Abstract
Humans, primates, and rodents with cervical spinal cord injury (SCI) show permanent sensorimotor dysfunction of the upper/forelimb as consequence of axonal damage and local neuronal death. This work aimed at characterizing a model of cervical SCI in domestic pigs in which hemisection with excision of 1 cm of spinal cord was performed to reproduce the loss of neural tissue observed in human neuropathology. Posture and motor control were assessed over 3 months by scales and kinematics of treadmill locomotion. Histological measurements included lesion length, atrophy of the adjacent spinal cord segments, and neuronal death. In some animals, the retrograde neural tracer aminostilbamidine was injected in segments caudal to the lesion to visualize propriospinal projection neurons. Neuronal loss extended for 4–6 mm from the lesion borders and was more severe in the ipsilateral, caudal spinal cord stump. Axonal Wallerian degeneration was observed caudally and rostrally, associated with marked atrophy of the white matter in the spinal cord segments adjacent to the lesion. The pigs showed chronic monoplegia or severe monoparesis of the foreleg ipsilateral to the lesion, whereas the trunk and the other legs had postural and motor impairments that substantially improved during the first month post-lesion. Adaptations of the walking cycle such as those reported for rats and humans ameliorated the negative impact of focal neurological deficits on locomotor performance. These results provide a baseline of behavior and histology in a porcine model of cervical spinal cord hemisection that can be used for translational research in SCI therapeutics.
Introduction
Approximately half of all human spinal cord injury (SCI) cases show incomplete tetraplegia caused by partial damage to the cervical spinal cord. 1 Chronic motor deficits after incomplete cervical SCI are greater in the upper limbs than in the lower limbs. 2,3 Whereas the impaired function of the lower limbs is attributable to interruption of spinal axonal systems, both axonal breakage and focal neuronal death are responsible for upper limb deficits. 4 Several animal species have been used to model incomplete cervical SCI and characterize the associated functional loss, particularly rats, 5 –14 cats, 15 –18 and primates. 17,19 –26 In these quadrupeds, severe and permanent sensorimotor impairments occur in one foreleg, whereas the other legs are much less dysfunctional and show motor adjustments that compensate for the most disabled one. Rehabilitation by treadmill locomotion or forced foreleg use produces further behavioral improvements. 27 –30
Pigs have several anatomical and physiological characteristics that resemble those of humans and make them attractive for testing neurotherapeutics and guide SCI clinical trials. 31 Despite being artiodactyls, pigs have a corticospinal tract (CST) with lateral and ventral fascicles travelling along the entire cervical spinal cord. 32,33 The pig brain is gyrencephalic and exhibits a perinatal growth spurt quite similar to that in humans, with the brain weight growing more rapidly from the last third of gestation to lactation. 34 Myelination of the pyramidal system likewise takes place in comparable developmental stages. 35,36 The size of the spinal cord and cerebrospinal fluid space in pig are also similar to those of a human, 37 which is of great value for assessing drug dosage and distribution before clinical translation. Similitude with humans in terms of circulatory system helps to reproduce the pathophysiology of SCI and the development of tissue cavitations. 38,39 Moreover, the anatomy of the pig vertebral column approximates that of humans regarding overall vertebral body height, body width, end-plate shape, and spinal canal dimensions, 40,41 making pigs useful for assessment of bone implants and spine surgical interventions in a biomechanical context relevant to humans. Despite the translational advantages of research data gathered in swine, the use of this model in the SCI field has not yet consolidated. Most studies using pigs deal with thoracic SCI, 37,39,42 –49 whereas a few have produced cervical lesions, mainly for contusion or transection injuries without animal recovery 50,51 and short-term studies focused on dorsal root lesions. 52
Incomplete cervical SCI can be produced experimentally by contusion, selective spinal tractotomy, or sectioning large parts of the spinal cord. Axotomy and axonal regenerative failure occur irrespective of the lesion type. However, other pathological features, particularly neuronal death, vascular damage, and connective tissue scaring substantially differ depending on the mechanism of injury. Contusive lesions reproduce the necrosis and cavitation of neural tissue observed in more than half human cases. 7,53,54 Although sharp cutting injuries are infrequent in the clinical setting, 1,55 the damage produced by sectioning parts of the spinal cord in animals is representative of lacerating SCI, which occurs in ∼20% of human cases. 53 Neurologically, hemisection of the cervical spinal cord in animals is similar to the Brown–Séquard syndrome described in human patients. 56 The present study investigated the feasibility of swine for producing a chronic model of cervical SCI by excising 1 cm of spinal cord on one side of the cervical enlargement. By removing the tissue, we attempted to model the cavitation with loss of neural elements characteristic of human pathology, with the future aim of using this model for investigating strategies to bridge the spinal cord and promote axonal regeneration and neural circuit reconnection across the tissue gap.
Methods
Animals
Eight 2-month-old Large White male pigs (Sus scrofa domesticus) weighting 10–15 kg at the time of lesion were used. The animals were purchased from a commercial supplier (Granja Agropardal, Toledo, Spain). Five pigs underwent a right-sided cervical spinal cord hemisection with excision of 1 cm of neural tissue, and three age-matched pigs were used as uninjured controls. The experimental protocols adhered to the recommendations of the European Commission and Spanish regulations for the protection of experimental animals (86/609/CEE, 32/2007 and 223/1988) and were approved by the Ethical Committee for Animal Research of the Hospital Nacional de Parapléjicos.
Anesthesia and surgical procedures
All surgical procedures were performed under inhalational anesthesia. Anesthesia was induced by intramuscular (IM) injection of ketamine (10 mg/kg), midazolam (0.1 mg/kg), and medetomidine (0.02 mg/kg), followed by intravenous (IV) administration of propofol (3 mg/kg). Then, a tracheal tube was placed, and the anesthesia was maintained with sevoflurane (1.7–2%) together with remifentanil (26 mg/kg/h IV) and rocuronium (1.2 mg/kg/h IV). Mechanical ventilation (Fabius Tiro, Dräger) was set at 12–14 breaths/min with a tidal volume of 10–15 mL/kg. Heart rate, blood pressure, exhaled carbon dioxide, blood oxygen saturation, and inspired and expired sevoflurane levels were monitored during the procedure (Dräger Infinity Delta).
For SCI, the back and neck of the anesthetized animals were disinfected with povidone iodine, and a 12-cm dorsal midline incision was made between C1 and T1 for exposing the cervical spine. Dorsal laminectomy of the C5 vertebra with partial removal of the rostral and caudal dorsal laminae allowed access to the C5 and C6 spinal cord segments. The pial surface was exposed by midline durotomy, and the ventral and dorsal right nerve roots of the targeted spinal segment were cut. Subsequently, a hemisection with excision of 1 cm of the right side of the spinal cord was performed (Fig. 1A). Our aim was to perform the lesion at C6, rostral to the bulk of the triceps brachii motoneuron nucleus, which is mainly located in C7–C8 in most mammalian species 57 in order to produce a clear deficit for elbow extension and weight-bearing foreleg force that could be precisely assessed by biomechanical and physiological techniques. 11 The lesion produced a transient but profuse hemorrhage at the lesion site that required some effort to be controlled. Hemostasis was achieved by introducing pieces of gelatin sponge (Gelfoam®) at the tissue cavity, waiting for about five minutes and then removing the sponge and rinsing the site several times with saline solution. Subsequently, the dura mater and the muscle planes and skin were separately sutured. Post-operatively, the animals received meperidine (4 mg/kg) subcutaneously (SC) every 12 h for 2 days for pain, marbofloxacin (2 mg/kg IM) as an antibiotic for 7 days, and meloxicam (0.2 mg/kg SC) as anti-inflammatory treatment.
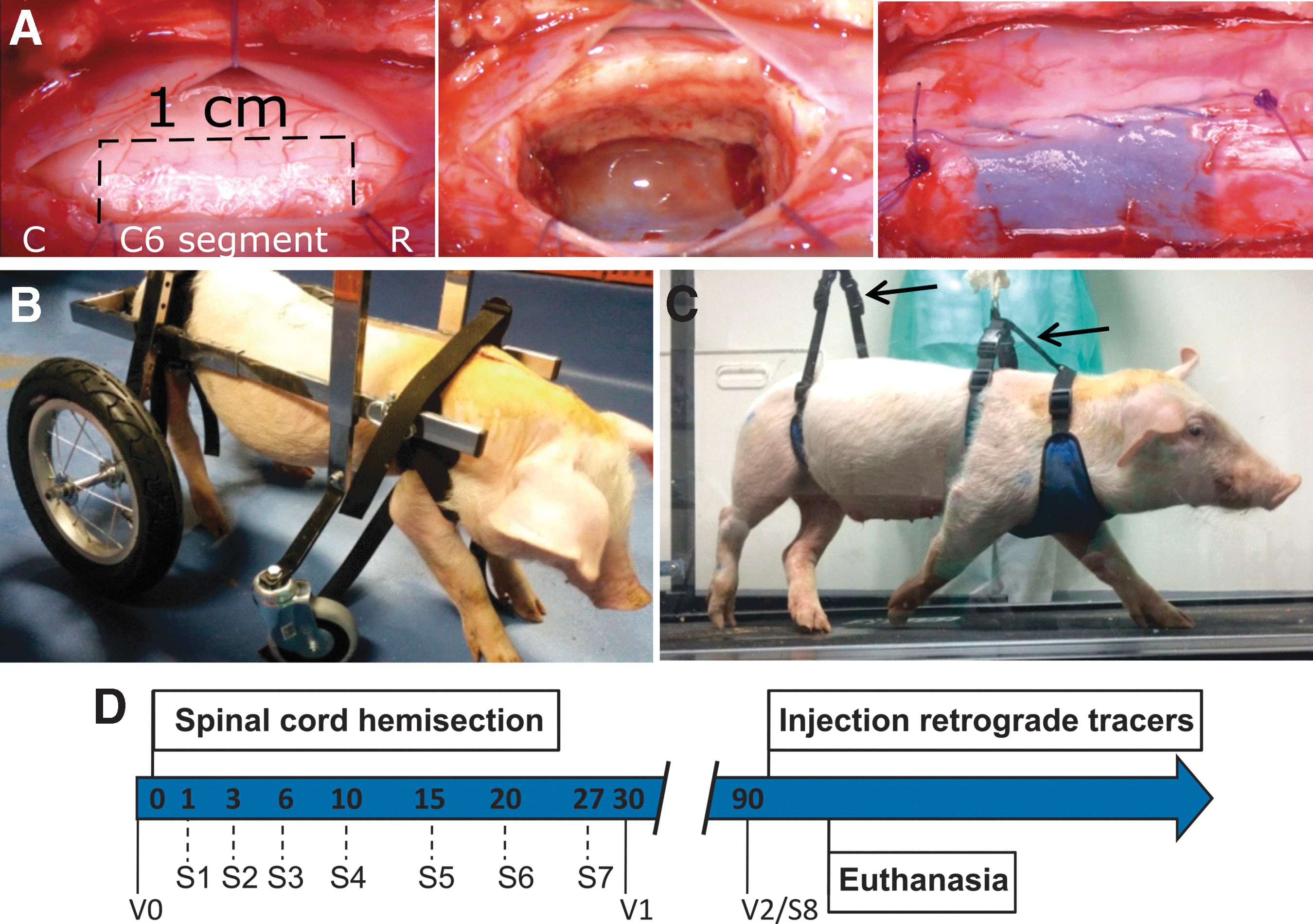
Porcine model of cervical spinal cord hemisection. (
Three months after SCI, three animals of the SCI group were anesthetized again, and the dorsal laminae of the C7 vertebra were removed to expose the spinal cord segment C8. Subsequently, 12 injections (4 μL each, at 2 mm and 4 mm depth in the dorsoventral plane, spaced 5 mm longitudinally) of aminostilbamidine (40 mg/mL, Sigma-Aldrich) were made into the right side of C8 at 2 mm lateral from the midline using a Hamilton syringe. The needle was let in place for 10 min to prevent reflux of the injected solution. We injected a large volume (48 μL) of tracer along the porcine spinal cord segment C8 expecting that diffusion of the tracer in the rostrocaudal direction would effectively make it available to most axons projecting to this segment. The placement of the injections was informed by our previous results in the rat spinal cord showing that aminostilbamidine diffused ∼1 mm from the injection site by 3 days. 11 In the porcine spinal cord, the retrograde neural tracing aimed at documenting the presence of propriospinal neurons (PNs) immediately caudal to the lesion site, given the potential showed by PNs to form synaptic relays after SCI. 58
Animal care and rehabilitation
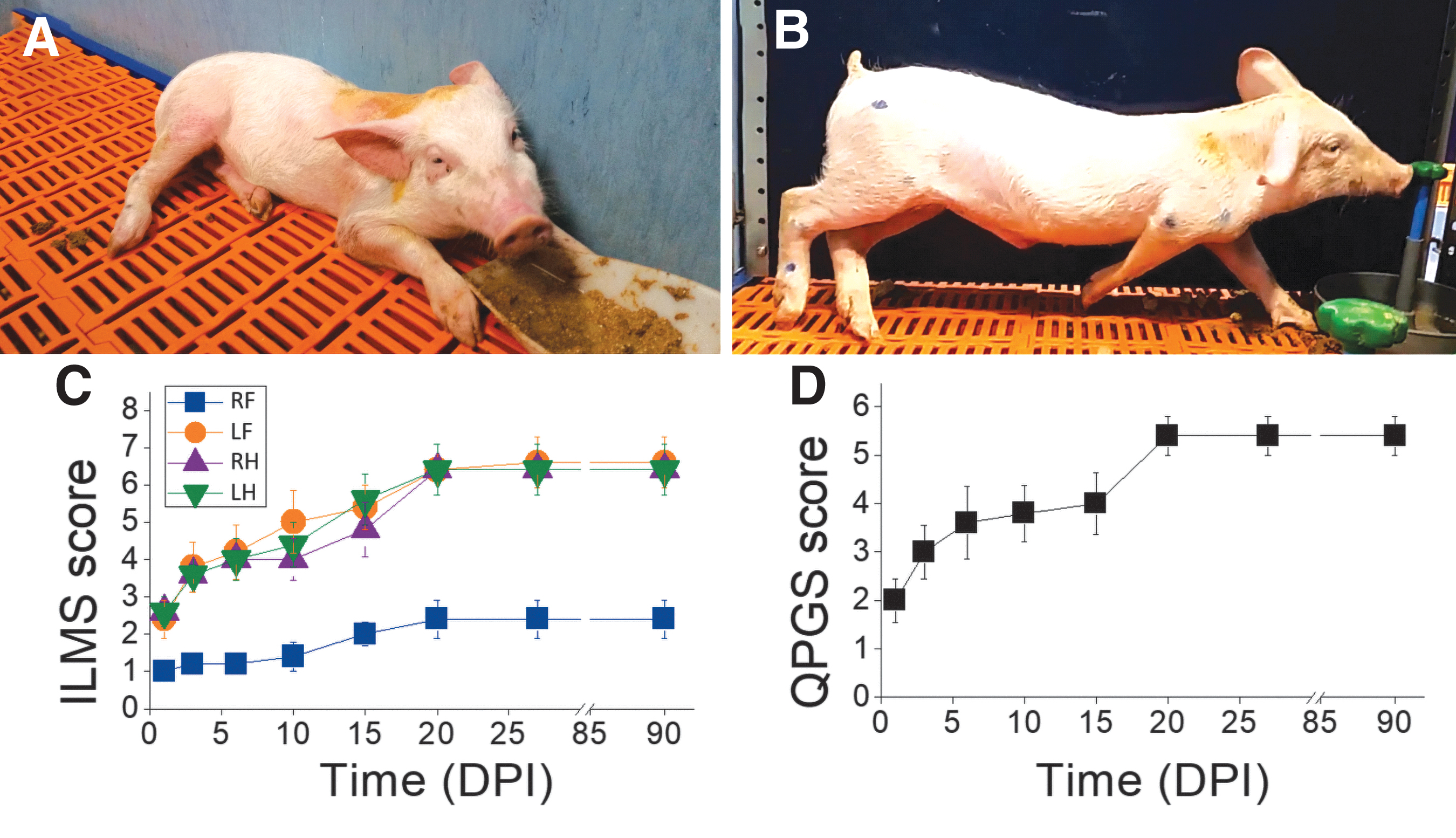
After arrival, the pigs could acclimate to our animal facilities for at least 3 weeks prior to SCI. Meanwhile, they were trained to walk in a treadmill at speeds of 1, 3, and 5 km/h for 30 min per day and to wear harnesses aimed at providing body support after SCI. For the first 15 days post-injury (DPI), they lay on a soft mattress and were helped in feeding and drinking. Assisted standing in wheelchairs for quadruped animals (Fig. 1B) in sessions of ∼45 min started by 3 DPI and continued every day up to at least 30 DPI. During those sessions, the hindlimbs and the forelimbs were moved to reduce tissue atrophy and prevent the appearance of pressure sores. When the animals could stand up continuously for 5 min without help, locomotor rehabilitation was performed on the treadmill in 30-min sessions, 3 times per week. One or two harnesses connected by strain gauges in series to a crane (Fig. 1C) allowed us to provide balance to the animal (i.e., keep its body elevated) and to obtain an estimation of the amount of weight support needed during the walking cycle.
Scales for motor assessment
We separately scored the individual movement of each leg and the posture of the animal, designing an Individual Limb Motor Scale (ILMS) (Table 1) and a Quadruped Position Global Scale (QPGS) (Table 2). The ILMS scores the movement, positioning, and weight bearing ability of each leg with values from 1 (flaccid, inactive limb) to 8 (apparently normal use of the limb); whereas the QPGS scores the axial muscle tone, capacity to acquire prone position and standing up, with values from 1 (hypotonia, absent or slight trunk torsion) to 6 (standing up for at least 5 min). The animals were assessed with these scales at 1, 3, 7, 10, 15, 20, 27, and 90 DPI (Fig. 1D).
Individual Limb Motor Scale (ILMS)
The ILMS is an eight-point scale designed for scoring each individual limb. The animals were assessed with this scale at 1, 3, 6, 10, 15, 20, 27, and 90 days post-injury (DPI).
Quadruped Position Global Scale (QPGS)
The QPGS is a six-point scale for scoring the evolution of the quadruped position after spinal cord injury (SCI). The animals were assessed with this scale at 1, 3, 6, 10, 15, 20, 27, and 90 days post-injury (DPI).
Kinematics of locomotion and measurement of weight support
As mentioned, pigs were trained to walk on a treadmill at different speeds and to wear harnesses in series with strain gauges for weight support measurement. The treadmill was enclosed with transparent methacrylate walls to restrain the pigs from falling or jumping outside. Video recordings of animal locomotion were captured at 125 frames/sec using a PCI 500 MotionScope camera orthogonal to the right side of the body. Skin markers were drawn on the right shoulder, elbow, wrist, metacarpus, hip, knee, ankle, and metatarsus, and their positions were tracked using automatic motion analysis software (WINanalyze V. 2.2, Mikromak GmbH, Münster, Germany). Video recordings were obtained at day 0 (just before SCI) and at 30 and 90 DPI. The analyses included joint angle measurements, variation of eye and hip height, and relative aft-to-forward movements of the distal part of the legs (ankle minus hip, and wrist minus shoulder) along the waking cycle. In addition to kinematics, errors in gait coordination were quantified from the video recordings. The force transducers in series with the harnesses were connected to a computer equipped with Bionomadix software for acquiring the vertical force signal (weight support) at 10 kHz, synchronized with the video recordings.
Normalization and analysis of biomechanical parameters
Normalization was performed to reduce data variability resulting from animal size and placement and movement of the skin markers. Most kinematic and force parameters were divided by their values at the beginning of the stance phase, except for wrist–shoulder and ankle–hip distances, which were divided by the total length of the forelimb or hindlimb, respectively. This procedure produced dimensionless numbers. Data were subsequently normalized to the duration of the right forelimb or hindlimb stride and smoothed (Savitzky-Golay filter, 10-point window, polynomial order 2). At least six walking cycles were averaged per animal and day of assessment. After SCI, some pigs could not place the right forehoof on the treadmill surface. In these cases, the kinematic parameters of the right foreleg (RF) were normalized to the duration of the left hindlimb stride, taking advantage of the almost in-phase coupling observed for the diagonal leg pair when the pig walked at 5 km/h (see Results section). The signals from the force transducers were converted to 125 Hz, normalized to the duration of the right forelimb stride, and smoothed as for kinematics. They were expressed as animal weight percent.
Histological procedures and analyses
After a 2-week period to allow for aminostilbamidine transport, inhalational anesthesia was administered as described, and the entire pig cervical spinal cord was exposed by a dorsal surgical approach. Subsequently, the animals were euthanized with pentobarbital (120 mg/kg IV) and the cervical spinal cord was quickly removed and fixed in 4% paraformaldehyde in 0.1 M, pH 7.35 phosphate buffer for 3 days. Each spinal cord segment was separated and cryoprotected by immersion in 30% sucrose for 4 days, and then was embedded in Optimal Cutting Temperature (OCT) embedding matrix for frozen sections (Pioneer Research Chemicals) and stored at -20°C until cut in the cryostat. Transversal spinal cord sections were cut at 20 μm for immunohistochemistry or at 50 μm for eriochrome cyanine and cresyl violet staining. Histological assessment was performed on the injured and adjacent segments at intervals of 2 mm. In the text and figures, the symbols “-” and “+” are used to identify the coordinates rostral or caudal to the lesion, respectively.
Eriochrome cyanine and cresyl violet-stained sections were photographed with a stereology BX61 microscope (Olympus, Hamburg, Germany) using a 4 × objective. The lesion epicenter was defined as the tissue coordinate with the greatest damage in the transverse plane. Images of cresyl violet-stained sections were used to delineate and measure the areas of the white matter (WM) and gray matter (GM) with ImageJ software, and to quantify neuronal profiles in the ventral horn. In addition, cresyl violet was useful as a counterstain for neurons retrogradely labeled with aminostilbamidine, thus providing a reliable identification of PNs when simultaneously visualized with transmitted light and fluorescence microscopy (Olympus BX51).
For immunohistochemistry, antigen retrieval was performed by heating the tissue sections at 90°C for 25 min in 0.01 M sodium citrate; subsequently, they were blocked for 1 h in phosphate buffered saline (PBS) containing 0.2% triton and 2% normal donkey serum, rinsed three times with PBS, and incubated overnight at 4°C with the primary antibodies: mouse anti-Neuron-Specific Nuclear Protein (NeuN, Chemicon MAB3377, 1:400), goat anti-Choline Acetyltransferase (ChAT, Millipore AB144P, 1:100), mouse anti-Neurofilament 200 (NF) (Sigma-Aldrich N0142, 1:350), or rabbit anti-Glial Fibrillary Acidic Protein (GFAP) (Dako Z0334, 1:400); and dissolved in PBS with 0.2% triton and 1% normal donkey serum. Then, the sections were washed with PBS and incubated for 2 h at room temperature with donkey secondary fluorescent antibodies (Alexa Fluor anti-mouse 594, Alexa Fluor anti-goat 488, or Alexa Fluor anti-rabbit 488; Abcam, 1:1000). Other tissue sections were incubated with Alexa Fluor 488-conjugated isolectin GS-IB4 (IB4, ThermoFisher Scientific I21411, 1:400). Neuronal profiles fluorescently stained for NeuN or ChAT were counted at 2, 4, 6, 8, and 16 mm rostral and caudal to the lesion, using images of the entire transverse extension of the GM acquired at 2776 × 2074 pixels with an Olympus MVX10 microscope equipped with a DP71 digital camera. Illustrations of fluorescence in Figure 2 are images obtained with the confocal microscope Leica TCS SP5 using a 20X objective, and illustrations in Figure 3 are mosaics from images of the ventral horn taken with the same microscope and objective, but applying 2.5 zoom during imaging.
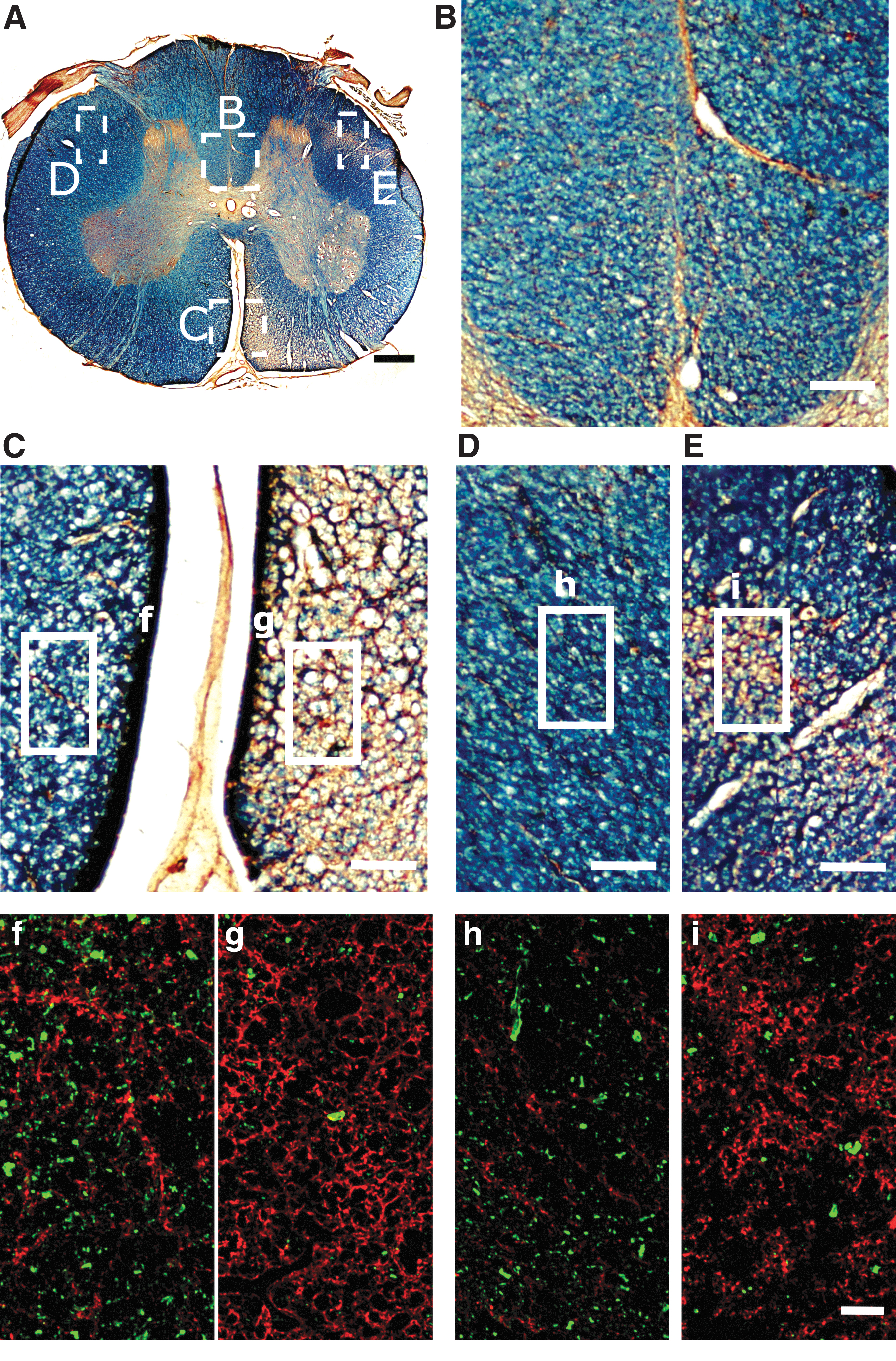
Wallerian degeneration caudal to the hemisection. (
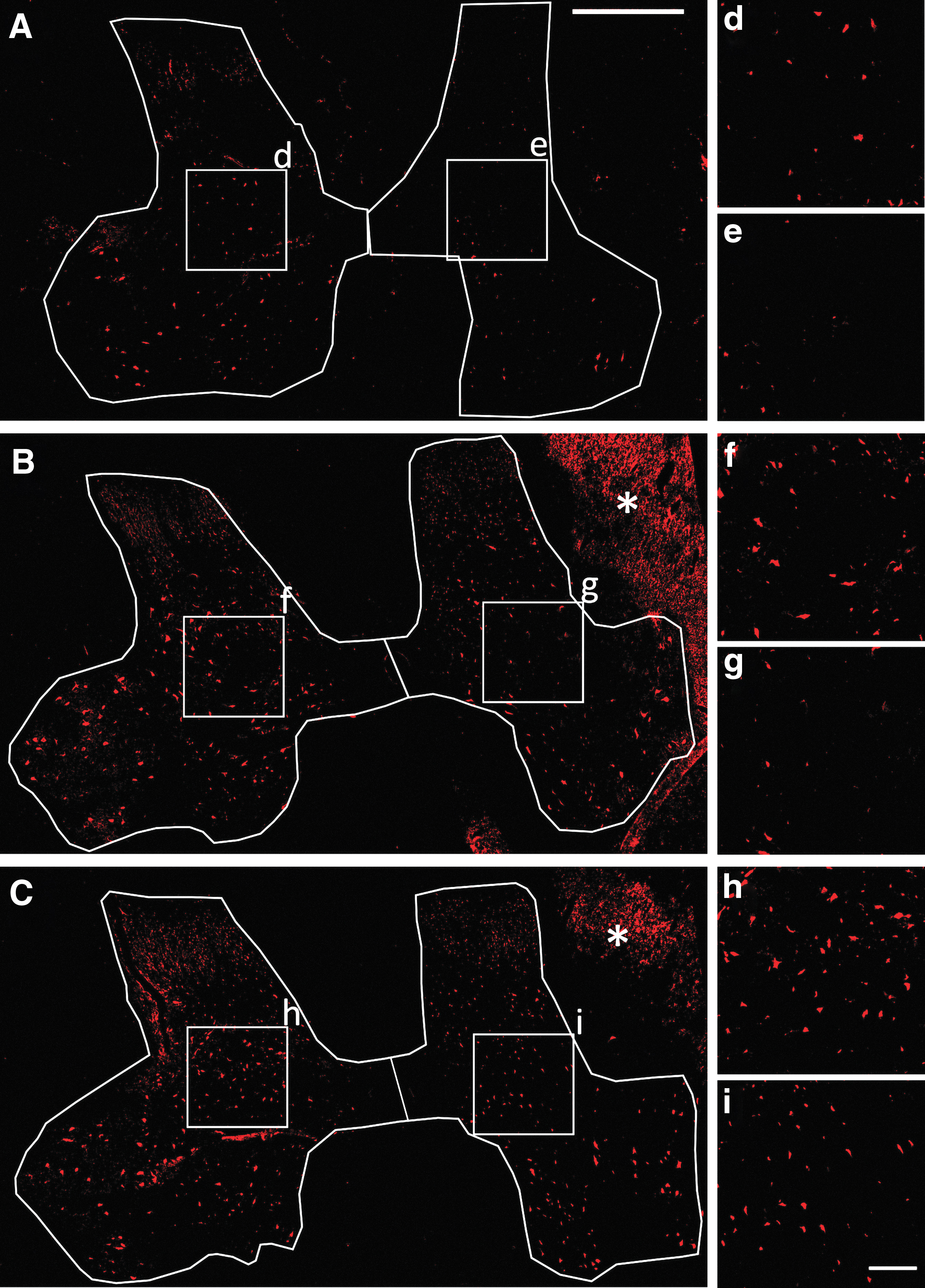
Representative images of spinal cord sections from injured pigs processed for NeuN-immunohistochemistry. (
Statistical analysis
Statistical analyses were conducted using SigmaStat. All values reported, unless otherwise stated, are means ± standard error of the mean (SEM). Differences were considered statistically significant at p < 0.05. WM and GM areas and total number of cresyl violet-stained neuronal profiles, ChAT-positive cells, and NeuN-positive cells were compared by two-way analysis of variance (ANOVA) and Holm–Sidak (HS) post-hoc test. Data from the injured side were usually averaged and compared with the uninjured side for the five animals or with data from the control, uninjured animals. For some parameters indicated in the text, data from the injured side were expressed as percentage of the uninjured side in each animal to reduce bias caused by variability in animal size and histological procedures. Usually, one-way, two-way, and repeated-measures ANOVA followed by the HS post-test were used to compare the average, maximum, and minimum values of biomechanical parameters, considering either the complete walking cycle or its phases as indicated in the Results section. Unpaired Student's t test was used for comparisons involving single measurements from injured and uninjured animals.
Results
General clinical and histological outcomes
All animals survived to cervical SCI without visible complications except for the development of small pressure sores in the skin of the most affected side, as well as minor trauma in other parts of the body when trying to stand up, which was treated with topical ointments and disappeared in ∼2 weeks when the animals moved actively. Micturition was apparently normal. The first DPI, the animals were usually in lateral recumbency on the side ipsilateral to the lesion (right), being unable to acquire the ventral decubitus position and needing manual postural changes. Because of the postural impairments, they also needed human help feeding and drinking for ∼10 days. In this regard, using the wheelchair daily even for short periods during the first DPI benefited the clinical condition of the disabled animals. With the pig in the wheelchair, the caregivers could reposition and mobilize its paralyzed and retracted RF to prevent ankylosis of the wrist, elbow, and shoulder; and it was easier to take care of the skin to prevent pressure sores. The wheelchair also helped the pigs in feeding, and served to motivate them to resume stepping with the hind legs. With the body completely supported by the wheelchair and without risk of falling, the pigs tried to propel their bodies by stepping with the hindlimbs to advance toward the food source by 3 DPI.
In contrast to the almost permanently paralyzed RF, the left foreleg (LF) was initially paretic but after a few days it recovered sufficient strength to support the fore trunk and head, thus allowing the animals to feed themselves without using the wheelchair. Balance loss was progressively compensated for by the less impaired legs and some pigs could stand up by 1 week after SCI. Animal body weight and rostrocaudal length continuously increased during the follow-up period, reaching ∼250% and 150% of the initial values, respectively. No statistical differences were detected in weight and length gain between injured and uninjured animals.
For a better examination of the spinal cord atrophy and neuronal loss caused by the lesion, it was useful to consider first the gross anatomy of the uninjured pig cervical spinal cord (Fig. 4). In the cervical enlargement, the transverse area of the spinal cord increased progressively from C5 to C7 (Fig. 4A, C, E) and the area occupied by WM was ∼80% of the total without significant differences between right and left sides. However, the WM enlarged only ∼14% in the transverse plane from C5 to C7, whereas the GM increased by 70% (Fig. 4B, D), and consequently the ratio of gray/white matter increased from 0.18 ± 0.006 at C5 to 0.26 ± 0.008 at C7 (one-way ANOVA, p < 0.0001). The total number of neuronal profiles (NeuN+ cells) across the entire transverse plane of the GM increased by 21% from C5 to C7 (one-way ANOVA, p < 0.001), showing a positive linear correlation (R = 0.999) with the enlargement of the GM (Supplementary Fig. S1). Nevertheless, cell densities per unit of GM area progressively lowered from ∼102 neuronal profiles per mm2 at C5 to ∼72 profiles per mm2 at C7, likely reflecting the addition of large cells along the cervical enlargement. In fact, neuronal profiles in the ventral horn increased as much as 83% from C5 to C7 (one-way ANOVA, p < 0.001, Fig. 4F). As in humans 59 and rodents, 60 several motoneuron columns were segregated in the ventromedial and ventrolateral GM of the porcine spinal cord (Fig. 4E). The ventromedial columns for axial muscles were present along all the studied segments, while the ventrolateral columns for the foreleg muscles made the lateral GM protrude progressively into the WM from C5 to C7, thus resembling the gross appearance of the human cervical spinal cord enlargement.
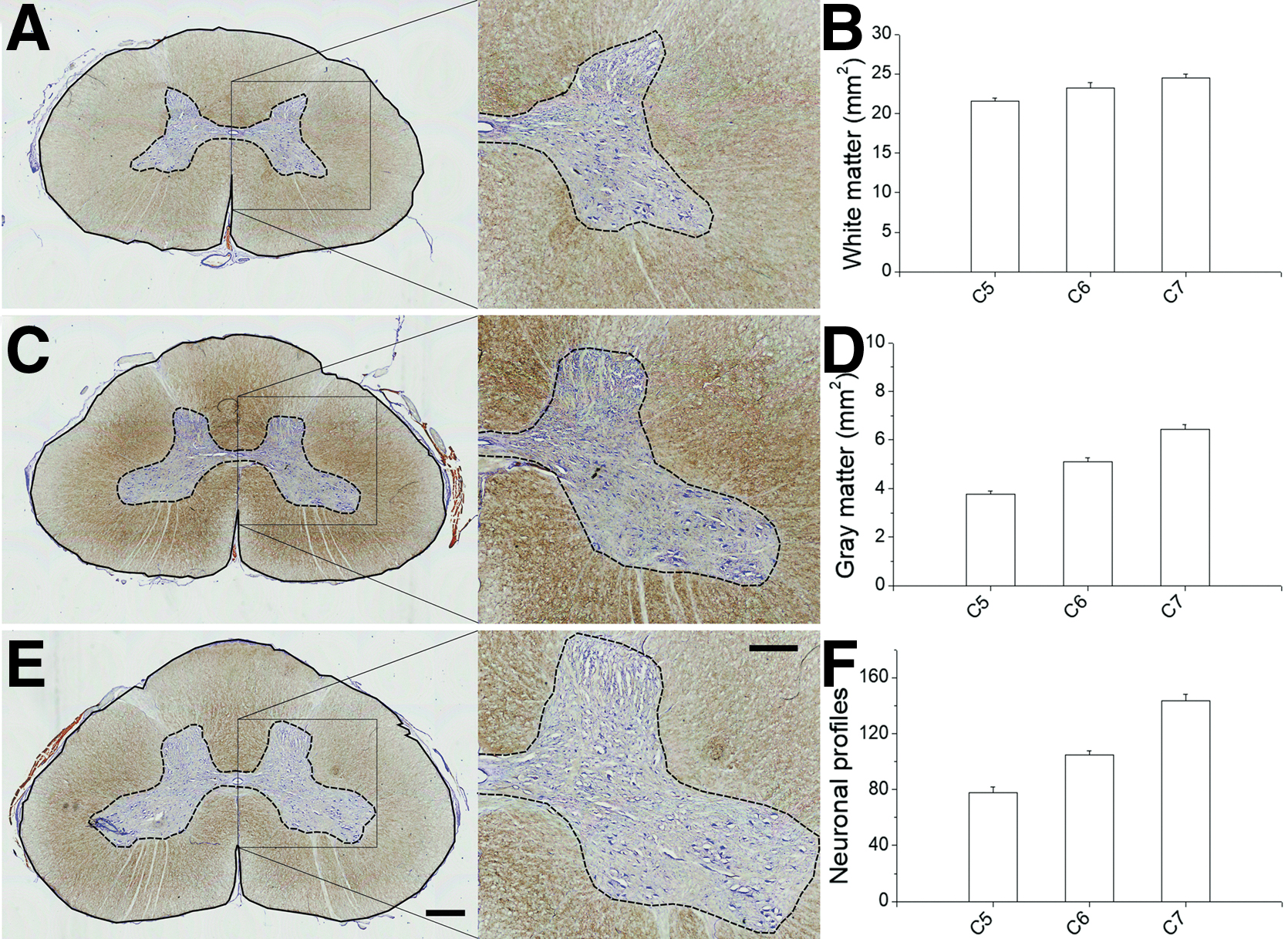
Anatomy of the cervical spinal cord enlargement in 6-month-old uninjured pigs. (
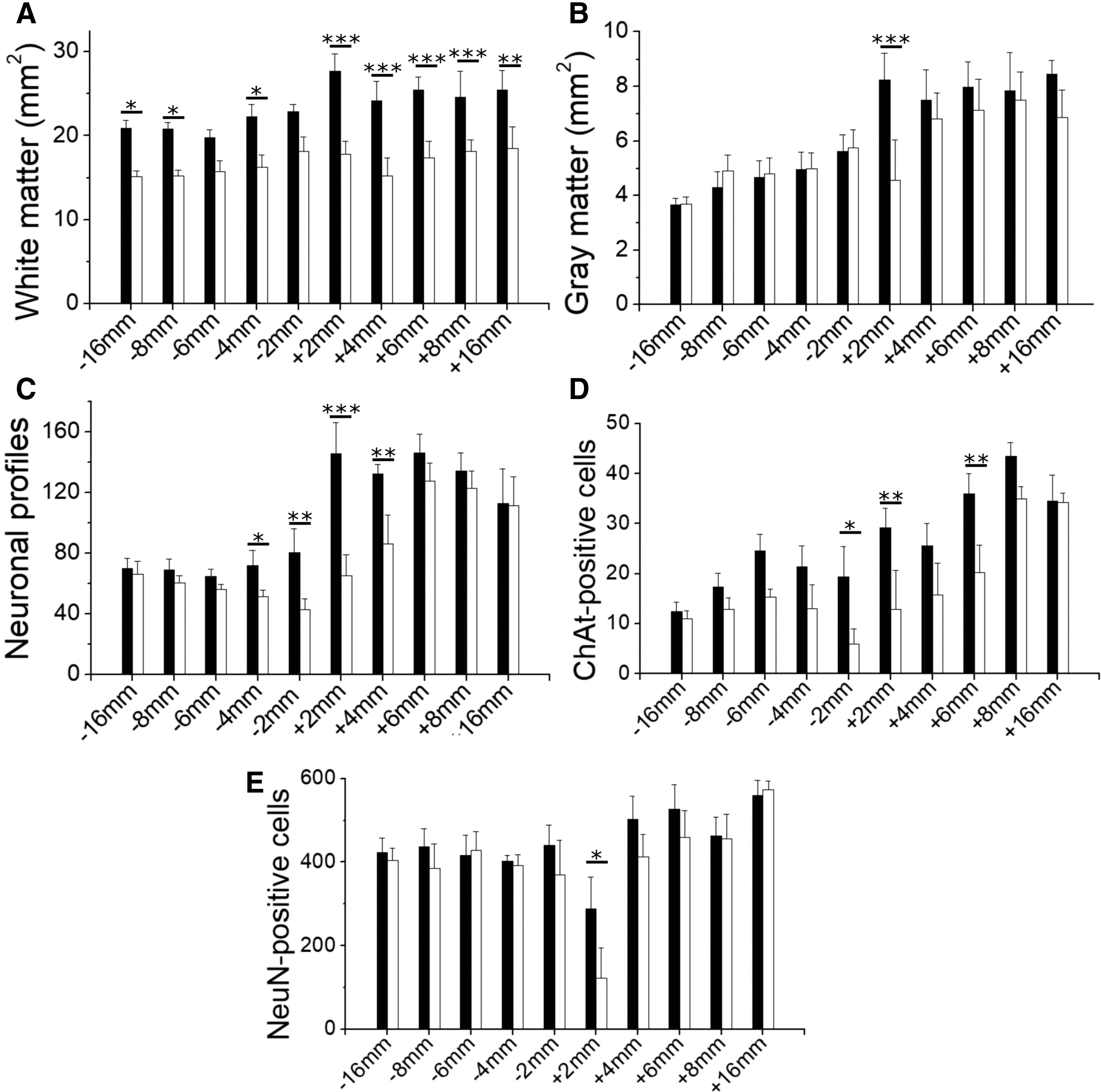
Quantification of histological changes in the spinal cord tissue rostral (-) and caudal (+) to the lesion, performed when the injured pigs were 6 months old. (
A complete right-sided spinal cord hemisection was verified in the five animals (Fig. 5). Additionally, some damage was inflicted to the left side of the spinal cord, mainly involving the gracilis fascicle and the GM, albeit a larger WM involvement occurred in two pigs (Fig. 5B and E). Injury to the untargeted side was most likely vascular in origin. Central branches of the anterior longitudinal arterial trunk penetrated through the ventral median spinal fissure, bifurcated, and spread out laterally into the white and gray matter. 61 Arterial branches supplying the left side might have been inadvertently cut during midline incision of the spinal cord, leading to ischemic tissue damage, further worsened by the hemorrhage and associated vasoconstriction, and by the actions taken to achieve hemostasis.
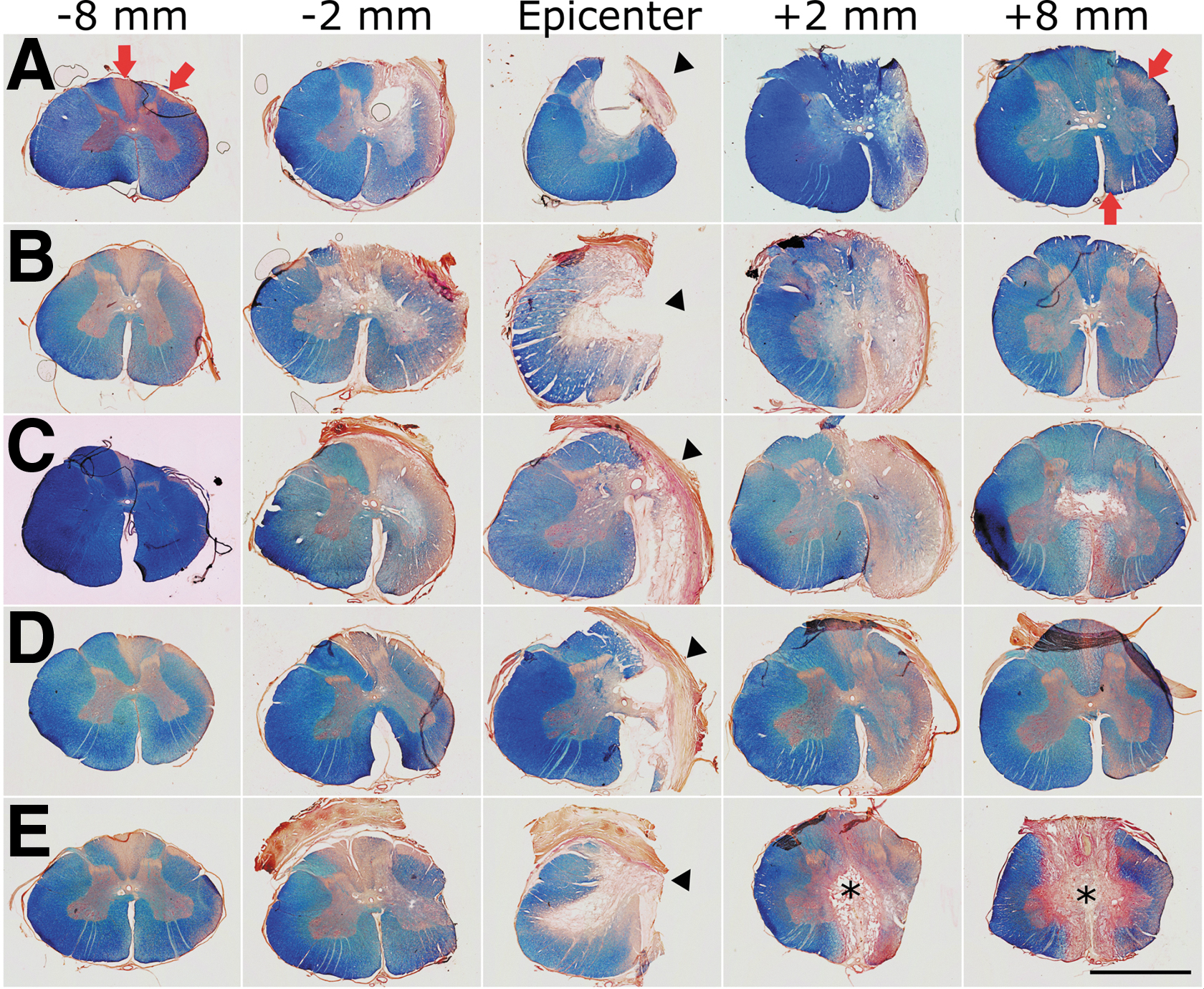
Appearance of the spinal cord at 15 weeks after injury. (
Lesion length, defined as the distance between the most rostral and the most caudal transverse tissue sections in which the spinal cord tissue appeared complete, was 1.24 ± 0.13 cm. Because only 1 cm of tissue was excised, it is possible that the additional 24% of lesion length measured in histology was the result of elongation of the spinal cord because of the continued growth of the animals as previously mentioned. However, the injured spinal cord segments were still 14% shorter than the same segments from age-matched uninjured animals (1.25 ± 0.06 cm for injured, vs. 1.45 ± 0.11 cm for healthy controls, t test p = 0.05).
The spinal cord tissue very adjacent (∼2 mm) to the lesion showed severe atrophy, particularly caudal to the lesion (Fig. 5). Additionally, several small cavities were present in the WM and GM for ∼6 mm longitudinally in the caudal stump (Supplementary Fig. S2). These degenerative changes were much less frequent in the rostral stump. While the gross appearance of the ipsilateral GM substantially improved with increasing distance from the lesion border (Fig. S2), atrophy of the WM with signs of axonal Wallerian degeneration (WD) was observed in the complete length of tissue analyzed (3 and 4 cm caudal and rostral to the lesion borders, respectively). Regions undergoing WD appeared as pale areas in eriochrome cyanine-stained sections because of myelin loss, and were almost devoid of axons as evidenced by neurofilament immunohistochemistry (Figs. 2 and 5, Supplementary Fig. S3). Moreover, hypertrophied astrocytes formed a honeycomb-patterned structure surrounding the microcavities that resulted from myelin and axonal degeneration (Fig. 2, Fig. S3), and putative reactive microglial (IB4+) cells apparently engulfed and contributed to the elimination of degenerating axons (Supplementary Fig. S4). As expected, the main regions affected by WD were different rostral and caudal to the lesion (Fig. 5). In caudal segments, they corresponded to the location of the major tracts descending from supraspinal centers, namely the putative rubrospinal and corticospinal tracts in the dorsolateral fascicle, and the reticulospinal and vestibulospinal tracts in the ventromedial fascicle (Figs. 2 and 5); in rostral segments they matched to the location of ascending tracts in the dorsal columns, as well as the putative spinothalamic and spinocerebellar tracts in the lateral area (Fig. S3). The transverse areas of the WM and GM were quantified for the segments rostral and caudal to the lesion and compared with the values of the contralateral side (Fig. 6A, B) and to those obtained for healthy controls (Supplementary Fig. S5). The ipsilateral WM area was smaller in almost all coordinates (two-way ANOVA, p < 0.001; Fig. 6A), whereas the shrinkage of the GM was statistically significant only at +2 mm (p < 0.05; Fig. 6B). The contralateral WM and GM (rostral and caudal to the lesion site) showed no reduction in transverse area compared with the spinal cord of uninjured pigs (Fig. 4B, D; Fig. S5).
Neuronal death adjacent to the lesion
Initially, neuronal profiles were counted in the ventral horn using cresyl violet-stained tissue sections (Fig. 6C), and a significant cell loss was found extending at least 4 mm from the lesion borders into both spinal cord stumps (two-way ANOVA p < 0.001). The reduction in cell counts was more pronounced just caudal to the lesion (55% of cell loss at +2 mm, p < 0.001), in agreement with the cavitation and atrophy of the GM observed at this coordinate (Fig. 6B and Fig. S2). Neuronal counts contralateral to the hemisection were not statistically different compared with values of healthy subjects.
To obtain further insight into the ventral horn neurons affected by the lesion, we counted cholinergic neurons as visualized by ChAT immunohistochemistry (Supplementary Fig. S6). Similarly to data reported for cresyl violet-stained cells, cholinergic neurons were significantly reduced from -2 to +6 mm of the ipsilateral perilesional tissue, with a more severe cell loss (∼ 50%) closest to the lesion borders (two-way ANOVA p < 0.001, HS post-test p < 0.05 and p < 0.01 for -2 mm and +2 mm, respectively, Fig. 6D). Cell counts were similar at +8 mm, although motoneurons seemed somewhat atrophic on the ipsilateral side (Fig. S6).
The large size of the motoneurons made them easier to be distinguished from other cells in cresyl violet-stained sections; however, small neurons of the intermediate and most dorsal laminae could be confused with other cells if not appropriately labeled. Therefore, we further quantified cells using immunohistochemistry for NeuN. Counting all NeuN+ cells in the dorsoventral plane of the GM (Fig. 6E), a statistically significant loss of neurons was detected only at +2 mm (p < 0.05, Fig. 6E). However, loss of NeuN+ cells was evident up to +6 mm in some animals (Fig. 3). To examine differences in neuronal loss among the ventral (VII, VIII, IX) laminae and the dorsal (I, II, III, IV, V, VI) laminae, we added the number of NeuN+ cells at these locations from coordinates +2, +4 and +6 mm for each animal. In addition to confirming the disappearance of ∼33% of neurons in the ipsilateral ventral horn (paired t test, p < 0.05), this procedure unveiled a significant neuronal death in the ipsilateral dorsal horn (paired t test p < 0.01). Within the dorsal horn itself, laminae I, II, and III were more severely affected than laminae IV, V, and VI (37% vs. 20% of cell reduction, respectively, p < 0.05).
PNs projecting to more caudal spinal cord segments are abundant in laminae VI and VII of the GM in the cervical spinal cord. 11,58 As described in the Methods section, we injected the retrograde neuronal tracer aminostilbamidine at specific coordinates in the ipsilateral spinal cord caudal to the lesion in an attempt to label PNs projecting from C7 to more caudal segments. However, the fibrosis around the dura and atrophy of the spinal cord precluded accurate placement of the injections and led to very variable neuronal labeling in the animals that received the tracer. Nevertheless, the preliminary results suggest that the procedure is appropriate to label PNs and that this neuronal type is also lost in the caudal spinal cord stump ipsilaterally to the lesion. Figure 7 illustrates cells in laminae VII caudal to the lesion. Neuronal death and tissue cavitation were observed close to the injury site in the ipsilateral but not in the contralateral side, the latter showing several retrogradely labeled cells with typical neuronal morphology. As with other histological procedures, there was no apparent cell loss at more caudal coordinates (Fig. 7). No signs of unspecific neuronal labeling by tracer spreading to adjacent rostrocaudal segments were observed. Fluorescent neurons appeared mostly in laminae VII and VIII as expected, 11 and none was labeled in lamina IX. On the other hand, although the tracer injections were made slowly and the volume was distributed in two tissue depths at each longitudinal coordinate, some of the tracer refluxed through the needle path, spilling into the cerebrospinal fluid, thus allowing its uptake by meningeal and perivascular cells, as well as by cells of microglial morphology in the WM far from the injection site.
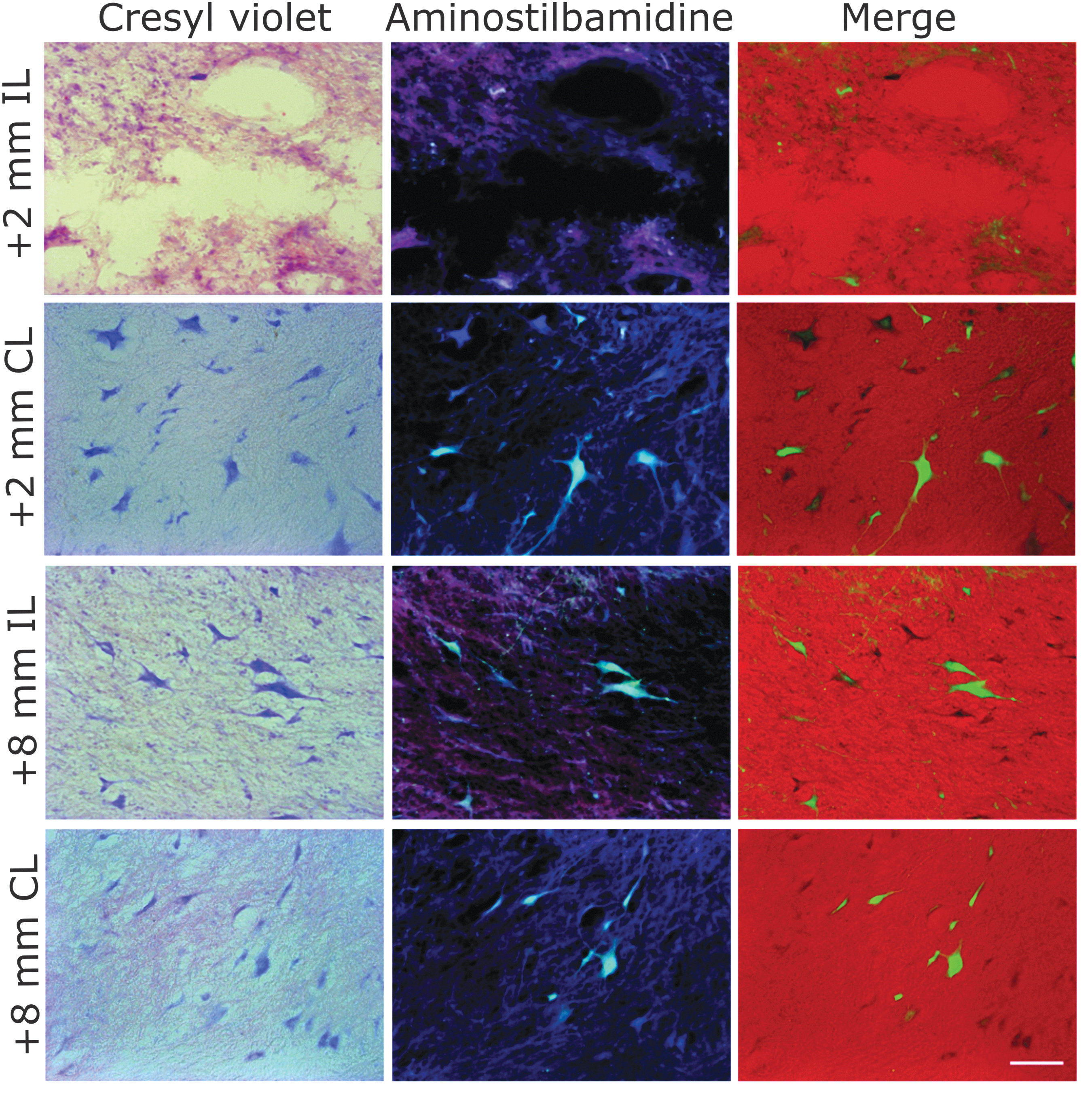
Propriospinal neurons from intermediate gray matter (GM) laminae caudal to the lesion as visualized after retrograde fluorescent labeling with aminostilbamidine injected at C8 and cresyl violet counterstaining. A marked neuronal loss was found close to the lesion border, whereas numerous “healthy” neurons were labeled at the same coordinates in the contralateral side, and bilaterally at more caudal regions. Scale bar, 100 μm. Color image is available online.
Our initial purpose was elucidating the state of neurons in the spinal cord stumps as a basis for future assessment of axonal growth and functional reconnection across the tissue gap. As already described, we focused our analysis to coordinates rostral and caudal to the injured segment, finding reduced neuronal numbers ipsilaterally. However, no cell loss occurred in the contralateral side at the same coordinates in comparison with healthy animals. Evidently, the two animals that, in addition to the hemisection, underwent substantial damage to the contralateral spinal cord (Fig. 5B and E) had reduced counts of cresyl violet, ChAT+, and NeuN+ cells in the contralateral side (not shown), but this was restricted to the injured region and did not extend to the rostral and caudal segments. Nevertheless, as shown in the next sections, unintentional damage to the contralateral spinal cord in those two pigs worsened their functional outcomes.
Postural and motor impairments
All injured animals had monoplegia or severe monoparesis of the RF immediately after SCI, with little recovery during the 90 DPI as assessed by the ILMS scale (Table 1, Fig. 8 A, C). The other three legs initially had an abnormal position and were very weak, but showed progressive motor recovery during the first 3 weeks and became strong enough to support the body for standing and stepping (Fig. 8 B) in the pigs with little damage to the contralateral (left) spinal cord. Usually, the LF recovered faster than the left hind leg (LH), which in turn recovered faster than the right hind leg (RH). Considering the complete group of injured animals, no statistical differences were found in the ILMS of the RF at 1 DPI compared with later days (i.e., it remained dysfunctional), whereas the LF scored significantly better from 10 DPI onward (two-way ANOVA p < 0.001), the LH scored significantly better from 15 DPI onward (p < 0.001), and the RH scored significantly better from 20 DPL onward (p < 0.001). Further, the RF scored much lower than the other legs on the ILMS from 3 DPI on (two-way ANOVA, p < 0.01). The two pigs with bilateral damage to the dorsal columns and GM (Fig. 5 B, E) showed greater motor impairment in all legs (Supplementary Figs. S7 B, E). In particular, none of the legs had an ILMS >4 in the pig shown in Fig. 5 B; whereas both hind legs showed a plateau of recovery at an ILMS of 6 in the pig shown in Fig. 5 E. For comparison, all legs except for the RF reached an ILMS score of 7 or 8 in the three pigs with mostly unilateral spinal cord damage.
Limb use and posture recovery after cervical spinal cord hemisection. (
The ILMS allowed us to document the permanent, focal impairment of the RF and the gross functional recovery of the other three legs. However, this scale provided no information regarding the movement of the trunk and its contribution to posture and body balance. These aspects of motor behavior were assessed with the QGPS (Table 2), showing an improvement during the first 3 weeks post-lesion (Fig. 8D) in parallel with the ILMS score for the three less-affected legs. All injured animals recovered from the hypotonia and disability of the trunk that initially impeded them from getting into the ventral decubitus position. As a result, statistical differences in the QGPS score started to be noted by 6 DPI (one-way ANOVA, p < 0.01) and increased later. The three pigs with almost unilateral spinal cord damage were able to stand up and keep the body elevated for long periods between 7 and 20 DPI, whereas those with the largest bilateral damage (Fig. 5 B, E) were unable to stand up by themselves for at least 5 min (i.e., had a QGPS <6) by 28 DPI (Fig S7 B, E).
Kinematics of locomotion
The kinematics of treadmill locomotion was studied in five uninjured pigs and the three spinal cord-injured pigs that reached a QGPS of 6. The other two injured pigs were unable to stand by themselves for sustained periods and therefore received no treadmill training or further locomotor analysis. Pigs moving at 1 km/h were easily distracted and moved irregularly, thus increasing kinematic data variability. This trouble magnified after SCI. Therefore, data from locomotion at 1 km/h and 3 km/h were only used for studying the influence of speed on normal gait kinematics, and data from locomotion at 5 km/h were used for comparisons of motor performance before and after SCI.
After 1 or 2 weeks of training, uninjured pigs were proficient in treadmill locomotion, and we proceeded to analyze their kinematics at different speeds (Fig. 9). Initially, we quantified the step cycle parameters for the four limbs, as well as interlimb coupling in stance. The duration of the stride, stance phase, and swing phase, as well as the duty factor, were very similar for the four legs at a given treadmill speed and showed speed dependence as described for quadruped mammals in overground locomotion, 62 as well as for Yucatan minipigs in treadmill locomotion. 63 The mean duration of the stance phase decreased exponentially with velocity, whereas the duration of the swing phase showed only a small linear decrease (Fig. 9 A). As a consequence, the duty factor decreased from 0.75 ± 0.03 at 1 km/h to 0.56 ± 0.01 at 5 km/h (one-way ANOVA, p < 0.001), indicating that the gait style changed from slow walking to trotting. 62 The supporting limbs mostly alternated in diagonal pairs with 77.60 ± 1.77% RF/LH overlapping at 5 km/h, whereas RF/LF and RH/LH overlapped only 22.56 ± 3.87% and 14.59 ± 4.91%, respectively. RF landing preceded LH landing 0.13 ± 0.04 s at 1 km/h, and this time was reduced to 0.05 ± 0.004 s at 5 km/h (one-way ANOVA, p < 0.05), favoring the diagonal stance coupling.
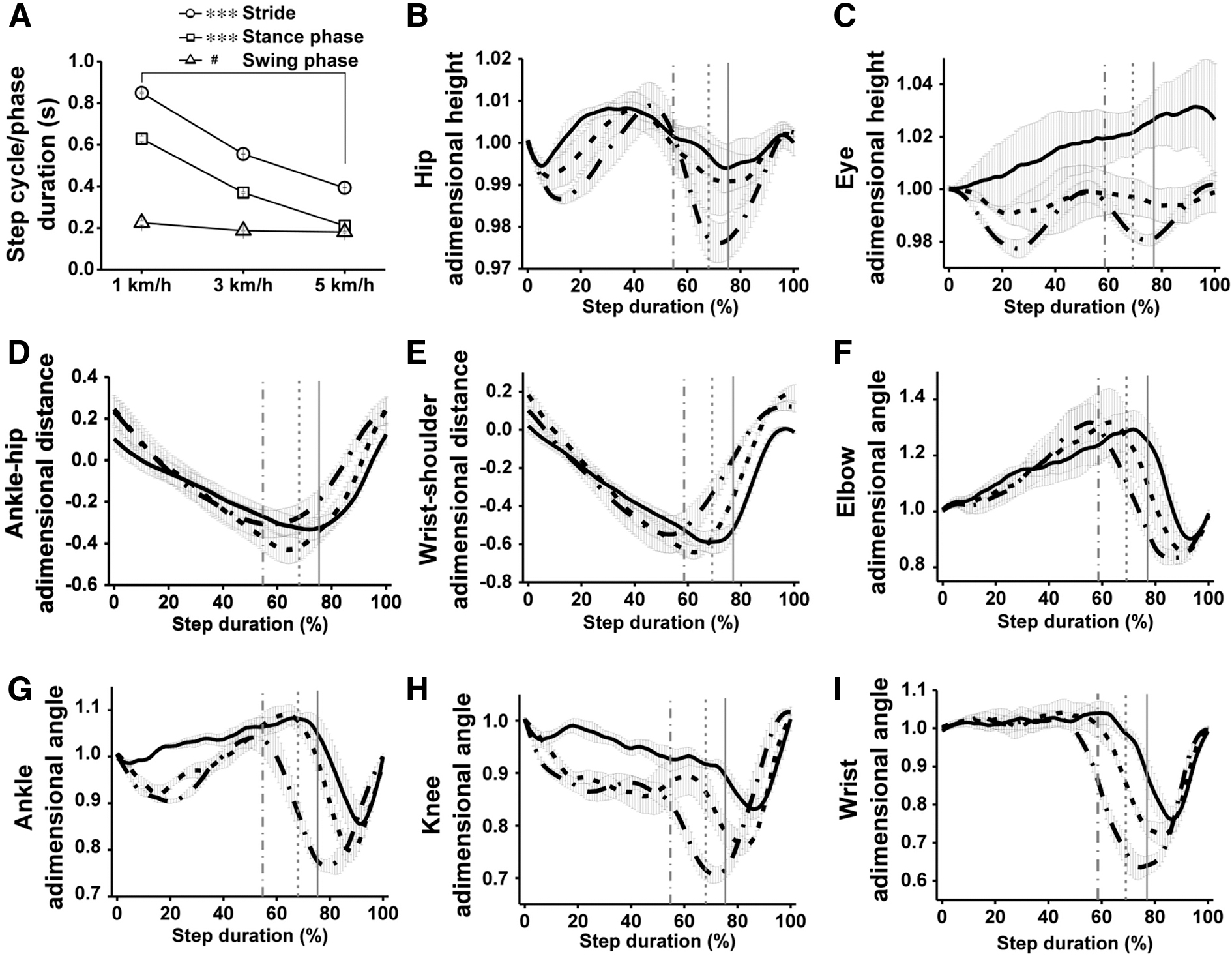
Kinematics of locomotion in uninjured, 2-month-old domestic pigs. (
Hip height and eye height are indicators of the capacity of the hindlimbs and forelimbs to keep the body elevated, and therefore their fluctuations along the step cycle provide information regarding balance. 11 Rhythmic hip and eye height oscillations (3–4% of the initial value) with two differentiated peaks per stride were produced at 5 km/h (Fig. 9 B, C) by the sequential action of the RF/LH and LF/RH diagonal pairs. Although the movement of the pig head during locomotion is complex and relies on the legs and on several neck muscles, the elbow contributed to raising the head as indicated by the coincidence of the peaks of eye height and elbow extension in the last third of the RF stance (Fig. 9 C, F). As described for rats, 11 one of the key dysfunctions after cervical spinal cord hemisection is the loss of elbow extension force that disables the forelimb to receive the accelerated body, impairing balance and leading to fall of the fore part of the body. A similar phenomenon was observed in pigs as will be shown.
The relative position of the distal joints to the proximal ones (wrist-shoulder, ankle-hip) in the aft-to-forward plane provides information about intralimb coordination and use of the limbs for locomotion. The representation of pig ankle-hip (Fig. 9 D) showed a smooth curve in which the ankle moves from ∼0.3 at landing to ∼ -0.3 at lift off, returning to the initial position at the end of the swing phase. This relative ankle-hip position is similar in the rat 64 and facilitates supporting and propelling the center of mass of the animal. Nevertheless, the pig forepaw was placed only little forward to the shoulder at landing and was retracted for a long distance during the stance (Fig. 9 E). The initial forepaw position almost beneath the shoulder likely prevented a large breaking force that would oppose forward movement, while the prolonged forelimb retraction allowed the body to stay elevated being transferred from the hindlimb. Angles recorded for the healthy pig right elbow, ankle, knee, and wrist are shown in Fig. 9 F–I. The maximum joint flexion during the swing phase consistently increased for all joints from a treadmill speed of 1 km/h to 5 km/h (one-way ANOVA, p < 0.05), but little or no changes occurred in the maximum joint extension during the stance phase. In fact, the hindlimb joints tended to increase their flexion also when supporting the body at high speeds, in agreement with the oscillations of hip height. Elbow angular movement in pig was like that in rat, 11,64 but wrist angular movement in the pig differed from that of rat because it did not further extend after landing, being essentially fixed at the maximum extension at the beginning of the stance (Fig. 9 I). Nevertheless, the wrist was actively flexed during the swing phase, increasing the flexion (minimum joint angle) from 0.76 ± 0.03 at 1 km/h to 0.62 ± 0.03 at 5 km/h (one-way ANOVA, p < 0.05).
Three to four weeks after SCI, the three pigs with little damage in the contralateral spinal cord could walk overground and on the treadmill at 1 km/h, albeit with difficulty because of the great impairment of the RF. As for the two pigs incapable of self-standing, two of the three pigs able to stand showed abnormal RF posture without placement of the right hoof on the ground during the 3 months of assessment (Fig. 10 A, middle and right panels). However, after a few sessions of motor retraining on the treadmill with the aid of harnesses, providing partial weight support to the forepart of the animals was sufficient to prevent their falling and to assess their locomotion at 3 and 5 km/h. Kinematic data from locomotion at 5 km/h showed less variability and therefore were used for statistical analyses. At this treadmill speed, the pigs required some external weight support along both the RF/LH and the LF/RH strides, but this aid was critical at the end of the RF/LH stance when the impaired RF could not hold the accelerated animal body, producing a large peak in the weight measured by the strain gauge connected to the harness (Fig. 10 C). The weight support provided by the harness during the stance of the RF/LH in the spinal cord-injured pigs peaked at 62.25 ± 20.34% of body weight at 90 DPI, a value substantially higher than the 9.58 ± 1.07% measured for healthy age-matched controls. The need of weight support showed no significant changes over the period of assessment (30–90 DPI, Fig. 10 D).
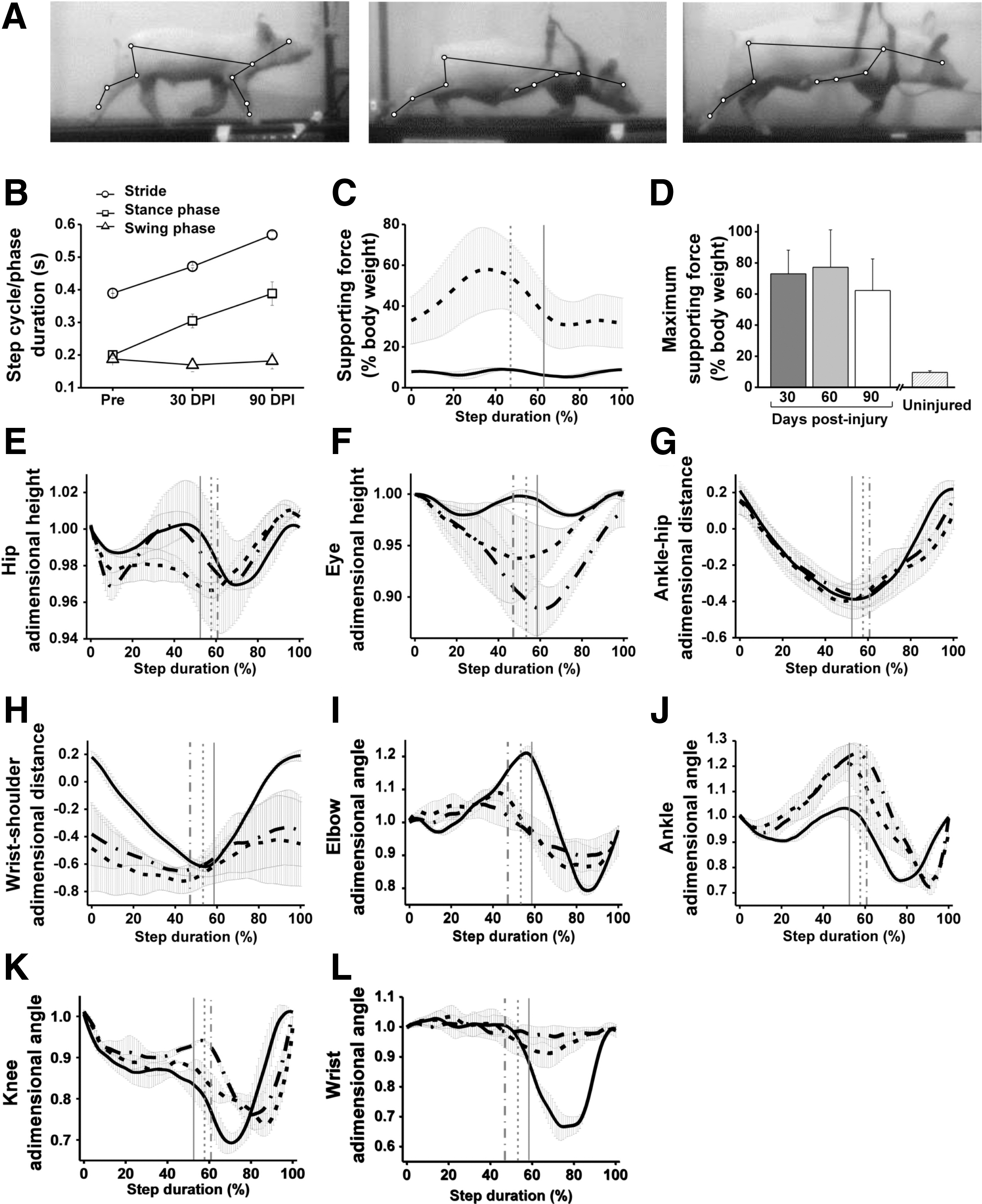
Pig gait kinematics and dynamometry at 5 km/h before and after C6 spinal cord hemisection. (
We likewise compared the kinematics of locomotion at pre-injury, 30 DPI, and 90 DPI, averaging the data from the three pigs. As already mentioned, the RF was chronically dysfunctional, but the three animals were able of stepping using the other three limbs and could adapt their movements to deal with the functional impairments. Stride and stance durations for the three limbs (LF, RH, LH) progressively increased after injury (Fig. 10 B), and by 90 DPI, they were ∼46% and 94% longer, respectively, than the pre-injury values (one-way ANOVA p < 0.01 and p < 0.001, respectively). Part of this increase was a compensation by the other legs for the RF impairment, and part was simply a consequence of body growth that demanded fewer steps (and therefore longer stride and stance durations) for the same velocity, as attested by a drop of 30% in step frequency by 90 DPI. As a reference, step frequency at 5 km/h also decreased by 23% in healthy age-matched pigs, accompanied by an increase of 33% and 45% in stride and stance durations, respectively.
On average, the RH was highly functional as indicated by the ability to elevate the hip during stance (Fig. 10 A, E) and the displacement of the ankle relative to the hip along the stride (Fig. 10 G), which were very similar to uninjured controls. Further, the RH was overused, as attested by the increased extension of the ankle and knee at the end of the stance phase (Fig. 10 J, K, one-way ANOVA, p < 0.05 for the maximum joint angles). On the contrary, the RF was retracted in the animals that could not land the right hoof, with a very negative wrist-shoulder position at the beginning of the stance that was maintained along the stride (Fig. 10 H). On average, elbow extension was markedly reduced after SCI (maximum extension of 1.21 ± 0.02 pre-injury, 1.09 ± 0.03 at 30 DPI, and 1.06 ± 0.04 at 90 DPI, one-way ANOVA, p < 0.05; Fig. 10 I), and there was almost a complete loss of wrist flexion (Fig. 10 L). As mentioned, RF sensorimotor impairment negatively affected balance during locomotion. Even when using the harness to provide partial weight support, the pigs could not elevate the forepart of the body during the stance of the diagonal pair RF/LH, and instead tended to fall, as reflected in the progressive negative values of eye height (1.0 ± 0.01 pre-injury, -0.92 ± 0.04 at 30 DPI, and -0.88 ± 0.02 at 90 DPI, one-way ANOVA, p < 0.05; Fig. 10 F). Nevertheless, eye height recovered to the initial values during the stance of the RH/LF.
Comparisons of motor data from individual pigs at 90 DPI against age-matched (5-month-old) uninjured controls were performed to identify common functional deficits as well as to confirm the suitability of the chosen kinematic parameters for describing different degrees of functional impairment. This information is reported in the Supplementary Text, as well as in Supplementary Figures S8 and S9.
Finally, it is worth mentioning that despite the relatively good performance of the hindlimbs for locomotion in the treadmill after SCI, they still showed persistent sensorimotor errors interfering with the stride. Those errors varied in presentation between cycles and animals, and included trips (when one of the hindlimbs hit the contralateral hindlimb during the swing phase), rear-falls (when the hindquarters dropped and got in contact with the treadmill surface as a result of collapse, trip, or lack of coordination of the hindlimbs), lateral imbalance with the pig hitting the methacrylate walls of the treadmill, and miscellaneous errors such as hindpaw scuffing, involuntary hindpaw slipping, hindpaw rotation, and tiptoeing (stepping on the very tip of the hoof). Taking all errors together, pigs with SCI made from 1 to 4.8 errors per stride at 5 km/h, and only the pig that was able to land the right hoof on the treadmill had less than one error per stride, whereas control pigs (either pre-injury or uninjured age-matched controls) usually showed no errors at all. Hindlimb trips are of interest because they may represent a kinematic correlate of the interruption of proprioceptive signals carried by the spinocerebellar tracts and dorsal columns. 65,66 By 30 DPI, injured pigs had 0.51 ± 0.15 trips per stride at 5 km/h, and this value remained essentially unaltered (0.49 ± 0.10 trips per stride) by 90 DPI, in line with other permanent functional impairments. No trips were detected in uninjured animals.
Discussion
This work characterized the neuropathology and motor impairments that ensue from a cervical spinal cord hemisection with excision of 1 cm of neural tissue in domestic pigs. A basic methodology of histological techniques combined with neural tracing documented the extent of neuronal loss in the adjacent spinal cord segments, whereas motor scales, kinematic analyses, and weight support measurements during treadmill locomotion allowed for a precise quantification of motor function loss and recovery. Although the pathological changes and the severity of functional impairments had some inter-subject variability, a typical pattern of neural damage and chronic motor disability was consistently observed in all animals and demonstrated the reliability of the SCI model. Significant neuronal loss was observed in the spinal cord tissue adjacent to the lesion, accompanied by WD and atrophy of the WM. All pigs showed severe and chronic paresis of the ipsilateral forelimb, whereas the hindlimb recovered substantially, thus manifesting a motor syndrome equivalent to that reported for rats, primates, and humans with asymmetric cervical SCI. These results provide a baseline for testing cervical SCI repair strategies with a translational perspective.
One important issue regarding the use of domestic pigs for SCI research is that weight gain makes them increasingly troublesome for handling and rehabilitation and may introduce bias in kinematic analyses that must be carefully controlled. Depending on breed and diet, a healthy white large pig can exceed 70 kg when reaching sexual maturity at ∼7 months of age and may increase to >200 kg in the next 12 months. This led us to produce SCI in 2-month-old pigs weighting between 10 and 15 kg, with the expectation of increasing weight to ≤50 kg during the follow-up period. It was then possible to take care of the pigs without great effort and allowed us to administer rehabilitation and study their neurological recovery. During the follow-up period, both injured and uninjured pigs became larger and could travel longer distances with each stride on the treadmill, subsequently reducing the number of steps for locomotion at a given velocity. This was achieved by increasing stride and stance durations, and these two parameters were similarly adjusted when the pigs walked at a low treadmill speed in the absence of body weight changes (i.e., when the gait of 2-month-old uninjured pigs was assessed the same day at different velocities). In fact, step cycle timing is the first parameter adjusted by legged animals in either treadmill or overground locomotion to cope with contextual needs in health and disease. Rats, 5,11,64 primates, 67 and humans 68,69 with SCI similarly increase stride and stance durations, thus spending more time in double or triple support, which improves gait stability at the cost of reducing forward velocity. However, balance progressively deteriorates as the subjects attempt to increase velocity. When assessing animal locomotion on the treadmill, the speed is chosen by the experimenter and the experimental subject must adapt to the requirements. Consequently, motor disability becomes more evident, and balance worsens in absence of compensatory mechanisms at sufficiently high speeds. Our data show that motor impairments and compensations following cervical SCI in piglets can be readily distinguished and quantified by kinematic techniques and force measurements during treadmill locomotion. The primary motor deficits of the RF, namely the loss of elbow and wrist angular movement, as well as its reduced weight-bearing force with the consequent loss of balance, persisted chronically despite the increase in body size. These locomotor impairments were the same as those of adult rats and monkeys with unilateral cervical SCI and will likely be common to all quadrupeds in which body weight bearing is shared by the hindlimbs and the forelimbs.
SCI was performed in piglets at an age of 2 months, which is equivalent to ∼4 years in humans (early childhood) in terms of sexual maturity; and the last motor assessment was performed when they were 5 months old, which corresponds to ∼10 years in humans. 70 The pig spinal cord doubles in weight from the second to the fifth post-natal month and increases in both length and transverse area, 71 but the neurobiological events subjacent to that growth have not been addressed, and neural circuit development in the porcine central nervous system (CNS) is much less known than in that of humans and rodents. The adult number of brain neurons in the domestic pig is established at birth, 72 and its brain weight is ∼70% of an adult by 2 post-natal months. 71 Considering the similar perinatal brain growth spurt in pigs and humans, and the fact that axonal systems projecting from the brain, including the CST, 73 arrive to the lower cervical spinal cord segments before birth in humans, it is safe to assume that all brain-spinal cord axons have similarly reached the target regions in 2-month-old pigs. We observed a marked atrophy of the spinal WM together with a differential distribution of WD rostral and caudal to the injury site, supporting the notion that the ascending and descending axonal tracts, including the CST, were already developed at the time of injury. Consequently, after SCI in 2-month-old pigs, axotomy and axonal regenerative failure likely occurs as in adults and may explain the persistent motor deficits reported in the present study. Nevertheless, spinal cord growth from 2 post-natal months onwards enlarges the lesion site, and this must be considered when planning and assessing therapeutic interventions. In addition, axonal myelination of the CST occurs in the post-natal period in humans and pigs, 35,36 reaching a mature state only by 6 post-natal months in the latter. 36 Because of the incomplete spinal cord myelination by 2 post-natal months, the possibility exists that axonal growth and guidance is still facilitated to some extent at this age. Notwithstanding that the present study was not focused on axonal growth, we found no histological indication of spontaneous spinal cord repair in the piglets, suggesting that their spinal cord milieu greatly resembles that of adult mammals. However, this issue warrants further investigation.
Motoneuron loss contributes to chronic paresis of elbow extensors 74 and hand muscles 75 in humans with SCI at the cervical enlargement. The same phenomenon is observed in rodents with contusion injuries, in which partial damage to the cervical or lumbar motoneuron pools causes permanent, segmental motor impairments. 7 Although the exact position of spinal motor nuclei for elbow extensors and wrist flexors has not been reported for pigs, they are mostly located from C7 to T1 in primates, rodents, and other mammals. 11,57,60,76 Consequently, the main motor deficits observed after cervical hemisection in pigs in the present study; namely, impaired elbow extension and wrist flexion, together with loss of weight-bearing force and balance, are mostly caused by the interruption of axons descending from the brain and upper cervical region, leading to denervation of the segments C7-T1. Nevertheless, similarly to studies in adult rats with spinal cord hemisection, 11 some neuronal death occurred adjacent to the lesion site in pigs, involving approximately half the length of a spinal cord segment and apparently affecting all GM laminae. This neuronal loss added to the neurons excised surgically with the 1 cm of spinal cord tissue, and likely caused some motor impairment in proximal forelimb joints. Moreover, reduced neuronal numbers adjacent to the neural tissue gap will worsen the functional impact of axonal regrowth by complicating the re-establishment of synaptic connectivity across the lesion. The distance of the cell body to the site of axotomy influences neuronal death and regeneration in rodents. 77 If the same happens in the piglet spinal cord, propriospinal neurons projecting from rostral to caudal spinal cord segments, and neurons from laminae I–IV projecting from caudal segments to supraspinal centers, might be more susceptible to dying after axotomy. However, motoneuron loss was also observed close to the lesion, and this cell type was not axotomized by SCI. Albeit that more rigorous identification and quantification of the affected neuronal types is necessary, our preliminary data point to the action of unspecific noxious events in the perilesional tissue, such as hemorrhage and hypoxia secondary to blood vessel damage. These events, together with factors conferring neuronal susceptibility, likely produced cell death close to the injury site.
Adult minipigs weight only between 20 and 85 kg depending on the species, 78 and therefore are gaining ground against domestic pigs as experimental models in SCI 37,39,42 –47,78 –80 and other biomedical research fields. 78 There is no doubt that adult pigs of manageable weight will be preferable to domestic piglets for SCI research unless the objectives of the study specifically relate to pediatric injuries. However, when considering their similarity to humans regarding CNS anatomy, cell biology, or pharmacology, there are no unequivocal data supporting the appropriateness of the available minipig strains as replacements for domestic pigs. Neurogenesis is pronounced in the cerebral cortex of the Göttingen minipig after birth, whereas it is essentially completed pre-natally in both humans and domestic pigs, 72 and hence the latter may be more suitable for the investigation of perinatal CNS pathologies. Moreover, fibroblasts from minipigs have a shorter lifespan, a slower proliferation rate, and more frequent chromosomal abnormalities than those of domestic pigs, 81 which is a relevant issue because proliferation of connective tissue cells is a major determinant of axonal regenerative failure after SCI. 82 The domestic pig seems superior to the Yucatan minipig for investigating transcutaneous drug delivery. 83 Therefore, different porcine strains may be suitable for studying specific aspects of CNS damage, axonal regeneration, and pharmacology. The anatomy of the spinal cord of minipig strains and domestic pigs has not been systematically compared so far. Also, most reports dealing with the porcine spinal cord have focused on the thoracic and lumbar regions, and information about the cervical segments is scarce. The lumbosacral spinal cord enlargements of domestic pigs and humans are largest and more similar in size (length and cross-sectional area) than those of rhesus monkeys and cats. 84 Moreover, the ratio of gray/white matter is quite similar in domestic pigs and humans, representing ∼0.35 at the largest lumbar cord cross-sectional area, whereas it is ∼0.38 and ∼0.4 in monkeys and cats, respectively. For the cervical spinal cord, our results seem to compare similarly well with those of humans and differ from monkeys, cats, and rats. In particular, the ratio of gray/white matter for the segment C6 was ∼0.22 in our domestic pigs, ∼0.22 for humans, 85 ∼ 0.25 for rhesus monkeys, 86,87 ∼ 0.37 for cats, 88 and ∼0.48 for rats. 11 Therefore, despite the disadvantages of a continuous increase in body size and weight, our data support the use of domestic piglets for studying the neuropathological and functional consequences of cervical SCI, as well as for assessing therapeutic interventions before proceeding to clinical trials.
In addition to their clinical relevance, animal models of cervical SCI offer the possibility of achieving precise correlations between anatomy and function at the neural circuit level. Motoneuron pools innervating forelimb muscles are topographically organized along the cervical region, 57,60 therefore allowing for the production of distinct functional deficits by selective axonal tract or neuronal damage at concrete spinal cord segments. This may be very valuable for assessing the effectiveness of therapies aimed at neuronal protection and/or replacement, as well as at axonal growth promotion, because the regenerated axons will have the possibility of reconnecting with the correct neuronal targets in the segments just caudal or rostral to the lesion. Most research in traumatic cervical SCI has been conducted in rodents, 5 –14,27 cats, 15,16,18 and primates, 17,19 –26 whereas work on porcine models is uncommon. 50 –52 Piglets were used for addressing the induction of c-fos gene expression after C1 spinal cord transection, 50 as well as for analyzing hemodynamic changes after C3-C4 complete SCI produced by severe contusion. 51 Those studies were limited to a few hours after the lesion because complete cervical injuries cause tetraplegia and loss of autonomic control, together with diaphragmatic paralysis in high cervical lesions, and hence preclude the long-term follow-up of the animals outside a specialized intensive care unit. Incomplete cervical SCI models lack this limitation as far as the animals are not severely disabled. Cervical spinal cord hemisection is frequently used experimentally 5,6,8,10,11,18,19,25,26 because unilateral neural damage enables the investigation of function loss and recovery mediated by the injured side, together with the compensatory mechanisms deployed by the uninjured tissue. To the best of our knowledge, only one study 52 investigated traumatic incomplete injuries at the cervical region in swine, albeit the lesions were superimposed on a brachial plexus injury. The authors avulsed the dorsal and ventral rootlets of the C5, C6, and C7 spinal levels, causing a complete paralysis of the ipsilateral foreleg. A ventrolateral myelotomy was performed in some of those pigs, but the foreleg paralysis already produced by root avulsion hid any additional impairment contributed by the SCI, except for some weakness that was observed in the hindlimbs.
We excised 1 cm of spinal cord to create a neural tissue gap somewhat comparable in longitudinal extent to the cavities observed in human cervical SCI. 89 In the pursuit of successful translation of neurotherapeutics from animal models to human patients, a given treatment must be effective to promote axonal regeneration and functional synaptic reconnection across a significant neural tissue gap in a large animal or it will likely fail in the clinical setting. Unilateral cervical spinal cord cavitation might be produced in pigs by hemi-contusion as performed in rats 9,12,14,90 and non-human primates. 91 Considering the high incidence of contusion SCI in humans, 53,54 developing a porcine hemi-contusion SCI model is relevant for translational medicine. However, hemi-contusions are more variable in terms of neuroanatomical damage and may complicate the assessment of axonal regeneration as a result of the sparing of white matter tracts on the injured side, whereas spinal cord hemisection with excision of tissue assures axotomy of the targeted systems and reproducibility in the longitudinal extent of the lesion. This will facilitate a systematic assessment of neural repair strategies while simultaneously informing the optimization of clinical trials. Further improvements of the cervical hemisection model might be achieved by performing subpial dissection of the anterior spinal artery and its major branches to prevent vascular injury and subsequent extension of the lesion to the contralateral side. Figure 11 summarizes the proposed SCI model and some current therapeutic approaches. Axonal growth facilitators and neuroprotective drugs will surely have a role in any neurorestorative scheme. Nevertheless, the concomitant implantation of biofunctionalized scaffolds providing cell guidance cues may become necessary to achieve neural regeneration through the tissue gap. 92
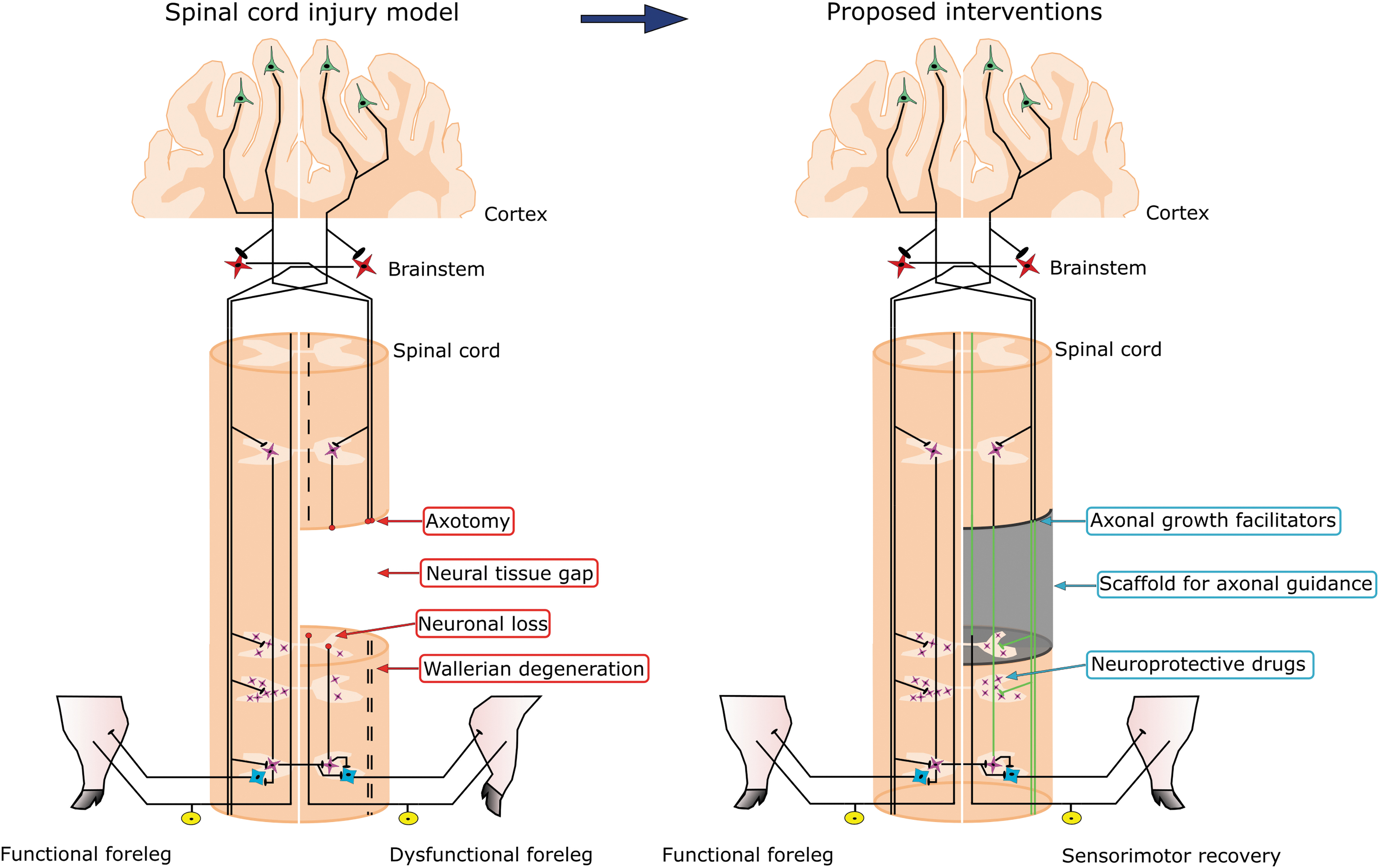
Diagram summarizing the neuroanatomical and motor impairments ensuing cervical spinal cord hemisection in pigs, and the therapeutic interventions proposed to achieve functional recovery. Pyramidal neurons are represented in green, brainstem neurons are represented are represented in red, propriospinal neurons are represented in pink, motoneurons are represented in blue and dorsal root ganglion neurons are represented in yellow. Wallerian degeneration appears as discontinuous lines and hypothetical axonal regeneration as green lines. Color image is available online.
Footnotes
Acknowledgments
We thank the Animal Care and Veterinary Service of the Hospital Nacional de Parapléjicos (HNP) for providing animal anesthesia and post-operative care; Alejandro Badajoz Barbero, Paloma Ania Benito, and Inés Pascual García-Núñez, for helping with animal motor training, histology, and video recording processing for kinematics; and the Servicio de Microscopía y Análisis de Imagen of the HNP for confocal microscopy.
Funding Information
This work was funded by the European Union's Horizon 2020 research and innovation programme (Neurofibres project, grant agreement 732344), the Spanish Ministry of Economy and Competitiveness and Fondo Europeo de Desarrollo Regional (FEDER) (Grant SAF2015-65236R), the Fundación Mutua Madrileña, and the Sociedad Española de Cirugía Ortopédica y Traumatología (SECOT).
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Text
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Figure S8
Supplementary Figure S9
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
