Abstract
Autoimmune profiling in rats revealed the antioxidant enzyme, peroxiredoxin 6 (PRDX6), as a target for autoantibodies evoked in response to traumatic brain injury (TBI). Consistent with this proposal, immunohistochemical analysis of rat cerebral cortex demonstrated that PRDX6 is highly expressed in the perivascular space, presumably contained within astrocytic foot processes. Accordingly, an immunosorbent electrochemiluminescence assay was developed for investigating PRDX6 in human samples. PRDX6 was found to be measurable in human blood and highly expressed in human cerebral cortex and platelets. Circulating levels of PRDX6 were elevated fourfold over control values 4 to 24 h following mild-to-moderate TBI. These findings suggest that PRDX6 may serve as a biomarker for TBI and that autoimmune profiling is a viable strategy for the discovery of novel TBI biomarkers.
Introduction
T
We utilized a discovery-based strategy that first involved screening for the expression of autoantibodies in rats in response to controlled cortical injury (CCI). 2 Immunoglobulins from injured rats were used to interrogate the entire rat brain proteome fractionated on large scale two-dimensional (2D) gel electropherograms. Of the approximately 3000 proteins visible in the array, only a small fraction of the proteins exhibited autoreactivity with immunoglobulin G (IgG) from the injured rats than exceeded background observed in controls. We mapped these immunoreactive features to replicate protein gels and identified them by mass spectrometry. 14 One of the most immunoreactive features was identified by mass spectrometry as peroxiredoxin 6 (PRDX6). PRDX6 is an important anti-oxidant enzyme that hydrolyzes the hydrogen peroxide (H2O2) generated during injury or disease. 15 –21 In brain, PRDX6 is primarily expressed in astrocytes, 22 although low levels of expression are also reported in neurons. 23 PRDX6 also is found in the periphery, including in the lung, kidney, skin, and neutrophils. The protein also is found in several forms of cancer. 24 –26 While several proteomics studies have identified PRDX6 as a central nervous system protein and have shown its release into cerebral spinal fluid following spinal cord injury, 27 –30 these studies did not explore the possibility of its role as a blood-borne candidate biomarker for TBI.
The present investigation demonstrates for the first time that PRDX6 is a normal constituent of human plasma where its levels are acutely elevated following mild-to-moderate TBI. While the specificity of this response to TBI has yet to be determined, these findings indicate that PRDX6 may contribute to a biomarker signature for acute mild brain injury.
Methods
Reagents
All reagents were obtained from Sigma-Aldrich (St Louis, MO) unless otherwise indicated.
Rodents, CCI treatment, and serum brain samples used in autoimmune profiling
Autoimmune profiling was carried out using serum obtained through Institutional Animal Care and Use Committee-approved tissue sharing protocols from control and CCI-treated male Sprague Dawley rats (250-300 g; Taconic Farms, Inc., Rockville, MD). The CCI procedure was carried out under 4% isoflurane anesthesia with a stereotaxic impactor (Leica, Buffalo Grove, IL) using a 5 mm impactor at a 20 degree angle to a depth of 2 mm at 5 m/sec with a dwell time of 500 msec as described previously. 2 Histologic evaluation confirmed that the CCI procedure consistently resulted in lesions that injured the cerebral cortex and the dorsal portion of the underlying corpus callosum. A severe CCI injury was used with the intention of maximizing an autoimmune response. Animals were euthanized and blood was collected 7 d post-CCI. Serum was prepared by centrifugation, mixed with an equal volume of glycerol to stabilize immunoglobulins, and stored at−20°C until used. Control animals received no treatments. Whole rat brain proteome was prepared from snap frozen brains (normal Sprague Dawley rats (250-300 g; Taconic Farms, Inc.). The tissue was thawed and homogenized by Polytron (three times for 10 sec, setting 6-7) in 10 volumes to weight 0.25 M sucrose, 20 mM TES, 1 mM EDTA, 0.6 M KCL (pH 7.0) containing 1×complete protease inhibitor cocktail. The homogenate was subjected to Potter-Elvehjem (glass/Teflon) homogenization (five down/up strokes) and centrifuged at 4900×G (4°C, 15 m) to remove cellular debris. The resulting supernatant was centrifuged at 150,000×G (4°C, 60 m). The supernatant was assayed for total protein (bicinchoninic acid assay [BCA]) and stored as aliquots at−80°C.
Human samples and sample preparation
Control plasma and serum
Control human plasma (sodium ethylenediaminetetraacedic acid (NaEDTA) anticoagulant) and matched sets of plasma (sodium ethylenediaminetetraacedic acid (NaEDTA) anticoagulant) and serum were prepared from single blood draws collected from healthy, normal male and female volunteers (78% Caucasian, 17% African-American, 5% Hispanic) ranging in age from 19 to 50 years old, and with a mean and median age of 25 years and 24 years, respectively (obtained commercially; Innovative Research, Novi, MI). Control samples for investigating differences between plasma and serum consisted of two separate matched sets of plasma and serum, collected from ten males and ten females each for a total of 40 samples. Control samples for the clinical analysis (Table 1) consisted of a third set of control plasma samples (n=30; 15 males and 15 females).
TBI; traumatic brain injury; SD, standard deviation; ED, emergency department; CT, computed tomography; MRI, magnetic resonance imaging.
TBI plasma
Post-TBI plasma samples were collected (NaEDTA anticoagulant) from patients at the time that neuroimaging was being performed (4-24 h and 2-7 d post-TBI). Patient demographics and clinical variables are presented in Table 1.
Cerebral cortex
Human cerebral cortex (male; 52 years old; African American; cause of death was atherosclerotic cardiovascular disease; postmortem interval was 2 h; neuropathologic evaluation revealed brain to be normal) was obtained from the Brain and Tissue Bank for Developmental Disorders of the National Institute of Child Health and Human Development, University of Maryland (Baltimore, MD). A homogenate of soluble protein was prepared from a wedge of tissue (∼5 g), spanning all layers of the frontal cortex, following the procedures described above for rat brain.
Platelets
Human platelets were prepared from Caucasian male and female normal volunteers (age 20 - 30 years old) as follows. Platelet rich plasma (60 mL average volume, prepared from 450 mL whole blood, Innovative Research) was combined with an equal volume of Tyrode's buffer (137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2, 1.8 mM CaCl2, 0.2 mM Na2HPO4, 12 mM NaHCO3, and 5.5 mM D-glucose) containing prostacyclin (100 ng/mL) and centrifuged at 1800×G for 15 min at 15°C. The pelleted platelets were gently resuspended in 22 mL of the Tyrode's buffer containing 1×complete protease inhibitor cocktail (Roche Applied Science, Indianapolis, IN) and assessed microscopically for purity (>99% routinely) and appearance (spherical with little sign of activation). Platelet proteins were prepared by three cycles of freeze/thawing and sonication. The extracts were then centrifuged at 14,000×G for 10 min at 4°C, and supernatants collected and assayed for protein content (BCA; Thermo Fisher Scientific Inc., Rockford, IL). Platelet extracts were diluted in phosphate buffer saline (PBS; pH 7.2) containing 1% bovine serum albumin (BSA) for evaluation of PRDX6 by immunoassay and immunoblot.
2D gel electrophoresis
Two-dimensional gel electrophoresis was performed according to the method of O'Farrell 31 with modification. 32 –34 A single run generally consisted of eight gels, two of which were silver stained to visualize proteins, while six were transferred to polyvinylidene difluoride (PVDF; Hybond-P PVDF Membrane, GE Healthcare, Buckinghamshire, UK) for immunoprofiling. These blots were probed with serum from either control rats or animals treated with CCI. Brain homogenates were de-lipidated by mixing one volume protein (2.4 mg/mL) sample with five volumes 2:1 chloroform/methanol followed by centrifugation. Protein samples (routinely 5 mg to 10 mg) were physically recovered as a pad positioned at the aqueous/organic interface; protein recovery was 95–99% through the extraction procedure (BCA assay; n=5). The extracted proteins were dissolved in isoelectric focusing rehydration buffer (1% dithiothreitol, 2% pH 3-10 pharmalytes (GE Healthcare Life Sciences, Pittsburgh, PA), 2 M thiourea, 6 M urea, and 1% CHAPS and a trace of bromophenol blue) and applied to pH 3-10, 24 cm isoelectric focusing Immobiline Dry Strips® (GE Healthcare Bio-Sciences, Uppsala, Sweden) (1 mg in 450 μL/strip). The strips were overlaid with mineral oil and proteins were focused for 22 h at 16°C using: 30 volts for 12 h, 500 volts for 1 h, 1000 volts for 1 h, and 8000 volts for 8 h.
Upon completion of the first dimension, proteins were reduced and alkylated by sequentially equilibrating the immobilized pH gradient strips in buffers containing 1% dithiothreitol and 2.5% iodoacetamide (both prepared in 6 M urea, 75 mM Tris-HCL, 29.3% gylcerol, 2% SDS, 0.1% Bromophenol Blue; 15 m each). Second dimension fractionations were performed on 255×196×1 mm gels (Dalt 12.5%; GE Healthcare Bio-Sciences) over 4 h. The gels were then either stained with silver stain 35 or transferred to PVDF membrane using a Pharmacia Multiphor II electrophoresis unit (GE Healthcare) operated at 600 volts/cm2, 400 mA limiting, for 60 m and a 3-(N-morpholino)propanesulfonic acid (MOPS) buffer system (40 mM MOPS, 10 mM NaAc, 1 mM EDTA) containing 10% methanol.
Immunoprofiling
Following transfer, immunoblots were blocked with 5% fetal bovine serum (FBS; Gibco, Grand Island, NY) in Tris-buffered saline containing Tween 20 (TBS-T; 50 mM Tris, 150 mM NaCl, 0.05% Tween 20, pH 7.6) for 60 min at room temperature and then probed overnight at 4°C with serum pooled from six to eight control or CCI rats diluted 1:250 in TBS-T/5% FBS. Blots were then washed three times with TBS-T, probed for 3 h at room temperature with horseradish peroxidase labeled anti-rat IgG secondary antibody (HAF005, R&D, Minneapolis, MN) diluted 1:5000 in TBS-T, washed three times with TBS-T and visualized by enhanced chemiluminescence (ECL+Plus; Amersham Searle Corp, Arlington Heights, IL) using a FUJI LAS 3000 imager (Fujifilm Medical Systems, Stamford, CT) and Multi Gauge v3.0 software (Fujifilm). Immunoreactive signals that were unique to CCI blots were mapped to the corresponding protein features on silver-stained gels. Proteins of interest were excised and processed for proteomic analysis. A total of six rounds of immunoprofiling were performed using separate pools of control and TBI serum.
Protein identification by peptide mass fingerprinting
The silver stained gel fragments were de-stained with 100 μL of a 1:1 mixture of 30 mM potassium ferricyanide and 100 mM sodium thiosulfate and rinsed with water until clear. The gel fragments were then equilibrated with 0.2 mL of 100 mM NH4HCO3/50% acetonitrile for 45 min at 37°C, dehydrated in 100 μL 100% acetonitrile, and dried under vacuum. The dried gel pieces were rehydrated with 40 mM NH4HCO3 /10% acetonitrile containing 20 ng/μL trypsin (Trypsin Gold, Mass Spectrometry Grade; Promega, Madison, WI) and incubated overnight at 37°C. Peptide fragments were recovered in sequential (60 m, room temperature) extractions with 1.0% trifluoroacetic acid (TFA; 75 μL) followed by two rinses with 50% acetonitrile/5% TFA (50 μL each). The three collections were pooled, dried under vacuum, and dissolved in 10 μL of 1% TFA. The peptides were then purified and concentrated using a C-18 Zip Tip® (Millipore Corporation, Billerica, MA) and mixed with alpha-cyanohydroxycinnamic acid matrix (10 mg/mL in 50% acetonitrile/0.1% TFA) containing bradykinin (1060.5692 daltons; 50 fmol/mL) and adrenocorticotropic hormone fragment 18-39 (2465.1989 daltons; 150 fmol/mL; AnaSpec, San Jose, CA) as internal standards. Samples were analyzed by matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) mass spectrometry using a Voyager MALDI-TOF DE STR instrument (PE Biosystems, Framingham, MA). The mass spectrometer was operated in reflectron mode with an accelerating voltage of 20,000 V, a grid voltage of 76.13% and a guidewire voltage of 0.003%. Peptide mass data were used to query the National Center for Biotechnology Information protein sequence database accessed through the ProteinProspector MS-Fit search engine (
Immunosorbent electrochemiluminescence assay
The single-plex immunosorbent electrochemiluminescence assay (IEA) was developed using 96-well, standard-bind electrochemiluminescence microtiter plates from Meso Scale Diagnostics (Rockville, MD). Combinations of seven monoclonal and five polyclonal anti-PRDX6 antibodies were screened for compatible pairs. A mouse anti-PRDX6 monoclonal antibody (Clone 1A11, GenWay Biotech, catalog #20-007-280008, San Diego, California) and a rabbit monoclonal anti-PRDX6 antibody (Clone EPR3755; Epitomics, catalog # 2769-1, Burlingame, California) were selected for optimization as capture and primary antibodies, respectively. The monoclonal capture antibody was first purified by Protein G affinity chromatography (Protein G HP SpinTrap; GE Healthcare BioSciences; vendors protocol) to remove glycerol, and then coated to the plates at a concentration of 4 μg/mL in PBS (25 μL/well, overnight, 4°C). The plates were blocked with 3% BSA in PBS for 2 h at room temperature. The wells were then washed three times with PBS containing 0.05% Tween-20 (PBS-T), and the samples (diluted in PBS-1% BSA) were added to wells in a total volume of 100 μL/well containing 25 μL plasma or serum and dilutions of either platelet and brain extracts. The standard curves were established using serial dilutions of PRDX6 recombinant human protein (catalog #GWB-F65D50; GenWay Biotech) in a final volume of 100 μL/well. Standard curves were prepared in PBS-1% BSA containing either 25% chicken plasma or 25% horse serum (both from Innovative Research) to control for the non-specific effects of serum or plasma matrix. These matrices were selected from a variety of species samples (Innovative Research) based upon their ability to: 1) mimic the backgrounds calculated for corresponding human samples, and 2) not interfere with the performance of recombinant PRDX6 protein standard in the IEA.
Plates were incubated for 2 h at room temperature and washed three times in PBS-T. Following the addition of primary antibody (2 μg/mL in PBS-1% BSA, 25 μL/well) plates were incubated for 1 h at room temperature and washed three times in PBS-T. The plates were then treated with goat anti-rabbit MSD Sulfo-Tag antibody (MSD, R32AB-1; 2 μg/mL in PBS-1% BSA, 25 uL/well, 60 min, room temperature). After three final PBS-T washes, the plates were developed with 150 uL/well of MSD Read Buffer T with Surfactant (MSD) and electrochemiluminescence read using a SECTOR Imager 6000 (MSD). The assay can detect 0.7 ng/mL (lower limit of detection) and is quantitative to 1 ng/mL (lower limit of quantitation) as defined by three and 10 times the standard deviation of readings obtained for 0 pg/mL standard, respectively (n=10). Intra- and inter-assay variability were 4% and 13%, respectively (n=10).
One-dimension immunoblotting
One-dimension (1D) immunoblot analyses were performed by standard methods as previously described. 36 Proteins resolved by SDS-PAGE were transferred to nitrocellulose membranes (iBlot® Gel Transfer Stacks; Invitrogen, Grand Island, NY). Membranes were blocked with 50 mM TBS-T containing 5% FBS (blocking buffer). Blots were probed overnight (4°C) with rabbit monoclonal anti-PRDX6 antibody (Clone EPR3755; Epitomics) diluted 1:1000 in blocking buffer. The blots were washed three times in TBS-T and incubated (two hr, room temperature) with horseradish peroxidase labeled goat anti-rabbit IgG antibody (#31210, 1:5000 in TBS-T; Thermo, Rockford, Il). Immunoreactive proteins were visualized with an enhanced chemiluminescence reagent system (ECL+Plus; Amersham Searle Corp, Arlington Heights, IL) using a FUJI LAS 3000 imager. Digital imaging quantification was performed with Multi Gauge v3.0 software (FujiFilm Medical Systems USA, Inc., Stamford, CT).
Immunohistochemistry
Anti-PRDX6 immunohistochemistry was performed as previously described 2 on sections of rat cerebral cortex (paraformaldehyde-fixed) using rabbit anti-PRDX6 antiserum (GWB-007PE6; GenWay Biotech; 1:250 dilution), or mouse monoclonal anti-glial fibrillary acidic protein (GFAP) (MAB360; Millipore; 1:1000) for co-localization to astrocytes. Alexa Fluor® 488 donkey anti-rabbit (green) or Alexa Fluor 488 donkey anti-mouse (red) detection antibodies (Invitrogen; 1:100 dilutions) were used to visualize PRDX6 and GFAP, respectively. Additional analyses of cellular co-localization were carried out using antibodies directed against Iba-1 (ab15690; Abcam, Cambridge, Massachusetts, 1:1000) and ED-1 (MCA341R; AbD Serotec, Raleigh, North Carolina; 1:150) for quiescent and activated microglia, respectively; and NeuN (MAB377; Millipore; 1:1000) for neurons. These data (not shown) confirmed that PRDX6 is primarily associated with astrocytes in rodent brain.
Statistical analysis
Data were analyzed by one-way analysis of variance (ANOVA) followed by the Tukey-Kramer multiple comparison test. Differences were considered significant at a p value of less than 0.05. Univariate ANOVAs were conducted to examine effects of mTBI on PRDX6 levels in injured patients, compared with controls at each time, between males and females, and among imaging groups.
Results
Identification of PRDX6 as a target of a TBI-induced autoimmune response
Figure 1 shows that PRDX6 is highly expressed in brain astrocytes, where it is particularly enriched in the perivascular space, presumably contained within astrocytic foot processes. Parallel analyses for co-localization of PRDX6 in microglia and neurons (not shown) confirmed that PRDX6 is primarily expressed by brain astrocytes. Figure 2 depicts the discovery of PRDX6 as a protein reactive with autoantibodies induced by brain injury in rats. Naive rat brain proteome was fractionated on large scale 2D gels, transferred to PVDF, and then probed with immunoglobulins present in serum from control (Fig. 2A) or TBI (Fig. 2B) rats. The control western blot (Fig. 2A) shows a characteristic pattern of IgG binding that reflects non-specific binding due to the relatively high concentration of serum (1:250 dilution) used to maximize the intensity of specific autoimmune signals. This investigation focused on immunologic signals that were either newly revealed, or greatly enhanced, on CCI blots, compared with control blots. One such prominent feature present on the CCI versus control blots (circled) was mapped to a replicate silver stained protein gel (Fig. 2C) and identified by peptide mass fingerprinting (Fig. 2D). The protein was identified with 77% sequence coverage as PRDX6, a 224-amino acid protein with a theoretical pI of 5.64 and molecular weight of 24.8 kDa. The identification of PRDX6 was confirmed in separate experiments and additional samples analyzed independently by the W.M. Keck Biotechnology Resource Laboratory, Yale University. Based upon the identification of PRDX6 as a candidate TBI-autoantigen, a sandwich IEA was established for measuring PRDX6 in human samples. This assay was based upon a matched pair of capture and primary monoclonal antibodies, each recognizing a separate epitope on the PRDX6 molecule. Figure 2E shows a representative standard curve for the assay utilizing recombinant human PRDX6 protein as standard (see Methods for details).
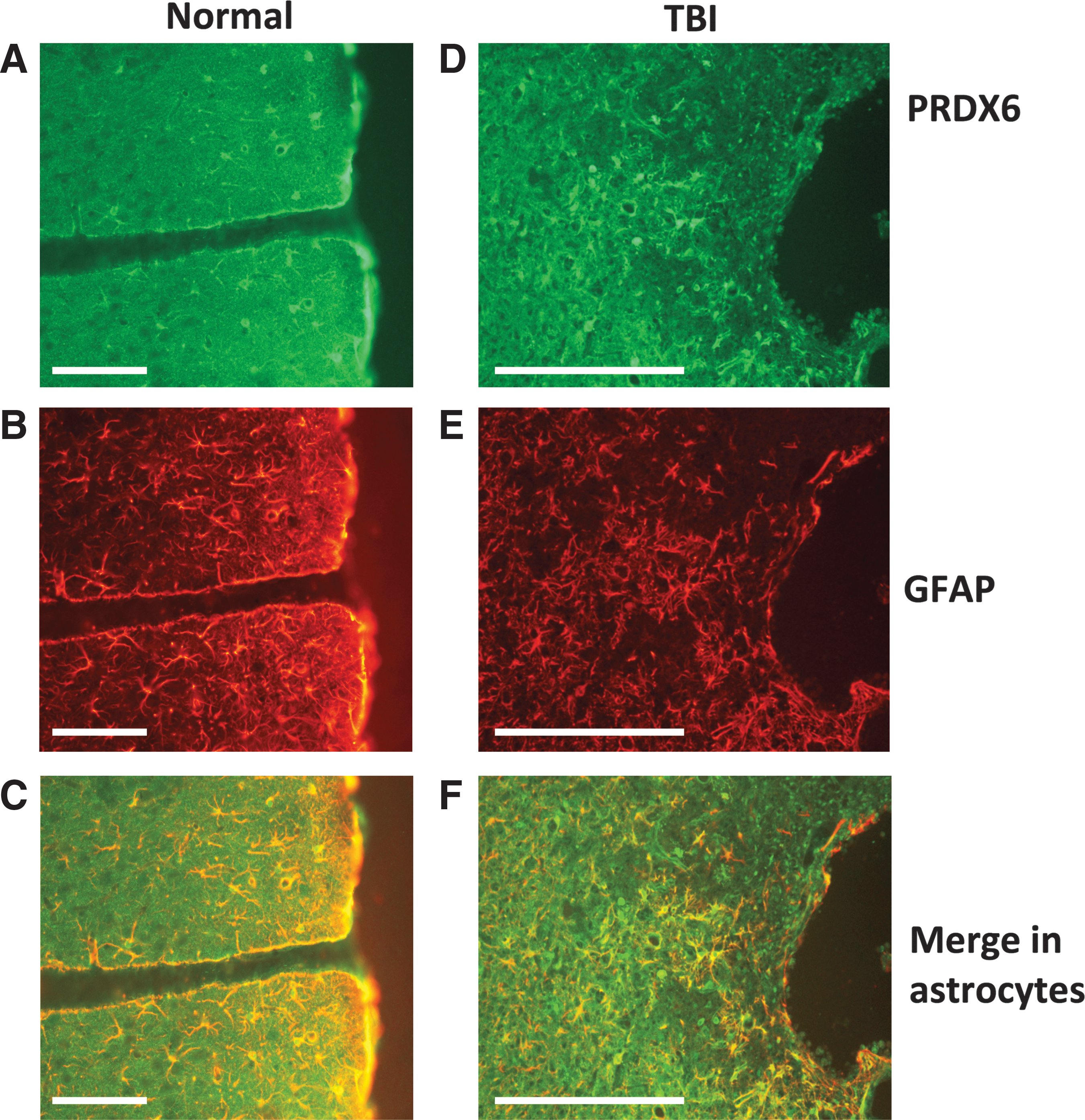
Peroxiredoxin 6 (PRDX6) is highly expressed in astrocytes in rat cerebral cortex and up-regulated following traumatic brain injury (TBI). Data show expression of PRDX6, glial fibrillary acidic protein (GFAP) and their co-localization in astrocytes in normal rat brain
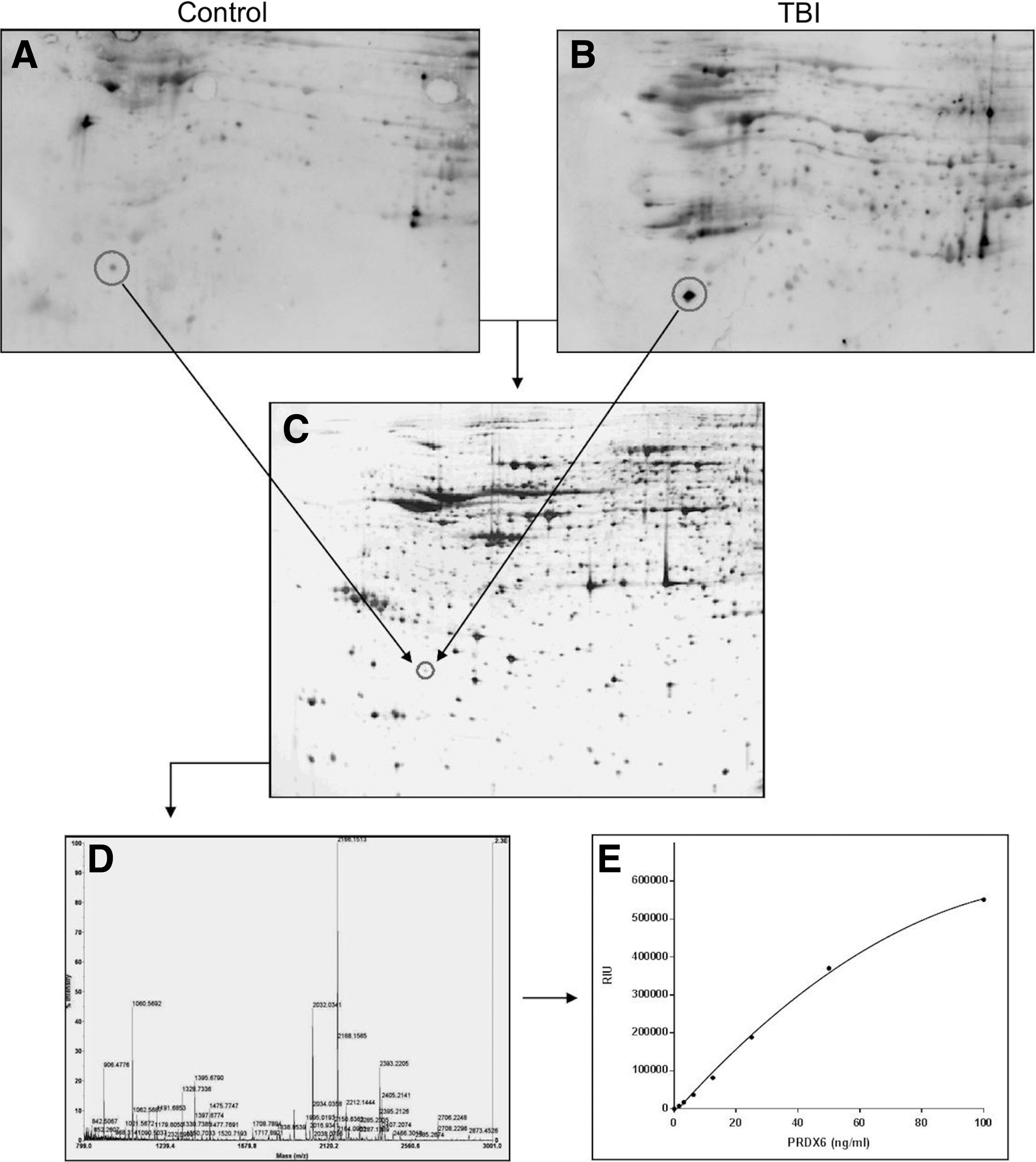
Discovery of peroxiredoxin 6 (PRDX6) as a candidate biomarker for brain injury. Rat brain proteome was fractionated by two-dimensional gel electrophoresis and transferred to polyvinylidene difluoride (PVDF). Blots were probed with serum from control and traumatic brain injury (TBI) rats (1:250), and visualized by enhanced chemiluminescence using pooled anti-rat immunoglobulin G (IgG) and IgM detection antibodies (
1D immunoblotting
The specificity of the capture and primary antibodies were evaluated by western blot using recombinant human PRDX6 protein and extracts of human cerebral cortex and platelets (Fig. 3). It is well established that human platelets function as circulating repositories for several candidate brain biomarkers. Accordingly, we investigated the presence of PRDX6 in platelets here. Figure 3A shows a replicate silver stained gel that depicts the complexity of the brain and platelet samples. While the capture antibody was not compatible with western blotting under the conditions used here, the primary antibody exhibited remarkable specificity for authentic PRDX6 present in highly complex samples of human brain and platelets (Fig. 3B). The recombinant PRDX6 standard exhibited a higher molecular weight, compared with tissue PRDX6, due to the presence of a histidine tag. Also, Figure 3A reveals the large amount of carrier protein present in the commercial standard; the recombinant PRDX6 was barely evident (arrow) at this protein loading. The concentration of PRDX6 in the single human brain sample was about double that of platelets, at 940 ng/mg soluble protein. Platelet samples making up the male and female pools were assayed individually for concentrations of PRDX6 and determined to be 500±29 ng/mg and 507±19 ng/mg soluble protein for males and females (n=8), respectively. The antibody was specific for human PRDX6 and did not cross-react with rodent PRDX6 present in samples of rat brain or blood (not shown).
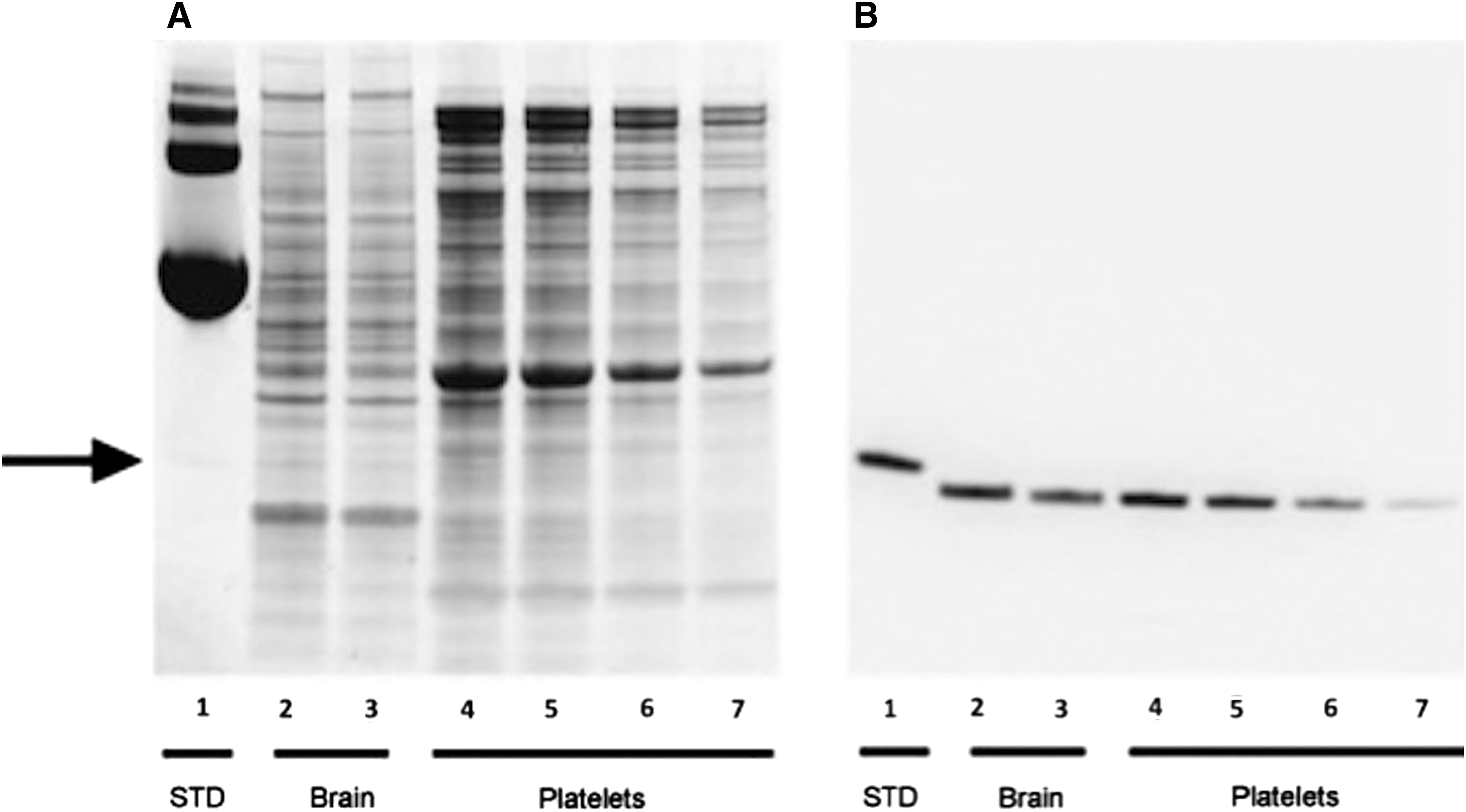
Western blot analysis of peroxiredoxin 6 (PRDX6) in extracts of human brain and platelets. Recombinant PRDX6 (200 ng, lane 1), human brain extract (20 μg, and 10 μg; lanes 2 and 3, respectively) and human platelet extract (20 μg, 10 μg, 5 μg, and 2.5 μg; lanes 4-7, respectively) were analyzed by silver staining
Immunoreactive PRDX6 in matched human plasma and serum
Figure 4 presents the mean values for PRDX6 in plasma and serum from normal male and female volunteers. The samples were prepared as matched sets from the same blood draws (n=10); NaEDTA was used as anticoagulant for the preparation of plasma. The data presented represent two separate experiments (Fig. 4A, 4B) carried out on two unique sets of matched plasma and serum samples prepared in the same fashion. Levels of PRDX6 were estimated using a standard curve that included an equivalent amount of either chicken plasma (NaEDTA anticoagulant) or horse serum to control for the non-specific effects of plasma or serum matrix, respectively. The presence of non-cross reactive (non-human) plasma or serum matrix increased nonspecific background signal by two- to threefold over buffer alone but did not interfere with the performance of recombinant PRDX6 protein standard in the IEA. Levels of PRDX6 in plasma were higher in females, compared with males (p<0.001) in both experiments (168±36 ng/mL vs. 65±10 ng/mL, and 178±30 ng/mL vs. 43±8 ng/mL, respectively). The process of coagulation dramatically reduced measured concentrations of PRDX6 in female blood samples (p<0.001). A tendency for this phenomenon also was observed in male samples; however, the effect here was not statistically significant due to the lower starting levels of PRDX6 present in male plasma.
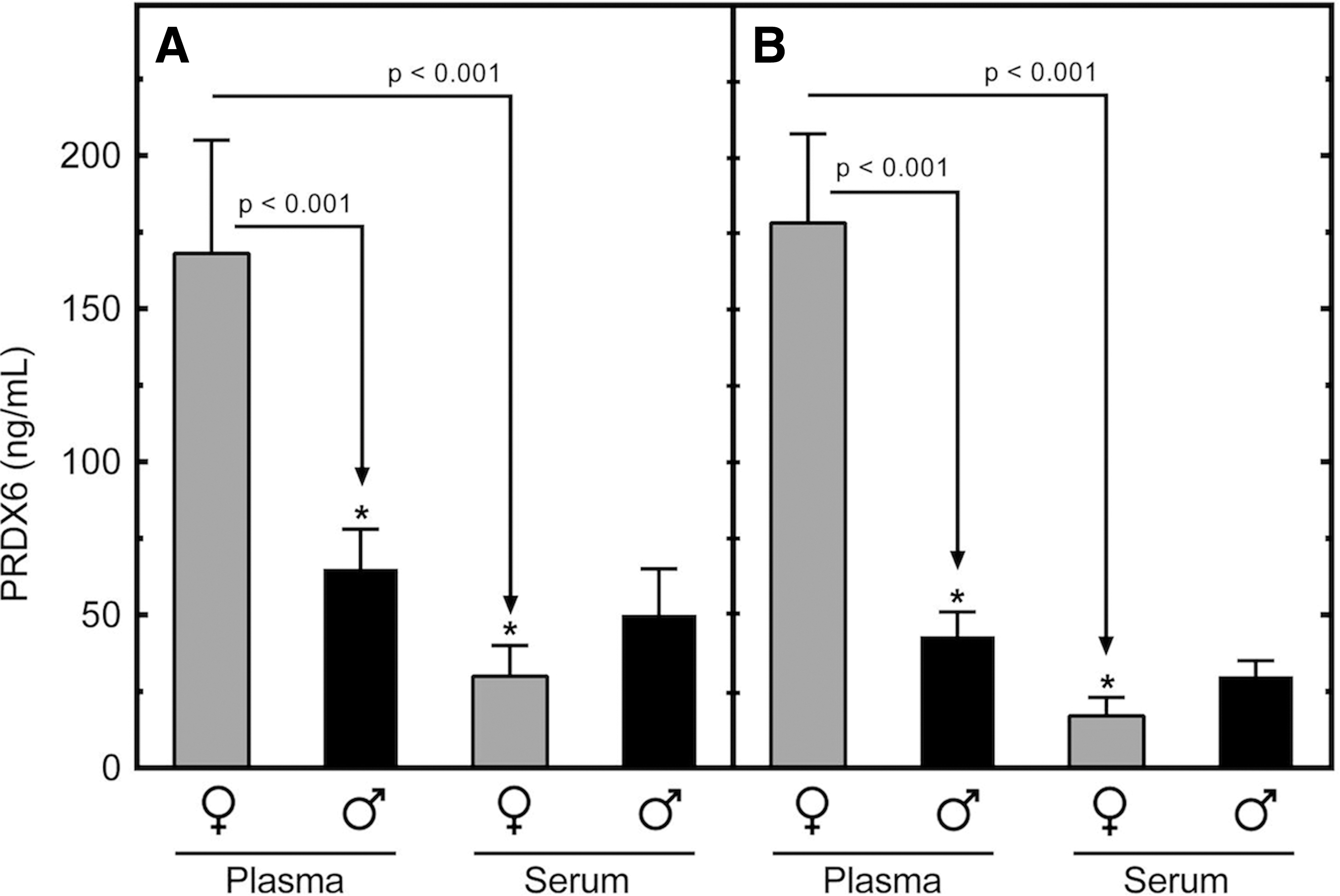
Comparison of levels of peroxiredoxin 6 (PRDX6) in human plasma and serum. Serum and plasma were prepared as matched sets from blood samples drawn from normal male and female volunteers (n=10 each). Levels of PRDX6 were measured by immunosorbent electrochemiluminescence assay (IEA). The experiment was replicated in a second, independent cohort of the same size
Effects of mild-to-moderate TBI on plasma levels of immunoreactive PRDX6
The effects of mild-to-moderate TBI on plasma levels of PRDX6 in humans is presented in Table 2. The patients participated in a clinical imaging study investigating computed tomography (CT) and magnetic resonance imaging (MRI) scanning for the diagnosis and assessment of TBI. Accordingly, plasma samples were collected at the times of imaging, which ranged from 4 to 24 h and 2 to 7 d post-injury. As summarized in Table 1, an analysis of the available CT and MRI data revealed that 51% (79 patients) of the TBI subjects presented with no discernable CT or MRI abnormalities, whereas 23% (36 patients) presented with CT and MRI abnormalities and 16% (24 patients) with MRI imaging abnormalities. All patients who were image positive on CT also were image positive on MRI. No imaging data was available on 10% (15 patients).While nearly half of the TBI subjects lost consciousness as a result of their accidents (45%), 51% presented with no discernible imaging abnormality of the brain. Scalp hematomas were observed by CT in 22% of the TBI patients. The median Glasgow Coma Scale (GCS) score at the time of hospital admission for all subjects was 15, consistent with mild TBI. The mean GCS scores for the imaging-positive group was not different from that of the CT/MRI-negative group (both GSC 15).
Significantly different from control values, p<0.01.
Data are expressed in ng / mL.
n=control male/female,15/15; TBI, male/female, 103/43
TBI, traumatic brain injury; PRDX6, peroxiredoxin 6.
While basal levels of PRDX6 were higher in females, compared with males (46±24 ng/mL vs. 141±24 ng/mL; p>0.01; Table 2), elevated levels post-TBI did not differ by gender (351±17 ng/mL [males] vs. 312±25 ng/mL [females]; Table 2). Accordingly, at the time of admission, fold increases in plasma levels of PRDX6 over control values differed between the genders with males increasing approximately sevenfold and females approximately twofold. There was no difference observed in plasma PRDX6 values measured in patients who were image positive for TBI versus those who were not. Considering all subjects combined, the mean circulating level of PRDX6 was increased nearly fourfold in response to TBI at time of admission (93±18 ng/mL vs. 322±15 ng/mL; p>0.01) and the elevation persisted for up to seven days post-TBI (372±15 ng/mL; p>0.01; Table 2).
Discussion
Autoimmune profiling was used as a strategy to identify candidate biomarkers for the diagnosis and assessment of TBI. We reasoned that the induction of autoantibodies by TBI might reveal a protein antigen that could be informative for the condition of brain injury. This screen led us to the identification of PRDX6 as one such candidate. Immunohistochemistry of rat brain confirmed that PRDX6 is highly expressed in astrocytes and is particularly enriched in the perivascular space, presumably contained within astrocytic foot processes. Based on these findings, we established an immunosorbent electrochemiluminescence assay for measuring PRDX6 in human samples and observed that PRDX6 is abundant in human cerebral cortex and platelets. Moreover, circulating levels of PRDX6 were measurable under basal conditions, tended to be higher in females, compared with males, and were significantly elevated (approximately fourfold) following mild-to-moderate TBI. These findings demonstrate that autoimmune profiling can be used to identify novel biomarkers which may prove useful in the detection of mild TBI.
There is a recent precedent in support of autoimmune profiling as a strategy for identifying candidate TBI biomarkers. Marchi and colleagues reported in collegiate football players that repeated sub-concussive hits to the head resulted in the formation of autoantibodies against the long-recognized TBI biomarker, S100B. 37 Responses in serum titers of anti-S100B antibodies were proportional to the number and severity of hits experienced and correlated with changes in brain structure detected by diffusion tensor imaging. The authors speculate that the autoimmune response was due to repetitive disruption of the blood–brain barrier and release of S100B with subsequent immune activation through an unknown mechanism. More recently, Zhang and colleagues 38 reported in humans that TBI induces the expression of autoantibodies against GFAP and its breakdown products. Like PRDX6, S100B and GFAP are highly expressed in astrocytes and also are constituents of blood where levels increase in response to TBI. 37,39 Therefore, the autoimmune response to all three candidate biomarkers may occur by similar mechanisms, possibly involving posttranslational modification and a reversal of peripheral immune tolerance by the structural modification. The oxidative stress of TBI can drive the formation of a variety of protein adducts, which can influence antigenicity. These modifications include citrullination, 40 –42 nitrosylation, 43 and the formation of 4-hydroxy-2-nonenal (HNE) adducts. 44 Interestingly, protein citrullination has important mechanistic implications in rheumatoid arthritis, 41 where autoantibodies against citrullinated connective tissue proteins appear to underlie the pathogenesis of the disease. 42 The same mechanism may be shared by PRDX6, GFAP, and S100B in response to TBI.
It is well established that human platelets function as circulating repositories for several established brain biomarkers, most notably brain-derived neurotropic factor (BDNF) 45,46 and neuron specific enolase (NSE). 47 –49 Because of this, we investigated the presence of PRDX6 in platelets of normal male and female volunteers. This analysis showed that levels of PRDX6 in human platelets approach those measured in human cerebral cortex. Accordingly, plasma levels of PRDX6 may reflect, at least in part, platelet secretion. If so, plasma levels of PRDX6 may serve as a marker of platelet reactivity and disturbances of hemostasis, rather than a specific marker of brain injury. Arguing against this proposal, however, is the present finding that levels of PRDX6 in serum were lower than those measured in plasma prepared from the same blood draw (Fig. 3). This was particularly true in the case of females, where levels of serum PRDX6 were approximately 20% those of plasma, suggesting that PRDX6 is not released from platelets during the clotting process and that PRDX6 may actually be sequestered into the developing thrombus or degraded. This is in marked distinction to BDNF, which is released from platelets during coagulation, resulting in serum levels that are elevated over those measured in corresponding plasma. 46,50 These findings on the gender difference observed in plasma levels of PRDX6 raise the possibility that males have a higher clearance rate for blood-borne PRDX6, compared with females. The observation that sample preparation can be a variable in the analysis of PRDX6 is important to its use as a candidate biomarker. Finally, at present, the origin(s) of circulating PRDX6 and their precise relation to TBI remain to be clearly defined.
It should be noted that many, if not all, of the candidate TBI biomarkers currently under investigation are not strictly brain-specific. For example, GFAP is expressed in the gastrointestinal tract and the liver, 51 BDNF and NSE are concentrated in platelets, 45,47 and S100B is expressed in adipocytes and the gastrointestinal tract. 52,53 Similarly, ubiquitin carboxyl-terminal hydrolase-L1 (UCH-L1, PGP9.5), perhaps the most promising prototype TBI biomarker under development, is expressed in the endometrium and secretory cells of the pituitary gland. 54 –57 Accordingly, the concept of a brain-specific response must be qualified when considering TBI biomarker proteins and should include the potential contribution of poly-trauma to a biomarker response. Additionally, we also recognize that a biomarker need not be specific to the brain or specific to traumatic injury in order to be useful in the clinical management of TBI. For example, a biomarker that detects traumatic vascular injury may be indicative of the need for therapy directed at promoting endothelial integrity, even if it is not specific to the cerebral vasculature. The brain, after all, does not exist in isolation from the rest of the body.
This study has several limitations. First, the study has a modest sample size and will need to be validated in a larger independent cohort. Second, pre-injury blood samples were not available for analysis. Accordingly, baseline measures were derived from the analysis of standardized control samples. While this approach provides for an important comparison across studies, within-subject responses are unavailable for individualized diagnoses. And third, the study does not include trauma controls to help define the specificity of the PRDX6 response specifically to brain injury. Accordingly, it is emphasized that PRDX6 represents a “candidate” TBI biomarker and that proof of PRDX6 as a definitive TBI biomarker will require its evaluation across a spectrum of TBI and non-TBI trauma paradigms, as well as in a variety of neuropathologies, including both psychiatric diseases and dementias. Finally, a biomarker does not necessarily need to be specific to traumatic injury, or even specific to the brain, in order to be useful in clinical practice. In this regard, a biomarker that detects vascular injury may be indicative of the need for therapy directed at promoting endothelial integrity, even if it is not specific to the cerebral vasculature. A similar case can be made for oxidative stress and anti-oxidant therapy.
A sensitive and specific IEA was developed and characterized for use with human samples. The analysis of human plasma and serum demonstrated that PRDX6 is a normal constituent of blood and that levels were higher in the plasma of females, compared with males. The process of coagulation reduced the relatively higher levels of immunoreactive PRDX6 observed in female plasma to those observed in the plasma and serum of males. Western and IEA analyses confirmed high concentrations of PRDX6 present in human platelets and cerebral cortex, approximately 0.5 μg/mg soluble protein and 1 μg/mg soluble protein, respectively. These data are the first to utilize autoimmune profiling as a strategy for the discovery of candidate biomarkers for brain injury. We have recently observed in ongoing studies investigating the full spectrum of TBI injury, from mild to very severe, that the magnitude of the acute plasma PRDX6 response is proportional to the severity of the injury (unpublished). Additional work is required to establish these findings and to determine the sensitivity and specificity of PRDX6 as a biomarker for TBI.
Footnotes
Acknowledgments
The authors would like to acknowledge the expert technical assistance of Ms. Tinghua Chen and Mr. James Freedy. Support for this work included funding from: Department of Defense in the Center for Neuroscience and Regenerative Medicine (G1703D), Defense Medical Research and Development Command (D61_I_10_J6_152) and TriService Nursing Research Program (N12-P12).
Author Disclosure Statement
No competing financial interests exist.
