Abstract
Studies have shown that brain lipid metabolism is associated with biological aging and influenced by dietary and genetic manipulations; however, the underlying mechanisms are elusive. High-resolution imaging techniques propose a novel and potent approach to understanding lipid metabolic dynamics in situ. Applying deuterium water (D2O) probing with stimulated Raman scattering (DO-SRS) microscopy, we revealed that lipid metabolic activity in Drosophila brain decreased with aging in a sex-dependent manner. Female flies showed an earlier occurrence of lipid turnover decrease than males. Dietary restriction (DR) and downregulation of insulin/IGF-1 signaling (IIS) pathway, two scenarios for lifespan extension, led to significant enhancements of brain lipid turnover in old flies. Combining SRS imaging with deuterated bioorthogonal probes (deuterated glucose and deuterated acetate), we discovered that, under DR treatment and downregulation of IIS pathway, brain metabolism shifted to use acetate as a major carbon source for lipid synthesis. For the first time, our study directly visualizes and quantifies spatiotemporal alterations of lipid turnover in Drosophila brain at the single organelle (lipid droplet) level. Our study not only demonstrates a new approach for studying brain lipid metabolic activity in situ but also illuminates the interconnection of aging, dietary, and genetic manipulations on brain lipid metabolic regulation.
Introduction
Aging is an almost universal phenomenon for living organisms and is associated with progressive deterioration of physiological functions and increased susceptibility to diseases and death. 1 Lipid metabolism has been associated with aging and age-related diseases. 2 The brain is a lipid-enriched organ, second only to the adipose tissue. 3 Previous studies reported a decline of omega-3 fatty acids and an increase in lipid peroxidation in aged brains.4,5 Dietary deficiency of omega-3 fatty acids may accelerate neurodegeneration,6,7 indicating the critical role of lipid metabolism in brain function. Genome-wide association study (GWAS) suggests that lipid metabolism changes in the brain may have impact on aging and longevity. 8
One group of brain lipids include sphingolipids, glycerophospholipids, and cholesterol, which serve as critical building materials for cell and organelle membranes, and are involved in cell signal transduction. 9 Another group of lipids that play nonstructural roles in the brain are triacylglycerol (TAGs) and steryl esters, which are wrapped in lipid droplets (LDs) for energy storage. Electron microscopy study demonstrated that LDs in the nervous system are mainly localized in glial cells.10–12 Many studies revealed valuable insights into the role of LDs play in aging and neurodegeneration. For example, reactive oxygen species (ROS) induced by mitochondrial dysfunction increase the activities of c-Jun-N-terminal Kinase and sterol regulatory element binding protein, leading to the accumulation of LDs in glial cells before or at the onset of neuronal loss.
LD accumulation in glial cells promotes neurodegeneration and represents as an early transient indicator of neuronal death. 13 LDs in glial cells can also protect polyunsaturated fatty acids in brain membranes from oxidation to maintain the proliferation of Drosophila neural stem cells under oxidative stress. 14 The accumulation of LDs in microglia has been reported involved in age-related and genetic forms of neurodegeneration. 12 These lipid-droplet-accumulating microglia, characterized as pathological glial cells, are defective in phagocytosis, produce high levels of ROS and secrete proinflammatory cytokines. The inability to transport lipids to glia for LD formation leads to accelerated neurodegeneration under stress. 15
Despite these efforts, how brain lipid metabolism is dynamically modulated by the aging process has not been fully understood. Alterations in nutrition balance is a notable cause of aging,16–18 but how diets and metabolic pathways regulate brain lipid metabolism remain elusive. One major reason for this is the lack of high-resolution imaging method to track brain metabolic activity in situ. Stimulated Raman scattering (SRS) imaging combined with deuterium water (D2O; DO-SRS) has been used to investigate the metabolic activity in Drosophila fat body.19,20
In this study, integrated with other bioorthogonal probes, for example, deuterated glucose and acetate, we used DO-SRS to investigate brain lipid metabolism at an even small single organelle level (1–2 μm LD) in Drosophila normal aging and longevity models. We discovered that the lipid metabolic dynamics and pathways are influenced and modulated by sex, diet, and insulin/IGF-1 signaling (IIS) pathway manipulation. These findings will project insights into mechanisms underlying age-related neurodegenerative diseases.
Results
Label-free SRS imaging visualized dynamic changes of lipid metabolism in Drosophila aging brain
As one of the classic aging model organisms, Drosophila has a short lifespan, shares ∼60% genes with humans (including about 75% of those linked to human diseases), and offers powerful genetic and molecular tools to finely dissect the gene functions and physiology. 21 Drosophila brain with its relatively small and simple structure but functionally analogous to mammals has the main advantage to be metabolically analyzed as one whole intact tissue. Drosophila brain can be roughly divided into two compartments: the central brain (CB) and the optic lobe (OL). Each compartment is subdivided into anatomically discrete cortex and neuropil regions (Fig. 1A).
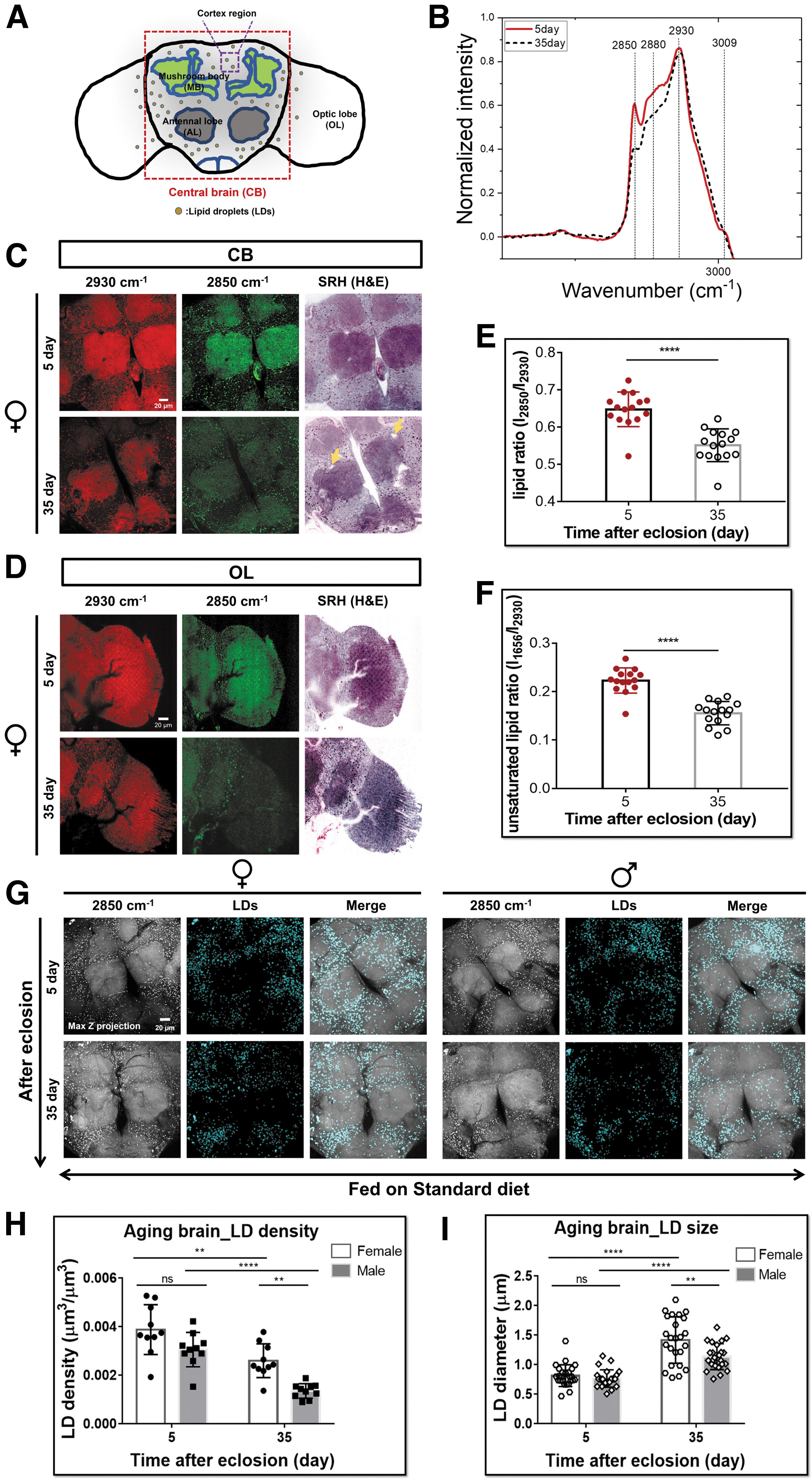
Based on its chemical selectivity, SRS can be used to visualize the subcellular localization of different types of molecules in cells and tissues without any labeling. To investigate the metabolic changes of Drosophila brain, we first used label-free SRS imaging and Raman spectroscopy to detect and track the endogenous lipids and proteins of the whole-mount brain during Drosophila aging process. The Raman spectra we collected displayed a dramatic reduction of the peak intensity at 2850 cm−1 (CH2 from lipids) in 35-day-old brains (Fig. 1B, E and Supplementary Fig. S1A). 22 Consistently, SRS imaging showed the global lipid signal (green channel at 2850 cm−1) was markedly weak in both the CBs and OLs of aged brains (Fig. 1C, D and Supplementary Fig. S1C, D).
This lipid reduction phenotype is consistent with that found in aged mice and human.12,23,24 In addition, the normalized abundance of unsaturated lipids (peak at 1656 cm−1) also showed a significant reduction in the aged brains (Fig. 1F and Supplementary Fig. S1A, B). Of note, stimulated Raman histology (SRH) images were generated from protein (2930 cm−1) and lipid (2850 cm−1) SRS channels, which mimicked histological hematoxylin and eosin staining. 25 SRH images clearly displayed structural changes in old Drosophila brains. Compared with young brains, old brains demonstrate increased vacuoles-like structures, shape alterations, and blurred boundaries in the neuropils (the highly dense compartments of CBs: mushroom bodies and antennal lobes; Fig. 1C, D and Supplementary Fig. S1C, D).
We noticed the lipid signal was dominantly located in the dot-like structures in the brain cortex regions (Fig. 1C, D and Supplementary Fig. S1C, D), which reminded us of brain LDs.10,12–14 We thus co-registered BODIPY staining with SRS imaging and verified these lipid signals were mainly from LDs (Supplementary Fig. S1E). The 3D high-resolved SRS imaging showed that these LDs were 1–2 μm in diameter and mainly localized on the surface of brain cortex (Supplementary Fig. S1F, G). 26 We then imaged and quantified the changes of brain LD density and size during aging in these regions (Fig. 1G–I). We found the LD density was decreased whereas the size was increased with aging in both females and males. Intriguingly, the LD density was consistently higher and LD size was increased more in females during aging than those in males. It indicates that brain lipid metabolism was altered in an age- and sex-dependent manner.
SRS imaging revealed LDs mainly localized in the glia of adult brain
LDs are dynamic organelles with heterogeneous size and location according to various contexts. 27 Previous studies showed that LDs in Drosophila larval and mouse brains were mainly localized in glia and consisted of TAGs.12,14 To examine the subcellular distribution of LDs in Drosophila brain, we used glia-specific Gal4-driven GFP to coregister with the SRS detected LDs, and found they were colocalized (Fig. 2A).
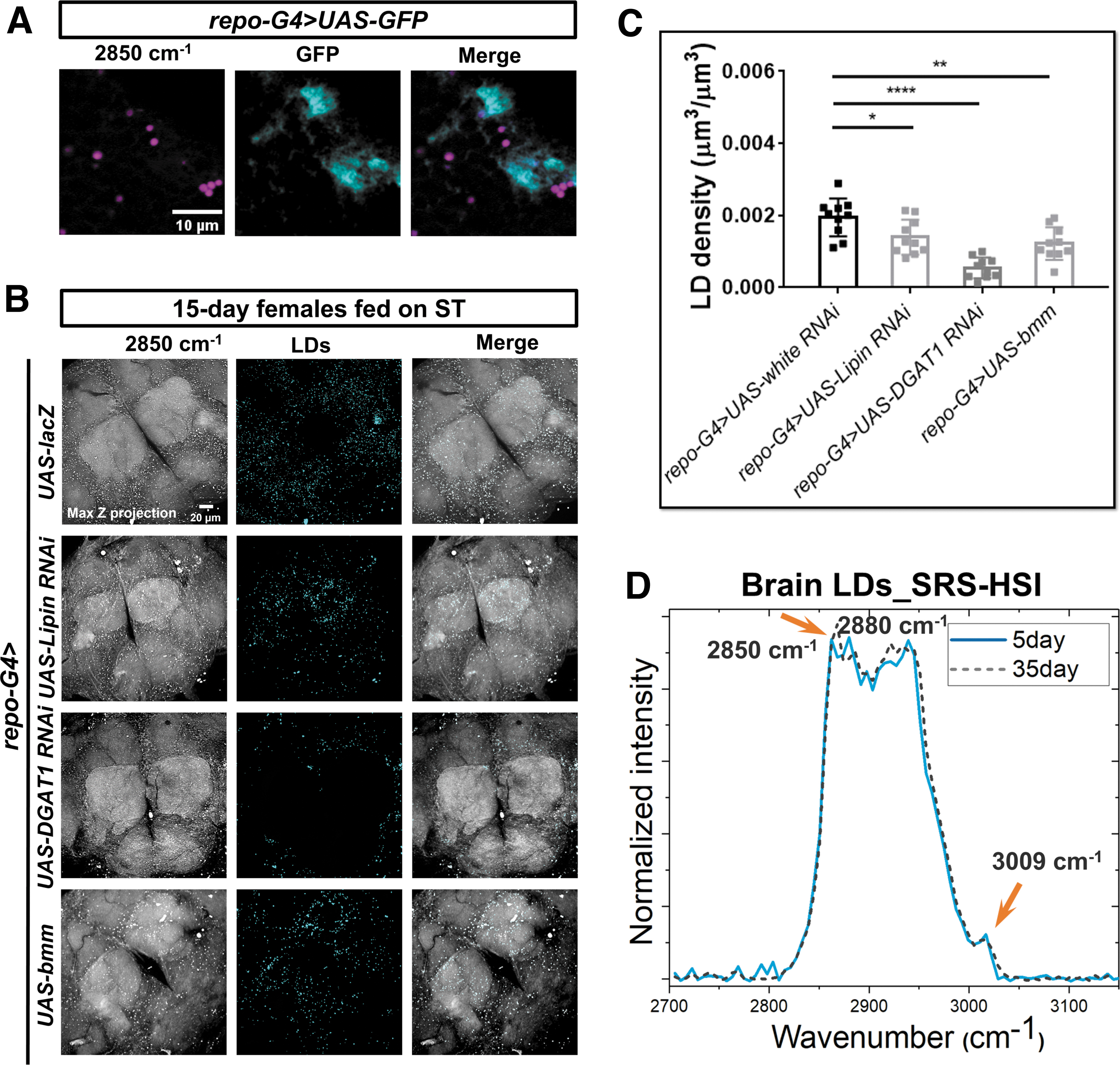
To illuminate the constitution of brain LDs, we manipulated the expressions of three key enzymes in TAG synthesis and mobilization specifically in glia, including Lipin (3-sn-phosphatidate phosphohydrolase), diacylglycerol acyltransferase 1 (DGAT1), and brummer (bmm, the fly homologue of adipose triglyceride lipase [ATGL]). We found that the brain LD density was significantly reduced in flies with Lipin or DGAT1 knockdown, or bmm overexpression (Fig. 2B, C), verifying that TAGs were the main contents of LDs, and further confirming these LDs were dominantly localized in glia.
The lipid contents inside LDs can vary greatly between cell types or even same type under different (age) contexts. 27 Any changes of LD content could be sensitively delineated by the shape changes of Raman spectra. 28 We, therefore, conducted SRS hyperspectral imaging (SRS-HSI) of single brain LD and compared LD contents in Drosophila at different ages and sexes (Supplementary Fig. S2). Dominant Raman peaks were found at 2850, 2880, 2930, and 3009 cm−1, respectively, which were consistent with those peaks in the spontaneous Raman spectra collected from a small volume (several μm3) of the whole brain tissues (Figs. 1B and 2D; Supplementary Fig. S1A).
SRS-HSI collects signal from individual pixel, which prevents the extraction of LD signal from neighboring cellular structures. LD spectra derived from SRS-HSI show no significant differences between young and aged brains, except for a slightly higher peak intensity at 2850 cm−1 in old brains, correspondingly, a slightly higher lipid to protein ratio (Fig. 2D). This is consistent with the larger LD size we previously observed in the old brains—since protein abundance is associated with LD surface area, it reduces when LD grows larger (reduced surface area to volume ratio), which leads to a higher lipid to protein ratio.
DO-SRS imaging revealed brain lipid metabolic activity gradually decreased with aging
Remodeling of LD size is regulated by lipogenesis and lipolysis.29,30 To understand the mechanism underlying dynamic changes of brain LDs, we examined the lipid metabolic activity at single LD level during fly aging by using DO-SRS imaging. 19 Once entering the body, D2O will be incorporated into brain lipid metabolism, and the deuterium (D)-labeled lipids in LDs can be detected by SRS in situ. After feeding flies at different ages (0, 10, 20, 30, and 40 days posteclosion) with diets containing 20% D2O for 5 days (Supplementary Fig. S3A), we imaged brain LDs and quantified lipid turnover by the ratio of CD signal (newly synthesized lipids) to CH signal (total lipids) collected at 2143 and 2850 cm−1, respectively.19,20
DO-SRS imaging revealed strong time-dependent deuterium incorporation from D2O-containing dietary water into LDs (Fig. 3A and Supplementary Fig. S3B), suggesting that de novo lipogenesis contributes neutral lipid cargo to LDs. DO-SRS images also showed nonuniform distribution of newly synthesized lipids inside individual LDs (Fig. 3A), which is consistent with those observed in Drosophila fat body. 20
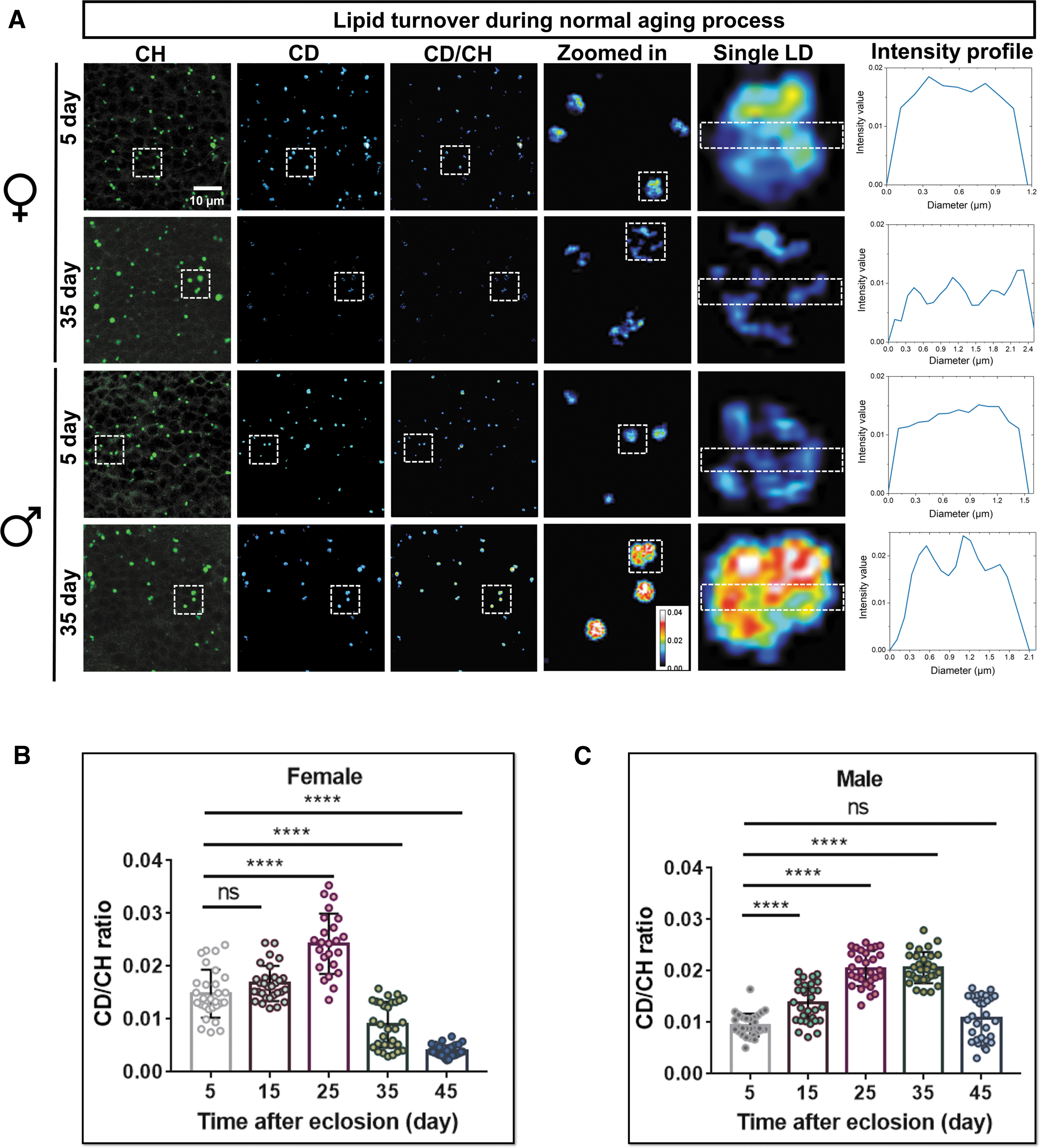
Quantification results displayed the same trend of lipid turnover rate (CD/CH ratio) changing with aging in both female and male flies (Fig. 3B, C). It gradually increased in young flies (5–15 days), to a maximum in mid-aged (15–35 days), and then declined in aged flies (35–45 days). Nevertheless, lipid turnover also displayed sexual differences during aging (Supplementary Table S2). It reached a maximum at an earlier stage in female flies than in males (25 days vs. 35 days), and the turnover rates were consistently higher in young and mid-aged females but lower in 45-day ones. Together, these indicate earlier and more active lipid metabolism in females, whereas males have the opposite trend during aging. It again verified the sex dimorphism in brain metabolism.
LD is a highly dynamic organelle, the inside lipid turnover rate measured was the result of the balance between lipogenesis and lipolysis. That is, the deuterium-labeled lipids (CD signal) observed were the net newly synthesized lipids. We measured lipid turnover decreased during aging, whereas LD size was enlarged in aged brains (Fig. 1G–I), suggesting that lipolysis in LDs was compromised. To test this hypothesis, we utilized a starvation assay that facilitated the mobilization of stored lipids. We treated 0-day (young) and 30-day (aged) flies with standard diet containing 20% D2O for 10 days to obtain saturated deuterium incorporation (∼2.7%) for DO-SRS imaging (Supplementary Fig. S3B), and then transferred the flies to 1% agar for starvation.
The CD/CH ratios in brain LDs were examined at 24, 48, and 72 h of the starvation, respectively (Supplementary Fig. S3C, D). The CD-chasing curves indicate that lipid turnover decreased faster in young flies than aged ones in both sexes, and faster in old males than in old females. The latter indicates old male flies had a higher lipolytic rate than age-matched females (52% vs. 76% of initial CD/CH ratio after 72 h starvation), that is, male mobilized more lipids than female. Data together indicated that, compared with young brains, old brains have inert lipid turnover. The dramatic decline of lipolysis in single LD of old flies may lead to the enlarged LD size. These data together indicate brain lipid metabolism is highly dynamic, especially at the young stages, and DO-SRS provides the sufficient sensitivity in the quantification of lipid turnover.
DO-SRS imaging illuminated brain lipid turnover significantly increased under dietary restriction
Dietary restriction (DR) has been considered an efficient approach to promote motor ability and extend healthy lifespan.31–34 Interestingly, DR benefits females' lifespan more than males' lifespan. 35 To investigate how DR modulates brain lipid metabolism in different sexes, we imaged and quantified brain lipid turnover rate in both female and male flies at different ages under DR (Fig. 4A, B). Similar to that in the normal aging process, the lipid turnover rate under DR increased first and then declined along aging.
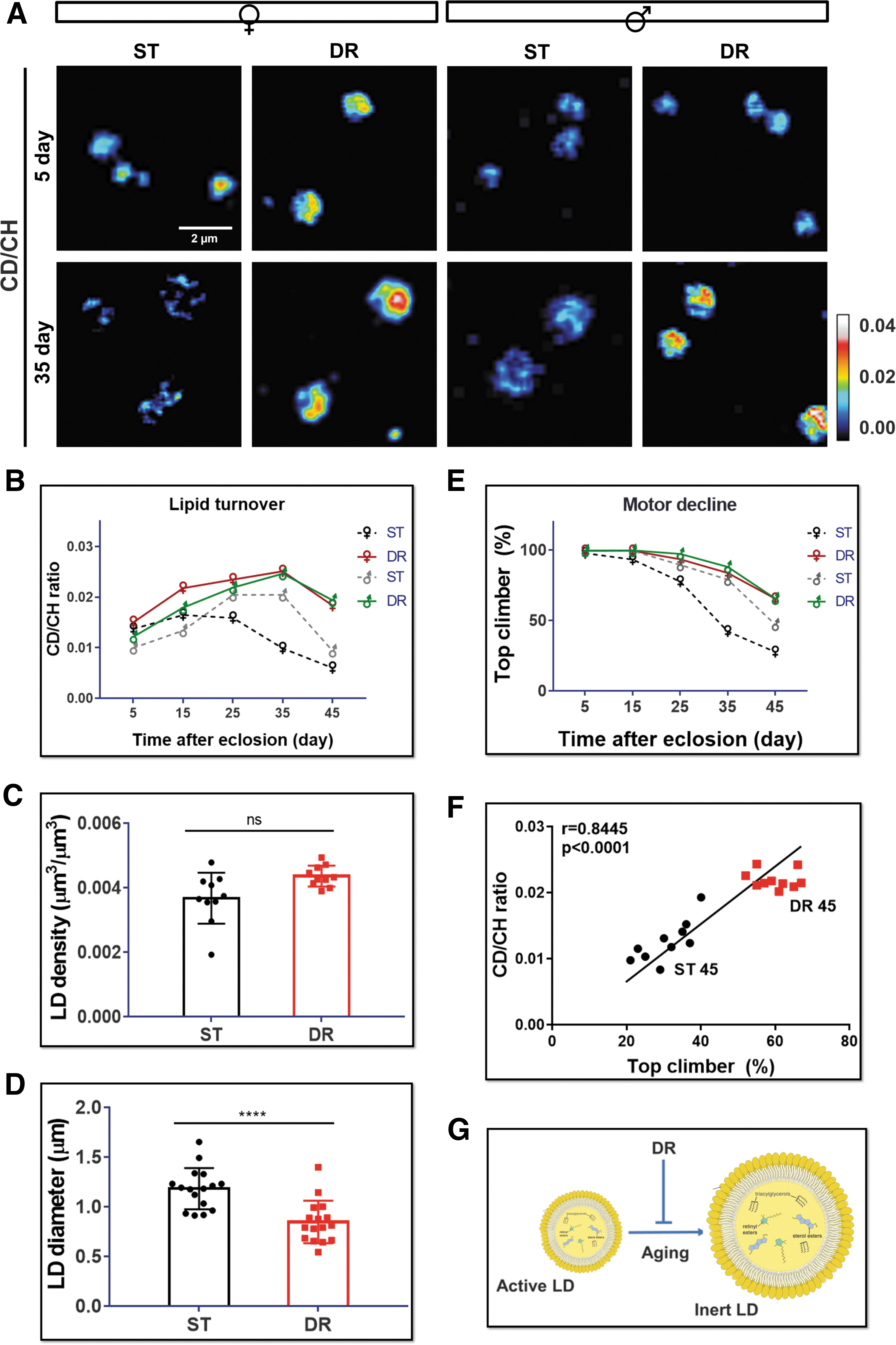
However, DR remarkably enhanced brain lipid turnover in 45-day old animals in both sexes, to a higher extent in females (3.6- and 2.1-fold in female and male, respectively) (Fig. 4B and Supplementary Table S3). That is, DR prevented LD metabolic activity from becoming inert with aging (Fig. 4G). We also detected average LD size was reduced under DR treatment, but no significant changes in LD density (Fig. 4C, D).
We next investigated if the lipid metabolic activity was correlated with brain function. By using the negative geotaxis climbing assay, we were able to assess the locomotor activity, which can partially reflect brain neuronal functions. 36 Consistent with previous studies, we observed an age-dependent decline of locomotor function with normal aging (Fig. 4E). DR decelerated the decline rate of motor function in both females and males (Fig. 4E and Supplementary Table S4). The correlation analysis further verified the association of locomotor function with LD lipid turnover rate in old females understandard diet (ST) and DR treatments (Fig. 4F). We thus concluded that DR might modulate the brain function through regulating brain lipid metabolism.
DO-SRS imaging illustrated reducing IIS activity upregulated lipid turnover
The IIS pathway is a critical regulator of development and aging that controls metabolic homeostasis of carbohydrates and lipids.37–40 Dysregulation of the IIS pathway can lead to insulin resistance, diabetes, and other metabolic disorders.41,42 Reducing IIS activity was reported to mediate DR-regulated lifespan extension in worms, flies, and mammals.37,43,44
To further understand the role of IIS in brain lipid metabolism during aging, we studied Drosophila mutants with reduced functional copies of chico, a homolog of insulin receptor substrate. The heterozygous adults with a loss of functional allele, genotypically chico 1 /+, were shown to have extended lifespan and improved locomotor function.36,45–48 We found that the density and size of brain LDs were reduced significantly in both old female (Fig. 5A and Supplementary Fig. S4A, B) and male chico 1 /+ flies (data not shown). This reduction reflects a decrease of lipid storage in the brain, which could be due to the enhanced lipid turnover.
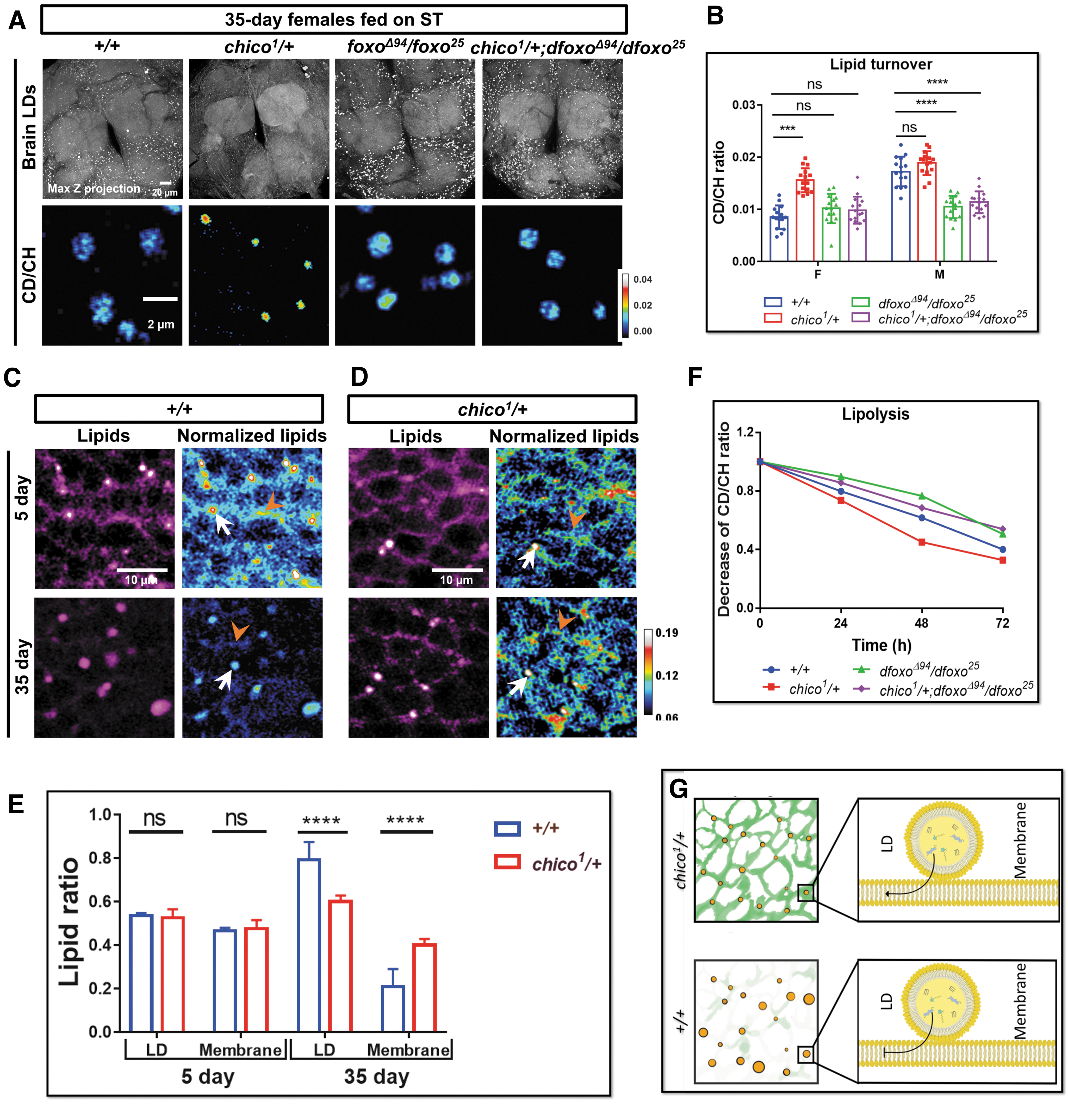
To verify this, we examined lipid turnover dynamics in chico 1 /+ mutant fly brains. DO-SRS imaging and quantification results indicate that, compared with w1118 (+/+) female controls, the CD/CH ratio in LDs increased significantly in both 7-day (by 15%; data not shown) and 35-day (by 85%) female chico 1 /+ fly brains (Fig. 5B). But CD/CH ratio in LDs from males only slightly increased in 7 and 35 days (Fig. 5B and data not shown). These results show that LDs in chico 1 /+ female brain maintained high lipid turnover rate during aging, and IIS reduction changed brain lipid metabolism in a sex-dependent manner.
Of note, label-free SRS images at lipid-related peaks of 2850 cm−1 can also detect the lipids on cell membranes (Fig. 5C, D). The lipid-related peaks were also confirmed by SRS hyperspectral images (the sharp peak at 2850 cm−1 of the yellow spectrum in Supplementary Fig. S4C). Removing lipids by methanol treatment in brain tissue abolished the lipid signal at 2850 cm−1, but the protein signal at 2930 cm−1 remained (Supplementary Fig. S4 C, D). In contrast, removing proteins by proteinase K treatment abolished the peak at 2930 cm−1, highlighting the 2850 cm−1 lipid peak (Supplementary Fig. S4C).
These results verified that the lipid signals on cell membranes were not due to the bleed through from protein signal at 2930 cm−1. Interestingly, we found that lipid content of brain membrane was significantly reduced during normal aging process in the control flies, but it was preserved in the chico 1 /+ fly brains (Fig. 5C–E). Together with the smaller LD size and larger lipid turnover in chico 1 /+ flies, we hypothesized that IIS downregulation promoted lipid exchanges between LD and membranes (Fig. 5G).
Downregulation of IIS pathway has been shown to alter metabolic profiles in Drosophila fat body and muscle, mainly by activating the downstream transcription factor Dfoxo.37,49–53 We then examined how brain lipid metabolism was modulated by dFOXO by using loss-of-function transheterozygous mutants, dfoxoΔ94 and dfoxo 25 Drosophila mutants. 54 Contrary to small LDs observed in chico 1 /+ flies, LDs in dfoxo mutants were significantly enlarged compared with control, especially at the old stages (35–45 days) (Fig. 5A and Supplementary S4A, B), which might be due to the increased lipid synthesis or reduced lipolysis. We first checked D2O labeled lipid turnover in mutants. Interestingly, we found no significant changes of lipid turnover in dfoxo females, but it was significantly reduced in males (Fig. 5B).
The enlarged LD size in dfoxo mutants reminded us of the inert LDs with reduction of lipolysis in the normal aging brains. We thus examined the lipolytic rate in these dfoxo mutants using the starvation assay. The lipid mobilization in dfoxo mutants was remarkably reduced during 72 h starvation in dfoxo mutant flies (Fig. 5F and Supplementary Table S5). We also compared the LD size and lipogenic activities in 0-day (the day flies eclosed from pupae) dfoxoΔ94/dfoxo 25 mutant with control and found no significant differences, suggesting that the change of lipid metabolism was not from development but adult-onset (Supplementary Fig. S4E, F). The data together suggest that enlarged LDs in dfoxo mutants were due to the reduced lipid mobilization during aging process.
We further examined brain lipid metabolism in the combined chico 1 /+; dfoxoΔ94/dfoxo 25 mutant flies. The LD size in these flies was enlarged and the LDs had same trend of lipid metabolic activity as dfoxoΔ94/dfoxo 25 mutants (Fig. 5A, B, and F; Supplementary Fig. S4A, B and Supplementary Table S5), suggesting the beneficial effects of downregulated IIS (chico 1 /+) functions through dFOXO on regulation of brain lipid turnover. The brain turnover gets increased in chico 1 females to a larger extent than males, but it is reduced more in dfoxo males than females, suggesting IIS pathway may be involved in regulating the sex differences of brain lipid metabolism.
Bioorthogonal SRS imaging resolved brain metabolic shifts upon DR and IIS manipulations
Glia-specific IIS inhibition was shown to extend lifespan in Drosophila. 55 To investigate how lipid metabolic activity was regulated by the glial IIS in adult brain (rather than during development), we applied mifepristone (RU486)-inducible glia-specific Gal4 driver GSG907 to modulate IIS activity in adult brain by overexpressing Pten (a lipid phosphatase counteracting PI3K enzymatic activity, GliaGS>UAS-Pten), and expressing mutant InR including GliaGS>UAS-InRDN (the dominant negative form of InR), with GliaGS>UAS-lacZ as the control. Both GliaGS>UAS-Pten and GliaGS>UAS-InRDN flies showed reduced LD density phenotype in the brains (Fig. 6A, B), similar to the chico 1 /+ flies, although to a lesser extent.
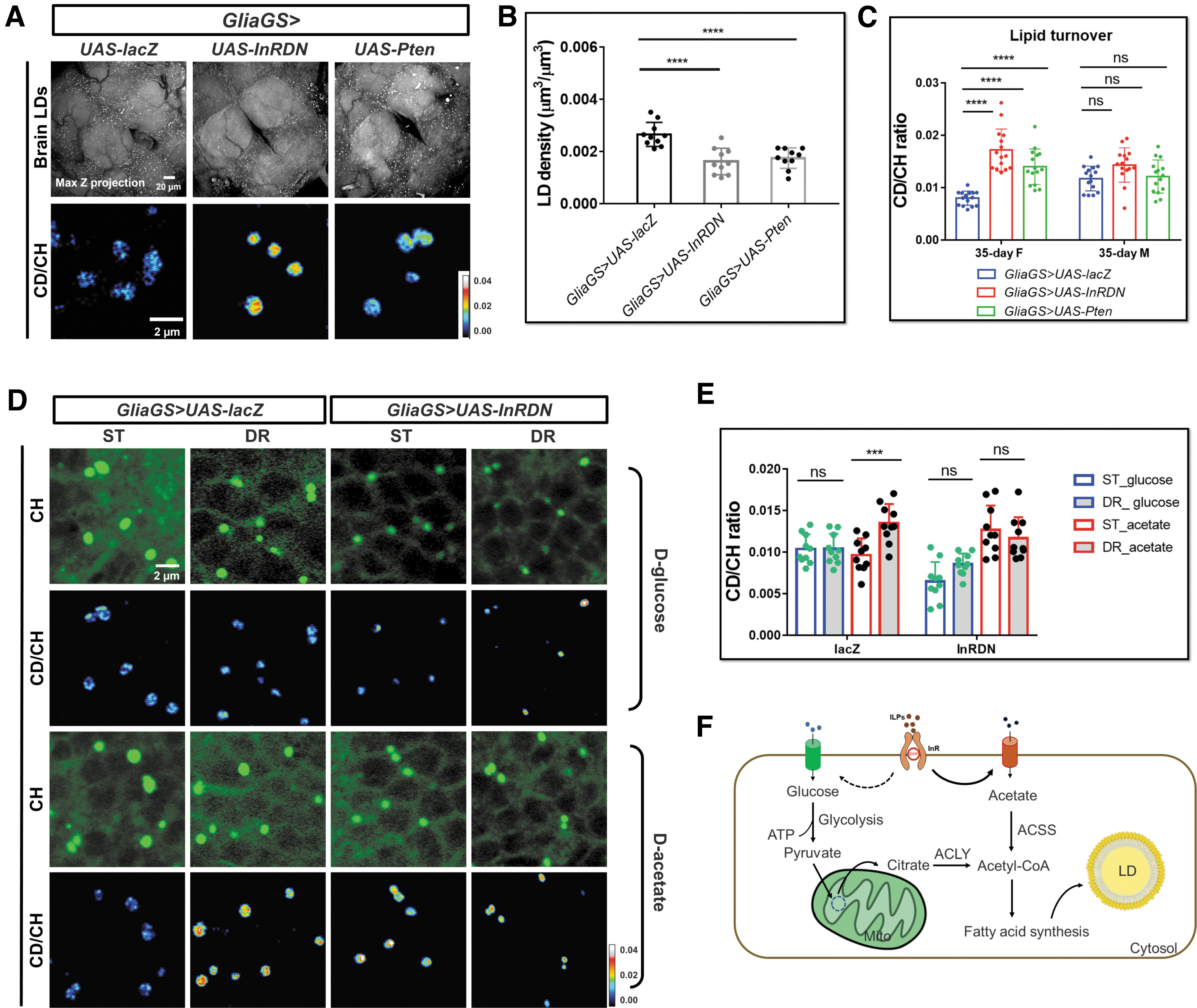
We next examined brain lipid metabolic activities in 35-day IIS transgenic flies. DO-SRS ratiometric images and quantification results showed significantly increased lipid turnover rate upon downregulation of IIS activity in both GliaGS>UAS-Pten and GliaGS>UAS-InRDN females, compared with age- and sex-matched control (GliaGS>UAS-lacZ) (Fig. 6A, C). However, there was no significant change of lipid turnover rate in the transgenic males (Fig. 6C), consistent with the aforementioned results that females are more sensitive to systemic IIS downregulation.
Similar with the phenotype of dfoxo mutants, we found significantly enlarged LD size and increased LD density in both GliaGS>UAS-dfoxoRNAi and GliaGS>UAS-InRDN,UAS-dfoxoRNAi flies (Supplementary Fig. S5A). The brain lipid mobilization was reduced in GliaGS>UAS-dfoxoRNAi and GliaGS>UAS-InRDN,UAS-dfoxoRNAi flies, suggesting that dFOXO knockdown was epistatic to InRDN overexpression (Supplementary Fig. S5B). These results again confirmed dFOXO plays an important role in IIS signaling-mediated regulation of brain lipid metabolism.
Using a neuron-specific elavGS driver to downregulate IIS in elavGS>UAS-InRDN and elavGS>UAS-dfoxoRNAi flies, we found no significant changes in LD density and lipid turnover compared with the elavGS>UAS-lacZ control (Supplementary Fig. S5C–E), suggesting neuronal IIS may not play a dominant role in brain lipid turnover. Taken together, our data demonstrate that glia-specific IIS regulates lipid metabolic turnover in Drosophila aging brain.
Aforementioned data obtained by DO-SRS imaging have shown that the general lipid metabolic activity was increased by downregulating IIS. Under prolonged DR or reduced IIS activity, it remains unclear how the reprogramming of lipid metabolism sustains neuronal survival and to what extent other metabolites, such as acetate and glutamine, contribute to support neurons. We next determined the metabolic origin of glial LDs by using SRS imaging combined with another two bioorthogonal probes, deuterated (D)-glucose and D-acetate, respectively.
Flies were cultured on diets containing D-glucose or D-acetate, major carbon sources for lipogenesis. The lipogenesis in glial LDs was then examined. SRS imaging revealed strong deuterium incorporation from dietary D-glucose or D-acetate into the core of LDs (Fig. 6D). CD/CH ratios in brain cross sections were quantified. The D-acetate-derived lipids were increased in the flies manipulated by DR and IIS downregulation. Meanwhile, D-glucose-derived lipids were significantly reduced upon IIS downregulation, even though no obvious changes were found in DR flies (Fig. 6D, E).
These results suggested that brain metabolism shifted toward using acetate as a major source for lipogenesis under DR or glia-specific IIS downregulation (Fig. 6F). In this study, we hypothesized that when IIS activity was downregulated, the precursor for lipid synthesis, acetyl-CoA, was mainly generated from acetyl-CoA synthetase (ACSS)-mediated pathway, rather than glycolysis-ATP citrate lyase (ACLY)-dependent pathway. 56
Discussion
For the first time we employed DO-SRS imaging to directly visualize and quantify spatiotemporal changes of lipid metabolic activity at organelle level (1–2 μm LD) in aging Drosophila brain. We examined interconnections between brain lipid metabolism, sex differences, dietary regulation, IIS pathway, and locomotor activity.
We observed abundant metabolically active small LDs and lipid rich membranes in young Drosophila brains. However, the LDs became enlarged and metabolically inert, whereas membrane lipids were reduced in the aged brains. Previous studies showed fatty acids from membrane phospholipids could redistribute to LDs during oxidative stress,14,57 suggesting active interaction between LDs and membranes. Our study suggested that lipid exchanges between LD storage and membranes were likely impaired in the aged brain, leading to lipid misallocation from membranes to LDs,58,59 which could be reversed by IIS downregulation.
Our results also demonstrated that regulation of lipid storage in the brain is a highly dynamic process, similar to that in the adipose tissue, 20 which is balanced between lipid synthesis and mobilization in LDs. Aging affected more than the process of lipogenesis, lipolysis, and translocation, but lipid shuttling between glia and neurons is also important to maintain brain energy homeostasis and function.11,60–66 Brain lipid homeostasis was intimately bound up with oxidative stress, injury, and immune challenges during aging process.10,12–14 Dysregulation of brain lipid metabolism can cause or accelerate brain aging and neurodegenerative diseases. Our in vivo SRS imaging of lipid metabolic dynamics at subcellular level could provide insights into the underlying mechanisms of aging-dependent neurodegenerative diseases.
We found remarkably improved lipid turnover under DR and IIS manipulations, as well as positive correlation between brain lipid turnover and locomotor activity. DR and the IIS pathway have been shown to regulate lipid metabolism and reduce the accumulation of harmful lipids, such as lipid peroxide. 67 Our study indicates that the improvement on brain lipid turnover or lipid translocation from LD to membrane could contribute to lifespan extension, as observed under DR and reduced IIS activity. However, more research is needed to fully understand the complex interactions between lipid metabolism and these pathways as well as their effects on aging and health span.
Previous studies showed that insulin receptor expression in glia is critical for brain function.39,68 Inhibition or downregulation of IIS specifically in glia extended the lifespan.55,69 It underscored the crucial role of brain lipid metabolism in maintaining not only nervous system function but also organism-wide health. dFOXO was supposed to mediate the health span extension from IIS downregulation.49,50,52,70 CNS-specific deletion of FoxO3 in 5xFAD mice led to altered brain lipid levels and exacerbation of synaptic loss and Abeta pathology, whereas astrocyte-specific overexpression of FoxO3 reversed these neurodegenerative phenotypes, suggesting a neuroprotective role of FoxO3. 71
The single-cell RNA-sequencing profiling of the adult Drosophila brain also showed that FOXO is endogenously expressed in glia but not neurons.72,73 Our study showed that glial downregulation of IIS activity enhanced lipid turnover and rejuvenated the enlarged inert LDs to small active ones in old animals. However, glia-specific knockdown of dfoxo is sufficient to deactivate the lipid turnover, resulting in larger LD size and number. These findings engender us to understand the underlying mechanism of glia IIS-regulated brain lipid metabolism. Whether inert LD-loaded glia have the same identity as the pathological microglia, 12 and are involved in age-related and genetic forms of neurodegeneration remains to be investigated.
It has been shown that IIS is low when nutrients are sparse and dFOXO translocate nuclear to active the downstream genes' expression. 74 Our results show sexual dimorphism in brain lipid metabolism during normal aging, diets, and IIS manipulations, consistent with those observed in peripheral tissues.35,75,76 It suggests the IIS sensitivity in brain is different between females and males. It has been reported that DR treatment exerted significant effects on extending lifespan in female flies more than male flies. And IIS plays a sex-dependent role in the body size and locomotor activity.47,77–81 How the lipid metabolism in local glia IIS is determined, and the correlation between IIS sensitivity, lipid turnover, and health span await more future studies.
In this study with bioorthogonal SRS imaging, we found acetate, instead of glucose, -derived lipids were largely increased under DR or reduced IIS activity. This acetate-mediated lipogenesis has been reported to promote cancer cell survival under stresses.82–85 However, the mechanism behind this metabolic shift and its role in DR/IIS-mediated lifespan extension needs further study.
Our study indicates that lipid turnover plays a crucial role in brain metabolic homeostasis and animal aging; however, the functions of different subtypes of glia and neurons in regulating brain lipid metabolism and healthy lifespan remain to be further determined. In addition to IIS-dFOXO axis, other signal pathways or factors may also participate in brain metabolic regulations.66,86,87 Our in situ metabolic imaging method will be highly useful to directly visualize and quantify the spatiotemporal alterations of metabolic profiles and to cast light on understanding the mechanisms underlying brain aging and diseases.
Materials and Methods
Drosophila genetics
Fly lines used in this study were originally obtained from the Bloomington Drosophila Stock Center (BDSC) unless otherwise stated. They have been maintained on standard diet (Nutri-Fly, Cat. No. 66-113; Genesee Scientific Corporation) in the laboratory for several generations. The wild type used for normal aging and diet treatment was w1118 (stock #5905).
Genetic elements used were repo-Gal4 (stock #7415), GliaGS-Gal4 (GSG550, stock #62085 or GSG907, stock #40310), elavGS- Gal4 (kindly provided by Dr. Xu Chen), UAS-lacZ (stock #8530), UAS-InRCA (also known as InRdel, stock #8248), UAS-InR.DN (stock #8253), UAS-Pten (stock #82170), UAS-ACC RNAi (stock #34885), UAS-white RNAi (was crossed out from stock #65409), UAS-LpR1 RNAi (stock #50737), UAS-LpR2 RNAi (stock #54461), UAS-Lipin RNAi (stock #63614), UAS-DGAT1RNAi (stock #65963), UAS-Bmm (stock #76600), and UAS-dfoxo RNAi (stock #32427). IIS pathway mutant alleles and reporter used were chico 1 (stock #10738), dfoxo 25 (stock #80944), dfoxoΔ94 (stock #42220), and dFOXO-GFP (stock #38644).
The Bigger Picture
SRS microscopy and hyperspectral imaging
The SRS and SRS-HSI images were collected from an upright laser-scanning microscope (DIY multiphoton; Olympus). It was equipped with a 25 × water objective (XLPLN, WMP2, 1.05 NA; Olympus), which was applied for near-IR throughput. The synchronized pulsed pump beam (tunable 720–990 nm wavelength, 5–6 ps pulse width, and 80 MHz repetition rate) and stokes beam (wavelength at 1032 nm, 6 ps pulse width, and 80 MHz repetition rate) were supplied by a picoEmerald system (Applied Physics & Electronics) and coupled into the microscope. After interacting with the samples, the pump and Stokes beams were collected in transmission by a high NA oil condenser (1.4 NA).
A high O.D. shortpass filter (950 nm; Thorlabs) was used to completely block the Stokes beam and transmit the pump beam only onto a Si photodiode for detecting the stimulated Raman loss signal. The output current from the photodiode was terminated, filtered, and demodulated by a lock-in amplifier at 20 MHz. The demodulated signal was fed into the FV3000 software module FV-OSR (Olympus) to form image using laser scanning. All images obtained were 512 × 512 pixels, with a dwell time 80 μs and imaging speed of ∼23 s per image. A background image was acquired at 2190 cm−1 and subtracted from all SRS images using ImageJ. The hyperspectral imaging stack of brain was taken at 75 spectral points covering 2800–3150 cm−1 at 40 μs pixel dwell time, then intensity profiles of interested regions were plotted in ImageJ.
LD analysis
3D Drosophila brain images were taken by SRS imaging system at 2850 cm−1. To enhance the detection precision, the pixel numbers were increased by two times along the lateral direction and four times along the axial direction and then A-PoD 26 was used to convert the preprocessed 3D images to the super-resolved images. From the super-resolved image, the numbers and sizes of LDs were measured with the 3D objects counter plugin 91 of ImageJ.
The information about volume and location of each LD were exported and delivered to a home-built Matlab code to visualize the data as color-coded 3D images. To reduce the background noise, band pass filter (threshold: 50 px) was applied to filtering high-frequency signal. The profile of the CBs including neuropiles was delineated by FeatureJ structure plugin. 92 The color-coded LD images and CB areas are combined into a single image stack to present the 3D distribution of each LD in the Drosophila brain.
Diet manipulation and locomotor function assay
The w1118 parents were raised in vials containing standard diet. To standardize the effects of parental age on offspring fitness, parents of experimental flies were of the same age (4–5 days and reared at a constant density for at least two generations). To synchronize larval development, we allowed flies to lay eggs on yeast apple juice plates for 1 h, discarded the first batch of embryos, and then collected for another 4 h. Groups of 20–25 embryos were put into vials containing standard diet and allowed to develop until pupae eclosion.
Newly eclosed flies were allowed to mature and mate for 48 h before the flies were briefly anesthetized with CO2 so that females and males could be separated for aging experiments. One hundred adult flies were randomly allocated at a density of 20 flies per vial using four cohorts for each diet: standard diet (ST), DR. For the detailed information of the food recipe (Supplementary Table S1). All eclosures were maintained at 25°C in a controlled light (12/12-h light/dark cycle) and humidity (>70%) environment. Flies were scored for survival daily and provided with fresh medium every 2 days. To minimize any density effects on mortality, two vials with cohorts were merged when the density of flies reached five or fewer individuals.
Eclosures were placed randomly in the incubator, and positions were rotated after each transfer to minimize the effects of microclimate. This process was followed until the end of experiments. The locomotor functions of flies were determined as reported. 93 Parental flies (w1118) were kept for 15 females and 10 males per vial and flipped every 2 days to prevent overcrowding. Progeny flies were collected within 24 h of eclosion and aged without further CO2 exposure. After 2 days' maturation, females and males were separated to different groups. For each diet condition, six vials of each sex with 20 flies per vial (totally 120 females, 120 males) were set up for examining the changes of motor activities during aging process.
The assay was performed approximately at the same time during the daytime to minimize circadian differences. Flies were transferred to the glass cylindrical vials with a line at the 5 cm mark and allowed to acclimate the circumstance for 10–15 min before testing. Flies were gently tapped down to the bottom of the vial, and the percentage of flies that crossed the 5 cm mark in 30 s was determined. Vials were placed horizontally and flies were retested 10–15 min later. The average of the two technical replicates for each vial was recorded and was plotted as a single point. The locomotor function was at 5, 15, 25, 35, and 45 days after eclosion.
D2O-labeling experiments
The changes of lipid turnover rated in wild-type flies at different ages were labeled by transferring the 0-, 10-, 20-, 30-, and 35-day adult flies to the 20% D2O labeled corresponding food conditions for 5 days, then the 5-, 15-, 25-, 35-, and 45-day aged flies were sacrificed, and their brains were dissected and subjected to Raman measurements and SRS imaging.
For investigating the metabolic activity of the IIS-related gene mutants, chico 1 /+ and dfoxoΔ94/dfoxo 25 flies aged at 2 days (young) and 30 days (old) were labeled by 20% D2O labeled for 5 days, then the 7- and 35-day aged flies were sacrificed, and their brains were dissected and subjected to Raman measurements and SRS imaging, respectively. The age paired w1118(+/+) flies treated in the same way were used as control.
For determining the metabolic activity of glia-specific IIS manipulated transgenic flies. Groups of 15 GliaGS virgin females were collected to cross with 15 UAS-InRDN, UAS-Pten of UAS-dfoxo RNAi males, respectively. All the crosses were maintained at 21°C on the standard diet to allow the normal embryo and larvae development. Eighty newly eclosed progenies (40 females and 40 males) with correct genotype were collected.
They were allocated to four cohorts (20 flies of each) and transferred to fresh food with RU-486 (Cat. No. 84371-65-3; Sigma) added at a final concentration of 200 μM and raised in 25°C. After 2 days maturation and mating, the 15 females and 15 males were separated and subjected to 5 day 20% D2O labeling experiments, then the brains from 7-day aged flies were dissected and measured by Raman or SRS imaging system. The rest of the flies (25 females and 25 males) were allowed to grow old to 30 days for D2O treatment and following experiments.
For the starvation assay, four groups of 2-/20-day old adult flies (15 flies per group) were transferred to fresh vials with 20% D2O labeled diets for 10 days labeling. Then the labeled (12- and 30-day) flies were starved on 1% agar. At each time point (24, 48, and 72 h after starvation), five flies randomly selected from the four vials were sacrificed, and brains were dissected and subjected to SRS imaging for the C–D signal quantification. The CD/CH ratios of LDs from three regions of interest in each brain were measured by ImageJ software and then the quantifications from 5 to 10 brains at each group were used to do statistical analysis.
D-glucose and D -acetate labeling experiments
Flies were fed isocaloric diets containing D-glucose (2 mM) and D-acetate (1 mM) (Cambridge Isotope Laboratories) for 5 days and the brains were dissected and subjected to SRS imaging.
Brain LD staining
Brains were fixed overnight in 4% paraformaldehyde in phosphate-buffered saline (PBS) in 4°C, then washed three times with PBS. One microgram per milliliter BODIPY 493/503 (Cat. No. D3922; Invitrogen™) was used as final concentration to stain overnight in 4°C. After one rinse in PBS, tissues were rinsed and mounted in PBS for two photon fluorescent imaging at 800 nm wavelength.
Statistical analysis
Statistical significance was tested by using Student's t-test, one-way or two-way analysis of variance (ANOVA) with Tukey's multiple comparison or Dunnett's comparison using GraphPad Prism software. Kaplan–Meier log-rank test was performed for survival assays. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; ns, nonsignificant difference.
Footnotes
Acknowledgments
We are grateful to Bloomington Drosophila Stock Center (BDSC) for fly stocks. We thank Drs. K. Zhang, C. Metallo, G. Haddad, D. Zhou, and Shi Lab group members for helpful discussions.
Authors' Contributions
L.S. and Y.L. conceived the idea, designed the study, interpreted data, and wrote the article. Y.L. conducted the experiments, analyzed the data, and performed statistical analyses with the help from P.C., S.S., H.J., Y.N., A.Z., S.H., J.Y.W., X.C., and L.S. Y.L., J.Y.W., and L.S. wrote and revised the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
We acknowledge support from UCSD Startup funds, NIH U54 2U54CA132378, NIH 5R01NS111039, NIH R21NS125395, Sloan Research Fellow, and Hellman Fellow Award.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
