Abstract
Lesions in specific areas of the rat motor cortex generate deficits related to fine movement performance affecting the forelimb. We have previously shown that transplants of embryonic frontal cortex ameliorate these motor deficits. Amelioration has been associated with a functional integration of the transplant due to the connections established between the host brain and the graft. In the current investigation, the electrophysiological properties of the transplanted cells and the connections both intra-transplant and with the adjacent host cortex are analyzed. For this purpose, adult rats with a motor cortical lesion plus a fetal cortical graft were used. Neurons in the transplant were recorded using sharp electrodes or whole-cell recordings in brain slices. Application of intracellular depolarizing pulses showed two patterns of cell firing: regular and burst spiking. Postsynaptic responses evoked by both, intra-transplant and adjacent host cortex stimulation were mediated by glutamic acid acting on non-NMDA and NMDA receptors, and were modulated by both cholinergic and GABAergic drugs. In some cells, supra-threshold intra-transplant stimulation generated an epileptiform-like discharge, suggesting an imbalance between excitatory and inhibitory synapses. As expected, immunohistochemistry for cholinergic and GABAergic markers confirmed the electrophysiological results. Thus we show electrophysiological and immunohistochemical evidence supporting the functional development and integration of grafted cells into the host neocortex of adult animals.
Introduction
A
In other studies, transplanted cells have been reported to be metabolically active, integrated, and topographically organized within the host cortex (Grabowski et al., 1993; Ebrahimi-Gaillard et al., 1995; Gaillard et al., 1998).
Electrophysiological transplant integration of fetal tissue has been studied in vivo by extracellular recordings in grafted rats, suggesting that restoration of some kind of capability to evoke synaptic potentials is needed for such integration (Xu et al., 1991; Chen et al., 2002). It is thought that functional improvement of transplanted tissue is time-dependent and appears between 45 and 60 days in adult rats, which is sufficient time for the establishment of anatomical graft-host connections (Fernandez-Ruiz et al., 1991; Plumet et al., 1991; Miranda et al., 1997). Hence these studies suggest that recovery requires a functional integration of the transplanted embryonic tissue into the host, with the presence of connections between the grafted tissue and the host brain.
Nevertheless, the electrophysiological properties of the transplanted neurons and the intra-transplant and transplant-host brain connections in adult animals have yet to be elucidated. In the present study we address these issues by carrying out both sharp electrode analysis and whole-cell recordings to study implanted neurons 3 months after transplantation, a period reported to be effective for host recovery (Kolb et al., 1988; Stein et al., 1988; Riolobos et al., 2001). We show the firing patterns and synaptic properties of neurons within grafts in frontal cortex slices. These results, also supported by immunohistochemical data, demonstrate that the transplanted embryonic tissue can be functionally integrated into the host.
Methods
Animals
All experiments in this study were carried out in accordance with the animal care guidelines of the European Communities Council (86/609/ECC), and every effort was made to minimize the suffering and number of animals used. Fifty male Wistar rats (Criffa Laboratories, Barcelona, Spain) were used, aged 3 months on delivery. They were housed, two per cage, under natural light/dark conditions at a temperature of 18–20°C. Water and food were available ad libitum.
Cortical lesion
All surgical procedures were performed under anesthesia with 20 mg/kg Equithesin (injected IP). The animals (n = 50) were unilaterally lesioned in the motor cortex. The lesion was made by aspiration as previously described in detail elsewhere (Riolobos et al., 2001). Briefly, deeply anesthetized animals were placed in a stereotaxic apparatus and the skull exposed at the level of the bregma. The animals were lesioned at the coordinates indicated by Neafsey and associates (1986) to remove the forelimb area of the motor cortex: anteroposterior 1–4 mm anterior to the bregma; lateral 1–3.5 mm right or left laterally to the midline; and dorsoventral the ventral limit of the lesion was the white matter underlying the cortex.
Transplantation
Seven days after lesioning the animals were subjected to unilateral transplantation. The donor tissue was obtained from 16-day-old rat embryos. Transplantation techniques were performed as previously described (Riolobos et al., 2001). Briefly, the pregnant rat was anesthetized with Equithesin and the embryos removed one at a time as needed. The donor's skull was cut at the midline and peeled back, the brain was dissected out, and the meninges were removed and placed in a dish with sterile glucose saline. Donor fetal tissue was set up while the host was being prepared to receive it. Two solid pieces of the fetal frontal cortex of approximately 1 mm2 each, including the total thickness of the cortex, were taken from the embryo and positioned at the bottom of the single motor cortex cavity in the host animal (Fig. 1A–C). The cavity containing the transplant was filled with a piece of gelfoam soaked in glucose-saline and the skin sutured. Pregnant rats were sacrificed by anaesthetic overdose.
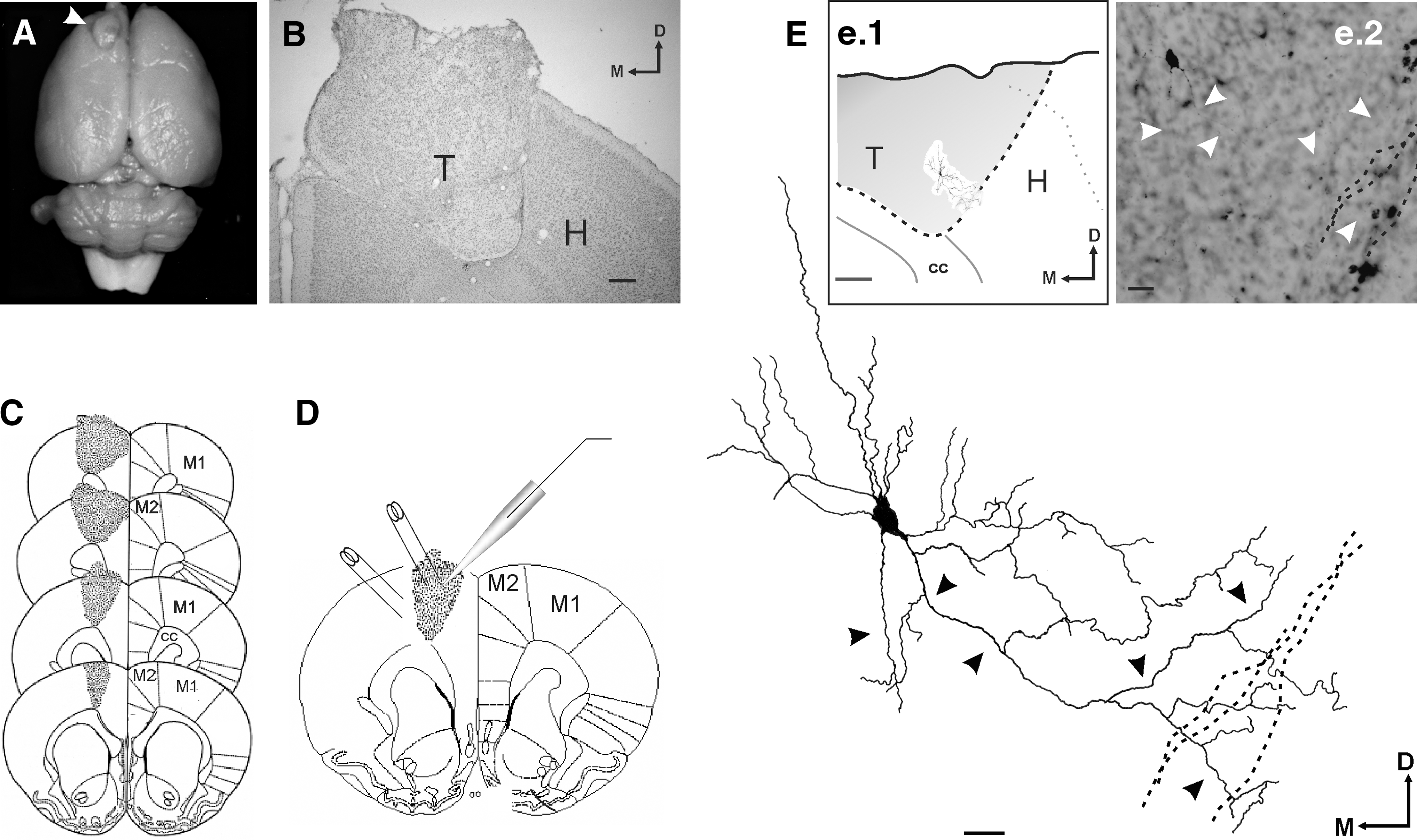
Experimental design: stimulation and recording sites in the grafted tissue. (
Slice preparation
Three months after transplantation the grafted rats (postnatal days ≥ 180) were processed to study the electrophysiological characteristics of the transplanted neurons in vitro (n = 38). The rats were deeply anesthetized with halothane gas, decapitated, and coronal slices (350 μm thick) containing the transplanted tissue were obtained as described previously (Yajeya et al., 2000). Briefly, slices were kept for at least 1 h at room temperature in aerated (95% O2 + 5% CO2) artificial cerebrospinal fluid (ACSF) before use. The ACSF was composed of 117 mM NaCl, 4.7 mM KCl, 2.5 mM CaCl2, 1.2 mM MgCl2, 25 mM NaHCO3, 1.2 mM NaHPO4, and 11 mM glucose (pH 7.3, 310–315 mOsm). For recordings, the slices were transferred to an interface-type chamber and continuously perfused with aerated ACSF.
Electrophysiological recordings
The transplant was composed of cell clusters separated by bundles of fibers (Fig. 1D). The recordings were made in these cell clusters distributed throughout the transplant. In the adjacent host cortex, recordings were made close to the transplant.
For firing properties studies, sharp electrode recordings were made (140–180 MΩ) by filling a pipette with 3 M potassium acetate and connecting it to an intracellular recording amplifier (VF180; Biologic, Claix, France). The neurons were characterized by supra-threshold depolarizing current pulses according to their firing pattern. No characterization of host neurons was made, as this was not the aim of this study.
In order to morphologically identify the neurons and recording sites in the grafts (Fig. 1E), at the end of electrophysiological recording some neurons (n = 6) were filled with biocytin (2%) in a potassium acetate solution (2 M). Positive current pulses of 0.2 nA, with a duty cycle (6 min) of 300 ms on/300 ms off, were used. After injection, the slices were transferred to an incubation chamber for 30 min and then fixed by immersion in 0.1 M phosphate buffer (PB) with 1.25% glutaraldehyde for 35 min. The fixed slices were placed in a 2% agar solution, cryoprotected in 30% sucrose in PB, and sections (45 μm) were cut using a freezing microtome (HM400R; Microm, Heidelberg, Germany). The sections were collected in PB, rinsed three times in the same buffer, and then incubated with avidin-biotin-peroxidase complex (Vector Laboratories, Burlingame, CA) for 3 h at room temperature. For visualization of the biocytin, 3,3′-diaminobenzidine was used as chromogen. The reaction was intensified with nickel ammonium sulfate. The sections were counterstained with cresyl violet to determine the position of the filled neuron in the graft. Neurons were reconstructed from serial sections. Drawings were made using a camera lucida (Labophot; Nikon, Kawasaki, Japan) for all sections that contained a part of the reconstructed neuron.
For connectivity studies, whole cell path-clamp recordings were mainly used. They were performed at room temperature at a holding potential of −70 mV. Evoked postsynaptic currents were elicited by intra-transplant or adjacent host cortex stimulation applied through a bipolar electrode (WPI TM33B10 with 1 MΩ of nominal impedance) connected to a programmable stimulator (Biologic SMP-311; Biologic) to generate single square-wave pulses of 100 μs duration and 100–500 μA intensity. Patch pipettes (5–10 MΩ) filled with 117.5 mM K-gluconate, 21.5 mM KOOCCH3, 4 mM NaCl, 10 mM HEPES, 0.2 mM EGTA, 6 mM Na2ATP, 0.3 mM Na3GTP, 2 mM MgCl2 (pH 7.2–7.3, 300–320 mOsm), were connected to a patch-clamp amplifier (Biologic RK 400; Biologic). Postsynaptic potentials were characterized according to their amplitude (as a function of the resting potential) and latency. A schematic representation of a graft showing the recording and stimulating electrode locations are represented in Fig. 1D.
Data analysis
Sharp electrode data were acquired online with the help of a CED 1401 interface (CED, Cambridge, UK), whereas patch-clamp recordings were acquired through an analog-to-digital interface (DTR-GPIB; Biologic), both sets of data were stored on a personal computer (sample frequency 12.5 kHz). All recorded currents were low-pass filtered with a cutoff frequency of 2 kHz. No compensation of capacitive transients and series resistance was performed during recordings of synaptic events. Analyses in both cases were performed using the MiniAnalysis Program, version 6.0.3 (Synaptosoft, Decatur, GA).
Chemicals
Drugs were applied using a gravity-fed system and added to the bath solution from previously prepared stock solutions stored at −20°C. Bicuculline (Tocris, Madrid, Spain) was applied to determine the presence of functional inhibitory γ-aminobutyric acid type A (GABAA) receptors in the preparation, and 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) and amino-5-phosphonopentanoate (APV) (Tocris) were added to characterize the excitatory glutamatergic responses. Carbachol, a cholinergic agonist, and atropine sulfate, a muscarinic antagonist (Sigma, Poole, UK), were used to assess the presence of functional cholinergic receptors. Finally, nifedipine and ω-agatoxin IVA (Peninsula Lab., Belmont, CA), were used to block different types of voltage-dependent calcium channels.
Immunocytochemistry and histology
In some animals (n = 9) the presence of choline acetyltransferase (ChAT) and glutamic acid decarboxylase (GAD), as well as GABAA and muscarinic acetylcholine M1 receptors in the grafted neurons were investigated. Deeply anesthetized animals were perfused through the ascending aorta with a wash solution composed of 2% dextran in PB, pH 7.4 at room temperature. This was followed by 500 mL of 4% paraformaldehyde (for ChAT), 4% paraformaldehyde/0.1% glutaraldehyde (for GAD), or 2% paraformaldehyde/2% glutaraldehyde (for GABAA and M1 receptors) in PB. After perfusion, the brains were removed from the skulls and post-fixed in the same fixative overnight at 4°C. Coronal sections cut on a vibratome or a freezing microtome at 40 μm were processed for ChAT, GAD, GABAA, or M1 receptors using the avidin-biotin procedure (Vector Laboratories). Sections were incubated overnight at 4°C in anti-ChAT monoclonal antibody (MAB5270 diluted 1:500; Chemicon, Billerica, MA), anti-GAD serum (AB5992 diluted 1:2000; Chemicon), anti-GABAA receptor, β-chain monoclonal antibody (MAB341 diluted 1:1000; Chemicon), or anti-muscarinic acetylcholine (M1) antibody receptor serum (M9808 diluted 1:200; Sigma). Six to 12 sections were analyzed per animal/antibody. Control sections were always processed without the primary antibody.
In some animals (n = 3) the morphological characteristics of the graft tissue were studied. Anesthetized animals were perfused with saline followed by 10% formalin in PB (pH 7.4). The brains were removed and post-fixed in fresh fixative at 4°C for 3 h. They were then soaked in 30% sucrose in PB for cryoprotection. Coronal sections 50 μm thick were cut on a freezing microtome. The sections were mounted on gelatin-coated slides and stained with cresyl violet for Nissl substance.
Statistics
Unless otherwise indicated the electrophysiological data are always expressed as the mean ± SEM. In all cases “n” represents the average number of neurons. Unless otherwise indicated action potentials and synaptic currents were averaged (five or more) before quantitative analysis. Statistical analysis of the data collected was performed using a paired Student's t-test, and when necessary by ANOVA test. Statistical significance was set at p ≤ 0.05.
Results
Firing properties of transplanted cells
To examine the electrophysiological properties of the transplanted neurons, intracellular recordings with sharp electrodes (n = 20) were obtained from cells in the graft. They did not present spontaneous action potentials at resting membrane potential (RMP) values (–69 ± 2 mV). The direct activation of these neurons by supra-threshold depolarizing current injection (0.1–0.5 nA) defined two main patterns of discharge: regular firing (n = 12, Fig. 2A) and burst firing (n = 8, Fig. 2B).
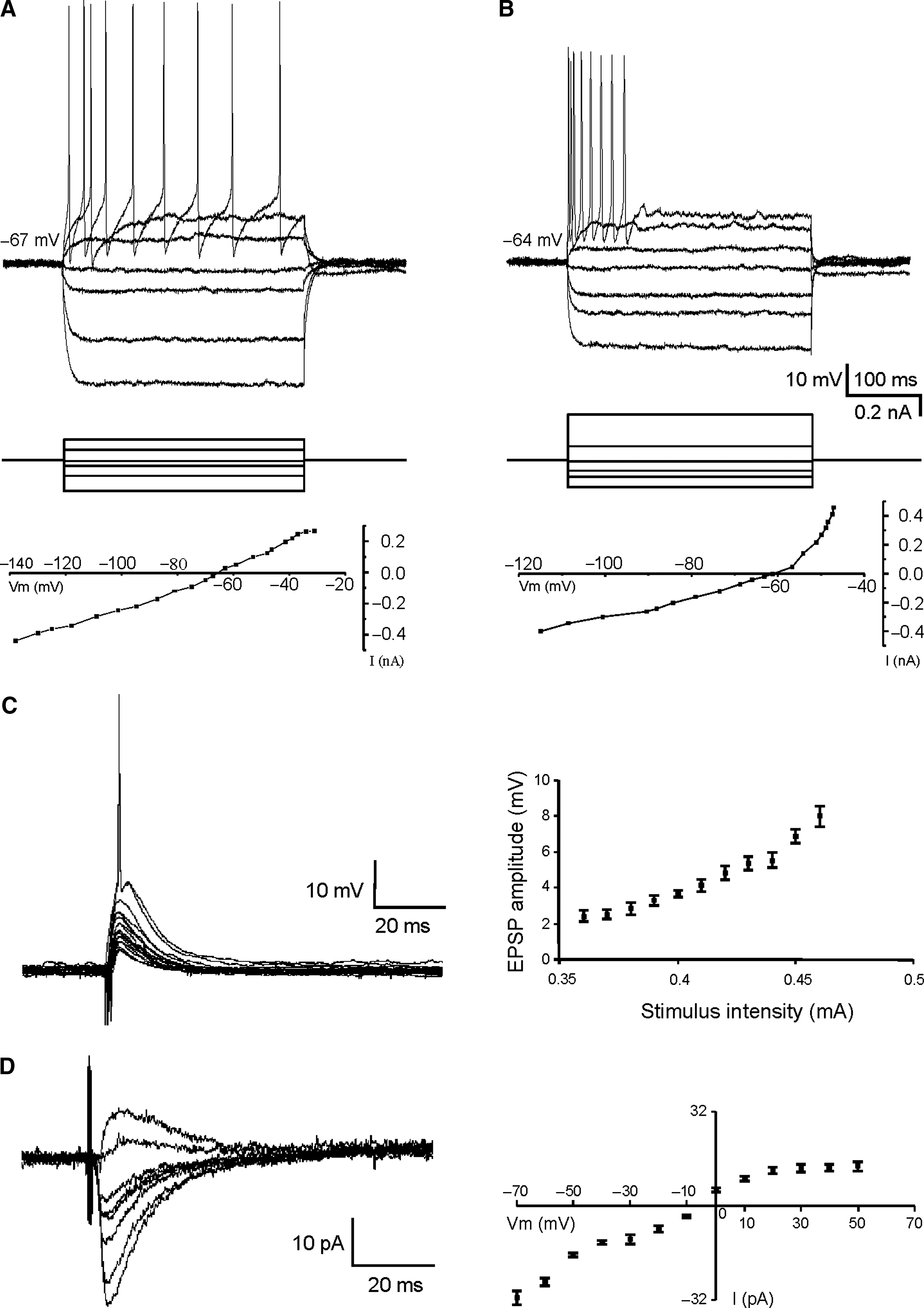
Electrophysiological properties of neurons in the graft. (
Regular firing neurons presented action potentials or a train of spikes with very slow adaptation (Fig. 2A). RMP was −71.4 ± 1.9 mV, input resistance of the neurons was 132.1 ±18.5 MΩ, the mean membrane time constant was 13.6 ± 3.4 msec, and the threshold potential was −47.9 ± 1.8 mV. The spike amplitude was 67.9 ± 3.5 mV, with a duration of 1.6 ±0.3 msec, a rise time of 0.25 ± 0.02 msec, a decay time of 0.59 ± 0.2 msec, and a half width of 0.89 ± 0.2 msec.
Burst-firing cells responded with consecutive action potentials (bursts) to the depolarizing pulse (Fig. 2B) The mean RMP was −66.1 ± 2.2 mV and the mean input resistance of the neurons was 153.4 ± 10.4 MΩ with a membrane time constant value of 6.5 ± 1.4 msec. The threshold potential was −45.2 ±2.1 mV and the spike amplitude was 56.5 ± 3.4 mV, with a duration of 2.4 ± 0.3 msec, a rise time of 0.31 ± 0.05 msec, a decay time of 0.52 ± 0.08 msec, and a half width of 0.82 ±0.13 msec.
These electrophysiological patterns of transplanted neurons are similar to those reported for some of the cellular types described in normal rat motor cortex (Degenetais et al., 2002). According to these results, fetal transplanted neurons were differentiated into at least two electrophysiological-firing-types: regular spiking and burst firing.
Intra-transplant evoked synaptic responses
In order to investigate the synaptic evoked responses on neurons in the graft, intra-transplant electrical stimulation was applied at subthreshold values. The synaptic responses evoked were recorded by using whole cell patch clamp (n = 12) or sharp electrodes (n = 10), and consisted of excitatory postsynaptic responses in all cases. This response showed a graded amplitude nature, depending on stimulus intensity (Fig. 2C) and membrane potential (Fig. 2D), suggesting a monosynaptic nature. They had a mean amplitude of 29.5 ± 1.9 pA, a mean latency of 2.1 ± 0.3 msec, a duration of 28.8 ± 20.6 msec, a rise time of 8.1 ± 3.2 msec, a decay time of 18.5 ± 9.8 msec, and a half width of 21.2 ± 9.2 msec (Fig. 2C and D).
In patch-clamp recorded neurons (n = 12) the evoked excitatory postsynaptic currents (EPSCs) displayed a rectifying voltage-current relationship that could be observed by changing the holding potential from −70 to + 40 mV, with a reversal potential between −10 and 0 mV (Fig. 2D).
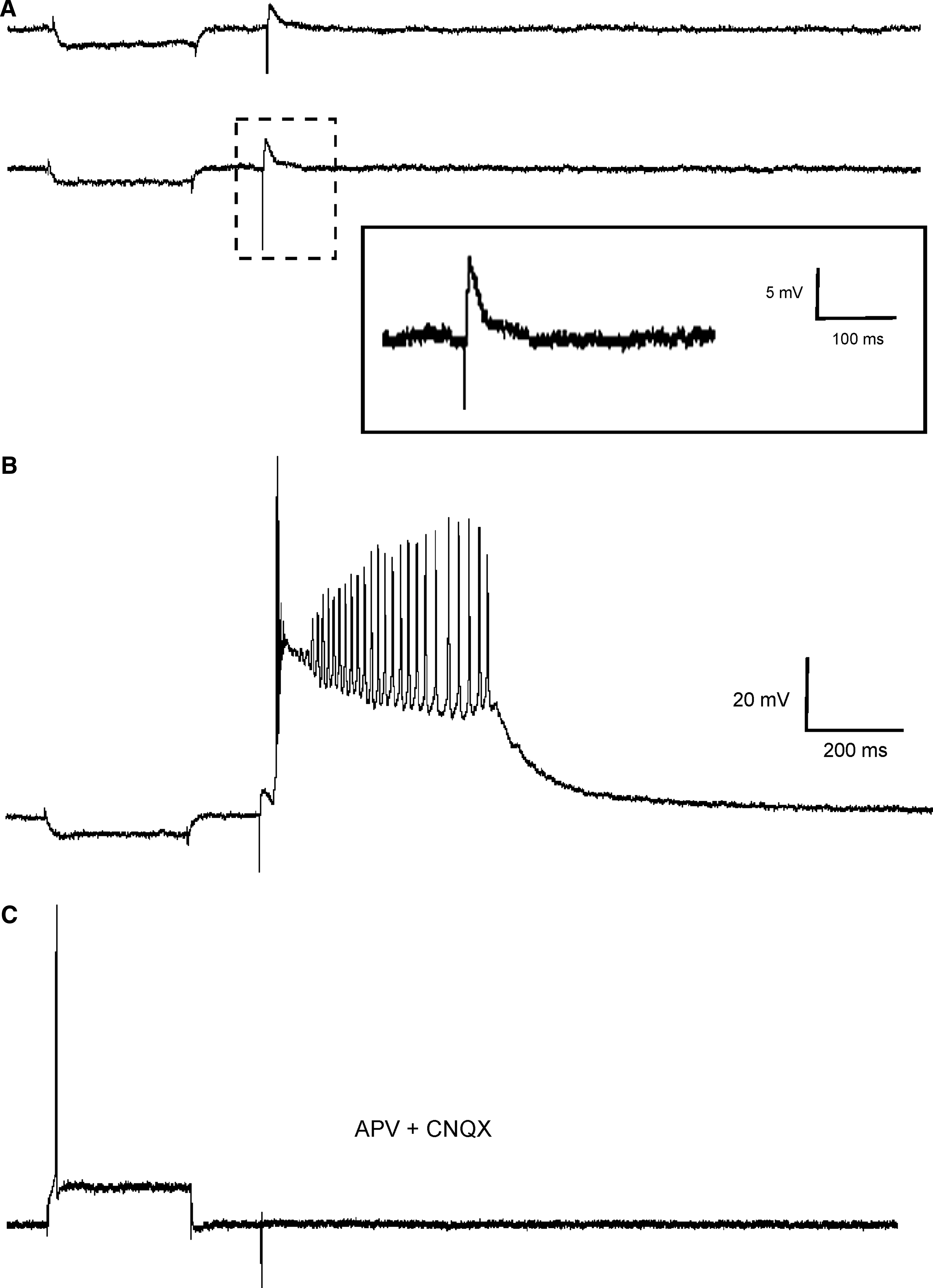
On the other hand, in 73% of the recorded cells the evoked responses consisted of excitatory postsynaptic potentials (EPSPs) that generated a single action potential when a sole stimulus reached suprathreshold (Fig. 2C). In the remaining 27% an epileptiform-like discharge (paroxysmal depolarization shift, PDS) was evoked (Fig. 3) instead of a single action potential. This response consisted of a long depolarization (0.9 ± 0.5 sec) with a variable number of superimposed action potentials, regardless of prior direct current (DC) injection (Fig. 3A, C.3, and D). The evoked epileptiform-like response was always generated by intra-transplant stimulation and never by intracellular stimulus or by adjacent host cortex stimulation (even when a high-frequency stimulus of 200 Hz and 100 msec was applied; Fig. 4E), suggesting that its substrates are the circuits formed in the transplanted tissue.
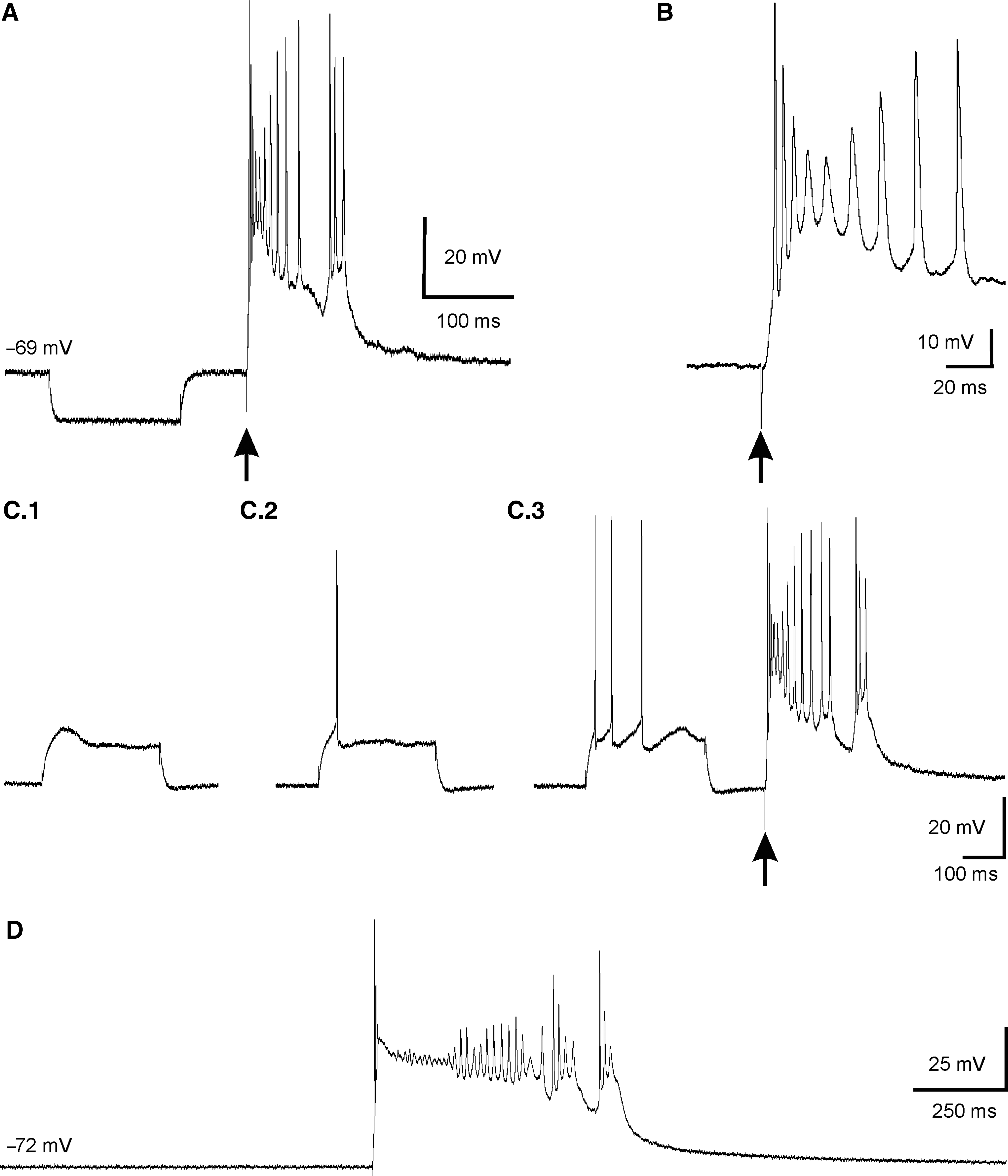
Epileptiform-like response to supra-threshold intra-transplant stimulation (paroxysmal depolarization shift, PDS). (
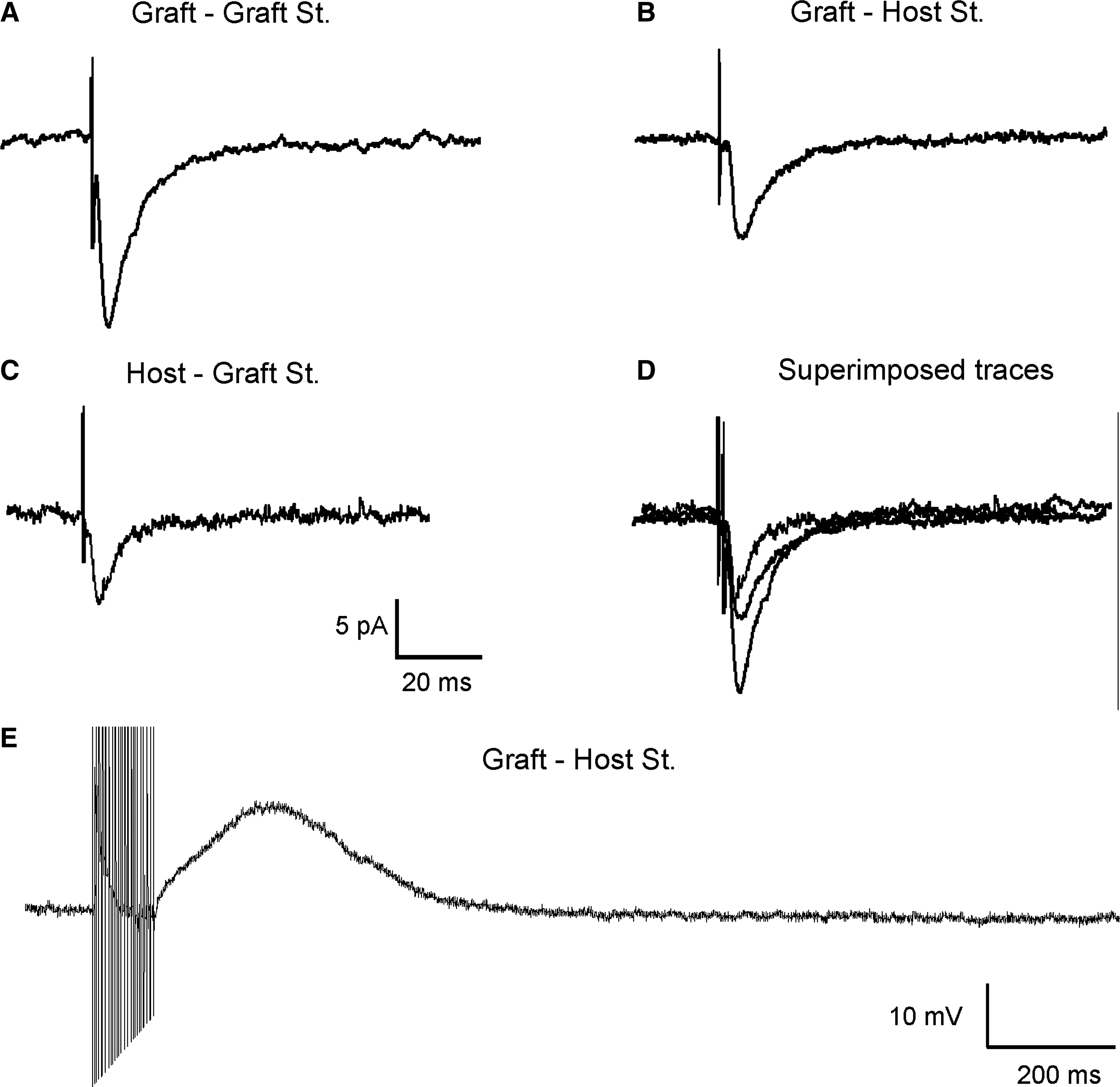
Electrophysiological characterization of the connections between the transplant and the adjacent cortex. (
Connections between the transplant and adjacent host cortex
Connections between adjacent host cortex and neurons in the graft were also explored (Fig. 4B–E). Stimulus applied in the host cortex (n = 9) 1 mm from the limit of the transplant, evoked postsynaptic responses that were always excitatory in 33% of the cases, showing successful afferent neurotransmission of transplanted neurons. The synaptic responses (Fig. 4B) had a mean latency of 1.46 ± 0.8 msec, a duration of 45.4 ± 8.9 msec, a rise time of 2.8 ± 0.6 msec, a decay time of 7.5 ± 1.3 msec, and a half width of 9.92 ± 1.8 msec. The amplitude also showed a graded nature, depending on stimulus intensity and on RMP (data not shown). Moreover, as shown in Figure 4E, train stimulation (200 Hz for 100 msec) of the adjacent cortex evoked a sustained depolarization of neurons recorded in the graft exceeding the end of the train by hundreds of milliseconds (amplitude of 15.8 ± 0.4 mV and a duration of 678.7 ± 83.1 msec), without any evident change in latency, suggesting its monosynaptic nature.
Finally, to characterize efferent synaptic signals from graft neurons to adjacent cortex, neurons in this latter area were recorded (n = 6). The graft region was electrically stimulated at subthreshold values and an excitatory synaptic response could be evoked (amplitude 7.35 ± 0.53, latency 1.6 ± 0.1msec, a duration of 19.4 ± 2.9 msec, a rise time of 1.2 ± 0.1msec, a decay time of 5.5 ± 0.3 msec, and a half width of 7.9 ± 0.4 msec; n = 1) (Fig. 4C).
Some neurons in the graft (n = 6) were iontophoretically injected with biocytin following electrophysiological recording with sharp electrodes. In some cases (n = 3) reconstruction of stained neurons allowed us to identify neuronal processes growing for hundreds of microns into the grafted tissue (Fig. 1E). Some of them could also be followed crossing the transplant limits into the adjacent motor cortex, supporting the existence of graft-host brain connections (Fig. 1E).
Synaptic transmission in the graft
In order to characterize neurotransmitters mediating the intra-transplant synaptic responses, we performed different pharmacological interventions. As shown in Figure 5, the EPSCs evoked by subthreshold electrical pulses applied intra-transplant (Fig. 5A) were only modified in duration by slice superfusion with APV (50 μM) (Fig. 5B; decay time: control: 9.42 ± 0.52 msec; versus APV: 6.59 ± 0.47 msec; p < 0.05, n = 5), but were completely removed by the application of CNQX (10 μM) (Fig. 5C). These results indicate that the EPSC evoked in neurons by intra-transplant stimulation was essentially mediated by glutamate acting on both NMDA and AMPA-kainate receptors. In addition, perfusion with CNQX (10 μM) plus APV (50 μM) also blocked the PDS response without affecting the capability of the cell to generate action potentials (n = 5; Fig. 6C). This effect suggests that the PDS response is not dependent on the intrinsic membrane properties of the transplanted cells, but depends on glutamatergic synaptic activity evoked by intra-transplant stimulation (Fig. 6). What is more, the fact that PDS was not affected by perfusion with nifedipine (5 μM) (n = 3) or ω-agatoxin IVA (100 nM) (n = 3) (data not shown) suggests its independence from calcium entering through voltage-dependent calcium channels.
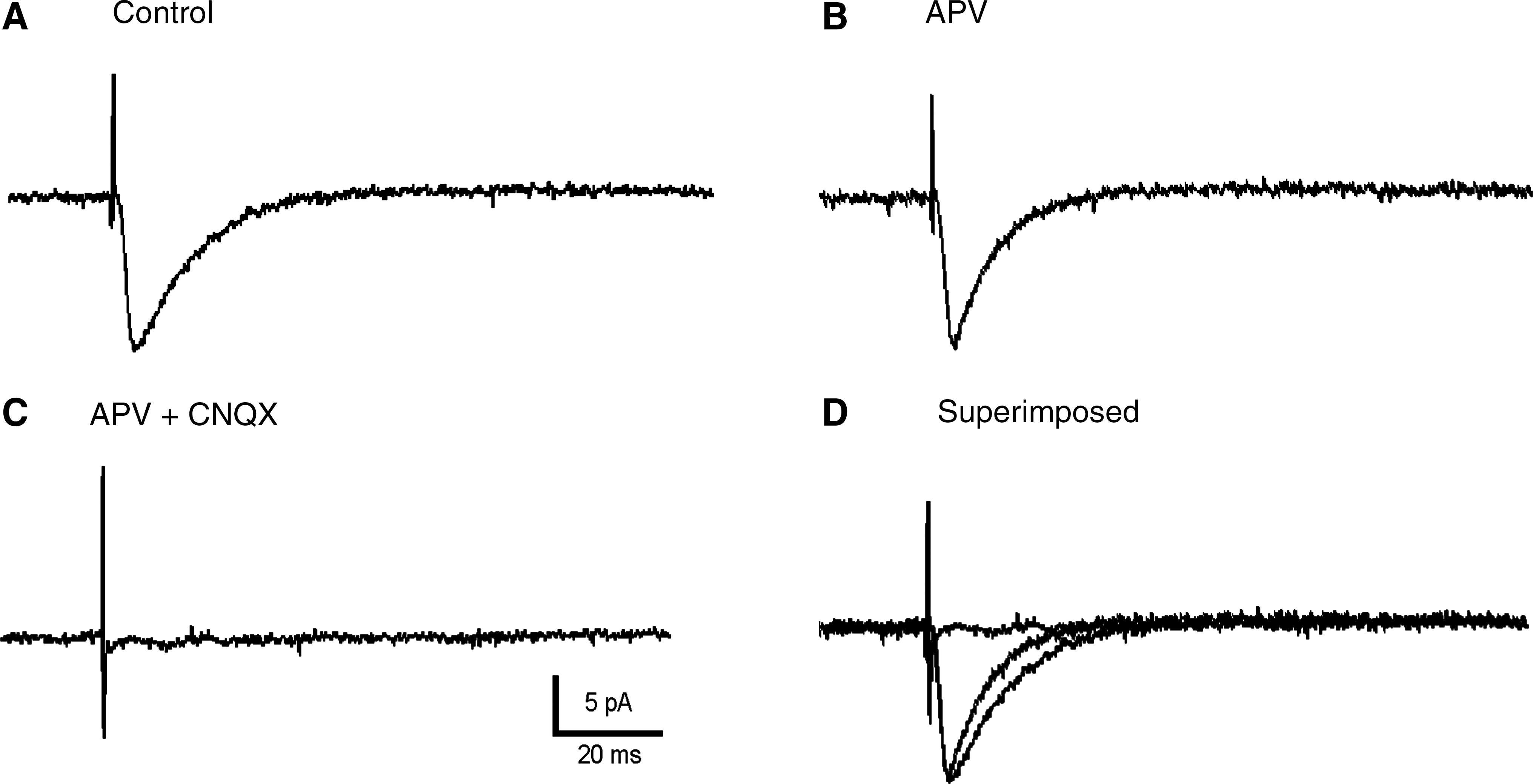
Graft-evoked synaptic responses were mediated through glutamatergic receptors. (
The epileptiform-like response showed a glutamatergic nature, acting on NMDA/non-NMDA receptors. The cell response generated by DC current followed by intra-transplant stimulation. From top to bottom: Synaptic response elicited by transplant stimulation with increased intensity. (
On the other hand, even when inhibitory postsynaptic currents were never elicited in the transplanted cells by extracellular stimulation, the application of bicuculline (10 μM) (n = 7) induced an increase in EPSC amplitude (control: 16.33 ± 0.86 pA; bicuculline: 19.17 ± 1.37 pA; p < 0.05) (Fig. 7A). In two out of seven recorded neurons treated with bicuculline, blocking GABAergic receptors induced a prolonged EPSC current, presumably mediated through activation of polysynaptic pathways (Fig. 7B). Taken together these results suggest that cells composing the transplant expressed functional GABAergic receptors.
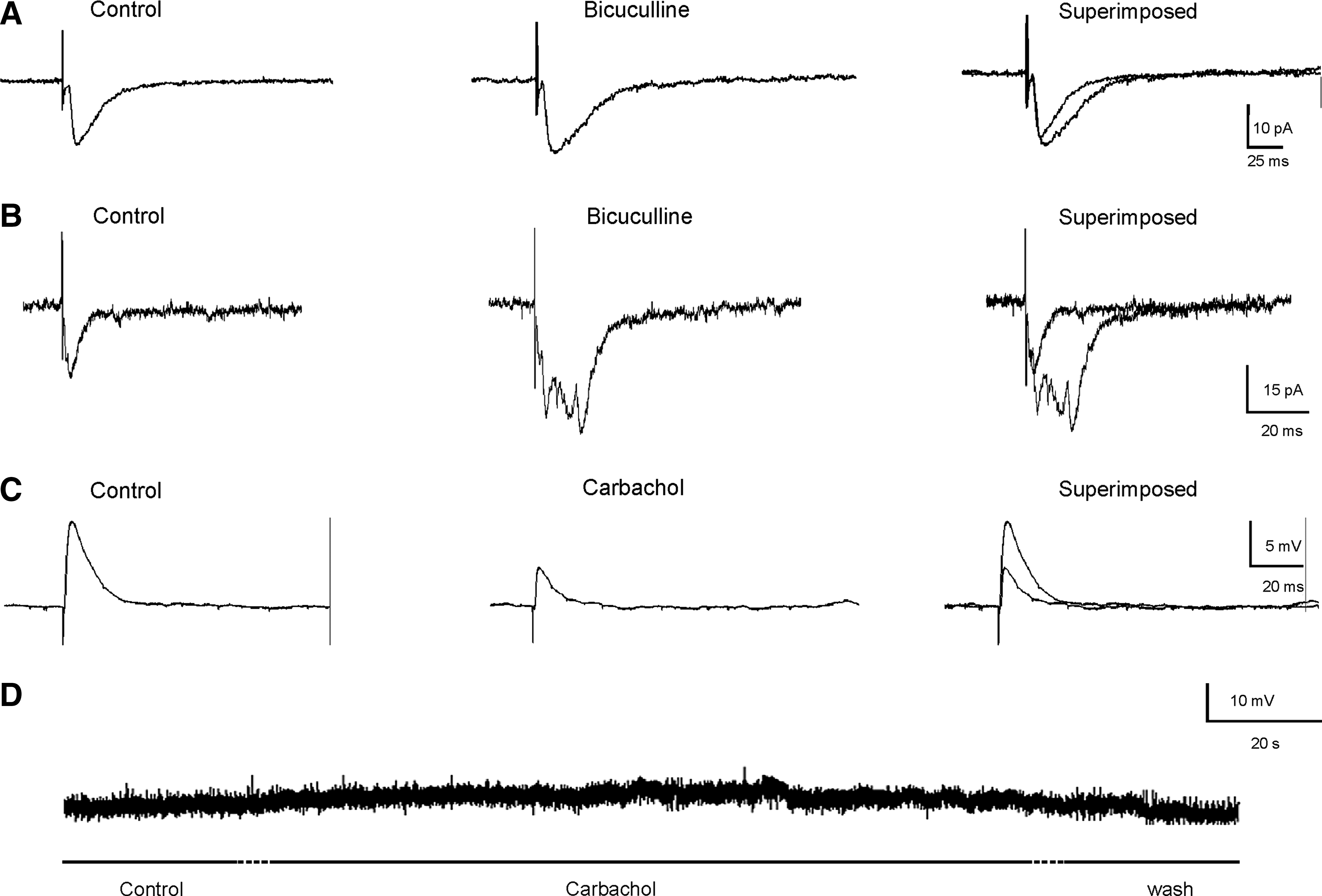
The evoked synaptic glutamatergic responses in the transplant are modulated by GABAergic and cholinergic drugs. (
In order to investigate whether the responses evoked on grafted neurons could be modulated by cholinergic drugs, carbachol and atropine were used. Superfusion of the slice with the cholinergic agonist carbachol (5 μM) was able to induce a slow-building and long-lasting depolarization of membrane potential (4.2 ± 0.31 mV; n = 5) in the recorded neurons (Fig. 7C and D). In addition, the amplitude of the excitatory response holding the membrane potential at resting values was depressed significantly (control: 10.18 ± 0.58 mV versus carbachol: 4.89 ± 0.31 mV; p < 0.001) (Fig. 7C). However, perfusion with the muscarinic antagonist atropine (5 μM) (n = 5) did not generate significant changes in the EPSC amplitude (control: 23.4 ± 0.86 pA; versus atropine: 22.4 ±0.83 pA) (data not shown). These results suggest the existence of cholinergic receptors in the graft with capacity to modulate the excitatory glutamatergic synaptic transmission, in spite of the graft not showing intrinsic cholinergic activity.
Neurotransmitter and receptor immunocytochemistry
All grafted animals presented surviving transplants, and the grafts were well embedded in the host cortical tissue. A photograph of a grafted brain is shown in Figure 1A. The cytoarchitectural features of the graft tissue were studied in sections stained with cresyl violet. In the Nissl-stained material all the transplants studied were seen to contain well-differentiated neurons of normal appearance. The transplant was composed of cell clusters separated by bundles of fibers (Fig. 1B). The characteristic stratification of the normal host cortex was not preserved within the transplant. The transplant–host interface was generally obvious and characterized by the presence of fiber laminae surrounding the transplant (Fig. 1B).
Choline acetyltransferase immunolabeling was identified in a small number of transplanted cells. Choline acetyltransferase–immunoreactive cells showed bodies of different sizes and shapes, and in some neurons dendrites radiated from the cell bodies (Fig. 8A).
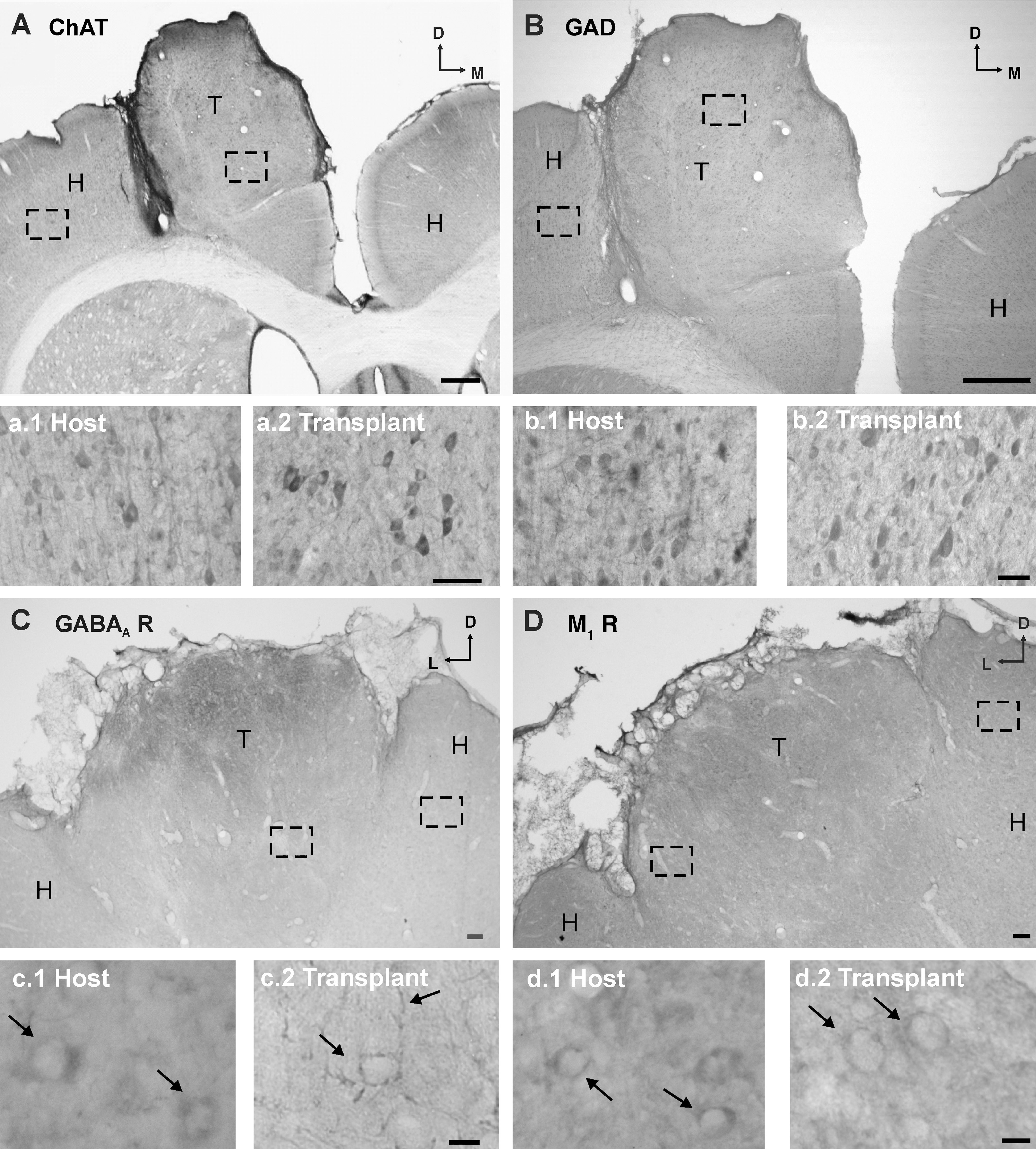
Immunolabelling of transplanted cells. (
GAD immunopositivity was identified in some grafted cells across the transplant. The cell bodies of GAD-immunoreactive cells tended to be rounded or pyramidal (Fig. 8B). Immunopositive dendrites were not consistently exhibited, but were present in some cases.
GABAA receptor immunoreactivity was identified outlining the soma and processes of some grafted neurons (Fig. 8C). Immunolabeling of muscarinic acetylcholine receptors (M1; a widely expressed receptor in the normal cortex) was present surrounding the somata of some transplanted cells. Anti-M1 antibody did not label the cell processes (Fig. 8D).
These results support the presence of GABAergic and cholinergic modulations of the glutamatergic neurotransmission as shown by the electrophysiological recordings carried out in the transplant.
Discussion
During the last century, many studies have shown the use of the grafting of tissue to the brain as a strategy to generate new neurons and glial cells capable of being integrated functionally during the repair process of the adult central nervous system (Bjorklund and Lindvall, 2000; Tuszynski, 2007). Until now, cell survival, metabolism, and connections inside the graft and between graft and host have been demonstrated by glucose uptake (Ebrahimi-Gaillard et al., 1995), neurotracer injections (Heredia et al., 1991; Grabowski et al., 1993; Garnier et al., 1997; Riolobos et al., 2001), and recently by the use of green fluorescent protein expression (Englund et al., 2002; Alvarez-Dolado et al., 2006; Gaillard et al., 2007). The physiological properties of early grafted neurons in young animals have been reported, and show that these cells can be integrated into the very young brain and used to modify or repair neural circuits (Englund et al., 2002; Wernig et al., 2004; Alvarez-Dolado et al., 2006). Nevertheless, to our knowledge, these kinds of studies in adult brain have not been reported, either for neuronal properties or connectivity of the transplant. We have previously shown, by using fetal homotopic transplants in the motor cortex area, a functional graft-dependent recovery by using behavioral studies (Riolobos et al., 2001). Very recently it has been demonstrated that, at least for cortex, there is substantial anatomical re-establishment of cortical circuitry following embryonic cortex grafting into the adult brain (Gaillard et al., 2007). Here we investigate whether the integration of transplanted cells into adults develops not only functional synaptic connectivity with the host tissue, but also electrophysiological properties that are similar to neurons previously described in the neighbor host tissue (Kawaguchi, 1993).
Our results have shown that transplanted neurons have membrane properties and firing discharges similar to neurons from normal cortex previously described in vitro and in vivo (Kawaguchi, 1993; Degenetais et al., 2002). Current depolarization characterized two different firing patterns: regular and burst firing neurons. These cell types could not be differentiated from normal cortex neurons by their electrophysiological characteristics, such as RMP, spike amplitude, or input resistance. Regular-spiking and burst cells have been reported in the rat frontal cortex (Chagnac-Amitai et al., 1990). Moreover, cortical cells that show membrane-rectifying properties and all-or-nothing burst discharge to depolarizing current pulses are present in sensory-motor cortical slices (Connors et al., 1982; Degenetais et al., 2002; Cho et al., 2004). Differences displayed by regular-spiking and burst neurons in the current-voltage (I-V) relationship could be due to different expression of receptor populations (e.g., AMPA receptors). Recently it has been shown that an immune response from TNF-α after the trauma or transplantation might remodel the AMPA receptor population from GluR2-containing to GluR2-lacking (Leonoudakis et al., 2008). This would likely change the I-V curve from linear to inwardly rectifying. Furthermore, regular-spiking and burst discharge action potentials generated by intracellular current injections have also been described in neurons within neocortical ectopias (Gabel and LoTurco, 2001). All recorded neurons were able to generate action potentials when positive current injection was applied. Hence, our results suggest that, at this time point, the transplanted cells had reached a degree of maturation similar to that of host neurons described in the literature (Kawaguchi, 1993; Degenetais et al., 2002), and may survive and behave like normal cortical cells.
The fact that electrical stimulation of adjacent host cortex elicited excitatory synaptic responses in grafted cells (Fig. 4B and E) suggests the presence of functional connections between the host and the transplant. The same observation can be made for the connections from the transplant to the adjacent host cortex. These results are supported by the anatomical evidence previously reported using different tracing methods (Heredia et al., 1991; Grabowski et al., 1993; Riolobos et al., 2001; Gaillard et al., 2007), and our current data on biocytin-labeled grafted neurons (Fig. 1E). The lack of evoked responses in some cases when studying host-transplant connections could be due to the sectioning of fibers during slicing procedures.
Evoked responses were mediated by glutamic acid, because perfusion with NMDA and AMPA-kainate receptor blockers completely removed the response. These results indicate that glutamate is the main excitatory neurotransmitter in the transplant, in agreement with previous reports (Englund et al., 2002).
Inhibitory GABA-mediated transmission in grafted neurons from young transplanted animals has been previously described (Englund et al., 2002; Alvarez-Dolado et al., 2006). In our experiments, none of the evoked synaptic responses was inhibitory, although GABA immunocytochemistry showed a number of GABA-positive grafted neurons in the homotopic transplants. Our results could be attributable to two factors. First, the fact that during the development of cortex, GABA-expressing cells migrate from the subcortical telencephalon into the neocortex. This migration occurs around the 17th day of rat gestation (Anderson et al., 1997; Xu et al., 2004), later than when we obtained our embryonic transplanted cells. Second, the decline of GABA cells in neocortical transplantation (Bragin et al., 1991). However, functional GABAergic receptors capable of regulating the evoked synaptic responses in the grafted neurons were present. Perfusion of the slices with bicuculline (a GABAA blocker) significantly increased the amplitude and duration of the excitatory synaptic response, suggesting the presence of functional GABAergic receptors in the synaptic complex. In addition, GABAA receptors were identified in the graft by immunocytochemistry. Similar synaptic changes to exogenously applied GABAergic drugs have been described in neocortex (Sutor and Luhmann, 1995; Englund et al., 2002; Alvarez-Dolado et al., 2006) and that quality seems to be conserved in homotopic cortical grafts in adults. Furthermore, when GABAergic receptors were blocked, some recorded neurons presented prolonged evoked responses with several peaks. This fast polysynaptic response suggests that in the transplant, some cells can be hyper-activated as a consequence of blocking inhibition.
In some cases, suprathreshold stimulation elicited synaptic responses described as epileptiform-like discharges, also called paroxysmal depolarization shift (PDS), which may be related to an imbalance between the excitatory and inhibitory neurotransmitters. Although immunocytochemistry demonstrated the presence of GABAergic cells, their functional role to modulate excitatory activity seem to be exiguous. PDS discharge in presence of GABAergic receptor antagonists has also been described for neocortical brain slices (Schiller, 2004), in neurons from neocortical ectopias (Gabel and LoTurco, 2001), or grafted neurons in young animal cortex (Englund et al., 2002). It has been proposed that in the neocortex PDS response may be mediated by calcium-activated non-specific cation current, since specific blockers for this current were very effective (Schiller, 2004). However, PDS responses evoked in neurons from cortex ectopias were mediated by NMDA-receptor activation (Gabel and LoTurco, 2001). We have shown that PDS in transplanted neurons was not affected by voltage-dependent calcium channel blockers, whereas it could be removed with glutamatergic-receptor antagonists. Taken together, imbalance of inhibitory circuitry, organization of cells in clusters within the graft, and the existence of NMDA calcium-permeable receptors may be factors involved in the generation of intra-transplant epileptiform discharges (Gabel and LoTurco, 2001; Englund et al., 2002; Schiller, 2004).
Furthermore, carbachol modulated the evoked responses and RMP, suggesting a plausible double location for acetylcholine receptors as in other cortical or brainstem structures (Yajeya et al., 2000; Navarro-Lopez et al., 2004). In the presynaptic terminal they would adjust the amount of neurotransmitter to be liberated, and postsynaptically they would control membrane excitability. The enzyme choline acetyltransferase was found in transplanted cells, suggesting that these cells are able to synthesize acetylcholine. Also muscarinic M1 receptors, very common cholinergic receptors in the cortex, were positively immunolabeled in the grafted neurons. Our results support a cholinergic participation in transplant functionality, a concept in agreement with the idea of acetylcholine being specifically involved in the process of behavioral recovery induced by homotopic cortical transplants (Miranda et al., 1997).
Our group has previously shown that homotopic cortical grafted animals established connections from the graft to the host tissue (Riolobos et al., 2001). Furthermore, in the present study we found grafted neuronal processes crossing the graft-host interface (Fig. 1E). Recently the anatomical re-establishment of damaged adult motor pathways by grafted embryonic cortical neurons has been shown (Gaillard et al., 2007). Other authors have reported that in young grafted animals, neuronal precursors can modify levels of electrophysiological activity in the host brain by establishing functional synapses with native neurons (Englund et al., 2002; Alvarez-Dolado et al., 2006). Thus much evidence seems to indicate that transplantation of tissue can lead to the establishment of functional connections, and could be used as a cellular vector to deliver therapeutic molecules to wide regions of the brain. Even more, it has been proposed that grafted tissue could lead to the appropriate reconstruction of damaged circuitry in the adult brain. Our demonstration that transplanted cells develop into normal cortical cells that express glutamatergic, cholinergic, and GABAergic receptors are concomitant with an imbalance between excitatory and inhibitory activity in the transplant. However, this latter fact, which has also been demonstrated in young grafted animals (Englund et al., 2002), does not seem to affect functional graft-dependent recovery (Plumet et al., 1993; Riolobos et al., 2001).
In summary our results support the hypothesis that development and differentiation of transplanted cells appear to be necessary for the functional integration of the graft into the damaged host brain. In this study we have shown that cells transplanted into adult damaged host brains establish interconnections and develop into normal cortical cells, which suggests that both phenomena could be part of the substrate underlying the functional graft-dependent recovery that has been previously described (Plumet et al., 1993; Riolobos et al., 2001). In any case, this study furthers our understanding of how transplanted cells integrate and fail to integrate, but further studies involving labeled transplanted cells are needed to establish a relationship between anatomical evidence (Gaillard et al., 2007) and functional implications for graft integration following brain injury.
Footnotes
Acknowledgments
This work was supported by grants FIS-03/0907 (Spanish Ministerio de Sanidad y Consumo, MSC), BFI 2003-01716 (Spanish Ministerio de Educacion y Ciencia, MEC), Red de Terapia Celular 2007–2010 (JCyL, Spain), Mapfre 2007 and Proyecto Jóvenes Investigadores 2007, and USAL2008A13 (University of Salamanca, Spain). J.S.-T. was a Ph.D. fellow from the Agencia Española de Cooperación Internacional (AECI). L.J.-D. is a Juan de la Cierva fellow from Spanish MEC (JCI-2005-1775-25) and J.N.-L. is a postdoctoral fellow from Spanish MSC (CD06/00175). We acknowledge Prof. Delgado-Garcia for helpful comments, the editorial help of G.H. Jenkins, and the technical assistance of Noelia Gonzalez.
Author Disclosure Statement
No conflicting financial interests exist.
