Abstract

Introduction
Synthetic biology—the design and construction of new biological parts, devices, and systems, and the redesign of natural biological systems for useful purposes—opens the richness of biological diversity to solve pressing, real-world problems for humanity and the planet: precisely targeted, personalized medicines; energy-rich molecules for sustainable fuels and industrial chemicals; remediation of polluted environments; and food supplies to meet an explosively growing global population. 1 Still, we are far from capitalizing on synthetic biology's fullest potential—a promise that also poses great ethical questions and risks. This places a critical onus on researchers, policymakers, regulatory bodies, industry, and all citizen stakeholders to engage in an intelligent dialogue about how to advance synthetic biology not only to sustain but also to enhance life on Earth.
With an expected global market of $10.8 billion by 2016, synthetic biology will play an important role in the bioeconomy with powerful implications for future US competitiveness and employment. 2 The US currently leads the world in intellectual conception, research, and commercial development in this field. 2,3 But in both industry and research in the US and worldwide, synthetic biology is hampered by a lack of standardized fundamental tools, inadequate regulatory policy, a need for both substantial public and private investment, and lack of education of stakeholders about its risks and benefits.
This Industry Report includes key findings and excerpts from the report “Synberc Sustainability Initiative: Initial Findings & Recommendations,” prepared by Nancy J. Kelley & Associates, February 2014, co-funded by the Alfred P. Sloan Foundation and Synberc. The full report is available at
This report reviews the development of synthetic biology from a historical perspective, within a global landscape of regulatory frameworks, funding initiatives, and social and ethical aspects. The full version of this report was prepared as part of a 1-year independent initiative funded by the Alfred P. Sloan Foundation and Synberc. 4 Much foundational research is yet needed, as well as supportive policy and sufficient financial investment, if the US is to maintain and grow its leadership position in this field it has pioneered.
Synbio: The First 15 Years
While research in synbio and adjacent fields has been underway for decades, the field emerged on its own around the turn of the century with a few seminal events, including Blue Heron's (Bothell, WA) and GeneArt's (Grand Island, NY) launch of the gene-synthesis industry (1999), Jim Collins' Nature paper on bacterial toggle switches (2000), the initial sequencing of the human genome (2001), and the introduction of the BioBricks concept by Tom Knight (2003). Since then, the pace of development has rapidly accelerated, with a cascade of research, funding, and corporate activity. This is obvious from the publishing volume over this period: in 2003, fewer than ten articles in the scientific literature related to synthetic biology; by 2011, that number had grown to approximately 350, including articles, reviews, proceedings, etc. (Fig. 1).
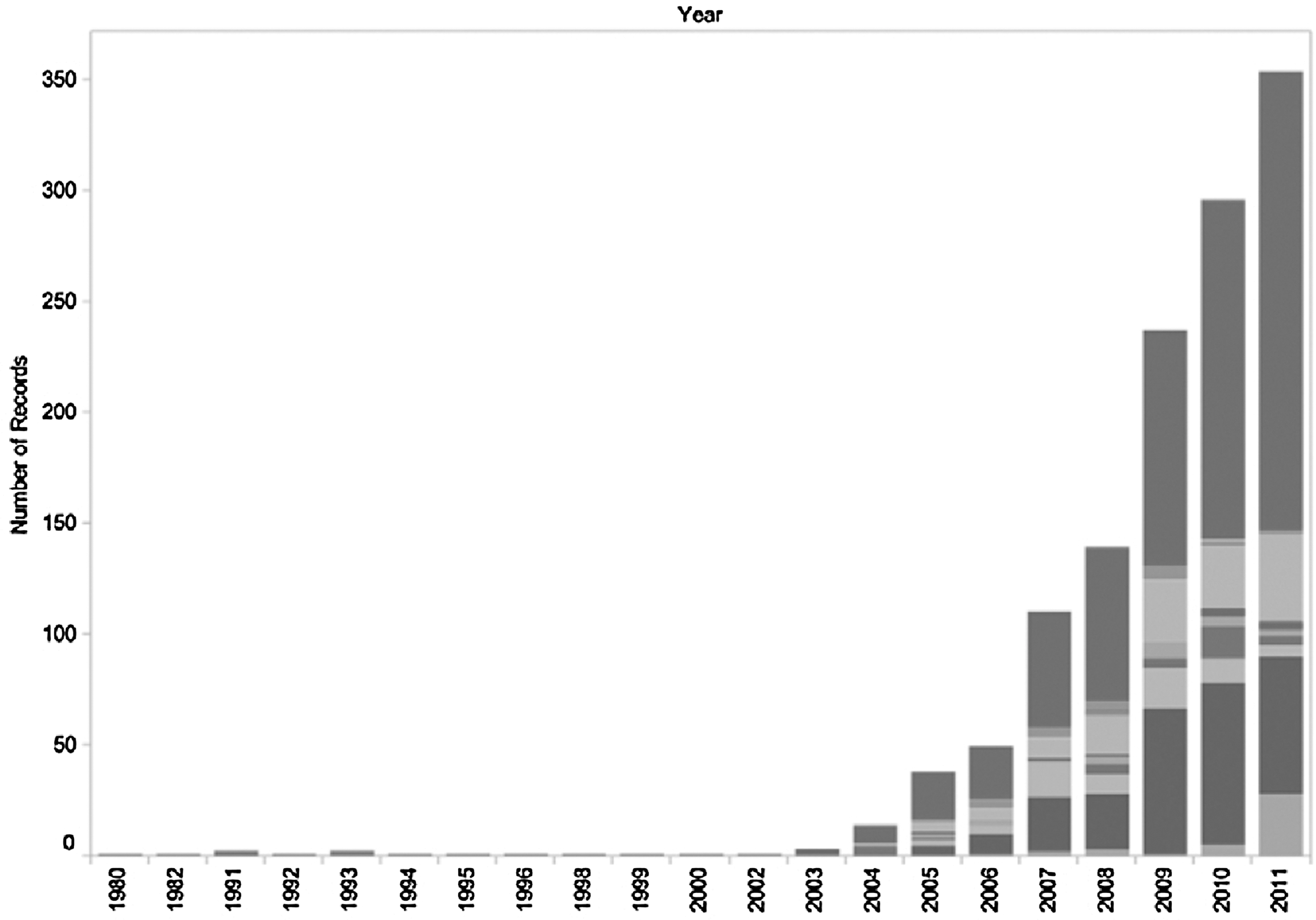
Growth in synthetic biology research: Web of Science listings for “Synthetic Biology.” (Reprinted with permission from Oldham P, Hall S, Burton G. Synthetic biology: Mapping the scientific landscape. PLoS One 2012;7:e34368)
The majority of these publications have stemmed from US research, but the field is growing rapidly elsewhere. Recent UK investments should lead to even greater outputs, and European efforts are gaining momentum. China, which intends to become a significant synbio player within 10 years, now contributes 10% of all synthetic biology papers published. 5,6 The timeline in Figure 2 highlights developments in synbio over the past 15 years.
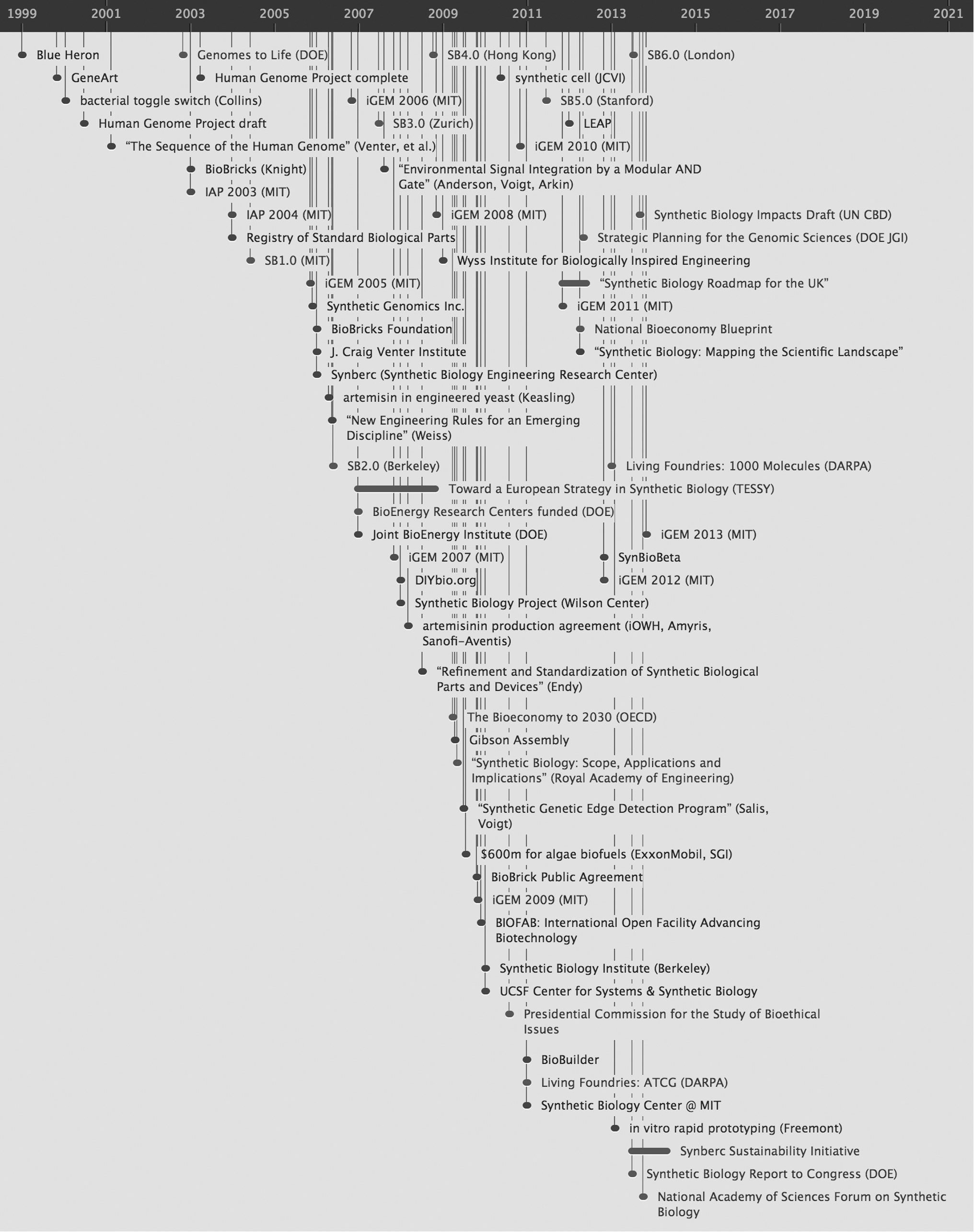
In 2002, the US Department of Energy (DOE) invested $12 million in Craig Venter's research as part of its Genomes to Life Program, followed by the Department of Defense's (DOD) Defense Advanced Research Projects Agency (DARPA) investigation into the intersection of computing and biology. US investment into synbio has totaled at least $500 million (by NJK&A estimates, up to $1 billion), spanning multiple agencies and supporting numerous research efforts and institutions.
9
The largest single investment to date has been National Science Foundation's (NSF's) support of the Synthetic Biology Research Engineering Center (Synberc,
Several institutional affiliates of Synberc have developed their own synbio-related programs and centers over the past decade, with other US schools following suit. Meanwhile, numerous multinational corporations—including Agilent Technologies (Santa Clara, CA), Life Technologies (now Thermo Fisher Scientific, Waltham, MA), BP (London, UK), Dow Chemical (Midland, MI), ExxonMobil (Irving, TX), and Goodyear Tire and Rubber (Akron, OH), pharmaceutical companies like GlaxoSmithKline (London), Novartis (Basel, Switzerland), Merck (White House Station, NJ), Pfizer (New York, NY), Roche (Basel), and Sanofi-Aventis (Paris, France), and consumer products companies such as L'Oréal (Paris, France) and PepsiCo (Purchase, NY)—are integrating synbio research into their R&D processes. A number of start-ups are emerging from synbio-related research activities and beginning to make their mark in the critically important areas of health, energy, environment, and food: e.g., Ginkgo BioWorks in Boston (engineered organisms), San Diego, CA-based Genomatica (sustainable chemicals), and Amyris in Emeryville, CA (energy and pharmaceuticals).
With research moving forward quickly and industry capitalizing on these developments, governmental regulatory bodies and funding agencies are struggling to keep up. The patchwork of US agencies and regulations can be confusing to practitioners and regulators alike. Furthermore, while both the US and the EU have established regulations to address synthetic biology, their approaches have not always aligned, and there is no agreement about what exactly is being regulated and what constitutes the boundaries of “synthetic biology.” The global community has mobilized around a few key initiatives, but the US has remained conspicuously absent from these discussions, choosing not to sign on. It is imperative that the synbio community continue its work with the US government to refine its regulatory strategy, both domestically and globally.
Considering the lack of clarity at the governmental level, it is not surprising that public perception toward synthetic biology is mixed. Most Americans (roughly two-thirds) are in favor of synbio research, a view shared by the UK public. 10,11 However, many are unfamiliar with exactly what synthetic biology entails, and their concerns only increase as they become more acquainted with the concept. 10 Questions about genetically modified organisms (GMOs) and environmental and biosafety risks are prevalent. Better communication and education are required to support the growth of synbio research and products, especially those with a direct consumer connection (e.g., pharmaceuticals, food).
Synberc has spearheaded numerous communication and educational initiatives as well as fundamental advances in synthetic biology. Synberc's principal investigators have been critical to creating a formative body of research, establishing synbio as an important and viable effort oriented to the public good. Beyond this, the organization has created innovative educational programs to train future scientists and leaders. The Synberc community now constitutes a national cadre of interdisciplinary researchers within a focused, collegial community. It has also established the foundation of a unique industrial-academic collaboration.
Synbio achievements to date—from the International Genetically Engineered Machines (iGEM) competition, to BioBricks, to registries like iGEM's Registry of Standard Biological Parts and the Joint BioEnergy Institute Inventory of Composable Elements—are inextricably linked to Synberc's mandate and activities. This highlights the pivotal role of the US in the overall global success of synbio going forward and the imperative for a strong central organizing force that will continue to support its growth (both downstream and upstream markets), and mobilize researchers, industry, government, and philanthropy.
Market Overview
Synthetic biology as an academic and research discipline is not yet 15 years old, and as an industry, even younger. Core technologies are in development, as foundational tools have yet to mature; applications are still emerging as markets ponder how best to profit from the vast potential; and synthetic biology's products and applications have only begun to penetrate the many markets it stands to disrupt. Regardless, a significant global synthetic biology market, however immature, has established itself across multiple fields. The sheer number and diversity of companies in the synbio space is telling. From 2009 to 2013, the estimated number of companies engaged in synbio worldwide more than tripled, from 61 to 192 (Fig. 3). 12 Two-thirds of these companies are in the US, where the number of synbio commercial entities grew from 54 to 131 in the same 2009–2013 period; at least 43 European companies are engaged in synbio, and 13 in Asia/Oceania. All regions are expected to see increases in the size of synbio-enabled product markets through 2016, with North America continuing to lead the industry. 2
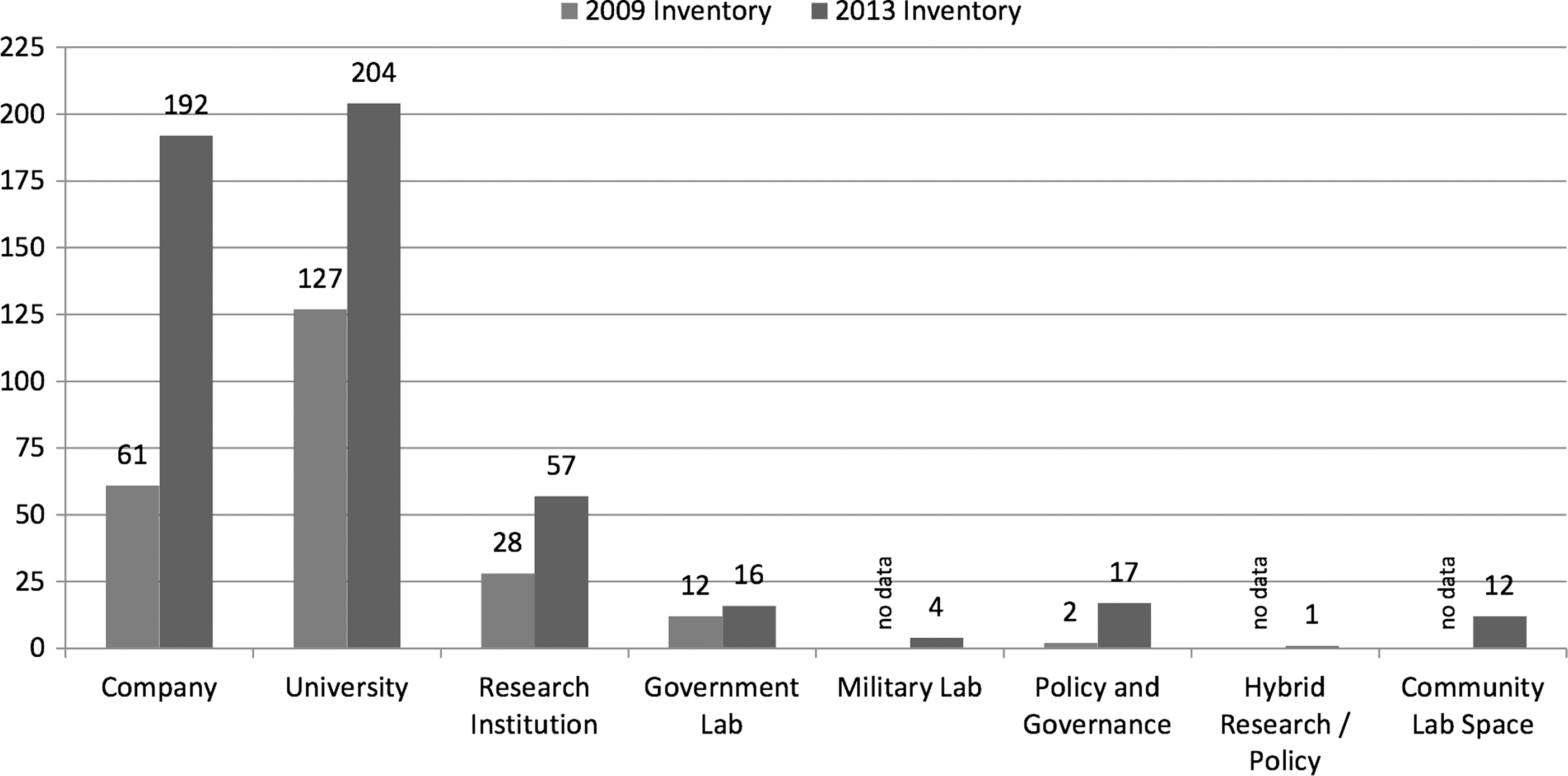
Entities conducting research in synthetic biology worldwide. 12
There is a dearth of directly relevant market research on synthetic biology. NJK&A surveyed available recent analyses and found that few comprehensively describe the synbio landscape; further, each seems to include market characterizations that differ from each other and our observations. 2,13,14 Despite understandable challenges in sizing industrial biotechnology markets, there is a clear need for more-incisive market studies. 15 Nonetheless, some trends are evident.
The global value of the synthetic biology market could grow from $1.15 billion in 2010 to $10.84 billion in 2016. 2,15 Rob Carlson recently calculated the size of the US bioeconomy—the “Genetically Modified Domestic Product (GMDP)” and a superset of BCC Research's synthetic biology market—as $350 billion in 2012, dwarfing these numbers by orders of magnitude. His data are drawn from a number of sources and divide the bioeconomy into three market sub-sectors: biotech drugs (biologics), genetically modified crops, and industrial biotechnology (fuels, enzymes, and materials), with particular attention paid to the value of end-user markets. 15 )
This market can be divided into three segments, each predicted to demonstrate sizable growth through the next decade: Enabling Technologies (basic technologies on which synbio relies); Core Technologies (those that define synbio endeavors); and Enabled Products (products, services, or platforms resulting from or incorporating the end results of synbio processes, or that include synbio components). 2 These segments constitute a simple value chain (Fig. 4).
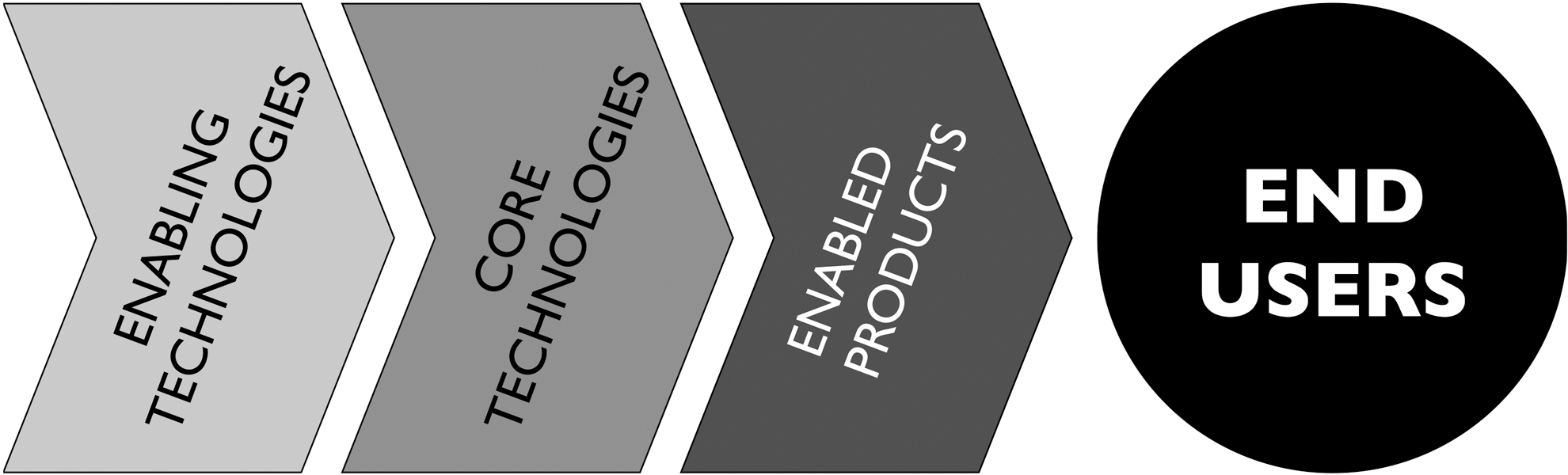
Synthetic biology product value chain.
Enabling and Core Technologies
Enabling Technologies comprise tools and platforms necessary for, but not specific to, synbio (e.g., DNA synthesis, sequencing, specialty media, bioinformatics, data management tools); their importance is in their enablement of downstream production. Marked reductions in cost for certain enabling technologies (specifically synthesis and sequencing), together with rapid increases in the amount of genomic information, are vastly increasing possibilities for industrial-scale synbio systems, expanding the potential for market growth in the other segments. 2,16 Sequencing of the human genome, for example, cost $2.7 billion and took 13 years in the first groundbreaking endeavor; some believe that within the next decade sequencing a whole genome will cost $100 and take 1 hour. 16 Core Technologies, including biological components (integrated systems, enzymes, DNA parts, metabolic engineering platforms), constitute a pivotal market segment. Investment in this area will catalyze development of more end-user products, and it is here that ongoing and future research efforts will have the most impact.
Enabled Products
Enabled Products are the dominant synthetic biology segment, valued at $1.4 billion in 2011, and anticipated to grow to as much as $4.5 billion by 2015, and $9.5 billion in 2016. 2,17 Enabled Products can be further subdivided into several primary markets, discussed below.
Chemicals
Chemicals dominate the synbio industry; some estimates suggest up to 20% of the current $1.8 trillion chemical industry could eventually be dependent on synbio.
18
Many available synbio chemicals are important products in the traditional petroleum-refinery value chain: succinic acid (BioAmber, Montreal, Canada; DSM, Heerlen, The Netherlands), 1,3-propanediol (Tate & Lyle, London; Genencor/DuPont, Palo Alto, CA), isobutanol (Gevo, Engelwood, CO),
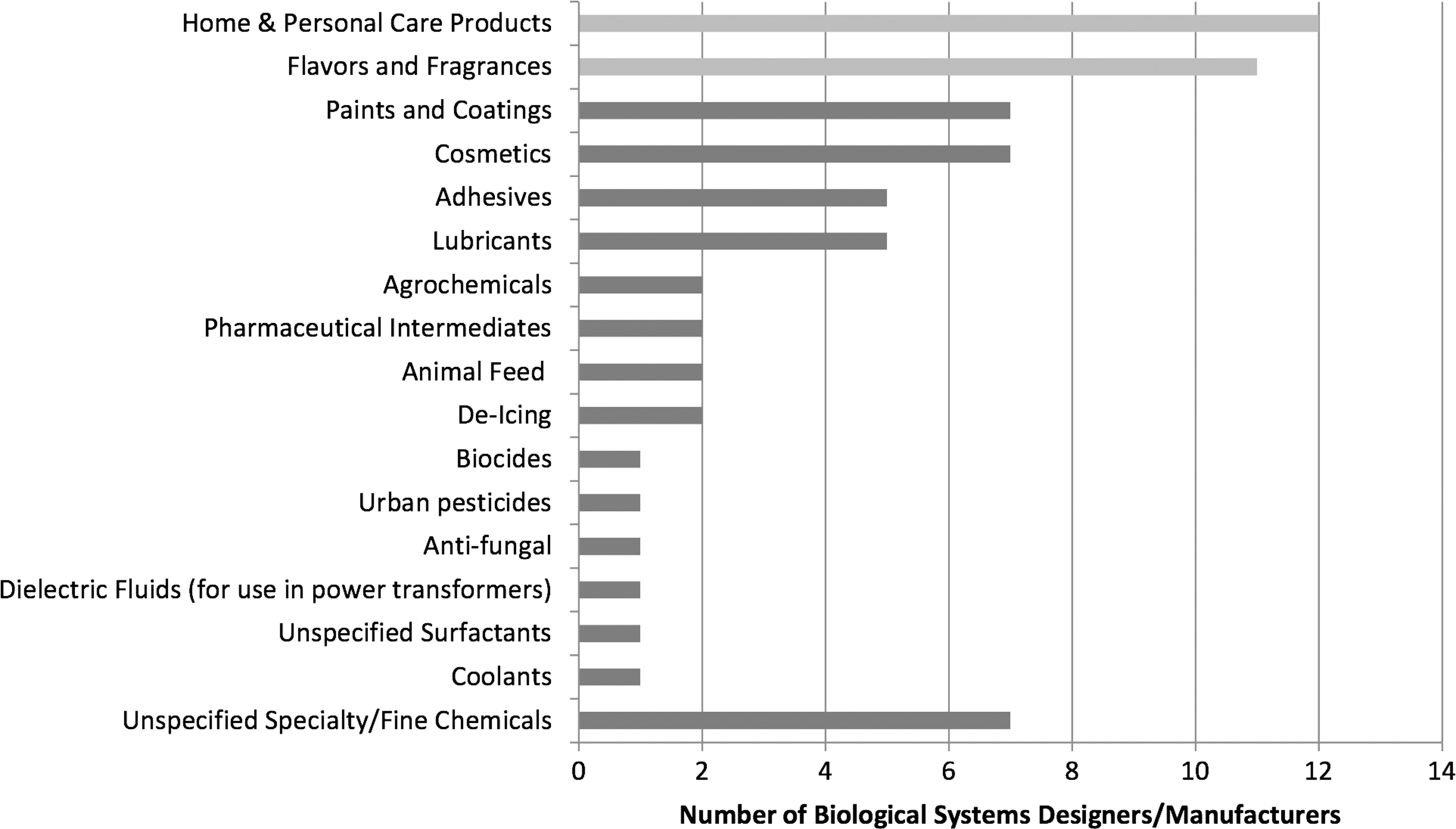
Breakdown of specialty/fine chemical applications worldwide. 12
Energy
Much of the history of applied synthetic biology is commingled with biofuels, with the result that disappointing shortfalls in anticipated targets for commercial-scale biofuels production have been setbacks for both. 20 Nonetheless, persistent US government interest in advancing next-generation (non-corn lignocellulosic, algae, and other) biofuels and biopower is supporting continued efforts by companies like Amyris, Solazyme (South San Francisco, CA), Qteros (Marlborough, MA), Genencor/DuPont, and Algenol (Fort Myers, FL), to develop biobased production platforms, feedstocks, and products. 21 As many as 530 biorefineries, each with mean capacity of 40 million gallons annually, could be required to meet the goals of the US Environmental Protection Agency's 2007 amended Renewable Fuel Standard (RFS2), which stipulated 36 billion gallons of renewable fuel blended into national transportation fuel supplies by 2036. 22,23
Agriculture
Engineering of complex traits in higher plants has yet to move beyond research endeavors. The need for mass-volumes of biofeedstocks with high energy yields, lower input requirements, and greater environmental hardiness, for large-scale production of chemicals and fuels, should drive continued efforts for agricultural synbio. One possible nearer-term use may stem from Synberc investigations to exploit the nitrogen-fixation capacity of Klebsiella oxytoca, with downstream potential for management of soil nitrogen from chemical fertilizers. 24
Other markets
Synthetic biology stands to be massively disruptive in several other markets. Current diagnostics and pharmaceutical applications center on improving existing methods to produce on-market drugs (e.g., Codexis' [Redwood City, CA] directed evolution for optimized production for Merck, Pfizer, Teva, Roche, DSM) and to facilitate vaccine production (Amyris and Sanofi-Aventis' antimalarial precursor production; Novartis' rapid-response H7N9 influenza vaccine). 25 –27 Future advances will include synthetic self-amplifying mRNA vaccines, cell-based therapeutics, and as yet elusive personalized medicines (including effective cancer treatments). 26,27
Designed microorganisms for environmental remediation have been demonstrated by Modular Genetics (partnering with Iowa State University), which developed microorganisms that produce soy-based biodispersants for oil-pollution management (e.g., the 2010 Deepwater Horizon oil spill, supported by an NSF Rapid Response Grant of $200,000). 28 The myriad sensing and response pathways of cells are being harnessed for development of biosensors, for applications from food safety to bioterror-agent detection. The synbio-enabled capacity to detect arsenic in drinking water was demonstrated by an iGEM team as early as 2006; a sophisticated evolution of this concept is in implantable, bioresorbable biosensors for frontline military, a project currently supported by DARPA. 29,30 While the field still awaits a steady stream of successes, the market for synbio products is nonetheless evolving and growing rapidly, as indicated by the number of technologies moving synbio companies to maturity, successful public offerings, and strategic ventures with established industry leaders and government agencies.
Technology Readiness
The growth of these applications critically depends on availability of foundational and translational technology. The US government has noted that synbio still requires “substantial basic and applied R&D to realize its full potential as an engineering and scientific discipline and as a viable tool set for commercial biotechnology.” 31 A recent international survey of approximately 150 self-identified synthetic biology researchers revealed a breadth of options available to synbio researchers, including various public registries, assembly standards, and software tools—many, well established and in the public domain and others available through non-exclusive licensing (Figure 6 ). 32
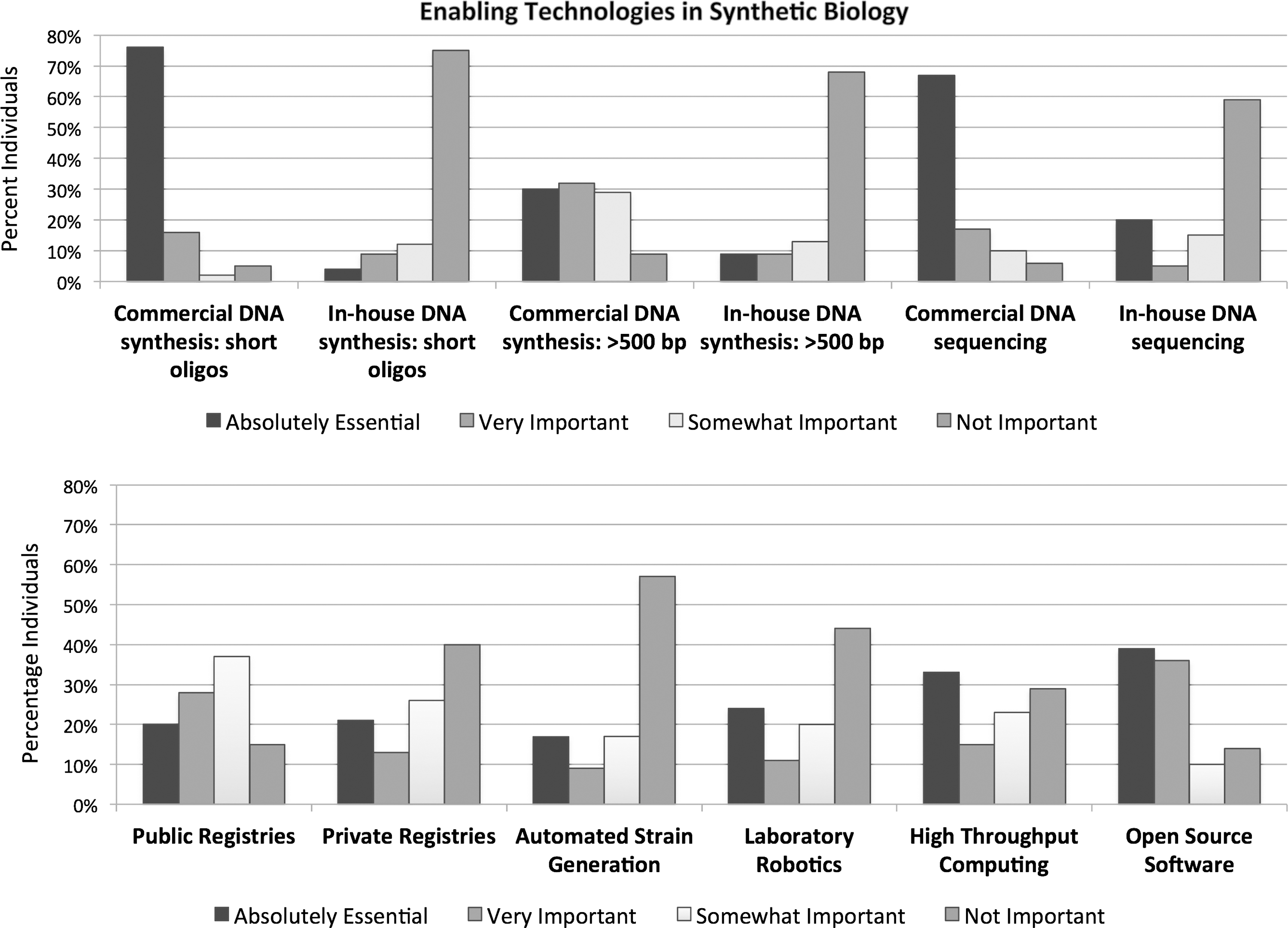
Enabling technologies in synthetic biology. 33
Still, several areas of need remain, calling for continued research investment. The DOE has identified critical fundamental knowledge gaps in methods and technologies (genome-scale engineering tools, DNA synthesis and assembly, analytical tools), biological platforms (biological design principles, genetically tractable organisms/chassis, minimal cell and in vitro systems, tools for plant systems, biocontainment), and computational tools and bioinformatics (information standards, databases). 31 Continued efforts must build on the foundational sciences established to date while addressing these key research needs—a concept that governments worldwide are acknowledging.
Public Research Landscape
United States
Challenges must be overcome before the needed research developments described can occur. In the US (and elsewhere), synbio research lacks a single, coordinated governmental funding plan. US funding is spread among agencies, and industry analysts note that concrete numbers on US funding are difficult to come by due to missing information, an uncoordinated government approach, and varied definitions for synthetic biology. Still, the US government has spent more money to support synthetic biology than has Europe or Asia: at least $500 million by 2010 and as much as $140 million per year 1 since the field's inception, by some estimates. 3,6,9
Important US agencies that impact the field include the NSF, DOE, Department of Health and Human Services (including NIH), DOD (including DARPA), Department of Agriculture (USDA), US Navy, Air Force, and National Aeronautics and Space Administration (NASA). The White House Office of Science and Technology Policy (OSTP) is increasingly looking to coordinate synthetic biology efforts across agencies. OSTP's Technology and Innovation Division has voiced a keen interest in synbio and adjacent disciplines (systems biology, bioprocessing, and biomanufacturing) and has engaged with DARPA and other agencies to identify collaboration opportunities. Still, a coordinated national effort on the scale of the National Nanotechnology Initiative seems unlikely.
Regardless, while the US may lack a solidly coordinated, integrated research strategy, there is some level of support for synthetic biology across many agencies that appears to be growing. A more coordinated and defined research effort, properly communicated and managed by the synbio research community, would likely translate into greater and more-coordinated government funding. Conversations with government representatives consistently identified the lack of a collective US vision for synbio, driven by research and commercial interests, to guide independent but aligned investments from US agencies—investments required to grow the base of foundational technologies that will allow synthetic biology to fulfill its greatest promise. (Also missing for the OSTP may be a more appropriate term than “synthetic biology,” which may do more harm than good in Washington, DC. Both OSTP and DARPA prefer “engineering biology,” which is perceived as more inclusive and less polarizing. Others in the community disagree that the name should be changed.)
European Union
European biosciences research is funded through a complex, tiered web of organizations and time-limited initiatives, at modest levels compared to the US (on the order of tens of millions of dollars, versus the 100-million-dollar scale of some US initiatives, like DARPA's Living Foundries Programs;
The take-away is that while synbio is becoming a cohesively orchestrated EU initiative it is still very much in early development. A European Commission EU-funded project, “Towards a European Strategy in Synthetic Biology (TESSY),” was used to outline a roadmap for European synthetic biology around scientific milestones, knowledge transfer, funding, and regulation. ERASynBio represents a major first initiative through which national Member State funding bodies aim to support common goals. ERASynBio is integrating emerging national activities into a coordinated European effort. Specific objectives include development of research programs, strategies, and infrastructures that preempt fragmentation of programs, regulatory frameworks, and policies; initiation and implementation of transnational research funding; and coherence of a fragmented European research landscape. In April 2014, ERASynBio published a report, “Next Steps for European Synthetic Biology: A Strategic Vision from ERASynBio,” which outlines its recommendations across five broad themes that encompass world-leading research, responsible innovation, global community, skilled workforce, and open data.
United Kingdom
The UK is second only to the US in synthetic biology research (by publication activity) and leads Europe in the number of synbio companies and dedicated institutes. 3,34 The country completed a synbio roadmap in 2012, and synthetic biology became an area of formal focus for the UK government when it was formally declared one of “eight great technologies” that will impel the country's economic growth (the eight being life sciences, genomics and synthetic biology; big-data revolution and energy-efficient computing; satellites and commercial applications of space; robotics and autonomous systems; regenerative medicine; agri-science; advanced materials and nanotechnology; and energy and its storage). 35 To date, maximum funding initiatives have typically been in the range of tens of millions of dollars; however, a government commitment of over $133.5 million in the coming years was announced in early 2013 by the country's Ministry of State for Universities and Science. 36 The two main synbio funding councils, The Biotechnology and Biosciences Research Council (BBSRC) and The Engineering and Physical Sciences Research Council (EPSRC), have demonstrated clear commitment to advancing both fundamental and translational research. More recently, in April 2014, the BBSRC announced investments of nearly $17 million to establish five DNA synthesis centers across the UK and $3.4 million to enhance student training in synthetic biology.
China
While China's economic policies have been nationalistic, the country's acknowledgment of critical needs for its population may be encouraging a more cooperative approach that could open opportunities for synbio within the country and with other nations. (China's 12th Five-Year Program, announced in 2010, formally recognizes the need for openness and international collaboration. 6 ) China's burgeoning citizenry of 1.3 billion represents one-fifth of the global population. Given urgent needs in health, nutrition, and resources management, the Chinese government is motivated to have synbio research advance to real-world applications. The country has drafted a strategic roadmap outlining opportunities over the next 5, 10, and 20 years, with goals for comprehensive databases of synthetic parts, and time frames both for commercial application of engineered parts and clinical application of devices and systems. 6 The Chinese Academy of Sciences (CAS) intends that synbio research in China be “among the best in the world in 5–10 years,” a likelihood given China's clear commitment to R&D overall; the country intends to increase its R&D spending from 1.5% of its GDP to 2.5% in 2020. 5,37
Funding for Chinese synbio research comes from many sources, including the National Natural Science Foundation of China, state-level laboratories, and the CAS Knowledge Innovation Program. Research expenditures by the country total approximately $100 billion, though only $32.5 million is allocated specifically for synthetic biology. 6 Unlike in the US, much of the research financing is state-controlled; China's central party holds powerful sway over research directions, with large budgets at its disposal allocated according to 5-year R&D priorities focused in areas “of national urgency.” 6
Regulatory Landscape
The regulatory landscape governing synbio is complex. International agreements are difficult to reach and sometimes criticized for not being comprehensive. Governments differ in domestic approaches, which may render compliance processes difficult and expensive to manage. Meanwhile, it is synbio scientists who have been at the forefront in developing and recommending solutions to problems and potential challenges that arise as the field evolves.
Regulatory approaches have both top-down and bottom-up aspects. Top-down regulations set standards for proper laboratory use and environmental safety standards, establish weapons non-proliferation practices, and further humanitarian research. Bottom-up approaches involve engineering biological safeguards into organisms, screening and tracking orders for DNA sequences, and developing safe and ethical laboratory practices.
The International Risk Governance Council (IRGC) has classified concerns of civic watchdog groups, industry experts, lawyers, ethicists, and governmental organizations into seven themes: • Insufficient basic knowledge about potential risks posed by designed and synthetic organisms • Uncontrolled release (accidental or deliberate) of novel genetically modified organisms with environmental or human health implications • Bioterrorism, biological warfare, and construction of novel, hostile organisms • Emergence of a “bio-hacker” culture, with unregulated individuals developing dangerous organisms • Patenting of genetic material and creation of monopolies controlling these resources • Trade and global justice (exploitation of indigenous resources, or distortion of land-use agendas for large-scale biomass feedstocks) • Creation of artificial life and related philosophical and religious concerns
38
–41
The challenge is that, similar to the research-funding landscape, regulatory oversight is unclear and disorganized across international, national, and regional levels. Several international approaches to biological weapons have emerged, such as “The Convention on the Prohibition of the Development, Production and Stockpiling of Bacteriological (Biological) and Toxin Weapons and on their Destruction” (commonly known as the Biological Weapons Convention), which formed “the first multilateral disarmament treaty banning the development, production and stockpiling of an entire category of weapons of mass destruction.” 42 Critics, however, are quick to note that international agreements' “slow consensual nature is not well suited to addressing unconventional risks posed by non-state actors.” 43
Additional international agreements have been negotiated, including the “Convention on Biological Diversity (CBD) Cartagena Protocol on Biosafety” and subsequent “Nagoya Protocol,” and the World Health Organization's “Biosafety Standards Manual.” 44 The US has signed but not ratified the CBD and is therefore precluded from ratifying the “Nagoya Protocol”—preventing the country from assuming its critical leadership in these important conversations.
Domestically, US synbio activity is subject to an overlapping but uncomprehensive patchwork of regulations across multiple agencies. As Synberc and iGEM leadership have concluded, existing regulations may not be either enforced or enforceable. This lack in oversight will be increasingly important as synbio progresses and the more-inventive possibilities materialize (of heretofore nonexistent genetic functions), especially in the hands of citizen researchers in the Do-It-Yourself (Synthetic) Biology (DIYbio) movement.
DIYbio has emerged over the past decade and presents both challenges and opportunities for regulatory oversight and public perception. It puts bioengineering tools in the hands of citizen scientists, under the premise “that wider access to the tools of biotechnology, particularly…related to the reading and writing of DNA, has the potential to spur global innovation and promote biology education and literacy.”
45
This community was inspired by Rob Carlson's home biology lab, gained a voice with
The DIYbio movement has obvious potential to stir concerns about public safety. However, there is a great opportunity for grassroots community efforts like these both to spur innovation and foster the public education needed for synbio to mature. Still, in order to succeed, new financial models for community labs must emerge, additional community resources will likely be required, and sufficient regulatory oversight must be established. With these in place, the potential for DIYbio to be a positive contributor to the field—and the greater good—is significant.
An additional regulatory concern is that NIH guidelines (the most comprehensive available to date) are not binding on all individuals conducting synbio research. 47 As one report states:
Products are currently covered by three different US agencies operating under four separate statutes. The result is a regulatory system marked by fragmentation, lack of coordination, and different standards for different types of products…Differences in statutory mandates, risk assessment methodologies, and agency cultures…create a system where the stringency of the risk assessment and approval process is more dependent on the specific product category than on the risk of the product. 47
There is a clear opportunity for the US to take a more focused, integrated, and global approach to synbio regulation—with support from the research community—in advocacy, education, and collaboration. Without this, ambiguous regulatory policies (as well as lack of capacity) may constrain the full potential of synbio in the US and the world going forward.
Public Awareness
The ultimate success of synbio will depend on acceptance by public stakeholders, both as citizens and consumers. Within the US, public opinion on synthetic biology is cautious but open: nearly two-thirds of Americans surveyed believe synthetic biology research should continue rather than be banned, even if its implications and risks are not fully understood. 10 Similar opinions exist in the UK (although public opinion there varies by the nature of the end product, means to achieving that product, and by gender, region, and age). 48
It is important to note that most Americans are unsure about what synthetic biology is. As the ultimate stakeholders in any governmental regulatory decisions, consumers must be equipped with factual information about the vast potential benefits of synthetic biology, as well as its risks, and the measures being undertaken to address these. Public education and engagement are essential to the future growth of this field.
Conclusions
The US has been a leader in the development and commercialization of synthetic biology. It has produced more high-impact foundational and translational research than any other nation, and its technology infrastructure is in many ways a model of industry, government, and academic cooperation. But the United States' continued leadership in synthetic biology is not guaranteed. Other countries, including the super economy of China, and some within the collective entity of the European Union, are investing heavily to assure their own strength in applying synthetic biology to address the needs of their citizenries.
The US must therefore renew and grow its commitment to synthetic biology, in investment, policy, and public education, or risk falling behind in an industry and research sector that it has pioneered. To realize the true promise of this emerging field, much more foundational research is needed, as are continued funding and rationalized regulations (both domestic and international) that facilitate the safe deployment of these technologies for the greatest good, with the public truly engaged. The time is now to bring public and private actors together to create a strategic direction for synthetic biology consistent with US national priorities and values.
