Abstract
Blast-induced neurotrauma (BINT) is an important injury paradigm of neurotrauma research. This short communication summarizes the current knowledge of BINT. We divide the BINT research into several broad categories—blast wave generation in laboratory, biomechanics, pathology, behavioral outcomes, repetitive blast in animal models, and clinical and neuroimaging investigations in humans. Publications from 2000 to 2023 in each subdomain were considered. The analysis of the literature has brought out salient aspects. Primary blast waves can be simulated reasonably in a laboratory using carefully designed shock tubes. Various biomechanics-based theories of BINT have been proposed; each of these theories may contribute to BINT by generating a unique biomechanical signature. The injury thresholds for BINT are in the nascent stages. Thresholds for rodents are reasonably established, but such thresholds (guided by primary blast data) are unavailable in humans. Single blast exposure animal studies suggest dose-dependent neuronal pathologies predominantly initiated by blood–brain barrier permeability and oxidative stress. The pathologies were typically reversible, with dose-dependent recovery times. Behavioral changes in animals include anxiety, auditory and recognition memory deficits, and fear conditioning. The repetitive blast exposure manifests similar pathologies in animals, however, at lower blast overpressures. White matter irregularities and cortical volume and thickness alterations have been observed in neuroimaging investigations of military personnel exposed to blast. Behavioral changes in human cohorts include sleep disorders, poor motor skills, cognitive dysfunction, depression, and anxiety. Overall, this article provides a concise synopsis of current understanding, consensus, controversies, and potential future directions.
Introduction
Blast-induced neurotrauma (BINT) has been identified as a signature wound during conflicts in Iraq and Afghanistan.1,2 More than 20 years have passed since the early research publications of BINT.3–5 Since then, significant efforts have been made to understand BINT. In the past 20 years, ∼$900 million USD has been spent on research funding related to BINT. 6 In this short communication, we summarize (Fig. 1) the current knowledge, consensus, controversies, and potential future directions. Additional details regarding this short communication are provided in the section Transparency, Rigor, and Reproducibility Summary at the end of the article.
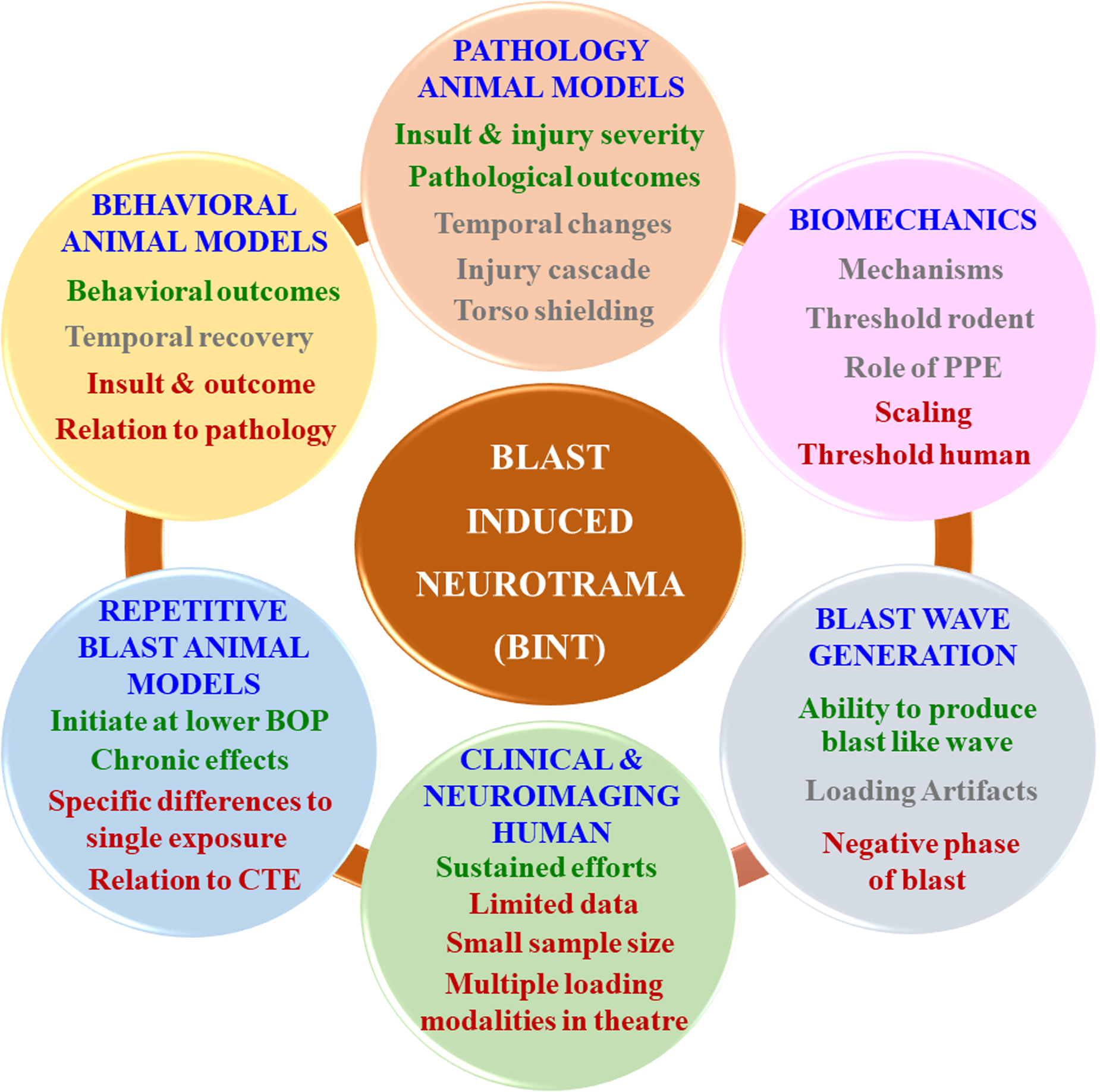
Schematic depicting the current state of knowledge of BINT in various subdomains. The text color for each aspect in individual subdomains is commensurate with the degree of understanding. Green-colored text indicates significant, unequivocal findings in the literature. Gray-colored text indicates that the findings are equivocal. Red-colored text indicates a lack of substantial literature. For example, in the “behavioral animal models” subdomain, unequivocal findings are reported in the literature for various behavioral outcomes observed because of exposure to the blast demonstrating/exuberating consensus across investigations. However, findings regarding temporal recovery of the behavioral outcomes are equivocal (or confounding), indicating a lack of consensus. There is a lack of substantial literature regarding the relationship between external mechanical insult and behavioral outcomes and the correlation between observed pathology and behavioral outcomes. Future work in each subdomain should focus on aspects highlighted using gray- and red-colored texts. BINT, blast-induced neurotrauma; BOP, blast overpressure; CTE, chronic traumatic encephalopathy; PPE, personal protective equipment.
Blast Wave and Its Generation in a Laboratory
An explosion is termed as a rapid expansion of gases, generally occurring because of the detonation of explosives. 7 Explosion creates a pressure pulse in the surrounding medium (e.g., air) that propagates at supersonic speed, generating an (almost) instantaneous rise in pressure known as a shock front. The rarefaction wave, generated because of the overexpansion of explosive gases or reflections from the ground, catches with the shock front, causing a decay in the pressure profile. At a sufficiently longer distance from the explosion, a blast wave takes the form of the Friedlander wave 8 (or so-called free-field blast wave). The free-field blast wave is characterized by an instantaneous rise in pressure followed by a non-linear decay. Broadly, the blast wave implied in BINT is a free-field blast wave referred to as a “primary blast” in BINT literature.
Given that the explosives are restricted, primary blast waves are typically generated in a laboratory using compressed gas-drive shock tubes .9–15 It has been shown, in these investigations, that shock tubes can generate primary blast waves in some form. Significant literature exists on the accurate generation (or lack thereof) of primary blast waves using shock tubes.16–28 The blast wave evolves considerably along the length of the shock tube,17,18,21 based on the design of the shock tube.22,24,25,27 It has also been demonstrated that the blast wave profile and specimen placement location critically affect biomechanical loading16,23,28 and injury outcome. 28 It is difficult to generate similar blast wave profiles across various shock tubes. Thus, it is crucial to report the blast-related measurements rigorously. Further, as many measurements as possible should be made and care should be taken to avoid jet wind (occurring near the exit of the shock tube) and secondary loading effects on the specimen.17,19–21,27
There is a lack of standard protocol for the blast measurements (i.e., incident blast overpressure [BOP], reflected overpressure, and intracranial pressure [ICP]). In the literature, BOP has been measured and reported at varying distances from the specimen, with different types of sensors, and with various data acquisition rates.9,11,13,20,29–32 This has caused a large scatter in reported incident overpressures.9,11,13,20,29–32 As a result, correlating BOP with the biomechanical response (e.g., ICP) and comparing data across various investigations become challenging. 33 We have collected and analyzed the experimental data from the literature across various models (head surrogates, post-mortem human subjects [PMHS], and rat). 33 Our analysis (Supplementary Fig. S1) suggests that, for a given model, a reasonable correlation can be established between reflected overpressure measured on the surface of the head (generally at the point of initial impact location) and ICP measured in the brain. Hence, in addition to the measurement of BOP, it is advisable to measure reflected overpressures at a few locations on the surface of the head.
Biomechanics of Blast-Induced Neurotrauma
Various mechanisms (direct transmission,34–37 skull flexure,38–40 thoracic,3,41,42 head acceleration,43–46 and cavitation,32,47–50) have been proposed for BINT. A large amount of literature (e.g., see previous works51,52 and references therein) exists on the dominance or lack of these mechanisms. Investigations in PMHS53,54 and computational human head models33–35,37,39,55–58 (CHHM) suggest the dominance of head transmission, with skull flexure accounting for high-frequency, low-amplitude oscillations in ICP response 39 and localized pressure gradients.38,40 Thoracic mechanism was one of the earliest candidates 3 of BINT; recent literature59–61 suggests that this mechanism may not be responsible for BINT. Compression of the thorax, however, can cause a sudden blood surge in the brain vasculature, leading to vascular pathology.59,62 Various researchers have observed either ameliorating or insignificant effects on animal pathology because of the shielding of the thorax.63–68 Well-designed protocols across the laboratories are probably needed to address the contribution of the thoracic mechanism to BINT.
Blast-induced head acceleration43–46 was observed as a dominant mechanism in several investigations using rodents. In these investigations, head acceleration alone led to diffuse axonal injury (DAI) pathology and memory deficits. Further, there is evidence of DAI in veterans with histories of blast exposure.69–72 Thus, there is a need to investigate the effect of blast-induced head acceleration and blast DAI pathology using primates, rodents, PMHS, and CHHM, given that it may offer significant new insights. Cerebrospinal fluid (CSF) cavitation was observed in investigations using PMHS 32 and CHHM.48–50 However, additional experimental investigations explicitly focused on CSF cavitation, biomechanical cascade, and BINT are needed to corroborate these findings fully.
Injury thresholds for BINT are in the nascent stages. A few investigations73,74 suggest that incident BOP of ∼145 kPa is a threshold for mild BINT in rodents. Animal-to-human scaling 75 has been attempted; however, robust thresholds for BINT in humans remain unavailable. Biofidelic, three-dimensional CHHM34,35,37,39,55,57,58 may eventually provide threshold values, provided significant clinical data and associated field measurements in humans become available. The use of blast dosimeter 76 to characterize blast exposure in military and law enforcement personnel will be a promising step.
The role of helmets and goggles in blast protection is a critical issue. Numerous investigations suggest that these accessories only marginally mitigate or have an adverse effect under certain situations (e.g., a gap between the head and helmet,40,77–79 with visor and high-density foam pads, 80 with better linking of the helmet with the head, 56 excessive compression of foam pads).81,82 Particularly, negative ICPs and brain strains have been shown to increase with these accessories.56,80 Carefully constructed strategies will be required to address the challenges associated with personal protective equipment (PPE).
Pathology of Blast-Induced Neurotrauma Using Animal Models
The pathology of BINT has been mainly studied using rodent models. Investigations, based on a single exposure, can be broadly categorized into mild (BOP, 85–145 kPa), moderate (BOP, 145–220 kPa), severe (BOP, >220 kPa), and fatal (>300 kPa).83–87 Fatal injuries represent animal mortality.85,88–90 For the BINT, blood–brain barrier (BBB) permeability and oxidative stress have been the most common pathological features. The degree of BBB permeability and oxidative stress depends on the intensity of the blast; these increase with an increase in the BOP.91,92 In cases of severe loading, BBB permeability is acute (i.e., manifesting almost instantaneously) and typically reversible in 3–4 days post-exposure.91,93,94 BBB permeability is subacute for mild loading, manifesting in 3–6 h post-exposure.83,86,92,95,96 BBB permeability is reversible, as evidenced by a significant decrease in extravasation observed beyond 24 h post-exposure.83,86,92,95,96 Numerous studies have noted both the acute and subacute phases for moderate loading.84,97,98 An investigation suggests that BBB permeability is not compromised below a BOP of 70 kPa. 99 The most substantial changes attributable to the BBB and oxidative stress have been observed in the frontal cortex, striatum, thalamus, hippocampus, and amygdala.95,98–102 Apart from these peculiar pathological features, hemorrhage, edema, inflammation, DAI, vasospasm, neural loss, and metabolic changes have been observed for moderate and severe loading.63,67,74,87,103–112 Mild loading mainly manifests neural degeneration, astrocyte and microglial activation, DAI, multi-focal axonal damage, and microvascular damage.43,64,83,101,113–123
Behavioral Outcomes of Blast-Induced Neurotrauma Using Animal Models
Most of the investigations of behavioral deficits are in rodents with BOPs in the range of 70–150 kPa.43,66,90,124–135 Anxiety,101,136–138 auditory127,128,139–142 and recognition memory deficits,43,66,134,143–145 and fear conditioning129,132,133 have been consistently reported behavioral deficits. These deficits were observed between 7 and 30 days post-exposure.43,66,129,132–134,143,144 Spatial working memory and motor deficits have also been reported, especially for animals kept outside the shock tube (and potentially subjected to the jet wind).36,103,125,126,130,131,146
Repetitive Blast-Induced Neurotrauma Using Animal Models
Experimental investigations involving repetitive blast exposure are in the early stages, with most of the studies utilizing rodents exposed to BOPs ranging from 40 to 207 kPa.29,95,121,132,147–162 Reported pathology of repetitive BINT is very similar to the pathology observed during a single exposure.95,121,147–158 Various investigations have observed diverse neuropathological outcomes, including BBB permeability, oxidative stress, chronic vascular impairments, neurodegeneration, neuroinflammation, and axonal damage.95,121,147–158,163 The notable differences of repetitive BINT, with respect to a single exposure, include manifestation of BINT at lower BOPs (≥40 kPa) and chronic effects lasting for several months.29,132,147 Investigations also indicate heightened anxiety, motor impairments, and fear responses during subacute and chronic time points.29,132,159–162,164–166
Clinical and Neuroimaging Investigations in Humans
Clinical studies have been conducted on combat veterans and law enforcement personnel to understand the effects of BINT. Human subjects have been exposed to multiple or repetitive blast.167,168 We could not find any investigation with a single exposure. Such a scenario is probably impractical in the combat theatre. Cerebral edema, intracranial hemorrhage, delayed vasospasm, perivascular damage, skull fracture, tissue damage, and intracranial hypertension have been observed in severely injured patients.88,123,169–171 In recent years, cases of mild BINT have increased.172–175 These studies have reported headaches, sleep disorder, poor motor skills, cognitive dysfunction, depression, and anxiety.176–182 These symptoms appear acutely and continue for months to years. Sustaining a mild BINT increases the likelihood of developing the risk of migraines, post-traumatic stress disorder, and neurocognitive impairment.72,89,167,176,180,183–189 Neuroimaging studies among military personnel have predominantly reported a loss of white matter integrity,71,190–195 and alterations in cortical volume and thickness.196–204 Other observed effects include DAI,69–72 alterations in the thalamic network, 205 small cerebral microhemorrhages, 193 and pituitary impairments. 193
Delineating the effects of a primary blast from blunt and ballistic trauma in humans remains a paramount challenge. 72 Recent investigations in special operations personnel, predominantly exposed to primary blast, are encouraging in this regard.195,206–211 These studies suggest that repetitive, low BOP (range, 7–90 kPa207–209 ) has the potential to cause neurological effects such as tinnitus, sleep imbalance, cognitive impairment, anxiety, insomnia, and headaches.195,208–211 Additional investigations (including longitudinal) in humans with a large sample size are needed to understand the short- and long-term effects of BINT. Such investigations will complement the laboratory studies.
In summary, reasonable progress has been made in each subdomain of BINT as discussed above. Pathology in rodents is most widely understood, followed by behavioral outcomes. Significant literature also exists on BINT mechanisms and the use of shock tubes for simulating primary blast waves. Clinical data in humans, exclusive to blast, are limited. Definitive injury thresholds for BINT in humans remain unavailable. Contemporary investigations of repetitive blast in targeted cohorts, exposed to primary blast, are encouraging. Future interdisciplinary research within and across various subdomains of BINT will probably lead to further progress and tangible outcomes.
Footnotes
Acknowledgments
S.G. acknowledges the funding from the Ordnance Clothing Factory, Avadi, India, and the Defence Metallurgical Research Laboratory, Hyderabad, India. T.S. acknowledges a fellowship from the Ministry of Human Resource Development.
Authors' Contributions
Conceptualization: S.G. Formal analysis: S.G. and T.S. Funding acquisition: S.G. Investigation: S.G. and T.S. Methodology: S.G. and T.S. Resources: S.G. Supervision: S.G. Writing–original draft: S.G. and T.S. Writing–review and editing: S.G. and T.S.
Transparency,Rigor,and Reproducibility Summary
This short communication is not a detailed review of various subdomains (![]() ) of BINT. Rather, it is a broad overview of the present state of knowledge in each subdomain. For a few subdomains, review articles51,52,89,100,146,168,212,213 are already available in the literature. Nonetheless, we have extensively gone through the literature in each subdomain. Relevant literature in each subdomain was considered while writing the manuscript; specifically, publications from 2000 to 2023 were included. Various search terms, such as “blast + rodent,” “blast + human,” “blast + imaging,” “blast + behavior,” “blast + TBI,” “blast + shock tube,” and “blast + biomechanical,” were used. The search items are a few sample search terms only. For blast wave generation and biomechanics subdomains, the corresponding author has been an active researcher for over a decade. Data collected and analyzed during this period were also considered during investigation and writing.
) of BINT. Rather, it is a broad overview of the present state of knowledge in each subdomain. For a few subdomains, review articles51,52,89,100,146,168,212,213 are already available in the literature. Nonetheless, we have extensively gone through the literature in each subdomain. Relevant literature in each subdomain was considered while writing the manuscript; specifically, publications from 2000 to 2023 were included. Various search terms, such as “blast + rodent,” “blast + human,” “blast + imaging,” “blast + behavior,” “blast + TBI,” “blast + shock tube,” and “blast + biomechanical,” were used. The search items are a few sample search terms only. For blast wave generation and biomechanics subdomains, the corresponding author has been an active researcher for over a decade. Data collected and analyzed during this period were also considered during investigation and writing.
Funding Information
This work was supported by the Ordnance Clothing Factory (Avadi, India), grant number 0005CA2122CONT, and the Defence Metallurgical Research Laboratory (Hyderabad, India), grant number: TD/21-22/DMR-327.02. T.S. was supported by a fellowship from the Ministry of Human Resource Development.
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
