Abstract
Geological evidence and atmospheric and climate models suggest habitable conditions occurred on early Mars, including in a lake in Gale crater. Instruments aboard the Curiosity rover measured organic compounds of unknown provenance in sedimentary mudstones at Gale crater. Additionally, Curiosity measured nitrates in Gale crater sediments, which suggests that nitrate-dependent Fe2+ oxidation (NDFO) may have been a viable metabolism for putative martian life. Here, we perform the first quantitative assessment of an NDFO community that could have existed in an ancient Gale crater lake and quantify the long-term preservation of biological necromass in lakebed mudstones. We find that an NDFO community would have the capacity to produce cell concentrations of up to 106 cells mL−1, which is comparable to microbes in Earth's oceans. However, only a concentration of <104 cells mL−1, due to organisms that inefficiently consume less than 10% of precipitating nitrate, would be consistent with the abundance of organics found at Gale. We also find that meteoritic sources of organics would likely be insufficient as a sole source for the Gale crater organics, which would require a separate source, such as abiotic hydrothermal or atmospheric production or possibly biological production from a slowly turning over chemotrophic community.
Introduction
The search for signs of life on Mars, either ancient or extant, is one of the primary pillars of astrobiological inquiry. It is widely thought that early Mars may have been more conducive to life—both in terms of its surface habitability (the ability for life to persist) and its genesity (factors promoting the emergence and evolution of life)—than present-day Mars (Westall et al., 2013; Amador and Ehlmann 2020; Wong et al., 2022). Geomorphological evidence for surface liquid water includes (1) valley networks that may be indicative of precipitation and, hence, an early martian hydrological cycle (e.g., Hynek et al., 2010); (2) giant outflow channels possibly due to the discharge of subsurface reservoirs and large-scale flooding (e.g., Baker et al., 1992); and (3) crater lakes or fluvio-lacustrine systems favorable to hosting life and the preservation of biomarkers (e.g., Cabrol and Grin, 1999). Geochemical evidence also points to extensive aqueous alteration of near-surface minerals, including hydrothermalism (e.g., Ehlmann et al., 2009, 2011; Mangold et al., 2012). Climate modeling suggests that early Mars may have retained at least transient periods of widely habitable states, potentially due to the warming effect of water ice clouds (Kite et al., 2021) or collisionally induced absorption caused by H2 and/or CH4 in a ≳1 bar background atmosphere of CO2 (Wordsworth et al., 2017). Finally, the measured D/H ratio of water on Mars indicates that a great deal of Mars' initial water inventory has been lost to space and/or crustal hydration since the Noachian period (∼4.1–3.7 Ga) (e.g., Villanueva et al., 2015; Scheller et al., 2021). The Mars Science Laboratory's (MSL) investigations in Gale crater have revealed an ancient, intermittent, fluvio-lacustrine environment that likely persisted for hundreds to tens of thousands of years at a time and up to tens of millions of years in total (Grotzinger et al., 2014, 2015).
This paleolake is considered to have been habitable because of its high water activity, neutral pH, availability of bioessential elements (CHNOPS), and suitable redox environment for chemotrophic life. Fluvio-lacustrine environments, especially deltaic sediments such as those found in Gale crater, are of great interest because they are also thought to have high biosignature preservation potential (Ehlmann et al., 2008; Summons et al., 2011; Hays et al., 2017; McMahon et al., 2018; Bosak et al., 2021). Indeed, MSL has detected and characterized complex organic molecules in the sediments of Gale crater (Freissinet et al., 2015; Eigenbrode et al., 2018), and the anomalously depleted carbon isotopic composition of samples from Gale crater could have a biological explanation (House et al., 2022).
If life existed on early Mars, it would likely have been chemotrophic in nature. Chemotrophic organisms rely on environmental redox couples—rather than electromagnetic radiation (i.e., phototrophy)—as their source of free energy. Some models for the origin of life favor a chemoautotrophic start whereby the redox disequilibrium between CO2 and H2/CH4 drives the emergence of core metabolism (e.g., Smith and Morowitz, 2004; Russell et al., 2013, 2014; Nitschke et al., 2023), aided perhaps by natural proton gradients in alkaline hydrothermal systems (e.g., Lane et al., 2010; Lane, 2017; Hudson et al., 2020), transition metal–bearing minerals (e.g., Nitschke et al., 2013; Russell, 2018; Vasiliadou et al., 2019), and the presence of nitrogen oxides (NO x ) as a high-potential electron acceptor and source of fixed nitrogen (e.g., Ducluzeau et al., 2009; Wong et al., 2017). Phylogenetic studies suggest that the last universal common ancestor of Earthly life was likely a chemolithoautotrophic thermophile that lived in an alkaline hydrothermal system (Weiss et al., 2016, 2018). Evidence for hydrothermal systems on Mars raises the possibility that such a chemotrophic emergence occurred on the Red Planet as well (Shock, 1996; Squyres et al., 2008; Ruff and Farmer, 2016). The origin of life is a hotly debated topic, and many other hypotheses also invite the possibility of an emergence of life on early Mars (e.g., Benner and Kim, 2015; Sasselov et al., 2020; Clark et al., 2021; Deamer et al., 2022).
Chemotrophic biospheres are dependent on environmental sources of reductants and oxidants to power primary productivity. Previous attempts to quantify the biomass of hypothetical martian biospheres have focused on atmospheric redox pairs of C-H-O species (e.g., Weiss et al., 2000; Krasnopolsky et al., 2004; Sholes et al., 2019) and more recently on iron redox reactions (Moore and Carr, 2021). While the earliest metabolisms may have acquired free energy from C–H–O redox pairs (e.g., CO2 + H2), it is reasonable to assume that life would evolve and take advantage of other, potentially more exergonic redox couples present in the environment.
In the present study, we consider a chemotrophic metabolism that uses ferrous iron (Fe2+) as its electron source and nitrate (NO3
−) as its electron acceptor, quantify its productivity in the context of early Mars, and examine whether a microbial community based on this metabolism could produce the abundance of organics that MSL measured in Gale crater sediments. Price et al. (2018) advanced the idea of nitrate-dependent Fe2+ oxidation (NDFO) as a plausible metabolism for early Mars. Stoichiometrically, NDFO can be represented by the following equations (Weber et al., 2006; Bryce et al., 2018):
or
NDFO is performed in anaerobic, mesophilic, neutrophilic bacteria on Earth (Weber et al., 2006; Oshiki et al., 2013; Bryce et al., 2018)—microbes that may well have thrived in the conditions of Gale crater's paleolake as evinced by recent laboratory experiments (Price et al., 2022). Both constituents of the NDFO redox couple would have been present during a period of Mars' history conducive to surface habitability. Mars' surface is an iron-rich environment, and although the top few centimeters are oxidized, below this rusty cover lies copious reduced iron (Vaniman et al., 2014); thus Fe2+ is expected to have been abundant in early anaerobic surface waters of Mars. Nitrate can be produced in wet, CO2–N2 atmospheres through lightning and photochemistry (Navarro-González et al., 1998; Wong et al., 2017). Adams et al. (2021) modeled nitrogen fixation under a range of plausible wet-and-warm early Mars atmospheres and found that on the order of ∼109 molecules of NO x cm−2 s−1 may have been delivered to the surface through the rainout of HNO x species. Figure 1 illustrates these processes.
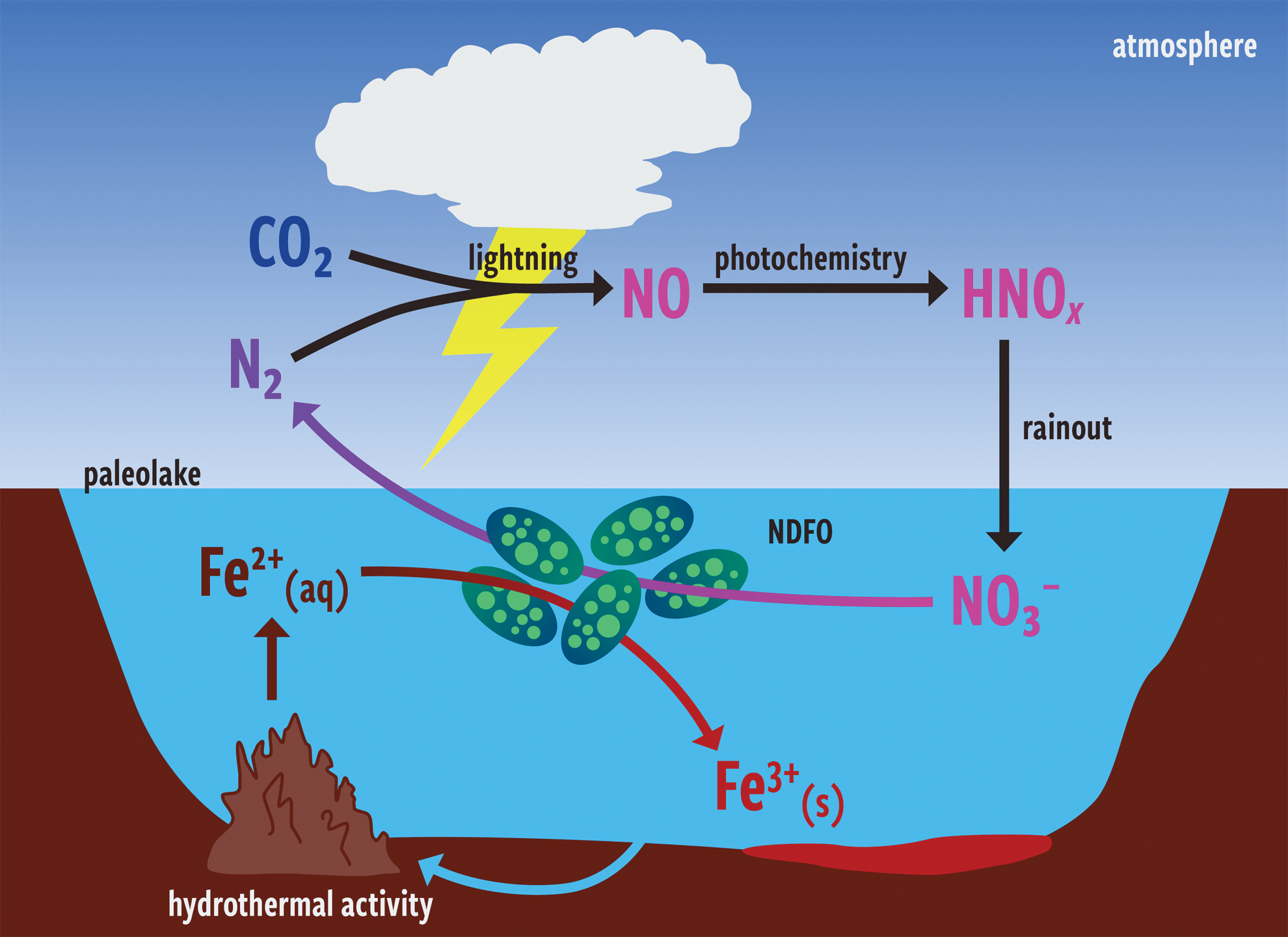
Sketch of hypothetical NDFO-organisms' role in biogeochemical cycles in an ancient martian paleolake environment, following Price et al. (2018).
In the present study, we use this theoretical NO x flux to constrain the hypothetical biological productivity of the NDFO metabolism on early Mars and its possible impact on the organic content of the fluvio-lacustrine sediments of Gale crater.
We outline here our numerical modeling approach to estimate the potential for an ancient NDFO community in a Gale crater lake environment, and the extent to which remnants of this community might currently exist if preserved in the former lake sediment. In subsection 2.1, we consider sources and sinks of vital NO x molecules to Gale crater lake to determine the available free energy for NDFO metabolism. In subsection 2.2, we estimate the size of an NDFO community that could be supported by this available energy. In subsection 2.3, we determine the potential for preservation of organic matter from dead NDFO microbes in mudstone sediments. Finally, in subsection 2.4, we consider the detrimental effects of radiation on preserved organic molecules in the sediment. We use a Monte Carlo approach through the majority of our calculations, and the ranges across which we vary our input parameters are given in Table 1.
Parameters Varied in Model of Gale Crater Biomass Production and Biosignature Preservation
Parameters Varied in Model of Gale Crater Biomass Production and Biosignature Preservation
Life performing NDFO reactions would require a continuous source of NO3
− (see Eq. 1). We use the modeled flux found by Adams et al. (2021) for atmospheric NO3
− precipitation of ∼5 × 108 to 2 × 109 cm−2 s−1. This estimate assumes efficient transformation of precipitated HNO
x
(primarily HNO) into dissolved NO3
− through the following reaction pathway (Adams et al., 2021):
In addition to quantifying the flux of metabolites, it is crucial to know the affinity, A, of the NDFO reaction, that is, the available free energy in the reaction (Eq. 1) that a life-form could exploit as a free energy source. The affinity is critically a function of the concentrations of the products and reactants of the reaction. Generally, high concentrations of the reactants and low concentrations of the products benefit life by allowing high affinity (A > 0), whereas a preponderance of reactants or an excess of waste products could lead to low or even nonexistent (A < 0) available energy. The affinity for Eq. 1a in the lake environment can be found via
Here, G
r
o is the standard Gibbs energy of reaction specific to Eq. 1a, R is the universal gas constant, T is the temperature, and the terms in brackets represent the molal concentrations (≈ activities) of each molecule. We take T to be 298.15 K for simplicity and because it is a reasonable approximation for the clement conditions that would have permitted liquid water on the surface of early Mars. The activities of iron hydroxide Fe(OH)3 and iron carbonate (FeCO3) are taken to be 1 because they are solid phases. The concentration of dissolved nitrogen (N2) is a function of the partial pressure of N2 in the atmosphere and the solubility of N2 in water. We adopt a 1 bar atmosphere of mostly carbon dioxide (CO2) and 10% N2 (an upper limit in the work of Adams et al. [2021]). This leads to pN2 of ∼104 Pa. Assuming equilibrium between the lake and the atmosphere, we can find the dissolved molal concentration of N2, or any gas species i, via Henry's law:
Here, pi
is the partial pressure of gas i in the atmosphere that is in equilibrium with the lake, and Hi
is the Henry's law solubility constant in units of mol kg−1 Pa−1 (Sander, 2015). We use Eq. 4 to determine the dissolved concentrations of both N2 and CO2 in the lake. Dissolved CO2 is necessary to find the concentration of bicarbonate (HCO3
−), which is a waste product (and therefore a determining factor in the affinity) of the NDFO reaction. We calculate this via the pH-dependent inorganic carbon speciation:
Here, K 1 is the dissociation constant of aqueous CO2. For these calculations, we assume a pH of 7, which is consistent with the pH of the ancient lacustrine environment at Gale crater (Grotzinger et al., 2014), to derive [H+]( = 10-pH). The affinities we calculate for the metabolisms in Eqs. 1a and 1b are comparable at the assumed pH of 7, with the two reactions producing ∼320 and 400 kJ mol−1, respectively, so in the rest of the paper we limit our modeling to the reaction in Eq. 1a. While the affinity for Eq. 1b is highly pH-dependent (ranging from ∼30 to ∼750 kJ mol−1 from pH 0 to 14), the affinity for Eq. 1a is relatively constant (ranging from ∼350 to ∼290 kJ mol−1 from pH 0 to 14). Therefore, changes in environmental pH would likely induce at most a ∼10% reduction to our biomass estimates, indicating that there should have been plentiful free energy for NDFO even under highly acidic or alkaline pH conditions at Gale crater.
The only remaining unknown on the right side of Eq. 3 is the concentration of nitrate in the lake, which will be a function of the precipitating flux that provides it, and the biological and photoreductive fluxes that consume or destroy it. Thus, we can write a mass-balance equation for the source and sinks of NO3
−:
Here, all production and destruction fluxes are in units of mol m−2 s−1. The photoreduction reaction is a first-order reaction so that
Here, k photored. is the rate of reaction in s−1, d photored. is the depth (in m) of the lake layer where photoreduction is able to take place, and ρ water is the density of liquid water.
We also define a biological efficiency factor, η = ɸ bio./ɸ precip., which represents the fraction of precipitating nitrate consumed by biology. A biological efficiency factor less than one would mean that NDFO-performing life does not make use of all the available precipitating nitrate, perhaps due to other limiting nutrients or unknown factors that limit metabolic rates. We assume that biology should generally be capable of outcompeting the abiotic destruction of nitrate via photoreduction (e.g., by inhabiting the uppermost layers of the lake), but a biological efficiency factor less than one could also represent a sluggish community that is only able to consume some fraction of the precipitated nitrate that escapes photoreduction. An ecosystem with η = 1 would be perfectly efficient and therefore solely energy-limited, that is, limited only by the available precipitating flux of nitrate from the atmosphere. In that perfectly efficient case, no nitrate would be left to be destroyed by photoreduction. We can reframe Eq. 6 in terms of the biological efficiency factor as below:
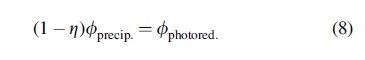
In our analysis, we treat η as a free parameter. By selecting a value for η and combining Eqs. 7 and 8, we can calculate the steady-state concentration of nitrate in the lake. We can then use Eq. 3 to find the affinity (the available free energy) for NDFO in the lake.
The previous section provides a method to determine the availability of biologically useful nitrate in the Gale crater lake system; in this section, we examine the potential size of a community feeding off of that chemical energy. We can first translate the biological consumption flux of nitrate, ɸ
bio., into a rate of free energy consumption via
Here, E is the net rate of free energy consumption by all NDFO organisms in the lake environment (in J s−1), A is the affinity calculated in the previous subsection, and S lake is the surface area of the lake, which we take to be 5.8 × 109 m2 (Palucis et al., 2016).
To find the biomass that would be supported by this rate of total free energy consumption, we must estimate the energy requirements of an individual NDFO microbe. For this value, sometimes called a “basal power requirement,” we adopt a minimum estimate of 3 × 10−23 kJ s−1 cell−1 from Lever et al. (2015), also used by Sholes et al. (2019) in their estimation of the maximum potential biomass in the modern martian subsurface. We can then find the total biomass in the lake via
Here, M total is the total biomass in the lake (kg), m cell is the dry mass of an average terrestrial single-celled organism (kg cell−1), and q BPR is the basal power requirement.
Using the total biomass found from Eq. 10, we estimate the preservation potential of the Gale crater lake environment and the concentration of preserved organics that this ancient microbial community could leave behind. To accomplish this, we consider the rates of organic matter and sediment buildup at the lakebed, the degree to which organic matter is incorporated into sediment, and the chemical preservation of said organic matter via sulfurization.
First, the rate of organic matter buildup is the product of the total biomass in the lake (M total, from Eq. 10) and the average turnover rate of the NDFO organisms (in yr−1). Considering the inherent unknowability of the lifetimes of martian organisms, we adopt a wide range of 10−2 to 104 yr−1 that is representative of the enormous variety in lifetimes seen in terrestrial microbes (Braun et al., 2017; Hoehler, 2022).
We estimate the sedimentation rate in Gale crater lake following the methodology of Grotzinger et al. (2014). In their consideration of the time taken to form the Sheepbed mudstone, Grotzinger et al. (2014) noted that the thickness of the exposed mudstone is ∼1.5 m, and they extrapolated from terrestrial sedimentation rates on the order of ∼1 m per 1000 years (Sadler, 1981) to conclude that the mudstone could have formed over a period of 1500 years (during which Gale was a lacustrine environment). However, as Grotzinger et al. (2014) pointed out, sedimentation rates in terrestrial lake environments vary by several orders of magnitude (Sadler, 1981), and the lake's lifetime (and therefore the period of mudstone formation) could have spanned 104 to 105 years. The lifetimes of lakes in Gale crater have more recently been constrained to be on that order (104 to 105 years), with a minimum cumulative lifetime of 30,000 years (Palucis et al., 2016). Using these lifetimes and the 1.5 m thickness of visible mudstone, we assume sedimentation rates on the order of 10−5 to 10−4 m yr−1, which are also broadly consistent with terrestrial rates (Sadler, 1981), sedimentation rates determined in other Gale crater features, and timescales of orbital obliquity variation that may have permitted clement conditions and surface lakes on early Mars (Lewis and Aharonson, 2014).
We can find the mass fraction of necromass to sediment arriving at the lakebed via the following equation:
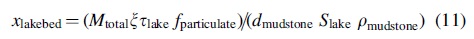
Here, ξ is the turnover rate of the organisms, τ
lake is the lifetime of the lake, f
particulate is the fraction of organic matter that is in the form of particles (as opposed to dissolved), d
mudstone is the thickness of the mudstone, S
lake is the surface area of the lake, and ρ
mudstone is the density of the mudstone. We take f
particulate to be 0.1, based on the average ratios of particulate:dissolved organic matter in both terrestrial lakes and oceans (Wetzel, 1984). We note, however, that not all particulate organic material arriving at the lakebed will be incorporated into the sediment. Betts and Holland (1991) found that the burial efficiency is a function of the sedimentation rate in the context of terrestrial ocean sediment, and we use their semi-empirical function here:
Here, s is the sedimentation rate ( = d mudstone/τ lake) in units of cm kyr−1.
We also seek to determine the fraction of buried organics that are chemically preserved, that is, incorporated into the sediment in such a way that they are protected from chemical degradative processes. Sulfurization reactions of organics on Earth have been found to be an efficient preservation pathway, preserving up to ∼25% of sinking organic matter in ocean sediments during anoxic periods (Raven et al., 2018). Sulfurization may have played a significant role in preserving the organic material found in the Mojave and Confidence Hills sample sites in Gale crater, as evidenced by the high sulfur content of these organics (Eigenbrode et al., 2018). In our calculations, we include a factor that represents the fraction of buried organics that are preserved via sulfurization (f
sulfurization), so the mass fraction of preserved organics to mudstone becomes
As a final step in our modeling, we consider the detrimental effects of radiation on organic biosignatures and the extent to which preserved organic matter from an ancient NDFO community could be seen in modern Gale crater sediments, as sampled by the Curiosity rover. The fraction of organics remaining after some period of irradiation can be described by the following equation (rearranged from Kminek and Bada [2006]):
Here, k is the molecule-dependent radiolysis constant (in Gy−1), R is the rate of radiation (Gy yr−1), and t is the time of exposure (yr). While k varies between molecules and generally increases with molecular weight (Kminek and Bada, 2006), we adopt a value of 1.2 × 105 Gy−1 that represents an average for amino acids (Kminek and Bada, 2006; Pavlov et al., 2012). For simplicity, we assume that the entire biomass of the microbial cell is composed of amino acids on the basis that proteins and their amino acid constituents account for roughly half the dry mass of terrestrial bacteria (Neidhardt et al., 1996; Beck et al., 2018).
Organic molecules in the near subsurface face degradation by both galactic cosmic rays (GCRs) and solar energetic particles (SEPs, aka solar cosmic rays). SEPs dominate the radiation dosage rates within 2 cm of the surface, while GCRs penetrate deeper into the subsurface (Pavlov et al., 2012). We consider the radiation level at 6.5 cm under the surface, which represents the depth of the Cumberland drill sample of the Sheepbed mudstone (Vaniman et al., 2014), where organic molecules in Gale crater were initially found (Freissinet et al., 2015).
We adopt radiation dosage rates in the near martian subsurface from numerical modeling studies that considered both GCRs and SEPs (Dartnell et al., 2007a; Pavlov et al., 2012). At 6.5 cm, we use a radiation rate of 0.054 Gy yr−1, from a combination of GCRs and SEPs (Pavlov et al., 2012). Deeper in the subsurface, the radiation rates would be less pronounced. For example, at a depth of 1 m, the radiation rate would be 0.017 Gy yr−1, and due to GCRs alone (Dartnell et al., 2007a), because SEP penetration is negligible at depths greater than ∼20 cm (Dartnell et al., 2007b). For the total time of radiation exposure, we use 78 ± 30 Myr, an exposure estimate from cosmogenic nuclides that were measured in the Sheepbed mudstone (Farley et al., 2014). Fluctuations in the thickness of Mars' atmosphere would have affected the radiative flux that reaches the surface, with a thicker atmosphere providing more shielding and a thinner one providing less (Pavlov et al., 2012). However, Mars' atmospheric pressure was likely relatively constant over the ∼100 Myr period of radiation exposure that we consider here (Thomas et al., 2023). Therefore, the modern radiation fluxes we use should be representative of the entire history of the Sheepbed mudstone's exposure.
Following the methods described in Sections 2.1–2.2, we use a Monte Carlo simulation to estimate the potential biomass concentration (cells mL−1) of an NDFO community in Gale crater lake (Fig. 2). In this calculation, we treat the biological efficiency factor, η, as a free parameter (see Table 1). While a maximum value of η = 1 defines a perfectly efficient, energy-limited community, a lower limit for η is less easily defined. We set the lower limit for η based on the cell concentrations found in subglacial Lake Vostok, Antarctica, as a representative sparse ecosystem. We find in our calculations that η = 10−5.5 leads to cell concentrations comparable to the minimum measured in the Lake Vostok environment, ∼0.6 cells mL−1 (Bulat et al., 2009). Notably, the upper limit for cell concentrations that we calculate for Gale crater lake (corresponding to the energy-limited case) is approximately equal to the average cell concentration in Earth's oceans, ∼106 cells mL−1 (Fig. 2). In other words, the maximum biomass per unit volume for an ancient martian NDFO community would be similar to that in the modern terrestrial ocean. Unlike the lower limit for biological efficiency (that we arbitrarily prescribe to correspond to Lake Vostok cell concentrations), the similarity between Earth's oceans and the maximum martian NDFO biomass case in our models appears to be coincidental.
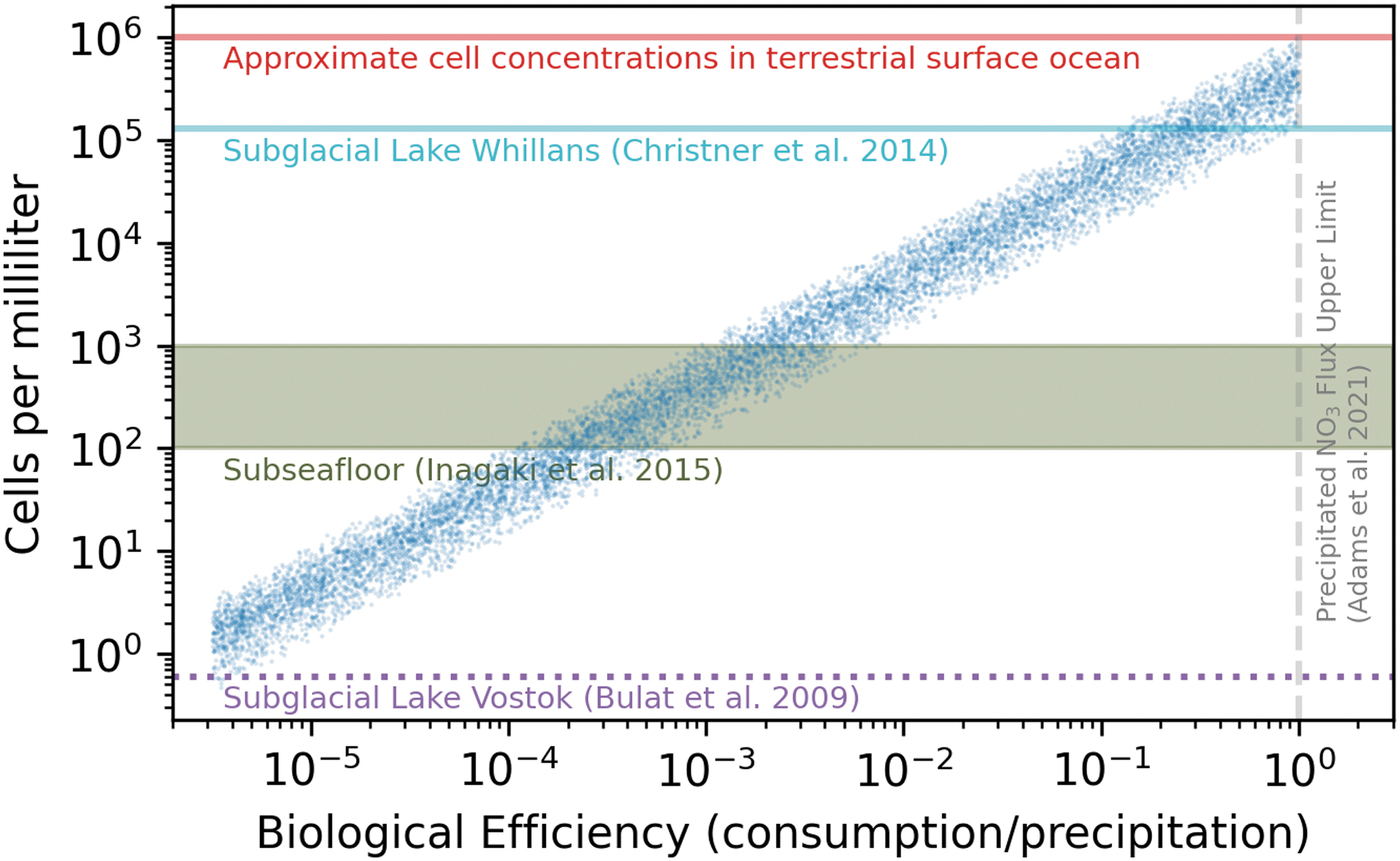
All model runs (blue points) plotted along axes of biological efficiency and cell concentration. Various terrestrial cell concentrations are plotted in horizontal lines/bands. The lower limit on biological efficiency (η = 10−5.5) is prescribed to produce a cell concentration that is comparable to that of Lake Vostok. However, the upper limit on biological efficiency (η = 1) is set by a perfectly efficient, energy-limited community, and the similarity between the cell concentration in this case and the terrestrial ocean is purely coincidental.
Incorporating the models of burial, sulfurization, and radiolysis described in Sections 2.3–2.4, we estimate a wide range in the possible amount of organic molecules preserved in the Gale crater sediment (Fig. 3). Radiation has a relatively minor effect over the 48–108 Myr timescale of exposure, as we estimate the fraction of organics to survive radiation (f remaining) over these time periods to be between 0.50 and 0.74. The biological efficiency (η, the fraction of precipitating nitrate consumed) and the turnover rate of organisms (ξ) are the two varied parameters with the greatest influence on the final mass ratio of preserved organic to rock (Fig. 4). Efficient communities (η closer to 1) tend to produce more biomass (and ultimately necromass), and communities with high turnover rates (in which organisms have short lifetimes) also produce high fluxes of necromass that settle to the lakefloor. By contrast, sinking necromass from an inefficient community (η ≪ 1) would have only a minor contribution to the lake sediment's composition, and low biological turnover rates would similarly allow the burial of necromass to be outpaced by sedimentation, leading to low concentrations of preserved organics in the lakebed. In edge cases, a perfectly efficient (η = 1), rapidly turning over (ξ = 104 yr−1) community could leave behind preserved organic matter at a ratio of ∼10:1 to the inorganic sediment (this case of the lakebed being more organic than rock is probably unrealistic, and certainly inconsistent with the organic fraction measured in the Gale sediments; see Section 4). On the other hand, a highly inefficient (η = 10−5.5), slowly turning over (ξ = 10−2 yr−1) ecosystem could leave behind as little as 0.01 ppb of preserved organic matter.
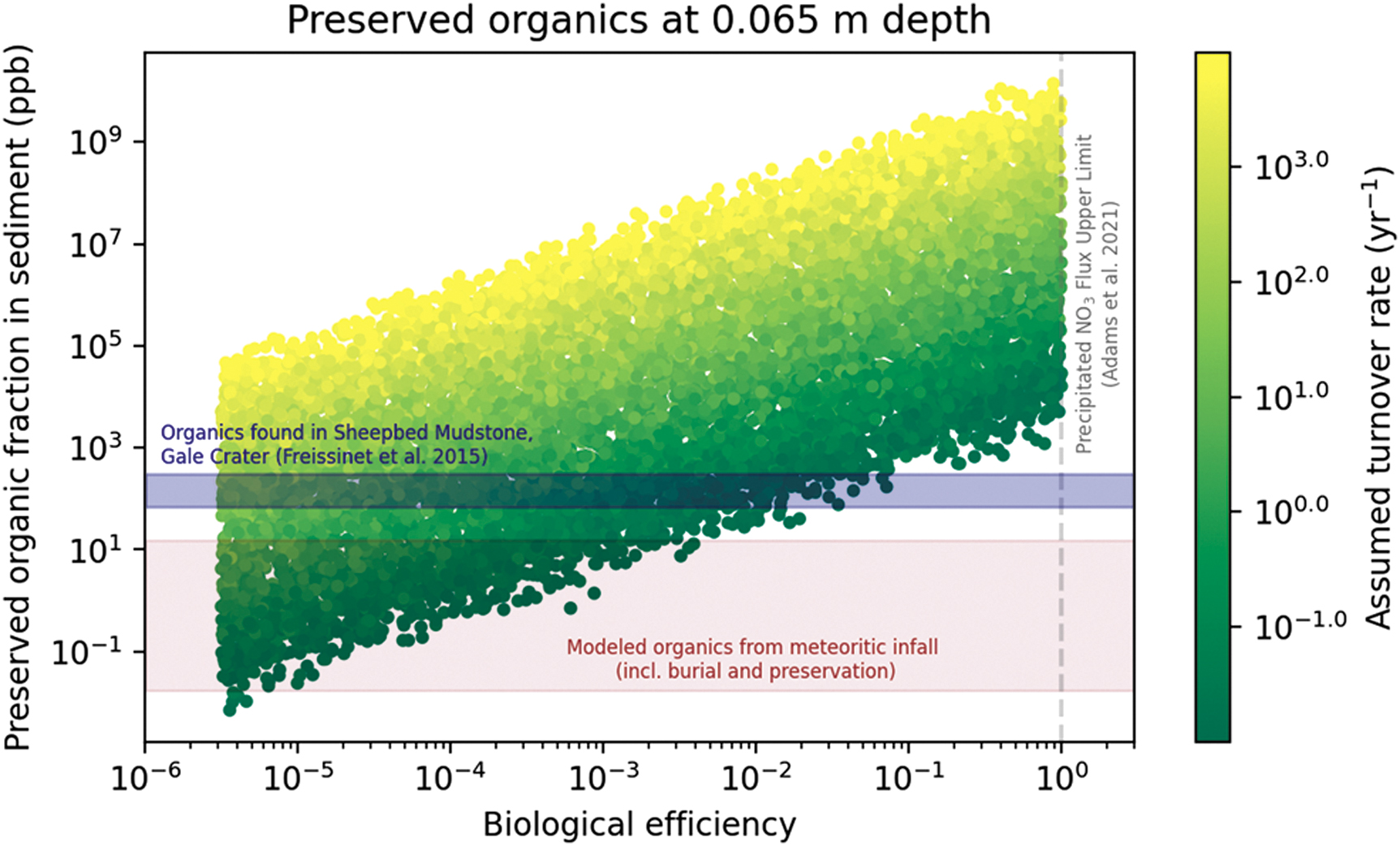
Estimates for the mass fraction of preserved organic matter in Gale crater sediment from an ancient martian NDFO community, incorporating the calculation of organic matter burial, sulfurization, and radiolysis (Sections 2.3–2.4). The x-axis shows the biological efficiency (η), or the fraction of precipitating nitrate that is consumed by biology, which is an input parameter to our model. The y-axis shows our predictions for the mass fraction of organic matter that could be found today in the Gale crater sediment. Each point represents a different case in our Monte Carlo simulation, with the color corresponding to the biological turnover rate (another input parameter, see Table 1). The shaded blue region indicates the mass fraction of organics measured by Curiosity in the Sheepbed Mudstone in Gale crater (Freissinet et al., 2015). The shaded red region is the range in organics that we estimate could be produced by meteorites alone (Section 4.1).
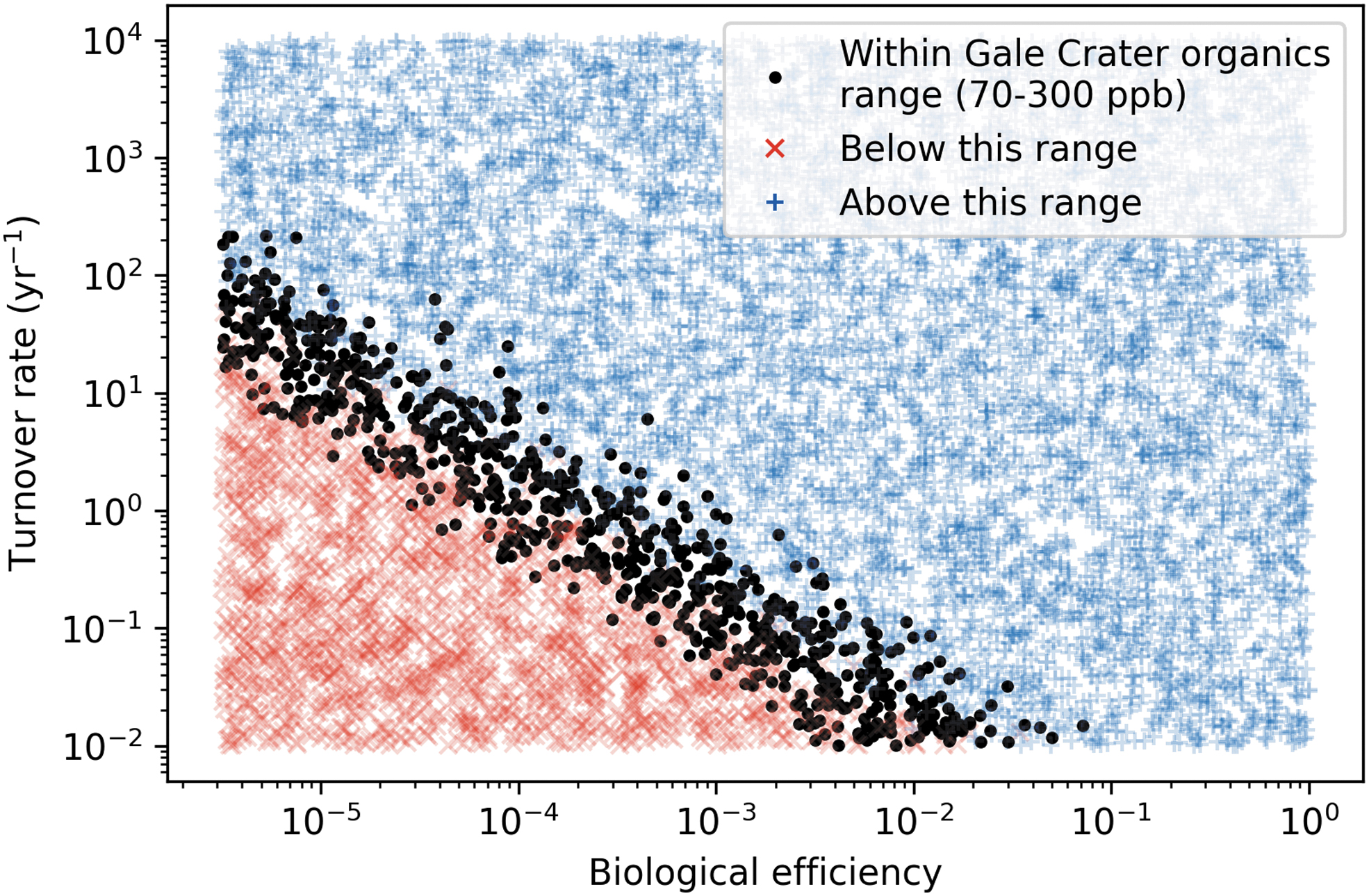
All model runs plotted against axes of biological efficiency and turnover rate. Black circles represent runs that matched the organic content of the Gale crater sediments; red X's represent runs that produced fewer organics than measured; blue +'s represent runs that produced more organics than measured. We predict a maximum biological efficiency of ∼10% and a maximum turnover rate of ∼200 yr−1.
Discrepancy between exogenous organic delivery and preserved organic inventory
Our predictions for preserved organics show that an NDFO community could produce enough biomass to fully account for the organics detected in Gale crater, even with confounding factors such as destruction via radiation. However, biology is only one of several possible explanations for organic-bearing sediments. Certain classes of meteorites, such as carbonaceous chondrites, are rich in organic matter (Mullie and Reisse, 2005) and could plausibly have supplied organics to the Gale crater sediments. Interplanetary dust particles (IDPs), which are approximately 10% carbon by mass (Flynn, 1996), represent another important source of organic carbon and a possible explanation for the organics in Gale crater (e.g., Stern et al., 2022).
Estimates for mass delivery rates of organic carbon to Mars via IDPs vary widely, from 5.1 × 103 kg yr−1 (Carrillo-Sánchez et al., 2020) to 2.6 × 105 kg yr−1 (Flynn, 1996). Estimates for how these delivery rates translate to organic ratios in the martian rock span an even wider range, from 10 ppm to 2.9% (Schuerger et al., 2012; Carrillo-Sánchez et al., 2020), depending on rates of soil production and whether oxidative destruction processes are considered. These predicted abundances could well exceed the 70–300 ppb detected in Gale crater, seemingly suggesting that meteoritic delivery could easily supply said organics. However, most predictions for organic content in Mars' regolith are for conditions preceding the detrimental effects of radiation. Additionally, published estimates often assume meteoritic delivery over billions of years, assume an approximate global average of regolith thickness (potentially overlooking local geological histories that could favor or disfavor organic buildup in specific areas), and assume a well-mixed regolith layer in some cases up to 100 m thick, without invoking a mechanism for mixing. Here, we make estimates for how much organic material could be delivered and preserved specifically in the mudstones of Gale crater.
To calculate the relative abundance of meteoritic organic material that could be preserved in Gale crater, we account for the same burial and preservation processes used in our estimates of biological production: sedimentation and organic burial over the lake's lifetime of <105 years, sulfurization as the primary mechanism for preservation, and radiation as a destructive force in the last ∼100 Myr since the Sheepbed Mudstone was exposed (Farley et al., 2014). We find that meteoritic infall can only account for up to ∼15 ppb of preserved organic material (Fig. 3), falling short of the 70–300 ppb organics measured in the Cumberland sample (Freissinet et al., 2015).
However, 15 ppb may be an underestimate if sulfurization is less crucial to the long-term preservation of meteoritic organics than it is for biological material. If sulfurization can be disregarded as a factor that governs the preservation of exogenous organics (either because meteoritic organics are chemically preserved without sulfurization, or 100% of meteoritic organics are sulfurized), then we predict up to 61 ppb of organics in a best-case scenario. While not wholly inconsistent with the 70–300 ppb measured in the Sheepbed Mudstone, it is apparent that meteoritic sources struggle to produce the observed organic signal. We consider a few possible explanations for this difference below.
In the above calculation, we assumed that meteoritic infall could only contribute to the Sheepbed organics over the relatively short time period when there was a lake in Gale crater, ∼100,000 years (Palucis et al., 2016). Could organics be delivered to the sediment by later meteorite impacts? While it is possible for subsequent emplacement of meteoritic organics to occur after Gale crater lake had dried up, this scenario is unlikely due to the efficiency of organic destruction at an unprotected martian surface by incoming radiation. The destructive potential of the UV flux at the surface is 2–3 orders of magnitude greater than fluxes of organic infall from meteorites (Stoker and Bullock, 1997; Schuerger et al., 2012), which suggests that only organics that are rapidly buried should survive. This justifies our assumption that the lacustrine era of Gale crater, during which the Sheepbed mudstone likely formed via sedimentation, was the only time during which organics could feasibly have been emplaced in the lakebed sediment. Once buried, organics in that layer would be relatively protected from radiation until their later re-exposure to the surface.
Other sources of martian organics
A separate source of organic material is required to explain the discrepancy between the organics expected to be delivered and preserved by meteoritic infall (≲61 ppb) and the organic content of the sediments of Gale crater (70–300 ppb). It is possible that this source could be abiotic—perhaps photochemical or hydrothermal in origin. Future investigations should carefully model these processes to understand whether abiotic sources of organics alone could produce the measured organic content at Gale crater.
Mars' early atmosphere may have contained significant amounts of reduced gasses—such as H2 and CH4, perhaps derived from the hydration of the early martian crust (Scheller et al., 2021)—which would have played a key role in warming the climate enough for liquid water to be stable at Mars' surface (Wordsworth et al., 2017). Photochemistry in CO2-dominated atmospheres with trace amounts of CH4 can produce organic aerosols, which would settle out of the atmosphere to be deposited on the surface (Trainer et al., 2004). The timing and duration of an organic haze layer in early Mars' atmosphere, the rate of production of photochemical organics, these compounds' interactions with the surface environment, and the survival of these molecules throughout potentially numerous redox transitions (Wordsworth et al., 2021) are all highly uncertain and remain outside the scope of this study.
Gale crater was initially excavated by a bolide impact—an event that likely also produced hydrothermal systems, potentially evinced by certain mineralogy within the crater (Yen et al., 2021). On Earth, hydrothermal systems are known to abiotically produce organic compounds through Fischer–Tropsch-type synthesis (e.g., McCollom et al., 1999) or Friedel–Crafts-type synthesis (e.g., Ménez et al., 2018). On early Mars, hydrothermal systems would have likely done the same (Shock and Schulte, 1998), and ongoing hydrothermalism in the subsurface may be the source of the putative methane in present-day Mars' atmosphere (e.g., Yung et al., 2018). The magnitude of hydrothermal organosynthesis on early Mars depends on the duration, pH, temperature, and mineralogy of the hydrothermal systems, as well as the prevalence of reducing compounds (e.g., H2 derived from serpentinization). As these quantities are currently uncertain, we leave an assessment of hydrothermal organic production on early Mars to future work.
Constraints on an NDFO community in Gale crater
If an NDFO-performing community was indeed responsible for generating a large fraction of the organics found in Gale crater sediments, then our analysis places important bounds on the biodynamics of that community. This ecosystem would likely have had a maximum cell density of a few × 104 cells mL−1, which is only ∼10−2 times the concentration of cells in surface terrestrial ocean water (Fig. 5). Interestingly, the range of cell densities we predict for an NDFO-driven martian ecosystem is consistent with those of Earth's subseafloor (Inagaki et al., 2015) but less than that of Subglacial Lake Whillans in West Antarctica (Christner et al., 2014).
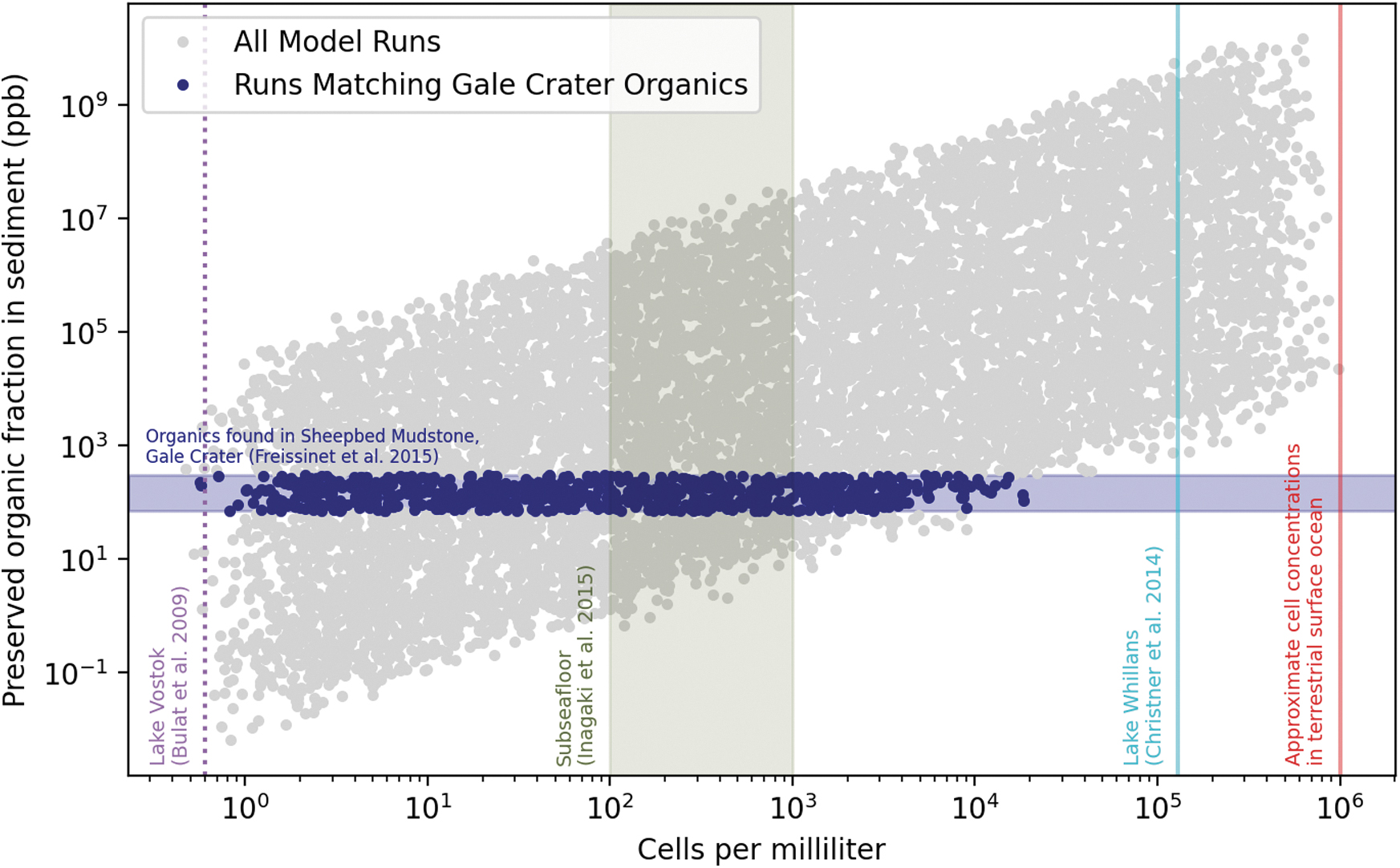
All model runs plotted along axes of cells per milliliter and preserved organic fraction in sediment. Blue circles represent runs that produced the measured organic content in Gale crater sediments; gray circles represent model runs that did not. Based on the observational constraints, we predict a maximum cell concentration of 2 × 104 cells mL−1.
The low cell density that we predict for a hypothetical NDFO-performing community on early Mars is a reflection of the low concentrations of organics found in Gale crater sediments. A purely energy-limited NDFO community could approach the cell density of modern Earth surface waters, but these cases are ruled out due to their inconsistency with the observed organic concentrations (Fig. 5). In other words, the organic content of Gale crater sediments is consistent with a relatively inefficient ecosystem, limited not by energy but something else. Similarly, many ecosystems on Earth are limited not by energy but by the availability of key nutrients, like nitrogen or phosphorus. If Fe2+ was somehow the limiting factor for this hypothetical NDFO community, then the proposed biosignature of Fe3+-minerals in association with complex organics (e.g., Miot et al., 2009; Price et al., 2018) might be elusive, because mineral encrustation on NDFO-performing microbes generally does not occur at lower Fe2+ concentrations (Chakraborty et al., 2011). However, Fe2+ was likely abundant in ancient Gale crater lake (Hurowitz et al., 2017; Price et al., 2018) and therefore unlikely to be limiting to life.
We are also able to place upper limits on the turnover rate (a few × 102 yr−1) and the biological efficiency of nitrate consumption (∼10−1) of a hypothetical NDFO-driven martian ecosystem that could be responsible for depositing the organics found at Gale crater. The fact that a maximum of ∼10% of the precipitating nitrate would be consumed by biology is consistent with the fact that, in addition to organic compounds, nitrate was also deposited and preserved in the sediments of Gale crater (Stern et al., 2015; Sutter et al., 2017). While deposition of nitrate might have occurred after Gale's lacustrine period via production within or upon ice crystals (as occurs on Earth [Honrath et al., 1999]), the contribution of this mechanism to nitrate at the surface and in sediment remains poorly characterized at this time. The amount of nitrate precipitated during more recent intermittent rainfall in Mars' drier period (Kite et al., 2019) is similarly unquantified. In any case, the detection of nitrates within the lacustrine mudstones of Gale crater (Stern et al., 2015; Sutter et al., 2017) suggests that nitrate rainout from the atmosphere (Adams et al., 2021) was concurrent with the lacustrine, potentially habitable window of time that we consider.
There are a number of assumptions in our calculations that could affect our resulting predictions for preserved biomass, which we describe below. For instance, we assume a radiolysis constant that has been adopted in previous studies of radiative degradation on Mars, which reflects an average value for amino acids in terrestrial life (Kminek and Bada, 2006; Pavlov et al., 2012). This average value is based on amino acid distributions in a typical terrestrial bacterial cell (Kminek and Bada, 2006), but we are extrapolating to hypothetical martian organisms for which we cannot know their molecular composition, their possible usage of amino acids, or the mass distribution of these or analogous organic molecules in their cells. However, the martian environment may inform our expectations for biomolecular composition of these hypothetical martian microbes. For example, radiation-resistant bacteria on Earth tend to utilize more low-mass amino acids (Sghaier et al., 2013). Because of the mass-dependence of radiolysis constants (Kminek and Bada, 2006), a microbe that adapted to a harsh radiation environment by using more low-mass amino acids would tend to have a lower average radiolysis constant. Therefore, the constant we assume based on terrestrial averages may be an overestimate, so a more realistic (lower) radiolysis constant could permit higher abundances of preserved organics. In other words, this particular adaptation to Mars' radiation environment would assist in preservation even after microbes were dead and buried, simply due to the biomolecular distribution skewing toward lower mass, less radiolytically vulnerable molecules.
Unavoidably, several other parameters in our calculations are drawn from terrestrial biological and geochemical contexts; therefore, the ranges in values we adopt here may be narrower than, or offset from, ranges that would be more accurate to an early martian biosphere. For example, we adopt turnover rates that range from 10−2 to 104 yr−1 (alternatively stated as turnover times from 10−4 to 102 years), reflecting terrestrial microbes that can turn over rapidly on the order of hours to days, for example, in laboratory environments, or on timescales as long as decades or hundreds of years, for example, at the seafloor (Braun et al., 2017; Hoehler, 2022). It is unclear whether we should expect comparable turnover rates for an ancient martian NDFO community. It is possible that the hypothetical martian community could have faster turnover rates than 104 yr−1, but microbial communities tend toward lower turnover rates over time (Hoehler, 2022), so a community that persisted in Gale crater over the lifetime of the lakes (∼105 years) would likely have a relatively slower turnover rate. An upper limit on the turnover rate could be calculated based on the influx of cellular building blocks and nutrients to the lake, which we leave to future work. A hypothetical lower limit to the turnover rate is less clearly defined. Extrapolating from the trends in Figs. 3 and 4, if the turnover rate is extremely slow (<10−2 yr−1), it would be possible for an extremely energy-efficient and slowly turning-over microbial community to produce comparable preserved organics in the mudstone.
While there is evidence for sulfurization of organics in Gale crater (Eigenbrode et al., 2018), the fractional sulfurization of organics might deviate from the range we use in this modeling study, due to differing concentrations of organic matter, or unknown chemical effects on the kinetics of sulfurization. Additionally, the model used by Raven et al. (2018) to quantify sulfurization of organic carbon on Earth incorporated heterotrophic organisms as a competing sink; that is, only organics that escaped heterotrophy were available to be sulfurized. The model of Raven et al. (2018) closely reproduces the abundances of preserved organic carbon in terrestrial samples from an ancient ocean basin in Pont d'Issole, France, but the translatability of their model to an ancient ecosystem in Gale crater is highly uncertain. We do not directly consider heterotrophs in our model, but their presence is implied by the range in sulfurization factors we include. Future work directly modeling the possible activity of heterotrophs could provide more predictive estimates for their role as a sink for organic matter in Gale crater lake. That said, if the NDFO organisms modeled here were the primary producers of a Gale crater ecosystem, and the turnover rates of NDFO organisms and heterotrophs in the lake were comparable, then the net production of organic matter and ultimately preserved organic matter would likely be similar to our model results.
The basal power requirement (i.e., the rate of energy consumption per cell) is another factor with uncertainty that is not fully captured in our model. Following the example of Sholes et al. (2019), we use a minimum value for the basal power requirement, so we are predicting a maximum biomass that could be sustained by an NDFO community in Gale crater lake. The particular value we use is based on the lowest observed basal power requirement among terrestrial microbes (LaRowe and Amend, 2015; Lever et al., 2015). Theoretically, a true minimum basal power requirement could be lower, but this would depend on the rates of biomolecule degradation through abiotic processes, and the minimum power requirements for microbes to successfully counteract that degradation. As such, a universal minimum basal power requirement is difficult to define even on Earth, due to the dependence on locally variable environmental conditions (e.g., pH, temperature, chemical composition) that govern abiotic degradation processes (LaRowe and Amend, 2015). A clear definition for a minimum power requirement in an ancient martian lake (that has long since evaporated) is even more elusive, but we hope that our calculation provides a framework for approaching the production and preservation of biological material, even as specific values and ranges are updated by continued empirical and theoretical study.
We emphasize that the amount of preserved organic matter in sediment that we estimate is specific to the Sheepbed mudstone in Gale crater. Our calculation accounts for the specific hydrologic and geologic history of Gale crater, including the lifetime of the lake, the sedimentation rate needed to build up this particular stratigraphic section, and time of recent exposure to radiation after uplift. Making similar estimates for preserved biosignatures in other depositional environments (on Mars or elsewhere in the Solar System) would require consideration of the conditions in that specific local context. A comparative study between environments could reveal which environments have the highest potential to preserve biological material and inform decisions on which targets to prioritize for astrobiological missions.
Additionally, our model estimates are limited to the productivity and biomolecular leavings of NDFO-performing microorganisms in Gale crater. Future work could incorporate additional metabolisms, for example, the variety of chemotrophic metabolisms described in Rucker et al. (2023) for another martian environment, Eridania basin. A complete survey of likely metabolisms and their productivity (e.g., as Ray et al. [2021] computed for Enceladus' ocean) coupled to a model of burial and preservation as we describe in this work could reveal a more complete picture of the net preserved biomass and which metabolisms contribute the greatest input.
Future work on isotopic fractionation may help determine whether the organics in Gale crater lake are biogenic in origin. Biological activity is known to impart isotopic fractionation on both carbon- and nitrogen-bearing compounds (e.g., Rothschild and Des Marais, 1989; Zhang et al., 2014). If the preserved organics are indeed necromass from an ancient ecosystem, we might expect that the carbon they contain would be depleted in 13C and 15N. Based on measurements of carbon isotopes thus far, the organics in the organic-rich lacustrine mudstones in Gale crater have a variety of possible sources, including diagenetically altered biomass (Stern et al., 2022). Similarly, if the preserved nitrates are the uneaten leftovers of nitrates devoured by NDFO-performing microbes, we might expect them to be enhanced in 15N. Therefore, we advocate for future measurements that determine the isotopic ratios of the molecular constituents of martian sediments, future studies that improve our understanding of the baseline isotopic ratios of carbon and nitrogen sources, and future research on the isotopic fractionation imparted by abiotic processes that affect the martian environment through time.
Additionally, targeted research on the oxidation states of Fe-bearing minerals in Gale crater could help reveal the possible role of NDFO in generating some of the Gale organics. Due to the uptake of Fe2+ and biomineralization of Fe3+ by NDFO-performing microbes, localized patterns in the iron oxidation states of minerals could serve as biomarkers for NDFO in terrestrial settings (e.g., Miot et al., 2009) or on Mars (Price et al., 2018). In addition to continued experimental work on microbial precipitation of Fe3+ minerals, a natural follow-up to our study would be to model microbial Fe-mineral production at Gale given our biomass estimates and the lake lifetimes and compare these to abiotic sources of minerals containing Fe2+ and Fe3+.
Conclusions
By modeling the potential productivity of an NDFO community on early Mars and the conditions for burial and long-term preservation of those organisms' biomolecules, we have placed some constraints on the possible contribution of this hypothetical ecosystem to the organics that Curiosity found at Gale crater. We also estimated the amount of organics that could be delivered via an alternative, abiotic source in the form of IDPs but found that this exogenous delivery alone would struggle to reproduce the organic-to-rock ratios in the Sheepbed mudstone. Other potential abiotic sources include photochemical reactions in the atmosphere and hydrothermal production at or near the surface; thus future work should consider these sources in detail.
The Gale crater organics may not require a biological explanation, but if an NDFO community was the primary source of those organics, then we have placed constraints on how such a community might perform under plausible conditions for early Mars. Nitrate and the associated energy in the NDFO redox reaction would be in abundance in an ancient Gale crater lake; thus an NDFO community alone could produce a comparable density of biomass to microbes in modern Earth's oceans.
Notably, a relatively inefficient community would be the most consistent with the measured concentration of organics in Gale's mudstones. Although we focus on a single metabolic pathway, the ramifications extend to heterotrophs or other chemotrophs living in concert with the “NDFO-ers.” Since even the NDFO community alone would only produce the Sheepbed mudstone concentrations if they were inefficiently consuming nitrate, other organisms in the lake could be similarly sluggish, or at the very least minimally productive in terms of necromass.
For an NDFO community to be so inefficient compared to its maximum potential, a factor besides free energy must have limited its growth. On Earth, ecosystem biomass is generally limited by key nutrients. The plausibility of a slow-turnover ecosystem in Gale crater lake could be tested by future in situ measurements and/or an in-depth model of sources and sinks of likely key nutrients in the lake environment. If other key nutrients such as phosphorus and sulfur are found to have been abundant in the ancient Gale crater lake, this would suggest that an NDFO community never existed there, because according to our model, an NDFO community with all the nutrients it needed would have surely left a larger mark.
Footnotes
Acknowledgments
We gratefully acknowledge the Optical Absorption of Water Compendium (![]() ) as well as useful discussions with Sebastian Haas, Lydia Kivrak, and Zack Cohen. During the course of this research, MLW was supported by (i) the Virtual Planetary Laboratory, a member of the NASA Nexus for Exoplanet System Science, which is funded via NASA Astrobiology Program grant No. 80NSSC18K0829; (ii) the Carnegie Postdoctoral Fellowship; (iii) NASA through the NASA Hubble Fellowship Program grant no. HST-HF2-51521.001-A awarded by the Space Telescope Science Institute, which is operated by the Association of Universities for Research in Astronomy, Inc., for NASA, under contract NAS5-26555.
) as well as useful discussions with Sebastian Haas, Lydia Kivrak, and Zack Cohen. During the course of this research, MLW was supported by (i) the Virtual Planetary Laboratory, a member of the NASA Nexus for Exoplanet System Science, which is funded via NASA Astrobiology Program grant No. 80NSSC18K0829; (ii) the Carnegie Postdoctoral Fellowship; (iii) NASA through the NASA Hubble Fellowship Program grant no. HST-HF2-51521.001-A awarded by the Space Telescope Science Institute, which is operated by the Association of Universities for Research in Astronomy, Inc., for NASA, under contract NAS5-26555.
Abbreviations Used
Associate Editor: Sherry L. Cady
