Abstract
Traumatic brain injury (TBI) in children often leads to poor developmental outcomes attributable to progressive cell loss caused by secondary injuries, including endoplasmic reticulum (ER) stress. Buprenorphine (BPN) is commonly used in children for pain management; however, the effects of BPN on ER stress in the pediatric population are still inconclusive. This study investigated the sex-specific effects of BPN on ER stress, abnormal protein accumulation, and cell loss in a mouse impact acceleration model of pediatric TBI. On post-natal day 20–21 (P20–21), male and female littermates were randomized into sham, TBI + saline and TBI + BPN groups. BPN (0.075 mg/kg) was administered to TBI + BPN mice at 30 min after injury and then every 6–12 h for 2 days. The impact of BPN was evaluated at 1, 3, and 7 days post-injury. We found that TBI induced more prominent ER stress pathway activation at 1 and 3 days post-injury in males, compared to females, whereas abnormal protein accumulation and cell loss were more severe in females at 7 days post-injury, compared with males. Although BPN partially ameliorated abnormal protein accumulation and cell loss in both males and females, BPN only decreased ER stress pathway activation in males, not in females. In conclusion, BPN exhibits sex-specific effects on ER stress, abnormal protein accumulation, and cell loss in a time-dependent manner at the acute phase after pediatric TBI, which provides the rationale to assess the potential effects of BPN on long-term outcomes after pediatric TBI in both males and females.
Introduction
Traumatic brain injury (TBI) remains a leading cause of morbidity and mortality in children and poses major concerns for public health.1,2 Infants and toddlers (0–4 years of age) are at high risk of poor developmental outcomes, such as impaired communication and problem solving, 3 even after mild TBI. 4 TBI induces endoplasmic reticulum (ER) stress,5–12 which contributes to neuronal loss, 13 abnormal protein accumulation,6,14 tauopathy, and cognitive deficits. 15 Upon ER stress, unfolded protein response (UPR) is activated to restore ER homeostasis.16,17 UPR consists of three major signaling cascades: inositol requiring enzyme 1 (IRE1), activating transcription factor 6 (ATF6), and double-stranded RNA-activated protein kinase-like endoplasmic reticulum kinase (PERK).18,19 IRE1 activation induces the unconventional splicing of transcription factor Xbox binding protein 1 (XBP1) to XBP1s (spliced X-box binding protein 1), which increases protein folding and ER biogenesis to restore ER homeostasis.16,17,20–22 PERK activation leads to phosphorylation of eukaryotic translation initiation factor-2α (eIF2α) and upregulation of activation transcription factor 4 (ATF4), which lead to greater ER protein folding capacity.23,24 ATF6 promotes protein folding and ER-associated degradation. 20
Although the primary function of UPR is to restore ER homeostasis, unmitigated persistent UPR activation leads to abnormal protein accumulation (e.g. beta-amyloid [Aβ] and phospho-Tau [p-tau])25–28 and cell death by the IRE1/TRAF2 (activation stimulates tumor necrosis factor receptor-associated factor 2)/ASK1 (apoptosis signal-regulating kinase) 29 and PERK/CHOP (C/EBP-homologous protein) pathways. 30
Buprenorphine (BPN), a synthetic opioid, has been used for pain management among children and adults with TBI 31 and in pre-clinical animal TBI models. 32 BPN is a partial agonist of μ-opioid receptors (MORs), and an antagonist of δ-opioid receptors and κ-opioid receptors (KORs). 31 Studies have also shown that opioids can inhibit the innate and adaptive immune systems 33 and exert protective effects in response to cytotoxic insults by reducing calcium overload and suppressing ER stress sensors.34,35 BPN treatments can limit chemokine (C-C motif) ligand 2–mediated monocyte transmigration 36 and alter microglial and astrocytes mediated neuroinflammation after TBI. 37 Our previous study indicates that BPN normalizes MOR expression in white matter astrocytes, improves sensorimotor function, and decreases oxidative stress after pediatric TBI. 38 To date, no study has investigated the sex-specific effects of BPN on ER stress, abnormal protein accumulation, and cell death after pediatric TBI. Here, we investigated the sex-specific effects of BPN on ER stress pathways, Aβ and tau accumulation, and cell loss after pediatric TBI in both males and females.
Methods
Animals
Male (M) and female (F) C57BL/6 mice (2–3 months of age; The Jackson Laboratory, Bar Harbor, ME) were in-house bred. All pups were delivered naturally and remained with their mother after birth until weaning. All animals (2–5 mice per cage) were housed under standard housing conditions (20°C–22°C, 40–60% relative humidity, and a 12-h light/dark cycle) with free access to food and water. All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Michigan.
Impact acceleration model of traumatic brain injury and buprenorphine administration
On post-natal days 20–21 (P20-21, equivalent to 2–3 years of age in humans), 39 animals from the same litter were randomized into sham (n = 64; 31M/33F), TBI + saline (n = 74; 37M/37F), and TBI + BPN (n = 73; 34M/39F) groups. Animals in the TBI + saline and TBI + BPN groups underwent TBI as previously described. 38 BPN was diluted in 0.9% NaCl (sterile) to a concentration of 0.01 mg/mL. The TBI + BPN group received intraperitoneal injections of BPN (0.075 mg/kg) at 30 min after injury and then every 6–12 h for 2 days as previously published. 38 The TBI + saline group received the same volume of saline at 30 min after surgery and then every 6–12 h for 2 days. The sham group did not receive any intervention. All animals were closely monitored as per IACUC guidelines.
RNA isolation and quantitative real-time polymerase chain reaction
mRNA expression of IRE-1α, XPB1s, PERK, ATF6, eIF2α, ATF4, TRAF2, ASK1, and CHOP were measured at the site of injury at 1 day (sham: n = 13, 6M/7F; TBI + saline: n = 12, 6M/6F; TBI + BPN: n = 13, 6M/7F), 3 days (sham: n = 10, 5M/5F; TBI + saline: n = 18, 8M/10F; TBI + BPN: n = 16, 8M/8F), and 7 days (sham: n = 11, 5M/6F; TBI + saline: n = 14, 8M/6F; TBI + BPN: n = 14, 5M/9F) post-injury. Brain tissues (approximately between bregma +2 mm and bregma −1 mm) were microdissected for RNA isolation as previously described. 38 Total RNA was extracted using TRIzol (Sigma-Aldrich, St. Louis, MO) and were quantified using the Nanodrop ND-2000 Spectrophotometer (ThermoFisherScientific, Waltham, MA). Single-stranded complementary DNA (cDNA) was reverse transcribed from RNA using the High-Capacity cDNA Reverse Transcription Kit with RNase inhibitor (ThermoFisherScientific). Primers were custom designed (Table 1) and ordered from Integrated DNA Technology (Coralville, IA). Quantitative real-time polymerase chain reaction (qPCR) was performed with iTaq(tm) Universal SYBR(R) Green Supermix (Bio-Rad Laboratories, Hercules, CA), and the comparative threshold cycle method was used to assess differential gene expressions. 38
Primers for qPCR
qPCR, quantitative real-time polymerase chain reaction.
Immunohistochemistry
Brains were post-fixed in 10% formalin for 48 h and cryoprotected in 30% sucrose (in phosphate-buffered saline [PBS]). Coronal sections (20 μm, 1:6 series) were prepared on a cryostat (Leica Microsystems, Buffalo Grove, IL). For double labeling of CHOP and p-tau, brain sections were incubated overnight at 4°C with mouse anti-CHOP (growth arrest- and DNA damage-inducible gene 153; 1:250, GeneTex, Inc., Irvine, CA) and rabbit anti-p-tau (1:250; ThermoFisherScientific). For double labeling of Aβ and Fluoro-Jade C (FJC),40,41 brain sections were washed three times in PBS for 5 min each, incubated in 0.001% FJC for 10 min, washed six times in PBS for 15 min each, and incubated with rabbit anti-Aβ (1:250; ThermoFisherScientific) overnight at 4°C. After wash, brain sections were incubated with fluorescent secondary antibodies (1:250; Life Technologies, Carlsbad, CA) for 2 h at room temperature. Slides were dried and cover-slipped with fluorescent mounting medium with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich).
Histology quantification
Histological quantification was performed at 1 day (sham: n = 10, 5M/5F; TBI + saline: n = 10, 5M/5F; TBI + BPN: n = 10, 5M/5F), 3 days (sham: n = 10, 5M/5F; TBI + saline: n = 10, 5M/5F; TBI + BPN: n = 10, 5M/5F), and 7 days (sham: n = 10, 5M/5F; TBI + saline: n = 10, 5M/5F; TBI + BPN: n = 10, 5M/5F) post-injury. Images were acquired using a Nikon Eclipse TS2R fluorescent microscope (Nikon Inc., Melville, NY) with same camera settings. All slides and images were coded, and the analysis was performed with personnel blinded to the experiments. Images (40 × , five images per animal) were randomly acquired from the injured cortex (or the matching area in the sham). Expression and distribution of CHOP, p-tau, Aβ, and FJC were evaluated as the percentage of area using particle analysis function in Fiji ImageJ software (National Institutes of Health, Bethesda, MD) as previously described.28,38,42 Percentages of CHOP+p-Tau+ and FJC+Aβ+ were measured using Fiji ImageJ (National Institutes of Health) as previously described.28,38,42 Percentages of CHOP+p-Tau+ and FJ-C+Aβ+ cells were calculated as follows:

Statistical analysis
Data were analyzed using GraphPad Prism 6 (Version 6.04; CA, USA). All data are presented as mean ± standard error of the mean. The D'agostino and Pearson omnibus normality test was used for normality measurement. The Student's t-test (two-tailed) or Mann-Whitney U test (two-tailed) were used for two-group comparisons. One-way analysis of variance and Bonferroni's post hoc tests were used for multiple group comparisons. Statistical significance was set at p < 0.05 for all analyses.
Results
Sex-specific effects of buprenorphine on endoplasmic reticulum stress pathways
We first compared the messenger RNA (mRNA) expression of IRE-1α, XPB1s, PERK, and ATF6 between male and female TBI + saline mice. We found that XBP1s significantly decreased at 1 day (p = 0.03) and 7 days (p = 0.01; Fig. 1B1) and ATF6 significantly decreased at 3 days post-injury (p = 0.0001) in female TBI + saline mice (Fig. 1D1). There was no difference in IRE-1α and PERK expressions (Fig. 1A1,C1). In males, IRE-1α significantly increased in TBI + saline at 1 day post-injury (F = 4.3, p = 0.03; vs. sham and TBI + BPN) and significantly increased in both the TBI + saline and TBI + BPN groups at 3 days (F = 7.7, p = 0.004; vs. sham) and 7 days (F = 9.9, p = 0.002; vs. sham) post-injury (Fig. 1A2). XPB1s significantly increased in TBI + saline at 1 day post-injury (F = 4.6, p = 0.03; vs. sham) and significantly increased in TBI + BPN at 3 days (F = 5.5, p = 0.01; vs. sham) and 7 days post-injury (F = 15.5, p = 0.0002; vs. sham and TBI + saline; Fig. 1B2). PERK significantly increased in TBI + saline at 1 day (F = 5.7, p = 0.01; vs. sham and TBI + BPN) and 3 days post-injury (F = 3.7, p = 0.04; vs. sham) and significantly increased in TBI + BPN at 7 days post-injury (F = 10.6, p = 0.001; vs. sham; Fig. 1C2). There was no change in ATF6 expression (Fig. 1D2).

mRNA expression of IRE-1α, XPB1s, PERK, and ATF6 in the injured brain regions at 1, 3, and 7 days post-injury. (
In females, IRE-1α significantly increased in TBI + saline at 1 day post-injury (F = 6.3, p = 0.04; vs. sham) and significantly increased in the TBI + BPN at 7 days post-injury (F = 5.9, p = 0.01; vs. sham; Fig. 1A3). XPB1s significantly increased in TBI + BPN at 7 days post-injury (F = 11.5, p = 0.0006; vs. sham and TBI + saline; Fig. 1B3). PERK significantly increased in TBI + saline at 1 day post-injury (F = 4.7, p = 0.02; vs. sham; Fig. 1C3). ATF6 significantly decreased in TBI + saline at 3 days post-injury (F = 11.0, p = 0.0006; vs. sham; Fig. 1D3).
We next investigated the mRNA expression of eIF2α, AFT4, TRAF2, ASK1, and CHOP between male and female TBI + saline mice. We found that eIF2α significantly increased in female TBI + saline mice at 1 day post-injury (p = 0.04; Fig. 2A1). ATF4 significantly decreased in female TBI + saline mice at 3 days post-injury (p = 0.04; Fig. 2B1). TRAF2 significantly decreased in female TBI + saline mice at 7 days post-injury (p = 0.007; Fig. 2C1). There was no significant difference in ASK1 (Fig. 2D1). CHOP significantly decreased in female TBI + saline mice at 3 days post-injury (p = 0.002; Fig. 2E1). In males, there was no significant difference in eIF2α expression (Fig. 2A2). ATF4 significantly decreased in TBI + saline at 7 days post-injury (F = 5.9, p = 0.01; vs. sham; Fig. 2B2). TRAF2 significantly increased in both TBI + saline and TBI + BPN at 7 days post-injury (F = 8.6, p = 0.003; vs. sham; Fig. 2C2). ASK1 significantly decreased in TBI + saline and TBI + BPN at 1 day (F = 19.9, p < 0.0001; vs. sham) and 7 days post-injury (F = 9.8, p = 0.002; vs. sham; Fig. 2D2). CHOP significantly increased in TBI + saline at 1 day post-injury (F = 11.1, p = 0.004; vs. sham and TBI + BPN) and significantly increased in both the TBI + saline and TBI + BPN groups at 3 days (F = 4.2, p = 0.03; vs. sham) and 7 days (F = 15.5, p = 0.0002; vs. sham) post-injury (Fig. 2E2).
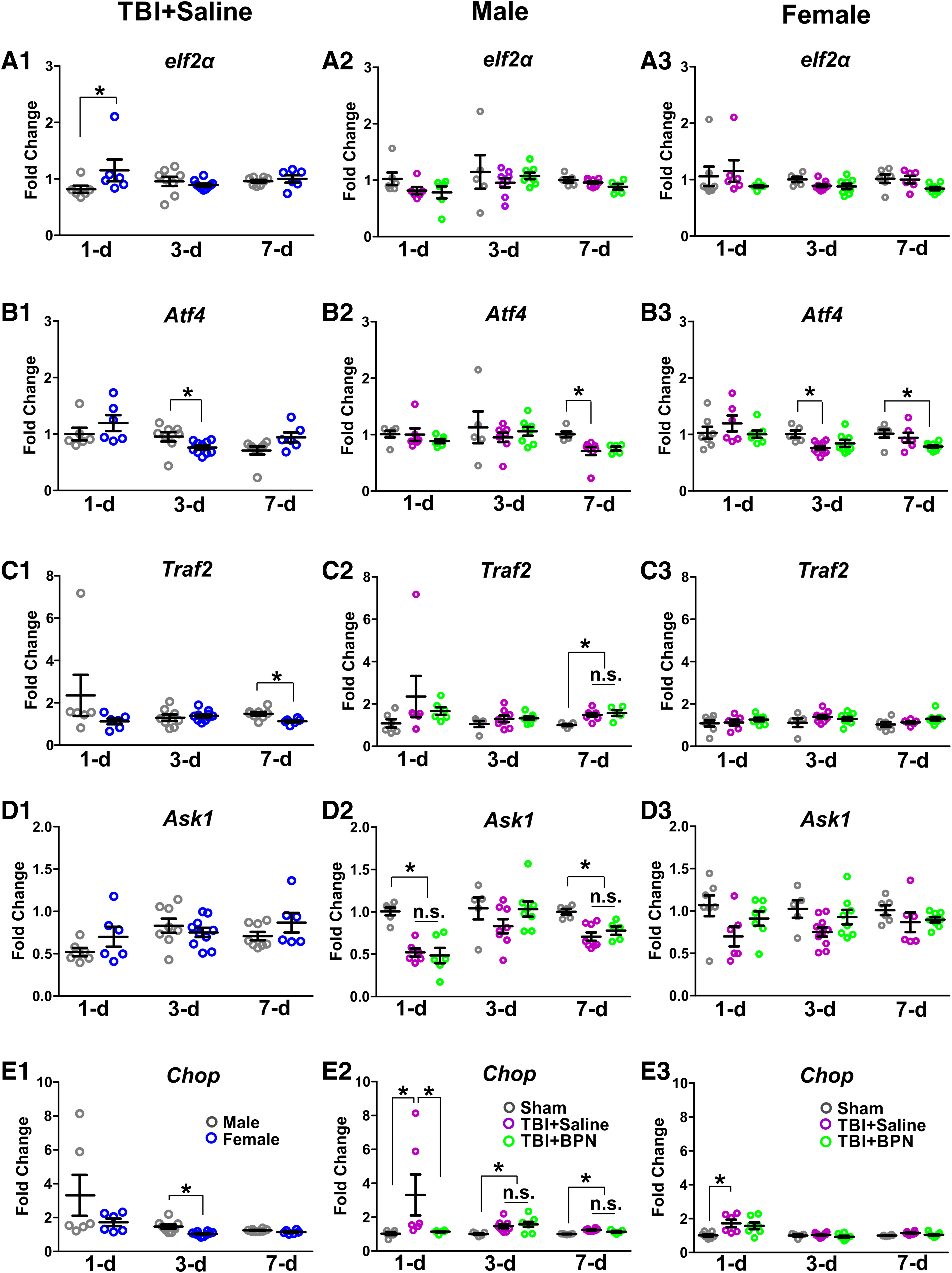
mRNA expression of eIF2α, ATF4, TRAF2, ASK1, and CHOP in the injured brain regions at 1, 3, and 7 days post-injury. (
In females, ATF4 significantly decreased in TBI + saline at 3 days post-injury (F = 5.1, p = 0.02; vs. sham) and in TBI + BPN at 7 days post-injury (F = 3.9, p = 0.04; vs. sham; Fig. 2B3). CHOP significantly increased in TBI + saline at 1 day post-injury (F = 4.8, p = 0.02; vs. sham; Fig. 2E3). There was no significant difference in eIF2α, TRAF2, and ASK1 expressions (Fig. 2A3,C3,D3).
Sex-specific effects of buprenorphine on p-tau
We first compared the expression of CHOP and p-tau, and the percentage of CHOP+p-Tau+ cells between male and female TBI + saline mice. There was no significant difference in CHOP and p-tau expressions (Fig. 3 and Fig. 5A1, B1). Percentage of CHOP+p-Tau+ significantly decreased at 1 day post-injury (p = 0.04) and significantly increased at 3 days post-injury (p = 0.02) in female TBI + saline mice (Fig. 3 and Fig. 5C1). In males, CHOP significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 138.0, p < 0.0001), 3 days (F = 150.0, p < 0.0001), and 7 days (F = 80.9, p < 0.0001) post-injury, compared with sham (Fig. 3 and Fig. 5A2). p-tau significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 61.3, p < 0.0001), 3 days (F = 58.4, p < 0.0001), and 7 days (F = 82.0, p < 0.0001) post-injury, compared with sham. Moreover, p-tau significantly increased in TBI + saline at 1 and 3 days post-injury, compared with TBI + BPN (p < 0.05; Fig. 3 and Fig. 5B2).

Expression of CHOP and p-tau and the percentage of colocalization of CHOP and p-tau at the cortex of injured brain regions at 1, 3, and 7 days post-injury. (
The percentage of CHOP+p-Tau+ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 38.3, p < 0.0001), 3 days (F = 46.1, p < 0.0001), and 7 days (F = 178.5, p < 0.0001) post-injury, compared with sham. Moreover, the percentage of CHOP+p-Tau+ significantly increased in TBI + saline at 1 day (p < 0.05) post-injury, compared with TBI + BPN (Fig. 3 and Fig. 5C2). In females, CHOP significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 124.2, p < 0.0001), 3 days (F = 75.5, p < 0.0001), and 7 days (F = 43.1, p < 0.0001) post-injury, compared with sham (Fig. 3 and Fig. 5A3). p-tau significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 31.1, p < 0.0001), 3 days (F = 99.5, p < 0.0001), and 7 days (F = 73.2, p < 0.0001) post-injury, compared with sham. Moreover, p-tau significantly increased in TBI + saline at 1, 3, and 7 days post-injury, compared with TBI + BPN (p < 0.05; Fig. 3 and Fig. 5B3). The percentage of CHOP+p-Tau+ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 39.0, p < 0.0001), 3 days (F = 57.7, p < 0.0001), and 7 days (F = 101.7, p < 0.0001) post-injury, compared with sham. Moreover, the percentage of CHOP+p-Tau+ significantly increased in TBI + saline at 1, 3, and 7 days post-injury, compared with TBI + BPN (p < 0.05; Fig. 3 and Fig. 5C3).
Sex-specific effects of buprenorphine on amyloid-beta accumulation and cell loss
We first compared the expression of FJC and Aβ and the percentage of FJC+Aβ+ cells between male and female TBI + saline mice. FJC significantly decreased at 3 days post injury (p = 0.01), but significantly increased at 7 days post-injury (p = 0.003) in female TBI + saline mice (Fig. 4 and Fig. 5D1). Aβ significantly increased at 7 days post-injury (p = 0.03) in female TBI + saline mice (Fig. 4 and Fig. 5E1). The percentage of FJC+Aβ+ significantly decreased at 1 day post-injury (p = 0.0002) in female TBI + saline mice (Fig. 4 and Fig. 5F1). In males, FJC significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 62.9, p < 0.0001), 3 days (F = 122.3.0, p < 0.0001), and 7 days (F = 121.5, p < 0.0001) post-injury, compared with sham. Moreover, FJC significantly increased at 1 and 3 days post-injury in TBI + saline, compared with TBI + BPN (p < 0.05; Fig. 4 and Fig. 5D2).

Expression of FJC and Aβ and the percentage of colocalization of FJC and Aβ at the cortex of injured brain regions at 1, 3, and 7 days post-injury. (
Aβ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 38.0, p < 0.0001), 3 days (F = 118.9, p < 0.0001), and 7 days (F = 48.0, p < 0.0001) post-injury, compared with sham. Moreover, Aβ significantly increased in TBI + saline at 3 days post-injury (p < 0.05), compared with TBI + BPN (Fig. 4 and Fig. 5E2). The percentage of FJC+Aβ+ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 343.3, p < 0.0001), 3 days (F = 1303.0, p < 0.0001), and 7 days (F = 2868.0, p < 0.0001) post-injury, compared with sham (Fig. 4 and Fig. 5F2).
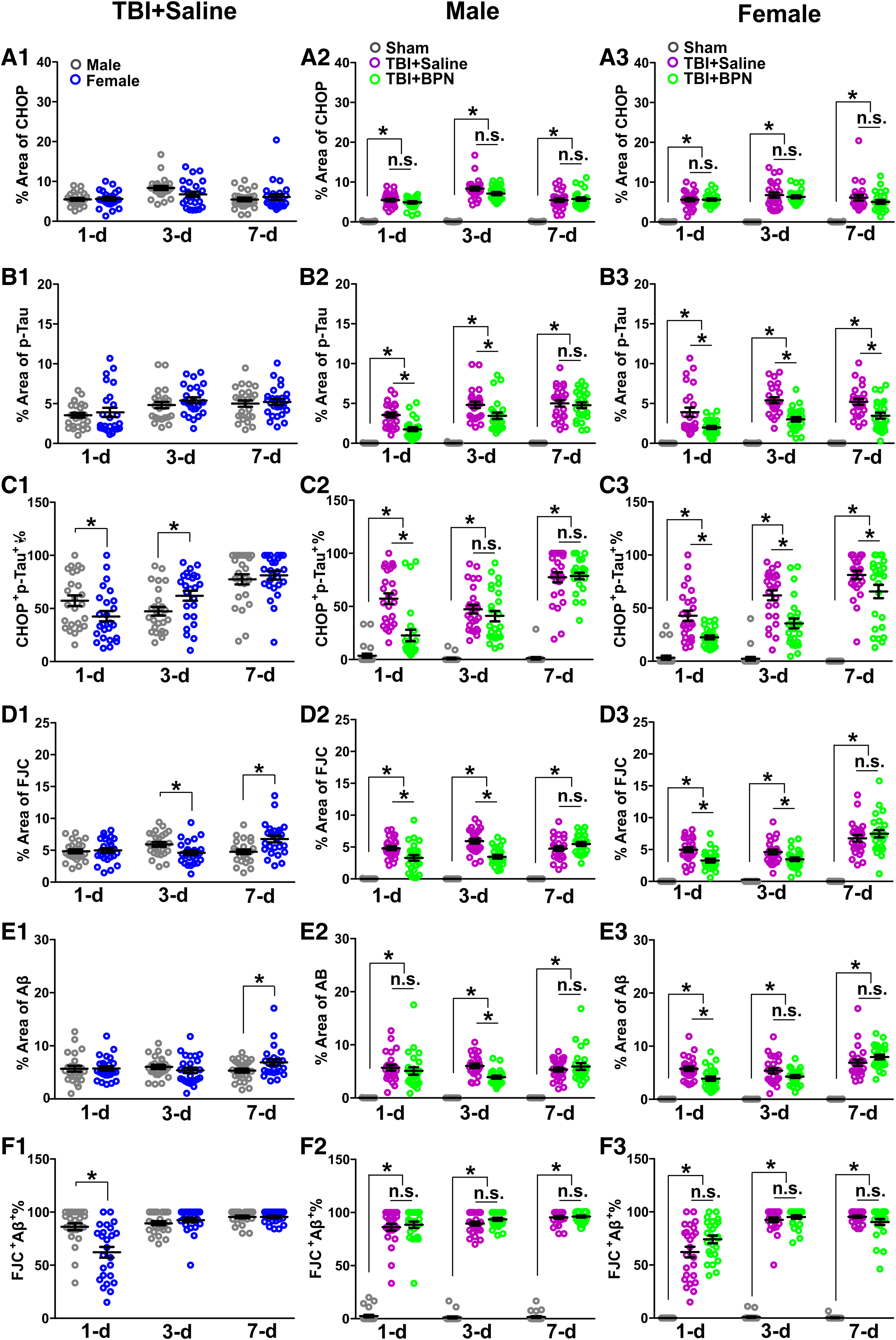
Expression of CHOP and p-tau and the percentage of CHOP+p-Tau+ cells (
In females, FJC significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 84.1, p < 0.0001), 3 days (F = 82.7.0, p < 0.0001), and 7 days (F = 83.6, p < 0.0001) post-injury, compared with sham. Moreover, FJC significantly increased in TBI + saline at 1 and 3 days post-injury, compared with TBI + BPN (p < 0.05; Fig. 4 and Fig. 5D3). Aβ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 74.1, p < 0.0001), 3 days (F = 71.1, p < 0.0001), and 7 days (F = 106.4, p < 0.0001) post-injury, compared with sham. Moreover, Aβ significantly increased in TBI + saline at 1 day post-injury (p < 0.05), compared with TBI + BPN (Fig. 4 and Fig. 5E3). The percentage of FJC+Aβ+ significantly increased in both TBI + saline and TBI + BPN at 1 day (F = 126.4, p < 0.0001), 3 days (F = 1085, p < 0.0001), and 7 days (F = 937.9, p < 0.0001) post-injury, compared with sham (Fig.4 and Fig. 5F3).
Discussion
Evidence has shown that ER stress responses are different between male and female.43,44 In the present study, we demonstrated that both sexes showed a significant upregulation of ER stress genes; however, the magnitude of gene upregulation was different between males and females in a time-dependent manner. For example, CHOP significantly decreased at 3 days and eIF2α significantly increased at 1 day post-injury in female TBI + saline mice, compared with males. CHOP expression increases after TBI and is a representative feature of ER stress-induced cell death,45,46 whereas eIF2α promotes cell survival in response to oxidative stress. 47 Therefore, the decreased CHOP and increased eIF2α may be partially responsible for the decreased cell loss at 3 days post-injury in the females.
Interestingly, BPN significantly decreased CHOP at 1 day post-injury in male mice, but not in females. These sex differences in BPN might be partially attributable to the differences in opioid receptor expression and distribution. 48 For example, we have previously demonstrated that MOR expression significantly increased at 1 and 7 days post-injury in the male TBI + saline mice after pediatric TBI, but not in females. 38 Evidence indicates that MOR activation can decrease ER stress and protect cells from excitotoxicity-induced apoptosis, 35 whereas KOR activation induces apoptosis through the enhanced ER stress pathway. 49 BPN can exert its effects through activation of MORs and inhibition of KORs, leading to decreased cell death. In addition, BPN significantly increased XPB1s at 7 days post-injury in both male and female TBI + BPN mice, compared to sham and TBI + saline mice. Evidence indicates that overexpression of XBP1s rescues Aβ neurotoxicity 50 and ameliorates tauopathy. 51 Therefore, increased XPB1s can be beneficial for reducing abnormal protein accumulation.
Interestingly, there is a discrepancy between mRNA and protein expression of CHOP. For example, in females, mRNA expression of CHOP was similar among groups at 3 and 7 days post-injury; however, protein expression of CHOP significantly increased in both the TBI + saline and TBI + BPN groups. This indicates that CHOP expression may be regulated transcriptionally and/or mRNA stability. 45
Studies have demonstrated acute Aβ and tau aggregation and cell loss in both TBI patients and animal models of TBI.52,53 Here, we demonstrated that TBI caused a persistent increase of CHOP-, p-tau-, Aβ-, and FJC-positive cells at the injured brain regions. Our results are consistent with a previous study, in which there is a positive correlation between CHOP, Aβ, and p-tau. 6 Interestingly, FJC and Aβ significantly increased in female TBI + saline mice at 7 days post-injury, compared with males. Studies have shown that females exhibit higher Aβ and tau burden than males, which is significantly associated with poor outcomes.54–58 We have previously demonstrated that female TBI + saline mice exhibit significantly increased depressive-like behaviors at 7 days post-injury, compared to males. 38 These behavioral deficits might be related to the increased Aβ burden and cell loss in females. We further demonstrate that BPN significantly decreased p-tau and FJC at 1 and 3 days post-injury, suggesting a neuroprotective effect against Aβ and p-tau toxicity.50,51 Together, our study provides a first line of evidence that BPN reduces ER stress, abnormal protein accumulation, and cell loss in a time- and sex-dependent manner after pediatric TBI. However, the underlying mechanisms of BPN actions need further investigation.
Conclusion
This study demonstrates that BPN treatment attenuated ER stress pathway activation, reduced Aβ and p-tau accumulation, and decreased cell loss in a time- and sex-dependent manner. Additional studies are warranted to study the long-term effects of BPN in both males and females after pediatric TBI.
Footnotes
Acknowledgments
We thank Mauda Abdullah for her assistance of the animal care.
Authors' Contributions
Megan B. Faulkner: investigation, writing–original draft. Mariam Rizk: investigation, writing–original draft. Zahraa Bazzi: investigation, writing–original draft. Robert C. Dysko: conceptualization, writing–review and editing, funding acquisition. Zhi Zhang: conceptualization, investigation, formal analysis, visualization, verification, resources, writing–original draft, writing–review and editing, supervision, project administration, funding acquisition.
Funding Information
This work was supported by the “Quality Improvement Funds (QIF)” grant number: U069304 (Animal Care and Use Office, University of Michigan–Ann Arbor) and “Start-up” grant number: U062955 (Department of Natural Sciences, CASL, University of Michigan–Dearborn) for Zhi Zhang.
Author Disclosure Statement
No competing financial interests exist.
