Abstract
Inhaler-based digital biomarkers can be objectively evaluated to indicate changes in response to therapeutic interventions. In chronic respiratory disease (CRD), measures of exhaled airflow play a central role in diagnosis and management, yet inhaled measures have not. Digital inhalers can passively collect inspiratory parameters such as inhaled volume (InhV) and peak inspiratory flow (PIF) during real-life patient use. Emphasis has been directed to the PIF, but little is known about the role of the InhV. InhV has now been observed to decrease prior to an exacerbation of asthma or chronic obstructive pulmonary disease and increase afterwards and be lower in uncontrolled asthma patients. These observations are consistent with the current understanding of the pathophysiology of CRD, whereby a strong correlation between InhV and forced expiratory volume in one second has been observed. Measurement of InhV has demonstrated accuracy and low variability, supporting the validity of interpreting individualized trends over time. After counseling, patient inhalation technique decays over time, and it is InhV that decreases more than PIF. Together, these findings suggest that InhV, captured by a digital inhaler, has utility as an effective digital biomarker to aid management of patients with CRD, with InhV being a major factor in the predictive value with respect to acute exacerbations.
Introduction—Unmet Needs in Chronic Respiratory Disease
Despite advances over decades in the treatment of chronic respiratory diseases (CRDs) such as asthma and chronic obstructive pulmonary disease (COPD), considerable unmet needs in the management of patients with these conditions remain to be addressed. Poor asthma control, inadequate management of COPD symptoms, and misdiagnosis are still widespread in real-world practice.1–3 Inhaled medications remain the mainstay of treatment for the majority of patients with these conditions.4,5 They were first introduced in 1956 and, immediately, problems with technique were identified. 6 From their introduction until now, inhaler technique has been frequently poor.7,8 Furthermore, adherence to prescribed maintenance therapies such as inhaled corticosteroids (ICS) is often suboptimal, 9 with and without poor inhalation technique. 10 Adherence has been found to be much lower in real life relative to that seen in randomized controlled trials. 11
Assessment of key diagnostic criteria for both asthma and COPD involves the measurement of airflow. While acknowledging that asthma is a heterogeneous disease, the core definition of the disease per the Global Initiative for Asthma (GINA) specifies the presence of “variable expiratory airflow limitation.” 4 For COPD, the Global Initiative for Chronic Obstructive Lung Disease (GOLD) states that the diagnosis of COPD is confirmed by the spirometric measurement of airflow obstruction that is not fully reversible. Both GINA and GOLD now acknowledge access limitations with respect to diagnostic spirometry,4,5 particularly in low- and middle-income countries; GINA also notes that lung function testing may be underused in the diagnosis of asthma due to clinical time constraints. 4
Following a diagnosis of asthma or COPD, ongoing assessment also poses substantial challenges. Other than among patients with the most severe disease, consultations with a health care professional (HCP) are typically occasional, with the patient necessarily having to self-manage their condition in the meantime. At the consultation, patients should usually be asked about their symptoms during the intervening period; however, responses to such enquiries are known to be subject to substantial recall bias, 4 including with respect to the past occurrence of exacerbations of the disease. 12
In recent years, recognition has grown of the potential for digital technologies to revolutionize CRD care and overcome these long-standing barriers.13–15 In addition to the now well-established utility of digital technologies in monitoring adherence to maintenance medications, potential exists to leverage inhaler-based digital biomarkers to provide hitherto unobtainable insights into the CRD state. The potential of digital inhalers, which can be supported by dedicated mobile apps, is now being identified. 11 Some only identify that a dose has been released and so are useful as a surrogate measure of compliance 11 and bronchodilator overuse. 16 More sophisticated digital inhalers use sensors to measure an inhalation profile. 11
Typical inhalation profiles are shown in Figure 1, illustrating the peak inspiratory flow (PIF), the inhaled volume (InhV), the length of the inhalation (Ti), and the time to the PIF (Tp) (Table 1) of patients making an inhalation from a dry powder inhaler (DPI) with fast and slow acceleration, respectively. These parameters can be measured in the clinic by an inhalation profile recorder (IPR)17–19 and, now, during real-life inhaler use 16 by digital inhalers 11 that have electronic sensors inside the actual inhaler. Measuring an inhalation profile, during real-life use, confirms that a dose was prepared (by a sensor, including a date stamp, noting the dose preparation step) and that it was actually inhaled.
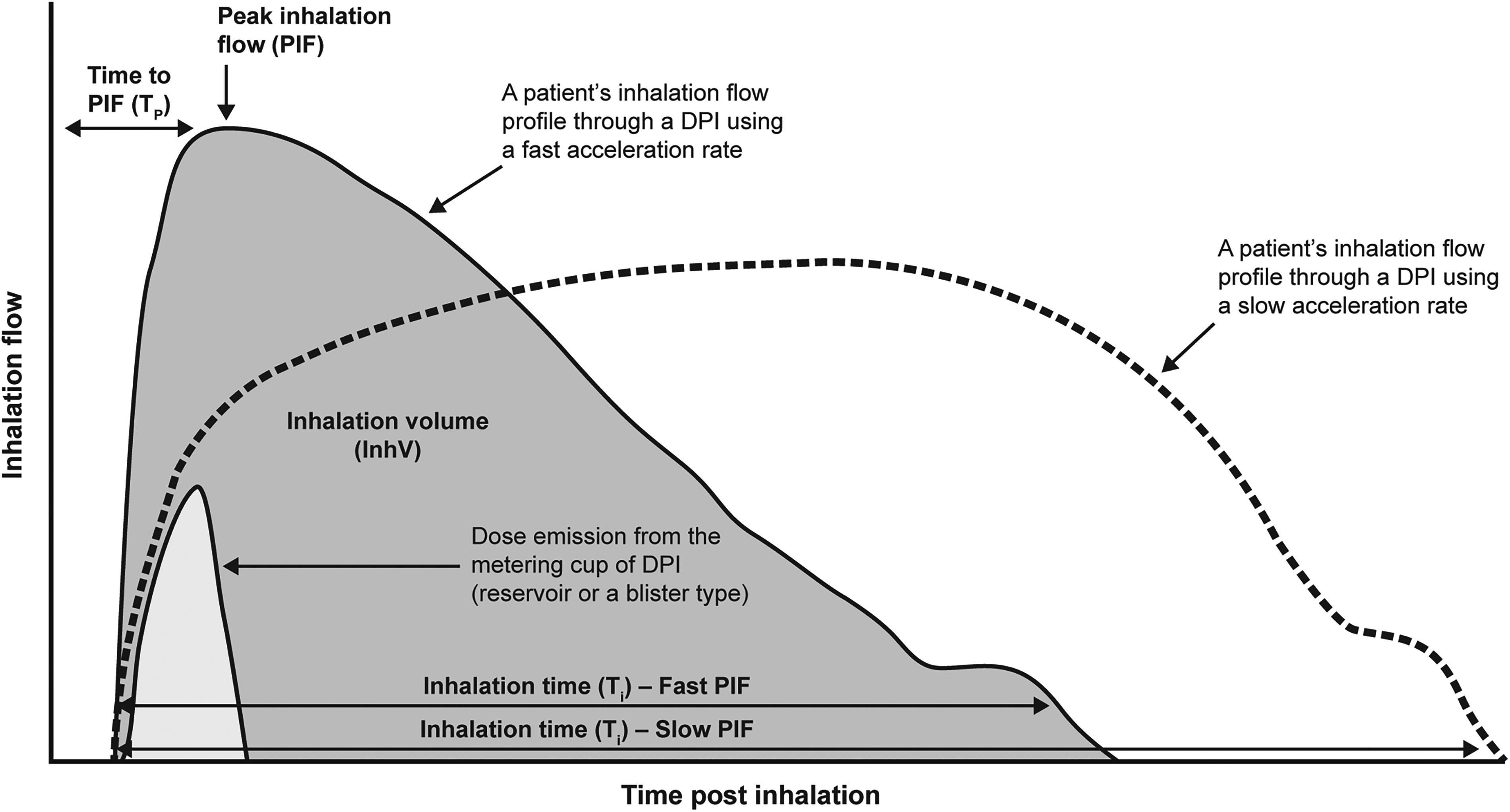
Inhalation profile showing characteristics relevant for dose emission from a dry powder inhaler. DPI, dry powder inhalation; InhV, inhaled volume; PIF, peak inspiratory flow; Ti, the length of inhalation; Tp, time of the PIF.
Inhaler Airflow Measures

Sometimes referred to as Vin.
InhV, inhaled volume; PIF, peak inspiratory flow; Ti, duration of inhalation; Tp, time to PIF; Vin, inhaled volume.
PIF, Tp, and InhV all play important roles in dose emission from a DPI (Fig. 1).18,20–23 Continuing the inhalation, the delivery of the particles (from the emitted dose) is facilitated by the InhV. Until recently, the focus has been on the PIF, because it can be easily measured, while the importance of the InhV has remained unknown. Now that InhV, as well as PIF, can be measured during real-life use of a digital DPI, the potential for this parameter to inform the management of patients using inhalers warrants exploration. Such is the purpose of this review.
Biomarkers in CRD
Biomarkers are measurable characteristics that can be objectively evaluated to indicate normal or pathological biological processes and/or biological changes in response to therapeutic interventions. 24 Both GINA and GOLD note the potential value of biomarkers in guiding treatment.4,5 For instance, the fraction of exhaled nitric oxide (FeNO) and blood or sputum eosinophil count are biomarkers of type 2 airway inflammation in asthma.4,25 FeNO has been shown to predict response to ICS and risk of exacerbation in patients with asthma, 25 while blood eosinophil levels are measured to determine the diagnosis of “eosinophilic asthma” that may be treatable with anti-interleukin-5 biologics. 4 The potential for patient-reported outcome measures to serve as digital biomarkers in asthma has also been explored. 26
In COPD, a blood eosinophil count ≥300 cells/mL has been shown to indicate a higher risk of exacerbation and an increased likelihood of benefiting from preventative treatment with ICS. 27 Devices for the measurement of oxygen saturation and/or respiratory rate may also have some value in COPD, although further study is needed. 28 Measures of exhaled airflow play a central role in the diagnosis and management of CRD.4,5 These measures include forced expiratory volume in one second (FEV1), forced vital capacity (FVC), and peak expiratory flow (PEF). The ratio of FEV1 to FVC provides the primary diagnostic criterion for COPD—a ratio of 0.7 or lower is indicative of the presence of the condition. 5 A reduction over time in an individual’s FEV1/FVC ratio reflects increasing expiratory airflow limitation, which is known to be a risk factor for exacerbation in both asthma and COPD.4,5
Changes in exhaled variables can signify obstructive or restrictive deficits 29 and disease severity, making them central to both the diagnosis and the ongoing management of CRD. The measurement of FEV1 and FVC requires spirometric equipment, which is seldom available for use by patients on a regular basis with good quality and reproducibility. 4 While patients can obtain and record PEF measurements using a peak flow meter at home, PEF is recognized as a suboptimal measure that lacks specificity (neither asthma nor COPD can be ruled out by measurement of a “normal” PEF 4 ). Furthermore, PEF has a lower correlation with measures obtained via spirometry than might be expected. 5 Importantly, the use of peak flow meters by patients necessitates an active maneuver that is separate from ordinary disease management. It is therefore itself subject to potential technical errors 30 and is often perceived as burdensome by patients, with associated impacts on adherence. 31 Conversely, almost all patients with asthma or COPD use at least one inhaler routinely to manage their condition and relieve symptoms on a day-to-day basis. If an inhaler-based digital biomarker were capable of providing insight into airflow limitation on an ongoing basis—analogous to that provided for glycemia by continuous glucose monitoring (CGM)—such a biomarker might offer invaluable insights to inform individualized care of patients with CRD. Additionally, if such a biomarker can be recorded at the time when a patient feels the need to use their inhaler, it is likely to capture a moment of physiological change without necessitating any additional maneuver.
The emergence of digital inhalers, 11 capable of collecting data electronically via sensors both inside and (critically) outside of the clinical environment on an ongoing basis, has given rise to the concept of digital biomarkers. 24 Such inhaler-based digital biomarkers, which can be monitored remotely by both patients and their physicians, 24 are by now well established in other chronic diseases, for example, the clearly evident value of CGM in diabetes. 32
Digital Technologies and the Inhalation Maneuver
Both GINA and GOLD highlight the importance of patient management of reviewing symptoms and assessing adherence and technique.4,5 However, the value obtained from such reviews is highly dependent on the quality and accuracy of the information supplied, which may not be an accurate reflection of typical day-to-day patient behavior. Digital technologies have the potential to greatly improve data quality and so add significant value to this process. 13 Furthermore, there is evidence to suggest that digitally enhanced adherence interventions in asthma and COPD are likely to be cost-effective, in view of the social and economic costs of poorly controlled CRD.9,33
The potential for digital technologies to help address long-standing challenges in the management of CRD is now well recognized by experts.14,34 Studies of digital adherence monitoring devices such as the Inhaler Compliance Assessment device have highlighted low levels of actual adherence among COPD 35 and asthma, 10 as well as demonstrated improvements in adherence among asthma patients when data from the device were used to guide treatment decisions. 36
In addition to monitoring adherence, some digital inhalers also have the ability to provide insight into the patient’s inhalation technique. 11 Poor inhalation technique can result in what is sometimes termed “unintentional” nonadherence, whereby the patient intends to adhere to the medication regimen and likely fully believes that they are doing so. 35 However, because of “critical” inhaler errors, the patient does not receive the full benefit (or, in some cases, any benefit) from the inhaled medication. 37
The Digihaler® inhaler (Teva Pharmaceuticals) is a Food and Drug Administration (FDA) and Medicines and Healthcare products Regulatory Agency (MHRA)-approved electronic multidose DPI, with integrated digital sensors (eModule) capable of accurately recording time-stamped quantitative data on inhalation parameters, including InhV and PIF (Table 1, Fig. 2). 21 Pressure changes generated by inhalation airflow are detected by the microelectromechanical sensors; these changes are used to calculate the aforementioned inhalation parameters. 21 Results from an open-label study in children and adults with asthma, and adults with COPD, comparing inhalation parameters measured using the Digihaler inhaler with readings made simultaneously using an IPR validated its ability to provide accurate measurement of inhalation parameters. 21
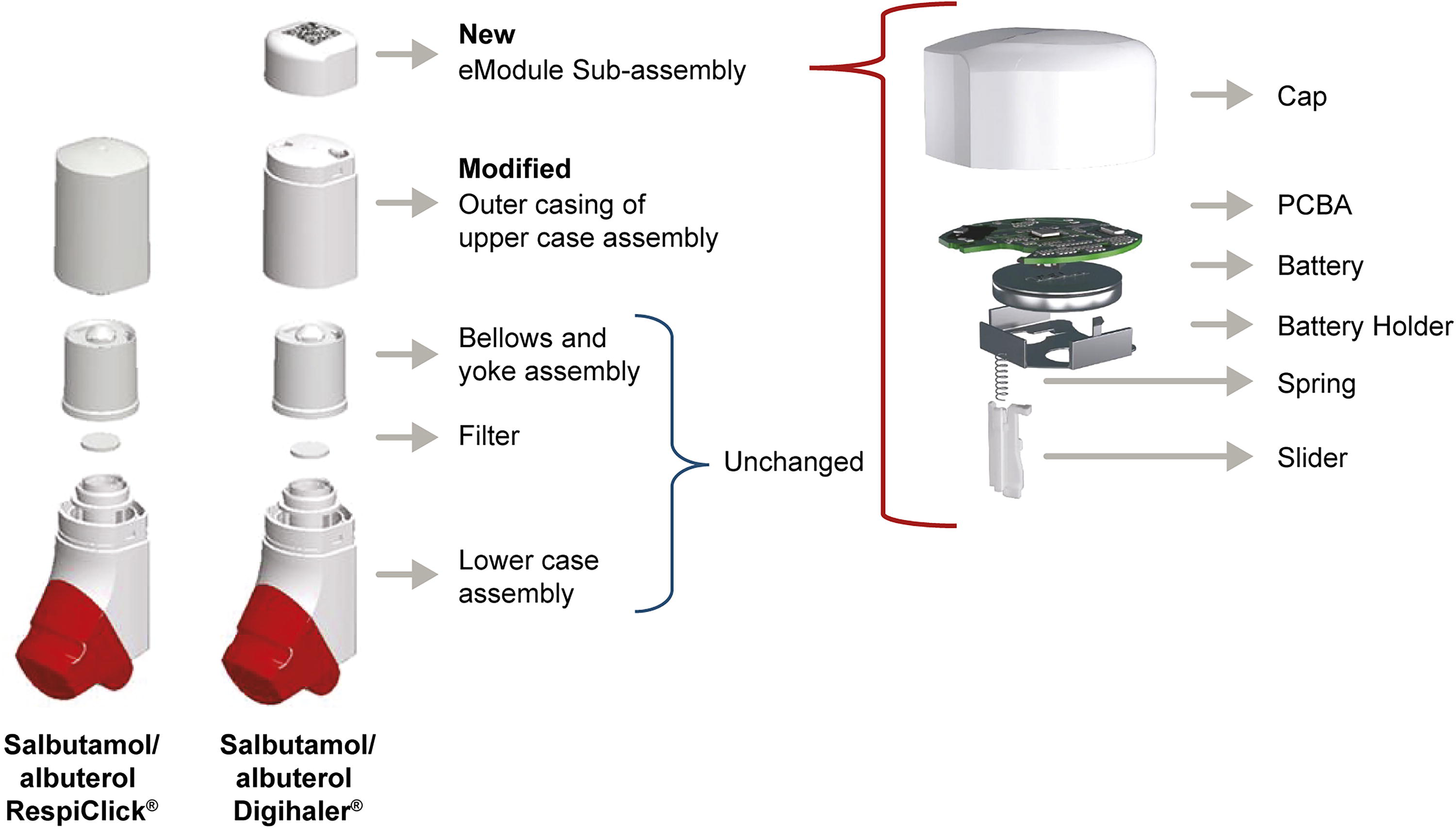
Exploded schematic views of the salbutamol/albuterol RespiClick® and Digihaler® inhalers and the Digihaler eModule. Reproduced with permission from Chrystyn H, Merchant R, Safioti G, et al. The Digihaler®: A new approach to manage asthma and COPD. Respir Drug Deliv 2020;1:249–260.
In the Digihaler System (DS), this DPI with integrated sensors is combined with a patient-facing mobile application (“app”) and a Dashboard, which provides HCPs with insights into each individual patient’s inhaler usage and inhalation parameters.34,38 In two recent randomized studies, patients with asthma using the DS were found to have greater odds of achieving clinically meaningful improvements in asthma control than those continuing to use nondigital standard of care inhalers.34,38 In the more recent of the two studies, in which patients used Digihaler inhalers for delivery of both maintenance and reliever medication, a reduction in weekly short-acting beta2 agonist usage was observed over the 24 weeks of the study. 34 Furthermore, objectively measured inhaler technique was observed to be maintained over the duration of the study, 34 in contrast with typical observations of rapid deterioration of technique following training. 7
Of note, the “app” and Dashboard that form part of the DS report inhalation technique in terms of PIF, with a “good inhalation” being reported if the PIF rate is >45 L/min and a “fair inhalation” reported if the PIF rate is between 30 and 45 L/min. 34 This is consistent with the literature on the airflow necessary for dose delivery from a breath-activated DPI.39,40 In addition to PIF, the sensors record InhV, Ti, and Tp. Importantly, these data are captured passively, as the patient uses their inhaler in the course of ordinary management of their disease and its symptoms. Through the DS, there is the potential to individualize each patient’s inhalation parameters and make recommendations or interventions when these change during routine use.
While InhV is almost certainly of lesser importance than PIF in regard to dose emission from the DPI, 41 there are several other contexts within which InhV is of substantial and largely underrecognized relevance for the understanding and management of CRD.
Dose Emission From a DPI—The Role of the InhV
When a patient uses a DPI, the PIF is dependent on their ability to inhale as fast as possible. This is related to the effort used by the patient and the lung disease status. Tp is linked to the PIF and is why, when using a DPI, the instruction is always to inhale as fast as possible from the start of the inhalation (Fig. 1). PIF and Tp are the drivers for the required deaggregation of the formulation during an inhalation. 42 PIF and the characteristics of the emitted dose can be easily measured; hence, it has been the focus of attention. Instructions for the inhalation maneuver typically ask the patient to first exhale gently as far as comfortable and continue inhaling for as long as possible. The volume inhaled during an inhalation maneuver is measured as the InhV (shown in Fig. 1). This volume is, therefore, dependent on how far the patient exhales and for how long. Like the PIF, it is also related to the condition/disease status. The CRITical Inhaler mistaKes and Asthma control (CRITIKAL) study revealed that in patients with asthma not exhaling as far as possible, not using a fast inhalation, not using a fast inhalation from the start, and not inhaling for as long as possible were related to poor disease control. 37
Hence, the InhV, as it pertains to the usage of a DPI, is the volume of air (usually measured in liters) that is sucked into the lungs during the course of a single usage of the device. On a typical inhalation profile (as exemplified in Fig. 1), whereby inspiratory flow plotted on the y-axis and time post-inhalation is plotted on the x-axis, InhV is represented by the area under the curve.21,23 Effective dose emission from a DPI is dependent on characteristics of the patient’s inhalation maneuver, encompassing InhV as well as inspiratory flow and acceleration rate.19,43,44 Although they do not require the coordination of inhalation and actuation needed for pressurized metered dose inhalers, DPIs necessitate a minimum inspiratory flow rate (generally considered to be a PIF >30 L/min) to release the dose and de-aggregate the formulation. 39 One factor of note that contributes to the device-specific airflow requirements is the internal resistance of the device.19,39 Perhaps counterintuitively, the airflow required for dose delivery from a device with high resistance is typically lower than for a DPI with low internal resistance. 39
The InhV facilitates the emission of the dose from the inhaler during an inhalation and then the delivery of the inhaled particles to all the airways. Figure 1 shows that dose emission occurs in the first part of inhalation, hence the importance of inhaling fast from the beginning. Studies indicate that dose emission from DPIs occurs within the first 500 mL of an inhalation.23,43,45 In our dose emission studies, using inhalation profiles, we did not use an InhV <1 L, which could explain why we did not show any effect related to the InhV.46–48 In these studies, we did find that not all the metered dose was emitted during each inhalation, whereas when a vacuum pump was used in accordance with the U.S. Pharmacopeia methodology 49 (hence with an acceleration of the inhalation with an “instant” Tp [in the order of milliseconds, rather than 0.5–1 second]), then all of the metered dose was emitted. The difference between the onset of turbulence intensity in the first few milliseconds of an inhalation between an in vitro vacuum pump and lungs could explain the difference between the dose emissions. 50
Clinical Relevance of InhV
PIF and InhV measurements during inhalation by patients at home have been shown to be lower than those measured in the laboratory. 23 One factor known to impact the InhV is the quality of the patient’s exhalation immediately prior to the measured inhalation. Residual volume (RV) is the volume of air remaining in the lungs after maximum forceful expiration, whereas functional residual capacity (FRC) is the volume remaining in the lungs after a normal, passive exhalation. It has long been known that lung bioavailability of an inhaled medication is substantially greater when the patient inhales from RV to total lung capacity versus FRC to total lung capacity.51,52
RV is associated with inspiratory airflow rate, and the depth of the exhalation made by a patient before using an inhaler has been shown to be associated with InhV and flow. 53 InhV has been found to be significantly affected by the type of exhalation that a patient makes before they inhale. 54 In a study of healthy volunteers and patients with asthma, InhV was lower for inhalation, preceded by “half-way” type exhalation compared with exhalation to RV. 55 The CRITIKAL study also showed that not exhaling before an inhalation is a critical inhaler usage error. 37
Inhaler technique tends to decay 7 within weeks of training, and improvements in technique achieved through training are typically short-lived in the absence of ongoing feedback. 56 When the salbutamol/albuterol Digihaler was used for 12 weeks by 360 patients with asthma who only received inhaler technique training at the time of study enrollment but did not have access to the Digihaler app, the natural deterioration in technique during real-life use was observable. 16 Figure 3 shows the decrease in the patients’ median PIF (panel A), InhV (panel B), and Ti (panel C) over the 12-week period, as objectively recorded by the sensors built-in to the Digihaler. The decrease in median InhV (−12.6%) was greater than the decrease in median PIF (−5.1%); the decrease in Ti (−15.9%) was comparable with that observed for InhV. These observations suggest that during real-life use, patients may have begun making errors, such as not exhaling before their inhalation, not inhaling as long as possible, or a combination of both of these. The observed decrease in PIF suggests that patients may also have begun not inhaling as fast as when they were trained.
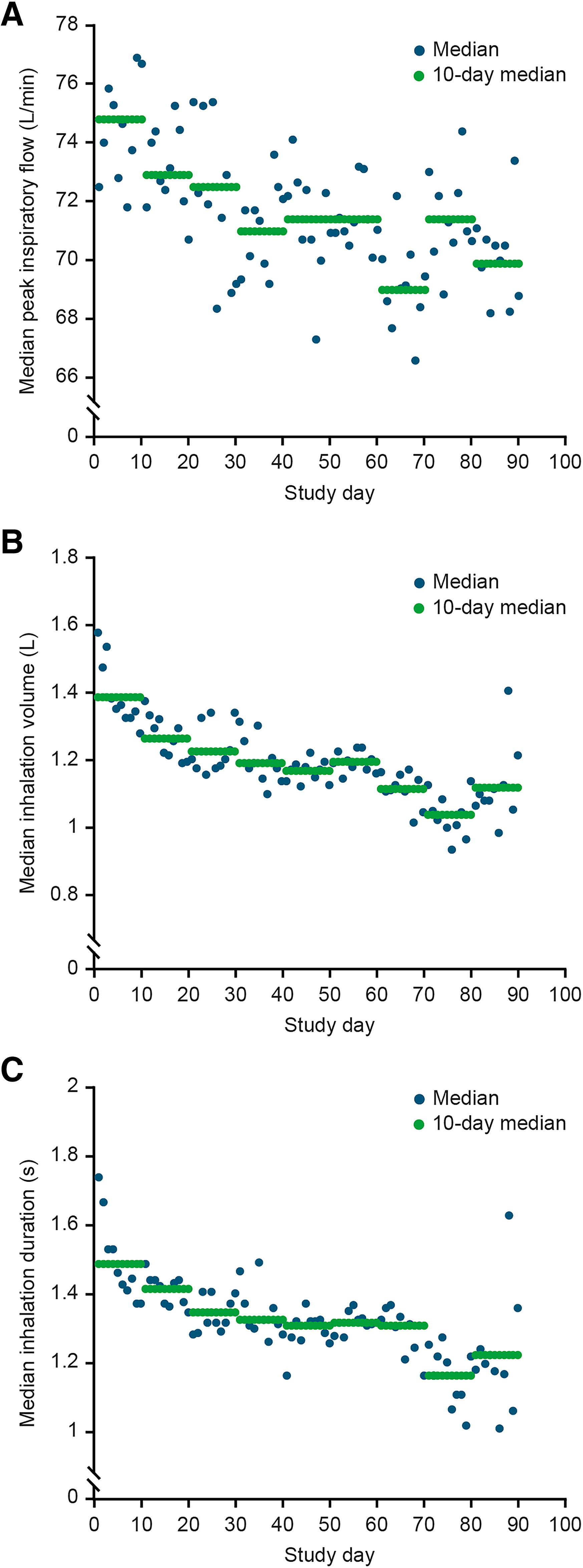
Decreases in median inhalation parameters observed during a 12-week study in which patients with asthma used the salbutamol/albuterol Digihaler® eMDPI:
In studies comparing inhalation profiles, no statistically significant difference was seen in InhV between healthy subjects and patients with asthma or COPD. 57 However, statistically significantly greater InhV measurements were recorded from patients with mild versus either severe or very severe COPD and with moderate versus very severe COPD. 57 Therefore, while InhV may lack the specificity required to serve as a diagnostic criterion, it shows promise as a potential inhaler-based digital biomarker, which could provide insight into the progress and/or status of disease.
For example, visual assessment of the inspiratory portion of the flow-volume loop can provide important insights on a patient’s muscle strength and upper airway patency. 58 Measurements of InhV may complement such assessment, with reduced InhV potentially providing evidence of intrathoracic or extrathoracic upper airway obstruction.
Associations have previously been reported between parameters of inhalation from a DPI (including InhV) and spirometric indices. In a study of healthy volunteers and patients with asthma which collected inhalation profiles from participants using a low-resistance DPI using an experimental modified spirometer setup with a flow transducer, InhV after exhalation to RV (V100%) was significantly correlated with FEV1 (p ≤ 0.003), FVC (p < 0.0001), and PEF (p ≤ 0.0001) in both participant groups. 54 InhV after exhalation partway to RV (V50%) was also found to be significantly correlated with all three spirometric parameters in both groups. 54 Consistent with this report are the findings of a multiple-regression analysis that was performed using data from 100 patients with asthma or COPD who used a Spiromax® (RespiClick® in the United States) DPI (this DPI is the nondigital version of the Digihaler) equipped with a pressure-sensing probe to determine inspiratory parameters as described previously17,59 and whose lung function was also assessed by spirometry. As shown in Figure 4, PIF from the DPI was found to be significantly and moderately correlated with PEF (r = 0.548; p = 0.006) (Fig. 4A). Furthermore, InhV was again found to be highly significantly (p < 0.001) and strongly correlated (r = 0.609) with FEV1 (Fig. 4B). 60
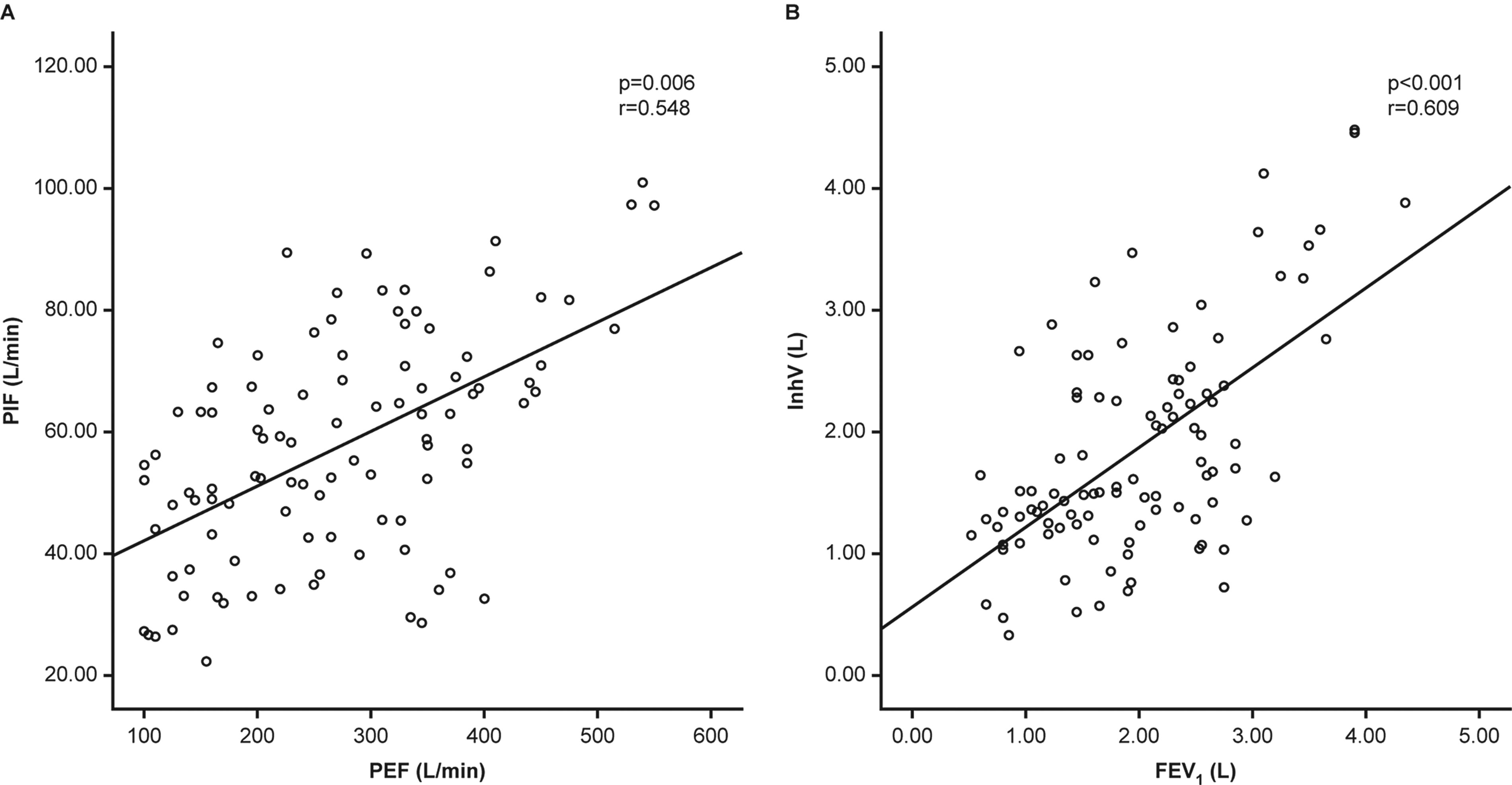
Correlations between inspiratory parameters from patients (n = 100) using the Spiromax dry powder inhaler (DPI) and spirometric parameters:
It has previously been suggested that visualization of inhalation profiles could aid in improving inhaler technique. 61 The implications of these observed associations potentially reach far beyond this, since, unlike spirometric indices, which require active effort to measure, PIF and InhV can be measured and recorded passively and on a continuous basis by a digital inhaler with integrated sensors. With suitable validation of accuracy and repeatability, these parameters might therefore fulfill the requirements to serve as inhaler-based digital biomarkers in CRD. 24
Accuracy and Repeatability of InhV Measures
In a single-visit, open-label study intended to assess the accuracy of inhalation profiles measured by the Digihaler inhaler, children (≥4 years of age) and adults with asthma, and adults with COPD, used a Digihaler inhaler connected to an IPR. The findings of this study have been reported previously 21 ; briefly, there was found to be a strong correlation between the Digihaler and IPR measurements for both PIF (Spearman’s correlation coefficient = 0.96) and InhV (Spearman’s correlation coefficient = 0.98).
A subsequent study explored the accuracy and repeatability of the InhV measurements recorded by the Digihaler eModule (integrated sensor) across a range of flow rates and inhalation durations reflecting use of standard flow (SF) and high flow (HF) DPIs. The accuracy of the InhV data recorded by the eModule was assessed through the use of a calibrated flowmeter, which applied a prespecified flow rate for a specified duration. Additionally, the InhV data displayed on the DS’s HCP-facing Dashboard were assessed and determined to be consistent with those recorded by the eModule (data not shown). 62 The accuracy findings are shown in Figure 5 62 ; and repeatability findings are shown in Table 2. 62 The mean (95% confidence interval) differences between the actual and eModule-recorded values were −0.011 L (−0.020; −0.003) and 0.038 L (0.030; 0.046) for SF and HF rates, respectively. Relative standard deviations ranged from 0.4% to 4.7% and from 0.5% to 4.2%, respectively. It was therefore demonstrated that the Digihaler eModule provides accurate and highly repeatable InhV measurements.
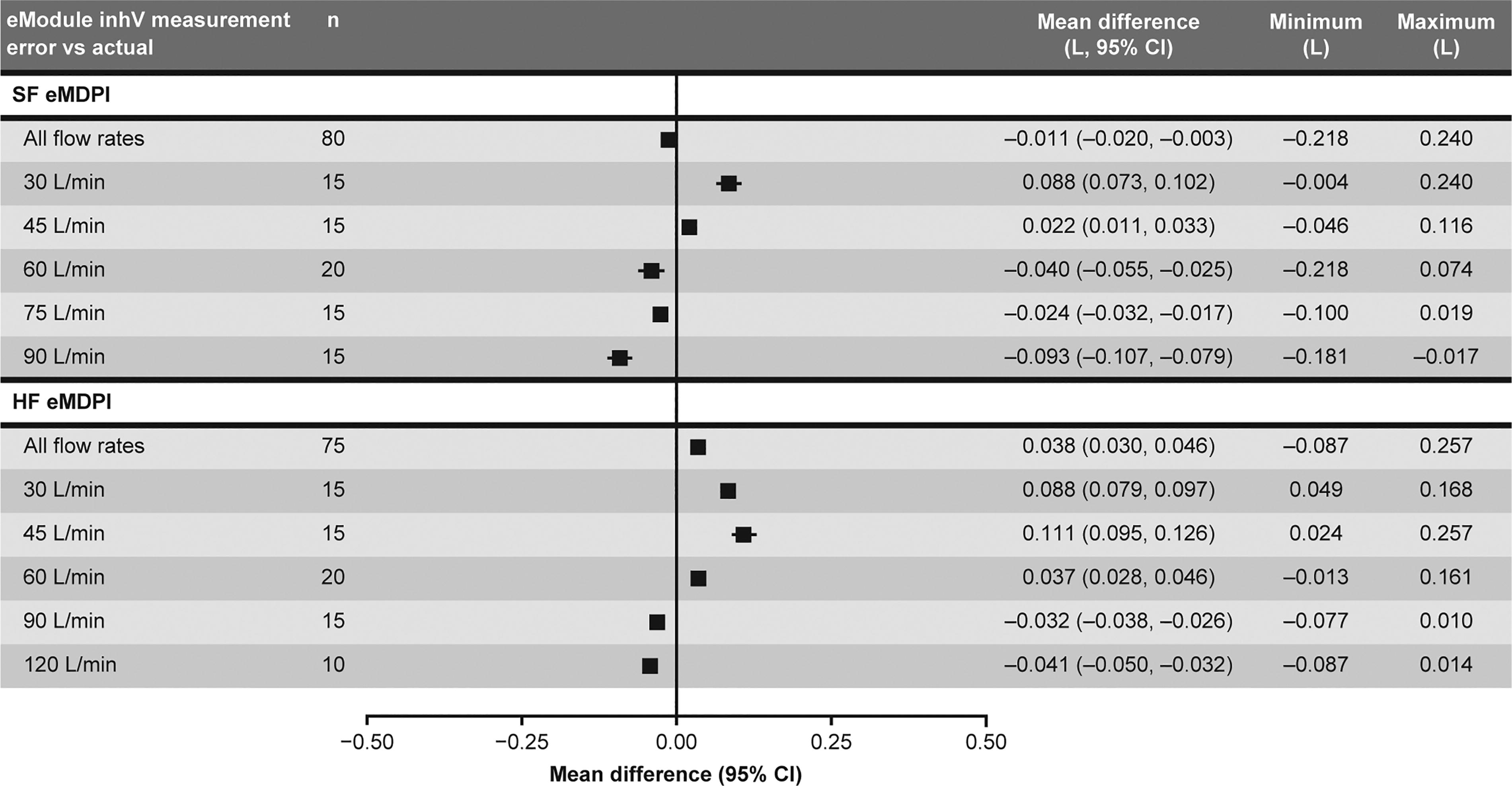
Accuracy of InhV measurements recorded by the Digihaler® eModule for a range of flow rates in standard flow (SF) and high flow (HF) eMDPIs. 62 Data from applied volumes <0.5 L and duration <0.5 seconds have been excluded from the analysis. eMDPI, electronic multidose dry powder inhaler; InhV, inhaled volume. Figure originally presented in a poster at the International Society for Aerosols in Medicine congress 2023. Reproduced with permission from Chrystyn H, Forrestal J, Yang D, et al. Accuracy and repeatability of inhalation volume data recorded by standard and high flow electronic multidose dry powder inhalers: An in vitro study. J Aerosol Med Pulmon Drug Deliv 2023;36:G.03.
Repeatability of Inhaled Volume Measurements Recorded by the Digihaler® eModule for a Range of Flow Rates in Standard Flow and High Flow Electronic Multidose Dry Powder Inhalers 62
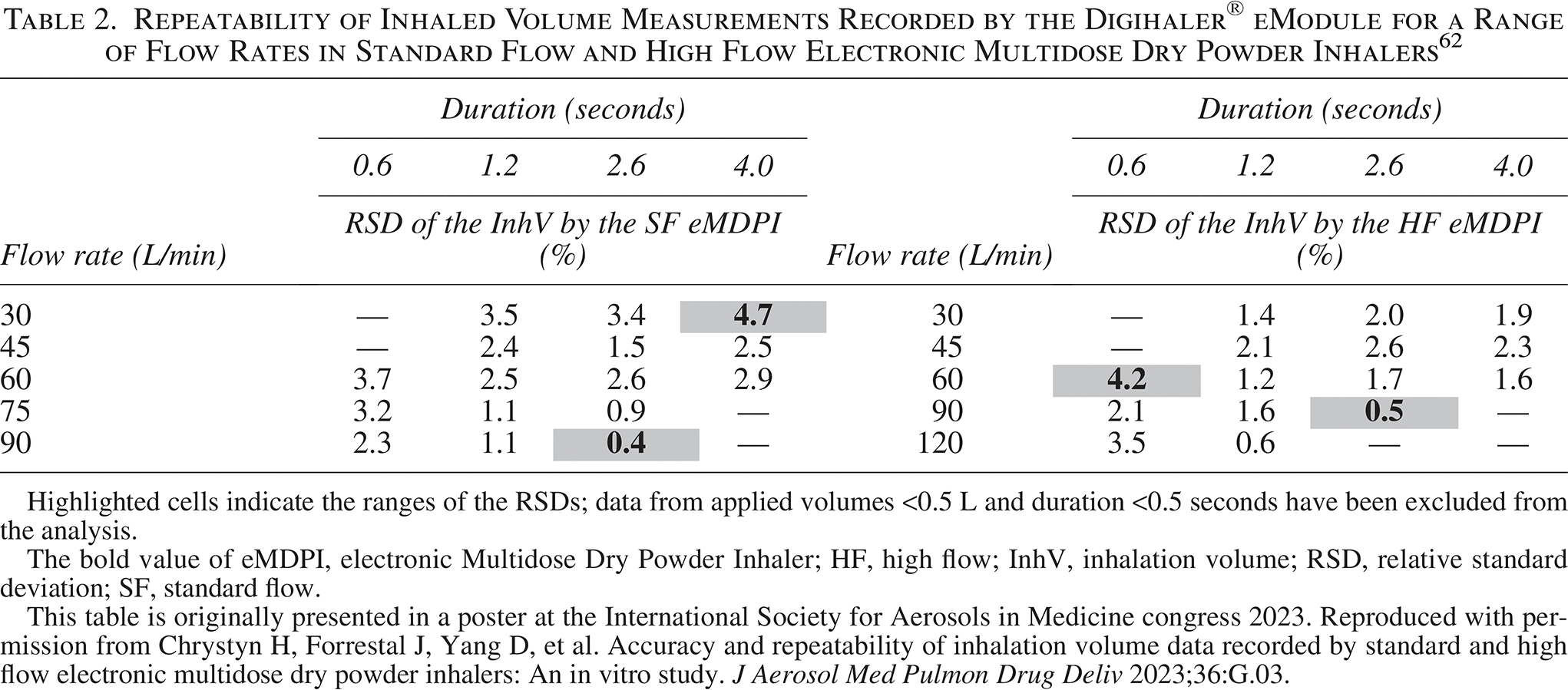
Highlighted cells indicate the ranges of the RSDs; data from applied volumes <0.5 L and duration <0.5 seconds have been excluded from the analysis.
The bold value of eMDPI, electronic Multidose Dry Powder Inhaler; HF, high flow; InhV, inhalation volume; RSD, relative standard deviation; SF, standard flow.
This table is originally presented in a poster at the International Society for Aerosols in Medicine congress 2023. Reproduced with permission from Chrystyn H, Forrestal J, Yang D, et al. Accuracy and repeatability of inhalation volume data recorded by standard and high flow electronic multidose dry powder inhalers: An in vitro study. J Aerosol Med Pulmon Drug Deliv 2023;36:G.03.
Predictive Value of InhV Measures
An analysis of adults with asthma (n = 18) and COPD (n = 7) who were admitted with an acute exacerbation was conducted. In addition to their traditional medication to manage their acute exacerbation, they inhaled through an empty Spiromax DPI equipped with an IPR, as well as undertaking spirometry during their recovery. Quantifiable improvements in PIF and InhV, as well as PEF and FEV1, were observed over the course of 3 days of recovery (Fig. 6). 60 Among the 18 patients with asthma, the percentage of improvement in mean InhV from Day 1 to Day 3 was 48.2%, compared with 29.2% for the FEV1, which may be due to inhalation against a resistance. It might therefore be deduced that similarly measurable deteriorations in these parameters may occur during the early phase of an exacerbation, giving rise to the potential for early intervention.
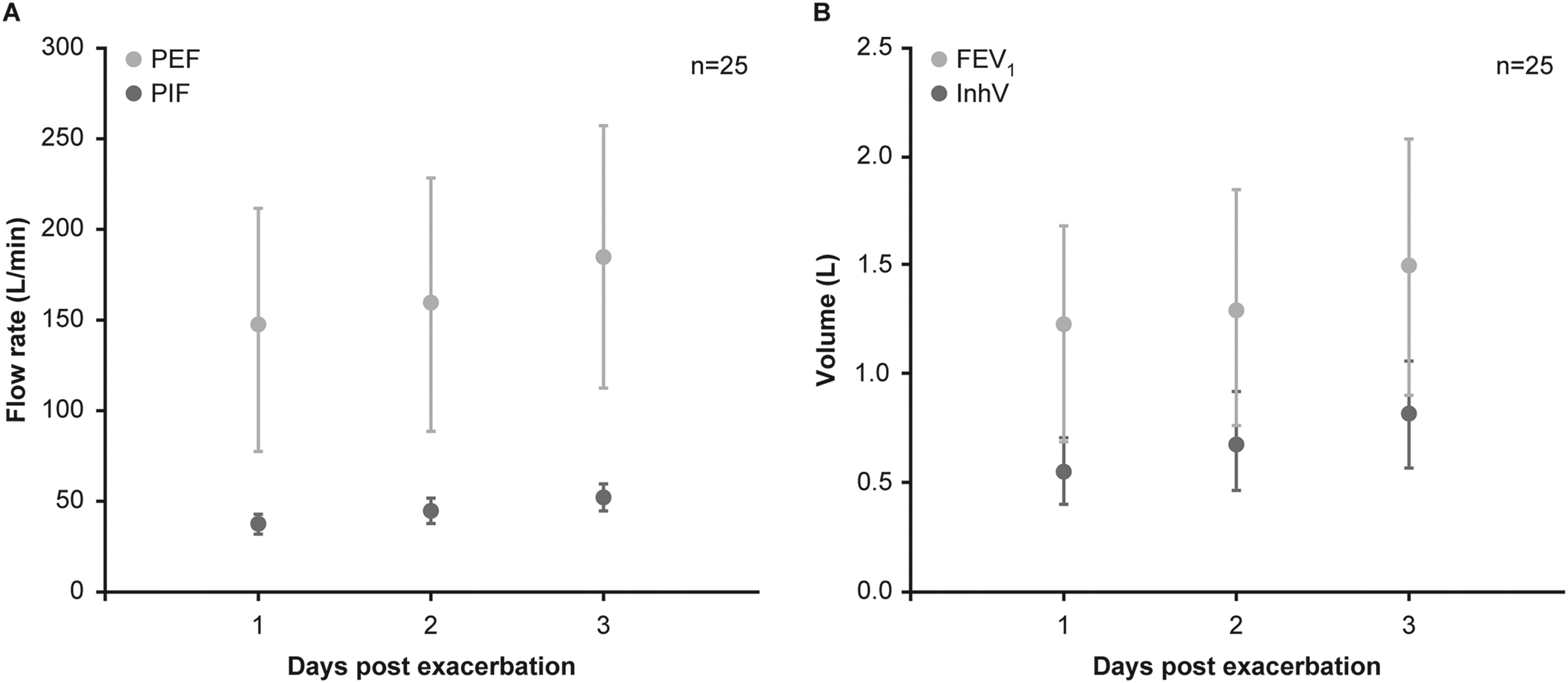
Mean (SD) inspiratory and spirometric parameters measured during the 3 days subsequent to an acute exacerbation in patients with asthma (n = 18) and COPD (n = 7):
The potential for digitally recorded inhalation parameters and inhaler usage data to predict exacerbations of CRD has been explored. Predictive models have been developed using machine learning techniques on data from usage of the salbutamol/albuterol Digihaler by patients with COPD 63 and asthma 64 participating in open-label studies. In these studies, exacerbation of COPD or asthma was defined according to applicable guidelines and involved worsening of symptoms with a requirement for systemic corticosteroid and/or antibiotic treatment together with an unscheduled health care event up to and including hospitalization.
In a study of 336 patients with COPD who used the salbutamol/albuterol Digihaler, the predictive model was able to predict COPD exacerbations within 5 days with overall accuracy of 0.77 receiver operating characteristic area under the curve (ROC-AUC) with sensitivity of 0.65 and specificity of 0.81 (Fig. 7A), 63 while in an analogous study of 360 patients with asthma, the predictive model was able to predict asthma exacerbations within 5 days with overall accuracy of 0.83 ROC-AUC with sensitivity of 0.69 and specificity of 0.89 (Fig. 7B). 65 In COPD, the inhalation parameters (including InhV) contributed to almost 80% of the accuracy of the model. In asthma, although the frequency of inhaler usage was found to be the most significant model feature, inhalation parameters contributed ∼40% to the model’s accuracy.63,64
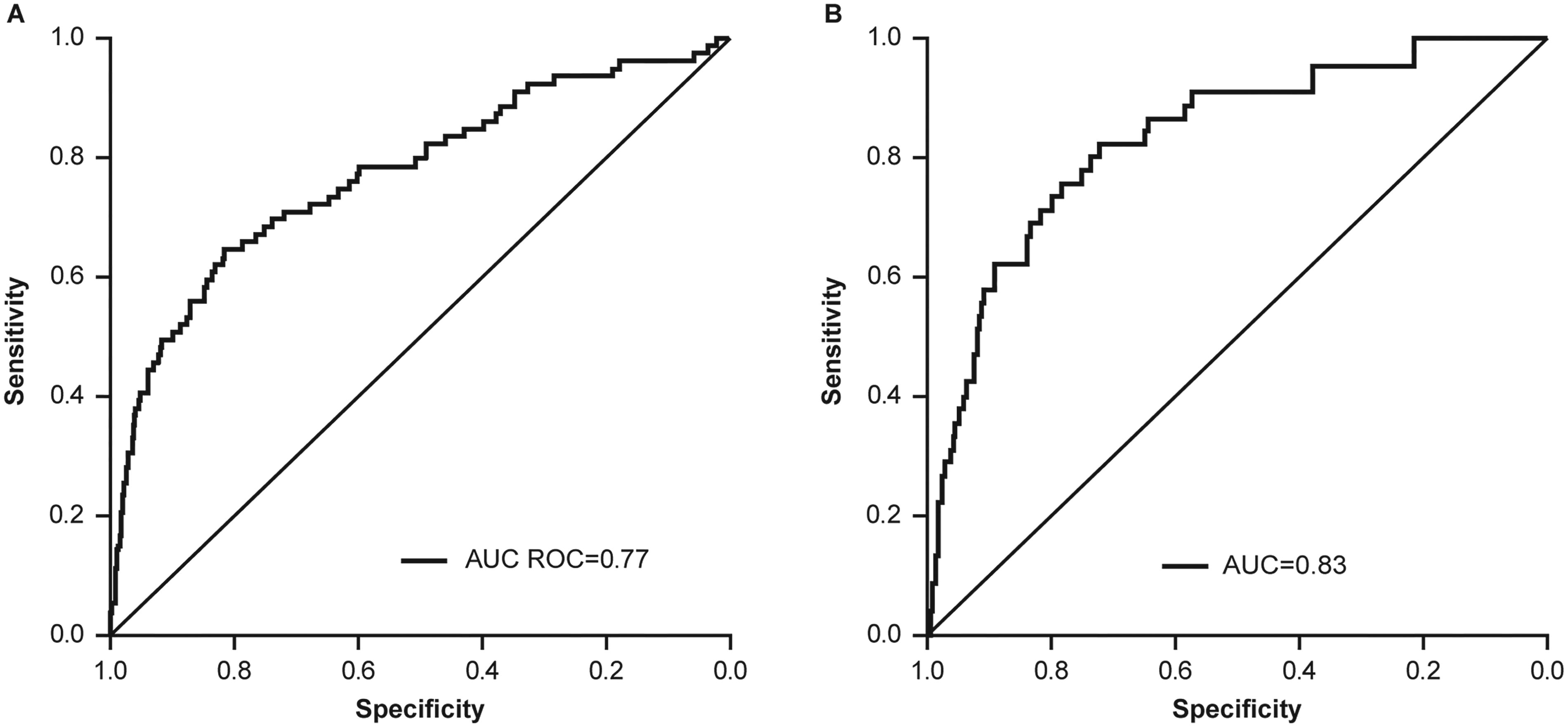
Accuracy of machine learning models informed by inhalation parameters, inhaler usage, and patient baseline characteristics in predicting
Across all patients in each of these studies who experienced an exacerbation, analysis of mean inhalation parameter values demonstrated a clear pattern of decrease in both InhV and PIF in the period preceding an exacerbation, followed by rapid return to baseline following treatment of the exacerbation (Fig. 8).64,66 Among the 98 patients with COPD who experienced exacerbations during the study, changes in InhV preceding and following the exacerbation were notably more marked than corresponding changes in PIF (Fig. 8A); among the 64 patients with exacerbation of asthma, similar changes were seen in each of these parameters (Fig. 8B).
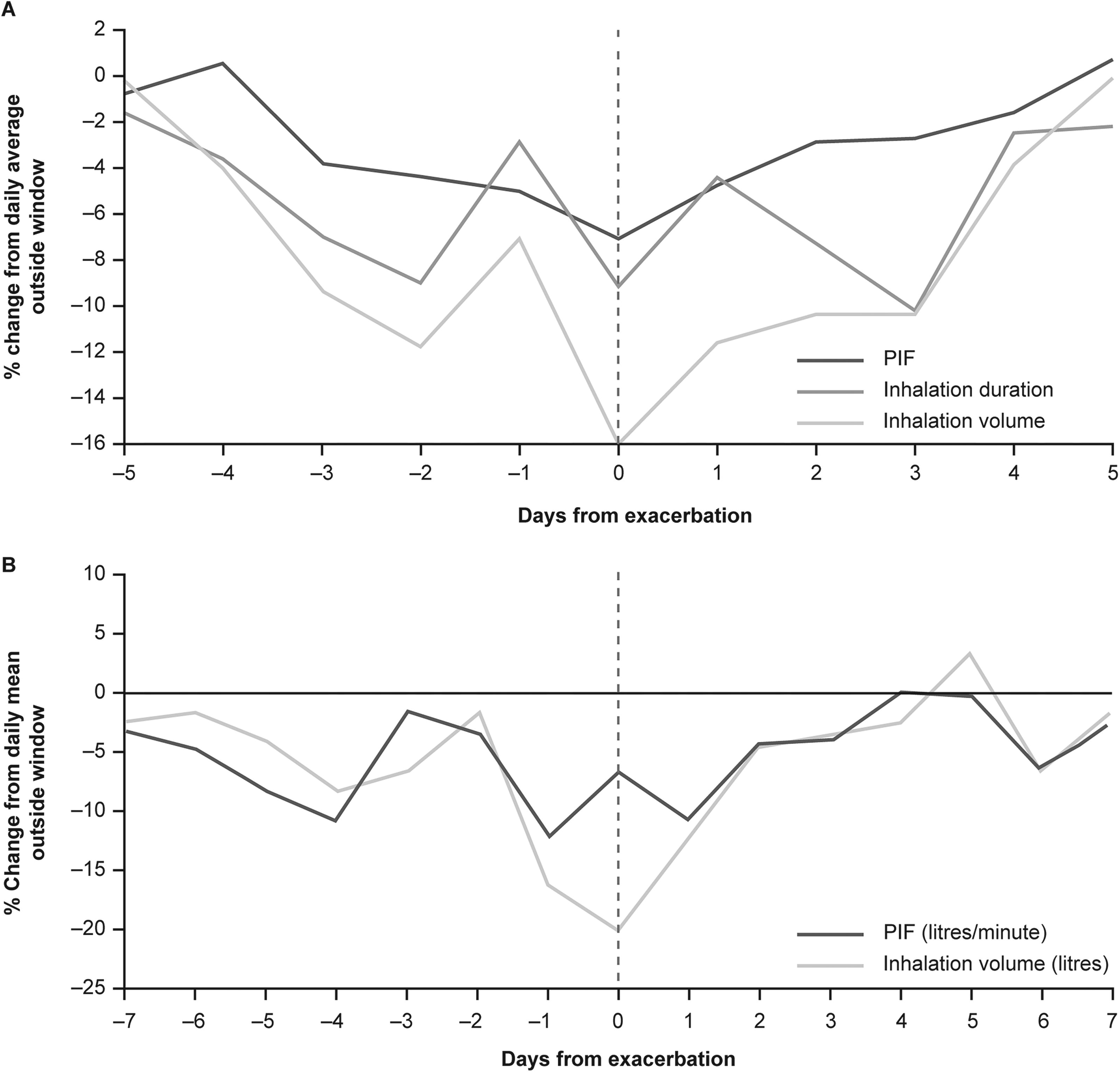
Mean InhV and PIF during lead-up to and recovery from exacerbations in
These findings have recently been supported by those of a 3-month observational study using the salbutamol/albuterol Digihaler in patients with COPD at elevated risk for exacerbations. In an interim analysis of data from 10 patients who experienced a total of 17 exacerbations, an overall decrease in mean InhV over the 14 days preceding the exacerbation of 24.4% was found. This decrease was observed to be progressive over the course of the 14 days (Fig. 9) 67 and appeared to be more sensitive for the prediction of exacerbation than PIF (for which only an 11.1% decrease over the same period was observed). Furthermore, in a separate pilot study in 20 patients using the salbutamol/albuterol Digihaler, exacerbations of COPD were found to be preceded by a ≥20% reduction in recorded InhV from patient baseline in the prior 14 days in 65% of instances; other inhalation parameters were not found to be as predictive of forthcoming exacerbations. 68
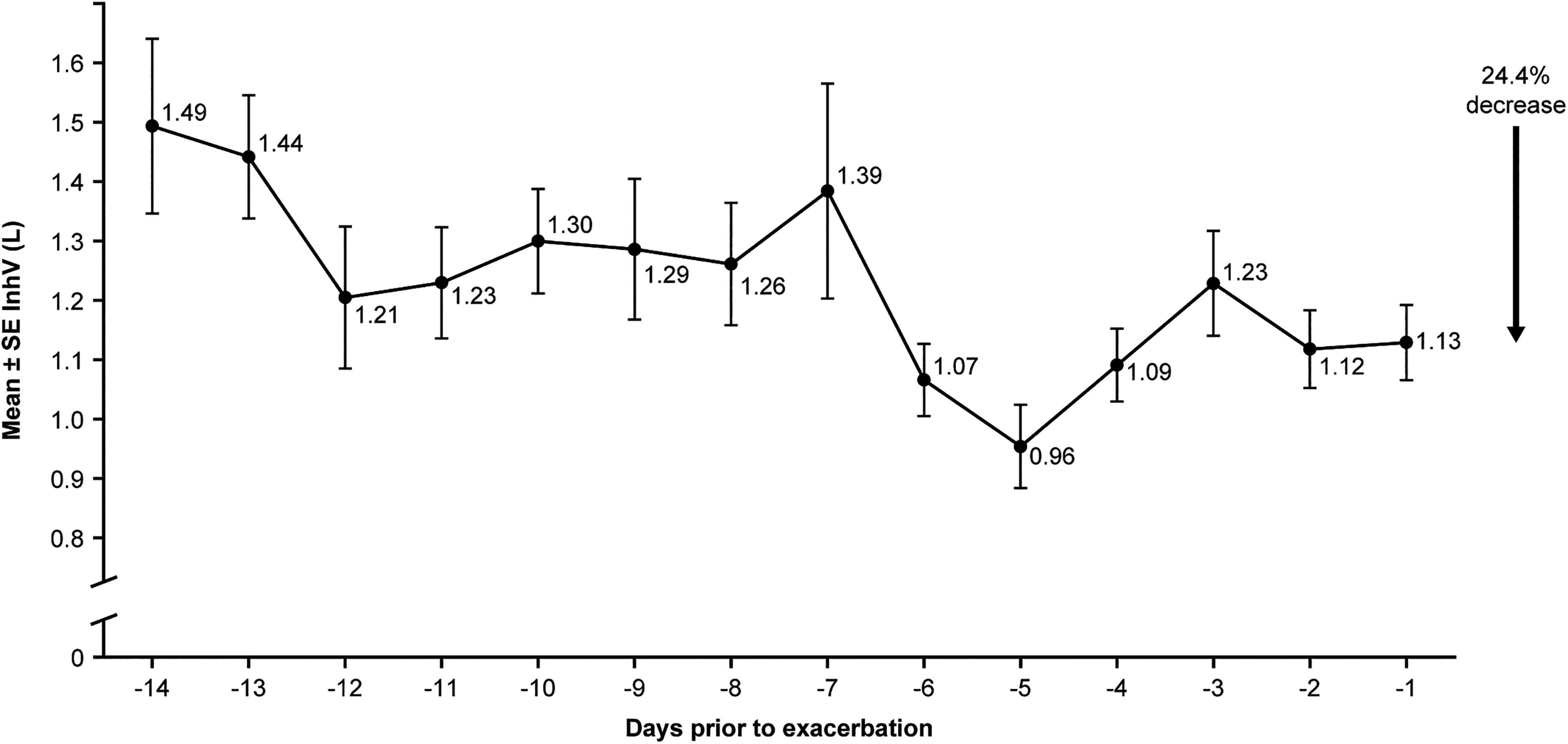
Decrease in daily mean InhV in the 14 days prior to an exacerbation observed in patients with COPD (n = 10). 67 Reproduced with permission from Ohar JA, Wang T, Hill T, et al. COPD, chronic obstructive pulmonary disease; InhV, inhaled volume. Objectively recorded peak inspiratory flow (PIF), inhalation volume and reliever medication use preceding COPD exacerbations. Am J Respir Crit Care Med 2023;207:A4495.
A separate study was conducted in patients with severe asthma who were eligible for treatment with biologics and whose asthma control was assessed using the Asthma Control Test (ACT). Patients with uncontrolled (ACT score <19) and controlled (ACT score ≥19) asthma used fluticasone/salmeterol (maintenance) and salbutamol/albuterol (reliever) Digihaler devices. Those with poorly controlled asthma were found in an interim analysis to have significantly (p < 0.001) lower mean InhV through both the maintenance and reliever inhalers, compared with well-controlled patients (Fig. 10A, B). 69 While significant differences between the two groups in PIF were also observed, the direction of these differences was inconsistent across the two devices (Fig. 10C, D). 69 This finding suggests that InhV may be of significant value, with greater reliability than PIF, for monitoring asthma control status.
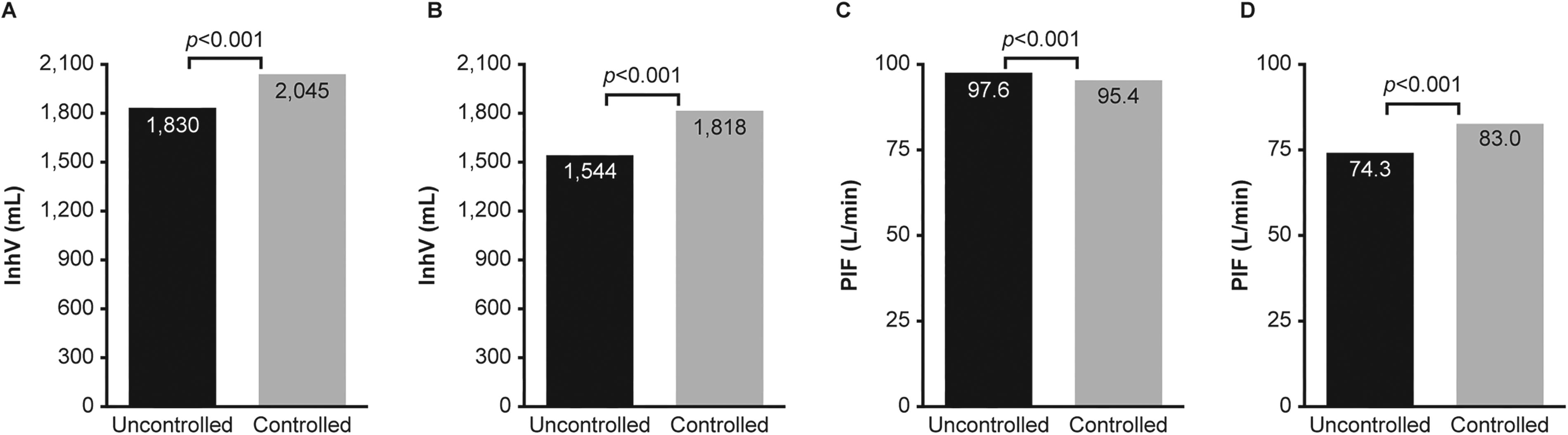
Mean inhalation parameters in patients with asthma defined as uncontrolled (ACT score <19; n = 52) or controlled (ACT score ≥19; n = 16) at baseline:
Future Outlook
Current best practice recommendations in both asthma and COPD emphasize the importance of individualization of management strategies and treatment goals, and of supporting patients with self-management of these chronic conditions through education, training, and personalized action plans.4,5 However, in contrast to other chronic conditions such as diabetes, the lack of reliable and objective data on the status and progression of these conditions is even now greatly hindering efforts to optimize treatment and improve self-management. 14 In numerous studies over recent years, digital tools have demonstrated benefits with respect to medication adherence and inhalation technique in patients with asthma and COPD, with associated cost savings 33 and potential to bring about substantial improvements in patient quality of life.13,70
Telemonitoring has already been shown to benefit disease control, treatment adherence, and quality of life in respiratory allergy care,71,72 and the potential for insights provided by artificial intelligence and machine learning to support the personalization of treatment plans has been recognized. 71 The value of such innovative approaches is, however, only as great as the quality of the inputs and the appropriateness of the interpretation of the outputs.
Together, the findings described here demonstrate that measurement of inspiratory parameters (specifically, InhV and PIF) by a digital inhaler with integrated sensors gives rise to a biomarker with great utility for the day-to-day management of CRD. These parameters are analogous to the spirometric measures that are central to the diagnosis and monitoring of asthma and COPD. Unlike spirometric measures, however, these inhalation parameters can be recorded passively and on an ongoing basis through the patient’s ordinary use of a digitally enabled inhaler with validated accuracy and repeatability of recorded measurements. They therefore fulfill the necessary criteria to serve as a digital biomarker.
Furthermore, the observation of changes in the values of these parameters from an individual patient’s baseline has been recognized as reflecting clinically meaningful changes in symptom status, including the progression toward exacerbations, with detectable changes in InhV being apparent from up to 14 days prior to the exacerbation in COPD. The use of data on these parameters, together with patient baseline information in population-based machine learning models to interpret individual patient data, has demonstrated potential to predict acute exacerbations of both asthma and COPD.63,64 InhV data may also be of value in identifying poor asthma control in an objective manner. 69
Importantly, the InhV parameter, which reflects the patient’s total air intake during an inspiratory maneuver, is critical to these models, in addition to being highly correlated with FEV1 and FVC. The value of InhV with respect to understanding CRD has largely been overlooked to date.
In conclusion, therefore, InhV measured in this manner has the potential to play a core role in understanding patients’ respiratory health and disease status now and in the future.
SUMMARY
Inhaler-based digital biomarkers are measurable characteristics that can provide insights into normal or pathophysiological processes, such as glycemia in diabetes or airflow in CRD. Lack of accurate, objective disease state data is a major barrier to effective personalized care and self-management in CRD. Exhalation parameters measured by spirometry, such as FEV1, are central to the diagnosis and management of both asthma and COPD; however, these are typically only recorded at occasional clinic visits and are impractical for patients to self-monitor on an ongoing basis. PEF can be recorded by patients at home, but it is a poor substitute for spirometry and is subject to significant adherence and data quality issues. In contrast to spirometry and peak flow measures, inhalation parameters can be passively recorded by a digital inhaler equipped with integrated sensors, without requiring active effort on the part of the patient beyond ordinary usage of their maintenance and/or reliever inhaler. These parameters include InhV and PIF. The accuracy and repeatability of the inhalation parameter measurements of one such digital device, the FDA and MHRA-approved DS, have been robustly validated. Analyses of inhalation parameter data, recorded using the Digihaler inhaler from patients with asthma and COPD who experienced clinical exacerbations, have shown that InhV typically decreases over the days preceding the exacerbation and increases again afterward. Furthermore, patients with poorly controlled asthma have been observed to have lower mean InhV than those with well-controlled disease. InhV appears to be more sensitive than PIF for the purpose of these analyses. Changes in InhV (and PIF) post-inhaler technique training occur during real-life use. The connected “app” will be able to advise/remind the patient about their technique, thereby saving clinic time having to retrain and help maintain good control of their lung disease. High-quality InhV data, passively recorded by a digital inhaler, therefore fulfill the requirements to serve as a digital biomarker for monitoring the asthma and COPD disease states and may have predictive value with respect to acute exacerbations of these CRDs.
Authors’ Contributions
H.C.: Conceptualization, methodology, validation, formal analysis, resources, investigation, visualization, supervision, and writing—reviewing and editing. M.M.-E.: Conceptualization, methodology, validation, formal analysis, resources, investigation, visualization, project administration, funding, and writing—original draft preparation.
Footnotes
Author Disclosure Statement
H.C. has no relevant competing interests to disclose. M.M.-E. is a former employee of Teva Pharmaceuticals and the founder and CSO of Digital Medical Innovations Ltd.
Funding Information
Medical writing and editorial support for the development of this article, under the direction of the authors, was provided by Ian C. Grieve, PhD, Jane Blackburn, PhD, and Sherriden Beard, MA, of Ashfield MedComms, an Inizio company, and was funded by Teva Branded Pharmaceutical Products R&D Inc.
