Abstract
Background:
Withania somnifera (Linn) or Ashwagandha is used in Ayurveda and other traditional medicine systems as an adaptogen and a neuroprotective supplement.
Objective:
The effect of Ashwagandha root extract (ARE) standardized for 2.5% full-spectrum withanolides as per The United States Pharmacopeia (USP) protocol with piperine (500 mg with 5 mg of 95% piperine) once a day (12.5 mg withanolides/day) was evaluated in individuals with mild to moderate depression and anxiety.
Methods:
In a randomized, double-blind placebo-controlled study, for 90 days, 70 participants were randomized to ARE (n = 34) or placebo (n = 36) once daily at night. Mean change in the Hamilton Depression Rating Scale (HDRS) and Hamilton Anxiety Rating Scale (HARS), Groningen Sleep Quality Scale (GSQS), and quality of life (QOL) from screening to days 30, 60, and 90 were evaluated. Safety was evaluated by monitoring any incidence of adverse events and laboratory parameters. Two-way analysis of variance (ANOVA) and repeated-measure ANOVA were used to compare ARE and placebo, and the changes within the group at different time points.
Results:
Seventy individuals were randomized and all of them completed the study. The HARS, HDRS, GSQS, and QOL scores improved significantly (p < 0.001) in all the participants taking ARE compared to placebo on days 30, 60, and 90. Anxiety and depression improved from baseline to end of the study in both groups, but the quantum of improvement was significantly higher in ARE. Serum levels of serotonin increased in ARE, but showed a decrease in placebo, the difference being statically significant (p < 0.001). Biochemical and hematological parameters remained in the normal range in all participants and ARE was well tolerated during the study.
Conclusion:
The results of the study suggest that 500 mg of ARE standardized for 2.5% withanolides with 5 mg piperine is beneficial in improving depression, and anxiety, by increasing serum serotonin levels. The trial was registered prospectively with the Clinical Trial Registry of India (CTRI) with the registration number CTRI/2022/05/042640, on May 18, 2022.
Introduction
Anxiety and depression are the most common comorbid psychological disorders having similar emotional profiles. 1 While anxiety is a negative emotional state dominated by fear and apprehension, depression is a mental health condition characterized by a persistent feeling of debilitating low mood, sadness, loss of interest, and impaired cognition. 2,3 According to the World Health Organization, 4.4% and 3.6% of the world's population is affected by depression and anxiety disorders. Interestingly, more women are affected by depression than men. 4
Depression affects the personal and social behavior of the affected individual and may also result in an inability to experience pleasure, changes in sleep and eating behaviors, and difficulty in concentrating and even suicidal thoughts over time. 5 Depression may develop due to social and psychological issues, but can also have a biological predisposition due to genetic abnormalities, stress, dysbiosis, sleep disorders, anxiety, and dysfunction of kynurenine pathway. 6,7 The deficiency of monoamine neurotransmitters like serotonin, noradrenaline, and dopamine is also believed to play a role in the development of depression. 7 In addition, a correlation between cortisol levels and low-grade inflammation with depression has been reported in experimental studies. 8 –10 The deficiency of monoamines is also involved in the development of anxiety disorders. 11 Apart from the disruption of gamma-aminobutyric acid (GABA), regulation of cortisol and the role of cholecystokinin have been implicated in regulating emotional states of an individual. 12,13
Tricyclic antidepressants, monoamine oxidase inhibitors, selective serotonin reuptake inhibitors, serotonin and norepinephrine reuptake inhibitors, norepinephrine and dopamine reuptake inhibitors, and serotonin antagonist and reuptake inhibitors are used for the pharmacological treatment of anxiety and depression. 14,15 Headaches, sexual dysfunction, addiction, seizures, and suicide are few of the common side effects associated with antidepressant and anxiolytic medications. 16 Natural products are a safer alternative with lower adverse effects and have seen a considerable increase in their use in the past decades. 17,18
Withania somnifera (Linn) Dunal, or Ashwagandha, is a traditional Ayurvedic adaptogen used to enhance muscle strength, endurance, and overall health. 19 Ashwagandha is mentioned as a “Rasayana” in ancient texts such as the Sushruta Samhita 20 and the Charaka Samhita, 21 written by Sage Sushruta and Sage Charaka around 1000 BCE and 300 BCE, respectively.
Withanolides are the most studied pharmacologically active phytoconstituents in the plant. 22 –24 Ashwagandha has been shown to possess anxiolytic, antidepressant, and neuroprotective effects in several animal models and clinical studies. 25 –30
In this study, we used a standardized root extract of W. somnifera (Linn), containing 2.5% withanolides (Shagandha®), and analyzed by using The United States Pharmacopeia (USP) monograph (High-Performance Liquid Chromatography [HPLC]). 31 Thus, the extract contains standardized amounts of withanolides compared with several uncharacterized other Ashwagandha extracts studied earlier and is fortified with the bioavailability enhancer, piperine. 32,33 The concept of bioavailability enhancers has been described in the traditional Ayurvedic system of medicine. Pepper derived from the plant Piper nigrum is one of the components of “trikatu,” along with long pepper and ginger, meaning three acrids. 34 Piperine has been shown to increase the absorption of drug in the intestine, reduce their metabolism, and inhibit drug efflux. 35 We evaluated the clinical efficacy and safety of this extract in improving the symptoms of depression and anxiety in adults with mild to moderate depression and anxiety.
Materials and Methods
Materials
The test material was a root extract of W. somnifera (Linn) (Shagandha) standardized to contain 2.5% withanolides. Each capsule contained 500 mg of extract with 5 mg of 95% piperine (BioPerine®) from P. nigrum. Piperine is known to increase the bioavailability of drugs and phytochemicals. 32,33 Microcrystalline cellulose capsules were used as placebo. The product was provided by Sami-Sabinsa Group Limited. The HPLC profile of the extract is given in Figure 1.
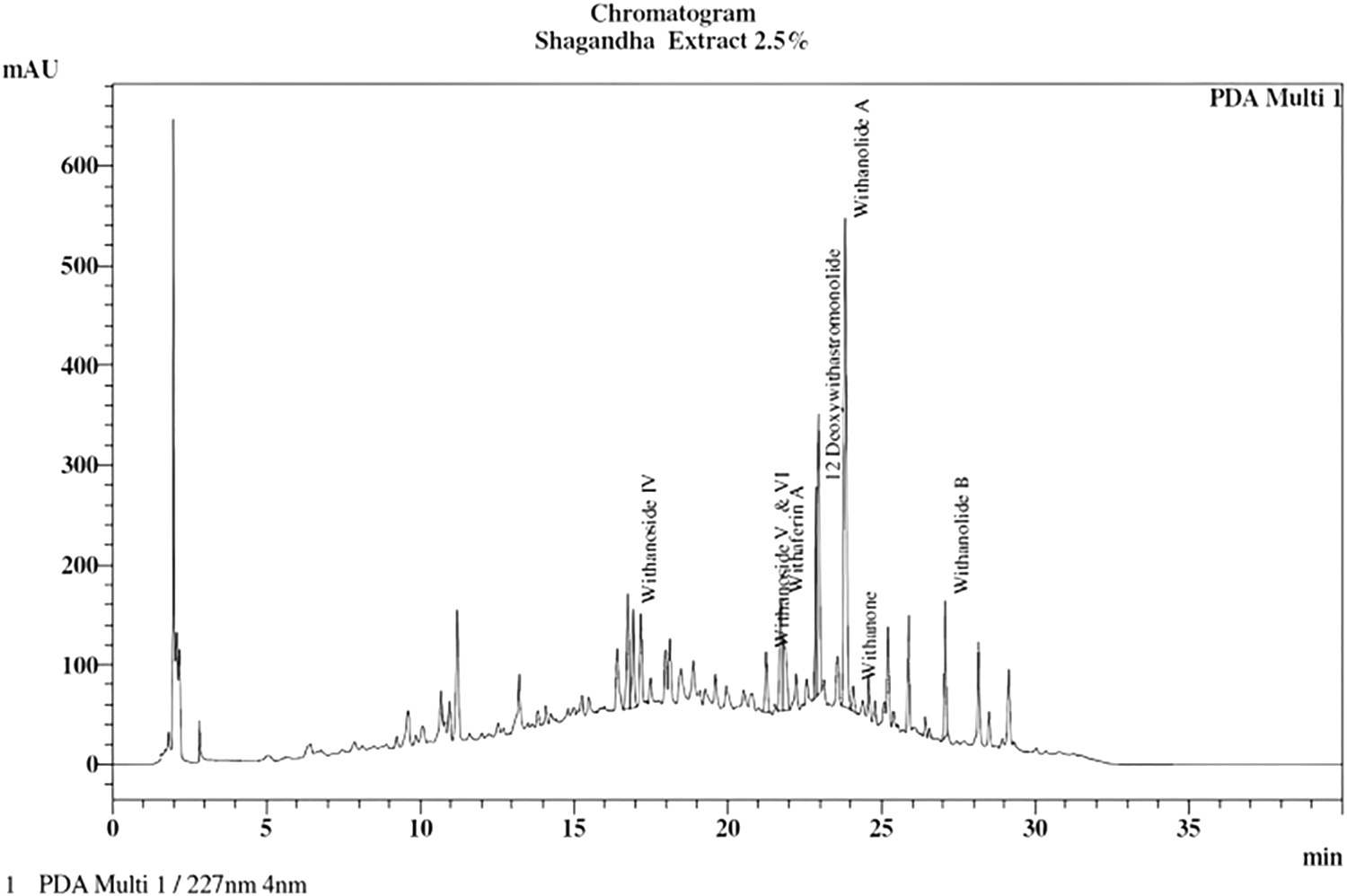
HPLC profile of standardized root extract of ashwagandha, containing 2.5% withanolides (Shagandha®), and analyzed by using The United States Pharmacopeia (USP) monograph. HPLC, High Performance Liquid Chromatography.
Study design and ethics
The study was conducted as a randomized, double-blind placebo-controlled design from May 19, 2022, to September 28, 2022, as per Declaration of Helsinki, and “Guidance on Good Clinical Practice” (GCP) requirements. The study was approved by the independent ethics committee of Apollo First Med Hospitals, Chennai, and DEC-Health Care Hospital, Nellore, for Protocol No. CW/103/ASH_DEPN/II/NOV/21. An informed consent form to participate in the study was signed by all the participants.
Sample size
The sample size was calculated for an alpha error of 0.05 and power at 80% and assuming a correlation of 0.48, based on the research publication by Chandrasekhar et al. 28 The sample size was calculated to be 60 for evaluation. Assuming a dropout rate of 15%, the sample size was fixed at 70, with 35 participants in each group. The sample size calculations are given in Supplementary Methods section.
Study population
Inclusion criteria
The study included adult participants in the age group of 18–60 years with a Hamilton Depression Rating Scale (HDRS) of 8–18 and Hamilton Anxiety Rating Scale (HARS) from 6 to 24.
Exclusion criteria
Individuals with major psychiatric, medical, or neurocognitive disorders that make participation infeasible or interfere with the adherence to the lifestyle intervention, using medication or psychotherapy for depression, or taking Ayurvedic/herbal/homeopathic/dietary supplements (including Vitamin E) or any alternative therapy in the last 1 month were excluded from the study. Participants with a history of severe allergies or anaphylactic reactions, alcohol abuse, excessive smoking, and hypersensitivity to the investigational product were also excluded from the study. Other exclusion criteria included the presence of chronic gastrointestinal and genitourinary diseases, severe immune deficiency, thyroid disorders, respiratory diseases, acute medical/surgical complications, uncontrolled diabetes, or hypertension and abnormal serum biochemical parameters. Those who participated in any clinical study during the previous 3 months, and pregnant/lactating women were excluded from the study. Informed consent to participate in the study was signed by all the participants.
Randomization and blinding
The randomization sequence was prepared by the statistician, using computer-based randomization software (SAS 9.3), independent of the sponsoring organization. The principal investigator assigned the participants to two different groups by using the alphabetic codes. A sealed envelope containing the treatment codes was provided to the Principal Investigator to be broken only in case of an emergency, that is, serious adverse event (AE).
The randomization sequence was prepared by a statistician, independent of the sponsoring organization and not involved in the conduct or reporting of the study. An alpha code was generated for both the active and placebo to improve the blindness of the study and concealment of allocations. Block randomization with a block size of 6 was followed, wherein the subjects were randomized to receive either a placebo or the ashwagandha extract. The randomization codes were kept strictly confidential and were accessible only to authorized persons on an emergency basis, as per the Sponsor's standard operating procedures, until the time of unblinding.
The investigator, sponsor, study monitors, and participants were blinded to the study intervention. The study supplements were exactly similar, coded and supplied to the study site, in a manner to keep the blinding throughout the study. The details of randomization are given in Supplementary Methods.
Intervention and compliance
The participants were instructed to take 500 mg of ashwagandha root extract (ARE), standardized for 2.5% withanolides as per The United States Pharmacopeia method (Shagandha) containing 5 mg of piperine (ARE), or an identical placebo capsule once a day after dinner for 90 days. The study did not include psychotherapy.
The study period consisted of five scheduled visits, including a screening visit, a baseline visit (day 0), and follow-up visits on days 30, 60, and 90 and a telephonic follow-up, 15 days from the date of the last visit to assess the occurrence of any AE and overall well-being. Empty or unused study medication bottles were collected, and Investigational Product accountability was checked and recorded in the case record form to assess study compliance. Compliance was assessed through record and review in the data collection tool used during the conduct of the clinical study. A record of the number of capsules dispensed to and administered by the subject and returned at each visit was kept in the Case Record Form.
Outcome
The primary endpoints of the study were mean change in the depression and anxiety scores, HDRS and HARS, from screening to days 30, 60, and 90. The secondary endpoint included mean change in Groningen Sleep Quality Scale (GSQS) and WHO-quality of life (WHO-QOL) Questionnaire from day 0 to days 30, 60, and 90 and serum serotonin levels from day 0 to 90. Safety was determined by changes in blood biochemistry, hematology, urine profile from day 0 to 90, and occurrence of AEs throughout the study.
Measures
HDRS is used to quantify the severity of symptoms of depression and is one of the most widely used and accepted instruments for assessing depression. The standard version of the HDRS contains 17 items rated on a 4-point scale, with the sum of all items making up the total score. An HDRS score of 0–7 is considered normal, 8–13 mild depression, 14–18 moderate depression, 19–22 severe depression, and more than 23 very severe depression. 36,37
HARS is a widely used 14-item rating tool to measure the severity of anxiety symptoms among individuals previously diagnosed with anxiety disorders. The 14 items reflect 13 categories of anxiety-related symptoms, including anxious mood, tension, fear, insomnia, intellectual/cognitive symptoms, depressed mood, general somatic (muscular and memory symptoms), cardiovascular, respiratory, genitourinary, and gastrointestinal symptoms, with one item capturing the rater's assessment of behavioral symptoms. Each item is scored on a scale of 0 (not present) to 4 (severe), with a total score range of 0–56, where <17 indicates mild severity, 18–24 mild to moderate severity, and 25–30 moderate to severe. 38
The GSQS-15 evaluates the subjective quality of sleep, covering the general quality of sleep, insufficient sleep, problems of falling asleep, and waking up feeling unrested. The scale was originally constructed to study sleeping problems of depressed patients. 39 GSQS score ranges from 0 to 14, a higher score indicating lower subjective quality of sleep.
The WHOQOL-Bref is a frequently used generic questionnaire for the assessment of QoL across different populations. It captures many subjective aspects of quality of life (QOL) and is one of the best-known instruments that has been developed for cross-cultural comparisons of QOL and is available in more than 40 languages. 40
Biomarkers and safety parameters
Serum serotonin levels were measured by ELISA at Suburban Diagnostics Lab as per the manufacturer's instructions.
Safety analysis
The principal investigator monitored safety data throughout the course of the study, which included monitoring of AEs and/or serious AEs, laboratory investigations, and vital signs. Regular serum biochemical tests and hematological and urine analyses were carried out at Apollo & Vijaya Diagnostic Centre.
Statistical analysis
The normality of quantitative variables was analyzed using the Shapiro-Wilks test, and the data are represented as mean, standard deviation, or median, and range based on their distribution. The differences within the group at different time points were compared by repeated-measures analysis of variance (ANOVA) and Dunnett's Multiple Comparison post hoc test. The differences between the groups at different time points were analyzed by two-way ANOVA and the change in values from day 0 to 90 by Mann–Whitney test. All the statistical analysis was performed by STATA Software version 16.0 by an independent statistician blinded to the study groups. The level of significance was defined as a p-value <0.05.
Results
Demographic and baseline characteristics
The principal investigators prescreened 94 probable subjects and 72 participants were considered for screening. There was one screen failure and one participant did not come for the screening visit. Thus, 70 were found eligible and were randomized for intervention, 34 in ARE and 36 in placebo groups. As per the statistical software (SAS 9.4), the randomization codes were generated in a block of 6. There were two sites. One site recruited 36 participants, 18 each in ARE and placebo. The second site recruited 34 participants, distributed as 16 in ARE and 18 in placebo as per the 6-block randomization schedule. All the participants completed the study (Fig. 2). The mean age of subjects was 40.69 ± 11.34 years, 43 (61.42%) were men and 27 (38.57%) were women. Table 1 represents the baseline characteristics of enrolled patients.
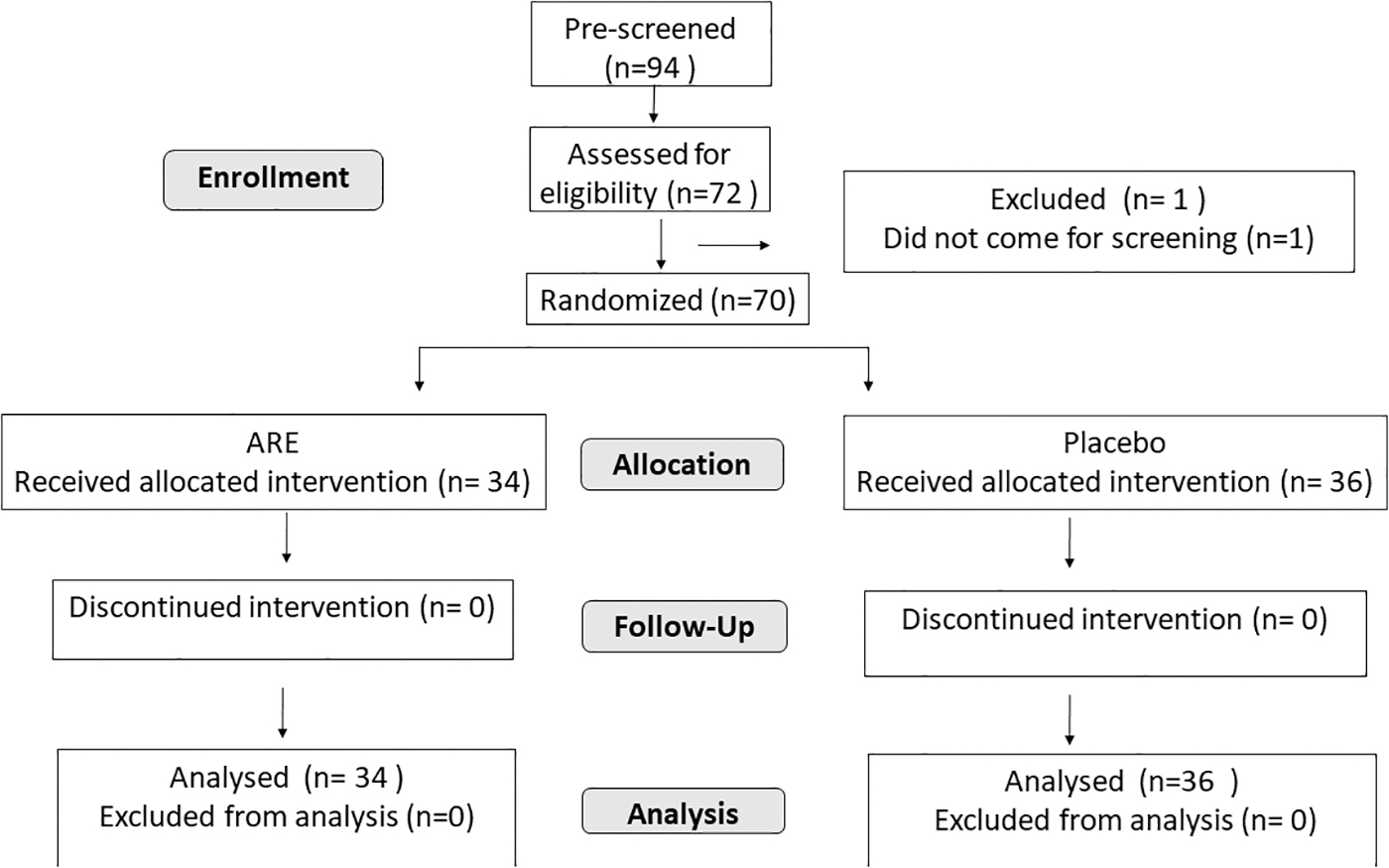
Consort flow diagram showing the participant disposition.
Demographic and Baseline Characteristics of Study Subjects
Data are represented as mean ± SD for anthropometric parameters, body temperature, and respiratory rate. Gender, race, smoking, alcohol, and drug abuse history as number of participants N (%), while BP and pulse rate are given as median and range.
ARE, Ashwagandha root extract; BMI, body mass index; BP, blood pressure; SD, standard deviation.
Hamilton Anxiety Rating Scale
The HARS scores were assessed on day 0, 30, 60, and 90, for all the participants. A significant decrease in the depression scores was observed as early as a month of supplementation with ARE. The scores decreased from 17.09 ± 4.45 on day 0 to 15.03 ± 3.08 on day 30, 11.53 ± 3.13 on day 60, and to 8.18 ± 3.20 on day 90. In comparison, the decrease in placebo was 18.68 ± 3.94 to 17.00 ± 3.76, 17.06 ± 3.50, and 16.14 ± 3.09 on days 0, 30, 60, and 90. The change in the score was −8.06 U (52.91%) on day 90, was significantly better in ARE (p < 0.001) compared to placebo (−1.78 U, 11.65%). Two-way ANOVA revealed a significant difference between ARE and placebo for HDRS scores on days 60 and 90 (Fig. 3A and Table 2).
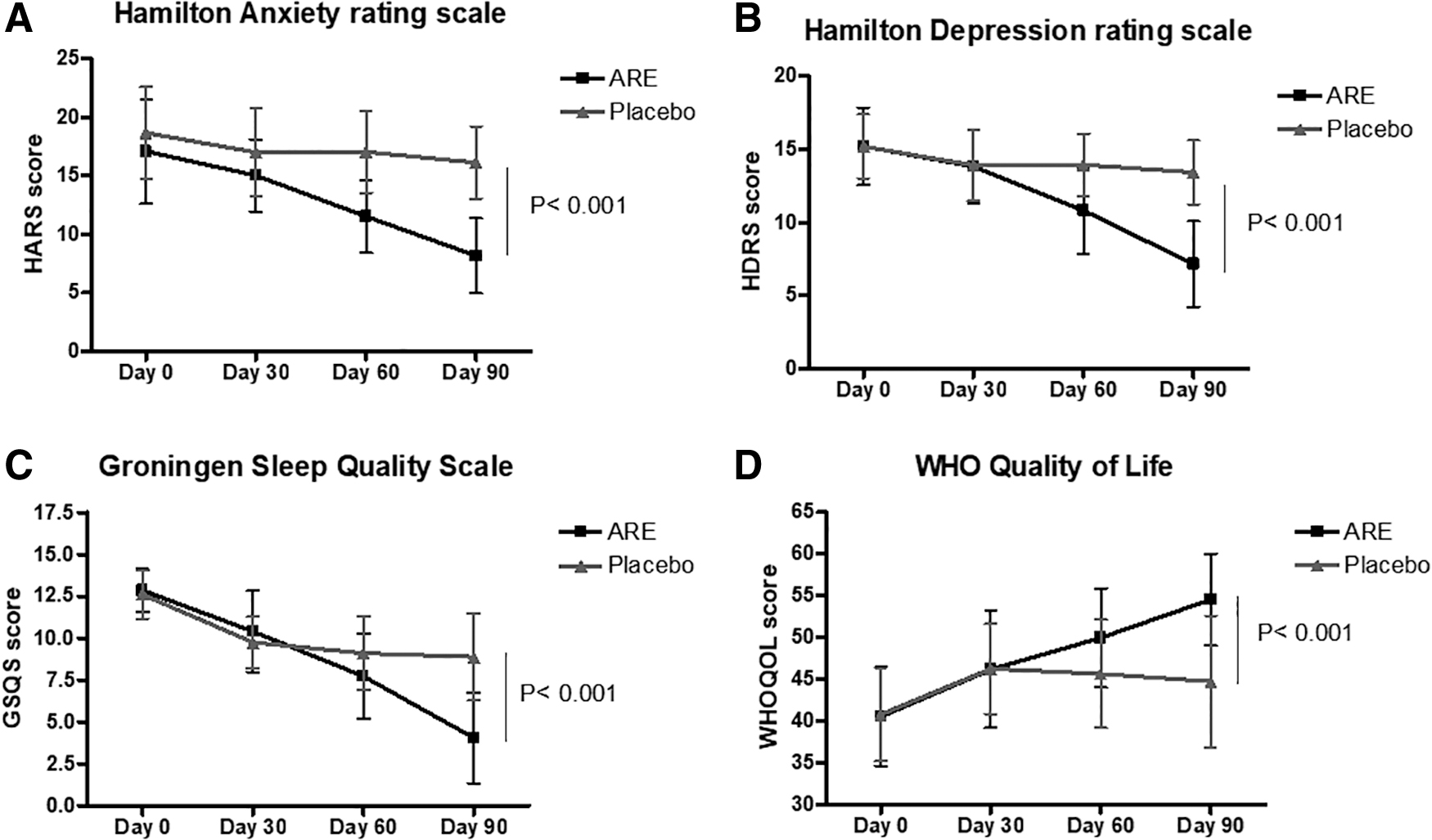
Change in anxiety, depression, QOL, and sleep from baseline to day 90. The HARS
Mean Differences in Efficacy Parameters from Baseline to End of the Study
Data are represented as mean ± SD. Serotonin levels were not normally distributed; hence median (interquartile range) is given. Significance (*** p < 0.001) within the group from baseline to end of the study. The significance (p value) of the mean change in scores between ARE and placebo are represented in the table.
GSQS, Groningen Sleep Quality Scale; HARS, Hamilton anxiety rating scale; HDRS, Hamilton depression rating scale; SD, standard deviation; WHOQOL questionnaire, world health organization quality-of-life questionnaire.
Hamilton Depression Rating Scale
The HDRS scores showed a similar trend as HARS in the reduction of anxiety in the ARE group. The HDRS scores decreased from 15.21 ± 4.45 on day 0 to 15.03 ± 3.08 on day 30, 11.53 ± 3.13 on day 60, and to 8.18 ± 3.20 on day 90. In comparison, the decrease in placebo was 15.19 ± 2.18 to 13.91 ± 2.45, 13.94 ± 2.14, and 13.42 ± 2.18 on days 0, 30, 60, and 90. The change in the score was −8.91 U (52.13%) on day 90 compared to day 0 in the ARE group and −2.56 U (13.59%) in placebo, which was significantly better (p < 0.001). Two-way ANOVA revealed a significant difference between ARE and placebo for HARS scores, on day 60 and 90 (Fig. 3B and Table 2).
The Groningen Sleep Quality Scale (GSQS-15)
The quality of sleep improved in both ARE and placebo, but the extent of improvement was significantly better (p < 0.001) in ARE compared to placebo. The score was comparable on day 0 at 12.85 ± 1.28 in ARE and 12.61 ± 1.44 in placebo. A gradual improvement was observed in ARE group (10.41 ± 2.43 on day 30, to 7.74 ± 2.53 on day 60 and 4.06 ± 2.70 on day 90) and placebo (9.75 ± 1.54, 9.11 ± 2.19, and 8.89 ± 2.53 on days 30, 60, and 90, respectively). The sleep index changed by −8.79 U (68.40%) in ARE and −3.72 U (29.50%) in placebo, which was significant between the groups (p < 0.001). Two-way ANOVA showed a significant difference between the groups on days 60 and 90 (Fig. 3C and Table 2).
Change in QOL (WHOQOL Score) from baseline to the final visit
The WHOQOL measures the physical, psychological, and social relationships, environment, and general (Q16) health status of the individual. An increase in the score indicates better QOL. The QOL showed an increase of 34.47% in ARE and 9.57% in placebo from day 0 to 90, which was significant (p < 0.001) between ARE and placebo by two-way ANOVA analysis. The WHOQOL scores on day 0 were 40.55 ± 5.94 in ARE and 40.82 ± 5.58 in placebo. The scores increased to 46.18 ± 7.01 in ARE and 46.25 ± 5.39 in placebo on day 30, to 49.95 ± 5.93 and 45.64 ± 6.48 in ARE and placebo, respectively, on day 60, and to 54.53 ± 5.46 and 44.73 ± 7.88 on day 90 in ARE and placebo, respectively (Fig. 3D and Table 2).
Serotonin levels from baseline to final visit
The serum serotonin levels increased in ARE, but showed a decreasing trend in placebo. The median serotonin levels in serum increased from 87.43 (56.39, 134.00) to 132.24 (91.43, 188.30) in ARE, while the values decreased from 86.00 (53.43, 159.50) to 65.93 (28.88, 131.23) in placebo. The mean difference from day 0 to 90 was significant between ARE and placebo (Fig. 4 and Table 2).
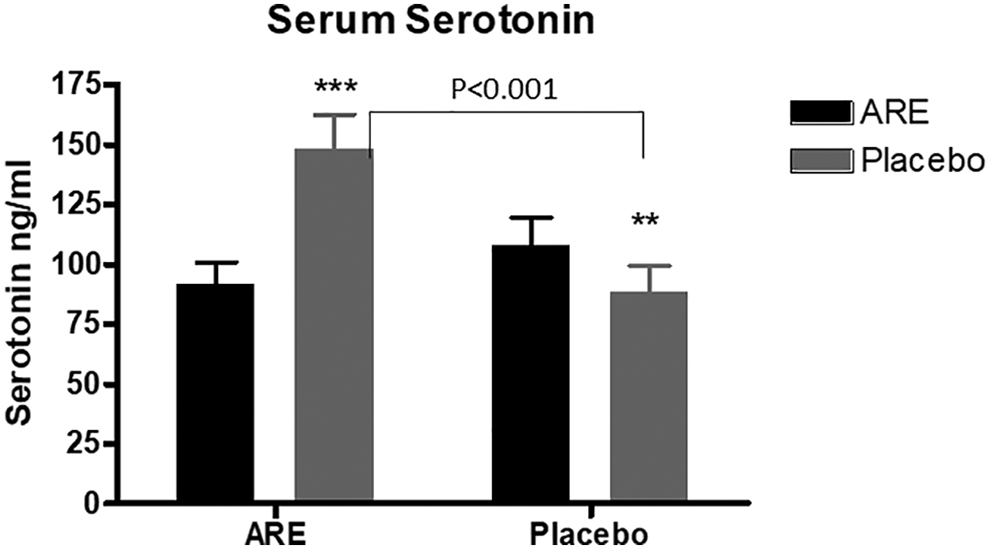
Change in serum serotonin levels from baseline to day 90 (Mean ± SEM). Serotonin levels in serum at baseline and on day 90 in ARE and placebo groups. **p < 0.01, ***p < 0.001. p Value represents the difference between ARE and placebo on day 90.
Safety
Eighteen subjects in ARE and 16 subjects in placebo reported mild discomforts like headache, nausea, diarrhea, drowsiness, fever, back pain, and stomach pain. All the events were transient and were resolved within 24 h (Table 3 and Supplementary Table S1). No patient discontinued the study due to AEs or reported any serious AE. The biochemical and hematological parameters were in the normal range in both ARE and placebo groups (Supplementary Tables S2–S4).
Summary Statistics of Safety Assessment by Adverse Events
AE, adverse event.
Discussion
We observed significant improvements in depression, anxiety, and sleep quality in individuals consuming 500 mg ARE standardized for 2.5% total withanolides (Shagandha) for 90 days in this randomized, double-blind placebo-controlled study. All participants (100%) in the ARE group reported an improvement in anxiety and depression compared to 66.6% in placebo. The magnitude of improvements was 52.1% for anxiety and 52.9% for depression in ARE compared to 13.6% and 11.6% in placebo. Improvement in sleep and QOL was reported by 100% and 97% of participants, respectively, in ARE and 83.3% and 55.5% in placebo, ARE being significantly better than the effect of placebo
Ashwagandha has been reported to alleviate anxiety and stress in several studies earlier. 28,29,41,42 In a recent meta-analysis, ashwagandha supplementation was reported to significantly reduce anxiety (standardized mean difference: −1.55, 95% confidence interval: −2.37 to −0.74; p = 0.005; I 2 = 93.8%). 43 Our results agree with these studies, at a lower dosage of total withanolides. The ARE used in this study was standardized for 2.5% of withanolides corresponding to 12.5 mg of withanolides in the 500 mg dose. Earlier studies have used ashwagandha extracts with 15–48 mg of effective withanolides.
The effect of ashwagandha in depression has been evaluated in fewer studies compared to anxiety and stress. Chauhan et al. showed a beneficial effect of ashwagandha (500 mg/day) for 6 weeks in moderately depressed elderly women. 44 ARE containing 5% withanolides at a dose of 300 mg twice a day was reported to significantly reduce in scores corresponding to depression by 79.2% after 60 days. 28 In another study, ashwagandha supplementation at 100 mg per day for 12 weeks showed promising effect on alleviating depression in patients with schizophrenia. 45 In rodent models of depression and anxiety, treatment with ashwagandha extract could demonstrate significant antidepressant effect. 46 –48
Glycowithanolides from ashwagandha roots were found to exhibit an antidepressant effect, comparable with that of imipramine in a rat model. 26 Our results are concurrent with these reports and demonstrate that even at a dose of 12.5 mg of withanolides per day, ARE was beneficial in improving the depression scores in individuals with mild to moderate depression.
Sleep disorders are frequently observed as comorbidity in depression and is a risk factor for both incident and recurrent episodes of depression. 49 In fact, depression and insomnia share a bidirectional relationship as the presence of one is a risk factor for the development of the other disorder. 50 A recent meta-analysis revealed moderate to large effect size improvement in depression in individuals treated for insomnia. 51 Ashwagandha extract has been shown to improve sleep quality in clinical studies. 52 We observed a significant improvement in sleep pattern in participants taking ARE, suggesting an overall benefit in improving anxiety depression and sleep pattern. It is worthwhile to mention that an improvement in sleep was also observed in the placebo, This could be due to placebo effect, which is known for perceived insomnia symptoms. 53 However, the effect of ARE was significantly better than placebo.
To further understand the possible mechanism of action of ARE, we estimated the change in serum serotonin levels during the study. A deficit in monoamine neurotransmitters (serotonin, dopamine, and noradrenaline) is responsible for the development of depression. 7 ARE supplementation increased the serum serotonin levels significantly compared to placebo, suggesting a possible mechanism of action through this neurotransmitter activity. Earlier studies have shown that withanolide A could increase serotonin mRNA in Caenorhabditis elegans and alleviate stress and depression. 54
In an experimental study in Equine stress model, Priyanka et al. observed an increase in serotonin levels in response to administration of ashwagandha root powder for 21 days. 55 Bhatnagar et al. observed that ARE suppresses corticosterone release and activates choline acetyltransferase, which in turn increases serotonin level in hippocampus in experimental rodents. 56 We also observed an increase in serotonin levels with ARE supplantation in our earlier study (unpublished observation). Ashwagandha supplementation has been shown to have an antioxidant and anti-inflammatory effect and reduce serum cortisol levels in stressed adults, 41,57 –60 which could have also helped in the increase in serotonin levels.
Participants taking ARE reported a significant improvement in WHOQOL and was well tolerated with no change in hematological and biochemical parameters and no major AE was reported over time.
The strength of the studies includes assessment of anxiety, depression, sleep quality serum, and serotonin levels in the same population to understand the effect of Ashwagandha on the interrelated parameters. The limitations of the study could be a relatively smaller sample size from a single ethnic population. We estimated only serotonin levels in this study, while cortisol, noradrenaline, dopamine, and GABA are also known to play an important role in the development of anxiety and depression, which could be considered a study limitation. Future studies in diverse populations with different degrees of depression, anxiety, and related disorders and follow-up after termination of supplementation will help to understand the sustained effects of ARE. Multiple, interdependent pathways are involved in the development of depression and anxiety. Future studies, including the effect of ARE on the hypothalamic-pituitary-adrenal axis, inflammation, and other neurotransmitters, would delineate the mechanism of action of the supplement.
Conclusions
In conclusion, the results of this study suggest a positive impact of ARE (Shagandha, standardized for 2.5% withanolides) at 500 mg with 5 mg of 95% piperine once a day on individuals with mild to moderate depression and anxiety. ARE increased serum serotonin levels, suggesting its mechanism of action through the monoamine neurotransmitters. The results of the study were comparable to earlier studies conducted with ashwagandha extracts containing a higher quantum of withanolides. Future studies in diverse populations would substantiate our results.
Footnotes
Acknowledgments
The authors thank Mr. Kamal Kammili and his team at Sanjeevani BioSolutions, Hyderabad, for the statistical analysis and the clinical team associated with the study.
Authors' Contributions
Conceptualization: M.M. and K.N.; methodology and validation: L.M. and A.M.; formal analysis: A.M. and L.M.; investigation: M.V.R. and T.V.D.; resources: M.M..; data curation: A.M. and L.M.; writing—original draft preparation: L.M.; writing—review and editing: K.N., M.M., A.M., and L.M.; supervision: K.N.; project administration: K.N.; and funding acquisition: M.M. All authors have read and agreed to the published version of the article.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Author Disclosure Statement
M.M., K.N., A.M., and L.M. are employees of Sami-Sabinsa Group Limited/Sabinsa Corporation that markets Ashwagandha products. M.V.R. and T.V.D. do not have any conflicts to declare. All the authors have agreed on the contents of the article. The study was conducted by the principal investigators: T.V.D. and M.V.R. The results were analyzed by an independent statistician. The investigators, sponsors, and the subjects were blinded to the study.
Funding Information
This research received no external funding. The study was funded by Sami-Sabinsa corporation.
Supplementary Material
Supplementary Methods
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
