Abstract
A sizable proportion of patients with mild traumatic brain injury (mTBI) have persistent symptoms and functional impairments months to years following injury. This phenomenon is continually observed despite an explosion of research and interest in improving mTBI clinical outcomes over the last two decades. All pharmacological clinical trials to date have failed to demonstrate improved outcomes for mTBI. One possible explanation for these continued failures is an overly myopic approach to treating mTBI (i.e., testing the effect of a single drug with a specific mechanism on a group of people with highly heterogenous injuries). Clinical presentation and prognosis of mTBI vary considerably between patients, and yet we continue to assess group-level effects of a homogenized treatment. We need to utilize an equally complex treatment approach to match the extraordinary complexity of the human brain. Dynamical systems theory has been used to describe systems composed of multiple subsystems who function somewhat independently but are ultimately interconnected. This theory was popularized in the motor control literature as an overarching framework for how the mind and body connect to interact and move through the environment. However, the human body can be viewed as a dynamical system composed of multiple subsystems (i.e., organ systems) who have isolated functions, which are also codependent on the health and performance of other interconnected organ systems. In this perspective piece, we will use the example of mTBI in the obese patient to demonstrate how broadening our approach to treatment of the individual (and not necessarily the injury) may ultimately yield improved outcomes. Furthermore, we will explore clinical and pre-clinical evidence demonstrating multiple system interactions in the context of obesity and TBI and discuss how expanding our understanding of the mechanistic interplay between multiple organ systems may ultimately provide a more personalized treatment approach for this mTBI patient subpopulation.
Introduction
Obesity is now considered a “global epidemic” with 13% of the global adult population considered obese. 1 In addition, traumatic brain injury (TBI) is often described as a “silent epidemic” with an estimated 50 million individuals suffering each year;2,3 however, these numbers are likely a gross underestimation. Emerging evidence suggests that obese patients subsequently exposed to TBI suffer from worse outcomes, including prolonged symptomology 4 and higher mortality rates,5–8 compared with patients with “normal” BMI. A majority of this evidence has been presented in the severe TBI population (i.e., those with a Glasgow Coma Scale [GCS] at presentation <8). However, based upon prior research and Centers of Disease Control estimates for obese BMI, approximately 1.1 million people in the United States have both functional limitations related to a “mild” TBI (mTBI; GCS = 13–15 at presentation) 1-year postinjury and obese BMI. 9 There is high potential that obesity could mediate outcomes from mTBI, as well as severe TBI.4,10,11 Higher post-traumatic levels of circulatory obesity-induced inflammation, higher long-term levels of inflammation, and association with myriad comorbid conditions may be the leading mechanistic hypothesis for this relationship (see Fig. 1).4,12

Schematic representation of our central hypothesis regarding adipose tissue-induced inflammation after mTBI.
Overview of dynamical systems theory and application to human physiology
Dynamical systems theory (DST) became popularized in the 1980s as a means for understanding how we learn to interact with the world around us. 13 Within the context of motor control and learning, DST describes accomplishing a given task as the interaction between multiple subsystems within the body, as well as interactions between our bodies and the environment within the constraints of the task itself. 14 On a more comprehensive scale, the theory suggests that the human condition is usually in a steady state of “health” or “disease.” While perhaps appearing “steady” on the surface, either of these states is characterized by a complex interplay of feedforward and feedback mechanisms seeking to reinstate homeostasis after perturbations.15,16 Human existence shifts back and forth between health and disease steady states. The intensity of the shift itself can be gradual (as in the increased likelihood of disease states observed as we age) or abrupt. 16 Abrupt shifts into a disease state can occur when one of the homeostatic mechanisms keeping us in a healthy state is rapidly disturbed by some stimulus.
A mechanical force resulting in mTBI is a clear example of an event which can rapidly shift our entire system into a disease state. The majority of research on understanding mTBI and how best to treat it have prioritized reductionist approaches aiming to treat the injury itself.17,18 Over the last 10–15 years, scientific and clinical communities treating patients with mTBI have broadened our understanding of mTBI recovery to include preexisting psychological, cognitive, and cortical-centric (e.g., headache/migraine, vestibular dysfunction) risk factors.19–24 Our knowledge of how to manage the postinjury sequelae of mTBI has also expanded, and it is now well-understood that mTBI can be “subtyped” for the purposes of targeting therapies to the individual’s specific needs. 25 The purpose of this article is to present evidence that incorporating health of the bodily systems below the cervical spine into personalized management of patients with mTBI can improve outcomes and quality of life on a broader scale. DST can provide a viable framework by which to conceptualize and apply treatments for this heterogenous injury.
Appreciating the dynamic interaction of systems is better understood within the context of severe TBI, as systemic effects following severe TBI have been repeatedly noted in the liver, gastrointestinal, pulmonary, cardiovascular, kidney, and endocrine systems.26–29 Multisystem interactions are less well-characterized in the mTBI patient, possibly because serious or life-threatening changes to other organ systems are less likely to occur as a direct result of mTBI. The endocrine system is the primary exception, as multiple studies have identified endocrine system issues following mTBI.30–32 Izzy et al. 30 recently conducted a study of 4,351 patients with mTBI and found increased risk for diabetes mellitus (hazard ratio = 1.9) and obesity (hazard ratio = 2.1). These studies have provided useful data for understanding how disturbing the central nervous system through mTBI can directly influence the downstream development of chronic issues in another organ system. In DST terminology, mTBI can act as an abrupt stimulus to transition the body into a disease system. However, we have very little information about how the current state of the system (at the time of injury) influences recovery from an mTBI.
Obesity has been considered a steady disease state, but, in terms of DST, a more precise term may be “stable attractor”. 13 As a person shifts into the obese disease state, a host of comorbidities and other medical conditions are “attracted” to the new conditions of the human body system (see Fig. 2). Multiple subsystems within the body dynamically adjust to excess calorie intake by storing adipose tissue within the capacity of that individual’s body. 33 As this pattern continues, inflammatory cytokines and other cells increase in concentration within the blood.33–35 Blood lipids become more prevalent, and adipose tissue is deposited at higher than normal rates around the liver, viscera, kidneys, and pancreas ultimately leading to multiorgan system dysfunction and medical conditions such as insulin resistance and cardiovascular disease. 33 Emerging evidence suggests that visceral fat itself should be viewed as an organ within the endocrine system, as it can self-generate hormone signals and inflammatory cells. 36 Critically, inflammatory cells secreted by adipose tissue can cross the blood–brain barrier, increasing overall neuroinflammation and worsening clinical symptoms and cognitive performance. 37 In other words, visceral fat is capable of creating a feedforward mechanism with the brain to maintain a stable disease state across multiple subsystems. The interaction of a physical trauma like mTBI into a stable disease system can theoretically exacerbate these feedforward processes into a deeper state of attraction.

Depiction of a gradual shift from healthy bodily systems (left) with low circulatory inflammation, progressing over time to pathological interactions between bodily systems associated with obesity (right), increased circulatory inflammation, and impairment of microglia and cognitive function.
Interplay between obesity and traumatic brain injury in the pre-clinical setting
The pathophysiology of both obesity38–40 and TBI41–44 is characterized by widespread inflammation throughout the body, effects of which are associated with long-term neurological consequences.39,44,45 Experimentally, comorbid models of diet-induced obesity and TBI are associated with exacerbated neuroinflammatory responses and worsened neurological outcomes.35,46–49 Specifically, preexisting diet-induced obesity leads to an exacerbation of microglial activation in several brain regions, including the cerebral cortex, corpus callosum, and hypothalamus, at 30 days following exposure to mTBI. 47 Notably, although exposure to mTBI in lean mice did not result in evident anxiety-like behavior, obese male mice subsequently exposed to mTBI exhibited increases in anxiety-like behavior. 47 Further evidence demonstrating an interaction effect of obesity/diet and mTBI is that obese mice subsequently exposed to mTBI exhibited an exacerbation of central insulin resistance, microglial activation, and deficits in memory and anxiety-like behavior. 35 Similarly, obese rodents subsequently exposed to moderate TBI displayed worsened short-term sensory and memory deficits 48 and motor and learning/memory. 46 Notably, such diet-induced exacerbations were associated with increases in expression of markers of neuroinflammation and oxidative stress and experimentally-induced lesion volume, up to one month postinjury. 46
However, despite this, we still lack a clear understanding of the interplay between these two disease processes. Increasing our knowledge is not only important at a basic physiological level but also for the development of novel therapeutic targets for mTBI patients with a preexisting metabolic condition. Potential molecular pathways of interest from a therapeutic viewpoint include obesity-induced changes in gut microbiota, adiposity, and subsequent metabolic dysfunction. 50 A recent study by Henry et al., demonstrated a potential bidirectional relationship between the adipose tissue and brain in a pre-clinical model of comorbid diet-induced obesity and moderate TBI. 49 In this study, high-throughput transcriptomic analysis demonstrated a moderate TBI-obesity interaction of central and peripheral microglia/macrophage responses, including super-additive changes in several canonical inflammatory pathways. Notably, such interaction effects in cellular responses were associated with an obesity-induced exacerbation of cognitive deficits following exposure to moderate TBI. These findings are of potential importance as they show for the first time a bidirectional relationship between the adipose tissue and brain in the context of comorbid obesity and TBI, one that may be pharmacologically targetable.
In agreement with this, a previous body of research demonstrated a vital role for adipose tissue inflammation, specifically that of NOD-, LRR- and pyrin domain-containing protein 3 (NLRP3)-induced inflammation, in driving obesity-induced cognitive deficits; effects of which were shown to be mediated through IL-1-mediated microglial activation. Thus, suggesting that NLRP3/IL-1β signaling may underlie correlations between visceral adiposity and cognitive impairments in human obesity. 36 Notably, findings from Henry et al. demonstrate that obesity-induced increases in visceral adipose tissue expression of NLRP3 and IL-1β were further exacerbated in the presence of moderate TBI. Thus, it is plausible to suggest that targeting of visceral adipose tissue in the context of comorbid obesity and mTBI may be a novel therapeutic target for patients with preexisting metabolic dysfunction, including obesity. However, it is important to note that these studies are focused on the impact of diet-induced obesity models on TBI outcomes; thus, perhaps not considering the complex nature of obesity etiology which may result from an array of factors, including genetics, stress, and other lifestyle factors. This will be an important consideration for future studies.
Interplay between obesity and mTBI in the clinical setting
One particularly interesting example of the multidimensional relationship between obesity as a disease state and mTBI can be found in the bariatric surgery literature. Recent case reports have studied severely obese patients who are suffering from chronic effects related to mTBI. 51 Following bariatric surgery, the desired effect of weight loss and reversal of diabetes mellitus was achieved. Interestingly, there were also notable improvements in executive function and alleviation of symptoms the patient associated with their mTBI. 52 These results are intuitive, as notable and sustained improvements in cognition, sleep, headache/migraine, and psychological health have been reported following bariatric surgery, 52 which are domains with a high degree of overlap with commonly reported symptoms of mTBI (see Fig. 3). This evidence had led clinicians and researchers to suggest that bariatric surgery may be a useful treatment option for the severely obese patient experiencing chronic issues from mTBI. However, more conservative treatment options for managing mTBI in the obese patient should be considered first.
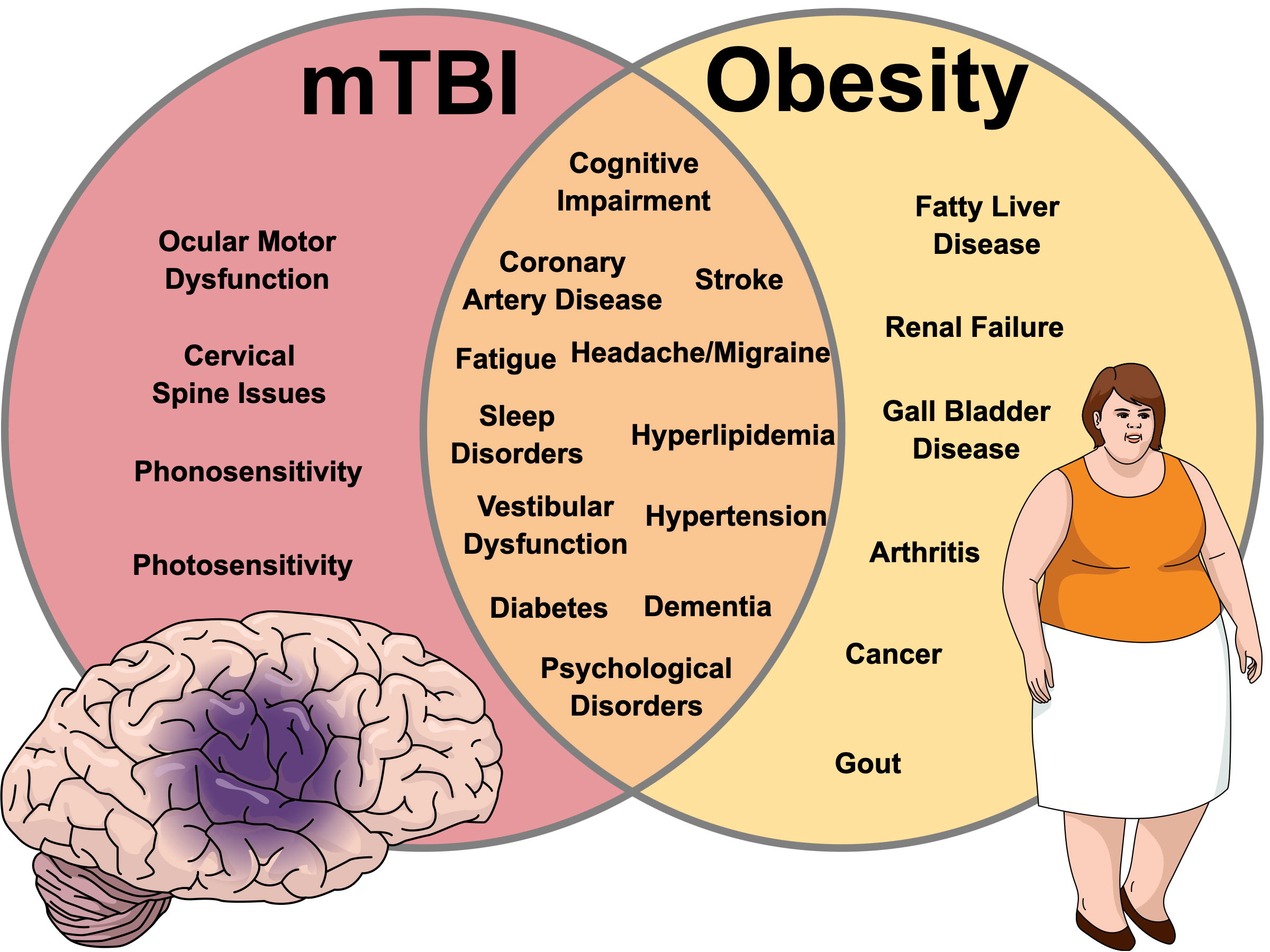
Venn diagram depiction of overlap between symptoms and medical conditions following mild traumatic brain injury (mTBI; left) and obesity (right).
Future directions for personalized mTBI treatments for patients with pre-existing metabolic dysfunction
Therapeutic potential for behavioral change and weight loss programs
Overwhelming evidence suggests that exercise and weight loss improve health and outcomes in the obese patient. A growing body of evidence also suggests that early aerobic exercise is beneficial to shortening recovery from mTBI. Early (following 24–48 h of relative rest) physical activity improves mTBI symptoms faster and reduces recovery time amongst adolescent athletes. 25 A 2023 systematic review of 7 studies reported that prescribed physical activity reduces recovery time following sport-related concussion by 4.6 days. 53 Amongst adolescents who adhered to a physical activity prescription following 72 h of relative rest, significant reductions in total symptoms were reported at 2-weeks postinjury in comparison to a control group, which waited to be asymptomatic at rest before initiating physical activity. 54
Conversely, strict rest prolongs recovery time from mTBI and is associated with higher depression and anxiety symptoms. 55 Varner et al. 56 conducted the only known randomized controlled trial of early (>48 h of relative rest) physical activity as an intervention in adults with mTBI. The authors reported that a prescription of 30 min light exercise 5x per week for 1-month postinjury did not elicit a treatment effect compared with those with no instructions to exercise. 56 Participants in the intervention group were instructed to perform light walking or stationary cycling “at a pace that does not cause you to sweat or breathe harder”. 56 It is possible that this prescribed intensity was not strong enough to elicit a treatment effect, as more vigorous forms of aerobic exercise prescribed in both adult outpatients with persistent symptoms and adolescent athletes have consistently yielded positive results.53,57
Despite the intuitive connection, there is little evidence available that demonstrates the efficacy of exercise and/or weight loss interventions on improvements after mTBI in the obese patient. One small randomized controlled trial by Driver et al. 58 in the moderate-severe TBI population showed that a prescribed weight-loss intervention significantly improved weight loss, blood pressure, triglycerides, and cholesterol in comparison to a control group. Results also suggested that the intervention group had better self-reported health habits than the control group within the first year. No data were reported from this study specific to TBI symptoms, such as cognition.
Pharmacological interventions for the obese patient with mTBI
A targeted anti-inflammatory drug for obese patients with mTBI is an obvious potential intervention for this population. Research has recently begun to investigate the impact of NLRP3-inhibitors on recovery from experimental brain injury in animal models. Ismael et al. 59 reported that administration of MCC950, a NLRP3 inhibitor, significantly improved neurological function and reduced cerebral edema at 1 and 3 h post-TBI. MCC950 may inhibit inflammasome priming by downregulating nuclear factor kappa B and caspase-1, which are key components of inflammasome activation. 59 Treatment with MCC950 also reduced NLRP3 expression and IL-1β release while improving short-term memory deficits in a blast exposure rat model. 60 Neurodegenerative disease is associated with NLRP3 inflammation, and utilizing MCC950 has also been of interest in animal models to potentially reduce risk for chronic neurodegenerative disease. Naeem et al. 37 recently demonstrated MCC950’s ability to reverse amyloid beta plaque formation and improve cognitive function in rats with Alzheimer’s Disease (AD). Future work should consider the potential role of NLRP3 inhibitors in this subpopulation of TBI patients of all severities.
Therapeutic potential of antiobesity drugs following mTBI
Other potential avenues of pharmacological intervention for this subpopulation of TBI patients of all severities may include antiobesity drugs. Glucagon-like peptide-1 (GLP) is a gastrointestinal peptide hormone that is secreted by the intestinal tract and mediates effects through the G-protein-coupled receptor (GLP1R). 61 The long-acting GLP-1R agonist, semaglutide, has been approved by the FDA in both oral and injectable forms for the treatment of T2DM and in injectable forms for the treatment of obesity. 62 In a double-blind Semaglutide Treatment Effect in People with Obesity (STEP) 1 trial, for 1961 nondiabetic adults with a BMI of 30 kg/m2, treated once weekly with semaglutide or placebo (2.4 mg; subcutaneous administration), for a total of 68 weeks, an average weight loss of 15.3 kg was reported in semaglutide treated participants. 63 Importantly, both randomized STEP 3 and 8 clinical studies have reported similar effects on weight loss.64,65 The mechanisms of action of GLP-1R agonists on weight loss include a decrease in appetite and food intake, increased satiety, decreased gastric emptying and gastrointestinal motility, and a decrease in insulin resistance. 61 However, there is now emerging evidence that GLP-1R agonists, in addition to effects on weight loss, can reduce inflammation in both mice and humans.66–68
Another potential antiobesity target that has received significant attention in recent years is the stress-regulated hormone, growth differentiation factor (GDF)-15, which is reported to decrease food intake, energy expenditure, and body weight in mice and nonhuman primates through the brainstem-restricted receptor, GDNF family receptor alpha-like.69–71 In a similar manner to that of GLP-1R agonists, GDF-15 is reported to exhibit anti-inflammatory effects in obese models. 72 Given the evidence that targeting of systemic inflammatory responses is associated with beneficial effects on brain function in obesity, 36 it is plausible to suggest that antiobesity drugs (GLP1R, GDF-15) may offer a potential novel avenue of investigation in the context of comorbid obesity and TBI of all severities, at least in part, using their ability to decrease inflammatory responses.
Conclusion
Given the ever-increasing rates of obesity in modern society, it is plausible to suggest that more obese patients will suffer an mTBI throughout their lifetime. There is now increasing evidence that the presence of individual comorbidities, including but not limited to metabolic dysfunction, may play an important role in determining patients’ outcomes following exposure to mTBI. As such, it is important that we move away from the notion of a ‘magic bullet’ treatment for mTBI patients to that of a personalized treatment approach based on individual health factors throughout the person’s entire body. Viewing management of this injury through the lens of dynamic systems theory can ultimately lead to an improvement in patient outcomes and quality of life following mTBI
Footnotes
Authors’ Contributions
Shawn R. Eagle conceived of this review topic, developed the initial outline, produced the figures, and wrote the clinical research sections of the review. Rebecca J. Henry critically reviewed the outline and first draft and wrote the preclinical and therapeutic sections of the review.
Author Disclosure Statement
The authors have no conflicts of interest to disclose.
Funding Information
No funding was received for this article.
