Abstract
This study evaluated the efficacy of nanoemulsified benzyl isothiocyanate (NBIT) and carvacrol (NCR) to reduce Salmonella contamination on alfalfa seeds and sprouts. Given the susceptibility of sprouts to microbial contamination during germination, improved interventions were needed to enhance food safety without compromising product quality. NBIT and NCR were applied to Salmonella-inoculated seeds at high (6.3 log CFU/g) and low (2.4 log CFU/g) inoculum levels and their impact on Salmonella populations, seed germination, sprout quality (length and weight), and visual appearance was assessed. Results revealed that both NBIT and NCR significantly reduced Salmonella populations and most treatments did not adversely affect seed germination or sprout quality. Treatment with 2.0% NBIT achieved reductions of up to 2.4 log CFU/g on seeds and 6.4 log CFU/g on sprouts after 4 days at the low inoculum level. However, treatment with 2.0% NBIT resulted in an 18.8% reduction in germination that occurred at the high inoculum level and a 10.7% reduction at the low inoculum level, along with a slight decrease in sprout size. In contrast, treatments with 0.5% and 1.0% NBIT, as well as 2.0% NCR, reduced Salmonella on seeds by 1.5–2.0 log CFU/g, regardless of inoculum level, and achieved reductions of 5.0–6.4 log CFU/g on sprouts at the low inoculum level. In addition, NBIT (0.5% and 1.0%) and NCR (2.0%) preserved seed germination rates and sprout quality metrics, including length, weight, and visual appearance, comparable with untreated controls. These findings highlight the potential of NBIT and NCR as effective antimicrobial agents for improving the microbial safety of alfalfa sprouts without compromising quality.
Introduction
The global consumption of sprouts has risen due to their high nutritional value and year-round availability, making them a popular choice for health-conscious consumers (Benincasa et al., 2019). Alfalfa sprouts, in particular, are valued for their rich content of vitamins, minerals, and antioxidants, which contribute to a balanced diet (Aloo et al., 2021). However, alfalfa sprouts also pose a potential risk as a vehicle for foodborne pathogens, primarily due to their growing conditions, which provide a warm and humid environment that supports microbial growth (Mir et al., 2021; Wang et al., 2020). Notably, Salmonella enterica and Shiga-toxigenic Escherichia coli have been associated with contamination incidents in the sprout industry, and documented outbreaks have implicated alfalfa sprouts as the source (Zheng et al., 2021).
To address safety concerns associated with sprout production, the U.S. Food and Drug Administration (FDA) has implemented specific regulations under the Produce Safety Rule within the Food Safety Modernization Act (U.S. Food and Drug Administration, 2022). The FDA endorses treatment of seeds with chlorine-based antimicrobial such as a 20,000 ppm calcium hypochlorite (Hong and Kang, 2016). However, while effective, chlorine-based treatments can adversely affect the sensory quality of sprouts and is associated with potential health and environmental concerns (Neo et al., 2013; Sikin et al., 2013). These limitations have prompted interest in exploring alternative treatments, including natural plant extract-based antimicrobials (Pinto and Ayala-Zavala, 2024), which may ensure microbial safety without compromising sprout quality.
Glucosinolates are naturally occurring compounds found in cruciferous vegetables such as cauliflower, broccoli, and cabbage (Connolly et al., 2021). Upon hydrolysis by the enzyme myrosinase, glucosinolates break down into bioactive compounds with antimicrobial properties, which have shown effectiveness in laboratory settings (Abdel-Massih et al., 2023; Patel et al., 2020). Among these compounds, benzyl isothiocyanate (BIT) at the concentration of 0.5–2%, presented a promising natural alternative for controlling Salmonella contamination on sprouting alfalfa seeds, offering a plant-derived solution to microbial safety in sprout production without compromising seed germination (Yin et al., 2020).
Despite the potential of natural antimicrobial agents such as BIT to combat various pathogens, their use in food applications faces challenges due to low water solubility, chemical instability, and sensory impacts (Pinto et al., 2023). Nanoemulsion technology can overcome these limitations, enhancing their effectiveness and suitability for food safety applications (Mushtaq et al., 2023). For example, carvacrol, a natural antimicrobial compound found in oregano and thyme (Mączka et al., 2023), has been effectively nanoemulsified and this nanoemulsified carvacrol (NCR) treatment demonstrated enhanced microbial reduction in leafy greens during washing treatment (Chen et al., 2021) and various sprouts (Landry et al., 2014, 2015).
This study formulated nanoemulsified BIT (NBIT) and evaluated its antimicrobial efficacy alongside NCR on Salmonella-inoculated alfalfa seeds, as well as on sprouts germinated from those inoculated seeds. The primary objective was to assess the effectiveness of NBIT and NCR in controlling Salmonella contamination while also evaluating their impact on sprout quality.
Materials and Methods
Bacterial strains and inoculum preparation
S. enterica serotype Newport (USDA4558) was obtained from our culture collection and prepared using a modified method of Yin et al. (2020). The strain was cultured twice in 10 mL of tryptic soy broth (BD Biosciences, Franklin Lakes, NJ) at 37°C for 24 h and optical density adjusted to 9 log CFU/mL and 5 log CFU/mL for high and low inoculation, respectively.
Inoculation of alfalfa seeds
Organic alfalfa seeds (Medicago sativa), purchased from Johnny’s Selected Seeds (Winslow, ME), were inoculated with Salmonella, and Salmonella enumeration from the seeds was performed using procedures described previously (Yin et al., 2020). Ten grams of seeds were placed in 100 × 15 mm Petri dishes (VWR, Radnor, PA) and soaked in 10 mL of high or low inoculum for 2 min. The seeds were then dried in a biosafety cabinet at room temperature for 24 h. After drying, Salmonella populations on the seeds were approximately 6.3 log CFU/g for high inoculation and 2.4 log CFU/g for low inoculation.
Preparation of nanoemulsified BIT and carvacrol
NBIT were prepared at 0.5%, 1.0%, and 2.0% (v/v) using Monolaurin (TCI America, Portland, OR), ethanol (Decon Labs, King of Prussia, PA), Tween 80 (TCI America), Span 80 (TCI America), and sterile deionized water. For the emulsifying solution, 1.0% monolaurin was dissolved in ethanol. A 9:1 (w/w) surfactant blend of Tween 80 and Span 80 was used. To prepare 0.5% NBIT, 50 mL emulsifying solution, 45 g surfactant blend, 5 mL BIT (Sigma-Aldrich, St. Louis, MO), and 900 mL ice-cold sterile deionized water were stirred at 200 rpm. Proportional amounts of ingredients were used for 1.0% and 2.0% NBIT. Our group previously utilized a 2.0% (v/v) NCR, prepared as described, and therefore, we adopted the same concentration for our experiment. (Chen et al., 2021; Chen and Zhong, 2015). Droplet sizes were measured with a Zetasizer Pro (Malvern Instruments, Worcestershire, UK), with Z-Average sizes ranging from 12 to 34 nm and Polydispersity indexes ranging from 0.16 to 0.57.
Alfalfa seed treatments and Salmonella enumeration
Ten grams of Salmonella-inoculated alfalfa seeds were soaked in 10 mL of treatment solutions (0.5%, 1.0%, 2.0% NBIT; 2.0% NCR) for 15 min at room temperature with agitation (600 rpm) using a Digital MicroPlate Genie Pulse (Scientific Industries, Bohemia, NY). An untreated control group was included. After treatment, seeds were transferred to Petri dishes and dried in a biosafety cabinet at room temperature for 2 h. Four grams of dried seeds from each group were transferred to sterile Seward Stomacher bags (15 × 23 cm, VWR), and 16 mL neutralizing buffer (Neogen, Lansing, MI) was added. Seeds were pummeled at high speed (12 strokes/s) for 2 min using a stomacher (MiniMix bag mixer, Interscience, Woburn, MA). For high inoculum tests, the buffer was serially diluted (1:10) in phosphate-buffered saline (PBS, Thermo Fisher Scientific, Waltham, MA), and 0.1 mL of the appropriate dilution was spiral-plated onto xylose-lysine-tergitol 4 agar (XLT4, BD Biosciences, Franklin Lakes, NJ) for Salmonella enumeration. For low inoculum tests, 1 mL undiluted buffer was spread-plated onto three XLT4 agar plates. Plates were incubated at 37°C for 24 h, and Salmonella populations were expressed as log CFU/g.
Seed germination and analysis of sprouts
A modified procedure (Yin et al., 2020) was used to assess the effect of NBIT and NCR on controlling Salmonella during alfalfa germination. Treated and control seeds were soaked in 10 mL sterile deionized water in Petri dishes for 24 h, then 200 seeds of each sample were transferred to an EasyGreen Light Sprouter (Danbury, CT) for germination at 25°C. After 4 days, seeds were considered germinated when the radicle exceeded 2 mm. Sprout weight and length (cotyledons to root) were measured. Cotyledon visual appearance, encompassing greening, expansion, and morphology, was assessed through direct visual observation, noting perceived color intensity, opening angle, and structural abnormalities, respectively. Germinated sprouts (2.5 g) were collected into sterile Seward Stomacher bags (15 × 23 cm, VWR) with 10 mL PBS. Samples were pummeled for 2 min using a stomacher, diluted in PBS as appropriate, and spiral-plated on XLT4 agar for Salmonella enumeration.
Statistical analysis
Salmonella populations (colony counts) were converted to log CFU/g of seeds or sprouts. Data were analyzed using a multifactorial analysis of variance model with fixed effects in the PROC GLM procedure in SAS version 9.3 (SAS Institute, Cary, NC). All experiments were repeated three times. Differences among means for treatments were considered significant at p < 0.05, as determined by Fisher’s least significant difference test. The effect of antimicrobial treatments on seed germination, sprout length, and weight were also analyzed for significance.
Results
Effectiveness of NBIT and NCR treatments against Salmonella on alfalfa seeds
Treatments using NBIT at concentrations of 0.5%, 1.0%, and 2.0%, as well as NCR at 2.0%, yielded Salmonella counts that were significantly lower than those on untreated seeds (Fig. 1A). The antimicrobial effect of NBIT was found to be concentration dependent. At the high inoculum level of 6.3 log CFU/g, populations of Salmonella from the 0.5%, 1.0%, 2.0%NBIT-treated, and 2.0%NCR-treated alfalfa seeds were 4.8 ± 0.1, 4.4 ± 0.1, 4.1 ± 0.2, and 4.6 ± 0.1 log CFU/g, respectively, compared with 6.3 ± 0.1 log CFU/g (providing 1.5 to 2.2 log CFU/g reduction) of Salmonella recovered from the untreated control seeds. At the low inoculum level of 2.4 log CFU/g, seed treatment with 2% NBIT reduced Salmonella to undetectable level. The populations of Salmonella from the 0.5%, 1.0% NBIT-treated, and 2.0%NCR-treated alfalfa seeds were 0.6 ± 0.3, 0.4 ± 0.3, and 0.6 ± 0.3 log CFU/g, respectively, compared with 2.4 ± 0.0 log CFU/g of Salmonella recovered from the untreated control seeds.
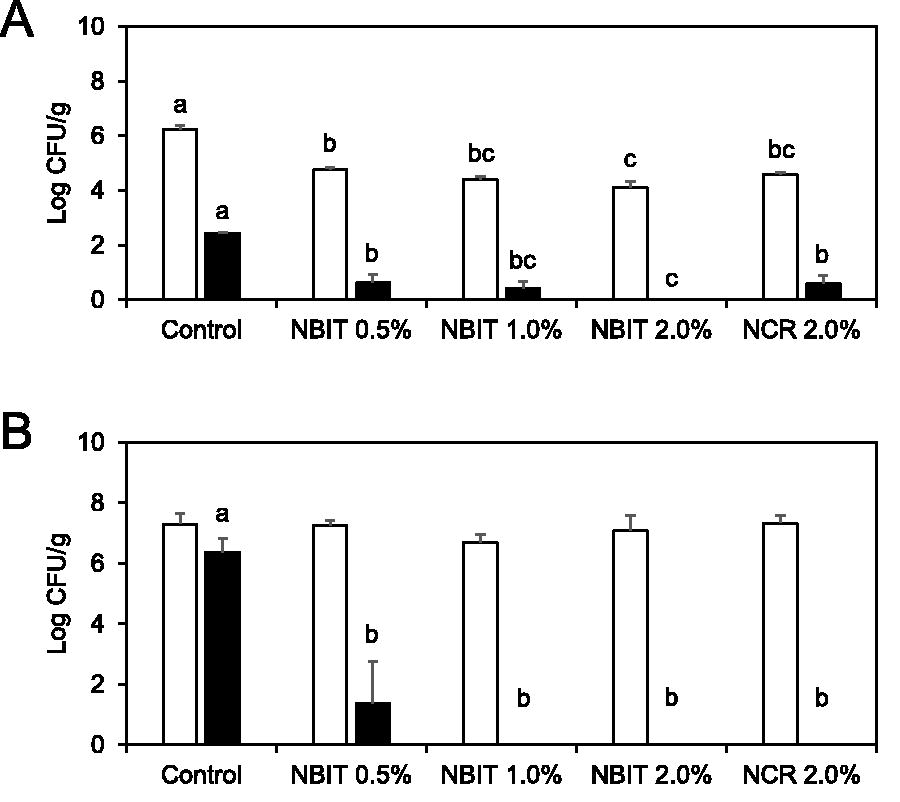
Effect of NBIT and NCR for inactivating Salmonella enterica serotype Newport on alfalfa seeds
Effectiveness of NBIT and NCR in reducing Salmonella during alfalfa sprouting
Figure 1B illustrated the survival of Salmonella on alfalfa sprouts throughout sprouting after treatments with NBIT or NCR. At a higher inoculum level (6.3 log CFU/g), Salmonella populations on the sprouts were in the range of 6.7–7.3 log CFU/g and similar (p > 0.05) across all treatments and the control (Fig. 1B). Conversely, at a lower inoculum level (2.4 log CFU/g), all treatments significantly reduced Salmonella on sprouts compared with control. A 6.4 log CFU/g Salmonella were recovered from control sprouts after 4 days. Salmonella populations in sprouts from seeds treated with 1.0% NBIT, 2.0% NBIT, or 2.0% NCR were below the detection limit (Fig. 1B).
Impact of NBIT and NCR treatments on alfalfa seed germination, sprout length, sprout weight, and visual appearance of sprouts
In the high inoculum level test, germination rates for untreated seeds (94.7%) and those treated with various NBIT concentrations or 2.0% NCR were mostly similar, with the exception of the 2.0% NBIT treatment, which resulted in an 18.8% decrease (p < 0.05) in germination compared with control seeds (Fig. 2A). In the low inoculum condition, the most substantial germination reduction occurred with 2.0% NBIT treatment, showing a 10.7% decline relative to untreated controls (Fig. 2B).
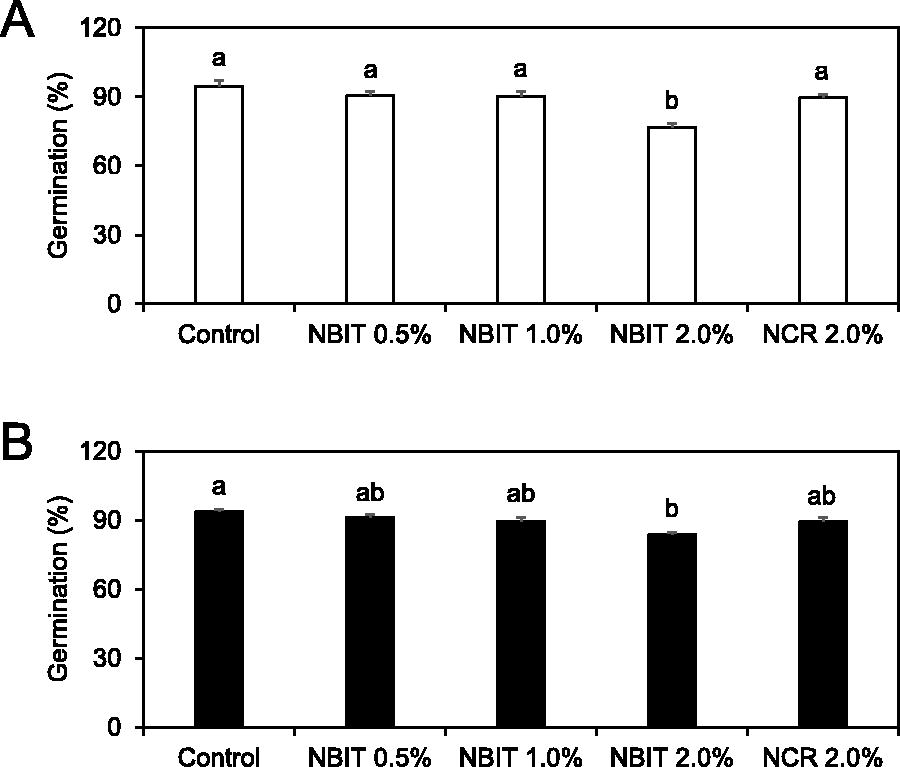
Effect of NBIT and NCR on the germination of alfalfa seeds. Alfalfa seeds were inoculated with Salmonella enterica serotype Newport at high inoculum level of 6.3 log CFU/g
For high inoculum seeds, treatment with NBIT concentrations of 0.5–2.0% or 2.0% NCR had no significant impact on sprout length, with treated sprouts measuring 3.5–4.3 cm, comparable with the control length of 4.4 cm (Fig. 3A). Under low inoculation, seeds treated with NBIT produced shorter sprouts, resulting in significant difference when treated with 2.0% NBIT (2.8 cm) compared with the control sprouts (4.2 cm). The length of sprouts from 0.5% NBIT or 2.0% NCR-treated seeds were similar to controls (Fig. 3A). The observed difference could be attributed to the interaction between the bacteria and the antimicrobial. Specifically, the high bacterial population might have detoxified or degraded the antimicrobial, thereby lessening its impact on the plant.
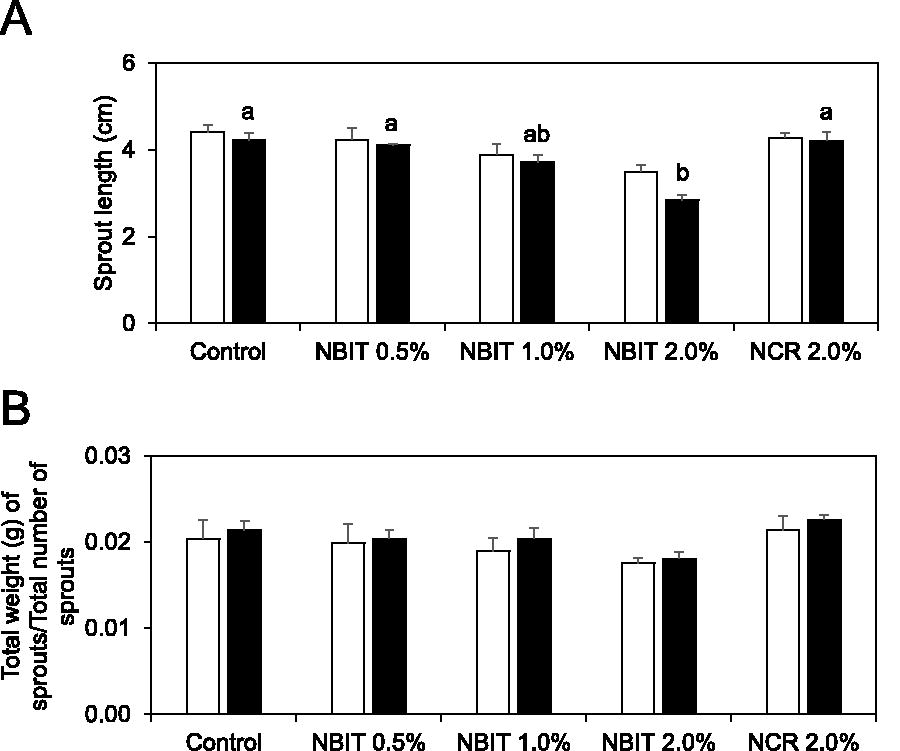
Effect of NBIT and NCR on alfalfa sprout length
Across both high and low inoculation tests, NBIT treatments at 0.5–2.0% and NCR at 2.0% did not lead to significant differences in sprout weight, which ranged from 0.018 to 0.023 g, in comparison with control sprouts at 0.020 and 0.021 g (Fig. 3B). In all experimental treatments, there were no visible defects in the sprouts’ appearance (Fig. 4), except for those treated with 2.0% NBIT, which exhibited a smaller architecture compared with other treatments. This suggested that while NBIT and NCR treatments generally maintained the visual quality of alfalfa sprouts, a higher concentration of NBIT (2.0%) may slightly impact sprout size.
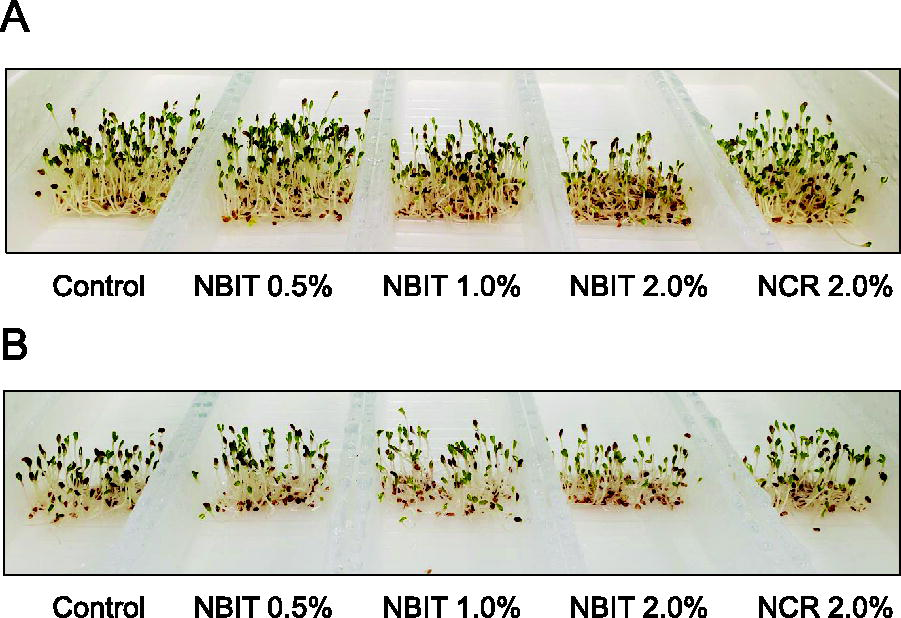
Effect of NBIT and NCR on visual appearance of alfalfa sprouts. Alfalfa seeds were inoculated with Salmonella enterica serotype Newport at high inoculum level of 6.3 log CFU/g
Discussion
At high bacterial inoculum level on seeds, NBIT exhibited a dose-dependent reduction in S. enterica serotype Newport populations, achieving up to a 2.1 log CFU/g reduction at a 2.0% concentration. Similar effects were observed at low inoculum level, where reductions reached 2.4 log CFU/g. These results aligned with those of Yin et al. (2020), who reported that non-nanoemulsified BIT effectively reduced S. enterica populations across multiple serotypes (Newport, Tennessee, Typhimurium, Braenderup, and Montevideo), achieving reductions of 0.7–1.9 log CFU/g at high inoculum level (5 log CFU/g) and 1.0–1.7 log CFU/g at low inoculum level (3 log CFU/g). While nanoemulsification achieved marginally higher Salmonella reduction, it may also improve uniform application on seeds. It has been reported that nanoemulsification can enhance BIT’s stability by protecting it from environmental degradation, potentially increasing its efficacy (Tian et al., 2021). However, despite these advantages, both studies indicated that BIT, whether in nanoemulsified form or not, had inherent limitations in completely eradicating Salmonella on alfalfa seeds contaminated at the higher level. Salmonella were below the detection limit in our study following 2.0% BIT treatment when seeds where contaminated at low level. This consistency across studies reinforced BIT’s reliability as an antimicrobial agent for reducing Salmonella on seeds but also suggested that additional complementary strategies may be necessary for more comprehensive control.
Consistent with our findings on the notable antimicrobial efficacy of NCR against Salmonella on alfalfa seeds, Landry et al. (2014, 2015) found that NCR effectively reduced Salmonella and E. coli O157:H7 on alfalfa, mung bean, broccoli, and radish seeds, demonstrating its potential for pathogen control during sprout production without compromising yield. The application of 0.4% NCR to alfalfa seeds inoculated with Salmonella Enteritidis for 60 min led to a reduction of 2.0–3.0 log CFU/g, depending on the initial inoculum sizes (Landry et al., 2014). This outcome represented a more significant reduction compared with our findings, which showed a decrease of 1.7–1.9 log CFU/g. The observed differences may be attributed to variations between strains (specifically Salmonella Newport versus Salmonella Enteritidis) or the duration of treatment, which was 15 min in our study compared with 60 min in their study. In addition, another study found NCR’s significant reduction of E. coli O157:H7 on leafy greens, achieving reductions comparablewith traditional chlorine treatments (Chen et al., 2021). The broad-spectrum efficacy of NCR against both E. coli and Salmonella, along with its increased stability under environmental stresses, supported its potential as a robust, natural antimicrobial agent for fresh produce sanitation.
Our results revealed that NBIT’s effectiveness in controlling Salmonella on alfalfa sprouts was dependent on the initial inoculum level. At a low inoculum (2.4 log CFU/g), NBIT achieved substantial Salmonella reduction, demonstrating high antimicrobial efficacy under low-contamination conditions. However, at a high inoculum level (6.3 log CFU/g), reductions were limited, suggesting that high bacterial loads may diminish the antimicrobial’s effectiveness during sprouting. One possible explanation was that higher bacterial concentrations promoted biofilm formation, which created a protective barrier around bacterial cells, hindering direct contact with antimicrobials (Liu et al., 2023). Biofilms are known to enhance bacterial resilience (Liu et al., 2024), making Salmonella more difficult to eradicate in high-contamination scenarios. In addition, at higher inoculum levels, Salmonella may internalize more deeply into sprout tissues, as shown in studies on leafy greens that linked increased bacterial internalization with higher initial inoculum sizes (Grivokostopoulos et al., 2022). This inoculum effect, documented across multiple bacterial species, indicated that higher bacterial densities can reduce antimicrobial efficacy (Diaz-Tang et al., 2022; Loffredo et al., 2021). The enhanced performance of NBIT at low bacterial concentrations likely reflected the unique advantages of nanoemulsification, which reduces droplet size, increases surface area, and improves BIT dispersal (Kumar et al., 2025), enabling more consistent bacterial contact. Yin et al. (2020) observed a reduction of up to 3.2 log CFU/g using non-nanoemulsified BIT under similar low inoculum conditions, while our study achieved up to 6.4 log CFU/g with NBIT. This comparison suggested that nanoemulsification significantly enhanced BIT’s efficacy in low-contamination scenarios. These findings underscored that nanoemulsified BIT offered considerable advantages for applications targeting low levels of microbial contamination, maximizing antimicrobial action possibly through improved dispersal and bacterial contact.
The application of NBIT and NCR as antimicrobial treatments showed minimal adverse effects on seed germination, sprout growth, and appearance, supporting their potential as safe treatments for sprout production. Most NBIT and NCR treatments did not significantly impact germination or sprout development, similar to the non-nanoemulsified BIT treatment on alfalfa seeds (Yin et al., 2020), except for 2.0% NBIT treatment which adversely affected germination rate and sprout length. Treated sprouts with NBIT or NCR exhibited no visible defects, preserving their physical appearance. This aligned with findings by Chen et al. (2021), and Landry et al. (2014, 2015) who reported no significant effect of NCR on the color of fresh produce leaves and germination rate of plant seeds. It was noteworthy that free carvacrol oil at a low concentration (e.g., 0.2 mg/mL) exhibited no phytotoxic effects on the germination performance of perennial ryegrass seeds, as reported by Saghrouchni et al. (2021). In contrast, high concentrations of free carvacrol oil (e.g., 3.0 mmol/L) were found to adversely affect the germination of both millet and lettuce (De Assis Alves et al., 2018). These findings highlighted the concentration-dependent effects of carvacrol on seed germination and subsequent seedling development.
Both BIT and carvacrol are GRAS (Generally Recognized as Safe) ingredients, offering potential for improving microbial safety in fresh produce. However, sensory impact remains a critical consideration. Yin et al. (2020) and Chen et al. (2021) have highlighted potential sensory challenges associated with these compounds and proposed solutions. Therefore, future studies should focus on optimizing application methods of NBIT and NCR to minimize sensory alterations while maintaining effective pathogen control.
Overall, NBIT and NCR effectively reduced Salmonella contamination on alfalfa seeds without significantly hindering germination, except at 2.0% NBIT, suggesting that seed-level decontamination could be a viable strategy for controlling microbial loads prior to sprouting. During sprouting, the treatments effectively controlled Salmonella at low inoculum levels while maintaining sprout growth and quality, with no significant reductions in length or weight across most conditions, thereby preserving the physical appearance and growth characteristics of the sprouts.
Conclusions
This study highlighted the practical value of NBIT and NCR in enhancing the microbial safety of alfalfa sprouts. NBIT demonstrated strong antimicrobial activity against Salmonella, particularly at higher concentrations, while minimizing adverse effects on sprout quality, making it a promising solution for pathogen control in fresh produce. Although the highest concentration of NBIT (2.0%) showed slight trade-offs with seed germination and sprout size, these findings suggested that further optimization could improve its applicability. Overall, NBIT and NCR offer a promising, consumer-acceptable approach to pathogen mitigation, curtailing product recalls, and supporting the safer consumption of alfalfa sprouts.
Authors’ Contributions
S.O. and J.P. developed the concept of the study, designed the experiments, performed the experiments, collected and analyzed the data, wrote the original draft, and revised the article. All authors read and approved the final article.
Footnotes
Acknowledgments
The authors thank Ashley Boomer and Ujjwol Subedi for technical assistance.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was funded by USDA research project No. 8042-32420-009-00D.
Disclaimer
USDA is an equal opportunity provider and employer.
