Abstract
(−)-Epicatechin (EC), a bioactive compound found in the plant kingdom contained in beans from the cacao tree, and other vegetables, has high flavonoid content. Traditionally consumed as chocolate, presents various benefits in human health. Many flavonoids have demonstrated therapeutic effects against osteoporosis by enhancing bone density; however, the specific impact of EC on bone regeneration remains unknown. Since bone regeneration involves a small group of responsive stem cells, we investigated the flavonoids’ effects on key bone precursors like mesenchymal stem cells (MSCs). We used a conventional osteogenic medium alone or combined with different EC concentrations. Our study revealed that low concentrations of EC could enhance the expression of osteogenic genes when combined with low concentrations of osteogenic medium and induced the highest differentiation of human MSCs compared with pharmacological concentrations. So, the increase in the expression of osteoblastic marker genes such as bone morphogenetic protein 2, Runt-related transcription factor 2, and secreted protein acidic and cysteine rich agreed with an increase in cellular calcium deposits. Therefore, we conclude that the natural flavonol (−)-EC improved the expression of genes involved in bone regeneration and could potentially serve as an adjunctive therapy for bone loss diseases.
INTRODUCTION
A bioactive compound, (−)-Epicatechin (EC) is prevalent in numerous species of the plant kingdom including cacao a seed that provides chocolate a product commonly ingested by humans due to its abundant flavonoid constitution. 1,2 It is of particular interest to investigate if flavonoids could be useful as a complement to regenerative therapies. 3 Some research proposes that patients with osteoporosis who include flavonoids in their diet augmented their bone density, but it has not been established specifically how to use these bioactive molecules in bone health. 4
Flavonoids are polyphenols, and within this group we can find (−)-EC, which is seen in significant concentration in food like chocolate or green tea and others 5 that produce positive effects in the prevention of different diseases such as heart attacks, 6,7 diabetes, 8 or osteoporosis. Early studies have proven that flavonoids such as catechin, 9 epigallocatechin, epigallocatechin gallate, and epigallocatechin 3-gallate 10 had an osteogenesis effect over mesenchymal stem cells (MSCs). At the same time, diverse investigations have proven that older women who frequently consume green tea plant-based foods, such as apples, berries, and cocoa, have major bone density. 5,9 –12 This has been attributed to the antioxidants of polyphenols present in several plants and nature products (as cacao) but other phytochemical properties remain unknown. 13 –17
Osteoporosis, a chronic condition characterized by bone tissue loss and increased fracture susceptibility by microarchitecture deterioration, poses significant challenges in treatment due to adverse effects becoming a public health topic. 18 –23 Several in vitro investigations in which bone differentiation is induced in MSCs 24 –27 traditionally uses commercial osteogenic mediums that includes in their formula some agents such as dexametasone, ascorbic acid, and others that for humans represent elevated pharmacological concentrations, which are considered high risk due at their long term on the health. 28
Since the above is a great limitation for therapy, the major reason of this work was to explore the effect of natural factors derived of plants (like flavonoids), in specific EC on osteoblasts differentiation as part of the investigation of its phytochemical properties compared with commercial mediums.
Some investigations have demonstrated osteogenic effects of flavonoids such as catechin and epigallocatechin on MSCs. 29 The specific impact of (−)-EC remains unclear. Conventional poliphenols used for MSC differentiation often induce cell death, limiting their clinical utility. 30 On the contrary, osteogenic mediums are used exclusively for in vitro research not for humans. Therefore, our study aimed to assess the effects of (−)-EC alone or in combination with osteogenesis medium on MSC cultures derived from human bone (MSC-hBM).
The significance of (−)-EC lies in its classification as a flavanol and includes the following compounds: catechin, EC, epigallocatechin, EC gallate, and epigallocatechin gallate. 31 Its antioxidant and anti-inflammatory properties, particularly in cocoa and dark chocolate, highlight its potential therapeutic benefits. 32 –35 Phytochemicals in food, as (−)-EC, modulate reactive oxygen species formation, thereby activating osteogenesis 24 by augment genes as BMP2, SPARC, and RUNX2. 36
Moreover, (−)-EC’s phytochemical properties are remarkable, given its classification as a flavanol and its abundance in plants such as Vitis vinifera, Camellia sinensis, and Theobroma cacao. 37 Our study used (−)-EC ≥90% purity, structurally identical to naturally occurring sources, including cocoa combined with low concentrations of osteogenic medium to obtain mixes that could be useful in therapeutic administration.
On the contrary, MSCs are a heterogeneous multipotent cellular group with the capacity to differentiate into diverse lineages such as osteoblast. 38,39 Therefore, this is proposed as an adequate model to elucidate the osteogenesis mechanisms related to polyphenols. 10,40 –42 However, it is not known yet if (−)-EC is capable of inducing osteogenesis differentiation by itself and the involved mechanism(s). Even though several antioxidants have been used to induce MSC differentiation, they also produce cell death, rendering them unsuitable for their use in patients. 43 –46 That is why in this work we employed (−)-EC alone or combined with osteogenesis medium in MSC-hBM of healthy patients to assess its effects.
Due to the significant limitations in current therapies, the main objective of this study was to evaluate the effects of (−)-EC—alone or in combination with osteogenic medium—on the osteoblastic differentiation of hBM-MSCs. This work forms part of the broader investigation into the phytochemical properties of dietary plant-derived natural compounds, such as flavonoids, in promoting bone regeneration.
RESULTS
Characterization of MSCs
Characterization of morphology of MSCs
MSC-hBM were characterized using the protocol established by the International Society for Cellular Therapy (ISCT). 47 MSC-hBM displayed adherence to the culture surface, forming organized fibroblast-type colony-forming units. Over successive passages (Passages 1, 2, and 3), the cells gradually formed a monolayer and adopted a spindle-shaped morphology as confluence increased (Fig. 1a–d, respectively). This morphological transition is indicative of the typical characteristics observed in MSC-hBM cultures during expansion.
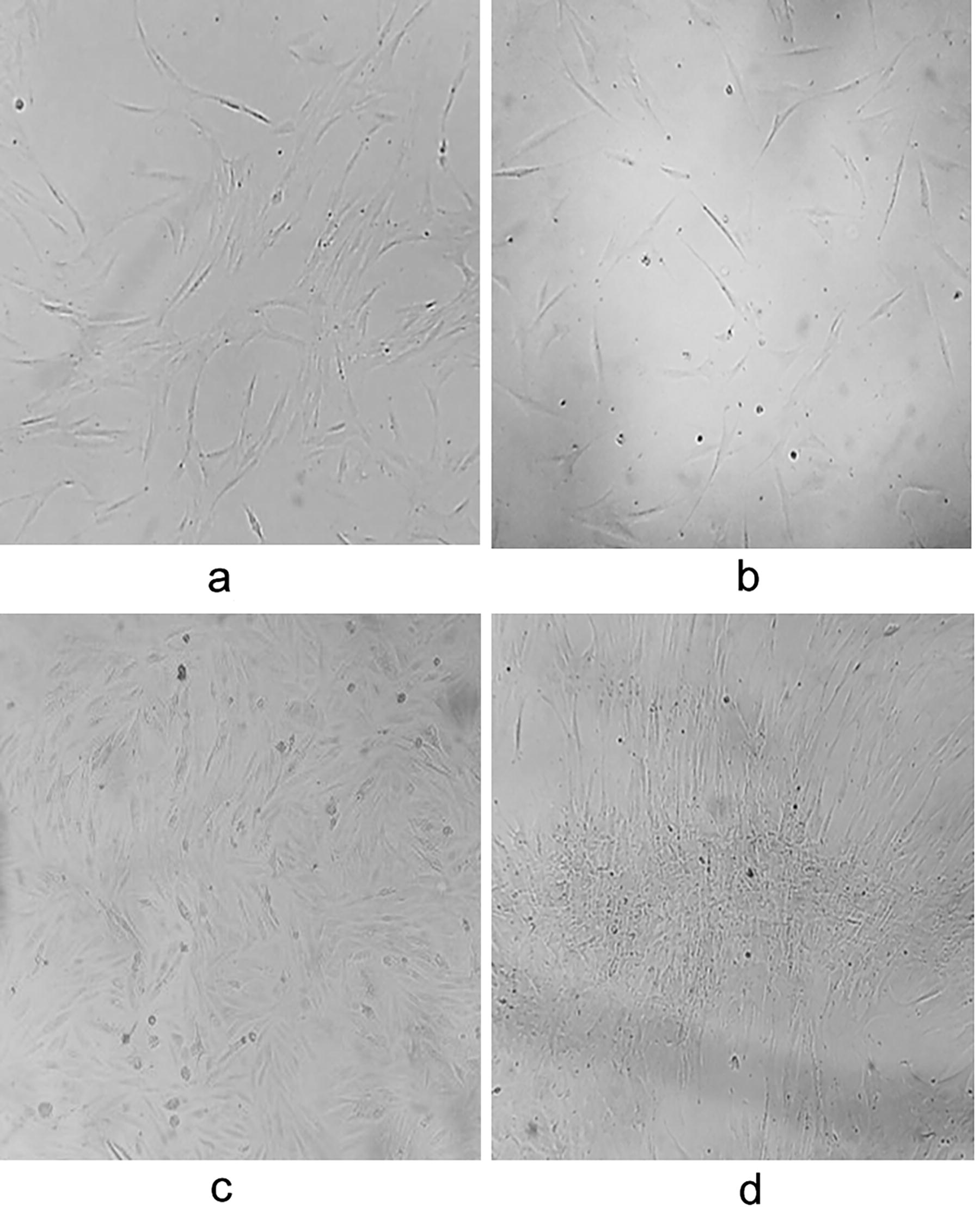
Isolated cells from human bone marrow exhibit elongated morphology with prominent and centrally located nucleus, resembling fibroblasts, which are characteristic of mesenchymal stem cells. The fibroblast-like shape persisted in our cultures up to passage 3:
Plasticity assays for MSCs
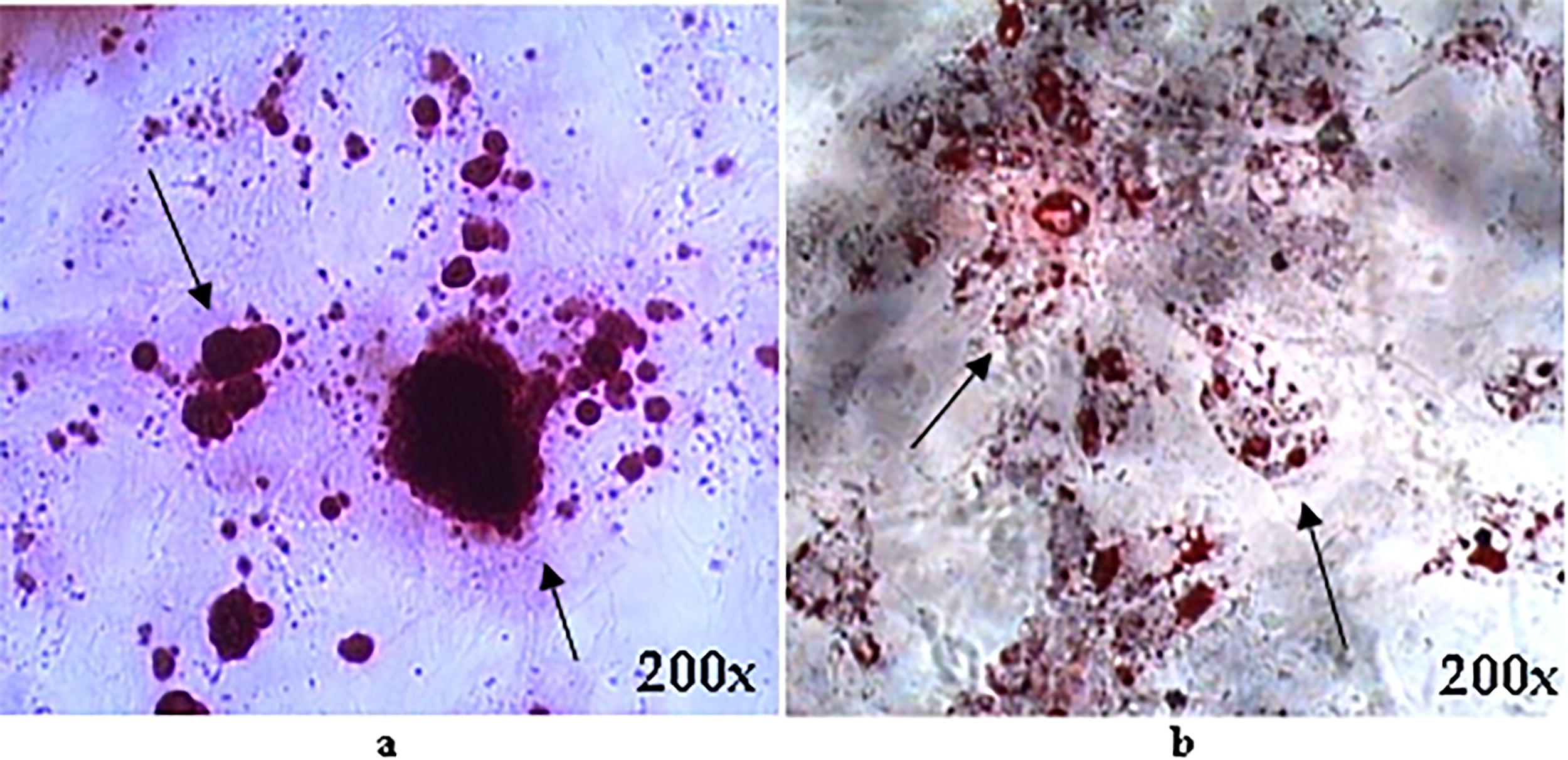
MSC-hBM were subjected to differentiation assays to investigate their plasticity. Osteogenic differentiation was induced using OSTEO, a medium rich in osteogenic-promoting factors, while adipogenic differentiation was induced using adipogenic medium, over a 15-day period. Following differentiation, cells were stained with alizarin red and Oil Red O (ORO) to visualize calcium deposits indicative of osteogenic differentiation and lipid vesicles characteristic of adipogenesis, respectively. Microscopic examination of stained cells revealed distinct outcomes: calcium deposits were observed in cells undergoing osteogenic differentiation (Fig. 2b), while lipid vesicles were prominent in cells undergoing adipogenic differentiation (Fig. 2a). These findings demonstrate the multipotent nature of MSC-hBM, capable of differentiating into various cell lineages and so highlight their promising potential for applications in regenerative medicine.
Molecular markers of MSCs by flow cytometry
In our study, we followed the recommendations set by the ISCT to identify and characterize MSC-hBM. Employing a panel of positive and negative cell surface markers, we conducted flow cytometry analysis across three passages of culture to ensure appropriate characterization. Figure 3 illustrates the different areas analyzed within the total bone marrow population. Evaluation of all samples revealed consistent positivity for the expression of the following markers: CD13, CD73, CD90, and CD105, with low expression observed for CD14, CD34, CD45, and HLA-DR (Table 1). Although percentages varied between different passages, indicating some degree of variability, this comprehensive characterization confirms the suitability of the MSC-hBM population for subsequent experimental investigations and underscores their potential for various therapeutic applications in regenerative medicine.
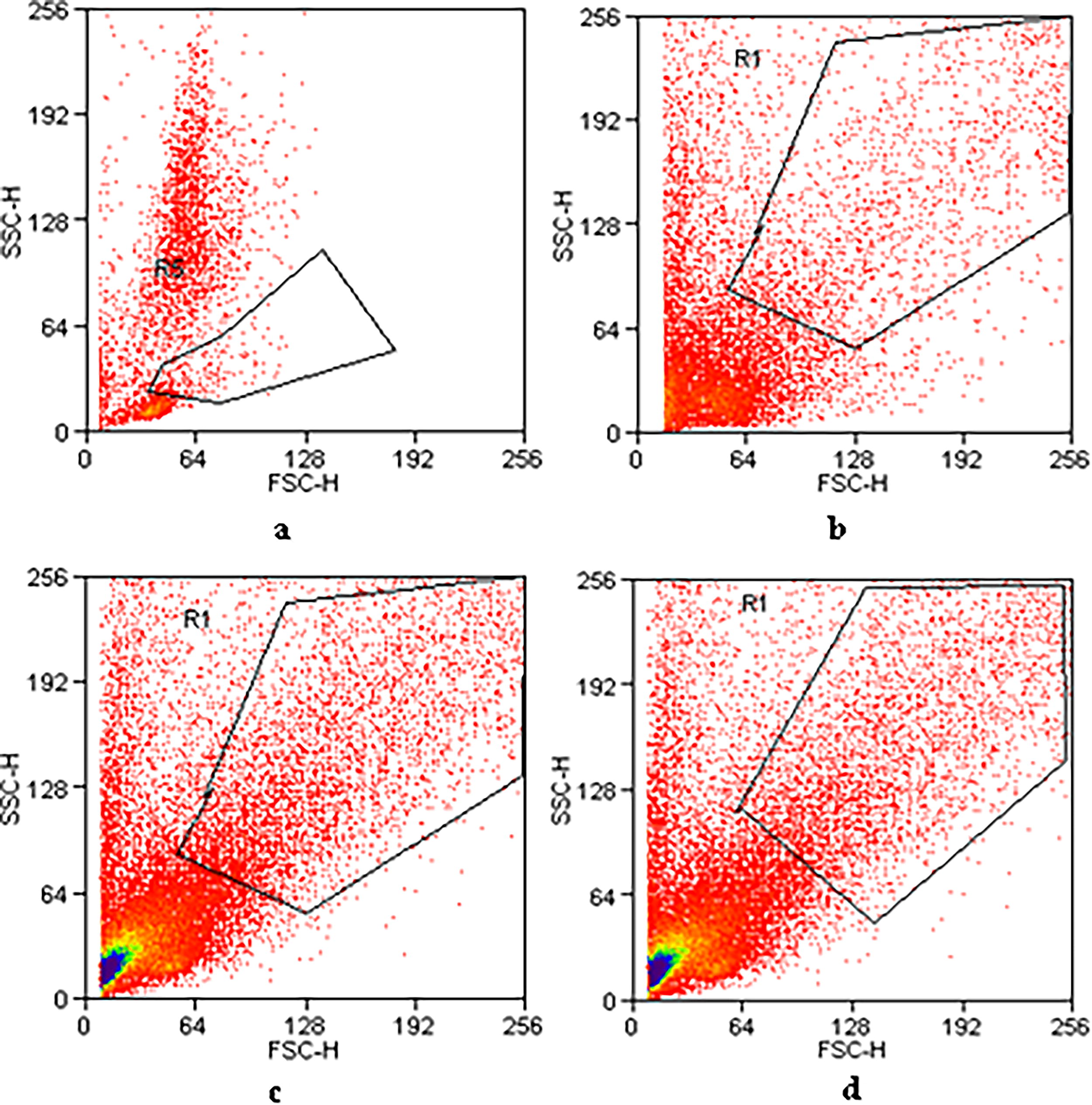
Flow cytometry analyses were performed on the selected area, showing the cell population studied for mesenchymal and hematopoietic markers in total bone marrow
Percentage of Mesenchymal and Hematopoietic Markers of Cellular Populations of MSC-hBM (BM) in Different Cell Culture Passes
Effect of the EC on the viability of MSC-hBM
To assess the impact of EC on the proliferative capability and viability of MSC-hBM, cultures were treated with concentrations of 1, 10, and 100 μM of EC for 72 h. After this period, cell proliferation was evaluated using crystal violet (CV) staining, while cell viability was determined via 3 (4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Finally, absorbance was measured and the results of both techniques were calculated just in a ratio that represents the real effect of EC in the cell population and mitochondrial activity of the cells (viability). Figure 4 shows no significant differences in the proliferation/viability of the cells in all concentrations used. Therefore, these concentrations were saved and later were used in combination with commercial mediums of differentiation (OSTEO).
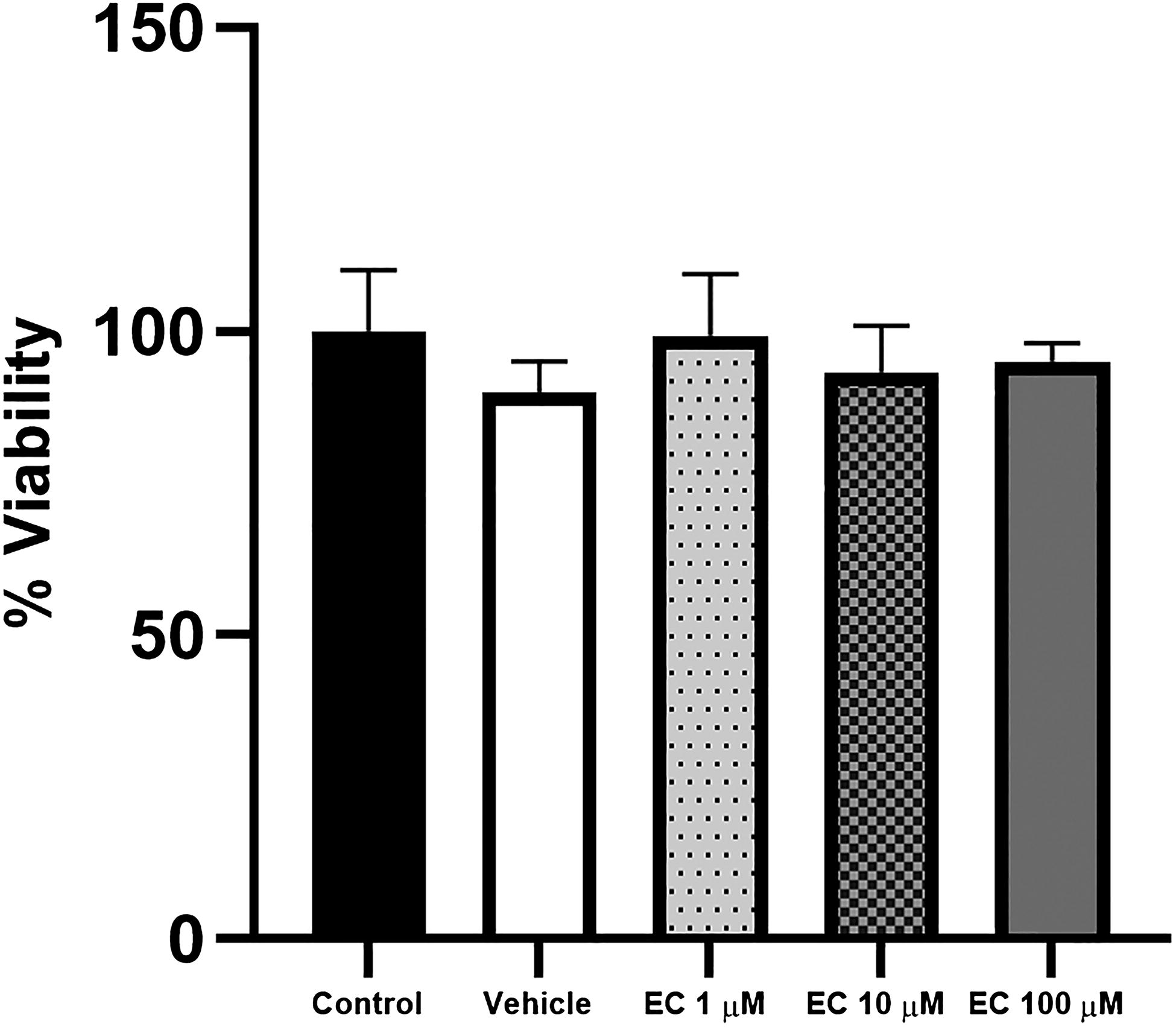
Effect of epicatechin (EC) on the viability of MSC-hBM. MSC-hBM cultures were treated with 1, 10, and 100 µM of EC for 72 h. Cell viability was determined using the MTT assay. No significant differences in cell viability were observed between the control and EC-treated groups.
EC-induced ostogenic differentiation markers
After observing that EC at various concentrations (1, 10, and 100 μM) did not induce toxic effects, we proceeded to investigate its effects on the gene expression of osteogenesis molecules: secreted protein acidic and cysteine rich (SPARC), transcription factor related to runt 2 (RUNX2), and bone morphogenetic protein 2 (BMP2), in comparison with expression induced by a commercial osteogenic medium (OSTEO, see Fig. 5a and b). Results from agarose gel electrophoresis demonstrated that the housekeeping gene expression remained constant under all cell culture conditions without any noticeable changes (Fig. 5a), serving as a reliable reference gene for comparison. EC at concentrations of 1 and 100 μM induced higher gene expression of the osteogenic molecule RUNX2 compared with the OSTEO medium. SPARC also exhibited elevated expression levels, although it did not reach the same level of expression as observed with the OSTEO medium (Fig. 5b). Unexpectedly, at a concentration of 10 μM, EC only induced a modest increase in the expression of RUNX2, ∼0.5-fold compared with the OSTEO medium containing differentiation factors.
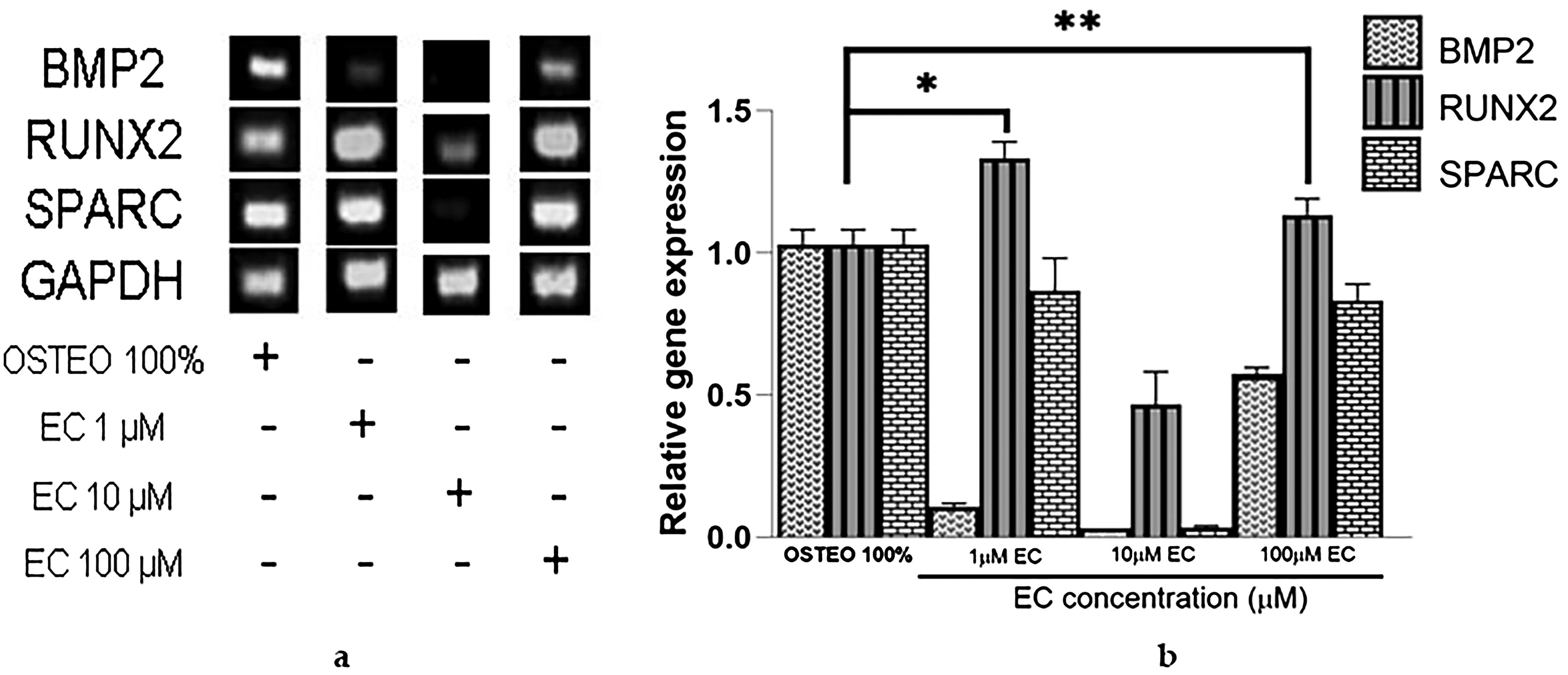
Since EC promoted high expression of RUNX2 and SPARC at a concentration of 1 μM but not at 10 μM, we tested both concentrations in combination with three different dilutions of osteogenic medium to investigate if the combination could improve BMP2 gene expression compared with their separate application. Figure 6 shows that combining 33% OSTEO with 1 μM EC did not result in any changes. Surprisingly, BMP2 gene expression increased more than twofold when 33% OSTEO was combined with 10 μM EC, exceeding the expression levels induced by 33% OSTEO alone and even reaching levels not observed with 10 μM EC applied independently. Following this, we used 50% OSTEO in combination with 1 and 10 μM EC. Figure 7 shows a higher BMP2 expression when combined with 1 μM EC. It became evident that when OSTEO and EC are present at higher concentrations, the inhibitory effect on BMP2 expression is stronger compared with their separate use (Fig. 6).
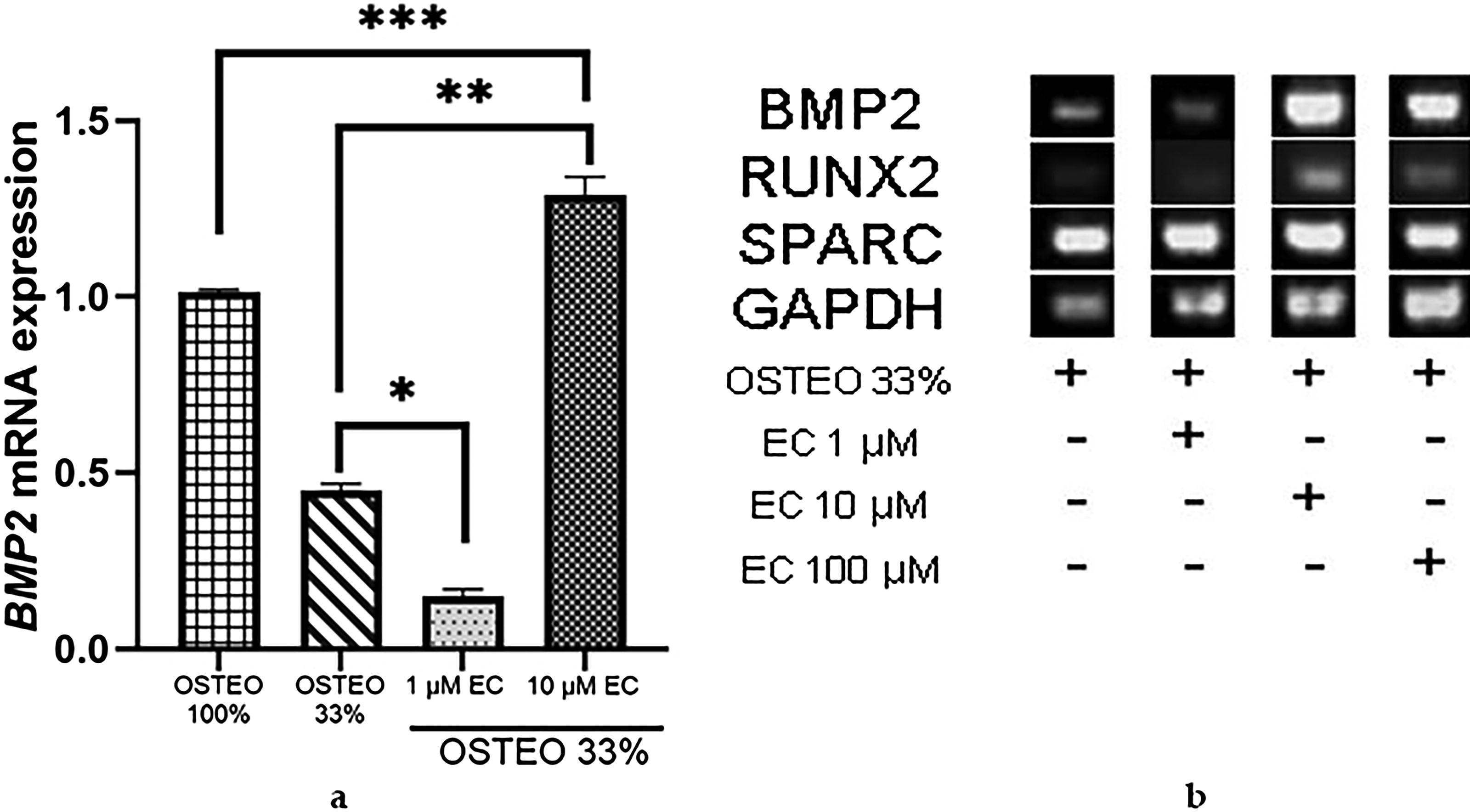
Effect of EC combined with 33% OSTEO.
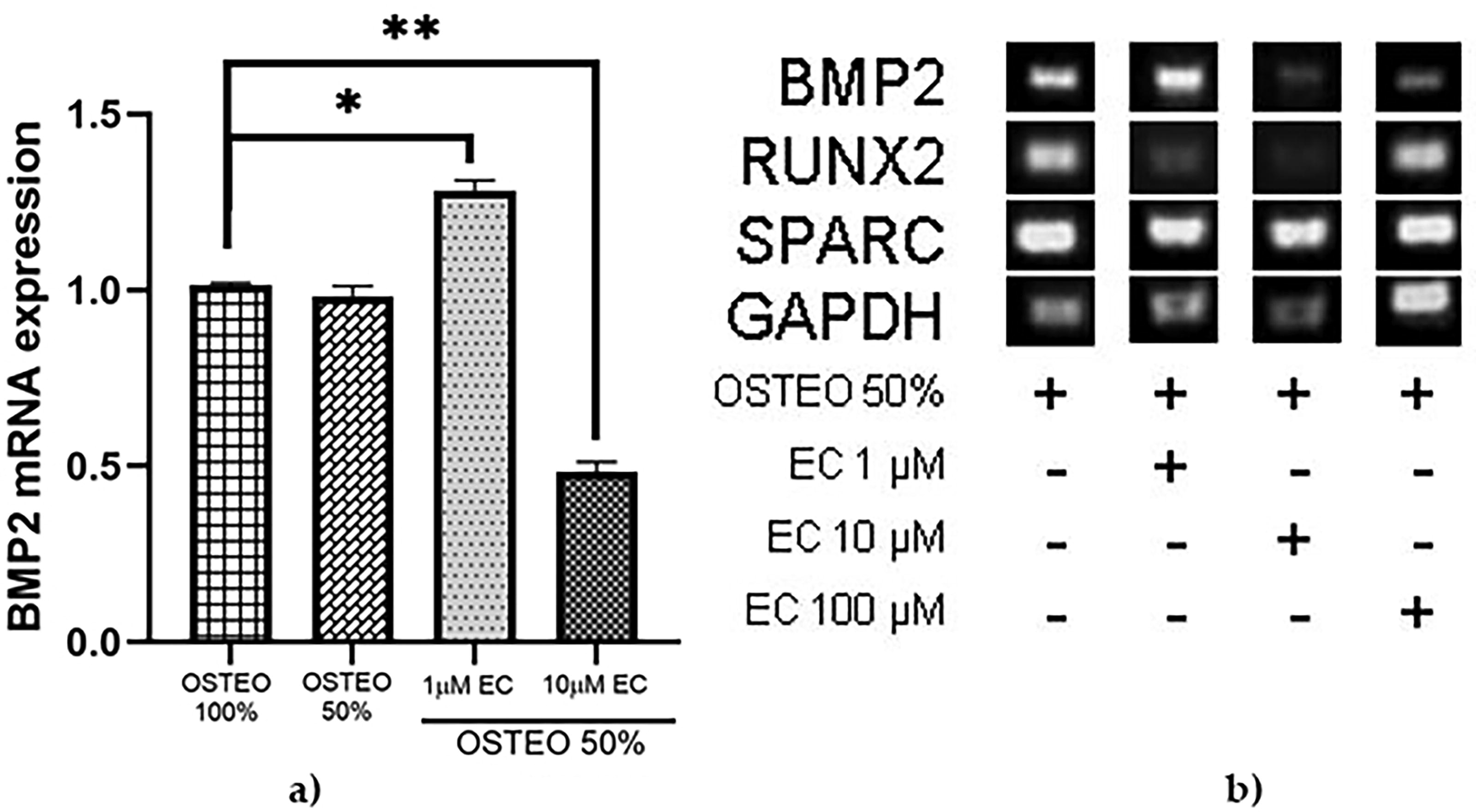
Effect of EC in combination with 50% OSTEO.
EC promotes Ca++ deposits as an indicator of osteogenic differentiation
We investigated the effect of EC at 1 and 10 μM combined with dilutions of OSTEO (33% and 50%) on Ca++ production in MSC-hBM (Fig. 8). Total intracellular Ca++ produced by differentiated cells treated with osteogenic medium without dilution (OSTEO) was taken as a reference standard (first bar, Fig. 8). Deposits of Ca++ were quantified by alizarin red stain. It was observed that OSTEO in both dilutions combined with EC either 1 or 10 μM were capable of inducing more production of Ca++; however, EC at 1 μM combined with OSTEO at 33%, followed OSTEO at 50% and 10 μM of EC were the best combinations to produce significantly more quantities of Ca++.
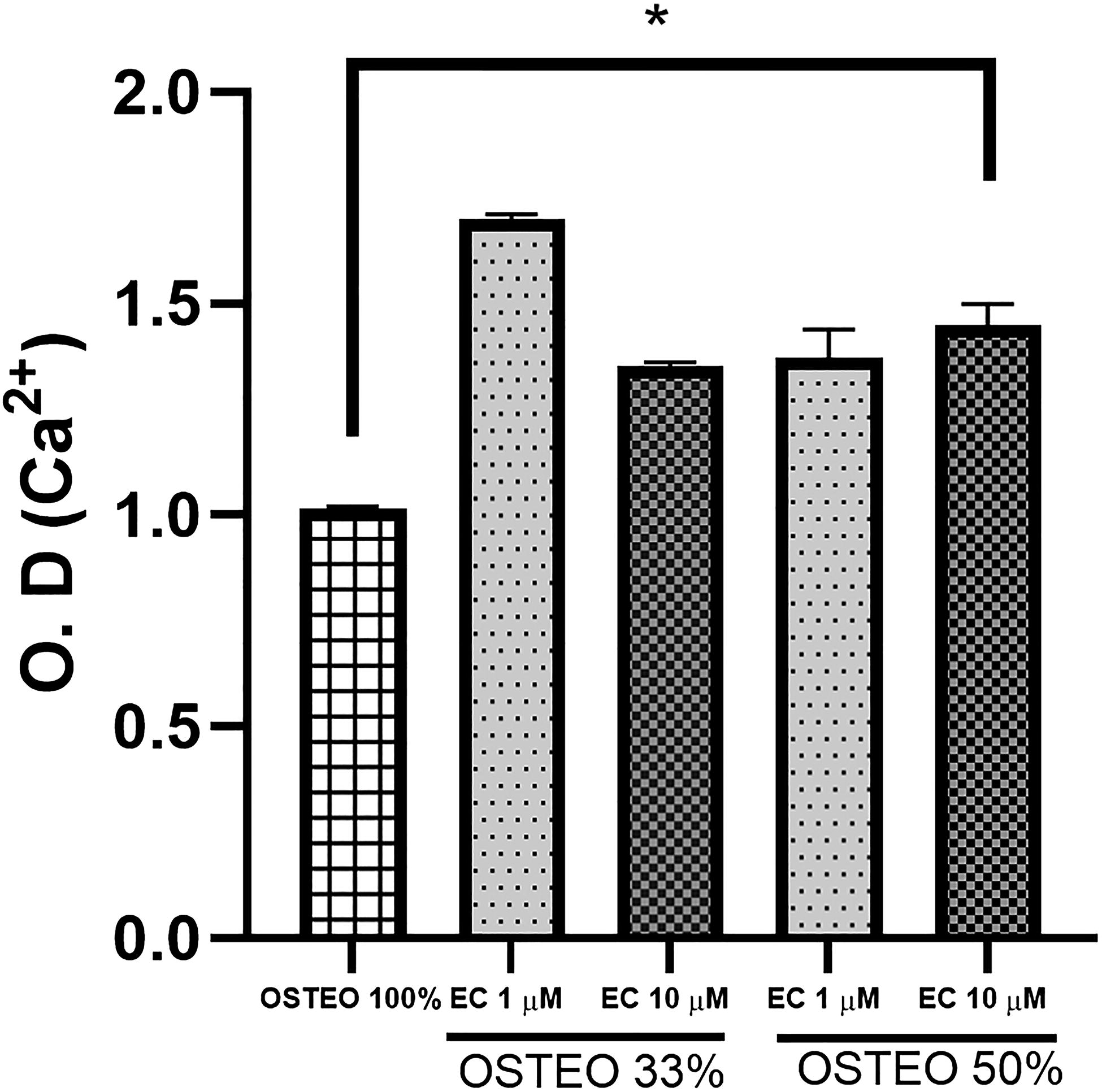
Analysis of osteogenic differentiation through the determination of extracellular Ca++, represented by relative optical density in cultures stimulated with osteogenic differentiation medium (33%, 50%, and 100%) plus 1 and 10 μM of (−)-epicatechin. MSC-hBM exposed to each condition were stained, and alizarin red was quantified by simple spectrometry. One-way analysis of variance P < .05.
DISCUSSION
Recently, research efforts to improve the quality of life for people with degenerative bone diseases (such as osteoporosis, brittle bones, and fractures) have gained importance because they address the main health issues in individuals ≥30 years old. 48 Therefore, compounds derived from natural products are proposed as suitable adjuncts to existing treatments. 49 –52 On the contrary, in vitro models employing MSCs differentiated into osteoblasts have been proposed to study cellular and molecular mechanisms related to bone regeneration. 53,54 For this reason, this study investigated whether (−)-EC (a polyphenolic compound), whose properties for bone regeneration have not yet been described yet induces osteogenic molecular markers alone or in combination with commercial osteogenic medium.
We used different concentrations of (−)-EC, and we found that all concentrations do not induce inhibition of proliferation, as is observed in the majority of polyphenols derived from foods. 55 Expression of genes closely related to the MSCs osteogenesis differentiation was evaluated for EC treatment in comparison with the complete commercial osteogenic medium.
It is well known that SPARC is one of the most abundant extracellular matrix proteins synthesized by osteoblasts 56 and has been perfectly described in differentiated MSCs toward this cell type. 57 EC alone at 1 and 100 μM induced expression of SPARC in cultures of MSC-hBM and reached similar expression levels of those of the osteogenic medium. However, at 10 μM it was not able to induce the same effect. Some molecules could present dual effects as was observed with 10 μM (mainly with antioxidants). 58,59 In molecules like estrogens, contrary effects could be appreciated in the same cell type.
Due to their structural similarity with estrogens, this phenomenon could be related. 60,61 These results are consistent with those observed by Lee et al., who identified SPARC as a commonly secreted protein by human MSCs derived from bone marrow, adipose tissue, and synovial membrane under both normal and inflammatory conditions. 62 We believe that the basal expression of SPARC is due to the gene expression’s relation to different cellular signaling pathways that are active during development, wound healing, adipogenesis, angiogenesis, and cell adhesion, as proposed by Cheng et al. 63 On the contrary, the related transcription factor RUNX2 is one of the most significant related to osteogenesis in MSC. 64 For this reason, RUNX2 was studied. Once more, concentrations of 1 and 100 μM of EC exhibited the highest induction of gene expression compared with the osteogenic medium. However, the 10 μM concentration resulted in only half the gene expression induced by the osteogenic medium.
BMP2 belongs to the group of morphogenic bone proteins or BMPs, which in turn are members of the transforming growth factor family β. 65 BMP2 is considered the osteogenic protein that orchestrate the switches in bone differentiation of MSCs to osteoblasts 66 –68 and is capable of inducing novo bone synthesis. 69 In some studies, transfection of BMP2 has been sufficient to induce osteogenic differentiation of MSCs. 70,71 Since the commercial osteogenic medium (OSTEO) contains factors that cannot be used in humans due to their pharmacological concentrations, we decided to use dilutions at 50% and 33% of OSTEO combined with 1 and 10 μM of (−)-EC. This decision was made because these concentrations alone did not induce BMP2 expression.
Successfully, we observed the expression of BMP2 mRNA in the MSC-hBM in the presence of 33% OSTEO (Fig. 6a) or 50% (Fig. 7a). In addition, this effect depends on the concentration, which agrees with previous work by Davis et al. 70 We observed that BMP2 mRNA expression was reduced with 33% OSTEO plus 1 μM of EC (Fig. 6). However, when combined with 10 μM, the BMP2 mRNA expression increased by around 30% compared with complete OSTEO. In the case of combination at 50% OSTEO with 1 μM of EC, the expression even surpassed the complete osteogenic medium. This was contrary to what occurred when using 50% of OSTEO combined with 10 μM EC, as the expression was inhibited by less than half of the positive control (Fig. 7).
Several studies done by Ko et al. 72 reported that (−)-epigallocatechin, (−)-EC-3-gallate, and (−)-EC possess osteogenic activity on rat MSC-BM at 10, 25, and 50 μm. On the contrary, Chen et al. 24 induced expression of osteogenic genes with 1 and 10 μM (−)-epigallocatechin-3-gallate on a MSC-hBM line; likewise, Wei et al. 26 reported that concentrations of 0.1, 1, and 10 μM catechin induce differentiation of MSC-hBM to osteoblasts.
We found that EC itself is able to induce expression of osteogenic genes at high concentrations (100 μM) than other poliphenols, but combined with the components of osteogenic medium, it enhances the expression of principal genes involved in bone differentiation.
In this study, the expression of the BMP2 messenger RNA was not observed when we used 50% of the osteogenic medium combined with 10 μM EC (Fig. 7). This suggests a competitive inhibition between (−)-EC and some of the concentrated components of the osteogenic medium. For example, dexamethasone (Dexa), a molecule known to favor osteogenesis, may be implicated. 73 –78
In contrast to this, some components of the osteogenic medium appear to show an additive effect when combined with EC at low concentrations (∼1 μM), as has been observed with various components combined with Dexa. We think that this inhibition and synergism phenomenon on the expression of BMP2 are related to the structural similarities between (−)-EC and Dexa and could be used as a target in common. We suggest further investigation into the possible molecular targets to confirm this hypothesis.
Finally, since gene expression leads to osteogenic differentiation, which is identified by the formation of calcium deposits within the cell, 79 MSC-hBM were cultured with OSTEO at 33% and 50% along with combinations of 1 and 10 μM of EC. Subsequently, calcium deposits were stained with alizarin red and quantified. All combinations enhanced the production of Ca++ deposits compared with those induced completely by concentrated OSTEO.
Interestingly, even though most of BMP2 was not expressed at a concentration of 1 μM combined with 33% of osteogenic medium, this condition resulted in the highest production of calcium deposits. This finding likely suggests the existence of other molecular interactions stimulated by the combination of factors in the osteogenic medium and EC, independent of osteogenic genes.
The mechanisms promoting differentiation and increased calcium deposits may involve the activation of signaling pathways such as ERK, 80 enhanced cellular metabolism, and synergistic interactions between factors present in the osteogenic medium and EC. However, further studies are necessary to fully comprehend these mechanisms and how they could be utilized to enhance osteogenic differentiation in therapeutic applications.
MATERIALS AND METHODS
Culture of the MSCs of human bone marrow
Isolation of the MSC-hBM samples was donated by the Orthopedic Service of the General Hospital of Puebla “Dr. Eduardo Vázquez Navarro, (Mexico)”. From these samples, the mononuclear cells were isolated by gradient difference using Ficoll Hypaque 1.077 g/mL (Sigma, USA), which consisted of diluting 20 mL of human bone marrow (hBM) with 20 mL of sterile 1× phosphate buffer (PBS) (dilution 1: 1 v/v).
In Falcon® conical tubes (Corning Centri Star™, USA), 5 mL of Ficoll Hypaque and 10 mL of hBM were added, taking care not to break the gradient. It was centrifuged at 1500 rpm for 40 min at room temperature (RT) centrifugation was carry out in a centrifugue Thermo Scientific Sorvall ST 8 (USA). Cells from the interface corresponding to the mononuclear phase were recovered and washed with sterile PBS. The cell button was resuspended in MSCBM™ medium (Mesenchymal Stem Cell Basal Medium PIoetics™) (Lonza Walkersville, Inc., USA) and cells were seeded in 100 mm × 20 mm Petri dishes (Corning, USA).
The cultures were maintained in a Nuaire incubator (USA) at 37°C, an atmosphere of 5% CO2 at saturated humidity. The change of medium was made every third day and at the end of the first week, the medium MSCBM) was replaced by DMEM-F12 medium (Gibco™, USA) supplemented with 10% FBS (Gibco®, USA) and 1% antibiotic-antimycotic (Gibco, USA). When the cultures showed an 80% confluence, cell culture passes were made. The cells were used from pass 3.
Characterization
Cellular plasticity of the cells were determined by culturing them with osteogenic differentiation medium (Stempro® Osteogenesis Differentiation Kit, USA, Cat. A10072-01) and adipogenic (Stempro Adipogenesis Differentiation Kit, USA, Cat. A10070-01) for 21 days and finally the following staining: Alizarin red for osteoblasts; the MSC-hBM differentiated to osteoblasts were stained with a solution of red alizarin S (1,2-dihydroxyanthraquinone) for 10 min and washed with PBS to remove excess dye, and then ORO to stain adipose cells: Mesenchymal cultures grown with adipogenic were added to an ORO solution in 60% isopropanol and left at RT for 10 min, finally the samples were washed with 60% isopropanol, respectively, to remove excess of the dye. Finally, the cells were observed and photographed using a light field microscope (Zeiss, CA, USA).
Flow cytometry
The MSC-hBM were detached with a 2 mM EDTA solution, subsequently incubated for 30 min with anti-human monoclonal antibodies: HLA-DR (Santa Cruz Biotechnology, CA, USA), CD34 (Santa Cruz Biotechnology, CA, USA), CD45 (Thermo Scientific, USA), CD105 (Biolegend, USA), CD73 (Biolegend USA), and CD90 (Biolegend, USA), subsequently the cells were washed with PBS and centrifuged at 700 g min−1.
The cell button was resuspended in 4% paraformaldehyde, read in a FACSCalibur flow cytometer, and analyzed with the CellQuest software (Becton Dickinson, CA, USA). The percentages were obtained considering the number of positive cells for each marker (fluorescein isothiocyanate or phycoerythrin) and its relation with the total population analyzed.
Proliferation and cell viability assays
The MSC-hBM were stimulated for 72 h with EC at 1, 10, and 100 μM in DMSO as a vehicle, analysis of cell proliferation by CV. After the stimulation, the cells were fixed with 1.1% glutaraldehyde in PBS, washed with PBS, and stained with 0.1% CV dye (Sigma Aldrich, USA) prepared in formic acid buffer (Sigma) pH 6.0; subsequently the MSC-hBM were repeatedly washed with PBS to remove excess dye; the assimilated CV was solubilized with 10% acetic acid in PBS and stirred for 5 min at RT.
Viability analysis by MTT; once the culture time had elapsed, the cells were incubated with a solution of (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide), MTT (Sigma) at 1 mg mL−1 × 3 h at 37° C, the supernatant was discarded, and the cells were washed with PBS, then 0.1 N isopropanol–HCl was added to dissolve the dye. Finally, the absorbance of both assays was determined in an ELISA reader (E1x800: Bio-Tek) at a wavelength of 595 nm, and the proliferation/viability ratio of the similar concentrations was calculated.
Osteogenic differentiation with EC and OSTEO
The MSC-hBM were cultured in the presence of different dilutions of osteogenic medium (OSTEO): 33%, 50%, and 100%, each with combinations of 1, 10, and 100 μM of EC. The dilutions were in DMEM-F12 until completing 100% of the volume. The medium of the cultures containing EC was replaced every day, while the medium of the cultures with OSTEO was replaced every third day; after seven days of culture, the osteogenic differentiation was evaluated with the different conditions.
The osteogenic media contains Dexa at high concentrations and is essential for osteogenic differentiation; however, the exact concentration used has not been reported by the manufacturer, although different studies have concurred with the approximate concentration of Dexa to be 10 nM. 81
Gene expression
The extraction of total RNA from the MSC-hBM was performed according to the TRIzol®-reagent extraction protocol (Invitrogen, USA). The synthesis of cDNA (retro-transcription or RT) was carried out from the total RNA according to the epicentre® protocol (Illumina company, USA). The primers used were designed using the Oligo Explorer 1.0 program, for forward-GAPDH (5′-TCAACGACCACTTTGTCAAG-3′) and reverse-GAPDH (5′-ACTGTGAGGAGGGGAGATTC-3′), F-SPARC (5′-CAGAGGAAACCGAAGAGGAG-3′) and R-SPARC (5′-GCAAAGAAGTGGCAGGAAG-3′), F-RUNX2 (5′-AGTGCGGTGCAAACTTTCTC-3′) and R-RUNX2 (5′-CTGCTTGCAGCCTTAAATGA-3′), and F-BMP-2 (5′-GGACGCTCTTTCAATGGAC-3′) and R-BMP-2 (5′-GGTGGGTCTCTGTTTCAGG-3′), the sequences of the genes were obtained from the database: http://www.ncbi.nlm.nih.gov/pubmed.
The specificity was determined with the BLAST and MEGA 5.0 programs. The polymerase chain reaction was performed in an Eppendorf Mastercycler® thermocycler with the following amplification protocol: denaturation; 94°C 1 min−1, alignment; 58–60°C 1 min−1, extension; 72°C 30 s−1 30, for 35 cycles for each gene. The products were visualized on 2% agarose gels in 1% TAE buffer (40 mM Tris-acetate + 1 mM EDTA) stained with 100 μM of ethidium bromide. The registration was carried out with the Gel Doc XR gel documentation system (Bio-Rad), where the intensity of the bands was assessed by densitometry using the ImageJ program version IJ 1.46r.
Determination of extracellular Ca++ in the cultures
The red alizarin S fixed by the MSC-hBM differentiated to osteoblasts was solubilized with cetylpyridinium (Sigma, USA) at 10% in PBS. The samples were read in an ELISA plate reader (E1x800, BioTek) at a wavelength of 550 nm. Finally, the values of DO were converted to percentages, taking 100% reference cells grown with 100% OSTEO.
Statistical analysis
The results were represented as the mean of the values ± the standard deviation. To determine the significance of the results, they were analyzed with the statistical test of analysis of variance of one way, considering P < .05 as significantly different. The GraphPad Prism 9 program was used to perform the statistical tests.
CONCLUSIONS
EC itself is capable of inducing osteogenic differentiation, but its effect is significantly enhanced when combined with diluted compounds from the osteogenic medium. This synergy leads to increased expression of key osteogenic markers such as SPARC, RUNX2, and primarily BMP2. Consequently, EC emerges as a promising plant-derived candidate for treatments aimed at improving bone health.
These findings are based on the osteogenic differentiation of MSCs, which exhibited morphological changes and increased calcium deposits, indicating calcium recovery and bone regeneration in calcium-deficient bone tissues, such as those affected by osteoporosis. Notably, these effects were achieved without requiring pharmacological concentrations of osteogenic factors, thereby potentially reducing adverse side effects. Given the critical role of BMP2 in bone regeneration, it is essential to investigate the molecular pathways by which EC activates this protein. Understanding these mechanisms may have clinical relevance, enabling the development of dietary regimens or therapeutic strategies using molecules with osteogenic activity similar to natural polyphenols present in the human diet.
Footnotes
ACKNOWLEDGMENTS
The authors extend their gratitude to the Orthopedic Service of the General Hospital of Puebla and Eduardo Vázquez Navarro for generously providing the human bone marrow samples used in this investigation.
AUTHORS’ CONTRIBUTIONS
C.C.C.M., E.R.L., and A.P.B. contributed to the conceptualization and supervision of the work. G.G.I., J.L.A.E., and E.M.J. were responsible for data curation and formal analysis. E.M.J., E.R.L., I.P.L., and G.G.I. contributed to visualization and methodology. I.P.L., G.G.I., A.P.B., and C.C.C.M. were involved in drafting the initial article and reviewing and editing the document. J.L.A.E., G.G.I., and A.P.B. contributed to the investigation and validation of the study.
AUTHOR DISCLOSURE STATEMENT
The authors declare no conflicts of interest related to this study.
FUNDING INFORMATION
The present work was funded by Secretaría de Investigación y Posgrado del Instituto Politécnico Nacional in support of project numbers 20162141 and 20230453.
