Abstract
Objectives:
Homeopathy (HOM) is a therapeutic method, which is widely used by patients and medical professionals. The medical conditions as well as the homeopathic medical products investigated vary strongly. There is an extensive amount of research, and this necessitates a bibliography that comprehensively presents the entire body of clinical evidence grouped according to medical conditions.
Design:
Thirty-seven online sources as well as print libraries were searched for HOM and related terms in eight languages (1980 to March 2021). We included studies that compared a homeopathic medicine or intervention with a control regarding the therapeutic or preventive outcome of a disease (classified according to International Classification of Diseases-10). The data were extracted independently by two reviewers and analyzed descriptively.
Results:
A total of 636 investigations met the inclusion criteria, of which 541 had a therapeutic and 95 a preventive purpose. Seventy-three percent were randomized controlled trials (n = 463), whereas the rest were non-randomized studies (n = 173). The leading comparator was placebo (n = 400). The type of homeopathic intervention was classified as multi-constituent or complex (n = 272), classical or individualized (n = 176), routine or clinical (n = 161) and isopathic (n = 19), or various (n = 8). The potencies ranged from 1X (dilution of −10,000) to 10 M (100− 10.000). The included studies explored the effect of HOM in 223 medical indications. We present the evidence in an online database.
Conclusions:
This bibliography maps the status quo of clinical research in HOM. The data will serve for future targeted reviews, which may focus on the most studied conditions and/or homeopathic medicines, clinical impact, and the risk of bias of the included studies.
Introduction
Homeopathy (HOM) is a therapeutic method using homeopathic medical products (HMPs), which are selected based on the therapeutic principle “Let like be cured by like” or “Simile Principle.” 1 When used according to homeopathic textbooks, the HMP is selected based on the similarity between the symptoms that it causes when given to healthy persons and the symptoms of the patients. HMPs are used in various administration forms and the clinical research comprises a large diversity of HMPs.
Although several systematic reviews and meta-analyses on HOM have been conducted in the last two decades, 2 –5 their results have been inconclusive. 6 Thus, the debate whether HOM is a beneficial complement to conventional medical care remains active. 7,8 Next to the heterogeneity of homeopathic practices, the inconsistent results and conclusions of previous systematic reviews and meta-analyses may be caused by differences in the process of study selection by the individual reviewers: 9 most reports or reviews restrict the inclusion of studies by various criteria, 3 –5,10 sometimes even without defined selection criteria. 3,10 Most major reviews have been limited to randomized controlled trials (RCTs) comparing HMP with a placebo control, 2 –5,10 with some systematic reviews also comparing other-than placebo controls, such as best-practice conventional medical care, treatment as usual, or nutritional counselling, with individualized or non-individualized homeopathic treatment, respectively. 11,12
Although RCTs are usually considered as higher level of evidence, 13,14 maximizing internal validity comes at the cost of reduced external validity. 15,16 Whether a given intervention works under real-life conditions and whether only evidence from RCTs is able to answer the relevant health care questions remain uncertain. 16 –18 Therefore, recent evidence-ranking schemes like GRADE, 19 judge the quality of evidence not only by the study design, but consider also directness, precision, and consistency.
Another factor to be aware of when reviewing
An alternative way of expanding the evidence of HOM in specific clinical conditions is to consider not only placebo-controlled or pragmatic RCTs (controlled by placebo or otherwise), but also controlled non-randomized studies (NRS), either with an active comparator or with placebo or no treatment (including usual care i.e., add-on
Moreover, conceptual flaws in the design of RCTs often lead to a lack of coherence with the homeopathic intervention as it is done in practice, which in turn may bias the results due to low model and external validity. 15,27 –29 The majority of studies on HOM has been repeatedly said to be of low study quality and thus overall effect size estimations were deemed inconclusive. 3 –5,10 Nevertheless, we propose that homeopathic intervention studies (HOMIS) should be evaluated by including evidence from both RCTs and NRS. Thereby, the twin requirement of considering internal and external validity together, of studying an intervention in experimentally well-controlled as well as in natural settings, can be met. 30
The necessary starting point for any scientific evaluation of the diversity of HOMIS in targeted systematic reviews or meta-analyses is a systematic bibliography, which arranges the empirical literature regarding specific interventions, comparators, diagnoses, and outcomes as suggested for conducting systematic reviews. 31 Such a bird's-eye perspective will result in a data collection of HOMIS that is condition related. It can then be accessed by interested medical practitioners or researchers and it will make obvious any research gaps by identifying conditions with scarce empirical evidence.
Our goal, therefore, was to provide a systematically processed bibliography of controlled HOMIS in treatment or prevention of diseases in humans as a basis for upcoming condition-related systematic reviews and meta-analyses of homeopathic interventions.
Methods
A study protocol for this bibliography had been published in 2020. 32
Study eligibility criteria
All controlled clinical investigations (RCTs or NRS), employing one or more HMPs on humans exhibiting an International Classification of Diseases (ICD)-10 listed disease (treatment interventions) or on humans in danger of developing a disease (prophylactic interventions), were eligible. 32 Combined products containing mother tinctures and potentized substances were included, whereas products containing mother tinctures only were excluded. We included studies which compared HOM with (1) placebo, (2) active controls, (3) standard care or treatment as usual, and (4) no treatment (including waiting list controls). Substantial research articles (either peer-reviewed or not), reports, conference proceedings, and master or doctoral theses of all languages, identified by the database search, were considered. The time limit was set as “date of publication” between January 1, 1980 and March 15, 2021. Further eligibility criteria and translation policies are described in the protocols. 32,33
Information sources
The information sources were the databases, Medline, Embase, Cochrane Central Register of Controlled Trials, CORE-Hom and CAM-Quest, SCOPUS, Science Citation Index, AMED and CINAHL, LILACS, the “AYUSH RESEARCH PORTAL” (ARP), and the Indian Journal of Research in Homeopathy (IJRH) websites, 34,35 as well as the print library of the Carstens-Stiftung, Essen, Germany, the Faculty of Homeopathy, Glasgow, United Kingdom, and of the Central Council for Research in Homoeopathy, New Delhi. Results were supplemented by searches in online databases for master and doctoral theses (Supplementary Data S1).
Search methods
The databases CAM-Quest and ARP were searched by librarians. CORE-Hom, HOMINFORM, and the IJRH-homepage were screened manually (Supplementary Data S1). For the other databases, search criteria have been developed following the PRESS recommendations for electronic search strategies. 36 The search criteria are provided in Supplementary Data S1. K.G. and one assistant conducted the search under supervision of an external advisor.
Study selection
Data records of the obtained references were managed with the software Endnote X7 and X8 (Clarivate Analytics, Boston). After removal of duplicates and screening by two reviewers, full texts were retrieved for all reports of interest. References were transferred into a standardized and piloted Excel® spreadsheet, Version 16.16.10 (Microsoft Corporation, Redmont) for further data extraction as described previously. 33
Data items and data collection process
Therapeutic and prophylactic studies were processed separately. The prophylactic use of HOM was defined as at least one single homeopathic intervention to which the study population was exposed before the exhibition of symptoms or in a symptom-free period. Characteristics (title, author, year, language, sample size, target population, study and intervention design, homeopathic intervention, potency, and outcomes) from studies obtained after title and abstract screening were extracted based on the authors' description as stated in the study protocol
33
and inclusion status was rechecked by full-text revision. In addition, the online serials directory, Ulrichsweb,
37
was consulted to determine the peer-review status of journal articles. The study design was coded as RCT or NRS. The randomization status was based on the original authors' definition. The comparator was categorized into placebo, placebo+ (as add-on to conventional therapy), active treatment, active treatment+, and no treatment, including waiting list and usual care (add-on
The homeopathic intervention was classified as “pluralistic” when more than one single or complex HMP was used at a time; as “routine” in case of pragmatic or so-called “clinical” prescriptions; as “isopathy” if the same substance which caused the disease was administered to the patient processed as an HMP; or as three types of “classical”
The sample size was extracted as the total number of patients in all treatment arms. The investigated diseases were categorized by the ICD-10, version 201638 as well as ciphered and grouped according to the checklist for International Classification of Functioning, Disability, and Health with regard to the primary outcome (ICF Checklist, version 2.1a clinical form 39 ). If no primary outcome was defined, a hierarchical approach to define the most suitable outcome was applied. 32 For the classification into the ICD-10 code, 38 the condition of study population and the main endpoint of the study were used. 32
Data management and statistics
The data were analyzed with descriptive statistics and summarized in tables or with graphs. The tables were deducted from the data extraction file (Excel) and double checked using Microsoft VBA Macros. The algorithms certified the integrity of the data in multiple levels. Each of the records was checked one by one for uniqueness and accuracy. Any possible gaps, double recordings, or spelling errors in the deduction process were eliminated. Figure 2 was built using Microsoft Excel 2019, Figure 5 was created with plotly (
Results
The study selection process is shown in Figure 1; the timeline and the reasons for excluding studies are presented in the Table A1 in Supplementary Data S1. The excluded references are listed in Supplementary Data S2. We included 636 studies of which 541 had a therapeutic purpose and 95 were preventive.
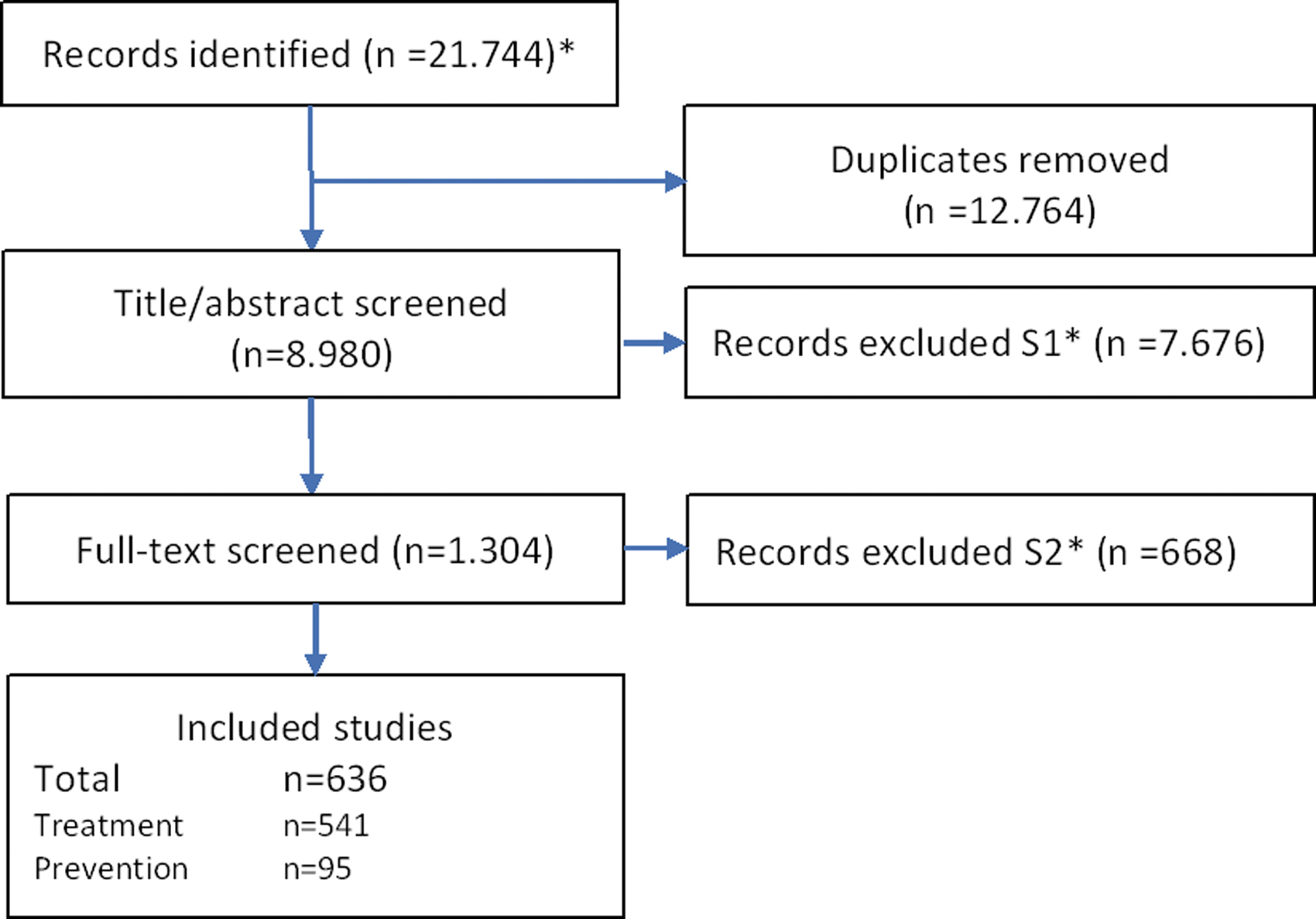
Flowchart of literature search. *See Table A1 in Supplementary Data S1 for details on literature search, including reasons for exclusion during screening S1 and S2.
Figure 2 displays the number of included studies per year from 1980 to March 2021 and the corresponding percentages of studies published in specific languages as well as the relative proportions regarding the publication type. Additionally, Figure 2B shows a treemap of the number of studies published in a given journal, categorized by the genre. The majority of studies was published in journals of a specific medical specialty (n = 186), followed by
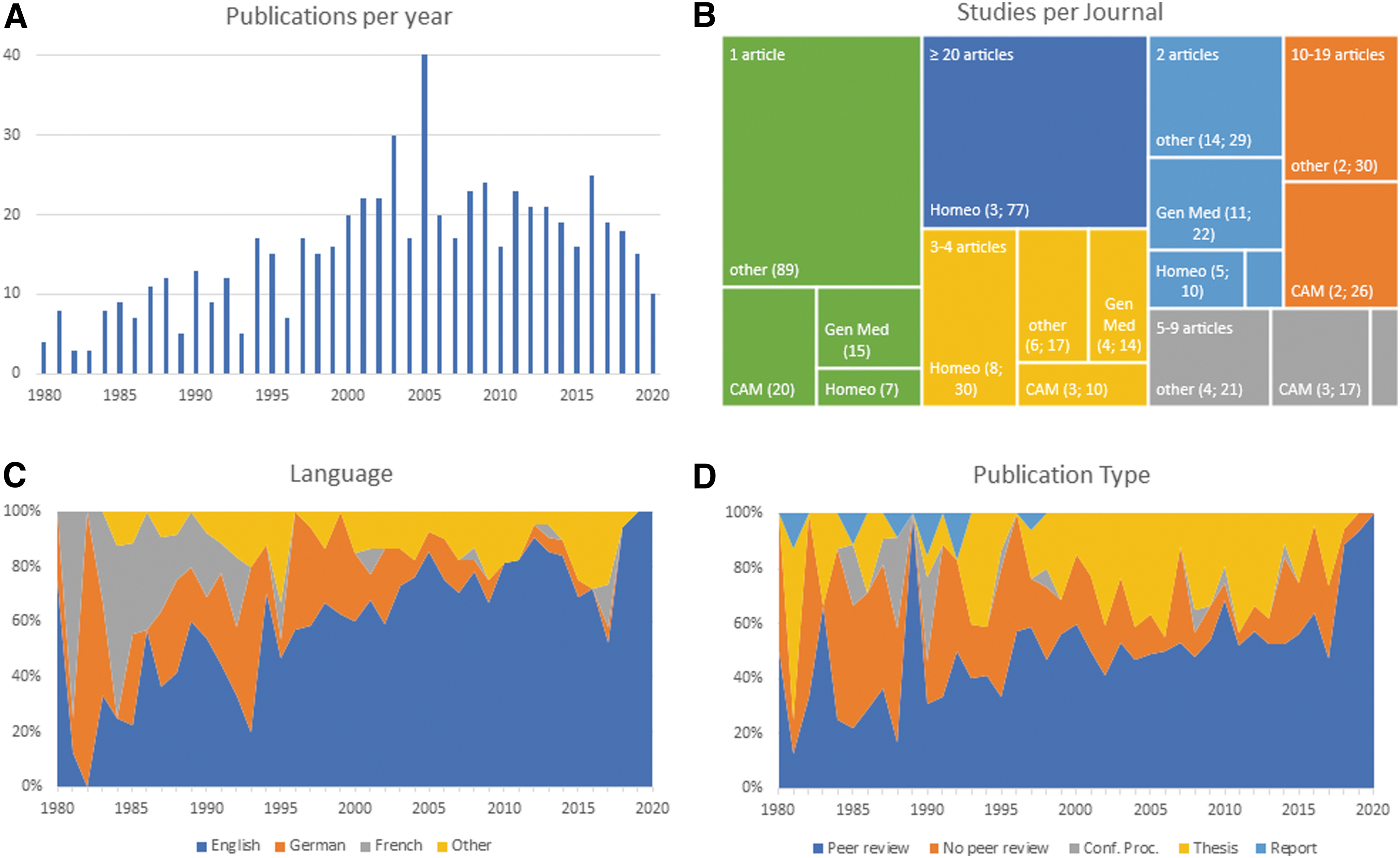
Characteristics of included studies.
Nearly three quarter of the studies were RCTs (n = 463) and the rest were NRS (n = 173) and the leading comparator was placebo (n = 400). The Sankey diagram in Figure 3 exhibits study characteristics with the affiliations between study design, medical sector, comparators, and sample size.
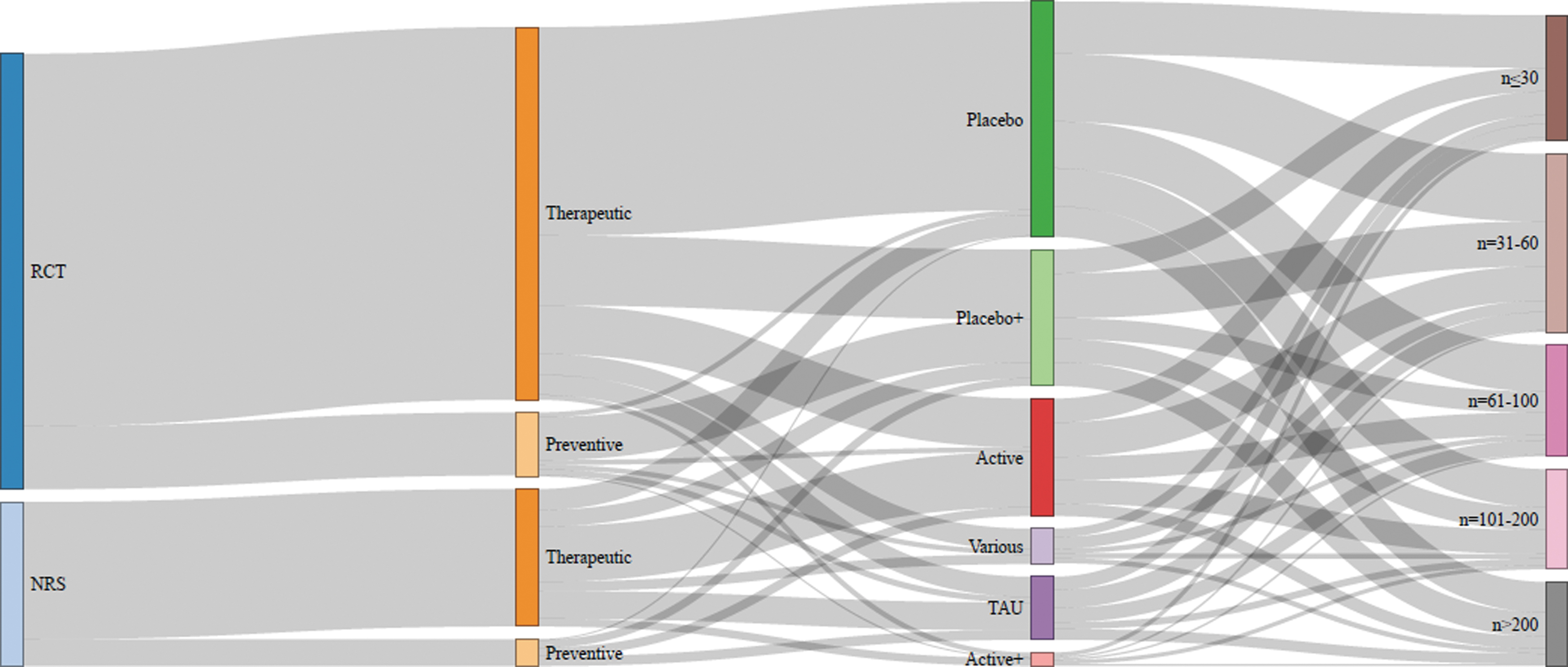
Sankey chart of study design characteristics: From left to right: (1) RCTs and NRS had either (2) preventive or therapeutic purpose; (3) the controls were placebo or an active treatment with (+) or without additional conventional therapy, TAU, or combinations (various); (4) the sample sizes ranged from n ≤ 30 to n > 200. NRS, non-randomized studies; RCT, randomized controlled trials; TAU, treatment as usual.
Clinical effects of HOM were analyzed for 223 different medical conditions. In 11 of those, HOM were tested for both, therapeutic and preventive approaches. All studies are gathered in an online database which can be accessed under this URL:
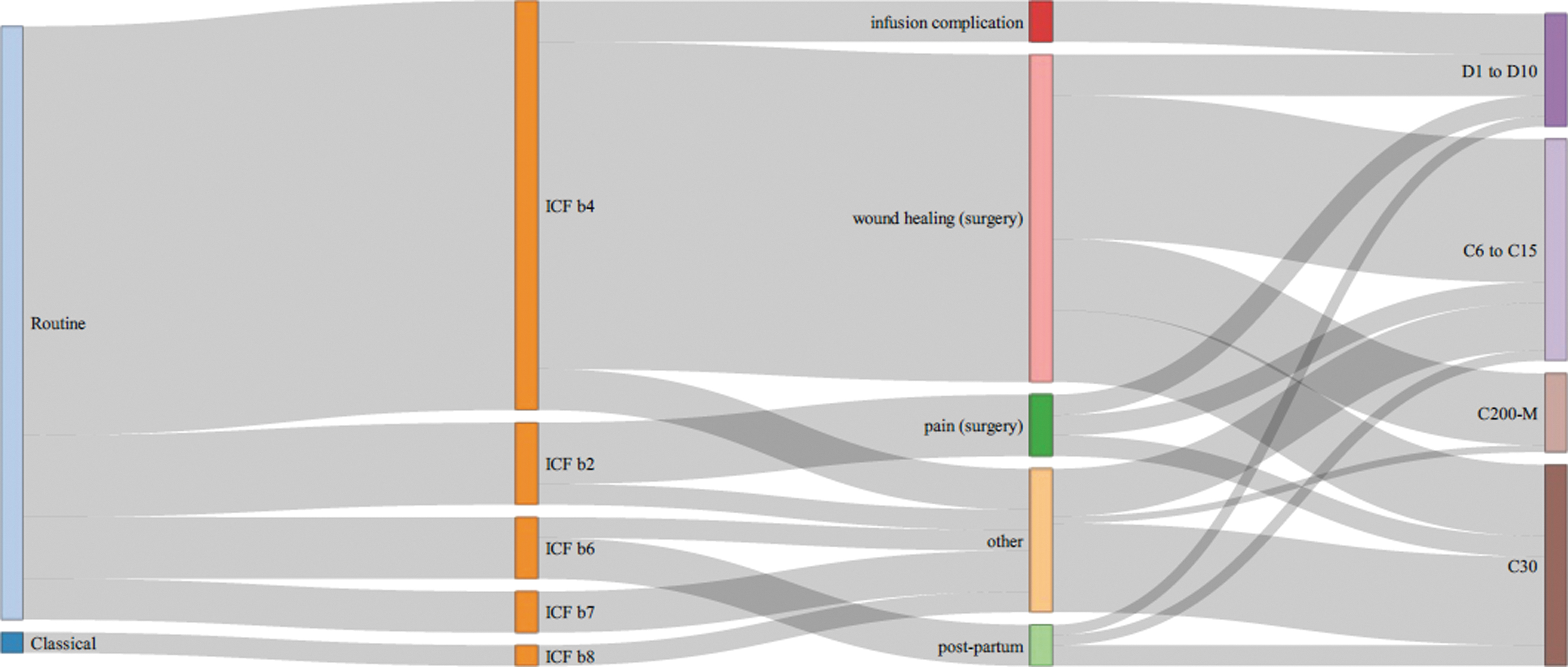
For each study, the database provides a detailed reference, ICD-10 code, trial design, comparator, and sample size. It can be screened through a search field and will be updated biannually. While the heterogeneity of medical disease according to ICD-10 limits a visualization beyond the tabular organization, the respective ICF categories can be related to the overarching medical goal and to the homeopathic type of intervention (Fig. 4). The active ingredients of complex HMP are shown in Supplementary Data S3.
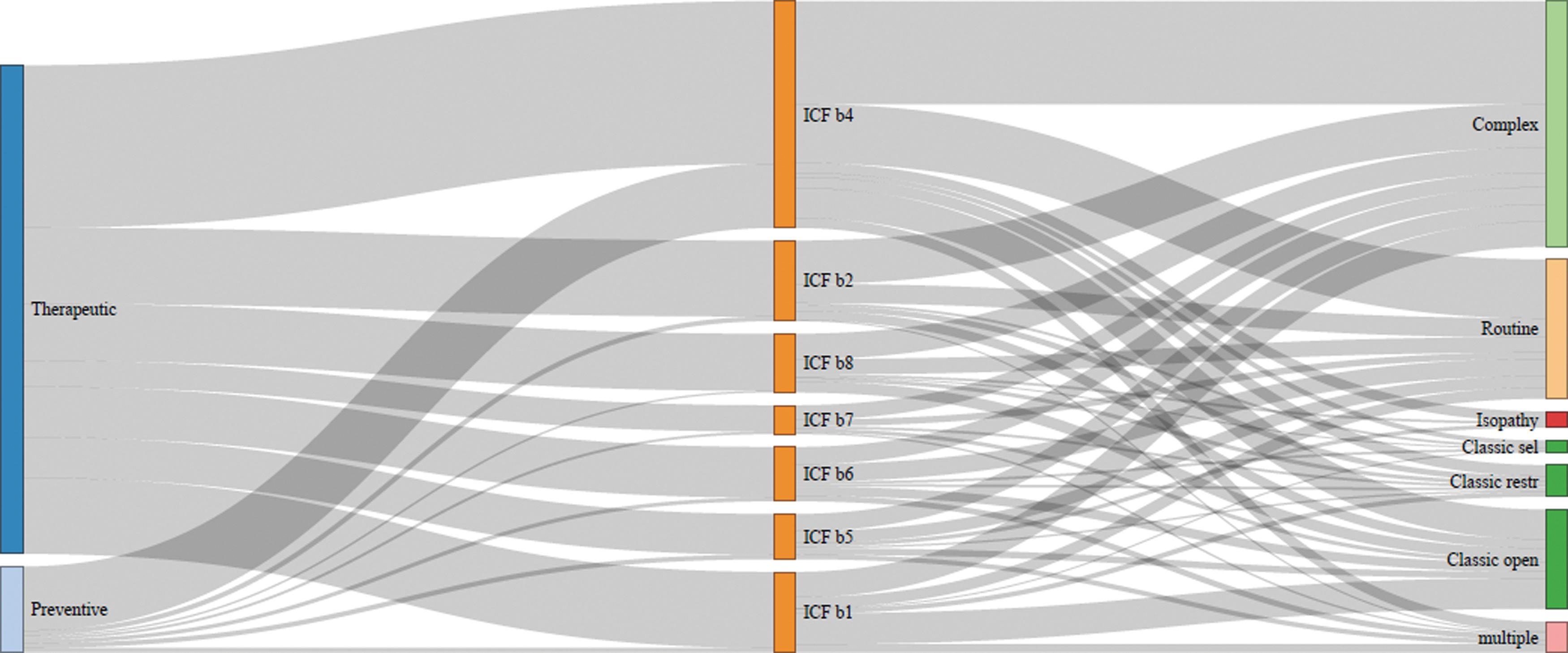
Sankey chart of WHO ICF classification and type of homeopathic intervention. ICF b1: mental function; b2: sensory functions and pain; b3: voice and speech; b4: cardiovascular, hematological, immunological, and respiratory systems; b5: digestive, metabolic, and endocrine system; b6: genitourinary and reproductive; b7: neuromusculoskeletal; b8: skin and related structures; Classic sel: predefined medicines are given to selected patients, Classic open: prescriber can choose any medicine, Classic restr: prescriber chooses from a limited group of medicines.
For 54 medical conditions at least two studies (range 2–19) testing a similar, specific HMP [originating from the same homeopathic application form or substance(s) in the identified studies] were found. For instance, there are 9 studies dealing with the influence of individualized
Figure 5 displays which HMPs are given in which range of potencies for the influenza-like syndrome as an example for an indication with a high number of studies (see Supplementary Data S4 for further indications).
The most frequently studied single HMP was A. montana (n = 47) in various potencies and 12 indications, administered either alone as a routine prescription or in combination with other HMPs. Figure 6 reveals that A. montana is most often used for wound healing and is administered in a wide range of potencies. Other frequently used single preparations were Oscillococcinum (n = 9), Arsenicum album (n = 7), Hypericum (n = 6), Pulsatilla pratensis (n = 5), and Sulfur (n = 5).
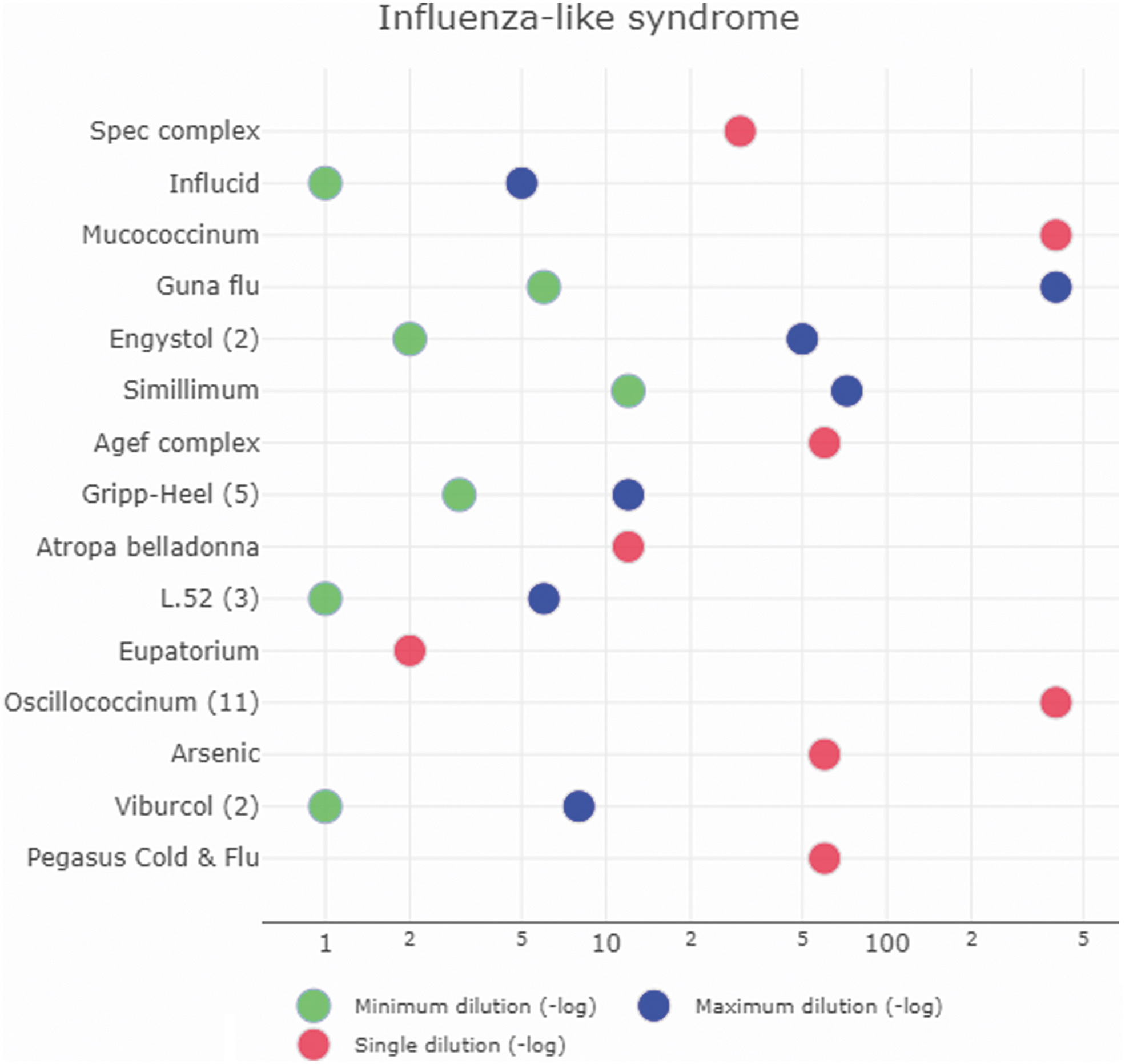
Homeopathic medicinal product and its dilution for influenza-like syndrome. Green and blue dots display the minimum and maximum dilution, respectively, red dots represent a single dilution. The potencies are shown as negative log-values (e.g., the potency C6 stands for a dilution of d = 1:1012 or d = 10−12. Thus, the dot would be at the position “12.” In the same vein, C30 would be at “60,” C200 at “400,” and D5 at “5”).
Sankey chart for studies in which Arnica montana was the single homeopathic medicine. ICF b2: sensory functions and pain, b4: cardiovascular, hematological, immunological, and respiratory systems, b6: genitourinary and reproductive, b7: neuromusculoskeletal, b8: skin and related structures; a potency Dx stands for a dilution of d = 10−x, Cx for d = 10−2x, and M for d = 10−2000.
Discussion
For this bibliography, a comprehensive literature search has been carried out in 10 databases and more than 25 additional sources. We identified nearly twice the number of studies compared with the most recent review of clinical trials on HOM. 6 The present overview encompasses the inclusion of NRS, which are of high impact for public health questions as their higher external validity are closer to real-world health care conditions. 15 –17 In addition, we also included theses and non-peer-reviewed articles to enhance the amount of available evidence regarding clinical effectiveness of HOM. 16 The 636 controlled studies found could be allocated to 223 medical conditions. In 54 conditions at least two homogeneous studies could be identified. Our online database allows access to this evidence for interested practitioners and researchers, or other stakeholders with specific clinical questions. Thus, the first substantive outcome of this bibliography is the surprisingly large number of research studies on HOM available.
This publication intentionally does not provide any information on the result of the studies, for example, effect sizes and statistical summaries. Reporting significance levels without proper risk of bias assessment would have been futile as they are dependent on the power and other quality criteria of the study. For instance, randomization aims at avoiding the influence of prognostic factors, so an assessment of its risk of bias is needed to interpret the relative effect of two interventions. Due to the high number of included studies such a comprehensive but consistent and meaningful evaluation of combined results was, contrary to the intent of the original research plan, 33 not feasible. As the identified studies are also highly heterogeneous, the originally planned subgrouping of pathologies (e.g., acute or chronic inflammatory vs. degenerative diseases), was replaced by this encompassing bibliography. Rather than focusing on potentially simplified criteria to conclude efficacy of HOM across all methods and conditions, we decided to provide a bird's-eye perspective of the empiric literature of HOMIS.
This will serve as a basis for future research regarding, for instance, the effectiveness of a specific homeopathic intervention for a defined clinical problem. Following that approach, a review and meta-analysis on homeopathic Arnica in postsurgical recovery is already published
40
and two others on individualized
This targeted evaluation of the various HMPs and administration forms is crucial to facilitate an evidence-based medical decision making instead of sticking to a dogma-based approach that all interventions with HOM are not effective 7 although empirical evidence might be different. 8 Taking evidence from both, RCTs and NRSs, into account, HOM is already recommended in clinical practice guidelines for specific medical conditions. Examples include additional homeopathic treatment in cancer patients 41 or fibromyalgia. 42
We suggest, therefore, to include evidence from RCTs and NRSs likewise for future condition-specific reviews and meta-analyses of HOM, as a comprehensive assessment of a therapeutic intervention with recommendations for clinical use. Evidence of efficacy can be obtained from placebo-controlled RCTs. Evidence of clinical effectiveness can be obtained from randomized or non-randomized comparative studies with conventional treatment or from cohort studies, as well as from pragmatic RCTs comparing homeopathic care to conventional care. 15
Many of the studies included in this bibliography and descriptive analysis may lack methodological quality and many studies have a small sample size. Furthermore, the body of evidence of HOMIS comprises various application forms of HOM, of which some may lack consistency with homeopathic treatment guidelines. 28,43 Several authors of previous systematic reviews and meta-analyses of trials investigating HOM have pointed out that in many studies, the type of homeopathic intervention researched may not reflect usual practice. 2,44,45 Homeopathic treatment plans are highly individualized. Patients suffering from the same condition often receive different HMPs and the specific HMP or potency is adjusted frequently, according to the treatment effect (feedback from the patient). Such feedback processes are difficult to implement in long-term trials and strict RCT conditions. This is an additional reason why open, pragmatic, or non-randomized practice-based HOMIS are important to consider.
With this in mind, it will be essential to evaluate to what extent HOMIS represent “good homeopathic practice” or routine homeopathic care alongside the methodologic quality (e.g., risk of bias) of the particular studies. Although clinical relevance and coherence of the intervention with the homeopathic principles are necessary elements when analyzing the quantitative effects, a suitable and complete quality assessment tool for HOMIS is still warranted. Therefore, our research group developed such an instrument, following a Delphi process with
Conclusions
The present map of clinical evidence may serve as a basis for future condition-related systematic reviews and meta-analyses of HOM. Suitable quality assessments are warranted.
Footnotes
Acknowledgments
Concept and Design: Prof. emer. Michael Kundi (Medical University of Vienna), Loredana Torchetti (Institute of Complementary and Integrative Medicine, University of Bern); Study search: Susanne-Monika Rehm (Deutsche Homöopathie Union—DHU); Polyxeni Kalafatis (Medical University of Vienna); data extraction: Loredana Torchetti and Alexandra Stöckli (Institute of Complementary and Integrative Medicine, University of Bern), Christina Leinert (Medical University of Vienna), Tania Simandopoulou (MD, Thessaloniki); data management: Thanasis Meliotis (surveying engineer, Vienna).
Authors' Contributions
K.G. developed the concept and design, coordinated the project, and drafted the article; M.L. analyzed the data and contributed to the article; M.F., H.W., K.v.A., M.F.-E., and S.B. contributed to the concept, the design, and the article. R.Mi., A.K., and
Ethics
No ethics approval has been needed because we retrieved and analyzed data from previous published studies in which informed consent was obtained by primary investigators.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The Tiedemann Foundation for Classical Homeopathy, the Homeopathy Foundation of the Association of Homeopathic Doctors (DZVhÄ), both in Germany, and the Foundation of Homeopathy Pierre Schmidt and the Förderverein komplementärmedizinische Forschung, both in Switzerland, provided project development and/or bridge funding, but did not have any role or influence in the design and conduct of the project, in its evaluation, decision to publish, and contents of the article.
Supplementary Material
Supplementary Data S1
Supplementary Data S2
Supplementary Data S3
Supplementary Data S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
