Abstract
The aim of our study was to investigate the biological underpinnings of persistent post-concussion symptoms (PPCS) at 3 months following mild traumatic brain injury (mTBI). Patients (n = 192, age 16–60 years) with mTBI, defined as Glasgow Coma Scale (GCS) score between 13 and 15, loss of consciousness (LOC) <30 min, and post-traumatic amnesia (PTA) <24 h were included. Blood samples were collected at admission (within 72 h), 2 weeks, and 3 months. Concentrations of blood biomarkers associated with central nervous system (CNS) damage (glial fibrillary acidic protein [GFAP], neurofilament light [NFL], and tau) and inflammation (interferon gamma [IFNγ], interleukin [IL]-8, eotaxin, macrophage inflammatory protein-1-beta [MIP]-1β, monocyte chemoattractant protein [MCP]-1, interferon-gamma-inducible protein [IP]-10, IL-17A, IL-9, tumor necrosis factor [TNF], basic fibroblast growth factor [FGF]-basic platelet-derived growth factor [PDGF], and IL-1 receptor antagonist [IL-1ra]) were obtained. Demographic and injury-related factors investigated were age, sex, GCS score, LOC, PTA duration, traumatic intracranial finding on magnetic resonance imaging (MRI; within 72 h), and extracranial injuries. Delta values, that is, time-point differences in biomarker concentrations between 2 weeks minus admission and 3 months minus admission, were also calculated. PPCS was assessed with the British Columbia Post-Concussion Symptom Inventory (BC-PSI). In single variable analyses, longer PTA duration and a higher proportion of intracranial findings on MRI were found in the PPCS group, but no single biomarker differentiated those with PPCS from those without. In multi-variable models, female sex, longer PTA duration, MRI findings, and lower GCS scores were associated with increased risk of PPCS. Inflammation markers, but not GFAP, NFL, or tau, were associated with PPCS. At admission, higher concentrations of IL-8 and IL-9 and lower concentrations of TNF, IL-17a, and MCP-1 were associated with greater likelihood of PPCS; at 2 weeks, higher IL-8 and lower IFNγ were associated with PPCS; at 3 months, higher PDGF was associated with PPCS. Higher delta values of PDGF, IL-17A, and FGF-basic at 2 weeks compared with admission, MCP-1 at 3 months compared with admission, and TNF at 2 weeks and 3 months compared with admission were associated with greater likelihood of PPCS. Higher IL-9 delta values at both time-point comparisons were negatively associated with PPCS. Discriminability of individual CNS-injury and inflammation biomarkers for PPCS was around chance level, whereas the optimal combination of biomarkers yielded areas under the curve (AUCs) between 0.62 and 0.73. We demonstrate a role of biological factors on PPCS, including both positive and negative effects of inflammation biomarkers that differed based on sampling time-point after mTBI. PPCS was associated more with acute inflammatory processes, rather than ongoing inflammation or CNS-injury biomarkers. However, the modest discriminative ability of the models suggests other factors are more important in the development of PPCS.
Introduction
Following a mild traumatic brain injury (mTBI), some patients experience persistent physical, cognitive, and emotional symptoms and/or sleep problems for over 3 months after injury, referred to as persistent post-concussion symptoms (PPCS). 1,2 The pathophysiological processes underlying PPCS are not well understood, and elucidating the predisposing factors, as well as finding cost-effective, minimally invasive, objective biomarkers of this poor outcome is an active area of research.
The prevailing biopsychosocial model of PPCS posits that a combination of biological (e.g., brain and cognitive reserve, sex, age, and pathophysiological consequences of brain trauma and/or of comorbid conditions), psychological (e.g., premorbid personality, emotional state, and cognitive factors such as illness beliefs and behavior), and social factors (e.g., social support and access to welfare systems) play a role in the etiology and maintenance of PPCS. 3,4 However, the prognostic importance of biological factors related to injury (e.g., findings on neuroimaging) and biomarkers linked to cellular damage or inflammation in peripheral blood for PPCS remains poorly understood, although these may represent important risk factors, triggers, and disease mechanisms. There are conflicting results from research on several biological factors, for example, age, 5 –8 Glasgow Coma Scale (GCS) score, 9 –11 presence of loss of consciousness (LOC), 5,12,13 duration of post-traumatic amnesia (PTA), 3,6,9,14 –16 and co-occurring extracranial injuries, 5,9,17,18 although female sex is one of the most consistently linked with higher risk of PPCS. 19 –22 Further, although diffusion tensor imaging (DTI) measures reflecting traumatic axonal injury (TAI) have been associated with PPCS, 23 –26 the findings regarding PPCS and traumatic intracranial findings (contusions, hematomas, TAI) determined from clinical scans are less consistent. 6,27 –31
Recent advances in the sensitivity of blood-based assay technologies have improved our ability to investigate blood-derived biomarkers, such as those associated with central nervous system (CNS) injury and inflammation. Multi-panels of blood biomarkers have shown diagnostic and prognostic utility in moderate-severe TBI and mixed-TBI cohorts, 32,33 but their utility in predicting post-mTBI outcomes such as PPCS calls for further study.
The most studied blood biomarkers of CNS damage are neurofilament light (NFL), glial fibrillary acidic protein (GFAP), S-100B, ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), and tau. All have shown ability to discriminate patients with mTBI from community controls and patients with traumatic intracranial findings from those without. 34 –38 Acutely measured UCH-L1 and GFAP are currently being used in the United States to assess the likelihood of intracranial injury in mTBI, 39 and S100B is recommended in Scandinavia for triaging patients with acute mTBI to computed tomography (CT) scanning. 40 Despite these proteins' well-evidenced diagnostic utility—which is confirmed in a previous article from our lab analyzing the same data 34 —there is little evidence regarding their prognostic utility for PPCS. NFL has shown the most promise as a biomarker of PPCS, with Shahim and colleagues demonstrating its discriminability for PPCS in a number of sports-related concussion cohorts, 41 –43 although replication in a mixed-mechanism mTBI cohort is needed.
Inflammation has been extensively examined in animal models of mTBI, where it has a well-validated role in modulating post-mTBI outcome. 44 Also in humans, evidence points to a role of inflammation in the pathophysiology of PPCS, 45 and possibly more so in persons with pre-injury vulnerability due to conditions known to involve the immune system, such as depression and headache. Other studies have shown the ability of inflammation biomarkers to discriminate sports-concussed patients from controls, 46 and emerging evidence suggests inflammation biomarkers may be able to discriminate patients with traumatic intracranial findings on CT and magnetic resonance imaging (MRI) from those without. 32,47 –49 Nevertheless, evidence linking systemic inflammation to PPCS is sparse, with recent research suggesting some inflammation biomarkers may be risk factors for PPCS, whereas others may reduce PPCS risk. 50,51 Many earlier studies were underpowered with a small number of inflammation biomarkers pre-selected for analysis, meaning potentially important inflammatory biomarkers may have been excluded. Further, in most previous studies blood was collected acutely, whereas few have studied inflammatory markers in a later phase, concurrent with the emergence of PPCS. It is therefore largely unknown to what degree inflammation is a hallmark of PPCS.
To help fill the knowledge gaps in the literature, our study (n = 192) investigated presence of PPCS at 3 months in a mixed-mechanism TBI cohort using a large assay of candidate biomarkers related to CNS injury and inflammation. The study is exploratory in nature, with the goal of guiding future human research on the feasibility of using blood biomarkers for predicting mTBI outcomes. Specifically, our aims were two-fold: To examine both single and multi-variable associations between PPCS at 3 months and demographic variables, injury-related characteristics, and blood biomarkers of CNS damage/inflammation, sampled at three different time-points (admission, 2 weeks, and 3 months), along with time-point differences, that is, delta value biomarker concentrations reflecting 2-week concentrations minus admission (Δ 2W) and 3-month concentrations minus admission (Δ 3M To determine the ability of candidate biomarkers to discriminate patients with PPCS from those without.
Methods
Participants and recruitment
The Trondheim mTBI study is a large-scale prospective cohort study with follow up for 12 months in patients with mTBI between 16 and 60 years of age. The study was approved by the Regional Committee for Medical Research Ethics (2013/754), and participants gave informed consent. Patients with mTBI (n = 378) were included from April 1, 2014 to December 15, 2015. They were recruited from two emergency departments (EDs): St. Olavs Hospital (Trondheim University Hospital), a regional Level 1 trauma center in Trondheim, Norway, and Trondheim Municipal Emergency Clinic, a general-practitioner-run, 24-h/7-day outpatient clinic.
Inclusion criteria were having sustained a mild TBI according to World Health Organization criteria, 52 that is, GCS score of 13–15; LOC >0 min and <30 min; and PTA >0 min and <24 h. Exclusion criteria were: (1) non-fluency in the Norwegian language; (2) pre-existing neurological, psychiatric, somatic, or substance use disorder, determined to be severe enough to interfere with follow-up and outcome assessment; (3) a prior history of a complicated mild (i.e., having trauma-related intracranial findings on CT or MRI), moderate, or severe TBI; (4) other major trauma that could interfere with follow-up or outcome assessment; or (5) presentation >48 h after the initial trauma. The sub-cohort selected for this investigation comprised patients with mTBI (see the publications by Skandsen and associates 53 and Einarsen and co-workers 54 for more details regarding patient enrollment and clinical ratings) who had blood data collected (see the publications by Clarke and colleagues 34 and Chaban and co-workers 55 for further details) and data available on PPCS.
Clinical information
Clinical information was obtained from patient interviews and medical records. LOC was rated as present only if observed. Duration of PTA was recorded as time after injury for which the patient had no continuous memory (> 0 min and <1 h, or 1–24 h). GCS score was assessed in the ED or inferred from records. 56 Presence of injuries to parts of the body other than the head (e.g., dislocations, fractures, soft-tissue injuries in need of treatment) was recorded based on self-report and ED/hospital records. Skin abrasions and contusions were not included in this rating.
Persistent post-concussion symptoms
The presence of post-concussion symptoms was assessed with the British Columbia Post-Concussion Symptom Inventory (BC-PSI) at 3 months, which is based on the International Classification of Diseases, 10th Revision (ICD-10) diagnostic criteria. 57 We used a Norwegian version that was developed in collaboration with a translator and the original author. 58 The BC-PSI contains 16 items that assess 13 core symptoms (headaches, dizziness, nausea, fatigue, noise sensitivity, irritability, sadness, nervousness, temper problems, poor concentration, memory problems, reading disability, and sleep problems), along with the severity of three life problems (alcohol tolerance, worrying about symptoms, and concern about having brain damage). Life problems were not applied in the PPCS definition in the current study. Symptom experience was rated on both frequency and intensity using 6-point Likert scales (frequency: 0 = not at all, 5 = constantly; intensity: 0 = not at all, 5 = very severe problem). Experience of life problems was rated on a 5-point Likert-scale (1 = not at all, 3 = somewhat, 5 = very much). The reported severity of each symptom and frequency of each symptom were combined into an item score between 0 and 4, following the principles outlined by Iverson and Lange. 57 Presence of PPCS was defined as having three or more total item scores of ≥3, which is considered a moderate symptom level, or a total score ≥13.
Neuroimaging
All subjects underwent a standardized brain MRI scan within 72 h of injury. 53 Because MRI has been shown to be more sensitive to intracranial traumatic findings, 54 the results from the clinical MRI readings are presented. All MRI scans were acquired with the same protocol on the same 3.0 Tesla Siemens Skyra MRI scanner with a 32-channel head coil. The protocol included three-dimensional (3D) volumes with T1-weighted (magnetization prepared rapid acquisition gradient echo); T2-weighted, fluid-attenuated inversion recovery (FLAIR); and susceptibility-weighted (SWI) scans. The clinical scans were read by neuroradiologists according to standard criteria, and the inter-rater reliability was moderate to good. 54 TAI was diagnosed and graded as described previously. 59 MRI results were dichotomized into intracranial traumatic lesions “yes” or “no” for analyses in this study—the “yes” category includes all possible findings (contusions, hematomas, and TAI). Detailed patient MRI results and their development over time are presented in a publication by Einarsen and co-workers. 54 One patient with a positive CT scan was unable to undergo MRI at inclusion, hence the reading of the CT scan was used instead of the MRI to describe TBI-related intracranial findings.
Blood samples
Time of blood sampling was measured as time from injury. Participants had their blood drawn either at admission (within 72 h post-injury), then at 2 weeks (± 3 days), 3 months (± 2 weeks), and 12 months (± 1 month). Plasma samples were obtained with ethylenediaminetetraacetic acid (EDTA) gel tubes, which were immediately put on ice and centrifuged for 10 min at 4°C at 2000g within 30 min of acquisition and then aliquoted into eight 0.5-mL Nunc tubes, which were immediately frozen at −80°C. The tubes remained stored at −80°C until two tubes were retrieved and transported in the frozen condition to the labs that analyzed the CNS injury and the inflammation makers, respectively. No freeze-thaw cycles were necessary.
Plasma GFAP, NFL, and tau concentrations were measured using the validated and commercially available Human Neurology 4-Plex A Assay (N4PA) on an HD-1 Single Molecule Array (Simoa) instrument, according to instructions from the manufacturer (Quanterix, Billerica, MA, USA). The measurements were performed in one round of experiments using one batch of reagents by board-certified laboratory technicians blinded to the clinical data. Patient samples were analyzed as singlicates. Assay variation was monitored using high and low internal control samples that were analyzed in duplicates in the beginning and end of each plate. Intra-assay coefficients of variation (CVs) were below 10% for GFAP, NFL, and tau (the N4PA assay also includes UCH-L1, but this biomarker showed CVs above 20% and was excluded from the study). All samples measured above the lower limit of quantification of the assay (GFAP: 0.467 pg/mL, NFL: 0.241 pg/mL, and tau: 0.053 pg/mL), as specified by the kit manufacturer (Quanterix, Billerica, MA).
For inflammation markers, the plasma samples were analyzed using a commercial fluorescence magnetic-bead-based immunoassay, with high-sensitivity detection range and precision (Bio-Plex Human Cytokine 27-Plex, Bio-Rad Laboratories, Inc., Hercules, CA, USA). Twenty-seven cytokines were analyzed in total: interleukin (IL)-1β, IL-1 receptor antagonist (IL-1ra), IL-2, IL-4, IL-5, IL-6, IL-7, IL-8 (C-X-C motif chemokine ligand 8; CXCL8), IL-9, IL-10, IL-12, IL-13, IL-15, IL-17, eotaxin-1 (C-C motif chemokine ligand 11; CCL11), basic fibroblast growth factor (FGF-basic), granulocyte colony stimulating factor (GCSF), granulocyte-macrophage colony stimulating factor (GM-CSF), interferon gamma (IFN-γ), IFN-γ-inducing protein 10 (IP-10; CXCL10), monocyte chemoattractant protein 1 (MCP-1; CCL2), macrophage inflammatory protein-1-alpha (MIP-1α; CCL3), macrophage inflammatory protein-1-beta (MIP-1β; CCL4), platelet-derived growth factor-BB (PDGF-BB), RANTES (CCL5), tumor necrosis factor (TNF), and vascular endothelial growth factor (VEGF).
The analyses were performed according to the manufacturer's instructions. Briefly, plasma samples were diluted 1:4 in Sample Diluent (Bio-Rad Laboratories, Inc.). A lower detection limit for the cytokines in the low picogram/milliliter range (< 20 pg/mL for all cytokines) was determined automatically by the software based on the standard curve for each inflammation marker. Only markers that were present in methodologically and clinically meaningful amounts, according to our previous experience, 60 in more than 75% of all samples during the observation period, were selected for further study (n = 12, see Results section). The remaining 15 were regarded as not present in the blood in meaningful amounts and therefore were excluded from further analyses.
Statistical analysis
Frequencies and percentages were used to summarize demographic and clinical categorical variables. Mean, standard deviation, median, interquartile range, and range are presented in Supplementary Table S1 for blood CNS-injury and inflammation biomarkers, separated by PPCS at each time-point (admission, 2 weeks, and 3 months).
Chi-square or Fisher exact tests were used to compare the proportions of male or female sex, causes of injury, GCS scores, LOC, PTA, and presence or absence of traumatic intracranial findings on MRI in patients with PPCS and those without.
To investigate whether there were group differences in biomarker levels between those with PPCS and those without, while accounting for temporal effects, linear mixed model (LMM) analyses were conducted. Time, group, and a time-by-group interaction were entered as fixed effects, with individual biomarkers as outcome. To account for within-subject correlations, a covariance structure for the total residuals was selected among a set of candidate models: (1) a model with an unstructured correlation matrix and homogeneous residual variance (UC-model); (2) a random intercept only model (RI-model), which implies a compound symmetry covariance structure; and (3) a random intercept model with heterogenous residual variances (HV-model).
A fully unstructured covariance structure, including heterogeneous variances, was ruled out due to lack of convergence of the fitting algorithm. Model fit was assessed using the Akaike information criterion (AIC) and log-likelihood ratio (LR) tests, aiming at selecting the most parsimonious model with an acceptable model fit. Models with lower AICs are considered to have better model fit, and LR tests assess the statistical significance of an improvement in model fit comparing two models. Due to multiple LR tests, the selection of covariance structures was based on a joint, pragmatic assessment of the AIC and the p-values from the LR-tests, without considering a specific threshold of significance. An effect of time with no effect of group was not of interest. Candidate biomarkers previously determined to deviate from normality 34,55 were log-transformed prior to being input into the model.
To determine the best combination of candidate biomarkers, injury-related and demographic characteristics in predicting PPCS, best-subset variable selection was used. This technique fits all possible logistic regression models using the given predictor input set and outputs a list of candidate models. Model fit was assessed using the Akaike Information Criterion corrected (AICc), 61 wherein lower values indicate better model fit. AICc values within 2 units from the best model are considered equally viable models with substantial support. Therefore, a model-averaged importance of terms plot was generated for the variables in the models with AICc values within 2 units from the best model. The plot indicates the relative importance weights of each variable, which is the proportion of times the variable was included in the assessed models (e.g., a relative importance weight of 0.8 indicates the variable was included in 80% of assessed models). The best model was chosen as the one in which all variables had relative importance weights >0.8. 62 If a candidate model where all variables had importance weights >0.8 was not available in the subset selections, the model including the next most important variable was chosen. 62 Separate models were calculated for each time-point of blood sampling (admission, 2 weeks, and 3 months), with all models including demographic and injury-related characteristics as possible predictors.
To assess the contribution of the biomarker temporal profiles in predicting PPCS, delta values were calculated, that is, biomarker concentrations at Δ 2W and Δ 3M. Single biomarker associations were assessed by regressing both calculated delta values for each candidate biomarker onto PPCS, controlling for age and sex. Full results for both standardized and unstandardized variables are presented in Supplementary Table S2. The delta values of each biomarker were then used as predictors in best-subset logistic regression models with PPCS as outcome, using the procedure described above, with one model for each time-point difference (Δ 2W and Δ 3M) and demographic and injury-related characteristics included as possible predictors. Therefore, a total of five best-subset models were computed (admission, 2 weeks, 3 months, Δ 2W, and Δ 3M). Continuous variables were standardized before inclusion in models. Unstandardized coefficients are presented in Supplementary Table S3. The model-averaged variable importance plots for each model are presented in Supplementary Figure S1.
The ability of each biomarker—at its respective time-points—to discriminate patients with PPCS from those without, was assessed with receiver operating curves (ROCs) and area under the curves (AUCs). The optimal pair of sensitivity and specificity was defined as the one corresponding to the Youden's J statistic. 63 To assess whether a combination of blood biomarkers could improve discrimination compared with single biomarkers, the best-subsets procedure described above was performed using only the 15 candidate biomarkers (the 12 inflammation and 3 CNS-injury biomarkers) as potential predictors (demographic and injury-related characteristics were not considered). Three models were generated, one for each time-point. Optimal AUCs were calculated on these best-subset models.
To provide protection against false-positives due to multiple comparisons, the significance level for all comparisons was set to 0.01.
Statistical analyses were performed using R version 4.2.2. 64 Chi-square tests were performed using the CrossTable function in the gmodels package. 65 LMMs were computed using the nlme package. 66 Single biomarker logistic regressions were performed using base R. 64 Best-subsets regressions were computed using the glmulti package. 67 AUC and ROC curves were computed using the pROC package. 68
Results
Figure 1 summarizes the group numbers and reasons for dropout. Two hundred seven had blood data at one or more time-points and 191 responses to the PPCS interview were recorded. By 3 months, 172 patients with mTBI remained in the study, giving a retention rate of 83%. The temporal profiles of all biomarkers, separated by PPCS group, are visually presented in Figure 2.
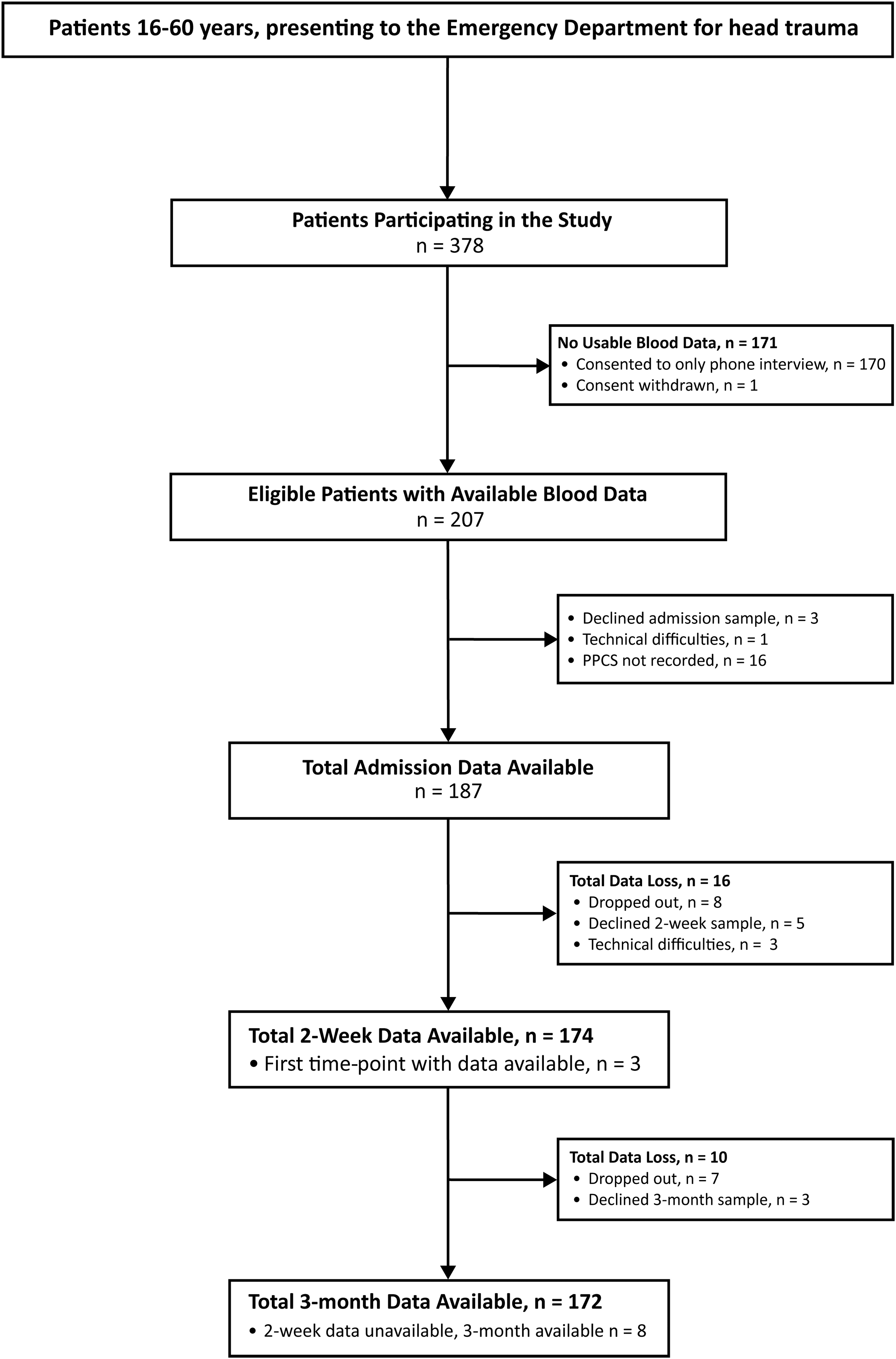
Enrollment and follow-up of patients with mTBI. mTBI, mild traumatic brain injury; PPCS, persistent post-concussion symptoms.
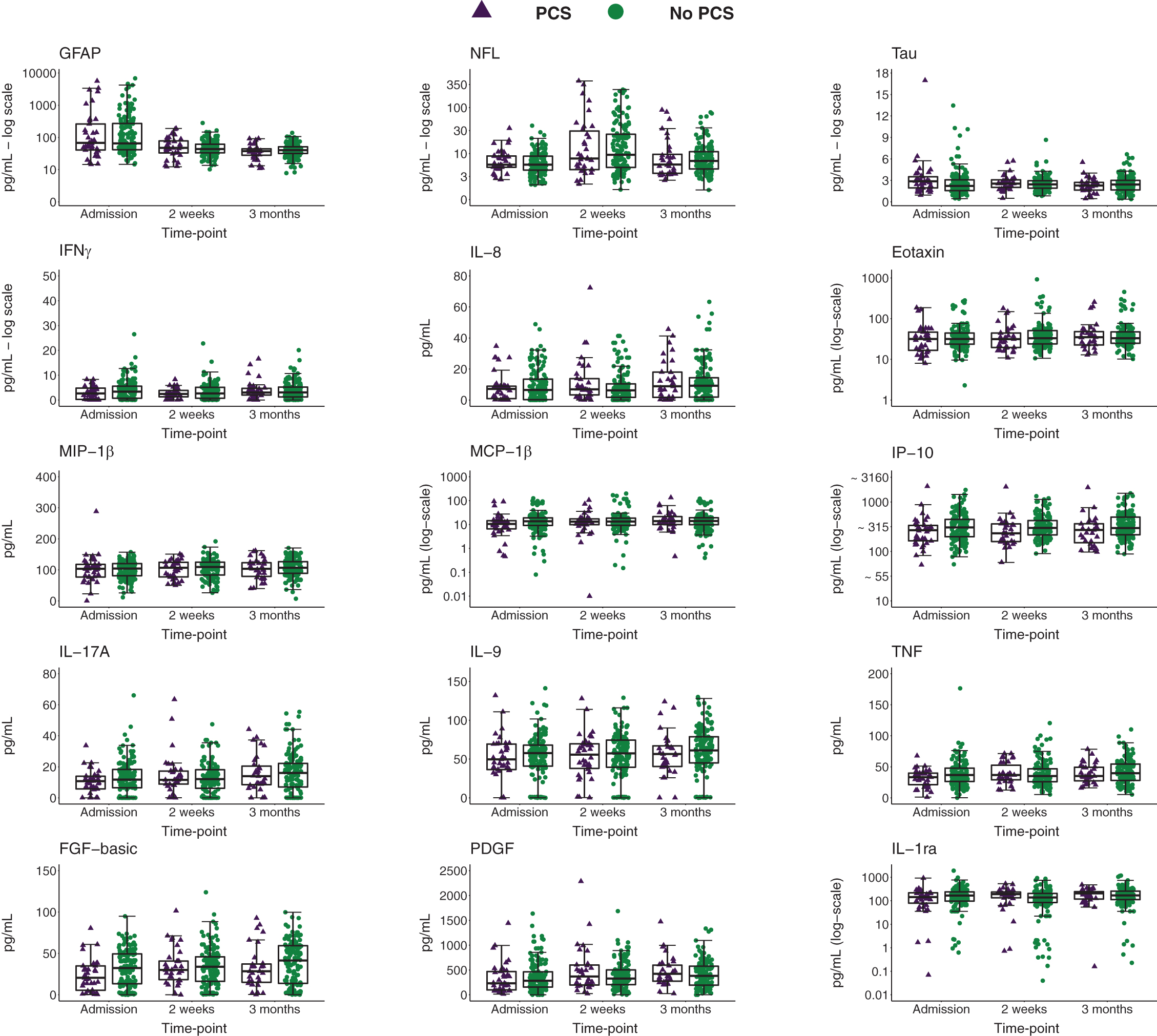
Biomarker concentrations over time in patients with PPCS versus without. Data are presented as boxplots with median as the midline, box borders representing the 25th and 75th percentile, and whiskers calculated as the 25th and 75th percentile +1.5 * interquartile range. Points above and below the whiskers represent outliers. Individual data points are presented within the boxplots. GFAP, NFL, eotaxin, MCP-1, IP-10, and IL-1ra are presented on a log-transformed scale. FGF-basic, basic fibroblast growth factor; GFAP, glial fibrillary acidic protein; IFNγ, interferon gamma; IL, interleukin; IP, IFNγ-induced protein; MCP, monocyte chemoattractant protein; MIP, macrophage inflammatory protein; NFL, neurofilament light; PDGF, platelet-derived growth factor; TNF, tumor necrosis factor.
Table 1 provides a detailed summary of the demographic and clinical characteristics of the patients with mTBI included. Most patients were male (62.8%). Most had less severe injuries, with GCS scores of 15 in 77.5%. LOC was observed in 48.7% and 29.8% had PTA between 1 h and 24 h. Traumatic intracranial findings on neuroimaging at 72 h were present in 11.0% of cases and 37.2% experienced concurrent extracranial injuries. The incidence of PPCS was 20.4%.
Participant Characteristics
Extracranial injuries refer to the presence of concurrent injuries to parts of the body other than the head (e.g., bone fracture).
mTBI, mild traumatic brain injury; GCS, Glasgow Coma Scale; LOC, loss of consciousness; MRI, magnetic resonance imaging; PPCS, persistent post-concussion symptoms at 3 months assessed with the British-Columbia Post-concussion Symptoms Inventory (BC-PSI); SD, standard deviation; TAI, traumatic axonal injury.
Factors associated with PPCS in single variable analyses
Patients with traumatic intracranial findings on MRI and PTA between 1 h and 24 h were significantly more likely to develop PPCS (Table 2). Table 3 presents the LMM results comparing concentrations of blood CNS-injury and inflammation biomarkers across time between patients with PPCS versus those without. No significant effects were found. In the delta value analyses, statistically significant effects on PPCS were found for Δ2W IL-17A (βstandardized = 0.67, p = 0.01, OR = 1.95 [1.21–3.33]) and Δ2W FGF-basic (βstandardized = 0.66, p = 0.01, odds ration [OR] = 1.94 [1.18–3.27]). No other significant differences were found. See Supplementary Table S2 for full results.
Differences in Demographic and Injury-Related Characteristics in Participants With and Without PPCS at 3 Months
Chi-square test used unless otherwise noted.
Fisher's exact test used. Significant differences are bolded (α = 0.01); bextracranial injuries refer to the presence of concurrent injuries to parts of the body other than the head (e.g., bone fracture),
GCS, Glasgow Coma Scale; LOC, loss of consciousness; MRI, magnetic resonance imaging; mTBI, mild traumatic brain injury; PPCS, persistent post-concussion syndrome as measured using the British-Columbia Post-concussion Symptoms Inventory (BC-PSI); PTA, post-traumatic amnesia.
Results of Linear Mixed Models Assessing Differences in Biomarker Concentrations in Those With PPCS Versus Those Without at Each Time-Point
P-values of the interaction effect from the fitted models; b p-value of the main effect from main-effect only models (no interaction term); cindicates base-10 log transformed data.
Estimate refers to mean time-point differences as estimated by the linear mixed model; 95% CI is the 95% confidence interval of the estimated time-point difference.
FGF-basic, basic fibroblast growth factor; GFAP, glial fibrillary acidic protein; IFNγ, interferon gamma; IL, interleukin; IP, IFNγ-induced protein; MCP, monocyte chemoattractant protein; MIP, macrophage inflammatory protein; NFL, neurofilament light; PDGF, platelet-derived growth factor; TNF, tumor necrosis factor.
Multi-variable combination of factors that best predict PPCS at 3 months
Table 4 presents the combination of biomarker concentrations at each time-point and each delta value, along with demographic and injury-related variables, determined to provide the best model fit for predicting PPCS. Unstandardized beta values are presented in Supplementary Table S3. In the admission model, the inflammation biomarkers IL-8, IL-9, MCP-1, IL-17A, and TNF were included in the best models. The associations with PPCS risk were positive (increased risk) for IL-8 and IL-9, whereas they were negative (decreased PPCS risk) for MCP-1, IL-17A, and TNF. At 2 weeks, the inflammation biomarkers IFNγ and IL-8 were included in the best models. The association was positive for IL-8 and negative for IFNγ. At 3 months, only PDGF was included in the model and the association was positive. Both greater Δ 2W and Δ 3M TNF concentrations, along with greater Δ 2W PDGF concentrations and greater Δ 3M MCP-1 concentrations, were positively associated with PPCS, indicating increased PPCS risk. Conversely, greater Δ 2W and Δ 3M IL-9 concentrations were negatively associated with PPCS, indicating decreased PPCS risk.
Results of the Best-Subsets Logistic Regression Predicting PPCS at 3 Months
Standardized regression coefficients (β), standard errors (
Variables assessed as possible predictors but not included in any models are: GFAP, NFL, tau, eotaxin, MIP-1β, FGF-basic, IL-1ra, IP-10, age, loss of consciousness, and extracranial injuries.
Reference category for Sex was male; breference category for PTA duration of between 1 and 24 h was PTA <1 h; creference category for MRI findings was No MRI finding; dreference category for GCS scores of 13–14 was GCS score of 15; eindicates log transformed data.
95% CI, 95% confidence interval; CNS, central nervous system; GCS, Glasgow Coma Scale; IFNγ, interferon gamma; IL, interleukin; IP, IFNγ-induced protein; MCP, monocyte chemoattractant protein; MRI, magnetic resonance imaging; PDGF, platelet-derived growth factor; PTA, post-traumatic amnesia; TNF, tumor necrotic factor.
All five models retained the variables traumatic intracranial MRI findings and GCS score in the best model, whereas sex was included in the 2-week, 3-month, and Δ 2W models and PTA duration was included only in the admission model. The estimated ORs in the best models indicate a positive association (OR >1) between female sex, longer PTA duration, presence of traumatic intracranial findings on MRI and lower (more severe) GCS scores, and PPCS, each adjusted for the other selected variables.
Discriminability of blood biomarkers for PPCS at 3 months
Figure 3 and Table 5 illustrate the ROC curves, sensitivities, specificities, and AUCs of the models resulting from the best combination of biomarkers in predicting PPCS at each time-point. Discriminability was moderate to poor (0.62 < AUCs <0.73). Supplementary Figure S2 shows ROC curves discriminating patients with PPCS from those without for each candidate biomarker at each time-point. Supplementary Table S4 provides the corresponding AUCs, biomarker thresholds, and sensitivities and specificities determined by Youden's J statistic. No biomarker at a single time-point was able to discriminate PPCS well, with many AUCs performing around chance (0.44 < AUCs <0.63).
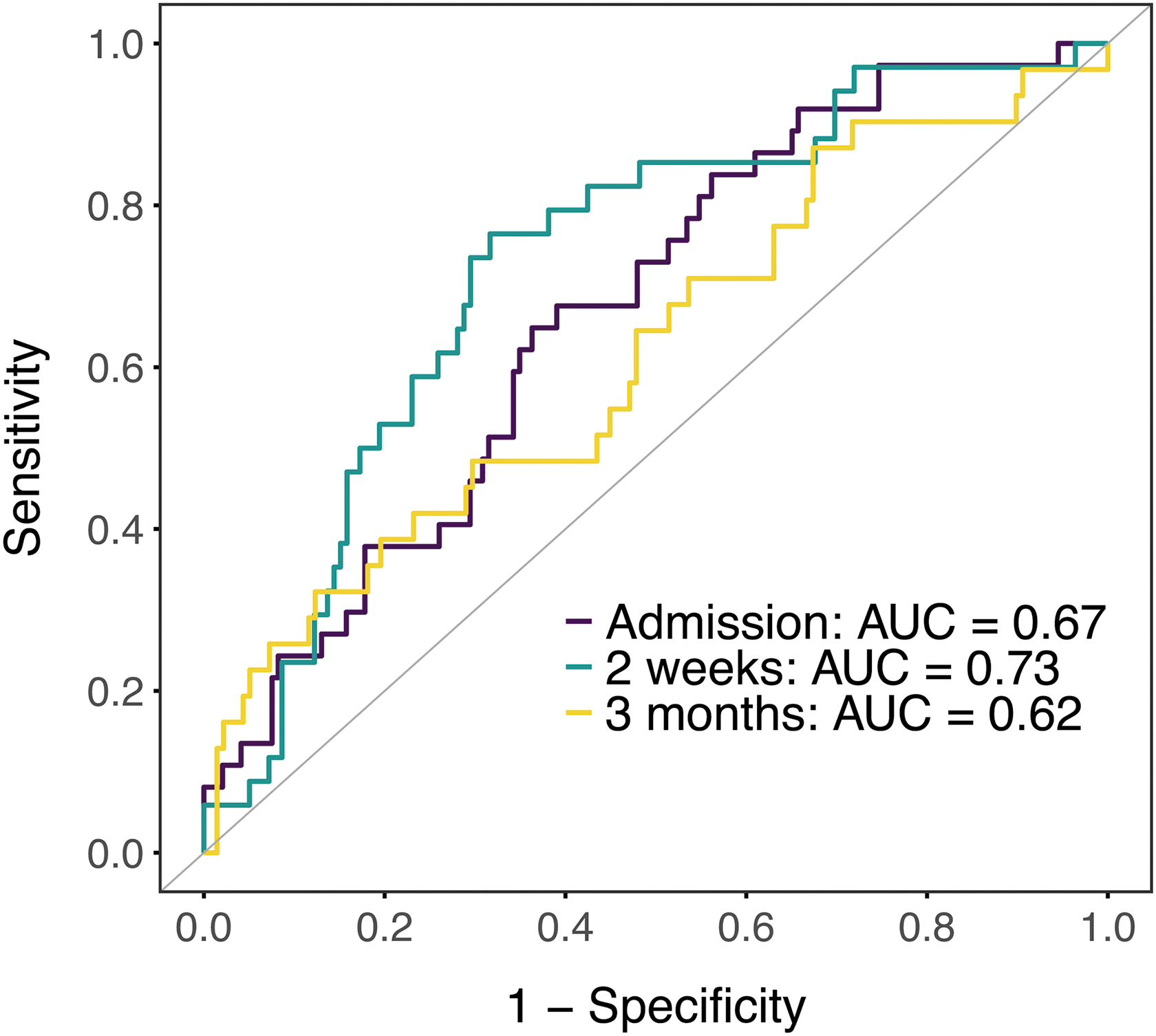
ROC curves indicating the diagnostic accuracy of the best-subsets combination of biomarkers for discriminating patients with PPCS from those without. ROC curves based on the best combination of biomarkers for predicting PPCS at each time-point are presented. AUC values for each time-point are indicated in the plot. Biomarkers included: admission: TNF, FGF-basic, and IL-9; 2 weeks: IP-10, eotaxin, IL-8, and IFNγ; 3 months: IP-10. AUC, area under the curve; FGF-basic, basic fibroblast growth factor; IFNγ, interferon gamma; IL, interleukin; IP, interferon gamma-induced protein; PPCS, persistent post-concussion syndrome; ROC, receiver operating characteristic; TNF, tumor necrosis factor.
Sensitivity, Specificity, and AUCs According to Youden's Index of the Best-Subset Models at Each Time-Point for Discriminating PPCS Cases
Only blood biomarkers and not demographic or injury characteristics are included as candidate predictors. Variables assessed as possible predictors but not included in any models are: GFAP, NFL, Tau, MIP-1β, MCP-1, IL-17A, PDGF, and IL-1ra.
95% CI, the 95% confidence interval of the estimated AUC; AUC, area under the curve; FGF-basic, basic fibroblast growth factor; IFNγ, interferon gamma; IL, interleukin; IP, interferon gamma-induced protein; PPCS, persistent post-concussion syndrome as measured using the British-Columbia Post-concussion Symptoms Inventory (BC-PSI); TNF, tumor necrosis factor.
Discussion
In this large-scale prospective study of patients with mixed-mechanism mTBI, we found novel multi-variable associations between blood inflammation biomarkers and PPCS at 3 months. Both positive and negative associations were found at single time-points, as well as when assessing changes in biomarker concentrations over time. These exploratory results will help inform future studies on the relation between inflammation and PPCS, and more broadly, mTBI. Further, we found associations between PPCS and injury characteristics, such as intracranial traumatic findings on MRI and PTA.
Single variable associations with PPCS at 3 months
Greater Δ2W IL-17A and FGF-basic values were significantly associated with PPCS. A substantial literature implicates a higher IL-17A level in adverse CNS outcomes, such as neuronal damage following ischemic brain injury 69,70 and neurodegenerative diseases. 71 A similar relationship could exist in mTBI with negative consequences following mTBI when IL-17A increases after the acute phase. Conversely, FGFs have been shown to play a crucial role in the repair of the brain following injury, and are currently being investigated as therapeutic targets in spinal cord injury. 72 Therefore, our findings could be an indication that delayed activation of otherwise beneficial mechanisms is associated with poorer outcome.
In terms of injury-related variables, longer PTA duration and intracranial findings on MRI were significantly more frequent in those with PPCS than those without. Findings regarding PTA are in line with a small subset of studies, 15,16 but not with the majority of the literature. 3,6,9,14 Given our sample size is larger than previous cohorts, and the fact that PTA is strongly related to outcome in more severe injuries, 73 we propose that similar pathophysiological consequences exist also in mTBI, but that studies may have previously been underpowered. The effect of PTA on PPCS in a larger sub-sample of the same cohort was previously reported on. 74 Our results regarding traumatic intracranial findings are in contrast to much of the literature, 6,28 –31 but in line with recent findings, 27,75 and with our previous work using a similar subset from the same cohort that linked intracranial findings on CT to PPCS. 74 We extend those findings here by evidencing contributions of intracranial findings measured using MRI on PPCS, in addition to the personal factors determined in the previous investigation, such as working less than full time before injury, pre-injury pain, and poor sleep quality. 74
We did not find a significant difference in the proportion of males and females with PPCS in this sample. Given the effect of sex is a well-validated finding, 19 –22 and authors from our lab have found significant sex effects analyzing a larger sample of the same data set including this subset of individuals, 74,76 it is likely that this effect would be significant with a greater number of PPCS cases.
Similarly, using LMMs, no single blood biomarker showed a significant group-by-time interaction or main effect of group, indicating it likely will not be viable to use a single biomarker to estimate the likelihood of developing PPCS. Nonetheless, from the contrasts at each time-point, we can see general trends that contribute to our understanding of biological mechanisms following mTBI. For instance, the difference in TNF was very large at admission, very small at 2 weeks, and larger again at 3 months. There were greater amounts of PDGF at admission in those who develop PPCS, but this difference reversed at 2 weeks, with greater PDGF in those without PPCS. Other group differences in biomarkers also reverse sign over time, for example, IFNγ, IL-8, MIP-1β, IL-17A, FGF-basic, and IL-1ra.
Multi-variable combination of factors that best predict PPCS at 3 months
Most interesting are the associations found between biomarkers of peripheral inflammation and PPCS in multi-variable models. The link between peripheral inflammation and neuroinflammation in TBI remains to be fully elucidated, although activation of peripheral immunity is believed to arise from reduced integrity of the blood–brain barrier (BBB). 77 The importance of inflammation biomarkers on PPCS indicated in this article suggests BBB breakdown could play a role in activation of peripheral immunity and PPCS onset. Further, some authors have suggested that PPCS is reminiscent of a “post-inflammatory brain syndrome” (PIBS), 44 given neuroinflammation's association with, for instance, post-traumatic headaches. 78 Our results lend credence to this, especially given a greater number of inflammation biomarkers were included in the admission model compared with 2 weeks or 3 months. However, we also demonstrate the complex nature of inflammation, given we found both positive and negative associations between inflammation markers and PPCS that differ based on sampling time-point.
PPCS was associated with lower concentrations of TNF at admission and IFNγ at 2 weeks indicating potential protective effects of both biomarkers. The mobilization of microglia to injured tissue is thought to represent a “first line” defense following neural insult. 79 Microglia in turn stimulate the release of pro-inflammatory markers such as TNF and IFNγ, which activate macrophages important for neuroprotection and repair. 79 A study using knockout mice showed that deletion of TNF-secreting genes led to reduced long-term neurological improvement following cortical contusion, suggesting a neuroprotective effect of TNF. 80 Our results support the presence of a similar effect in humans, and are in line with a recent study of mTBI in veterans, showing a negative association between TNF and a measure of PPCS symptom severity. 81
Similarly, intraperitoneal injection of IFNγ at 1, 3, and 7 days following brain injury in a mouse model of TBI led to increased number of astrocytes and microglia and enhanced production of neurotrophins, thereby promoting survival of cortical neurons. 82 The association between IFNγ and PPCS found at 2 weeks (but none at the acute time-point) suggests that protracted or continued production of IFNγ, rather than early elevations, could have beneficial effects in mTBI. Further, the TNF results taken together show that TNF concentrations in the PPCS group were lower than in the non-PPCS group at admission, becoming similar by 2 weeks and remaining so at 3 months. This provides further evidence for the importance of TNF elevation at admission for recovery following mild TBI.
PPCS was associated with lower IL-17A and MCP-1 concentrations at admission, indicating elevated early concentrations may be a protective factor for PPCS. Recent studies have shown IL-17A can prevent apoptosis during acute neuroinflammation. 83,84 IL-17A also stimulates secretion of MCP-1, 85 which is a cytokine important in recruiting monocytes to the area of injury. 86 Studies have shown that MCP-1 antagonism delays lesion reduction and reduces macrophage accumulation in an animal model of severe TBI. 87 Our results regarding IL-17A are in line with Vedantam and colleagues, 51 who showed higher post-concussion symptoms at 1 week in those with lower IL-17A. These findings must be considered in conjunction with the single biomarker effects of Δ2W IL-17A and Δ 3M MCP-1, which conversely show that increased concentrations of both markers at later time-points compared with admission are associated with increased PPCS risk. As previously described, delayed elevations of otherwise beneficial cytokines may therefore be associated with poorer outcome.
PPCS was associated with higher concentrations of both IL-8 and IL-9 at admission and IL-at 2 weeks. IL-8 is a known mediator of the pro-inflammatory response, inducing chemotaxis and phagocytosis of neutrophils, which are vital for cleaning up neural debris. 88 However, prolonged IL-8-mediated inflammation is suggested to contribute to persistent neural damage. 89 Our study provides novel evidence of blood IL-8's association with poor outcome in mTBI, in both acute and subacute phases. Increased IL-9 in the brains of TBI rodent models has been evidenced in a few studies, 90,91 and using much of the same data we confirmed a similar elevation in blood in human TBI versus a control cohort. 55 In general, IL-9 has not often been researched in relation to mTBI outcome as in the current study. Additionally, both Δ 2W and Δ 3M for IL-9 were negatively associated with PPCS, indicating that higher concentrations of IL-9 in the acute injury phase may be detrimental, but when IL-9 concentrations increase in the subacute and chronic phases, this could represent beneficial mechanisms. The Δ 3M beta value (−1.27) was the largest of all delta differences, warranting further investigation of IL-9 in relation to biological effects on the brain and outcomes over time.
The 3-month model assessed blood biomarker levels measured concurrently with PPCS. The only biomarker included in the 3-month model was PDGF, where there were increased levels of PDGF associated with PPCS. Increases in PDGF at 2 weeks compared with admission (Δ 2W) were also associated with PPCS. PDGF is a growth factor that has an essential role in tissue regeneration and wound healing by promoting angiogenesis and normal coagulation, 92 suggesting a prolonged repair process mediated by PDGF may be associated with poor outcome in mTBI. Further, PDGF signaling has been associated with aberrant BBB permeability following stroke, 93 but to which extent this could extend to mTBI remains speculative.
Traumatic intracranial findings and GCS were included in all models and PTA duration was included in the admission model, adding to the evidence for biological contributions to PPCS. Nevertheless, most patients presenting to health care with PPCS do not have any traumatic findings on neuroimaging, as was also seen in this study, which underscores the multi-faceted nature of PPCS.
Blood biomarkers' discriminability for PPCS at 3 months
In this study, we report discriminability coefficients of PPCS close to chance when using individual candidate biomarkers. Discriminability improved by using a combination of biomarkers (Table 5), although none attained the suggested clinically meaningful levels of >0.80. 63 The modest discriminative ability of the models and individual biomarkers support the increasing literature showing that other factors, such as personal factors, 74 are also involved in the development of PPCS.
Biological factors not associated with PPCS at 3 months
It is also pertinent to discuss which variables showed no significant group differences and were not included in multi-variable models predicting PPCS. Using our data-driven best-subsets approach, we can infer that the group of variables included in the models predict PPCS above and beyond variables not included. Therefore, the combined predictive power of the included clinical injury variables, sex and the inflammation biomarkers, eclipses that of age, extracranial injuries, and the CNS-injury blood biomarkers GFAP, NFL, and tau. Given previous studies have shown effects of NFL 41 –43 and age 5,7,8,94 on PPCS, our results inform the interpretation of the relative importance of these variables when injury-related information is available, whereas the lack of association between GFAP and tau is in line with previous literature. 95
The inflammation biomarkers MIP-1β and IL-1ra were not included in any models, suggesting the detrimental effects of MIP-1β found in some studies of moderate-severe TBI 96 do not extend to mTBI, which is also in line with a recent articcle showing no relation of MIP-1β to days to recovery following sports concussion. 97 Moreover, the negative effects of IL-1ra evidenced on other measures of functional outcome following mTBI 45 may not extend to PPCS. Further, although FGF-basic, eotaxin, and IP-10 were not included in the best-subset models with demographic and injury-related variables, they were included in models without those variables as candidate predictors, indicating they may have prognostic utility for PPCS independent of injury-related variables. Lastly, in line with some studies, we found no association between extracranial injuries and PPCS. 5,9,17 Although this may seem counterintuitive, our results suggest more direct brain-injury-related consequences and activation of peripheral inflammatory processes have greater relevance for determining PPCS.
Limitations
There are several limitations to our study. First, the sample consists of those who consented to comprehensive data collection and may therefore not be generalizable to all patients with mTBI. The number of PPCS cases compared with the number of candidate predictors was relatively small, which may be reflective of our sample coming from a relatively “milder” subject pool of mTBI. The small number of cases compared with non-cases increases the possibility of statistical overfitting, thus potentially reducing the reliability of our findings. This is also reflected in the wide confidence intervals seen in the multi-variable models. Therefore, we present our findings in the hopes they can be used to inform future, even larger studies (such as the CENTER-TBI and TRACK-TBI cohorts) about which blood biomarkers to focus investigative efforts on.
Further, only 11% of patients exhibited intracranial findings on MRI, therefore determined effects are based on small subsets of patients. However, we consider this a consequence of recruiting a more representative sample from both the ED and ambulatory clinics. Age <60 years was an inclusion criterion chosen to reduce the burden of age-related findings on brain MRI; however, age-related effects on PPCS beyond 60 years can therefore not be determined. Regarding the assessed biomarkers, total tau was assessed, although previous literature suggests the use of phosphorylated tau or the ratio of phosphorylated tau:total tau may be more relevant in cases of TBI. 98 Similarly, IL-6 and IL-10 are two of the most studied inflammatory biomarkers in mTBI 45 ; however, they were not expressed in sufficient quantities to be detected in our multiplex assay, de facto implying no associations. All-in-all, the limitations of our article highlight the need for pooling of data across labs and rigorous meta-analyses.
Conclusions
This study contributes new evidence on the underpinnings of PPCS, evidencing effects of biomarkers of inflammation and baseline injury characteristics on PPCS at multiple time-points. Our findings indicate potentially important associations between early and prolonged peripheral inflammation on the development of PPCS, with both detrimental and beneficial effects implied depending on the inflammation biomarker. This adds to the literature demonstrating the complex nature of inflammation, showing that if an inflammation biomarker is beneficial at admission, increased concentrations at later time-points compared with admission may be detrimental, and vice versa. Further, only one association was found between an inflammatory marker sampled at 3 months and PPCS, suggesting early, rather than sustained inflammation, is more important in the development of PPCS. As a caveat to our findings, the modest discriminative ability of the models suggests other factors not measured here play a significant role in the development of PPCS.
Transparency, Rigor, and Reproducibility Summary
Data from this study come from the larger Trondheim mTBI study. The analysis plan was not formally pre-registered, but Turid Follestad (lead statistician) and Asta Håberg (lead author) certify that the analysis plan was pre-specified (and slightly adjusted based on suggestions from reviewers). Power calculations, related to the overarching study indicated that with a hypothesized frequency of PPCS of 5% among patients with mTBI with normal conventional MRI, and 20% among patients with positive findings on MRI, we needed to include 140 patients to achieve a power of 0.8 to detect a difference, given a significance level of 0.05. A total of 1095 with head CT due to trauma were evaluated for inclusion in the Trondheim mTBI study. Of those, 624 met criteria for mTBI and 459 met further inclusion criteria. A further 299 with head CT were enrolled, along with 79 patients without head CT (total = 378 patients with mTBI). Of all patients with mTBI enrolled, 207 had blood data collected and analyzed.
All biofluid samples passed systematic quality assessment. Blood from one less participant was analyzed for inflammation markers due to a transportation issue. One hundred ninety-one samples were assessed for PPCS using the BC-PSI. Fluid biomarker measurements, quality control, and analyses were performed by investigators blinded to relevant characteristics of the participants. Samples were acquired between April 7, 2014 and April 26, 2017. Sampling was performed between 8:00am and midnight. Blood was collected at the ED, neurosurgical ward, or the MRI facility. Blood biomarker assays were performed according to the guidelines from the company. Key inclusion criteria and clinical outcomes are standards in the field and were assessed by investigators with extensive experience in the field. Following the conventional rule of thumb regarding categorical data comparisons, if the expected frequency of a cell in comparisons with PPCS was less than 5, a Fisher's exact test was performed instead of chi-square. Multiple comparisons were handled by setting the alpha level to 0.01. Internal validation was not performed because our chosen analysis methods do not require it.
Data, including on biofluids, from the Trondheim mTBI study used in this article can be accessed by contacting the corresponding author (
Footnotes
Acknowledgments
The authors thank the staff at the Trondheim Municipal Emergency Department, the Department of Neurosurgery, the Department of Anaesthesiology, and the Department of Intensive Care Medicine for their cooperation during patient recruitment. Thanks to Stine Bjøralt for study coordination and to Jonas Stenberg, Simen Berg Saksvik, and Migle Karaliute for recruitment of trauma controls and help with the blood samples. Thank you to Biobank 1 for the storage of our blood samples and thank you to the laboratory technicians of the Clinical Neurochemistry Laboratory at the Sahlgrenska University Hospital. We thank Marit Kristina Indergaard and Ina Møller for assistance with the blood testing procedures for inflammation biomarkers. Lastly, we thank Benedikte Emilie Vindstad for her graphical design contributions.
Authors' Contributions
GC organized blood data results, performed all statistical analyses, and drafted the manuscript for intellectual content. TS (principal investigator of the Trondheim mTBI study), AV, and AH designed the study, oversaw all data collection, contributed to analysis, and planned and revised the manuscript. HZ and KB selected and oversaw CNS injury biomarker analyses, quality assessed data, and revised the manuscript. SP and TM selected and oversaw inflammation biomarker analyses, quality assessed data, and revised the manuscript. CE was involved in recruitment of participants, collected and organized demographic, clinical, and blood sample data, organized MRI data files, provided data files, and revised the manuscript. TF is the statistician who approved all statistical methods and presentation of results in writing and figures/tables. AV provided neurosurgical expertise and revised the manuscript. AH was the principal supervisor of the manuscript. All authors carefully revised the manuscript and approved the submitted version.
Funding Information
The Trondheim mBI study was funded by the Liaison Committee between the Central Norway Regional Health Authority and NTNU. In addition, the MRI study was partly funded by the National Norwegian Advisory Unit for functional MRI and the Department of Radiology and Nuclear Medicine, St. Olavs Hospital, Trondheim University Hospital. Additionally, general financial support was obtained from The Odd Fellow Foundation and The Simon Fougner Hartmann Family Fund. GC is supported by the Center for Innovative Ultrasound Solutions (CIUS) funded by the Research Council of Norway (grant 237887).
HZ is a Wallenberg Scholar supported by grants from the Swedish Research Council (2022-01018 and 2019-02397), the European Union's Horizon Europe research and innovation program (under grant agreement 101053962), and Swedish State Support for Clinical Research (ALFGBG-71320). KB holds the Torsten Söderberg Professorship in Medicine at the Royal Swedish Academy of Sciences and was supported by the Swedish Research Council (2017-00915), the Swedish Alzheimer Foundation (AF651 742881), Hjärnfonden, Sweden (FO2017-0243), and a grant (ALFGBG-715986) from the Swedish state under the agreement between the Swedish government and the County Councils, the ALF-agreement.
Author Disclosure Statement
HZ has served on scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Annexon, Apellis, Artery Therapeutics, AZTherapies, CogRx, Denali, Eisai, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave; has given lectures in symposia sponsored by Cellectricon, Fujirebio, Alzecure, Biogen, and Roche; and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). KB has served as a consultant or on advisory boards for Alzheon, CogRx, Biogen, Lilly, Novartis, and Roche Diagnostics; and is a co-founder of Brain Biomarker Solutions in Gothenburg AB, a GU Venture-based platform company at the University of Gothenburg. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
