Abstract
Understanding how organics degrade under galactic cosmic rays (GCRs) is critical as we search for traces of ancient life on Mars. Even if the planet harbored life early in its history, its surface rocks have been exposed to ionizing radiation for about four billion years, potentially destroying the vast majority of biosignatures. In this study, we investigated for the first time the impact of simulated GCRs (using gamma rays) on several types of lipid biosignatures (including hopane C30, sterane C27, alkanes, and fatty acids [FAs]) in both the presence and absence of salts (NaCl, KCl, and MgCl2). We measured that the lipids degraded 6–20 times faster than amino acids in similar conditions; moreover, when irradiated in the presence of a salt substrate, degradation was at least 4–6 times faster than without salt, which suggests that salty environments that are often preferred targets for astrobiology warrant caution. We detected radiolytic by-products only for FAs—in the form of alkanes and aldehydes. These results expand our understanding of the degradation of organic molecules in Mars analog environments and underscore the urgent need to direct rover missions to sampling sites protected from GCRs, for example, sites on Mars that have been recently exposed by a wind scarp retreat or meteoritic impact.
Introduction
Mars was, at one time, habitable (Grotzinger et al., 2014). Early in its history, the planet was protected from cosmic radiation by a magnetic field and a dense atmosphere. Around 3.9–4.1 billion years ago, however, the magnetic field dissipated and the atmosphere was progressively lost into space (Acuna et al., 1999; Mittelholz et al., 2020), which turned Mars into the irradiated frozen desert we know today. Two types of ionizing radiation impact the Martian surface: first, solar cosmic rays (SCRs), which are composed of protons and alpha particles, are emitted during solar events, and their abundances and energies vary depending on solar activity (up to several hundreds of MeV/nucleon). Also, galactic cosmic rays (GCRs) from outside the solar system from supernovas, for example, are composed of 85% protons, 14% alpha particles, and ∼1% heavy nuclei (HZE). GCRs have much higher relative energies—in the range of 0.5–1 GeV/nucleon (Dartnell et al., 2007). Because of this high energy, models suggest that GCRs can penetrate up to several meters into the martian soil (Pavlov et al., 2012). When GCRs reach Mars, they react with the soil (as well as the martian atmosphere) and degrade into showers of secondary radiation (protons, neutrons, gamma rays, and electrons, which degrade further into muons, gamma rays, and electrons; Dartnell et al., 2007).
Two current NASA rovers, Curiosity and Perseverance, and the future ESA rover Rosalind Franklin (currently planned for launch in 2028) are, or soon will be, searching for clues of habitability and signs of ancient life on the planet’s surface. In concert, there is the Mars Sample Return effort, collecting samples with the Perseverance rover for return to Earth for analyses using state-of-the-art instrumentation. The NASA rovers drill to 5 cm deep (Moeller et al., 2021; Okon, 2010), which is deep enough to be shielded from SCRs but not from heavily irradiation by way of GCRs. The ESA rover is designed to drill to much greater depths (up to 2 m deep; Vago et al., 2015), where the samples will receive significantly less radiation (Pavlov et al., 2022). Yet it remains uncertain as to whether that 2 m will be deep enough for optimal preservation of organics. For this reason, it is critical to understand the impact of radiation on biosignatures in Mars analog environments, including the rate of biosignature degradation as well as potential by-products we could search for on Mars.
At present, most studies on the impact of GCRs on biosignatures have focused on amino acids (Gerakines and Hudson, 2013; Kminek and Bada, 2006; Pavlov et al., 2022; Pavlov et al., 2016; Pavlov et al., 2012), kerogen (Eigenbrode et al., 2015; Fox et al., 2023; Fox et al., 2019), pigments (Baqué et al., 2020; Baqué et al., 2018),
The conditions of irradiation have an enormous impact on the radiolytic rate. For example, radiolytic decay in cold conditions (e.g., −50°C) is slower than at room temperature (Pavlov et al., 2022). Mineral matrices have also been shown to have an impact on the radiolytic rate. Several studies using perchlorate salts under simulated GCRs (Eigenbrode et al., 2015; Pavlov et al., 2022) have shown a faster degradation of amino acids in contact with these salts under ionizing radiation. In the present study, we considered four different geochemical matrices: no-salt, NaCl, KCl, and MgCl2. Chloride deposits have been detected on Mars orbitally (Osterloo et al., 2008); halite has been found in martian meteorites (Bridges and Grady, 2000), and halite has been observed in situ at Gale crater (Rampe et al., 2020). Yet these salts have never been investigated for biosignature preservation under gamma radiation. These three salts are often associated with environments either favorable for life, such as salty brines that allow liquid water at low temperatures, or are preferential for biosignature preservation, such as salt deposits that trap organics (McMahon et al., 2018; Schreder-Gomes et al., 2022). We compared the loss of organics in said different matrices after exposing our samples at room temperature to three doses of gamma radiation—0.2 MGy, 0.5 MGy, and 0.9 MGy (Megagray).
Materials and Methods
Organics standards and salts
In the present study, we focused on four synthetic standards as follows: a C30 Hopane [17b(H), 21a(H)-Hopane] from Chiron (Trondheim, Norway), a deuterated C27 Sterane (5a-cholestane-2,2,4,4-d4, a standard used routinely in our laboratory) from Sigma (Burlington, MA), a mix of linear alkanes (even C number, C10 to C40) from Restek (Bellefonte, PA), and a mix of FAs (even C number, C10 to C24) that we prepared from individual FA solutions and powders from Sigma (all structures in Fig. 1).
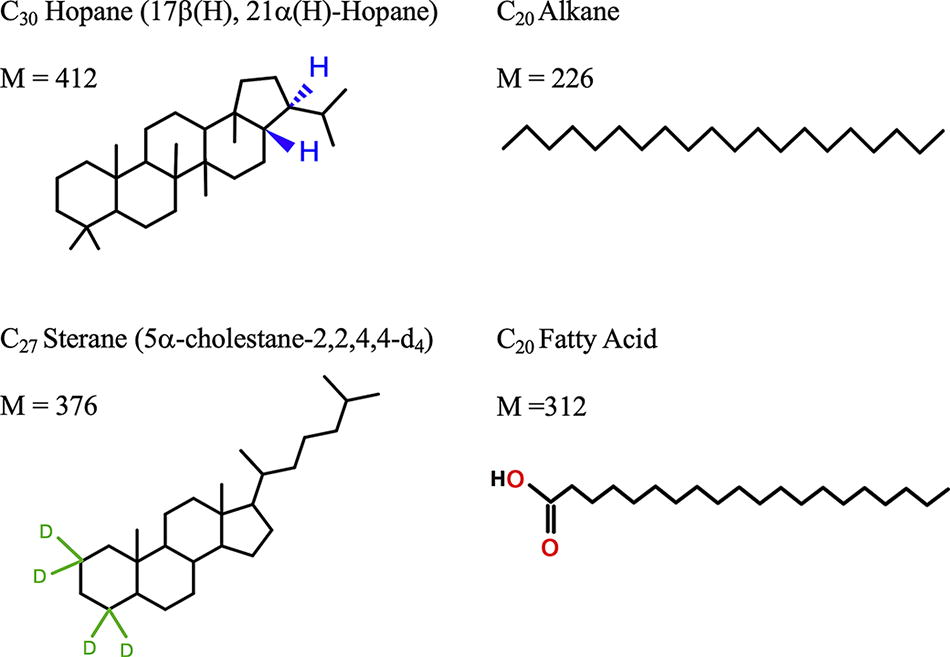
Chemical structures of the studied compounds and their molecular weights (M, in g/mol). A C
We selected three synthetic salts to use as matrices: NaCl at 99.0% purity from Fisher Scientific (Waltham, MA), KCl at 99.7% purity from Baker Analyzed Reagent (Phillipsburg, NJ), and MgCl2 anhydrous at 98% purity from Sigma.
Sample preparation is illustrated in Fig. 4. We spiked 2 µg of hopane, 2 µg of sterane, and 2 µg of each alkane in the alkanes mix. For the FA mix, we injected 50 µg of each FA. We injected this excess amount of FA mix to compensate for (1) the loss due to room temperature exposure (quantified in Supplementary Fig. S7) and (2) the incomplete quantification of FA because samples were not derivatized. The organics were dissolved in 40 µL of hexanes and injected in a small glass insert (100 µL volume) for the standards irradiated without matrix. For the standards irradiated in salts, we prepared 13 mm (outer diameter) borosilicate glass tubes with 1 g of salt, injected at the top of the 40 µL of hexanes containing the standards, and mixed the organics with the salt by turning the tube for homogenization. All tubes were then air-dried overnight in a fume hood and kept in a minus 20°C freezer during the rest of sample preparation. We then added quartz wool to block the salt or glass insert at the bottom of the tube and prevent sample loss during flame sealing under vacuum.
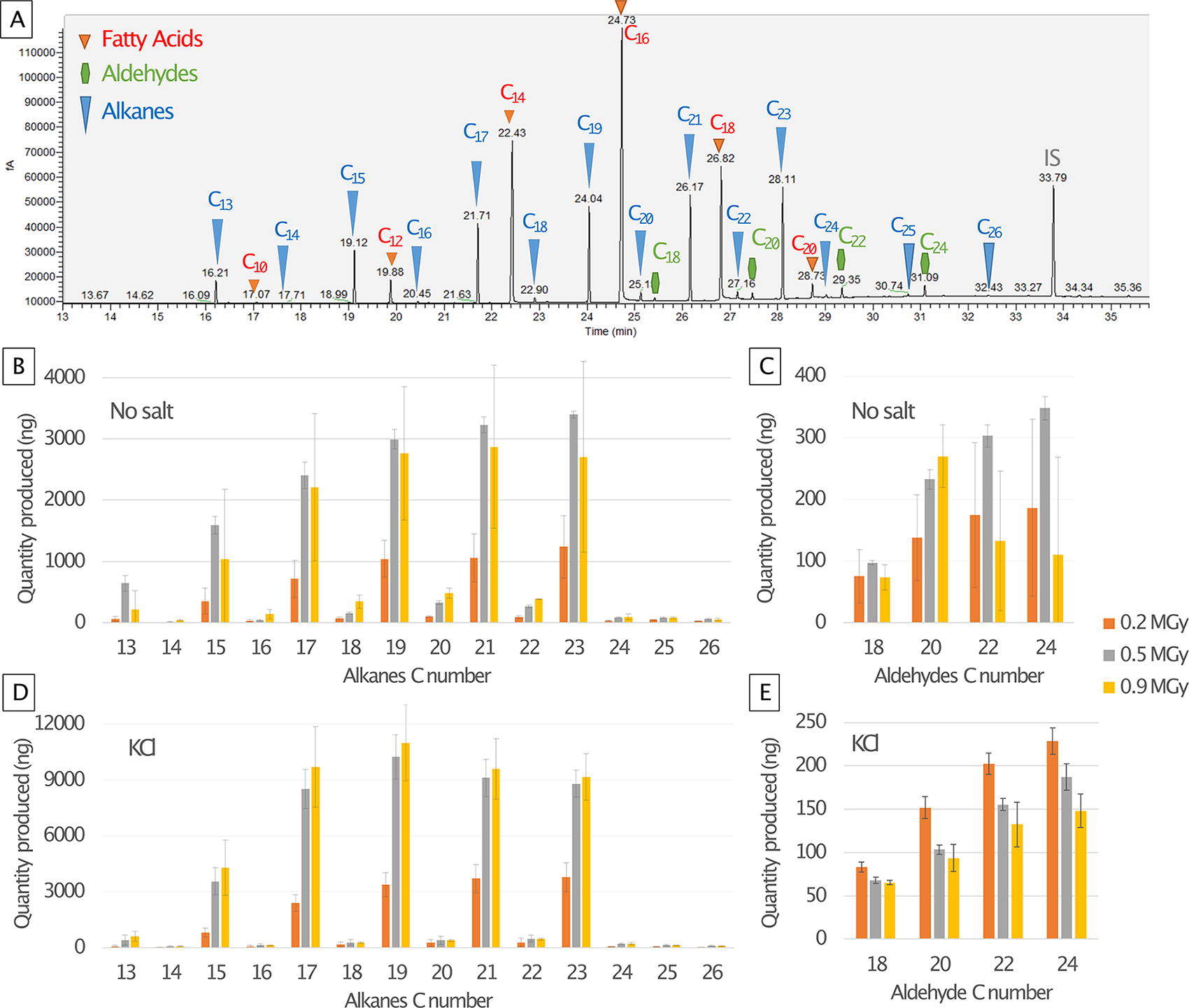
Identification and quantification of FA mix by-products. Upper panel:
For each combination of salt and organics, we prepared three replicates to take into account variation due to preparation of the standards, solvent recovery, and analytical error (Supplementary Table S1). We also analyzed one replicate of each salt alone to ensure absence of contamination (we did not detect any signs of contamination). We also tested for potential contamination due to sealing and radiation with an irradiated sand blank (again, we did not detect any signs of contamination).
The samples were irradiated with 1 MeV gamma radiation from a 60Co source at the Radiation Effects Facility at NASA Goddard Space Flight Center (GSFC). Three doses were achieved at a rate of 37.4 Gy/min—0.2 MGy, 0.5 MGy, and 0.9 MGy—with a maximum variation of 10% of the total accumulated dose due to the experimental setup. The tubes were dynamically pumped until ∼30 mTorr and flame sealed to remove the atmosphere and avoid extra organic degradation due to radicals formed by the water and air in the atmosphere under ionizing radiation (Dartnell, 2011; Pavlov et al., 2022). The samples were kept at room temperature during radiation, and we ensured that the ambient temperature never rose above 30°C. To guarantee a laterally uniform radiation dose across the samples, the tubes were rotated several times over the duration of radiation.
All standards were stable at room temperature except for the FA mix. Prior to the irradiation, we quantified the loss of FAs over 4.5 weeks at room temperature (one quantification per week, see details in Supplementary Fig. S1). We calculated significant concentration decreases after 37 days at room temperature for most FAs, so we prepared radiation tubes with an excess of FA to ensure there would still be enough material for quantification after a decrease in concentration due both to radiation and several weeks at room temperature. To be able to compare our controls to irradiated samples and measure the effects of radiation only, we kept all controls at room temperature during the entire time of sealing and radiation (38 days). As soon as the radiation was over, we transferred all samples into a minus 20°C freezer until the organics were recovered and analyzed.
Organics recovery from glass cones and salts
A schematic of the organics recovery pathway is presented in Fig. 2. All the surfaces (e.g., aluminum foil), containers (e.g., glassware), and tools (e.g., metal tweezers) used at all preparation steps were cleaned of organics by baking them at 500°C overnight. After radiation, we cleaned the outside of the irradiated tubes sequentially with methanol (MeOH) and dichloromethane (DCM) and then cracked them open with a gentle hit using a rock hammer wrapped in ashed foil. This prevented any external contaminant from entering our samples during the opening of the glass tubes. We removed the glass wool using tweezers and transferred either the glass inserts into a 2 mL vial or the salts into a 40 mL vial. In the case of salt samples, we rinsed the walls of the broken irradiated bottom of the tube with ∼100 µL hexanes in case any standard stuck to the glass during homogenization of salt.
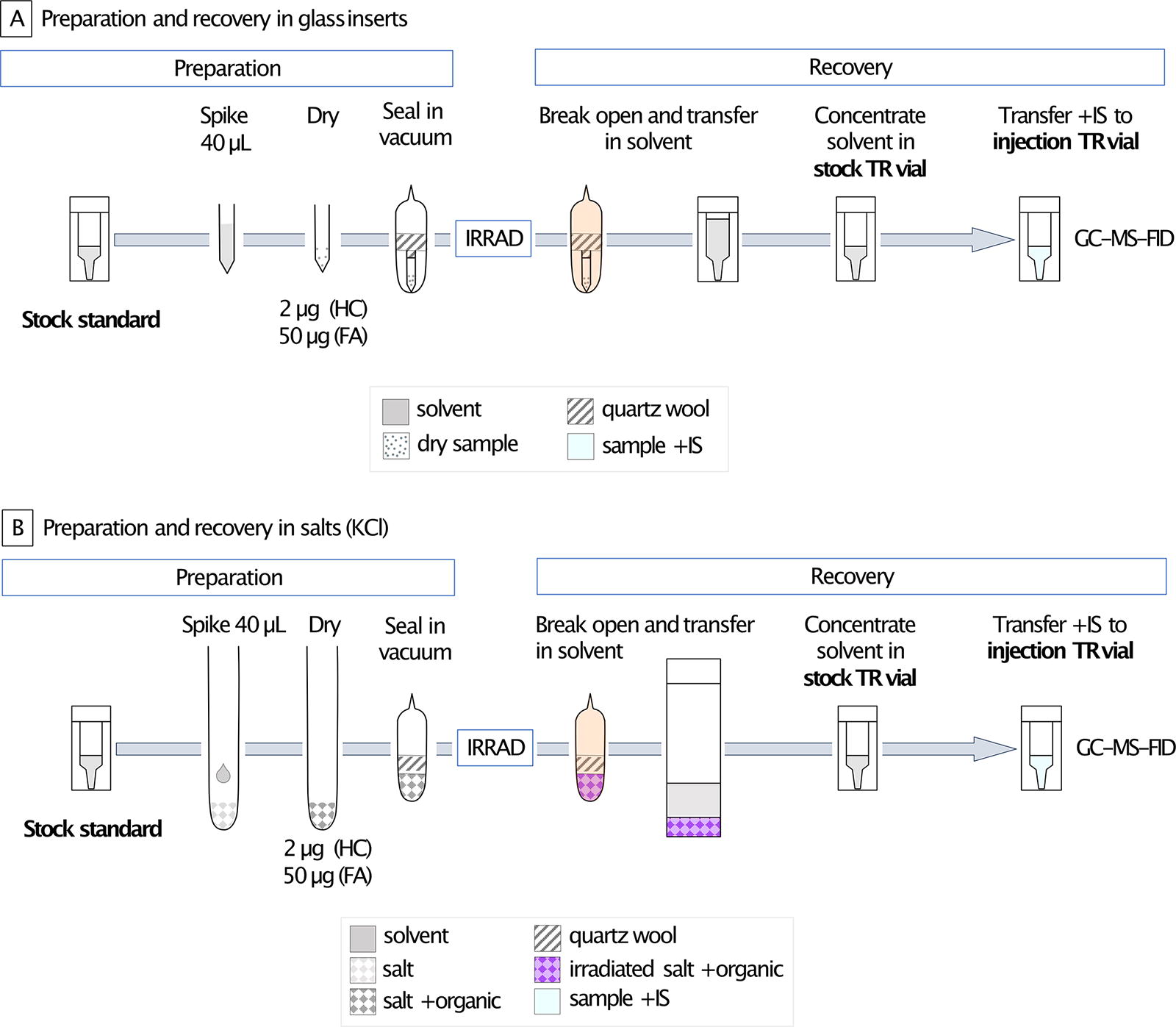
Samples preparation before irradiation and organics recovery after. Flowchart for samples in glass insert
The following protocols for the recovery of organics on glass and slats were optimized, and the optimization results are reported in Supplementary Figure S8. Alkanes were more efficiently recovered from a glass insert with hexanes compared with DCM:MeOH and with high repeatability (Supplementary Tables S2 and S3, respectively). The FA mix was detectable on our instrument without derivatization (Supplementary Fig. S2). No salt was dissolved in hexanes, and no damage to the gas chromatography (GC) column was observed (Supplementary Fig. S3). Alkanes and FA recoveries were tested in all salts (detection in Supplementary Fig. S4). Quantification of products is reported in Supplementary Tables S4 and S5. To quantify the hopanes, alkanes, and FAs, we used the C27 sterane as an injection standard. To quantify the steranes, we used the hopane C30 as an injection standard. As all irradiated data were normalized with the data from their corresponding control, different efficiency in organics recovery did not impact our results and allowed us to compare radiation damages between samples.
To recover the standards on glass (Fig. 2A), we washed the glass inserts with ∼400 µL hexanes that we transferred into a total recovery (TR) 1 mL vial. After completely drying this volume under ultra-high purity (UHP) N2 flow, we added 100 µL of hexanes (500 µL for the FAs) to make up our stock solution. We then transferred 50 µL of the stock solution (100 µL for the FAs) into a TR injection vial, and then the injection standard was added for quantification. The final volume of the TR injection vial was 100 µL, and the injection standard concentration was 10 ng/µL for all samples. The control concentration of hydrocarbon standards (alkanes mix, hopane, and sterane) in the injection TR vial was 10 ng/µL and 100 ng/µL for the FA mix. The excess concentration of FAs was added to compensate for the loss of molecules due to long exposure to room temperature to ensure detection.
To recover the standards from the salts (Fig. 2B), we first transferred the salts out of the broken tubes into 40 mL vials, and then we rinsed with ∼2 mL of hexanes the walls of the bottom of the tube. We added ∼10 mL hexanes to the salt, and the mix was vortexed. We then transferred the solvent to another 40 mL vial, dried it under UHP N2 flow, and transferred the organics with hexanes into a 1 mL TR vial with a final volume of 100 µL (for all standards). A total of 50 µL was transferred into an injection TR vial where the injection standard was added. The final volume of the injection TR vial was 50 uL and that of the injection standard concentration was 10 ng/uL. The control standard concentration was 20 ng/µL for hydrocarbons and 500 ng/µL for FAs. We increased our standard concentrations in the injection TR vials compared with the glass standards because we expected a lower recovery of organics on the salts compared with the glass inserts. Derivatization is a chemical preparation for polar organics, which makes them more volatile and more likely to transition into the gas phase and travel along the apolar GC column (Shantha and Napolitano, 1992). In this study, we did not derivatize the FA mix before analyses so did not detect the total amount of FAs present. However, because we always detected the same proportions of FAs, we could still compare the results of the controls with those of the irradiated samples. To compensate for this lower detection, we increased the concentration of FAs in the injection TR vials.
All samples were injected with a Thermo AI 1310 Auto-Sampler in a split/splitless injector at 300°C through a splitless glass liner. All injections were made in splitless mode with a split duration of 1 min and a He carrier flow of 1.2 mL/min, leading to a 60 m Rxi-5ms capillary column (0.25 mm ID, 0.25 µm). The Thermo trace 1310 GC oven temperature ramped from 60°C to 320°C at different rates and with a different final temperature hold. (1) For the hopane and the sterane standards, the ramp was 30°C/min and the final temperature was held for 15 min. (2) For the alkanes mix, the ramp was 9°C/min, and the final temperature was held for 20 min. (3) For the FAs mix, the ramp was 9°C/min as well, and the final temperature was held for 10 min only. For identification and quantification of the compounds, the column was then split into two disactivated shorter columns: a 150 µm × 0.363 mm × 240 cm column leading to the Thermo ISQ mass spectrometer (MS) and a 170 µm × 0.363 mm × 60 cm one to the Thermo flame ionization detector (FID). The MS source and transfer line temperature were set at 250°C and 300°C, respectively, and the electron energy was 70 eV. The FID detector temperature was 330°C, and the air and N2 and H2 flows were set at 350, 40, and 35 mL/min, respectively.
As mentioned above, each sample was analyzed in triplicate. To determine whether there was a significant decrease of biosignature between two doses of irradiation, we performed a Student’s t test (after confirming normally and equality of variances). When the p value was below 0.05, we concluded that the biosignature decrease was statistically significant.
When applicable, we calculated the radiolitic constant k to compare radiolysis of organics in different samples. The amount of a molecule being destroyed by radiation follows an exponential decay, described by the formula:
Results
Degradation of organics in different matrices
Organic molecule identification
All compounds were identified by using their fragmentation patterns and mass spectra, as shown in Supplementary Fig. S5, and all chromatographs for the samples irradiated without salts appear in Supplementary Fig. S6. Both for controls and experimental runs, we detected the alkanes C18 to C38 (in the alkane mix of C10 to C40) and the FA C10 to C18 (in the FA mix of FA C10 to C24).
The degradation of our organics exposed to radiation in different matrices is presented in Fig. 3, where the data are normalized over the mean of the controls (listed in Supplementary Table S6). All k values calculated are listed in Supplementary Table S7.
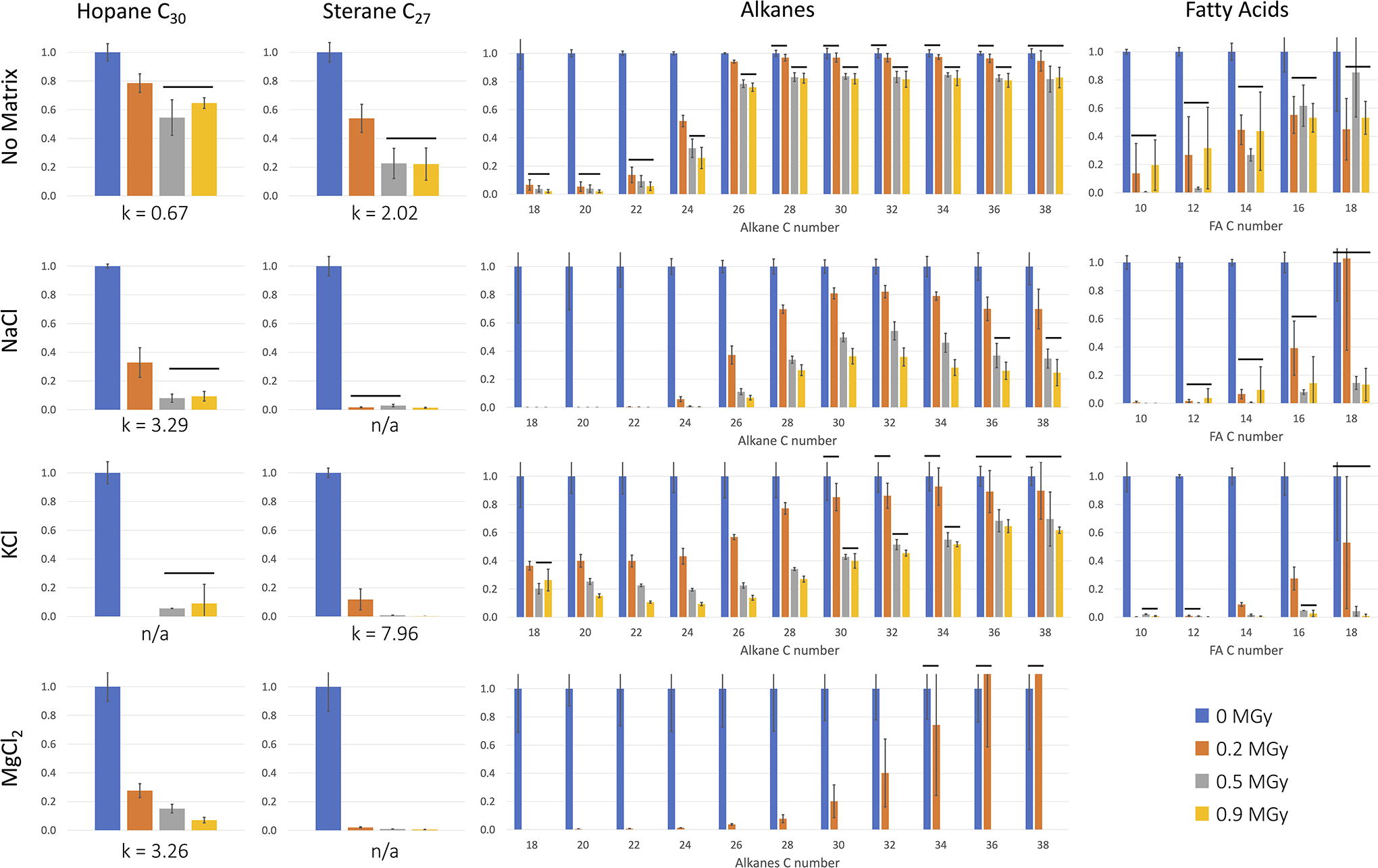
Histograms of different types of organics decreasing in several matrices (no-salt, NaCl, KCl, and MgCl2). The four organics studied were a C30 hopane, a C27 sterane, an alkanes mix (C10–C40), and a fatty acid mix (C10–C24). The data are normalized over the mean of the controls, and the error bars represent standard deviation between triplicates. The horizontal black lines indicate no significant differences between doses (p value >0.05). The radiolytic constant k (MGy-1) is listed for the hopane and sterane, and n/a means that we were unable to calculate k (See Supplementary Table S7). For all plots, the y axis is the survived fraction. For the FA and alkanes mixes, the x axis represents the C number of each molecule. There are no data for the FA mix in MgCl2, where the control was not recovered. There are no data for hopane in KCl at 0.2 MGy.
In the samples that were irradiated without salts, we measured significant degradation of all organics with increasing radiation dose (p-value <0.05).
The hopane and sterane decreased significantly after only 0.2 MGy of gamma radiation, and these compounds stayed stable between 0.5 MGy and 0.9 MGy.
In the alkanes mix, we notice that the shorter C chains degraded much more rapidly: C18 to C22 decreased by 90% after only 0.2 MGy, C24 decreased by 75% after the 0.9 MGy dose, and all longer alkanes decreased at most by 20%.
All FA in the mix significantly decreased between the control and irradiated samples (up to 90% loss), though all three doses presented no significant differences. Again, we observed that the long chain FAs were more stable (at 0.9 MGy 50% loss of C38, while 80% loss C18).
Organics in salt matrices
The presence of salt matrices during irradiation dramatically increased the radiolysis rate for all organics.
The hopanes and steranes in salts decreased about four to six times faster than without salt, with radiolytic constant k = 7.9 for sterane in KCl versus 2.0 without salt, and k = 3.2 for the hopane in both NaCl and MgCl2 versus 0.6 without salts. For hopanes and steranes, degradation rates were generally comparable among the three salts, and we noted that the hopane was more stable than the sterane (overall lower k values).
Alkanes degraded faster in NaCl than KCl (Supplementary Table S7), though we could not calculate enough k values for FA in KCl to determine whether it was at a faster or slower rate than in NaCl.
We could not recover any FA from the MgCl2 salt and only recovered a small amount of alkanes close to our limit of detection, which we hypothesize is because the salt is hygroscopic. The alkanes decreased about twice as fast in KCl compared with the samples without salts and twice more in the NaCl matrix (complete loss of C18 to C22 and C38 decreased by 80% at 0.9 MGy compared with 40% in KCl). The trend was opposite for the FA mix, where the degradation was faster in KCl: for example, the C18 FA decreased by 50% without salt, 90% in NaCl, and 99% in KCl. We note again that, both for alkanes and FA, the larger C chains were more stable under radiation in all salts.
Production of FA by-products under irradiation
We did not observe any degradation products from the hopane, sterane, or alkanes mix with our analytic setup and parameters (solvent extraction, GC-MS-FID), but in all the FA mix samples, we detected newly formed alkanes and aldehydes. These are identified in Fig. 4A using their mass spectra listed in Supplementary Fig. S5. The initial FA mix was composed of eight straight FA chains from C10 to C24 (only even C numbers). The 14 alkanes formed were C13 to C26, with a clear preference for odd C numbers, and the four aldehydes formed were C18 to C24 (only even C numbers).
We identified the same FA mix by-products in all matrices (no-salt and KCl results in Fig. 4), and all quantities are listed in Supplementary Table S8. Both with and without salts, the alkane quantities increased with the radiation dose (Fig. 4B and D), up to 3 µg produced without salts, 8 µg in NaCl, and 10 µg in KCl. The maximum amount of aldehydes synthesized was 200–300 ng without salts, 100–150 ng in NaCl, and 100–200 ng in KCl. The aldehydes increased with the dose in the samples without salt (Fig. 4C), but for NaCl and KCl, the trend was the opposite: after a large production of aldehyde at 0.2 MGy, the amount decreased at the two higher doses (Fig. 4E).
The alkane and aldehyde by-products are unlikely to be the result of contamination, as they were only detected in the irradiated FA samples (pure and in salt matrices), and their abundances were influenced by radiation dose (Fig. 4).
Color change of irradiated salts
All salts changed color after exposure to gamma radiation (Fig. 5), increasing in intensity with the dose. The NaCl turned to a bright orange, the KCl to a deep purple, and the MgCl2 to a light beige (see mechanisms in Section 4.5).

Salt color change after irradiation.
Degradation of organics without salts
In the samples without salts, the organics degraded at a much faster rate than amino acids in similar conditions (no matrix, room temperature, and gamma radiation). Experimental radiolitic constants k for small amino acids (glycine and alanine) are around 0.1 MGy−1 (Kminek and Bada, 2006; Pavlov et al., 2022). In this study, the hopane C30 degraded six times faster and the C27 sterane more than 20 times faster (Fig. 3). This faster degradation of hopane and sterane is likely due to their larger cross sections (masses about four times the masses of amino acids), which makes them more likely to break down under radiation (Kminek and Bada, 2006). Amino acids are not only smaller than our targeted molecules, but they are also more reactive and could potentially form hydrogen bonds with the surrounding materials. Here, we compare our radiolytic constants with previous results for amino acids not as a precise comparison but to shed light on the broad difference magnitude in radiolytic constants between different organics. In natural samples, additional interactions between minerals and organics may also have an impact on the rates of organic degradation under radiation (Baqué et al., 2018; Ertem et al., 2021; Roussel et al., 2024).
The sterane had a degradation rate significantly higher than the hopane (Fig. 3, Supplementary Table S6). We are uncertain about the exact mechanism behind this difference. The slower degradation of hopane is consistent with the extra ring in its structure, which makes it more stable under radiation (Ivanov, 1992), but further research is needed to explain this difference of rate. We observed no significant destruction of either the hopane or the sterane between 0.5 MGy and 0.9 MGy, potentially suggesting that a back reaction could reform the initial molecule after initial breakage.
For the alkanes and FA mixes, we did not detect some of the smaller and larger ones (alkanes C10 to C18, plus the longer C40, and FA C18 to C24). Our hypothesis is that we missed the largest compounds because of their difficulty to be volatilized and move into the gas phase, and the small alkanes were likely not detectable due to our GC-MS-FID method and setup (Dettmer-Wilde and Engewald, 2014).
The precise k value for most of the FA and alkanes irradiated without salts could not be calculated because their degradation was either too fast (short alkanes), or there was not enough statistical differences between the three doses (long alkanes), or no differences among the three doses (FA).
Overall, hydrocarbon biosignatures are generally considered the most resilient in time, especially hopanes and steranes (Brocks and Summons, 2003), yet here we show that as individual isolated molecules, they are more fragile than amino acids under radiation.
Effect of salts on radiolysis
The addition of any of the three salts (NaCl, KCl, and MgCl2) dramatically increased the degradation rate of all of the organic molecules studied. Almost all hopanes and steranes were lost after the first dose of 0.2 MGy, which corresponds to an order of magnitude of around 3 million years at the martian surface (estimated age calculated using the GEANT4 model, Pavlov et al., 2012). The calculated k values for hopane are around 3 MGy−1, which is comparable with amino acids exposed to gamma rays in fused silica and perchlorate salts at room temperature (Pavlov et al., 2022). We also determined that steranes degraded twice as fast (KCl) or more (NaCl and MgCl2; Fig. 3). Perchlorate salts are highly reactive and, in general, they are studied to understand how they damage biosignatures on Mars. Here, we showed that any irradiation of organics together with these chloride salts can accelerate the loss of organics at a similar, if not faster, rate.
Another parameter that could have accelerated the degradation of organics in salts in the present study is the property of trapping water from the atmosphere in salts, and the fact that water subsequently creates oxidizing radicals under ionizing radiation (Dartnell, 2011), which would further destroy the organics. As we flame sealed our samples under vacuum, reaching pressures as low as 10−3 Torr (see the Methods section), most of the adsorbed water was removed, though traces of water left in the salts may nevertheless have enhanced biomarker degradation. Similarly, it is possible that trace contaminants in the salts may have played a role in accelerating biomarker degradation. According to the vendors, both the NaCl and KCl had ppm-level traces of heavy metals and iron (ppm) as well as oxygen present in the form of chlorate (ClO3), nitrate (NO3), sulfate (SO4), and phosphate (PO4) (∼0.001%). The MgCl2 salt had <3 wt. % water, according to the manufacturers. Further work is needed to fully characterize the potential impact of these trace contaminants in biomarker degradation under ionizing radiation.
Notably, salty environments are a favorable target in the search for extraterrestrial life (McMahon et al., 2018) in that salty brines allow water to be stable in liquid form at lower temperatures, and salty deposits can trap and preserve biosignatures (Schreder-Gomes et al., 2022). Even if we had focused in this study on synthetic mineral matrix materials instead of natural samples, we suggest that targeting salt-rich environments in an irradiated context might be challenging for the detection of the types of lipid biosignatures studied.
FA by-products and potential formation
We did not detect any hopane, sterane, or alkane by-products (such as terpanes produced under thermal degradation of hopanes, Peters et al., 2013; Peters et al., 2005). Our first hypothesis as to why this is the case is that all the missing organics were lost in the gas phase (CH4 and CO2). Further studies analyzing the gases produced under radiation (such as the study by Rojas Vivas et al., 2021) would help in understanding the degradation steps of these organics. Alternatively, there could have been larger by-products, though they may have been too fragile and, therefore, destroyed before accumulating enough to be detected. An experiment with lower irradiation doses would show if such intermediates exist. Another hypothesis is that these hydrocarbons polymerized under radiation, building macrostructures undetectable with our methods, and a study with a pyrolysis step preceding analysis could detect fragments of such macrostructures. Lastly, our sample preparation and GC-MS-FID methods may have been unable to detect some by-products such as organic acids, for example, after oxidation of hydrocarbons (Benner et al., 2000).
The only by-products observed are from the FA mix (all samples, with or without salts): linear aldehydes (even C numbers only) and linear alkanes (all C numbers and a large preference for odd C numbers; Fig. 4). A reason why we are able to detect degradation products for FA and not the other organics could be because of the initial larger quantity (50 µg instead of 2 µg). Even if only a small fraction turns into a by-product, there was a larger amount formed until it was destroyed. Additionally, we may be missing other polar by-products that were not detectable without derivatization. We propose a formation mechanism in Fig. 6, where an even C number FA first loses its OH group, which creates an even C number aldehyde, and second loses its CO group to form an odd C number linear alkane. Additionally, in salts samples where the radiolysis was faster than without salts, aldehydes decreased with the increasing dose (Fig. 4E), which indicates that they are probably an intermediate by-product. Interestingly, we detected alkanes that were longer (C26) than the longest FA (C24), which suggests a potential polymerization of alkane chains under radiation and the possibility of even longer alkane chains that we cannot detect with our methods. Even if they are a minority, we detected even C number alkanes; this could be due to the loss of a methyl group of an odd C number chain, or the polymerization of two short odd C number alkanes. We also identified by-products formed just by storing the FA mix at room temperature (Supplementary Fig. S7) that were not present in the irradiated samples. An explanation for this may be that the room temperature by-products were destroyed quickly under radiation, as the radiation damages are more intense than room temperature exposure.
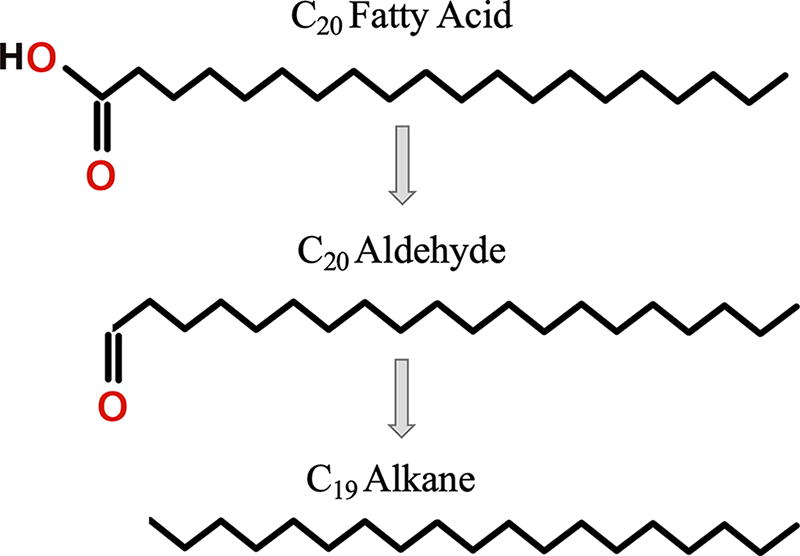
Tentative breakdown steps of FA under radiation and by-products formation. An even C number FA loses its OH group to form an even C number aldehyde. When this aldehyde loses its CO group, there is only an odd C number alkane left.
A previous study (Kminek and Bada, 2006) experimentally demonstrated a positive correlation between the molecular weight and the radiolytic constant k for amino acids, arguing that a larger molecular cross section decreases a molecule’s stability under radiation. However, in the present study, we observed a smaller decrease in large irradiated FA and large alkanes compared with shorter molecules, which implies that larger polymers might be more stable under radiation. A notable difference is that the Kminek and Bada (2006) amino acid results were obtained by irradiating each amino acid separately, whereas, in this study, the alkanes and FA were irradiated in mixes, which allowed for more reactions. For example, our smaller alkanes could polymerize into larger ones, compensating the degradation of the original large alkanes. Further investigation is needed by irradiating alkanes and FA not in mixes but in separate tubes to confirm the trend we report here.
Mechanisms for salts color change and uses in astrobiology
The mechanism behind salt color change under radiation is illustrated in Fig. 7. Under radiation, anions from the crystalline structure can be dislocated and leave an empty space. This space would be filled with electrons to maintain balanced charges, and the excitation of these trapped electrons could give the salt its new color (Ramos-Ballesteros et al., 2021).
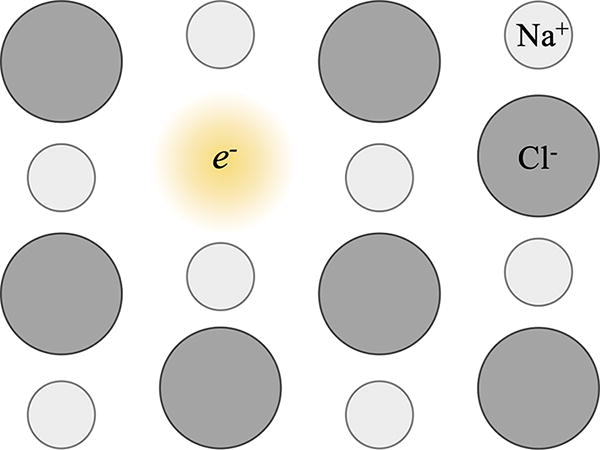
Crystalline structure of NaCl changed after irradiation. A Cl- anion was removed from the structure under radiation and left a hole that was then filled with electrons to maintain the charge balance and gave a new color to the salt.
Salt color change has been used in astrobiology to characterize the surface composition of nonice material on Europa. For example, experimental studies have irradiated mixtures of salts and reproduced the spectral signatures of the moon’s southern hemisphere orange stripes (Hibbitts et al., 2019). Similarly, this phenomenon has been utilized in hypothesizing the composition of its underlying ocean (Hand and Carlson, 2015), as ocean salts are ejected and deposited on the surface and are subsequently exposed to radiation. In addition, as the intensity of the resulting color is proportional to the radiation dose received, these color changes in NaCl have been used as a tool to date young features on Europa’s surface (Poston et al., 2017). This technique has potentially exciting applications for Mars; however, caution is warranted, as color can also be lost by heating the sample (thermal bleaching).
Because average surface temperatures on Europa are much lower than those of Mars (−160°C to −220°C vs. 20°C to −153°C), the biomarker degradation we describe in this study is not directly applicable to icy worlds, where temperature is thought to have a large effect on radiolysis rates (Pavlov et al., 2022). Further research is needed with similar samples and irradiation at icy moon temperatures to evaluate the radiolytic constants in the outer solar system.
Several rover missions are looking for biosignatures on Mars, which has given rise to a need to understand the impact of GCRs on a large range of organic molecules. While primarily amino acids have been studied previously, here we concentrated on four types of lipid biosignatures exposed to gamma rays to mimic GCRs: hopane C30, sterane C27, alkanes, and FAs. The organics were exposed to three doses (0.2 MGy, 0.5 MGy, and 0.9 MGy) at room temperature, in vacuum, and in several matrices (no-salt, NaCl, KCl, and MgCl2). We compared the degradation of different types of organics, by themselves and on different chloride salt substrates, and found that all these molecules degrade much faster under gamma radiation than amino acids in similar conditions despite their resilience in terrestrial samples over geological timescales. Saline environments are often targeted in astrobiology because of their capacity to entrap and preserve biosignatures and because they can imply the former presence of a water-bearing, habitable environment (McMahon et al., 2018; Schreder-Gomes et al., 2022). However, the addition of salt dramatically increased the rate of degradation of the studied organic molecules, which suggests that environments such as brines or salt deposits may be nonoptimal locations for preservation when exposed to radiation. To minimize the possibility of atmospheric water trapped in salts serving as an accelerating agent in the degradation of organics, we sealed our samples under vacuum. Our findings suggest that the presence of NaCl, KCl, and MgCl2 may be just as deleterious for organic preservation as perchlorate salts (Pavlov et al., 2022), although future radiation studies are called for.
Lastly, we did not detect any distinguishing by-products of the irradiated hydrocarbons (hopane, sterane, and alkanes mix) that might have been used on Mars as clues to the presence of irradiated biosignatures or that could have given rise to ratios to illuminate the radiation exposure of a sample. Only FAs produced by-products under irradiation, that is, an intermediate aldehyde after the loss of an OH group, which can then turn into an alkane after the loss of the CO group, though further research is necessary. Overall, our results suggest that we may be currently underestimating the scale of biosignature degradation under GCRs on Mars, and this underscores the importance of targeting sampling sites that are protected from radiation.
Footnotes
Acknowledgment
The authors thank Martin A. Carts and the Radiation Facility at NASA GSFC.
Authors’ Contributions
A.R. designed the study, developed the methods, and completed the GC-MS-FID analyses. A.A.P. assisted with the results’ interpretation. J.P.D. flame sealed the samples under vacuum. S.S.J. supervised the study. A.R. wrote the original article, and all coauthors reviewed and edited it.
Data Sharing Statement
All the raw GC-MS-FID data for this study have been deposited in FigShare repositories. They are available at https://figshare.com/s/c95e409995822ee016c0 (no salt), https://figshare.com/s/ae4e634cd0e4259f8c94 (NaCl), https://figshare.com/s/dea904541366d54ec051 (KCl), and ![]() (MgCl2).
(MgCl2).
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
This work was supported by Future Investigators in NASA Earth and Space Science and Technology (FINESST) NASA grant 80NSSC23K1387 (A.R. and S.S.J.), NASA Planetary Science Division Funding through the Goddard Center for Astrobiology, the Internal Scientist Funding Program through the Fundamental Laboratory Research (FLaRe) (J.P.D.), NASA award number 80GSFC21M0002, and NASA’s Planetary Science Division Internal Scientist Funding Program through the Fundamental Laboratory Research (FLaRe) work package (A.A.P.).
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Figure S8
Abbreviations Used
Associate Editor: Mary Beth C. Wilhelm
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
