Abstract
Concussion is a heterogeneous injury that relies predominantly on subjective symptom reports for patient assessment and treatment. Developing an objective, biological test could aid phenotypic categorization of concussion patients, leading to advances in personalized treatment. This prospective multi-center study employed saliva micro-ribonucleic acid (miRNA) levels to stratify 251 individuals with concussion into biological subgroups. Using miRNA biological clusters, our objective was to assess for differences in medical/demographic characteristics, symptoms, and functional measures of balance and cognition. The miRNAs that best defined each cluster were used to identify physiological pathways that characterized each cluster. The 251 participants (mean age: 18 ± 7 years; 57% male) were optimally grouped into 10 clusters based on 22 miRNA levels. The clusters differed in age (χ2 = 19.1, p = 0.024), days post-injury at the time of saliva collection (χ2 = 22.6; p = 0.007), and number of prior concussions (χ2 = 17.6, p = 0.040). The clusters also differed in symptom reports for fatigue (χ2 = 17.7; p = 0.039), confusion (χ2 = 22.3; p = 0.008), difficulty remembering (χ2 = 22.0; p = 0.009), and trouble falling asleep (χ2 = 17.2; p = 0.046), but not objective balance or cognitive performance (p > 0.05). The miRNAs that defined concussion clusters regulate 16 physiological pathways, including adrenergic signaling, estrogen signaling, fatty acid metabolism, GABAergic signaling, synaptic vesicle cycling, and transforming growth factor (TGF)-β signaling. These results show that saliva miRNA levels may stratify individuals with concussion based on underlying biological perturbations that are relevant to both symptomology and pharmacological targets. If validated in a larger cohort, miRNA assessment could aid individualized, biology-driven concussion treatment.
Introduction
Concussion assessment lacks robust, objective diagnostics and remains primarily based on self-report of subjective symptoms. 1 Challenges to accurate diagnosis may be attributed to the mechanistic, symptomatic, and biological heterogeneity among concussions. The dearth of clinically useful measures to personalize diagnosis, prognosis, and clinical management hampers successful treatment. Current guidelines 1 –3 for the assessment and treatment of concussion offer broad strategies that are not individualized and have limited validation in prospective trials. Individual differences in response to concussive injury (as quantified by symptoms, neurological function, and biological measures) may reflect concussive profiles or phenotypes, which could improve screening, diagnosis, and treatment.
Clinical studies have sought to characterize concussions by identifying distinct symptom profiles, 4,5 balance and neurocognitive deficits, and sleep, mood, and dysautonomic problems. This has led to various proposed taxonomies of concussion phenotypes. Several classification schema still rely primarily on self-reported symptom clusters, with some approaches including neurocognitive, autonomic, balance, vestibular, or visual assessments. 6 –8 Some of the current clinical tools in use may have limited ability to discriminate among these phenotypes because of their lack of sensitivity to mild physiological perturbations as well as the heterogeneous etiologies of concussion.
Investigations of concussion that include body fluid or physiological biomarkers, paired with detailed phenotyping of the clinical manifestations, may provide opportunities to more precisely categorize concussions. Non-coding RNAs represent a novel approach to understanding concussion phenotypes. Measures of RNA levels may provide insight into underlying disturbances that occur after concussion. Increasing evidence suggests that salivary microRNA (miRNA) levels may reflect underlying biological changes following concussion. Saliva miRNA levels change after head impacts, 9,10 differentiate individuals with concussion from non-concussed peers, 11 and display longitudinal patterns that mirror recovery trajectories. 12 These patterns may be useful in diagnosing concussion, 13 -15 or predicting the duration of symptoms. 16 However, it is unclear whether miRNA patterns may be used to identify concussion phenotypes and guide individualized, biologically driven treatment plans.
The objective of this study was to determine if saliva miRNA profiles could differentiate concussed individuals based on clinically relevant medical/demographic traits, symptomology, or functional measures of balance and cognition. We hypothesized that a set of 22 salivary miRNAs previously implicated in concussion pathophysiology (see Table 3 of cited article 14 ) would identify clusters of concussion phenotypes with physiological and clinical relevance to concussion. Sixteen of these 22 miRNAs have been identified in multiple studies, and 12 have been identified in multiple biofluids. 17
Methods
The study protocol received institutional review board (IRB) approval (Western IRB 1271583). Ethical approval was also provided by individual IRBs at the Penn State College of Medicine (STUDY00003729), SUNY Upstate Medical University (1070727), SUNY Buffalo (#00004347), Vanderbilt University Medical Center (#181814), and the Department of the Army, Regional Health Command-Atlantic (#1510001-1). Written, informed consent was obtained for all participants and written assent was provided by participants <18 years of age.
Participants
This prospective multi-center study included a convenience sample of 251 individuals (age 18 ± 7 years). All 251 individuals received a clinical diagnosis of concussion, as defined by the 2016 Concussion in Sport Group criteria. 1 Participants included individuals with sport-related and non-sport-related mechanisms of injury, who were enrolled from emergency departments (ED), sports medicine clinics, urgent care centers, concussion specialty clinics, and outpatient primary care clinics at initial clinical presentation (within 14 days of injury). At enrollment, saliva was collected from each participant via swab, medical/demographic data was collected via survey, concussion symptoms were assessed via the post-concussion symptom scale (PCSS 18 ), and balance/neurocognitive performance was measured with the ClearEdge Toolkit (Quadrant Biosciences, Syracuse, NY 19,20 ). Concussion symptoms were re-assessed ≥3 weeks post-concussion using the PCSS. Participants were enrolled from April 2017 through February 2020 at seven institutions: Adena Health System (Chillicothe, OH; n = 37), Colgate University (Hamilton, NY; n = 42), the United States Army (Fort Benning, GA; n = 6), Penn State College of Medicine (Hershey, PA; n = 112), State University of New York (SUNY) at Buffalo Jacobs School of Medicine (Buffalo, NY; n = 11), SUNY Upstate Medical University (Syracuse, NY; n = 26), and Vanderbilt University Medical Center (Nashville, TN; n = 17). Exclusion criteria for all participants were primary language other than English, pregnancy, active periodontal disease, neurological disorder (e.g. epilepsy, multiple sclerosis, hydrocephalus), drug or alcohol dependency, upper respiratory infection, having a legally appointed guardian or inability to provide consent/assent because of intellectual disability, Glasgow Coma Score (GCS) ≤12 at the time of initial injury, penetrating brain injury, hospitalization for current concussion, skull fracture, unresolved concussion symptoms, additional concussion within the previous 3 months, or intracranial bleed on imaging (if performed).
Demographic and medical data collection
Participant characteristics were collected via survey and administered by research staff in the clinical care setting. Parents assisted children (≤ 12 years) with survey completion. When possible, survey responses were confirmed through review of the medical record. The following medical and demographic characteristics were collected: age (years), sex (male/female/non-conforming), race (White, Black or African American, Asian, American Indian or Alaskan Native, and Native Hawaiian or Pacific Islander), ethnicity (Hispanic or Latinx/not Hispanic), body mass index (BMI) (kg/m2), chronic medical issues (presence/absence of attention-deficit/hyperactivity disorder [ADHD], anxiety, depression, and chronic headache disorder). The following concussion-related characteristics were collected: number of previous concussions, time since most recent concussion (days), presence/absence of loss of consciousness. For a subset of all participants (n = 208) 22 symptoms were self-reported on a Likert scale (0–6) using the PCSS. Total symptom severity (sum of all Likert scores), and total symptom burden (sum of all symptoms >0) were calculated for each participant. Presence/absence of persistent post-concussion symptoms (PPCS) >3 weeks after initial injury was assessed through self-report on the PCSS (≥ 5 symptoms), or through electronic medical record review (where available).
Balance and neurocognitive assessment
Balance was assessed for a portion of participants (n = 177) using the validated ClearEdge system, as we have previously reported. 14 ClearEdge measures body sway in three dimensions during eight different stances: two legs eyes open (TLEO), tandem stance eyes open (TSEO), two legs eyes closed (TLEC), tandem stance eyes closed (TSEC), two legs eyes open on a foam pad (TLEOFP), two legs eyes closed on a foam pad (TLECFP), tandem stance eyes open on a foam pad (TSEOFP), and tandem stance eyes closed on a foam pad (TSECFP). A scaled score of 0 indicates poor balance and 100 indicates superior balance. There were 21 participants who were unable to complete balance testing because of orthopedic injury, or extreme postural instability. The remaining participants were unable to complete the assessment because of time constraints in the clinical setting of enrollment. Computerized neurocognitive assessment was performed in the same subset of participants (n = 177) using the following tests: simple reaction time (SRT1), procedural reaction time (PRT), go/no-go (GNG), and a repeat of simple reaction time (SRT2). This battery is part of the Defense Automated Neurobehavioral Assessment (DANA 21 ). The four cognitive tests were scored for speed and accuracy using a mean throughput measure of mental efficiency, where higher scores reflect better performance. Individual scores were objectively calculated with computerized software, and overall test battery means were determined for balance and neurocognition.
Salivary RNA assessment
Saliva swabs (DNA Genotek, Ottawa Canada) were collected from all participants (n = 251) in a non-fasting state, as previously reported. 22 Following RNA extraction (miRNeasy Kit; Qiagen, Germantown, MD), RNA quality was assessed (Agilent Bioanalyzer, Illumina, San Diego, CA), sequencing libraries were prepared (TruSeq Stranded Small RNA Kit, Illumina), and sequencing was performed at a depth of 10,000,000, 50 base-pair reads/sample (NextSeq500, Illumina). Fastq files were trimmed using Cutadapt v1.2.1, 23 aligned to miRBase22 using Bowtie v1.0.0, 24 and quantified using a custom bio-informatics architecture (Human Alignment Toolchain [HATCH]). Aligned reads were quantile normalized and each miRNA feature was mean-center scaled (divided by feature standard deviation). The miRNA profile for each sample was reduced to a panel of 22 miRNAs previously shown to differentiate concussed and non-concussed individuals. 14 Missing values (1.5 % of survey measures, 0.0% of miRNA measures) were imputed with the singular value decomposition imputation method. Quantitative polymerase chain reactions (qPCR) were used to validate directionality and proportion of change for a subset of miRNAs.
Statistical analysis
Saliva miRNA levels were used to divide the 251 participants into biological clusters with a K-means clustering approach. K-means clustering was selected because it provided optimal internal validity (Davies Bouldin Index), stability (figure of merit), and biological validity (biologic homogeneity) for this data set, when compared with graph-based clustering and model-based clustering. 25 The optimal number of clusters (between 3 and 20) was determined using the elbow method. 26 Initial cluster centroids were selected to optimize the distance between clusters (Euclidean distance metric). A maximum of 1000 iterations was used to compute cluster centroids. K-means clustering analyses were performed in Partek Flow (Partek Flow, St. Louis, MO). Levels of the 22 miRNAs were compared among the biological clusters via Kruskal Wallis testing with Holm step-down multiple testing correction. For three miRNA candidates (miR-27a-5p, miR-192-5p, miR-4510), Spearman's correlations were used to repeatability with qPCR, and directionality of change across clusters was visualized using a hierarchical heat map with a Euclidan distance metric. Next, medical and demographic characteristics were compared across groups using analysis of variance (continuous variables) or χ 2 test (nominal variables). Finally, linear regression was used to assess the relationship of cluster group with subjective symptoms (scores on the PCSS) and functional symptoms (balance scores and neurocognitive scores), while controlling for medical factors that displayed between-cluster differences (age, days post-concussion, number of prior concussions). Between-group comparisons were performed using Jamovi v2.2 software. 27 Finally, miRNA function was used to explore the physiological characteristics of each cluster. Cluster-specific miRNAs (i.e., miRNAs with 1.5-fold up- or downregulation relative to the mean value for the entire cohort) were identified for each cluster group. High confidence gene targets (microT-CDS score ≥0.95, adjusted p value <0.040) were determined for each set of cluster-specific miRNAs in DIANA miRPATH v3.0 online software. 28 Over-representation of Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways by these target genes (adjusted p < 0.05) was determined using a Fisher's exact test with Bonferroni correction.
Results
Participants characteristics
Participants were predominantly adolescent (18 ± 7 years) and male (143/251, 57%) (Table 1). Underlying ADHD, anxiety, depression, or chronic headache disorders were uncommon (present in <10% of participants, respectively). Approximately one third of participants had had a prior concussion (71/251, 30%). On average, participants were enrolled 4 (± 4) days post-injury. The most common mechanism of injury was sport-related (185/251, 74%). Few participants (40/251, 16%) reported loss of consciousness immediately following their injury. There were 37 participants (15%) who reported post-concussive symptoms ≥3 weeks beyond the time of injury.
Participant Characteristics
Denotes p < 0.05 on analysis of variance (ANOVA) or χ 2 testing. No individual cluster comparisons demonstrated significant differences on Tukey's post-hoc testing. Percentages are based on total number of respondents (not total participants in the group). OTMB is determined from t-scores for all sub-tests in a performance category.
ADHD, attention deficit hyperactivity disorder; BMI, body mass index; MVC, motor vehicle collision; OTBM, overall test battery mean; PPCS, persistent post-concussion symptoms; SD, standard deviation.
Biological clusters
Using the elbow method, it was determined that the 251 concussion participants were optimally divided into 10 clusters based on their levels of 22 concussion-related saliva miRNAs (Davies Bouldin Index = 1.131; Fig. 1A). Clusters ranged in size from 6 participants (cluster 10) to 49 participants (cluster 1) (Fig. 1B). Of the 22 salivary miRNAs, 21 displayed a significant difference in expression (adjusted p < 0.05) across the 10 clusters (Fig. 1C). Clusters 2, 8, and 9 displayed the greatest similarity in miRNA expression, and generally segregated the 22 miRNAs into 10 upregulated and 12 downregulated features. Levels of miR-181c-5p displayed the greatest degree of difference across clusters (F = 46.1, adj. p = 3.0 × 10−46) (Table S1). The most significant up-regulation of miR-181c-5p was in cluster 8 (Fig 1C). RNA sequencing and qPCR results were significantly correlated (p < 0.05), and qPCR values differentiated participants into similar clustering patterns (Fig. S1). Although several additional miRNAs showed between-clusters differences, a clustering approach using 22 randomly selected miRNAs yielded inferior clustering validity (Davies–Bouldin Index = 1.407; Fig. S2).
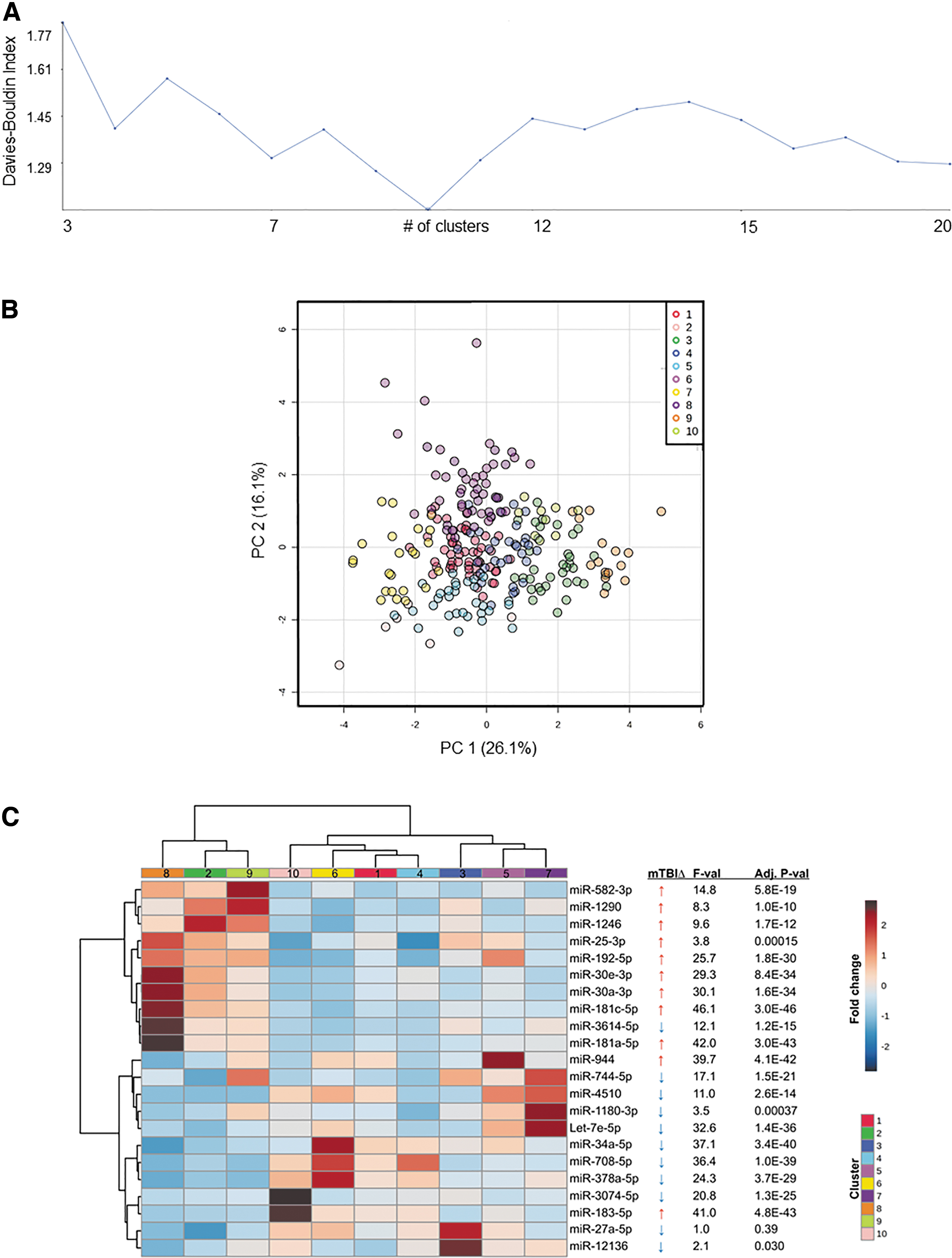
Saliva miRNA profiles divide concussions into 10 clusters. The 251 participants were divided into 10 clusters based on the salivary profile of 22 concussion-related miRNAs using the elbow method
Cluster clinical features
Medical and demographic features for the 10 cluster groups are displayed in Table 1. The 10 biological clusters differed in age (χ 2 = 19.1, p = 0.024), days post-injury at the time of sample collection (χ 2 = 22.6; p = 0.007), and number of prior concussions (χ 2 = 17.6, p = 0.040) (Fig. 2). The clusters did not differ in sex (χ 2 = 10.1, p = 0.34), race (white vs. non-white; χ 2 = 9.5, p = 0.38), injury mechanism (sport vs. non-sport; χ 2 = 10.7, p = 0.30), BMI (χ 2 = 11.6, p = 0.23), or loss of consciousness at the time of concussion (χ 2 = 28.2; p = 0.059). There was no difference in overall test battery mean for balance (F = 0.2; p = 0.9), or neurocognitive performance (F = 0.9; p = 0.5).
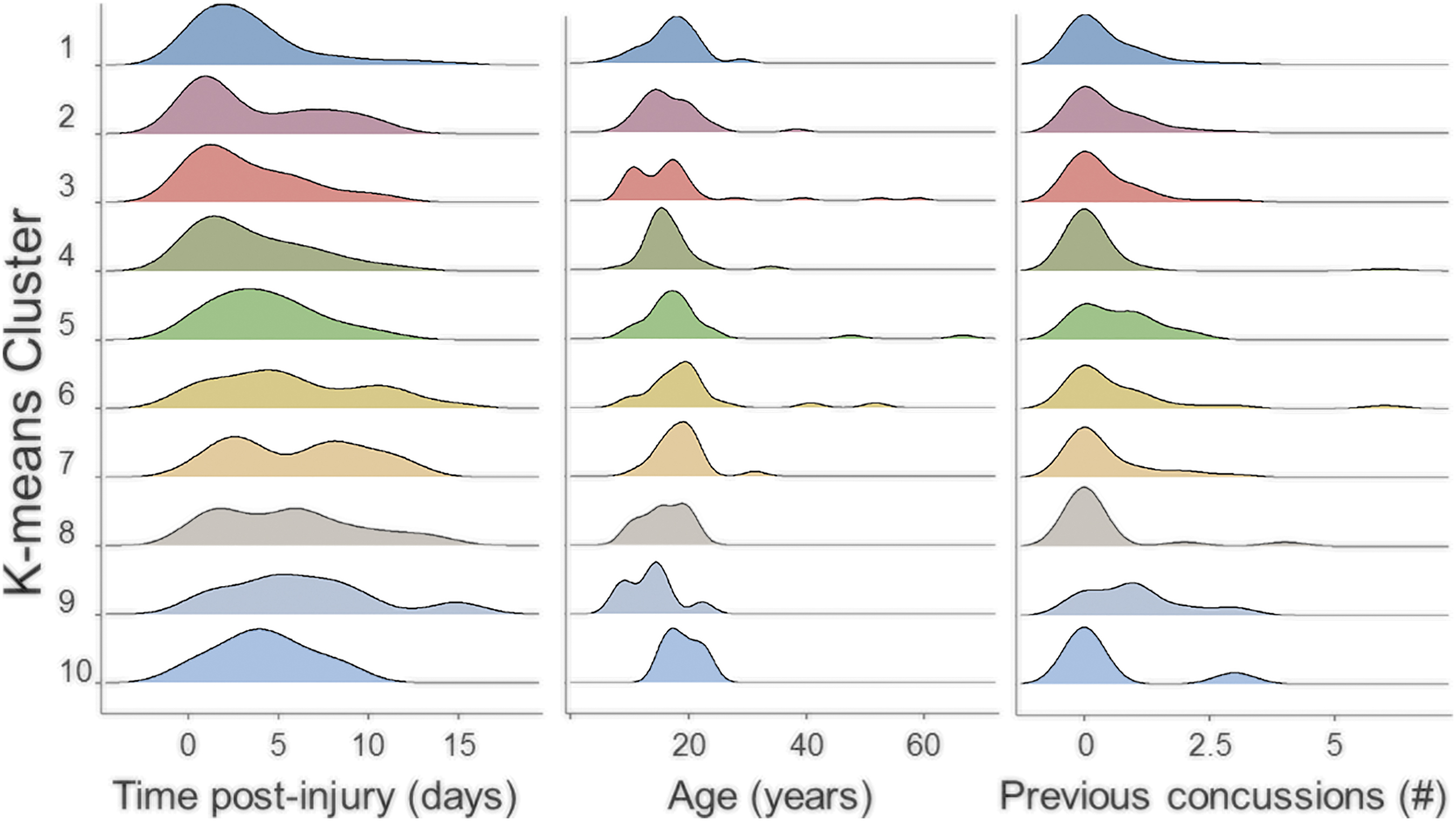
The 10 clusters differed in age, time since injury, and number of previous concussions. Density histograms display the distribution of age (in years), days since concussion at the time of saliva collection, and number of previous concussions for the 10 cluster groups. These were the only medical/demographic features that differed across groups on analysis of variance testing.
Linear regressions assessing the relationship between biological cluster and balance or neurocognitive performance (while controlling for age, days post-injury, and number of prior concussions) found no difference (p > 0.05) among clusters (Fig. 3). Ordinal regressions assessing the relationship between biological cluster and subjective symptoms (while controlling for age, days post-injury, and number of prior concussions) identified differences among the biological clusters in fatigue (χ 2 = 17.7; p = 0.039), confusion (χ 2 = 22.3; p = 0.008), difficulty remembering (χ 2 = 22.0; p = 0.009), and trouble falling asleep (χ 2 = 17.2; p = 0.046) (Fig. 4). Post-hoc tests for these four symptoms revealed the greatest degree of difference among the following biological clusters: fatigue – cluster 3 vs. 4 (odds ratio [OR] = 0.19, 95% confidence interval [CI]: 0.069–0.494; p = 0.001); confusion – cluster 5 vs. 4 (OR = 0.10, 95% CI: 0.020–0.380; p = 0.002); difficulty remembering – cluster 3 vs. 4 (OR = 0.13, 95% CI: 0.041–0.36; p = 0.001); and sleep difficulties – cluster 1 vs. 3 (OR = 8.0, 95% CI: 2.3–37.6; p = 0.002) (Fig. S3).
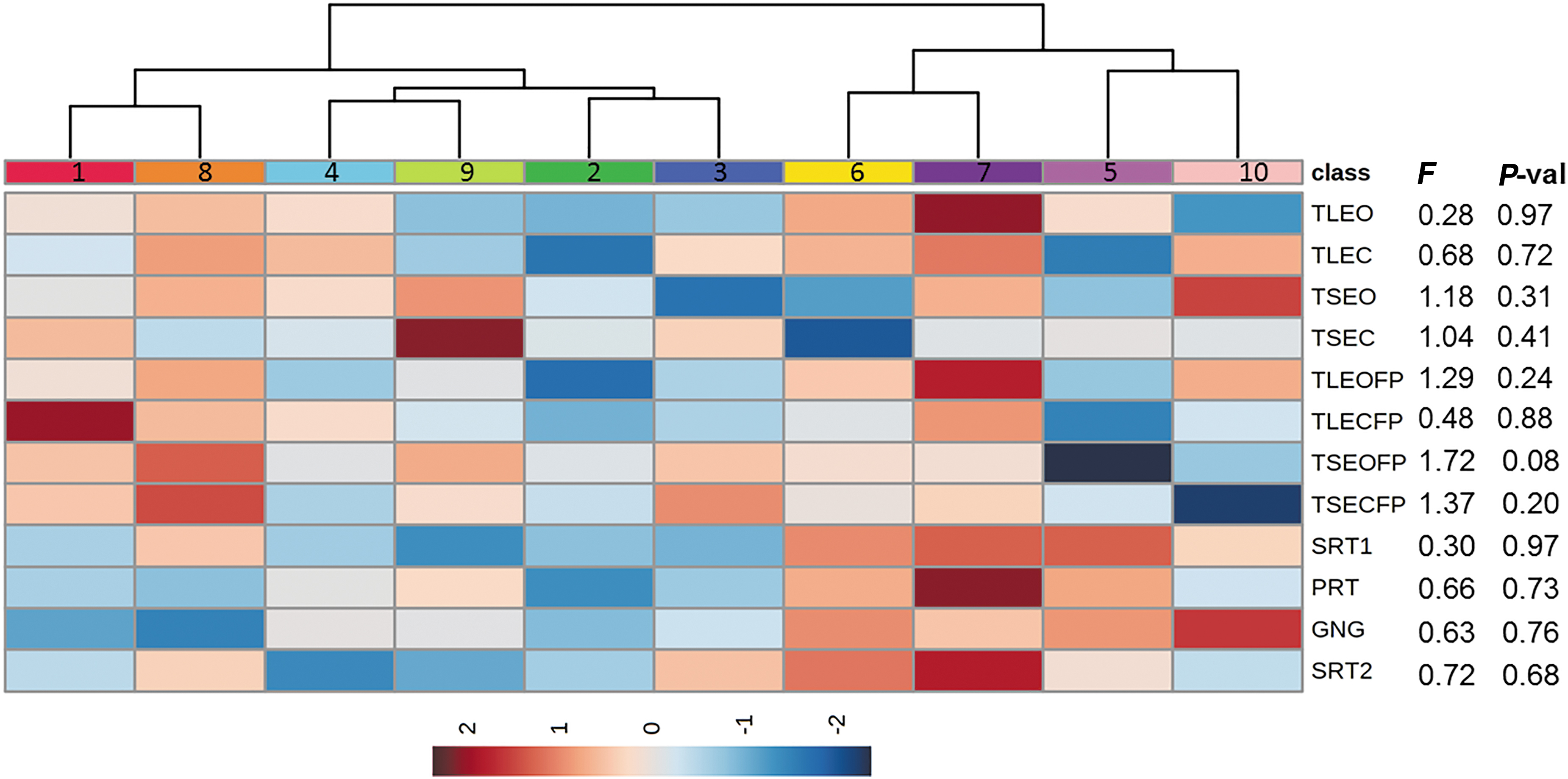
Patterns of neurocognitive and balance performance across the 10 clusters. A linear regression analysis was used to assess the relationship of cluster group with neurocognitive and balance performance, while controlling for age, days post-injury, and prior concussions. No functional tests displayed a significant difference (p < 0.05) among clusters on omnibus analysis of variance (ANOVA) (F statistic and p value are shown). A hierarchical clustering heat map with a Euclidean distance metric was used to visualize functional performance patterns across the 10 clusters, where red denotes superior performance on balance or neurocognitive testing.
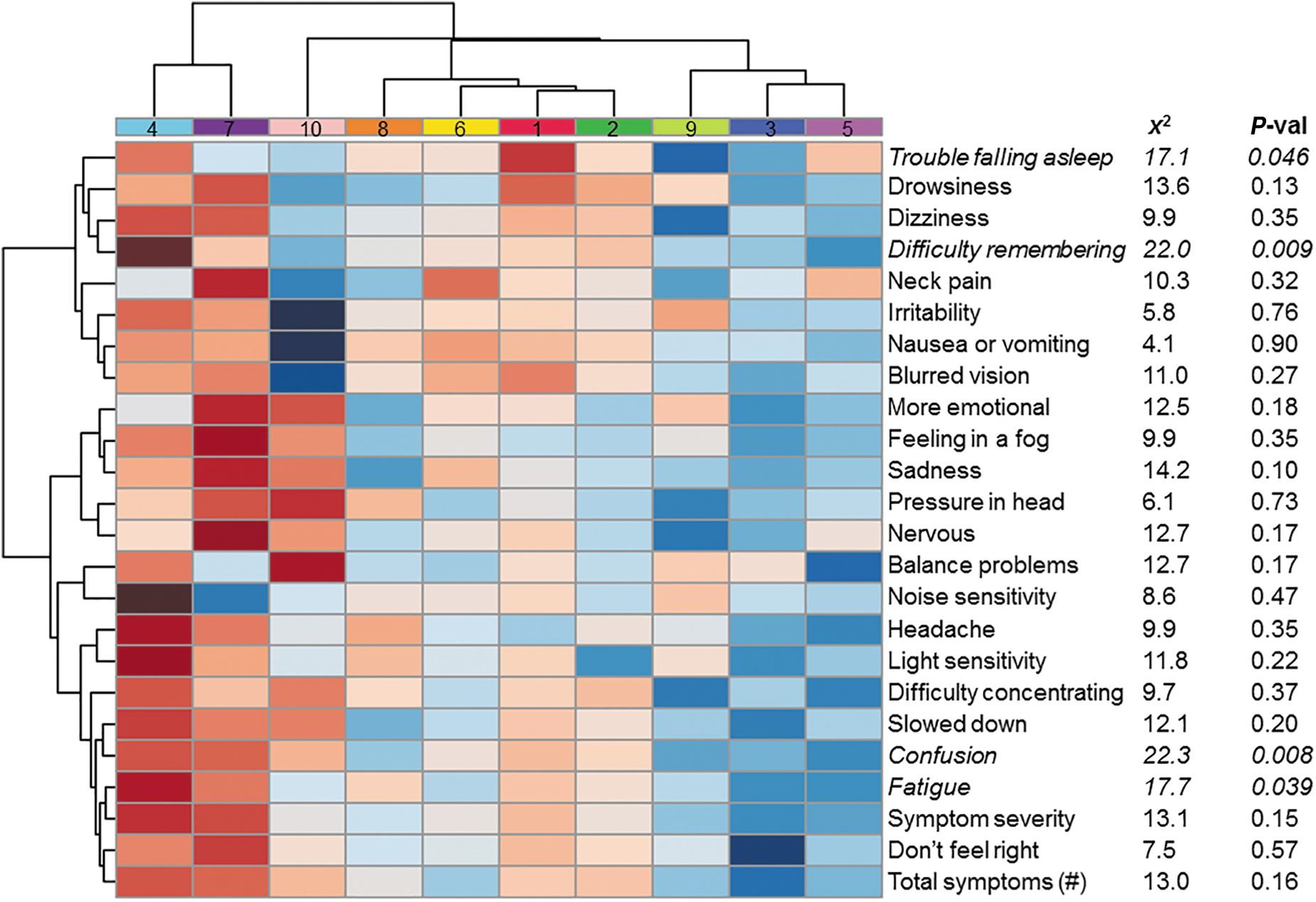
Fatigue, confusion, difficulty remembering, and trouble falling asleep differ across the 10 clusters. An ordinal regression analysis was used to assess the relationship of cluster group with subjective symptom severity, while controlling for age, days post-injury, and prior concussions. Subjective reports of fatigue, confusion, difficulty remembering, and trouble sleeping were reported on the post-concussion symptom scale, and differed across clusters. Chi-square statistics and p values are provided from omnibus likelihood ratio tests. Red denotes elevated symptom reports.
Cluster physiological features
For each cluster, the miRNAs that displayed significant differences (1.5-fold change relative to other clusters), and paralleled the direction of change previously reported in concussion literature (relative to non-concussed controls 14 ) were used to characterize physiological pathway enrichment. (Table 2). Across all 10 clusters, there were 16 pathways targeted by miRNAs with greater frequency than would be expected by chance alone (adj. p < 0.05). Cluster 1 demonstrated the greatest number of physiological targets (n = 7), while clusters 7 and 10 displayed the least (n = 2, respectively). The most commonly targeted pathway was signaling, which was implicated in 8 of the 10 clusters. Notably, the majority of pathways targeted by miRNAs in each cluster have been previously implicated in the pathophysiology of traumatic brain injury, including adrenergic signaling, 29 AMP kinase, 30 estrogen signaling, 31 fatty acid metabolism, 32 GABAergic signaling, 33 synaptic vesicle cycling, 34,35 and transforming growth factor (TGF)-β signaling. 36
Physiological Targets of the 10 microRNA Clusters
The number of genes targeted within each pathway is displayed, followed by the adjusted p value in parentheses. Pathways with adjusted p < 0.040 are shown. Pathways are listed.
TGF, transforming growth factor; ECM, extracellular matrix; AMPK, AMP activated protein kinase.
Discussion
In this investigation, using 22 salivary miRNAs previously implicated in concussion, we identified 10 distinct biological clusters. These clusters display miRNA perturbations that mirror trends from previous studies, 9 –16 and each cluster is associated with unique biological pathways that have been implicated in concussion pathophysiology. With regard to medical/demographic characteristics, there were differences among clusters in age, days post-injury (of saliva collection), and history of prior concussions, indicating that miRNA profiles may be sensitive to acute changes following injury and, perhaps, cumulative concussion effects. 9 There were no differences in miRNA profiles when comparing between sexes, races, mechanisms of injury, or loss of consciousness. These results provide a biological foundation for stratification of concussed individuals in future research, in lieu of injury mechanism, or subjective symptom reports. Such an approach might allow researchers to uncover biological subpopulations in which novel therapies show benefit in randomized clinical trials.
In addition to the relationship to demographic features, miRNA profiles were also found to differentially associate with certain clusters of subjective symptoms. Specifically, the clusters differentiated individuals with high levels of fatigue, confusion, difficulty remembering, and trouble falling asleep. Although these symptoms are often inter-related in clinical practice (i.e., insomnia leads to fatigue, and downstream difficulties with concentration), these miRNA results suggest that each symptom may arise from a distinct biological pathway. Further validation of these clusters could yield insight into specific miRNA profiles that indicate potential targets for treatment (such as melatonin for disordered sleep phenotype). In contrast, we did not find any correlations among the 10 miRNA clusters with standard clinical performance measures of balance or neurocognition. This may be because performance on these assessments is multi-factorial, and not simply related to brain function in a single neurological pathway. Balance has multiple contributing domains, including musculoskeletal strength and coordination. Neurocognition also represents a state function, influenced by multiple factors, such as fatigue, hunger, or mood. The dynamic characteristics of these assessments may account, in part, for the challenges in identifying useful treatments for concussion.
In terms of understanding the underlying biology of concussion, miRNA cluster 2 displayed robust expression patterns that have been previously described in the literature. 14 –17 This cluster performed most poorly on functional assessments of neurocognition and balance. In addition, they displayed moderate elevations in subjective symptom reports, but lacked substantial emotional symptoms. This profile may reflect a “typical” concussion phenotype, in terms of our historical understanding of concussion as a constellation of neurocognitive and balance deficits. In contrast, miRNA cluster 4 was characterized by subjective symptom reports that were primarily cognitive in nature (confirmed by low neurocognitive scores on functional assessment), but without clinical balance deficits. Interestingly, this cluster lacked “classic” changes in concussion-related miRNAs, and may reflect a more specifically cognitive phenotype of concussion. In contrast, cluster 5 was characterized by functional balance problems with generally low self-reported symptom burden and relatively typical neurocognitive performance. Cluster 5 did display several miRNA changes that are consistent with previous studies, perhaps indicating that the phenotype manifesting in this cohort reflects a broader collection of concussion-related biological pathways (e.g., those impacting the cerebellum or visual cortex). Lastly, cluster 7 contained almost no signature miRNA changes observed in previous concussion studies and lacked substantial neurocognitive or balance deficits, but had notably high subjective symptom scores. This may represent a cohort in which subjective concern for injury may be the primary feature, rather than actual biological injury from concussion. In our current era when there is heightened awareness of concussion, it would be important for us to identify patients for whom reassurance is the appropriate approach to management, rather than unnecessary interventions, such as rest or rehabilitation. However, it is also possible that cluster 7 represents a phenotype with physiological deficits that were not captured in this study (such as vision deficits).
Although this is the first study to utilize miRNAs for concussion endophenotyping, similar approaches have been proposed using gene polymorphisms, microvascular injury, neurohormonal perturbations, histopathology, and neuroimaging. 37 –40 Endophenotyping provides an opportunity to glean objective, personalized information about underlying pathophysiology that can be used to individualize treatment. It is likely that successful enophenotyping will ultimately converge on a limited number of physiological pathways relevant to brain injury and repair. For example, miRNA clusters 1 and 6 demonstrated perturbations in adrenergic signaling, which could be associated with the autonomic dysfunction reported in a subset of concussed individuals. 29 Estrogen signaling was specifically targeted by miRNAs in cluster 6. This may explain, in part, sex-specific symptoms that have been well described in the concussion literature. 31 Finally, biosynthetic pathways involving glycosphingolipids and fatty acids were uniquely targeted in clusters 1 and 2, respectively. Such patterns may actually represent unique neuro-metabolic cascades, 32 requiring similar, but distinct treatments that would only be discernible through endophenotypic analysis.
This study has several limitations that should be noted. Participants were largely adolescent with mild traumatic brain injury. It is unclear whether the miRNA profiles identified in this population would generalize to older individuals, or to those with more severe injury. Although 46% of samples (117/251) were collected within 2 days of injury, some were collected as late as 14 days post-injury. Since “time-since-injury” differed among certain miRNA clusters, future studies seeking to validate these findings should employ a tighter enrollment window with clinical relevance (e.g., within 72 h of injury). We note that the functional assessments utilized in this study were not employed for all participants (177/251; 71%) because of the time required to perform them in busy clinical settings. In addition pre-injury baseline function was not available for any study participants. Therefore, functional testing results should be interpreted with caution.
This investigation highlights the difficulty our field has experienced when utilizing non-specific clinical and subjective symptom assessments to accurately stratify concussion patients into phenotypes. These findings have important implications for prognosis and, in turn, targeted treatment interventions. The miRNA profiles identified in this exploratory study indicate that a more robust understanding of the underlying biology of injury may be an important additional factor to take into consideration when optimizing the design of future therapeutic clinical trials. Further large-scale cohort studies are required to validate this approach.
Footnotes
Acknowledgments
The authors thank Allison Iles, Arianna Montefusco, Rhianna Ericson, (Quadrant Biosciences), Kevin Zhen, Raymond Kim (Penn State), Dr. Christopher Neville (SUNY Upstate), Aaron Roberts, and Samantha Johnson (Adena) for aiding with participant enrollment and sample collection. Rhianna Ericson, Mackenzie Metras (Quadrant Biosciences), and Karen Gentile (SUNY Upstate) performed downstream sample processing, RNA extraction, and RNAseq. We thank Gregory Fedorchak and Shivani Kamath Belman (Quadrant Biosciences) for assistance with bioinformatics processing.
Authors' Contributions
All authors certify that they have participated sufficiently in the conception and design of this work, as well as the writing of the manuscript, to take public responsibility for it. All authors have reviewed the submitted manuscript and approved it for publication. Neither this manuscript nor one with substantially similar content has been published or is being considered for publication elsewhere.
Funding Information
This research was funded by a grant from the National Institutes of Health to Quadrant Biosciences (R42NS119119). S.D.H.'s time was supported by grant R01NS115942.
Author Disclosure Statement
S.D.H. serves as a consultant and advisory board member for Quadrant Biosciences. J.L. is on the scientific advisory board for Neurolign, Stage 2 Contract Engineering, Highmark Innovations, Neuronasal, and is a minority owner of 360 Concussion Care. Material has been reviewed by the Walter Reed Army Institute of Research. There is no objection to its presentation and/or publication. The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or as reflecting true views of the Department of the Army or the Department of Defense. The investigators have adhered to the policies for protection of human subjects as prescribed in AR 70–5.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
