Abstract
Sex hormone replacement therapy provides several advantages in the quality of life for climacteric women. However, estrogen-induced cell proliferation in the uterus and mammary gland increases the risk of cancer development in these organs. The lower incidence of mammary cancer in Asian women as compared with Western women has been attributed to high intake of soy isoflavones, including genistein. We have previously shown that genistein induces an estradiol-like hypertrophy of uterine cells, but does not induce cell proliferation, uterine eosinophilia, or endometrial edema. It also inhibits estradiol-induced mitosis in uterine cells and hormone-induced uterine eosinophilia and endometrial edema. Nevertheless, genistein stimulates growth of human breast cancer cells in culture; therefore, it is not an ideal estrogen for use in hormone replacement therapy (HRD). The present study investigated the effect of another soy isoflavone, daidzein (subcutaneous, 0.066 mg/kg body weight), in the same animal model, and its effect on responses induced by subsequent treatment (1 h later) with estradiol-17β (E2; subcutaneous, 0.33 mg/kg body weight). In addition, we investigated the effects of daidzein (1 μg/mL) or E2 on the growth of human breast cancer cells in culture. Results indicate that daidzein stimulates growth of breast cancer cells and potentiates estrogen-induced cell proliferation in the uterus. We suggest caution for the use of daidzein or formulas containing this compound in HRD. Future research strategies should be addressed in the search for new phytoestrogens that selectively inhibit cell proliferation in the uterus and breast.
Introduction
M
It is generally accepted that HRT with estrogen in postmenopausal women, apart from many beneficial therapeutic effects, increases the risk of breast or endometrial cancer, at least after a prolonged treatment. 1 –3 It was also reported that HRT with a combined estrogen–progestin regime also increased breast cancer risk. 4,5 In addition to many other possible mechanisms, this effect seems to be caused by target organ cell proliferation stimulation by estrogens used in HRT, 6 which is similar to the action of estrogen-related receptor alpha (ERRα) that also induces cell proliferation in a ligand-independent manner. 7 Therefore, the finding of a compound mimicking beneficial responses to estrogen but not inducing cell proliferation is desirable.
If all responses to estrogen were mediated by the same mechanism and all estrogen receptors (ERs) were identical, as was first proposed for the cytosol-nuclear, receptor-mediated genomic responses, 8 there would be scarce possibilities to selectively induce therapeutically convenient responses to estrogen, but not those at risk (cell proliferation). In contraposition to the possibility just mentioned, the existence of several ERs involved through different mechanisms of action in separate responses to estrogen (vide infra) allows the possibility to induce beneficial responses and selectively inhibit those considered untoward effects. In fact, apart from the classical cytosol-nuclear ERs, 8 now α-ERs, 9 –11 the existence of additional ERs was proposed: eosinophil leukocyte ERs, 12 –14 membrane ERs, 15,16 cytoplasmic and nuclear type II ERs, 17 specific antiestrogen receptors, 18 and β-ERs. 9 –11 The finding of the estrogen-binding proteins just mentioned—presumably ERs—in the uterus opens the possibility of a dissociation of responses by agents displaying selective agonist or antagonist action on some but not all receptors. This dissociation was reported under several experimental conditions in the rat uterus. 13,14,19 –23
The lower incidence of several hormonally dependent cancers in Asian women than in Western women 24 was attributed to the higher amount of diet phytoestrogens—including soybean products—consumed by Eastern Asian women, 24,25 and suggested their possible role in breast cancer prevention. 26 For this purpose, several plant extract preparations were registered as pharmaceuticals or nutraceuticals recommended for hormone-dependent cancer prevention in climacteric women. 27,28 Among them, in the pharmaceutical market, Glycine max, Cimicifuga racemosa, Vitex vinifera, and other estrogenic isoflavone-rich preparations containing genistein (and its glucoside genistin), daidzein (and its glucoside daidzin), and other phytoestrogens were available. 29 –32
We had previously reported that one of the phytoestrogens present in these preparations, genistein, induced several genomic responses to estrogen in the rat uterus, but induced neither uterine cell proliferation nor uterine eosinophilia and endometrial edema; therefore, it could be considered a selective estrogenic agonist. 33 Further, genistein almost totally inhibited estrogen-induced cell proliferation and partially inhibited uterine eosinophilia and edema; therefore, it can also be considered a selective estrogenic antagonist. 34
In contraposition to convenient effects of genistein in the uterus, which can be used for endometrial cancer prevention or treatment, 34 potential adverse effects were reported for the mammary gland. Although some reports suggest that genistein (or high soy bean isoflavones intake) reduces breast cancer risk 26,35 and decreases mortality and recurrence of the disease in treated patients, 27 many other reports indicate that genistein, at concentrations 0.1–10 μM or higher, stimulates the proliferation of human breast cancer MCF-7 cells in culture, 36,37 an effect which is mediated by alpha ERs. 37 Similarly, genistein stimulates estrogen-dependent human breast tumor growth 38 and negates the effectiveness of tamoxifen treatment. 39 The stimulation of MCF-7 cells growth in culture by genistein at concentrations of 7.5 μg/mL, or by soy extract at 5.5 μg genistein/mL, was recently confirmed in our laboratory.* All of these findings discourage the use of genistein in the treatment of climacteric syndrome.
Daizdein is another phytoestrogen present in G. max and in several plant species commonly used to obtain phytoestrogenic isoflavone-rich preparations for HRT in climacteric women. This report describes the effects of daidzein in the rat uterus as well as in human breast cancer cells.
Materials and Methods
Experimental animal procedures
The experimental protocol was approved by the local ethics committee. Sprague–Dawley rats were breaded and maintained in the vivarium of the University of Chile Medical School. The prepubertal rat model was chosen, as the very low endogenous estrogen levels in control animals assure absence of estrogenic responses; at that age, all ERs and mechanisms were reported as fully responding to hormone stimulation. 40 Ten rats were used for each experimental condition. Female prepubertal (21-day-old) animals were pretreated subcutaneously with high-performance liquid chromatography standard quality daidzein (0.066 mg/kg body weight; Sigma-Aldrich, St. Louis, MO, USA) or control vehicle (0.3 mL 1:9 ethanol/saline), and 1 h later, they were treated with estradiol-17β (E2; 0.33 mg/kg body weight; Merck, Whitehouse Station, NJ, USA) or control vehicle. The dose of E2 was chosen from former studies in the same prepubertal rat model, assuring the maximal responses to hormone stimulation for all analyzed parameters of estrogen action in the uterus. 41 –43 The daidzein dose was chosen to assure an approximation to E2 molar concentrations inducing the maximal response to hormone stimulation (0.01–0.3 mg/kg). Uteri were excised under ether anesthesia 6 or 24 h after treatment and fixed in neutral formalin for further histological processing.
Histological, histochemical, and morphometrical procedure
Each uterine formalin-fixed and dehydrated horn was cut in three pieces (superior, medium, and caudal), which were paraffin embedded together in a single paraffin block, so that 5 μm-thick uterine cross sections from these three pieces could be observed together in the same histological slide and evaluated. For each animal, one group of hydrated uterine cross sections was stained 1–3 min in hematoxylin, washed in several changes of tap water, transferred to a saturated lithium carbonate solution, and kept for 1 min. Subsequently, they were stained in 1% eosin Y aqueous solution, washed quickly in distilled water, and dehydrated in a graded series of ethanol and xylene. 44 The stained slides were microscopically evaluated for eosinophil quantification, 45,46 mitoses counting in the various uterine histological layers, 42,43 and endometrial edema morphometrical estimation. 22,44
Quantification of estrogenic responses
The following estrogenic responses were quantified: uterine eosinophilia, percentage of eosinophils according to the distribution in different uterine histological layers and to their degree of degranulation; edema in deep endometrial stroma; and increases in cell size and number of mitotic figures in uterine luminal epithelium, glandular epithelium, and myometrium. The various parameters of estrogen stimulation were evaluated at the times during which their maximal response to E2 occurs. 22,44,45 Uterine eosinophilia reaches maximal response at 6 or 24 h; endometrial edema reaches maximal response at 6 h and declines thereafter; and uterine luminal epithelial, stromal, and myometrial cell proliferation reaches maximal level responses at 24 h after hormone treatment. To allow a comparison between the effects of daidzein among the different parameters of estrogen stimulation, all responses were expressed as % of response to E2, that is, the value of vehicle-treated animals was considered 0% response, and the value of response to E2—100% response.
For each animal, uterine eosinophilia 45 was assessed in 30 uterine sections, distributed along the uterus (proximal, medial, and caudal); eosinophils were classified according to their location within the different uterine histological layers and to their degree of degranulation. 42,46 Edema in deep endometrial stroma 44 was evaluated by counting the number of nuclei in thirty-six 1054 μm2 areas delimited by a standard circle located in the ocular piece of the microscope. As shown earlier, 44 an increase in the reciprocal value of cell density (a decrease in cell counts within a standard area) in a location containing a few cells and mainly extracellular space reflects edema, as an increase in its volume, but not in the cellular volume, results in an increase in the reciprocal value of cell density. Estrogen-induced mitotic response was evaluated as an increase in the number of mitotic figures in luminal epithelium, glandular epithelium, and myometrium, and quantified for each animal in 18 uterine cross sections by considering all three uterine pieces. 42,43 Cell hypertrophy (increase in cell size) was evaluated morphometrically in the above cell-types. 22,33,42,44
Human breast cancer cell culture procedure
The human breast cancer cell lines were obtained from American Type Culture Collection (Rockville, MD, USA). MCF-7 cells (expressing both ERs α and β) and MDA-MB-231 cells (only expressing ERs β, considered hormone-independent cells) were grown in Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal calf serum, 100 U/mL penicillin, 100 μg/mL streptomycin, and 1 mM glutamine. Cells were seeded into 96-well microtiter plates in 100 μL at a plating density of 3×103 cells per well. After 24 h incubation at 37°C under a humidified 5% carbon dioxide to allow cell attachment, the cells were washed with phenol red-free and the medium was changed to phenol red-free DMEM supplemented with 4% dextran-charcoal-stripped fetal calf serum. Then, the cells were treated with vehicle, E2 (5 μg/mL; Sigma-Aldrich, Cat. E8875, molecular weight=272.4), daidzein (1 μg/mL; Sigma-Aldrich, Cat. D7802, molecular weight=254.24), or both, and incubated for 72 h under the same conditions. Stock solutions of compounds were prepared in ethanol, and the final concentration of this solvent was maintained constant at 1%. Control cultures received 1% ethanol alone.
In vitro growth inhibition assay. The sulforhodamine B assay was used according to the method of Skehan et al. 47 with some modifications. 48 Briefly, the cells were set at 3×103 cells per well of a 96-well, flat-bottomed 200 μL microplate. Cells were incubated at 37°C in a humidified 5% CO2/95% air mixture and treated with the compounds at different concentrations for 72 h. At the end of drug exposure, cells were fixed with 50% trichloroacetic acid at 4°C. After washing with water, cells were stained with 0.4% sulforhodamine B (Sigma-Aldrich), dissolved in 1% acetic acid (50 μL/well) for 30 min, and subsequently washed with 1% acetic acid to remove unbound stains. Protein-bound stains were solubilized with 100 μL of 10 mM unbuffered pH 10.2 Tris base, and the cell density was determined using a fluorescence plate reader (wavelength 540 nm).
Statistics
According to previous studies 42 using the Tukey test of additivity, 49 for some parameters of estrogen stimulation parameters of estrogen stimulation, the data need to be submitted to logarithmic or square-root transformation to normalize distribution. Accordingly, uterine eosinophilia numbers were subjected to log transformation. Transformed (uterine eosinophilia) and non-transformed data (the remaining parameters of estrogen stimulation in the uterus) were subjected to further statistical analysis. Since multiple comparisons were performed between the different experimental conditions, transformed and non-transformed data were subjected to the least significant difference a posteriori test (LSD test): Two-tailed P-values of <.05 were taken to indicate statistical significance. The common variance needed for this test was estimated from a one-way unbalanced analysis of variance; in all reported results, it was P<.05. In uterine eosinophil degranulation and distribution proportion studies, the χ 2 statistic was chosen to evaluate differences between the proportions. Results on eosinophil degranulation and distribution within the various uterine histological locations were not considered in control animals without E2 treatment, due to the extremely low number of eosinophils observed in this experimental condition.
Results
Daidzein does not induce cell hypertrophy in uterine luminal epithelium or glandular epithelium, as E2 does, but it strongly potentiates the response to E2 in the luminal epithelium (Fig. 1). The apparent increase in glandular epithelium under the effect of daidzein pretreatment in E2-treated animals is not statistically significant (P=.063). As shown in Figure 2, in the myometrium, daidzein but not E2 causes an important increase in cell volume at 6 h of treatment. Further, daidzein pretreatment followed by E2 treatment induces an important increase in cell volume, 6 h after treatment with E2, which is not observed in animals treated with E2 alone. At 24 h after treatment, daidzein by itself does not induce any change in cell volume and that daidzein pretreatment does not modify the response to E2 treatment.
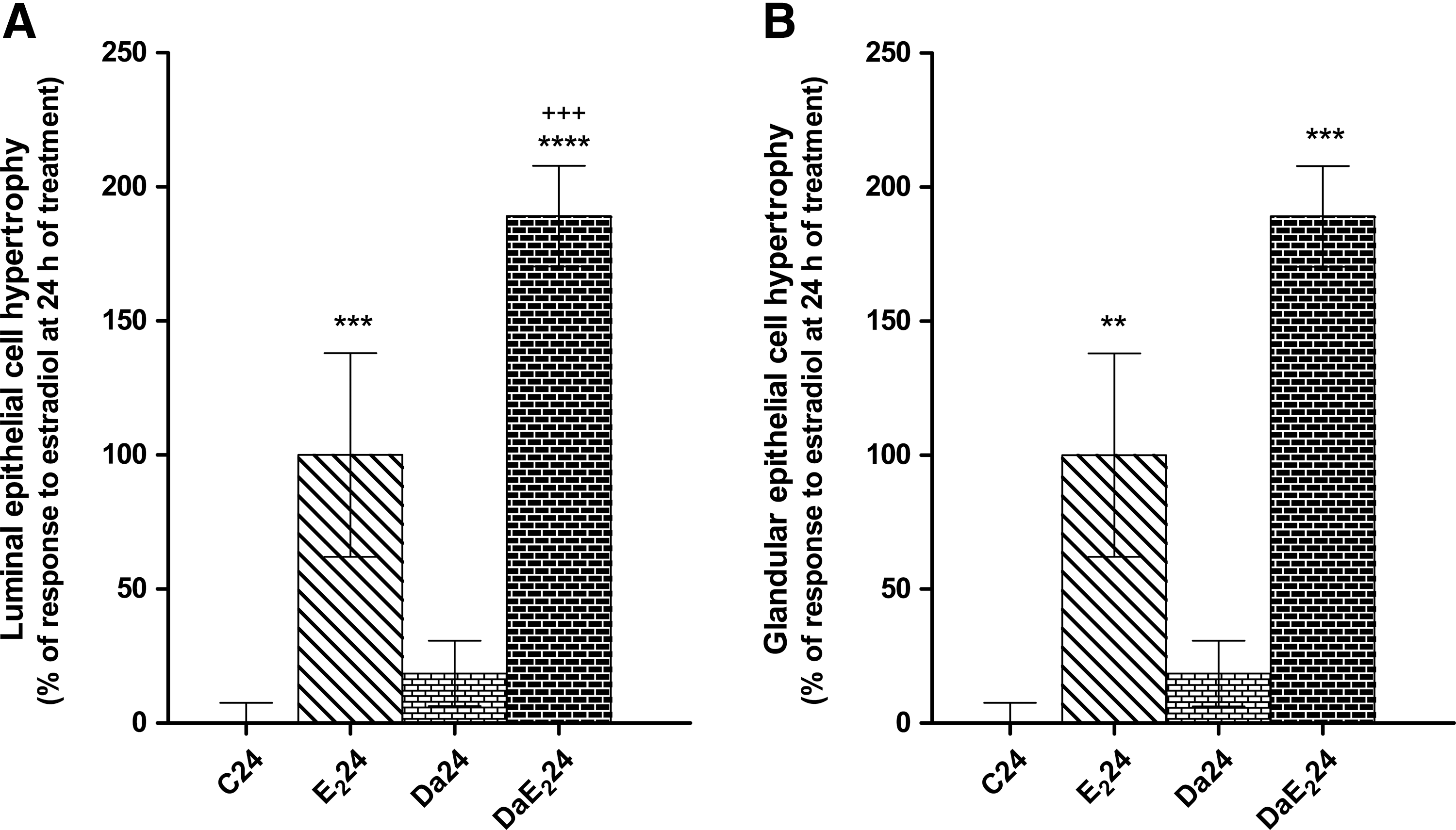
Effects of daidzein (Da) pretreatment on estradiol-17β (E2)-induced increase in uterine luminal epithelial
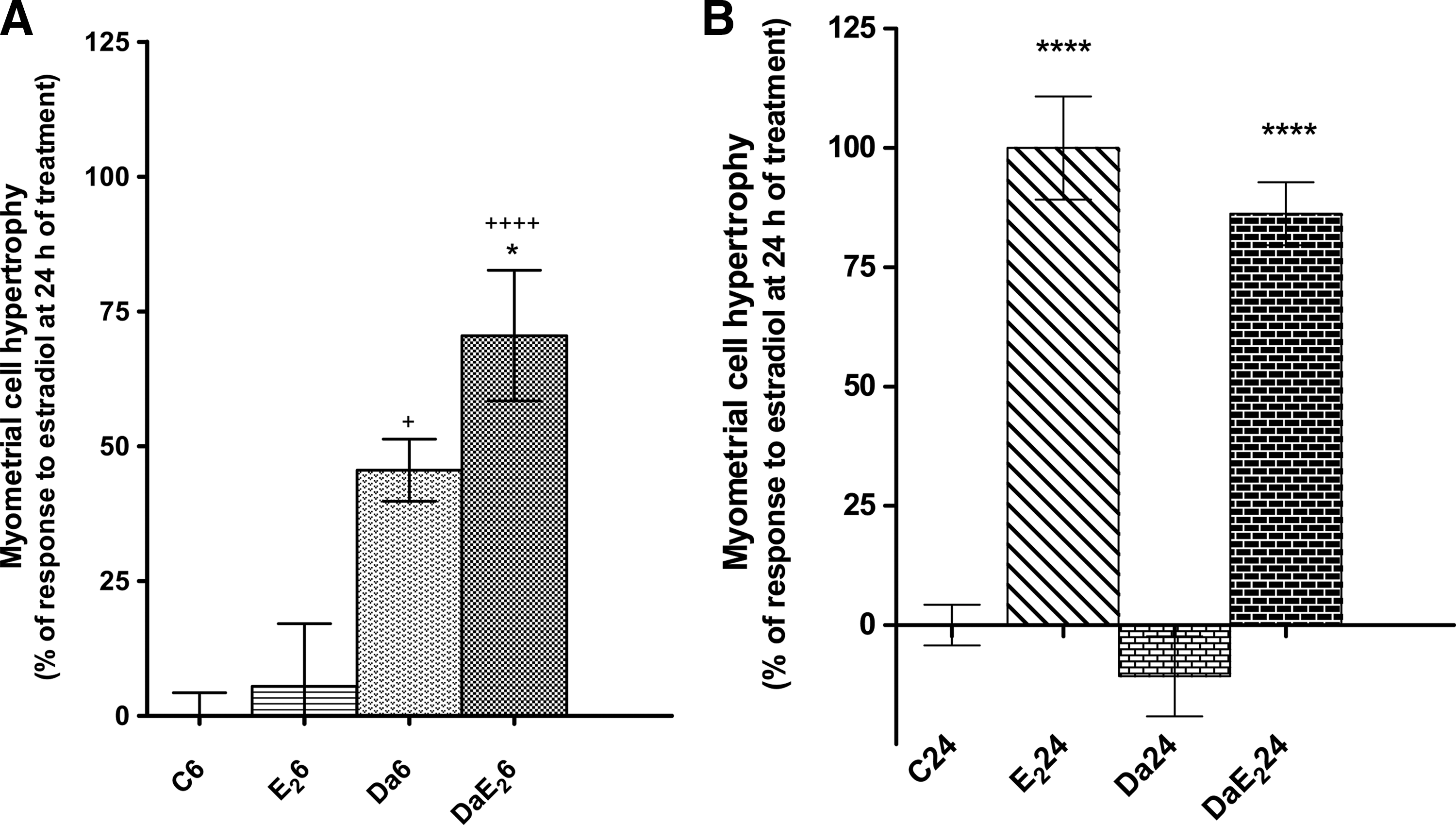
Effects of daidzein (Da) pre-treatment on E2-induced increase in myometrial cell volume at 6 or 24 h of treatment with E2. Prepubertal rats received sc 0.066 mg daidzein/kg bw or control vehicle, and 1 h later, they received sc 0.33 mg E2/kg bw or control vehicle; the uteri were excised 6 h
Daidzein by itself does not induce uterine eosinophilia, but its pretreatment strongly potentiates the response to stimulation by E2 at 6 h after treatment (Fig. 3). At 24 h after E2 administration in daidzein-pretreated animals, values are similar to those in animals without pretreatment. Figure 4 shows that daidzein pretreatment of E2-treated animals increases the proportion of eosinophils located in the endometrial layer of the uterus at 6 h after treatment, and decreases the proportion of degranulated eosinophils at 24 h after hormone administration. These proportions were not analyzed in animals without E2 treatment because of very low eosinophil numbers that would invalidate any statistical analysis.
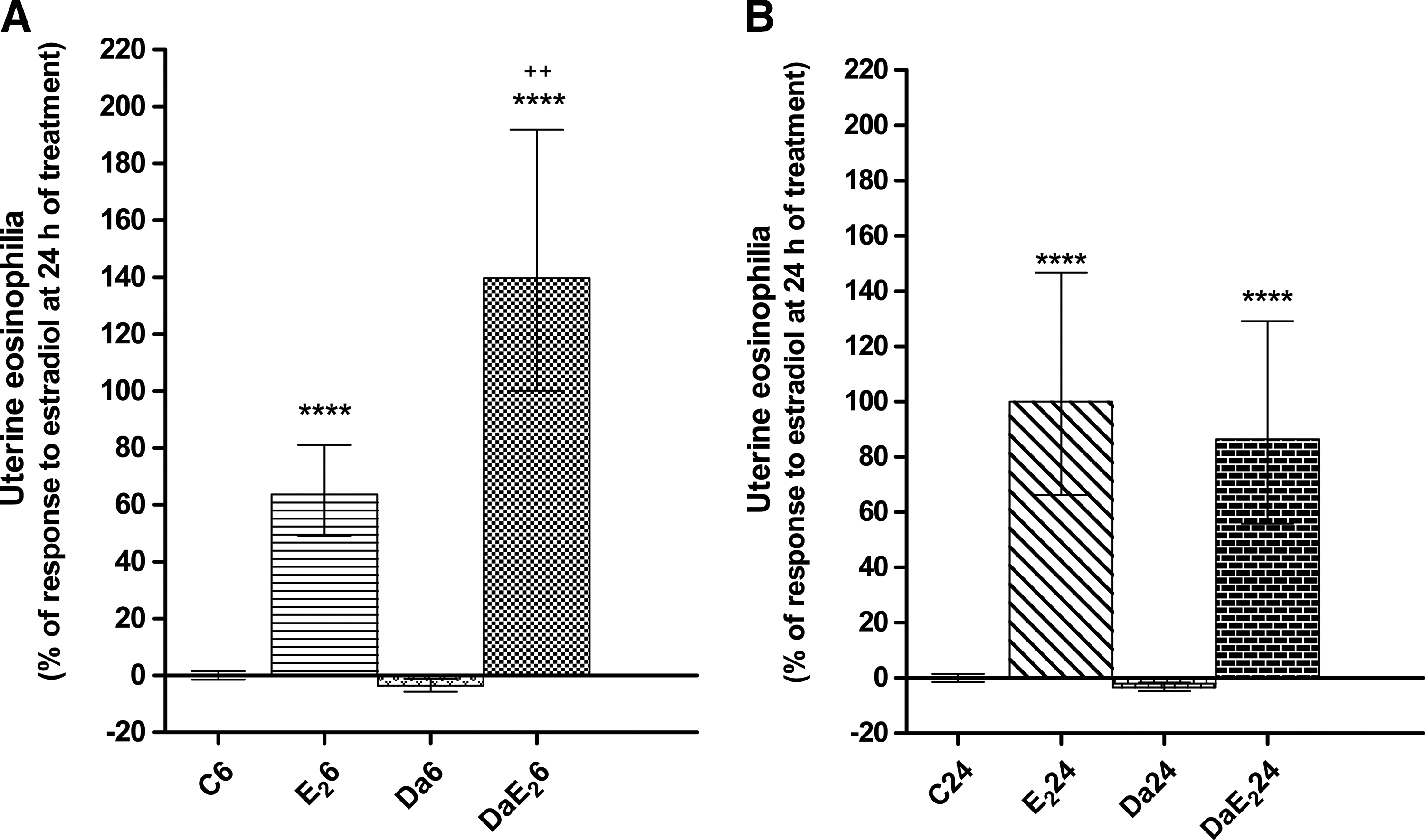
Effects of daidzein (Da) pre-treatment on E2-induced uterine eosinophilia at 6 or 24 h of treatment with E2. Prepubertal rats received sc 0.066 mg daidzein/kg bw or control vehicle, and 1 h later, they received sc 0.33 mg E2/kg bw or control vehicle; the uteri were excised 6 h
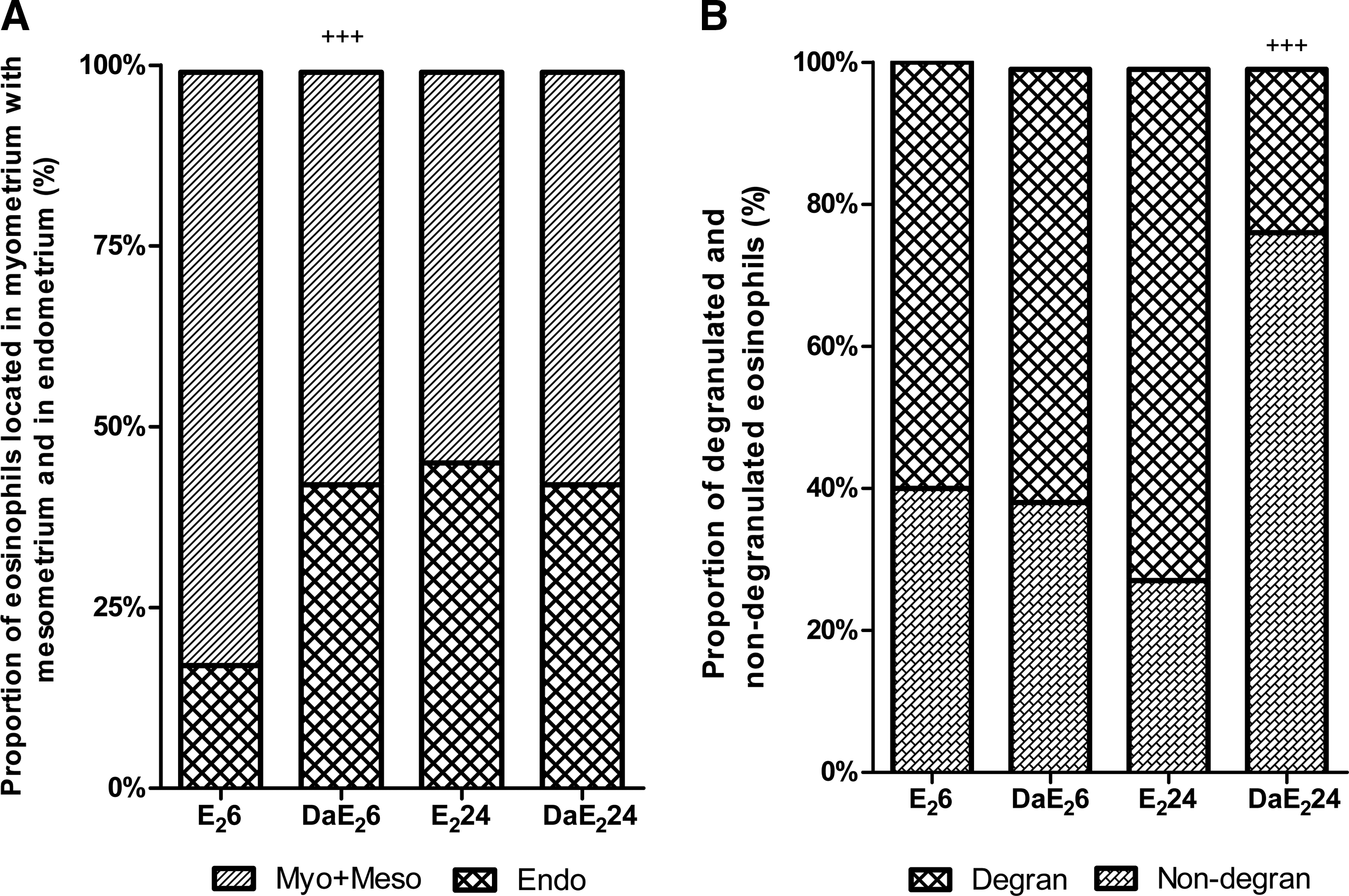
Effects of daidzein (Da) pre-treatment on E2-induced changes in proportions of uterine eosinophils located in the myometrium with mesometrium (Myo+Meso) and those in the endometrium (Endo)
Daidzein itself does not induce water imbibition in the uterus, but it strongly potentiates endometrial edema induced by E2 at 24 but not at 6 h after treatment (Fig. 5). Figure 6 shows that daidzein, in the absence of E2 treatment, increases the number of mitotic figures in the uterine glandular epithelium and in the myometrium. Further, daidzein pretreatment strongly potentiates the response to E2 in uterine luminal and glandular epithelium, at 24 h after treatment.
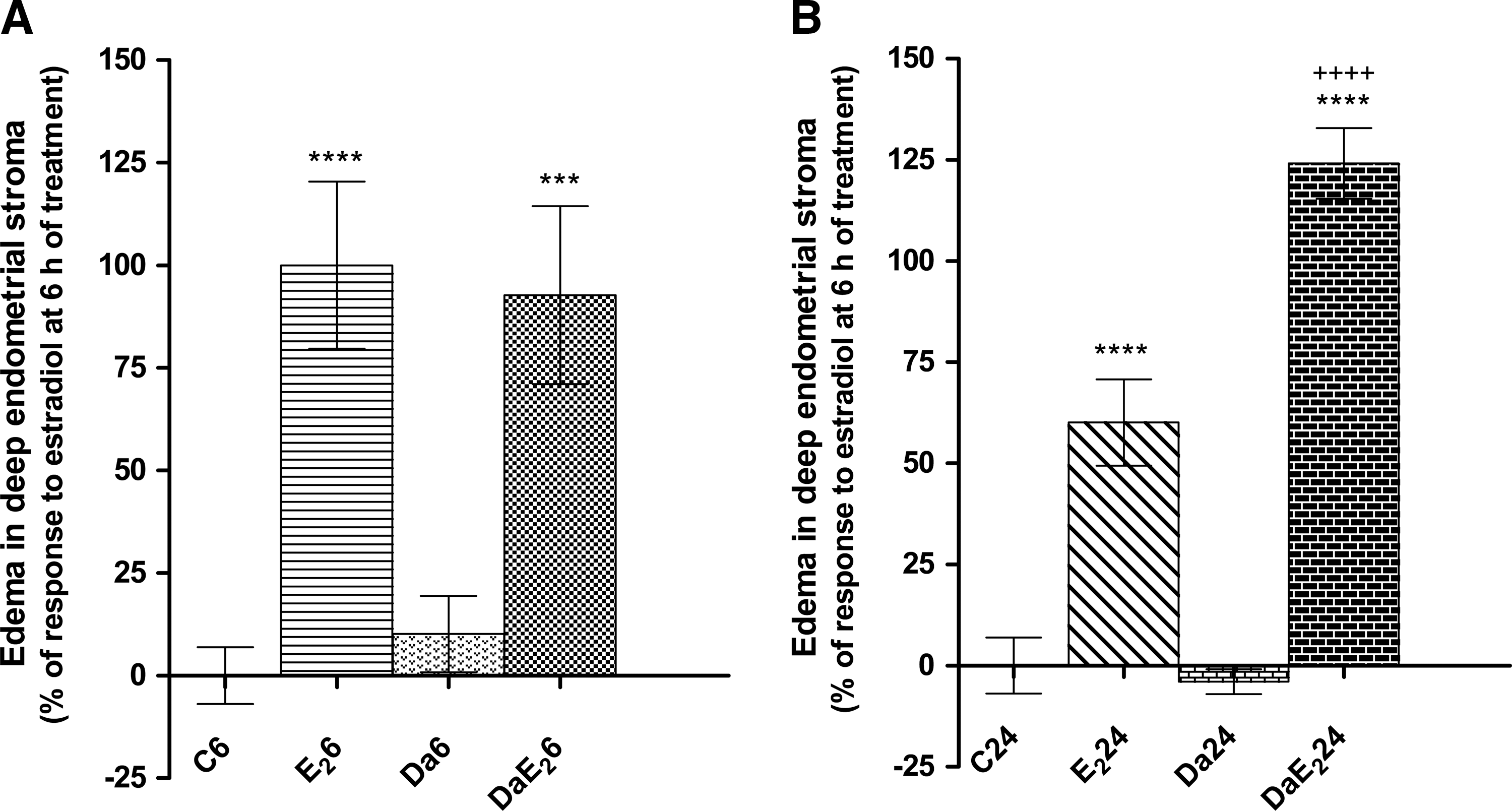
Effects of daidzein (Da) pretreatment on E2-induced edema in deep (yuxtamyometrial) endometrial stroma at 6 or 24 h of treatment with E2. Prepubertal rats received sc 0.066 mg daidzein/kg bw or control vehicle, and 1 h later, they received sc 0.33 mg E2/kg bw or control vehicle; the uteri were excised 6 h
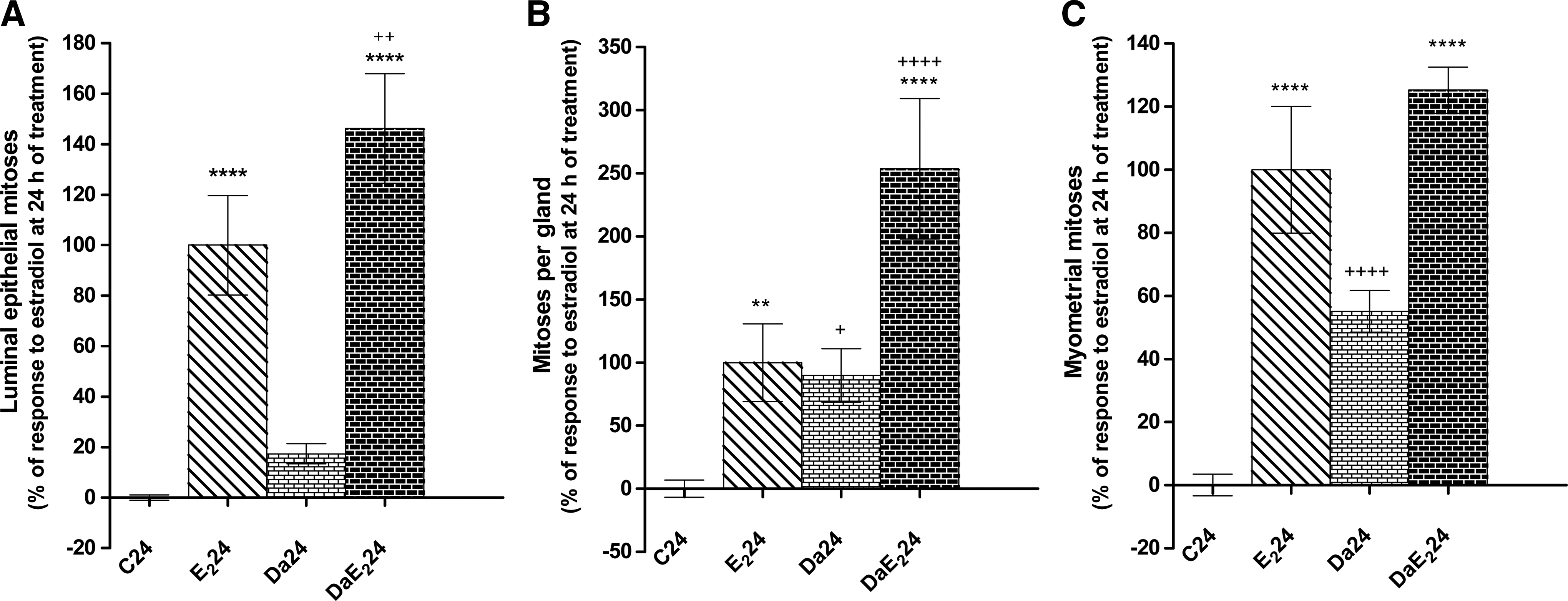
Effects of daidzein (Da) pretreatment on E2-induced increase in the number of mitotic figures in the uterine luminal epithelium
As shown in Figure 7, daidzein dramatically increases human breast cancer MCF-7 cell growth; this increase is stronger than the growth observed in cell cultures under stimulation with E2. On the contrary, E2 slightly decreases human breast cancer MDA-MB-231 cells, while daidzein itself does not cause any change to MDA-MB-231 cell growth.
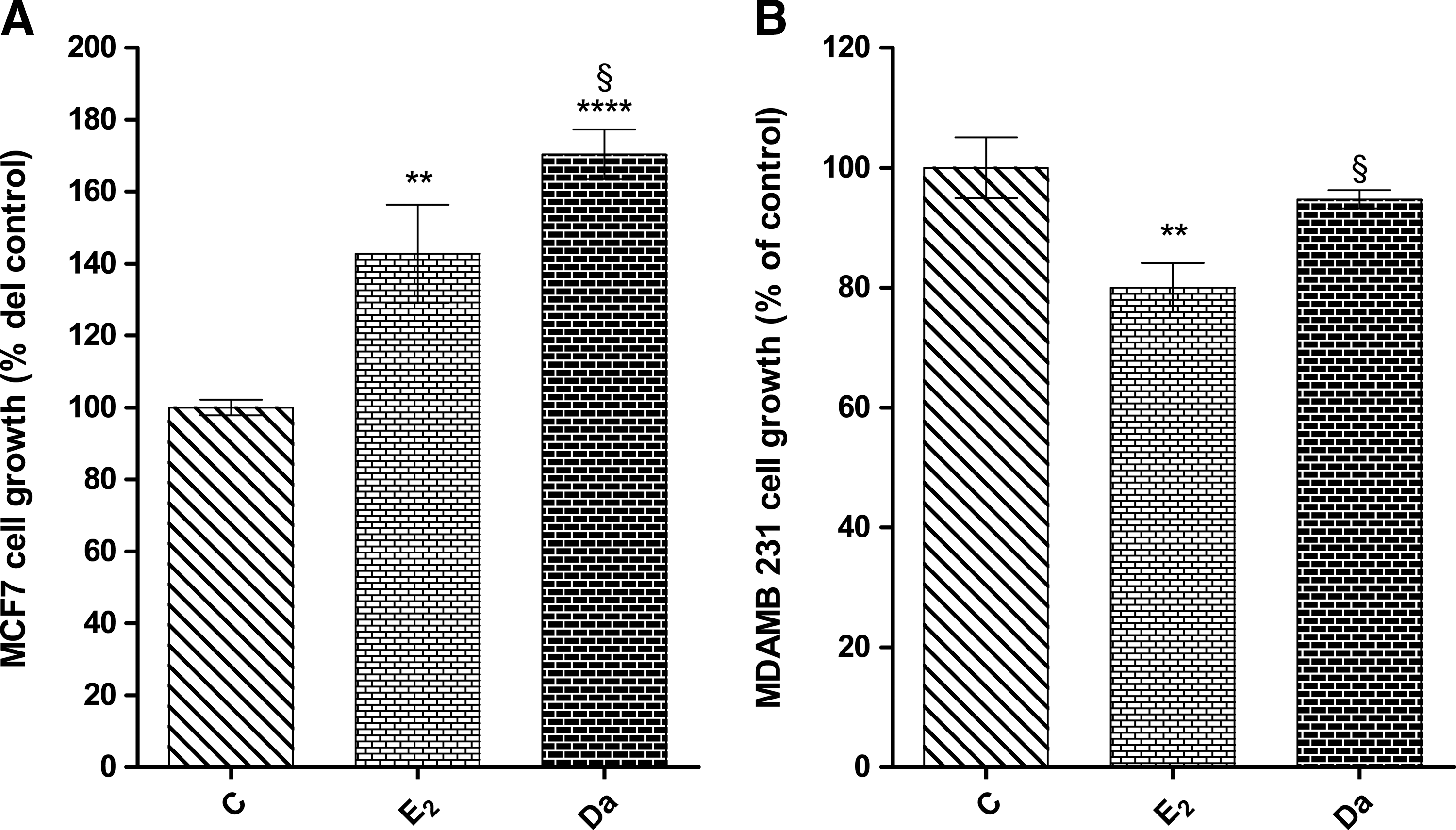
Effects of daidzein (Da) and/or E2 on growth of human mammary cancer MCF-7
Discussion
The present study shows that daidzein mimicks some, but not all, responses to estrogen in the immature rat uterus. It induces cell hypertrophy in myometrial cells, evaluated at 7 but not at 25 h after phytoestrogen administration, and increases cell proliferation in uterine glandular epithelial and myometrial cells, a response that was evaluated as an increase in the number of mitotitic figures at 25 h of daidzein administration. On the contrary, no effect of daidzein administration was observed, in the absence of E2 treatment, for uterine luminal and glandular epithelial hypertrophy, endometrial edema, uterine eosinophilia, and cell proliferation in uterine luminal epithelial cells. It also increases growth of human hormone-responsive MCF-7 breast cancer cells in culture, but does not display any effect on the non-hormone-responsive MDA-MB-231 cell line.
In addition, it is shown that daidzein selectively modifies most responses to E2 treatment in the rat uterus: cell hypertrophy in uterine luminal epithelium and myometrium (at 6 h after E2 treatment), uterine eosinophilia, uterine eosinophil redistribution (towards endometrium) and degranulation of the uterine eosinophils, endometrial edema, and an increase in cell proliferation in uterine luminal and glandular epithelium. Changes in the remaining responses to E2 did not reach statistical significance but may suggest a possible tendency for these responses, particularly for glandular epithelial hypertrophy. Considering the proposed role of uterine eosinophils in the mechanism of estrogen induced endometrial edema, 13,14 the increase in this response at 24 h of E2 treatment may be explained by both the potentiation of the estrogen-induced uterine eosinophilia at 6 h and the increase in the migration of the eosinophils from external histological layers of the uterus toward the endometrium. The increase in the proportion of non-degranulated eosinophils at 24 h may reflect an increase in eosinophil turnover, which also contributes to the potentiation of endometrial edema.
The present study reveals a selective induction of some responses to estrogen in the rat uterus in vivo and in human breast cancer cells in culture, and a selective potentiation of some responses when they are induced by E2. These results are not in disagreement with our hypothesis that the various responses to estrogen are mediated by different kinds of ERs (or binding proteins) separately through independent mechanisms (vide supra).
Apart from the existence of different ERs, many other cellular components may contribute to the selective induction or stimulation of estrogenic responses in some uterine cell types and differences between results when comparing MCF-7 and MDA-MB-231 human breast cancer cells. For instance, subtle differences in ER conformations caused by (1) interaction with different ligands interacting with different co-activators or co-repressors within the cell; 50 (2) the existence of spliced receptor variants such as ER36 51,52 or ER46; 53 (3) recruitment of co-regulators by the orphan nuclear ERRα in a ligand-independent manner, 54 –56 thus decreasing their availability for interaction with remaining ERs in some cell types but not others; (4) presence of different molecular chaperones such as FKBP51, FKBP52, and Cyp40, which may be incorporated in the inactivated ER complexes from the different cell types; 57,58 or simply, (5) that some of the responses are not ER-mediated responses. In the case of differences between human breast cancer cell lines MCF-7 and MDA-MB-231, differences had been explained by the role of the ERα46 isoform in cellular proliferation. 53,59
The following findings of the present study add information for the analysis of the current controversy for the use of plant preparations containing daidzein or its metabolites (for instance, equol), whether they are convenient or dangerous for a prolonged HRT. The increase in proliferation of luminal and glandular epithelial cells of the uterus points to a possible risk for endometrial malignancies after administration of these agents. This risk faced by uterine tissues is not displayed by genistein or soy extracts. 33,34 Unpublished results from our laboratory suggest a risk for breast cancer in MCF-7 cells, as 2×10−5 M genistein in the culture media stimulated growth of these cells.* These results are in disagreement with reports from Wang et al., who described that while lower concentrations (10−8–10−6 M) of genistein stimulate growth of MCF-7 human breast cancer cells in culture, higher concentrations (>10−5 M) inhibit MCF-7 growth. 36
Our present finding of an increase in human breast cancer MCF-7 cell growth in a culture medium containing daidzein at 5×10−7 M, which is significantly greater than the increase in cell growth under the effect of E2, suggests an additional risk for breast cancer development under HRT with preparations containing daidzein. The protective effect of E2 on growth of the non-hormone responsive breast cancer cells MDA-MB-231 under the effect of E2 is not observed in cultures containing daidzein. Further studies are necessary to find an explanation to this finding.
Several authors reviewed the pros and cons of HRT with soy phytoestrogens with regard to breast cancer. 26,36 –39,60 –62 Among the beneficial effects of soy estrogenic isoflavones, it was reported that daidzein could induce breast cancer cell apoptosis through the mitochondrial caspase-dependent cell death pathway. 61 Among the effects that may involve risk of breast cancer development, it was reported that high doses of the daidzein metabolite equol exert clear mammotropic effects in ovariectomized rats, 62 that dietary genistein can stimulate the growth of a mammary carcinogen 1-methyl-1-nitrosourea-induced estrogen-dependent mammary tumors, 38 and that genistein and the daidzein metabolite equol elicit a delayed activation of ERK1/2, which appears to be involved in breast cancer cell proliferation and estrogen-dependent transcriptional activation. 37 Further, it was reported that dietary genistein negated/overwhelmed the inhibitory effect of tamoxifen on MCF-7 tumor growth. Therefore, postmenopausal women consuming dietary genistein while on tamoxifen therapy for estrogen-responsive breast cancer may not respond to the treatment due to antagonism exerted by dietary genistein. 39
Taking all of these factors into consideration, we suggest caution for the use of the different soy isoflavone formulas for HRT, at least until the reasons for reported discrepancy in results could be understood. At least for daidzein itself, present results suggest that its use is not convenient in HRT, as it stimulates the proliferation of hormone-responsive human breast cancer cells in culture, antagonizes the proliferation-inhibiting effect of E2 on non-hormone responsive breast cancer cells, and that, in the rat uterine model in vivo it also induces the proliferation of glandular epithelial and myometrial cells and potentiates estrogen induced proliferation of luminal and glandular epithelial cells. In this context, research should be directed toward investigating the conditions required for safe usage of genistein, daidzein, or other soy isoflavones.
However, based on the possibility to selectively stimulate or inhibit estrogen action on some target cell types but not others, we suggest that future research strategies should be addressed toward searching for compounds present in plants inhibiting E2-induced proliferation in uterine and cancer breast cells, or at least not inducing cell proliferation in the absence of E2, but inducing other beneficial responses to estrogen stimulation.
Footnotes
Acknowledgments
This research was financed by the Research Team Grant in Science and Technology ACT07, Bicentennial Program in Science and Technology, CONICYT, Chile. The authors thank Ms. Iris Rodríguez (ICBM, University of Chile Medical School, Santiago, Chile) for the technical help rendered.
Author Disclosure Statement
No competing financial interests exist.
*
Villena J, Tchernitchin AN, Gaete L, Bustamante R, Lemus I: unpublished observations, Universidad de Valparaiso, Valparaiso, Chile, and University of Chile, Santiago, Chile, 2009.
*
Villena J, Tchernitchin AN, Gaete L, Bustamante R, Lemus I: unpublished observations, Universidad de Valparaiso, Valparaiso, Chile, and University of Chile, Santiago, Chile, 2009.
