Abstract
Currently, few interventions have been shown to successfully limit the progression of secondary damage events associated with the acute phase of spinal cord injury (SCI). Docosahexaenoic acid (DHA, C22:6 n-3) is neuroprotective when administered following SCI, but its potential as a pretreatment modality has not been addressed. This study used a novel DHA pretreatment experimental paradigm that targets acute cellular and molecular events during the first week after SCI in rats. We found that DHA pretreatment reduced functional deficits during the acute phase of injury, as shown by significant improvements in Basso-Beattie-Bresnahan (BBB) locomotor scores, and the detection of transcranial magnetic motor evoked potentials (tcMMEPs) compared to vehicle-pretreated animals. We demonstrated that, at 7 days post-injury, DHA pretreatment significantly increased the percentage of white matter sparing, and resulted in axonal preservation, compared to the vehicle injections. We found a significant increase in the survival of NG2+, APC+, and NeuN+ cells in the ventrolateral funiculus (VLF), dorsal corticospinal tract (dCST), and ventral horns, respectively. Interestingly, these DHA protective effects were observed despite the lack of inhibition of inflammatory markers for monocytes/macrophages and astrocytes, ED1/OX42 and GFAP, respectively. DHA pretreatment induced levels of Akt and cyclic AMP responsive element binding protein (CREB) mRNA and protein. This study shows for the first time that DHA pretreatment ameliorates functional deficits, and increases tissue sparing and precursor cell survival. Further, our data suggest that DHA-mediated activation of pro-survival/anti-apoptotic pathways may be independent of its anti-inflammatory effects.
Introduction
T
Injury to the spinal cord results in immediate mechanical primary damage, which is followed by secondary injury mediated by a complex cascade of molecular events (Hulsebosch, 2002). These immediate and acute secondary mechanisms include neurogenic shock, vascular insults, excitotoxicity, calcium-mediated injury, inflammation, cell death, and additional cellular and molecular disturbances (Dumont et al., 2001). Based on evidence showing that the major occurrence of apoptosis, white matter degeneration, and inflammatory responses occur during the first week after SCI (Beattie et al., 2000; Liu et al., 1997; Norenberg et al., 2004; Popovich et al., 1997), we focused this study on investigating the neuroprotective efficacy of prophylactic docosahexaenoic acid (DHA; 22:6 n-3) during this acute injury phase. DHA is a promising intervention to prevent acute neurodegeneration because it can simultaneously target several pathways that contribute to SCI pathophysiology. For instance, DHA and its derivatives show anti-inflammatory (De Smedt-Peyrusse et al., 2008; López-Vales et al., 2010; Mullen et al., 2010), and neuroprotective attributes (Bazan, 2006,2005; Belayev et al., 2009). Moreover, DHA activates both Akt and CREB, which have been associated with neuroprotection and myelin stability in vitro and in vivo (Akbar et al., 2005; Mukherjee et al., 2004; Salvati et al., 2008; Wu et al., 2008). These beneficial effects have been reported in a number of neurological studies (Lauritzen et al., 2000; Lim et al., 2005; Pan et al., 2009; Rodriguez de Turco et al., 2002), including SCI (Huang et al., 2007; King et al., 2006; Lang-Lazdunski et al., 2003; Ward et al., 2010). However, there is a lack of consensus regarding the primary neuroprotective mechanisms of DHA and whether the protection is primarily due to direct activation of pro-survival pathways or to modulation of inflammation.
Initial experiments from our lab showed that two low-dose intravenous injections of DHA, administered 1 week and 1 h before SCI, significantly improved important functional outcomes without inhibiting microglial activation at 1 week after SCI. Based on these findings, we focused this study on the characterization of these protective prophylactic effects to provide strong support for the idea that DHA pretreatment may preferentially target various secondary injury processes associated with cell death and acute neurological impairments after SCI. This study further supports our initial observations and demonstrates, for the first time, that prophylactic administration of DHA is neuroprotective and ameliorates behavioral deficits, even in the presence of an inflammatory environment during the acute phase of SCI. Our results suggest that this neuroprotection may be attributable, at least partially, to the activation of Akt and CREB signaling pathways, and support the use of DHA to successfully target the secondary injury responses associated with acute SCI.
Methods
Experimental procedures were performed in compliance with Loma Linda University and University of Puerto Rico School of Medicine regulations and institutional guidelines consistent with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Animals
Young adult (200–250 g) female Sprague-Dawley rats were used based on their behavioral and anatomical traits; females are normally less aggressive and less prone to urinary tract infections than their male counterparts. Animals were obtained from Hilltop Lab (Scottsdale, PA) and Charles River Laboratories (Portage, MI). The animals were housed with a light/dark cycle of 12 h/12 h, and fed ad libitum. Following preliminary experiments to investigate dose- and time-dependent responses, four groups of animals received a tail vein injection of either vehicle or docosahexaenoic acid (DHA; compound ID 445580; Sigma-Aldrich, St. Louis, MO; IUPAC: 4Z, 7Z, 10Z, 13Z, 16Z, 19Z-docosa-4, 7,10,13,16,19-hexaenoic acid; MW: 328.488280 g/mol; MF: C22H32O2) at a dose of 250 nmol/kg (∼82 μg/kg) in a volume of 5 mL/kg before undergoing contusive SCI or spinal cord laminectomy only (sham). The DHA stock was dissolved at 1 M in ethanol, diluted with sterile saline, adjusted to pH 7.4, and used immediately after preparation. This bolus administration leads to a concentration of ∼3.8 μM of DHA in the blood (50 nmoles DHA/13 mL blood), and has been shown to be neuroprotective while promoting recovery in rodents at 7 days and 6 weeks when administered 30 min after contusive SCI (Huang et al., 2007; King et al., 2006; Ward et al., 2010). We performed pilot experiments to evaluate the effects of multiple DHA doses, routes of administration, and times of intervention. The results reported herein include animal groups that were pretreated 1 week and 1 h before undergoing trauma to the spinal cord (see Fig. 1A). Animals did not receive further DHA administration after injury. The number of animals per group was 4–6 in the sham and preliminary study groups, and 11–12 rats in the SCI groups.
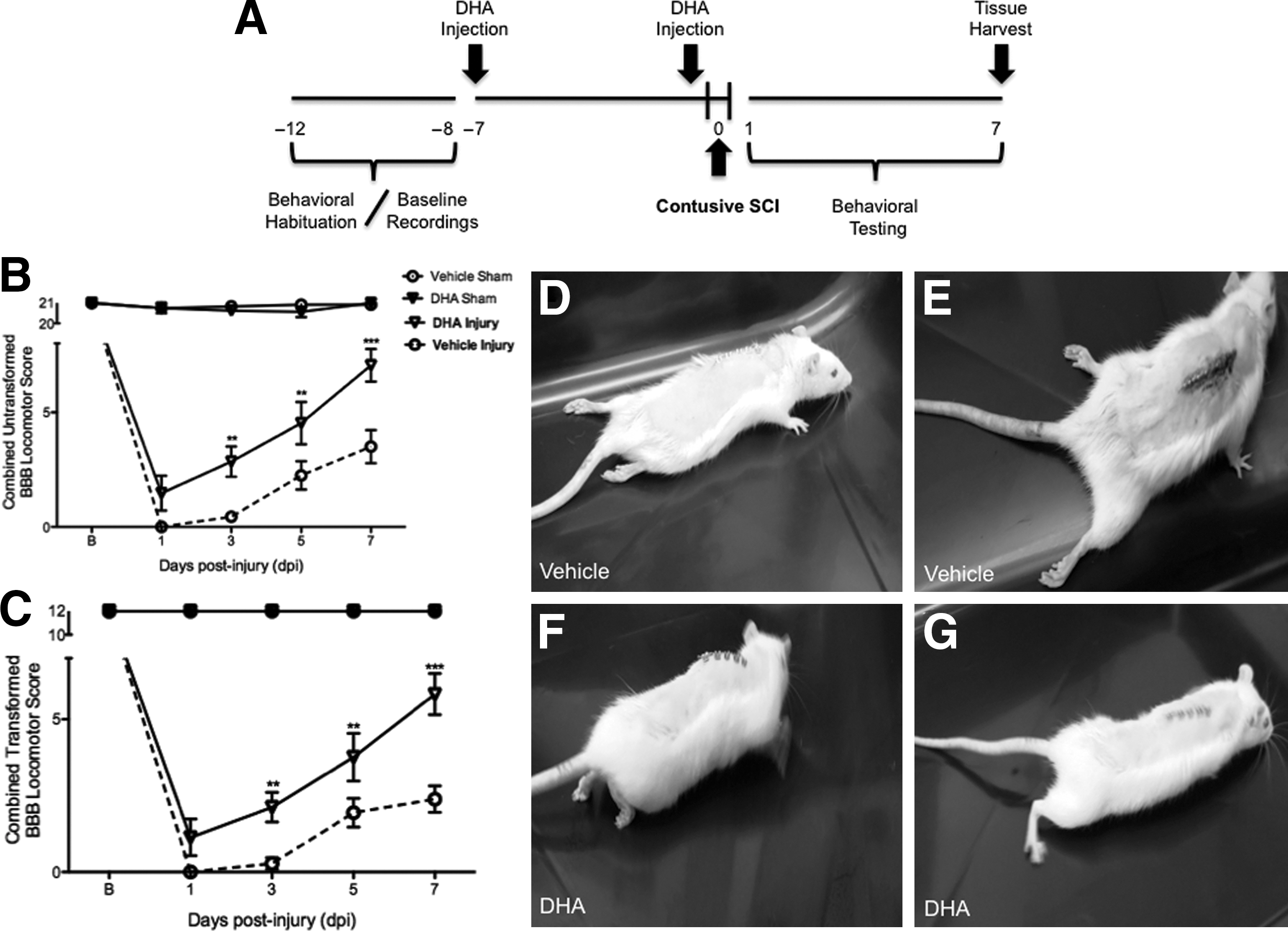
Acute beneficial effects of docosahexaenoic acid (DHA) pretreatment on the hindlimb neurological function of rats after a moderate injury, as assessed by the Basso-Beattie-Bresnahan (BBB) locomotor rating scale. (
Surgical procedures
To perform the injury to the spinal cord, the animals were deeply anesthetized with a mixture of ketamine (80 mg/kg) and xylazine (10 mg/kg). It is noteworthy that ketamine has been shown to induce metabolic changes and increased lipid turnover (Saranteas et al., 2005). However, this finding seems to be significant only at higher doses than the one used in this study. The spinal cord was injured at thoracic level 10 (T10) using the NYU/MASCIS impactor device, a reliable and consistent model, which exhibits a progressive secondary injury expansion, including apoptotic cell death and activation of inflammatory pathways (Figueroa et al., 2006; Gruner, 1992). Briefly, the skin and muscles overlying the spinal column were incised and a laminectomy performed at T10, leaving the dura intact. The T8 and T12 spinal processes were clamped to the impactor and contusion injuries were produced by releasing a 10-g rod from 12.5 mm onto the exposed T10 cord. Analysis of the impact parameters was used to identify potential outliers. Behavioral analyses were also used to corroborate differences in injury severity within groups. Two were excluded from the study based on these criteria. Sham animals received only a laminectomy to expose the spinal cord. After surgery, muscle layers were sutured and skin layers closed with wound clips.
Post-injury care
Each animal received 5 mL of sterile saline injected subcutaneously. Cefazolin (25 mg/kg SC; Bristol Myers Squibb, New York, NY) and Buprenex® (buprenorphine, 0.05 mg/kg SC; Reckett and Colman Pharmaceuticals, Inc., Richmond, VA) were given to all rats for 5 and 3 consecutive days, respectively. The bladders of injured rats were expressed using Crede's maneuver (abdominopelvic compression) three times a day until the voiding reflex was restored. All animals survived for 7 days after surgery and behavioral testing was performed during this acute phase. Severe urinary tract infections, autophagy, mortality, and morbidity are some of the major problems in post-operative care of spinal cord-injured animals. Three out of 52 animals were excluded from the data analysis for these reasons.
Analysis of locomotion function
Rats' spontaneous open-field locomotion was evaluated using the 22-point (0–21) Basso-Beattie-Bresnahan rating scale (BBB; Basso et al., 1995,1996). Briefly, the animals were acclimatized to the open-field environment during several sessions before testing. Subsequently, two double-blinded and trained observers assessed the locomotive function, joint movement, paw placement and rotation, coordination, and tail and trunk position and stability for 4 min. In this scale, a completely paralyzed rat scores 0, a rat with increasing joint movements but without weight support scores between 1 and 8, a rat with abnormal locomotion but with weight-supported steps (plantar or dorsal) and graded coordination patterns scores between 9 and 20, and a normal (and sham) rat scores 21.
Because our data were focused on the behavioral effects of DHA during the acute injury phase, locomotor scores were transformed to help assure that the data were amenable to parametric analyses and to increase statistical power (Ferguson et al., 2004). This transformation produces a continuous distribution by pooling together scores from 2–4 animals and thus avoids potentially suspect measures of performance in the lower end of the scale. Additional statistical power was also achieved by obtaining a measure of locomotor performance during earlier injury stages at 1, 3, and 5 days post-injury (dpi). This provides a behavioral index of the injury extent that is correlated with long-term recovery (approximate values of r>0.40, 0.60, and 0.70, respectively; p<0.05; Hook et al., 2004).
Electrophysiological assessment of recovery
Analyses of axonal conductance were performed using transcranial motor evoked potential (tcMMEP) studies using electrophysiological methods described previously (Figueroa et al., 2006; Loy et al., 2002a,2002b). The animals were sedated with a cocktail containing ketamine (40 mg/kg IM; Fort Dodge Animal Health, Fort Dodge, IA), and acepromazine (2.5 mg/kg IM; Vetus Animal Health, Rockville Center, NY). Once restrained, the magnetic transducer was placed over the skull and a short magnetic pulse (70 μsec, clockwise direction) was delivered using a Maxim 2002 stimulator (Magstim Company Limited, Spring Gardens, Whitland, United Kingdom) attached to a 50-mm-diameter hand-held magnetic transducer. Magnetically evoked electromyographic EMG responses were recorded using monopolar needle electrodes placed on both gastrocnemius muscles. Data were amplified using a Magstim Neurosign 100 (Magstim Company Limited). Afterward, the signal was converted and analyzed using the Digidata 1322A (Molecular Devices Corporation, Union City, CA), and Axoscope 8.2/Axograph X software, respectively (Molecular Devices Corporation). Only responses with amplitude measurements above 130 μV, and latencies shorter than 25 msec appearing at 70%, 85%, and maximum stimulation intensity were considered. Following our behavioral and electrophysiological assessment, the animals were processed for mRNA and histological analyses to evaluate acute outcome measures.
Tissue preparation
To prepare the tissue for histopathological analyses, we followed previously published procedures (Figueroa et al., 2006; Willson et al., 2002). The animals were submitted to fast and humane euthanasia with Fatal-Plus™ (Vortech, Dearborn, MI), and perfused transcardially with phosphate-buffered saline (PBS), followed by 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer. During rostro-caudal tissue harvest, the area of visible bruising of the cord from rod impact at T10–T11 was marked. The spinal cords were removed and post-fixed for 3 h in 4% PFA, cryoprotected in 30% sucrose for 12–16 h at 4°C, embedded in Tissue-Tek® O.C.T.™ compound (Sakura, Torrance, CA), and immediately frozen on dry ice. A series of transverse 20-μm coronal cryodissections were cut on a Richart-Jung Cryocut 1800 cryostat (Leica, Deerfield, IL) and kept at −20°C. Every fifth section was collected and placed on consecutive microscope slides (Superfrost Plus; Fisher Scientific, Pittsburgh, PA). Each slide contained sections 1 mm apart. The slides were stored at −20°C until use.
Myelin staining and quantification of white matter sparing
To study the integrity of myelin and cell survival after the DHA intervention, histological analysis was performed on coded Luxol fast blue/cresyl violet-stained coronal sections, as previously described (Figueroa et al., 2006). The stained sections were visualized with an Olympus BX50 microscope and digitized with a SPOT color cooled CCD camera (Diagnostic Instruments, Sterling Heights, MI). Unbiased morphometric analyses were performed using ImageJ software (NIH, Bethesda, MD;
Immunofluorescence
Immunofluorescence double labeling has been described previously (Figueroa et al., 2006). Spinal cord coronal sections were dried at room temperature for 10–15 min, washed with PBS, and post-fixed with 4% PFA for 10 min. The sections were washed, blocked, and incubated in either mouse anti-NeuN monoclonal antibody (clone A60, 1:250; Millipore, Billerica, MA), mouse anti-APC-7 monoclonal antibody (clone CC-1, 1:500; Calbiochem, San Diego, CA), mouse anti-NG2 chondroitin sulfate proteoglycan monoclonal antibody (1:250; Millipore), and mouse anti-neurofilament, heavy tail domain monoclonal antibody (clone RMO-24, 1:100; Millipore) to examine the immunoreactivity (IR) and cell numbers of neurons, mature oligodendrocytes, oligodendrocyte precursor cells, and phosphate-dependent NF-H domains in axons, respectively. Alternatively, sections were incubated in mouse anti-GFAP (1:200; Millipore), mouse anti-CD11b (OX42, 1:100; AbD Serotec, Raleigh, NC), and mouse anti-CD68 (ED1, 1:100; AbD Serotec) to examine the infiltration, proliferation, and recruitment of reactive astrocytes, monocytes/macrophages, and microglial cells, respectively. For double-labeling immunohistochemistry, sections were concomitantly incubated with either anti-NeuN, anti-APC-7, and anti-NG2 antibodies, and either rabbit anti-pAkt or rabbit anti-pCREB (1:100, 1:50, respectively; Cell Signaling, Danvers, MA). The antibody solutions were applied to the sections overnight at 4°C. On the following day, the sections were incubated with Alexa Fluor® 488 or 594-conjugated donkey anti-mouse or anti-rabbit antibodies (1:250; Invitrogen, Carlsbad, CA). Immunoblots, primary antibody omission controls, antibody preabsorption with immunizing peptide, and normal donkey serum controls were used to further confirm the specificity of the IR. The slides were examined with an Olympus Optical Fluoview FV1000 confocal microscope. The images were prepared for publication with Photoshop CS4 software (Adobe Systems, San Jose, CA).
TdT FraEL labeling (TUNEL) and quantification of cell death
To investigate the oligoprotective role of DHA after SCI, NG2+ cells were visualized and quantified after being exposed to the terminal deoxynucleotidyl transferase (TdT) FragEL DNA fragmentation detection kit as previously described (Figueroa et al., 2006). Briefly, for double-label immunofluorescence, TUNEL, and NG2+ cell analyses, sections were respectively labeled using Fluorescein-FragEL (Oncogene Research Products, Boston, MA), and a primary antibody against mouse anti-NG2 chondroitin sulfate proteoglycan (1:250; Millipore, Billerica, MA), that was subsequently labeled with Alexa Fluor 594-conjugated donkey anti-mouse antibody (1:250; Invitrogen). The sections were then cover-slipped with Hoechst 33342 mounting media and observed under an Olympus FluoView™ FV1000 confocal microscope (Olympus America, Inc., Center Valley, PA). Photomicrographs were captured and analyzed for co-localization by blinded investigators using NIH ImageJ software.
Unbiased stereological methods and histological analysis
Our unbiased stereological methods were based on the following steps: (1) estimation of the injury area before quantification. Spinal cord epicenters were marked during tissue harvest and sections were collected in successive slides thus providing a clearer estimation of the spinal cord regions. In addition, investigators used Luxol fast blue-cresyl violet staining and qualitative morphometric analyses to determine the regions of interest before proceeding with quantification. The lesion epicenter was defined as the spinal cord sections containing the least amount of spared white matter. (2) Randomization of location and orientation within the ventrolateral funiculus (VLF), corticospinal tract (CST), and ventral gray matter (VGM) using appropriate sampling techniques. (3) Counting of cells, pixel profiles, and areas by a defined test area within an unbiased counting frame. (4) Normalization of the time required for image acquisition to prevent photobleaching. (5) Digitalization and image quantification was carried out by blinded observers in at least four animals per group. For staining analyses, a minimum of four images per area per animal was used from the epicenter, and six areas immediately adjacent to the lesion site. Image analyses of similar areas were averaged and the total sample number used for statistical analysis equaled the number of animals used. (6) Cells identified as IR-positive were manually quantified for cell counts. Automated particle analyses were used after threshold correction to detect the levels of phosphorylated Akt and CREB in spinal cord sections. (7) To assess the extent of white matter content, automated particle detection was performed using ImageJ. Briefly, the images were converted to a binary outline image and adjusted to a similar intensity threshold. Integrated density measurements (area×mean gray values) were performed in spinal cord sections. The number of particles in an unbiased VLF rectangular selection was quantified using the particle analysis tab on ImageJ. (8) Nucleic acid fragmentation in NG2+ cells was quantified using NIH ImageJ, adapted from previous methods (Figueroa et al., 2006). The total FragEL fluorescence and the number of NG2+ cells that co-localized with Fluorescein-FragEL staining were quantified using the co-localization finder plug-in of NIH ImageJ software.
RNA extraction and real-time PCR
The animals were submitted to humane euthanasia with Fatal-Plus, and perfused transcardially with ice-cold 0.01 M PBS solution, pH 7.4 (Sigma-Aldrich) as previously described (Figueroa et al., 2006). Spinal cord segments containing the laminectomy/injury area (5 mm) were collected at 7 days post-operation (dpo). Total RNA was extracted using Trizol reagent (Invitrogen) according to the manufacturer's instructions, and the RNA concentration was determined on a NanoDrop spectrophotometer (Thermo Scientific). One microgram (μg) of total RNA from each sample was used for the first-strand cDNA synthesis. The first-strand cDNA synthesis was primed using oligo (dT) based on the SuperScript II First-Strand synthesis kit (Invitrogen). The synthesized cDNA was used as a template for relative quantification of target transcription in spinal cord tissue by real-time PCR. The cDNA was amplified by PCR using a pair of primers specific for protein kinase B (Akt, Akt1 primers were used in this study; FWD: 5′-TAC CAT GAA CGA CGT AGC CA-3′ and REV: 5′-AGG TGC CAT CAT TCT TGA GG-3′), and the cyclic AMP responsive element binding protein (CREB; FWD: 5′-CAT GGA CTC TGG AGC AGA CA-3′ and REV: 5′-GGG CTA ATG TGG CAA TCT GT 3′). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH; FWD: 5′-TGC CAC GAA GAC TGT GG-3′ and REV: 5′-TTC AGC TCT GGG ATG ACC TT-3′) and β-actin (FWD: 5′-GGG AAA TCG TGC GTG ACA TT-3′ and REV: 5′-GCG GCA GTG GCC ATC TC-3′) served as internal controls for data normalization. Real-time PCR amplification and analysis were carried out on a CFX96 Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA). SYBR green I (P/N 4309155; Applied Biosystems, Foster City, CA) was used as a fluorescent reporter dye for the presence of double-stranded DNA. After optimization of PCR conditions, 25-μL reactions containing cDNA, SYBR Green PCR Master Mix, and forward/reverse primers were prepared in triplicate. Each PCR run was repeated twice. Cycles consisted of 10 sec at 95°C for denaturation, and 30 sec at 60°C for annealing/extension. A negative (mock) control reaction without reverse transcription was also performed to verify genomic DNA contamination in our samples. The specificity of real-time PCR products was confirmed by melting curve analysis. A single melting curve peak was used to determine specificity for each gene target. Since the PCR efficiencies of our target and control genes were similar, the relative levels of target mRNAs were calculated using the comparative Ct (crossing threshold) values as previously described (Almaguel et al., 2009; Figueroa et al., 2006; Liu et al., 2008).
Statistical analysis
Data are presented as mean±SEM. Two-way analysis of variance (ANOVA), followed by Bonferroni post-hoc comparisons was used to determine the effect of our intervention and time on open-field locomotion scores. Fisher's exact test was used to determine the effects of our DHA pretreatment on the tcMMEP conduction latencies, followed by a relative risk estimate of the data. All other data were assessed by t-test. Statistical analyses were performed using Prism 5 Software (GraphPad Software Inc., San Diego, CA). Outliers were identified using the Grubbs' method, also known as ESD (extreme studentized deviate). Alternatively, values falling more than 2 SD from the mean were excluded from all statistical analyses. Only two animals were excluded from the study after using these exclusion methods. Statistical differences were considered significant at p<0.05.
Results
Effects of DHA pretreatment on hindlimb recovery during the acute phase of injury
After a 5-day training and behavioral habituation period, female Sprague-Dawley rats were given two DHA injections: one at 1 week and an additional challenge dose at 1 h before contusive SCI. Our rationale for the pretreatment schedule was based on long-standing pharmacokinetic analysis showing that administration of DHA results in fast incorporation (Yamazaki et al., 1991), and accumulates in neurons and glia up to 7 days after injection (Kabuto et al., 2009). Since our initial objective was to investigate the effects of DHA on the inflammatory responses in advance of neurodegenerative trauma, we used a pretreatment schedule that is based on routine interventions for inflammatory conditions, such as the use of methotrexate for rheumatoid arthritis (Cronstein, 2005). The animals were allowed to survive for a full week after injury, while behavioral data were collected at 1, 3, 5, and 7 dpi. At the end of the study, spinal cords were harvested and tissues were used for inmmunohistochemical studies, RNA, and protein analyses (Fig. 1A summarizes the study timeline).
The BBB locomotor grading scale was used to assess the effects of DHA pre-administration on the locomotive behavior of injured rats. Analysis of BBB scores during the acute injury phase demonstrate significant improvements in locomotor behavior in DHA-pretreated rats (Fig. 1B; untransformed data means±SEM: 21±0, 1.5±0.8, 4.5±0.9, and 7.1±0.7, at baseline, 1, 3, 5, and 7 dpi, respectively; n=10) compared to animals treated with vehicle injections at 1, 3, 5, and 7 dpi (untransformed data means±SEM: 21±0, 0±0, 0.4±0.2, 2.3±0.6, and 3.5±0.7, at baseline, 1, 3, 5, and 7 dpi, respectively; n=9; F(3,100)=373.19, MSE=2289; **p<0.01 at 1, 3 and 5 dpi, ***p<0.0001 at 7 dpi).
To validate our acute behavioral results, we used a simple post-hoc transformation that improves the metric properties of the lower portion of the BBB scale (Ferguson et al., 2004). Transformation (pooling) of BBB scores 2–4 removes the discontinuity in the grading scale to obtain an ordered scale and improves statistical power. Two-way ANOVA followed by Bonferroni post-hoc testing after employing this approach demonstrated very significant differences in locomotor behavior between treatment groups. Our data transformation and analysis revealed improved BBB scores in the DHA-pretreated group (Fig. 1C; transformed data means±SEM: 12±0, 1.1±0.6, 2.1±0.5, 3.8±0.8, and 5.8±0.7, at baseline, 1, 3, 5, and 7 dpi, respectively; n=10) compared to controls (transformed data means±SEM: 12±0, 0±0, 0.3±0.2, 1.9±0.5, and 2.4±0.4, at baseline, 1, 3, 5, and 7 dpi, respectively; n=9; F(3,100)=176.5, MSE=687.6, **p<0.01 at 3 and 5 dpi, ***p<0.0001 at 7 dpi).
A video recording of each subject's performance in the open field was obtained at 1, 3, 5, and 7 dpi. Still images from representative vehicle subjects showed limited movement, dragging of hindlegs (Fig. 1D), and slight movement of hindlimb joints (Fig. 1E) at 7 dpi. Remarkably, DHA-pretreated animals began to show signs of intermediate locomotor recovery as indicated by their ability to produce extensive movements of their joints with dorsal-stepping patterns (Fig. 1F). Further, end-point BBB score analysis revealed a group of DHA-pretreated animals with scores at or above 8, as evidenced by weight-supported locomotive behavior (Fig. 1G).
DHA pretreatment significantly improves axonal conduction at 7 days post-contusive spinal cord injury
tcMMEP in vivo electrophysiology was used to evaluate the integrity of thoracic ventrolateral spinal myelinated tracts, which are involved in rodent locomotion (Loy, 2002; Loy et al., 2002a). Electrophysiological responses were recorded before lesion (baseline) and at 7 dpi. Figure 2 shows representative tcMMEP traces for baseline (Fig. 2A) and treatment groups (Fig. 2B and C). Animals receiving vehicle injections displayed no tcMMEP responses at 7 dpi (Fig. 2B). Remarkably, 67% of the DHA-pretreated animals showed latencies below 25 msec and amplitudes above 130 μV after transcranial magnetic stimulation at 7 dpi (Fig. 2C). Data from baseline and treated animals are summarized in Figure 2D and E. Contingency table and statistical analyses revealed a p value of 0.0303 using Fisher's exact test (n=12). Strength of association analysis demonstrated an odds ratio of 23.40 CI (0.8925,613.5), with a sensitivity of 1.000 CI (0.5407,1.000), and a specificity of 0.6667 CI (0.2228,0.9567).
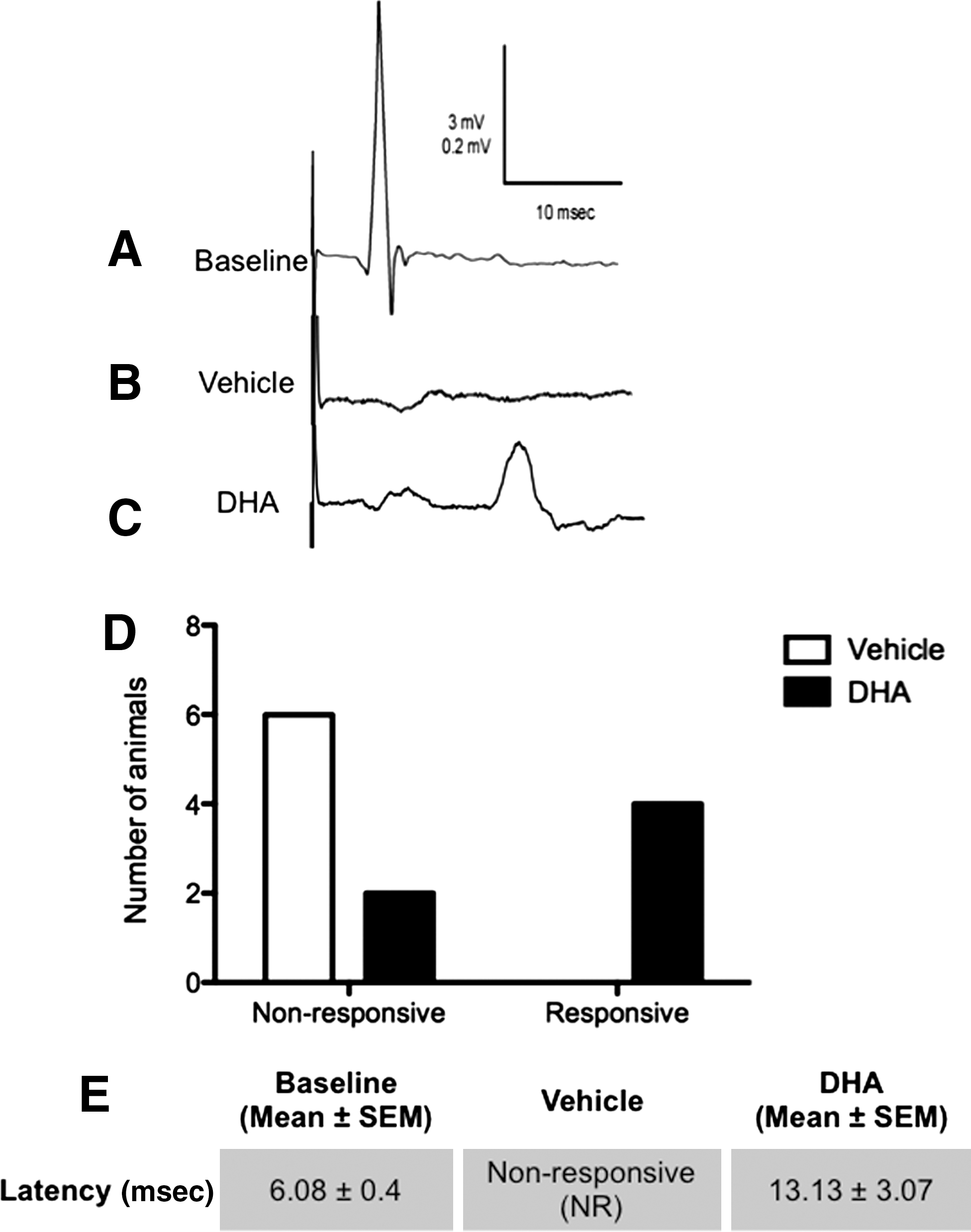
Docosahexaenoic acid (DHA) administration before spinal cord injury (SCI) reduces some of the conduction blockade associated with SCI at 7 days post-injury (dpi). Transcranial magnetic motor evoked potential (tcMMEP) responses recorded bilaterally from gastrocnemius muscles at 7 dpi. (
DHA pretreatment increased the density of the ventrolateral funiculi (VLF) spared white matter at 1 week post-injury
To evaluate the histopathology of functionally significant ventrolateral white matter tracts, and to validate our behavioral and electrophysiological findings, we performed Luxol fast blue (LFB) staining in spinal cord sections of DHA-pretreated and control animals. Figure 3A shows representative gray-scaled LFB-stained transverse sections encompassing seven distinct areas at 1-mm intervals that extend 3 mm rostrally and caudally from the lesion epicenter. The lesion epicenter (0 mm) was defined as the section exhibiting the greatest white matter damage at 7 dpi. Closer examination showed contusion-induced white matter degeneration as evidenced by reduced LFB staining (Fig. 3B and C). The staining density of randomized ventral white matter areas was quantified using automated particle counter analysis on binary photomicrographs from different VLF regions (Fig. 3B′ and C′). Quantitative histological analyses revealed increased myelin staining in the epicenter of DHA-pretreated rats compared to vehicle-treated animals at 7 dpi (Fig. 3D; Student's t-test, n at least 4 animals, *p<0.05). Measurements of the total volume of white matter demonstrated a significant increase in percent volume of spared white matter (∼10%) after DHA pretreatment compared to vehicle-pretreated animals at 7 dpi (Fig. 3E; Student's t-test, n at least 4 animals, **p<0.01). Immunohistochemistry with anti-neurofilament H (NF-H) was used to assess axonal integrity after our DHA intervention. Qualitative analysis of immunostained sections revealed more NF-H immunoreactivity in the ventrolateral white matter of DHA-pretreated animals compared to our control group (Fig. 3F and G). Control experiments were carried out to determine antibody specificity (Fig. 3H and I). Arrows denote NF-H-positive axons in white matter.
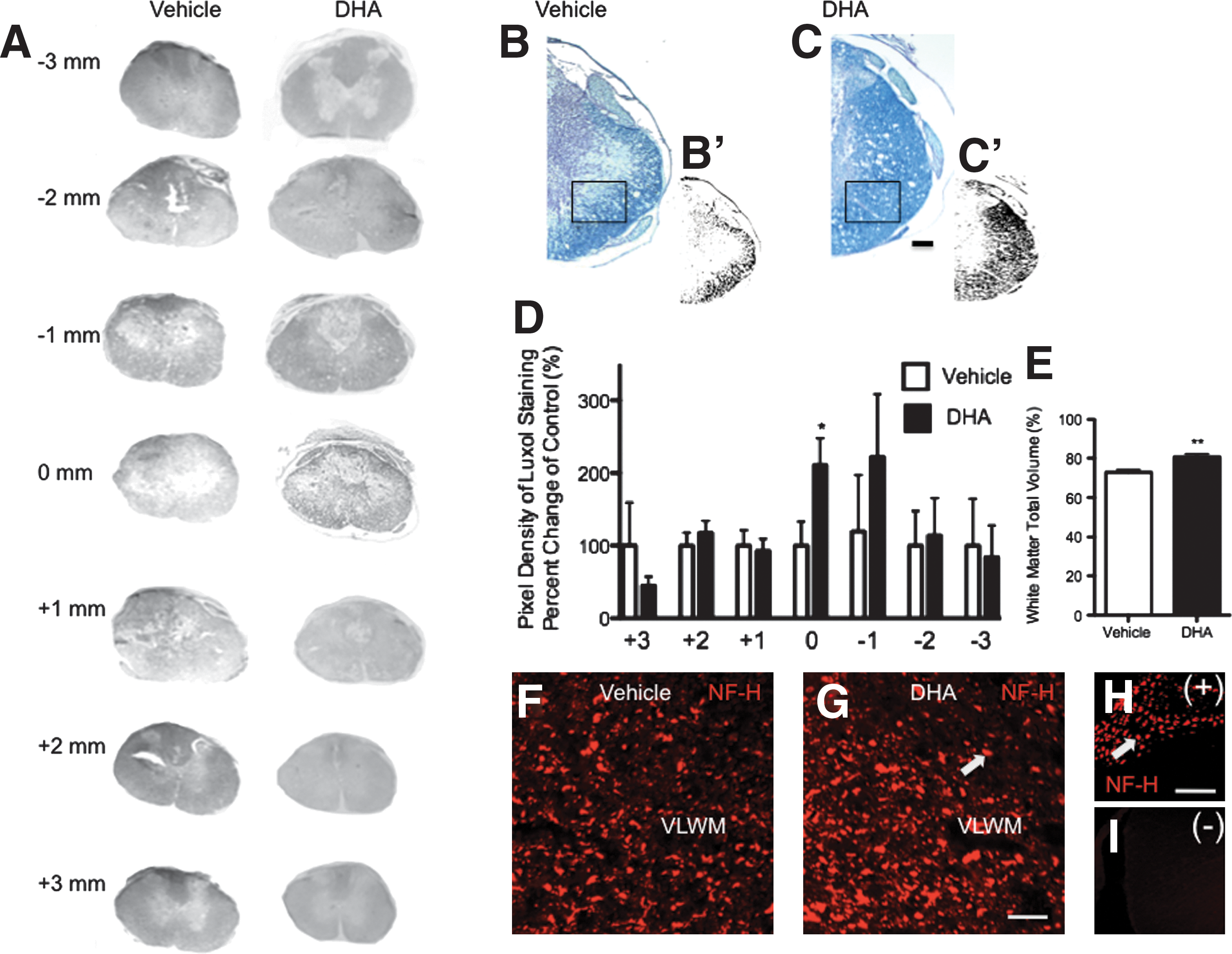
Docosahexaenoic acid (DHA)-pretreatment increased white matter sparing of functionally significant areas of the spinal cord at 7 days post-injury (dpi). (
DHA pretreatment decreases cell death in functionally relevant white matter areas and in oligodendrocyte precursor cells after SCI
To investigate whether DHA pretreatment reduces the activation of cell death pathways after SCI, we assessed DNA fragmentation in the white matter using fluorescence terminal-deoxy-transferase-mediated dUTP nick-end labeling (TUNEL). We observed a marked reduction in the number of TUNEL-positive cells in the white matter compared to those found in vehicle controls at 7 dpi (Fig. 4A; **p=0.0043; n=8 per group). Representative photomicrographs of white matter spinal cord regions stained with Fluorescein-FragEL and counterstained with 4,6-diamino-2-phenylindole (DAPI; B and E). Images demonstrate increased DNA fragmentation in vehicle-treated animals (B) compared to DHA-pretreated animals (D).
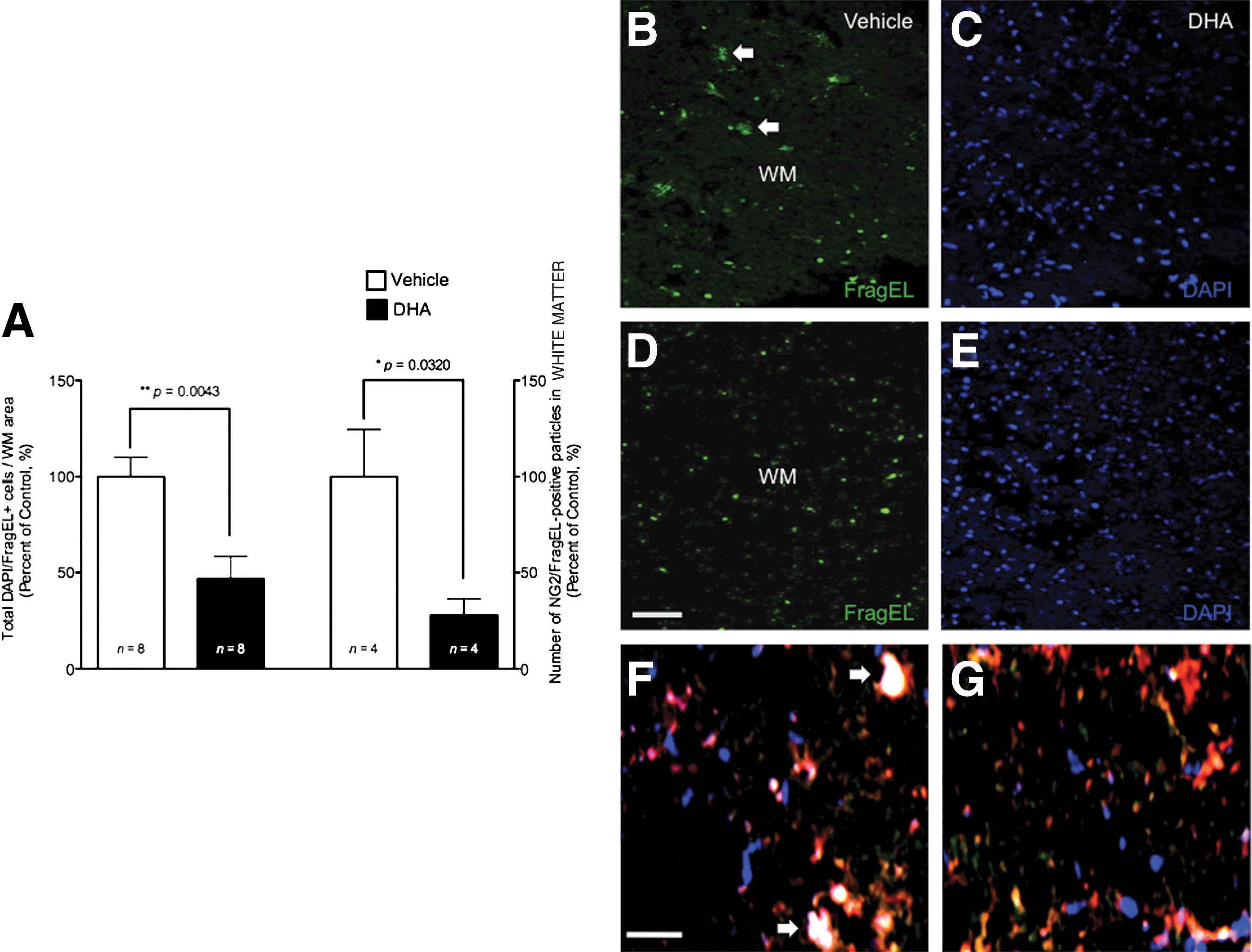
Prophylactic intervention with docosahexaenoic acid (DHA) reduces the number of white matter cells undergoing cell death after spinal cord injury (SCI). (
To investigate whether DHA decreases the susceptibility of oligodendrocyte precursor cells (OPCs) to undergo cell death after SCI, we quantified the DNA fragmentation in NG2+ cells using immunohistochemical analysis. Examination of immunoreactivity in the VLF revealed that the number of NG2+ cells undergoing DNA fragmentation was significantly reduced in our DHA-pretreated group at 7 dpi (Fig. 4A; *p=0.0320; n=4 per group). In these analyses, cells undergoing DNA fragmentation (Fluorescein-FragEL+) were examined and scored as positive only if they co-localized with NG2+ and DAPI immunoreactivity (Fig. 4F and G).
DHA pretreatment preserves NeuN+, APC+, and NG2+ cell numbers
To determine the quantity of neurons, oligodendrocytes, oligodendrocyte lineage cells, and inflammatory cells in areas involved in the observed locomotor and electrophysiological responses, immunohistochemical analyses were performed at the critical apoptotic stage of 7 dpi. Detection of neuronal and oligodendrocyte phenotypes was assessed by anti-NeuN and anti-adenomatosis polyposis coli (APC) antibodies, respectively. Alternatively, the anti-NG2 antibody was used to determine the number of potential OPCs. Cell counts were made in relevant areas involved in rodent locomotion such as VGM, VLF, and dorsal corticospinal tract (dCST; Fig. 5A). Our results revealed a significant increase in the number of NeuN+ cells in the ventral gray matter of DHA-pretreated animals (*p<0.05; vehicle, n=10; DHA, n=9). In addition, the relative number of oligodendrocytes and OPCs was doubled in the DHA-pretreated groups at 7 dpi (both *p<0.05; vehicle, n=6 rats; DHA, n=7 rats). Representative images from sections labeled with anti-NeuN (Fig. 5C and D), anti-APC (Fig. 5E and F), and anti-NG2 (Fig. 5G and H) show preservation of neurons, oligodendrocytes, and OPCs in the DHA-pretreated group at 7 dpi.
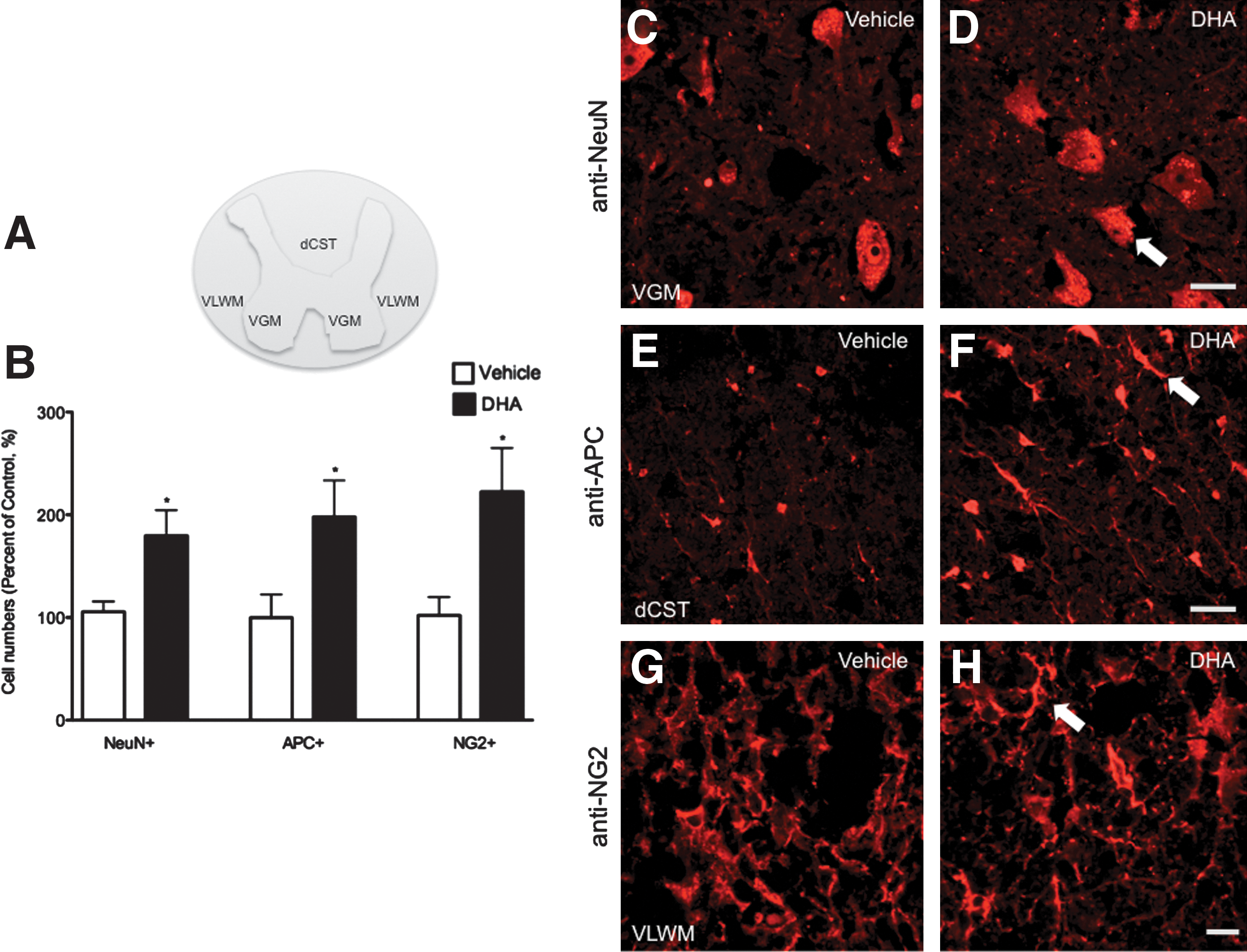
Pretreatment with docosahexaenoic acid (DHA) preserves neurons, oligodendrocytes, and oligodendrocyte precursor cells at 1 week after spinal cord injury (SCI). (
Prophylactic injection with DHA did not result in significant anti-inflammatory effects at 1 week following compression injury
Although the beneficial effects of inflammation in SCI remain controversial, it has been increasingly recognized that the recruitment and activation of inflammatory cells during the first few days after trauma may play a critical role in modulating functional recovery after SCI (Popovich and Jones, 2003). To determine whether DHA pretreatment reduces the expression of these inflammatory markers in physiologically relevant areas, we quantified the number of immunoreactive particles using immunohistochemical analysis. Surprisingly, DHA-pretreated animals showed no significant differences in the immunoreactivity to ED1 (monocytes/macrophages/microglia; Fig. 6A–E), and GFAP (astrocytes; Fig. 6F–J), compared to vehicle controls (p>0.05; n=at least 4 animals per group). To validate our ED1 results and determine whether our DHA prophylactic intervention reduced the activation of microglia, OX42 antibody was employed to examine the expression of CD11b/c in microglia in the spinal cord. No significant differences were observed between the studied groups (data not shown).
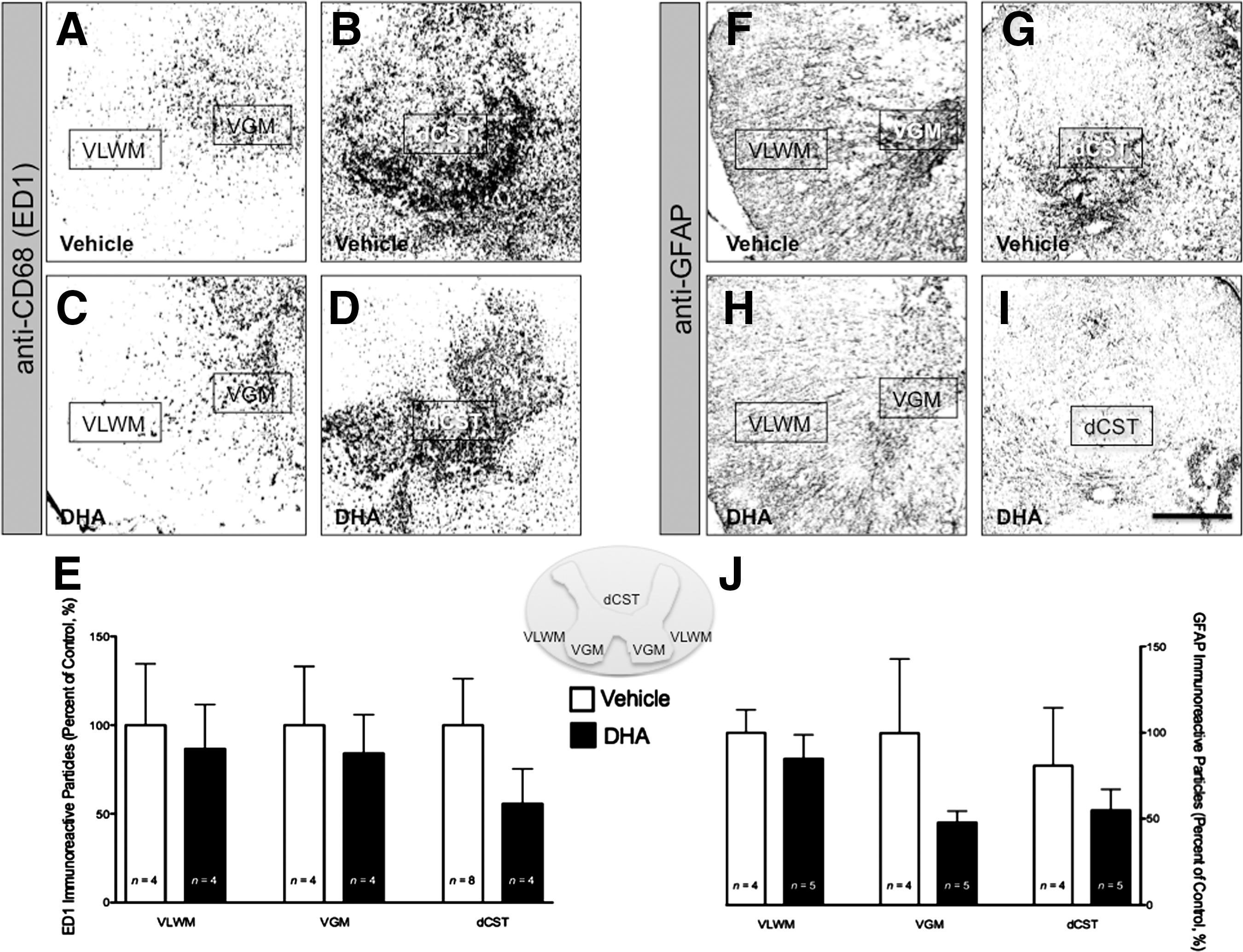
Pretreatment with docosahexaenoic acid (DHA) did not affect inflammatory markers after spinal cord injury (SCI). Photomicrographs from ED1-labeled monocytes/macrophages in animals injected with vehicle (
DHA pretreatment induces the upregulation and activation of Akt and CREB after contusive SCI
Akt and CREB have been associated with DHA protective effects on survival and myelin stability in vitro and in vivo (Akbar et al., 2005; Mukherjee et al., 2004; Salvati et al., 2008; Wu et al., 2008). We examined whether DHA pretreatment increases the mRNA and protein phosphorylation levels of Akt and CREB after acute SCI. Real-time PCR analysis revealed that DHA pretreatment increased Akt and CREB levels 1.61- and 2.03-fold, respectively, at 7 dpi compared to vehicle controls (Fig. 7A; both *p<0.05; n=4 animals/group). Quantitative determination of Akt and CREB protein phosphorylation was performed by measuring the relative immunoreactivity to anti-pAkt and anti-pCREB antibodies. Gray-scale calibration of 0–255, representing white-to-black, permitted measurements of optical density values in immunoreactive sections. Quantitative analysis of pAkt and pCREB immunoreactivity in our DHA-pretreated group demonstrated a 1.59- and 1.70-fold increase, respectively, when compared to controls (Fig. 7B; both *p<0.05; vehicle, n=4 animals; DHA, n=5 animals). Interestingly, DHA pretreatment did not change the levels of pAkt and pCREB in the uninjured spinal cord (data not shown). Gray matter photomicrographs show pAkt (Fig. 7C and D), and pCREB (Fig. 7F-G), immunoreactivity in neuron like cells (arrows). These cells had an intensely positive pAkt and pCREB signal in nuclear and perinuclear regions (Fig. 7D and G). Antibody preabsorption with blocking peptides shows reduced immunoreactivity, thus confirming the specificity of each antibody (Fig. 7E and H).
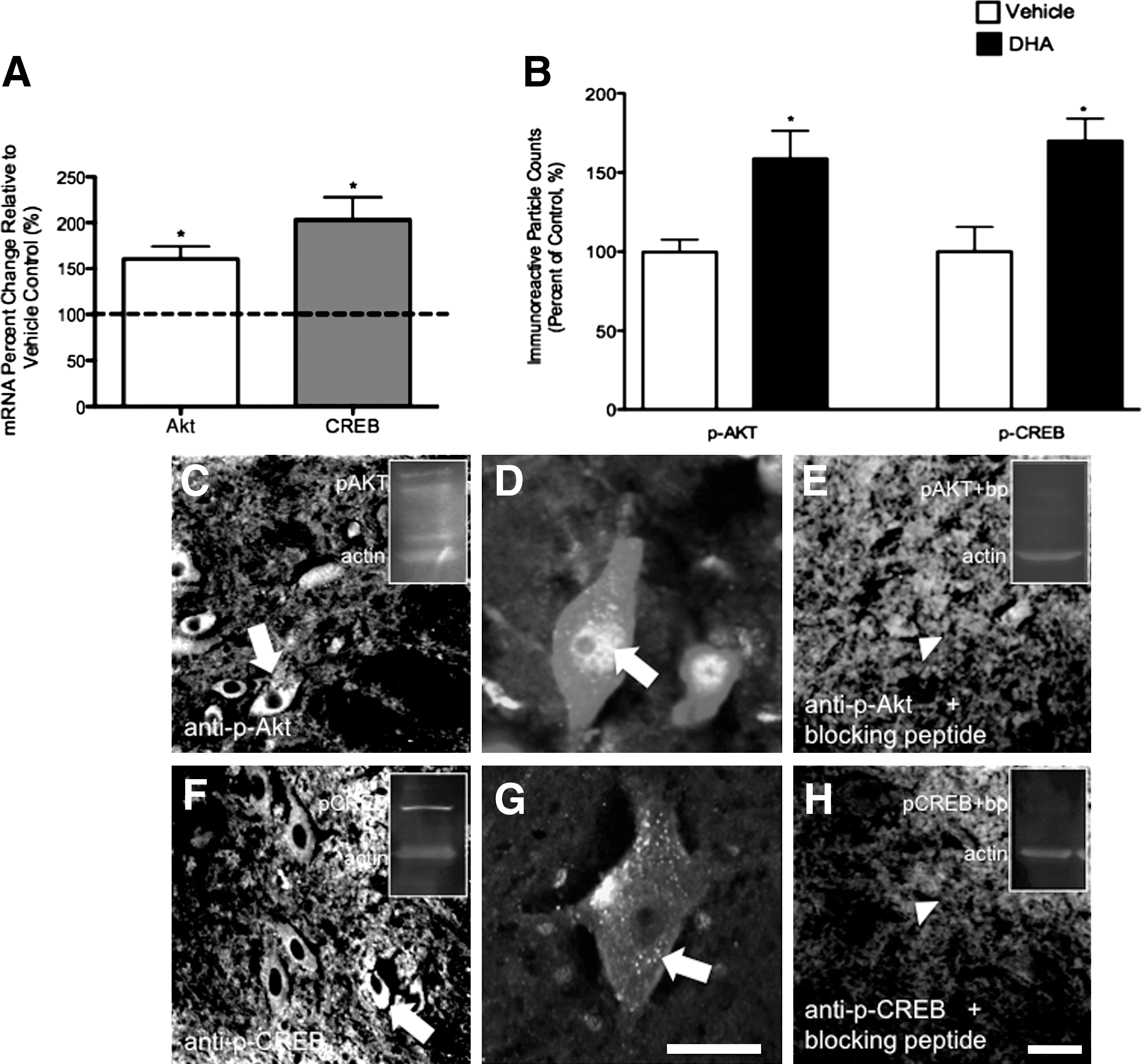
Intravenous administration of docosahexaenoic acid (DHA) before spinal cord injury (SCI) results in increased mRNA and phosphorylated protein levels of Akt and CREB at 7 days post-injury. (
We also examined whether DHA pretreatment resulted in increased pAkt and pCREB levels in the different cellular phenotypes that were neuroprotected at 7 dpi. Gray matter double-labeling immunohistochemistry showed immunoreactivity for pAkt (Fig. 8A–L) and pCREB (Fig. 9A–L) in NeuN+ cells. White matter photomicrographs also demonstrated concomitant expression of pAkt and pCREB in NG2+ (Fig. 8M–R; Fig. 9M–R, respectively), and APC+ cells (Fig. 8S–X; Fig. 9S–X, respectively).
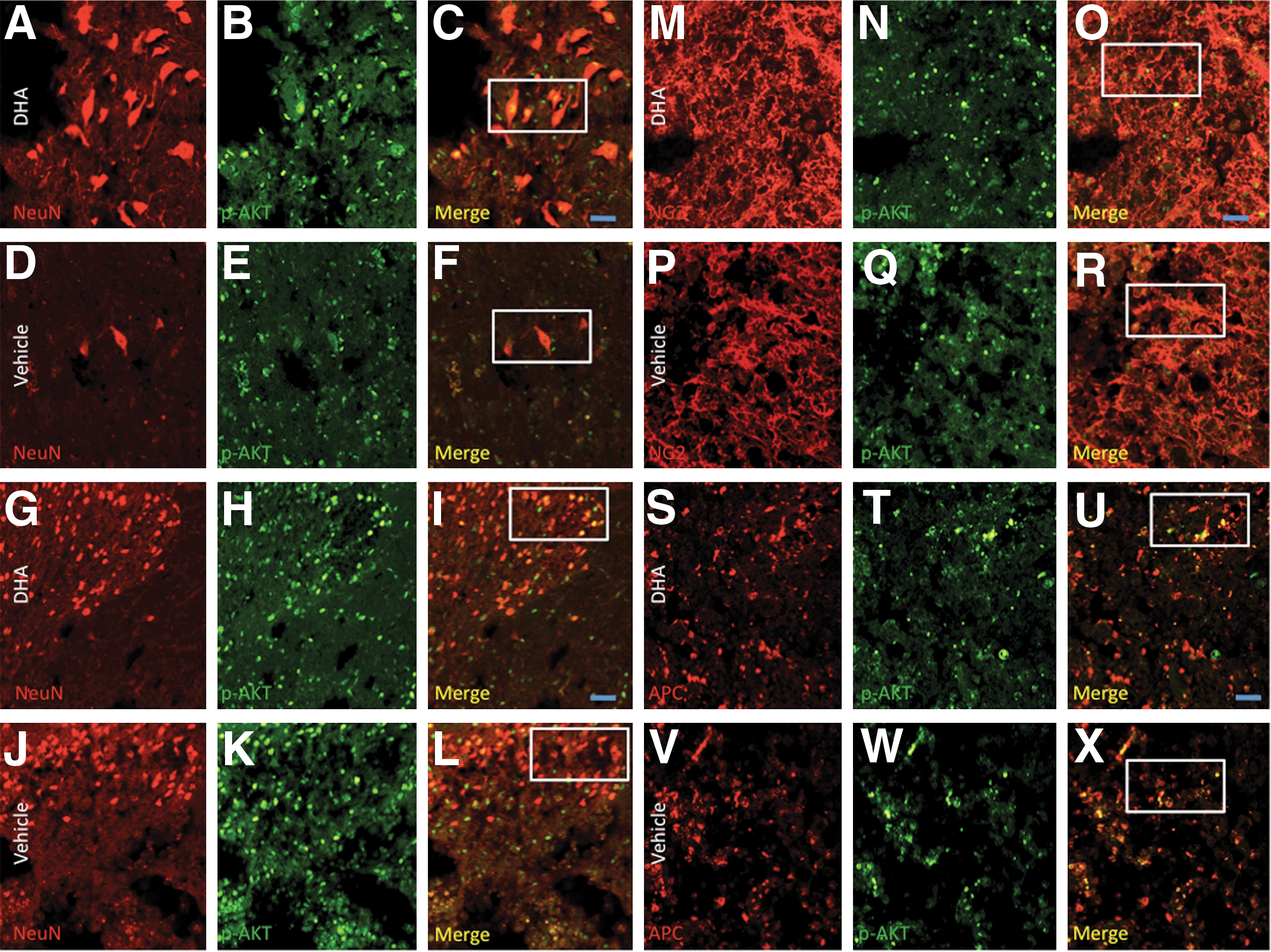
Double labeling and merge photomicrographs of NeuN, adenomatosis polyposis coli (APC), and NG2 cells immunoreactive for pAkt in transverse sections at the ventral and dorsal gray matter, ventrolateral funiculus, and dorsal corticospinal tract of the T10–T11 spinal cord segment. pAkt expression and neuron cell numbers were higher in ventral gray motor neurons of docosahexaenoic acid (DHA)-pretreated (
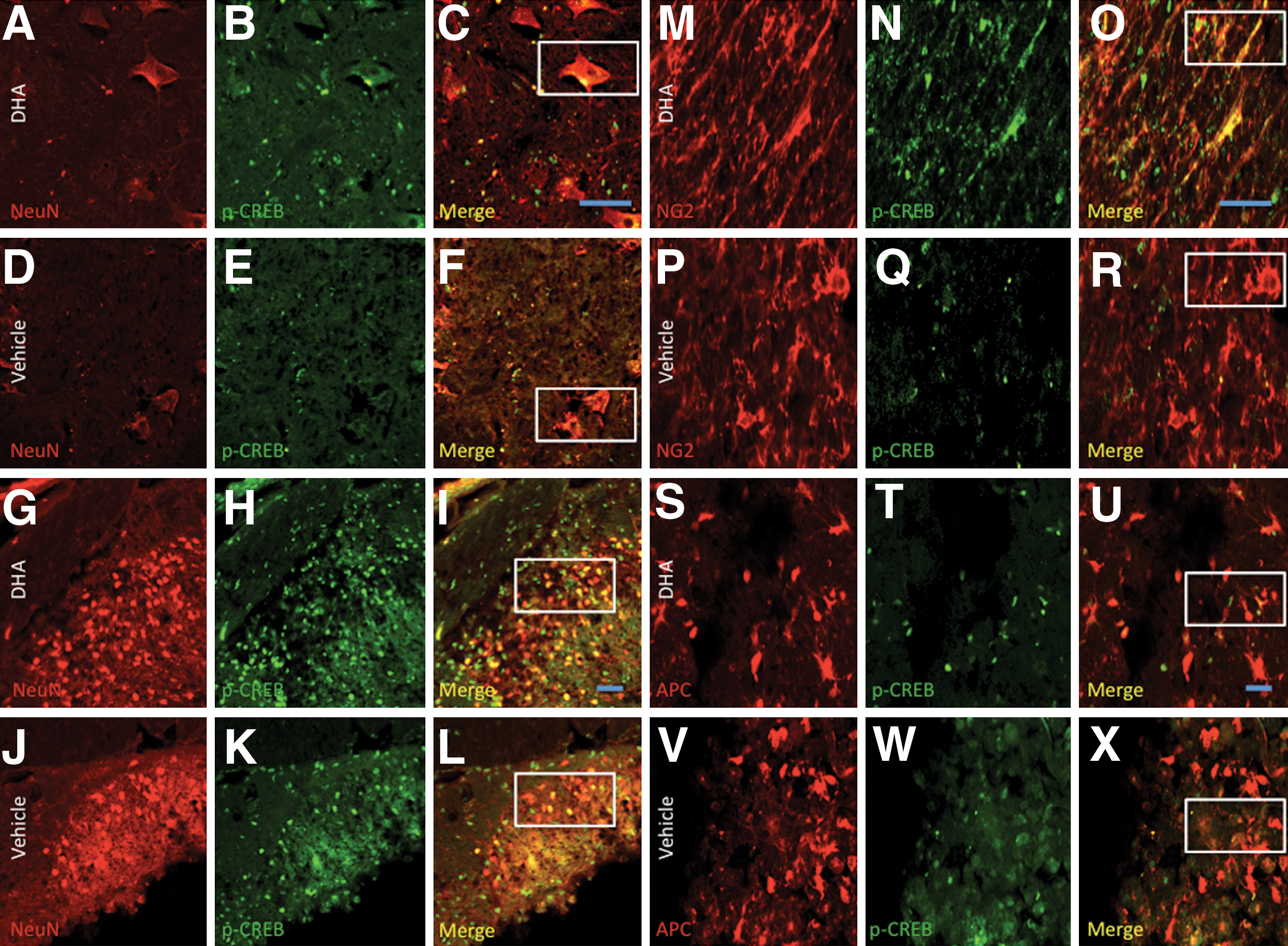
Double labeling and merge photomicrographs of NeuN, adenomatosis polyposis coli (APC), and NG2 cells immunoreactive for pCREB in transverse sections at the ventral and dorsal gray matter, ventrolateral funiculus, and dorsal corticospinal tract of the T10–T11 spinal cord segment. Photomicrographs show increased pCREB expression and neuron cell numbers in ventral (
Discussion
The results of this study show that docosahexaenoic acid (DHA), when injected in a prophylactic manner, resulted in amelioration of locomotor and conduction deficits observed during the acute phase of SCI. These acute beneficial effects were evidenced by (1) improved BBB scores, (2) the emergence of motor evoked potentials, (3) white matter tissue preservation, (4) increased numbers of neurons, oligodendrocytes, and precursor cells, (5) activation of pro-survival and anti-apoptotic responses without reduction in inflammatory markers, and (6) increased gene expression and activation of Akt and CREB. Altogether, these data suggest that DHA-mediated neuroprotection can occur even under significant inflammatory conditions, and supports the prophylactic use of DHA in situations of anticipated high risk of damage to the spinal cord.
There are numerous clinical and occupational scenarios that are known to exhibit a significant risk for SCI. Examples of these conditions include cerebral palsy, spina bifida, amyotrophic lateral sclerosis, vitamin B12 deficiency, herpes simplex viral infection, multiple sclerosis, atherosclerosis, iatrogenic ischemia, syringomyelia, spondylolysis, disc herniations, radiation toxicity, tumors, contact sports, and warfare (Boden and Jarvis, 2009; McDonald and Sadowsky, 2002; Weaver et al., 2009). Although the primary pathophysiology of non-traumatic SCI is disease-specific, the clinical signs, symptoms, and general therapeutic principles are similar to traumatic SCI. The clinical safety and neuroprotective properties of DHA make it an ideal prophylactic treatment in these situations because of its ability to simultaneously inhibit multiple pathways involved in cell death and dysfunction such as apoptosis (Lukiw et al., 2005), excitotoxicity (Hogyes et al., 2003), ROS generation (Sarsilmaz et al., 2003), and inflammation (Serhan et al., 2008). Interestingly, contrary to dietary DHA, a single low-dose DHA injection results in acute blood–brain barrier penetration and incorporation in the brain (Polozova and Salem, 2007), and is rapidly metabolized and accumulates in neurons and glial cells, even at 7 days after the injection (Kabuto et al., 2009). This is consistent with recent studies revealing that DHA injections are neuroprotective at 7 dpi when administered after injury to the spinal cord (King et al., 2006), whereas dietary supplementation with DHA did not confer neuroprotection (Huang et al., 2007). It is reasonable that injection pretreatment with DHA could lead to a significant inhibition of very early damaging events and promote recovery after SCI.
A striking finding of this study is that DHA pretreatment led to a significant reduction in locomotor deficits associated with contusive SCI, as evidenced by the improvements in BBB locomotor scores at different time points during the acute phase of SCI. Remarkably, DHA-pretreated animals exhibited extensive movements in all three joints and weight-supported steps at 7 dpi. Our findings are supported by studies showing that pretreatment with polyunsaturated fatty acids (PUFAs) provides tolerance against ischemic-induced neurodegeneration (Blondeau et al., 2002), reduces disabilities after traumatic brain injury in rats (Wu et al., 2004), confers neuroprotection from brain hypoxia-ischemia (Berman et al., 2009), and increases resistance to pentylenetetrazol-induced seizures (Taha et al., 2010).
Another important finding of our study is the presence of transcranial magnetic motor evoked potential (tcMMEP) electrophysiological responses at 7 dpi. The appearance of these responses provides a reproducible and objective assessment of axonal conduction of descending motor pathways in both the normal and injured spinal cord (Figueroa et al., 2006; Linden et al., 1999; Magnuson et al., 1999). Moreover, this measurement is useful to evaluate the conduction capacity and integrity of descending myelinating pathways located in the medial ventrolateral funiculus (VLF; Loy et al., 2002a,2002b). Our results are the first demonstration of in vivo electrophysiological improvements after acute administration of DHA before SCI. This may represent preservation of myelinated fibers and return of function in spared reticulospinal fibers, which run mainly in the VLF and carry important motor information. Our anatomical and histological data support this idea by showing significant sparing of white matter tissue and increased axonal integrity in DHA-pretreated animals compared to vehicle controls at 7 dpi.
Both neurons and oligodendrocytes are extremely vulnerable to cell death following SCI (Bunge et al., 1993). Several lines of evidence demonstrate that during the first few days after injury, acute apoptotic and autophagic waves contribute to tissue damage and loss of function after SCI in animals (Abe et al., 1999; Beattie et al., 2000; Kanno et al., 2009; Li et al., 1999; Liu et al., 1997; Lu et al., 2000), and humans (Emery et al., 1998). DHA has been shown to be neuro- and oligoprotective when administered and supplemented after SCI (Huang et al., 2007; King et al., 2006; Lang-Lazdunski et al., 2003; Ward et al., 2010). Here, we demonstrated that DHA administration before SCI significantly increased neuronal and oligodendrocyte cell survival at 7 dpi. Previous in vitro work from our laboratory supports these protective effects, after showing that DHA pretreatment protects NGFD-PC12 cells from lipotoxicity-induced apoptosis (Almaguel et al., 2009). In agreement with our findings, evidence demonstrates that inhibition of early apoptotic events, such as cytoskeletal degradation, is neuroprotective and results in improved neurological function after SCI (Schumacher et al., 2000).
Proliferation of progenitor cells expressing NG2 proteoglycans may play a role in replenishing oligodendrocytes, and may determine neurological outcomes after acute SCI (Lytle and Wrathall, 2007; Sellers et al., 2009; Tripathi and McTigue, 2007). It was recently proposed that these precursor cells might also have a broader relevance for gray matter physiology, based on the observation that some of these cells can mature into cortical projection neurons (Rivers et al., 2008). Therefore, strategies targeting precursor cells with beneficial pro-survival agents may represent a viable mechanism for neuronal and oligodendrocyte replacement, spinal cord homeostasis, and functional recovery. Our results show, for the first time, not only that DHA pretreatment leads to increased numbers of NG2
Although DHA plays an important role in neuroprotection and functional recovery, we lack information regarding its underlying mechanisms after SCI. Previous reports suggest that therapeutic treatment with DHA after SCI may exert its neuroprotective effects, at least partially, through negative modulation of cycloxygenase-2 protein expression, and by means of reducing peroxidation and oxidation products (Huang et al., 2007). This same group demonstrated that DHA administration after SCI results in improved recovery associated with a significant reduction of inflammatory markers, such as ED1 (Huang et al., 2007; King et al., 2006). Interestingly, our study showed no significant differences in the immunoreactivity levels of various inflammatory makers, including GFAP (astrocytes), CD68 (macrophages), and CD11b (microglia). This discrepancy suggests that DHA pretreatment may have different targets early after injury. DHA is rapidly and selectively incorporated into the central nervous system (CNS) when injected in rats (Polozova and Salem, 2007), and it is also acutely released from cell membranes after SCI (Demediuk et al., 1989). Thus it is plausible that acute accretion of DHA in advance of SCI might promote neuroprotection by triggering the docosanoid pathway and DHA-derived messengers, including docosatrienes, resolvins, and neuroprotectins, such as neuroprotectin D1 (Bazan, 2006). Also, while DHA multiple double bonds are excellent targets for lipid peroxidation and could potentiate neurotoxicity, DHA may also function as a free-radical scavenger to reduce the neuronal and glial susceptibility to oxidative stress after trauma. We cannot disregard the possibility that DHA could reduce the effects of primary injury by modulating the biophysical properties of membranes and axons (Mayurasakorn et al., 2011). Future work is needed to study the immediate targets of DHA when administered before injury to the spinal cord.
Dietary DHA increases the levels of activated Akt and CREB in rats (Wu et al., 2008). These signaling pathways play a major role in DHA-mediated neuronal survival (Akbar and Kim, 2002), provide resistance to CNS insults (Kitagawa, 2007; Nakajima et al., 2002; Rybnikova et al., 2008), and increase endogenous protection in the retina (Dreixler et al., 2009). Our findings showing that Akt and CREB expression was upregulated in DHA-pretreated rats are in agreement with the involvement of these molecules in estrogen-mediated neuroprotection after SCI (Yune et al., 2008). Current work in our lab is aimed at investigating whether Akt/CREB activation is required to exert DHA neuroprotective effects in a prophylactic manner (Descoberth et al., unpublished observations).
In summary, our findings are the first to demonstrate that acute pretreatment with DHA renders neural tissue more resistant to SCI-induced secondary damage, possibly by limiting the expansion of the apoptotic pathways (See summary in Table 1). This report substantiates the relevance of DHA as a preventive neuroprotective agent, which may confer a clear advantage in situations of anticipated neuronal damage, and deserves further consideration as an innovative approach to prevent neurodegeneration after neurotrauma. Further studies testing the durability of these neuroprotective effects over longer post-SCI survival times are being tested.
DHA pretreatment in advance of contusive SCI results in relevant physiological, anatomical, histological, and molecular changes. Our electrophysiological, immunohistochemical, and molecular biology assays indicate that two bolus injections of DHA given 1 week and 60 min before SCI reduces locomotor impairments and conduction latencies, increases neuroprotection, and results in increased mRNA and phosphorylated protein levels of the pro-survival proteins Akt and CREB (DHA, docosahexaenoic acid; SCI, spinal cord injury; CREB, cyclic AMP-responsive element binding protein).
Footnotes
Acknowledgments
We are thankful to Laurivette Mosquera and Luz Arocho for their support during the tissue collection. Thanks to Odrick Rosas and Jose Santiago for their technical support, and to Dr. Jo-Wen Liu and Lorena Salto for their critical reading and helpful comments. This work was supported in part by grants from the National Institutes of Health 5P20MD001632 and 5R25GM060507 (to M.D.L.), the Montogmery Street Foundation (to M.D.L.) R25-GM061838 and S06-GM008224 (to J.D.M.), and National Institute for Neurological Disorders and Stroke INDS 39405 (to J.D.M.).
Author Disclosure Statement
No competing financial interests exist.
