogeneity analyses (p = 0.30, I2 = 17%); for 3 months, there was a statistically significant heterogeneity (p < 0.01, I2 = 73%). The previously mentioned unsatisfactory performance for 12-month prognosis was ratified in the meta-analysis with pooled AUC (0.592) and no heterogeneity within studies (I2 = 0%). That said, the best S100B predictive capacity for functionality was for 6 months and its worst performance for 12 months after trauma.
The literature shows that the victims recovery is a dynamic process, dependent on education, socioeconomic level, previous illnesses, severity of injuries, interventions applied, and, especially, time, since the injured brain tissue may not have been destroyed and gradually resume normal function as the clinical state stabilizes and neural connections remodel.85–87
In this process, there is a marked improvement in patients up to 3 months post-trauma, continuous recovery up to 6 months, and little progress up to a year, configuring stability in the TBI recovery process at 6 months postinjury.85–87 It is believed that the prognostic performance of S100B identified in this review, with emphasis on the 6-month period, reflected the recovery pattern of TBI victims. Authors point out that the most appropriate period to establish outcomes of moderate and severe TBI is 3 and 6 months after injury, since at this stage functional capacity shows spontaneous improvement associated with recovery from injuries and the influence of rehabilitation interventions. 34
Deaths are other outcomes with great social impact after moderate/severe blunt TBI and are very common among severe cases.32,88,89 This frequency reaches levels above 60% until hospital discharge. 90 In the meta-analysis, it was found that the grouped AUC of S100B in relation to the prediction of death up to hospital discharge and at 6 months were similar (0.818), presented a lower value at 12 months (0.731) and presented unsatisfactory levels 3 months after injury (0.687). Study results heterogeneity was low in the 3-, 6-, and 12-month analyses; however, at hospital discharge, it was high and statistically significant (p < 0.01 and I2 = 74%). Given these findings, it was considered that the best predictive capacity was considered to be 6 months after the traumatic event.
One of the challenges of predicting results after TBI, highlighted in the literature, concerns the ideal time to collect the biofluid used to analyze the S100B concentration. The initial sample of S100B can be considered promising for predicting results, since its release of S100B occurs immediately after injury. 80
In this literature review, a meta-analysis of early and late samples was performed for vital status at hospital discharge and for functional capacity at 3 months. In both analyses, the pooled AUC presented higher values for late samples (>6 h). Furthermore, meta-analysis was applied to studies with early samples (≤6 h) for vital status and functional capacity at 6 and 12 months; for 6 months, the discriminatory capacity of S100B was good, for death and functional capacity (pooled AUC = 0.808 and 0.841, respectively). For the 12-month prognosis, for both outcomes, the performance of the protein was unsatisfactory. Therefore, late samples have shown better prognostic capacity than the early ones; the isolated analysis of this last type of sample indicated that it presents poor performance for outcomes after 12 months.
Higher mean and peak values of S100B in patients with TBI who become dependent or die may be related to the worsening of the injury that can occur hours and even weeks after severe and moderate TBI. Among the worsening factors, we can mention, in addition to secondary injuries, the process of attempting to repair the injury, which begins immediately after the trauma and triggers inflammatory responses that can worsen it, especially due to changes in vascular permeability.6,85
Furthermore, in the first weeks after TBI, there are different levels of cerebral edema, changes in calcium influx and glutamate levels. During this period, processes of cell death and oxidative stress are present in the central nervous system.6,91 Therefore, it is believed that the better performance of late samples is associated with the pathophysiology of TBI, specifically with the process of attempting to repair the injury and worsening factors, which occur over a period of >6 h.
As limitations of this investigation, although the study provides an update on known facts, most of the studies were published in 2016 (n = 4). Between 2019 and 2023, eight studies were identified. Furthermore, the high risk of bias in the overall assessment of most of the studies analyzed should be highlighted. The only study presenting low risk of bias was included in the meta-analysis of vital status until hospital discharge; a study with a moderate risk of bias was included in the meta-analysis for death at 12 months, functional capacity at 6 and 12 months; and a single study with moderate risk was included. Therefore, subgroup and sensitivity analysis strategies were not feasible in this review.
Furthermore, among the limitations, there is the fact that several studies presented the AUC; however, they did not present confidence interval or standard error, making their inclusion in meta-analyses impossible. No study was found that applied the AUC to assess the discriminative capacity of functionality until hospital discharge. For late samples, the meta-analysis was not performed for the 6- and 12-month periods for both outcomes analyzed. In addition, in early and late samples this analysis was not carried out for vital status at 3 months after TBI. Moreover, late samples were collected over a wide range of time after TBI, between 24 and 96 h.
Conclusion
This is an interesting update on already known facts. The systematic review indicated that S100B is a promising tool for estimating the prognosis of functional capacity and vital status in patients with moderate and severe TBI, since in most studies the levels of this protein were significantly higher in dependent patients or in those who died after this injury. The best discriminative performance of S100B for functional capacity and vital status was observed for the 6-month period after moderate and severe TBI, when low heterogeneity in study results and good prognostic capacity were observed (pooled AUC 0.841 and 0.818 for functional capacity and vital status, respectively). However, for the analyses 12 months after injury, the discriminatory capacity of S100B for functional capacity was unsatisfactory in all studies and the pooled AUC was 0.592. For death at 3 months, a similar result was observed with a pooled AUC of 0.687. Blood samples collected later, 6 h or more after TBI, performed better to predict functional capacity at 3 months and vital status at hospital discharge than early samples. However, the information from the studies was insufficient to carry out a meta-analysis of samples from other periods. In addition, the heterogeneity in the results of the studies included in some meta-analyses carried out was high. Therefore, further studies are required that indicate the AUC values associated with CI or standard error, especially in samples collected late after injury.
Footnotes
Acknowledgments
The authors thank the Library of the School of Nursing at the University of São Paulo for their support in developing the search strategies, selecting the databases, and providing guidance on reference management tools.
Author’s Contributions
L.C.F. conceived and designed this study, contributed to the investigation and methodology (bias analysis and literature search), and was responsible for drafting, editing, and submitting the article. D.A.S. was involved in the investigation and methodology (literature search). T.O.R. was involved in the investigation and methodology (bias analysis) and wrote the original draft. R.A.O. carried out the statistical analysis and provided significant inputs into the formation of the analysis group. L.S.N. provided her professional opinions on systematic reviews and contributed to the overall research and interpretation of the results, as well as to the investigation and methodology (bias analysis and literature search). A.M.T.C. played a significant role in data curation, formal analysis, and visualization (meta-analysis and forest plot). R.C.A.V. led the validation and was responsible for reviewing and editing the article. W.S.P. provided his professional opinions on neurosurgery and TBI research. L.L.M. contributed her expertise in biomarkers and protein measurement techniques and contributed to the interpretation of results. R.M.C.S. conceived and designed this study, was responsible for project administration, validation, analysis, and interpretation, and contributed to the investigation and methodology (bias analysis and literature search), as well as to drafting and editing the article. This article has been approved for publication by all authors.
Author Disclosure Statement
The authors have no competing interest to disclose.
Funding Information
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasil, Finance Code 001.
