Abstract
Managing traumatic brain injury (TBI) patients with a cerebral perfusion pressure (CPP) near to the cerebral autoregulation (CA)-guided “optimal” CPP (CPPopt) value is associated with improved outcome and might be useful to individualize care, but has never been prospectively evaluated. This study evaluated the feasibility and safety of CA-guided CPP management in TBI patients requiring intracranial pressure monitoring and therapy (TBIicp patients). The CPPopt Guided Therapy: Assessment of Target Effectiveness (COGiTATE) parallel two-arm feasibility trial took place in four tertiary centers. TBIicp patients were randomized to either the Brain Trauma Foundation (BTF) guideline CPP target range (control group) or to the individualized CA-guided CPP targets (intervention group). CPP targets were guided by six times daily software-based alerts for up to 5 days. The primary feasibility end-point was the percentage of time with CPP concordant (±5 mm Hg) with the set CPP targets. The main secondary safety end-point was an increase in therapeutic intensity level (TIL) between the control and intervention group. Twenty-eight patients were randomized to the control and 32 patients to the intervention group. CPP in the intervention group was in the target range for 46.5% (interquartile range, 41.2–58) of the monitored time, significantly higher than the feasibility target specified in the published protocol (36%; p < 0.001). There were no significant differences between groups for TIL or for other safety end-points. Conclusively, targeting an individual and dynamic CA-guided CPP is feasible and safe in TBIicp patients. This encourages a prospective trial powered for clinical outcomes.
Introduction
It is increasingly recognized that patients with traumatic brain injury (TBI) may benefit from individualized clinical management. 1,2 Cerebral autoregulation (CA) may allow for maintaining cerebral blood flow for adequate energetic requirements in response to changes in mean arterial pressure (MAP), by means of vasoconstriction and vasodilation. 3,4 In TBI patients, CA is frequently affected, and impaired CA is associated with poor clinical outcome. 5 Given that dynamic slow variations in cerebral blood volume produced by CA are transmitted into changes in intracranial pressure (ICP), the relationship between slow changes in MAP and ICP (named the pressure reactivity index [PRx]) can be considered a surrogate method for the assessment of CA. 6
The Brain Trauma Foundation (BTF) guideline recommends a target cerebral perfusion pressure (CPP) value between 60 and 70 mm Hg. The “optimal” CPP target within this range is, however, uncertain and may depend upon the patient's autoregulatory status. 7 In 2019, a consensus ICP management algorithm was published that incorporated the assessment of CA using a discrete MAP challenge to define individual CPP goals in TBI patients. 8 However, given the well-recognized temporal evolution of physiology after TBI, a continuous assessment of CA status and range might allow for improved dynamic and precise MAP/CPP titration over the disease narrative. In this regard, a continuously updated PRx and its derived “optimal” CPP (CPPopt) value could be advantageous. CPPopt is the CPP value where patients' CA is best preserved and is derived automatically from the U-shaped (parabolic) CPP-PRx relationship typically observed. 9,10 Retrospective data demonstrated an association between improved outcome in patients who had a CPP value concordant with the CPPopt value. 9,10
Accordingly, we performed the first randomized controlled clinical trial—the CPPopt Guided Therapy: Assessment of Target Effectiveness (COGiTATE)—to assess feasibility as the percentage of monitoring time with CPP concordant with CPPopt in the intervention group and whether this leads to a potentially harmful need for higher therapeutic intensity levels (TILs) in TBI patients with ICP monitoring (TBIicp).
Methods
Trial design
The protocol has been published previously. 11 An extended summary of the methodology and (statistical) analysis can be found in Supplementary Appendix S1. COGiTATE was a multi-center, international, non-blinded, randomized controlled parallel phase II trial (RCT). Four tertiary hospitals that provide acute neurocritical care for TBI patients were involved. Ethical approval was obtained at all participating institutions.
Participants
Eligible participants were patients with TBIicp meeting the following inclusion criteria: 1) adults (age ≥18 years) indicated for ICP-directed therapy for at least 24 h; 2) randomization within 24 h after intensive care unit (ICU) admission; and 3) signed proxy informed consent. Exclusion criteria were: 1) primary decompressive craniectomy; 2) injury deemed to be unsurvivable at presentation; and 3) known pregnancy. Block randomization, with stratification according to trial site, was used to ensure a uniform patient distribution. Randomization was performed electronically using a centralized electronic case report form (eCRF) by a researcher. The study was powered to achieve a 20% increase in percentage of monitored time with CPP concordant ±5 mm Hg of the CPPopt values; from 30% (historical cohort) to 36% in the intervention group, resulting in a sample size of 60 patients. 11
Data collections
Baseline and daily patient characteristics were collected. The daily characteristics included the TIL score, 12,13 laboratory and diagnostic organ function parameters, measures of hemodynamic and ventilatory support intensity, and pre-defined adverse events (AEs) and serious adverse events (SAEs). The aim of the summary (daily) TIL score is to provide a quantitative estimate of the ICP lowering interventions used in a given period (recommended 24 h) by assigning numerical scores to each therapy intensity level of each intervention and summating these (0–38). 12,13 The 6-month neurological outcome was assessed using the Glasgow Outcome Scale (GOS). 11 In addition, the International Mission for Prognosis and Analysis of Clinical Trials in TBI (IMPACT) score was calculated. 14 High-frequency monitoring data were collected using the research software, ICM+® (Cambridge Enterprise, Cambridge, UK), at the bedside. MAP was monitored by invasive arterial cannulation in the radial or femoral artery and zeroed at the tragus. ICP was recorded by a parenchymal microsensor.
Interventions
Patients were randomized to either the BTF CPP range (60–70 mm Hg) 7 or targeting a “dynamic” CPP value (defined as the CPPopt). According to the randomization group, the software displayed the appropriate physiological targets and CPP review information (Supplementary Fig. S1) at the bedside. 11 CA information, including PRx, CPP-PRx curve, and the updated 1-min trend of CPPopt (further referred to as “CPPopt trendline”), was displayed for the intervention group only (Fig. 1). 11,15 In the intervention group, patients were managed according to the calculated CPPopt, that is, the CPP value extracted from the CPPopt trendline at the regular review time points (further referred to as the “CPP target”). How to achieve the CPP target was left to the discretion of the attending clinical team. The protocol ended after a maximum of 5 study days or after reaching pre-defined end-points (Supplementary Appendix S1). 11
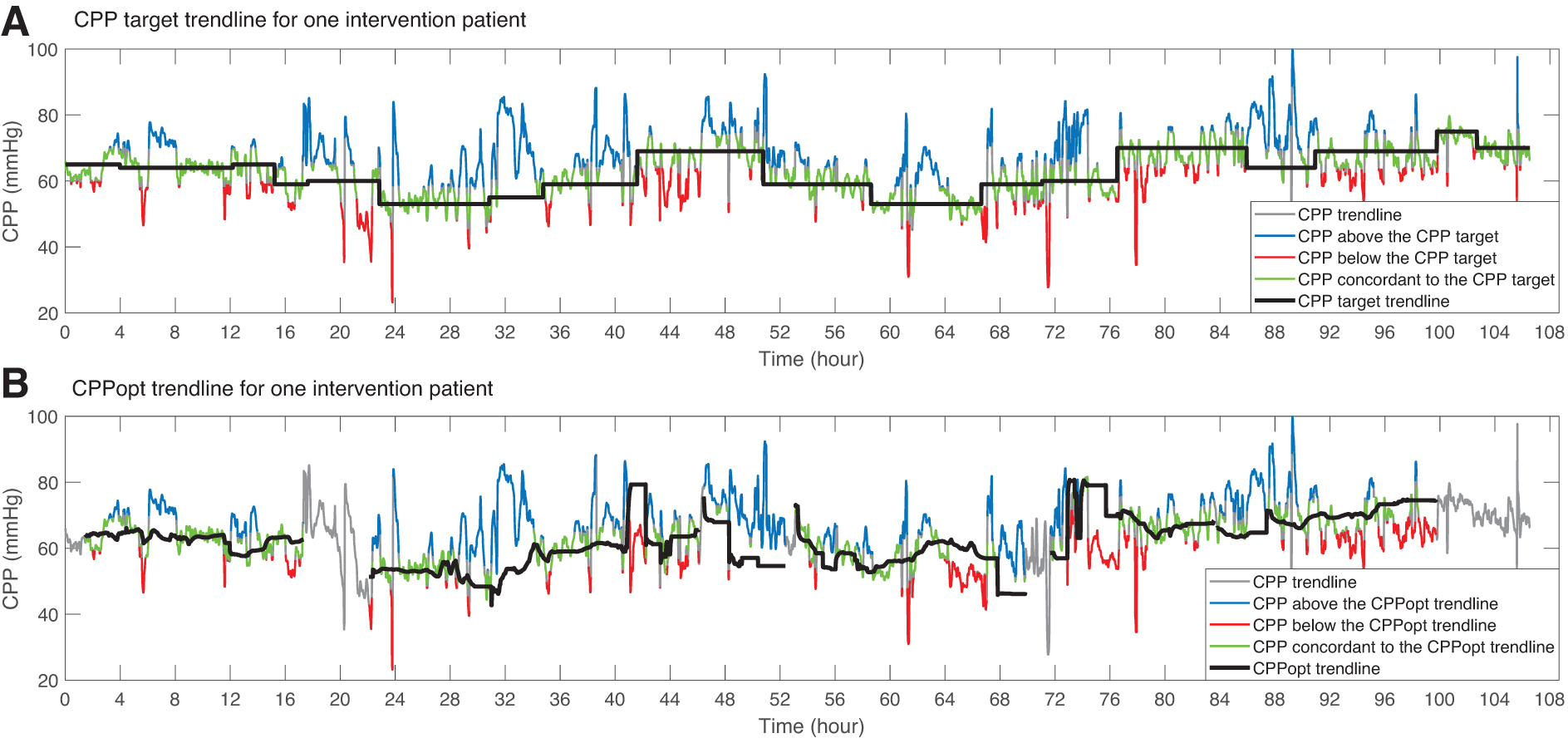
Illustrative example of a recording from a patient in the intervention group. Time t = 0 is the start of the intervention protocol, which is the time that a CPP target can be provided. (
Cerebral perfusion pressure reviews
The clinical team reviewed the CPP targets according to visual alerts generated by the software six times daily. Until the first review alert, the protocol recommended to keep CPP between 60 and 70 mm Hg in both groups (the period up to the first review alert was not used for data analysis). For the intervention group, a CPP was recommended at each review, which was either 1) the calculated CPPopt trendline value or 2) a “clinical” CPP value. The latter was suggested when the CPPopt calculation was not available, and the clinical team were to choose an appropriate target themselves. 11 In both groups, the clinical team was allowed to deviate from the software recommendation, but requested to record their rationale and the chosen CPP target value in real time.
Outcome measures
To assess the feasibility of the study protocol, we aimed to increase the percentage of the monitored time with measured CPP within a range of 5 mm Hg above or below the CPP target to values above the pre-defined 36% in the intervention group (primary end-point). 11 To examine safety, we identified a clinically relevant difference in daily TIL score as an averaged daily TIL score difference ≥3 between the groups (main secondary end-point). Between-group differences of physiological, diagnostic, treatment, and AE/SAEs were assessed for additional evidence for safety. Finally, clinical outcome parameters were assessed.
Data analysis
Data preparation
High-frequency monitoring data, including the 1-min average physiological data (including ICP, MAP, CPP, PRx, CPPopt) and the six times daily review results, were anonymized and packaged into HDF5 files, 16 using ICM+, and imported into MATLAB (Release 2019b; The MathWorks, Inc., Natick, MA) for further calculations. Patients' total mean (covering the study protocol period) and 1-h mean physiological values were calculated after automated removal of non-physiological values in MATLAB. The eCRF and averaged physiological data were imported in R (version 4.0.3; R-Core Team). 17
Statistical analysis
Data are presented as mean ± standard deviation (SD) or median and interquartile range (IQR) for continuous variables. We used frequencies (%) for categorical variables. The primary feasibility end-point was evaluated using a one-sample t-test. All other between-group comparisons were based on the two-sample t-test/Mann-Whitney U test for continuous variables, and analyses of categorical variables were based on the chi-squared/Fisher's exact test. Levene's test was used to test equality of variances between groups. A p value <0.05 was considered statistically significant. All statistical analyses were performed in R.
Data availability
Not applicable.
Results
The enrollment flowchart and study timeline are summarized in Supplementary Figures S2 and S3. From February 2018 until January 2020, 28 patients were randomized to the control group and 32 patients to the intervention group. Admission patient characteristics were slightly imbalanced between the groups (Table 1). Figure 1 shows a patient example with trends of patients' CPP, CPP targets, and CPPopt over time. The CPPopt trendline was available for 76.6% (SD, 16.5) of the monitored time in the intervention group. MAP was significantly higher for the intervention group (control, 81 [SD, 6] vs. intervention 85 [SD, 8] mm Hg; p < 0.05). Also, mean CPP was significantly higher (control 68 [SD, 4.4] vs. intervention 73 [SD, 6.6] mm Hg; p < 0.05), with significantly increased variability for the intervention group (p < 0.05). In the intervention group, PRx was, on average, lower compared to the control group (Table 2).
Presented percentages may not be equal to 100% as a result of rounding. n = number.
The IMPACT score for moderate-to-severe TBI. Included patient characteristics in the prognostic model for mortality prediction at 6 months using the Core model: age, GCS Motor score, and pupillary reactivity. IMPACT is validated for TBI patients having an initial GCS ≤12 (control, n = 27; intervention, n = 26). In addition, 3 patients with missing Glasgow Outcome Scale at 6 months (control, n = 1 and intervention, n = 2) were not included for the average IMPACT outcome prediction scores.
When the software recommendation was to use a “clinical target,” the question “adopting CPPopt target” was not presented.
Exact question: “Will you be adopting the advised CPPopt target/CPP range (yes/no)?” The percentages were computed in the intervention group without the “clinical target” reviews (n = 144).
Exact question: “Where there any clinical problems with reaching the previous CPP target?”
Exact question: “Which interventions are you planning to achieve the new CPP target?”
ICP monitoring discontinuation was a clinical decision and not influenced by the intervention protocol.
SD, standard deviation; IQR, interquartile range; GCS, Glasgow Coma Scale; CT, computed tomography; TBI, traumatic brain injury; IMPACT, the International Mission for Prognosis and Analysis of Clinical Trials in TBI; CPP, cerebral perfusion pressure; CPPopt, optimal CPP; ABP, arterial blood pressure; ICP, intracranial pressure; NA, not applicable.
Unpaired sample t-test, Mann-Whitney U test, or chi-squared or Fisher's exact test was used.
Levene's test was used to test equality of variances between groups.
CPP target. At a review alert, a CPP target was set by the clinical team either by adopting the recommended CPPopt or by taking a “clinical” CPP target.
CPPopt trendline is the 1-minute updated CPPopt over time.
Calculated as the percentage of time CPPopt was available given the CPP value being present.
The number of outcome values missing were one in the control group and two in the intervention group.
Statistics are added in Suppplementary Figure S7.
SD, standard deviation; IQR, interquartile range; CPP, cerebral perfusion pressure; MAP, mean arterial pressure; HR, heart rate; ICP, intracranial pressure; PRx, pressure reactivity index; CPPopt, optimal cerebral perfusion pressure; GCS, Glasgow Coma Scale; ICU, intensive care unit; GOS, Glasgow Outcome Scale; NA, not applicable.
Feasibility end-point
In the intervention group, CPPopt-based software recommendations were provided in 74% of the 552 reviews (i.e., no CPP targets were given in 26% of the reviews, where the clinicians were to choose an appropriate “clinical” target until the next review; Table 1). Figure 2 shows the distribution of the CPP recommendations and final CPP targets in the intervention group (with the individual CPP targets and distributions over time in Fig. 3 and Supplementary Fig. S4). The number of deviations from the CPP recommendations in both groups was low (control 6.8% vs. intervention 8.3%, respectively; Table 1).
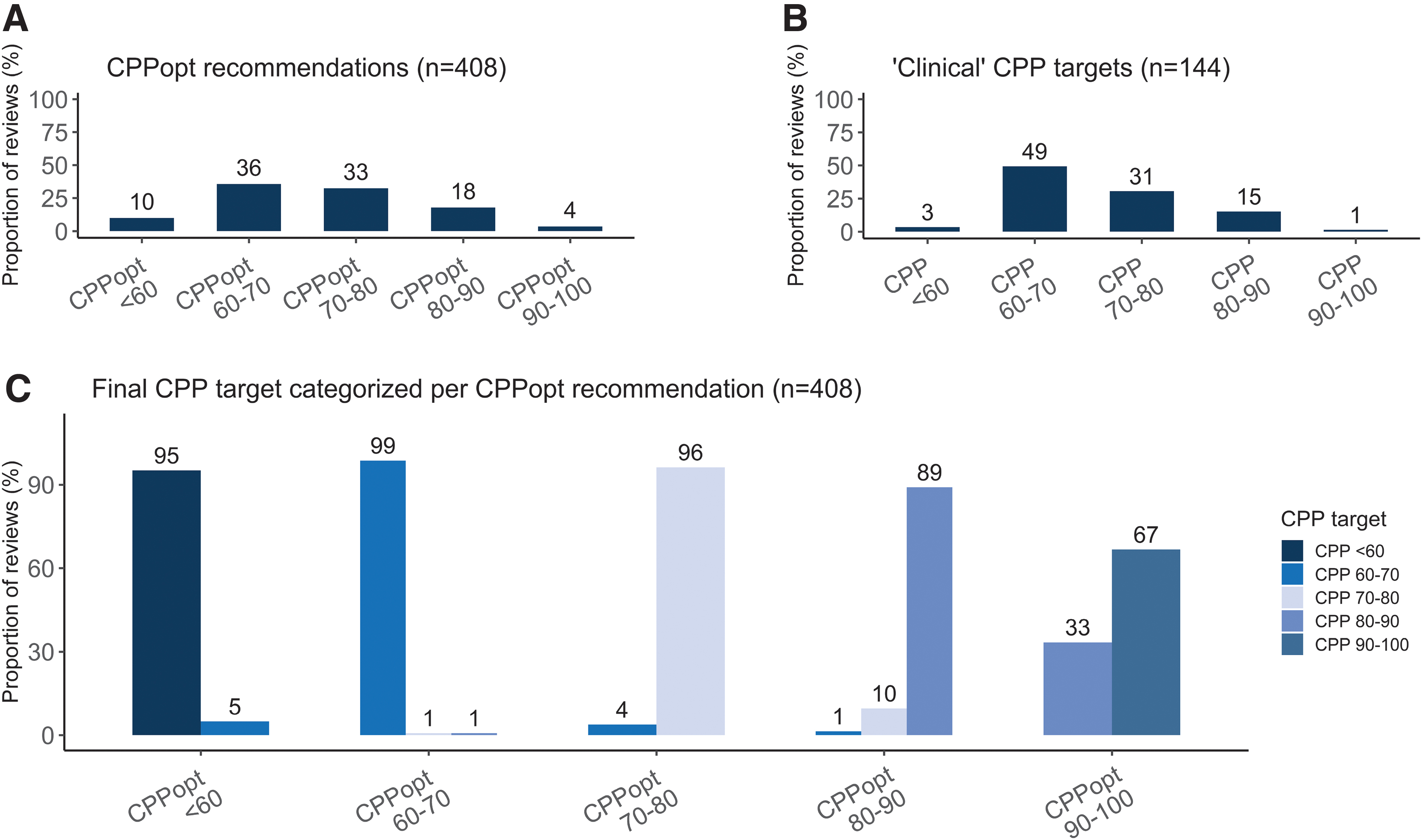
Six times daily CPP review recommendations and targets for the intervention group. In total, 552 reviews were done in the intervention group. The software suggested a CPPopt in 74% of the reviews (n = 408 reviews; median CPP, 71 [IQR, 64–78] mm Hg). The clinical team adopted this target in 92% of the reviews or rejected this recommendation (8%). In 26% of the reviews, the software was not able to provide a CPPopt. In this case, the clinical team had to choose a “clinical” CPP target for the upcoming period (n = 144 reviews; median CPP, 66.5 [IQR, 65–74] mm Hg). (
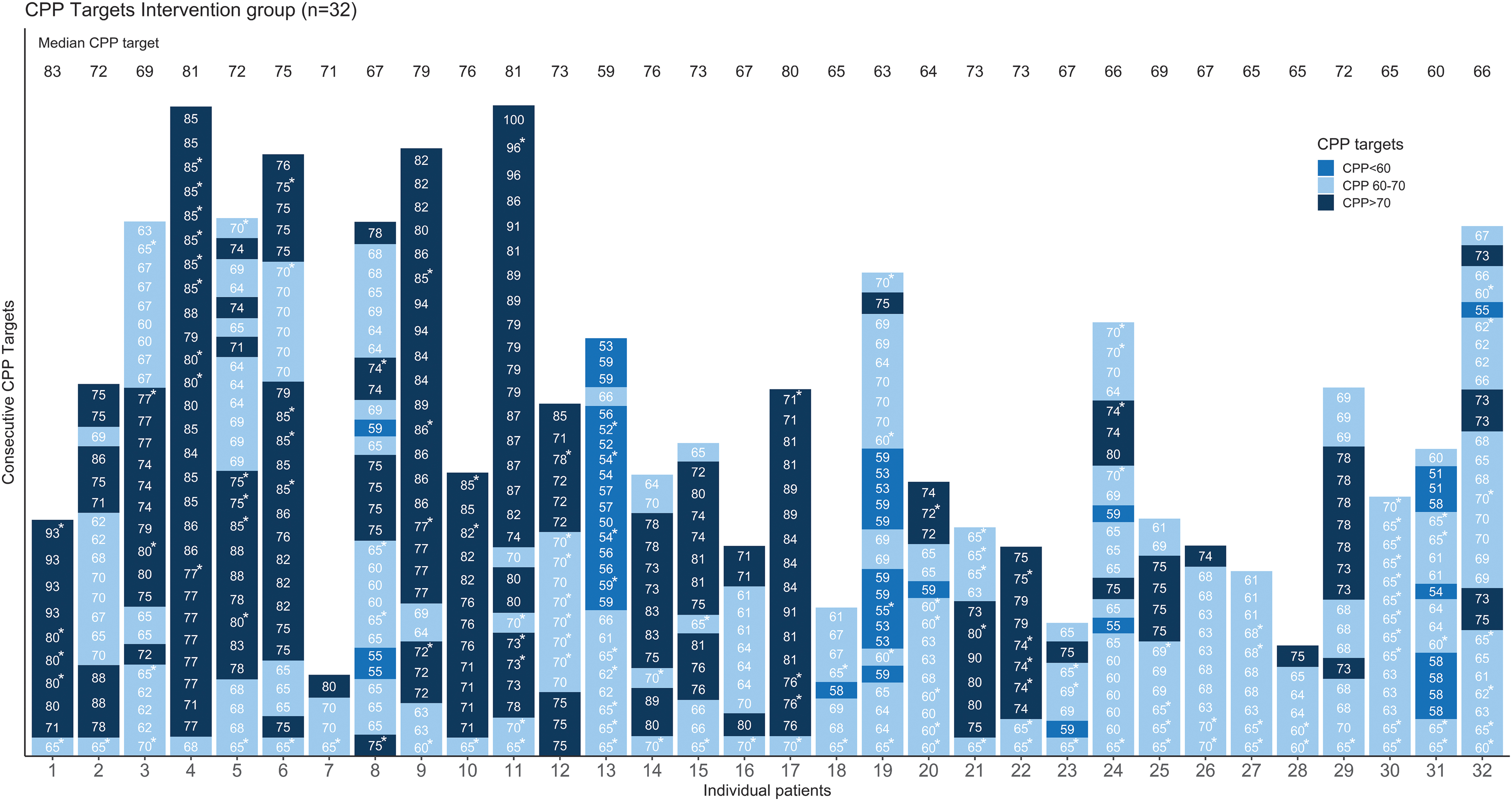
CPP targets in individual patients over time. The individual CPP targets (updated six times a day) for patients randomized to the intervention group are shown over time. Each bar represents a patient. The CPP targets are grouped by three colors: 1) below 60 mm Hg (blue); 2) above 70 mm Hg (dark blue); or 3) within 60–70 mm Hg (light blue). Numbers in the bars represent the specific CPP targets. “Clinical” targets are marked with an asterisk (*). Median CPP target values for each individual are shown on top. Seventeen patients (53%) had a median CPP target outside the BTF CPP guideline (60–70 mm Hg) range. BTF, Brain Trauma Foundation; CPP, cerebral perfusion pressure.
The median CPP target was 70 mm Hg (IQR, 66–75) in the intervention group. In 37.9% (IQR, 18–58) of the time, the target was within the BTF CPP range (Table 2 and Supplementary Table S1). The median CPPopt trendline was slightly higher in the intervention group (control 69 [IQR, 67–73] vs. intervention 72 [IQR, 66–77] mm Hg; p = 0.448). In the intervention group, patients spent 46.5% (IQR, 41.2–58) of the monitoring time with CPP concordant with the set CPP targets. This is significantly higher than the powered 36% (p < 0.001; Table 2 and Supplementary Table S2). Table 2 and Supplementary Table S2 show the between-group comparisons of the retrospectively available CPPopt trendline values. Patients in the intervention group spent more time with their CPP concordant and above the CPPopt trendline (p = 0.150 and p = 0.573, respectively), but significantly less time below the CPPopt trendline (control 34.6% [IQR, 22.4–43.5] vs. intervention 19.1% [IQR, 13.8–29.3]; p < 0.001). Patients in the intervention group spent less time with CPP values <60 mm Hg (control 11.7% [IQR, 5.46–21.5] vs. intervention 6.71% [IQR, 1.5–10.4]; p < 0.05), but more time with a CPP >70 mm Hg (control 30.7% [IQR, 23–46.6] vs. intervention 64.9% [IQR, 44–82.5]; p < 0.001).
Safety end-points
The median TIL score showed no significant difference between the groups (control 7 [IQR, 6–10] vs. intervention 7 [IQR, 5–9]; p = 0.882). Data on individual TIL items are available in Supplementary Table S3. In addition, no significant between-group differences in parameters suggestive for lung, cardiac, and kidney damage were observed (Table 3). Both groups received similar amounts of fluids, and the median daily dose of noradrenaline was comparable between the groups (control 12.2 mg [IQR, 7.42–18.8] vs. intervention 10.4 [IQR, 4.51–20.7]; p = 0.514). No significant between-group differences over time were found for any of the secondary end-points, including TIL (p = 0.245) and CPPopt (p = 0.106; Supplementary Table S4; Supplementary Figs. S5 and S6). Similar numbers of AEs (control 7 vs. intervention 9) and SAEs (control 1 vs. intervention 1) were observed. (Table 3 and Supplementary Table S5).
The laboratory and fluid administration values in Table 3 concern median values. The values were first calculated per patient over the intervention period (values collected per day). Then, the study group median values are reported. n = number. The control and intervention group had n = 107 and n = 122 measurement days, respectively.
Unpaired sample t-test, Mann Whitney U test, or chi-squared or Fisher's exact test was used.
Missing days: control group, 1 day (0.9%); intervention group, 3 days (2.5%). Missing patients, n = 0.
High-sensitive troponin, including high-sensitive troponin T and high-sensitive troponin I, were divided in values above/below a critical threshold (high-sensitive troponin T, 14 ng/L; high-sensitive troponin I, 58.1 ng/L (male); 39.6 ng/L (female). Missing patients, 0. Missing days: control group, n = 8/107 (7.5%); intervention group, n = 9/122 (7.4%).
Missing days: control group, 12 (11%) days; intervention group, 11 (9%) days. Missing patients, n = 0.
Levene's test was used to test equality of variances between groups.
The specific adverse events and serious adverse events can be found in the Supplementary Materials, Supplementary Table S5.
IQR, interquartile range; NT-proBNP, N-terminal pro-brain natriuretic peptide; PaO2/FiO2, partial pressure of oxygen/ fraction of inspired oxygen ratio; ICP, intracranial pressure; CPP, cerebral perfusion pressure.
Outcome
The distribution of 6 months' GOS of both groups is shown in Supplementary Figure S7. Fewer patients died in the intervention group (23%) compared to the control group (44%). None of the outcome results reached statistical significance (Table 2 and Supplementary Figure S7).
Discussion
The current study represents the first RCT evaluating CA-guided CPP management in TBIicp patients. Compared to retrospective pilot data, 11 we showed that patients in the intervention group spent a significantly higher percentage of time with CPP concordant with the set CPP target given by CPPopt. Given that most CPP review recommendations were adopted (>90%), these results demonstrate the feasibility of using CPPopt as a novel digital biomarker for precision medicine in TBIicp management. Safety was demonstrated by a similar between-group TIL scores, number of AEs/SAEs, and biomarkers of organ damage. Although the study was not designed to assess clinical outcomes, we found these to be comparable. The hemodynamic support, represented by the amount of administered fluids and vasopressors use, was (non-significantly) lower in the intervention group, although these patients ended up with significantly higher mean absolute MAP/CPP values. Dynamic CPP targets probably explain the increased CPP variability in patients in the intervention group.
The BTF guideline mentions that TBIicp patients with intact autoregulation are best served by higher CPP values whereas pressure-passive patients with dysfunctional pressure autoregulation do better with lower CPP values. 7 The recent Severe Traumatic Brain Injury Consensus Conference consensus ICP treatment suggests the use of intermittent MAP challenges for CA assessment. 8 This represents an effort to integrate CA into ICP management. However, MAP challenges are infrequent, occupy nursing and medical time, and may provoke ICP elevation when CA is compromised. Further, MAP challenges are only likely to be undertaken when ICP control is a problem, which neglects the optimization of CPP as a continuous parameter. PRx offers a more, dynamic, safe, automated, and repeated evaluation of CA. Perhaps most critically, the protocol we suggest moves CA-related management beyond just a means of ICP control, to ensuring optimal physiology and wider beneficial impact on outcome. Moreover, PRx requires only invasive ICP and MAP for its calculation. However, this also implies that PRx values may be unreliable after periods with limited MAP fluctuations. This presumed effect might have contributed to CPPopt trendline values not being available in ∼25% of the monitoring time (Table 2).
Mean CPP in the intervention group was 73 (SD, 6.6) mm Hg. However, mean values might mask intra- and interpatient variability over time as shown by the individual target distribution (Fig. 3), increased CPP variability (Table 2), and large proportion of time with CPP targets outside the BTF guideline range in the intervention group (Supplementary Table S1). We postulate that these individual dynamic targets allow continuous fine-tuning of perfusion pressure, potentially leading to outcome benefit. Both groups spent <1% of the monitoring time with a CPP outside our pre-defined safety ranges (50–100 mm Hg), which is reassuring. These periods were attributed to drops in arterial blood pressure (ABP) given that ICP was, on average, well controlled in both groups (Table 3). Patients in the intervention group spent significant less time with their CPP below the CPPopt trendline and below the CPP threshold of 60 mm Hg (group difference of 15.5% and 5%, respectively). In the intervention group, the chosen CPP target was in only 37.9% (IQR, 18.2–58.3) of the monitored time between 60 and 70 mm Hg (Table 2 and Supplementary Table S2). This supports the potential benefits of our intervention protocol.
Other retrospective TBIicp analyses demonstrated that impaired CA reduces the tolerability for low CPP and high ICP. 18 –21 On average, CPP targets <60 mm Hg were less common (10%; Fig. 2) compared to targets ≥70 mm Hg (55%; Fig. 2). This might indicate that TBIicp patients indeed need (on average) higher CPP targets. An alternative explanation is that our understanding of CA and its monitoring are both imperfect. Tzeng and colleagues hypothesized that the CA may be better adapted to compensate for increasing than for decreasing MAP. 22 Moreover, animal studies have described less clear or more variable (individualized) upper CA limits. 23,24
Multimodality neuromonitoring allows for a better understanding of the pathophysiology and therefore for the application of individualized, targeted treatments in TBIicp patients. 1,25 Large RCTs are underway to prove the clinical benefit of using combinations of monitoring signals and/or physiological concept protocols. The Brain Oxygen Optimization in Severe TBIicp phase-II (BOOST-II) multi-center RCT reported that PbtO2 augmentation could be safely implemented in patients where such monitoring was available. Interventions to optimize CPP were restricted to the ICP and PbtO2 group only, showing parallels to our intervention study. 26 The main difference is that in COGiTATE, the CPP management target was generated using an automated, dynamically adaptive algorithm as opposed to fixed, pre-defined thresholds.
Although the difference in CPP between the intervention and control groups was small across the study population, this does not exclude the possibility that, at an individual level, CA-guided CPP management results in a more individualized treatment given that we know that the CA varies substantially between patients and over time. 27 Given that the study centers have a history of using CPPopt in clinical research settings, it is also possible that these institutions already tend to target higher CPP values (i.e., closer to 70 mm Hg) in control patients. Our study set out to examine feasibility and safety end-points. A detailed examination of the effect of CA-guided management on neurological recovery or suitable surrogate outcome will require further prospective, randomized studies.
Our study has limitations. First, our sample size limits conclusions about physiological interactions, organ complications, and outcome results. Second, both groups still spent a considerable amount of time away from the CPP target (±5 mm Hg) or outside the BTF target range. This is likely related to the lack of a continuous feedback about significant deviations from the target to the nursing team. 28 Adding a continuous feedback may improve the percentage of time CPP concordant with CPPopt and probably the clinical relevance of the therapy. Third, our study was necessarily not blinded, and the outcome assessors were aware of the group assignment. Fourth, in the intervention group, in 26% of the reviews CPPopt was not available. This is a limitation of the current methodology. 29 Fifth, we did not treat the patients according to a standardized hemodynamic management protocol. Sixth, we noticed a considerable delay before starting the intervention protocol after trauma ictus in both groups (Supplementary Fig. S3). For a phase II study, this is less important, but in a phase III study, the delay between ICU admission and start intervention can be minimized with application of a deferred consent procedure in all eligible patients. 30,31
Seventh, the study was performed in centers with experience in ICM+ research software. This limitation has to be taken into account with future study setup in less-experienced centers. Finally, the software used in the intervention protocol is labeled as research software and therefore not commercially available for clinical purposes. Our study also has several unique strengths. The study protocol was pre-specified, 11 with a pragmatic design to test, for the first time, the interaction between a software algorithm and clinical team at the bedside six times daily. This innovative approach was studied in four centers with different clinical teams involved over the 24-h patient care. No additional invasive monitoring was required. Lastly, we limited our protocol to a maximum of 5 days to overcome the over-representation of certain patients in the analysis.
Conclusion
Individualizing care by targeting a dynamic optimal CPP using CA guidance six times daily is feasible and safe in TBIicp patients. These findings encourage a larger phase III outcome study of this novel digital biomarker for precision medicine in these patients.
Footnotes
Acknowledgments
We thank the nursing staff, clinical fellows, and intensive care consultants for their contribution to the daily reviews and data collection. In particular, we thank the following people for their support and advices: Gert Bellen, Melisa Borg, Dominike Bruyninckx, Manuel Teixeira Cabeleira, Erwin Derhaag, Marta Fedriga, Angelos Kolias, Phaedra Lebegge, Jo Lodewyckx, Joanne Outtrim, Chiara Robba, Eric Thellin, and Carole Turner. We thank the patients and their families.
Authors' Contributions
All authors contributed to the study conception and design. Material preparation and data collection by Jeanette Tas, Erta Beqiri, Ruud C. van Kaam, Analisa L. Liberti, C.W.E. Hoedemaekers, Bart Depreitere, Peter Smielewski, Ari Ercole, David K. Menon, Geert Meyfroidt, and Marcel J.H. Aries. Data analysis was performed by Jeanette Tas, Erta Beqiri, Marek Czosnyka, Sander M.J. van Kuijk, David K. Menon, Peter S. Smielewski, Ari Ercole, and Marcel J.H. Aries. The first draft of the manuscript was written by Jeanette Tas and Marcel J.H. Aries, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding Information
This research was supported by the NIHR Cambridge Biomedical Research Centre (BRC-1215-20014). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
The COGiTATE study was funded with a grant from the European Society of Intensive Care (ESICM).
Jeanette Tas is supported by “HersenStrijd fonds” (translated as “Brain Battle foundation”), a non-profit foundation of the University Maastricht, Maastricht, The Netherlands.
Erta Beqiri is supported by the Medical Research Council (grant no.: MR N013433-1) and by the Gates Cambridge Scholarship.
Marek Czosnyka is supported by National Institute for Health Research (NIHR), Cambridge Biomedical Research Centre.
David K. Menon is supported by the National Institute for Health Research (NIHR), Cambridge Biomedical Research Centre, an NIHR Senior Investigator Award, and by a European Union Foundation Program 7 Grant (CENTER-TBI; award no.: 602150).
Peter J. Hutchinson is supported by the National Institute for Health Research (NIHR): research professorship, Biomedical Research Centre and Global Neurotrauma Research group and the Royal College of Surgeons of England.
Geert Meyfroidt is supported by the Research Foundation, Flanders (FWO) as a senior clinical researcher (1843118N) and receives project funding from the KU Leuven (C24/17/072) and the Belgian Health Care Knowledge Center (COV201003).
Author Disclosure Statement
Peter Smielewski and Marek Czosnyka receive part of the licensing fees for multi-modal brain monitoring software ICM+, licensed by Cambridge Enterprise Ltd, University of Cambridge, Cambridge, UK.
David K. Menon reports research funding or consultancy agreements with the following entities: Calico Inc; GlaxoSmithKline Ltd; Integra LifeSciences; Lantmannen AB; NeuroTrauma Sciences; and PressuraNeuro Ltd.
Supplementary Material
Supplementary Appendix S1
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
