Abstract
Curcumin, the fat-soluble active ingredient and major compound of curcuminoids contained in the curcuma root, is known for its physiological low absorption and bioavailability. Various formulations and galenic technologies are currently available on the market. In this study, the product tested was provided as a soft gelatin capsule containing curcuminoids in an oily matrix mixed with phospholipids (oil/phospholipids [PL]-based, no new technologies applied or artificial excipients added). This was intended to improve bioavailability of curcuminoids as well as to mimic the natural digestion process of fat-soluble substances. In particular, the oral bioavailability of curcuminoids in the oil/PL-based formulation was compared with the pure curcuminoids extract alone (reference product), in a randomized, cross-over, single oral dose study design. Twelve healthy subjects were administered 200 mg curcuminoids under fasting conditions. Pharmacokinetic parameters were analyzed from individual concentration–time curves of total curcuminoids, as well as the curcumin metabolite tetrahydrocurcumin (THC). Results showed significantly higher AUC0-8h levels after the intake of the oil/PL-based formulation for total curcuminoids (205.60 vs. 112.50 ng/mL*h, P = .0001) as well as for THC (347.30 vs. 118.90 ng/mL*h, P < .0001) in comparison to the pure curcuminoids extract. Cmax was also significantly higher for both parameters analyzed (total curcuminoids: 47.54 vs. 21.16 ng/mL, P = .0001; THC: 96.69 vs. 29.83 ng/mL, P < .0001). In addition, the uptake kinetic of total curcuminoids was significantly fastened with the oil/PL-based curcuminoids formulation compared with the pure curcuminoids extract (P = .0446). These data suggest an improved impact on curcuminoids uptake of the oil/PL-based formulation and confirms its good tolerability.
INTRODUCTION
Curcuminoids are the fat-soluble active ingredients of curcuma root, which are known for their anti-inflammatory, antioxidant, antithrombotic, and antiangiogenic properties. 1,2 Recent clinical studies demonstrated beneficial effects of curcumin (CC) in several health disorders such as cardiovascular diseases, osteoarthritis, diabetes, etc. 3 –5 CC is the major compound of curcuminoids. Because of their high lipophilicity, curcuminoids exhibit low absorption and bioavailability in the bloodstream. The majority of the ingested CC is excreted unmetabolized through the feces. The absorbed CC is rapidly converted to its water-soluble metabolites, glucuronides, and sulphates. 5,6
During the physiological process of digestion, fat-soluble substances are normally absorbed in the upper small intestine. The uptake requires the presence of dietary fats as transporters for lipophilic molecules and bile acids as emulsifiers to increase solubility. During digestion, micelles are formed. These mixed micelles are then absorbed into cells of the small intestinal epithelium via passive diffusion. Absorption is strongly dependent on the type and amount of fats supplied at the same time. In the intestinal cells, fat-soluble substances are absorbed into chylomicrons (lipoproteins rich in fats), which transport them via the lymph into the peripheral blood circulation. 6
It is known from nutritional science that fat-soluble substances, such as vitamin D or curcumin, should be dissolved in oil or taken with a high-fat diet to increase absorption in the small intestine via the lymphatic system. Curry dishes, which contain curcuma as an essential ingredient, are also traditionally roasted with oils during cooking to promote the absorption of the lipophilic curry ingredients. 7
The main objective of this study was to compare the bioavailability of an oil/phospholipids (PL)-based curcuminoids formulation (mixture of a pure curcuminoids extract, lecithin, and olive oil) with the pure curcuminoids extract alone, both presented as soft gelatin capsules. The galenic formulation tested should not only increase bioavailability but also mimic the natural digestion process for fat-soluble substances such as curcuminoids, aiming at achieving their natural physiological absorption through the lymphatic system and not through the liver. This aimed to reflect a common diet in the form of a mixture of normal food ingredients and not to represent a new technology.
MATERIALS AND METHODS
Study design and objectives
The study was conducted as a randomized, controlled, cross-over mono-center study with a 14-day lasting washout period. In the study, 12 healthy men and women were enrolled. The clinical trial was performed to investigate the bioavailability of total curcuminoids from a new oil/PL-based curcuminoids formulation (tested product) in comparison with the pure curcuminoids alone (reference product). Plasma concentrations of total curcuminoids and tetrahydrocurcumin (THC) were measured over an 8-h period after administration of a single dose of the two study products, respectively. Tolerability and adverse events were also assessed.
The study was performed between January and March 2022 at the study site of the independent nutritional clinical research organization BioTeSys GmbH (Esslingen, Germany).
Study subjects
Healthy men and women between 18 and 50 years with a body mass index (BMI) between 19 and 30 kg/m2, nonsmoker, were eligible to participate in the study. The main exclusion criteria for study participation were relevant history or presence of any severe medical disorder potentially interfering with the study (e.g., malabsorption, chronic gastrointestinal diseases, heavy depression, diabetes, acute malignant disease in the past 3 years, except basal cell carcinoma of the skin), known allergy or hypersensitivity to ingredients of the study products, blood donation until 1 month prior to study start or during the study, significant changes in lifestyle or medication intake in the past 3 months, surgical intervention or surgical procedure such as bariatric surgery and clinically relevant abnormal laboratory values, and vital signs or findings following physical examination at screening, which could interfere with the foreseen study assessments.
Overall, 33 people were prescreened for study eligibility. Twelve of them (6 men and 6 women) were screened, enrolled in the study, and randomized to treatment. All 12 subjects completed the study successfully. Figure 1 depicts the subjects’ disposition.
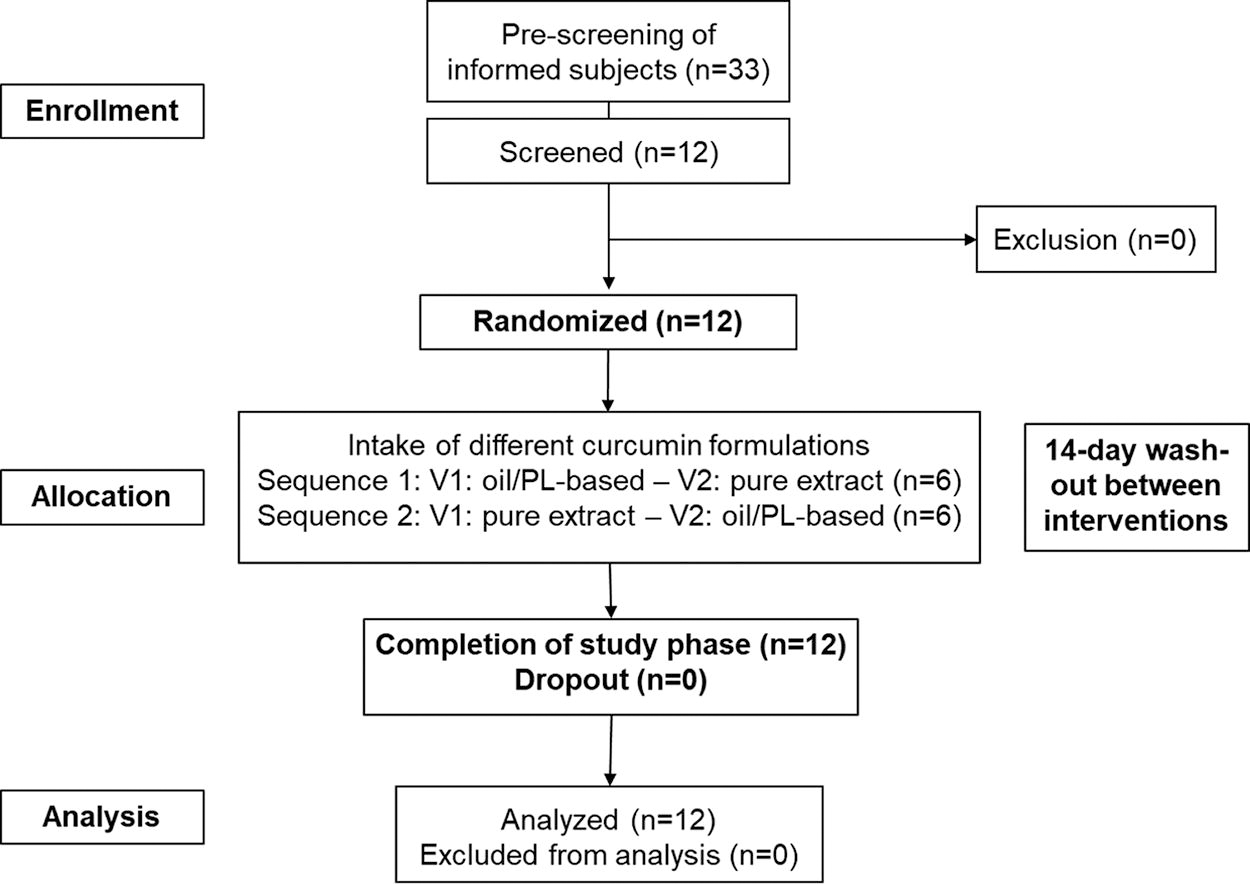
Subject’s disposition. V1: Visit 1; V2: Visit 2.
Composition of study products
The tested product (oil/PL-based) consisted of a pure curcuminoids extract, corresponding to 100 mg curcuminoids (curcumin 65–80%, demethoxycurcumin [DMC] 15–25%, bisdemethoxycurcumin [BDMC] 2–5%, minimum 95% curcuminoids, see Fig. 2), mixed and emulsified with soy lecithin and olive oil in a ratio of 1:1:8.
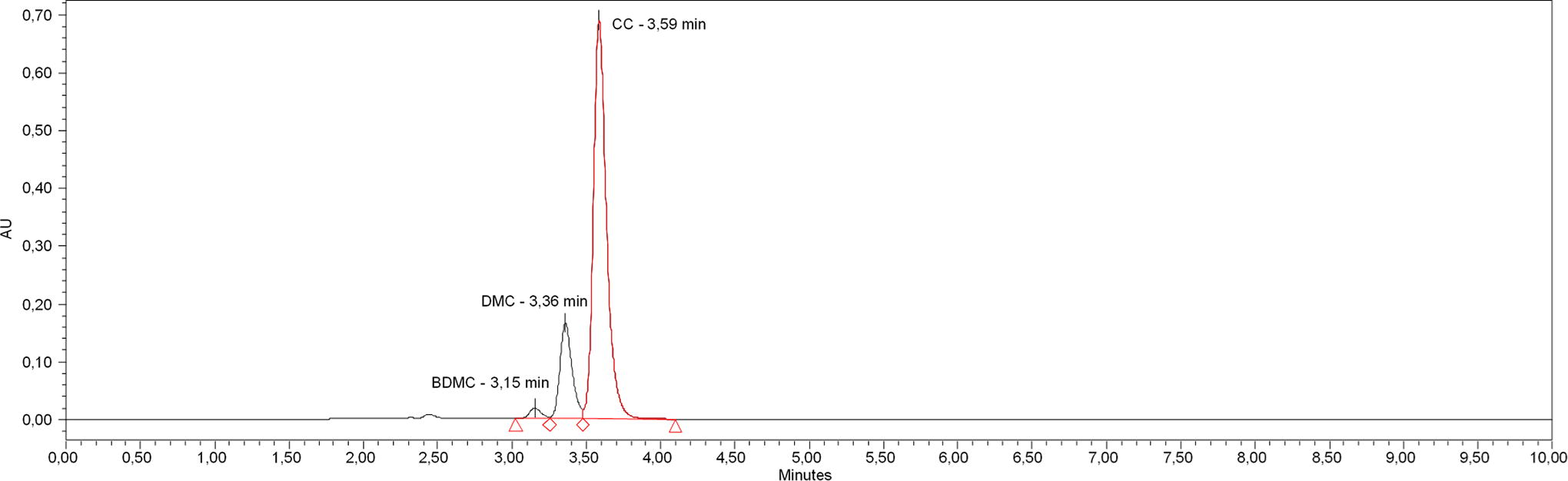
HPLC-UV/Vis-Chromatogram of the curcuminoid extract used at 425 nm wavelength. BDMC, bisdemethoxycurcumin; CC, curcumin; DMC, demethoxycurcumin; HPLC-UV, high-performance liquid chromatography-ultraviolet.
The reference product consisted of the pure curcuminoids extract. Both products were provided in a soft gelatin capsule.
Study products administration
The study products (test and reference) were administered orally with a single dose each of 200 mg curcuminoids (content per 2 capsules) with 250 mL water, after baseline blood sampling (0 h), following at least 12 h of fasting. The fasting period was continued for 4 h after intake of the study products. During the kinetic days, meals were standardized.
Methods for sampling and data collection
Blood samples were collected at defined timepoints over an 8-h period (before, 0.5 h, 1 h, 1.5 h, 2 h, 2.5 h, 3 h, 4 h, 6 h, and 8 h after intake of the study products). Total curcuminoids and their metabolite THC were detected in plasma samples. The pharmacokinetic parameters AUC, Cmax, and Tmax were determined to describe bioavailability of curcuminoids and its metabolite THC from the different formulations.
Tolerability and adverse events were assessed at the end of each kinetic day.
Analysis
Levels of total curcuminoids (sum of BDMC, DMC, and CC) and its metabolite THC were determined by ultra-performance liquid chromatography (UPLC) coupled to a mass spectrometer (Waters Xevo TQ-S micro mass spectrometer and Acquity H-class UPLC). For sample preparation, a 400 µL aliquot of blood plasma was diluted with 200 µL sodium acetate buffer (pH 4.5). Glucuronidase treatment with Glucuronidase from Helix pomatia (Merck KGaA, Darmstadt) corresponding to 4000 units of glucuronidase activity and 300 units of sulfatase activity per sample was performed 60 min at 37°C. The incubation was stopped by adding 20 µL concentrated acetic acid, cooling it down, and adding 1 mL water, 400 µL solvent mixture (50% acetonitrile in 0.1% formic acid in H2O), and 200 µL internal standard curcumin d6 (Cayman Chemical, Ann Arbor) into the solvent mixture. Samples were extracted by adding 3 mL of ethyl acetate using ultrasonic treatment followed by centrifugation. The supernatant was transferred to a glass vial, dried with nitrogen, reconstituted in 100 µL solvent mixture, and centrifuged at 16,000 g prior to injection to liquid chromatography-mass spectrometry (LC-MS/MS). Quantification of plasma samples was performed using matrix adjusted internal standard calibration with a range of 10–1000 ng/mL for THC, 2.5–280 ng/mL for CC, and 1–100 ng/mL for BDMC and DMC. All standards were purchased from Merck KGaA, Darmstadt.
Total curcuminoids are outlined as sum of CC and its derivates DMC and BDMC. Detailed instrument settings of the Waters Xevo TQ-S micro mass spectrometer and the Waters UPLC system Acquity H-Class are listed in Supplementary Data.
Statistical analysis
From concentration-time curves, pharmacokinetic endpoints were calculated. Statistical analysis was performed using the software GraphPad Prism (Version 5.04, San Diego, United States) and SPSS 24.0 (Version 24.0, IBM Statistics, New York, United States). Within figures, mean levels with 95% confidence interval (CI) are depicted.
The comparison of single pharmacokinetic data AUC and Cmax was based on log-transformed data.
Results were analyzed using a two-step analysis strategy to consider the cross-over analysis by strict separation of treatment effects from period effects. This was achieved via computing the treatment effects separately in two sequence groups formed via randomization. The difference between treatment effects was assessed by means of a Student t-test for independent samples using the intraindividual differences between the outcomes in both periods as raw data. In case of nonnormal distribution, the Wilcoxon rank sum test was applied. Differences between Tmax were evaluated by Wilcoxon rank sum test. In addition, the 90% CI of the geometric mean was evaluated for the ratio of the oil/PL-based curcuminoids formulation and the pure curcuminoids extract alone.
Ethical review and informed consent
The study was conducted in orientation with the International Conference on Harmonization-Good Clinical Practice (ICH-GCP) guidelines and the Declaration of Helsinki regarding the treatment of human subjects in a study. All subjects gave their informed consent before they participated in the study. The study protocol was reviewed by the Ethics Committee of Landesärztekammer Baden-Württemberg without any concern (approval number: F-2021-165). The study was registered in the German Clinical Trials Register (DRKS00027929).
RESULTS
Subject characteristics
Baseline characteristics of the study population are summarized in Table 1. Overall, study collective was healthy with no clinically relevant findings. Chronic use of medication was allowed only if considered not interfering with the study protocol.
Demographic Data and Baseline Characteristics (n = 12, at Screening)
Data shown as means with 95% CI.
BMI, body mass index; BP, blood pressure; GOT, glutamic oxaloacetic transaminase; GPT, glutamate pyruvic transaminase; LDL, low-density lipoprotein.
Concentration-time curve of total curcuminoids
Figure 3 summarizes the concentration-time curves of total curcuminoids in plasma as a mean value (± 95% CI) from all subjects at each timepoint. In all subjects, curcuminoid concentrations in plasma were below limit of detection in baseline samples.
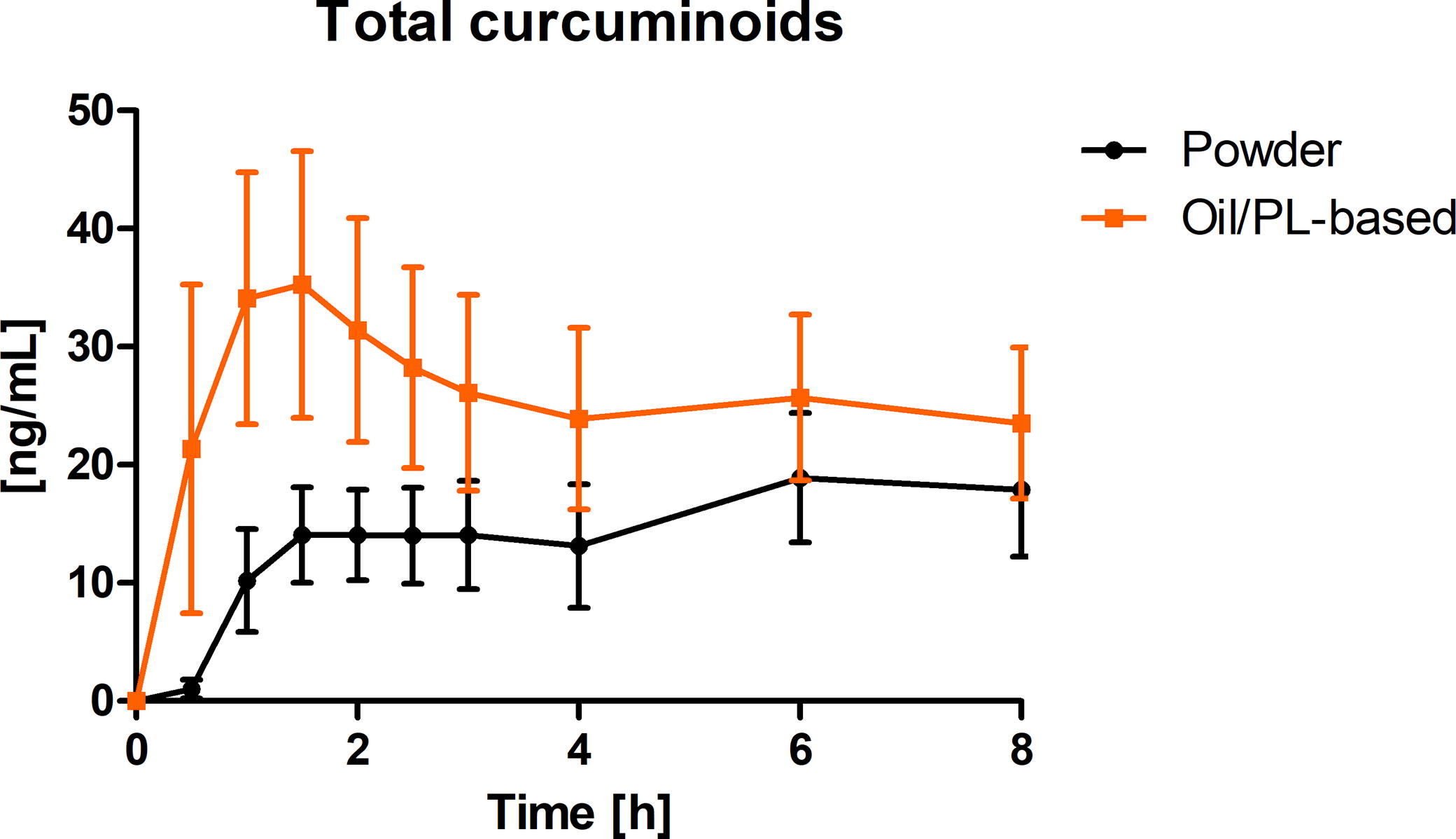
Total curcuminoids plasma concentration-time curve (ng/mL) after a single oral dose of 200 mg total curcuminoids administered as oil/PL-based curcuminoids formulation (orange) and the pure curcuminoids extract (powder) alone (black) depicted as summary curves of mean values at single timepoints (mean ± 95% CI) for all subjects (n = 12). PL, phospholipids.
The concentration-time profile of total curcuminoids shows a higher concentration in plasma for oil/PL-based curcuminoids formulation compared with the pure curcuminoids extract alone. For both study products, total curcuminoids concentrations were still elevated at the end of the observation period.
Pharmacokinetic parameters of total curcuminoids
The AUC0-8h of total curcuminoids after the intake of a single oral dose of the oil/PL-based formulation was significantly higher than the AUC0-8h after the intake of the pure curcuminoids extract (Fig. 4).
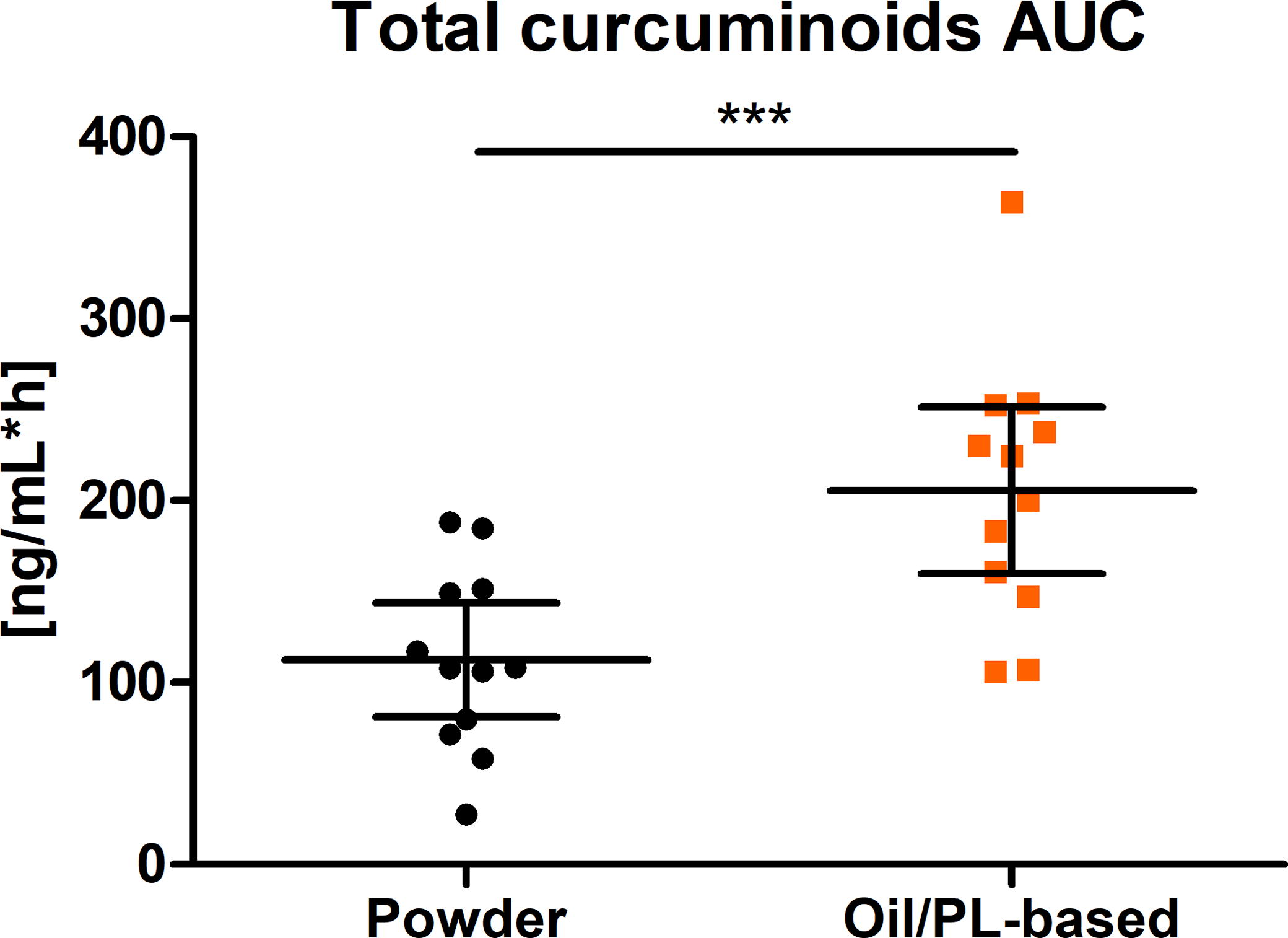
Distribution of AUC0-8h of total curcuminoids (ng/mL*h) (scatter plot with mean ± 95% CI); ***P = .0001; n = 12. PL, phospholipids.
AUC0-8h was 205.60 ng/mL*h and 112.50 ng/mL*h (1.8-fold, P = .0001) for tested and reference product, respectively (Table 2). These observations were also confirmed for Cmax (P = .0001). Cmax values were 21.16 ng/mL and 47.54 ng/mL for the pure curcuminoids extract alone and the oil/PL-based formulation, respectively. The ratio of geometric means of total curcuminoids for AUC was 1.93 (1.62–2.30) and that for Cmax was 2.41 (1.92–3.02).
Pharmacokinetic Parameters of Total Curcuminoids After a Single Dose of 200 mg Curcuminoids Administered as Pure Curcuminoids Extract (Powder) Alone and as Oil/PL-Based Curcuminoids Formulation
PL, phospholipids.
In addition, absorption of total curcuminoids from the oil/PL-based formulation was significantly faster compared with the absorption of the pure extract alone. Tmax was 6.0 h for the pure powder and 1.0 h for the new formulation (Table 2).
Concentration-time curve of THC
Figure 5 summarizes the concentration time curves of the curcuminoids metabolite THC in plasma as a mean value (± 95% CI) from all subjects at each timepoint. The concentration time profile of THC shows a higher concentration in plasma for the oil/PL-based curcuminoids formulation in comparison to the pure curcuminoids extract alone. Consistent with total curcuminoids, the plasma concentrations were still elevated at the end of the observation period.
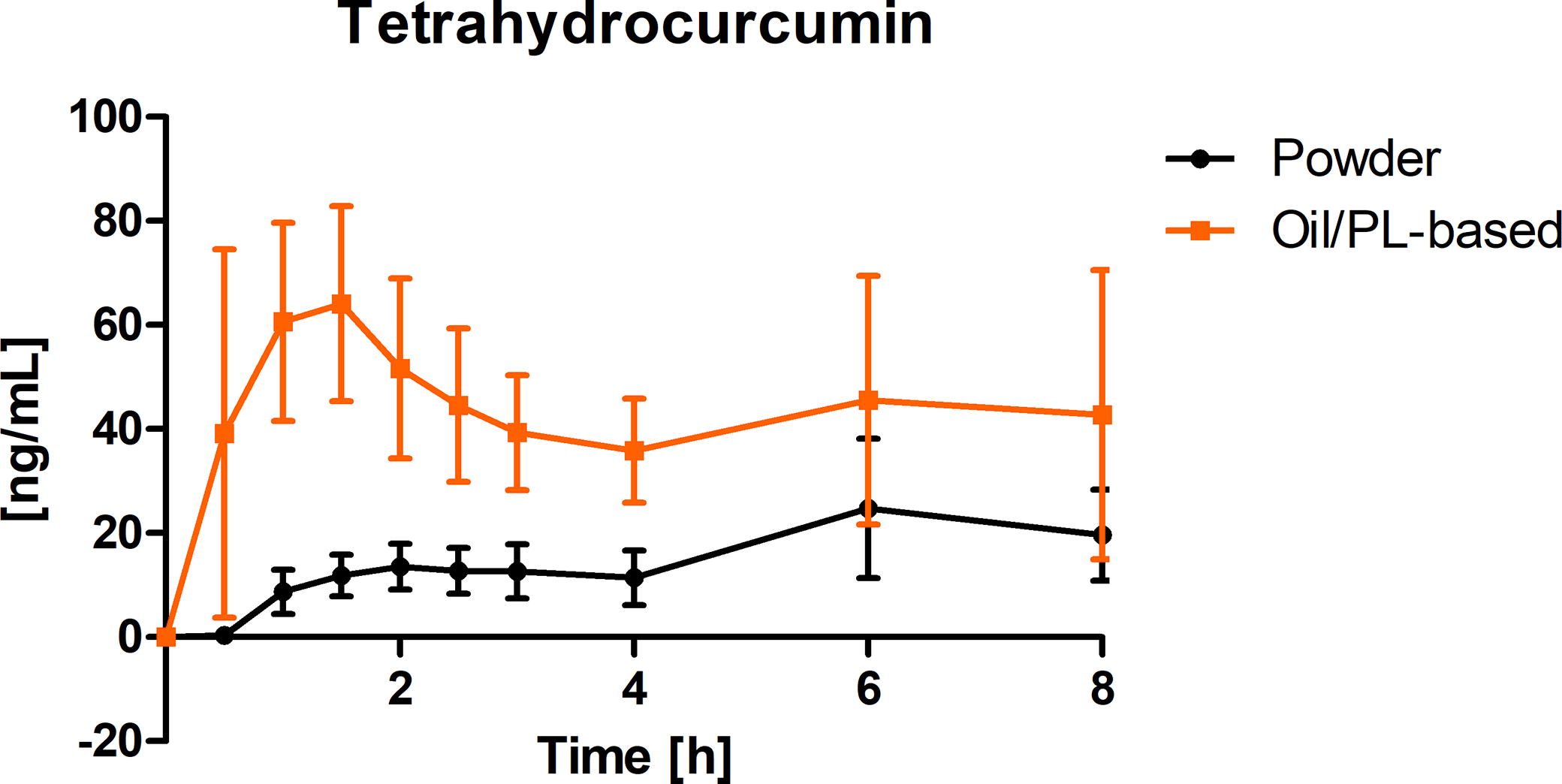
Tetrahydrocurcumin (THC) concentration time–curve in plasma (ng/mL) after a single oral dose of 200 mg total curcuminoids administered as oil/PL-based curcuminoids formulation (orange) and pure curcuminoids extract alone (black) depicted as summary curves of mean values at single timepoints (mean ± 95% CI) for all subjects (n = 12). PL, phospholipids.
Pharmacokinetic parameters of THC
The AUC0-8h of THC after the intake of a single oral dose of the oil/PL-based curcuminoids formulation was significantly higher than the AUC0-8h after the intake of the pure curcuminoids extract (P < .0001) (Fig. 6). AUC0-8h was 347.30 ng/mL*h and 118.90 ng/mL*h for tested and reference products, respectively.
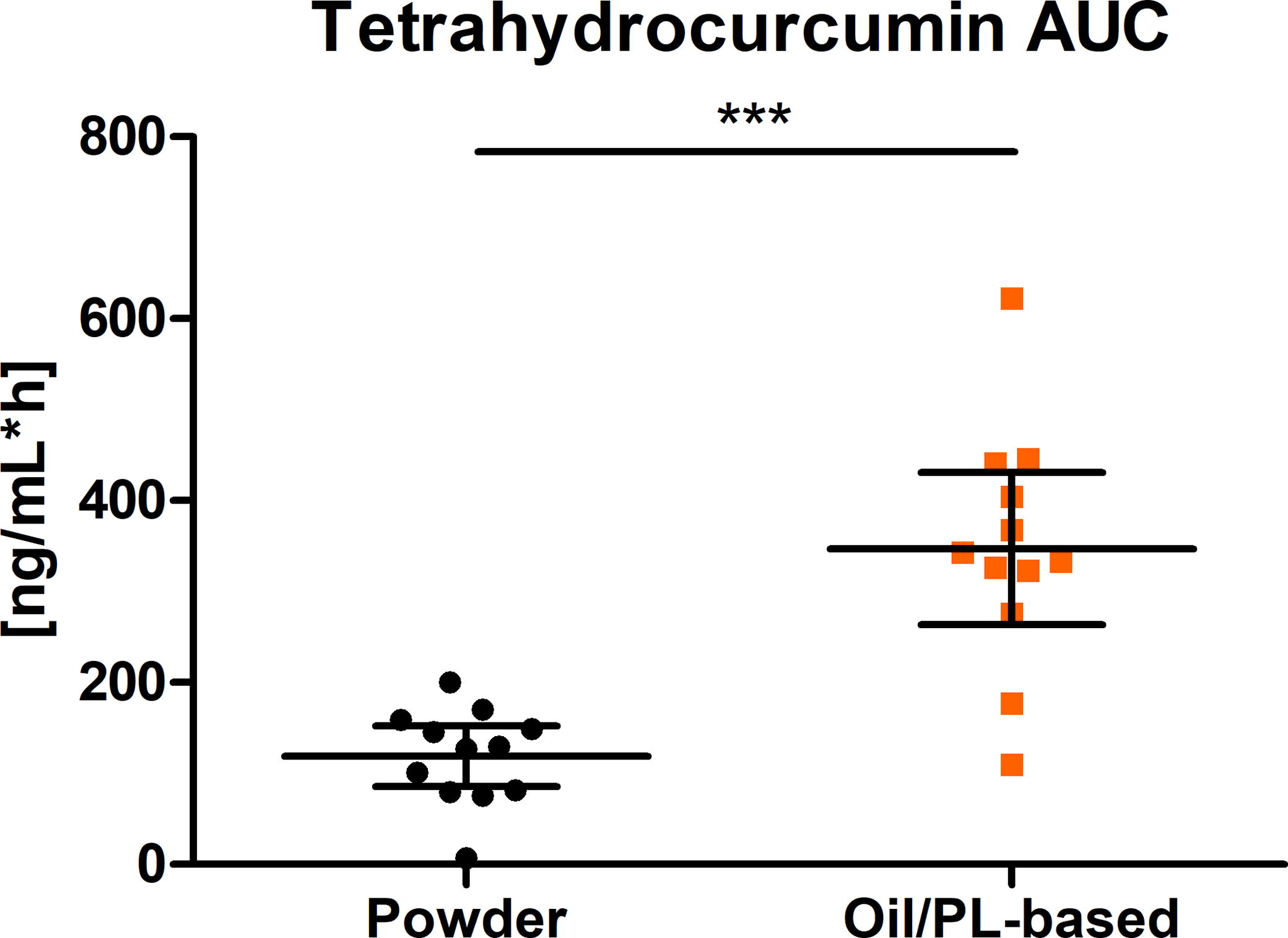
Distribution of AUC0-8h of THC (ng/mL*h) (scatter plot with mean ± 95% CI); ***P < .0001; n = 12. PL, phospholipids; THC, tetrahydrocurcumin.
The THC maximal plasma concentration (Cmax) was also significantly higher after the oil/PL-based formulation compared with the pure extract alone. Cmax values were 29.83 ng/mL and 96.69 ng/mL for the pure extract alone and oil/PL-based formulation, respectively (Table 3).
Pharmacokinetic Parameters of Tetrahydrocurcumin (THC) After a Single Dose of 200 mg Curcuminoids Administered as Pure Extract Alone and as Oil/PL-Based Formulation
PL, phospholipids.
The ratio of geometric means resulted in 3.31 (2.38–4.59) for AUC and 3.67 (2.57–5.52) for Cmax.
In addition, maximum concentrations of the metabolite THC were detected significantly earlier after intake of the oil/PL-based formulation in comparison to the pure extract alone (powder: 5.0 h and oil-PL based formulation: 1.5 h).
Safety
No treatment-related adverse events were reported during the study. Both study products were rated as “well tolerated” by all subjects.
DISCUSSION
In this study, a new oil/PL-based curcuminoids formulation was presented, attempting to reflect a diet-like absorption including a mixture of commonly used food ingredients. More specifically, a curcuma extract (curcuminoids) was mixed with olive oil and soy lecithin (as a natural emulsifier) and filled in a soft gelatin capsule. Through this composition, the curcuminoids were emulsified, aiming to achieve a physiologically natural absorption via the lymphatic system.
Numerous factors including low water solubility, poor intestinal permeability, fast metabolism, as well as rapid systemic elimination contribute to curcumin’s limited oral bioavailability. 8
The main objective of this study was to compare the in vivo bioavailability of an oil/PL-based curcuminoids formulation with a pure curcuminoids extract alone, both administered as a single dose of 200 mg curcuminoids in 12 healthy subjects. Results indicate a significantly higher bioavailability of curcuminoids (particularly its metabolite THC), but in a physiological range with 1.9-fold for total curcuminoids and 3.3 for THC following the intake of the oil/PL-based formulation compared with the pure extract alone. This is likely based on the fact that PL mimic the natural formation of micelles during digestion of fat-soluble ingredients, favoring in this way the intestinal uptake. 6
In addition, the uptake kinetic of total curcuminoids was significantly fastened with the oil/PL-based formulation, with a distinct increase already in the first half an hour following intake. As the focus of this study was on the comparison of formulations with main interest on the absorption phase, the observation phase was limited to 8 h. The limitation to 8 h restricts the comprehensive assessment also covering metabolization and elimination especially for the metabolite THC. Anyhow, main differences in absorption of formulations are covered in the selected observation time-window. For the pure extract alone, it is noteworthy mentioning that a second increase of total plasma curcuminoids after serving the standardized meal 4 h post-dosing was detected. This was most likely linked to the ingestion of fat-containing meals at this timepoint which probably increased the absorption of lipophilic curcuminoids in the intestinal mucosa. A similar increase but less pronounced was also observed for the oil/PL-based formulation.
The THC plasma concentration was even higher in comparison to total curcuminoids concentration, indicating a high metabolism of curcuminoids in the body. 6 Considering that the factor of AUC0-8h for THC in relation to AUC0-8h for total curcuminoids following the intake of the oil/PL-based formulation was 1.7, while this was only 1.1 for the pure curcuminoids extract alone, it may be deducted that the release of THC was highly influenced by the type of formulation, being the administered dosages of curcuminoids identical for both study products.
Some published studies have implied the crucial roles of PL in improving bioavailability of small molecules with poor bioavailability. In general, amphipathic PL complexes act as bioactive components that facilitate transport through gastrointestinal cells into the blood. The CC molecule is found to have high affinity toward biological membranes and tends to penetrate them rapidly to form dimeric biological complexes. Despite being a phenolic and poorly soluble compound, CC can link with PL (particularly phosphatidylcholine) by forming noncovalent adducts. At last, formation of these curcumin-PL complexes could enhance the CC pharmacokinetics by stabilizing at intestinal pH values and protecting CC from retro-Claisen hydrolysis, the major mechanism of degradation in water. In addition, pharmacokinetic study results revealed that a PL-curcumin complex implied significantly high plasma concentrations and was found to be more stable when compared with natural curcumin. 9 –12
The curcuminoids provided in an oily matrix mixed with PL tested in our study also underwent these metabolic processes, aiming to mimic physiological digestive conditions. The final study outcome suggested an improved impact on curcuminoids uptake and therefore on its bioavailability.
Currently, various other formulations techniques as well as galenic technologies have been developed to increase the bioavailability of curcumin, such as coadministration with piperine, polysorbates, or cyclodextrins. Moreover, various technological micelle formulations, incorporation into micelles, micro/nanoemulsions, nanoparticles, liposomes, solid dispersions, spray drying, and noncovalent complex formation with galactomannosides have been investigated with in vitro cell culture models, in vivo animal models, and humans. These advances in chemistry and technologies have provided the versatility and tools to develop a range of innovative CC formulations with considerable improvement in oral bioavailability and safety. 6,13,14
In the commercialized product Longvida®, particle-based technology (solid lipid CC particle [SLCP]) is used for preparing CC with a mixture of lecithin and PL from soy. This formulation was administered versus unformulated CC in healthy volunteers and, to determine its tolerability and dose-plasma concentration relationship, in late-stage osteosarcoma patients. Doses of 0.65, 2, 3, and 4 g of SLCP were evaluated. In healthy subjects, the mean peak concentration of CC is achieved by administration of 650 mg of SLCP 22.43 ng/mL, whereas plasma concentration of CC could not be detected when an equal amount of unformulated 95% curcuminoid extract was administered. Despite the high interindividual variability in pharmacokinetics and the observed nonlinear dose dependency, it has been shown that the SLCP technology boosts CC bioavailability. 8,9,15,16
The product Meriva® contains a curcumin-phosphatidylcholine complex made with soy lecithin (standardized mixture of natural curcuminoids and lecithin in a 1:2 ratio, plus microcrystalline cellulose to improve the physical state). In a randomized, double-blind, cross-over clinical study, the complex versus the standardized CC extract alone was tested. The complex has been shown to boost CC bioavailability by 29-fold over the standardized extract. 8,16,17
In these two studies, the administration of CC in combination with PL sources confirmed to improve curcuminoid uptake and bioavailability.
The incorporation of CC in other lipophilic matrices such as liposomes, phytosomes, and lipid micro- and nanoparticles or encapsulation with micellar surfactants or casein has also been tested. Many of these formulations have led to dramatic improvement in the absorption of CC in animal experiments, but only limited human data exist. 9
A combination with other adjuvants capable of increasing the absorption of curcumin, like piperine, quercetin, or turmeric oil showed to increase the bioavailability of CC in humans. 9
Adjuvants such as piperine are able to neutralize detoxification enzymes that are active in CC metabolism and thus contribute to improving the oral bioavailability of curcumin. On the other hand, it has been argued that this improvement comes at the expense of an increased potential of interaction with mainstream drugs, since piperine is an inhibitor of phase-1 and phase-2 xenobiotic metabolizing enzymes, and CC has also been shown to inhibit various classes of cytochromes (including CYP3A4) as well as P-glycoprotein activity. 9
Only a few exact comparisons (limited to the comparison of 2–4 products at a time) have been published for head-to-head evaluations within the same study. 18 –20 Comparisons of bioavailability among different studies are often inexact owing to dissimilarities in the dose, duration of treatment, varying clinical study design, formulation type, the method used for analysis, pharmacokinetic parameters measured and population disparity, as well as other relevant details of each formulation.
Despite these drawbacks, a varying range of enhanced absorption and bioavailability of curcuminoids is reported in literature, revealing clear trends, with several products exhibiting much higher bioavailability versus standard unformulated curcumin, in some cases also far exceeding the physiological amounts, potentially raising safety concerns. 13 –15,21
In this study, an enhanced bioavailability of curcuminoids was achieved by combining the pure curcuminoids extract with commonly used food ingredients such as lecithin and oil. No new technology was applied. Curcuminoids with their well-established safety profile are recognized for their multiple bioactive properties and health benefits underlining the popular dietary use of curcuma extracts. 2,7,22
Traditionally, curcuma is used in food preparation in combination with vegetable oils or lecithin-rich ingredients (as sources of PL), e.g. eggs (as served in the 4-h post-dosing standardized meal). 6,17 It is generally recognized that the concomitant intake of food can affect compounds pharmacokinetic profiles by altering dissolution, absorption, metabolism, and excretion behavior. Dietary status can therefore also dramatically influence the bioavailability of curcumin. 23 The approach adopted in this study to administer an oil/PL-based formulation, attempted to mimic the physiological digestive process to overcome the limitations of curcuminoid bioavailability of pure extracts.
CONCLUSION
The oil/PL-based curcuminoids formulation tested was intended not only to increase bioavailability but also to mimic the natural digestion process of fat-soluble substances such as curcuminoids. The formulation aimed to reflect a common diet in the form of a mixture of normal food ingredients and did not represent a new technology. The data indicate a significantly higher bioavailability of the oil/PL-based curcuminoids formulation mimicking the natural formation of micelles during digestion of fat-soluble ingredients, compared with the pure curcuminoids extract alone. Thus, the oil/PL-based formulation is deemed suitable and appropriate to be included in food supplements for its higher in vivo bioavailability, good tolerability, and for the various health benefits. To what extent the patterns of the oil/PL-based formulation can be transferred to other fat-soluble plant ingredients with low bioavailability needs to be investigated in further studies.
Footnotes
ACKNOWLEDGMENT
The authors are grateful to the study participants for their valuable contribution to this research.
AUTHORS’ CONTRIBUTIONS
T.W., T.K., A.K., and C.S.: conceptualization. T.K. and T.W.: methodology. T.W. and C.S.: clinical study conduct and analysis of data. R.W.: curcumin analysis. All authors contributed to writing, discussion of results, critical review, and editing. All authors have read and agreed to the published version of this article.
AUTHOR DISCLOSURE STATEMENT
T.K. and A.K. declare competing interests as employees of Apomedica Pharmazeutische Produkte GmbH. All other authors declare no conflict of interest.
FUNDING INFORMATION
This research was funded by Apomedica Pharmazeutische Produkte GmbH.
SUPPLEMENTARY MATERIAL
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
