Abstract
Rocky planets may acquire a primordial atmosphere by the outgassing of volatiles from their magma ocean. The distribution of O between H2O, CO, and CO2 in chemical equilibrium subsequently changes significantly with decreasing temperature. We consider here two chemical models: one where CH4 and NH3 are assumed to be irrevocably destroyed by photolysis and second where these molecules persist. In the first case, we show that CO cannot coexist with H2O, since CO oxidizes at low temperatures to form CO2 and H2. In both cases, H escapes from the thermosphere within a few 10 million years by absorption of stellar XUV radiation. This escape drives an atmospheric self-oxidation process, whereby rocky planet atmospheres become dominated by CO2 and H2O regardless of their initial oxidation state at outgassing. HCN is considered a potential precursor of prebiotic compounds and RNA. Oxidizing atmospheres are inefficient at producing HCN by lightning. Alternatively, we have demonstrated that lightning-produced NO, which dissolves as nitrate in oceans, and interplanetary dust particles may be the main sources of fixed nitrogen in emerging biospheres. Our results highlight the need for origin-of-life scenarios where the first metabolism fixes its C from CO2, rather than from HCN and CO.
Keywords
Introduction
Understanding the prevalence of life in our galaxy is an inherently multidisciplinary endeavor that binds together research efforts from a large range of scientific fields. Astronomical observations have now detected a significant number of small rocky planets beyond the Solar System (Kossakowski et al., 2023), with successful characterization so far of their orbits, masses, and/or radii as well as the effective temperature (e.g., Anglada-Escudé et al., 2016; Gillon et al., 2017). The imminent characterization of the atmospheric pressure and composition will be key to assessing the potential habitability of such rocky exoplanets. Recently, thermal flux measurements with the flagship James Webb Space Telescope have provided evidence for the lack of atmospheres around the rocky planets TRAPPIST-1b (effective temperature 400 K) and for either a thin atmosphere or lack of an atmosphere on TRAPPIST-1c (effective temperature 340 K) (Greene et al., 2023; Zieba et al., 2023). With the first characterization of an atmosphere of a rocky exoplanet within reach, this emphasizes a fundamental research question: What is the expected composition of the secondary atmosphere outgassed from the magma ocean of a rocky planet after its formation? And how does that composition reflect on possible scenarios for the origin of life on Earth and on other planets?
Many early Earth environments have been proposed as the cradle of life on our planet. These environments range from submarine hydrothermal systems (Russell and Hall, 1997; Sojo et al., 2016) to surficial hydrothermal geysers (Damer and Deamer, 2020) and volcanic pools (Fox and Strasdeit, 2013) and include more niche environments such as floating pumice rafts (Brasier et al., 2011), nuclear-powered geysers (Ebisuzaki and Maruyama, 2017), mica clay sheets (Hansma, 2013), and hydrothermal-sedimentary settings (Westall et al., 2018). Each of these environments has its strong and weak points when it comes to promoting or hampering certain types of prebiotic chemistries that are universally deemed as important (e.g., nitrogen and carbon fixation, C-N bond formation, C-C bond formation, condensation/polymerization reactions). We will not delve into deeper discussions of the pros and cons for each locale and hypothesis here. Rather, we will contextualize our findings on the oxidation state of primitive atmospheres with the associated types of prebiotic chemistry that in turn become more or less feasible as a consequence.
Regardless of the primordial environment in which organic molecules (i.e., molecules that host carbon bonded to other carbon, nitrogen, or hydrogen atoms) began to self-organize and display life-like behaviors, it is important to address whether the environmental feedstock molecules necessary to form such compounds were synthesized within Earth (endogenous source), delivered from extraterrestrial (exogenous) sources, or a mixture of both. It can be argued that the relevance of exogenous delivery as a prebiotic chemistry promoter relies not so much on the type of organics delivered but on the sheer quantity of fixed carbon and nitrogen being delivered versus that which is endogenously synthesized. However, recent research indicates that a nuanced cross-play can occur between many prebiotic molecules and their complex network chemistry (Xavier and Kauffman, 2022), which suggests that the type of organics (that is, not only their quantities) may have had an important impact on the pathway to abiogenesis (the emergence of life from non-living matter).
There is currently no consensus on which organic (as well as inorganic) species are prebiotically relevant as feedstock molecules. Some authors prefer to use ancient life as a guide so that the jump between prebiotic chemistry and the universally conserved core of biochemistry is as short as possible (e.g., Harrison and Lane, 2018). This school of thought explores the nonenzymatic chemistry between molecules and reaction types that are directly represented in (or are very close to) the chemistry of current biomolecules and their biological precursors, for example, using N2/NH3 and CO2 for the synthesis of biomolecules that contain both C and N atoms, such as amino acids and nucleotides. Alternatively, the main competing school of thought favors the usage of more exotic (to life-as-we-know-it) chemical precursors such as HCN and its derivatives in a highly reducing atmosphere (e.g., Haldane, 1929; Oparin, 1938; Wu and Sutherland, 2019), since this type of simple C-N-bearing molecules are very reactive and thus readily build up larger biomolecules such as amino acids and nucleotides. This type of chemistry bears a strong parallelism to that explored in the famous Miller and Urey (1959) experiment, which considered lightning in a heavily reducing atmosphere. Such a scenario must then postulate that once biomolecules are obtained and the system eventually becomes a living one, Darwinian evolution will—over generations—radically modulate the original ‘alien’ prebiotic chemistry toward the CO2-based metabolism used by the Last Universal Common Ancestor (LUCA, see Boyd and Peters, 2013). The prebiotic plausibility of these two approaches relies heavily on the chemical composition (and thus the overall redox state) of the primitive atmosphere. More neutral or mildly oxidizing atmospheres (dominated by CO2) will naturally promote prebiotic chemistry that uses CO2 and N2, whereas a reducing atmosphere (dominated by CO, CH4, and H2) will much more easily promote HCN-related reactivity.
In this study, we focus therefore on first-principles modeling of the potential composition of the first atmosphere of Earth and other rocky planets. We are particularly interested in understanding the oxidation state of the atmosphere. The oxidation state is perhaps the most important unknown in origin-of-life discussions, since the very nature of equilibrium chemistry changes fundamentally between slightly reducing atmospheres where H2, CO, CH4, and NH3 can exist in significant amounts in oxidizing atmospheres that consist almost entirely of CO2, H2O, and N2.
The study is organized as follows. In the section “Atmosphere Equilibrium Model”, we present our chemical equilibrium model for the primordial atmosphere and elucidate the role of atmospheric mass loss in oxidizing initially reducing atmospheres. In the section “Fixation of Nitrogen by Lightning”, we discuss the fixation of nitrogen in atmospheres of various degrees of oxidation and confirm the known result that N2 is fixed by lightning to HCN in reducing atmospheres and to NO in oxidizing atmospheres. We show that the internal production of NO in atmospheres with a realistic oxidation state provides an emerging biosphere with comparable amounts of usable nitrogen relative to external delivery by interplanetary dust particles. In the “Discussion” section, we discuss the implications of our model for the origin of life; in particular, we emphasize that we expect that rocky planet atmospheres will either be oxidizing at the magma ocean outgassing stage or they will undergo later self-oxidation by escape of the H component by XUV irradiation. We conclude briefly on our results in the following section. Appendix 1 describes our XUV mass loss model and discusses various parameter variations to the nominal model. Appendix 2 contains a brief estimate of the possible contribution to fixed N from early asteroid impacts.
Atmosphere Equilibrium Model
The differentiated core-mantle structure of the terrestrial planets in the Solar System provides direct evidence that these objects reached the melting temperature of metal and likely silicate rock during their formation as well. The formation channel of Earth is debated, with classical giant impact models yielding a formation timescale of up to 100 million years (Raymond and Morbidelli, 2022). In contrast, the pebble accretion model postulates Earth formation within the few-million-years lifetime of the gaseous protoplanetary disc (Johansen et al., 2021), which must be supplemented by a single moon-forming giant impact after at least 35 million years to comply with the low amount of 182W in Earth’s mantle (Johansen et al., 2023b; Olson and Sharp, 2023; Yu and Jacobsen, 2011). Mars, in contrast, has been inferred from the same Hf-W (Hafnium-Tungsten) system to have formed its core within just a few million years after the formation of the Sun (Dauphas and Pourmand, 2011). The immense rate of accretion energy released in the compilation of such rocky bodies must, under all circumstances, lead to their partial or full melting and prevalence of global magma oceans on newborn rocky planets (Matsui and Abe, 1986; Elkins-Tanton, 2008; Elkins-Tanton, 2012).
We assume that the atmosphere over the magma ocean evolves in chemical equilibrium with volatiles dissolved in the underlying magma (Elkins-Tanton, 2008; Sossi et al., 2020; Johansen et al., 2023c). These volatiles are delivered to a growing planet by both planetesimals and pebbles. Enstatite chondrites and ordinary chondrites (the meteorite classes that represent the likely composition of planetesimals that formed in the 1 AU region around the Sun) contain significant amounts of hydrogen, carbon, and nitrogen (Grewal et al., 2019; Piani et al., 2020), although measurements of a high hydrogen abundances in enstatite chondrites were criticized recently by Peterson et al. (2023). Johansen et al. (2021) nevertheless demonstrated that these volatiles can also be delivered during the protoplanetary disc phase via rims of ice and organics on small pebbles. Ambient conditions above ≈400 K within the protoplanetary disc would lead to the sublimation of water and destruction of most organic carriers of C and N (Gail and Trieloff, 2017). Formation of rocky planets under such hot conditions could lead to a largely volatile-free magma ocean, as was likely the case for Mercury. Mercury’s mantle indeed has a very low fraction of oxidized iron (Vander Kaaden and McCubbin, 2015), which is in agreement with the idea that Mercury provides an example from the Solar System of a planet formed well interior of the water ice-line. Such a planet could later obtain an atmosphere by delivery of volatiles via impacts of icy asteroids and comets that formed under much colder conditions. We focus instead in this work on studying rocky planet atmospheres that are outgassed from the magma ocean, under the assumption that this was the case for Venus, Earth, and Mars and that magma ocean degassing provides the dominant pathway for rocky exoplanets to acquire their most primordial atmosphere.
We consider here an atmosphere model that comprises four basic volatile-building elements: H, C, N, and O. We assume that the crystallization of the magma ocean occurs from the bottom and up toward the surface (the crystallization takes place over 0.01–1 Myr years after the protoplanetary disc dissipates and planetary accretion terminates 1 ). This leads to efficient degassing of the volatiles from the magma and fixes the respective numbers of H, C, N, and O in the atmosphere, with only minor H, C, and N persisting in the solid mantle but with major amounts of mantle oxygen remaining bound to iron and silicates. The molecular speciation will nevertheless continue to undergo changes as the temperature sinks toward its final equilibrium with stellar incoming radiation. We analyze this equilibrium speciation in the first four subsections for a range of possible atmospheric oxidation states from strongly oxidizing to strongly reducing.
We assume that H, C, and N dissolve in the magma ocean in concentrations that are in equilibrium with partitioning to the core and with the partial pressures of their molecular hosts in the atmosphere (Fischer et al., 2020; Johansen et al., 2023b; Li et al., 2020; Lichtenberg et al., 2021; Speelmanns et al., 2019); with the magma ocean composition assumed to be constant with depth due to mixing by vigorous convection (Rubie et al., 2003; Elkins-Tanton, 2008; Armstrong et al., 2019). While the magma ocean is liquid, we further assume that the partial pressure of O2 over the magma ocean surface is set by buffer reactions in the magma between oxygen and iron in its three oxidation states: Fe0 (metallic Fe), Fe2+ (FeO) and Fe3+ (FeO1 . 5) (Armstrong et al., 2019; Deng et al., 2020). The oxygen pressure, in turn, determines the speciation of H between H2 and H2O and of C between CO and CO2 (Ortenzi et al., 2020). We therefore calculate realistic atmospheric oxidation states as a function of the planetary mass in the subsection “The oxygen fugacity of the magma ocean”.
Young stars have X-ray luminosities up to 600 times the current solar X-ray output and EUV luminosities up to 100 times the current value (Johnstone et al., 2021). These high-energy luminosities typically decay by an order of magnitude over the first 100 million years of rotational slow-down. Molecules and atoms have large cross sections to XUV absorption, which leads to heating of the thermosphere. As the lightest element, H escapes most easily when heated and drags along the heavier atoms and molecules of the atmosphere (Sekiya et al., 1980; Zahnle and Kasting, 1986; Erkaev et al., 2014). In the section “Atmosphere loss and self-oxidation”, we finally expose our atmospheres to mass loss by XUV irradiation and conclude that loss of H drives an efficient self-oxidation of the atmosphere, regardless of its initial oxidation state.
Quantifying the oxidation state
We chose to quantify the oxidation degree of the atmosphere through the ratio R/O, with O denoting the number of oxygen atoms and R denoting a reducing “pseudo-element” that we introduce here and define as:
For R/O < 1, the atmosphere must contain an excess of free O2, while R/O >1 entails that there is not sufficient O to fully oxidize H2 to H2O and C to CO2. With additional knowledge of the bulk atmospheric C/H ratio, the oxidation parameter R/O provides both the C/O and H/O ratios through the relations:
Since most of the original carbon content of Earth’s primordial atmosphere is now stored in the mantle by subduction of carbonates (Sleep et al., 2001; Kadoya et al., 2020), we calculate the probable C/H ratio of the primordial bulk silicate Earth (which excludes any volatile reservoirs in the core) by dividing the C contents of Venus’ atmosphere with the H contents of Earth’s oceans. Estimates of the C content buried in Earth’s mantle are comparable to the measured C contents of the Venus atmosphere (Dasgupta and Hirschmann, 2010), so we prefer to use the more direct measurement from Venus rather than rely on indirect estimates. Our approach yields (C/H)BSE ≈ 0.07 (by number). We emphasize that the majority of the H and C budgets of the terrestrial planets likely reside in their cores (Fischer et al., 2020; Li et al., 2020; Johansen et al., 2023c), but we assume that these reservoirs are not in contact with the magma ocean since it crystallizes from the bottom up (Elkins-Tanton, 2008) and is hence not relevant for estimating the C/H ratio of the primordial atmosphere. This estimate of the primordial C/H ratio also assumes that Earth’s surface reservoir of water has not been depleted significantly with time by hydrogen escape, that it was not changed by impacts with volatile-rich asteroids after the main accretion phase, and that only insignificant amounts of volatiles are trapped in the mantle as the magma ocean crystallizes (we refer to Hier-Majumder and Hirschmann, 2017, for a perspective on this assumption).
An additional assumption on the N/H ratio yields the atmospheric mixing ratios of H, C, N, and O through the expressions:
All quantities involved in evaluating the right-hand sides of these equations can be obtained from C/H, N/H, C/O, and H/O, the latter two obtained from R/O through Equations (2) and (3). We set here N/H = 0.008 by dividing the N contents of Venus’ modern atmosphere with the H contents of Earth’s surface water reservoir. Together with the total pressure, the four quantities X H, X C, X N, and X O now completely describe the atmosphere after the crystallization of the magma ocean. We do not consider here any further exchange of material between the atmosphere and mantle after the crystallization of the magma ocean.
Equilibrium speciation
In our nominal model, we calculate the equilibrium speciation of the four basic elements into molecules using the simplified thermochemical model of Ortenzi et al. (2020) that conserves all four included elements. The chemical equilibrium is given by the balance of the two-way reaction set:
In this model, N is hosted entirely in the strongly bound N2. We ignore here any exchange of O between Fe2+/Fe3+ in the mantle and H2/H2O in the atmosphere, under the assumption that the magma ocean crystallizes rapidly after the end of the accretion (Kite and Schaefer, 2021) and that Fe in the crust does not react with H2O or O2. At low temperatures (but not too low pressure, see Tian and Heng, 2024), two more reactions become relevant for determining the main host of C and N (see discussion in the work of Heng and Tsai, 2016),
In oxidizing atmospheres, with R/O < 1 and negligible free H2, the reactions with H2 in Equation (10) and (11) are unimportant. However, when transitioning to reducing conditions with R/O >1, the equilibrium C host molecule shifts from CO to CH4, and the equilibrium N host shifts from N2 to NH3 at the temperatures below 500–600K relevant for the early atmosphere (Hirschmann, 2012; Sossi et al., 2020). Both CH4 and NH3, nevertheless, have large cross sections to destruction by UV photolysis (Kuhn and Atreya, 1979; Kasting, 1982; Romanzin et al., 2005; Zahnle et al., 2013); NH3 is photolyzed at photon wavelengths between 160 nm and 230 nm and CH4 below 145 nm (Kasting, 2014). Zahnle et al. (2020) concluded that oxidation of CH4 to CO is the major destruction route of CH4 in models of reducing impact-produced atmospheres, particularly when the top of the atmosphere contains significant amounts of water vapor.
Line et al. (2011) found efficient photolysis of CH4 at pressures below 10−5–10−6 bar in models of the hot Neptune GJ 436 b. Kasting (2014) estimated the timescale to photolyse and oxidize 10 bar of CH4 to scale as:
This equation is normalized here, as in the work of Kasting (2014), to a UV flux of 5 times the modern value, relevant for the 0.1 to 1 Gyr epoch of a relatively slowly rotating star (see Fig. 5). In the section “Atmosphere loss and self-oxidation”, we discuss that, during the earliest epoch of stellar evolution out to 100 Myr after planet formation, the XUV fluxes were 1–2 orders of magnitude higher than that of the modern Sun. Therefore, the CH4 photolysis timescale would likely have been ∼1 Myr during the crystallization of the magma ocean and the cooling of the mantle. Importantly, the surface temperature in our models is set to 500 K and, hence, reformation of CH4 in the main atmosphere is kinetically inhibited (see discussion in Section 2.4). Other works (Zahnle et al., 2020; Wogan et al., 2023) have reported longer lifetimes of CH4 compared to Equation (12). To bracket reality, we therefore present in Section 2.4 the results of a model where the abundances of both CH4 and NH3 are allowed to reach their equilibrium values in the absence of photolysis. 2
Regarding the second route to CH4 formation, abiotic serpentinization by reaction of water with the primitive mantle, kinetic models that include the formation of methane by serpentinization and destruction by photolysis find very low CH4 concentrations, at the ppm level, in oxidizing atmospheres (Guzmán-Marmolejo et al., 2013) and extremely low HCN concentrations near the surface at the 10−16–10−14 level (Pearce et al., 2022). We discuss the kinetic freeze-out temperatures of CH4, NH3, and HCN in more detail in Section 3.
Chemical equilibrium of CH4- and NH3-free atmospheres
We start by analyzing the case where CH4 and NH3 are assumed to be efficiently dismantled by photolysis. The equilibrium ratios of H2, H2O, CO, and CO2 change with the ambient temperature. Particularly, the so-called water-gas shift reaction:
In the top panel of Figure 1, we show the relative abundances of 2O2/O, 2H2O/H, and CO2/C as a function of the reduction parameter R/O and C/H at a total pressure of 100 bar and temperature of 500K. As expected, O2 becomes an important O carrier for R/O < 1, irrespective of C/H, but vanishes for R/O ≥ 1. Increasing R/O further causes first H2O to convert to H2 while CO2/C is always at its maximal value of unity. Remarkably, the equilibrium chemistry dictates that, only when the atmosphere is already completely depleted of H2O, will any further increase of R/O allow CO to co-exist with CO2. This transition happens at C/O = 1/2, since higher values of C/O imply an oxygen budget insufficient to fully oxidize all C to CO2. The atmosphere finally becomes ultra-reducing at C/O ≥ 1. We do not model this H2O- and CO2-free state here, given that such a reducing composition is not plausible for realistic magma ocean oxidation states (Hirschmann, 2022).
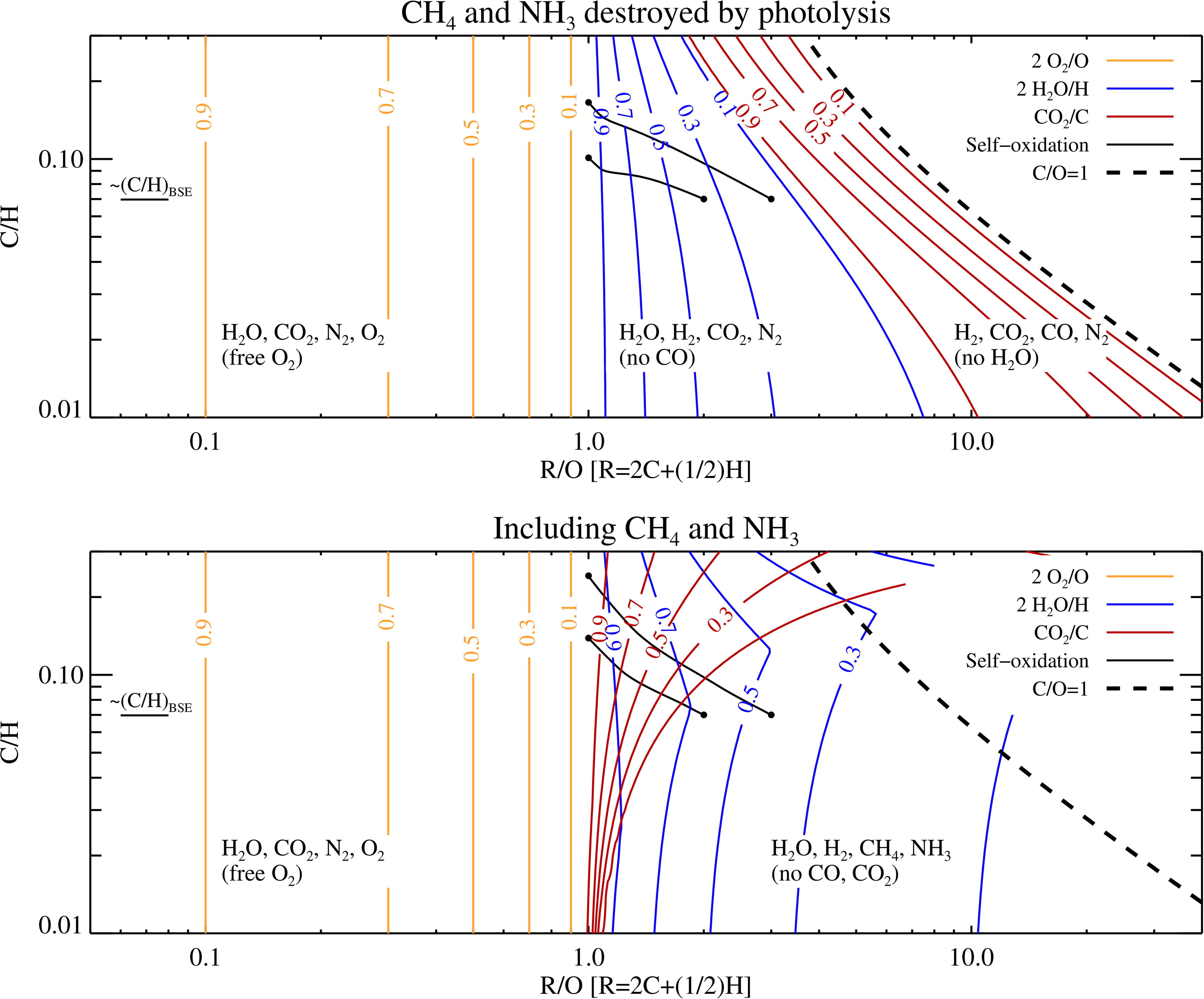
The top panel shows that, for a model where CH4 and NH3 are assumed to be destroyed by photolysis, the equilibrium abundances of O2, H2O, and CO2 at a total pressure of 100 bar as a function of the reduction parameter R/O and the C/H ratio, calculated at 500 K and normalized to their characteristic elemental constituent (O, H, and C, respectively). For R/O < 1, free oxygen must exist and O2 even becomes the dominant O-carrier for very low values of R/O. At R/O > 1, H2O is first deprived of O and transformed to H2, oxidizing in the process all CO molecules to CO2. However, at large values of R/O, the O budget becomes insufficient to maintain 100% CO2; this leads to an increase in the fraction of C that resides in CO until the critical point of C/O = 1 is reached. The black lines with dotted start and end points indicate self-oxidation tracks by H loss (from the data in Fig. 6, starting at R/O = 2 and R/O = 3 for the 50% rotator host star and including the diffusion limit). The bottom panel shows results when including CH4 and NH3 in the chemical equilibrium. These two molecules become the dominant C and N hosts for high R/O and low C/H. CO is nearly absent in the lower panel for the low C/H ratios tested here, which implies that the CH4 abundance can be read off directly from the CO2 curves through CH4/C ≈ 1-CO2/C.
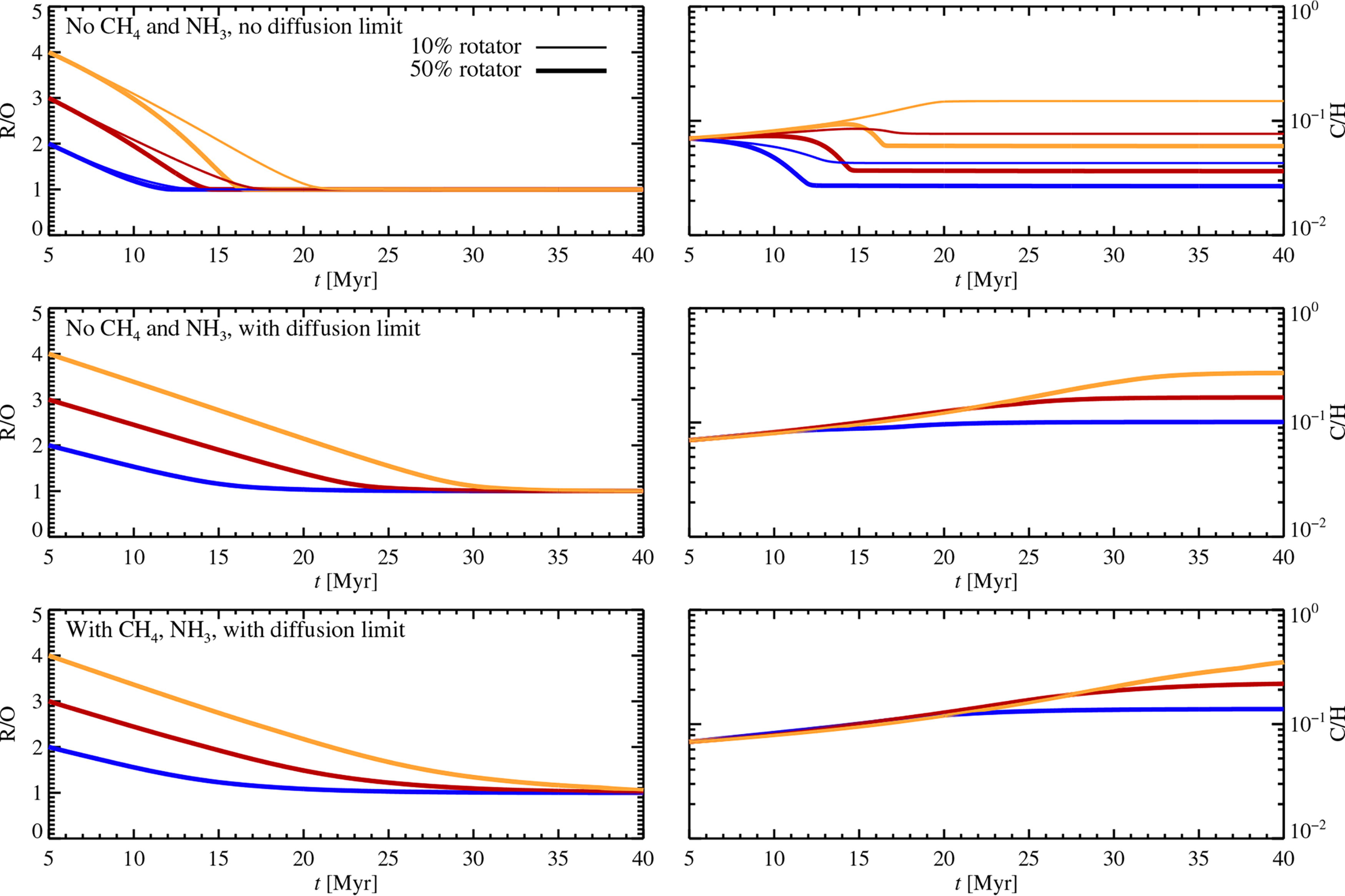
The evolution of R/O (left panels) and C/H (right panels) due to mass loss by XUV irradiation, for models without CH4 and NH3 (top and middle panels) and a model including those molecules (bottom panel). In the top panel, we do not apply a diffusion limit to the H escape flux. The atmospheres undergo self-oxidation (moving towards R/O = 1) within 10–20 Myr for the first case and within 20–40 Myr for the two cases that include the diffusion limit, due to the escape of H. This H flux drags out the heavier C, N, and O atoms as long as the flux is sufficiently high. The higher H flux in the top panel leads to a decrease of C/H for the 50% rotator star; this is a result of the efficient loss of C (hosted in CO and CO2) combined with an inefficient loss of H2O that is cold-trapped in the lower atmosphere. In contrast, C/H always increases with time in the middle and bottom panels where the diffusion limit is applied, and heavier atoms and molecules fractionate substantially in the weaker escape flow.
The equilibrium of the water-gas shift reaction precludes the coexistence of H2O and CO at low temperatures. We nevertheless need to analyze the speed (or kinetics) of the reaction to assess the minimum temperature range at which the equilibrium is reached during the relevant evolution timescale of the atmosphere. The slowing of chemical kinetics at low temperatures can cause an atmosphere to maintain the composition of an effective equilibrium temperature that is much higher than the actual temperature. The kinetics of the water gas shift reaction were studied experimentally by Graven and Long (1954), who found the following production rate of CO2 given known concentrations of CO, H2O and H2,
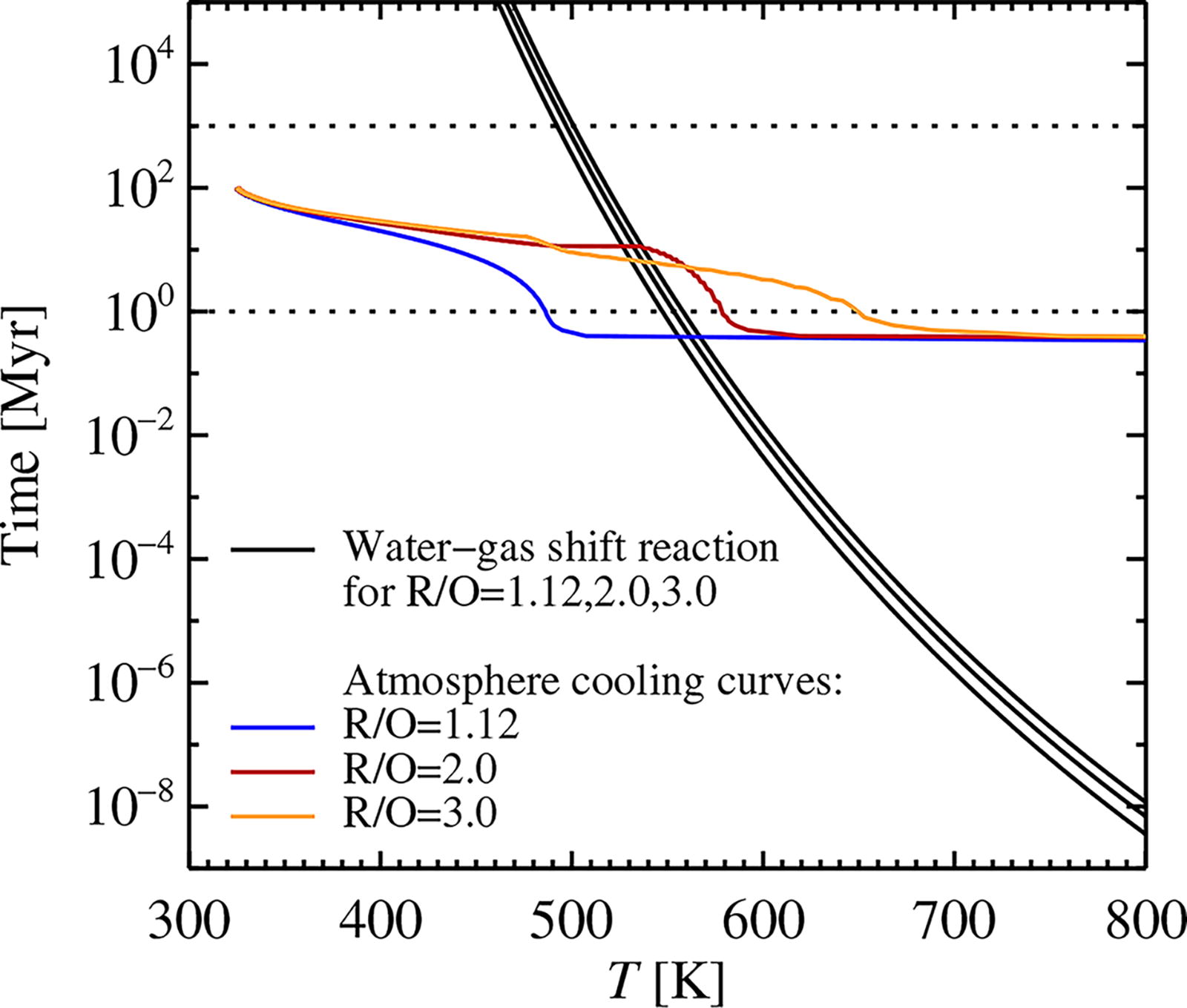
The reaction timescale for the water gas-shift reaction, calculated from the work of Graven and Long (1954), for three reduction states R/O = 1.12, 2.0, 3.0, and an Earth/Venus-like H, C, and N reservoir. We overplot the cooling curves for the three R/O values during and after termination of planetary accretion, assuming that the accretion luminosity drops on a timescale of 0.1 Myr and that H2 and CO2 are subsequently removed (by XUV irradiation and carbonate burial, respectively) on a 10 Myr timescale. The freeze-out temperature of the water-gas shift reaction lies generally in the interval between 500 K and 550 K.
We over plot in Figure 2 the cooling curves for atmospheres with three different values of the oxidation state R/O (1.12, 2.0, and 3.0). We wish to compare these reaction rates with the rate of cooling of the atmosphere. For computing cooling curves, we use the atmosphere equilibrium model of Johansen et al. (2023b), which integrates the atmosphere from the surface to the photosphere under hydrostatic equilibrium and appropriately chooses either the radiative or the convective temperature gradient. The code uses a gray approach to radiative transfer, with the opacity of H2O and CO2 proportional to the pressure (κ H2O = 1.0 m2 kg−1 at 1 bar pressure for H2O and κ CO2 = 0.01 m2 kg−1 for CO2). The latent heat of water is taken into account in the convective temperature gradient (Leconte et al., 2013). We initially decrease the accretion luminosity (i.e., the energy release rate of the accreted material) over a timescale of 105 yr to mimic the dissipation of the protoplanetary disc and the decline of the pebble accretion rate in the terrestrial planet region, until stellar irradiation becomes the dominant heat source. 3 We set the planetary albedo to A = 0.5 and the stellar luminosity to 0.7 times the modern solar luminosity. Both H2 and CO2 are subsequently removed on a timescale of 10 Myr, to mimic the loss of H2 by XUV irradiation (see Section 2.6) and the loss of CO2 by sedimentation of carbonates in the ocean. Sleep et al. (2001) derived a CO2 removal timescale between 10 Myr and 100 Myr (which depends, among other things, on the availability of cations to bind C in mineral form); we chose here the lower limit to reduce the computational time of the model.
The cooling curves in Figure 2 show that the more reducing models obtain a higher temperature in equilibrium with stellar irradiation. This is due to the lower amounts of water in these models, which decreases the cooling effect of moist convection. We also tested the effect of our choice of the opacity level of CO2, by comparing the nominal level of κ CO2 = 0.01 m2 kg−1 at 1 bar pressure (Badescu, 2010) and a lower level of κ CO2 = 0.001 m2 kg−1, which we found to be more consistent with the temperature of modern Venus (Johansen et al., 2023b). The opacity level of CO2 nevertheless has a negligible effect on the temperature structure, which is mainly set by the opacity of H2O and the moist adiabat of the H2O/CO2/N2 mixture. Overall, Figure 2 demonstrates that the water-gas shift reaction freezes out at a temperature interval between 500 K and 550 K. This value is mainly dependent on the removal timescale of CO2 in the two reducing cases with R/O = 2.0 and R/O = 3.0. Under all circumstances, this temperature range is low enough that CO is effectively converted to CO2 during the cooling.
Chemical equilibrium of atmospheres with CH4 and NH3
The kinetic timescale of Equations (10) and (11) to reach equilibrium was reported by Zahnle et al. (2020) and Liggins et al. (2023). At an initial cooling time of 100,000 yr, Liggins et al. (2023) found that the CH4 equilibrium freezes out at 700 K and NH3 at 1000 K. As we demonstrate later in our lightning heating calculations, CH4 and NH3 become significant C and N hosts at those temperatures (Fig. 9). The equilibrium timescales at 500 K rise steeply to ∼109 yr for CH4 and ∼1010 yr for NH3. Hence any destruction of those species by photolysis will not be replenished at the likely surface equilibrium temperature of the early atmosphere.
We nevertheless consider here a model that includes both CH4 and NH3 in the chemical equilibrium in the bottom panel of Figure 1. We use the FastChem package, described in Section 3.3, to find the chemical equilibrium. FastChem does not include water condensation, so we ignore this effect, as we did as well for the calculations in Section 2.3. The nature of reducing atmospheres changes character drastically compared to the nominal model (shown in the top panel of Fig. 1), with CH4 and NH3 taking over as the dominant carriers of C and N for high R/O and low C/H. Atmospheres with a higher C/H value show significant amounts of CO2 co-existing with CH4. Here, H2O is present at all values of R/O, in contrast to the nominal model, since H2 is oxidized by the excess O released when C is transferred from CO2 and CO to CH4. We demonstrate in Section 2.6 that XUV-driven mass loss leads to efficient self-oxidation in both the nominal chemical equilibrium model without CH4 and NH3 and in the model analyzed here that includes these molecules; hence, the main result of our paper is relatively insensitive to whether CH4 and NH3 are destroyed by photolysis.
The oxygen fugacity of the magma ocean
We have so far considered R/O to be a free parameter in our chemical equilibrium models, but in reality, this value is set by outgassing of C, H, and O from the magma ocean. We calculate the equilibrium composition of the outgassed atmosphere over the magma ocean at a temperature of T = 1900 K, for a range of magma ocean oxygen fugacities relative to the iron-wüstite buffer reaction Fe + (1/2)O2 ⇌ FeO (Ortenzi et al., 2020; Hirschmann, 2021). The oxygen fugacity (i.e., the chemical activity or effective partial pressure of the gas measured in bar) relative to a strongly oxidizing magma ocean with 100% FeO is denoted in logarithmic form by ΔIW (Huang et al., 2021); we vary ΔIW from −2 (strongly reducing) to +4 (strongly oxidizing). Figure 3 shows the composition of the outgassed atmosphere as a function of ΔIW at a total pressure of 100 bar. The figure also shows the composition when the temperature is lowered from the initial T = 1900 K to either T = 500 K or T = 550 K (the likely interval of the freeze-out temperature, see Fig. 2), under conservation of H, C, N, and O and under the assumption that CH4 and NH3 are irrevocably destroyed by photolysis. The water-gas shift reaction very efficiently destroys CO such that CO2 becomes the dominant C-carrier even at low oxygen fugacities.
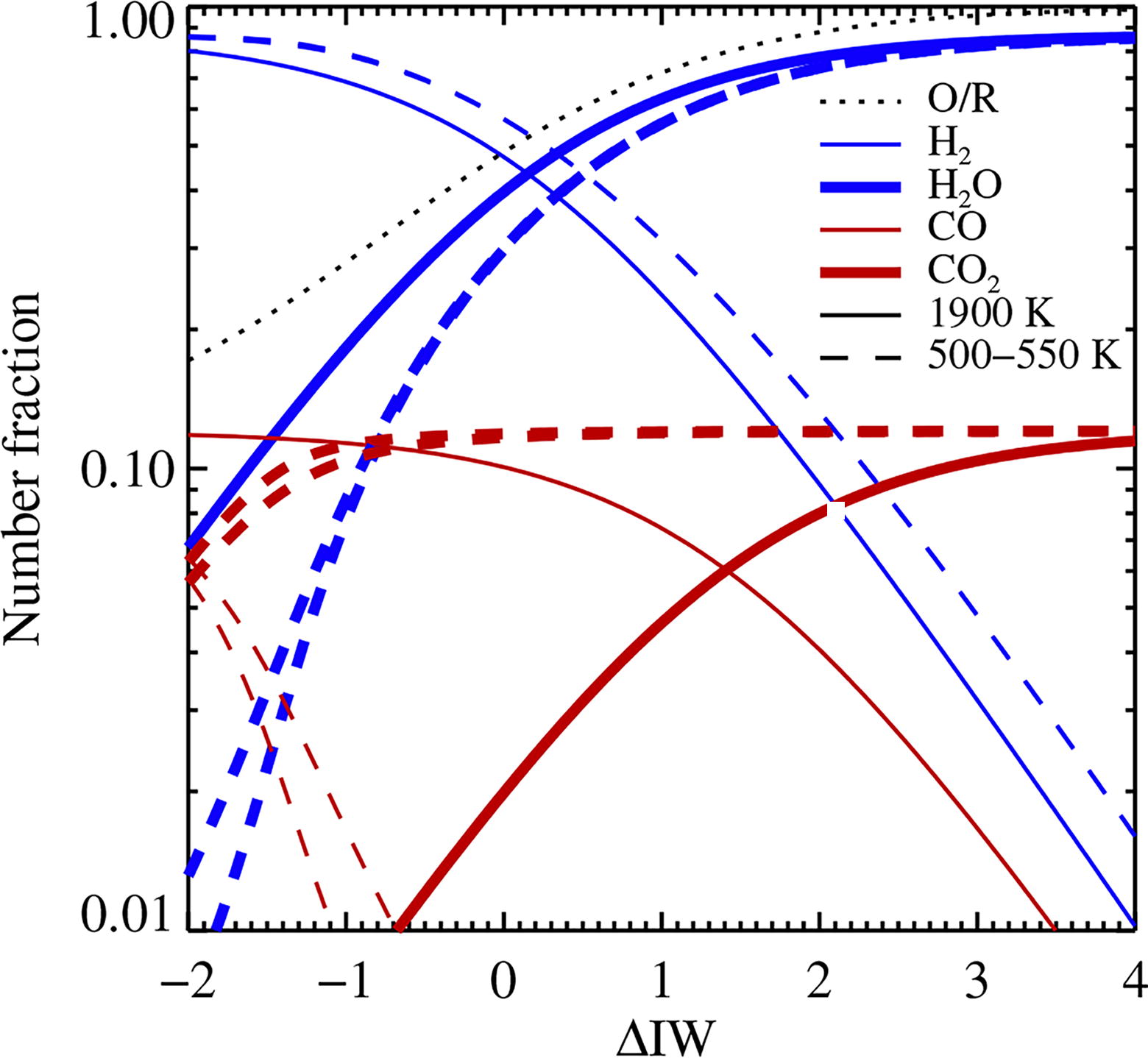
The equilibrium mixing ratios of H2, H2O, CO, and CO2 for an atmosphere with 100 bar pressure and C/H = 0.07 buffered in oxygen by the magma ocean at T = 1900 K (full lines) and after cooling down to T = 500 K or T = 550 K (dashed lines) under conservation of elements. We exclude here CH4 and NH3 from the equilibrium. The composition is given as a function of the magma oxygen fugacity relative to the iron-wüstite buffer (IW), ΔIW = log(P O2 /bar) − log(P IW /bar). The dotted line shows O/R = (R/O)−1; the atmosphere becomes strongly oxidizing for ΔIW ≳ 1…2. The water-gas shift reaction efficiently destroys CO by reacting with H2O when the temperature is decreased. H2O and CO clearly cannot coexist in significant quantities at low temperatures; CO2 becomes the dominant C-carrier even at low oxygen fugacities.
The oxygen fugacity of the magma ocean relative to the iron-wüstite buffer is expressed as a function of the magma composition as:
This expression is valid when FeO in the magma is in balance with free metal (Huang et al., 2021). Here a denotes the chemical activity of the dissolved species (approximately equal to the mole concentration in the relevant solvent): a FeO denotes the activity of FeO in the silicate melt and aFe denotes the activity of iron in the descending metal droplets. Earth’s mantle FeO fraction (8%) implies differentiation at IW − 2, while Mars likely differentiated at IW − 1.5 due to this small planet’s higher FeO fraction of 14–18% (Hirschmann, 2022; Johansen et al., 2023a; Yoshizaki and McDonough, 2020. The reaction FeO + (1/4) O2 ⇌ FeO1 . 5 within the silicate melt nevertheless becomes increasingly important at high pressures. As planetary accretion terminates and free metal sediments from the magma ocean, the current leading hypothesis to explain the high oxidation state of Earth’s mantle is that the ratio of Fe3+ (in FeO1 . 5) to Fe2+ (in FeO) was set by the conditions at the core-mantle boundary (CMB) where a high Fe3+ fraction is necessary for the silicate liquid to remain in equilibrium with core metal (Armstrong et al., 2019). Efficient convective flows, driven by the hot lower mantle that has been heated by the sinking of metal to the core, dictate a constant Fe3+/Fe2+ ratio from the CMB up to the magma ocean surface (Armstrong et al., 2019). The increase in the mean Fe oxidation state near the surface yields a high oxygen fugacity there and, hence, the outgassing of an oxidizing atmosphere. Here, we follow the calculations of Deng et al. (2020) and Hirschmann (2022), who used thermodynamical modeling of the equation state of FeO and FeO1 . 5 valid up to a pressure of approximately 140 GPa, to calculate the oxygen fugacity of the magma ocean.
The calculated oxygen fugacity at the surface is shown as a function of pressure in Figure 4 for an Earth-like core-mantle boundary oxygen fugacity at IW − 2. We base the calculations here on the composition of Earth, since the mantle equation of state for the Mars composition in Hirschmann (2022) is fit to pressures only up to the Mars core-mantle boundary; we checked that up to this pressure, the results based on either the Earth or the Mars composition yield relatively similar surface oxygen fugacities. Figure 4 shows that the magma ocean of a Mars-mass planet has a surface oxygen fugacity of IW − 1 after the termination of accretion (which would rise to IW − 0.5 for the actually higher FeO fraction of Mars, while an Earth-mass planet outgasses its atmosphere under very oxidizing conditions at IW + 2 (Sossi et al., 2020). These conditions are slightly less oxidizing than the results of Armstrong et al. (2019), who used an equation of state valid at low pressures (see discussion in Hirschmann, 2022).
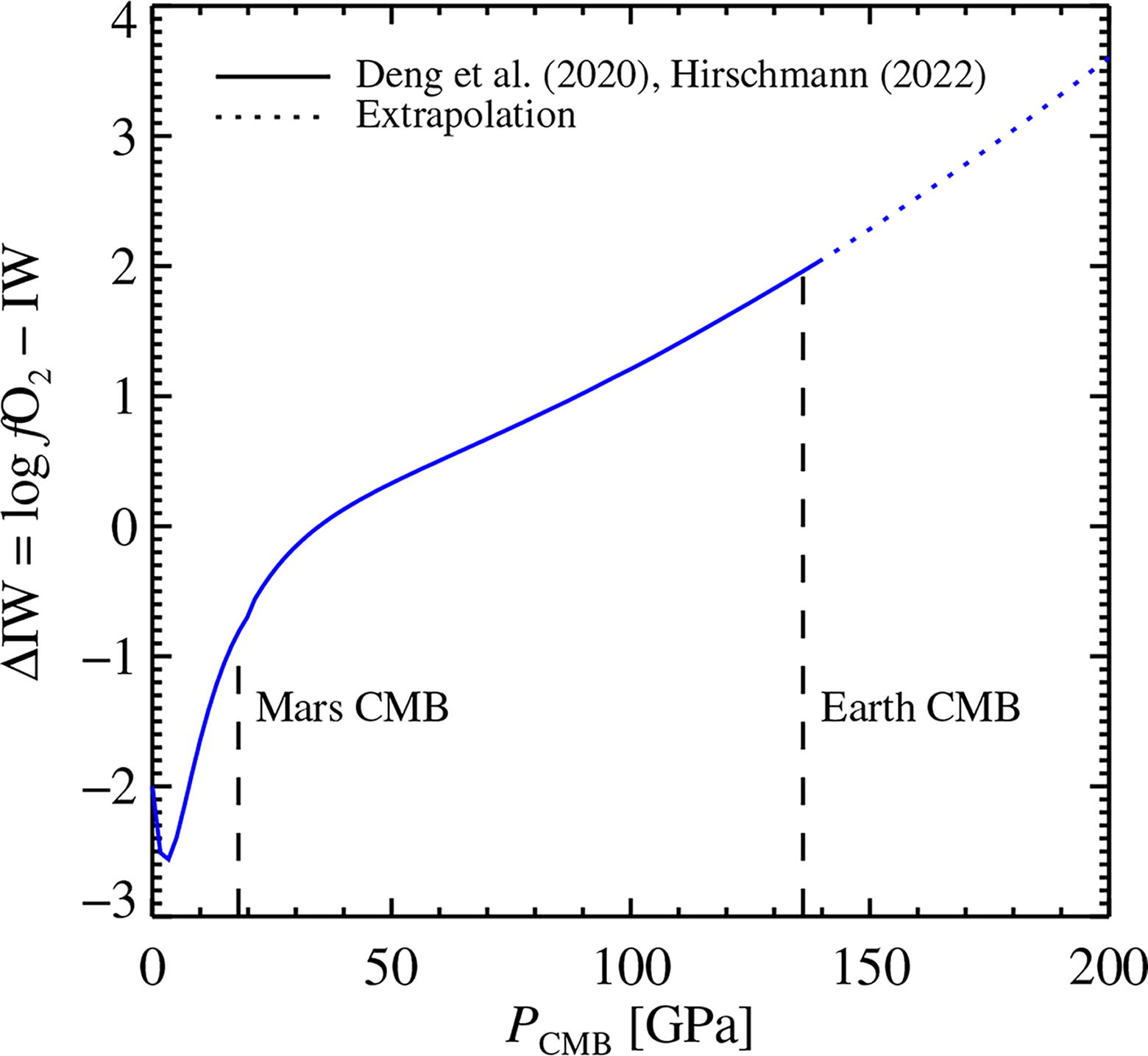
The oxygen fugacity, relative to the iron-wüstite buffer (IW), at the surface of the magma ocean after the termination of accretion, as a function of the pressure at the base of the magma ocean. We make the calculation here following the approach of Deng et al. (2020) and Hirschmann (2022); the calculation was done for the composition of bulk silicate Earth. The equation of state is valid up to 140 GPa (Deng et al., 2020), and we indicate extrapolation to higher pressures with dots. The relevant pressures at the core-mantle boundary (CMB) of Mars and Earth are shown in dashed lines.
Extrapolating these results to rocky exoplanets with generalized masses ranging from below Mars mass to super-Earth masses, the results in Figure 4 predict that smaller planets should have low mantle oxygen fugacities that correspond well to their FeO (Fe2+) fraction, see Equation (16), with no increase of the FeO1.5 (Fe3+) fraction at the core-mantle boundary. Planets as massiveor more massive than Earth, on the other hand, are predicted to have strongly oxidizing mantles. This extrapolation nevertheless depends on the FeO fraction of the mantle material, since the oxygen fugacity in Figure 4 is anchored at the value of Equation (16) at the core-mantle boundary. In the work of Johansen et al. (2023a), the authors demonstrated that the FeO fraction in the pebble accretion model for rocky planet formation will be a decreasing function of the mass. This is due to the accretion of pebbles that contain metallic iron with an intrinsically low oxidation degree. If, on the other hand, rocky planets form by accretion of planetesimals that experienced extensive iron oxidation by aqueous alteration (Rosenberg et al., 2001; Zolensky et al., 1989; Zolensky et al., 2008), then the FeO fraction of a rocky planet could be significantly higher. Also, the reaction between FeO and H2 sourced from the gas envelope accreted from the protoplanetary disc could change the Fe—FeO—H2—H2O distribution significantly on both planetary embryos and fully formed planets with magma oceans (Ikoma and Genda, 2006; Kite et al., 2020; Young et al., 2023). However, even if rocky exoplanets have a factor three lower or higher FeO fraction in their mantle compared to Earth, then this would yield at most a change of logarithmic core-mantle boundary oxidation state by 1 (Equation 16), which would reflect in a surface oxygen fugacity displaced by at most ±1 in Figure 4. Changing the oxygen fugacity of an Earth-mass planet from ΔIW = 2 to ΔIW = 1, by lowering its FeO mantle fraction, would only change R/O slightly from 1.1 to 1.3 (Fig. 4); these are still oxidizing conditions.
Atmosphere loss and self-oxidation
Here, we evolve the number of H, C, N, and O atoms in the atmosphere through XUV mass loss. The atmosphere model is assumed to instantaneously shift its chemical equilibrium to the prevalent atomic composition during the mass loss. We describe the XUV mass loss rates model in detail in Appendix 1. Importantly, we assume that the mixing ratio of H2O above the photosphere is equal to its value at the photosphere (where the atmosphere temperature equals the skin temperature), due to cold-trapping by cloud formation. We also apply a limit to the H escape either by scaling the mass-loss efficiency with the mixing ratio of H in the thermosphere or by imposing that the H flux cannot be higher than the diffusion rate from the lower atmosphere. For simplicity, we fix the temperature at 500 K during the mass loss. We include now in the models without CH4 and NH3 the condensation of H2O vapor to form oceans. Ocean condensation is, nevertheless, still not included in the FastChem calculations that incorporate those and other additional species. Since water vapor experiences strong cold-trapping in our models, the condensation of oceans does not significantly impact atmospheric mass loss rates.
We input the evolution of the X-ray and EUV luminosity of young stars of different initial rotation speeds from precalculated tables provided by Johnstone et al. (2021). The initial rotation speed is, in turn, quantified by the position of the star in the relative distribution function of rotation speeds. The temporal decline of the X-ray luminosity and the EUV luminosity of a solar-mass star is shown in Figure 5 for initial rotation rates of 10%, 50%, and 90% along the normalized cumulative distribution. Rocky planets that orbit in the habitable zone are exposed to a very high XUV flux during the first billion years of stellar rotation slow-down, with substantial differences between the luminosity curves in the 10–1000 Myr epoch.
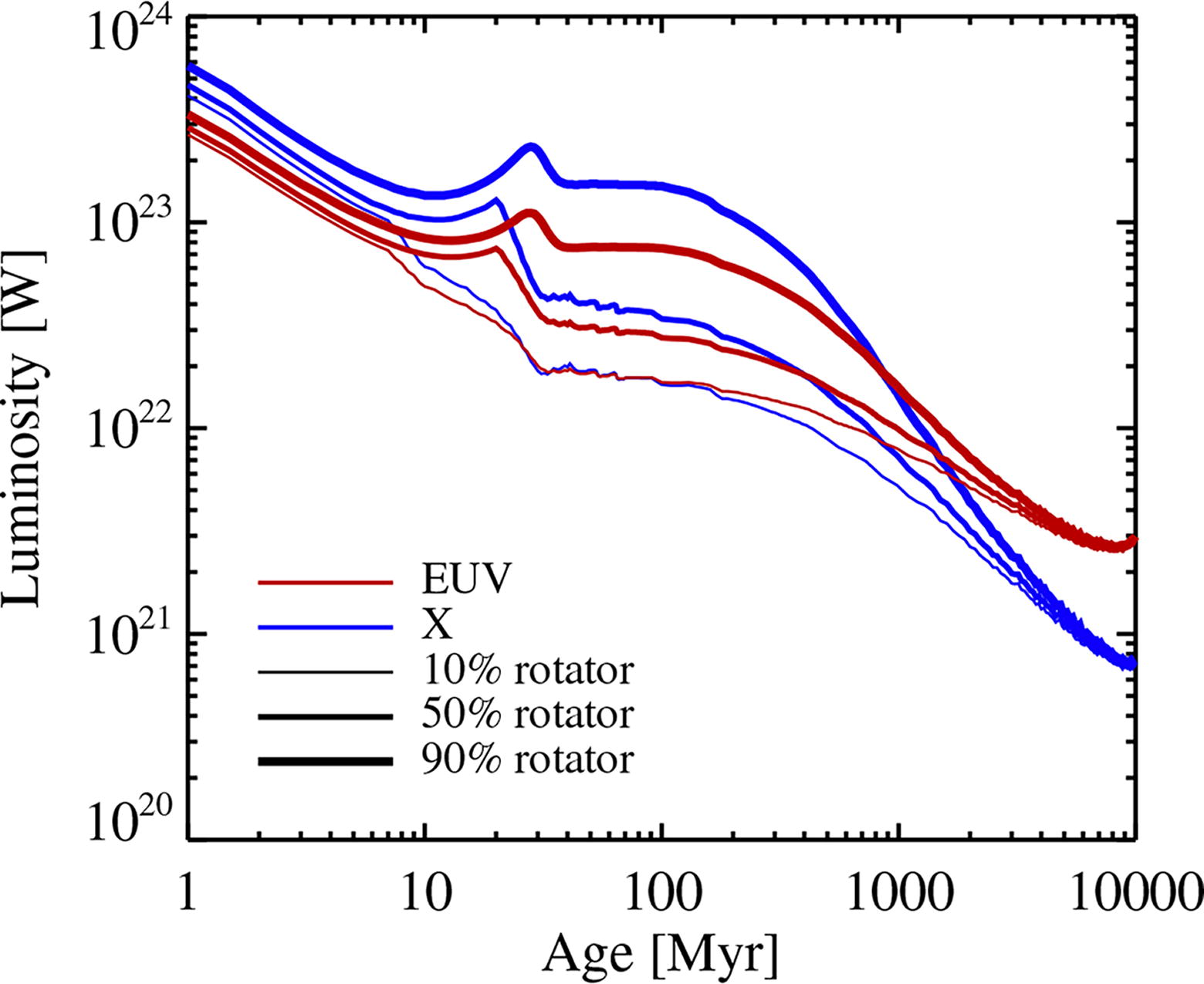
The evolution of the X-ray luminosity and the EUV luminosity with age for a solar-mass star, from the tables provided by Johnstone et al. (2021). We show results for stars with positions of 10%, 50%, and 90% along the normalized initial distribution of rotation speeds. The X-ray luminosity drops by a factor of approximately 400–600 between 1 and 5000 Myr of stellar age, while the EUV luminosity drops by a factor of approximately 100.
We expose atmospheres with three initial values of R/O (=4.0, 3.0, 2.0), and Earth/Venus-like C/H and N/H values, to XUV irradiation for up to 40 Myr after the formation of the star. We assume that the protoplanetary disc has been accreted and photoevaporated to oblivion after 5 Myr and that the atmosphere thereafter quickly cools down to an equilibrium temperature of 500 K, which we assume for simplicity to remain constant even when the atmospheric pressure and composition change with time. We evolve the number of H, C, N, and O atoms as a function of time by integrating Equations (20) and (21) for the 10% and 50% rotator, noting that the 90% rotator gives even more efficient mass loss. Figure 6 shows the evolution of R/O and C/H with time, both for the model without CH4 and NH3 (top and middle panels) and for the model that includes those molecules (bottom panel). The composition quickly drops toward R/O = 1 for both models. This evolution takes only 10–20 Myr for the top panel where we do not include a diffusion limit to the H escape flux. In the middle and bottom panels, where the diffusion limit is applied to the model without and with CH4 and NH3, the self-oxidation timescale is 20–30 Myr and 25–40 Myr, respectively, depending on the initial value of R/O. Including the diffusion limit, mass loss rates are independent of the initial rotation speed because diffusion from the lower atmosphere sets the maximal mass loss rate in that case.
The C/H ratio displayed on the right panels of Figure 6 drops only in the top panel for the 50% rotator without the diffusion limit, due to the high H fluxes that can, in that case, efficiently drag out C (plus N and O) with little fractionation. This leads to a decreased C/H ratio because the bulk part of the H2O component is cold-trapped and does not participate in the atmosphere escape. In the models with the diffusion limit, C/H increases by a factor of 2–5 due to extensive fractionating in the mass loss flux. Some representative self-oxidation tracks based on Figure 6 are also indicated in Figure 1.
In Figure 7, we show the evolution of the atmospheric species in the 50% rotator model that starts at R/O = 2 and includes the diffusion limit, for models with and without CH4 and NH3. For the model without these molecules (top panel), the dominant reducing molecule is H2, with CO playing only a minor role due to the water-gas shift reaction. Both H2 and CO fall to trace levels in ∼30 Myr of mass loss evolution. The first oceans condense out after 22 Myr when the atmospheric pressure has fallen to a low enough value that water vapor can no longer be present at its equilibrium pressure for the nominal mixing ratio. CO2 and N2 masses fall by nearly a factor of two, as these species are dragged along with the H outflow. The complete loss of H2 nevertheless leads to an increase in the bulk C/H ratio (see middle right panel of Fig. 6). The model that includes CH4 and NH3 (calculated using FastChem) loses its free H2 budget very rapidly. The subsequent oxidation of the atmosphere happens by loss of H donated by CH4 destruction in the thermosphere. The excess C is oxidized in equilibrium to CO2 by reaction with water vapor.
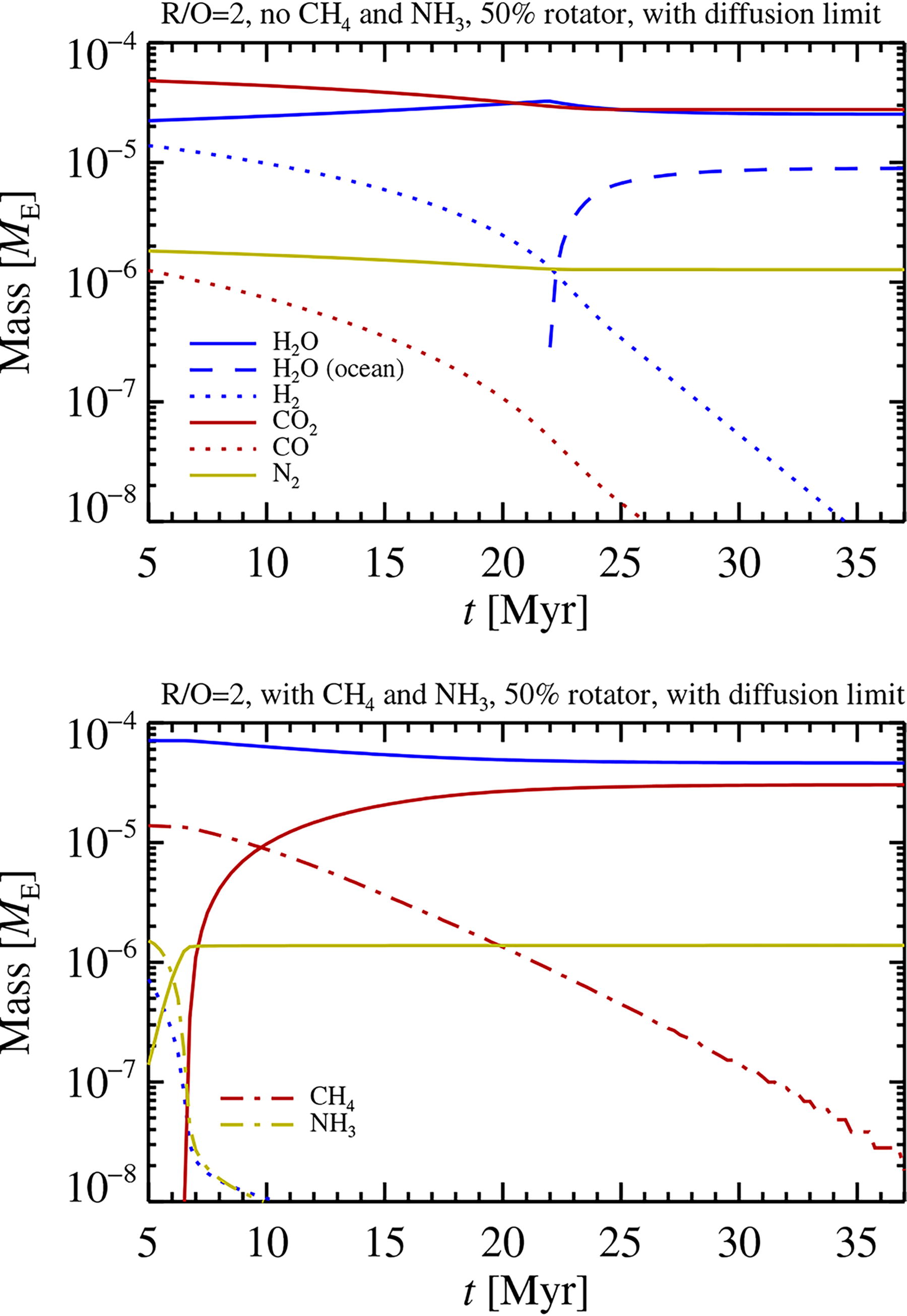
The evolution of the molecular species in the middle model and bottom models of Figure 6 starting at R/O = 2. In the top panel (no CH4 and NH3), the XUV irradiation leads to escape of the reducing H2 and CO molecules within approximately 30 Myr. CO2 and N2 experience some mass loss too as these molecules are dragged out together with the escaping H. The first oceans condense out after 22 Myr when the pressure of the atmosphere has dropped enough that water vapor can no longer be sustained at its equilibrium pressure. In the bottom panel (where we use FastChem to include a wider range of molecules), the initial small amount of H2 escapes quickly, and subsequent oxidation of the atmosphere is driven by loss of H donated by CH4 destruction. The surplus C is oxidized to CO2 by reaction with water vapor, while the amount of CO formed in equilibrium is insignificant (at the 550–600 K surface temperature range of these reducing conditions, see Fig. 2).
Overall, the mass loss calculations presented in Figure 6 show that atmospheres undergo a rapid self-oxidation process within a few 10 Myr. This conclusion holds relatively irrespectively of the chemical equilibrium model, variations to the mass loss calculations, and variations in the initial oxidation state of the outgassed atmosphere. We further test the robustness of mass loss in Figure A3 of Appendix 1, where we vary the efficiency of the energy-limited mass loss and perform an additional calculation where molecules such as CO2 are assumed indestructible and, hence, harder to lift by the H flux.
Outlook on self-oxidation calculations
The atmospheric self-oxidation calculations presented here employ an end-member approach to modeling thermospheric escape, by assuming either (a) complete atomization of all molecules in the thermosphere (in this section) or (b) destruction of only H2 (see Appendix 1 where we explore the latter approach to the model without CH4 and NH3). The goals of our paper bear some similarities to the work of Zahnle et al. (2020) who considered the evolution of impact-generated atmospheres using chemical equilibrium combined with quenching temperatures and a kinetic photolysis model. A direct comparison to that work is nevertheless difficult due to the very different starting points: we begin with an atmosphere outgassed from the magma ocean, while Zahnle et al. (2020) considered the reducing effect of an iron-rich impactor on an existing atmosphere. The short cooling timescale following an impact, of order ∼103 years, importantly motivated Zahnle et al. (2020) to consider a freeze-out (quench) temperature of 800 K for the full “H2–H2O–CH4–CO–CO2” system, while we demonstrate in Figure 2 that the reducing models have equilibrium surface temperatures in the range 550–600 K where the water-gas shift reaction timescale is much shorter than the cooling time of the atmosphere. At these temperatures, we expect CO to be rapidly converted to CO2 by reaction with H2O. The reduction power of the atmosphere is thus transferred in our model almost entirely to H2, which escapes most easily, while the post-impact models of Zahnle et al. (2020) exhibit significant amounts of remnant CO after the H2 component of the atmosphere has escaped.
We demonstrated in this section the efficiency of the self-oxidation process for an Earth-mass planet, but we note that rocky planets of Earth-mass and higher will undergo self-oxidation already at the magma ocean stage, as shown in Figure 4, and hence outgas strongly oxidizing atmospheres. Smaller planets undergo less magma ocean oxidation but have higher mass loss efficiencies and lower gravities (Salz et al., 2016). A Mars-mass planet in an Earth-like orbit will, therefore, undergo atmospheric self-oxidation even more rapidly than our nominal Earth-mass planet. We show in the next section that self-oxidation has major implications for the origin of life scenarios.
Fixation of Nitrogen by Lightning
In this section, we explore lightning as a pathway to fixing both nitrogen and carbon. By “fixation,” we refer to any chemical process that transfers C or N away from the strongly bound CO2 and N2 molecules into more reactive forms. Kinetic barriers are of great importance in allowing disequilibrium levels of molecules that contain fixed N and C to persist in the atmosphere at low temperatures. At elevated temperatures, the equilibrium host of N shifts to include significant amounts of NO/NO2 in an oxidizing atmosphere and to HCN/HNC in a reducing atmosphere. We will discuss the relevance of these fixed nitrogen host molecules for prebiotic chemistry in the following section.
Molecular freeze-out temperatures
Shifting the atmosphere locally to very high temperatures by lightning, the equilibrium speciation of molecules (and atoms) will change. The “memory” of this high-temperature equilibrium depends in turn on the approximate freezeout temperature, below which the reaction rates back to the low-temperature equilibrium proceed too slowly to matter on Gyr timescales. We discuss here the freeze-out temperatures of NO/NO2, HCN/HNC, CH4, and NH3.
NO/NO 2. The NO abundance peaks at a temperature of around 4000 K and freezes out between 2000 K and 2250 K (Mancinelli and McKay, 1988). In Earth’s strongly oxidizing atmosphere, O2 is the main donor of oxygen to create NO from the reaction between N and O (Hill et al., 1980). In atmospheres devoid of O2, O comes mainly from H2O and CO2 (Chameides and Walker, 1981; Kasting and Walker, 1981; Yung and McElroy, 1979), forming NO through the reactions O + N2 → NO + N and N + CO2 → NO + CO (Navarro-González et al., 2001) and NO2 by subsequent oxidation of NO (Kasting and Walker, 1981). Lightning in Earth’s modern atmosphere produces approximately 109 kg of fixed nitrogen per year (Navarro-González et al., 2001). This gives an atmospheric depletion timescale of approximately 6 Gyr; the main return of fixed nitrogen back to N2 is through bacterial denitrification that harnesses energy from lowering the energy level of nitrogen back to strongly bound N2 (Mancinelli and McKay, 1988).
HCN/HNC. In reducing atmospheres, HCN is produced in large quantities by lightning heating. The lightning energy destroys N2 and CH4 molecules to form the radicals N and CH3 that readily combine with HCN (Pearce et al., 2022). Chameides and Walker (1981) reported an HCN freeze-out temperature of 2000–2500 K. This contrasts with Jupiter models that find that the HCN level freezes out at a temperature below approximately 870 K (Moses et al., 2010). Borucki et al. (1988) reported an HCN freeze-out of approximately 950 K from lightning experiments and found much higher HCN yields for their Titan atmosphere models than for Jupiter, due to the destruction of HCN by H2 under Jupiter conditions (Moses et al., 2010). The contrast between the high freezeout temperature of Chameides and Walker (1981) and the lower value favored by Borucki et al. (1988) may be due to different assumptions about the strength of lightning; indeed Chameides and Walker (1981) also reported an elevated freezeout temperature of NO at 3000–3500 K. We, therefore, take here the freeze-out temperature of HCN to lie in the interval 1000–2000 K.
CH 4. Methane forms by consecutive reactions of CO and its products with H2 (Zahnle and Marley, 2014). Methane is the energetically favorable C-host at low temperatures in reducing atmospheres. The reaction CH3OH + H2 ⇌ CH4+H2O nevertheless appears as a bottleneck for CH4 production; this reaction becomes kinetically inhibited below a freeze-out temperature in the range of 900–1,000K (Sossi et al., 2020; Moses et al., 2011; Zahnle and Marley, 2014).
NH 3. Ammonia forms by consecutive reactions of N2 and its products with H2. NH3 is the dominant carrier of N in reducing conditions. The equilibrium between NH3 and N2 freezes out at a temperature of 1600–1700 K, where the NH3 ratio is relatively low (Moses et al., 2011).
Heating in lightning
Lightning is the result of charge separation in water clouds (Christian et al., 2003; Yair, 2012). The low conductivity of planetary atmospheres allows the electric field to build up to its breakdown strength, where the electrons accelerated by the field are fast enough to cause an ionization cascade. The rapid release of electrons and increase in conductivity lead to charge neutralization by lightning discharge in narrow discharge channels. The lightning channel reaches a temperature up to between 20,000 K and 30,000 K within a few microseconds.
The channel subsequently cools by mixing of cold air with the surroundings (Hill et al., 1980). At high temperatures, the chemical reactions are extremely rapid, and, hence, chemical equilibrium is maintained at the instantaneous temperature. As demonstrated above, many chemical reactions nevertheless have freeze-out temperatures that are significantly above the ambient temperature of the passively irradiated atmosphere.
Lightning yield tests
The yield (or production rate) of a given molecule i (in our case, NO/NO2, HCN/HNC, CH4, and NH3) can be quantified as the number of molecules ΔNi
formed per input energy ΔE (Mancinelli and McKay, 1988). With a molecular freeze-out temperature of T
f, the yield in an atmosphere of equilibrium temperature T
0 and mass M
atm is defined as:
We use the FastChem code to calculate the equilibrium speciation of molecules in the gas phase as a function of temperature, pressure, and elemental abundances (Stock et al., 2018; Stock et al., 2022). We test our numerical calculation of chemical equilibrium by measuring the HCN yields produced by FastChem for Jupiter and Titan analogs for a range of temperatures. For Jupiter, we approximate the composition of the atmosphere as solar but with the abundances of H and He reduced by a factor of three (Guillot, 2005). We consider a pressure level of 5 bar to represent the depth of Jupiter’s water cloud layer (Yair, 2012). For Titan, we take an atmosphere that consists of 95% N2 and 5% CH4 and a pressure of 1.5 bar (Hörst, 2017). We show the HCN concentration as a function of temperature in Figure 8. As expected, the Jupiter model produces a far lower HCN concentration than Titan. This is partially due to the low abundance of N relative to H in this atmosphere of nearly solar composition, but more importantly we see the effect of HCN destruction by reaction with ambient H2 (Moses et al., 2010). The right axis of Figure 8 shows the yield of HCN in the Titan atmosphere. A yield of slightly above 1017 molecules per joule of input energy agrees well with the calculation presented for Titan in the work of Borucki et al. (1988).
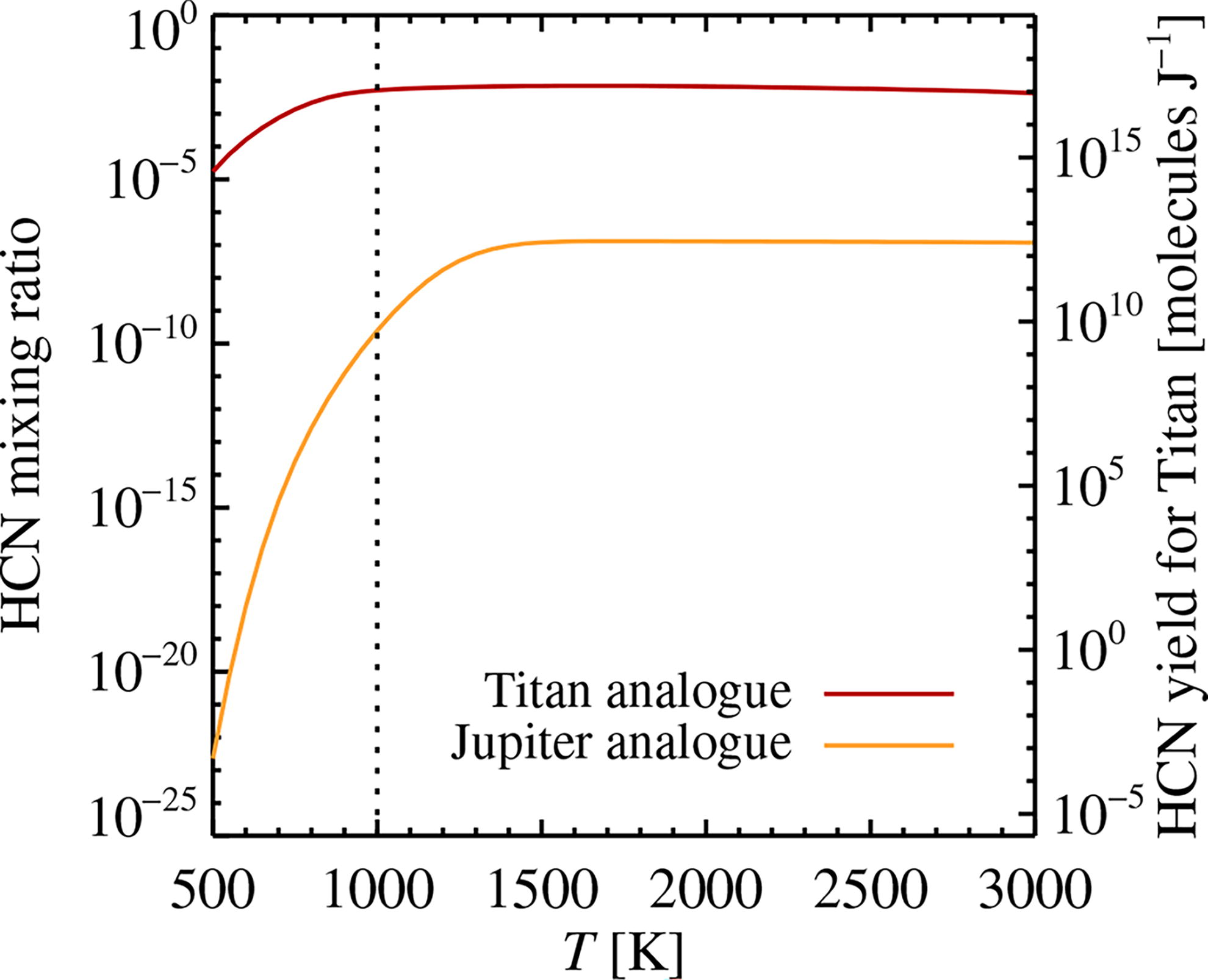
Test of HCN concentration obtained with FastChem for a Jupiter analog, where H and He abundances are reduced by a factor of three in abundance relative to the solar composition, and a Titan analog consisting of 95% N2 and 5% CH4. The Jupiter analog atmosphere gives a far lower HCN concentration than Titan; this is mainly due to the destruction of HCN by reaction with H2 in this nearly solar composition environment. The molecular yields of HCN in the Titan analog are indicated on the right axis and agree well with the work of Borucki et al. (1988).
Lightning on rocky planets
We now turn to modeling lightning in the outgassed atmospheres of young rocky planets. We fix the pressure at P = 100 bar and test temperatures of 1000 K, 2000 K, and 2300 K. We run simulations for a range of R/O values, but with C/H fixed at 0.07 and N/H fixed at 0.008 (see discussion in section “Atmosphere equilibrium model”). In Figure 9, we show the resulting concentrations of NO and NO2, HCN and HNC, CH4 and NH3 as a function of R/O and for three different values of the temperature. The value of R/O is clearly a key determinant for the yield of fixed N and C carriers at elevated temperatures. For strongly oxidizing atmospheres with R/O ≤ 1, mainly NO/NO2 is formed, accompanied by a tiny fraction of NH3 (at the 10−12 level). For R/O > 1, NO/NO2 levels drop slowly, while the levels of HCN/HNC, CH4, and NH3 increase. CH4 becomes the dominant carrier of carbon at its freeze-out temperature of 1000 K, while NH3 dominates as carrier of fixed nitrogen for R/O > 1. The relatively low concentration of HCN found in these models (at the 1–1000 ppm level) is due to the presence of large amounts of H2 for R/O > 1, which is similar to what we observed for the Jupiter model in Figure 8. We have experimented with larger values of C/H and observed an increased HCN production at the expense of a decrease in NH3.
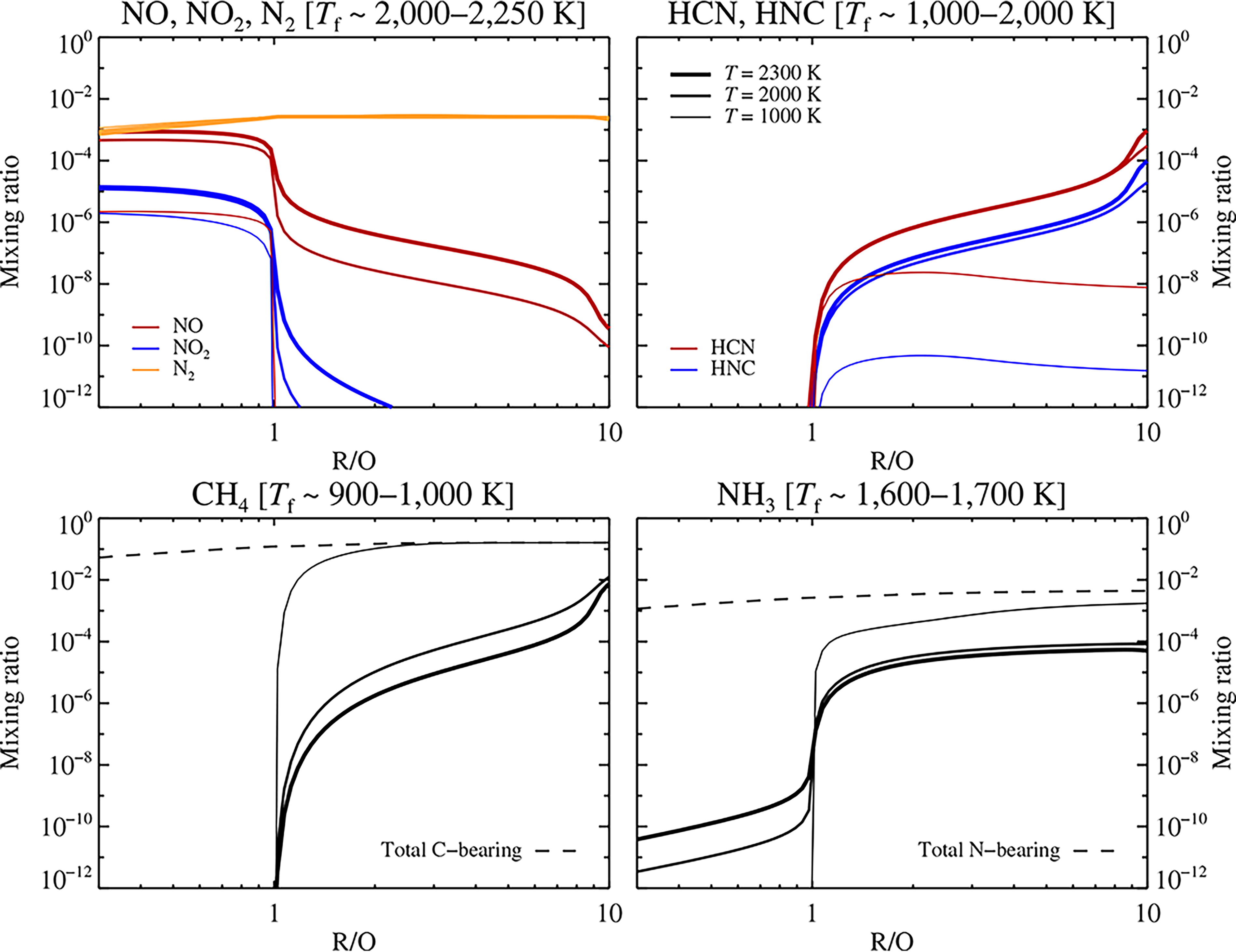
Mixing ratio of the C and N host molecules NO, NO2, and N2 (top left); HCN and HNC (top right); CH4 (bottom left); and NH3 (bottom right) for C/H = 0.07 (the approximate value for bulk silicate Earth), as a function of R/O and at three different temperatures. The approximate freeze-out temperatures of the chemical kinetics are indicated in the plot titles. The production of NO and NO2 molecules drops slowly when transitioning from oxidizing atmospheres with R/O ≤ 1 to reducing atmospheres with R/O > 1. With increasing R/O above unity, HCN and HNC concentrations in turn rise slowly. CH4 becomes the main carrier of carbon at its freeze-out temperature for R/O > 1, while NH3 dominates over HCN and HNC as the main carrier of fixed nitrogen for all R/O > 1.
Discussion
NO yields in the early atmosphere
The mixing ratios in Figure 9 were calculated at a fixed total pressure of P = 100 bar. We present now an additional set of simulations performed with FastChem where we vary the partial pressure of CO2, while keeping the partial pressure of nitrogen fixed at P N2 = 0.8 bar and the partial pressure of water vapor fixed at either P H2O = 1 bar or P H2O = 0.01 bar. These represent the equilibrium vapor pressure in atmospheres at temperature level 100°C and a more temperate 20°C, respectively. In Figure 10 we show the mixing ratio as well as the partial pressure of NO at a temperature of T = 2000 K. The mixing ratio increases significantly with lower CO2 pressure, mostly driven by the decrease in the total number of molecules when decreasing the pressure, reaching a plateau or a slight decline below P CO2 = 1 bar, below which H2O and N2 dominate the pressure. The partial pressure of NO, which represents the total NO mass, drops by 1–2 orders of magnitude over a CO2 pressure drop of four orders of magnitude, as the oxygen source for NO switches from CO2 to H2O.
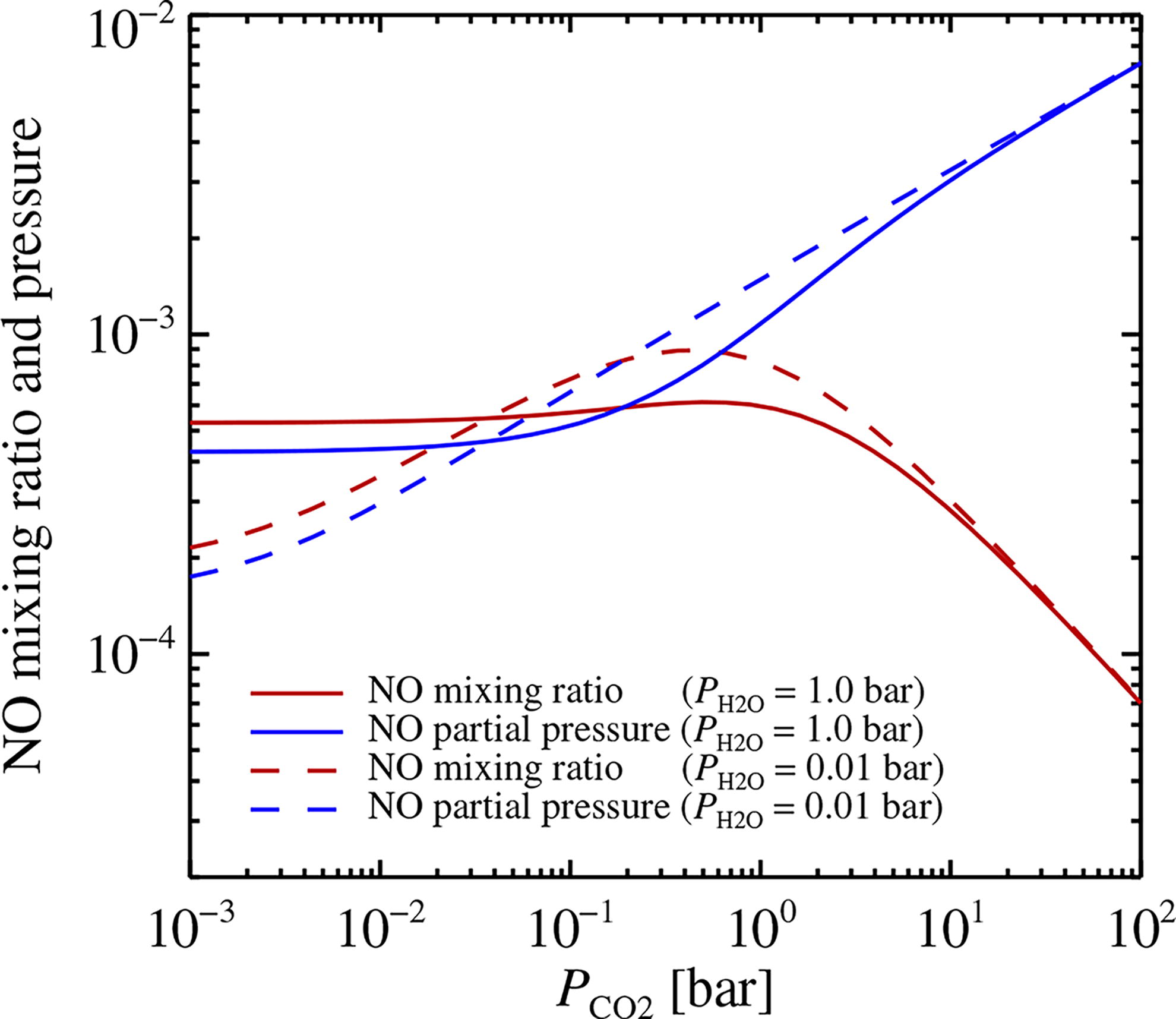
The NO mixing ratio and partial pressure for atmosphere models with a range of CO2 partial pressures (x-axis), a fixed partial pressure of N2 (0.8 bar), and two pressure values of H2O (1 bar and 0.01 bar). The temperature is considered here to be T = 2000 K, representing the freeze-out temperature of NO. The NO mixing ratio increases significantly as the CO2 partial pressure is decreased. Below 1 bar CO2 pressure, the mixing ratio reaches a plateau for P H2O = 1 bar and even falls for P H2O = 0.01 bar, as lightning energy is then spent on heating H2O and N2 rather than CO2. The partial pressure falls considerably over the considered interval of CO2 pressure, mainly due to the decrease in the total oxygen budget carried by CO2 for NO production.
We use now the yield Equation (17), together with an NO concentration from Figure 10 of C NO∼7 × 10−5 at 100 bar CO2 pressure, to estimate an NO yield of Y NO∼4 × 1014 J−1 (NO molecules produced per Joule) in an early atmosphere dominated by 100 bar of CO2. The yield depends only on the mixing ratio and, thus, increases to Y NO∼3 × 1015 J−1 for lower CO2 partial pressure. This estimate agrees well with the plateau at high CO2 mixing ratios found in lightning laboratory experiments by Navarro-González et al. (2001), who considered a CO2-N2 mixture at a fixed pressure level of 1 bar.
The rate of lightning strikes on modern Earth has been measured at a level of 44 s−1 (Christian et al., 2003). With a mean energy of ∼109 J per lightning strike (Chyba and Sagan, 1991), this results in an energy dissipation rate of 4 × 1010 W or approximately 1018 J yr−1. This lightning energy is ultimately derived from the energy carried through the atmosphere by convection (Borucki et al., 1982); hence, we can assume that the lightning energy dissipation rate on young Earth was not substantially different from the modern value (and, hence, was to first order, not affected by effects such as Earth’s higher primordial rotation frequency). This leads to an estimated NO production of ∼1026 s−1 under early Earth conditions. At this rate, it would have taken ∼70 Gyr to process the entire N2 budget present in Earth’s modern atmosphere into NO. However, as discussed above, the dense CO2 atmosphere is not expected to have persisted for more than a few 100 Myr. The drop of the CO2 partial pressure to less than 0.1 bar is inferred to have been reached by the beginning of the Archean eon 4 Gyr ago (Catling and Zahnle, 2020), together with a drop in the partial pressure of H2O vapor at milder surface temperatures would have lowered the lightning yields substantially and perhaps triggered the evolution of bacterial nitrogen fixation (Navarro-González et al., 2001).
Delivery of fixed nitrogen by interplanetary dust particles
Interplanetary dust particles (IDPs) recovered on Earth exhibit typical concentrations of C N∼10–100 ppm in organic nitrogen (Marty et al., 2005). These values lie within the range of the N concentration in ordinary chondrites and enstatite chondrites (Grewal et al., 2019). Atmospheric entry heating of organics to more than 600°C–800°C nevertheless leads to alteration and loss of organics (Riebe et al., 2020); only IDPs in the mass range 10−15–10−9 kg maintain their organics against UV photolysis (lower range) and silicate evaporation (upper range) (Anders, 1989; Chyba and Sagan, 1992). Terrestrial weathering could also have contributed significantly to nitrogen loss. Hence, the pre-atmospheric-entry nitrogen concentration level may have been closer to 1,000 ppm (Hashizume et al., 2000; Marty et al., 2005), with some grains of cometary origin reaching even higher levels of nitrogen concentration (Aléon et al., 2003).
Pyrolysis (i.e., thermal decomposition) of complex organic matter during atmospheric entry transfers the nitrogen to simple compounds such as NH3 (Nakano et al., 2003) and HCN (Okumura and Mimura, 2011). Hence, IDPs delivered both intact organics and simple N-carrying compounds to the young Earth. Both could have served as a source of fixed nitrogen needed by an emerging biosphere that exploited geological serpentinization reactions as a source of H2 to fix C from atmospheric CO2. NH3 is nevertheless photolyzed at even longer wavelengths than CH4 and transforms to N2 in a CO2-rich atmosphere (Kuhn and Atreya, 1979; Kasting, 1982) on a timescale that is much shorter than the CH4 photolysis timescale from Kasting (2014) repeated here in Equation (12).
The current flux of IDPs at the top of the atmosphere is estimated at F
IDP = 1.5 − 4.5 × 107 kg yr−1 (Peucker-Ehrenbrink and Ravizza, 2000). That gives a nitrogen delivery rate from IDPs of
This is approximately three orders of magnitude lower than the internal nitrogen fixation on early Earth calculated in Section 4.1. Chyba and Sagan (1992) discussed evidence that the IDP delivery rate has been relatively constant in time back to at least 3.6 Gyr ago, based on analysis of IDPs from lunar soils. There is currently no known direct way to infer the IDP flux further back in time, but Chyba and Sagan (1992) nevertheless estimated that the IDP flux could have been 3–4 orders of magnitude higher at the earliest epochs of the history of Earth. We, therefore, suggest that delivery of fixed nitrogen by IDPs was likely comparable in rate to fixation by lightning on Hadean Earth.
In Appendix 2, we calculate the delivery rate of fixed nitrogen from asteroid bombardment. As with interplanetary dust particles, the asteroid impact rate during the early Hadean is very uncertain, but it was likely orders of magnitude higher than today. Asteroid impacts are nevertheless prone to conversion of the fixed N present in their organics into N2 in the immense impact heat (as demonstrated in Fig. 9), particularly if the impactor material mixes with the oxidizing atmosphere during the cooling.
Relevance of our results for origin-of-life scenarios
Life as we know it requires plenty of fixed nitrogen (often incorporated in biomolecules as a −NH3 amino group) since N is an important constituent part of central biomolecules such as proteins, nucleic acids, and many cofactors. Because of this, all different proposed pathways to life’s emergence include nitrogen as one of the focal points. For instance, there are two main known prebiotic pathways for the synthesis of amino acids. The first one relies on the chemistry of nitriles and uses HCN in a reaction mechanism known as Strecker synthesis where amino acids are produced (Ruiz-Bermejo et al., 2013). This mechanism, which is chemically unrelated to that used by extant biology, is thought to be responsible for at least a portion of amino acids found in extraterrestrial environments such as asteroids and comets (Giese et al., 2022). The alternative prebiotic mechanism, which is chemically analogous to how cells synthesize amino acids, occurs through reductive amination or transamination of simple ketoacids (Harrison et al., 2023; Mayer et al., 2021). The prebiotic synthesis of nucleotides has been historically dominated by HCN-related approaches (e.g., Powner et al., 2009; Teichert et al., 2019). However, there have recently been advances in the prebiotic synthesis of nucleobases when using biologically relevant simple precursors such as carbamoyl phosphate (Yi et al., 2022), which are HCN-independent and, thus, fully compatible with a CO2-dominated atmosphere.
Our results are relevant for the state-of-the-art in the origin of life studies mainly by pointing out the most abundant forms of N and C that were available for prebiotic chemistry: oxidized NOx species dissolved in the oceans (producing NH3 after interaction with marine hydrothermal environments locally rich in H2) as well as CO2. In contrast, the synthesis of HCN requires an atmosphere that is rich in already-fixed carbon (e.g., CH4) or free hydrogen (H2), none of which are abundant according to our model. Hence, we propose that the prebiotic chemistry of N that was globally available on the Hadean Earth would have relied on reductive amination of small organics (themselves probably derived from CO2 reduction by H2, see Beyazay et al., 2023; Hudson et al., 2020) by NH3. These geochemical mechanisms, which echo what we see in modern biology, aid in closing the gap between early geochemistry and ancient biochemistry.
The dichotomy between a submarine and surficial origin of life has been a main topic of discussion during the past few decades, with HCN-dominated chemistry usually invoked for surficial environments, and CO2/NOx-dominated chemistry for submarine ones. This environmental division nevertheless does not necessarily correspond exactly to the two distinct chemical pathways, both of which are technically possible in each scenario (e.g., reactivity of HCN has also been explored at alkaline hydrothermal vents, see Villafane-Barajas et al., 2021). Hence, our results, which show a primitive CO2-dominated atmosphere, do not specifically preclude either geographic locale. Instead, the early dominance and availability of CO2 demonstrated here is suggestive of a prebiotic chemistry based on carbon and nitrogen fixation in a way reminiscent of ancient biology (CO2- and NH3-based). It is worth noting that HCN-based prebiotic chemistry would still be plausible in the Hadean Earth after large impactors (Itcovitz et al., 2022); however, whether these transient reducing conditions prevailed for long enough for a putative HCN-based life to emerge remains an open question.
Implications for exoplanets
Atmospheres of rocky planets become oxidizing both from magma ocean processes that increase the Fe3+ fraction in the mantle (Armstrong et al., 2019; Frost et al., 2004) and from XUV mass loss that efficiently removes any reducing H that is not bound in cold-trapped H2O (this work). From that perspective, rocky planet atmospheres should always obtain an oxidation state of R/O ≈ 1. Rocky planets close to their host star may subsequently undergo additional destruction and loss of H2O during a later run-away greenhouse process (Turbet et al., 2021; Way et al., 2016), potentially leading to abiotic pile-up of O2 (Luger and Barnes, 2015) and thus Earth-like oxidation conditions with R/O < 1.
We worked in the present study with Earth/Venus-like C/H and N/H ratios in the atmospheres and an Earth-like mantle FeO fraction (8%), but we expect the results to be relatively independent of these parameters unless the chemistry becomes C-dominated with C/O >1. Planets more massive than Earth can accrete a primary atmosphere of H2 and He on top of their outgassed atmosphere (Ikoma and Hori, 2012). We did not consider any interactions between the H2/H2O in such an envelope and the Fe/FeO contents of the magma ocean (Kimura and Ikoma, 2020; Kite and Schaefer, 2021; Schlichting and Young, 2022; Young et al., 2023). A realistic span of FeO mantle fractions from three times below Earth’s value to three times above will change the oxygen fugacity by one logarithmic unit up or down, but this would not change the picture that low-mass planets outgas reducing atmospheres and high-mass planets outgas oxidizing atmospheres, nor would it change our conclusion that reducing atmospheres undergo rapid self-oxidation by XUV-driven mass loss. We also fixed the initial atmosphere pressure at 100 bar. Higher atmosphere masses would be much harder to erode by XUV irradiation. Mini-Neptunes that maintain either a primordial gas envelope or an equally massive outgassed atmosphere, nevertheless, are unlikely to be habitable, to an Earth-like biosphere at least, since the blanketing effect of the envelope keeps the magma ocean from crystallizing (Benneke et al., 2019; Kite and Barnett, 2020; Kreidberg et al., 2022; Tsiaras et al., 2019).
We note that a possible way to override the primordial self-oxidation processes would be through late delivery of significant amounts of reduced material by asteroid impacts (Pearce et al., 2022; Wogan et al., 2023; Zahnle et al., 2020) after a few hundred million years, when the XUV luminosity of the young star has faded enough to slow down the H2 loss. The likelihood that such extensive bombardments occur in a planetary system is nevertheless hard to assess since it depends on the dynamics of one or more giant planets (Martin and Livio, 2021) as well as on the masses and locations of remnant planetesimals rich in unoxidized, metallic iron.
Conclusions
In the present study, we have considered molecular speciation in the atmospheres of rocky planets as they cool down after the magma ocean phase. We have particularly focused on the molecular host of nitrogen, since this has strong implications for the origin of life scenarios. Our main findings can be summarized as follows: The water-gas shift reaction in combination with loss of H from H2, CH4, and NH3 to XUV irradiation will transform any strongly or mildly reducing atmosphere outgassed from the magma ocean into a marginally oxidizing atmosphere consisting of H2O, CO2, and N2. Strongly reducing atmospheres convert most of their outgassed water reservoir to H2 (which is lost) and CO2 in this self-oxidation process. Self-oxidized atmospheres, as well as primordially oxidizing atmospheres, are not prone to HCN production by lightning. This finding contrasts with the view that HCN was a key prebiotic feedstock molecule (Miller and Urey, 1959). Instead, lightning in CO2-rich atmospheres produces copious amounts of NO. This NO will dissolve in the oceans and transform to nitrate that carries fixed nitrogen available as feedstock to an early biosphere. Our results, therefore, highlight serpentinization (producing H2 and metallic FeNi alloy that drive prebiotic carbon fixation) and lightning as key planetary processes that converted oxidizing atmospheric components to fixed carbon and nitrogen ripe for the abiogenesis process. As an alternative to the serpentinization scenario, C may be fixed from CO2 using metallic iron from meteorites and volcanic particles as catalysts (Peters et al., 2023). The external delivery of fixed nitrogen by interplanetary dust particles to an emerging biosphere was likely comparable to the lightning fixation, under reasonable assumptions about the early flux of such dust particles. Interplanetary dust particles nevertheless display a large range of nitrogen concentration, and the actual delivered amount would depend on their source region (inner or outer Solar System). Extrapolating to rocky planets around other stars, our proposed self-oxidation mechanism suggests that the atmospheres of cool rocky planets are likely dominated by CO2. The discovery of rocky exoplanets with CO-rich or H2-rich atmospheres would be surprising, given the efficiency of self-oxidation by hydrogen escape through XUV irradiation.
These conclusions are necessarily based on a number of model assumptions, which come with a varying degree of certainty. We chose to study two endmembers of the chemical model: one where the molecules CH4 and NH3 are assumed to be irreversibly destroyed by photolysis and another where those molecules are assumed to survive. We could then demonstrate that self-oxidation by loss of H is efficient under both these assumptions. Two salient limitations of our work are the assumption of chemical equilibrium and our simplified approach to the XUV irradiation of the thermosphere where, we assume, all molecules are atomized. Future studies should, therefore, include realistic photochemical reactions (Kopparapu et al., 2012; Line et al., 2011; Moses et al., 2022; Zahnle et al., 2020) both during cooling and during XUV-driven mass loss. The energy-limited approach to XUV mass loss comes with significant uncertainties in both the components included in the model as well as in the efficiency factor and the planetary cross-section for XUV absorption. We nevertheless showed that loss of H on a timescale of a few ten million years is a robust outcome of our calculations relatively irrespectively of the parameter choice for the XUV mass loss.
We additionally neglected the role of asteroid impacts in generating temporary reducing atmospheric conditions (Gaillard et al., 2022; Hashimoto et al., 2007; Itcovitz et al., 2022; Pearce et al., 2022; Schaefer and Fegley, 2017; Wogan et al., 2023; Zahnle et al., 2020). While such impacts can certainly occur, we would question an origin-of-life pathway that starts from HCN in a temporary reducing atmosphere and later completely changes its biochemistry to mimic chemical reactions in a serpentinization-related hydrothermal system, the proposed metabolism for LUCA (Weiss et al., 2016), after the loss of the reducing atmosphere and re-outgassing of an oxidizing atmosphere by volcanism. We nevertheless recognize that the origin of life is sufficiently complex that both planetary processes and external forcing (i.e., impacts of small and large objects) likely played significant roles.
Footnotes
Acknowledgments
The authors would like to thank the referees Edwin Kite and Kevin Zahnle for their many constructive comments on the original article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
A.J. acknowledges funding from the European Research Foundation (ERC Consolidator Grant 724687-PLANETESYS), the Knut and Alice Wallenberg Foundation (Wallenberg Scholar Grant 2019.0442), the Swedish Research Council (Project Grant 2018-04867), the Danish National Research Foundation (DNRF Chair Grant DNRF159), the Göran Gustafsson Foundation and the Carlsberg Foundation (Semper Ardens: Advance grant FIRSTATMO). E.C. thanks the University of Texas System for a STARs award. E.v.K. acknowledges support from the Villum Young Investigator Grant (project no. 53024). H.J.H. acknowledges partial financial support from The Fund of the Walter Gyllenberg Foundation.
Appendix 1
Appendix 2
Associate Editor: Christopher P. McKay
