Abstract
Background and Objective:
Mental health complaints have significantly increased in Europe over recent years. This study evaluated the effects and safety of a 4-month online, multimodal, complementary intervention on the mental and physical health of home-based adults.
Method:
Ninety-eight home-based adults with mild to severe mental health and related physical symptoms were recruited across Europe through digital advertisements. This single-arm repeated-measures study utilized three individualized treatment groups to evaluate the impact of the Ayurveda Program for Detox and Lifestyle. The online program included a personalized Ayurveda diet, daily routine, yoga exercise, herbal detoxification procedures, and herbal preparations. Standardized online tests were administered monthly for 4 months to assess anxiety, depression, physical symptoms (Patient Health Questionnaire for Somatic, Anxiety, and Depressive Symptoms), fatigue (PROMIS), and body mass index (BMI).
Results:
The study population was predominantly female (85%), with a mean age of 58 years. By the end of the fourth month, primary outcomes indicated a significant decrease in anxiety (55%, p < 0.001) and depression symptoms (45%, p < 0.001). The secondary outcomes showed a significant reduction in physical symptoms, including musculoskeletal, neurological, and digestive issues (42%, p < 0.001); fatigue (31%, p < 0.001); and BMI (2%, p < 0.001). Improvements were consistent across all treatment groups, with no significant differences observed. The intervention was feasible and generally safe, with 91% retention, 70% compliance, and 6% adverse effects.
Conclusion:
The initial results suggest that the Ayurveda Program for Detox and Lifestyle is safe and is associated with significant improvements in the mental and physical health of participants. This trial highlights the potential benefits of integrating complementary lifestyle protocols into standard primary and mental health care practices. Future randomized controlled trials are recommended to control for confounding factors and validate these findings in larger clinical populations.

Introduction
Mental health problems significantly impair cognitive, social, and physical functioning. 1 Research from 2015 to 2019 indicates that in Europe, approximately 11% of adults reported psychological symptoms, 14% experienced anxiety disorders, and 12% suffered from depressive disorders. 2,3 These issues are linked to an increased risk of chronic diseases and suicide, accounting for 15% of all years lived with disability and resulting in 119,000 lost lives in Europe in 2019. 4 –8 During the COVID-19 pandemic, the prevalence of psychological symptoms, depression, and anxiety surged to as high as 31%. 9,10 Despite advancements in treatments, mental health problems and their associated health care costs remain a significant public health concern, with annual costs in the EU exceeding 600 billion EUR, representing more than 4% of the GDP. 8,11
As reported by the World Health Organization (WHO), approximately 65% of the population in developed countries and more than 80% in developing countries use Complementary and Alternative Medicine (CAM) therapies for chronic and treatment-resistant conditions such as anxiety, depression, headaches, and back pain. The WHO recognizes Ayurveda as a CAM system for prevention, diagnosis, and treatment of physical and mental illnesses. 12
Research indicates that unhealthy diet and sleep habits, sedentary lifestyle, and stress contribute to psychological and physiological disorders. 13 –15 Prior studies targeting these risk factors have shown that CAM can effectively decrease mental and physical health symptoms. 16 –20 However, most CAM trials have focused on single-treatment methods. Few studies have investigated the effects of traditional whole-system (WS) and multimodal CAM interventions that simultaneously address multiple risk factors. Therefore, the overall objective of this study was to evaluate the effects and safety of a comprehensive online Ayurveda WS intervention on the mental and physical health of home-based adults, before conducting a larger, definitive trial.
Methods
Study design
The study employed a single-arm repeated-measures design with three individualized treatment groups to enhance the precision of the effects of a 4-month online Ayurveda program. The treatment protocols were designed according to individual psychophysiological principles (Doshas) in Ayurveda. Participants were allocated into three treatment groups based on the Ayurveda Dosha Self-Assessment: Vāta (n = 32), Pitta (n = 42), or Kapha (n = 24). 21 By incorporating personalized protocols, the study facilitates the evaluation of the intervention’s effects across various psychophysiological constitutions.
In phase 2 trials, disease response and safety are the primary endpoints, providing direct insights into the treatment’s effects and safety. 22 A total of 98 home-based adults were assessed for anxiety, depression, physical symptoms, fatigue, body mass index (BMI), compliance, and side effects every month for 4 months. In the repeated-measures design, subjects serve as their own controls, increasing sensitivity to changes and reducing variability. 23
The experiment was conducted in two stages with different participants to ensure the reliability of the results. The first stage, conducted in English with 51 European subjects (March–June 2021), gathered preliminary data. The second stage, in German with 47 European subjects (March–June 2022), confirmed initial findings and assessed generalizability across languages and cultures. The pilot trial concluded within the planned 4 months as all primary objectives were achieved. The study followed the CONSORT guidelines for pilot trials. 24 Interim analyses were conducted after the first month, examining anxiety, depression, and side effects. Stopping guidelines included halting the trial if any participant experienced a severe adverse event or a significant increase in adverse reactions.
Prespecified criteria for determining the feasibility of a future definitive trial included: At least 70% of participants adhere to the intervention and complete the study. A statistically significant reduction in anxiety and depression scores, as measured by validated scales. No serious adverse events related to the intervention.
Procedure
The Center for Maharishi AyurVeda and Holistic Medicine in Austria recruited home-based adults across Europe through digital advertisements to enhance generalizability. Recruitment occurred in two phases: December–March 2021 and December–March 2022. Data were collected through an online survey at baseline and then monthly over 4 months to minimize observer bias. The protocol was approved by the Institutional Review Board of Maharishi International University, Fairfield, IA (Approval No. IRB-MIU-PH-2020-1008, Date: December 04, 2020). The study was registered at ClinicalTrials.gov (NCT06551467). All participants provided electronic informed consent before inclusion.
Participants
Baseline data were collected through an online survey, which also screened for suicidal ideation and health conditions. Participants with potential contraindications underwent teleconference interviews with Ayurveda-trained medical doctors to assess their suitability. Eligible home-based adults had mild to severe anxiety, depression, or physical symptoms. Table 1 outlines the inclusion and exclusion criteria.
Inclusion and Exclusion Criteria
The table provides a detailed explanation of the criteria used to include or exclude participants from the study.
Participants continued their standard medical care for existing or new health conditions during the study. Six participants on psychiatric medication had been on a stable regimen for at least 3 months before enrollment.
Outcomes
The primary outcomes were a reduction in anxiety and depression, while the secondary outcomes included physical symptoms, fatigue, and BMI. Anxiety, depression, and physical symptoms were assessed using the Patient Health Questionnaire for Somatic, Anxiety, and Depressive Symptoms (PHQ-SADS), which combines the GAD-7 (for anxiety), 25 PHQ-9 (for depression), 26 and PHQ-15 (for physical symptoms). 27 Physical symptoms assessed included the most prevalent musculoskeletal, neurological, and digestive symptoms in primary care. Higher scores on these scales indicate greater severity of symptoms, with cut-off points at 5 for mild, 10 for moderate, and 15 for severe symptoms. Studies have confirmed the validity and reliability of the PHQ-SADS in clinical and nonclinical samples. 28 The minimal clinically important difference (MCID) is 4 points or 50% change for GAD-7; 25 5 points or 50% for PHQ-9; 29 and 2–3 points for PHQ-15. 30
Fatigue was assessed using the PROMIS Fatigue Short Form (8a), where higher scores indicate greater fatigue. Scores of 22, 26, and 36 correspond to mild, moderate, and severe fatigue, respectively. 31 Validation studies confirm the questionnaire’s reliability across various populations, with a minimally important difference of approximately 3–5 points. 32,33 Participants reported their height and weight, and BMI was calculated using a standardized formula.
An Ayurveda Self-Assessment questionnaire determined individual physiological constitution and imbalances.
21
Compliance, facilitators, barriers, and side effects related to the intervention were monitored using a self-report questionnaire over 4 months. Demographic data and potential confounders were collected through an online survey. This study adhered to WS's research criteria, employing a variety of psychological and physiological outcome measures.
34
To minimize bias and improve validity: Participants were recruited from diverse sources. Scientifically validated questionnaires were used. Anonymous online survey completion was ensured. Participants were instructed to report measurements immediately. The online survey included additional wellness questions, keeping participants unaware of the study’s focus on specific conditions, potentially reducing placebo effects.
17
Interventions
The Online Ayurveda Program for Detox and Lifestyle is easily replicable and includes personalized herbal remedies (Dravyaguna), detoxification procedures (Shodhana), dietary guidelines (Ahara Vidhi), and daily routine practices (Dinacharya), all based on traditional Ayurvedic medicine principles. 35 –37 While clinical studies on Ayurveda multimodality treatments for anxiety and depression are scarce, some studies have shown significant improvements in mental and physical health, emphasizing the need for further research. 18,38 –43
According to Ayurveda texts, three fundamental principles—the three Doshas: Vāta, Pitta, and Kapha—govern all cognitive and physical functions. The intervention aimed to restore and maintain health by considering individual psychophysiological constitution (Prakriti), imbalances (Vikriti), and lifestyle factors. 37 Home-based adults received personalized Dosha-specific (Vāta, Pitta, or Kapha) treatment protocols and herbal compounds based on their Dosha Self-Assessment questionnaire, which were sent by the Center for Maharishi AyurVeda and Holistic Medicine in Austria (see Table 2).
Treatment Protocol
The table comprehensively explains the treatment protocol, detailing the specific doses and administration methods.
Two Ayurveda-trained medical doctors led the Ayurveda Program, which included eight interactive live webinars over ten days and a lifestyle program lasting up to 4 months. Each 1.5-hour webinar addressed the etiological factors contributing to mental and physical ill-health: Sessions 1–2: Focused on digestion, nutrition, and an individualized, Dosha-specific healthy diet. Sessions 3–4: Elaborated on individualized, Dosha-specific daily routines, oil massages (Abhyaṅga), yoga exercises, and behavioral recommendations. Sessions 5–6: Explained individualized, Dosha-specific herbal detoxification procedures, gastrointestinal oleation, and elimination therapies (Snehana, Virechana).
During webinars, Ayurveda-trained doctors offered motivational coaching and personalized treatment adjustments. These sessions were as effective as daily doctor visits in a residential setting, providing a cost-effective alternative. After the ten-day detoxification, clients continued to follow diet and lifestyle recommendations, along with the herbal program for mental and physical health, for up to 4 months. During this period, participants were encouraged to contact leading medical doctors for guidance at all hours. All herbal preparations used in the intervention were mild in dose and impact to avoid unwanted reactions.
Statistical analysis
The primary and secondary outcomes were assessed using repeated-measures multivariate analysis of covariance (MANCOVA) with the intention-to-treat method in SPSS. The primary outcomes focused on the severity of anxiety and depression, with three Dosha-specific individualized treatment groups as between-factor groups. Repeated measures were conducted at baseline and at four monthly posttests. The secondary outcomes included physical symptoms, fatigue, and BMI. Covariates for both primary and secondary analyses included meditation practice, vitamin use at baseline, study year, and gender.
If significant interactions were observed, Bonferroni-adjusted simple main effects or ANOVA was conducted as follow-up analyses. The effect size was computed using Cohen’s d, defined as the difference in mean change scores divided by the sample standard deviation of the mean difference. 46
Based on preliminary data, power analysis indicated that for within-group analyses, eight subjects were needed to detect significant changes in anxiety [effect size (d = 1.22)] and 12 subjects for depression [effect size (d = 0.91)] with 80% power. For between-group analyses with three subgroups, approximately 36 subjects were required for anxiety and 120 participants for depression.
Results
The CONSORT Flow Diagram (Fig. 1) illustrates participant flow through the study phases. 47 Nine percent of the data were missing completely at random, confirmed by Little’s MCAR test, χ2(270) = 261.700, p = 0.630. Missing values were replaced using the expectation-maximization method in SPSS, enhancing accuracy and statistical power.
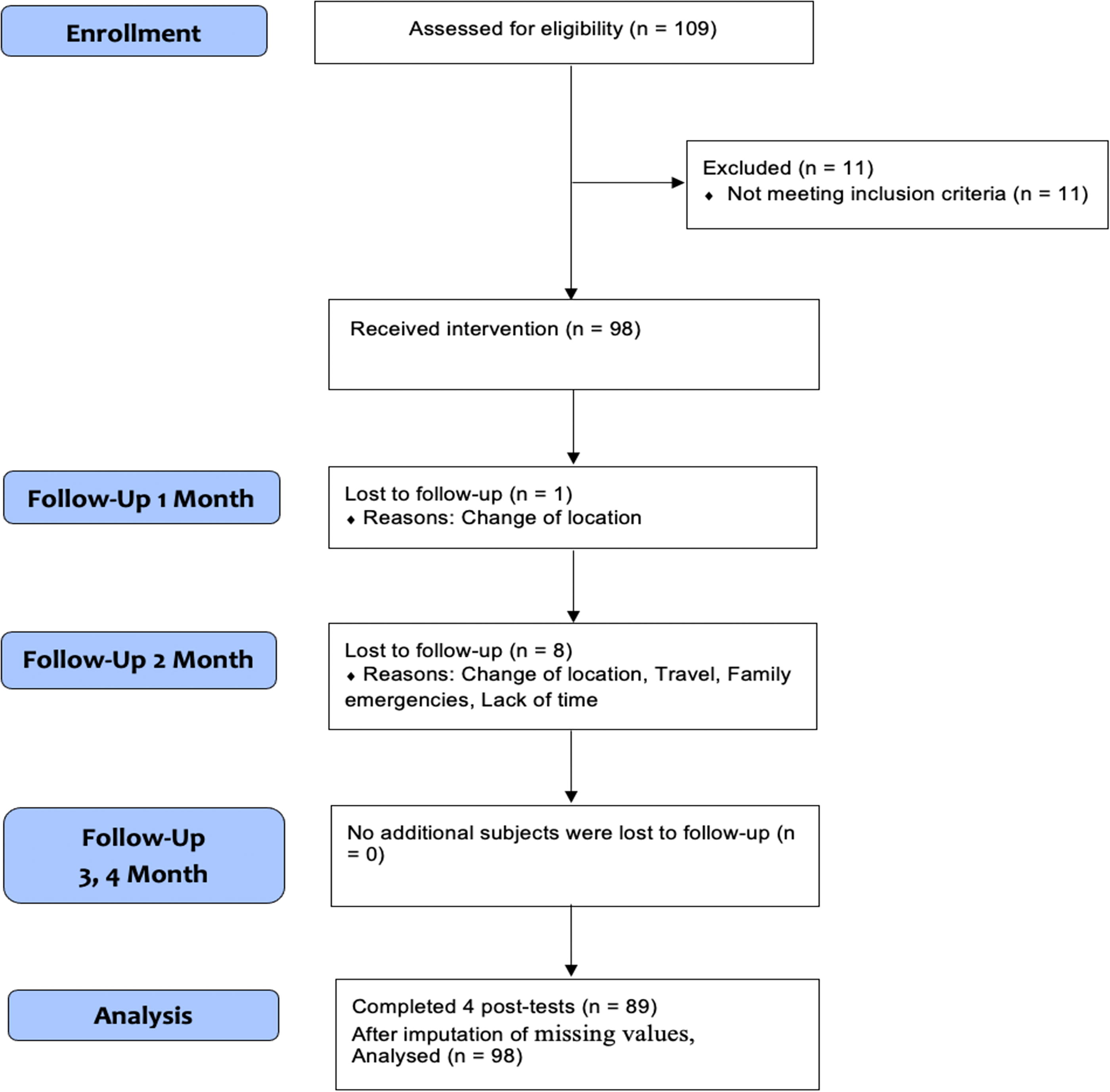
Adjusted CONSORT Flow Diagram. Adjusted Consolidated Standards of Reporting Trials Flow Diagram illustrates the flow of participants through the study phases.
Baseline demographics
Table 3 presents the baseline demographics of the 98 eligible participants, predominantly female (85%) with a mean age of 58 years.
Baseline Characteristics
The table summarizes the baseline demographic and clinical characteristics of the 98 participants in the study.
Primary outcomes
The repeated-measures MANCOVA, adjusted for meditation, vitamin use, study year, and gender, revealed a significant main effect for anxiety and depression scores [Pillai’s trace = 0.195, F(8,84) = 2.547, p = 0.016, η2 = 0.19]. There were no significant differences between the effects of Dosha-specific treatment groups [Pillai’s trace = 0.200, F(16,170) = 1.179, p = 0.290]. However, a significant interaction effect was found in the meditation groups [Pillai’s trace = 0.181, F(8,84) = 2.316, p = 0.027]. The ANOVA test did not confirm a significant interaction for anxiety [F(1,91) = 1.499, p = 0.224] or depression [F(1,91) = 0.176, p = 0.676]. Bonferroni-adjusted pairwise comparisons showed significant differences for each month compared to baseline (p < 0.001), with mean scores remaining stable throughout the 4-month follow-up period (Table 4 and Fig. 2)
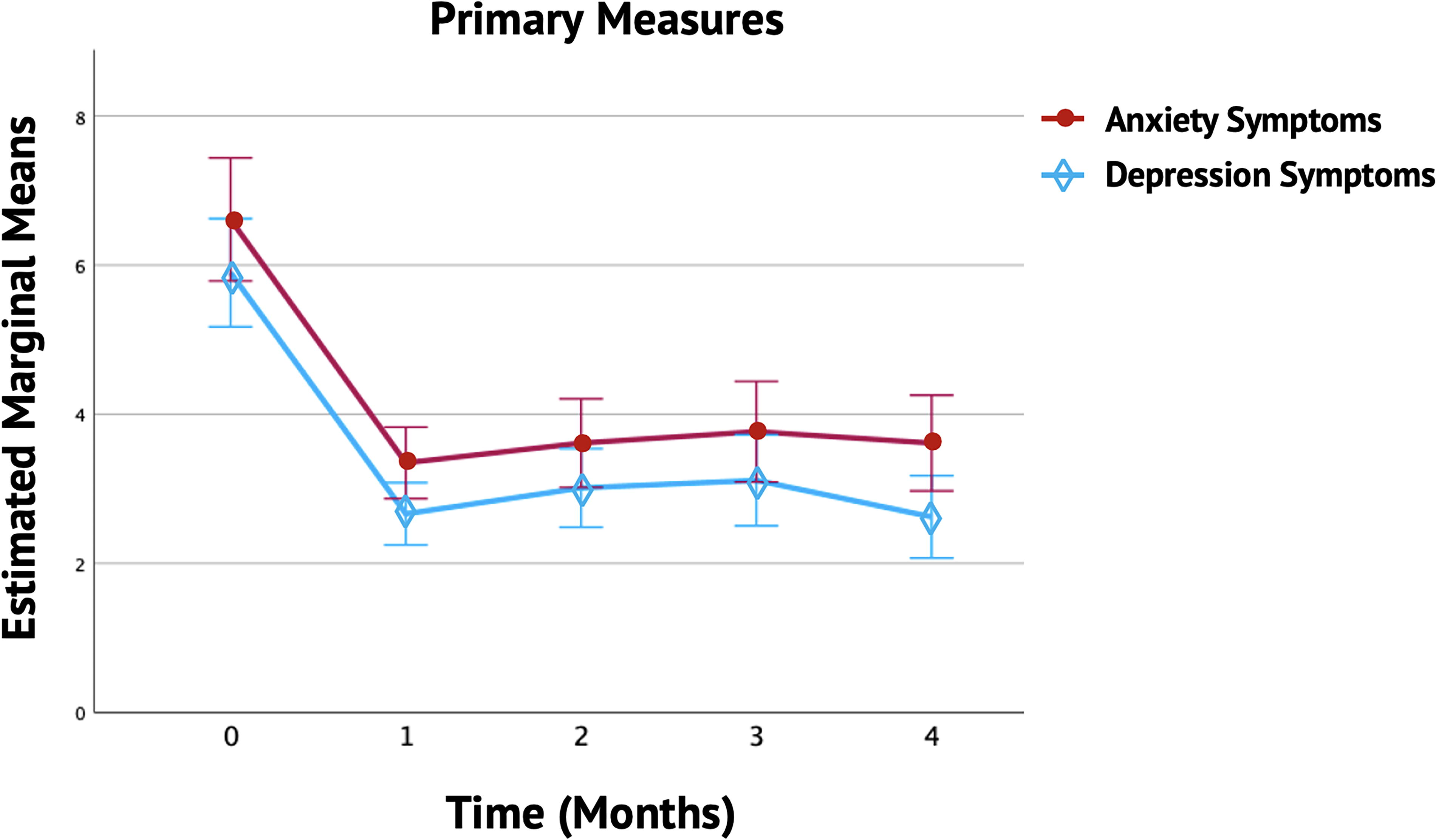
Monthly Variations in Mean Anxiety and Depression Symptoms. The figure illustrates the monthly variations in anxiety and depression symptoms over 4 months among participants in the Ayurveda Program. The solid lines show trends in anxiety and depression symptoms, with error bars representing the standard deviation and 95% confidence intervals (CI). Each month showed a significant reduction in both anxiety and depression symptoms compared to baseline (p < 0.001). The Estimated Marginal Means (EMMs) reflect the average change scores for anxiety and depression, adjusted for study year, vitamin use, meditation, and gender.
Mean and Mean Change Scores of Primary Measures
This table presents the mean and mean change scores of primary measures for each month over a 4-month period, including effect sizes and percentage changes compared to baseline.
Secondary outcomes
The repeated-measures MANCOVA, controlling for meditation, vitamin use, study year, and gender, revealed a significant main effect for all secondary outcomes [Pillai’s trace = 0.299, F(12,80) = 2.840, p = 0.003, η2 = 0.29]. However, there were no significant differences between the Dosha-specific treatment groups [Pillai’s trace = 0.225, F(24,162) = 0.857, p = 0.660] or other interactions. A Bonferroni correction was applied to control for Type I error, setting the significance level at p < 0.01 for secondary analyses. Table 5 and Figure 3 illustrate the Bonferroni-adjusted significant mean differences for all secondary measures compared to the baseline (p < 0.001). These initial results were sustained over a 4-month follow-up period.
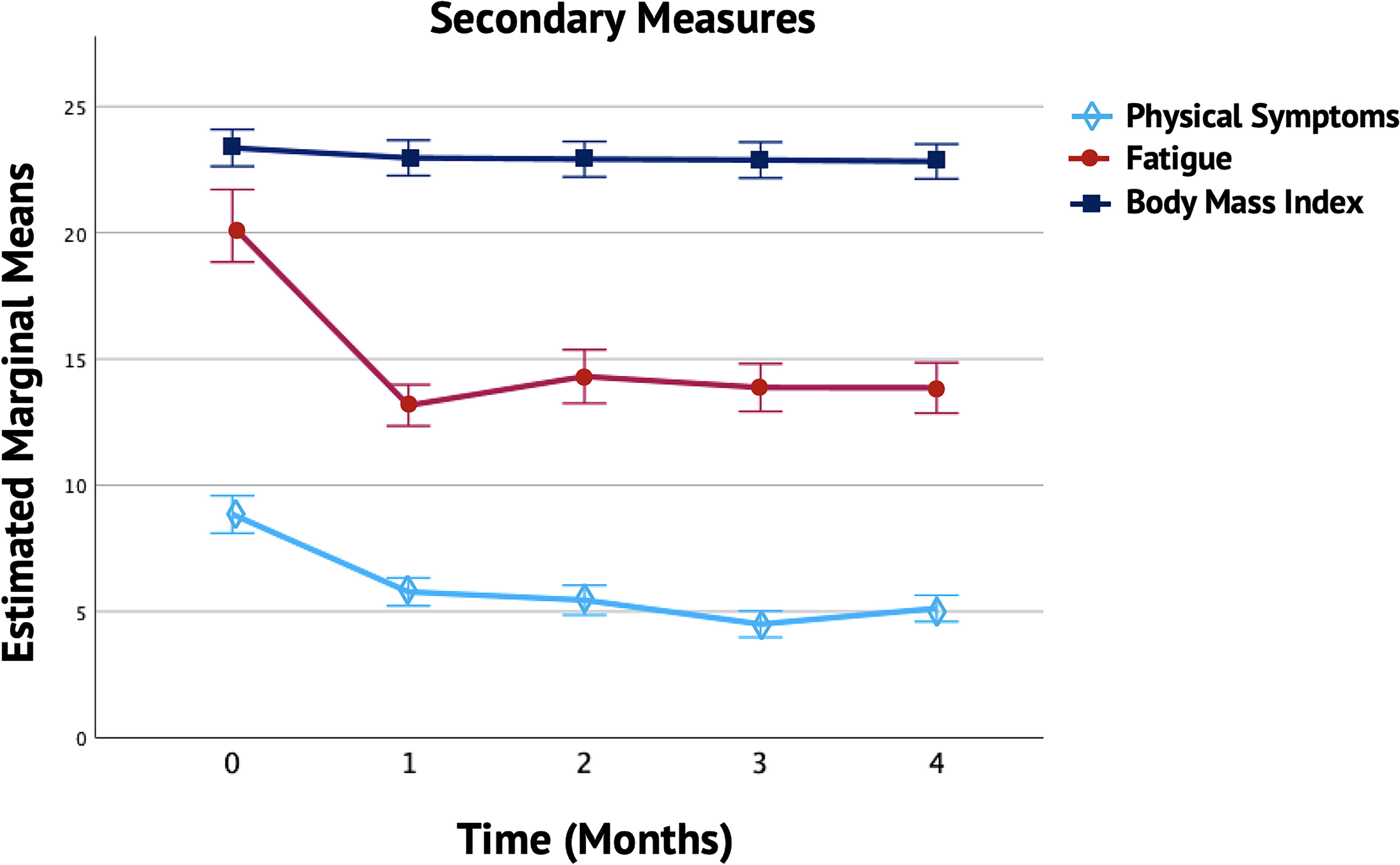
Monthly Trends in Secondary Measures. The figure presents the monthly trends in physical symptoms, fatigue, and body mass index (BMI) over 4 months among patients enrolled in the Ayurveda Program. The solid lines show trends in anxiety and depression symptoms, with error bars representing the standard deviation and 95% confidence intervals (CI). Each month showed a significant reduction in physical symptoms, fatigue, and BMI compared to baseline (p < 0.001). The Estimated Marginal Means represent the average levels of these secondary measures, adjusted for the effects of study year, vitamin use, meditation, and gender.
Mean and Mean Change Scores of Secondary Measures
The table illustrates the monthly mean and mean change scores of secondary measures over a 4-month period, including effect sizes and percentage changes compared to baseline.
Tertiary analyses
A repeated-measures ANCOVA tested the mean differences in the severity groups for anxiety, depression, and physical symptoms. Levene’s test was insignificant for most months across all variables. The alpha level was set to 0.004 and 0.003 to account for multiple tests and potential violations of assumptions.
Anxiety symptom groups
The results of the repeated-measures ANCOVA, adjusted for meditation, vitamin use, study year, and gender, showed a significant main effect for anxiety [Huynh–Feldt F(4,333) = 17.449, p < 0.001, η2 = 0.16]. Significant between-subjects effects for anxiety symptom groups were also observed F(2,91) = 67.210, p < 0.001, η2 = 0.59. No other covariates or interactions were significant.
Figure 4 shows that the adjusted mean anxiety scores were highest in the moderate to severe anxiety symptoms group (Mean = 7.03, SE = ±0.45), followed by the mild symptoms group (Mean = 4.02, SE = ±0.21), and lowest in the minimal symptoms group (Mean = 1.41, SE = ±0.25). Bonferroni-adjusted pairwise comparisons revealed significant decreases in anxiety scores each month compared to baseline for the moderate to severe and mild symptoms groups (p < 0.001), and for the minimal symptoms group (p = 0.003 to p < 0.001). These improvements were maintained over the 4-month period.
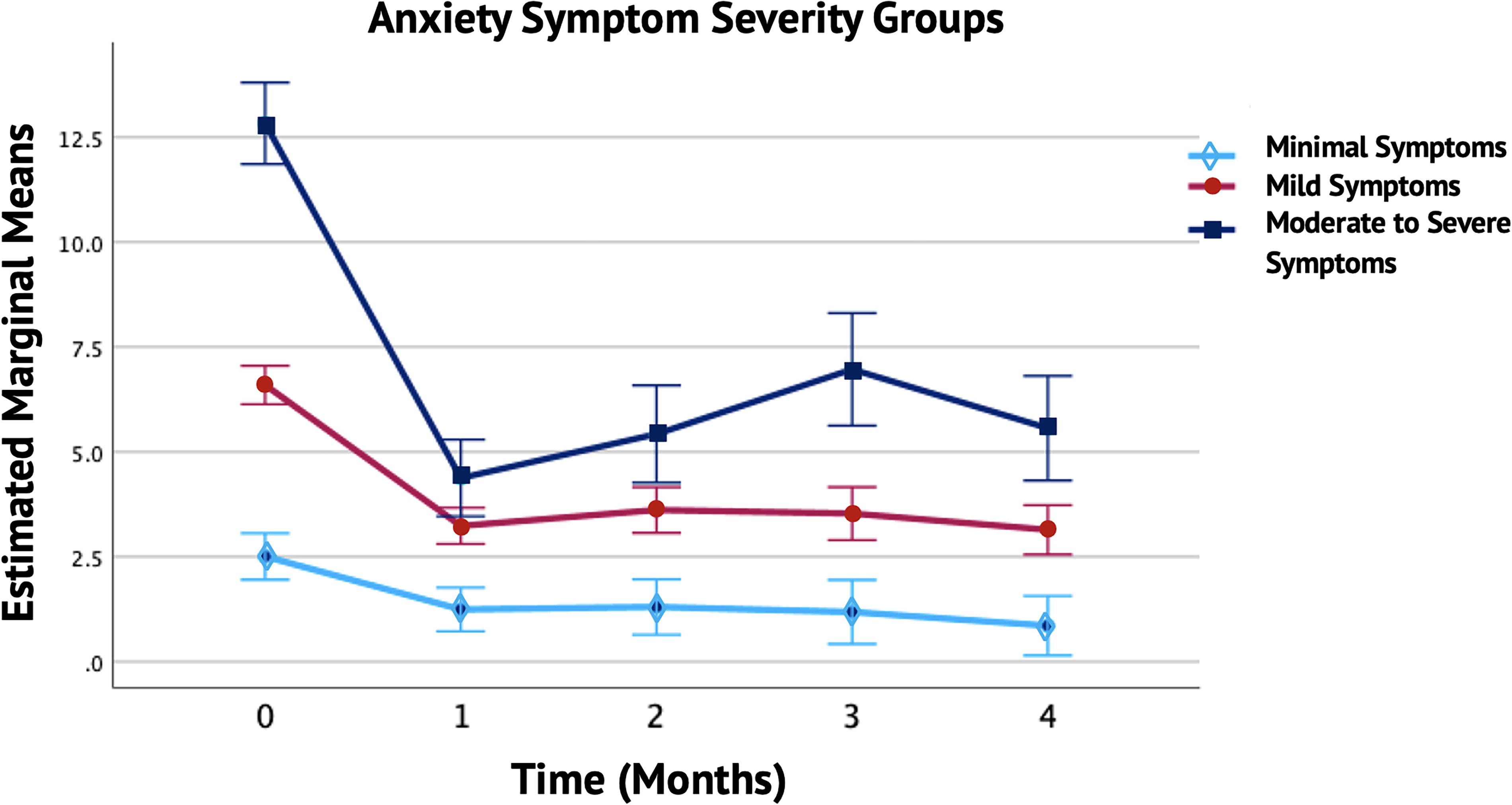
Monthly Trends in Anxiety Symptom Severity Groups. The figure depicts the monthly trends in anxiety symptoms over a 4-month period across different severity groups (Minimal, Mild, and Moderate to Severe). The solid lines represent changes in anxiety symptoms for each group, with error bars indicating the standard deviation and 95% confidence intervals (CI). Each month showed a significant reduction in anxiety symptoms in each group compared to baseline. The Estimated Marginal Means reflect the average anxiety levels for each severity group, adjusted for study year, vitamin use, meditation, and gender. Covariates appearing in the model were evaluated at the following values: vitamins = 0.29, study year = 1.48, meditation = 1.04, and gender = 1.14.
Depression symptom groups
The repeated-measures ANCOVA, controlling for meditation, vitamin use, study year, and gender, revealed a significant main effect for depression, Huynh–Feldt F(4,363) = 10.464, p < 0.001, η2 = 0.10. Significant between-subjects effects for depression symptom groups were also observed, F(1,91) = 66.712, p < 0.001, η2 = 0.59. No other significant interactions were found.
Figure 5 shows the adjusted mean depression scores, highest in the moderate to severe depression symptoms group (Mean = 7.72, SE = ±0.38), followed by the mild symptoms group (Mean = 4.09, SE = ±0.27), and lowest in the minimal symptoms group (Mean = 1.78, SE = ±0.33). Bonferroni-adjusted pairwise comparisons indicated significant changes in depression scores each month compared to baseline for the moderate to severe and mild depression symptom groups (p < 0.001). Improvements remained constant over the 4-month period. However, depression scores in the minimal symptom group did not significantly decrease when correcting for the alpha level (reaching p = 0.003 in the fourth month).
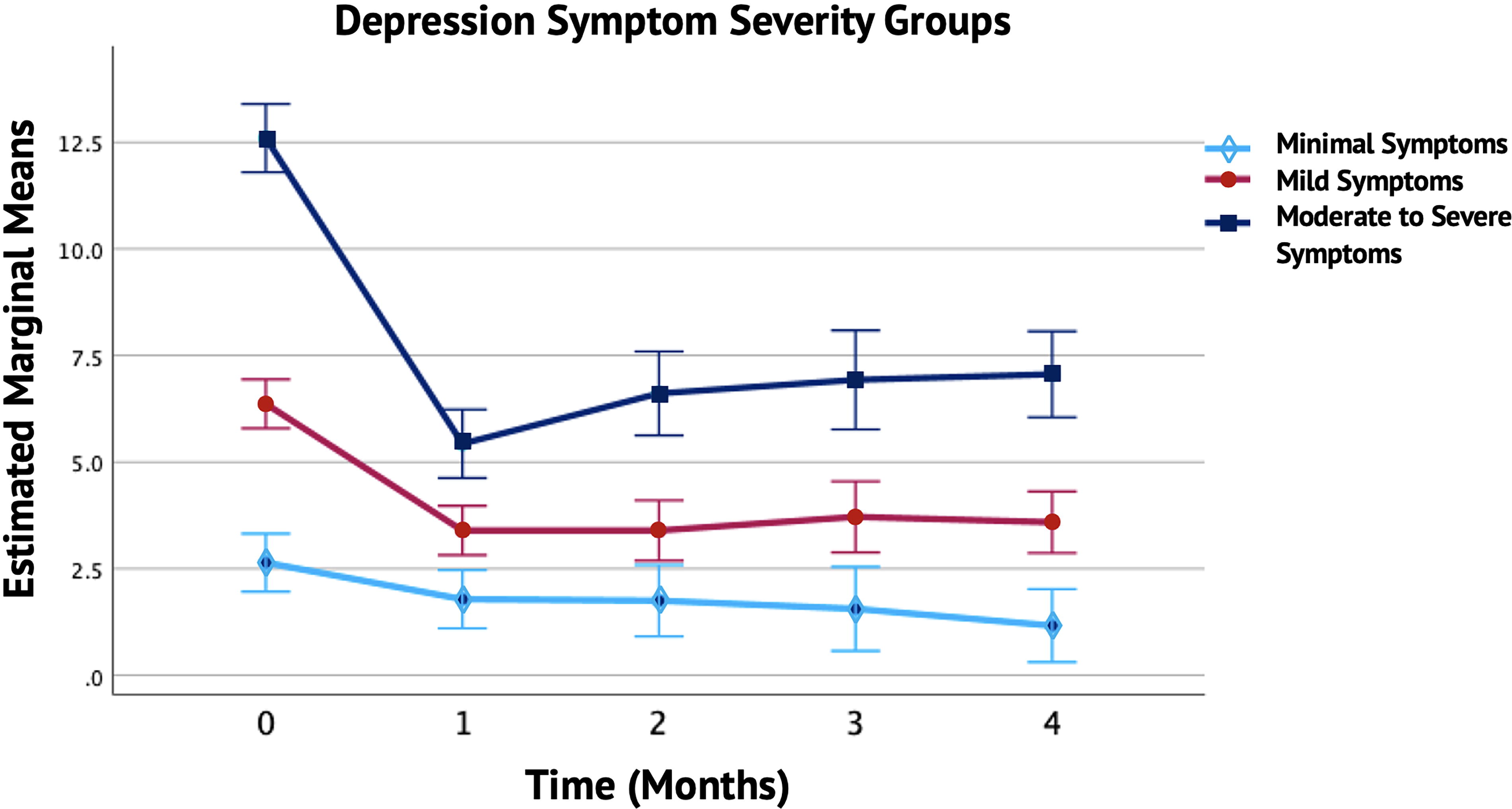
Monthly Trends in Depression Symptom Severity Groups. The figure shows the monthly trends in depression symptoms over a 4-month period for different severity groups (Minimal, Mild, and Moderate to Severe). The solid lines illustrate the changes in depression symptoms for each group, with error bars indicating the standard deviation and 95% confidence intervals (CI). In the Mild and Moderate to Severe groups, there was a significant reduction in depression symptoms compared to baseline, whereas the Minimal symptom group did not show a statistically significant change. The EMMs indicate the average depression levels for each severity group, adjusted for study year, vitamin use, meditation, and gender. Covariates appearing in the model were evaluated at the following values: vitamins = 0.29, study year = 1.48, meditation = 1.04, and gender = 1.14.
Physical symptom groups
The repeated-measures ANCOVA, adjusting for meditation, vitamin use, study year, and gender, revealed a significant main effect for physical symptoms, Huynh–Feldt F(4,364) = 12.048, p < 0.002, η2 = 0.04. Significant between-subjects effects for physical symptom groups were also observed, F(1,91) = 43.026, p < 0.001, η2 = 0.48. No other significant interactions were found. Figure 6 indicates that the adjusted mean physical symptom scores were highest in the moderate to severe group (Mean = 8.01, SE = ± 0.29), followed by the mild group (Mean = 4.99, SE = ± 0.24), and lowest in the minimal group (Mean = 3.39, SE = ± 0.56).
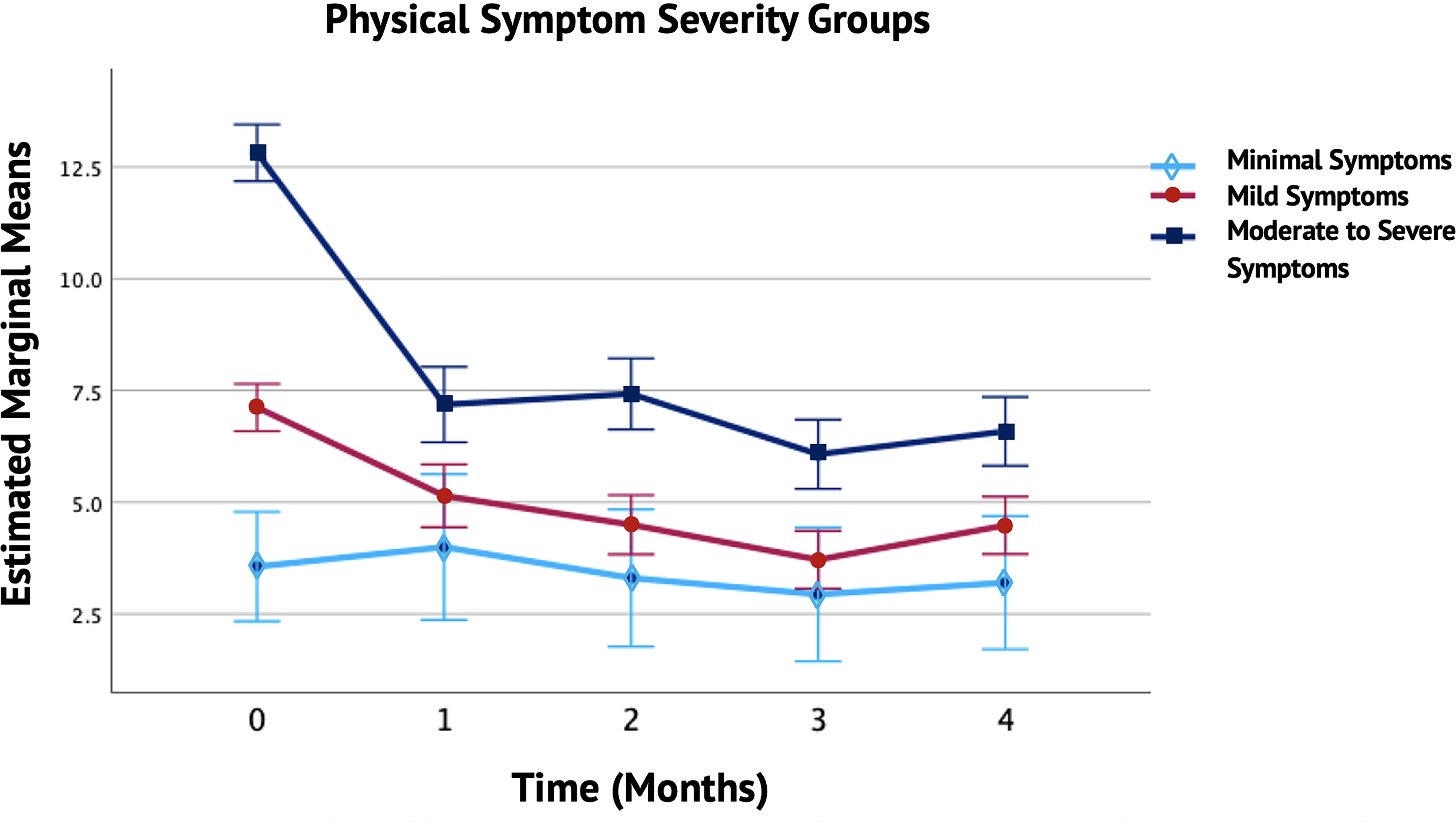
Monthly Trends in Physical Symptom Severity Groups. The figure illustrates the monthly trends in physical symptoms over a 4-month period across different severity groups (Minimal, Mild, and Moderate to Severe). The solid lines represent the changes in physical symptoms for each group, with error bars showing the standard deviation and 95% confidence intervals (CI). Significant reductions in physical symptoms were observed in the Mild and Moderate to Severe groups compared to baseline, while the Minimal symptom group did not exhibit a statistically significant change. The EMMs reflect the average levels of physical symptoms for each severity group, adjusted for study year, vitamin use, meditation, and gender. Covariates appearing in the model were evaluated at the following values: vitamins = 0.29, study year = 1.48, meditation = 1.04, and gender = 1.14.
Bonferroni-adjusted pairwise comparisons indicated significant changes in the moderate to severe and mild physical symptom groups each month compared to baseline (p < 0.001), with mean scores remaining stable. The minimal symptom group did not show a significant decrease in physical symptoms over 4 months (p = 1.00).
Feasibility and safety results
Treatment compliance
The Treatment Compliance Questionnaire, developed for this study, included items on diet (14), lifestyle (9), and herbs (1). Patients rated their adherence to each treatment component on a 5-point scale over 4 months, with (5) indicating “daily use” and (0) “never.” Compliance was highest in the first month, with scores of 50.92 ± 9.71 (SD) for diet, 30.92 ± 6.09 (SD) for lifestyle, and 4.21 ± 1.34 (SD) for herbs. By the fourth month, compliance decreased to 47.57 ± 10.32 (SD) for diet (69%), 29.94 ± 6.53 (SD) for lifestyle (67%), and 3.84 ± 1.26 (SD) for herbs (77%).
Side effects, facilitators, and barriers to intervention implementation
Participants described and graded their side effects using a survey, rating them on a 4-point scale: mild, moderate, strong, or severe. They also described the facilitators and barriers to intervention implementation and acceptability. Table 6 provides a detailed summary of the feasibility and safety outcomes from the online survey.
Feasibility and Safety Results
This table provides an overview of the feasibility and safety results, including participant compliance, barriers and facilitators, adverse events, and overall satisfaction with the treatment protocol.
Discussion
Given the significant public health concern posed by mental health issues and the potential benefits of CAM therapies, this study evaluated the effects and safety of the Ayurveda Program for Detox and Lifestyle on the mental and physical health of home-based adults. The preliminary findings indicated a significant reduction in mild-to-severe anxiety (55%) and depression (45%) symptoms by the end of the fourth month. Secondary analyses revealed a significant decrease in mild-to-severe physical symptoms, including musculoskeletal, neurological, and digestive symptoms (42%), fatigue (31%), and BMI (2%).
These changes in patient-reported outcomes are clinically relevant, reflecting significant improvements that meet the MCID for GAD-7 (4 points, score <10, ∼50% change), 25 PHQ-9 (5 points, score <10, ∼50% change), 29 PHQ-15 (2–3 points), 30 and PROMIS F-SF (3–5 points). 33 While PHQ-9 scores showed slightly less than a 50% improvement in depression for the entire group, they achieved a 5-point change in the moderate to severe depression symptom groups. Larger changes in BMI were limited as most subjects were within a healthy BMI range. However, meta-analyses indicate that even a 1 kg (2.2 pounds) reduction in overweight can improve fasting blood glucose, blood pressure, serum lipid parameters, and reduce the risk of coronary heart disease. 48 These findings underscore the clinical importance of even small reductions in overweight.
The effect sizes were large for all measures, with participants showing improvement in four out of five measures (see Supplementary Appendix A1). The Vāta, Pitta, and Kapha treatment groups improved similarly, with no significant differences. Results were consistent over 4 months. Initial improvements were significant during the first month due to the detox program, diet, daily routine, and coaching. In the post-detox phase, herbs, diet, and daily routine adjustments stabilized these changes. Participants’ clinical anxiety, depression, and physical symptoms decreased to mild, nonclinical levels. Minimal post-detox symptoms may have limited further symptom reduction.
The Ayurveda Program for Detox and Lifestyle showed improvements comparable to other treatments, with effect sizes and percentage changes ranging from 30% to 65%. 49 –51 The program’s multimodal approach enhances physiological homeostasis and enables the body to normalize suboptimal functions in the gastrointestinal, neuroendocrine, and immune systems. Research indicates that a healthy diet, yoga, daily routines, phytotherapy, oil massage, and detoxification can reduce stress, anxiety, and depression., 17 –19,43,52 –54 These practices improve gastrointestinal health, modulate neuroendocrine-immune responses, and promote neuroplasticity (Figure 7). 55 –62 Evidence also suggests that Ayurvedic treatments may influence gene expression and enhance healthy epigenetic mechanisms. 63
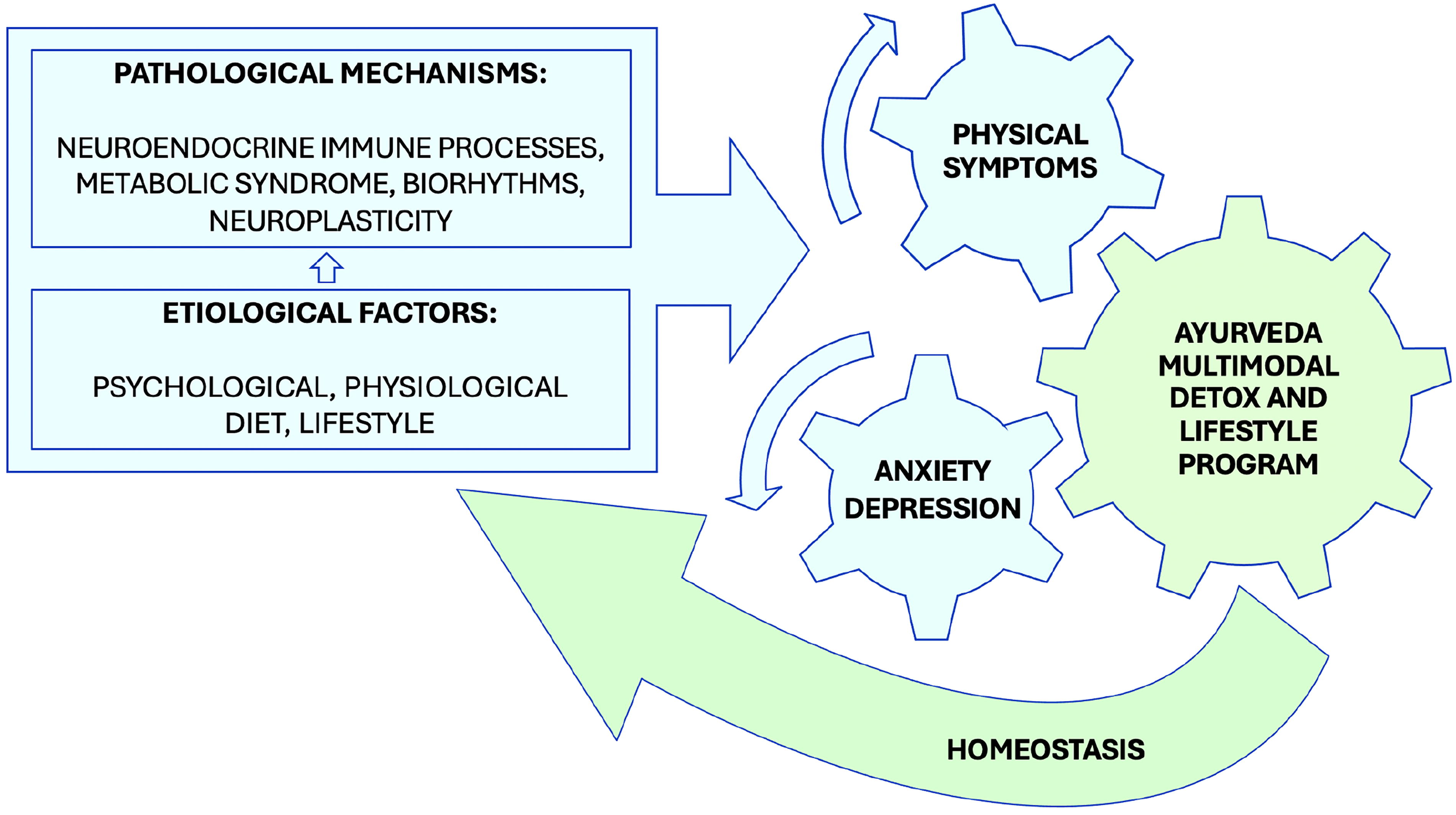
Hypothesized Mechanisms of Ayurveda Treatments. The figure illustrates the hypothesized mechanisms of the Ayurveda multimodal approach, which integrates dietary, lifestyle, herbal treatments, and detoxification to address the underlying causes and pathological mechanisms of anxiety and depression symptoms.
At the end of the first month, 6% of participants reported side effects, including weakness, constipation, tiredness, low mood, and headaches. Most side effects were mild (grade 1) or moderate (grade 2), with none being severe. Headaches were linked to caffeine withdrawal, while other side effects were associated with the castor oil cleanse. All side effects were transient, lasting no more than 2–3 days. Ayurveda-trained experts were available around the clock to provide guidance. Compared to the average 30% incidence of common side effects (e.g., gastrointestinal side effects, memory impairment, sexual dysfunction, and insomnia) associated with mental health-related pharmacological treatments, 64 –66 Ayurveda detox-related side effects were minimal, transient, and easily remedied.
The intervention was generally safe, feasible, and cost-effective, with high participant retention of 91% and compliance rates of 70%. The intervention was well-received, with 96% of participants expressing satisfaction and 93% recommending the webinar. Among the 98 participants, 83% experienced facilitating factors, while 67% encountered barriers to intervention implementation. Future studies should prioritize offering participants more frequent coaching from Ayurvedic-trained medical doctors during follow-up periods to help them overcome these barriers.
Additionally, the sample had fewer male participants (14%) and non-meditating participants (16%), and a higher mean age (58 years) than expected. Controlling for meditation, vitamin use, study year, and gender showed no significant interaction effects, indicating stable treatment effects across these variables. This consistency supports the generalizability and reliability of the findings. Future trials should enhance recruitment efforts and target specific demographics.
The trial significantly advanced the understanding of the potential clinical benefits of complementary multimodal lifestyle protocols, paving the way for larger trials and their promising integration into primary and mental health care practices.
Limitations
The single-group design cannot control for confounding factors, including placebo effects, or confirm efficacy. 67 Additionally, self-report questionnaires may introduce bias. The predominantly female, older adult, nonclinical sample could underestimate effects and limit generalizability. Furthermore, potential selection bias is a significant limitation. Online assessments and interventions also have limitations and may not address all comorbidities. Moreover, the short-term study may not capture long-term outcomes. The multimodality program prevents the determination of the contribution of each treatment, although Ayurveda traditionally uses a holistic approach. Therefore, future studies should include diverse, randomized samples, physical measures, and longer follow-up periods to enhance validity and reliability.
Conclusions
The trial found significant reductions in anxiety, depression, physical symptoms, fatigue, and BMI over 4 months in 98 home-based adults with mild to severe symptoms. These improvements had large effect sizes, indicating clinically significant effects. Future randomized controlled trials are needed to control for confounding factors and validate these findings in larger clinical populations. This study offers health professionals protocols for natural, holistic, accessible, and complementary treatments for anxiety, depression, and physical symptoms.
Footnotes
Acknowledgments
The authors gratefully acknowledge Dr. Wolfgang Schachinger, Dr. Valeria Schachinger, and the team at the Center for Maharishi AyurVeda and Holistic Medicine, Soma, Austria, for their collaboration in intervention development, delivery, and participant enrollment. The authors also sincerely appreciate the expertise of Dr. Marcia Murphy, Dr. Peter Swan, Dr. Jane Bialosky, Dr. William F. Sands, and Dr. Susan L. Dillbeck.
Authors’ Contributions
V.L.: Conceptualization, Methodology, Investigation, Resources, Project administration, Data Curation, Formal analysis, writing-original draft, and visualization. F.T.: Supervision and Methodology. C.A.G.: Critical review. G.M.: Critical review. R.H.S.: Supervision and Critical review
Data Availability Statement
The research data supporting this study’s findings are not publicly accessible due to privacy and ethical constraints related to patient consent. However, anonymized data may be available from the authors upon request with permission from the institutional review board of Maharishi International University, Fairfield, Iowa.
Author Disclosure Statement
The authors declare no personal financial interests, such as stocks, shares, consulting fees, or patents, that could gain or lose value from the publication of this article. The research was not supported by organizations that may gain or lose financially from its publication. The authors are employed by a university where they teach and consult on Ayurveda. They commit to maintaining objectivity and transparency in their reporting and declare no conflicts of interest that may influence decisions related to the study design, data analysis, reporting, or publication of this article.
Funding Information
The study did not receive any funding. All volunteer study participants personally purchased the herbal preparations.
Supplementary Material
Supplementary Appendix A1
Supplementary Appendix A2
Supplementary Appendix A3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
