Abstract
Neurostimulation protocols are increasingly used as therapeutic interventions, including for brain injury. In addition to the direct activation of neurons, these stimulation protocols are also likely to have downstream effects on those neurons’ synaptic outputs. It is well known that alterations in the strength of synaptic connections (long-term potentiation, LTP; long-term depression, LTD) are sensitive to the frequency of stimulation used for induction; however, little is known about the contribution of the temporal pattern of stimulation to the downstream synaptic plasticity that may be induced by neurostimulation in the injured brain. We explored interactions of the temporal pattern and frequency of neurostimulation in the normal cerebral cortex and after mild traumatic brain injury (mTBI), to inform therapies to strengthen or weaken neural circuits in injured brains, as well as to better understand the role of these factors in normal brain plasticity. Whole-cell (WC) patch-clamp recordings of evoked postsynaptic potentials in individual neurons, as well as field potential (FP) recordings, were made from layer 2/3 of visual cortex in response to stimulation of layer 4, in acute slices from control (naive), sham operated, and mTBI rats. We compared synaptic plasticity induced by different stimulation protocols, each consisting of a specific frequency (1 Hz, 10 Hz, or 100 Hz), continuity (continuous or discontinuous), and temporal pattern (perfectly regular, slightly irregular, or highly irregular). At the individual neuron level, dramatic differences in plasticity outcome occurred when the highly irregular stimulation protocol was used at 1 Hz or 10 Hz, producing an overall LTD in controls and shams, but a robust overall LTP after mTBI. Consistent with the individual neuron results, the plasticity outcomes for simultaneous FP recordings were similar, indicative of our results generalizing to a larger scale synaptic network than can be sampled by individual WC recordings alone. In addition to the differences in plasticity outcome between control (naive or sham) and injured brains, the dynamics of the changes in synaptic responses that developed during stimulation were predictive of the final plasticity outcome. Our results demonstrate that the temporal pattern of stimulation plays a role in the polarity and magnitude of synaptic plasticity induced in the cerebral cortex while highlighting differences between normal and injured brain responses. Moreover, these results may be useful for optimization of neurostimulation therapies to treat mTBI and other brain disorders, in addition to providing new insights into downstream plasticity signaling mechanisms in the normal brain.
Introduction
A wide variety of neurological and psychiatric effects result from brain injuries like mild traumatic brain injury (mTBI). 1 –6 The capacity of the brain to adapt to injury and undergo restorative plasticity throughout the lifespan is well-documented. 7 –14 Up- and downregulation of the strength of synaptic transmission, such as long-term potentiation (LTP) and long-term depression (LTD), represent processes implicated as biological substrates of learning and memory. 15 –27 Such changes have been observed throughout the brain, including the: hippocampus, 28 –31 visual cortex, 32 –34 olfactory cortex, 35,36 somatosensory and motor cortices, 37 –39 striatum, 40,41 and cerebellum; 42,43 LTP and LTD are also implicated in functional reorganization after brain injury 44 –49 and during neurorehabilitation for brain injury or disease. 50 –56 It is of interest to learn whether these processes may be selectively evoked to facilitate a compensatory rebalancing of the relative strengths of particular synaptic pathways after mTBI and thus become a therapeutic target for restoring functionality. 57 –59 Moreover, with the availability of various neurostimulation technologies, such as deep brain stimulation (DBS), 60,61 transcranial magnetic stimulation (TMS), 62 –64 transcranial electrical stimulation (tES; e.g., direct or alternating current stimulation), 65 –67 transcranial focused ultrasound (tFUS), 68 –70 and more, 71 to treat conditions such as Parkinson’s disease, 72 –74 Alzheimer’s disease, 75 –77 seizure disorders, 78 –80 obsessive compulsive disorder (OCD), 81 –83 and major depression, 84 –87 there is value in better understanding the effects of these interventions on synaptic plasticity in a well-controlled experimental environment.
Although some clinical uses of DBS have begun to take into account temporal pattern, 88 –90 most experimental approaches to studying the induction mechanisms for LTP and LTD hold temporal patterns regular and constant, with a brief period of high frequency afferent stimulation, 91,92 a protracted period of low frequency presynaptic stimulation, 31,93 or a series of conjunctions of temporally coincident pre- and postsynaptic activation. 94 –96 While varying stimulation frequency, these protocols maintain a regular interval between each stimulus (i.e., all interstimulus intervals, ISIs, are equal). These temporally regular epochs of synaptic stimulation have been widely applied in isolated in vitro experimental preparations where the neurons are usually quiescent until stimulated. However, in vivo many neurons exhibit ongoing activity, continuously receiving synaptic inputs, many of which are evoked by presynaptic action potentials. 97 –101 Moreover, the temporal pattern of action potentials and subsequent evoked synaptic activity is often highly irregular with varying distributions of ISIs. 102 –107 Thus, it is important to understand the contribution of temporal pattern (vs. frequency alone) of evoked synaptic activity per se to the induction of synaptic plasticity in the normal brain and whether there are different temporal pattern requirements for accessing downstream synaptic plasticity signaling cascades after mTBI.
Because the neocortex is often subjected to mTBI and the synaptic circuitry of the visual cortex is well-characterized, we chose this area to evaluate the role of the temporal pattern of evoked synaptic activity. In particular, we were interested to learn whether the major synaptic drive from the neurons of the primary thalamic input zone (layer 4, L4), onto the layer 2/3 (L 2/3) pyramidal neurons that transmit information over long distances between cortical areas, reacts differently to variations in the temporal pattern of conditioning inputs in the normal brain and after mTBI. We focused on three stimulation frequencies known to play a role in plasticity in neocortex (1, 10, 100 Hz), three temporal patterns (perfectly regular: all ISIs are identical with a coefficient of variation [CV] = 0.0, slightly irregular: Gaussian distribution of ISIs with a CV = 0.2, and highly irregular: Poisson distribution of ISIs with a CV = 1.0), and two types of conditioning epochs (continuous or discontinuous).
METHODS (Details in Supplementary Data S1)
Animal use and experimental design
All procedures were approved by the Institutional Animal Care and Use Committees of Virginia Tech or Baylor College of Medicine and followed the National Research Council’s Guide for the Care and Use of Laboratory Animals. At 8–9 weeks of age, male Long-Evans rats were single housed and assigned to one of three treatment groups as follows: control (naive), sham (sham operated), or mTBI (controlled cortical impact, CCI). Two to three weeks later, these animals were used in righting reflex, electrophysiology, and/or calcium imaging experiments.
Mild traumatic brain injury
We used a well-established model of mTBI (a direct lateral CCI with a depth of 2.5 mm, velocity of 3.0 m/s, duration of 100 ms, angled at 45° from vertical). 108 –110 This model produces a brief spike in intracranial pressure (during impact) and a transient decrease in cerebral blood flow, but no gross abnormalities in brain histology. 108 –110 It also transiently impairs performance on motor function tasks, which recovers to baseline within 14 days postinjury. 110
At 8–9 weeks of age, the mTBI group underwent surgery under isoflurane anesthesia (1–4%). Animals were placed on a homeostatically controlled heating plate and mounted in a stereotaxic frame. The scalp was shaved, cleaned with alcohol and Betadine, and incised to expose the skull. A 7 mm diameter craniotomy was made over the right parietal cortex immediately posterior to bregma and lateral to midline. A direct lateral CCI was delivered through intact meninges using a Benchmark stereotaxic impactor (Leica) with a 6 mm diameter spherical head. After the CCI, we administered a long-lasting analgesic (buprenorphine SR, 1.2 mg/kg SC; ZooPharm), the craniotomy and incision were repaired, anesthesia was discontinued, and the rat monitored until fully recovered. Shams received identical treatment except for the CCI.
Righting reflex
At 10–12 weeks of age (2–3 weeks after treatment: control, sham, or mTBI) the first cohort of 45 rats was examined to assess righting latency. Individuals were placed in an induction chamber and anesthetized with isoflurane (4% for 120 s). The rat was removed, placed in a supine position, and righting latency (the time required for the rat to right itself to an upright posture standing on all four paws) was measured.
Brain slice preparation
At 10–12 weeks of age (2–3 weeks after treatment: control, sham, or mTBI) a second cohort of 389 rats was anesthetized with ketamine/xylazine (75 and 10 mg/kg, IM) and then decapitated. The brain was removed and placed in ice-cold cutting solution containing (in mM): 124 NaCl, 2 KCl, 3.5 MgCl2, 0.2 CaCl2, 1.25 KH2PO4, 26 NaHCO3, 11 dextrose, with pH 7.4 and saturated with carbogen (95% O2 + 5% CO2). Coronal slices (300 µm) were prepared from the right visual cortex (ipsilateral to the mTBI) and placed in artificial cerebral spinal fluid (ACSF—containing in mM: 124 NaCl, 2 KCl, 2 MgCl2, 2 CaCl2, 1.25 KH2PO4, 26 NaHCO3, 11 dextrose, with pH 7.4 and saturated with carbogen). Slices were incubated in ACSF at 33 ± 1°C for 20 min and then at room temperature for > 40 min prior to recording.
In vitro electrophysiology
For recording, slices were transferred to a submersion chamber perfused with 3 ml/min of ACSF at 33 ± 1°C. A bipolar stimulating electrode (FHC #30213) was placed in L4. Glass micropipettes (OD 1.5 mm, ID 1.12 mm; A-M Systems) were pulled on a vertical puller (PC-10; Narishige) to make field potential (FP) pipettes (1 ± 0.5 MΩ) or whole-cell (WC) patch pipettes (4 ± 1 MΩ). FP pipettes were filled with ACSF and placed in L2/3. WC patch pipettes were filled with pipette solution (containing in mM: 115 K-gluconate, 20 KCl, 10 Hepes, 4 NaCl, 4 Mg-ATP, 0.3 Na-GTP, and 4 phosphocreatine-Na; pH 7.4; 280–290 mOsm) and placed in L2/3 (∼100 µm deep to the FP pipette). Biocytin (0.5%; Sigma-Aldrich) added to the pipette solution allowed post hoc identification of patched cells (only pyramidal cells located in L2/3 were analyzed).
Recordings were made using a MultiClamp 700B amplifier, digitized at 20 kHz with a Digidata 1440A, and collected and analyzed with pCLAMP 10 software (Axon Instruments, Molecular Devices). Recordings of evoked postsynaptic potentials (PSPs) and FP amplitudes were measured as the difference between baseline and peak. Recordings with unstable preconditioning response amplitudes, membrane potentials more positive than −60 mV, high access resistance (> 40 MΩ, or > 20% of the membrane resistance for that cell), or a > 20% decrease in input resistance were discarded. Intrinsic firing properties of patched cells were tested by injecting a 100 ms depolarizing current pulse, and cells that did not exhibit regular spiking typical of excitatory cells 111,112 were discarded. Only one recording was made from each slice, so that a single stimulation protocol was applied in each case.
Stimulation protocols
Afferent stimulation consisted of constant current square waves (100 µs, 30–500 µA; set to evoke ∼40% of the maximal PSP response) output by a stimulus isolation unit (SIU; A365, World Precision Instruments). Stimulation was optimized to produce PSPs, but in some cases (∼30%) also evoked FPs, which were recorded simultaneously. Preconditioning baseline recordings of evoked PSP (and FP) peak amplitudes were made at 0.1 Hz for 10 min. This was followed by a 90 or 900 sec period of synaptic conditioning, consisting of 900 pulses at 1, 10, or 100 Hz. Conditioning was either continuous (1 Hz for 900 sec, or 10 Hz for 90 sec; Fig. 1A–B) or discontinuous (9 epochs of 100 pulses each at 10 or 100 Hz, separated by 8 equal rest intervals consisting of probe trials at 0.1 Hz, with a total duration of 900 sec; Fig. 1C). We included the discontinuous protocols since continuous stimulation at high frequency can induce presynaptic fatigue that might interact with long-term synaptic plasticity. 113,114 Postconditioning evoked PSP (and FP) peak amplitudes were evaluated at 0.1 Hz for 20 min. Conditioning stimulation was delivered with one of three temporal patterns as follows: perfectly regular (CV 0.0; Fig. 1D–E), slightly irregular (CV 0.2; Fig. 1F–G), or highly irregular (CV 1.0; Fig. 1H-I) each with the same mean frequency. Conditioning stimulation was generated using MatLab (MathWorks), transformed with a digital analog converter (BNC 2090A, National Instruments), triggered with a pulse stimulator (Master-9, A.M.P.I.), and output through the SIU, giving us precise control of frequency, continuity, and temporal pattern.
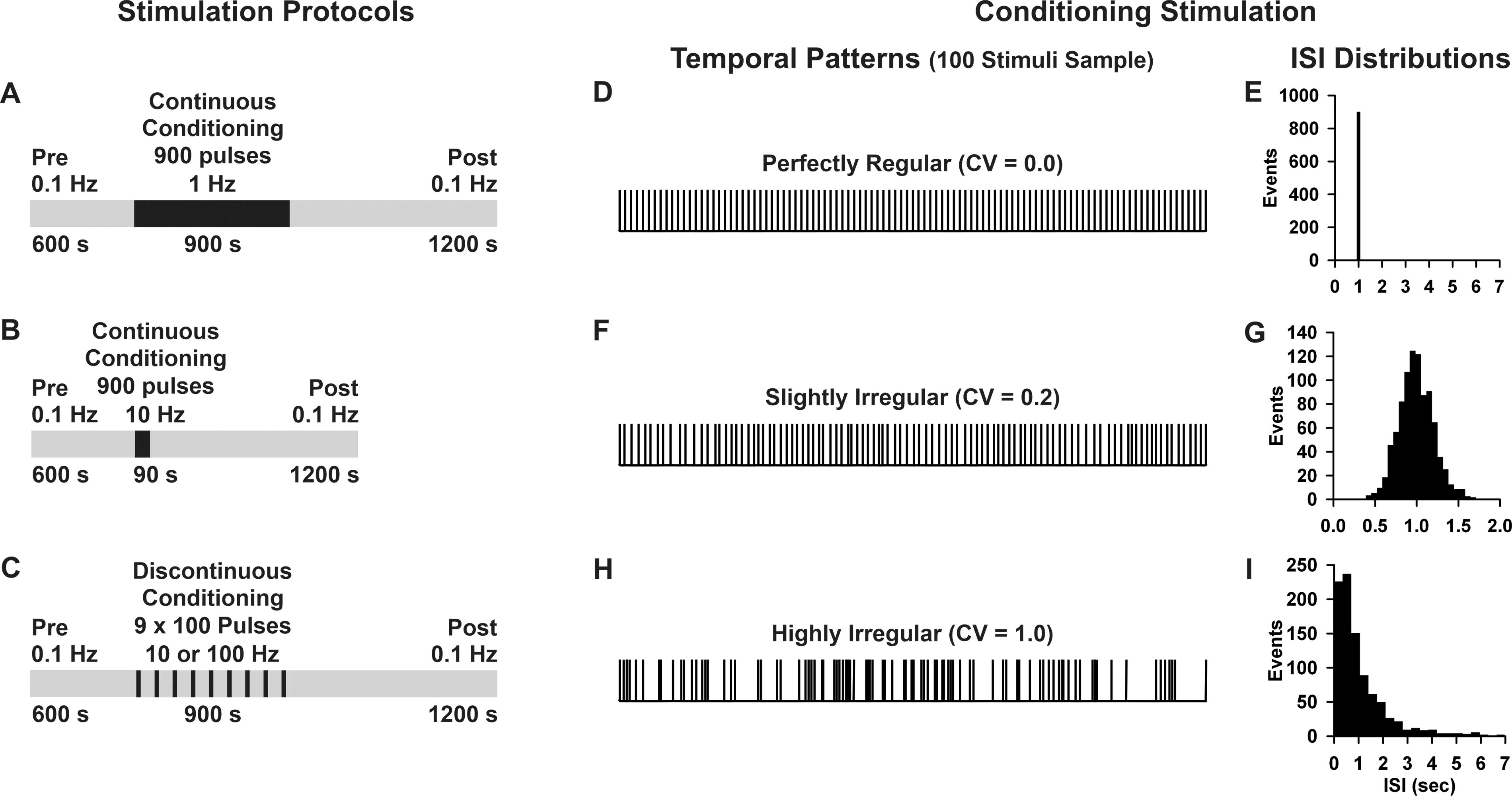
Stimulation protocols, temporal patterns, and interstimulus interval distributions.
Temporal patterns of conditioning stimulation
Conditioning stimulation temporal patterns were generated by creating ISIs according to the family of distributions given by:
We randomly assigned numbers drawn uniformly on the unit interval to ISIs in the following way. For each random number, r, we inverted equation (4) to find a t that represented an ISI affiliated with a given mean and CV. By specifying a different mean and CV (and therefore different η and τd, or equivalently, different η and w) in equation (4), we derived related sets of ISIs, using a single set of random numbers.
Analyzing electrophysiology data
We measured evoked PSP (or FP) peak amplitudes during the last 5 min of pre- and postconditioning (using pCLAMP) and calculated a post/pre ratio. We evaluated the effects of mTBI, as well as stimulus frequency, temporal pattern, and continuity on the induction of synaptic plasticity [defined as: LTD, a significant (p < 0.05, t-test) decrease (≥10%) in post/pre ratios; LTP, a significant increase (≥10%) in post/pre ratios; or no change (NC), post/pre ratios that changed <10% and/or were not significantly different]. For these same time periods (the last 5 min of pre- and postconditioning), we used pCLAMP to calculate the average PSP: half width (at half height), rise time (10–90%), and decay time (90-10%); as well as the membrane potential and input resistance for each cell.
We also analyzed the kinetic profiles of conditioning responses for individual recordings in the groups that showed a significant difference between control and mTBI. For continuous stimulation protocols, we took all data points generated during the conditioning period and calculated exponential and linear best fits, from which we determined the slope. For discontinuous stimulation protocols, best fits were calculated for each of the nine conditioning subepochs and the slopes averaged. Furthermore, we calculated the mean of all conditioning response amplitudes (average), as well as the average of the initial 10% (initial) and final 10% (final). For all recordings within a given stimulation protocol, a linear regression analysis was performed to determine the degree to which each factor (exponential slope, linear slope, average amplitude, initial amplitude, and final amplitude) was correlated with plasticity outcome (post/pre ratio).
Calcium imaging
In a subset of the WC patch experiments, the ratiometric calcium sensitive dye Fura-4F (100 µM, Thermo Fisher Scientific) was added to the pipette solution. Baseline somatic calcium concentrations were calculated as the ratio of bound to unbound calcium minus background (AxioVision; Zeiss) during the last five minutes of preconditioning.
Statistics
We used SigmaPlot (Systat Software) to determine statistical significance, except where noted. Normality was assessed with a Shapiro–Wilk test. For normally distributed data, we used a t-test or an ANOVA followed by Fisher’s least significant difference method for multiple comparisons (Fischer’s LSD). For datasets with non-normal distributions, we used a Mann–Whitney U test (MWU) or a Kruskal–Wallis one-way ANOVA on ranks (Kruskal–Wallis) followed by Dunn’s method for multiple comparisons with Sidak correction (Dunn’s corrected; statskingdom.com/kruskal-wallis-calculator.html). Group data are presented as the mean ± SEM. To assess whether specific characteristics of conditioning responses were predictive of plasticity outcome, for each factor, we calculated a correlation coefficient and determined significance using linear regression. We considered differences statistically significant at p < 0.05 (except for Dunn’s correction, which was considered significant at p < 0.017).
Results
Efficacy of our mTBI model
To establish the efficacy of our CCI model for mTBI, we used several independent methods. First, we used a behavioral measure, recovery of righting reflex, to assess motor and cognitive function in control, sham, and mTBI rats. Righting latency was significantly impacted by treatment group (p = 0.04) and was longer in mTBI than in control (p = 0.04) or sham (p = 0.02; Fig. 2A) groups. Note that for this and all subsequent figures we show control data in black, sham data in blue, and mTBI data in red. Second, we measured intracellular calcium concentration in a subset of the L2/3 pyramidal neurons used for our WC patch-clamp recordings. Intracellular calcium concentrations were significantly different between treatment groups (p < 0.001) and were higher in the mTBI than in the control (p < 0.001) or sham group (p < 0.001; Fig. 2B). Third, we measured the intrinsic properties of this same subset of cells and found that input resistance was significantly affected by the treatment group (p = 0.005), being higher in the mTBI group than in the control (p = 0.001) or sham (p = 0.008; Fig. 2C) groups; whereas membrane potential did not differ between the control, sham, or mTBI groups (p = 0.38; Fig. 2D).
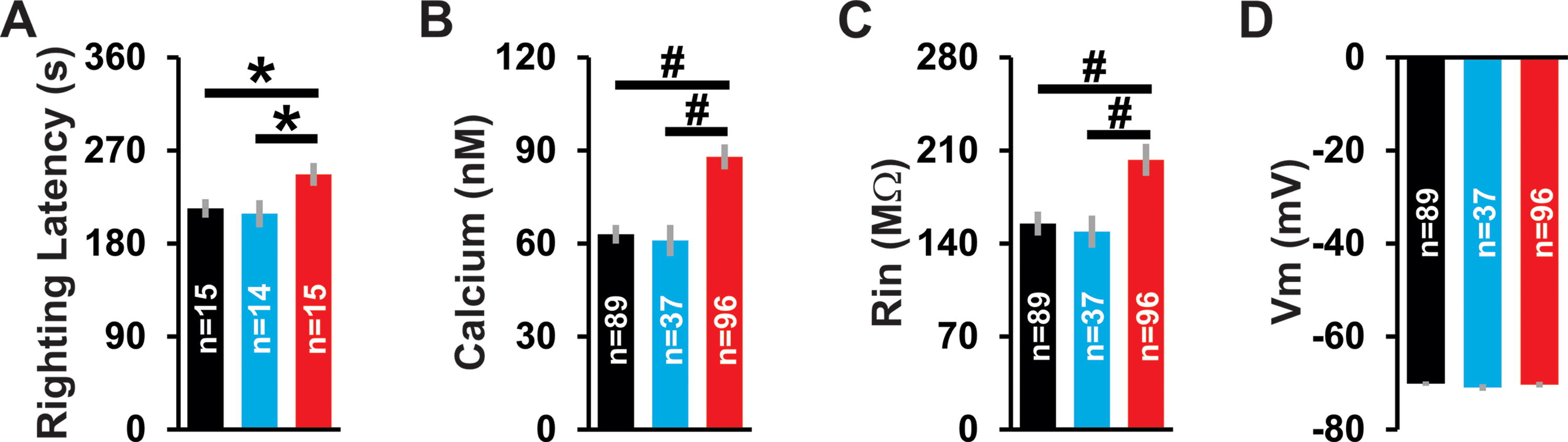
Efficacy of our CCI model for mTBI.
Effect of temporal pattern and treatment condition on plasticity outcome
WC patch-clamp experiments were conducted to investigate the role of temporal pattern on synaptic plasticity in control, sham, and mTBI groups. Figure 3 shows example recordings of evoked PSPs and time plots of individual recordings in response to 1 Hz continuous highly irregular conditioning (cf. Fig. 1A, 1H-I). Note the substantial decrease in postconditioning PSP amplitude, indicative of LTD, in control and sham examples (Fig. 3A–B). In contrast, there was a substantial increase in postconditioning PSP amplitude, indicative of LTP, in the mTBI example (Fig. 3C).
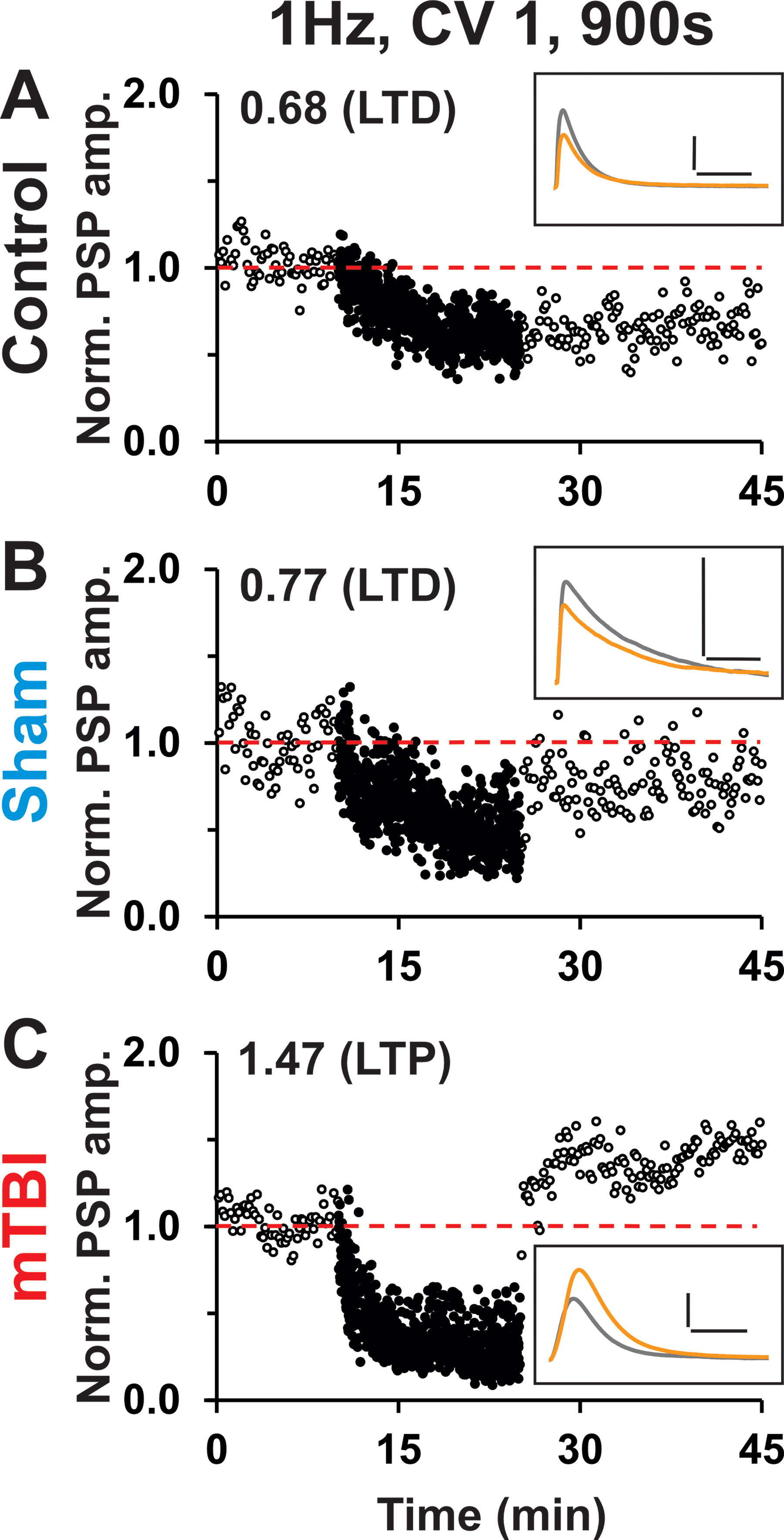
Individual time plots of WC responses to highly irregular 1 Hz continuous conditioning.
Grouped results for our WC recording experiments for the various frequencies, temporal patterns, and treatment groups are shown in subsequent figures. Continuous conditioning at 1 Hz induced LTD in both control groups (Fig. 4A–C) and mTBI groups (Fig. 4D–E) in all cases except one—highly irregular conditioning (CV 1.0) in mTBI rats resulted in LTP (Fig. 4F). Temporal pattern (CV 0.0, CV 0.2, CV 1.0) had no effect on plasticity outcome in control groups (p = 0.50; Fig. 4A–C, 4G). However, for mTBI groups, there was a significant difference in plasticity outcome between the three temporal patterns (p = 0.002; Fig. 4D–F, H) with highly irregular conditioning (CV 1.0) showing significant potentiation (versus: CV 0.0 p = 0.007, CV 0.2 p < 0.001). Notably, within each of the six stimulation groups, there was heterogeneity in plasticity outcomes among individual cells (Fig. 4G–H). Importantly, plasticity outcomes in control and mTBI rats were significantly different only when the temporal pattern was highly irregular (p = 0.008; cf. Fig. 4C, F), but not when it was perfectly regular (p = 0.76; cf. Fig. 4A, D) or slightly irregular (p = 0.66; cf. Fig. 4B, E). For highly irregular conditioning control, sham and mTBI groups were significantly different (p = 0.004; Fig. 4I). Pairwise comparisons showed that mTBI was significantly different from both control (p = 0.002) and sham (p = 0.002), but control and sham groups did not differ (p = 0.49; Fig. 4I). The potentiation produced by highly irregular 1 Hz conditioning in mTBI rats reflects both an increase in the proportion (control = 18%, sham = 6%, mTBI = 47%) and magnitude (post/pre ratio: control = 1.31, sham = 1.13, mTBI = 1.62) of LTP outcomes (Fig. 4I).
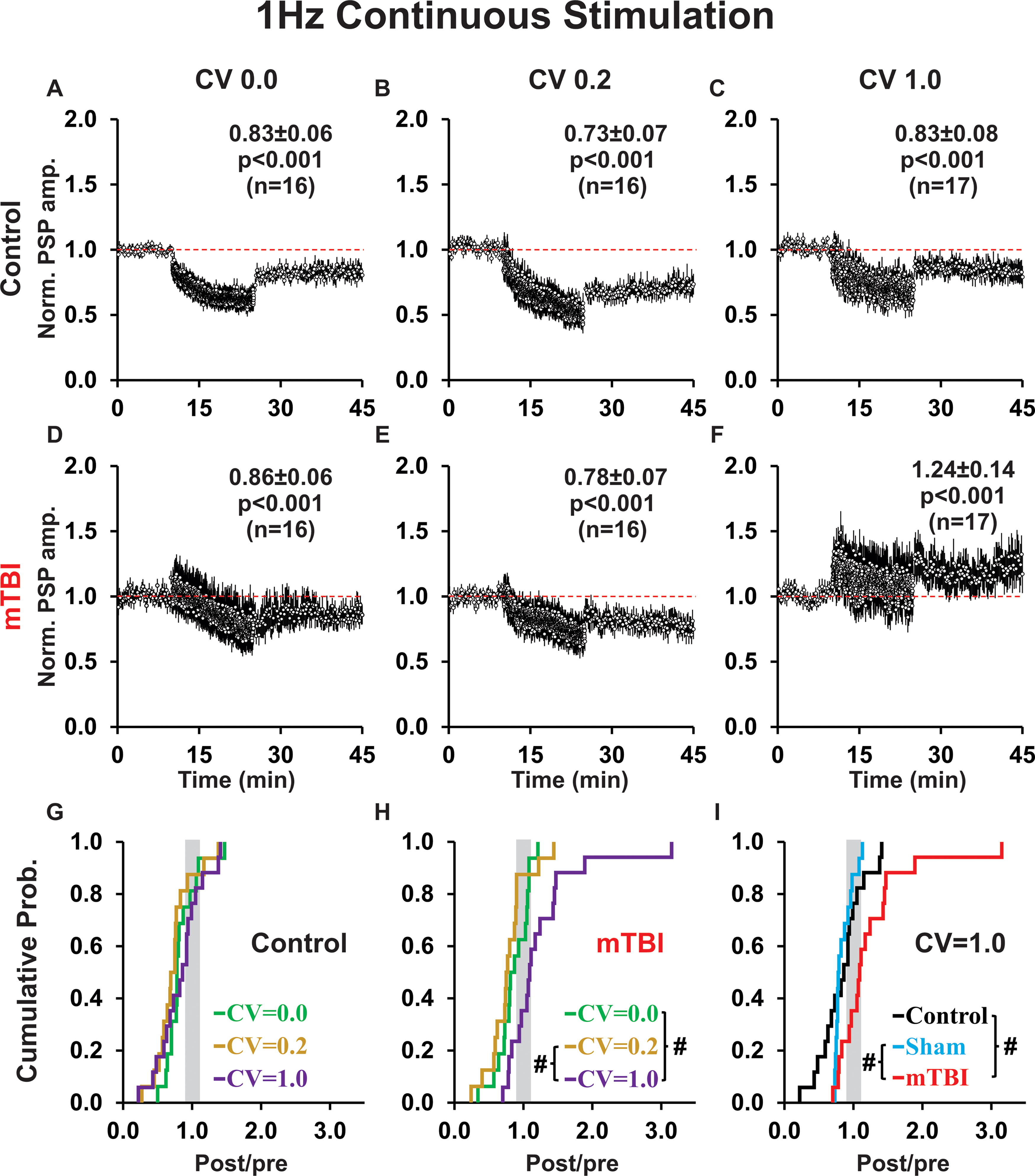
Continuous 1 Hz conditioning induces LTD in every case except one—highly irregular conditioning in mTBI rats results in LTP.
In addition, highly irregular 1 Hz continuous conditioning differentially affected the evoked PSP shape indices (Supplementary Fig. S1A) in controls, showing a significant decrease in decay time (DT p = 0.03) and half width (HW p = 0.03; Supplementary Fig. S1B), and this effect occurred primarily in the responses that underwent LTD (DT p = 0.045, HW p = 0.03; Supplementary Fig. S1C, cf. Supplementary Fig. S1B, S1D). Shape indices in shams showed a similar decrease in DT (p = 0.09) and HW (p = 0.02; Supplementary Fig. S1B), which occurred primarily in responses that underwent LTD (DT p = 0.03, HW p = 0.02; Supplementary Fig. S1C, cf. Supplementary Fig. S1B, S1D). Interestingly, although the shape indices of the responses in the mTBI group as a whole did not change significantly after conditioning (DT p = 0.49, HW p = 0.54; Supplementary Fig. S1B), among just the responses that underwent LTP, they significantly increased (DT p = 0.01, HW p = 0.01; Supplementary Fig. S1D, cf. Supplementary Fig. S1B-C).
Continuous conditioning at 10 Hz produced a rapid rundown in PSP amplitudes, but was less likely to induce LTD, than 1 Hz continuous conditioning (cf. Figs. 5A–E, 4A–E). In particular, for controls, slightly irregular continuous conditioning produced LTD for 1 Hz, but NC for 10 Hz conditioning (p = 0.01; cf. Figs. 4B, 5B); whereas after mTBI, perfectly regular conditioning induced LTD for 1 Hz continuous, but LTP for 10 Hz continuous conditioning (p = 0.05; cf. Figs. 4D, 5D). Temporal pattern had no effect on plasticity outcome in control groups (p = 0.58; Fig. 5A–C, 5G). However, for mTBI groups, there was a significant difference in plasticity outcomes (p = 0.002; Fig. 5D–F, 5H) between slightly irregular conditioning, which showed a shift toward LTD, and perfectly regular conditioning or highly irregular conditioning, both of which showed a shift toward LTP (p = 0.015; p < 0.001; Fig. 5H). In addition, we found that 10 Hz continuous conditioning (similar to 1 Hz continuous conditioning) produced significantly different plasticity outcomes for control and mTBI groups only when the temporal pattern of conditioning was highly irregular (p = 0.003; cf. Fig. 5C, F), but not perfectly regular (p = 0.35; cf. Fig. 5A, D) or slightly irregular (p = 0.10; cf. Fig. 5B, E). Accordingly, for highly irregular conditioning, we analyzed control, sham, and mTBI groups (p = 0.003; Fig. 5I). Pairwise comparisons showed a significant potentiation of responses in mTBI relative to control (p = 0.001) and sham (p = 0.004) groups, but no difference between controls and shams (p = 0.30). The potentiated response to highly irregular conditioning (like that observed for the 1 Hz continuous group) reflected a change in both the proportion (control = 0%, sham = 12%, mTBI = 52%) and magnitude (post/pre ratio: control = no LTP responses, sham = 1.24, mTBI = 1.56) of LTP outcomes. However, unlike 1 Hz continuous conditioning, there were no effects of 10 Hz continuous conditioning on the shape indices of PSP responses (data not shown).
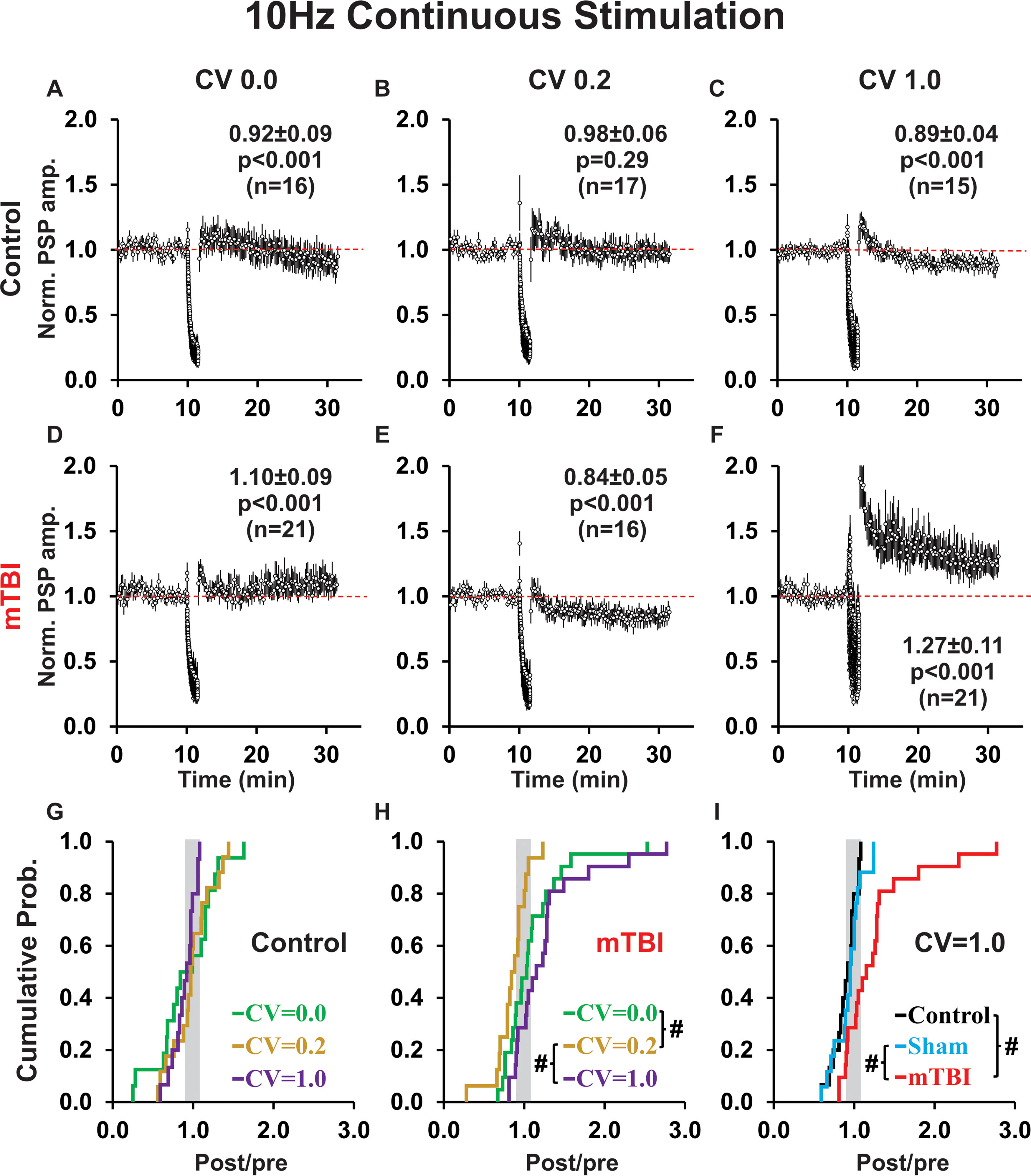
Continuous 10 Hz (compared to 1 Hz) conditioning produces a rapid rundown in PSP amplitudes, is less likely to result in LTD in controls, and more likely to result in LTP after mTBI.
For 10 Hz discontinuous stimulation protocols, the brief conditioning subepochs mitigated the rundown of response amplitudes that occurred for 10 Hz continuous conditioning protocols (cf. Figs. 6A–F, 5A–F). Moreover, probe stimuli administered at 0.1 Hz during the rest periods (Fig. 6A–F, yellow symbols) showed that PSP amplitudes quickly recovered, in most cases showing a brief period of post-tetanic potentiation, which suggests that any synaptic fatigue induced by an individual subepoch of higher frequency conditioning was transitory. Interestingly, probe trail response amplitudes were sensitive to the temporal pattern of conditioning in control groups (depressed for CV 0.0 and 1.0, but potentiated for CV 0.2; cf. Fig. 6A–C) and mTBI groups (potentiated for CV 0.0, neutral for CV 0.2, and potentiated for CV 1.0; cf. Fig. 6D–F). Notably, the continuity of 10 Hz conditioning influenced plasticity outcome and this was also dependent on temporal pattern. In controls, 10 Hz highly irregular conditioning induced a significantly more robust LTD when conditioning was discontinuous rather than continuous (p = 0.02; cf. Figs. 5C, 6C). In mTBI groups, 10 Hz perfectly regular conditioning induced LTD for discontinuous conditioning, but LTP for continuous conditioning (p = 0.06 trend; cf. Figs. 5D, 6D).
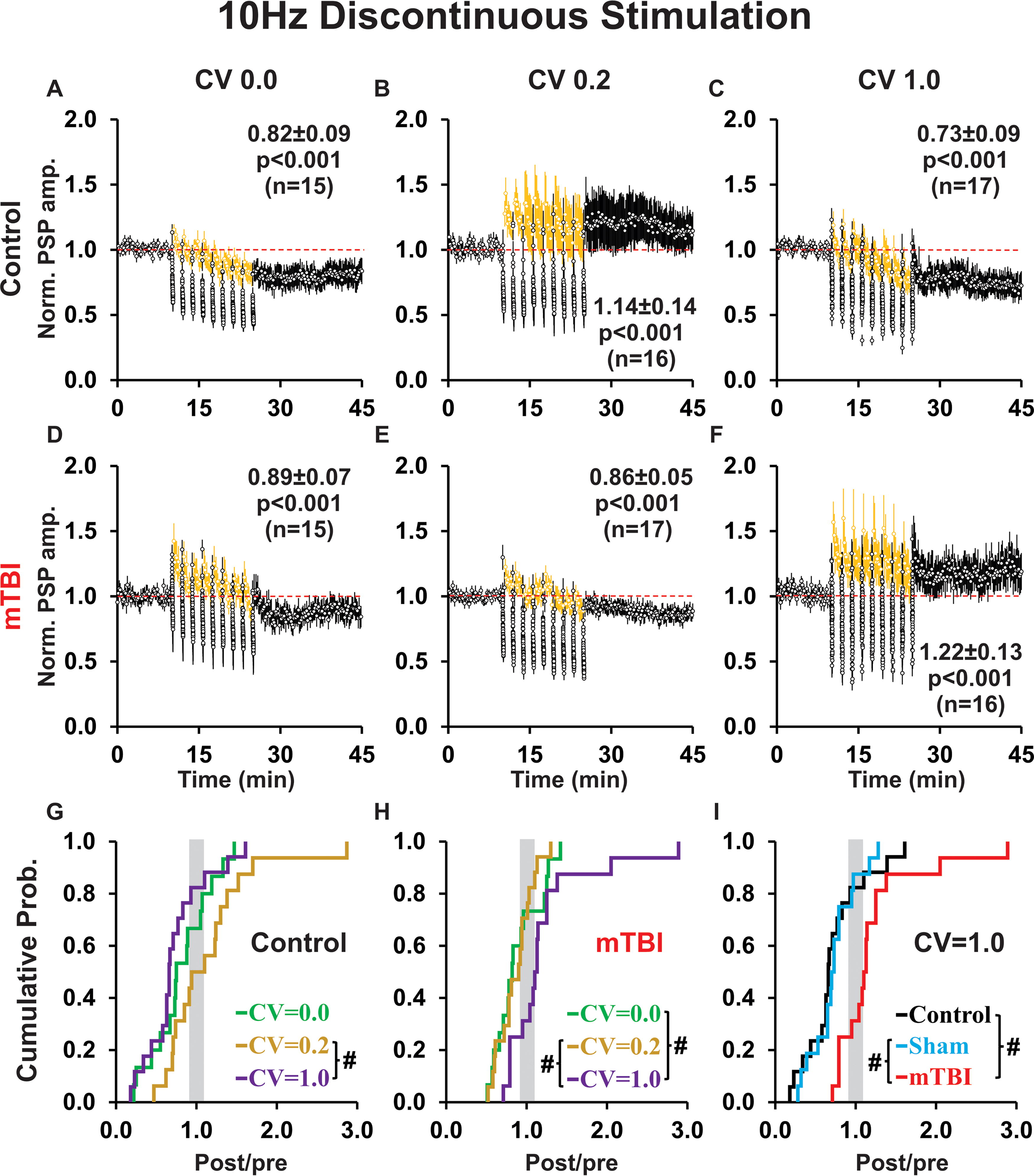
Discontinuous 10 Hz conditioning primarily induced LTD, except for slightly irregular conditioning in controls and highly irregular conditioning in mTBI rats.
For 10 Hz discontinuous (unlike 10 Hz continuous) conditioning, there was a significant effect of temporal pattern between control groups (p = 0.04; Fig. 6A–C, 6G), as well as between mTBI groups (p = 0.02; Fig. 6D–F, H). Pairwise comparisons showed a significant effect of temporal pattern in controls, with LTD in response to highly irregular conditioning (Fig. 6C) compared to LTP elicited by slightly irregular conditioning (p = 0.006; Fig. 6B, G). In mTBI groups, there was a significant difference between highly irregular (Fig. 6F) and both: slightly irregular (p = 0.006; Fig. 6E, H) and perfectly regular (p = 0.012; Fig. 6D, H) conditioning. Moreover, control and mTBI groups differed for highly irregular conditioning (p < 0.001; cf. Fig. 6C, F), but not for slightly irregular (p = 0.17; cf. Fig. 6B, E) or perfectly regular (p = 0.56; cf. Fig. 6A, D) conditioning. Accordingly, for highly irregular conditioning, we analyzed control, sham, and mTBI groups (p < 0.001; Fig. 6I). Pairwise comparisons showed a significant difference between treatment groups—with a robust shift toward LTP in mTBI relative to both control (p < 0.001) and sham (p < 0.001) groups (Fig. 6I). The potentiation reflected an increase in the proportion (control = 12%, sham = 12%, mTBI = 56%), but (unlike 1 Hz and 10 Hz continuous conditioning) not magnitude (post/pre ratio: control = 1.50, sham = 1.23, mTBI = 1.48) of LTP outcomes. There were no effects of 10 Hz discontinuous conditioning on PSP shape indices (data not shown).
For 100 Hz conditioning, we used only a discontinuous protocol which allowed for rapid recovery of PSP amplitudes during rest intervals [with attendant enduring post-tetanic potentiation-see yellow probe trial data during conditioning (gray bar)-Figure 7A–F]. Note that data from the nine conditioning subepochs are not shown because most PSPs were embedded in a wave of depolarization and/or had ISIs too short to allow accurate quantification. In control groups, 100 Hz discontinuous perfectly regular and slightly irregular conditioning produced no change in PSP amplitude (cf. Fig. 7A, B), whereas highly irregular conditioning induced LTD (Fig. 7C) similar to 1 Hz continuous, 10 Hz continuous, and 10 Hz discontinuous conditioning (cf. Figs. 7C, 4C, 5C, 6C). In contrast, for mTBI groups, perfectly regular conditioning produced NC (Fig. 7D), slightly irregular conditioning produced LTP ([Fig. 7E], which differed from 1 Hz continuous, 10 Hz continuous, and 10 Hz discontinuous conditioning, all of which exhibited LTD [cf. Figs. 7E, 4E, 5E, 6E]), and highly irregular 100 Hz continuous conditioning resulted in NC (Figure 7F; which differed from 1 Hz continuous, 10 Hz continuous, and 10 Hz discontinuous conditioning—all of which induced LTP [cf. Figs. 4F, 5F, 6F]). Importantly, despite these modest differences, comparisons between control groups (p = 0.17) or between mTBI groups (p = 0.93) showed no significant effect of temporal pattern on plasticity outcome for the 100 Hz discontinuous conditioning protocol. In addition, there was no significant difference in plasticity outcomes between control and mTBI groups for any temporal pattern (CV 0.0 p = 0.40, CV 0.2 p = 0.97, CV 1.0 p = 0.28).
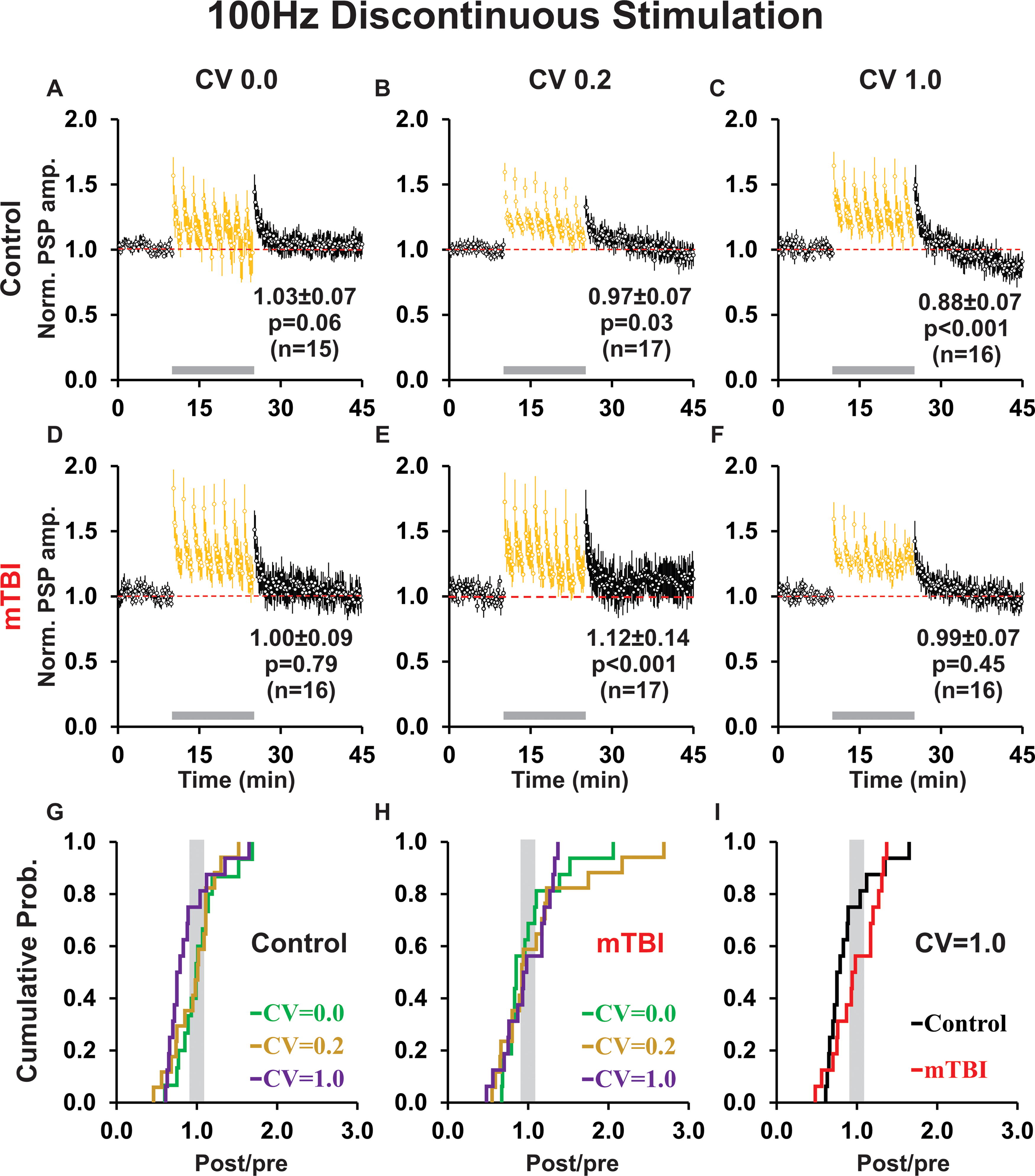
Discontinuous 100 Hz conditioning resulted in little plasticity, except for highly irregular conditioning in controls which produced LTD and slightly irregular conditioning after mTBI which induced LTP.
Interestingly, for 100 Hz highly irregular conditioning, the shape indices of PSPs among the mTBI group showed a significant increase in HW overall (p = 0.03; Supplementary Fig. S1E) that was nonspecific for plasticity outcome (LTD p = 0.40, LTP p = 0.26; cf. Supplementary Fig. S1E-G), whereas in controls there was no significant change in DT overall (p = 0.54; Supplementary Fig. S1E), but a significant decrease for those cells responding with LTP (p = 0.04; Supplementary Fig. S1G) but not LTD (p = 0.39; Supplementary Fig. S1F). In contrast, for 100 Hz slightly irregular (CV = 0.2) conditioning, the mTBI group showed an increase in RT overall (p = 0.002; Supplementary Fig. S1H) which was consistent across all plasticity outcomes (LTD p = 0.035, LTP p = 0.006; cf. Supplementary Fig. S1H-J), whereas in controls RT was unchanged overall (p = 0.06) or for any particular plasticity outcome (LTD p = 0.18, LTP p = 0.47; cf. Supplementary Fig. S1H-J).
Kinetic properties of conditioning period responses
We analyzed the kinetic profiles of conditioning responses for individual recordings, in the groups that showed a significant difference between control and mTBI, to determine if they were predictive of plasticity outcome. For 1 Hz continuous highly irregular conditioning, we show example time plots of conditioning responses for each treatment group (Fig. 8A–C). Notably, the slope of an exponential best fit of conditioning response amplitudes showed a significant correlation with plasticity outcomes only in control (p = 0.02; Fig. 8A, D) and sham groups (p = 0.048; Fig. 8B, D), whereas the slope of a linear best fit showed a significant correlation with plasticity outcomes only in the mTBI group (p = 0.02; Fig. 8C, E). The slopes of conditioning response amplitudes did not significantly differ in magnitude for either linear (control = −0.013 ± 0.003, sham = −0.012 ± 0.003, mTBI = −0.007 ± 0.004; p = 0.41 ANOVA; data not shown) or exponential best fits (control = −0.019 ± 0.005, sham = −0.018 ± 0.004, mTBI = −0.011 ± 0.004; p = 0.39 ANOVA; data not shown). However, relative to control and sham groups, the mTBI group exhibited a significant increase in conditioning response amplitude: initial value (overall p = 0.002, mTBI vs. control p < 0.001, mTBI vs. sham p = 0.003), final value (overall p = 0.006, mTBI vs. control p = 0.001, mTBI vs. sham p = 0.007), and average value (overall p = 0.01, mTBI vs. control p = 0.003, mTBI vs. sham p = 0.019—trend; Fig. 8F). In addition, in the mTBI group, all of these values: average (p = 0.004), initial value (p = 0.007), and final value (p = 0.001) were predictive of plasticity outcome (Fig. 8G-I), whereas in the control and sham groups, only the average (control p = 0.04, sham p = 0.03) and final (control p = 0.007, sham p = 0.009) amplitudes were predictive of plasticity outcome (Fig. 8G-I).
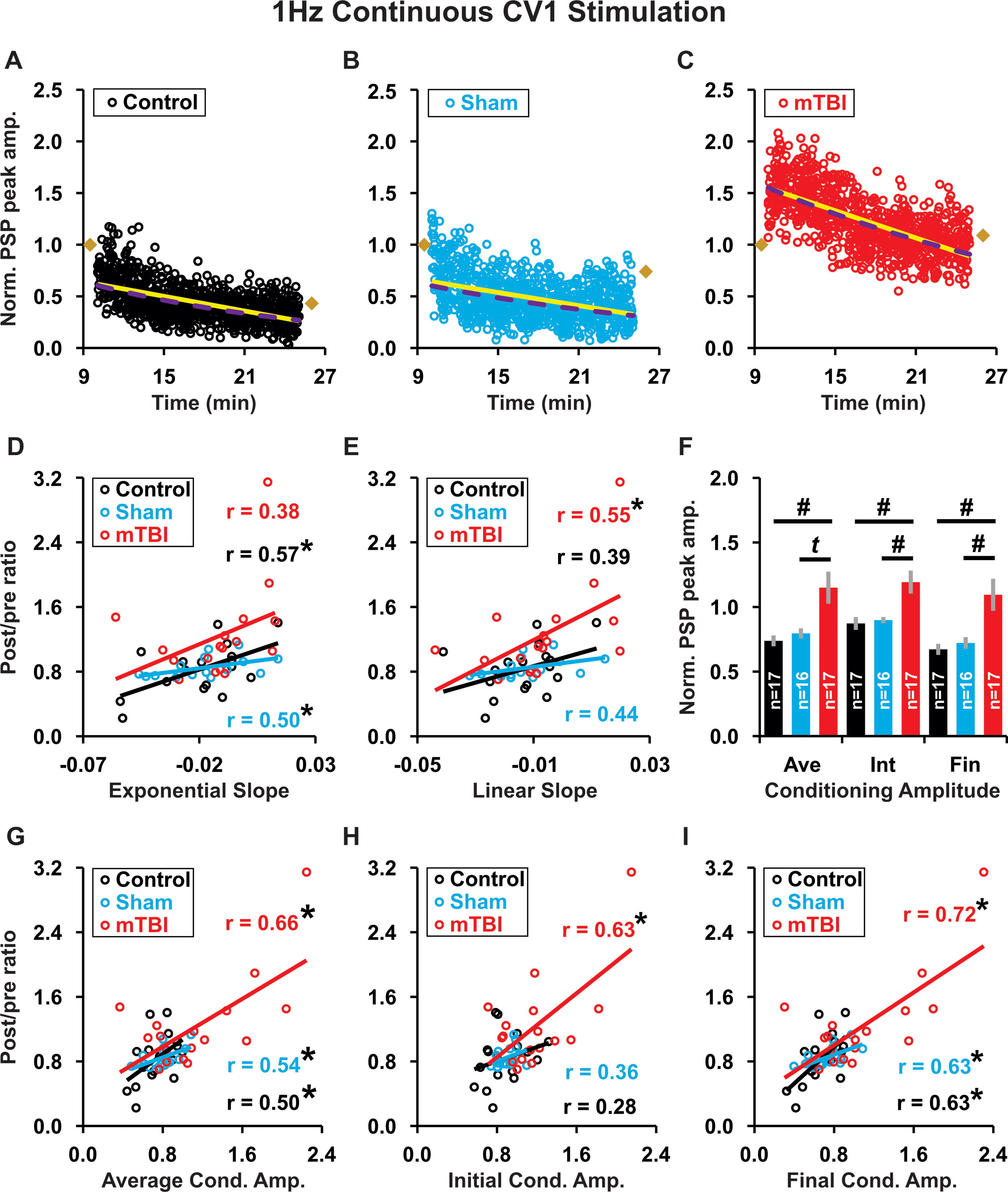
During 1 Hz continuous highly irregular conditioning, the kinetic profile of response amplitudes was predictive of plasticity outcome.
In contrast, for 10 Hz continuous highly irregular conditioning, the kinetics of the conditioning responses (slope Fig. 9A–E; amplitude: Fig. 9G-I) were not predictive of plasticity for any treatment group (control, sham, or mTBI). However, exponential best fits of conditioning amplitude had a shallower slope in mTBI than in control or sham groups (overall p < 0.001 ANOVA; with mTBI = −0.580 ± 0.096 vs. control = −1.045 ± 0.090, or sham = −1.125 ± 0.114; p = 0.002, p < 0.001, Fisher’s LSD; data not shown). Moreover, although the conditioning response amplitude was not significantly different between treatment groups initially (p = 0.15; Fig. 9F), both the average (overall p = 0.006, mTBI vs. control p = 0.009, mTBI vs. sham p = 0.002) and final (overall p < 0.001, mTBI vs. control p = 0.001, mTBI vs. sham p < 0.001) amplitudes were significantly higher in the mTBI group than in the control or sham groups (Fig. 9F). Thus, during 10 Hz continuous highly irregular conditioning, response amplitudes decreased less and more slowly in mTBI than in control and sham groups.
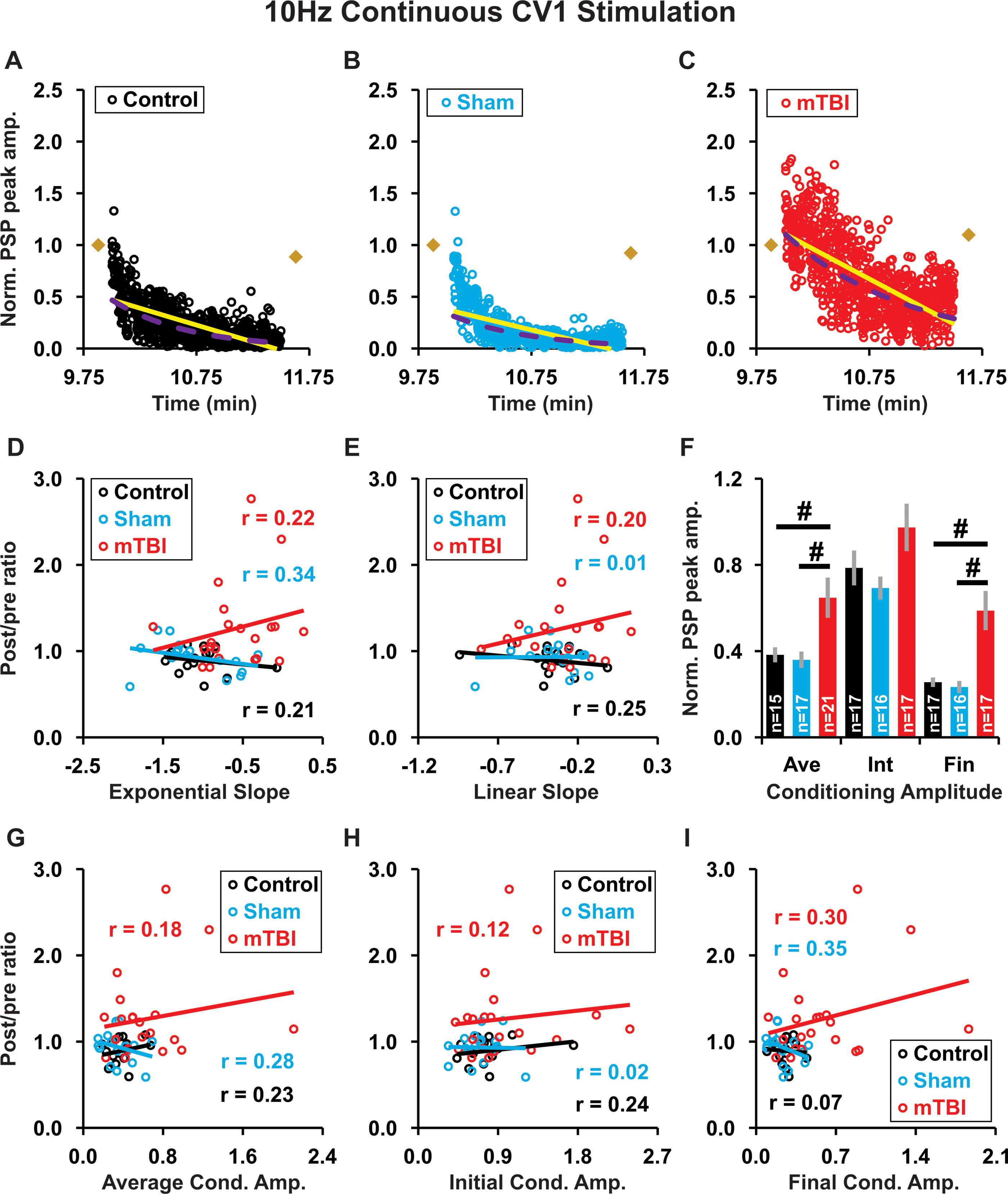
During 10 Hz continuous highly irregular conditioning, the kinetic profile of response amplitudes was not predictive of plasticity outcome.
For 10 Hz discontinuous highly irregular conditioning, the kinetic profile of conditioning response amplitudes measured as the average slope of exponential or linear best fits (Fig. 10A–C) was predictive of plasticity outcome only in control and sham groups—showing significant negative correlations for both linear slope (control p < 0.001, sham p = 0.02) and exponential slope (control p < 0.001, sham p = 0.04; Fig. 10D–E). This result differs from both 1 Hz continuous highly irregular conditioning (significant positive correlation, cf. Fig. 8D–E) and 10 Hz continuous highly irregular conditioning (no correlation, cf. Fig. 9D–E). There was no significant difference in the magnitude of conditioning response slopes between treatment groups, for either: linear (control −0.560 ± 0.376, sham −0.908 ± 0.233, mTBI −1.157 ± 0.155; p = 0.19 Kruskal–Wallis; data not shown) or exponential best fits (control −0.899 ± 0.671, sham −1.555 ± 0.297, mTBI −1.959 ± 0.388; p = 0.24 Kruskal–Wallis; data not shown). In addition, there was no significant difference in the magnitude of conditioning response amplitude: average (p = 0.75), initial (p = 0.95), or final values (p = 0.41; Fig. 10F). However, in the mTBI group, conditioning response amplitude: average (p = 0.02), initial value (p = 0.03), and final value (p = 0.02) were all predictive of plasticity outcome (Fig. 10G-I). In contrast, for the control and sham groups, conditioning response amplitude was not predictive of plasticity outcome for: average (control p = 0.71, sham p = 0.35; Fig. 10G), initial (control p = 0.17, sham p = 0.24; Fig. 10H), or final values (control p = 0.69, sham p = 0.44; Fig. 10I).
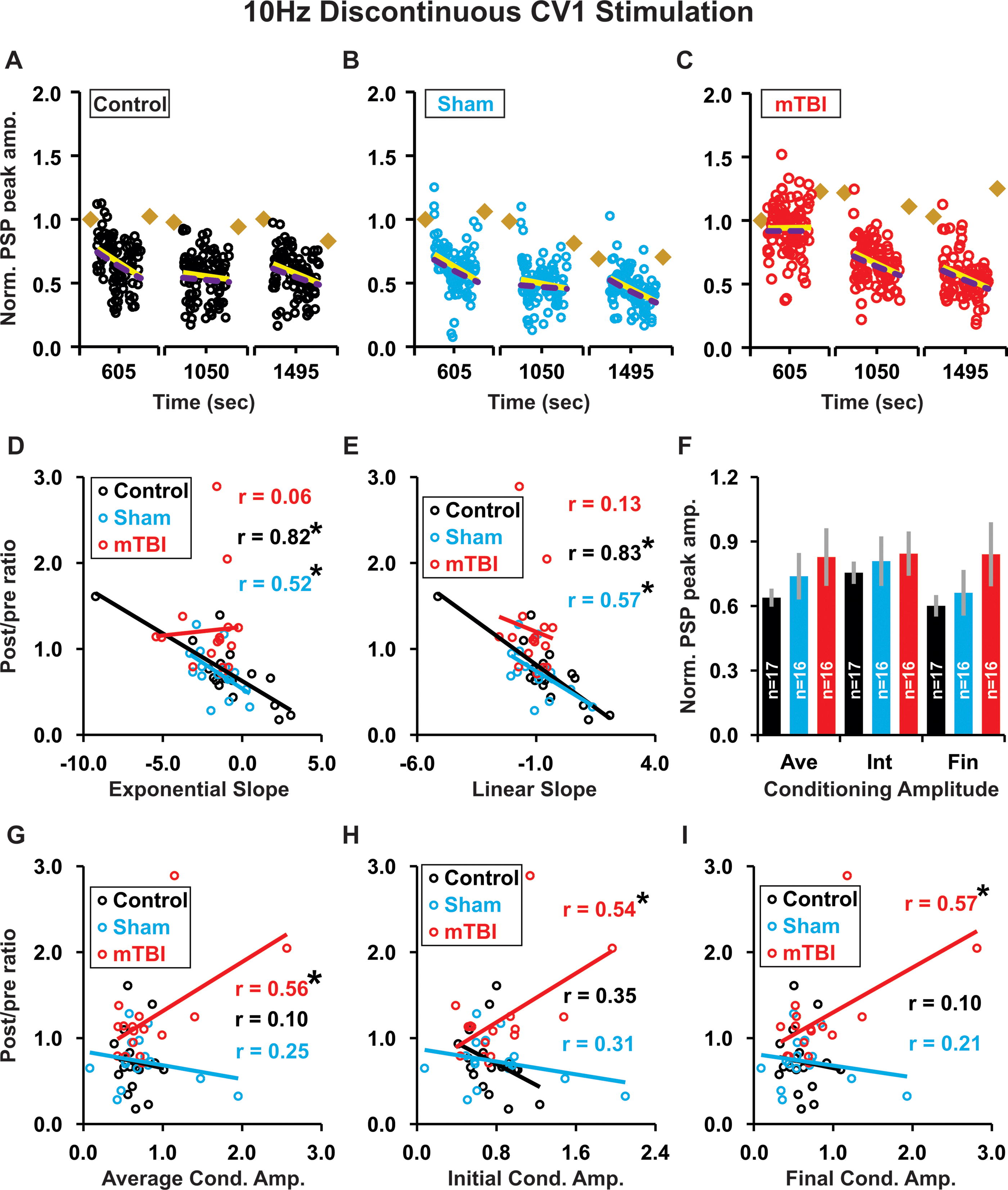
During 10 Hz discontinuous highly irregular conditioning, the kinetic profile of PSP responses had: slopes (exponential and linear) that were significantly correlated with plasticity outcome in control and sham, but not in mTBI groups; and amplitudes (average, initial and final) that were significantly correlated with plasticity outcome in mTBI groups, but not control or sham groups.
Data from our WC recordings appear broadly representative
In order to assess whether our WC results were reflective of the larger population of synapses in L2/3, we also made simultaneous FP recordings. Representative examples of individual recordings and time plots for paired WC and FP recordings are shown in Supplementary Figure S2 for: 1 Hz continuous (Supplementary Fig. S2A–B), 10 Hz continuous (Supplementary Fig. S2C-D), and 10 Hz discontinuous (Supplementary Fig. S2E-F) conditioning. Notably, plasticity was highly correlated between the simultaneous WC and FP recordings regardless of treatment group (control p < 0.001, sham p < 0.001, mTBI p < 0.001; Supplementary Fig. S2G) or temporal pattern (CV 0.0 p < 0.001, CV 0.2 p < 0.001, CV 1.0 p < 0.001; Supplementary Fig. S2H).
Discussion
We found that mTBI dramatically affected the dependency of plasticity on the temporal pattern of conditioning. At all frequencies studied, with highly irregular temporal patterns of synaptic conditioning, where neurons from controls underwent LTD (Fig. 4C, 5C, 6C, 7C), those from mTBI animals underwent LTP (for 1 Hz or 10 Hz; Fig. 4F, 5F, 6F) or NC (for 100 Hz; Fig. 7F). Moreover, with slightly irregular or perfectly regular temporal patterns of conditioning, neurons from mTBI animals underwent different plasticity outcomes than those from controls (LTP vs. NC for 10 Hz continuous conditioning, CV 0, cf. Fig. 5D, A; LTD vs. NC for 10 Hz continuous conditioning, CV 0.2, cf. Fig. 5E, B; and LTD vs. LTP for 10 Hz discontinuous conditioning, CV 0.2, cf. Fig. 6E, B). Finally, for slightly or highly irregular 100 Hz discontinuous conditioning, neurons from mTBI rats displayed different plasticity outcomes from controls (LTP vs. NC for CV 0.2, cf. Fig. 7E, B; and NC vs. LTD for CV 1.0, cf. Fig. 7D, A).
To our knowledge previous studies have not examined the effect of temporal pattern on the induction of LTP or LTD following TBI, but they have shown that temporal pattern affects induction of synaptic plasticity in the normal brain. Studies using 2 Hz Markovian stimulation with different second order statistics reported that strong negative correlations produced LTD, whereas more positive correlations produced LTP. 115 –117 In addition, studies using a spike timing dependent plasticity protocol evoked by paired pre-post synaptic stimulation at 0.5–3 Hz observed LTD if ISIs were regular or had a Gaussian distribution, but no plasticity (NC) if ISIs had a Poisson distribution. 118,119 A direct comparison with these studies is difficult because we used a different approach in generating our stimulation protocols, but one study 120 used a similar approach—varying the CV for ISIs—and similar to them, we found that 1 Hz perfectly regular conditioning produced LTD, but inconsistent with them, we found that 1 Hz highly irregular conditioning often did too. This discrepancy in plasticity outcomes for highly irregular conditioning may reflect subtle but important differences in the temporal pattern of ISIs in the two studies or could reflect differences in: stimulus pulse duration, species, or degree of brain maturation, including the development of synaptic inhibition.
Analyses of PSP shape index and conditioning response kinetics suggest underlying plasticity mechanisms in mTBI
Our WC patch clamp recordings of compound PSPs from L2/3 pyramidal neurons evoked by stimulation of L4 probably included a mixture of excitatory and inhibitory components to L2/3 pyramidal neurons. 121 –125 Therefore, changes in evoked PSP amplitude after conditioning could reflect an alteration in any of these components. For example, LTP of the PSP could be due to an increase in the strength of excitatory transmission, a decreased strength of inhibitory transmission, or both.
We chose not to pharmacologically isolate either component as we aimed to have the experiments directly address the type of neurostimulation that would be used in a clinical situation. However, we did apply an analysis of PSP shape indices 120,126 –129 (half width, rise time, and decay time) to our WC recordings (Supplementary Fig. S1). These results were generally consistent with LTP of the PSP being due to an increase in excitatory transmission strength and LTD being due to a decrease in excitatory transmission strength. Moreover, the FP results (Supplementary Fig. S2G–H) that are generally thought to reflect excitatory synaptic transmission 130,131 were consistent with this result.
Our results show that the frequency, continuity, and temporal pattern of conditioning stimulation interact to modify synaptic plasticity, but that interaction of these factors produces different plasticity outcomes in control (naive and sham) rats versus those with mTBI. Application of these results can also be informed by our findings on the utility of the kinetic profile of synaptic responses during the conditioning (Fig. 8–10) for predicting a desired plasticity outcome. Our utilization of probe trials (Fig. 6–7) to assess synaptic strength throughout the conditioning period could be useful to adapt a treatment protocol in real time to achieve the desired plasticity outcome. 132 –137
The increased intracellular calcium concentration and input resistance we observed following mTBI (Fig. 2) are indicative of increased neuronal excitability. 138,139 Evidence suggests that differences in the magnitude, duration, rate of rise, source, and spatial distribution of intracellular calcium signals are important for triggering LTD vs. LTP. 134,140 –146 These distinct calcium signals may act differentially on downstream signaling cascades, 147 –151 to selectively alter postsynaptic AMPA receptors 152 –157 and translation/transcription factors. 158 –163 Moreover, intracellular calcium levels can increase over larger spatial and temporal scales, for example, by entry through voltage-gated calcium channels and/or release from intracellular stores, 164 –168 which can affect processes such as gene expression and protein synthesis that contribute to more persistent synaptic plasticity. 169 –172
Implications of our results for potential therapeutic interventions in the treatment of mTBI
One potential application of our findings is to inform the design of stimulation parameters for clinical interventions such that different patterns of synaptic conditioning characterized by specific combinations of temporal pattern, frequency, and continuity could be deployed to modulate plasticity outcomes, strengthening or weakening affected neural circuits, to promote recovery of cortical function after mTBI. Recent clinical studies have demonstrated the efficacy of neurostimulation for treatment of several neurological and psychiatric conditions, including the use of: tES for Parkinson’s disease, 173 Alzheimer’s disease, 174 seizures, 175 OCD, 83 and major depression; 176 –178 TMS for Alzheimer’s disease, 174 OCD, 81 and postconcussive symptoms; 179 and DBS for Parkinson’s disease, 180 seizure disorders, 181 OCD, 182 and major depression. 183 –185 However, the relationship of various stimulation parameters to treatment efficacy is not well understood. 186 In order to optimize such approaches, the identification of the most effective stimulation parameters, including those that may elicit desired up- or downregulation of downstream synaptic networks, would be useful. Indeed, emerging methods and technologies for different stimulation modalities—DBS, 89,187 –189 TMS, 190 tES, 191 tFUS 192 –194 —have the potential to provide new insights for optimizing neurostimulation protocols. In addition, TMS, tES, and tFUS have the important advantage of eliminating the need for invasive brain surgery.
Limitations and future directions
Our study design encompassed an extensive parameter space, comprised not only of different treatment groups (control, sham, mTBI) but also different stimulation as follows: frequencies (1, 10, and 100 Hz), temporal patterns (perfectly regular, slightly irregular, highly irregular), and continuities (continuous, discontinuous). Because of this, we chose to constrain several other factors, including: sex (we used only males), age at injury (8–9 weeks), and the duration of recovery (2–3 weeks). As our study was confined to males, it remains to be determined whether similar effects would occur in females as reports have indicated time- and sex-dependent differences in cerebrovascular responses to mTBI in rodents. 195 It would also be of interest to evaluate the effects of repetitive mTBIs on synaptic plasticity. Previous studies in rodents have shown that repetitive mTBIs with shorter interinjury times involve larger tissue volumes and result in greater tissue pathology, although the effects on synaptic plasticity have not been reported. 196 Similarly, recovery time after injury could potentially be an important variable for the effects of mTBI on synaptic plasticity as other metrics of injury have been shown to change with time. 197 In addition, while our study was focused on characterizing the effects of mTBI on synaptic plasticity outcomes, an important extension of this work going forward will be to identify the mechanisms underlying these effects. For example, determination of the underlying molecular signal detection and induction processes that differentiate the regular versus irregular rises in intracellular calcium, 198 which likely occur during synaptic stimulation, may provide clues to better understanding the functional differences in normal versus mTBI brains.
Transparency, Rigor, and Reproducibility Summary
The study design and analytical plans were not formally preregistered, but they were prespecified. The planned sample size for the righting reflex experiment was 12–15 rats/treatment group (control, sham, mTBI) based on data from similar studies. 199,200 For the electrophysiology experiments the planned sample size was 15–20 cells for each of the 24 experimental groups (4 stimulation protocols—1 Hz continuous, 10 Hz continuous, 10 Hz discontinuous, 100 Hz discontinuous; 3 temporal patterns—perfectly regular, slightly irregular, highly irregular; and 2 treatment groups—control, mTBI) based on data from similar studies. 120,143,201 Given that the typical yield of successful WC recordings in these studies was about 1.5 cells/animal, we estimated that 320 rats would be required for the electrophysiology experiments. In addition, when control and mTBI results for four of the stimulation groups appeared divergent, we amended our experimental design to include 55 sham operated controls.
The actual number of rats used for the righting reflex experiments was 45 (including 1 sham excluded for a disruption during testing), whereas that for the electrophysiology experiments was 389 rats (including: 1 sham and 5 mTBI rats excluded for surgical complications; and 67 rats excluded from WC experiments because recordings were incomplete, but retained for calcium experiments because preconditioning was successful). From these animals, we obtained 446 successful WC recordings and 222 successful calcium imaging experiments. This corresponded to 15–21 cells/experimental group for WC experiments and 37–96 cells/treatment group for calcium imaging experiments. Statistical significance was determined using SigmaPlot (Systat Software) or an online service (statskingdom.com/kruskal-wallis-calculator.html). Normality was assessed with a Shapiro–Wilk test. For normally distributed data, we used a t-test or a one-way ANOVA followed by Fisher’s LSD method for multiple comparisons. For non-normally distributed data we used a MWU test or a Kruskal–Wallis test followed by Dunn’s method for multiple comparisons with Sidak correction (Dunn’s correction). Differences were considered statistically significant at p < 0.05, except for Dunn’s corrected which was considered significant at p < 0.017.
At 8–9 weeks of age, rats were randomly assigned to treatment groups by an investigator who also performed the surgeries (mTBI or sham). Two to three weeks later, a different set of investigators blinded to treatment group performed the righting reflex, electrophysiology, and calcium imaging experiments. All data collection and analysis of individual experiments (including criteria-based exclusions—see methods) were performed by the investigators blinded to treatment group.
Data are available from the corresponding author upon request. The authors have agreed to publish this article under a Creative Commons Open Access license, and upon publication it will be freely available at: liebertpub.com/loi/neu.
Footnotes
Acknowledgments
The authors thank Susanna Kiss for administrative and technical support with: blinding procedures, sham and mTBI surgeries, righting reflex experiments, and histological processing. In addition, the authors express their gratitude to: members of Dr. Claudia Robertson’s laboratory for instruction and technical assistance with mTBI and sham procedures and Dr. P. Read Montague for helpful discussions regarding the development of temporal pattern distributions.
Authors’ Contributions
Conceptualization: Q.S.F., D.K., G.V.D.P., P.R.B, and M.J.F.; Methodology: Q.S.F., D.K., G.V.D.P., P.R.B., and M.J.F.; Validation: Q.S.F. and D.K.; Formal analysis: Q.S.F., D.K., G.V.D.P., and C.A.W.; Investigation: Q.S.F., D.K., G.V.D.P., and C.A.W.; Software: Q.S.F., C.A.W., and P.R.B.; Data curation: Q.S.F., C.A.W., and P.R.B.; Writing—original draft: Q.S.F. and M.J.F.; Writing—review and editing: Q.S.F., D.K., G.V.D.P., C.A.W., P.R.B. and M.J.F.; Visualization: Q.S.F., D.K. and M.J.F.; Supervision: Q.S.F. and M.J.F.; Project administration: Q.S.F. and M.J.F.; Resources: M.J.F.; Funding Acquisition: M.J.F. All authors contributed to the article and approved the submitted version.
Author Disclosure Statement
The authors have a patent pending (U.S. Patent Application No.: 63/610,701) for the software code (scripts and algorithms) used to generate the temporal patterns of stimulation, as well as the specific temporal patterns which were used in this study. A preprint of this article is available at: biorxiv.org/content/10.1101/2023.12.13.571587.
Funding Information
This work was supported by the Department of Defense (CDMRP grant
Supplementary Material
Supplementary Data S1
Supplementary Figure S1
Supplementary Figure S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
