Abstract
Introduction
l-theanine is a nonproteinogenic amino acid contained in green tea
1
and mushrooms.
2
Among the different tea varieties in Japan, gyokuro and matcha contain more
The epidemiologic Nakajima 8 and Tsurugaya studies 9 in Japan indicated that green tea may improve age-related cognitive impairment. In addition, the Hisayama study warned about a recent increase in the prevalence of Alzheimer's disease (AD). 10,11 Prevention of AD is an important problem worldwide, and it is necessary to seek a solution.
Green tea contains several substances that affect cognitive function, including caffeine,
12,13
The purpose of this study was to clarify whether the intake of
Materials And Methods
The study was conducted at the Tokyo Skytree Station Medical Clinic (Tokyo, Japan) and was approved by the Research Ethics Committee of Nihonbashi Egawa Clinic (Tokyo, Japan; Approval No.: food-18071704). The study was conducted in accordance with the Declaration of Helsinki from August 8, 2018 to December 6, 2018. The study was registered with University Hospital Medical Information Network (Tokyo, Japan).
Test food
Subjects
Sixty-nine Japanese men and women certified as healthy by a physician based on hematological and biochemical blood test results and with self-assessed declined cognitive function were initially enrolled.
Of these, subjects with a Mini Mental State Examination-Japanese version (MMSE-J) score ≥24 were included, while those who had food allergies or were taking medication or undergoing treatment were excluded.
The subjects participated in the study on their own initiative after receiving a full explanation of the study.
Study design
A double-blind, randomized placebo-controlled parallel group study was conducted. The primary end points were the results of MMSE-J and Cognitrax, and the secondary end points were blood levels of amyloid β 1–40 [Aβ (1–40)], Aβ 1–42 [Aβ (1–42)], secreted form of amyloid-β precursor protein α (sAPPα), amyloid-β precursor protein 770 (APP770), and brain-derived neurotrophic factor (BDNF). A computer-generated stratified randomized schema (Huma R&D Corp., Tokyo, Japan; Contract Research Organization) was used to assign the subjects to either the placebo or the
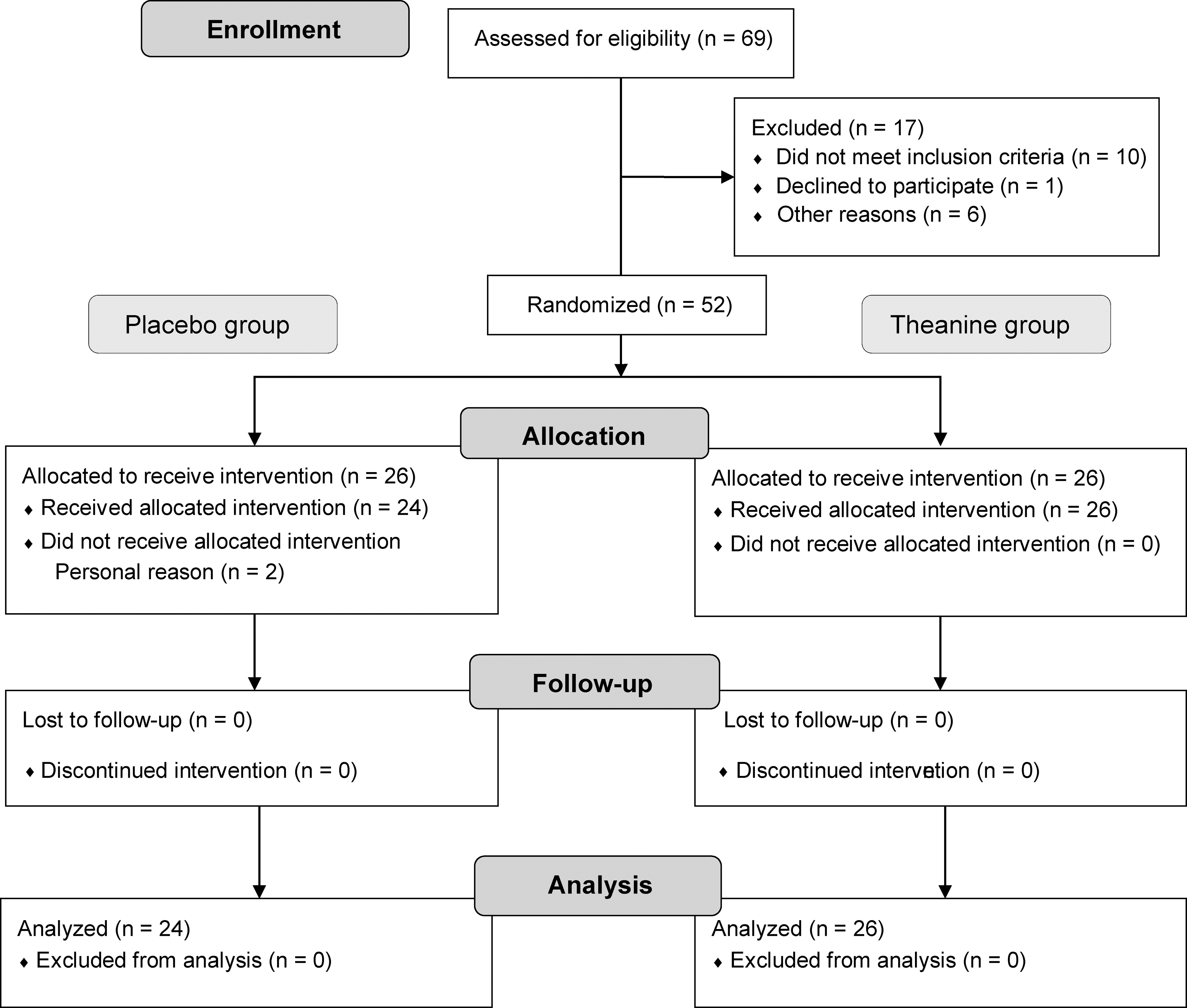
Study flow diagram.
Subjects took one capsule per day of placebo or
Evaluation items
The evaluation items are shown in Table 1. On the day of the single-dose study, the Cognitrax test was started ∼50 min after capsule intake. Except for taking the capsule before undergoing the tests, the tests were performed in the same order at week 12. Hematologic tests and biochemical blood parameter measurements were conducted at baseline and 12 weeks as a safety evaluation at SRL, Inc. (Tokyo, Japan).
Evaluation Scheme of the Clinical Trial
On the day of the test, evaluations marked with circles were implemented. Tests were carried out in the order listed in the table.
Blood sampling was performed for hematologic tests (white blood cell count, red blood cell count, hemoglobin concentration, hematocrit, platelet count, mean corpuscular volume, mean corpuscular hemoglobin, and mean corpuscular hemoglobin concentration) and evaluation of biochemical blood parameters (total protein, triglycerides, total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, gamma-glutamyl transpeptidase, lactate dehydrogenase, uric acid, urea nitrogen, total bilirubin, albumin, creatinine, fasting blood glucose, and glycated hemoglobin).
MMSE-J, Mini Mental State Examination-Japanese version.
Mini Mental State Examination-Japanese version
MMSE-J (Nihon Bunka Kagakusha Co., Ltd., Tokyo, Japan) is a Japanese version of MMSE, 22 and its validity and test–retest reliability have been confirmed in Japan. 23 The test consists of 11 items: time orientation, location orientation, memorization, attention and calculation, recall, naming, repetition, three-stage command, reading, writing, and copying, and it is evaluated with the total score. Two tests of attention and calculation were used: the backward spelling task and the serial sevens task, respectively. The score for the backward spelling task was used for allocation of subjects into the placebo or theanine group. 22,23
Cognitrax test
Cognitrax 24 is a test to evaluate cognitive function, developed by the U.S. company CNS Vital Signs (Morrisville, NC, USA). It comprises 10 test items with accompanying instructions. It measures both reaction time and number of responses. Reaction time is measured in milliseconds. The test items were ordered as follows: verbal memory (VBM), visual memory (VIM), finger tapping test (FTT), symbol digit coding (SDC), Stroop test (ST), shifting attention test (SAT), continuous performance test (CPT), perception of emotions test (POET), nonverbal reasoning test (NVRT), four-part CPT (FPCPT), VBM, and VIM. These items assess various cognitive functions—VBM and VIM assess memory; ST, SAT, CPT, and FPCPT Parts 1 and 2 assess attention; POET assesses facial expression recognition; FPCPT Parts 3 and 4 assess working memory; SDC and NVRT assess visual information processing; and FTT assesses motor function.
The first VBM and VIM performed are indicative of immediate memory, while the last is indicative of delayed memory. There were ∼50 min between the first-performed VBM and VIM and the last-performed VBM and VIM. Details of the test are shown in Table 2.
Cognitrax Test Instructions
SDC, symbol digit coding; VBM, verbal memory; VIM, visual memory.
Biomarkers related to dementia
On the day of the test, the subjects were restricted from eating from 6 h before arriving at the hospital until the completion of the test. Serum blood collection tubes were used to estimate BDNF levels, while ethylenediamine tetra-acetate disodium tubes were used for Aβ (1–40), Aβ (1–42), sAPPα, and APP770 measurements. The blood was centrifuged at 3000 rpm (Kokusan Co., Ltd., Tokyo, Japan, H-19Ra). The measurement was performed using kits with the following dilutions: the blood sample for Aβ (1–40) was diluted 20 times with human Aβ (1–40) Full Length (FL) Assay Kit-IBL; the blood sample for Aβ (1–42) was diluted 4 times with human Aβ (1–42) (FL) Assay Kit-IBL; the blood sample for sAPPα was diluted four times with sAPPα (highly sensitive) Assay Kit-IBL; the blood sample for APP770 was diluted 50 times with human APP770 Assay Kit-IBL; and the blood sample for BDNF was diluted 20 times with human BDNF Enzyme-linked Immunosorbent Assay Kit (Quantikine-R&D Systems). Any measured value below the kit range was excluded due to inaccuracy. The measurement was carried out by Skylight Biotech, Inc. (Akita, Japan). The results are shown in Table 3.
Effects of l-Theanine on Biomarkers
Values are presented as mean ± SD.
Aβ, amyloid-β; APP770, Aβ precursor protein 770; BDNF, brain-derived neurotrophic factor; sAPPα, secreted form of Aβ precursor protein α; SD, standard deviation.
Statistical analysis
The values are presented as mean ± standard deviation. Normality was tested using the Shapiro–Wilk test. The unpaired t-test or Mann–Whitney U test was used to calculate P values, with Bonferroni correction (P < .05/3 = 0.017). This was performed at baseline, single-dose test, and 12 weeks. Data were analyzed using SAS 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
The final analysis comprised 24 subjects in the placebo group (11 men and 13 women; average age, 57.9 ± 6.3 years) and 26 subjects in the theanine group (12 men and 14 women; average age, 57.7 ± 4.8 years).
MMSE-J (interactive test)
There was no significant difference in MMSE-J scores between the theanine and placebo groups before and after the intervention (placebo group: before the intervention, 27.7 ± 1.8; after the intervention, 28.3 ± 1.5; theanine group: before the intervention, 27.7 ± 1.4; after the intervention, 28.0 ± 1.6).
Cognitrax test (PC-based cognitive function test)
Attention task
In the single-dose study, ST (Part 1) reaction times were significantly lower in the theanine group than in the placebo group (Table 4). Therefore, a single dose of
Performance on Attention Tasks
Values are presented as mean ± SD.
P < .05/3 = 0.017 versus placebo group. P values were calculated using the unpaired t-test or Mann–Whitney U test and Bonferroni correction.
CPT, continuous performance test; FPCPT, four-part continuous performance test; SAT, shifting attention test; ST, stroop test.
Working memory tasks
In the single-dose FPCPT (Part 4), correct responses were significantly higher and omission errors were significantly lower in the theanine group than in the placebo group. In FPCPT (Part 4), compared to baseline, the theanine group had a significantly lower change in average incorrect response time in the single-dose study than the placebo group (Table 5). After single dose ingestion,
Performance on Working Memory Tasks
Values are presented as mean ± SD.
P < .05/3 = 0.017 versus placebo group. P values were calculated using the Mann–Whitney U test and Bonferroni correction.
Performance on Memory Tasks
Values are presented as mean ± SD.
Performance on the Facial Expression Recognition Task
Values are presented as mean ± SD.
Performance on Visual Information Processing Tasks
Values are presented as mean ± SD.
Performance on Motor Function Tasks
Values are presented as mean ± SD.
FTT, finger tapping test.
Blood biomarkers
No significant difference was observed between the placebo and theanine groups (Table 3).
Discussion
This study evaluated the effects of
A cup of green tea contains about 25 mg of theanine.
25
Kuriyama et al. reported that people who drink green tea have less cognitive dysfunction.
9
In the previous report, the amount of theanine that led to an improvement in attentional function was 50.3 mg. In this study, the dose of theanine was set at 100.6 mg for the purpose of clearly showing the effect of theanine alone. A single dose of
It is interesting that caffeine,
26
which acts as a stimulant for neural activity, and
Studies that explored brain activity have previously reported that
According to a study by Baddeley,
33
working memory is composed of three factors: a phonological loop for language information processing, a visuospatial sketch pad for visuospatial information processing, an episodic buffer, and a central executive system that integrates the other three parts. The central executive system is considered to be related to attention control. The effects observed in the two-back task (FPCPT, Part 4) are the results of
Because attention and working memory are closely related,
34
it is difficult to consider them separately. It is therefore necessary to examine, in more detail, attentional function and working memory, which were found to be improved by
The limitations of this study involve subject age and cognitive function status. This study was conducted in middle-aged and older subjects who were aware of a decline in their cognitive function. Previous studies tested elderly people, including those who had mild cognitive impairment
35
or vascular dementia,
36
to verify whether green tea powder could improve cognitive function. In the future, a comparative study on the preventive or restorative effects of
Footnotes
Acknowledgments
The authors gratefully acknowledge the individuals who participated in the study and the clinical research coordinator, Huma R&D Corp. The authors thank Editage for English language editing.
Author Disclosure Statement
ITO EN, Ltd. covered the testing costs and provided the test food as the study sponsor. Huma R&D Corp., Contract Research Organization, conducted subject recruitment, selection, allocation, observation, and data collection. Kureha Special Laboratory Co., Ltd. conducted statistical analysis. T.K. was the principal investigator for this study and was contracted by Huma R&D to conduct the study. The study was performed by a third party. The remaining authors report that no competing financial interests exist.
Funding Information
The authors have no conflicts of interest directly relevant to the content of this article.
