Abstract
Objective:
To describe the endocannabinoid system, cannabinoid receptors, its agonism/antagonism in neurodegenerative disorders.
Introduction:
The endocannabinoid system (ECS) represents a highly complex biological system that synthesize and degrade the endocannabinoids. Cannabinoids are endogenous lipid-based neurotransmitters, work in retrograde manner, that bind to a class of G protein-coupled receptors (GPCRs) known as CBRs, of two types: CB1R and CB2R.
Results:
The literature review suggests that the endocannabinoids regulate cognitive processes, fertility, pregnancy, immune homeostasis, appetite, pain, pain sensation, memory, and behavior. Both CB1 and CB2 couple mainly to inhibitor G proteins and having same pharmacological influences as other GPCRs. Thus, selective/partial agonism, functional selectivity/activity, and inverse agonism all play vital activities in determining the cellular response to specific CBR and ligand interactions.
Conclusion:
Improved therapeutic modalities for neurological disorders and the development of novel molecular targets for therapeutic intervention of neuronal diseases require a full understanding of the cross-talk between CB1, cannabinoids, and pathophysiology of neurodegenerative disease.
Introduction
The endocannabinoid system (ECS) is an intriguing cell-signaling system that typically comprises endocannabinoids, cannabinoid receptors (CBRs) and involves biosynthetic machinery for their biosynthesis and degradation. Endocannabinoids, also called endogenous cannabinoids, comprised CBRs and the enzymes for their synthesis and degradation. 1 Anandamide (AEA) and 2-arachidonoylglycerol (2-AG) are well-characterized examples of endocannabinoids.
Several other lipid ligands, chemically similar to AEA and 2-AG, act like endocannabinoids. 2 The role of ECS in biological systems is still not fully understood. 3 The ECS regulates a multitude of biological functions and processes, which include sleep, mood, appetite, fertility, and pain.4–6 The ECS participates in the regulation of motor activity, neurogenesis, synaptic plasticity, and inflammatory response, maintains positive energy balance, and phases of memory processing. 7
The major physiological function of the cannabinoid signaling system is to modulate neuronal behavior. 8 Endocannabinoids regulate these biological activities through their interaction with endocannabinoid receptors (CB1 and CB2). Many, if not all, of the CBR type 1 (CB1 receptor) and its endogenous lipid ligands regulate brain activities (endocannabinoids). 9 The CB1 receptors are found throughout the brain, and their distribution reflects the ECS's complexity and range of activities in neuronal activity. 10 Unlike CB1, the CB2 receptor is mainly expressed in immune cells and regulates immune and inflammatory responses. 11
Endocannabinoids
These are classes of lipid mediators that include amides, and esters of long-chain polyunsaturated fatty acids. The primary endogenous agonists of CBRs are AEA and 2-AG, which can imitate various pharmacological outcomes of -9-tetrahydrocannabinol, the active ingredient of Cannabis sativa. The mechanisms that cause neuronal and non-neuronal cells to synthesize and release AEA and 2-AG are yet unclear.
Instead, cellular absorption of AEA is known to be limited via a particular membrane transporter, followed by an intracellular breakdown by a fatty acid amide hydrolase (FAAH). 12 Cannabis is a highly complex substance that contains terpenoid-like compounds referred to as phytocannabinoids. The major psychoactive phytocannabinoid found in cannabis is Δ(9)-tetrahydrocannabinol (THC), which exerts its pharmacological effects mainly via CB1 and CB2 receptors.
After the discovery of CB1 and CB2, many novel cannabinoids were chemically synthesized, which subsequently led to the identification of the endocannabinoids (or endocannabinoid) system.11,13 Within the human body, endocannabinoids are enzymatically produced and released on demand. It is now clear that many phytocannabinoids including cannabidiol (CBD) and cannabinol (CBN) can modify the functional and regulatory activities of the central nervous system through their interaction with CB1R and CB2R (Table 1). 14
Major Components of the Endocannabinoid System
ECS, endocannabinoid system; FAAH, fatty acid amide hydrolase; NAPE, N-acylphosphatidylethanolamine.
In this review article, we attempted to understand the role of the ECS and approaches to its targets and selectivity to treat or alleviate neurological disease processes. 15
Biosynthesis and degradation of endocannabinoids
AEA and 2-AG, unlike traditional neurotransmitters and neuropeptides, are generated on demand by receptor-stimulated cleavage of lipid precursors. This is under the strict control of FAAH, which breaks down AEA, and monoacylglycerol acid lipase (MAL), which typically breaks down 2-AG. The N-arachidonoylphosphatidylethanolamine (NArPE), AEA precursor, is formed from arachidonic acid (AA) via a calcium-dependent N-acyltransferase (trans-acylase). The N-acylphosphatidylethanolamine (NAPE)-specific phospholipase D then cleaves NArPE, releasing AEA and phosphatidic acid. However, because their precursors, N-acylethanolamine phospholipids, are widespread elements of animal and human cells, tissues, and body fluids, a similar technique can be used to make the other cannabimimetic NAEs.
The AEA's biological action is stopped when it is removed from the extracellular space, which is accomplished in two steps:
intracellular breakdown by a FAAH Cellular uptake by a high-affinity transporter.
The AEA membrane transporter (AMT) is bidirectional and independent of energy and sodium with facilitated diffusion. The CB1 receptors agonism or activation by AEA produces Nitric oxide, which then activates AMT, so driving the AEA clearance. This AEA's stimulation of CB1 receptors results in AEA's activity termination and potential to operate in reverse, which expels AEA outside the cell.
FAAH, a membrane-bound enzyme having esterase activity, regulates AEA absorption in cells by maintaining an inward concentration gradient that promotes AEA diffusion via AMT. The FAAH enzyme is primarily present in mitochondrial fractions and is critically involved in AEA degradation.
The FAAH enzyme has three domains, a transmembrane domain, a serine and glycine-rich domain, and proline-rich domain. These three domains regulate protein oligomerization at the N terminus; the typical amidase signature sequence is homologous to the class II SH domains, respectively. Several synthetic and natural cannabinoids have been identified that can be used for pharmacological intervention of ECS. 16 However, there is a need to understand the more precise mechanism of how CBS functions at the molecular level.
Here, we attempted to understand the mechanistic role of Cannabinoids in neuroprotection as well in pathological pain. A better understanding of the cannabinoid-based signaling system may provide a rationale for improving existing neuroprotective strategies and identifying novel targets for therapeutic intervention of neuronal disorders. This may significantly improve the therapeutic applicability of targeting the ECS, for managing neurodegeneration, pain, and mental illness.
Cannabinoid receptors
The CB1 CBRs, which are mostly found on central and peripheral neurons, the CB2 CBRs, which are mostly found on immune cells, the non-CBRs, and the vanilloid receptors are the molecular targets of AEA and 2-AG. The CBRs belong to the class G protein-coupled receptor (GPCR) family, signal through inhibitory Gαi/o heterotrimeric G proteins, and interact with β-arrestins. 17 CBR 1 (CB1R, encoded by the CNR1 gene) is a G protein-coupled CBR in humans and is expressed in both the central nervous and peripheral nervous systems. 18
CB1 is activated in response to endocannabinoids and plant-based phytocannabinoids (such as THC, which is a key component of cannabis) and THC-derived synthetic analogs. 19 In humans, the primary agonist of CB1 is AEA. 20 Cannabis has an excellent spectrum of therapeutic effects such as anti-nociceptive, anti-inflammatory, and anticonvulsant, which has largely limited its medical application. 21 Despite varying etiologies, neurodegenerative disorders share key characteristics such as neuro-inflammation, excitotoxicity, deregulation of intercellular Ca2+ homeostasis, mitochondrial dysfunction, and disruption of brain tissue homeostasis. 22
CB1 and CB2 receptor signaling participates in the control of Ca2+ homeostasis, trophic support, mitochondrial dynamics, and inflammatory situations. Pharmacological intervention of endocannabinoid signaling is thus a valid therapeutic option in neuro-degeneration. 23
CB1R signaling and its effects depend on cellular and subcellular localization and are pleiotropic in nature. The brain is a very complex system but the intrinsic components of the brain behave as static that combines to perform its function. 24 CB1R activates as well as inhibits complex series of intracellular cascades and these effects can happen to any receptor at different locations.
These effects largely emerge as CB1R can regulate different G-proteins, depending on their functional state, localization, and so on. 25 CB1R expression levels are variable among different locations, for example, CB1R is expressed high in cortical neurons, expressed lower in cortical glutamatergic neurons, and has no expression in hypothalamic regions. TheCB1R agonist, in intrinsic signaling, increases its expression while recruiting G-proteins. 26
This should happen in the CB1R neuroprotective strategy; however, due to its pleiotropic nature, this might not happen. Some studies have shown that the hypothalamus (CB1R has very low or null expression) displays higher levels of CB1R significantly and even much higher expression. 27 Some CB1R signaling, for example, cortical GABAergic neurons express high CB1R levels but have very low efficient CB1R signaling, thus considering silent CB1R expression. 28 Some more recent studies have shown that even if the CNR1 gene is removed from mice, still they express CB1R much more stronger, and only activation of G-proteins got decreased.
This suggests that CB1R signaling is G-protein dependent. 25 The causes for these differences are not known at this moment and need further intervention to CB1R signaling. CB1R can also propose to form homo/hetero dimers with CB2R and these interactions can modify its signaling, with new therapeutic strategies.
Glutamate (l -glu) signaling in brain
Glutamate serves as the primary excitatory neurotransmitter in almost all nervous system functions and plays a role as a free amino acid in almost all nervous system functions.
In addition to iGluRs, there are eight isoforms of metabotropic glutamate receptors that belong to the G-protein-coupled receptor family and signal through diverse second messenger systems rather than forming ion channels.
31
A postsynaptic excitatory potential is generated as a result of
Extrasynaptic NMDA receptor stimulation suppresses CREB activity, whereas synaptic NMDA receptor activation stimulates it.
35
Excitotoxicity is caused by the overactivation of glutamate receptors, which results in a loss of post-synaptic structures such as dendrites and cell bodies in neuronal cells.
36
Extracellular
As we know, the maintenance of neuronal and astroglial membrane potentials is energy-dependent, and the sudden loss of energy supply caused by the cessation of blood flow to the brain causes them to break down (Fig. 1).38,39 The release of vesicular
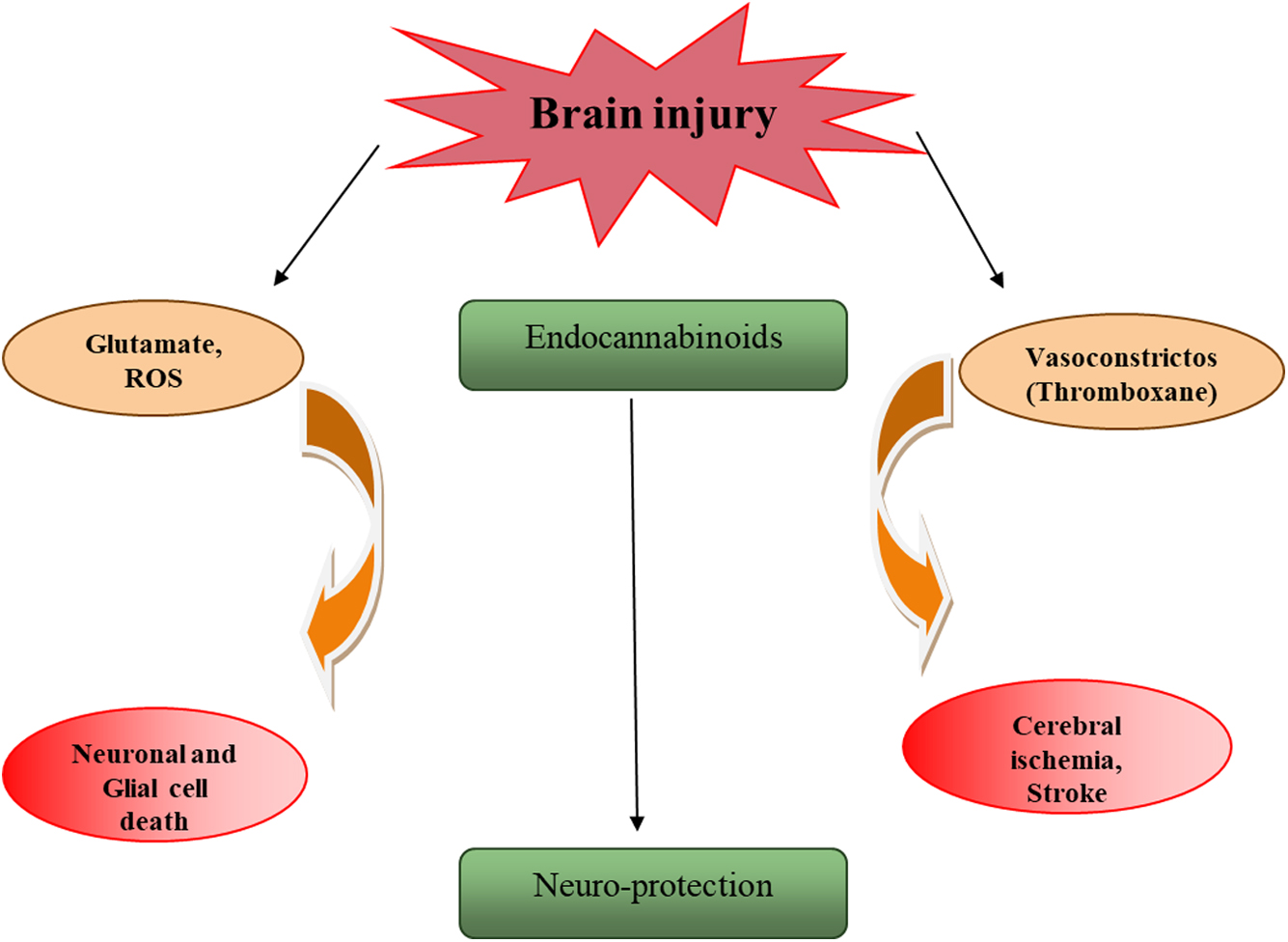
Glutamate, cytokines, and ROS are released after a brain injury, causing neuronal and glial cell death. After a brain injury, vasoconstrictors such as thromboxane are raised, which contributes to ischemia. After trauma, endocannabinoids can rise, inhibiting the production of glutamate, reactive oxygen species, and cytokines. Endocannabinoids play a role in neuroprotection through this method. ROS, reactive oxygen species.
Link between the ECS and neuropathic pain
Chronic neuropathic pain affects nearly 7% to 10% of the general population. Pain of this type shows higher incidence in aged people and causes a huge burden on the global health care system. 41 Patients with neuropathic pain are less productive and developmental disorders. 42 The ECS is an important neurotransmitter system that has recently received tremendous attention due to its role in the control of neuropathic pain. 43
The components of this system are present in neuron cells and, as such, play a critical role in the development of neuropathic pain. 44 Several preclinical studies have shown that the ECS could be targeted for the management of pain. 45 Many cannabinoid agonists have shown analgesic effects in multiple neuropathic pain models. Many specific targets exist in this system that can be used to develop more efficacious and biologically safe therapeutic agent. 46
Likewise, several cannabinoids have entered into human intervention trials and have been significantly found to reduce pain in peripheral neuropathies. Many of these trails, however, failed due to a lack of required standards of quality.47,48 Therefore, more refined clinical trials with an appropriate number of patients, and control subjects are needed to have an adequate profile of the effectiveness and safety of cannabinoids in the management of neuropathic pain. Neuropathic pain involves spontaneous pain, and signs of sensory loss accompanied by the gain of function such as allodynia and hyperalgesia. 49
Pharmacology of Biologically Important Cannabinoids
The modulators of the CB1 and CB2 receptor systems (Fig. 2) offer a multitude of therapeutic opportunities for the treatment of neurological disorders. AEA and 2-AG are a class of endocannabinoids (ECs) that activates CBR type-1 (CB1), type-2 (CB2) agonist, partial agonist as well as antagonist, to stimulate their action (Table 2). Many inhibitors for the main EC hydrolytic enzymes FAAH, monoacylglycerol lipase (MAGL), COX2, and α,β-hydrolase domain-6 (ABHD6) and α,β-hydrolase domain-12 (ABHD12) have been synthesized and characterized.50,51

Structure of few pharmacologically important synthetic cannabinoids.
Behavior of Few Biologically Important Cannabinoids at the Cannabinoid Receptor Site
Bold indicates the activity specific to CB1/CB2 receptor.
2-AG, 2-arachidonoylglycerol; Δ9-THC, Δ(9)-tetrahydrocannabinol.
Delta 9-tetrahydrocannabinol
Δ9-THC is the major psychoactive component of marijuana. Marijuana is a drug that is addictive and consists of dried leaves, flowers, stems, and seeds of the C. sativa or Cannabis indica plant.51,52 Δ9-THC is considered the principal psychoactive component of cannabis. 53 The psychotropic effects of this compound are mediated primarily through its interaction with CB1 receptors (Fig. 3).54,55

A simplified mechanism of the neuromodulatory function carried out by the endocannabinoid system shows subsequent neurotransmitter exocytosis across the synapse, synthesis of endogenous cannabinoids after postsynaptic receptor activation, and retroactivation of CB1 receptors, which leads to an inhibition of TRPV channels and a reduction in vesicular exocytosis of neurotransmitters. TRPV, Transient Receptor Potential cation channel subfamily V.
CP55,940
CP-55,940 is an excellent example of nonclassical, bicyclic cannabinoids. This drug is a full agonist for both CB1 and CB2. CP 55,940 and WIN 55,212-2 have been shown to induce distinct conformational changes in CB1 receptors on binding. 56
Abnormal CBD
Abnormal CBD (abn-CBD) is a synthetic regioisomer of CBD without producing any psychoactive effects. Unlike other most other cannabinoids, abn-CBD produces vasodilator effects, lowers blood pressure, and induces cell migration, cell proliferation, and mitogen-activated protein kinase activation in microglia. Some actions of abnormal CBD are mediated through other than CB1 and CB2 receptor sites. 57 O-1602 is a synthetic compound that resembles abnormal CBD. It does not have any affinity to the CBRs CB1 or CB2; it serves as an agonist at some other receptors, particularly GPR18 and GPR55. Orphan receptors are major targets for many endogenous cannabinoid compounds, they mediate non-CB1, and non-CB2 effects. 58
CID16020046, an inverse agonist at GPR55, blocks a number of its responses like activation of the immune system. 59 It shows good selectivity over the CB1 and CB2 receptors and has other targets. 60 This compound has anti-inflammatory actions, both GPR55 and CB1 mediated activity. 61
PSB-CB5 (CID-85469571) –This molecule is the selective antagonist, at the orphan receptor GPR18, with an IC50 of 279 nmol/L. 62 GPR18 antagonize GPR55, and work synergistically, therefore, GPR18 antagonist-mediated compounds, are expected to be useful in research. 63
WIN55,212-2
The CBR agonist WIN55,212-2 prevents nigrostriatal cell loss in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) mouse model of Parkinson's disease. 64
Anandamide
AEA, a fatty acid amide, is a CB1 receptor ligand. Arachidonic acid and ethanolamine combine to form AEA. 65 AEA can be present in practically every tissue of a wide variety of mammals. The FAAH enzyme that transforms AEA into ethanolamine and AA is the enzyme that degrades it the most. As a result, inhibitors of FAAH cause an increase in AEA levels, which is why they are being studied for therapeutic purposes.
URBB47
URB447 is a synthetic CB1/CB2 receptor ligand substituted pyrole residues numerously in all positions. It has a good stereoselectivity (CB2/CB1 ratio 9 < 10) and acts as a CB1 antagonist and CB2 agonist. It has periphery CB1 antagonism, so it is able to lower food intake and body-weight gain in mice, an effect also shown by the CB1 antagonist rimonabant (SR141716A). 66
SR141716
SR141716 is a selective CB1 receptor antagonist. This molecule has been shown to increase the mRNA expression of Acrp30 in adipose tissue of obese fa/fa rats and in cultured adipocyte cells. 67 The cannabinoid CB1 receptor antagonist rimonabant (SR141716) inhibits cell proliferation in cultured mouse 3T3 F442A preadipocytes. 68 Neuroprotective CBR antagonist SR141716A prevents the downregulation of excitotoxic NMDA receptors in the ischemic penumbra. 69
SR141716A reduces infarct volume by ∼40% after experimental stroke. Although it is suggested that SR141716A may exert neuroprotection besides its CBR-blocking effect, SR141716A may possess additional intrinsic neuroprotective properties independent of receptor-coupled pathways or due to action as a partial agonist. 70 The CB1 antagonist, SR141716A, is protective in permanent photothrombotic cerebral ischemia. Neurological function correlated favorably to the reduction in stroke size when SR141716A was given as a pre-treatment. 71
UCM707
UCM707, a 3-furyl arachidonoyl analog, is a selective inhibitor of endocannabinoid uptake that potentiates the antinociceptive effects of AEA. UCM707 for the inhibition of AEA, with IC50 of 0.8 μmol/L uptake into human U937 cells. It has a low affinity for FAAH, exhibiting an IC50 value of 30 μmol/L. This molecule acts as an endocannabinoid transporter inhibitor, with the highest selectivity and potency and makes this compound useful in endocannabinoid signaling, having less severe side effects in treating neuronal disorders. 72
UCM707 significantly activates the action of AEA that did produce fewer effects when it was administered alone. 73 UCM707 compound may be considered a promising therapeutic tool, used in combination with various endocannabinoids, in treating neuronal disorders. It also exhibits anti-hyperkinetic activity in a rat model by the application of 3-nitropropionic acid. 74 UCM707, on the other hand, did not protect against the loss of GABAergic neurons caused by the unilateral application of malonate in rats, which is important for testing neuroprotective strategies. 74 One possible explanation is that UCM707 lacks anti-oxidant properties. 75
URB597 (SR141716A)
URB597 is the FAAH inhibitor and behaves as a potent stimulant as it suppressed the sleep recovery period after notably long waking. URB597 has been shown to reverse some addiction-related behavioral and neurochemical effects of nicotine in rats. This molecule is a CB1 CBR antagonist. 76
AM6538
(CB1 receptor antagonist) stabilizes the receptor-ligand complex for crystallographic analysis. It is a derivative of AM251, a widely used rimonabant analog. In contrast to AM251, the synthetic compound, AM6538 shows enhanced affinity for CB1 receptor. 77
Neuroinflammation, Pain, and Neurodegeneration
One important symptom of human disease is pain. Chronic pain is the cause of disability among neurodegenerative diseases. 94 Nociceptive pain occurs due to non-neural damage whereas neuropathic pain is caused by diseases among neuronal disorders, involving the central nervous system. 95 Chronic and non-neuropathic pain (nociceptive pain) are two conditions for which there is a high unmet need in clinical settings.
Although acute pain can be managed easily by using NSAIDS or opioids, the management of chronic pain is often challenging.96,97 Nociceptive pain is perceived by high-threshold sensory neurons and involves the action potential transfer to the spinal cord, with upward transmission to the brain as the warning signal. In contrast, neuropathic pain, which is caused by nerve injury, is characterized by hyperalgesia so that normally innocuous stimuli produce pain. 97
Chronic pain often induces depression that may be characterized by physical and psychological symptoms such as low mood, lack of energy, insomnia, lack of motivation, and so on. These disorders are complex and difficult to treat as they involve neuroinflammation, which is defined as the innate immune response of the nervous system to any injury or neurodegenerative disease. 98
Further, chronic neuropathic pain usually persists for several months, being treatable or identifiable but without the underlying cause. Indeed, only one in four patients experience over 50% pain relief. 99 So far, no single therapeutic target has been identified as being uniquely associated with the development of neuropathic pain. However, endocannabinoids and endovanilloids, as well as enzymes that regulate their energy balance, represent a therapeutic avenue to successful pain treatment. 100
CBRs activated by its selective/partial agonists modulate pain thresholds, inhibit pro-inflammatory molecules, and have synergistic effects with the endogenous opioid system, therefore helping in the treatment of neuropathies.100,101 During the past several years, it has become clear that for many years, it was considered that cannabinoids, the chemical components of the cannabis plant, induce analgesia by activating certain receptors throughout the body, particularly CB1, which is found mostly in the CNS, and CB2, which is found primarily in immune cells. 102
Therapeutic Potential of Cannabinoids
The therapeutic role of cannabinoids and the ECS have introduced various innovative strategies, based on several approaches. The belief that perhaps the ECS is engaged in emotional behavior, particularly depression, is based on the fact that C. sativa use has dramatic impacts on mood in humans. This system was first identified as the target of THC, a hallucinogenic component of C. sativa (THC). C. sativa has been utilized for mood-raising and euphoric effects by cultures all over the world for age. 103
Endocannabinoid signaling is a significant regulator of emotions in humans. Endocannabinoids function as retrograde messengers, preventing the release of neurotransmitters through presynaptic CB1 receptor. The ECS further plays a role in regulating neuron survival by activating the CB1 receptor, which shields neurons against sudden brain damage. Therefore, it has been demonstrated that activating the CB1 receptors protects neurons from short-term brain damage as well as from neurodegenerative disorders. The discovery that the brain overproduces endocannabinoids on damage supports the idea that cannabis neuroprotective effect depends on the regulation of glutamatergic neurotransmission as well as other different mechanisms.
The neuroprotective effects of cannabinoids may be influenced by the coupling of neuronal CB1 receptors to cell survival pathways such as the phosphatidylinositol 3-kinase/Akt and extracellular signal-regulated kinase pathways. This cross-talk between CB1 receptors and growth factor tyrosine kinase receptors can be partially responsible for these pro-survival signals. Inhibitors of eCB transport and degradation as well as synthetic and endogenous CBR agonists have both been used in these investigations. THC, AEA, and 2-AG treatment lessens neuronal loss and brain damage in excitotoxicity and ischemia models.
In addition, management of the WIN-55,212-2, a synthetic CBR agonist, the provision of neuroprotection in a model of neonatal hypoxia brain damage is caused by ischemia. 104 The endocannabinoid deficit theory suggests that depression will be accompanied by abnormalities in endocannabinoid signaling; both rodent models and human research support this idea. 105
The ECS in Parkinson's disease
Parkinson's disease, a neurodegenerative illness with both motor and non-motor issues, has resting tremor, muscular rigidity, and bradykinesia as the three main symptoms. 106 Some non-motor symptoms of the disease include autonomic alterations, cognitive deterioration, perceptional problems, depression, and sleep disorders. 107 Parkinson's disease affects an estimated 6.3 million people around the world. The average age of onset is over 60, however roughly 1 out of every 10 people develop the disease before they are 50. 108
Very few brain stem cells as well as neurons in the pars compacta of the substantia nigra show pathological indications of dopaminergic neuron degeneration with Lewy Bodies. 109 Glutamatergic, cholinergic, dopaminergic, and GABAminergic neurons all have CB1 receptors in their synapses. TRPV1, a member of the Transient Receptor Potential cation channel subfamily V, is an inotropic cannabis receptor that is also strongly expressed in dopaminergic areas. 110
Disease progression can produce excitotoxicity, oxidative stress, and glial activation, and its downregulation could be essential in the development of the disease and progression of Parkinson's disease. With the progression of the illness, the ECS is upregulated. 111 This upregulation, which has been observed in both animal and human studies, could be regarded as either causative or compensation for a loss in dopaminergic signaling in the basal ganglia. 112
Complex interactions can occur between the ECS and dopamine, acetylcholine, and glutamate signaling. 113 In the short term, dopamine availability in the prefrontal cortex is increased by CB1 receptor agonists. Long-term agonist activation, on the other hand, causes a decrease in dopamine activity, which may be linked to motivation and cognitive decline. 114 A cholinergic depressive effect has been linked to high dosages of CB1 agonists, which could be linked to cognitive difficulties.101,115
ECS in Alzheimer's disease
Alzheimer's disease (AD) is the most common age-related neurodegenerative condition, morphologically characterized by the presence of senile plaques, which causes dementia and progressive cognitive deterioration. It consists of β-amyloid (Aβ) produced by the cleavage of the Aβ precursor protein by β- and γ-secretases. This Aβ triggers plaque formation, hyperphosphorylation of tau protein, accumulates abnormal proteins, and causes disease progression.
Nerve cell exhaustion may be caused by mitochondrial functional defects, reactive oxygen species and RNS increase, and enzyme defects that are involved in energy metabolism. The Endocannabinoid signaling system modulates excitotoxicity, mitochondrial dysfunction, and oxidative stress, in neurodegenerative disorders. 116 Some Cholinesterase inhibitors (donepezil, galantamine) and Memantine, an NMDA antagonist, are two FDA-approved drugs for mild to moderate and moderate to severe AD, respectively. 117
These treatments just alleviate particular cognitive symptoms. 118 Other treatments, such as anti-inflammatory medications, antibodies against peptides, antioxidants, and PPAR agonists, have also been tested as optimistic tactics for the prevention of AD, but they have had minimal success. 119 The ECS is gaining popularity as a prospective source of medications for a variety of neurodegenerative illnesses, including AD. 120 The ECS is involved in the clearance regulation, oxidative stress, inflammation, and acetylcholine (ACh) homeostasis.121,122 CB1 receptors contribute to cannabinoid's effects on learning and memory as well as in cognition, which are reduced in AD.
In Alzheimer's patients, CB1 expression is diminished and upregulation of these ECS elements appears to be a cell-specific process.6,122,123 Cannabinoid autophagy potential or redox balance state was also a likely mechanism underlying the decrease in tau deposition.
Cannabinoids protection in oxidative stress
Mitochondria are required for ATP formation and calcium balance in the cell. As we know, neurons are high-energy cells that rely on oxidative phosphorylation. Cannabis derivatives, particularly CBD, have been shown to have anti-oxidant properties in vitro exposure to toxic glutamate levels. Cannabinoids also interact with neurotropic factors and increase brain-derived neurotrophic factor, which protects cells against excitotoxicity and helps in neurogenesis.
Conclusion
CBRs (CB1 and CB2), the endocannabinoids (AEA and 2-AG), and cannabinoid metabolizing enzymes constitute the ECS. ECS impacts the survival and homeostasis of cells, tissues, and organ systems. Given that endocannabinoid signaling regulates cytokine production from microglia cells, brain development, neurotransmitter release, and synaptic plasticity, any alteration in this signaling cascade leads to the development of neurological disorders.
Many activators and inhibitors of ECS have been developed for therapeutic effects in preclinical models. However, there is a need to develop clinically relevant models to better understand the therapeutic efficacy of therapeutic agents. Depending on the condition and the involvement of CB1 and CB2 in it, many models are available, although with limitations.
Often, targeting CBS for the therapy of neurological disorders is challenging, because cannabinoids activate different receptors and their metabolic pathways are connected with multiple mediators. The existence of other mediators in the ECS (also known as endocannabinoidome) limits the therapeutic targeting of cannabinoid metabolizing enzymes (anabolic and catabolic enzymes).
However, agonists of the ECS and CB1 and CB2 allosteric modulators are being studied intensively in the treatment and management of neurological diseases (such as multiple sclerosis and epilepsy) and neuropathic pain. The goal of this review was to simply show how new observations may be useful regarding CB1R signaling-based neuroprotective strategies that continue to open new perspectives on the complex nature of the brain in neuronal disorders.
With this supposition, we believe that CB1R agonists, with new surprises, await us, with new austere data and can resist these compacted current views. In the evolving discoveries of ECS, it is hoped that this additional material will be incorporated into this brief review.
Footnotes
Acknowledgments
The first author is thankful to University Grants Commission, New Delhi, India for Junior Research Fellowship (UGC-JRF).
Authors' Contributions
S.K.S. and S.T. conceived the idea. S.T. wrote the article with the assistance from S.K.S. All authors contributed to the review and approved the final submitted version.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Their is no funding information associated.
