Abstract
Induced cells of Rhodococcus rhodochrous DAP 96253, when placed in the proximity of climacteric fruit (peaches, bananas, avocados) delay the ripening of the fruit. Examination of the delayed ripening shows that the induced cells possess multiple mechanisms that have the potential to impact the Yang Cycle in climacteric plants and processes such as volatile flavor and fragrance compounds related to the climacteric process. The suggestion is brought forward that these induced R. rhodochrous cells possess the ability to modify/modulate an important plant process.
Introduction
This review provides a current summary of our work regarding the delayed ripening of climacteric fruit by induced cells of Rhodococcus rhodochrous DAP 96253. In the process of developing this technology, we believe a picture is emerging that illustrates a close relationship between the Yang Cycle, its role in climacteric plants (Fig. 1), and the biochemistry/physiology of an induced environmental prokaryote. 1
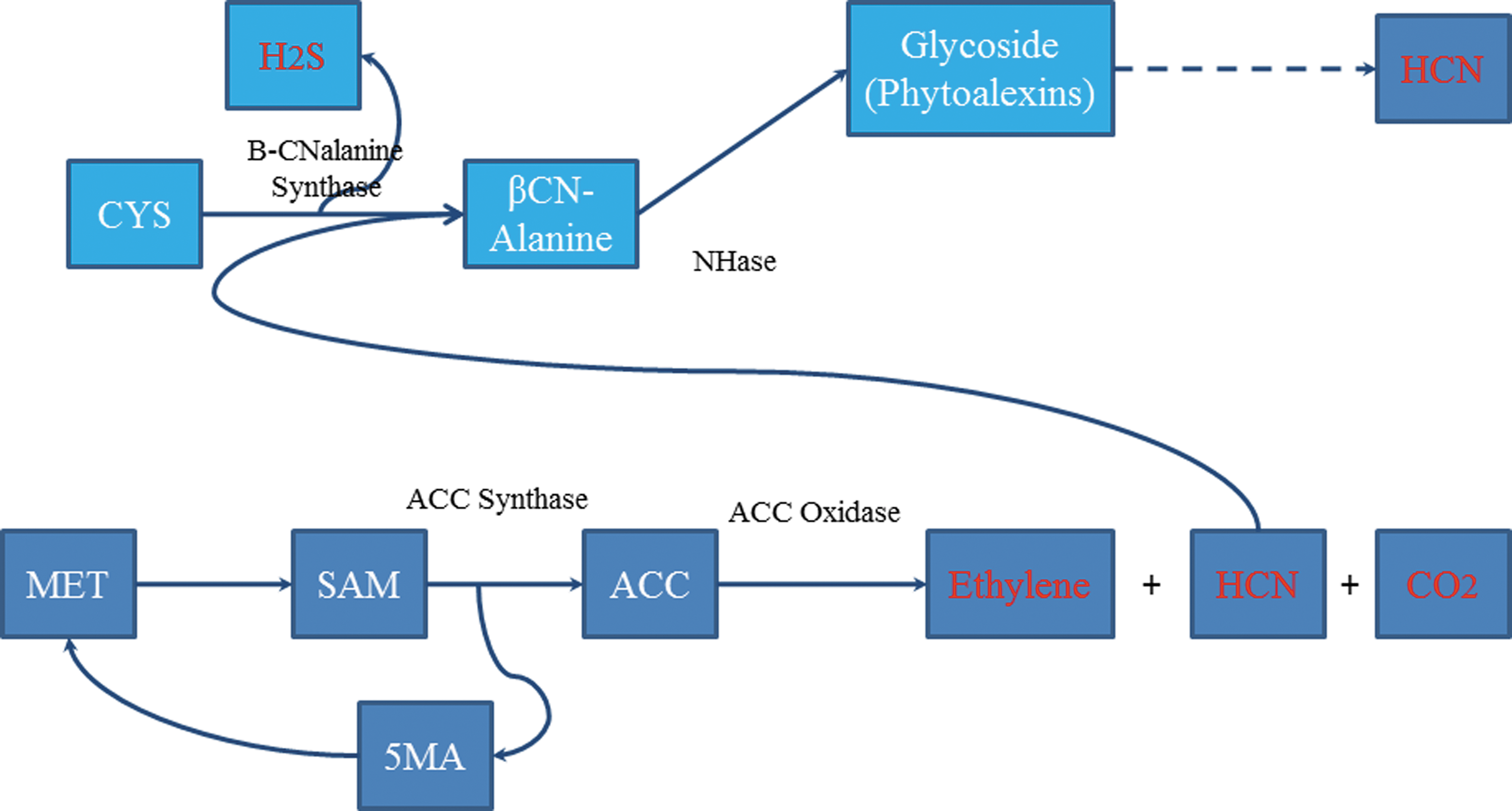
Yang Cycle (dark blue boxes), including the integration of β-cyanoalanine synthase activity (light blue boxes); red text indicates compounds with defined volatility. 1
The role of communication (plant signaling) in the ripening of climacteric fruit or in the response of climacteric fruit to chill injury is well established. 1 –4 Equally well established is the complex intraspecies communication present in a number of plant diseases such as Dutch elm disease or Ambrosia fungi/beetles. In all of these communication schemes, volatile organic compounds play an important role.
Our investigation of nitrile/amide metabolism in nature led to the discovery that induced cells of R. rhodochrous strain DAP 96253, when placed in proximity to harvested peaches (Fig. 2), significantly delays ripening. 5 –7 Subsequent studies showed delayed ripening of yellow bananas (Fig. 3) and avocados. Studies conducted with numerous peach varieties grown in the state of Georgia showed that delaying the ripening of peaches also delayed the onset of mold growth (Fig. 2). 5 –8 It was also observed that the effects of chill injury in peaches, after extended periods of refrigeration at transport temperatures recommended by the United States Department of Agriculture, could be significantly mitigated by exposing the refrigerated peaches to induced cells of R. rhodochrous DAP 96253. 8

Comparison of Big Red Peaches held for 6 days at 25–27°C.

Yellow-ripe bananas placed with a certain amount of live and immobilized fermented cells in a sealed plastic box for 14 d.
Initially, all experiments were conducted using live cells of induced R. rhodochrous DAP 96253. But, to investigate practical methods, the cells were immobilized using glutaraldehyde and then cross-linked using polyethylenimine (PEI), a method that renders the cells non-replicating. 8 –11 While no viable cells could be recovered from glutaraldehyde/PEI immobilization, the immobilized cells were still capable of delaying the ripening of peaches and/or bananas. 5 –8
Materials and Methods
Microorganisms
The bulk of the delayed ripening work was conducted using R. rhodochrous DAP 96253. The maintenance, cultivation, and induction are detailed in Pierce et al. 8 –11 R. rhodochrous DAP 96253 cells were grown on yeast extract, malt extract agar (YEMEA) medium (ISP medium 2 as recommended by Dietz and Thayer). 12 Full induction was achieved by supplementing YEMEA with urea (7 g/L) and cobalt (as CoCl2), at 10–88 ppm with 50 ppm preferred. 5 R. rhodochrous DAP 96622 was similarly maintained. Rhodococcus erythropolis 47072 was obtained from the American Type Culture Collection (ATCC, Manassas, VA), and was initially revived and maintained as recommended by the ATCC before transfer to YEMEA-based media.
R. rhodochrous DAP 96253 has been deposited with the ATCC and has been subsequently assigned the strain designation ATCC 55889.
Enzyme Assays
Nitrile hydratase
R. rhodochrous DAP 96253 cell concentrate (equivalent to 50 mg) was first suspended with 1 mL 50 mM phosphate buffer (pH 7.4) and then diluted to 50 mL with the phosphate buffer. Sufficient cell suspension was prepared for all enzyme assays to be conducted in triplicate using identical cell suspensions. The method is based upon the method of Pierce et al., as follows. 8 Cell suspension (1 mL) is combined with 9 mL of acrylonitrile solution (5,000 ppm in phosphate buffer) and vigorously mixed for 2 min at 30°C, at which time the reaction is terminated by the addition of 20 μL of 4 N H2SO4. After mixing for 1 min, the reaction was neutralized with 4 N NaOH. Cells were separated either by centrifugation or filtration, and 2.5 units of amidase (Sigma-Aldrich, St. Louis, MO) was added to the cell-free solution. The solution was then incubated for 30 min at 37°C. The solution was transferred into a new test tube, and 2 mL sodium phenate, 3 mL 0.01% sodium nitroprusside solution, and 3 mL 0.15% sodium hydrochloride solution were added, in that order. (The phenate solution was prepared by melting 25 g phenol in a waterbath at 50°C, followed by addition of 800 mL double distilled water and 78 mL 4N NaOH.) The test tube was vortexed for 30 sec and incubated in the dark at room temperature for 30 min. After 30 min, the optical density (OD) was measured at 600 nm.
Amidase and cyanidase assays
Using the cell solution prepared above (1 mg cells/mL), 1 mL of cell solution was added to 9 mL of acrylamide solution (1,000 ppm in phosphate buffer) for the amidase assay—or 9 mL of HCN solution (50 ppm in phosphate buffer) for the cyanidase assay. The solutions were vigorously mixed for 2 min at 30°C, at which time the reactions were terminated by the addition of 20 μL of 4 N H2SO4. After mixing for 1 min, the reactions were neutralized with 4 N NaOH. Cells were separated either by centrifugation or filtration, and 2.5 units of amidase (Sigma-Aldrich) were added to the cell-free solution, which was then incubated for 30 min at 37°C. After the solution was transferred to a new test tube, 2 mL sodium phenate, 3 mL 0.01% sodium nitroprusside solution, and 3 mL 0.15% sodium hydrochloride solution were added, in that order. The tube was vortexed for 30 sec and incubated in the dark at room temperature for 30 min. After 30 min, the OD was measured at 600 nm.
1-Aminocyclopropane-1carboxylic acid (ACC) deaminase assay
Based upon the method of Nagatsu and Yagi, and using the cell solution prepared above (1 mg cells/mL), 1 mL of cell solution was added to 9 mL of ACC solution (100 ppm in phosphate buffer) and vigorously mixed for 2 min at 30°C. The reaction was then terminated by the addition of 20 μL of 4N H2SO4. 13 After mixing for 1 min, the reaction was neutralized with 4 N NaOH. Cells were separated either by centrifugation or filtration, and 2.5 units of amidase (Sigma-Aldrich) were added to the cell-free solution, which was then incubated for 30 min at 37°C. The solution was then transferred into a new test tube, and 2 mL sodium phenate, 3 mL 0.01% sodium nitroprusside solution, and 3 mL 0.15% sodium hydrochloride solution were added, in that order. The test tube was vortexed for 30 sec and incubated in the dark at room temperature for 30 min. After 30 min, OD was measured at 600 nm.
β-cyanoalanine synthase assay
Based upon the method of Ezzi and Lynch, 1 mL of the cell suspension (1 mg/mL) was added to 1 mL 50 mM phosphate buffer (pH 7.4). Subsequently, 0.5 mL of 0.05 M HCN (in 0.1 M Tris-HCl, pH 8.5) and 0.5 mL of 0.01 M L-cysteine solution (in 0.1 M Tris-HCl, pH 8.5) were added. 14 The solution was then immediately capped and incubated at 30°C for 20 min. The cells were removed by centrifugation at 13,000 rpm for 2 min. The supernatant was transferred to a new tube, to which 0.5 mL of 0.03 M FeCl3 in 1.2 M HCl was added followed by rapid addition of 0.5 mL of 0.02 M N,N-dimethyl-p-phenylenediamine sulfate solution (in 7.2 M HCl). The capped samples were incubated at room temperature in the dark for 25 min. The absorbance of each was then read at 600 nm.
Isocitrate lyase
Based upon assays performed by McFadden and Howes, and modifications by Hiller and Charnetzky, 0.1 mL of cell suspension—1 mg cell/0.1 mL in 3-(N-morpholino)propanesulfonic acid buffer (pH 7.7) containing 5 mM MgCl2+1 mM ethylenediaminetetraacetic acid—was incubated with 5 mL of 2.2 mM isocitric acid for 30 min at 30°C. 15,16 The cells were then removed by centrifugation at 42,000 rpm for 5 min, and 0.5 mL of the supernatant was transferred to a test tube, to which 1.0 mL of 1% phenylhydrazine hydrochloride solution was added and mixed for 30 sec. Concentrated HCl (4 mL) was then added, followed by 1 mL 5% K3Fe(CN)6. The solution was then mixed for 1 min and allowed to rest for 7 min. The OD was read at 520 nm.
Monooxygenase and epoxide hydrolase
Based upon the method of McCay et al., styrene oxide stock solution (10 mM) was prepared by adding 2.85 μL of styrene oxide to 2.5 mL acetone. 17 Standards of 1, 2, 3, 4, and 5 mM were then prepared in acetone. Then 50 μL of each standard was added to 5 mL of phosphate buffer (50 mM, pH 7). Cell suspension (0.1 g/mL) in phosphate buffer (50 mM, pH 7) was transferred to a 40-mL amber glass vial (outer vial); 100 μmol (11.5 μL) of styrene was added to each sample. For assaying epoxide hydrolase, 350 μL of 3 mmol/L of styrene oxide solution was added to each sample. Next, 500 μL of 4-(4-nitrobenzyl) pyridine solution (100 mmol/L) in ethylene glycol was added to a 4-mL transparent glass vial (inner vial), which was then placed into the outer vial. The outer vial was crimp-sealed using a crimp with a Teflon-faced butyl rubber septum. The sealed vials were incubated at 30°C and 150 rpm for 24 h. To each inner vial 500 μL of triethylamine solution (1:1 v/v) in acetone was added. The OD was read at 600 nm.
Monooxygenase gas chromatography method
Head-space gas (500 μL) was injected into a DB 624 column (75 m × 0.53 mm). A Perkin Elmer (Waltham, MA) gas chromatograph, with inlet and flame ionization detector set to 250°C, was run isothermally at 180°C with carrier gas (He) set to 4 mL/min.
Results
Induced R. rhodochrous DAP 96253 and Delayed Ripening of Climacteric Fruit
Figures 2 and 3 provide images of delayed ripening of peaches and bananas, respectively. In the comparative images in Fig. 2, a catalyst derived from induced cells of R. rhodochrous DAP 96253 was formulated with an edible wax. This wax/catalyst formulation was applied to the inner surface of a standard, commercial peach box; the control box was only wax-coated. The peaches were Big Red Variety from Georgia. (This type of test has been repeated over several seasons). To date, seven different commercial varieties of peaches from Georgia and South Carolina have been evaluated with similar results. In Fig. 3, yellow-ripe bananas were placed in a container alongside a petri dish containing the catalyst derived from the induced cells of R. rhodochrous DAP 96253. In the container experiments, yellow-ripe bananas have been evaluated as single fingers, multiple-single fingers, and as hands with up to eight fingers. All bananas evaluated originated in South America.
Induced R. rhodochrous DAP 96253 and the Potential Relationship to the Yang Cycle
The biosynthesis of ethylene in plants (Yang Cycle) is shown in Fig. 1. The precursor to ethylene in the Yang Cycle is ACC. Induced cells of R. rhodochrous DAP 96253 possess increased levels of ACC deaminase (Table 1), enabling these cells to convert ACC to α-ketobutyric acid and ammonia. In the Yang Cycle, the biosynthesis of ethylene results in the equi-molar production of HCN and CO2. In plants, the conversion of HCN to a nontoxic molecule can be accomplished through the action of β-cyanoalanine synthase (β-CAS). 18 While there are typically several substrates in plants for this reaction, β-CAS is most often depicted as involving the conversion of methionine to β-cyanoalanine. Several routes from β-cyanoalanine are possible, including the production of phytoallexins or the conversion to asparagine. Induced cells of R. rhodochrous DAP 96253 possess both cyanidase and a β-cyanoalanine synthase-like enzyme (Table 1). In addition, induced cells of R. rhodochrous DAP 96253 contain very high levels of NHase, which is capable of converting β-cyanoalanine to asparagine.
Enzyme Activity (Units/Mg-Cell Dry Weight) of Three Rhodococcus Strains When Grown on Inducing Medium a
Except for isocitrate lyase, all units are μmoles/min/mg-cell dry weight. For isocitrate lyase, units are nmoles/min/mg-cell dry weight;
ND=not determined
A role for monooxygenase(s)?
Catalysts derived from either induced viable cells or immobilized induced cells (where the immobilization has rendered the cells non-replicating) are capable of delaying the ripening of climacteric fruit. Direct headspace analysis has confirmed monooxygenase-like activity against ethylene and the subsequent epoxide hydrolase activity. Spiking with styrene and styrene epoxide has confirmed monooxygenase-like activity and epoxide hydrolase activity levels.
Subsequent aerobic experiments with yellow-ripe bananas and induced cells of R. rhodochrous DAP 96253 using gas-liquid chromatography with direct injection of the head-space (GLC-HS) positively identified a decrease in ethylene and concomitant increase in ethanol.
Observing delayed ripening with a catalyst containing ostensibly dead cells presents a quandry. While investigating the proteins that are altered in abundance in induced cells of R. rhodochrous DAP 96253, we uncovered a protein that upon analysis of fragments obtained through matrix-assisted laser desorption/ionization tandem time of flight (MALDI-TOF-TOF) mass spectrometry has been putatively identified as an oxido-reductase bearing a very high degree of identity to the N,N-dimethyl-4-nitrosoaniline (dependent) oxido-reductase, which was reported by Ohhata et al. to be increased by 20–100-fold in the extreme oligotroph R. erythropolis N9T-4 when grown on tetradecane oligotrophically. 19 Luque-Almagro et al. recently indicated that for the cyanide-degrading pseudomonad P. pseudoalcaligenes CECT 5344, HCN combines with oxaloacetate to form the corresponding cyanohydrin. 20 The cyanohydrin is then deaminated in a stepwise fashion through the aldehyde to the corresponding hydroxy-acid by nitrile hydratase/amidase. Oxalacetate is especially noteworthy, as this compound will form cyanohydrin at room temperature over a pH range of 6.5–9.5. The cyanohydrin also forms at ambient conditions with α-ketoglutarate.
Luque-Almargro et al. further noted that when P. pseudoalcaligenes was exposed to cyanide there was a significant increase in the enzyme isocitrate lyase. 20 Isocitrate lyase is the key enzyme for initiating the glyoxylate bypass. The glyoxylate bypass permits the use of C2 compounds as energy sources. This includes fatty acids and alkanes that degrade via beta-oxidation, producing acetyl CoA.
In separate experiments, the published sequence for the amo-C gene of ammonia monoxygenase (AMO) was used to make a knockout of the homologous AMO in R. rhodochrous DAP 96253. AMOs are members of the copper membrane monooxygenase (CuMMOs) family. While the amo-C knockout showed reduced delayed ripening activity, the knockout still retained significant activity (50%).
In examining protein gels of induced cells of R. rhodochrous DAP 96253, we have confirmed—through excision of this protein and subsequent sequence analysis by MALDI-TOF-TOF—the presence of an alkene monooxygenase. (Alkene monooxygenases do not belong to the CuMMO family). This finding confirms that R. rhodochrous DAP 96253 has at least two distinct monooxygenases.
A role for fruit volatiles?
That ethylene plays a major role in the ripening of climacterics is well recognized. It has been suggested that the rate of respiration and ethylene production together profoundly affect ripening in harvested climacteric fruit. 3,4,21 It is further noted that cyanide insensitive respiration contributes to this increase in respiration. 22 –24 The events of system 2 ethylene biosynthesis influence the aromatic (flavor and fragrance) chemicals, which are distinctive to many climacteric fruit. 3 For example, the distinctive flavor and aroma compounds have been defined for bananas, apples, and peaches. 25 –28
It is equally true that distinctive volatiles also may affect ethylene biosynthesis; jasmonates represent such an example. Liu et al. showed that increased jasmonate concentrations resulted in increased ethylene biosynthesis in tomatoes. 29 Fan et al. showed that jasmonate concentrations would influence the aroma chemicals in tomatoes and also in apples. 30 Subsequently, Zhang et al. showed that the cold storage temperature regime (up to 3 weeks and then up to 3 days after return to ambient) used for storing peaches also influenced the aroma chemicals of the peaches. 31
Wasternack suggested that in the case of the jasmonates, the metabolic fate was influenced by the many alternate pathways and the concentration of key components. 32 The complexity of the alternate pathways available are clearly shown by Sanchez et al. in peaches. 28 Soto et al. have shown that methyl-jasmonates interfere with ethylene and auxin biosynthesis in peaches. 33 Earlier, Koch et al. had shown that the biosynthesis of cis-jasmone relieved the stress jasmonate. 34
Notwithstanding that the bulk of our experiments with climacteric fruit involved catalysts placed in proximity to fruit—and thus in these experiments the ability of the catalyst to react to plant molecules is essentially limited to volatile plant molecules, where the catalyst and fruit are near—the reactivity of the catalyst is no longer limited to volatile chemicals. Members of the Mycolata, including Rhodococcus, Nocardia, and Mycobacterium, are well known for their biotransformation potential, as evidenced in both the scientific and patent literature.
In parallel experiments with yellow-ripe bananas exposed to induced cells of R. rhodochrous DAP 96253, the head-space was analyzed using both direct head-space analysis onto GLC and solid-phase microextraction (SPME) fibers that were desorbed onto the GLC column. Figure 4 shows changes in the volatiles of yellow bananas during ripening. In Fig. 5, the GLC head-space profile of induced cells (Fig. 5a) and stage 4,yellow-ripe bananas (Fig. 5b) are provided. Figure 5c shows the head-space profile of identical stage 4 bananas when exposed to the induced R. rhodochrous DAP 96253 cells. Figure 5c shows a significant reduction (70–90%) of the major banana volatiles present. This reduction is more clearly shown in Table 2, in which the component concentrations of the major peaks are compared. Figure 5d and Table 2 show the effect on banana volatiles when partially induced cells of R. rhodochrous DAP 96253 were used.
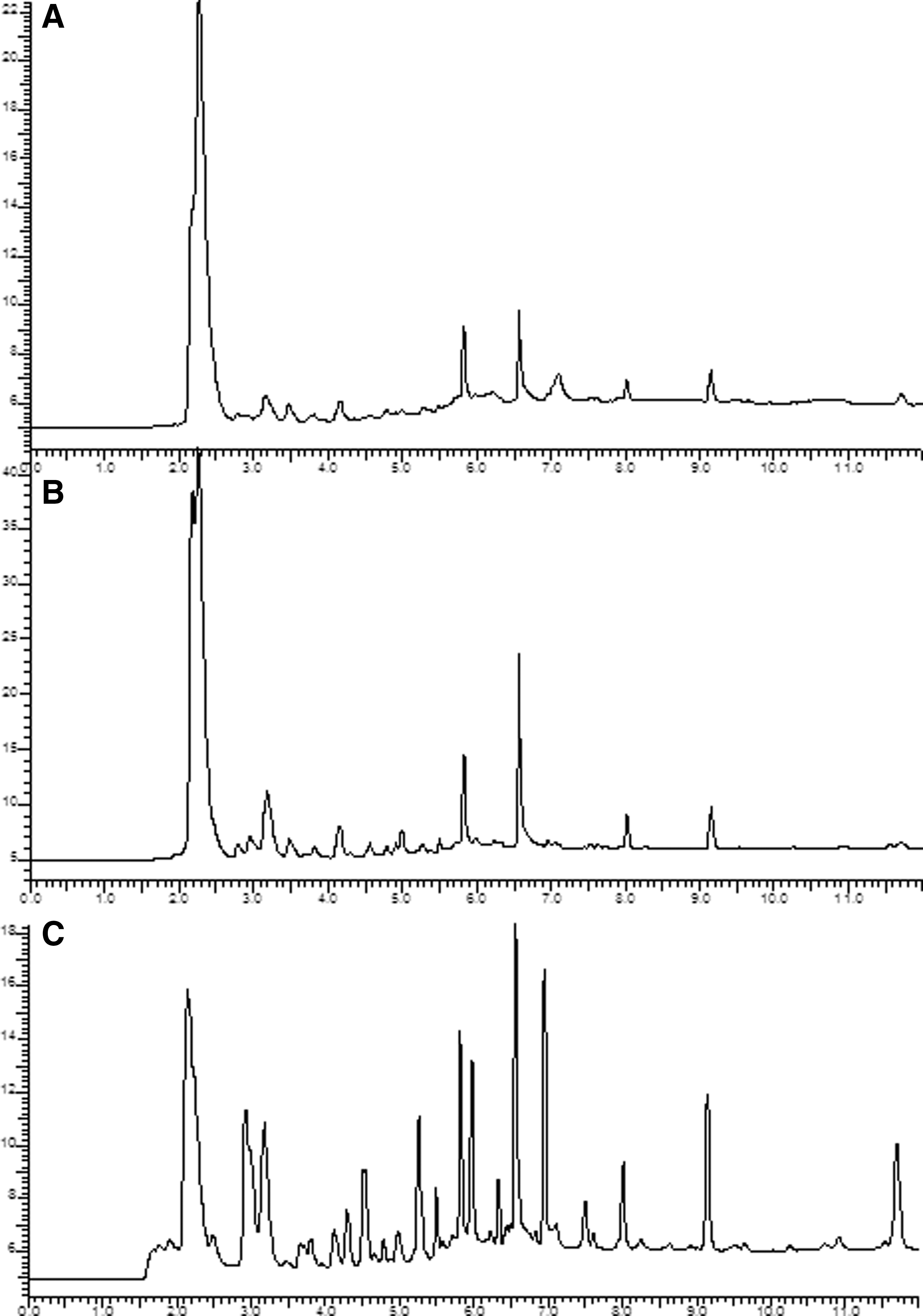
Comparison of volatiles (by GLS-HS) of yellow-ripe bananas.
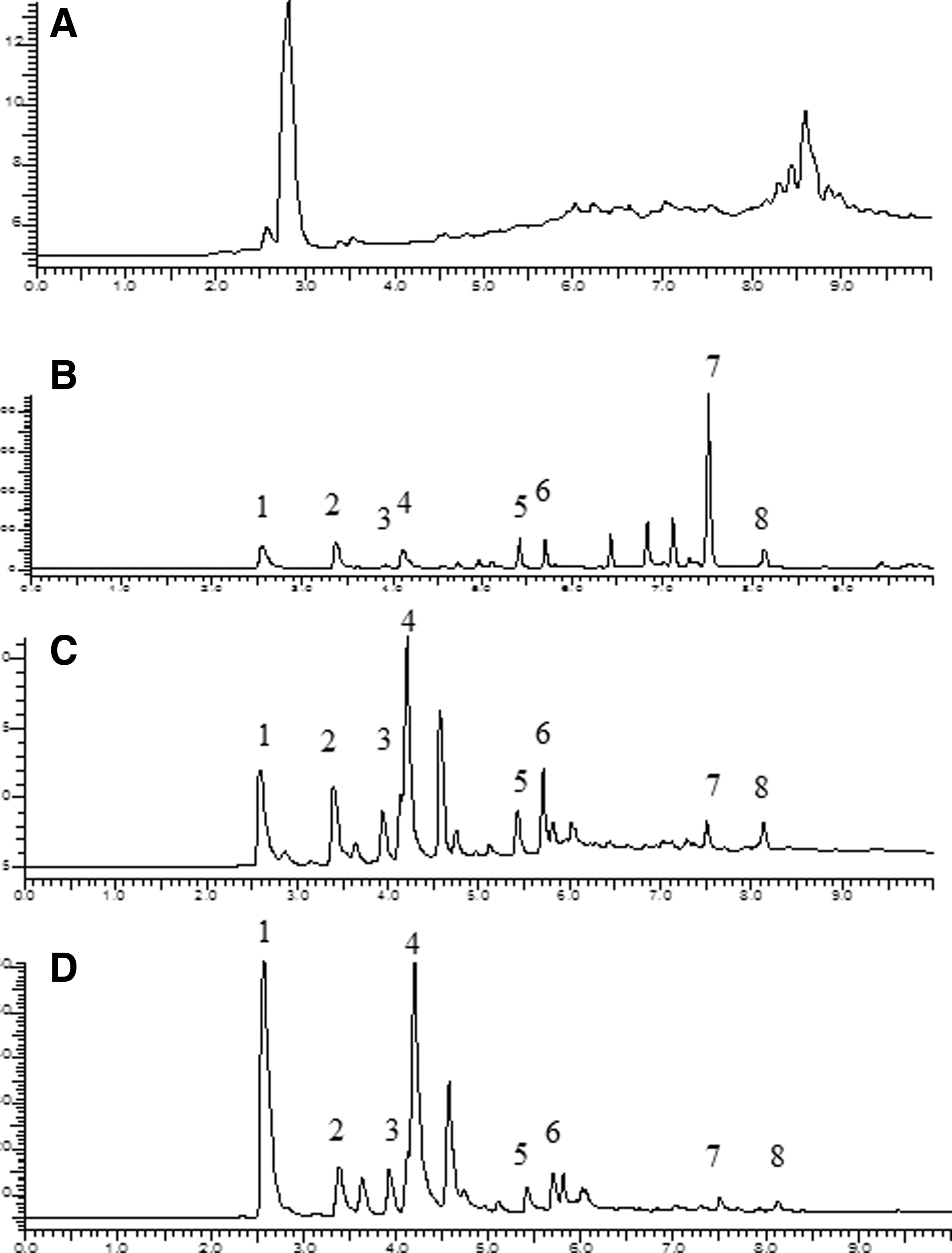
Comparison of volatiles by GC-SPME.
SPME, solid-phase microextraction.
Concentration of major banana constituents normalized to 100%, except for peak 4.
These experiments were conducted in triplicate, on three separate occasions with bananas from the same source and same stage (by visual comparison to commercial banana stage rating). Experiments are currently underway with fruit using different varieties and different harvest lots to expand our understanding of catalyst activity with fruit volatiles.
In the initial thinking regarding the metabolism of the products of ethylene biosynthesis (ethylene and cyanide), the combination of HCN and ethylene epoxide to form the cyanohydrin would result in a product that could easily be metabolized by NHase and amidase to the corresponding acid and ammonia. This approach does not involve molecular oxygen and is, therefore, both oxygen and cyanide insensitive.
Delayed ripening benefits of R. rhodochrous DAP 96253 (as well as the countering effects of chill injury, mechanical injury, etc.) may be viewed as being accomplished by the following mechanism (Fig. 6): • The presence of monooxygenases capable of converting ethylene to the corresponding epoxide, and the subsequent hydrolysis of the ethylene epoxide by epoxide hydrolase; AMOs are oxygen indifferent (provided the reduced pyridine nucleotide cofactor can be regenerated) and do not require molecular oxygen. Using the longer-life styrene we noted both epoxide formation and hydrolysis by R. rhodochrous, and our protein analysis and sequencing confirm the presence of an oxido-reductase in induced cells of R. rhodochrous DAP 96253. While preliminary in nature and confirmation and validation are required, this is most intriguing. • We note the increase of isocitrate lyase in cells exhibiting delayed fruit ripening. This is confirmed in gels of cells exhibiting delayed ripening versus cells that do not exhibit delayed ripening. • The creation of the cyanohydrin of oxaloacetate provides a mechanism whereby volatile cyanide can react with oxaloacetate forming the cyanohydrin. We have shown cyanohydrin degradation of cyanoalanine, and acetone cyanohydrin by NHase/amidase. We have previously shown that the NHase in cells grown with cobalt is even more insensitive to cyanide than iron-NHase. We further note that when cells having shown delayed ripening activity are exposed to cyanide the cyanide concentrations rapidly fall to undetectable levels. • Preliminary GLC and mass spectrometry data suggest that, in addition to being able to react with both ethylene and cyanide, the R. rhodochrous cells capable of delayed fruit ripening alter the secondary metabolites (e.g., jasmonates) of the ethylene/cyanide cascade system. Given that these aroma chemicals can influence ethylene biosynthesis, the temporary modification of fruit volatiles could potentially influence ethylene biosynthesis.
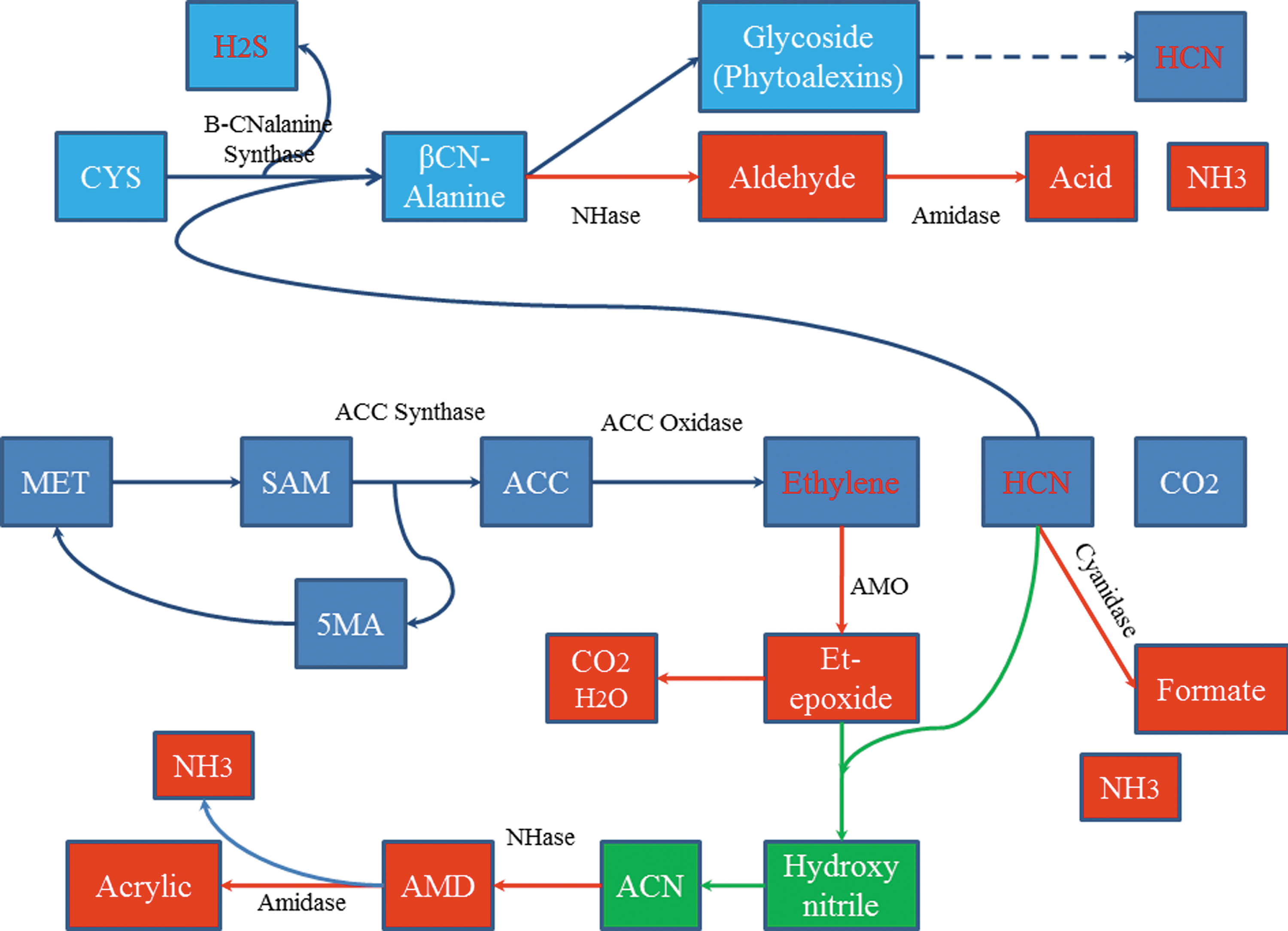
Yang Cycle with potential mechanisms for the interaction of induced cells of Rhodococcus rhodochrous DAP 96253 (red boxes) with the Yang Cycle in climacteric fruits; green boxes represent potential abiotic mechanisms. 1
If the above were to be confirmed, it would suggest that reducing ethylene to very low levels may not be the sole prerequisite for delayed fruit ripening. By partially reacting with ethylene, and by reacting with cyanide, and by further reducing the secondary metabolites of the ethylene cascade system, ripening management can be affected.
A role for cyanide metabolism?
The cyanide metabolism of induced cells of R. rhodochrous DAP 96253 is quite robust. In addition, these cells contain β-cyanoalanine synthase (β-CAS) activity. While the levels of β-CAS in induced DAP 96253 is significantly lower than in the other Rhodococcus strains shown in Table 1, the β-CAS activity in DAP 96253 is very stable (from 4°C to 55°C), and levels of β-CAS remain essentially constant throughout the experiments with climacteric fruits. In experiments in which the induced cells are simultaneously exposed to both ethylene and cyanide, the cyanide is quickly reduced to undetectable levels.
Catalyst-formulated cell preparations
The stabilization of enzymatic activity of the R. rhodochrous DAP cells is a very important practical consideration. Figure 7 shows that calcium alginate immobilized cells have significantly lower enzymatic levels (for the five enzymes selected for evaluation) than immobilization based upon either glutaraldehyde/polyethylenimine cross-linking or immobilization in an edible-wax formulation.
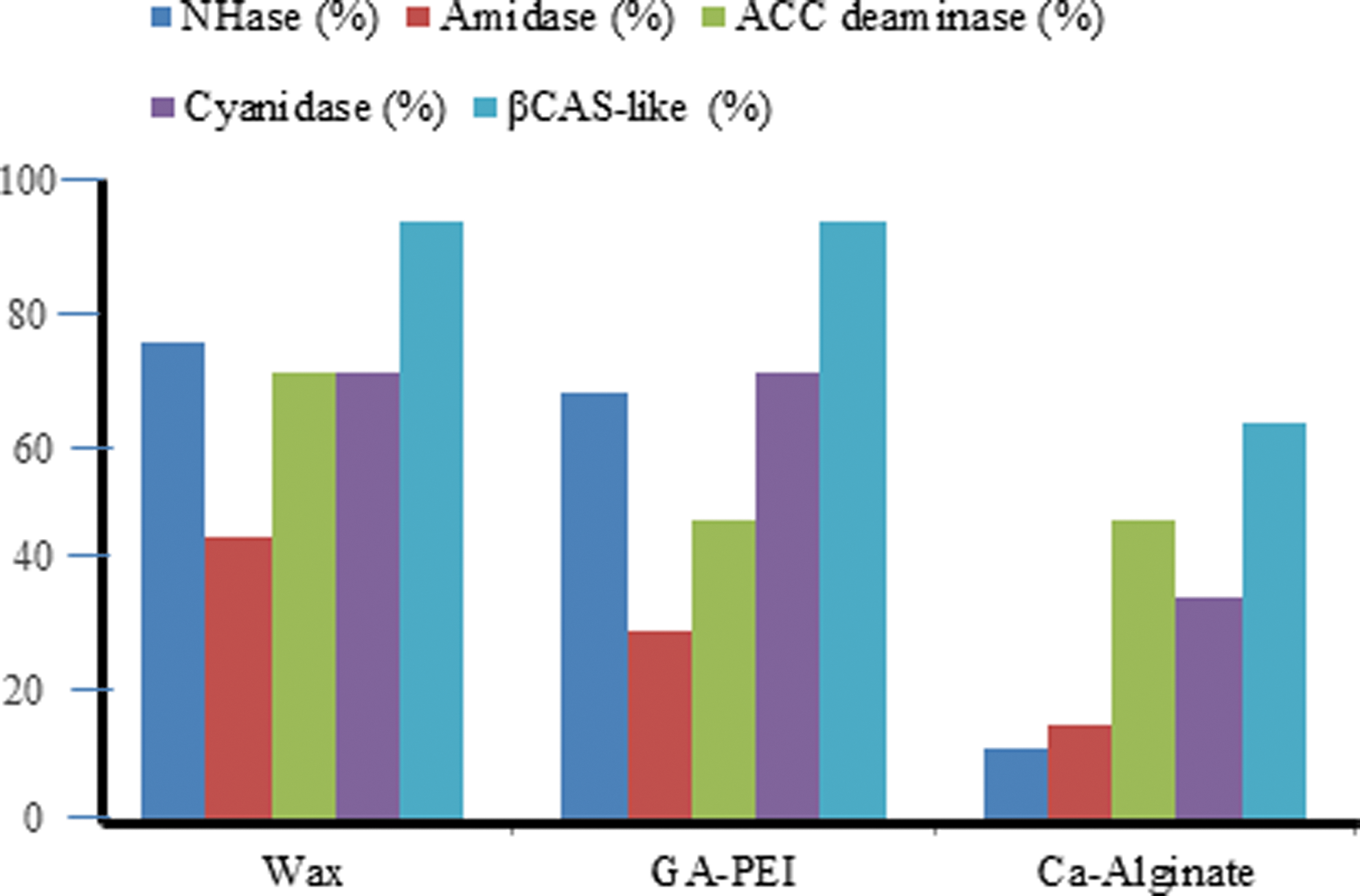
Effect of edible wax, glutaraldehyde (GA-PEI), and CA-alginate (beads) immobilization on enzyme activity.
Discussion
We have consistently shown delayed ripening in peaches (seven commercial varieties from Georgia and South Carolina) and in bananas (yellow-ripe Cavendish bananas). We also have shown significant reduction of chill injury in peaches and in apples (Granny Smith).
Our analyses show that fruit in which ripening is delayed can achieve the same level of ripening as that seen in control fruit. In the case of peaches and bananas, our experience has shown that the ripening process can be extended by 7–14 days While we see considerable variability in commercial fruit, we have always seen delayed ripening in those fruit exposed to catalyst derived from induced cells of R. rhodochrous DAP 96253.
While we are not at a point in our research where we would categorically state the mechanism of action of delayed ripening, we are struck by the extent to which the biochemistry of R. Rhodochrous DAP 96253 overlaps and is integrated into the biochemistry of climacteric fruits. Our perspective has focused on that of the bacteria and the atmosphere of the bacterial-plant microcosm. Future work will reflect that catalyst performance is influenced by the nature and the variety of the fruit.
Footnotes
Acknowledgments
We wish to thank the following for providing partial support for our research: State of Georgia Department of Agriculture, Georgia Research Alliance (Atlanta), Georgia State University Research Foundation (Atlanta), Georgia State University Office of Research (Atlanta), Georgia Peach Council (Fort Valley), Georgia Fruit and Vegetable Growers Association (LeGrance), and multiple industry sponsors.
Author Disclosure Statement
All inventions related to the delayed ripening of fruit discussed in this manuscript are the sole property of the Georgia State University Research Foundation. There is no conflict with any commercial entity.
