Abstract
Declines in estrogen levels occur in women transitioning to menopause. Estrogen hormones play important roles in multiple systems of the body, and estrogen loss is associated with a variety of symptoms that can decrease quality of life. The gut microbiota is involved in regulating endogenous estrogen levels. A portion of estrogen glucuronides can be reactivated in the gut by the microbial enzyme β-glucuronidase, and the resulting free estrogens can return to the bloodstream. Here, we carried out in vitro screening of β-glucuronidase activities for 84 strains belonging to 16 different species of lactic acid bacteria and bifidobacteria and found that one and three strains of Levilactobacillus brevis and Lacticasebacillus rhamnosus, respectively, can deconjugate estrogens. Among these strains, L. brevis KABP052 had the highest β-glucuronidase activity. Moreover, in an exploratory, randomized, double-blind, placebo-controlled trial, we demonstrated that serum estrogen levels in healthy peri- and postmenopausal women given a probiotic formula containing KABP052 were maintained over time, whereas levels significantly decreased in the group given a placebo. Significantly higher levels of estradiol (31.62 ± 7.97 pg/mL vs. 25.12 ± 8.17 pg/mL) and estrone (21.38 ± 8.57 pg/mL vs. 13.18 ± 8.77 pg/mL) were observed in the probiotic versus placebo group after 12 weeks of intervention. This clinical study demonstrated for the first time the estrogen modulation capacity of a probiotic formula containing a bacterial strain having β-glucuronidase activity in women during the menopausal transition and formed the basis for future investigations using probiotics in the menopausal population.
INTRODUCTION
Menopause is defined by the permanent cessation of menstrual periods that happens as a consequence of natural aging of the ovaries. Most women undergo menopause between 45 and 55 years of age. 1 This crucial milestone is preceded by perimenopause, a phase that can last several years when estrogen secretion from ovarian follicular cells decreases. 2 Perimenopause is thus characterized by a drastic hormonal change that affects physical and mental health. 3
In addition to their role in reproductive health, estrogens have many beneficial effects in multiple systems in the female body. Estrogens influence functions of the brain, sexual and urogenital systems, skin and mucosal barriers, adipose tissue, the musculoskeletal system, and the gastrointestinal tract, among others. 3,4 Declines in circulating levels of estrogens during peri- and postmenopause coincide with the onset of a variety of symptoms such as hot flashes, insomnia, depression, decreased libido, vaginal dryness, fragility of skin, and mucosal barriers or weight gain, which all can negatively impact quality of life. Moreover, declines in estrogen levels may have long-term health consequences including increased risk of cardiovascular disease and osteoporosis. 3
Estrogens may also affect the composition of gut microbiota, the community of microorganisms that inhabit the gut, since the menopausal transition is associated with a loss of gut microbiota diversity. However, the relationship between estrogens and the gut microbiota is bidirectional. 5 In some metabolic pathways, estrogens undergo glucuronide conjugation in the liver, and a fraction of the conjugated forms are excreted via the bile into the intestinal tract for evacuation with feces. Some bacterial species in the gut secrete the enzyme β-glucuronidase (GUS) that deconjugates the inactive glucuronide form of estrogen, restoring it to the active form. Reactivated estrogens can then be reabsorbed into the enterohepatic circulation and subsequently enter the systemic bloodstream to reach target tissues. 6 Gut bacterial functions involved in estrogen deconjugation are referenced as the estrobolome. 7 Several commensal gut bacterial species from all the major phyla have been shown to exhibit GUS activity. 8 Of note, a recent study found that menopause is associated with an altered estrobolome as postmenopausal women have decreased abundance of microbial GUS relative to premenopausal women. 9
Therapies to restore hormonal levels and alleviate symptoms during menopause include hormonal replacement therapy and phytoestrogens. However, these approaches are not suitable for everyone and alternative options are needed. 10,11 In this context, new strategies such as probiotic interventions have recently gained attention. Probiotics are defined as living microorganisms, which, when administered in adequate doses, confer a benefit to the host. 12
In this study, we aimed to develop a probiotic therapy to modulate estrogen levels in peri- and postmenopausal women. We first screened the GUS activity of 16 distinct lactic acid bacteria and bifidobacteria species and confirmed in vitro the estrogen-deconjugation capacity of the GUS-positive strains. We then combined the strain having the highest enzymatic activity with a probiotic blend that was tested in an exploratory, randomized, double-blind, placebo-controlled trial to evaluate its capacity to maintain serum estrogen levels in healthy peri- and postmenopausal women.
MATERIALS AND METHODS
Expanded materials and methods are provided in the Supplementary Data S1.
Bacterial strains and routine growth conditions
We selected 84 bacterial strains of 16 species among source isolates and culture collections from healthy human samples and fermented foods (Supplementary Table S1). Lactobacilli, Pediococci, Lactococci, Enterococci, and Leuconostoc spp. strains were cultured in Man–Rogosa–Sharpe (MRS) broth under aerobic conditions. Bifidobacterium longum strains were propagated in MRS broth supplemented with 1 mg/mL cysteine-HCl under anaerobiosis. All the strains were cultured at 37°C.
Screening of bacterial strains with GUS activity
We screened GUS activities of the 84 strains according to a previously described method.
13
In brief, we cultured strains in MRS broth supplemented with 200 µg/mL of the chromogenic substrate 5-bromo-4-chloro-3-indolyl-β-
Enzymatic assay for GUS activity
We carried out an enzymatic assay to test the GUS activity of the selected bacterial strains using a previously described method with some modifications. 6,13 Briefly, strains were washed twice with GUS buffer (100 mM sodium phosphate 2.5 mM EDTA, pH 6.0), resuspended in the same buffer, and disrupted using a Multi-bead Shocker. Following centrifugation, the protein concentration of each extract was determined using a protein assay kit. Extracts were kept on ice until the assay.
We incubated extracts (0.25 mg protein/mL) at 37°C for 1 h in GUS buffer containing 1 mM 4-nitrophenyl-3-β-
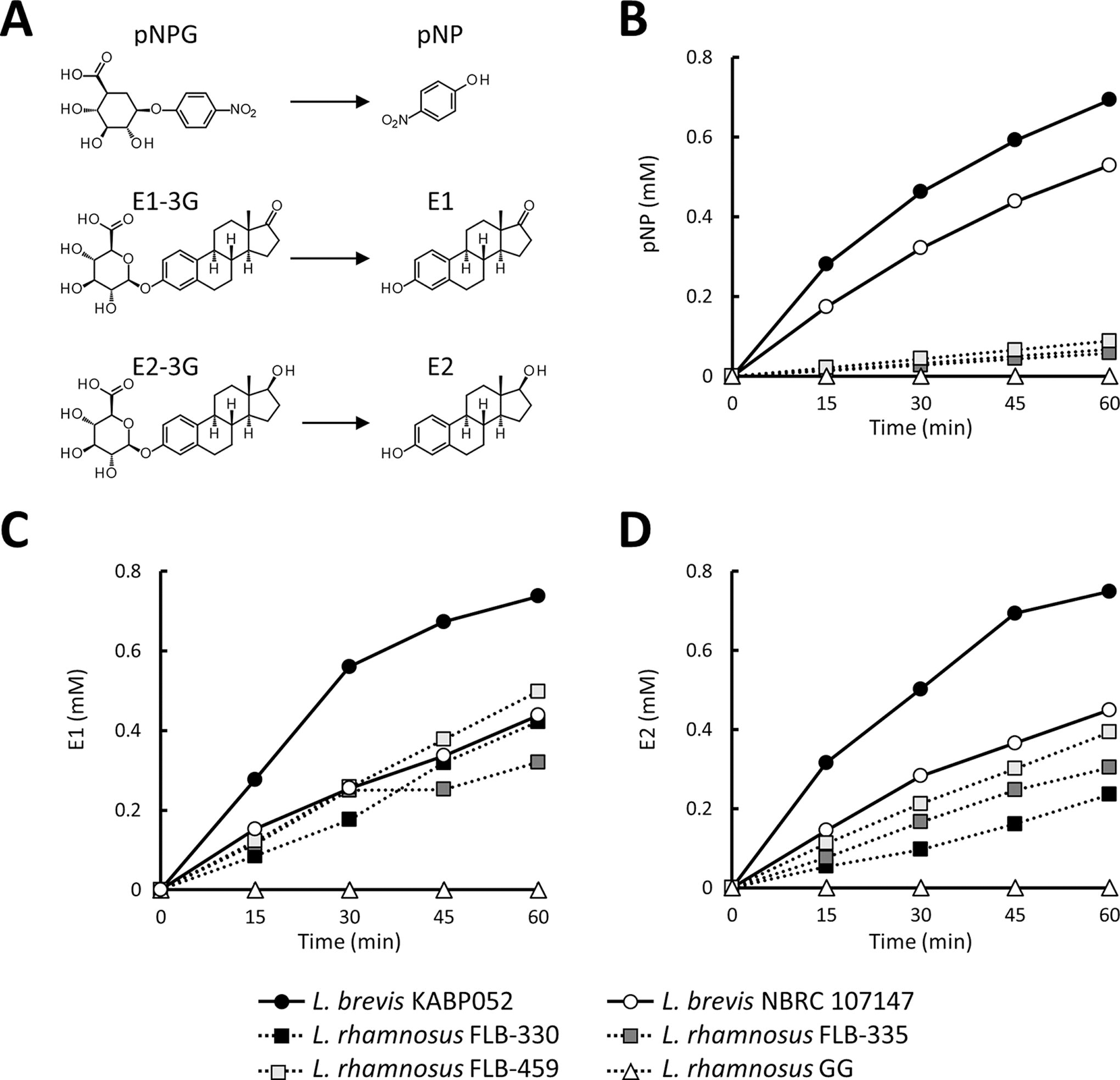
Enzymatic analyses for four probiotic strains able to deconjugate glucuronide components (GUS activity).
Protocol design and ethics for the clinical study
The main objective of this clinical study was to investigate how supplementation with probiotics affected serum E1 and E2 levels in healthy peri- or postmenopausal women. The prospective, double-blind, randomized, placebo-controlled study involved two parallel groups and was performed at two clinical centers under the control of a contract research organization (Mibyou Life Science, Inc., Tokyo, Japan) commissioned by Kaneka Corporation (Osaka, Japan). We performed this clinical study in Tokyo, Japan, in accordance with the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects.
The study protocol was approved by the Mibyou Research Ethics Review Committee (September 17, 2021; approval no. MRS2021091702) and was registered at the University Hospital Medical Information Network Clinical Trials Registry (UMIN-CRT; ID: UMIN000045547).
Test foods for the clinical study
Based on our in vitro experiments, Levilactobacillus brevis KABP052 (CECT 7480) had the highest GUS activity and was used in the probiotic supplement. Lactiplantibacillus plantarum KABP051 (CECT 7481) and Pediococcus acidilactici KABP021 (CECT 7483) were also included in the probiotic supplement based on their ability to inhibit opportunistic pathogens 14,15 that, in turn, could improve gut homeostasis. The probiotic blend comprised three bacterial strains at a ratio of 2:1:1 colony-forming units (CFU) of L. brevis KABP052, L. plantarum KABP051, and P. acidilactici KABP021, respectively. Each capsule contained ≥1 billion CFU. The placebo contained starch instead of the probiotic blend.
Participants in the clinical study
We recruited healthy volunteers according to the inclusion/exclusion criteria (Supplementary Data S1). In brief, we enrolled healthy Japanese women between 45 and 55 years of age who reported having some differences in menstruation compared with their 20s and were experiencing mild menopausal symptoms that did not interfere with daily life. All volunteers provided written informed consent to participate.
Randomization was performed by the permuted block method using a modified version of the Kupperman menopausal index 16 for assessment of Japanese women (mKMI) 17 as well as menopause status as stratification factors. Each mKMI question was scored according to four levels (severe, moderate, slight, and absent), and the individual score was calculated using the weighting method for each symptom. The total mKMI score is 51, and higher scores indicate more severe conditions. We collected total mKMI scores for all participants. The allocation manager kept the study allocation list, and blinding was maintained for all parties until completion of the study.
Intervention and measurements of biological/biochemical parameters in the clinical study
Eligible subjects took one capsule containing placebo or probiotics daily for 12 weeks. Serum samples to evaluate estrogen content were assessed by ASKA Pharma Medical Co., Ltd. (Kanagawa, Japan) according to the company’s methods. Fecal samples for GUS activity were prepared using a previously described method with slight modifications. 18 Briefly, fecal samples were resuspended in GUS buffer and centrifuged. Aliquots of the supernatants (0.50 mg protein/mL) were used for a pNP enzymatic assay as described earlier.
Menopausal symptoms were also monitored using the mKMI. Biological and biochemical parameters for safety evaluation were measured according to standard procedures recommended by the Japanese Ministry of Health, Labor and Welfare at the time of health examination. Adverse events were monitored throughout the study period.
Statistical analyses
In vitro enzymatic experiments were performed in duplicate, and the mean GUS activity of all strains at each test point was calculated. Clinical parameters were evaluated using parametric (Student’s t-test or Welch’s t-test) and nonparametric (Mann–Whitney U test or Wilcoxon signed rank test) statistical methods. Linear regression was used to identify factors having the highest influence on estrogen levels, and the factors were then evaluated using analysis of covariance (ANCOVA) analyses by combining the covariates age and baseline E1 and E2 concentrations. Fisher’s exact test was used to evaluate the balance of the number of participants per menopausal stage and adverse events. Two-sided P values <.05 were considered statistically significant.
RESULTS
Screening of GUS activity-positive strains
Screening of 84 bacterial strains for GUS activity using X-Gluc showed that nine L. brevis strains (KABP052/CECT 7480, NBRC 107147, NBRC 3345, NBRC 12005, NBRC 13109, NBRC 3960, NBRC 12520, and NBRC 13110) and three Lacticasebacillus rhamnosus strains (R-253, R-256, and FLB-459) had potential activity (Table 1 and Supplementary Table S1). Based on the intensity of blue color in the broth, we examined L. brevis KABP052 and the three L. rhamnosus strains in subsequent analyses. We also used L. brevis NBRC 107147 and L. rhamnosus GG, which were both major strains, in the enzymatic assays.
Species and Strains Screened for GUS Activity
GUS, β-glucuronidase.
Selection of the strain with the highest estrogen deconjugating activity
We next tested the deconjugating activities of the selected GUS-positive L. brevis and L. rhamnosus strains (Fig. 1). From experiments using pNPG, time-dependent release of pNP as the deconjugated product was observed for all four strains. L. brevis KABP052 produced the highest amount of pNP among the four strains (Fig. 1B). Next, we tested production of two conjugated forms of estrogens. All strains produced deconjugated E1, but L. brevis KABP052 had the highest hydrolyzation capacity at all time points (Fig. 1C). Similar results were observed for E2-3G (Fig. 1D). Therefore, we further evaluated the capacity of L. brevis KABP052 to modulate estrogen levels in the human body.
Efficacy and safety of a probiotic formula containing L. brevis KABP052 in peri- and postmenopausal women
Women in the peri- or postmenopausal stage (n = 115) were enrolled after screening and randomly divided into two groups (Supplementary Fig. S1). Two participants withdrew before the start of the intervention for personal reasons. A total of 113 participants consumed the test foods for the complete 12-week intervention period without changing their lifestyle. During the intervention period, two subjects in the probiotics group contracted COVID-19, and these participants were excluded as judged by the principal investigator. The two groups had no significant differences in major physical and clinical characteristics or the number of subjects in each menopausal stage at baseline (i.e., before the intervention) (Table 2).
Baseline Characteristics of the Clinical Study Population (per Protocol Set)
Values are presented as means ± SD.
Comparison between groups. Statistical significance of differences between groups was determined by unpaired Student’s t-test (hormones and physical parameters except body weight); Welch’s t-test (body weight); and Mann–Whitney U test (total mKMI score). The placebo and probiotic groups had no significant difference in the number of participants in each menopausal stage (Fisher’s exact test, P = 1.000).
BMI, body mass index; E1, estrone; E2, estradiol; FSH, follicle-stimulating hormone; mKMI, modified Kupperman menopausal index for Japanese women; SD, standard deviation.
At baseline, regression analysis showed a strong correlation between age and both E1 (r = −0.45, P <. 001) and E2 (r = −0.49, P <. 001). Based on this finding, we adjusted the analysis of estrogen concentrations throughout the study period according to age and estrogen level (E1 and E2) at baseline. After the 12-week intervention, levels of both E1 and E2 in the probiotic group were significantly higher than those for the placebo group when adjusting for baseline estrogen concentrations and age (E1: 31.62 ± 7.97 pg/mL vs. 25.12 ± 8.17 pg/mL, P <. 05; E2: 21.38 ± 8.57 pg/mL vs. 13.18 ± 8.77 pg/mL, P <. 05; Fig. 2). Furthermore, estrogen levels in the group given the probiotic were maintained over time, whereas those for the placebo group significantly decreased (Supplementary Table S3). Meanwhile, the groups showed no significant difference in GUS activity in fecal samples from the subjects either at baseline or after 12 weeks of the intervention (Table 3).
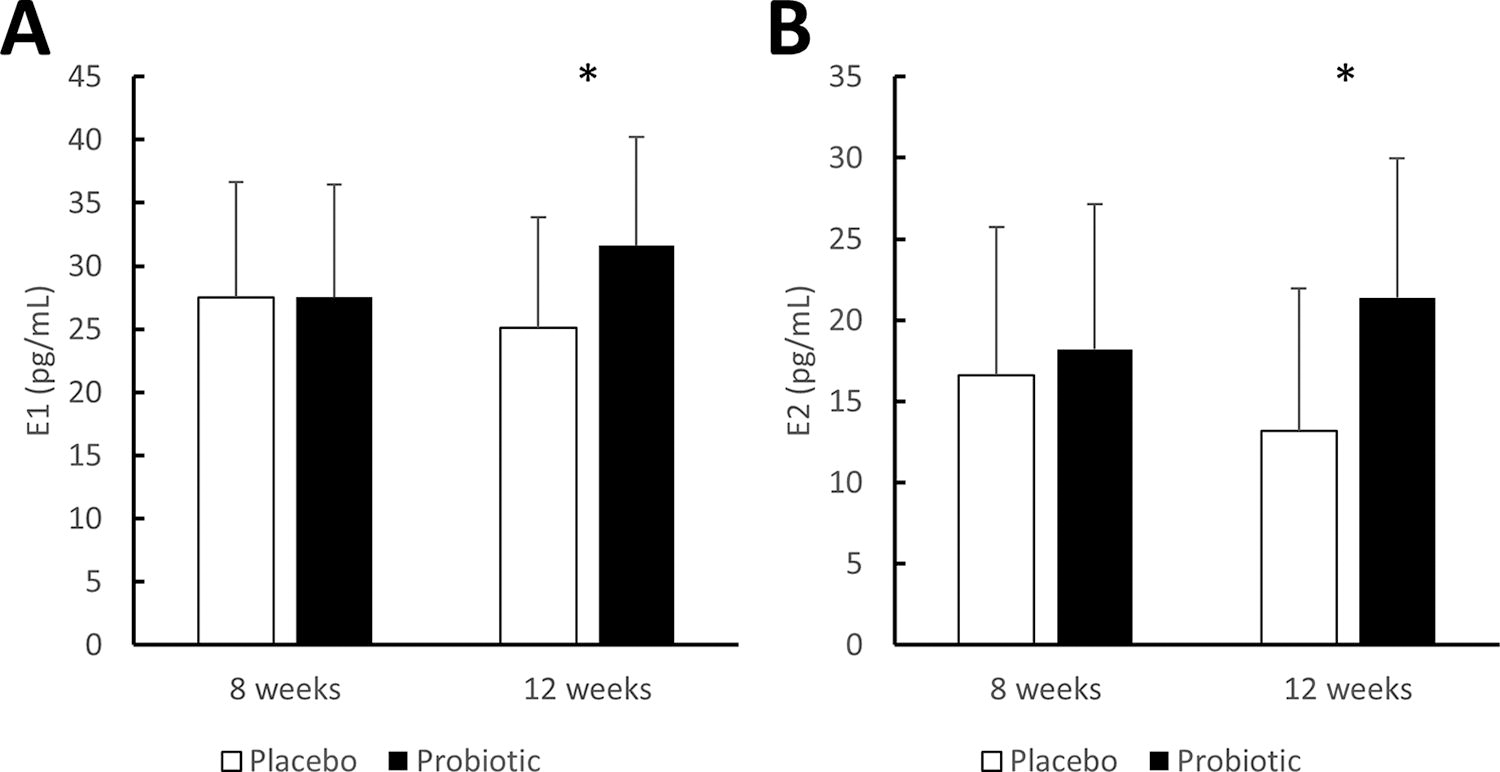
Serum estrogen levels after the intervention. The concentrations of
GUS Activity in Fecal Samples
Values are presented as mean ± SD.
P < .01 vs. baseline.
Comparison between groups. Paired Student’s t-test was used to determine statistical significance of differences between values before and after the intervention period for each group. Unpaired Student’s (at 12 weeks) or Welch’s (baseline) t-tests were used to compare groups.
GUS, β-glucuronidase.
Menopausal symptoms assessed by mKMI scores improved significantly at all time points during and after the intervention in both the probiotic and placebo groups compared with those at baseline (Table 4). The comparison between the groups at each test point revealed no significant differences.
Total mKMI Score to Evaluate Menopausal Symptoms
Values are presented as the median with first and third interquartile (parentheses) of the individual total mKMI scores.
P < .05 vs. baseline.
Comparison between groups. Statistical significance of differences between groups was analyzed by Wilcoxon signed-rank test to compare baseline values for each group or Mann–Whitney U tests for comparison between groups. mKMI, modified Kupperman menopausal index for Japanese women.
Blood and urine biomarkers and physical parameters were essentially unchanged throughout the study period (Supplementary Table S2), and the probiotic blend was well tolerated by study participants. The number of adverse events was similar between the groups (27 in the probiotic group vs. 28 in the placebo group), and all adverse events were mild or moderate in intensity and classified as unrelated to the probiotics.
DISCUSSION
Accumulating evidence suggests that gut microbiota is an important player in estrogen regulation. Menopause is linked to alterations of the estrobolome with loss of specific microbial functions (i.e., GUS activity). 9,19 As such, use of specific probiotic strain(s) having the ability to regulate estrogen levels could be a potential therapeutic approach to counteract estrobolome changes during the menopausal transition.
Our in vitro studies showed that several L. brevis and L. rhamnosus strains isolated from the human gut and fermented foods have GUS activity. Among the strains we tested, L. brevis KABP052 had the highest GUS activity. Furthermore, none of the Lactobacillus gasseri strains in our set had GUS activity. Notably, the L. gasseri strains we tested were vaginal isolates, whereas a previous study showing GUS activity of L. gasseri strains used isolates from the human gut, 13 suggesting that the deconjugation capacity of this species might be associated with intestinal origin. Similarly, our set of L. acidophilus, Enterococcus Faecium, B. longum (isolates from human gut), or Leuconostoc spp. (isolates from fermented foods) had no GUS activity, which is in contrast to the results of earlier studies, suggesting that these species do exhibit GUS activity. 13,20,21 Our findings for species that were positive for GUS activity were otherwise consistent with those of several previous studies. 6,22,23 Taken together, GUS capacity might be a conserved trait in L. brevis and L. rhamnosus that is independent of the isolation source.
We demonstrated that daily supplementation with the probiotic formula containing L. brevis KABP052 significantly sustained serum concentrations of both E1 and E2 in healthy peri- and postmenopausal women compared with women who received placebo. The probiotic formula was safe and well tolerated. In addition, the estrogen levels of subjects who took this probiotic formula were maintained within the normal range of healthy, young adult premenopausal women, according to the criteria of the clinical inspection organization. This result supports the safety of the supplementation using the current dosage and period of intervention. This is the first time, to our knowledge, that a probiotic blend has been demonstrated to maintain serum estrogens levels in peri- and postmenopausal women. This result contrasts with those of several recent studies that did not demonstrate modulation of hormone levels in study participants given probiotics containing various species. 24 –26 The menopausal transition is also associated with altered gut transit. 27,28 Thus, our probiotic formulation included L. plantarum KABP051 and P. acidilactici KABP021, which lack GUS activity (Supplementary Table S1) but can modulate gut homeostasis. 14,15 The inclusion of these two strains may have impacted the gut environment and contributed to changes in hormone levels. However, one limitation of this clinical study is its inability to evaluate the effect of the probiotic formulation on the gut environment and microbial content as the gut microbiota was not examined. Future studies that include gut microbiota composition analyses are thus needed to investigate interactions between the bacterial strains and changes in the gut environment because of simultaneous supplementation with a combination of bacterial strains.
The GUS activity in fecal samples was unchanged after the intervention in the group given the probiotic formula. This outcome might indicate that probiotic function occurs in the small intestine rather than the colon. Indeed, Lactobacilli populations are found in high numbers in the small bowel. 29 Interestingly, other studies using L. gasseri ADH, a GUS-positive strain, reported that consumption of live, but not killed, bacterial strains significantly decreased fecal GUS activity in elderly subjects. 30,31 More investigation is needed to understand the interactions that regulate GUS activity in the intestinal tract and to confirm the outcomes observed in our trial, including analysis of gut microbiota composition and function. In addition, we observed no correlation between estrogen levels and menopausal symptoms. This result may be because of the inclusion of volunteers who are all healthy but exhibit a variety of menopausal symptoms. No members of our study cohort had total mKMI scores that were severe, and/or a placebo effect associated with improvement in menopausal symptoms in the placebo group was similar to those in the probiotic group. Further investigations involving more participants, with focused symptoms, different intervention strategies, and different dosing regimens, may provide additional insights into the most effective, safest, and most sustainable methods for supporting women with menopause symptoms.
In conclusion, we identified L. brevis and L. rhamnosus strains as having GUS activity in vitro and demonstrated that a probiotic blend including GUS-positive L. brevis KABP052 maintained the serum concentration of estrogens in peri- and postmenopausal women. These results pave the way for future studies that evaluate symptomatology improvement in specific populations of women undergoing menopause.
Footnotes
ACKNOWLEDGMENTS
The authors greatly appreciate the helpful suggestions and comments from Dr. Iwao Funahashi (Kaneka Corporation, Japan) and Dr. Fumiki Aoki (AB-Biotics S.A., Spain).
AUTHORS’ CONTRIBUTIONS
S.H.: Conceptualization (equal); methodology (equal); investigation (in vitro—lead, clinical—equal); formal analysis (equal); visualization (lead); writing—original draft (equal); writing—reviewing and editing (supporting). Y.T.: Conceptualization (equal); methodology (equal); investigation (in vitro—supporting, clinical—equal); formal analysis (equal). J.E.-M.: Validation (equal) and formal analysis (equal). P.H.: Conceptualization (equal); validation (equal), formal analysis (equal). M.A.: Formal analysis (supporting); visualization (supporting); writing—original draft (equal). M.P.: Formal analysis (supporting); visualization (supporting); writing—original draft (equal). T.U.: Supervision (lead); conceptualization (equal); investigation (supporting). J.S.: Project administration (lead); conceptualization (equal); methodology (equal); investigation (in vitro—supporting, clinical—equal); formal analysis (equal); visualization (supporting); writing—original draft (equal); writing—reviewing and editing (lead).
AUTHOR DISCLOSURE STATEMENT
All the authors are employees of Kaneka Corporation or AB-Biotics S.A. as a partner company of Kaneka Corporation and declare no conflicts of interest. None of the co-authors would receive financial benefits from commercial development of the probiotic formulation.
DECLARATION
The experiments in this study complied with the current laws of the country in which they were performed.
FUNDING INFORMATION
This study was funded by Kaneka Corporation.
SUPPLEMENTARY MATERIAL
Supplementary Data S1
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
