Abstract
Introduction:
Reporting of aromatherapy-focused research often lacks sufficient quality and detail for replication and subsequent application of results. To our knowledge currently, no quality appraisal tool exists for aromatherapy research reporting. To address this gap, the Aromatic Research Quality Appraisal Taskforce (ARQAT) composed of aromatherapy professionals with varied expert backgrounds came together. Presented here is the Transparent Reporting for Essential oil and Aroma Therapeutic Studies (TREATS) checklist, which is a result of this collaborative effort.
Methods:
Creation of TREATS followed a three-stage process, including determination of interest/need, development, and dissemination. The shortcomings of existing aromatherapy research reporting quality were evaluated and responses to address these shortcomings were used to create checklist items that were then grouped into sections. Items for each section were brain-stormed with reference to the aromatherapy literature and ARQAT's expert knowledge, and the development of each section followed an iterative process until agreement was reached. An explanatory document was also created to assist more accurate use of the tool; it and the checklist were reviewed by a group of aromatherapy experts.
Results:
The TREATS checklist with 38 items in four sections was developed along with the explanatory document. The ARQAT and a global group of aromatherapy experts reviewed the TREATS. Their results and comments assisted development of the current version. The TREATS identifies key components of research involving essential oils, their application, and olfactory considerations that ARQAT considers the minimum necessary for high-quality aromatherapy research.
Conclusion:
The TREATS, explanatory document, and associated website (www.arqat.org) contribute to thorough aromatherapy research critique. The TREATS checklist aids appraisal of quality and can be used with any study design. It lays the foundation for the future development of aromatic research reporting guidelines.
Introduction
The term “aromatherapy” is used to describe many different application methods and practices involving the use of concentrated fragrant volatile compounds, often referred to as essential oils, for therapeutic purposes. Aromatherapy is currently used for mood management, perioperative nausea, pain relief, coping in childbirth, topical inflammation, and emotional support during palliation. 1,2
In recent years, use of essential oils and aromatherapy has grown in popularity. 3 –5 Likewise, the amount of research and subsequent publication of aromatherapy studies has increased 6 with promising evidence for the benefits for psychological and physiological health and well-being. However, many published studies lack appropriate methodological details related to the aromatherapy and essential oil elements of the studies. This lack of detail inhibits replication and limits incorporation of the study results into evidence-based practice. 7
Much health care research is conducted by individuals for whom research is not their first occupation and this often affects the quality of their research. 7 The need for a checklist specific to aromatherapy research emerged from observing that most aromatherapy research is conducted either by aromatherapy practitioners with little research training, or by clinical researchers with little aromatherapy understanding or training. Many peer-reviewers and editors of journals publishing aromatherapy research may also need support of an aromatherapy-specific checklist as they appraise the quality of reports before publication.
During review of the literature, it was discovered that the essential oil and aromatherapy-focused aspects of many of the studies were sub-optimally reported. Reviewed articles lacked adequate descriptive elements such that the results from the studies could not be used to generalize or replicate the findings 8 (Supplementary Material S1).
Similar concerns over poor reporting quality in published research in other therapeutic modalities have prompted the creation of multiple quality appraisal and reporting guidelines. 7 The purpose of this article is to report the development of the Transparent Reporting for Essential oil and Aroma Therapeutic Studies (TREATS) checklist containing elements considered vital to the complete and consistent critique of aromatherapy research studies that are conducted in humans.
Methods
Creation of the TREATS followed a similar three-stage process of a peer-reviewed framework for creation of quality assessment checklists as outlined by Whiting et al. The three stages are (1) assessment of need/interest, (2) development, and (3) dissemination 9 (Fig. 1).
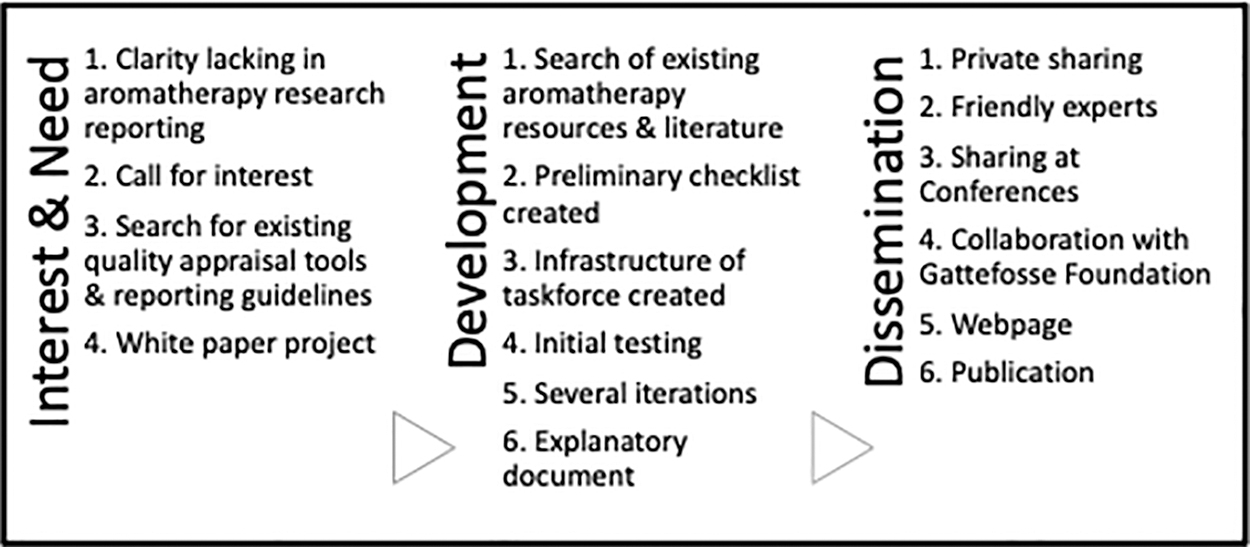
TREATS creation using framework. 14 TREATS, Transparent Reporting for Essential oil and Aroma Therapeutic Studies.
Ethics approval
Exempt status was granted by West Virginia University Institutional Review Board (IRB) for this study (Protocol 2205571104). Design and analysis involved members of the task force and others within the aromatic community who voluntarily agreed to provide input.
Assessment of need and interest in aromatherapy quality appraisal
Development started with assessment of need and determination of interest. Author M.E.R. invited a diverse cross-section of colleagues to meet. Inclusion criteria were knowledge of aromatherapy and the research supporting it.
A broad spectrum of experience was found in members of leading aromatherapy organizations such as the Alliance of International Aromatherapists (AIA) https://www.alliance-aromatherapists.org/, the National Association for Holistic Aromatherapy (NAHA) https://naha.org/, and the International Clinical Aromatherapy Network (ICAN) https://clinicalaromatherapynetwork.com/. Participants included medical and integrative practitioners, nurses of undergraduate and advanced practice, chemists and scientists, and educators of both conventional topics and aromatic practice.
The first meeting was held in January 2021, and 20 attended. From those 20, a core group of 11, who are the authors on this article, agreed to work on an aromatherapy research white paper 8 to address common concerns, and subsequently formed the Aromatic Research Quality Appraisal Taskforce (ARQAT). As research for the white paper continued in monthly meetings, it became apparent that a checklist of critique items would be beneficial to clarify what was needed in quality appraisal of aromatherapy research in humans.
To determine whether a new checklist was needed, the Enhancing the QUAlity and Transparency Of Health Research (EQUATOR) website (www.equator-network.org) and aromatherapy-focused publications, books, and websites were searched. No checklists or guidelines specific to critiquing aromatherapy research were found. Consequently, ARQAT proceeded to create a novel checklist.
While searching for aromatherapy research support materials, several well-known quality appraisal tools and reporting guidelines were reviewed, such as the Consolidated Standards of Reporting Trials (CONSORT) 10 and the Consolidated Standards of Reporting Trials for Nonpharmacologic Treatments. 11 In addition, the Template for Intervention Description and Replication checklist providing guidance specific for intervention descriptions was reviewed. 12 Guidelines for other integrative health modalities were also found, such as: The CheckList stAndardizing the Reporting of Interventions For Yoga 13,14 and extensions for acupuncture, 15 nutrition, 16 and herbal medicine. 17 The TREATS checklist incorporates ideas and insights from all of these (Appendix A1).
Items for each section were brain-stormed with reference to contemporary and extant aromatherapy literature, including but not limited to foundational works from Gattefossé and Valnet that have been translated into English and others. 8,18,19 The taskforce found that most aromatherapy-focused texts included at least some reference to what to include in the critique of aromatherapy-focused research; however, no one resource had compiled a checklist. Over 9 months, the checklist evolved into its current form, with 38 items in four sections. To improve clarity and promote understanding of each item, an explanatory document was also created (Supplementary Material S2).
Defining the scope
In addition to determining interest and need, Whiting et al. recommend that authors of quality assessment tools define the tool's scope and type of assessment the tool is intended for, such as risk of bias (internal validity); applicability (external validity); and reporting quality. 9 ARQAT determined the scope of TREATS to encompass research involving essential oils and aromatic interventions using topical and inhalation application methods in human subjects, and that TREATS should be used to critique quality, but not risk of bias or applicability.
ARQAT also determined that the TREATS should be applicable to all types of study designs such as randomized controlled trials, observational studies, and case reports, evaluating the quality relating to the aromatic components of these studies.
Refinement of TREATS
Taskforce members used the resultant TREATS checklist to review several studies, evaluating responses for consistency in the way the checklist was used. Reflection on the results allowed ARQAT to make changes to the checklist to improve consistency of results, and this was repeated several times. Global experts in aromatics, research, education, and publication known to members of the ARQAT were consulted to help further refine and improve the TREATS (Supplementary Material S2).
As the checklist evolved, a scoring system was adopted to provide more objective feedback. Each item is evaluated as “met,” “partially met,” or “not met,” receiving numerical scores of 1, 0.5, and 0 respectively. Points are tallied within each section to determine a sub-score and then sections are totaled to give the study an overall score.
Some sections may receive high scores whereas others may not. The overall score provides objective feedback and helps the reader determine how well the aromatic and essential oil aspects of the study were reported. The explanatory document helped strengthen the consistency of interpretation of checklist items by people using the TREATS.
Interrater reliability testing
Once the taskforce agreed that to the best of their knowledge all necessary items found in the literature were included in the checklist, the critique using TREATS was completed by taskforce members for four articles examining aromatherapy interventions in humans for stress, premenstrual syndrome, anxiety, and mood states 20 –23 (Table 1). In addition, a wider audience was recruited to critique two of the four articles.
Interrater Reliability
Interrater reliability as the ICC and CI 95% based on a mean-rating (Group 1, k = 12; Group 2, k = 17), absolute-agreement, two-way mixed-effects model, using item analyses.
CI, confidence interval; ICC, intraclass correlation coefficient.
This group of 17 represented a convenience sample from various backgrounds and countries, providing diversity, including aromatic researchers (7), educators (3), and a pharmacist (1), and were located in the United States, Canada, Cook Islands, Ireland, Madrid, Poland, Singapore, and the United Kingdom. Data from both groups were manually entered into an Excel (Microsoft 365) spreadsheet by two task force members (M.E.R. and B.K.L.).
Interrater reliability estimates and their 95% confidence intervals (CIs) were calculated using IBM SPSS Statistics (Version 28) predictive analytic software. The IRR estimates were based on a mean-rating (Group 1, k = 12; Group 2, k = 17), absolute-agreement, and 2-way mixed-effects model, using item analyses. The intraclass correlation coefficient & CI 95% were calculated. This model was used because it is designed to examine a specific set of raters and is concerned with consistency 24,25 (Table 1).
Dissemination of the TREATS
Dissemination of this work has taken place through presentations at scholarly conferences, seminars, and symposia. A website has been created to house information about ARQAT, the TREATS, and efforts to support high-quality aromatherapy research critique using essential oils in human health-focused studies.
Results
The TREATS checklist Explanatory and Example document is found in the Supplementary Materials S2. The four sections pertain to essential oil identification; application methods; aromatherapy intervention; and olfactory function considerations, and there are a total of 38 items. Each item receives a score of 1 for fully met; 0.5 for partially met; and 0 for not met. The overall score summed from the totals of each section is then ranked poor, 0–10; fair, 11–20; or good, 21–38, with space to allow for comments that can further qualify what the score indicates and where improvements may be made.
Section one aims at addressing clear identification of which essential oil(s)* were used with eight items that prompt complete identification. This section was considered of greatest importance in this work. Without complete and clear identification of the essential oil, the study is incomplete and replication made difficult if not impossible. 26 –30
*NOTE: The scope of the TREATS project is to address clear identification of which essential oil(s) were used and is not intended to redefine or replace formally recognized industry standards and Pharmacopoeias that offer clarity and historical grounding regarding essential oils. Please refer to the ISO (International Organization for Standardization) https://www.iso.org/about-us.html and AFNOR (Association Française de Normalisation [AFNOR, English: French Standardization Association]) https://www.afnor.org/en/and the European Pharmacopoeia regarding essential oil https://pheur.edqm.eu/home definitions and to Heinrich et al., for more about herbal extractions and methods.
An example of the importance of knowing as much as possible about the essential oils used relates to variations in constituents and chemotypes. “Chemotype indicates visually identical plants but having different chemical components, sometimes significantly so, resulting in different therapeutic properties.” 31 Geographic location, time of harvest, rainfall, and a myriad of other environmental conditions impact the chemical variations in plants, thus the distilled or mechanically expressed essential oils. 32 According to Benomari et al., chemical variability in essential oils impacts their therapeutic properties and thus their use. It is imperative to have an analysis of the chemical composition of all essential oils used in studies. 33
Variations of constituents lead to differences in therapeutic properties illustrated by the calming effect of linalyl acetate and linalool commonly found in Lavandula angustifolia (Lavender) though not present in Lavandula stoechas (Spanish Lavender, also called French Lavender) with a main chemical constituent of camphor that is not known for calming but instead for clearing sinuses and congestion. 34 Researchers performing their own analysis on materials used in their study should be aware that if at all possible obtaining a third-party analysis could decrease the appearance and risk of bias.
Sections 2A and 2B of TREATS cover description of application methods. The focus of this work is on topical and inhalant methods though TREATS is flexible enough, with room for comments, to lend itself to essential oil studies in humans where ingestion is the delivery method. All methods of application require varying levels of training and those completing research using any delivery method should identify their credentials and qualifications to practice at that level. Qualifications vary throughout the world. More is discussed in section three related to “Qualified Aromatherapist.”
If application methods are not adequately described, replication of the study and inclusion of it in meta-analyses is not possible. Items included are the dose of aromatic delivered (including details of any dilution in excipient or carriers), frequency of treatment, duration of exposure to the essential oil, and delivery systems such as a diffuser or vaporizer. If diluents or carriers are used, full characterization of them is expected as is for the essential oils. 26,28 –31,34 –38
Section three concerns the description of the aromatherapy intervention, which must be clear and detailed enough to allow for replication by other researchers. Section three of TREATS includes the rationale for use of the chosen essential oils and application methods, the choice of a theoretical and conceptual framework, consultation with or qualifications of one who has training and expertise to work with essential oils in aromatherapy-type applications, reports of any allergic or adverse reactions, and consideration of safe storage and use of essential oils during the study. 11,12,26,28,34,39,40
The past two decades have seen an increase in understanding and appreciation for olfaction and the sense of smell. 41 Section four of TREATS includes olfactory considerations such as pre-trial evaluation of olfactory ability and identification of anosmia, previous exposure to essential oils, evaluation of odor recognition, expectancies about the odor's therapeutic qualities, the perceived intensity of the odor, and any adverse effects from olfactory testing. These aspects of aromatic research are potential sources of bias and information related to causality is enhanced by inclusion of olfaction. 42 –44
Discussion
Methodology varies widely for creation of quality appraisal tools. 45 The purpose of this project was to determine items considered vital to the consistent and complete critique of the essential oil, treatment methods, and olfactory aspects of aromatherapy research in humans. For the TREATS checklist, the course followed a similar one as outlined by Whiting et al. with identification of need and interest, development of a list of items, and dissemination continuing in an iterative fashion throughout the project 9 (Fig. 1). As in other quality appraisal tool development studies, an explanatory document was created to help users better understand the items and reduce variability in scoring. 45
Working to achieve consensus is a crucial characteristic of quality appraisal checklist creation. For the TREATS, similar methods as those used by Lucas et al., including examination of principles, existing contemporary and extant literature, expert collaboration, and brainstorming, were chosen. 46 One limitation was that the taskforce was only able to examine and critique literature in the English language.
Recommendations for tool development often include at least one face-to-face meeting and the need for funding. 9,45,46 Although ARQAT members were unable to meet in person due to time, distance, and schedules, online meetings made it possible to move this work forward. Online meetings also reduced our need for funding to achieve the goal of creating the TREATS.
The TREATS was created over a 9 month period (Supplementary Material S1). This rapid creation was largely due to the use of input from a convenient though diverse group of colleagues and those known to core members of ARQAT. This fills a gap at this time when no critique checklist exists to support the critique of aromatherapy studies using essential oils in humans. Recent aromatherapy-focused studies using essential oils have cited use of the herbal extension for the CONSORT. 47,48 Until a reporting guideline is completed, the TREATS helps to fill this gap.
The TREATS is intended as a quality appraisal checklist of the essential oil and aroma therapeutic aspects of research and is meant to be used in conjunction with other tools that fit study methodology including other quality appraisal checklists such as the Grading of Recommendations, Assessment, Development, and Evaluations 49 framework and the Critical Appraisal Skills Programme 50 tool. Those creating and reporting all types of research involving essential oils and aromatherapy in humans are strongly encouraged to use the most appropriate quality appraisal tools and reporting guidelines at their disposal. 51
In common with a reported 49% of critical appraisal tools incorporating scoring systems, 45 the TREATS employs scoring. Whiting et al. discourage use of a scoring system due to concerns that users will simply calculate quality scores from an overall total. 9 They recommend the use of a domain-level structure that ARQAT has used to create a more nuanced scoring system. The TREATS has four domain-like structures, titled sections, each of which receives a sub-total. Like other checklists, the TREATS uses met, partially met, and did not meet. 52
The overall score summed from the totals of each section is then ranked poor, fair, or good with space to allow for comments that can further qualify what the score indicates and where improvements may be made. It is acknowledged that a higher score may not necessarily indicate a superior study when risk of bias and applicability are also evaluated using other quality appraisal tools. 53
The TREATS is slightly longer than some other quality appraisal checklists. 52,54,55 While 38 items may seem excessive, studies meeting all or most of these items are more likely to have meaningful, accessible, and replicable results in relation to the aroma and essential oil aspects of these studies. To address researchers' concerns about journal word count limits, items could be reported in the form of tables or online Supplementary Materials S1 and S2.
Conclusion
Until a reporting guideline is created, the TREATS helps to fill a gap for researchers wishing to create and critique aromatherapy-focused studies using essential oils in humans. Increased reporting quality in aromatherapy research will ultimately benefit future research, clinical recommendations, and health policy decisions. Use of the TREATS as a quality appraisal checklist by peer reviewers and journal editors may lead to improved research reporting and confidence in the findings of aromatherapy research studies. Looking to the future, the insights and knowledge gained through creation of the TREATS serve as the foundation for a broader consensus-based study to create an aromatherapy-specific extension to be used with other reporting guidelines.
Footnotes
Acknowledgments
The authors wish to thank all those who participated in the design and revision of the TREATS. Specific attributions are made in the white paper available at ![]() . Also, special thanks are due to the Gattefossé Foundation and Delphine Marchand; and to William McGilvray for his expertise and depth of knowledge. Finally, the authors express great appreciation to Roger Carpenter and Steffany Moonaz for reviewing earlier versions of this article.
. Also, special thanks are due to the Gattefossé Foundation and Delphine Marchand; and to William McGilvray for his expertise and depth of knowledge. Finally, the authors express great appreciation to Roger Carpenter and Steffany Moonaz for reviewing earlier versions of this article.
Authors' Contributions
M.E.R. was involved in the conceptualization, project administration, supervision, validation, and visualization of the article. E.J.B., D.D.A., and M.M.C. were involved in conceptualization and validation. J.T. was involved in conceptualization, resources, and visualization. B.K.L. was involved in supervision. D.J.J. was involved in funding acquisition. All authors, including M.E.R., E.J.B., D.D.A., M.M.C., D.J.J., B.K.L., A.M.F., M.P.C., J.A.R., J.M.T., and B.J.U., were involved in the investigation, methodology, writing of original, and writing, reviewing, and editing of the article in its current form.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Financial support for open access publication was provided by the Gattefossé Foundation.
Supplementary Material
Supplementary Material S1
Supplementary Material S2
Appendix A1. Transparent Reporting for Essential Oil & Aroma Therapeutic Studies (TREATS) Checklist
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
