Abstract
The discussion concerning the measure of the quality of a biobank should focus not only on the number of stored samples and their quality but also on the assessment of their access arrangements and governance. This article aims at contributing to the ongoing debate on samples and data access governance in biobanking by presenting the case of the Telethon Network of Genetic Biobanks (TNGB). We attempt to contribute to the need for clear and available access criteria and harmonization in access arrangements to maximize the influence of biobanks in the progress of biomedical research. We reviewed all the sample requests submitted to the TNGB from 2008 to 2020, focusing on those rejected by the Access Committee and the reasons behind the rejections. The analysis of the reasons behind the rejected requests allowed us to analyze how those relate to the issues of scientific misconduct, prioritization, and noncompliance with the biobank's mission. We discuss those issues in light of the actions and motivations used by TNGB in the access decision-making process. Based on this analysis, we suggest that a cross-implementation of a checklist for access assessment would improve the whole access process, ensuring a more transparent and smoother governance. Finally, we conclude that the TNGB's Charter and approach toward access governance could contribute as an important reference point to deal with the issues that have emerged in the international discussion on the topic.
Introduction
It has been said that a good biobank should be an empty biobank. This assumption seems consistent with the primary mission of biobanking, which is to facilitate access to the collection of human biospecimens and associated data for the scientific community. Therefore, in this article, we suggest that the measure of the quality of a biobank should be based not only on the number of stored samples and their quality but also on the efficacy of their access governance.
The governance of access to biospecimens and data is a set of rules and functional norms set up by each biobank to provide ethical guidelines and practical procedures to handle the requests of external researchers to access the biobank's samples and data.
Given that biobanks represent important infrastructures for the progress of biomedical research, ensuring appropriate and transparent governance mechanisms to manage access to their resources is crucial for successful international sharing and the progress of research.1,2
Access arrangements generally comprise basic rules for access, prioritization, and compensation criteria 3 and, among other governance possibilities, commonly rely on the Access Committee (AC) responsible for evaluating the requests submitted by researchers, ensuring the adequacy of the informed consent signed by biobank participants and the quality of project proposals. 4
Despite several international guidelines on biobanking that offer recommendations on setting access arrangements,5–8 the need to address unsolved challenges and issues persist at the level of individual biobanks.3,4,9,10 In particular, the latest literature on this topic has moved the debate on the adequacy of governance mechanisms to manage access to biobank data and its importance in a growing data-driven biomedical research. Biobanks, as is the case for other research infrastructures, should better define access arrangements that help overcome undeveloped data access criteria, the lack of sufficient oversight mechanisms, and the need of fairness and transparency in terms of access decisions. 1
Alongside these issues related to data access, the debate on access governance in biobanking—to which this article aims to contribute—seems to focus mainly on the need for clear and publicly available information on access conditions 11 and the importance of increasing harmonization in access arrangements between biobanks to implement large-scale biomedical research. 12
We aim at contributing to this ongoing debate by presenting the case of the Telethon Network of Genetic Biobanks (TNGB), which represents a compelling case study to shed light on how biobanks set and pursue improved access agreements. Indeed, the biobanks of the Network benefit from a long-lasting experience as they are among the oldest in Europe.13–15 Second, the TNGB has been repeatedly elected as a reference point for the implementation of international guidelines and recommendations.16–18 Third, TNGB's resources have contributed over time to achievements in many biomedical fields. 19 Fourth, the experience of TNGB with rare diseases provides insights into the access policy of samples presenting special concerns, due to their scarcity and value. 20 Finally, TNGB stands out for its commitment to patient organizations. 21 Particularly, although patient representatives are not part of the AC, they participate in the evaluation process with the Advisory Board and when samples and data are shared under the specific agreements between TNGB and Patient Organizations (cfr. Charter 6.1).
In this article, we present the strategies and solutions used by the TNGB to manage access to its collection. To do so, we analyzed all the requests submitted to TNGB, focusing on those that have been rejected by the AC, composed of the 11 directors of the Biobank partners of TNGB (Table 1). The discussion of the reasons behind these rejections offers the opportunity to reflect on open issues and concerns in access governance in biobanking, and to discuss the actions implemented by TNGB.
Telethon Network of Genetic Biobanks Governance Structure as Described Under Point 2 of the Charter
ELSI, ethical, legal and societal issues; RD, rare disease.
TNGB’ access arrangements
The TNGB is a nonprofit network created in 2007 interconnecting 11 Italian disease-oriented biobanks 22 through a centrally coordinated IT web-based platform which also includes a specific tool—the Request Control Panel (RCP)—for request management. TNGB's mission is to support research on rare diseases by facilitating access to high-quality biospecimens and associated data of interest to the scientific community, patients, and families.21,23
The TNGB originated as a research project supported by Fondazione Telethon—an Italian charity fostering research on rare diseases—and is regulated by its own charter, 24 which reflects the mission of the network and describes the governance 25 (Table 1), the rules for decision-making processes for accessing the samples, and the ethical guidelines and policies. The first draft of the charter was inspired by documents and guidelines available at the time.26–28 The value of the charter lies in the fact that it represents a common ethical framework for the network and a guarantee of transparency outward.
In the following, we describe the set of access arrangements that TNGB has implemented over the years. Its main pillars are the following:
A strong governance structure (Table 1) which constitutes an effective oversight and protective mechanism of access management, ensuring the objectivity of access review and the prevention of any conflict of interests in decision-making. A user-friendly sample online request form (Fig. 1) that contributes to promoting and facilitating access to the Network collection and guides researchers through the whole access process. Clear and publicly available rules of access (Table 2) that are equally applied to all researchers whether they work for for-profit or nonprofit organizations, including the Biobank's personnel within the Network (cfr. Charter 6.2, 6.5). A transparent evaluation process (Fig. 2) conveyed by the RCP which allows a shared visualization and management of the requests by all the members of the Network (cfr. Charter 6.1).
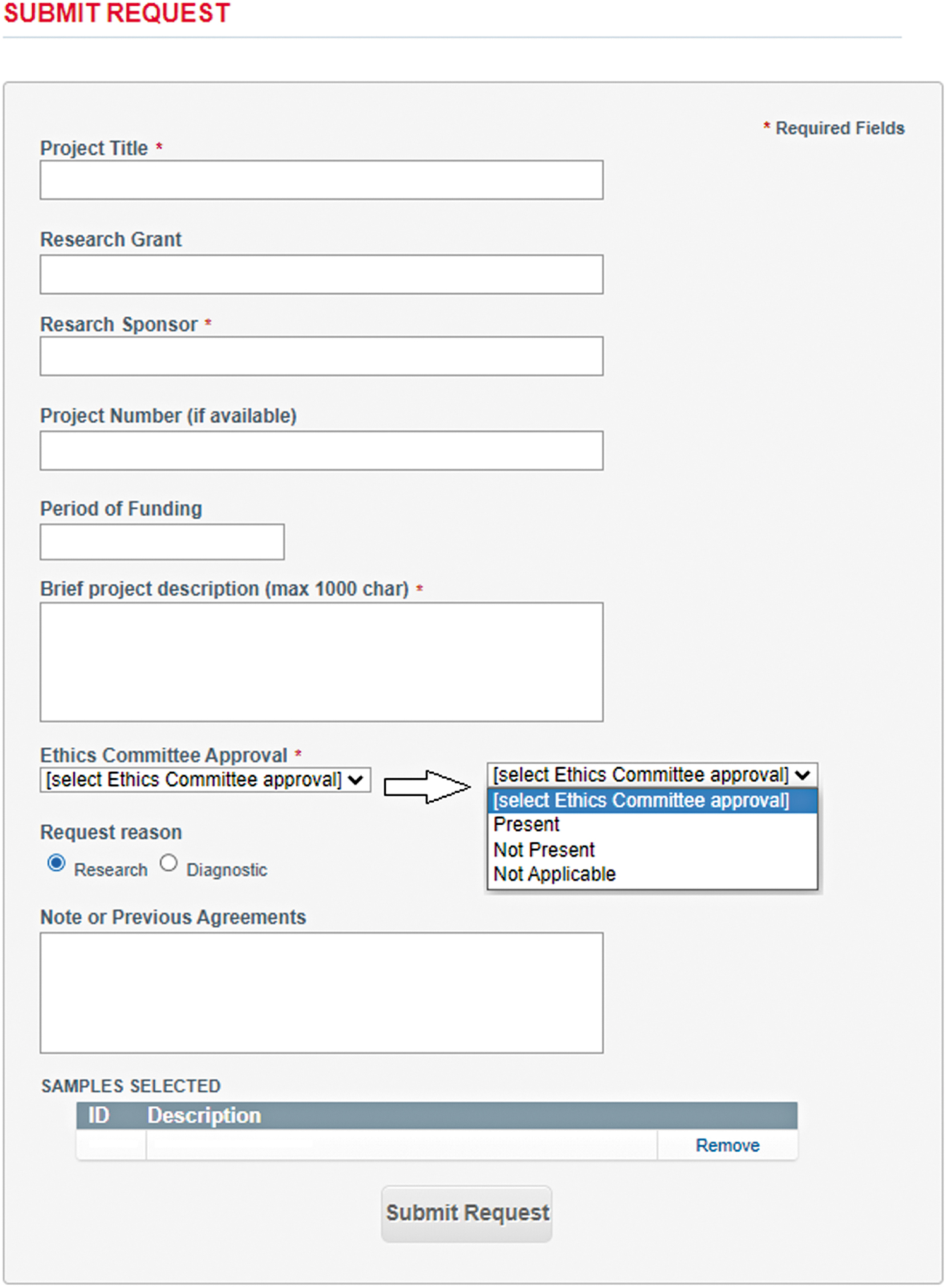
Sample request online form. The figure shows the sample request online form publicly available on the TNGB's website. TNGB, Telethon Network of Genetic Biobanks. Color images are available online.
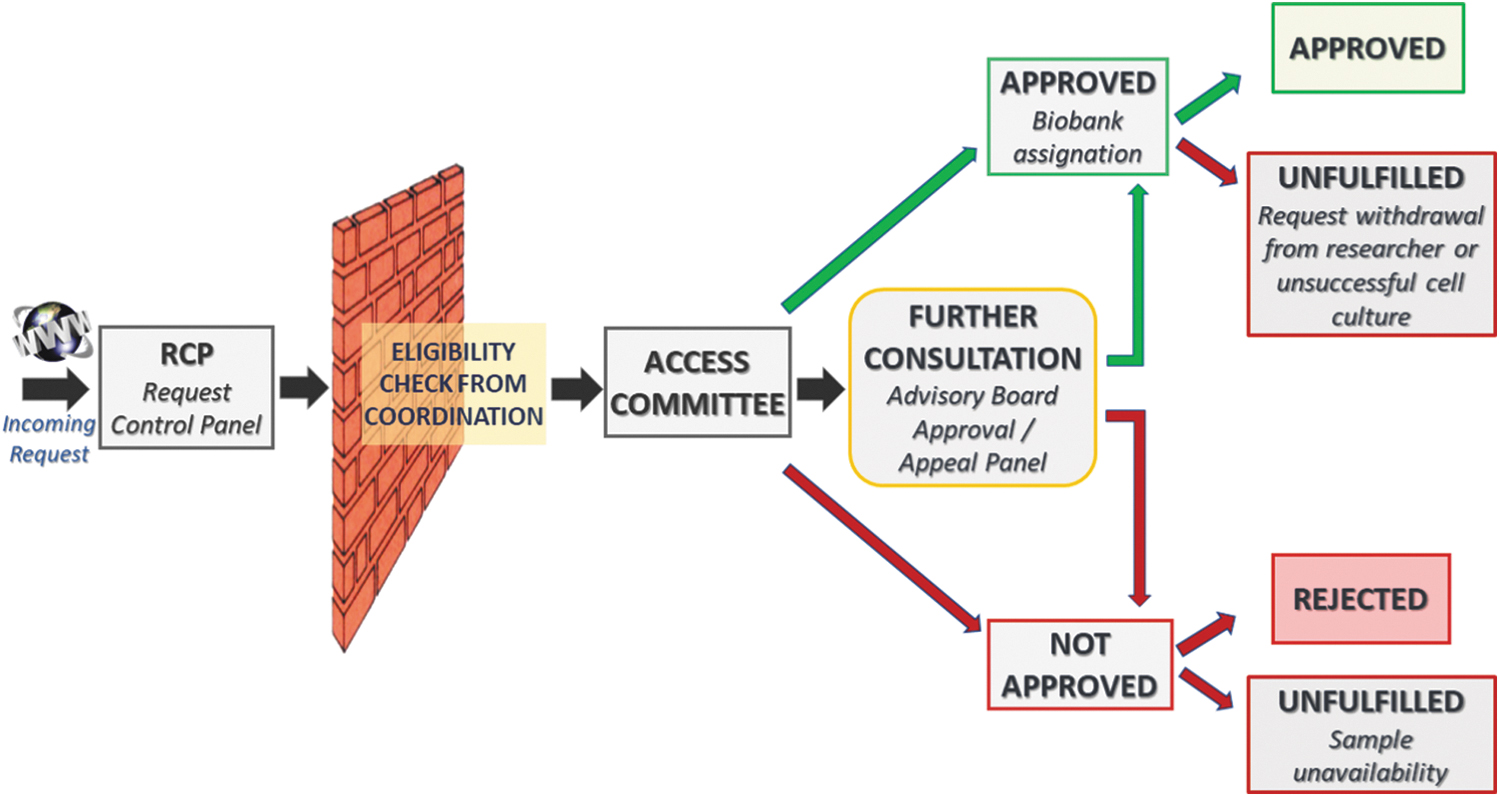
The flowchart of the request evaluation process by the Telethon Network of Genetic Biobanks. AC, Access Committee. Color images are available online.
Rules of Access by the Telethon Network of Genetic Biobanks
MTA, material transfer agreement.
It is worth noting that this set of arrangements ensures transparent access governance given that all the requests are assessed through the same process and by the same criteria regardless of the users' affiliation and the legal status of its home institution.
Moving to the practical side, for accessing the samples, researchers (from now on “users”) can request through the online form any samples listed in the TNGB online catalog. 29 The request must contain a description of the research project, the identification of the funding body, and the presence of the Ethics Committee approval (as shown in Fig. 1).
Once the request has been submitted, the evaluation process begins (Fig. 2). In brief, once the eligibility check is passed, the request is referred to the AC, which has 5 days to respond. In the case of a consensus, the request is assigned to the biobank(s); otherwise, in case of doubts about acceptance, a follow-up discussion among the AC is opened. The outcome of the discussion can result in two scenarios: the request is rejected following the TNGB's access rules or an external consultation is requested, that is, the Advisory Board or the Approval/Appeal Panel, which has to date never been consulted since the establishment of the TNGB.
During this phase, all communications to users are conducted centrally by the Coordinator or through the RCP.
There may be situations in which the request is unfulfilled, and this may occur in the case of sample unavailability, incomplete bureaucratic procedures (e.g., unsigned/unreturned documentation), or sample processing failure (e.g., unsuccessful cell culture).
Materials and Methods
We reviewed all the sample requests submitted to the TNGB from 2008 to 2020, analyzed the requests' flow and examined specifically those rejected during the evaluation process. Rejected requests are those which went through the whole evaluation process but were eventually declined.
The choice to focus on the rejected rather than successful requests offers the opportunity to reflect on the reasons for the refusals and, in particular, to understand the most common issues. Accordingly, this allows us to engage in a discussion regarding access governance in the light of the actions put into practice by TNGB and to understand values and motivations embedded in the access criteria that inform the AC's decision-making process.
Results
In the course of 10 years, the TNGB has received a total of 2731 requests for access for diagnosis and research purposes by researchers, clinicians, and patients from all over the world. This number also includes duplicate entries, technical tests, and user cancellation before the evaluation process. Out of all the requests, 2700 were evaluated in compliance with the TNGB access policy, and the samples were shipped according to the recommendations and laws in force. Fifty-seven of these requests, which are the focus of this article, were rejected.
Table 3 compares the accepted and rejected requests, highlighting their scope, the type of institution where the applicant works and, finally, at what level of the governance policy they were approved/rejected. It also shows that the majority of the samples were requested for research purposes, which was consistent with the mission of the network.
Comparison Between Accepted and Rejected Requests Considering Their Scope, the Type of Requesting Institution and the Level of Decision During the Evaluation Process
n.a., not applicable.
In Table 4, we grouped the rejected requests in five main categories. Three of them (scientific misconduct, prioritization, and noncompliance with TNGB's mission), worthy of being more closely examined, are the specific focus of our analysis, while the remaining categories (researcher withdrawal and sample unavailability) have been excluded because they do not require further explanations.
Reasons for Request Rejection
TNGB, Telethon Network of Genetic Biobanks.
Table 5, therefore, describes in detail the selected categories of rejected requests and links them back to the Network access rules (Table 2).
Comparison Between Rejected Requests and Telethon Network of Genetic Biobanks' Access Rules
Specifically, under the first category (“scientific misconduct”), we collocated the cases in which the AC rejected a request because of some kind of misbehavior or mistake on the user's side. In the second category (“prioritization”), there are the cases in which the AC detected issues regarding priority setting and fair allocation of biospecimens in the requests. Finally, under the third category (“non-compliance with TNGB's mission”), we collocated the access requests that the AC considered noncompliant or in conflict with the general mission of the TNGB.
Discussion
This section discusses the above results by stressing the actions used by the TNGB to deal with its access governance. We suggest that those practical solutions are embedded in the access criteria that inform AC's decision-making process and they reflect the mission and the principles of the Network (Table 6). At the same time, the discussion of the results is an opportunity to reflect on the need of further implementation.
Actions and Motivations Informing Access Committee's Decision-Making
Scientific misconduct
The ongoing debate on access arrangement in biobanking has repeatedly focused on the need to provide clear criteria for researchers who seek to gain access in the easiest way possible through a transparent evaluation process. 2 As such, TNGB's Charter already offers an unambiguous set of basic rules for access to its collection and a user-friendly access procedure.
Nevertheless, the AC is very committed to preventing any researchers' scientific misconduct in accessing a biobank's collection by putting in place the following actions:
a rigorous check of the proposed research project and of the quality and safety procedures applied by the User;
a careful review of the User's CV and scientific production and obtaining information on the procedures applied in the sample management;
a review of the previous requests (if any) from the same User to reveal missing acknowledgments in scientific products or reports on their research results or other misbehavior.
Behind those practical actions, we can appreciate the commitment of TNGB to ensuring the highest standards of the research and the control over researcher's qualifications concerning the respect of patients contributing their samples and data. Furthermore, the emphasis on the rule of reporting back the results to, and acknowledging the biobank, is fundamental to promote the impact and visibility of the biobank within the scientific community 2 and to ensure transparency and accountability toward different stakeholders, especially patients. 20 In other words, reporting the results to the Biobank Directors and acknowledging the service provided in their scientific articles (or any other scientific product) are considered a good practice among researchers and a form of compensation for biobank services,30–32 contributing to the enrichment of the sample value by having updated linked data and contributing to the sustainability of the biobank. 33
Finally, it is worth noting that the interest behind those solutions is to foster the relationship between the researcher and biobanks, improve the spirit of scientific collaboration as well as maintain the commitment to the patients who have provided the biospecimens and their consent for research.
Prioritization
An emerging topic in the debate surrounding access governance in biobanking is the need to introduce priority setting criteria in the access decision-making process.3,9 Especially regarding its commitment to rare disease research, TNGB has implemented some actions to ensure it does not waste precious and extremely rare samples that can be used for patients' diagnosis:
a rigorous check of the expertise of the User in the specific topic of the proposed research project;
verification of the consistency of the sample number/type with the disease prevalence in order not to waste precious and rare samples that can be used for patients' diagnosis and more valuable research projects;
ensure that valuable and scarce samples are devoted only to important and ambitious projects.
These solutions reflect some of the core values of the TNGB, which are, at the same time, a strong commitment to patients and patient associations and the effort to maximizing the scientific and social impact of biobanks' service and mission.
Furthermore, the actions put in place by TNGB are aimed to respect the dual role of biobanks, which is managing the collections of biospecimens and acting as a service provider, trying to find a balance between the importance of the network and the need for the local research and clinical context.
Noncompliance with TNGB's mission
Sharing biobank resources entails the concrete risk of misuse of samples and data collected by a biobank clashing with its mission. 34
Accordingly, TNGB's access governance has been implemented to:
avoid the situation that any commercial interests, disguised as research purposes, are in place when sharing biobank samples and data;
detect companies acting as intermediaries in seeking out and, in some cases, collecting biobank resources in place of their customers.
Those strategies are consistent with the foundational mission and values of biobanking activities, focusing on the common benefits of biomedical research and public health. 35 In particular, those actions implemented by TNGB to guarantee that samples and data are available to researchers for biomedical research purposes are based only on the respect of the principle of gratuity of the human body, which is consistent with an understanding of biobanking as a system based on altruism and solidarity34,36 and, at the same time, on the promotion of individual responsibility of each researcher in the needs of the Network to keep track of the samples provided.
In light of the discussion, and considering both the issues faced and the solutions that emerged through our analysis, we suggest that further studies and efforts are needed for the development of a checklist for access evaluation. Having an international discussion for a shared understanding regarding the main elements to be evaluated within the access procedures would render governance in biobanking more transparent and standardized. In this regard, TNGB will continue to share its implementation with the existing Rare Disease infrastructures both at the national and international level.
Coming as an output of the whole access procedure review, the proposed checklist should assess and summarize the adequacy and completeness of the user's request, relying on a quantitative evaluation system, indicating thresholds to be reached to have the sample request approved.
Different parameters (e.g., conformity with access rules, originality and feasibility of the project, user's CV, previous publications, and so on) will be selected on the basis of the Network access criteria and related scores will be identified. The qualitative evaluation will support the final AC's response (request accepted/rejected). Furthermore, the checklist will make clearer the difference between those rejections due to noncompliance by default (i.e., conflict with biobank mission) and those for which the rejection resulted from failure to reach the threshold of other parameters (i.e., CV, originality of the project).
In our vision, this evaluation checklist should be seen as a dual-purpose tool. In the first place, it represents a standardized summary of the access evaluation process for the single biobank or network, necessary to ensure transparency and fairness in decision-making and to keep track of the requests' history.
Second, for the users—both internal and external researchers—the checklist will aid in understanding the reasons behind the rejection through a clear exhibition of the scores obtained for each parameter.
In conclusion, in this study, we performed an analysis of the flow of the sample requests submitted to the TNGB from 2008 to 2020. The objective was to identify the main reasons behind the rejections of the requests and to present the strategies used by the Network to face them.
Specifically, we have highlighted the actions used by the TNGB to tackle the problem of scientific misconduct, prioritization, and noncompliance with its mission and we have shown the values and motivations embedded in its access policy and decision-making process.
Considering this, we believe that the current approach of the TNGB toward access decisions—along with the implementation of a checklist for access evaluation which we have proposed in this article—could contribute to overcoming some of the issues identified by the international debate on the topic.
In particular, regarding the issue in the biobanking community of the lack of clear and available access criteria, the TNGB has faced and overcome this deficiency by making its charter and the rules of access publicly available from the start through the Network's website. We believe that, along with the implementation of the checklist of evaluation, it should represent an example of shared access criteria to foster transparency.
Second, concerning the need for harmonization of access arrangements between biobanks aiming at facilitating sample and data sharing, and to foster research and innovation derived by biobanks, TNGB's access arrangements should be seen as an international reference point. It is not a coincidence, indeed, that it has already been endorsed by all the partner biobanks and included as a use case in European and national infrastructures/projects (i.e., BBMRI-ERIC, BBMRI.it, RD-Connect, etc.), including the Italian Ministry of Health which has selected TNGB as an example to be included in their recommendations for biological material management. 37
Footnotes
Acknowledgments
The authors thank current and former members of the TNGB Advisory Board for their advice, particularly, in the sample request evaluation process. The authors also give special thanks to Lucia Monaco, promoter of the TNGB initiative, Manuela Battaglia and Ermanno Rizzi of the Research Office of Fondazione Telethon (Italy) for their continued support and assistance. Telethon Network of Genetic Biobanks members (current directors): Domenico Coviello, Anna Lena Zecchinelli, Alessandra Renieri, Elena Pegoraro, Monica Sciacco, Francesca Andreetta, Giuseppe Merla, Vincenzo Nigro, Barbara Garavaglia. Telethon Network of Genetic Biobanks members (former directors): Mirella Filocamo, Chiara Baldo, Stefano Goldwurm, Roberto Cilia, Corrado Angelini, Maurizio Moggio, Marina Mora, Luisa Politano.
Author Disclosure Statement
No conflicting financial interests exist.
Funding Information
This work was supported by the TNGB (Project No. GTB18001) and partially by grants from the Ministero della Salute “Ricerca Corrente 2021” (to Istituto Ortopedico Rizzoli, Bologna, Italy).
