Abstract
Introduction:
Postoperative pain control is a challenge in enhanced recovery after surgery (ERAS). The current study reviewed the efficacy and safety of incorporating acupoint stimulation for postoperative pain control in ERAS.
Methods:
Ten databases for relevant randomized controlled trials (RCTs) published in English or Mandarin Chinese were searched from 1997 to 2022. The quality of each article was appraised using the Cochrane Collaboration Risk of Bias Criteria and the modified Jadad Scale. The primary outcome was pain control, measured using the visual analog scale 24 h after surgery.
Results:
Eleven trials met the eligibility criteria and were included in the study. Acupoint stimulation was found more effective than control treatments in terms of pain intensity (standardized mean difference [SMD] −0.94; 95% confidence interval [CI] −1.35 to −0.53), analgesic drug consumption (SMD −1.87; 95% CI −2.98 to −0.75), postoperative nausea (PON; SMD 0.31; 95% CI 0.13 to 0.73), postoperative vomiting (POV; SMD 0.57; 95% CI 0.11 to 2.92), and PON and POV (PONV; SMD 0.29; 95% CI 0.16 to 0.53). The Zusanli (ST36) and Neiguan (PC6) were the most-used acupoints in the included trials (8/11). The reported adverse reaction was only one case of bruising.
Discussion:
Acupoint stimulation improved pain control in patients undergoing ERAS more than control treatments. The findings provide an evidence-based premise for incorporating acupoint stimulation into ERAS strategies. More rigorous RCTs are needed in the future.
Introduction
Enhanced recovery after surgery (ERAS), a surgical strategy based on the pathophysiology of postoperative recovery, achieves optimal outcomes utilizing integrated, multidisciplinary, and multimodal perioperative protocols. 1 ERAS has yielded substantial benefits reducing medical complications and hospitalizations without increased readmissions 2 and has become a standardized perioperative care across surgical subspecialties. 3,4 However, challenges such as gastrointestinal dysfunction and postoperative pain persist, and a comprehensive analgesic plan is critical for ERAS success. 5,6 With over 200 million annual global surgeries being performed, 1 80%–86% of patients experience postoperative pain, with <50% receiving sufficient pain relief and ∼10% developing chronic pain. 7
Adequate analgesia poses challenges due to limited effectiveness and potential adverse effects of pharmacologic options. 8 The use of pain prophylaxis significantly predates ERAS, 9 particularly in children and older people, who are more prone to complications such as cognitive impairment. 10 However, pain control effective measures remain limited. 11 –13 Although opioids remain a crucial analgesic in clinical settings, 14 the global opioid crisis and side effects 15,16 require collaborative efforts to optimize perioperative nonopioid analgesic strategies. ERAS now emphasizes opioid-sparing multimodal strategies for adequate pain management. 12 Using different mechanisms, multimodal analgesic strategies optimize perioperative pain control and avoid opioid-related side effects. 17,18 Postoperative multimodal analgesia commonly includes non-narcotic agents, patient-controlled intravenous analgesia (PCIA), periarticular tissue injections of local anesthetics, and epidural and nerve block analgesia. 19 Non-narcotic agents have been considered integral to the success of ERAS. 20
Nonopioid analgesics such as gabapentin, acetaminophen, ketamine, and peripheral nerve blocks are increasingly used alongside opioids in perioperative settings. 21 Nevertheless, specific protocols remain premature considering clinical heterogeneity. Furthermore, treatment durations, ideal dosing, and more favorable outcome measures, including long-term effects, need to be investigated in future trials. 18,22 –24 All local anesthetics act on all types of axons nonselectively. In addition to pain relief, they also cause motor paralysis, hypoesthesia, and autonomic blocks. 25 In summary, ideal analgesic strategies require further discussion despite many published reviews and guidelines. Multimodal, opioid-sparing techniques are crucial to improve pain control using safe and effective analgesic strategies. 1 Considering the global opioid crisis and the ultrarestrictive ERAS nonopioid strategy, 20 nonpharmacologic acupoint stimulation therapy may be a good choice for pain control in ERAS.
Spinal cord stimulation, a validated treatment for pain syndromes, can cause trauma at the stimulation site and lose efficacy over time. 26 The effects of acupoint stimulation on pain have also been widely demonstrated, and its effects can persist over time. 27 Acupoint stimulation is a nonpharmacologic and minimally invasive therapy that has been practiced for thousands of years to treat many disorders in Asia, especially in China. 28 It involves stimulation of sites on the body (acupoints) through the skin and provides multiple benefits with few adverse effects. 29 –35 Acupoint stimulation has gained acceptance worldwide, with increasing evidence for its efficacy in postoperative pain management. 36 Acupoint stimulation in patients undergoing ERAS has proved to be a feasible option for pain control in a variety of surgical settings, and increasing evidence from randomized controlled trials (RCTs) indicates that it can be incorporated into ERAS and should be considered for a standardized ERAS strategy.
Very few RCTs have been summarized for assessing the quality of acupoint stimulation on pain control in ERAS. The main objective of this systematic review and meta-analysis was to summarize relevant evidence and further evaluate the effects of acupoint stimulation for pain control in ERAS.
Materials and Methods
Protocol and registration
This study followed procedures from the Systematic Review Registration (https://www.crd.york.ac.uk/prospero; identifier, CRD42022381235) and was performed with the guidance of Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA). RCTs using visual analog scale (VAS) scores 24 h after surgery reported were included. The details of the inclusion and exclusion criteria are shown in Table 1.
Eligibility Criteria Related to PICO of Inclusion and Exclusion Criteria
P, population; I, intervention; C, comparison; O, outcome; S, study design; L, language.
ERAS, enhanced recovery after surgery; RCTs, randomized controlled trials; TEAS, transcutaneous electrical acupoint stimulation; VAS, visual analog scale.
Search strategy
With the guidance of a librarian, the authors searched China National Knowledge Infrastructure, China Online Journals, Chinese Biological Medical Database (SinoMed), Chongqing VIP Chinese Science databases, PubMed, Web of Science, EMBASE, Cochrane Library, Wiley Online Library, and Scopus for articles published from January 1997 through December 2022 related to acupoint stimulation for pain management. RCTs were included. There were no restrictions on the age or sex of the included patients.
MeSH terms, titles/abstracts, or other comparable terms for databases were used. These included combinations of “Acupuncture,” “electroacupuncture,” “Electro-Acupuncture,” “Acupuncture Point,” “Acupuncture Points,” “Point, Acupuncture,” “Points, Acupuncture,” “Acupoint,” “Acupoints,” and “Enhanced Recovery After Surgery,” “ERAS,” “Enhanced Recovery Protocols,” “ERP,” “Enhanced Postsurgical Recovery,” “Postsurgical Recovery, Enhanced,” “Postsurgical Recoveries, Enhanced,” “Enhanced Recovery Program,” “Recovery, Enhanced Postsurgical,” “Fast-Track Surgery,” “Accelerate Recovery Surgery,” “Accelerated Rehabilitation surgery,” “Fast Track,” and “ERATS.” Bibliographies of relevant websites and articles were screened, and authors were contacted for detailed outcome data when necessary. Searches were limited to published studies in humans.
Data extraction and quality assessment
Data screening was managed using Endnote (X8; Clarivate, London, United Kingdom). Studies with duplicate data were detected and removed via Endnote and manual screening. According to the inclusion and exclusion criteria for initial inclusion, two authors (J.C. and L.L.) independently screened the titles and abstracts. The disagreements were adjudicated by further reviewing the full texts and discussing with a third author (Y.X.). The screening and selection process is shown in a detailed PRISMA flowchart (Fig. 1). Two authors (J.C. and L.L.) extracted data and assessed the quality of each article independently.
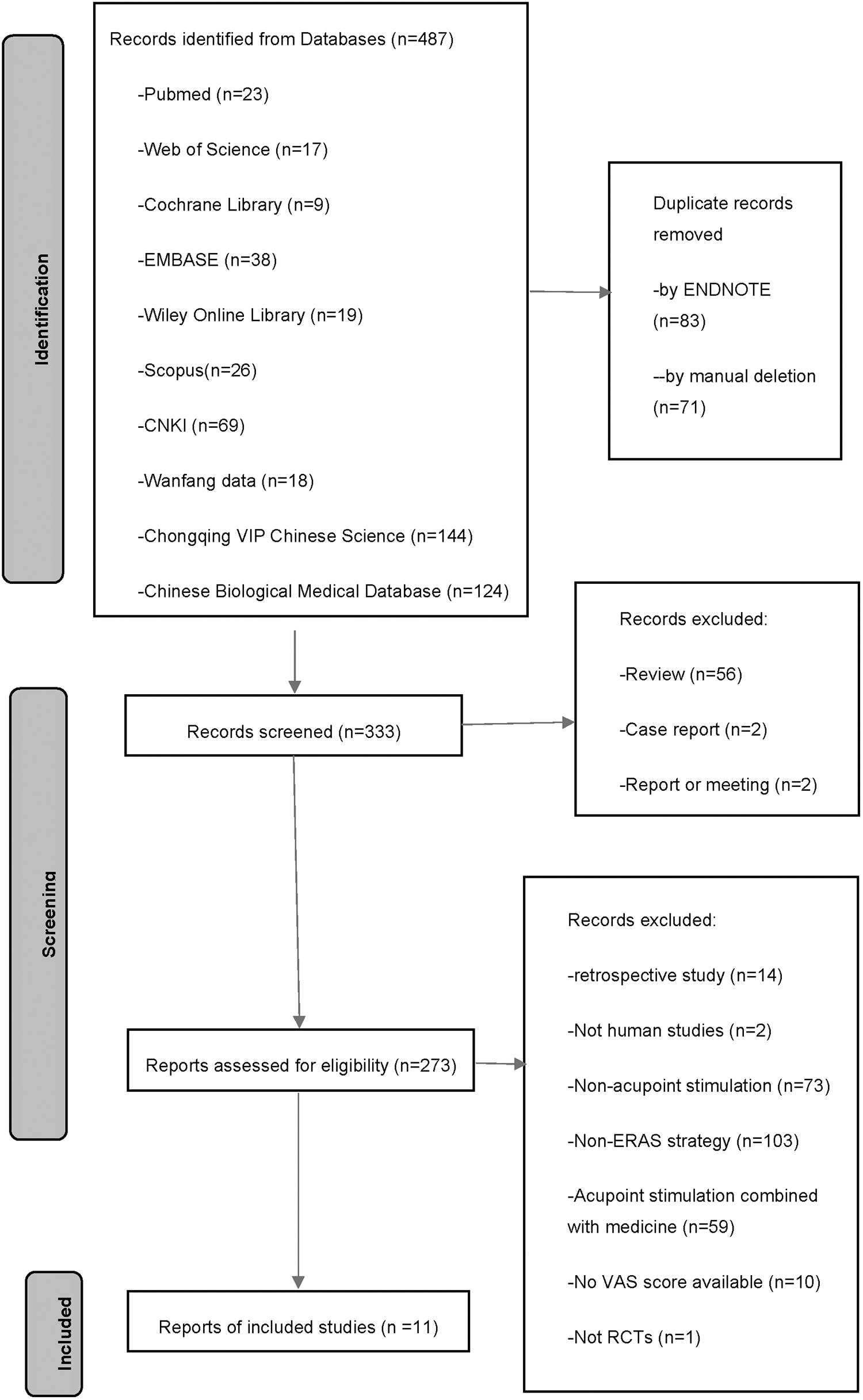
A flow diagram of the systematic review according to PRISMA guidelines. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analysis.
The modified Jadad Scale with the following items was used to evaluate the quality of the included trials 37 : (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel or outcome assessment, and (4) reporting of the number of dropouts and reasons for withdrawal. Each study was scored on a scale of 0–7, with 0–3 indicating low quality, 4–5 moderate quality, and 6–7 high quality. 37 Using the Cochrane Collaboration criteria, the risk of bias (RoB) was rated. The seven domains of the Cochrane tool include the following: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessment, (5) incomplete outcome data, (6) selective reporting, and (7) other bias. 38
Assessment of reporting biases
Publication bias was assessed using funnel plots and Egger tests. Using funnel plots, small-study effects were expressed intuitively for the primary outcome of pain intensity. Symmetry was assessed for potential reporting bias, true heterogeneity of intervention, or poor methodological quality. Egger tests were performed to investigate the degree of asymmetry, and p < 0.05 was set as the level of significance. Any disagreements were resolved by involving a third author (Y.X.) or by discussion.
The authors extracted information about the study country, authors, patient ages, surgical systems or organs, study design, treatment period, sample size, acupoint stimulation protocols, acupoint stimulation styles, adverse effects, and significant differences in outcomes between acupoint stimulation and control groups for each RCT. A summary of the included RCTs is shown in Table 2. Any disagreements noted during study selection or data extraction were discussed by the authors until agreement was reached. VASs are the most frequently used instruments to measure pain intensity in clinical settings. VAS scores ranging from 0 to 10 indicate pain ranging from none to the most severe. Postoperative nausea (PON) was defined as an unpleasant subjective feeling that was always associated with a sense of vomit urgency. Postoperative vomiting (POV) was defined as the forceful expulsion of gastric contents, usually by sustained contraction of abdominal muscles. The combination of PON and POV was defined as PONV.
Summary of Included Studies
1: Pain; 2: analgesic drug consumption; 3: PON; 4: POV; 5: PONV.
PON, postoperative nausea; PONV, postoperative nausea and vomiting; POV, postoperative vomiting.
Meta-analysis and synthesis of evidence
This meta-analysis was conducted using both Rev-Man (v.5.4.1; The Cochrane Collaboration, The Nordic Cochrane Centre, Copenhagen, Denmark) and R 3.4.3. 39 Acupoint stimulation groups were compared with control groups for each item. For the effect sizes of each item, standardized mean differences (SMDs) and the corresponding 95% confidence intervals (CIs) were measured. Using the I 2 statistic, heterogeneity was quantified. Random-effects methods were used in cases of high heterogeneity (I 2 > 50%), and fixed-effects methods were used in cases of low heterogeneity (I 2 ≤ 50%).
Only trials with fully available data were included in the meta-analysis, and the authors of the original RCTs were contacted by mail or phone if data were incomplete. Analysis proceeded if trial authors did not respond within 4 weeks. For trials with overlapping data, the authors selected those with the largest populations. Overall p-values were obtained, and statistical significance was defined as p < 0.05.
Results
Literature search and selection
The search of the initial databases yielded a total of 441 journal articles, of which 274 remained for further screening after omitting 167 duplicates. A total of 11 RCTs studying acupoint stimulation for pain control were included in the final analysis (Fig. 1).
Characteristics of studies and quality of evidence
The 11 studies analyzed included a collective total of 972 patients, and all of the RCTs were conducted in China. One was written in English and the other 10 were written in Chinese. Three types of acupoint stimulation were reported in the studies: acupuncture, electroacupuncture, and transcutaneous electrical acupoint stimulation (TEAS). Study sample sizes ranged from 40 to 184. All acupoint stimulations were conducted during perioperative periods. The type of control group in seven studies was acupoint stimulation compared with usual care, and only four studies compared acupoint stimulation with sham stimulation. 39 –42 Most (7/11) of the studies were focused on surgery involving the digestive system. Although different acupoint stimulations were used in different studies, all studies used standardized acupoint stimulation protocols for certain types of ERAS. Table 3 shows the details of the locations for the used acupoints.
Description of Acupoints
1 cun ≈3.33 cm.
The most widely used acupoints were Zusanli (ST36; n = 8) and Neiguan (PC6; n = 8), followed by Sanyinjiao (SP6; n = 4), Hegu (LI4; n = 4), Shangjuxu (ST37; n = 3), Gongsun (SP4; n = 2), Xiajuxu (ST39; n = 2), and Quchi (LI11; n = 1).
Modified Jadad scores were determined for each of the 11 RCTs included, in the following 5 domains: random sequence generation, allocation concealment, blinding of participants and personnel or outcome assessment, and reporting of the number of dropouts and reasons for withdrawal. Two of the studies had modified Jadad scores ≥6, indicating high quality, and the other nine had Jadad scores of 4 or 5, indicating moderate quality. RoB was determined with the following seven domains included in the Cochrane Collaboration criteria; random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and “other bias.” The acupuncturists were blinded in 4/11 studies. 39 –42 One study evoked some RoB concerns due to incomplete cases, 43 but the remaining 10 RCTs were all evaluated and showed low RoB. Figure 2 provides a summary of the included trials and details of domain-specific RoB assessments.

Risk-of-bias summary in included studies.
Funnel plots and statistical analyses yielded no evidence of publication bias with respect to pain intensity, with an Egger's test t-value of 0.12 (degrees of freedom [df] 9, p = 0.908). Additional Egger's test results were t = −3.29 (df 3, p = 0.046) for analgesic drug consumption, t = 4.40 (df 1, p = 0.142) for PON, t = −0.02 (df 1, p = 0.989) for POV, and t = 1.23 (df 2, p = 0.342) for PONV (Fig. 3).
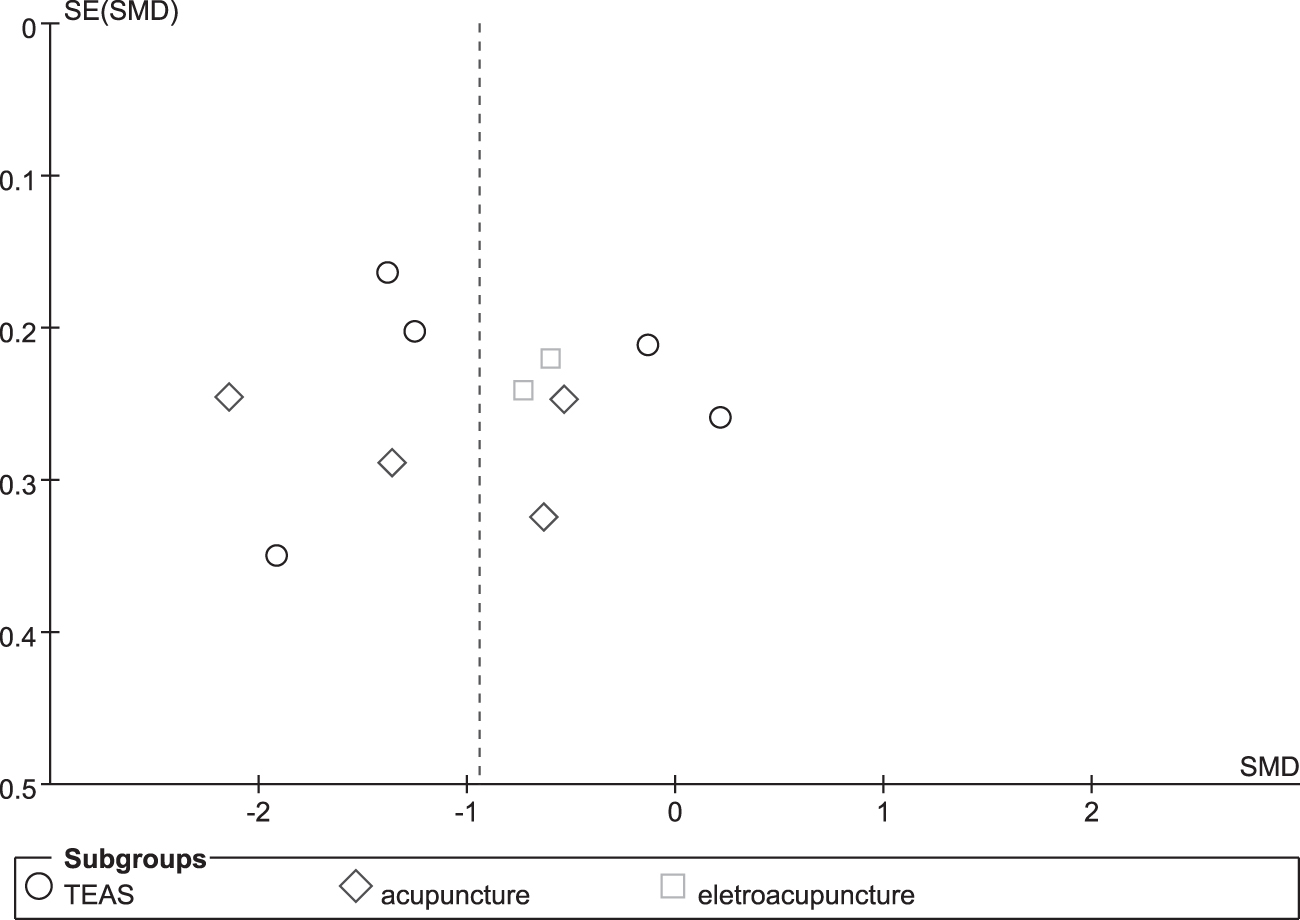
The funnel plots of publication bias of pain intensity.
Adverse events
Only one case of bruising was reported as an adverse event related to acupoint stimulation. 43 Other trials did not mention or report any adverse events. Based on available data and the long history of acupoint stimulation, it can be seen as a generally safe treatment with negligible adverse events. 44
Meta-analysis results of acupoint stimulation
For each item, forest plots for acupoint stimulation compared with control groups are shown in Figures 4–8. There were statistically significant effects of acupoint stimulation on pain intensity (SMD −0.94; 95% CI −1.35 to −0.53), analgesic drug consumption (SMD −1.87; 95% CI −2.98 to −0.75), PON (SMD 0.31; 95% CI 0.13 to 0.73), POV (SMD 0.57; 95% CI 0.11 to 2.92), and PONV (SMD 0.29; 95% CI 0.16 to 0.53), with respective statistical heterogeneity of 88%, 94%, 0%, 71%, and 0%. In subgroup analyses of transcutaneous electrical nerve stimulation (TENS), acupuncture, and electroacupuncture for pain control, compared with TENS and electroacupuncture, acupuncture was more effective with an SMD of −1.17 (95% CI −1.95 to −0.39; TENS: SMD −0.88; 95% CI −1.58 to −0.17; and electroacupuncture: SMD −0.66; 95% CI −0.98 to −0.34).
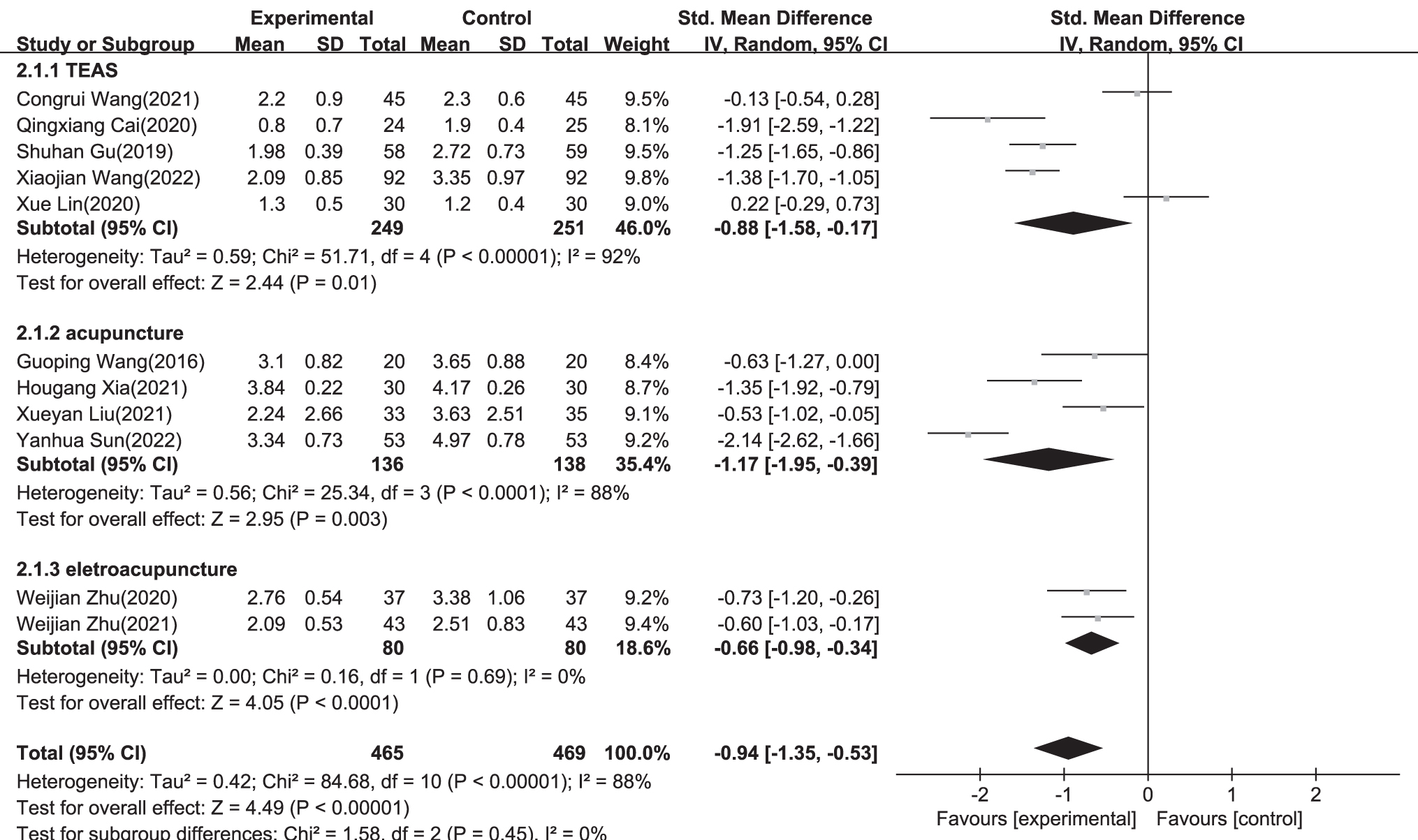
A forest plot of subgroup analysis showing the effect estimates and confidence intervals of acupoint stimulation versus control treatments on pain control in included studies.
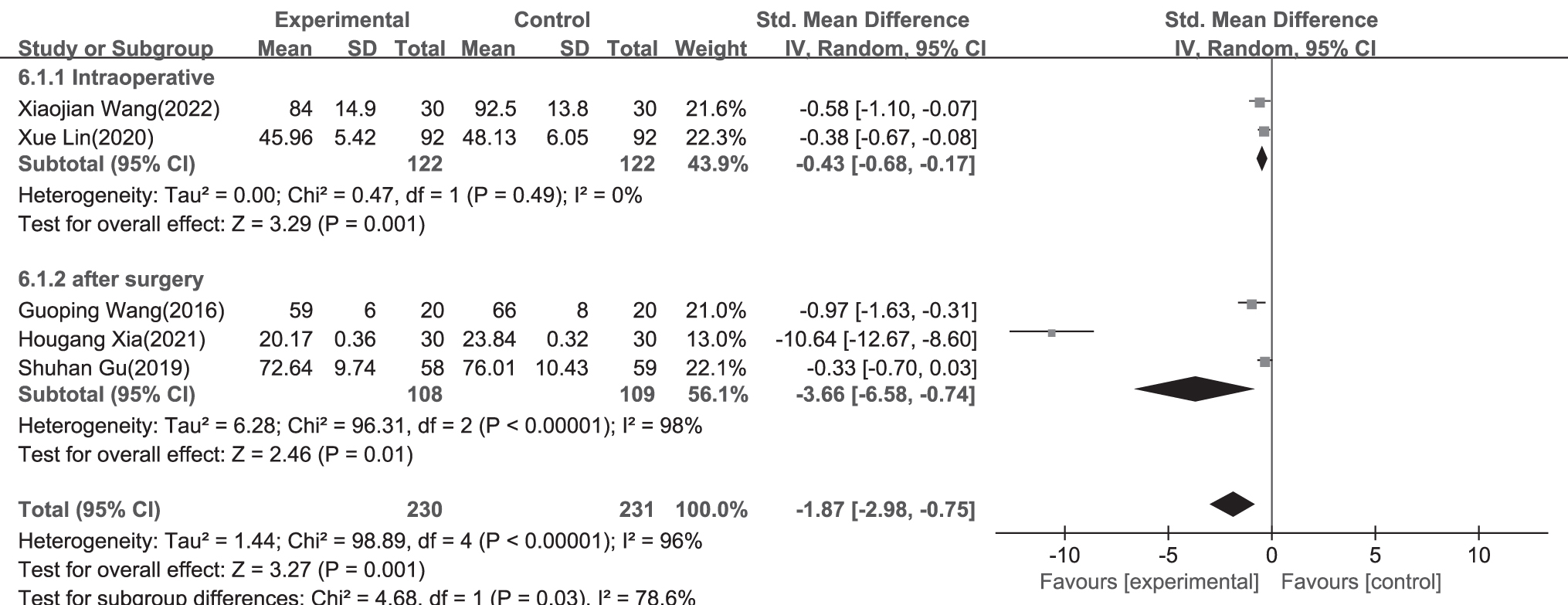
A forest plot of subgroup analysis showing the effect estimates and confidence intervals of acupoint stimulation versus control treatments on analgesic drug consumption in included studies.
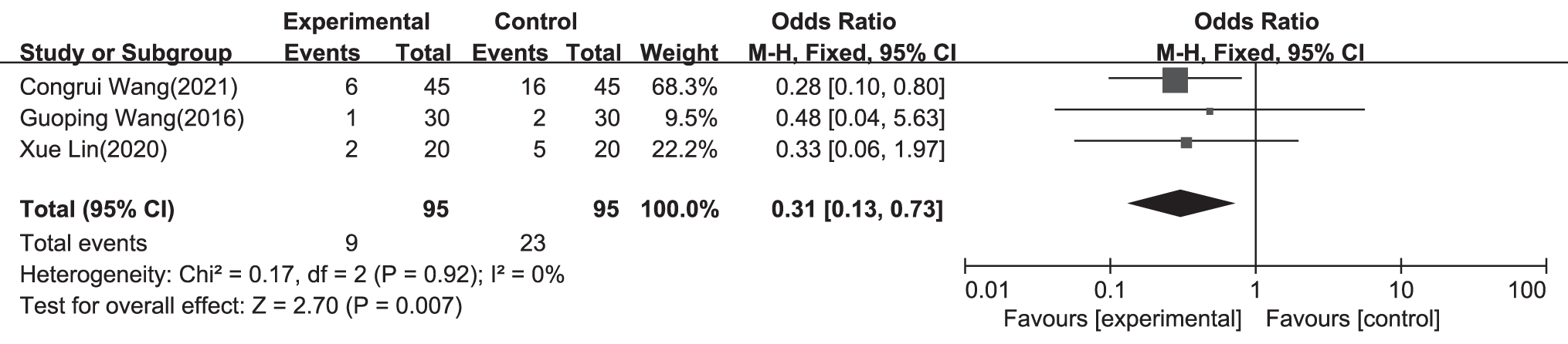
A forest plot of subgroup analysis showing the effect estimates and confidence intervals of acupoint stimulation versus control treatments on PON in included studies. PON, postoperative nausea.
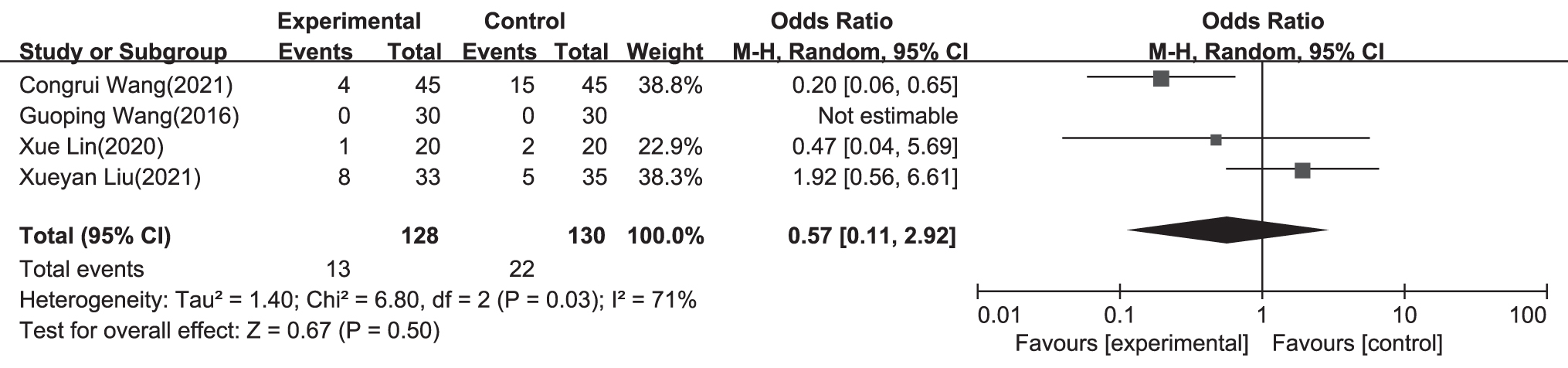
A forest plot of subgroup analysis showing the effect estimates and confidence intervals of acupoint stimulation versus control treatments on POV in included studies. POV, postoperative vomiting.
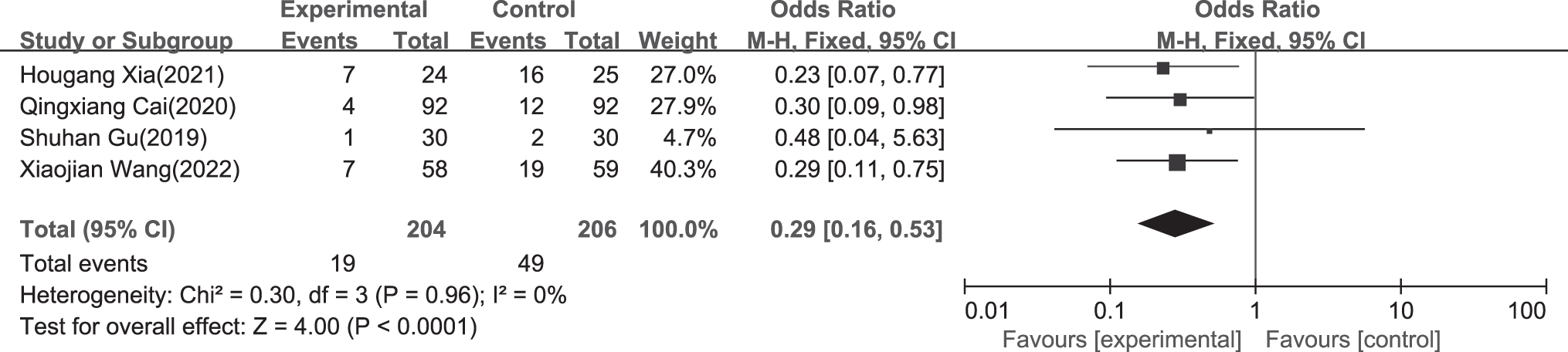
A forest plot of subgroup analysis showing the effect estimates and confidence intervals of acupoint stimulation versus control treatments on PONV in included studies. PONV, postoperative nausea and vomiting.
Limitations
The main strength of this systematic review and meta-analysis was its exhaustive search of Chinese databases, as acupoint stimulation originated and developed in China. However, this study was also limited by the inaccessibility of databases in other languages. The authors only included RCTs published in Mandarin Chinese or English, and all included RCTs were performed in China with Chinese patients. This limited the generalizability of the analysis, as other RCTs on acupoint stimulation may be published in other languages. More studies in other languages should be included in future systematic reviews and meta-analyses. In addition, this analysis assessed pain control, analgesic drug consumption, PON, POV, and PONV, while other symptoms such as abdominal distension and those associated with first postoperative exhaust and defecation were not assessed. Multiple symptoms, using comprehensive scales, should be compared in future studies.
More efficient and valid methods will be available for meta-analysis when more published studies are available. Finally, only symptoms before discharge were included in this systematic review and meta-analysis, and no RCTs reported long-term or follow-up items. These items related to postdischarge characteristics should be included in future studies.
Discussion
A total of 11 RCTs from 441 primary studies were included in the current system review and meta-analysis. Heterogeneity was evident with regard to pain intensity, analgesic drug consumption, and POV, with an I 2 > 50%. No publication bias was detected. Compared with control treatments, acupoint stimulation was more effective for reducing pain intensity, with an SMD of −0.94, and there was only one reported case of bruising. Acupoint stimulation was effective and safe for reducing pain intensity. Considering the limitations of current analgesic methods, acupoint stimulation—a technique developed in Traditional Chinese Medicine—has been used in ERAS strategies, and there is strong evidence for its use as an effective and safe means of analgesia. This should be considered for incorporation into guidelines on ERAS strategies.
The mechanisms behind postoperative pain control via acupoint stimulation are complex and involve multiple systems. In 2020, pain was defined as “an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage.” 45 The experience of pain is affected by personality, emotional state, sex, culture, and pain behavior. As a type of peripherally driven pain, 46 postoperative pain is usually unavoidable because of actual mechanical damage to tissue caused by surgery. The damage to tissue results in the release of bioactive substances such as bradykinin, prostaglandins, and substance P. These bioactive substances then bind to nociceptive receptors, which are present in terminal nerve fibers.
Signals are transmitted via terminal nerve fibers, afferent neurons, the dorsal horn of the spinal cord, ascending spinothalamic pathways, and thalamocortical projections and finally reach neural centers in the cerebral cortex. The thalamus and cortical modulation of neural activity are at the core of pain experiences. The medulla, descending pathways, and dorsal horn of the spinal cord play important roles in the further modulation of pain. 36,47 A previous study showed that acupoint stimulation could relieve many kinds of postoperative pain, such as abdominal, spinal/neurologic, and gynecologic/pelvic pain. 35 As with spinal cord stimulation, the pain relief associated with acupoint stimulation likely involves the activation of inhibitory interneurons in the dorsal horn (i.e., gate control theory), 26,48 but it is less invasive than spinal cord stimulation.
The exact mechanisms of the analgesic effects of acupoint stimulation are still being investigated, with the endorphin theory already being widely accepted. Stimulation of the relevant acupoints can reportedly cause the nerves next to those points to release endorphins that can ameliorate pain. 49 Transient receptor potential vanilloid (TRPV) channels and adenosine triphosphate are involved in stimulation at acupoints. The analgesic effects of neurotransmitters, including opiates, serotonin, norepinephrine, orexin, and endocannabinoids, can be modulated by acupoint stimulation in the central nervous system. Moreover, by acting on the hypothalamic–pituitary–adrenal axis, mediating peripheral opioid release, acupoint stimulation reduces cyclooxygenase-2 and prostaglandin E2 levels in the periphery. By modulating dopamine release in the nucleus accumbens, acupoint stimulation can also reduce the effects of positive and negative reinforcements. 36
Subcutaneous mast cells are sensitive to mechanical stimulation and play an important role in the analgesia induced by acupoint stimulation because multiple types of mechanosensitive channels are expressed by mast cells, such as chloride channels, TRPV1, TRPV2, and TRPV4.
Via the collagen network, the torque and force generated by acupoint stimulation indirectly activate mast cells. Various biological active substances such as adenosine triphosphate, histamines, serotonin, and adenosine are subsequently released from activated mast cells into the interstitial space. The corresponding receptors situated at local nerve terminals of sensory neurons in peripheral ganglia can be activated by these biological active substances and their downstream products. These neurons project to pain-relevant regions in the spinal cord and/or brain. 50 The analgesic effects are thought to be generated by reducing the electrical activity of primary sensory neurons. It has long been thought that the somatic afferents in the acupoints convey sensory signals successively to the spinal cord, brain stem, and hypothalamus once the acupoints are stimulated.
Multiple additional signal pathways are integrated in the brain and then stimulate multiple pathways, including neuroendocrine and neuroimmune pathways, ultimately affecting the functional activity of the nervous, digestive, and endocrine systems. 29,31,51 –54 In summary, the mechanisms of acupoint stimulation may involve the regulation of several systems. Acupoint stimulation-induced changes in bioactive substances and signal transduction in the nervous system could be the basis of postoperative pain control. Effective analgesia facilitates patient mobility and oral intake of food and water, 6 and reduces host stress responses, the risk of morbidity, the length of hospital stays, health care costs, 3 the risk of persistent pain, 55 and postsurgical complications. 56
Acupoints are specific or nonspecific sites located throughout the body. There are ∼361 specific acupoints located along 12 bilateral meridians and 2 midline channels (Du and Ren Meridians) and another 34 specific acupoints elsewhere in the body. In addition to specific sites, there are nonspecific sites such as Ashi acupoints. Ashi acupoints refer to those that can produce a painful or comfortable sensation when pressed appropriately. The ERAS strategy has been used in most surgical subspecialties. However, the RCTs included in this systematic review and meta-analysis mainly involved surgery of the digestive system (n = 7) and standardized acupoint stimulation protocols. ST36 and PC6 were the most widely used acupoints.
In Traditional Chinese Medicine, ST36 belongs to the Foot–Yangming Meridian, and PC6 belongs to the pericardium channel of the Hand–Jueyin Meridian. The Foot–Yangming Meridian is responsible for the stomach, and the Hand–Jueyin Meridian is responsible for the pericardium. The pericardium is partly responsible for the functional activity of the nervous system. ST36 has usually been used to treat stomach pain, vomiting, hiccups, bloating, abdominal pain, indigestion, diarrhea, constipation, and dysentery. PC6 has usually been used to treat heart pain, palpitations, chest tightness, chest pain, stomach pain, vomiting, and hiccups. Both ST36 and PC6 have been used in clinical settings for pain control and improving gastrointestinal function in modern studies. 57 –60 In addition to routine therapeutic effects, ST36 has been shown to cause consistent signal attenuation in the secondary somatosensory cortex, including the marginal and surrounding structures of the telencephalon, interbrain, and brain stem. 61,62
There are many types of acupoint stimulation used in clinical settings, such as acupuncture, moxibustion, massage, electroacupuncture, TEAS, acupressure, acupoint thermal therapy, acupoint injection, auricular acupoint sticking, auricular acupressure, acupoint pressing needle therapy, and acupoint sticking therapy. 63 Acupuncture, electroacupuncture, and TEAS are the most commonly used methods in clinical studies, and these three methods were included in this systematic review and meta-analysis. Acupuncture is the traditional method for acupoint stimulation and involves stimulation of acupoints via insertion of fine metal needles into the skin to stimulate muscles, nerves, and connective tissue throughout the body. This is followed by manipulations involving lifting and thrusting or rotating until a sensation of de qi (sense of soreness, numbness, fullness, or heaviness) is achieved. 36
Through the needles penetrating points on the wrist or the ankle, wrist–ankle acupuncture is considered a type of superficial acupuncture. The body is divided into six longitudinal zones and needle points on the left and right wrists and ankles. The wrist is the upper zone (1–6) with the right and left hands symmetrical. The ankle is the inferior zone (1–6) with the right and left ankles symmetrical. 19 As a special type of acupuncture, the acupoints in wrist–ankle acupuncture do not have specific names and are usually named zones 1–6. Wrist–ankle acupuncture has been used in orthopedic surgery and has exhibited advantages with respect to pain relief, the occurrence of postoperative adverse events compared with control treatments, and the use of PCIA. 19
Electroacupuncture, which was developed in the 1950s, is a modified type of acupuncture where needles with attached electrodes are inserted to deliver either low-frequency or high-frequency electrical impulses, or a combination of both. By using electrode tabs on the target acupoints, the stimulation is provided from an electrical stimulator. Instead of needles, TEAS uses electrode tabs placed on the acupoint surface, which release electrical pulses targeting acupoints, and it has a combination of the effects of TEAS and acupoint therapy. 38
Compared with acupuncture and electroacupuncture, without penetrating the skin, TEAS can be more easily accepted by children and people with puncture contraindications. Acupoint stimulation is generally characterized by simplicity, convenience, and low clinical cost; however, the most effective methods remain controversial. Previous studies have shown positive short-term effects of acupuncture, but not electroacupuncture, for related pain, disability, and strength in lateral epicondylalgia. 64 In the present meta-analysis, compared with electroacupuncture and TEAS, acupuncture was the most effective method for pain control. Acupuncture is the conventional method for acupoint stimulation and involves penetrating the skin and “liuzhen,” the state of subcutaneous quiescence. Both TEAS and electroacupuncture use continuous stimulation via electric currents. Compared with electroacupuncture, TEAS was safer and more effective for pain control in the current meta-analysis.
One possible explanation for this is that patients may feel less pain because the skin is not punctured. Pain is affected by pain behavior, emotional state, sex, personality, and even culture. 47 Although TEAS was moderately effective in comparisons of the three above-described methods, considering its convenience and that is it not influenced by platelet counts or coagulation function, it is a preferred method for acupoint stimulation. However, the use of electrical currents limits its applicability for certain patients, such as those with cardiac pacemakers. 65
Blinding is relatively simple in studies using TENS, as the current can be easily controlled, but there are difficulties surrounding adequate blinding and control group designs for acupuncture and electroacupuncture. Because it is difficult to blind participants and acupuncturists in studies investigating acupuncture and electroacupuncture, it can be difficult to adequately control those studies. Two types of sham acupuncture or electroacupuncture have been widely used in investigative studies. The first type of sham acupuncture is a kind of device with nonpenetrating placebo needles such as Park, Streitberger, or Takakura needles, but the current systematic review and meta-analysis used none of these nonpenetrating placebo needles. Compared with Park or Streitberger needles, Takakura needles consist of a set of one placebo skin touch needle and one penetrating needle and are considered to blind both acupuncturists and participants, as nobody can determine the type of needles being used by touch or sight.
Notwithstanding monetary costs and accessibility limitations, the effectiveness of Takakura needles requires further investigation because of its difficulty in blinding especially experienced acupuncturists. On the whole, however, compared with the other two sham acupuncture methods, it is the best option for blinding and should be considered for use in the future clinical studies. Another type of sham acupuncture uses needles that penetrate the skin at nonacupuncture points that are close to acupuncture points. However, this type of sham acupuncture has been criticized because it may have therapeutic effects due to the accidental stimulation of nearby acupoints.
POV occurs in ∼30% of patients, and the incidence of PON is ∼50%. 66 PON, POV, and PONV are distressing complications, and the use of opioid analgesics for pain control is considered a main contributing factor to these symptoms. 67 As well as pain control, another key aspect of ERAS is the control of gastrointestinal adverse reactions such as PON, POV, and PONV, especially in patients who have undergone surgery involving the digestive system. Acupoint stimulation is a U.S. Food and Drug Administration-approved therapy for nausea. A systematic review concluded that acupoint stimulation reduced postoperative pain and opioid consumption along with PON and other complications compared with sham controls. 36
The present meta-analysis evaluated perioperative analgesic drug consumption (during surgery and after surgery) and gastrointestinal reactions (PON, POV, and PONV) in acupoint stimulation and control groups. Opioid analgesics remain the most commonly used pain relievers. Opioid analgesics are a major contributing factor to PON, POV, and PONV. Once PON, POV, and PONV have been reduced, pain is also reduced, providing relief to patients.
Acupoint stimulation has not been incorporated into global ERAS guidelines. Previous clinical trials have tested acupoint stimulation, and reported its effectiveness for pain control in surgical pastients receiving ERAS. 39 –42,65,68 –73 In the present systematic review and meta-analysis, the effects of acupoint stimulation for pain control were assessed in patients receiving ERAS, providing objective evidence for its usefulness as part of ERAS strategies. The present study included 11 RCTs (972 patients), and acupoint stimulation was more effective than control treatments with respect to pain intensity. Furthermore, the present study analyzed postoperative analgesic drug consumption, PON, POV, and PONV, and acupoint stimulation was more effective than control treatments for all of these. Drug consumption, both during and after surgery, was compared in a subgroup analysis. PCIA was the most widely used form of drug consumption after surgery, and acupoint stimulation reduced total PCIA dosages compared with control treatments.
By reducing pain and facilitating the early recovery of gastrointestinal function, acupoint stimulation treatment can improve clinical outcomes and patient experiences associated with surgical treatment and recovery.
Conclusions
Acupoint stimulation promoted pain control in surgical patients receiving ERAS, and ST36 and PC6 were the most widely used acupoints. It also reduced analgesic drug consumption, PON, POV, and PONV. The safety of acupoint stimulation was not completely described in the studies analyzed. However, acupoint stimulation should be recommended to patients receiving ERAS who seek nonpharmacologic analgesic methods for pain control. More rigorous RCTs are needed in the future to confirm the efficacy and safety of acupoint stimulation for promoting pain control due to blinding issues in some of the included RCTs. Additional large-scale, high-quality RCTs are required to facilitate the development of a standardized ERAS strategy incorporating acupoint stimulation. The results of the present analysis may help to improve surgery patient recovery worldwide.
Footnotes
Acknowledgment
The authors thank Charlesworth Author Services for providing English language editing of the article.
Authors' Contributions
J.C. and Y.X. were responsible for acquisition, interpretation, and drafting the article. L.L. substantially contributed to the data analysis. G.Y. and X.Z. critically revised the work for important intellectual content. G.Y. was included in the article drafting and also critically revised the work. All authors provided final approval of the version to be published and agree to be accountable for all aspects of the work.
Disclaimer
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Disclosure Statement
The authors have no competing financial interests to report.
Funding Information
This study was supported by the Zhejiang Provincial Natural Science Foundation of China (award No. LBY22H270001); Clinical Research Fund of Zhejiang Medical College (award No. 2017ZYC-A13), and Seventh Batch of Nationwide Famous TCM Doctors Succession Program.
Supplementary Material
Supplementary Data
Supplementary Figure S1
PRISMA_2020_checklist
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
