Abstract
This systematic review focuses on an increasing subset of traumatic brain injury (TBI) survivors who develop post-traumatic parkinsonism (PTP), characterized by slowness of movement (bradykinesia), rigidity (stiffness), postural instability, and resting tremors caused by obstruction or damage to deep brain structures of the basal ganglia. PTP is rare, and one hypothesis to explain PTP rarity is that TBIs severe enough to affect deep brain structures are often lethal; however, with increasing survivability of TBIs, these numbers are expected to increase. The goal of this review is to raise awareness of an expected global increase of a subgroup of TBI patients who are treatment responsive and report therapeutic results aiding providers in diagnosing, educating, and treating PTP patients. Literature over the past 100 years was considered, and 44,663 peer-reviewed articles were identified. Inclusion criteria required a clinical indication of parkinsonian signs and TBI. Twenty-six case reports were ultimately included from which 36 individual patient data points were extracted for this review. Between 1980 and 2010, there has been an increase in reporting of PTP decade after decade. Forty-seven percent of PTP cases have 1–6 months of latency to symptom onset, and 83% of cases were male. PTP can occur with or without presence of brain lesions, and the most common type of injuries that cause PTP are motor vehicle accidents followed by falls. PTP patients are responsive to surgery or medication treatments. Further detail on PTP symptomology, treatment responsiveness, and injury types is provided.
Introduction
Traumatic brain injury (TBI) remains the most common neurological disorder and a global health concern. 1 TBI is defined by the Centers for Disease Control and Prevention as an injury to brain tissue that affects how the brain works resulting from a bump, jolt to the head, or skull penetrating injury. Data from epidemiological studies published between 2015 and 2020 indicate that the global incidence of TBI ranges from 476 per 100,000 in South Korea 2 to 787 per 100,000 in the United States. 3 In other words, the incidence of TBI is more than double the combined incidence of stroke, Alzheimer's disease, Parkinson's disease (PD), and multiple sclerosis. Factors affecting TBI survival include pre-hospital care, in-hospital care, access to health insurance, and early intervention. 1 Although the most drastic TBI survival improvements are reported in low- to middle-income countries, through improved access to healthcare and improved infrastructure, there exists significant room for improvement among wealthy countries with robust healthcare systems. For example, statewide implementation of pre-hospital TBI guidelines in Arizona doubled the survival among patients with severe TBI and tripled survival among patients with TBI requiring intubation. 4
The new guidelines emphasized improved ventilation and avoidance/treatment of hypotension. Increased TBI survival is followed by years of life with disability. Early and continuous care after a TBI is vital and one of the key takeaways from the 2022 Lancet Neurology Commission on TBI. Access to long-term care and rehabilitation often goes unmet. 5 The commission has also recommended identifying subgroups of TBI patients most likely to be responsive to specific treatments. 1 Identifying these groups can improve timely access to effective care and treatments, which will improve recovery and rehabilitation.
Even under the best of circumstances, outcomes after a TBI remain heterogenous. 6 History of TBI is strongly associated with development of PD years later.7–11 PD is typically diagnosed in the sixth decade of life by motor deficits that include progressive slowness of movement (bradykinesia), rigidity (stiffness), postural instability, freezing gait, and resting tremors, collectively referred to as parkinsonism. However, in rare cases, TBI patients can develop post-traumatic parkinsonism (PTP), sharing many of the signs and symptoms of idiopathic PD, after a TBI event. Although TBIs are common, affecting millions of persons each year, the vast majority of TBIs do not produce PTP. The prevailing hypothesis for the rarity of PTP is that TBIs severe enough to compromise the deep brain structures of basal ganglia responsible for orchestrating fine motor movement are often lethal. Motor deficits resulting from brain damage are a common sequelae after a TBI. 12 Persistent motor weakness resulting specifically from brain lesions after a TBI is also a common finding. 13 Deficits that cause PTP differ in that they are extrapyramidal in nature, with bradykinesia, rigidity, and resting tremors being the hallmark features. PTP has been described as a disorder of consciousness (DOC) given that loss of consciousness (LOC) often occurs with TBIs and as such may be responsive to amantadine hydrochloride, a prodopaminergic drug commonly used to treat other DOCs. 14
More recently, the relationship between TBI and parkinsonism has been the subject of reviews that provide insight and offer suggestions for animal modeling to better understand the underpinning biological relationship.11, 15 As strategies that improve TBI survival around the world continue to be implemented, the prevalence of PTP is also expected to increase, necessitating an increased awareness and better understanding to expedite treatment for this specific subpopulation of TBI patients.
In most cases, PTP is responsive to pharmacological and surgical intervention, but, if left untreated, may progress to permanent disability. PTP has been reported over the past 100 years, with few published retrospective studies. 16 Increasing survival of TBI accompanied by neurological deficits, and increased reporting in recent years of PTP, warrants an examination of our current understanding. To this end, we summarized the available PTP literature (over the past 100 years) with a particular focus on the nature of TBIs most likely to cause PTP, varying latency to development of symptoms, sex differences, as well as duration and severity of symptoms. Treatment options for PTP and considerations for future research efforts are presented.
Methods
Systematic searches were performed using Google Scholar and PubMed search engines with pre-defined query terms (Fig. 1). A total of 44,663 articles were identified as of February 15, 2023. The final titles were selected based on pre-defined inclusion criteria (indication of parkinsonian signs and indication of trauma, either patient reported or diagnosed in imaging). Twenty-six articles were selected from which 36 cases were extracted, and data were manually extracted from these articles and included in Table 1. Case report data from the 26 studies are presented as percentages of total reports rounded to the nearest whole percent. Prism software (version 9.5.1 for mac OS; GraphPad Software Inc., La Jolla, CA) was used for graphing and generating figures. Lesion cases or lesions are defined as hypointense abnormalities, similar to those observed in stroke or in neurodegenerative disorders. Non-lesion cases or non-lesions are defined as transient subdural hematomas (SDHs) or contusions that compress or transiently obstruct brain regions and resolve after decompression surgeries. A detailed clinical PTP case definition preceding our study was not available. Therefore, definition during literature searches was limited to our inclusion and exclusion criteria because of a dearth of peer-reviewed publications. Case definition as a result of this study is detailed in the Discussion section.

Review strategy and data extraction. Thirty-six cases, from 26 studies, were examined and underwent analysis.
Reviewed Case Details and Literature Sources
Data points were extracted from the manuscripts.
DBS, direct brain stimulation; LD, lumbar drain; LOC, loss of consciousness; MVA, motor vehicle accident; N/A, not applicable; NR, not reported; PD, Parkinson's disease; SDH, subdural hematoma; TBI, traumatic brain injury.
Data sharing
This systematic review article includes all the data points and articles from which the data were extracted in Table 1. Data used to generate graphs are available from the corresponding author upon reasonable request.
Results
Post-traumatic parkinsonism reporting increases with increased traumatic brain injury survival
From 1980 to 2010, the total number of PTP cases has been increasing decade after decade, with a dip in 2020 coinciding with COVID-19 (Fig. 2A). Available published data from 2006 to 2015 report an increase in total TBI emergency room visits and a decrease in total TBI mortality (Fig. 2B). From 2007 to 2015, hospitalizations from moderate-to-severe TBIs increased, whereas mortality remained relatively unchanged during the same period (Fig. 2C).
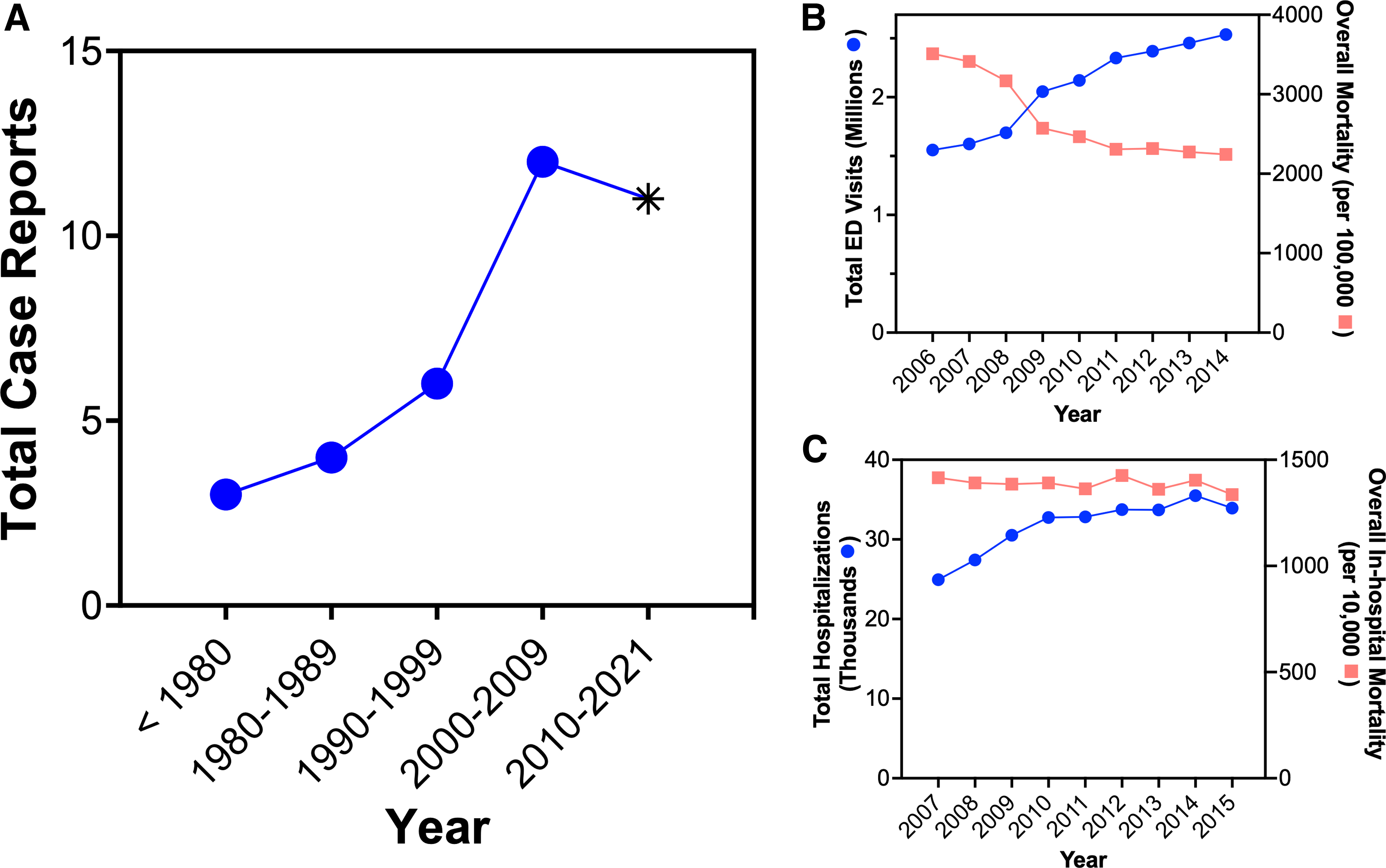
TBI survival and incidence of PTP are increasing. Reporting of PTP in the literature has increased decade after decade from 1980 to 2010. COVID-19 stay-at-home orders issued 23 (
Post-traumatic parkinsonism is most commonly caused by injuries from motor vehicle accidents with loss of consciousness and latency to symptom onset of 1–6 months
Thirty-one percent of PTP cases occurred because of motor vehicle accidents (MVAs), followed by falls which account for 25% of cases (Fig. 3A). Many PTP cases were of unknown trauma cause, although trauma was indicated in 33% (Fig. 3A). LOC was reported in 33% of cases, whereas only 8% of PTP cases reported no LOC (Fig. 3B). Forty-two percent of PTP cases did not report LOC (Fig. 3B). PTP symptom onset is most commonly reported 1–6 months after injury, with 53% of cases occurring with the presence of lesions (Fig. 3C).
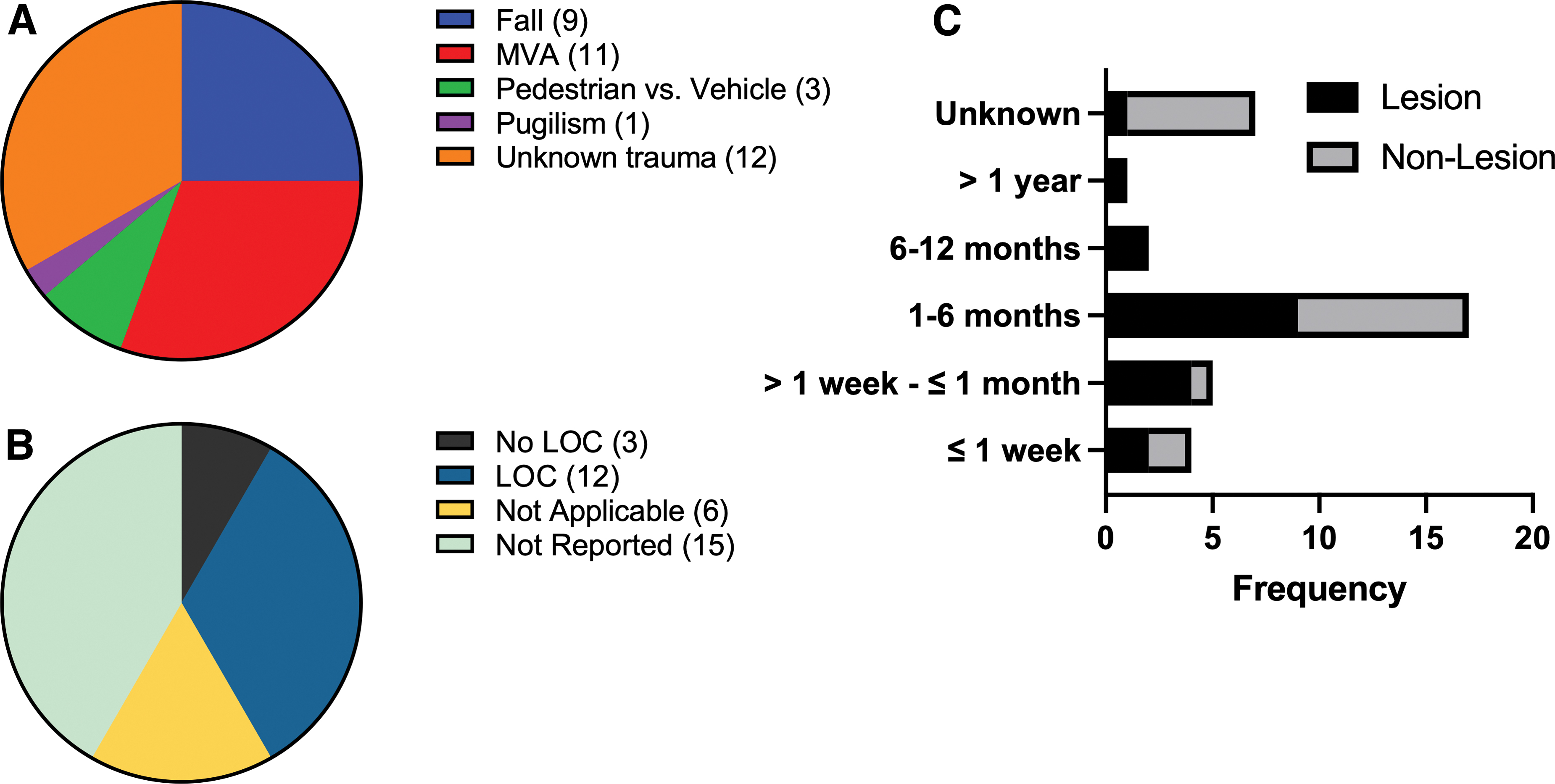
MVAs and falls are the major causes of PTP, with symptom onset most commonly occurring 1–6 months after injury. Unknown TBI (33%), MVA (31%), and falls (25%) are the most common causes of PTP (
Post-traumatic parkinsonism can occur in all age groups, with and without the presence of lesions, and most frequently in male traumatic brain injury patients
Fifty-three percent of PTP cases had a confirmed lesion (Fig. 4E). Fifty-three percent of lesions were localized specifically to the substantia nigra whereas 21% were reported more broadly in the midbrain (Fig. 4D). PTP with lesions occur more frequently in patients under the age of 60 (Fig. 4F). Males are 5 times more likely to develop PTP, regardless of lesion presence or absence (Fig. 4A–C).
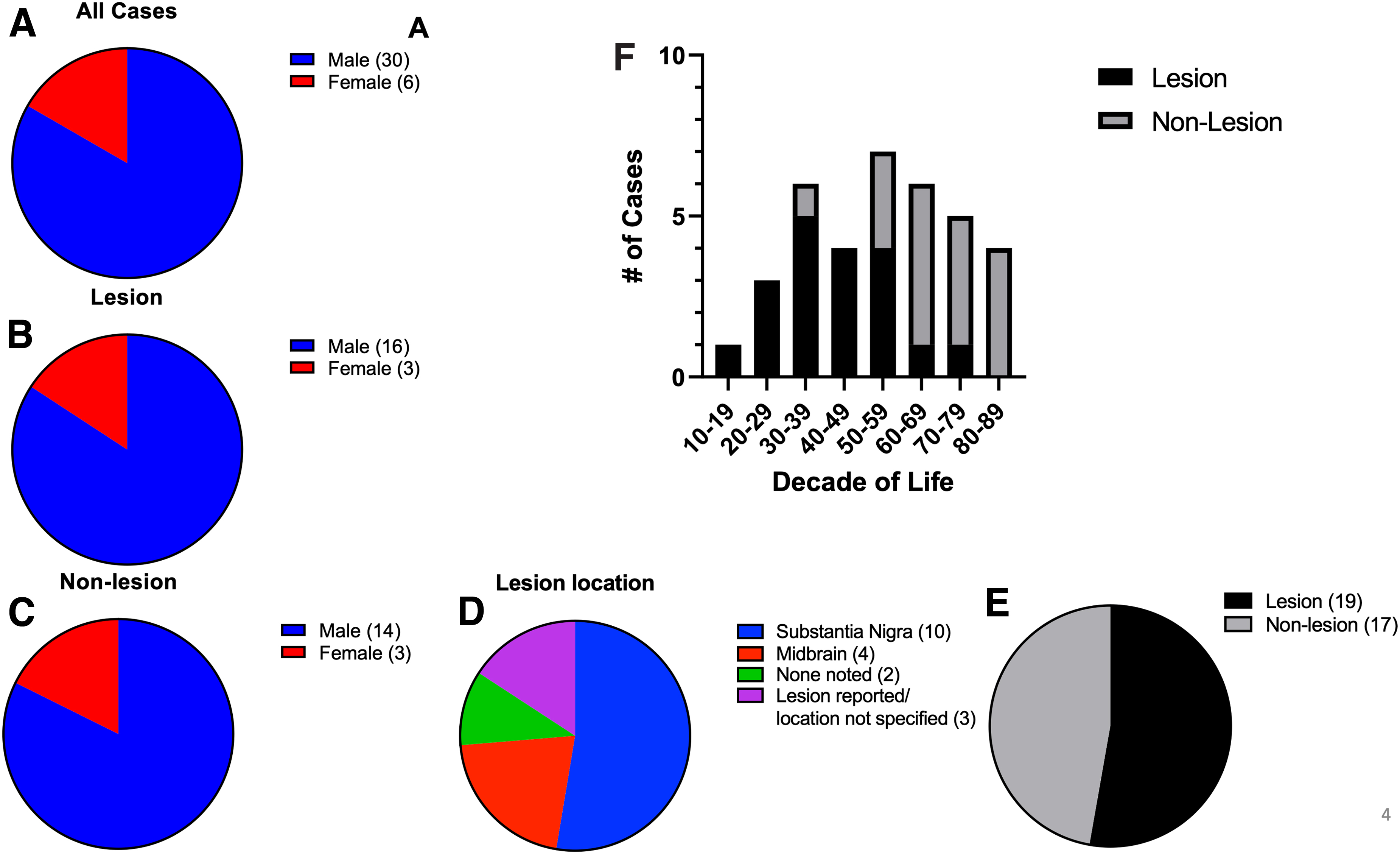
PTP occurs most frequently in males between the ages of 50 and 59, and the diagnosis is accompanied with a report of lesion in the substantia nigra pars compacta. PTP with or without identified brain lesions is reported 5 times more in males than females (
Post-traumatic parkinsonism can have parkinsonian symptomology with good treatment responsiveness
Sixty-eight percent of lesion PTP (Fig. 5A1–A2) and 88% of non-lesion PTP (Fig. 5B1–B2) cases met at least two of the four common parkinsonian diagnostic criteria (bradykinesia, rigidity, tremor, and postural instability). Listed in descending order of sign prevalence for lesion PTP are: tremors 63% (Fig. 5C3); rigidity 58% (Fig. 5C2); bradykinesia 52% (Fig. 5C1); and postural instability 16% (Fig. 5C4). Prevalence of signs for non-lesion PTP in descending order are: rigidity (Fig. 5C2); bradykinesia 59% (Fig. 5C1); tremors 58% (Fig. 5C3); and postural instability 24% (Fig. 5C4). Lesion PTP had mostly unilateral presentation (Fig. 5A3) whereas non-lesion PTP had mostly bilateral presentation (Fig. 5B3). PTP with lesions were responsive to medication treatment (carbidopa/levodopa), with >47% of cases having reported complete, majority, or transient recovery (Fig. 6A,B). Patients with non-lesion PTP were responsive to surgical intervention to alleviate compression caused by SDHs (Fig. 6C). Ninety-four percent of non-lesion PTP patients reported complete resolution of symptoms after decompression surgery (Fig. 6D).
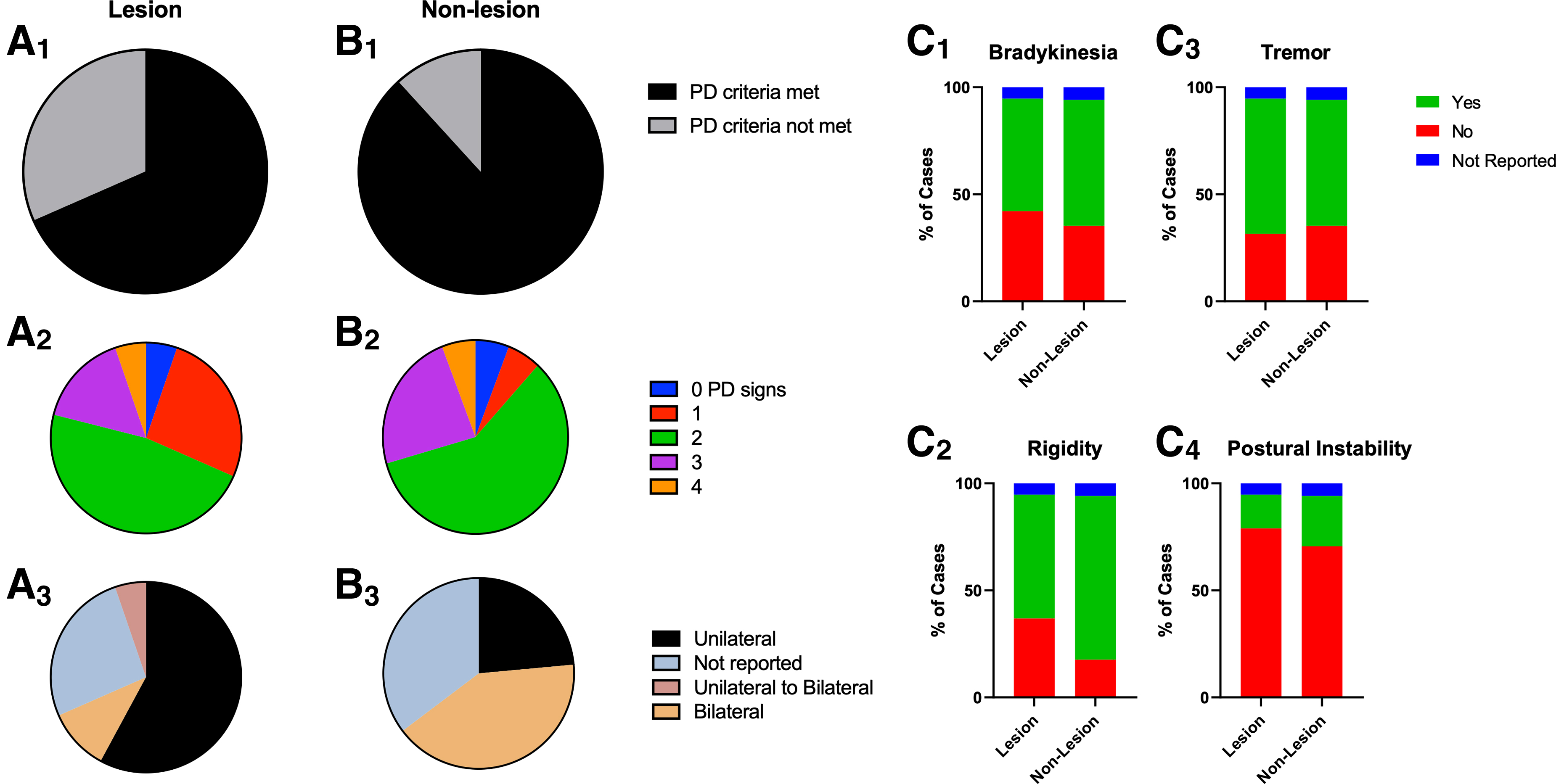
Majority of PTP cases met at least two parkinsonian signs. Sixty-eight percent of PTP cases with an identified lesion met PD diagnostic criteria of at least two parkinsonian signs (

PTP patients are responsive to surgery or medication treatments. Carbidopa/levodopa was the predominant treatment for PTP patients with identified lesion(s) (
Discussion
Impetus for this systematic review comes from an increase in TBI survival and our ongoing studies into the biological relationship between the history of TBI and PD. Investigation of TBI literature indicated that, along with an increase in TBI survival, there is an increase in reports of PTP. Both the number of TBI survivors and persons with consequences after a TBI are expected to increase, and according to the 2022 Lancet Neurology Commission on TBI, identifying TBI subgroups responsive to specific treatment is an effective strategy to address this growing problem. 1 Patients who develop PTP are one such group, sharing presentation and treatment options with parkinsonian disorders. As such, it is important to better understand this group to provide more effective treatments and help design more effective research studies by identifying knowledge gaps that remain.
Increase in post-traumatic parkinsonism reporting coincides with increase in traumatic brain injury survival
The prevailing hypothesis for the rarity of PTP is that injuries severe enough to cause PTP are lethal. It stands to reason that if TBI survival is improved that there would also be an increase in PTP reporting. Indeed, we observe that between 1980 and 2009 there has been a steep increase in PTP reporting (Fig. 2A) coinciding with improvements in healthcare that lead to an increase in TBI survival (Fig. 2B,C). If this trend continues, we expect to observe a continued increase in PTP reporting. The 2022 Lancet Neurology Commission on TBI recommended that identifying TBI subgroups responsive to specific treatment is an effective strategy to help address the growing number of TBI survivors with lifelong disabilities. 1
Improved case definition
Based on the data collected from case reports, a clearer clinical definition of PTP is emerging, as a parkinsonian disorder that most frequently occurs in men (83%), 1–6 months after a TBI event, and having at least two of the four common parkinsonian signs. Bradykinesia, rigidity, and tremors are common in PTP with or without identified lesion(s), whereas postural instability is reported only in 16% of lesion and 24% of non-lesion PTP cases. Though PTP can occur at any age between 10 and 89, most cases were over the age of 29 (Fig. 4F). MVAs were the most common cause of reported PTP cases followed by falls (Fig. 3A). Without definitive biomarkers, a clinical TBI history remains paramount to distinguish PTP from other parkinsonian disorders, particularly among persons over the age of 50, when other parkinsonian disorders are likely to develop. Neuroimaging findings show either an SDH causing transient compressive obstruction of the basal ganglia or hypointense lesions confined to the basal ganglia, similar to what is observed in neurodegenerative diseases. These are referred to as lesion versus non-lesion PTP. SDH PTP is resolved by decompressive surgery, and lesion PTP is responsive to dopamine replacement therapy (DRT).
Some missing details could be filled with more complete patient history. For instance, in 33% of published PTP cases, the specific trauma type was unknown (Fig. 3A). Similarly, state of consciousness (SOC) was not reported in 42% of cases (Fig. 3B). Inclusion of trauma causes and SOC histories in future reporting would significantly improve our understanding of PTP and help us further refine a clinical definition.
Post-traumatic parkinsonism treatments responsiveness and outlook
PTP is responsive to DRT in cases where lesions were detected (Fig. 6A,B), whereas decompression surgeries were effective in cases where hematomas were present (Fig. 6C,D). Forty-two percent of lesion cases had complete or majority resolution of symptoms (Fig. 6B) whereas lesion cases that required decompression surgery had 94% complete and 6% had majority symptom resolution (Fig. 6D). We delineate between PTP cases that have detectable lesions on neuroimaging and those with transient hematomas as structural abnormalities, which we refer to as non-lesion. Lesions were not reported in cases where hematomas were detected and managed, resulting in resolution or improvement of symptoms.
It is important to recognize that in order to develop future targeted therapeutics for PTP, better reporting of long-term PTP outcomes from current treatments (e.g., surgery and DRT) needs to be implemented. To emphasize, surgical intervention had better, 100% reporting overall of complete or major symptom resolution (Fig. 6C,D); by comparison, 53% of lesion cases reported complete, major, transient, or no resolution (Fig. 6B). None of the published cases report long-term follow-up to determine long-term patient outcomes. A fundamental question remains unknown, whether parkinsonism in treatment-responsive patients resolves or worsens with time. Moreover, it remains unknown whether patients placed on medication(s) remain on symptomatic treatment indefinitely.
Future research direction
It remains unknown whether patients responsive to treatment continued the treatment indefinitely or whether clinical benefits persisted after treatment cessation. In other words, it is unknown whether post-PTP is progressive or static. Additionally, it remains unclear whether PTP occurred during the prodromal period of PD pathology already present in the patient. TBIs, resulting mostly from falls, increase in patients before their PD diagnosis, likely attributable to undiagnosed motor deficits. 17 Future studies with the aid of effective biomarkers will be required to better understand this dynamic. Because of the clear link to compromised basal ganglia, PTP patients are of particular interest to determine whether they are at a higher risk for developing PD later in life. PTP is delayed from the point of injury by up to a year, and maybe even longer, which impedes diagnosis and treatment. PTP is responsive to PD treatments and does not follow rapid progression like other more aggressive parkinsonian disorders. In the absence of definitive diagnostic biomarkers in living patients, many cases of PD linked to TBI may be misdiagnosed as PD instead of PTP. Identifying additional cases of PTP could present an opportunity to study the effectiveness of drugs to slow or stop PTP progression or development of PD after PTP. Currently, these drugs are tested in persons who have developed PD. Alpha synuclein (α-Syn) amplification assay technology has been shown to detect PD biomarkers in the cerebrospinal fluid of PD patients.18,19
More recently, these assays have been used to differentiate between leucine-rich repeat kinase-2 variant and idiopathic PD. 20 α-Syn amplification assays have the potential to distinguishing between PTP and idiopathic PD, particularly in patients >60.
One theme confirmed in this review involves the diversity of responses to TBI. Genetic variants could account for many differences in post-injury neurodegeneration. Many genes have already been connected directly to parkinsonian disorders. Variants within these and other genes could help predict potential post-injury outcomes 21 and identify therapeutic targets to explore. It remains unclear whether pre-clinical models accurately predict the mechanism of PD progression after PTP. However, if true, those developed therapeutics may prove useful for patients long term and therefore is an important area of continued research to prevent future disability in PTP patients. We have found in pre-clinical rat models of α-Syn fibril-induced PD that pTau is localized in Lewy bodies (LBs) within dopaminergic neurons. 22 Similarly, we also found pTau aggregates within dopaminergic neurons of rats that underwent repetitive mild TBI. 22 If PTP induces pTau aggregation in surviving dopaminergic neurons, it could predispose cells to development of PD-specific proteinopathy by cross-seeding aggregation of α-Syn into LBs and Lewy neurites. Similarly, pTau-targeted therapeutics developed for Alzheimer's disease could potentially be used to limit or slow the progression of PTP to PD.
Limitations
Although PTP case reports have increased decade after decade, the number reported in the literature is still small, totaling 36 reported cases from 26 studies. Some cases of PTP that develop after 1 year, particularly in patients >60, may be misdiagnosed as PD, thereby limiting the numbers of PTP cases. Long-term follow-up of PTP cases beyond 1 year can further delineate between PTP and PD in older persons, but is absent from the literature. The majority of PTP case reports did not specify the cause of trauma.
Conclusion
Increase in PTP reporting decade after decade is likely a result of increased TBI incidence and increased TBI survival. These numbers are expected to rise. PTP is responsive to either DRTs or surgeries depending on whether PTP is a result of a lesion or from an SDH. Males are 5 times more likely to develop PTP, and the most common injury type that causes PTP is an MVA with LOC. PTP occurs most commonly after a 1- to 6-month latency period. More comprehensive reporting and long-term follow-up studies are needed to determine whether PTP is static or progressive.
Footnotes
Acknowledgments
We acknowledge Dr. Kevin Beck for useful discussion and Dr. Walter Husar for mentoring resident trainees who contributed significantly to this work.
The opinions, interpretations, conclusions, and recommendations are those of the authors and are not necessarily endorsed by the Department of Defense, and the contents do not represent the views of the Department of Veterans Affairs or the U.S. government.
Authors' Contributions
C.R.: execution, analysis, writing, and editing. G.R.A., E.F.: writing and editing. M.G.: writing and editing. B.A.C.: writing and editing. V.D.: conceptual design, execution, analysis, writing, and editing.
Transparency,Rigor,and Reproducibility Summary
This systematic review was designed to summarize clinical reports of post-traumatic parkinsonism that occurs after a traumatic brain injury. Systematic searches were performed using Google Scholar and PubMed search engines with pre-defined query terms (Fig. 1). A total of 44,663 manuscripts were identified as of February 15, 2023. The final titles were selected based on pre-defined inclusion criteria (indication of parkinsonian signs and symptoms, and indication of trauma either patient reported or diagnosed in imaging). Twenty-six manuscripts were selected from which 36 cases were extracted, and data were manually extracted from these manuscripts and included in ![]() . Case report data from the 26 studies are presented as percentages of total reports rounded to the nearest whole percent. Prism version 9.5.1 for mac OS was used for graphing and generating figures. Inclusion criteria were not pre-registered. Any ambiguity about article inclusion was discussed among authors, and agreement was reached before inclusion.
. Case report data from the 26 studies are presented as percentages of total reports rounded to the nearest whole percent. Prism version 9.5.1 for mac OS was used for graphing and generating figures. Inclusion criteria were not pre-registered. Any ambiguity about article inclusion was discussed among authors, and agreement was reached before inclusion.
Funding Information
The Department of Veterans Affairs (Veterans Health Administration, Office of Research and Development), Biomedical Laboratory (I01BX001515) and Rehabilitation (IK2RX003253) Research and Development. Dr. Citron is the recipient of a Research Career Scientist award (no. IK6BX006188) from the Department of Veterans Affairs.
Author Disclosure Statement
No competing financial interests exist.
