Abstract
Venus has become a target of astrobiological interest because it is physically accessible to direct exploration, unlike exoplanets. So far this interest has been motivated not by the explicit expectation of finding life but rather by a desire to understand the limits of biology. The venusian surface is sterilizing, but the cloud deck includes regions with temperatures and pressures conventionally considered compatible with life. However, the venusian clouds are thought to consist of concentrated sulfuric acid. To determine if any fundamental features of life as we understand them here on Earth could in principle exist in these extreme solvent conditions, we tested several simple lipids for resistance to solvolysis and their ability to form structures in concentrated sulfuric acid. We find that single-chain saturated lipids with sulfate, alcohol, trimethylamine, and phosphonate head groups are resistant to sulfuric acid degradation at room temperature. Furthermore, we find that they form stable higher-order structures typically associated with lipid membranes, micelles, and vesicles. Finally, results from molecular dynamics simulations suggest a molecular explanation for the observed robustness of the lipid structures formed in concentrated sulfuric acid. We conclude with implications for the study of Venus as a target of experimental astrobiology.
Introduction
The catalog of confirmed exoplanets continues to grow (Christiansen, 2022), and the James Webb Space Telescope has improved our ability to measure the transmission spectra of exoplanet atmospheres (e.g., Ahrer et al., 2023; Tsai et al., 2023). However, characterizing the physical properties of exoplanets that are relevant to habitability remains extraordinarily challenging. In contrast, our solar system can be explored directly, and Mars, Europa, Enceladus, and more recently Venus are proving of special interest. An astrobiological perspective on the solar system seeks to compare what we know about Earth biology with what we are learning about non-Earth environments through experiments and space missions. Some of our solar system neighbors, like Venus, may appear at first glance to be completely uninhabitable, yet there is nonetheless active speculation about the potential habitability of Venus’s sulfuric acid clouds (see, e.g., Bains et al., 2024b, 2021a; Grinspoon and Bullock, 2007; Kotsyurbenko et al., 2024, 2021; Limaye et al., 2018; Mogul et al., 2021a; Morowitz and Sagan, 1967; Patel et al., 2022; Schulze-Makuch and Irwin, 2006; Seager et al., 2021). However, considering whether a planet is habitable requires an understanding of whether the conditions on that planet are consistent with the conditions required by life (regardless of whether life actually exists there). Earth life remains the basis for such planetary habitability assessment, but the extent to which specifics of Earth life can be considered universal is disputed.
Venus is a critical test case for such astrobiological studies because the clouds consist of liquid concentrated sulfuric acid, which is considered incompatible with life, and contain almost no water, which is considered essential for life (e.g., Hallsworth et al., 2021). Despite these challenges, speculation about life in the clouds of Venus persists because of anomalies in its atmospheric chemistry (e.g., Bains et al., 2021b; Limaye et al., 2018; Mogul et al., 2021a, 2021b; Petkowski et al., 2024b). A rigorous astrobiology research program, centered around organic chemistry, should consider the fundamental features of life as we know it and seek to explore its limits with reference to known environments on other worlds, including Venus.
Organic chemistry in concentrated sulfuric acid is rarely studied yet surprisingly rich, with recent work supporting the notion that complex organic molecules, including amino acids and nucleobases, can be stable in this unusual solvent (Petkowski et al., 2024a, 2025; Seager et al., 2024a, 2023, 2024b; Spacek et al., 2024). These results build on isolated reports from decades ago that considered the chemical behavior of organic molecules in concentrated sulfuric acid long before its importance for planetary science was realized or the sulfuric acid composition of the venusian clouds was known (e.g., Albright et al., 1972; Habeeb, 1961; Miron and Lee, 1963; Reitz et al., 1946; Schumacher and Günther, 1983; Steigman and Shane, 1965; Wagner and von Philipsborn, 1971).
That simple organic molecules can be stable in concentrated sulfuric acid is an interesting observation, and without such stability, life could not be possible in it. But life also requires complex molecular structures for biological function. One fundamental feature of life is cellularity: the differentiation of “inside” (the contents of a cell, including information, molecules, and all their interactions) and “outside” (the environment), in addition to a mechanism for communication and exchange between the two. Cellularity also enables the distinction between encoded genotype (information internal to the cell) and expressed phenotype (the relationship of that information with the environment), a prerequisite for Darwinian evolution. Interpreting cellularity as a feature of life does not necessarily imply Earth biology (a DNA-encoded genotype, for example).
Biology uses lipid membranes to define cells. Unlike proteins and nucleic acids, biological lipids exhibit a broad range of chemistries. They must form effective membranes that can grow, divide in response to the cellular division machinery, and remain stable in the given cell’s environment. The primary role of cell membrane lipids is to physically define the cell and thereby demarcate inside from outside. The cell membrane is especially important in extreme environments because it must help maintain the homeostasis of the intracellular environment against otherwise harsh external conditions. Reasoning that with our current understanding of biology, we cannot envision life without cells, we here consider whether simple lipids might be resistant to sulfuric acid, and further, whether they can form higher-order structures such as membranes and vesicles typically considered a prerequisite for life-like phenomena.
The formation of micelles or membranous structures in nonaqueous solvents has attracted little attention. We identified only 11 papers that report the behavior of surfactants and other amphiphilic molecules in concentrated sulfuric acid (McCulloch, 1946; Menger and Jerkunica, 1979; Müller, 1991b, 1991c, 1991a; Müller and Burchard, 1995; Müller and Giersberg, 1992, 1991; Müller and Miethchen, 1988; Steigman and Shane, 1965; Torn and Nathanson, 2002). Several of these studies report the formation of micelles and other lipid structures.
Our research has also been inspired by the extensive literature on the potential role of fatty acids in the emergence of life on Earth (e.g., Apel et al., 2002; Deamer, 2016; Gebicki and Hicks, 1973; Hargreaves and Deamer, 1978; Mansy, 2009). These simple single-chain lipids can form vesicles made of canonical lipid bilayers if the pH of the aqueous solution is near the pKa of the fatty acid carboxylic acid head groups (typically ∼ pH 8 in the context of a membrane) (Kanicky and Shah, 2003). Under these conditions, approximately half of the headgroups will be deprotonated, and the other half protonated, so that charge interaction networks among the headgroups stabilize the hydrophilic bilayer surfaces.
Here we identify several simple lipids that appear to have similar properties in the context of aqueous sulfuric acid solvents in concentrations up to 90% (v/v). We challenge the assumption that concentrated sulfuric acid is inherently incompatible with basic properties of the chemistry of life and specifically show that complex solvent-enclosing lipid vesicles can form, are stable, and can dynamically rearrange and form higher-order structures in concentrated sulfuric acid. While the extreme corrosiveness of sulfuric acid limits our experimental toolkit and ability to comprehensively characterize the biophysical properties of these structures, we posit that they are consistent with lipid bilayer membranes. Our data suggest that micelles, vesicles, oil droplets, and under some conditions extensive membrane networks may be forming. These results signal an opportunity to experimentally push the boundaries of prebiotic chemistry into a regime that is relevant for studying Venus from an astrobiological perspective.
Materials and Methods
General
Lipids were obtained from Sigma-Aldrich (decyltrimethylamine, octadecyltrimethylammonium, decylphosphonate) or Combi-Blocks (octadecyl sulfate, decylsulfate) at the highest available purity without further purification. Sulfuric acid was from Sigma-Aldrich (nominally 95–99%). Experimental sulfuric acid concentrations are reported as volume/volume (v/v) percentages (volume sulfuric acid/total volume sulfuric acid + water) unless otherwise noted. Water was from a Millipore Milli-Q filtration system with a polisher filtration attachment, rated to dispense water with a resistivity of 18.2 MΩ·cm at 25°C, with total organic carbon ≤5 ppb. Glass vials for sample preparation were from ChemGlass, with PTFE-backed caps. Glassware to be used with sulfuric acid was rinsed with sulfuric acid prior to experimental use. Deuterated NMR reagents and internal standards were from Sigma-Aldrich or Cambridge Isotope Laboratories.
NMR spectroscopy
Samples were prepared by measuring out the appropriate mass of dry lipid in a 4-mL glass vial and adding the previously prepared aqueous sulfuric acid. The vial was sealed with a PTFE-lined cap, and the mixture was gently inverted by hand and allowed to incubate at room temperature for at least 1 h. For extractions, the CDCl3 was added directly to the vial, the mixture inverted gently by hand, and the phases separated over several minutes. The solution was transferred to prerinsed NMR vials with a prerinsed glass Pasteur pipette. NMR spectra were acquired on a Varian INOVA NMR spectrometer operating at 400 MHz for 1H spectra and 161 MHz for 31P spectra (pulse sequence s2pul). 1H spectra in water were referenced to sodium 2,2-dimethyl-2-silapentane-5-sulfonate at 0 ppm. 1H spectra in CDCl3 were referenced to tetramethylsilane at 0 ppm. Measurements were locked to D2O or CDCl3. Chemical shifts (δ) are shown in parts per million (ppm). Spectra were processed and analyzed with MestReNova v14.2.0 (Mestrelab Research) (Willcott, 2009). All full-spectrum NMRs used for preparing figures are in the Appendix to the Supplementary Data.
Dynamic light scattering measurements
Samples were prepared by measuring the appropriate mass of dry lipid in a 4-mL glass vial and adding 2 mL of 70%, 80%, or 90% aqueous sulfuric acid. Samples were placed on a rocker for ∼2 h at 20 rpm, after which they were transferred to a glass cuvette with a PTFE cap (Malvern PCS1115), and the day 0 measurements were taken. For the first set of experiments, readings were taken after 24 h and after 5 days. For subsequent analysis of the 50/50 mix of decylsulfate and decyltrimethylamine C10 (SDS, DTMA) or octadecyl sulfate and octadecyltrimethylammonium C18 (SOS, TMO) lipids, measurements were taken hours after preparation, after 24 h, 3 days, and 7 days, unless otherwise indicated. After each measurement, the sample was immediately returned to the rocker. Before each use, vials and cuvettes were cleaned by rinsing with Milli-Q water, detergent, another rinse of Milli-Q water, and then acetone with a vacuum cuvette washer. Measurements were acquired on a Zetasizer Nano ZS at a backscatter detection angle of 175° at 25°C. Three measurements were taken in three runs each for 60 s in the first set of experiments, and for the 50/50 mix of decylsulfate and decyltrimethylamine measurements were taken in three runs for 1000 s per run. The viscosity and refractive index for the indicated sulfuric acid concentrations were taken from the literature (Beyer et al., 1996; Liler, 1971; Rhodes and Barbour, 1923) and used as dispersant parameters in the respective measurements, and attenuator position was determined automatically. Particle sizes were taken from the resultant intensity distribution.
Confocal microscopy
For visualization of 70%, 80%, or 90% aqueous sulfuric acid samples prepared identically as in dynamic light scattering (DLS) experiments, 2 µL of sample was placed in the center of a µ-slide 18-well sterilized, uncoated glass bottom plate (Ibid.). For subsequent additional confocal experiments, vesicles were first prepared in water by measuring the appropriate amount of lipid for a final concentration of 75 mM in a 4-mL glass vial and adding 2 mL of water. After vortexing for ∼5 s, 75 mM KOH was added to reproduce conditions used in previous fatty acid studies, and the sample was again vortexed. Aliquots of this sample were added to a µ-slide 18-well glass bottom plate (Ibidi) to five wells, and sulfuric acid was added for a 10%, 50%, 70%, 80%, or 90% final concentration of sulfuric acid. For imaging of samples between glass slides, slides were placed in 1 M NaOH for 5 min, 1 M HCl for 5 min, then washed with deionized water. Two microliters total (lipids plus sulfuric acid) were placed in the center of an imaging spacer (VWR) between slides. For all samples, BODIPY (Sigma) was added in a final concentration of 2.5 µM. Samples were excited at 568 nm and imaged by confocal microscopy on an A1R/Ti setup (Nikon) using a 1.45 NA 100× CFI Plan Apochromat objective.
Molecular dynamics simulations
Membrane patches (100 × 100 Å) of the pure decylsulfate (Sys1), pure decyltrimethylamine (Sys2), and a 50/50 mix of decylsulfate and decyltrimethylamine (Sys3) were generated by the CHARMM-GUI (Jo et al., 2008) membrane builder. TIP3P waters were added 25 Å above and below the bilayer using the autosolvate plugin in visual molecular dynamics (VMD) (Humphrey et al., 1996). In-house Tcl scripts were used to replace all water molecules with sulfuric acid and hydronium ions such that the final concentration of sulfuric acid was ∼70% by volume with a net zero charge. Two water molecules were removed for every sulfuric acid added, and one water molecule was removed for each hydronium ion added. The ratio of protonated and deprotonated sulfuric acid molecules was adjusted for each system to give a net zero charge; this resulted in 2672 deprotonated and 1838 protonated sulfuric acid molecules for Sys1, 3228 deprotonated and 1506 protonated for Sys2, and 2894 deprotonated and 1840 protonated for Sys3. The total number of sulfuric acid and hydronium molecules was 4510 and 6012 for Sys1, 4734 and 5788 for Sys2, and 4734 and 5788 for Sys3, respectively. The standard CHARMM36 (Huang and Mackerell, 2013; Vanommeslaeghe and MacKerell, 2012) force field was used for the lipids and sulfuric acid, while parameters for the hydronium ions were taken from previously published results (Sagnella and Voth, 1996). The NAMD 2.14 (Melo et al., 2018; Phillips et al., 2020) program was used to perform all classical and hybrid quantum mechanical/molecular mechanical (QM/MM) simulations, while ORCA (Neese et al., 2020) was used to perform quantum calculations. Analysis was performed, and images were generated using VMD. Results of simulations are reported as significant when two populations exhibited a significant difference when evaluated using a pair sampled t-test for means with a hypothesized difference of zero.
To prepare for QM/MM simulations, all systems were minimized for 1000 steps and equilibrated in the NpT ensemble at 1 atm using a hybrid Nosé–Hoover Langevin piston method, and temperature was controlled using Langevin dynamics with a damping coefficient of γ = 1. Nonbonded interactions were switched off at 12 Å, and long-range interactions were calculated using the Particle Mesh Ewald method. Bonds involving hydrogen atoms were fixed using the SHAKE algorithm. At the end of the equilibration, quantum regions were selected to include three interacting lipids: two decylsulfate and one decyltrimethylamine in Sys3, one sulfuric acid molecule and five hydronium molecules for Sys1, two sulfuric acid molecules and one hydronium molecule for Sys2, and three hydronium ions and one sulfuric acid molecule for Sys3. Three such regions were selected for each system, which resulted in nine total QM/MM simulations (Sys1a-c, Sys2a-c, and Sys3a-c). The hydronium ions, the sulfuric acid molecules, and the head groups of all lipids to the C1 carbon atom of the lipid tail were all treated at the QM level (49 atoms total for Sys1a-c, 62 atoms total for Sys2a-c, and 49 atoms total for Sys3a-c), which resulted in a multiplicity of 1 and a net zero charge for all systems. The HF-3c semiempirical Hartree Fock method was used to simulate the QM region (Sure and Grimme, 2013); it was chosen due to its ability to describe large biomolecular systems to an accuracy that approaches large-basis methods at a fraction of the computational cost. Interactions between the QM and MM region were treated with an electrostatic embedding scheme, and covalent bonds split at the QM/MM boundary were treated with a Charge-Shifting method. Charge distribution in the QM region was taken from ORCA and updated at every step. A 0.5 fs timestep was used, and the remaining parameters for the classical region were identical to those used for the equilibration. Self-consistent field electron density and electrostatic potential maps were generated using the orca_plot command and the molecular orbitals output by ORCA and plotted with an isovalue of 0.09.
Results
Lipid selection
We began by selecting a set of simple lipids that are commercially available at high purity, with saturated hydrophobic tails, and without acid-labile ethers and esters. We limited the tail lengths to 10 or 18. Lipids with C10 and C18 tail lengths were chosen because they should generally be soluble but still hydrophobic enough to promote bilayer formation. Moreover, tail lengths in this range are frequently employed in experimental protocell models, which enables more straightforward comparisons to earlier studies. We sought to include head groups that could in principle form charge–charge interactions, analogous to what is observed with biological lipids, even under heavily protonating conditions. Here, we chose to work with trimethylamine, sulfate, and phosphonate head groups (Caschera et al., 2011; Liu et al., 2018; Walde et al., 1997). We note that other lipids and combinations could be considered in the future.
Nuclear magnetic resonance
We used NMR to test the resistance of each C10 lipid to attack by sulfuric acid at room temperature. At 100 mM, all the selected lipids are poorly soluble in water and concentrated sulfuric acid. However, at 50 mM, decyltrimethylamine (C13H30N+) is soluble in both water and concentrated sulfuric acid (Fig. 1). We incubated 50 mM decyltrimethylamine with D2O, D2O + 1% H2SO4, or 30% D2O + 70% D2SO4 for at least 1 h before acquiring an 1H spectrum. One percent (v/v) aqueous H2SO4 corresponds to a measured pH ≅ 0.8. We observed no additional peaks or changes to the peaks identified in water under the acidic conditions. Further, the proton ratios remained stable across all conditions. We conclude that decyltrimethylamine is resistant to sulfuric acid in concentrations up to 70%.
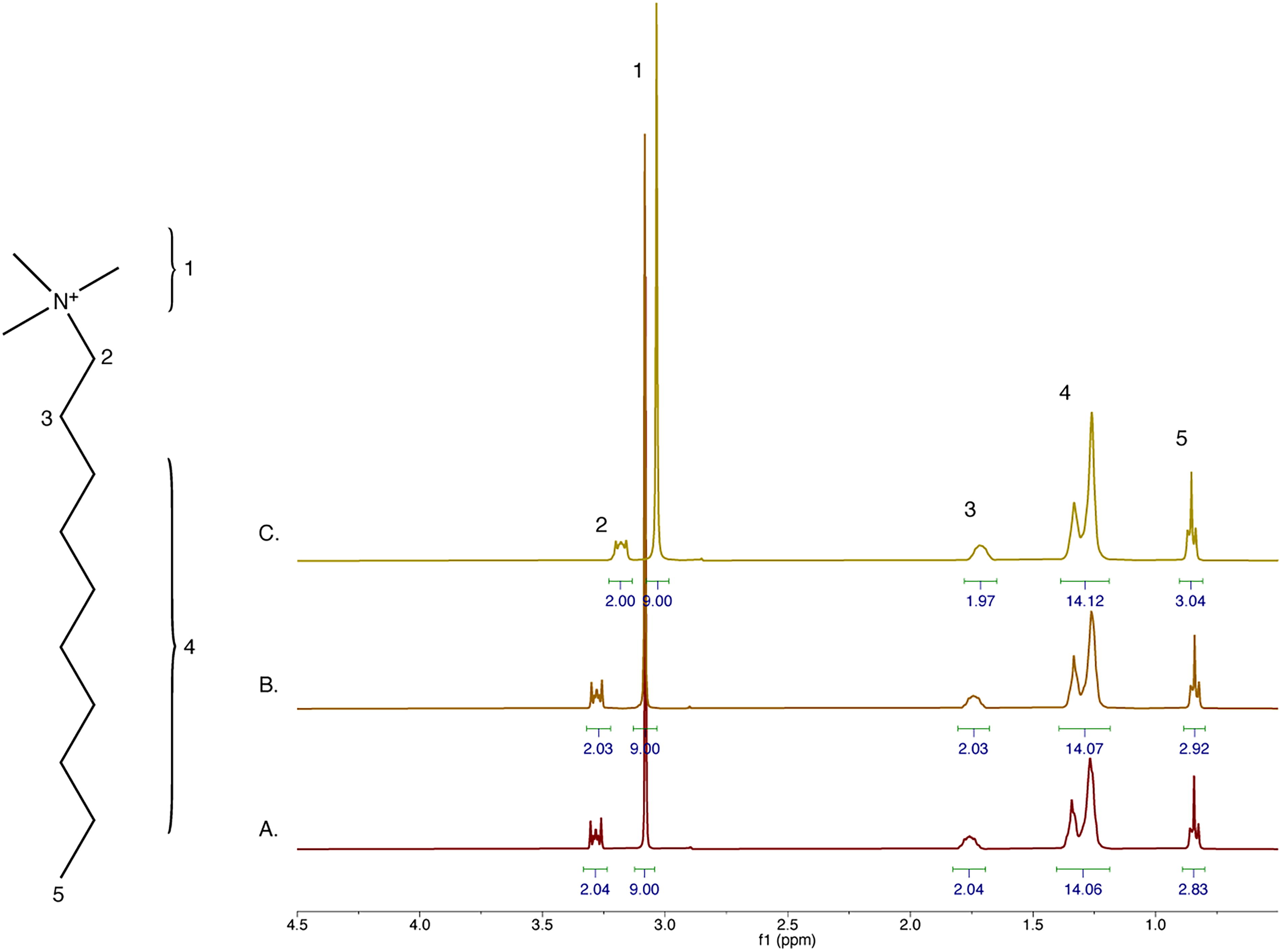
Decyltrimethylamine is resistant to 70% sulfuric acid. 1H NMR spectra of 50 mM decyltrimethylamine in
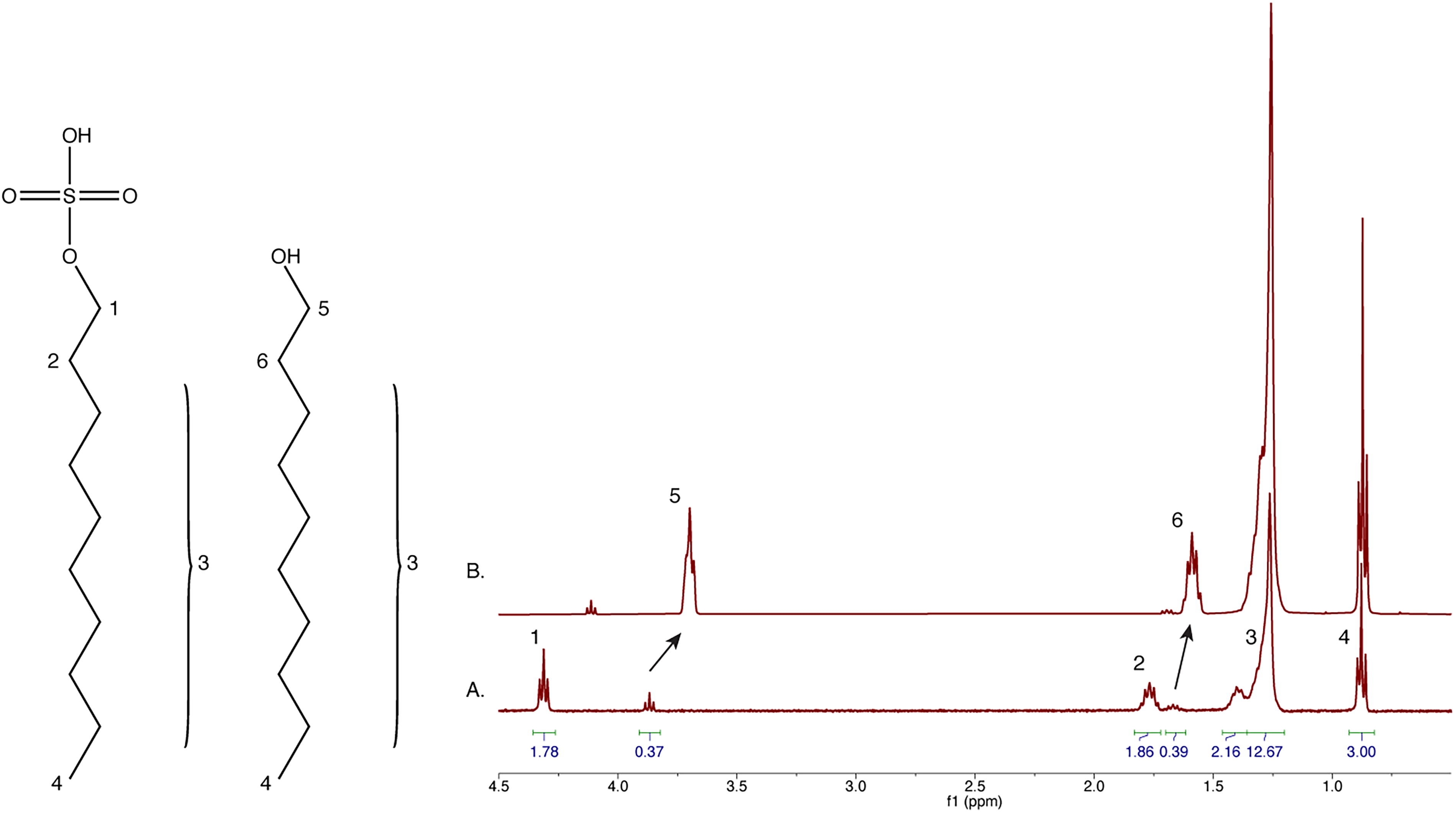
Decylsulfate (C10H22O4S) is resistant to 1% H2SO4 (Fig. 2). However, at 50 mM it is poorly soluble in 70% sulfuric acid and yields low-quality NMR data. Therefore, we incubated decylsulfate in 70% H2SO4 for 1 h and extracted the mixture into CDCl3 for NMR analysis (Fig. 3). Several new peaks appeared after 70% acid treatment (compare Fig. 2A to Fig. 3A) which we hypothesized arise from decyl alcohol. A spike-in of authentic standard confirmed the assignment (Fig. 3B). We conclude that after 1 h in 70% sulfuric acid, approximately 20% of the sulfate headgroups were hydrolyzed to the alcohol form. This was probably due to acid-catalyzed hydrolysis of protonated sulfate. Decyl alcohol can participate in lipid bilayer formation in the context of other lipids.
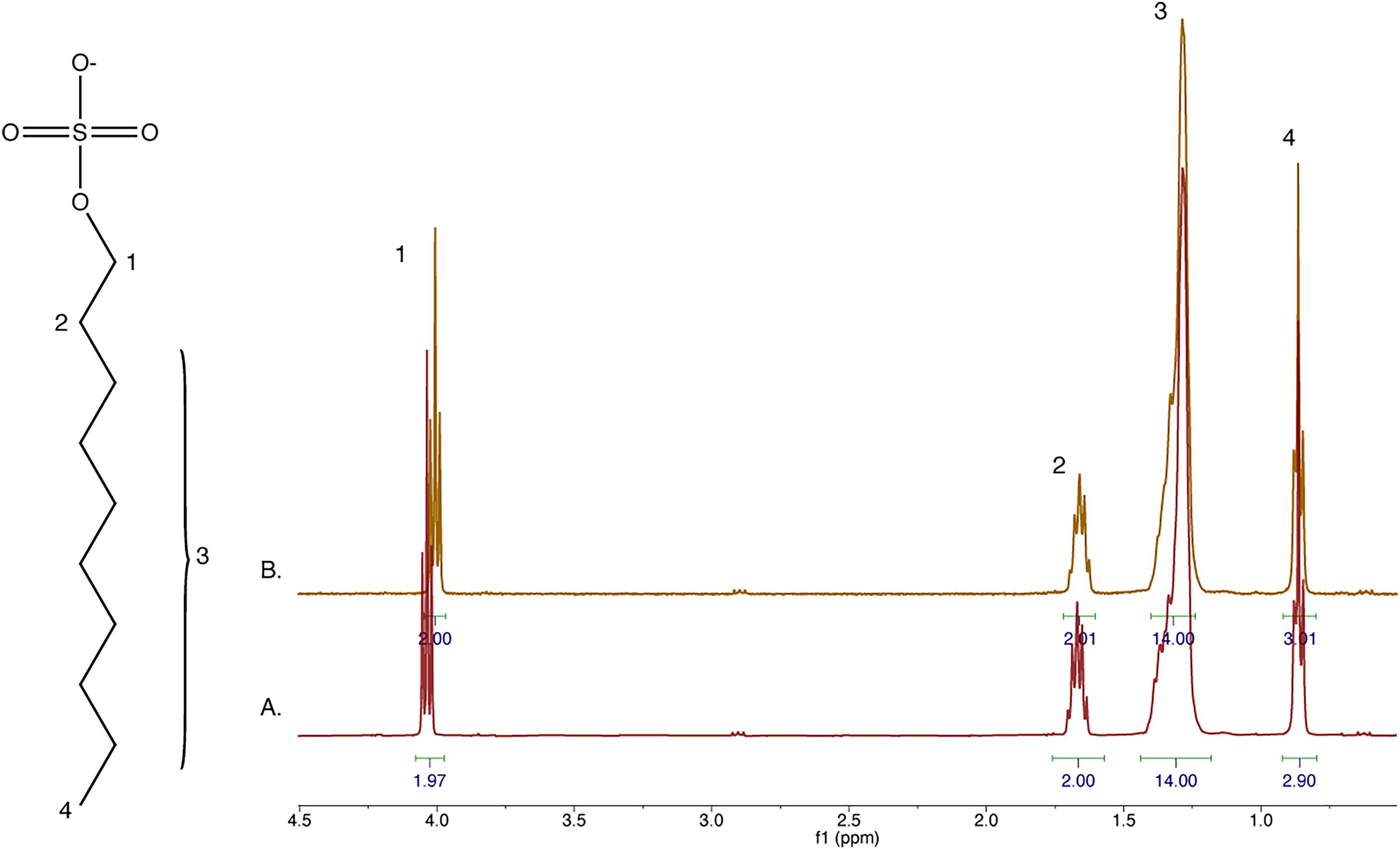
Decylsulfate is resistant to low-concentration sulfuric acid. 1H NMR spectra of 50 mM decylsulfate in
Decylsulfate is largely resistant to 70% sulfuric acid. 1H NMR spectra of
Decylphosphonic acid (C10H23O3P) is too poorly soluble in both water and 70% sulfuric acid to generate quality NMR spectra, so we applied the CDCl3 extraction approach for all conditions (Fig. 4). We found that decylphosphonate is stable in up to 70% sulfuric acid, and we note the presence of fully protonated phosphonate at the highest tested acid concentration (Supplementary Fig. S1).
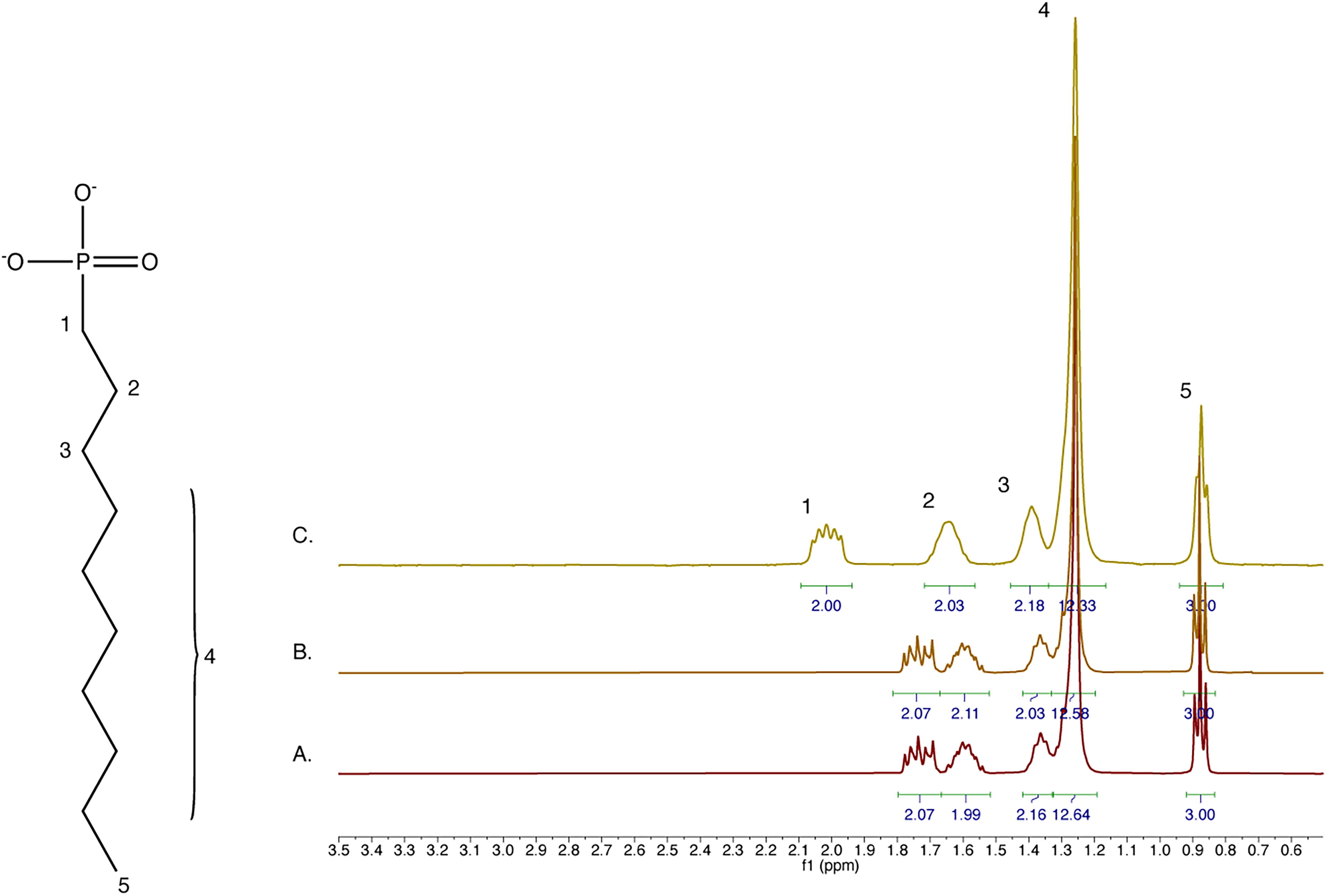
Decylphosphonate is resistant to 70% sulfuric acid. 1H NMR spectra of 50 mM decylphosphonate incubated in
We conclude that sulfuric acid in concentrations up to 70% does not affect decyltrimethylamine, protonates the sulfate head group in decylsulfate, catalyzes the formation of ∼20% decyl alcohol, and protonates the phosphonate of decylphosphonate. With the exception of the ∼20% decyl alcohol, we observed no acid-catalyzed degradation of any bonds in any of these compounds at room temperature over the 1 h duration of the experiments.
To determine if phosphonate, trimethylamine, and sulfate lipids form higher-order structures in concentrated sulfuric acid, DLS was employed to obtain the size distribution of lipid aggregates in solution. Initial measurements of 25 mM decylphosphonate, decyltrimethylamine, decylsulfate, an equimolar mix of all three lipids, and a 50/50 equimolar mix of decylphosphonate and decyltrimethylamine were taken in 70% sulfuric acid 24 h after preparing samples and 5 days after preparing samples. Intensity peaks were observed between 100 and 1000 nm for all four samples, which indicates the presence of higher-order structures larger than micelles, regardless of the identity of the lipid head group (Supplementary Fig. S2). (The sizes of micelles and vesicles are determined by the physical properties of the lipids. Simple single-chain lipids form micelles of ∼5–20 nm and vesicles of ∼100–1000 nm in aqueous solution. The size distributions of micelles and vesicles observed in concentrated sulfuric acid are similar to those observed for fatty acids in water.) In addition, signals were observed above 1000 nm for all samples, which suggests the presence of larger structures. While dust particles or other contaminants could potentially contribute to signals above 1000 nm, microscopy results (see below) showed lipid aggregates at this scale, which suggests that these signals are likely not due to contaminants.
Decylphosphonate exhibited larger aggregate sizes at both days 2 and 5 compared to all other samples except for the mix of all three lipids at day 2, which suggests that the identity of the headgroup has an effect on the biophysical properties of the lipid structures in sulfuric acid solution. The mix of decylsulfate and decyltrimethylamine exhibited the most stable and least variable signal between measurements, and between days 2 and 5, so lipids with these head groups were selected for further analysis.
To better characterize the changes in size distribution over time and at higher concentrations of sulfuric acid, we prepared a 50/50 equimolar mix of 75 mM C10 SDS-DTMA in 70%, 80%, and 90% sulfuric acid. Furthermore, to determine the effect of chain length on the size distribution of these lipids, a 50/50 equimolar mix of 1 mM C18 SOS-TMO was also tested in 70%, 80%, and 90% sulfuric acid. DLS readings were taken a few hours after preparation (day 0) and again after 1, 3, and 7 days. Three independent replicates were prepared and measured for all samples (Fig. 5; Supplementary Fig. S3). Signals between 100 and 1000 nm, which would roughly correspond to the size of lipid vesicles, were recorded at the peak intensity value. SDS-DTMA (and to a lesser extent SOS-TMO) changed color in concentrated sulfuric acid over the course of 7 days. SDS-DTMA in 90% sulfuric acid became increasingly dark and ultimately almost black (Supplementary Fig. S4). Despite this change in color, the average sizes of lipid structures were not significantly different between chain lengths or over the 7-day incubation as determined by a regression analysis of average size over time (p > 0.5). A pair sampled t-test for average size between successive measurements and between measurements at different sulfuric acid concentrations showed no significant difference (pcorrect null > 0.05 for all measurement pairs, with the null hypothesis of 0 difference between means). In addition, with three exceptions (SDS-DTMA in 70% sulfuric acid at days 0 and 3, SOS-TMO in 70% sulfuric acid at day 3; Fig. 5A), the average sizes of SDS-DTMA and SOS-TMO lipid aggregates in 70% or 80% sulfuric acid were not significantly different than the initial 25 mM SDS-DTMA readings (Supplementary Fig. S2E). However, at 90% sulfuric acid, only SDS-DTMA and SOS-TMO at day 0, and SOS-TMO at day 7 exhibited peaks between 100 and 1000 nm on more than one repeat (Supplementary Fig. S5), which suggests that at higher concentrations of sulfuric acid, these structures become more heterogeneous or destabilized. The high viscosity of sulfuric acid may also contribute to the size of vesicle-like structures. Together, the DLS results suggest that the size distribution of lipid structures in concentrated sulfuric acid below 90% is largely unaffected by lipid concentration or chain length and remains stable on the timescale of days.
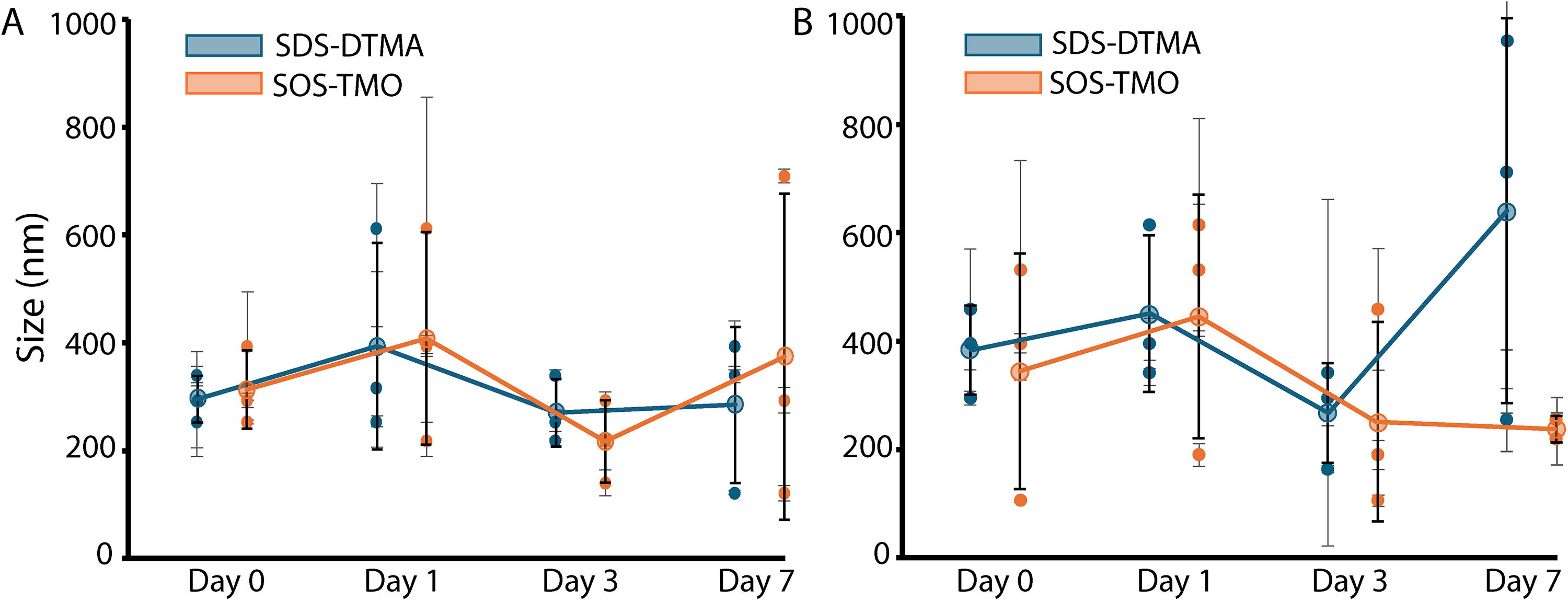
Lipids form stable structures in concentrated sulfuric acid. DLS readings of 75 mM decylsulfate and decyltrimethylamine (SDS-DTMA, blue) and 1 mM octadecyl sulfate and octadecyltrimethylammonium (SOS-TMO, orange) lipids prepared in
Our DLS measurements suggest that structured lipid aggregates can form in concentrated sulfuric acid. To determine the morphology of these structures, we used confocal microscopy to visualize 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY) stained lipids. Samples were prepared identically as in the DLS experiments and placed in the center of an 18-well uncoated, sterilized glass-bottom slide such that samples did not contact the walls of the chamber. We then added 2.5 µM BODIPY to each sample. Surprisingly, the organic BODIPY dye showed robust fluorescence despite the concentrated sulfuric acid solvent. In all samples, vesicle-like structures of various sizes and lipid morphology were observed (Fig. 6), which corresponded to the range of sizes observed in the DLS signals. The presence of these structures is strictly dependent on the addition of lipids; thus, this eliminates the possibility that the observations were an artifact of sulfuric acid interaction with the slide surface. In 70% and 80% sulfuric acid, vesicle-like structures were distributed throughout the sample, while in 90% sulfuric acid, they were segregated to the periphery of the sample. This suggests a possible gradient in sulfuric acid concentration within the sample with vesicles only able to form in a local region of lower acid concentration. This may have been an artifact of the hygroscopic sulfuric acid absorbing moisture from the air. These results suggest that SDS-DTMA lipids can form membranous, vesicle-like structures at sulfuric acid concentrations up to 90%. Additionally, all samples exhibited aggregates of widely varying sizes up to several microns, which agrees with the polydisperse samples suggested by the DLS readings (Fig. 5; Supplementary Fig. S2).
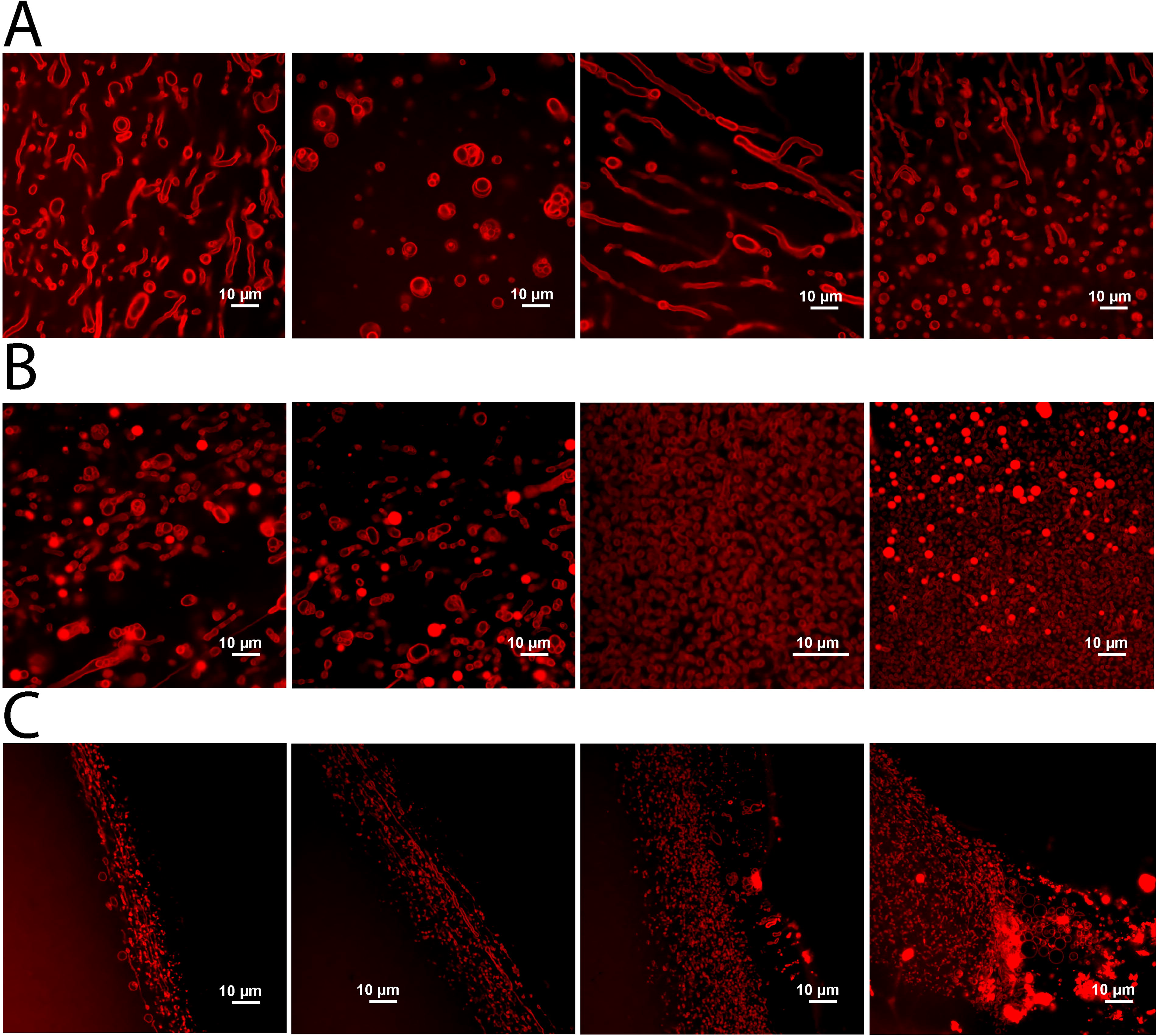
Vesicle-like structures form after the addition of concentrated sulfuric acid to solid lipids. Confocal images of BODIPY-stained SDS-DTMA lipids from samples prepared in
Next, we tested the ability of preformed SDS-DTMA vesicles in water to resist destabilization upon exposure to sulfuric acid. Lipids were prepared in water, and aliquots from this initial sample were added to individual glass-bottom wells. Sulfuric acid was added to each well for a final concentration of 10%, 50%, 70%, 80%, or 90%, and BODIPY was added to a final concentration of 2.5 µM. Images were taken shortly after preparing samples and 3 days after prepaing samples (Fig. 7). In the sample without sulfuric acid, multilamellar structures were abundant (Fig. 7A) and adopted a relatively consistent size, which indicates that the SDS-DTMA mix readily forms vesicle-like structures in an aqueous environment. In the presence of 10% or 50% sulfuric acid, a change in morphology was observed relative to the 0% (water-only) sample (Fig. 7B and C). These samples exhibited a more heterogeneous composition, with the presence of generally thicker membranes and a more irregular spatial distribution of aggregates and nonmembranous droplets. Vesicle-like structures were present in both 10% and 50% sulfuric acid at day 0 and day 3. A further change in morphology was observed in 70% and 80% sulfuric acid (Fig. 7D and E). Mostly aggregates or droplets were observed at day 0, but between day 0 and day 3, these droplets coalesced to form a series of multilamellar structures, which resemble vesicles, with an apparent internal volume. A three-dimensional image of these structures shows that they adopt a distinct ovoid shape (Fig. 7E, final panel). Finally, at 90% sulfuric acid, no membranous structures were observed at day 0 or 3 (Fig. 7F). These results suggest that vesicles formed by SDS-DTMA lipids remain stable when exposed to the addition of sulfuric acid below 90%.
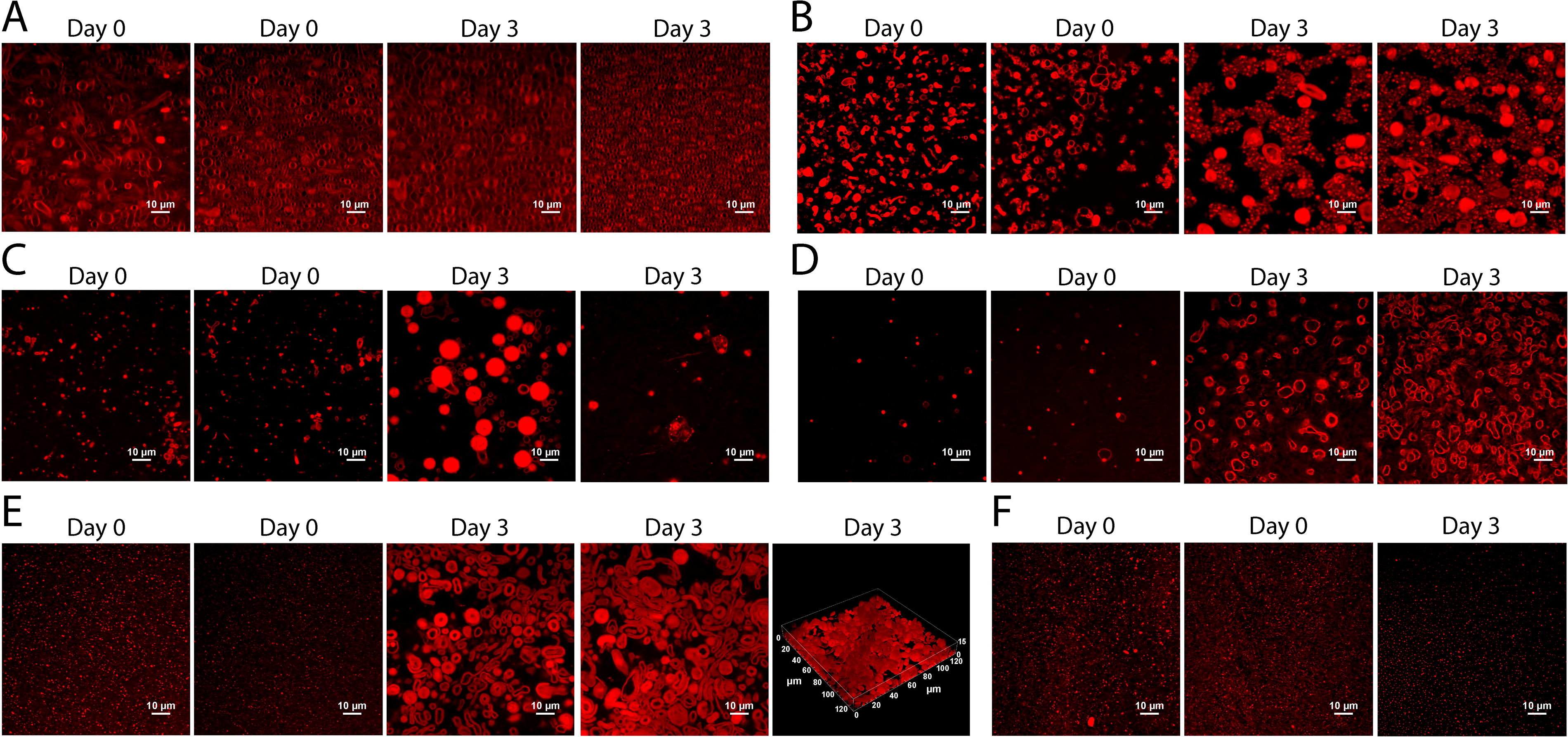
Vesicle-like structures formed in water persist after the addition of sulfuric acid. Confocal images of BODIPY-stained SDS-DTMA lipids at day 0 and day 3 at
In all samples besides those in 0% or 90% sulfuric acid, a pronounced change in membrane morphology was observed between days 0 and 3 (Fig. 7). This change could be the result of a structural rearrangement of lipids within the membrane due to the change in solvent properties, the heat released upon the addition of sulfuric acid, or osmotic shock upon addition of sulfuric acid. To determine if heat can induce a morphology change, 75 mM SDS-DTMA vesicles prepared as above were subjected to a heat shock of 65°C for 1 h. While some change may have occurred (Supplementary Fig. S6A), the size and morphology appeared similar to that without heat shock (Fig. 7A). Additionally, adding water in the same volume and ratio to samples prepared in 70%, 80%, and 90% sulfuric acid did not result in the morphological change observed upon addition of the same volume of sulfuric acid (Fig. 7). These results indicate that the changes observed between days 0 and 3 in the presence of sulfuric acid were likely due to changes in lipid–solvent interactions, not the heat released upon addition of sulfuric acid or osmotic shock.
To ensure the structures observed in Figure 7 were not the result of interaction between sulfuric acid and the walls of the sample well, and to better characterize the temporal determinant of the observed morphology changes, we prepared SDS-DTMA samples in 70%, 80%, and 90% sulfuric acid as above but placed between glass slides such that sulfuric acid only contacted clean glass. Images were taken on days 0, 1, 3, and 7 (Fig. 8A–D). All samples exhibited membranous vesicle-like structures of various sizes and morphologies with internal volumes (Fig. 8E and F). As in Figure 7, a change in morphology was observed after day 0 for all three samples. For all three samples with sulfuric acid, but not in the aqueous sample, a large, dense, and three-dimensional cluster of vesicle-like structures with diverse sizes and lamellarity was observed on day 3 or 7 (Fig. 8B–D, final frame). This clustering of vesicles may indicate a local exclusion of sulfuric acid in the interior of the cluster that has the effect of protecting the vesicles from disruption to their membranes. The mechanism behind the clustering could be positive surface charges bridged by sulfate anions, but further analysis will be required to explore this possibility. Vesicles were present outside of the large cluster structures in all samples, both adhered to the surface and free-floating, which indicates that while clustering among vesicles may provide some degree of protection, vesicles can exist individually in concentrated sulfuric acid. Additionally, elongation of the membrane up to tens of microns in length was seen in all three sulfuric acid samples but not in aqueous solvent (Fig. 8G), often with associated pearling along the elongated segment. This behavior has been observed upon growth and mechanical perturbation of oleic acid vesicles in aqueous solvent and was proposed as a mechanism for vesicle growth and division (Zhu and Szostak, 2009). These results suggest that a similar cycle can be achieved in concentrated sulfuric acid with SDS-DTMA vesicles.
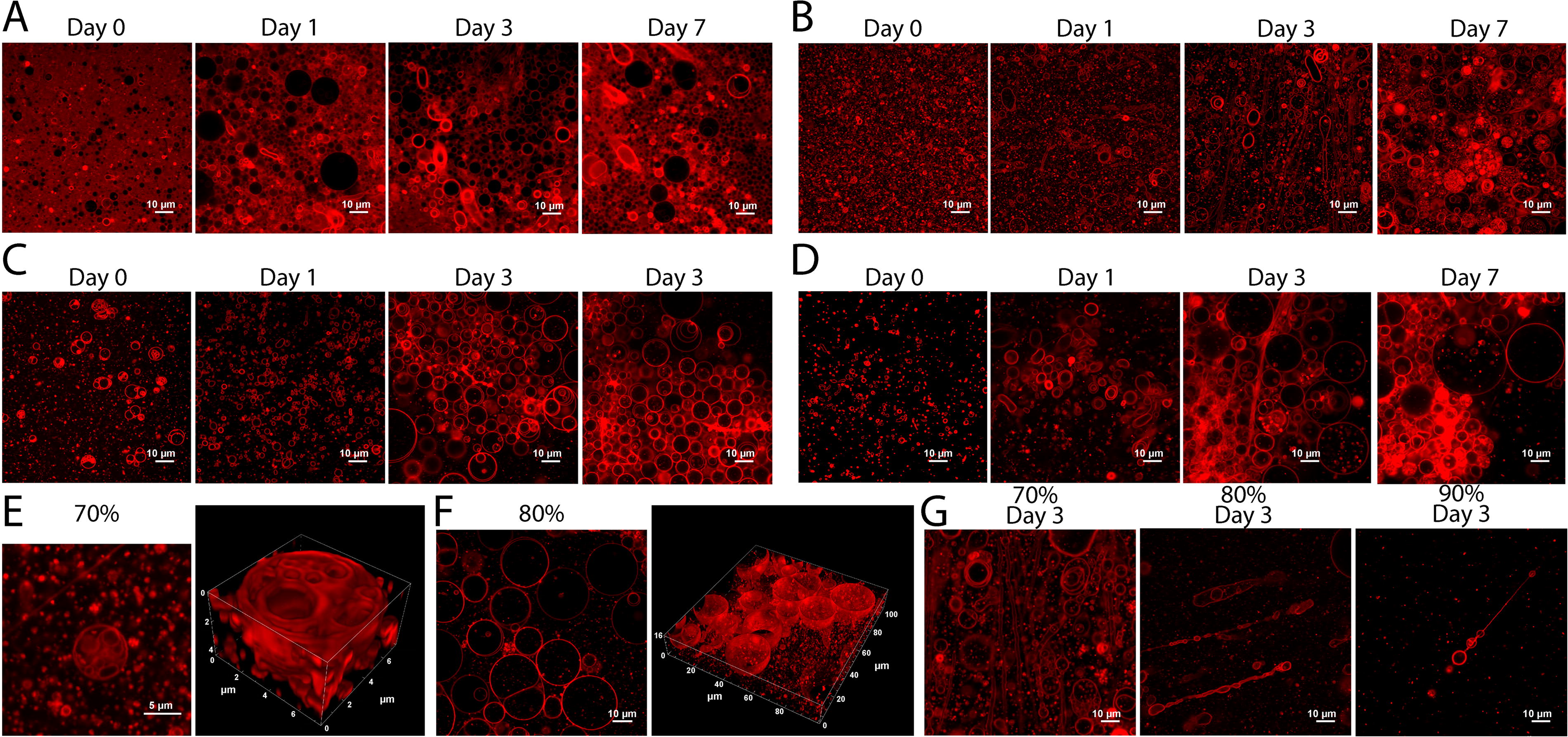
Vesicle-like structures persist in sulfuric acid over the course of 7 days. Confocal images of BODIPY-stained SDS-DTMA lipids at day 0, day 1, day 3, and day 7 at
Results of the confocal imaging suggest that SDS-DTMA membranes remain stable in the presence of sulfuric acid. To determine how the head groups of these lipids interact with each other and with concentrated sulfuric acid to maintain a stable membrane, three independent QM/MM simulations were run for a total of 125 ps per system on a membrane patch composed of DTMA, SDS, or a 50/50 mix of SDS-DTMA lipids for a total of nine simulations (Supplementary Fig. S7). For each system, three lipid head groups and interacting solvent molecules were treated at the QM level. At the end of each simulation, electron density and charge distribution were calculated. As expected, SDS head groups exhibited strong negative charge and DTMA head groups strong positive charge (Fig. 9A–C); this results in a repulsion between head groups and an attraction between oppositely charged head groups. The attractive forces result in a more energetically stable system with SDS-DTMA. Because the bilayer surface is perpendicular to the z-axis, deviation of the plane formed by the three lipid heads from the x–y plane can indicate the propensity for membrane deformations to form, which makes the membrane more susceptible to solvent penetration. The angle between an axis perpendicular to the plane formed by the three lipid heads and the z-axis was tracked during all simulations, and the SDS-DTMA and DTMA systems exhibited a significantly lower angle than SDS-only (Fig. 9D, left panel; p < 0.05). This indicates that the mixed lipid and pure DTMA systems remained most planar.
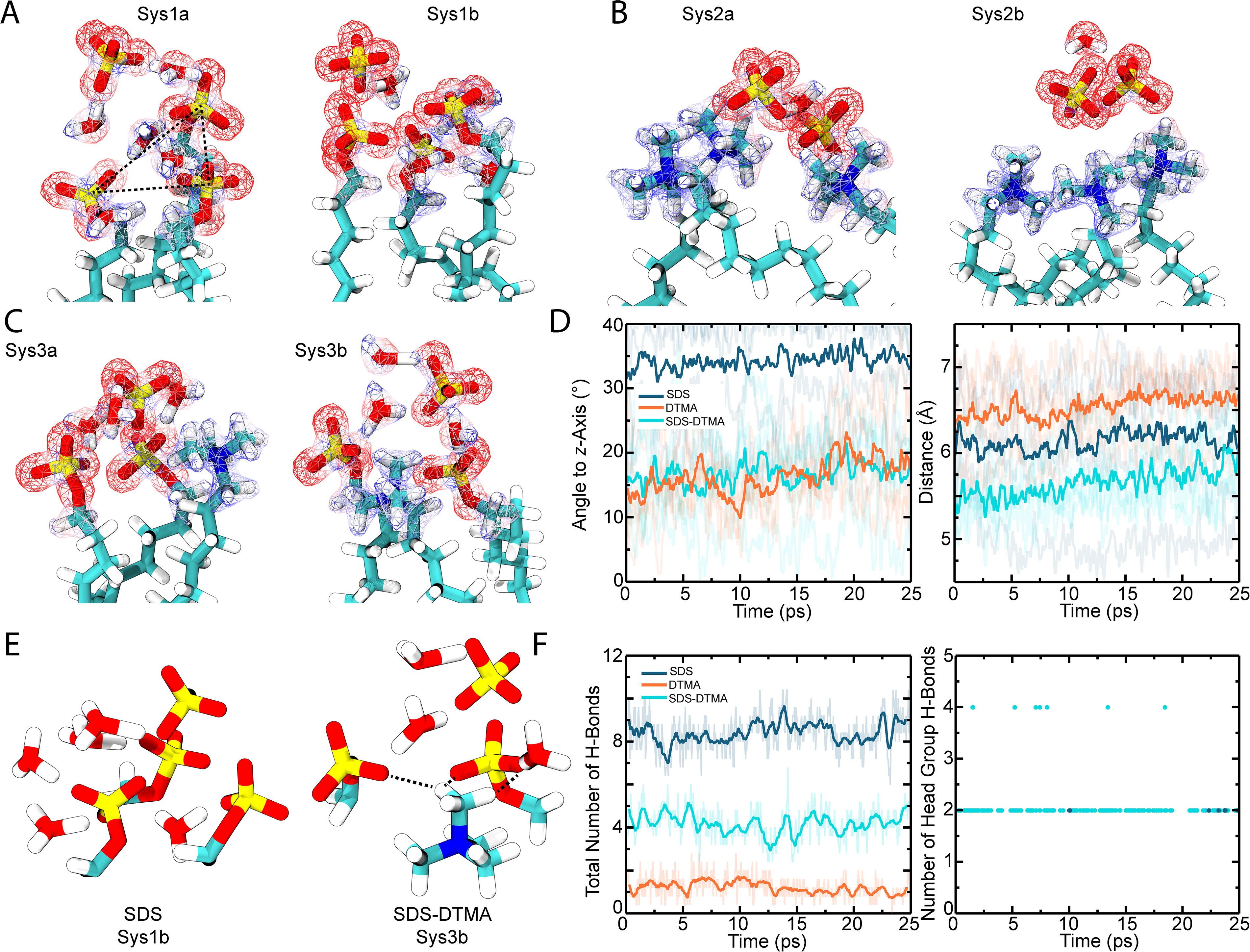
Modeling and simulation of SDS and DTMA membranes. The final frame of the QM/MM simulations shows the electron density for the
Next, average distance between the center of mass of head groups was monitored during simulations. The SDS-DTMA system exhibited a significantly lower average distance between head groups (Fig. 9D, right panel; p < 0.05). These results suggest that, based on the dynamics of the lipid groups and their interactions, the stronger interactions in the mixed SDS-DTMA system make the bilayer more resistant to disruption by sulfuric acid. This disruption likely comes from perforation by sulfuric acid and hydronium ions, which compromises membrane continuity. In addition to interlipid interactions, interactions between lipids and solvent also suggest a mechanism by which a mix of DTMA and SDS lipids are more robust to destabilization by sulfuric acid. In the SDS-only systems, interactions between lipids were not direct but facilitated by hydronium ions (Fig. 9A; Fig. 9E, left). This in turn leads to the penetration of hydronium molecules into the bilayer, which was not observed in the DTMA-only or SDS-DTMA systems (Fig. 9B and C). Furthermore, while the total number of hydrogen bonds was highest in the SDS-only system (Fig. 9E and F, left panel), almost none of the hydrogen bonds in the SDS-only system came from interaction between head groups throughout the simulations (Fig. 9E and F, right panel). In the SDS-DTMA system, there is a robust network of hydrogen bonds between both the solvent and head groups and between head groups themselves, preventing solvent molecules from penetrating beyond the head groups (Fig. 9E). Very few hydrogen bonds, either interlipid or lipid–solvent, were observed in the DTMA-only system (Fig. 9F). Collectively, the simulation results suggest the SDS-DTMA system forms stronger interlipid interactions and a more extensive hydrogen bonding network between lipids and solvent facilitated by a more even charge distribution across lipids, which results in a more stable bilayer.
Stable membranes that can form vesicles are likely to be an essential requirement for cellular life, no matter its chemical makeup. Water is highly favored as a medium for life because, in addition to being an excellent solvent for polar molecules and salts, many molecules are insoluble in water, which allows for the hydrophobic effect that drives the formation and the stability of membranes (Pohorille and Pratt, 2012; Pratt and Pohorille, 1992; Tanford, 1978). However, the ability to facilitate the formation of membrane structures is not a unique property of water. For example, ammonia (Griffin et al., 2015) and hydrogen fluoride (Roth et al., 1995) also have the dense hydrogen bonding networks required to drive a solvophobic effect in the liquid state (reviewed in Bains et al., 2024a). Here, we show the unexpected stability of complex membranous structures in another polar solvent: concentrated sulfuric acid.
Concentrated sulfuric acid as a planetary solvent could be widespread on exoplanets (Ballesteros et al., 2019), either on exo-Venuses or on other rocky planets that are desiccated as a result of the stellar activity of their host star (Ostberg et al., 2023). Concentrated sulfuric acid is also present in our immediate planetary vicinity, as a dominant liquid in the clouds of Venus (Titov et al., 2018), which further emphasizes its importance for planetary science, planetary habitability, and astrobiology.
Our results suggest that the membranous structures observed in concentrated sulfuric acid are apparently resistant to sulfuric acid degradation and in some cases are dynamic. We cannot rule out that membrane dynamics through time could be due in part to some chemical degradation of the lipids. We emphasize that even if in some cases the tested lipids are reactive in concentrated sulfuric acid (Supplementary Fig. S4), this reactivity does not prevent the formation of higher-order structures. It is also possible that membranes may protect otherwise reactive lipids from degradation in concentrated sulfuric acid. Further work will be required to explore this possibility. Some of the tested lipids, for example, decylphosphonate, exhibited larger aggregate sizes compared to other samples, which suggests that the properties of a membrane could be tuned in sulfuric acid by changing the membrane lipid composition. This observation has implications for the potential biological plausibility of such membranes, as it is analogous to the characteristics of biological membranes in water.
Confocal microscopy showed the formation of vesicles and other structures in solution. The microscopy also revealed that, once formed, the lipid membranes adopt a variety of morphologies and can undergo structural changes, such as elongation up to tens of microns in length, often with associated pearling along with the elongated segment. The pearling behavior is especially noteworthy as it could facilitate division of membranous structures. Our results show that such division could in principle happen in concentrated sulfuric acid and that this solvent is compatible with complex and dynamic membranous systems.
Our experimental results are also in agreement with the QM/MM simulations. The simulations showed that in the SDS-DTMA lipid membrane system, the formation of a robust network of charge–charge interactions between both the head groups themselves and the solvent efficiently prevent the sulfuric acid solvent from passing through the SDS-DTMA membrane. This result raises the possibility that the SDS-DTMA membrane (or other similar lipid systems) could be robust enough to retain an aqueous vesicle lumen, or at least a distinct internal solvent environment, while keeping sulfuric acid solvent molecules from penetrating the membrane. Such membrane characteristics could in principle allow for protection and retention of acid-labile components, such as DNA, RNA, or proteins, within the vesicle.
By demonstrating the stability of lipid membranes in this aggressive solvent, we have taken a step forward in exploring the potential habitability of the concentrated sulfuric acid cloud environment on Venus. Future work should consider the experimental verification of the QM/MM simulations, as well as laboratory testing of the leakage of lipid membranes resistant to concentrated sulfuric acid.
Footnotes
Acknowledgments
The authors are grateful to Andy Yu Zhi Li for assistance with confocal imaging. Simulations were performed using HPC resources on the Midway3 supercomputer at the Research Computing Center at the University of Chicago. This article was prepared without the use of artificial intelligence software.
Authors’ Contributions
Conceptualization: D.D., C.N., W.B., J.J.P., and S.S.; methodology: D.D., C.N., and C.K.K.; software: C.N.; formal analysis: D.D. and C.N.; investigation: D.D. and C.N.; writing—original draft preparation: D.D., C.N., and J.J.P.; writing—review and editing: D.D., C.N., J.J.P., S.S., W.B., J.W.S., and C.K.K.; supervision: S.S., D.D., and J.J.P.; and funding acquisition: S.S., J.W.S., and D.D. All authors have read and agreed to the published version of the article.
Data Availability
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
Partial funding for the presented work came from MIT and Breakthrough Initiatives. The writing and editing of the article were supported in part by a Marie Skłodowska-Curie FRIAS COFUND Fellowship (to D.D.) at the Freiburg Institute for Advanced Studies (European Union Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement No. 754340). J.W.S. is an investigator of the Howard Hughes Medical Institute.
Supplementary Material
Supplementary Data
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Associate Editor: Mary Beth Wilhelm
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
