Abstract
Listeria (L.) monocytogenes is an environmental pathogen that can contaminate a wide variety of food products, leading to foodborne listeriosis outbreaks. In this context, ready-to-eat (RTE) foods of animal and plant origin are of particular relevance as the source of infection. In Germany, the official food and veterinary control laboratories, under the sovereignty of the federal states, examine foodstuffs for the presence of foodborne pathogens, including Listeria. The German National Reference Laboratory for L. monocytogenes regularly organizes inter-laboratory proficiency tests (PTs) to monitor the accuracy and comparability of analyses performed by these official control laboratories. In our study, we provide summary data on frequently contaminated RTE food products in the European Union and present the outcome of five consecutive PTs. RTE food matrices provided by the German National Reference Laboratory for L. monocytogenes included processed meat, fish, vegetable, and dairy products. Overall, the official control laboratories achieved excellent results in the PTs. In total, 91 participants were documented. Flawless results were reported in 93.4% of the cases. Only four times, participants did not pass the PT in the first attempt. By performing official food and veterinary controls along the food chain, control laboratories make a significant contribution to food safety. To keep the excellent standard of the official control laboratories in Germany in line with future challenges in food safety, PTs with relevant RTE food matrices are indispensable.
Keywords
Introduction
Listeriosis is a rare, but severe and potentially life-threatening foodborne disease caused by Listeria (L.) monocytogenes and rarely by L. ivanovii (de Noordhout et al., 2014; Guillet et al., 2010). In immunocompetent individuals, the non-invasive form might cause self-limiting febrile gastroenteritis, whereas the invasive form mainly affects the elderly, immunocompromised patients, and pregnant women and may lead to sepsis, meningitis, and encephalitis or neonatal infections and abortion (Koopmans et al., 2023). Within the European Union (EU), a total number of 20,552 listeriosis cases were reported between 2013 and 2021, corresponding to an average notification rate of 0.455 per 100,000 population per year. The high proportion of hospitalizations (41.1%) and infections with fatal outcomes (15.8%) raise concern for public health in the EU. In the same period, the incidence was even higher (0.721 per 100,000 population) in Germany, with a peak at 0.874 in 2017 (Table 1). Listeriosis continues to be an issue, especially in adults ≥65 years of age with case fatality rates of 13% (ECDC, 2022; Wilking et al., 2021).
Confirmed Invasive Listeriosis in the EU and Germany
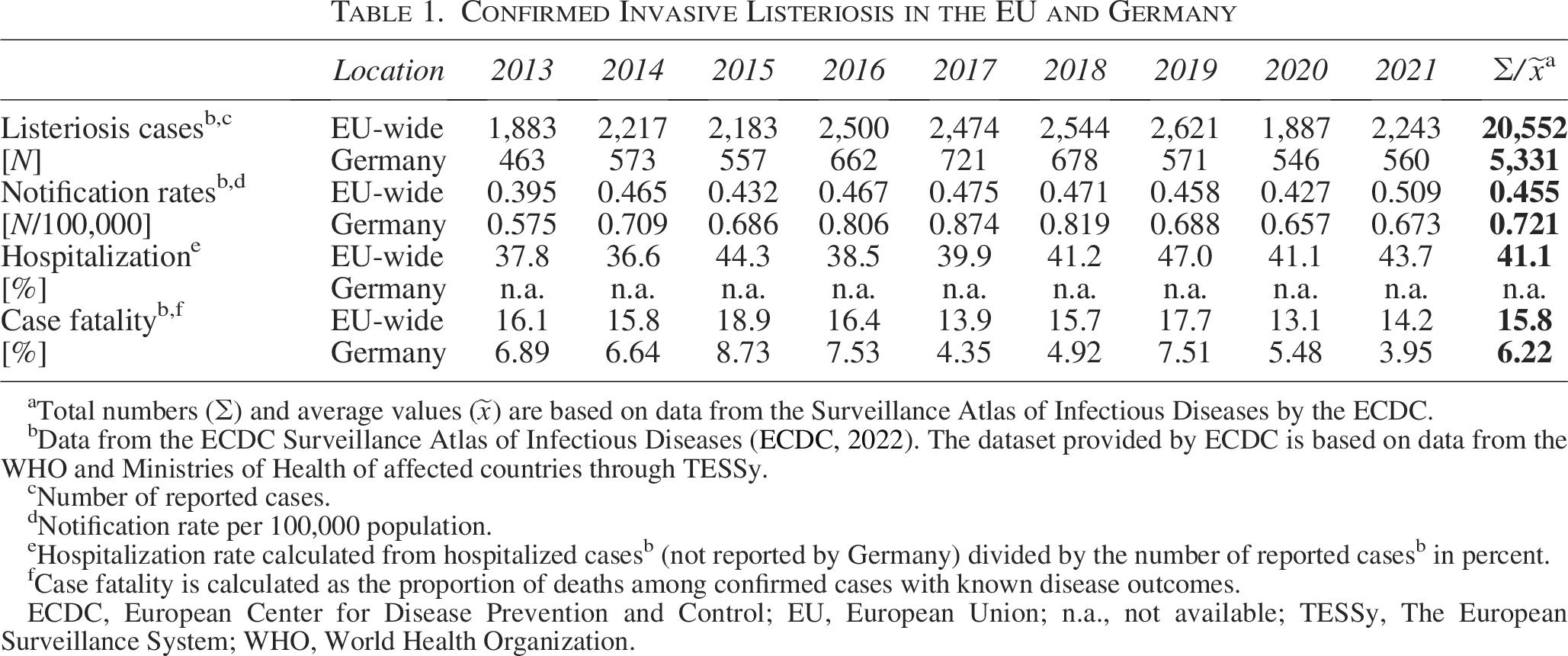
Total numbers (∑) and average values (
Data from the ECDC Surveillance Atlas of Infectious Diseases (ECDC, 2022). The dataset provided by ECDC is based on data from the WHO and Ministries of Health of affected countries through TESSy.
Number of reported cases.
Notification rate per 100,000 population.
Hospitalization rate calculated from hospitalized casesb (not reported by Germany) divided by the number of reported casesb in percent.
Case fatality is calculated as the proportion of deaths among confirmed cases with known disease outcomes.
ECDC, European Center for Disease Prevention and Control; EU, European Union; n.a., not available; TESSy, The European Surveillance System; WHO, World Health Organization.
L. monocytogenes can be found in a variety of raw food products of animal and plant origin, but also in heat-treated and subsequently processed food products (Fig. 1A) (EFSA, ECDC, 2022). The pathogen is widely distributed in the environment and can be easily introduced into food processing plants. There, the pathogen can survive and persist in ecological niches due to its ability to adapt to abiotic stress and can contaminate food products during and/or after processing (e.g., slicing, packaging) (Gandhi and Chikindas, 2007; Hansen et al., 2006; Wilks et al., 2006). In particular, ready-to-eat (RTE) foods are at higher risk for contamination with L. monocytogenes. Meat and fish products show the highest prevalence of L. monocytogenes contamination (Fig. 1A) and account for the majority of food matrices associated with strong-evidence foodborne outbreaks of human listeriosis in the EU (Fig. 1B–D). In addition, cheese, vegetables, and food products of no further specification, here labeled as “diverse foods”, have been frequently reported as causes of listeriosis outbreaks in the EU (Fig. 1C and D).
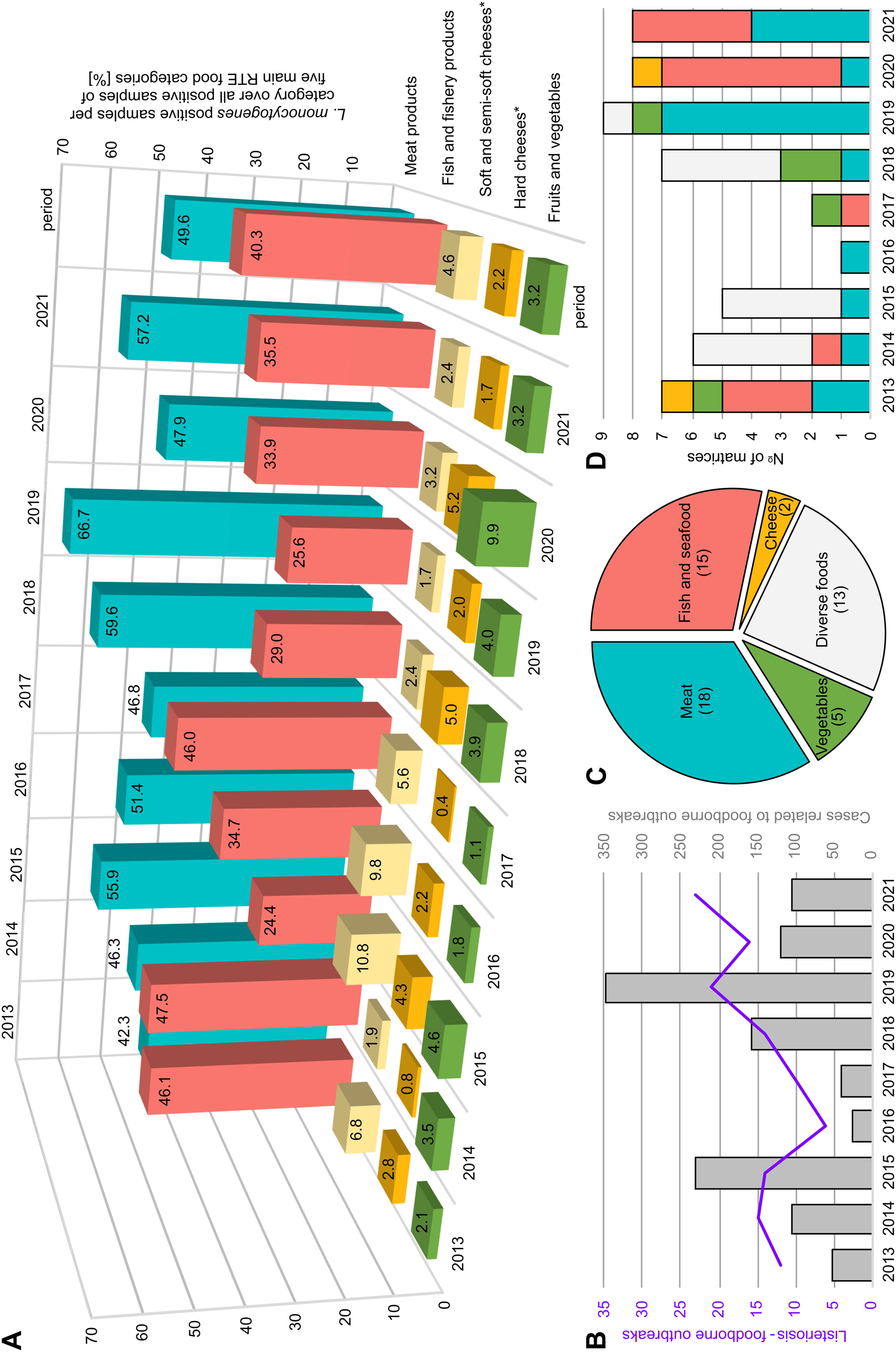
Listeria monocytogenes-positive ready-to-eat (RTE) food matrices as well as human listeriosis outbreaks and associated food matrices in the EU between 2013 and 2021 (EFSA, ECDC, 2015a; EFSA, ECDC, 2015b; EFSA, ECDC, 2016; EFSA, ECDC, 2017; EFSA, ECDC, 2018; EFSA, ECDC, 2019; EFSA, ECDC, 2021a; EFSA, ECDC, 2021b; EFSA, ECDC, 2022).
Official food and veterinary controls represent an important tool in monitoring Listeria along the food chain and verifying that food manufacturers comply with the microbiological criteria for foodstuffs laid down in Regulation (EC) No 2073/2005. Thus, they make a significant contribution to food safety. In Germany, the official food and veterinary control laboratories, under the sovereignty of the federal states, carry out these controls.
National Reference Laboratories organize inter-laboratory proficiency tests (PTs) to monitor the accuracy and comparability of analyses performed by official control laboratories. Furthermore, PTs aim to assist laboratories with unsatisfactory results by guiding them in implementing corrective actions and monitoring their effectiveness. In this context, the German National Reference Laboratory for L. monocytogenes (NRL), located at the German Federal Institute for Risk Assessment (BfR), conducts PTs to monitor the performance of official control laboratories in detecting L. monocytogenes in RTE food.
In our study, we present the outcome of five PTs organized by the NRL and discuss the overall competence of the laboratories to produce reliable and consistent results.
Materials and Methods
Proficiency tests and participating laboratories
PTs were organized yearly for the official food control and veterinary laboratories and were announced in advance, providing the main details on the scope, the registration deadline, and the expected start date. The PTs addressed throughout this article are numbered consecutively from “1” to “5,” starting with the PT in 2013 (PT1) and ending with the PT in 2017 (PT5; Table 2). Each laboratory was assigned a unique laboratory identifier (Lab-ID) to ensure performance traceability across the analyzed PTs.
Overview of PTs Organized by the NRL, Including Matrices and Sample-Set Composition
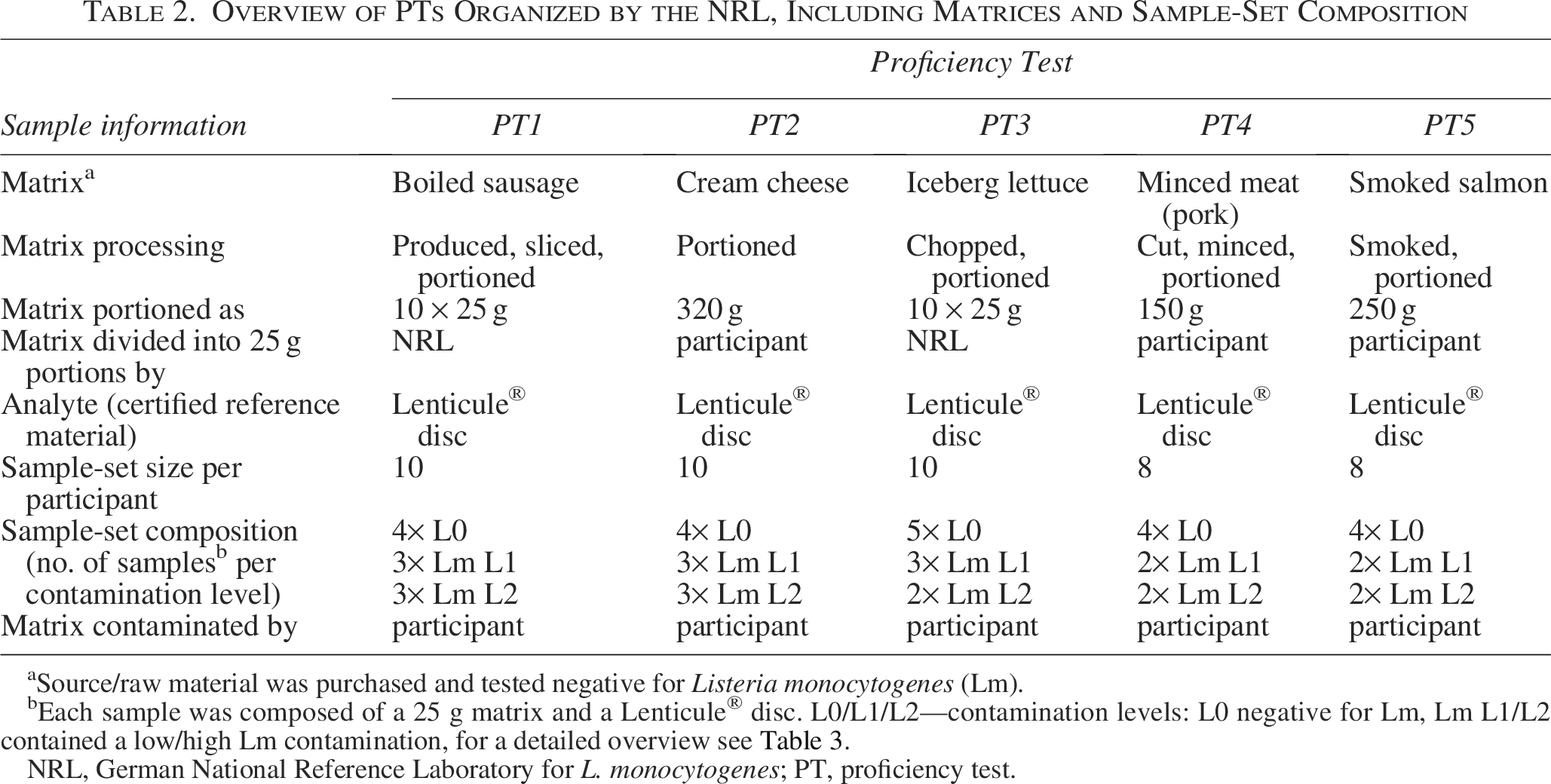
Source/raw material was purchased and tested negative for Listeria monocytogenes (Lm).
Each sample was composed of a 25 g matrix and a Lenticule® disc. L0/L1/L2—contamination levels: L0 negative for Lm, Lm L1/L2 contained a low/high Lm contamination, for a detailed overview see Table 3.
NRL, German National Reference Laboratory for L. monocytogenes; PT, proficiency test.
Detailed Record of Inocula with Different Contamination Levels for Proficiency Tests Provided by the NRL
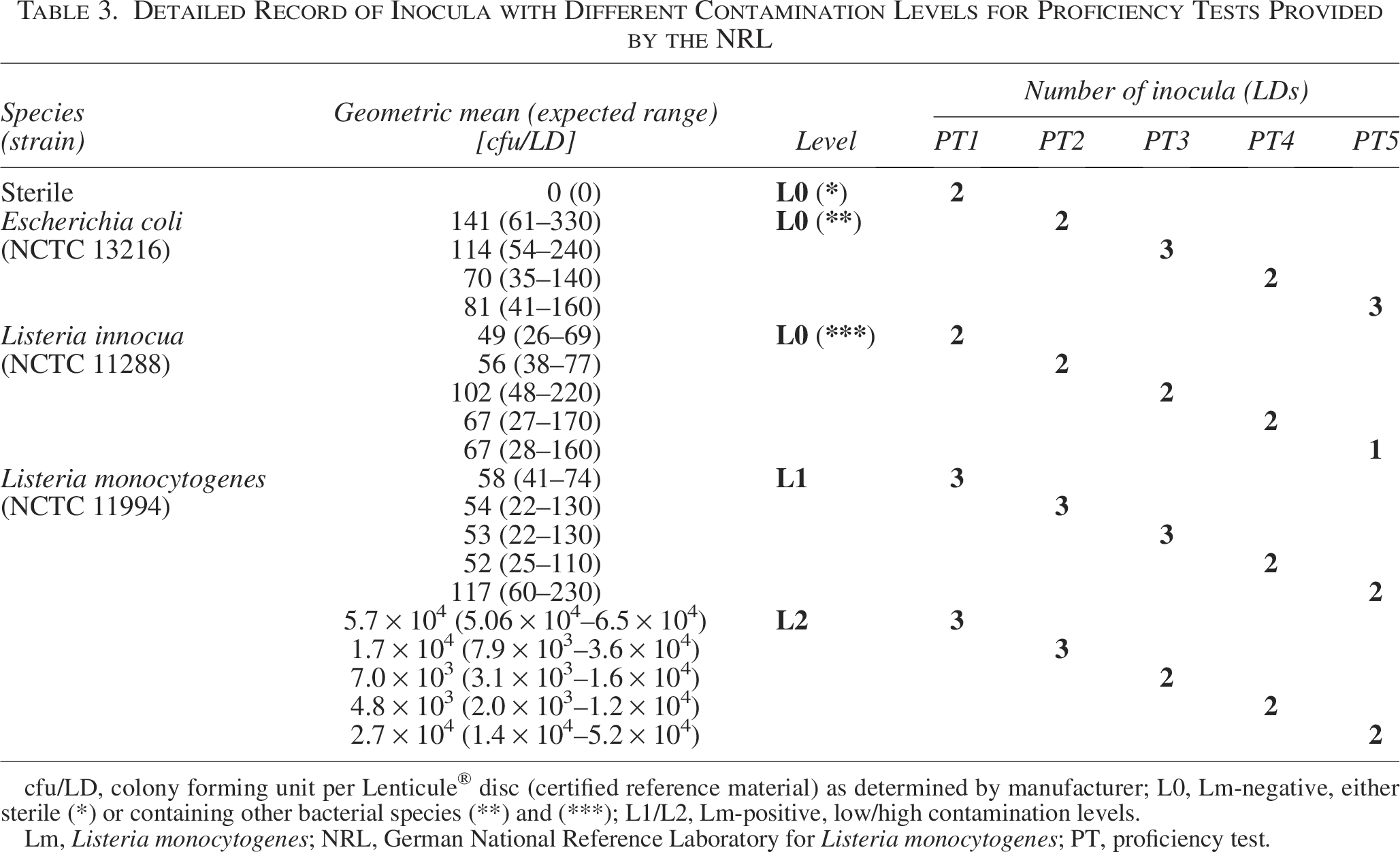
cfu/LD, colony forming unit per Lenticule® disc (certified reference material) as determined by manufacturer; L0, Lm-negative, either sterile (*) or containing other bacterial species (**) and (***); L1/L2, Lm-positive, low/high contamination levels.
Lm, Listeria monocytogenes; NRL, German National Reference Laboratory for Listeria monocytogenes; PT, proficiency test.
Matrices
Sample matrices examined in the PTs were chosen from foodstuffs typically contaminated with L. monocytogenes (Table 2). These foodstuffs were processed in-house in cooperation with the Food Technology Center at the BfR. Before assembling the sample-sets, the NRL tested samples from all matrix batches using the horizontal detection method of L. monocytogenes (ISO 11290-1:2005-1) to confirm the absence of L. monocytogenes. The PT-participants were supplied with L. monocytogenes-free material stored at −20°C until shipment.
Analytes and bacterial strains
The NRL provided the participants with Lenticule® discs (LDs; Public Health England, London, UK), which are certified as reference material (ISO 170134, ISO/IEC 17025). Sterile (L0*) LDs and LDs containing Escherichia coli (L0**) and L. innocua (L0***) were obtained as L. monocytogenes-negative analytes (L0). The same L. monocytogenes strain (LDs with low [L1] and high [L2] concentrations) was used to determine the laboratories’ ability to identify L. monocytogenes in different contamination levels consistently over the five successive years under study (Table 3).
Sample-set compilation, instructions, and shipping
For each PT, participants were provided with identical sets of matrix samples and LDs in blind coded tubes as analytes (Tables 2 and 3).
Prior to shipment, detailed instructions on how to proceed after package arrival, namely processing directives, confirmation of sample receipt, and result submission forms, were sent via e-mail to all participants.
The Styrofoam packages contained ice packs and from PT2 onwards EN 1283 certified temperature loggers (tempmate GmbH, Heilbronn, Germany) to monitor sample-set temperatures during overnight shipment. The packages were shipped as “Biological substances, Category B” (UN3373) within 24 h by an authorized parcel service.
Following receipt, the participants were asked to check the content of their packages for completeness and intactness and to report to the NRL. If the analysis was not started immediately, all matrices and analytes had to be stored at −20°C.
Sample contamination and analysis
The sample contamination procedure carried out by each laboratory was set in a standardized protocol. In brief, the selective enrichment medium (defined by the detection method) had to be added to sample bags containing thawed and portioned matrix. The frozen analytes were incubated at room temperature (RT) for 10–15 min and added to the samples by inverting the tube. While being fully immersed in the suspension, the LDs were re-hydrated at RT for 10–15 min before the samples were mixed thoroughly. Further sample processing was defined by the detection method chosen by the participants.
Result submission
To be considered for evaluation, results had to be submitted by the deadline. Additional information was requested to be reported in the result submission form to determine the actual analysis period in the laboratory (from sample contamination to final result validation) and the overall result submission period (from sample receipt to result submission) (Table 4).
Analysis Parameters and Overall Results of Proficiency Tests Determined with Qualitative Detection Methods Chosen by the Participants
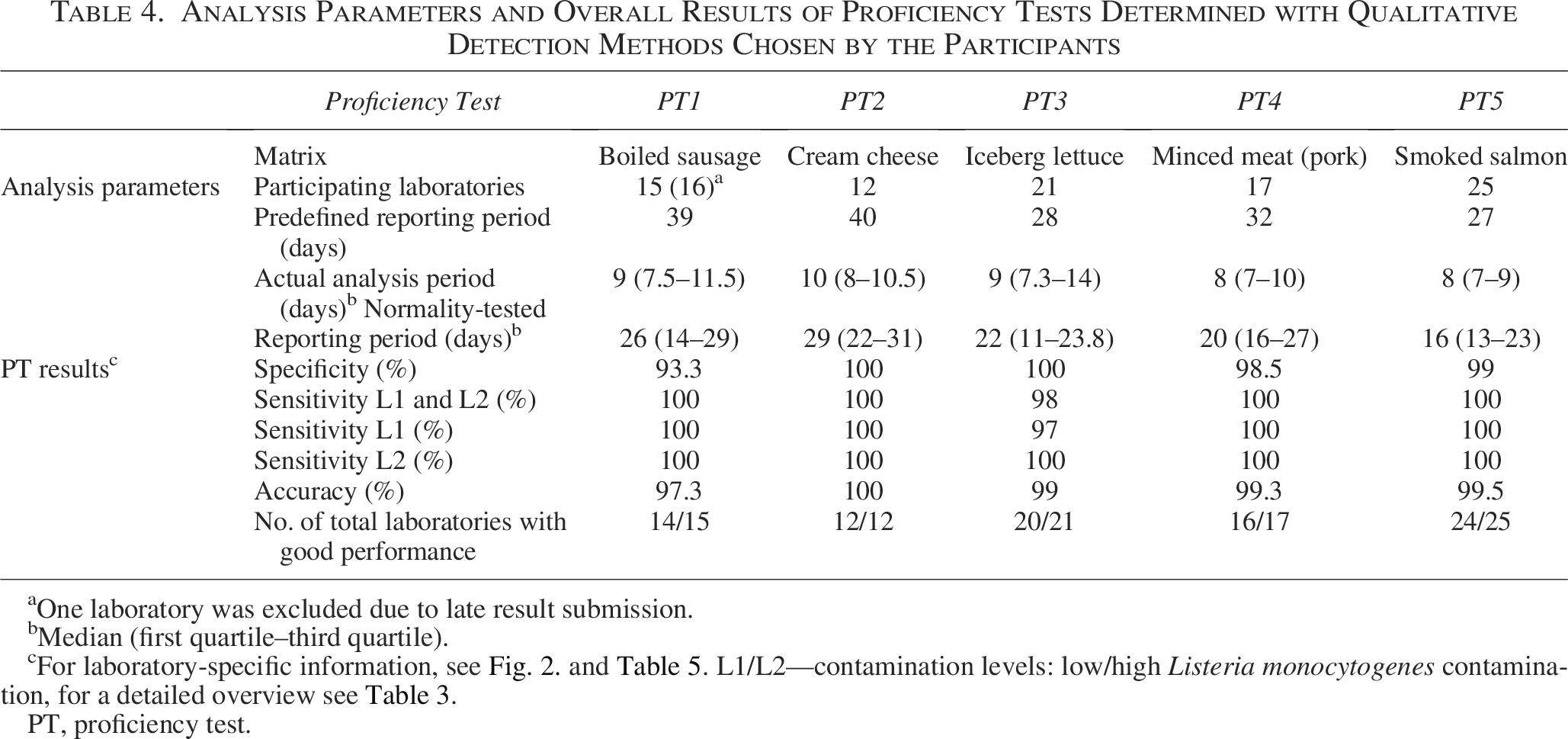
aOne laboratory was excluded due to late result submission.
Median (first quartile–third quartile).
For laboratory-specific information, see Fig. 2. and Table 5. L1/L2—contamination levels: low/high Listeria monocytogenes contamination, for a detailed overview see Table 3.
PT, proficiency test.
Performance evaluation
To evaluate the performance of participants, specificity, sensitivity, and accuracy were determined by the NRL for each laboratory (Fig. 2; Table 5) and each PT (Table 4). The specificity was calculated according to the formula [1]:
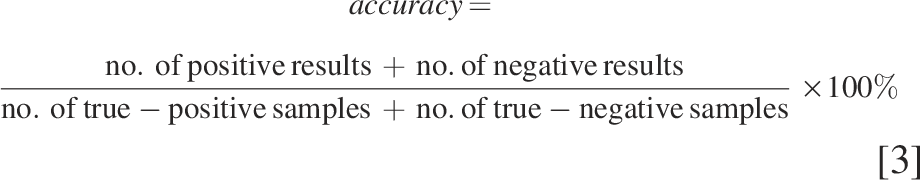
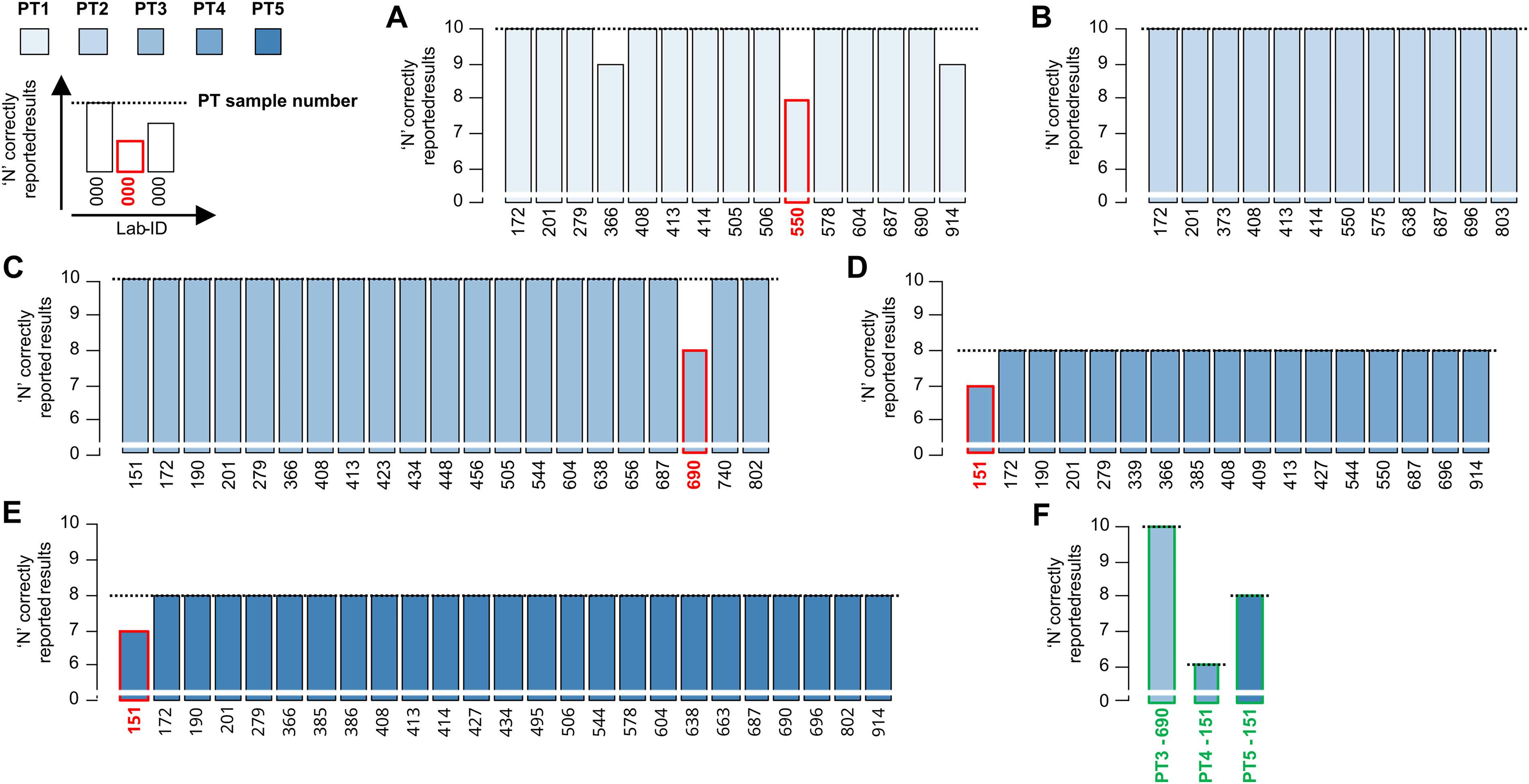
Performance of all laboratories in the proficiency tests organized by the NRL.
Performance Evaluation Based on the Parameters (Values in %) of Each Laboratory That Participated in the Proficiency Tests

Performance sequence examples: (1) exemplary laboratory participated in five PTs and achieved GP in all PTs (PT1-PT5). (2) Exemplary laboratory participated in PT3 only and achieved GP. (3) Exemplary laboratory participated in three PTs and achieved GP in PT1 and PT5 but failed in PT3. L1/L2, contamination levels: low/high Listeria monocytogenes contamination, for a detailed overview see Table 3.
ex., participant excluded from PT; GP, good performance; Lab-ID, laboratory identifier; n.a., not applicable, since laboratory did not participate in the respective PT; “N”-PTs, total number of PTs a laboratory participated in; PT, proficiency test.
The provided sample matrices were chosen from foodstuffs known to be frequently contaminated with L. monocytogenes either naturally or during food processing and were not decontaminated to preserve the microbial flora. The distribution of L. monocytogenes within the food matrix can be uneven. Thus, even though all matrices were pre-tested negative for L. monocytogenes by the NRL, there was a low probability of contamination with L. monocytogenes in the sample-sets. Therefore, a single false-positive test result reported per participant was accepted in PTs comprising larger sample-sets (n = 10). Because of the reduced number of samples (n = 8) per set in PT4 and PT5 (Tables 2 and 3), all samples supposed to be negative (L0) had to be correctly reported.
The sensitivity for the L. monocytogenes-positive contamination levels was calculated separately (sensitivity L1 or L2) and combined (sensitivity L1 and L2) according to the formula [2]:
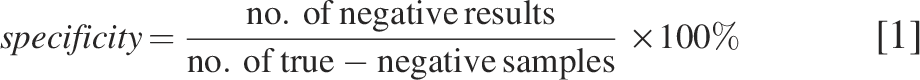
The accuracy was calculated according to formula [3]:
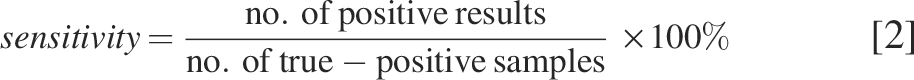
In general, the participants achieved good performance (GP) if specificity met or exceeded 75% (PT1 and 2), 80% (PT3), or 100% (PT4 and 5), and sensitivity was consistently 100%. All others were considered unsuccessful. From PT2 onwards, participants were offered a retest if they failed.
All participants received individual reports that included the overall PT results, a sample-specific evaluation of the participant’s results as well as feedback on specificity, sensitivity, accuracy, and ultimately on their performance.
Results
PT-wide analysis parameters and results
The number of laboratories registered per PT varied, ranging from 12 in PT2 to 25 in PT5 (Table 4). Temperature deviations of shipped sample-sets were not observed (data not shown). Transport damage to the shipped packages was only reported in PT2. The respective laboratories received a replacement.
Throughout 5 years of PTs with 91 registrations from 37 different laboratories, only one laboratory (Lab-ID 638) failed to report its results within the predefined reporting period and was consequently excluded from PT1 and the summary analysis (Tables 4 and Table 5). The predefined reporting period differed between PTs, from at least 27 days in PT5 up to 40 days in PT2 (Table 4). The median reporting period ranged from 16 days in PT5 to 29 days in PT2, whereby the actual sample analysis period in the laboratory was subjected to less pronounced fluctuations with a median of 8, 9, and 10 days in PT4/PT5, PT1/PT3, and PT2, respectively (Table 4).
The PT-wide results, including specificity, sensitivity, accuracy, and number of laboratories with GP, are presented in Table 4. The PT1-wide sensitivity was 100% regardless of the contamination level. Specificity (93.3%) and consequently accuracy (97.3%) were reduced due to false-positive results reported by three participants. The laboratories participating in PT2 reported all sample results correctly on the first attempt, achieving 100% specificity, sensitivity, and accuracy across PT2. One laboratory reported at least one incorrect result in PT3-5. The PT3-wide specificity and L2 sensitivity were 100%, but L1 sensitivity (97%) and consequently combined L1+L2 sensitivity (98%), as well as accuracy (99%), were slightly reduced due to the false-negative results reported in PT3. The overall sensitivity across PT4 and PT5 was 100%, regardless of the contamination level. However, overall specificity was impacted by a false-positive result in each of PT4 and PT5.
Laboratory-specific results
The number of correctly reported results per laboratory in each PT is summarized in Figure 2A–E. Laboratories that failed to achieve GP in PT3-5 were offered a retest as shown in Figure 2F. A detailed overview of the performance parameters per PT and throughout all PT participations, excluding the retest results, is given for each laboratory in Table 5.
Only 5 out of 37 laboratories (Lab-IDs 172, 201, 408, 413, 687) participated in all five PTs offered by the NRL. Their performance was flawless with 100% for all performance parameters in the five consecutive PTs.
Three laboratories participated in four out of five PTs. Laboratory 279 performed flawlessly in these PTs, while laboratory 366 achieved GP in all four PTs, with 90% accuracy in PT1 and error-free results in PT3-5. Laboratory 638 was the only laboratory that once failed to report its results within the mandatory reporting period, but reported flawless results in three additional PTs.
In addition to these 8 laboratories, a total of 20 laboratories performed exceptionally well in their PTs, achieving 100% across all evaluation parameters. Of these, 7laboratories participated in two PTs, while 13 took part in only one.
Nine laboratories participated in three out of five PTs. Among this group, four laboratories (Lab-ID 914, 550, 690, and 151) reported false results most frequently across PT1-5. Laboratories 914 and 366 reported one L0 (*) sample as L. monocytogenes-positive and still achieved GP in PT1. The performance of laboratories 914 and 366 was flawless in the other PTs (PT4-5 and PT3-5, respectively). Laboratory 550 failed in PT1 because it reported both L0 (*) samples as L. monocytogenes-positive, but performed flawlessly in the other PTs (PT2 and PT4). Across all PTs, the participating laboratories reported no false-negative results, demonstrating excellent sensitivity (100%), except for laboratory 690, which failed to correctly identify two L1 samples (low-level L. monocytogenes-positive) in PT3, resulting in an L1 sensitivity of 33%. However, laboratory 690 demonstrated excellent sensitivity (100%), regardless of the contamination level (L1/L2), in the other PTs it participated in (PT2 and PT4). Furthermore, laboratory 690 reported 100% correct results in the retest of PT3 and thus achieved GP. Laboratory 151 is the only laboratory that reported false-positive results, with one L0 (**) sample in both PT4 and PT5. Laboratory 151 also participated in retests of their failed PTs and ultimately reported 100% correct results, thereby achieving GP.
Conclusively, we counted 91 participants in the PTs organized by the German National Reference Laboratory for L. monocytogenes. Only five laboratories reported false results, of which four reported false results once and one laboratory twice. Thus, 85 out of 91 participations (93.4%) revealed excellent results (100%) in all three performance parameters. Only 3 (one twice) out of 37 participants failed to achieve GP in the first run, and every laboratory that was offered a retest then reported flawless results and finally achieved GP.
Discussion
In our report, we presented the results of PTs organized by the German National Reference Laboratory for L. monocytogenes (NRL). Overall, the official control laboratories have achieved outstanding results over several years, with only a few participants (4.4%) failing to achieve GP on the first attempt. In the following, we will discuss the observed pitfalls, the relevance and importance of PTs—particularly in relation to the guidelines, the provided matrices, and certified reference materials—and, ultimately, the implications for future PTs.
Similar to ring trials, PTs are used to verify the performance of laboratories by assessing specific performance criteria (Johnson and Cabuang, 2021). However, the goal of ring trials is typically an inter-laboratory comparison of a specific, often newly developed diagnostic method. In contrast, PTs can be conducted without restricting the diagnostic procedures used to achieve the performance criteria. In the presented PTs, the NRL permitted the participants to choose any diagnostic method to detect L. monocytogenes in the samples provided. The reason for this approach was (1) to enhance the attractiveness of our PTs, as many different PTs are available on the market, and (2) to allow participants to re-validate their current methods or to validate new or modified standard methods—an essential step, if these methods are to be accredited. By allowing any type of detection method, the participating laboratories were able to expand their range of validated methods, which could ultimately be included in their accreditation, if desired.
None of the PTs described above systematically required participants to report the methods applied. Nevertheless, participants frequently reported that they had isolated L. monocytogenes according to the standards of the “International Organization for Standardization” (ISO-11290-1), with modifications. Additionally, matrix-assisted laser desorption/ionization time-of-flight mass spectrometry, broad range diagnostic screening systems, or targeted polymerase chain reactions were used for species confirmation by some participating laboratories. In future PTs, a systematic query of the methods used by the participating laboratories will improve data quality, evaluation of results, and the comparison of performance with respect to the applied methods.
The majority of the official control laboratories achieved exceptional results, highlighting the reliability of official food control in Germany for detecting L. monocytogenes in food samples. However, one laboratory (Lab-ID 151) reported false-positive results in two consecutive PTs; probably due to cross-contamination during sample processing. We did not conduct troubleshooting with the participating laboratory as these issues did not recur in the retests, which resulted in excellent outcomes.
As part of a continuous improvement concept, participants in future PTs must submit a detailed report in case of deviations. This will help identify potential pitfalls and initiate sustainable corrective actions by the participating official control laboratories. The main goal is to start a process of improvement to enhance the reliability of diagnostic laboratory methods and prevent issues in routine practice.
The matrices provided in the PTs can all be allocated to the RTE food categories that are most frequently contaminated with L. monocytogenes according to the routine food monitoring samples collected in the EU (Fig. 1A) and therefore are investigated by the official monitoring laboratories on a regular basis. Between 2013 and 2021, a total of 230,173 samples belonged to the five RTE categories, of which approximately 5.0% (n = 11,524) tested positive for L. monocytogenes. About half of the L. monocytogenes-positive samples (n = 5,720) were from the “meat products” category within RTE foods (Fig. 1A). In Germany, 10–17% of “other preserved meat products,” a subcategory of “meat products” that includes among others “spreadable raw sausage” products, have been reported to be contaminated with L. monocytogenes (BfR, 2021). At the national level, the food safety authorities of the federal states collect and annually publish data on the prevalence of zoonotic pathogens and related antimicrobial resistances in food products and animals used for food production within the German National Zoonoses Monitoring Program (BVL, 2022a) . In this way, essential information for risk assessment is gathered, helping to better protect consumers from foodborne infectious diseases by guiding official food surveillance authorities in future food inspections. The food matrices investigated in the PTs were selected in accordance with the National Zoonoses Monitoring Programs on L. monocytogenes in food (Supplementary Table S1) (BVL, 2015; BVL, 2016a; BVL, 2016b; BVL, 2017; BVL, 2018; BVL, 2019; BVL, 2020; BVL, 2021; BVL, 2022b).
Sliceable raw sausage, an “other preserved meat product,” was sampled in the Zoonoses Monitoring in 2018 and was originally the selected matrix for a PT in the same year. However, this PT was never implemented because LDs were not commercially available at the time. This discontinuation of reliable certified reference material posed an issue for the upcoming years. Providing a reliable and stable reference inoculum that can be shipped to a large number of participants at feasible financial and environmental costs remained a challenge. Our experience underlines the importance of easily accessible and reliable certified reference material for controlled PTs and ring trials.
The herein analyzed PTs organized by the NRL addressed qualitative methods. However, the proper execution of quantitative L. monocytogenes methods is crucial in the EU. According to Regulation (EC) No 2073/2005, the EU allows the presence of L. monocytogenes in RTE food products, unlike the United States, which follows a zero-tolerance strategy (Regulation 9 CFR part 430, also known as the “Listeria Rule”). In the EU, L. monocytogenes is tolerated as long as the bacterial load is below 100 CFU in 25 g of the product matrix at the end of its shelf life. Products intended for infants or special medical purposes must be free from L. monocytogenes. Hence, official control laboratories in Germany/Europe need to reliably determine the L. monocytogenes counts in a food sample. This important diagnostic skill will be addressed in future PTs organized by the NRL.
The previous PTs included matrices representing the main RTE food products contaminated with L. monocytogenes. Future PTs will continuously comprise these most relevant food categories, with specific emphasis on “fish and fishery products” and “other conserved meat products.” Hence, we may include food matrices such as hot-/cold-smoked or marinated fish and spreadable raw sausages or minced pork and beef steak, which have been reported to be contaminated with L. monocytogenes more frequently in recent years (BfR, 2021). These RTE products were the cause of many foodborne outbreaks in Germany (Fig. 1D). Recently, we showed that the consumption of smoked and graved salmon provoked 22 outbreaks in Germany, with some having affected other European countries as well (Lachmann et al., 2022). RTE meat products have also been identified as the source of several protracted invasive listeriosis outbreaks. In southern Germany, different meat products, including smoked pork belly, from a single producer were associated with a large outbreak comprising 57 cases between 2012 and 2016 (Kleta et al., 2017). Between 2013 and 2018, several clonal lineages of L. monocytogenes were responsible for another large invasive listeriosis outbreak (83 cases), linked to plastic-packaged RTE meatballs (Luth et al., 2020). A comparatively small outbreak (Sigma1) of invasive listeriosis, with at least 13 cases between 2014 and 2019, was linked to the consumption of RTE sausages in German health care facilities (Adler et al., 2020; Lachmann et al., 2021). The largest listeriosis outbreak (Epsilon 1a; 112 cases) of the last decades in Germany was caused by a biocide-tolerant L. monocytogenes isolate. The outbreak was stopped after the product (blood sausage from a single producer) was withdrawn from the market (Halbedel et al., 2020).
Because of the increasing relevance of RTE foods in dietary habits associated with the high public health risk of L. monocytogenes, we will continue to offer relevant PTs to keep the excellent standards of the official control laboratories in Germany in line with future challenges in food safety.
Authors’ Contributions
C.-M.U.: Conceptualization, data curation, formal analysis, visualization, writing—original draft, review, and editing. M.A. and S.L.: Writing—review and editing. S.A.D.: Conceptualization, writing—review and editing. S.K.: Organization, conduction, and analysis of proficiency tests, conceptualization, writing—original draft, review, and editing.
Footnotes
Acknowledgments
On behalf of the National Reference Laboratory for Listeria monocytogenes, the authors thank the official control laboratories for their participation in their PTs. Furthermore, the authors are grateful to Dirk Meyer from their in-house Food Technology Center (Unit 41, BfR) and Marett Splett, Christine Fester, and Anne Stephan (Unit 47, BfR) for their excellent laboratory support.
Funding Information
This work was funded by the German Federal Institute for Risk Assessment (BfR project no. 47-003).
Disclaimer
The views and opinions of the authors expressed herein do not necessarily state or reflect those of the ECDC. The accuracy of the authors’ statistical analysis and the findings they report are not the responsibility of ECDC. ECDC is not responsible for conclusions or opinions drawn from the data provided. ECDC is not responsible for the correctness of the data and for data management, data merging, and data collation after the provision of the data. ECDC shall not be held liable for improper or incorrect use of the data.
Disclosure Statement
The authors have no conflicts of interest to disclose.
