Abstract
Ultraviolet (UV) light is likely to have played important roles in surficial origins of life scenarios, potentially as a productive source of energy and molecular activation, as a selective means to remove unwanted side products, or as a destructive mechanism resulting in loss of molecules/biomolecules over time. The transmission of UV light through prebiotic waters depends upon the chemical constituents of such waters, but constraints on this transmission are limited. Here, we experimentally measure the molar decadic extinction coefficients for a number of small molecules used in various prebiotic synthetic schemes. We find that many small feedstock molecules absorb most at short (∼200 nm) wavelengths, with decreasing UV absorption at longer wavelengths. For comparison, we also measured the nucleobase adenine and found that adenine absorbs significantly more than the simpler molecules often invoked in prebiotic synthesis. Our results enable the calculation of UV photon penetration under varying chemical scenarios and allow further constraints on plausibility and self-consistency of such scenarios. While the precise path that prebiotic chemistry took remains elusive, improved understanding of the UV environment in prebiotically plausible waters can help constrain both the chemistry and the environmental conditions that may allow such chemistry to occur.
Introduction
It is likely that surface environments on early Earth were subjected to significant amounts of UV light, given the lack of shielding from ozone and the list of probable species in the atmosphere (Farquhar et al., 2001; Pavlov and Kasting, 2002). UV light has often been invoked as a potential source of energy for driving prebiotic chemical synthesis (Sagan and Khare, 1971; Chyba and Sagan, 1992; Powner et al., 2009; Sarker et al., 2013; Patel et al., 2015; Green et al., 2021). However, UV light can also damage potentially important biomolecules, leading some to suggest that life originated in UV-shielded environments (Sagan, 1973). In addition to sources of feedstock molecules for synthesis, prebiotic chemistry also generally needs an energy source to drive reactions. Various sources of energy have been postulated, including lightning (e.g., Miller, 1953), mineral redox reactions (e.g., Wächtershäuser, 2008), and UV light (Chyba and Sagan, 1992) or other high-energy radiation (Pastorek et al., 2020).
The transmission of UV light through prebiotic atmospheres has been investigated, with light >204 nm likely present on Earth's surface (Ranjan and Sasselov, 2017). However, the transmission of UV light through realistic prebiotic waters is less constrained. Many estimates of the UV transmission in waters on early Earth are based on pure water or modern natural waters (Cockell, 2000; Ranjan and Sasselov, 2016; Pearce et al., 2017), neither of which are likely representative of the waters on early Earth. UV transmission has been considered for Martian waters where Fe(II) undergoes photooxidation to drive precipitation of oxidized iron species (Hurowitz et al., 2010; Nie et al., 2017). While UV light is transmitted nearly unattenuated through pure water at wavelengths >207 nm (Quickenden and Irvin, 1980), the presence of additional species in water may significantly increase extinction. In particular, as more complex molecules are synthesized prebiotically, it is possible that the UV transmission decreases significantly in the environments where prebiotic synthesis occurs, due to the increased UV absorption of species (Cleaves and Miller, 1998; Cockell and Knowland, 1999).
In previous work, we investigated the potential of simple geologically derived inorganic ions to attenuate UV transmission in prebiotic terrestrial waters. We found that diverse absorbers could have attenuated shortwave UV (e.g., halide salts; Br-, Cl-) but identified relatively few inorganic absorbers that could have attenuated broadband UV (Ranjan et al., 2022). However, the waters in which the chemistry that ultimately led to the origin of life occurred must also have included organic molecules, and organic molecules are often strong UV absorbers (Cleaves and Miller, 1998; Cockell and Knowland, 1999). In this study, we set out to investigate the UV transmission in waters containing organic feedstock molecules which have been suggested as sources in the synthesis of building blocks for the origins of life. We do not restrict ourselves to a single scenario or vision for prebiotic chemistry and the origins of life; rather, we identify key molecules that have been used in more than one independent prebiotic synthesis to provide a more robust and scenario-independent study.
Prebiotic chemistry has seen significant progress in recent years, now with many possible suggestions for the synthesis of the building blocks of life (e.g., Patel et al., 2015; Becker et al., 2016, 2019; Kim and Benner, 2017; Xu et al., 2018, 2019, 2020, 2021; Teichert et al., 2019). While the precise pathway for synthesis of prebiotic building blocks and the origins of life on Earth is not currently known, substantial clues from the planetary environment may indicate the various possibilities of relevant conditions for such chemistries to occur. Many of these prebiotic syntheses make use of similar molecules or have overlapping synthetic themes (Benner et al., 2019). Regardless of the precise path that synthesized the building blocks of life, in general, it is thought that geochemically available, fairly simple feedstock molecules were the inputs to prebiotic synthesis; intermediates with increasing complexity are formed, followed by the ultimate synthesis of prebiotic monomer building blocks (e.g., Fig. 1A). If we assume that the origins of life occurred with the canonical biomolecules still used by life today, these monomeric building blocks would have consisted of nucleotides and amino acids; however, a wider range of possibilities certainly exists (e.g., PNA, Nielsen, 2007; TNA, Orgel, 2000; depsipeptides, Fiahlo et al., 2021; noncanonical nucleotides, Fiahlo et al., 2020; noncanonical amino acids, Burton et al., 2012).
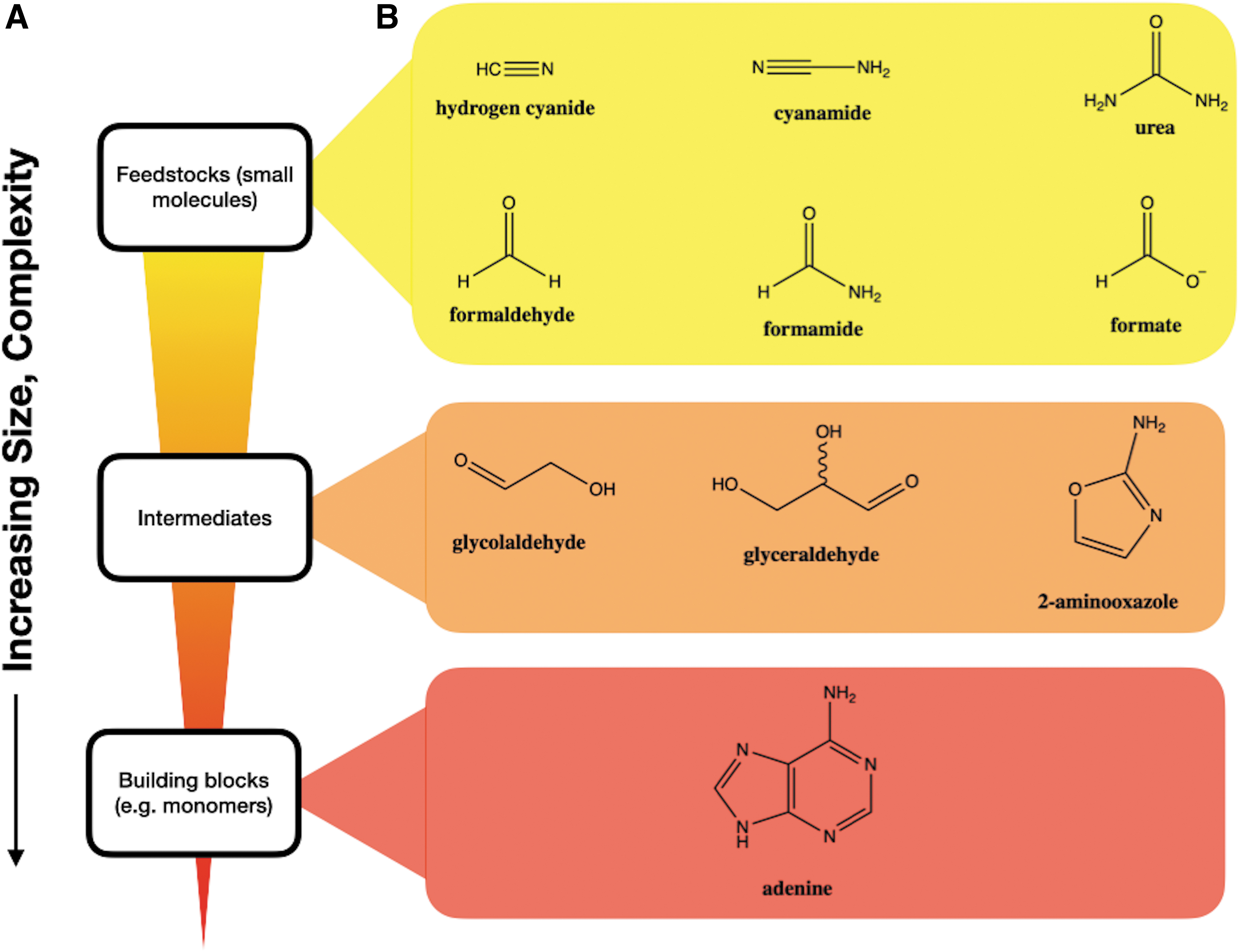
(
Here, we specifically study the UV absorption of the following molecules: formamide, formate, urea, cyanamide, 2-aminooxazole (2AO), glycolaldehyde, glyceraldehyde, cyanide, formaldehyde, and adenine (see Fig. 1B). These molecules are multiply invoked in diverse recent scenarios for origins of life chemistry. We then assess the implications of the measured results under the UV environment available on early Earth. Regardless of whether a given prebiotic scheme invokes the presence or absence of UV light, surface environments will likely be subjected to UV light. Therefore, understanding the UV transmission of prebiotic waters in a self-consistent manner is essential for constraining various prebiotic chemistries in surface waters on early Earth. Furthermore, understanding the UV transmission in environments where prebiotic chemistry may be occurring could aid in constraining various exoplanetary environments that may be conducive to prebiotic synthesis.
Measurements of molar decadic extinction coefficients
All chemicals were purchased at the highest available purity (see Supplementary Information Section 1 for details). All samples were dissolved in liquid chromatography–mass spectrometry (LC-MS) grade water (LiChrosolv, Millipore Sigma). Solid amounts of the chemicals were weighed and dissolved in LC-MS water to create the highest concentration solution for each sample. Concentrations did not exceed 10% of the saturation concentration to ensure samples could be fully dissolved, and scattering due to incomplete dissolution was not an issue. The UV-VIS absorption was measured from 200–360 nm using a Shimadzu UV-1900 or an Agilent Cary-60 UV-VIS spectrophotometer relative to a blank water sample. The molar decadic extinction coefficients were calculated as a function of wavelength according to the Beer-Lambert law (IUPAC, 1997):
where A is absorbance, P
0 is the incident spectral irradiant power, P is transmitted spectral irradiant power,
To adequately sample the molar decadic absorption coefficients in the linear region of the spectrophotometer, several dilutions were performed and measured spectrophotometrically. All measurements were repeated in triplicates. For details on measurement procedures, analysis, and error propagation, see Supplementary Information Section 2.
We follow Cockell (2000) in using scalar irradiance,
where
We first measured the molar decadic absorption coefficients for nine feedstock or intermediate molecules implicated in various prebiotic syntheses, as well as adenine for comparison. Of the feedstock or intermediate molecules, cyanamide and 2-aminooxazole have the highest molar decadic extinction coefficients, with peak extinction near ∼220 nm for both (Fig. 2). Cyanide, formamide, formate, and urea all have substantially smaller molar decadic extinction coefficients that peak at 200 nm and decrease at longer wavelengths. Similarly, glycolaldehyde and glyceraldehyde have maximum extinction at 200 nm but differ from the other molecules in that they show an additional extinction peak near ∼270 nm. The nucleobase adenine, measured for comparison to simpler feedstocks or intermediates, has the largest molar decadic extinction coefficient across all wavelengths. Adenine also differs from the other measured molecules in that its extinction at longer wavelengths (e.g., 260 nm) is several orders of magnitude larger than even the most UV-absorptive other molecules measured here at 260 nm. The molar decadic extinction coefficients of adenine and the other canonical nucleobases are available in the literature (see, e.g., the PhotochemCAD database, Dixon et al., 2005 and Taniguchi and Lindsey, 2018), and are shown in Supplementary Information Section 3, Supplementary Figure S1, along with our measurement of adenine. The broad absorption features of the canonical nucleobases, typically near ∼260 nm, are due to
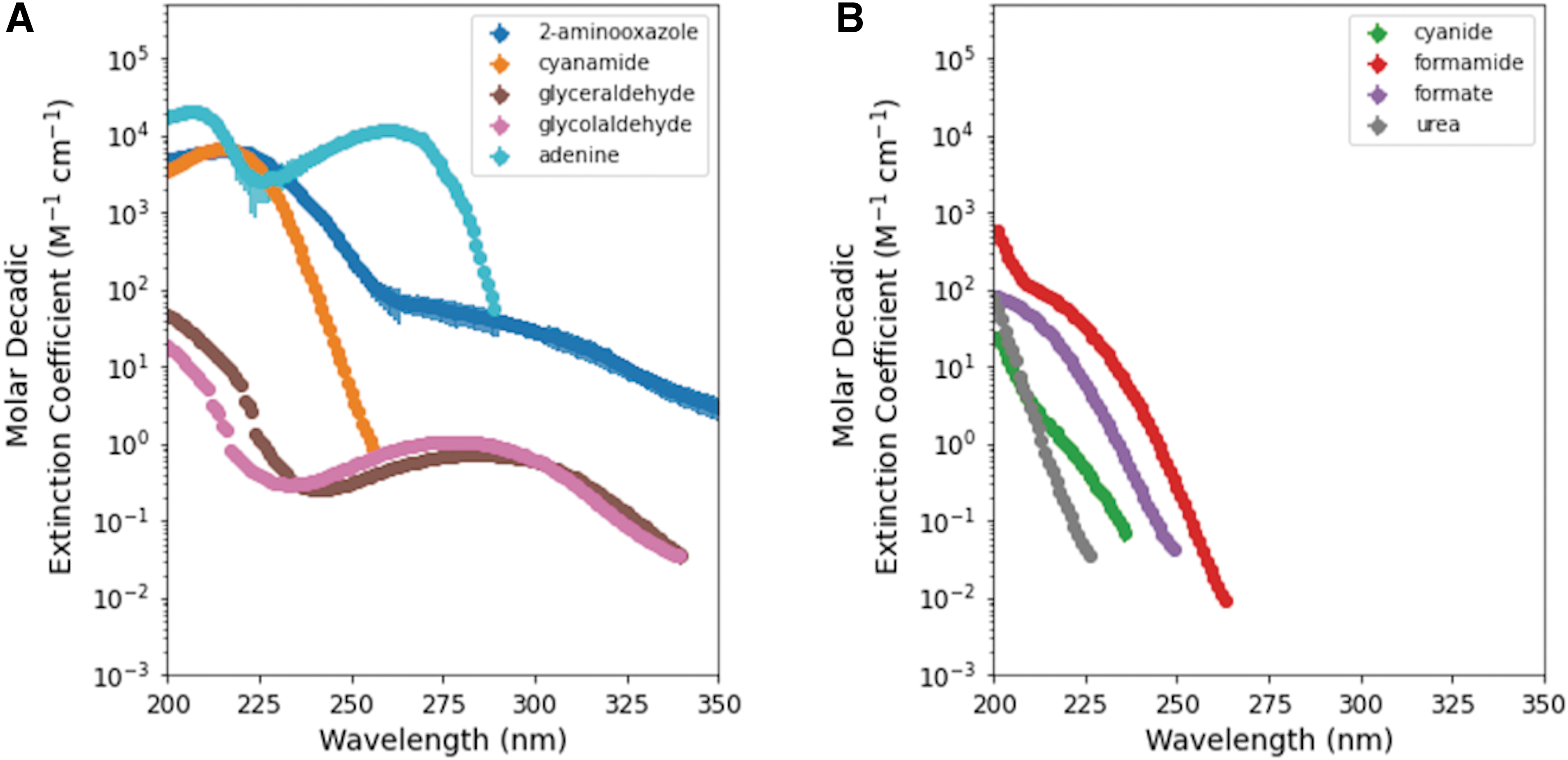
Molar decadic absorption coefficients measured here as a function of wavelength for all nine molecules measured in this study. (
In aqueous solution, formaldehyde nearly completely equilibrates to its hydrated form, H2C(OH)2. In addition, due to its instability to polymerization, formaldehyde solutions typically contain some amount of methanol as a stabilizing agent. Therefore, we are unable to calculate molar decadic extinction coefficients for formaldehyde itself; however, we have measured the absorption spectrum of formaldehyde solution (37%), stabilized with 10% methanol (Fig. 3). While the UV absorption of formaldehyde is highest at the shorter wavelengths, overall, formaldehyde is not significantly absorptive in the UV range of interest.
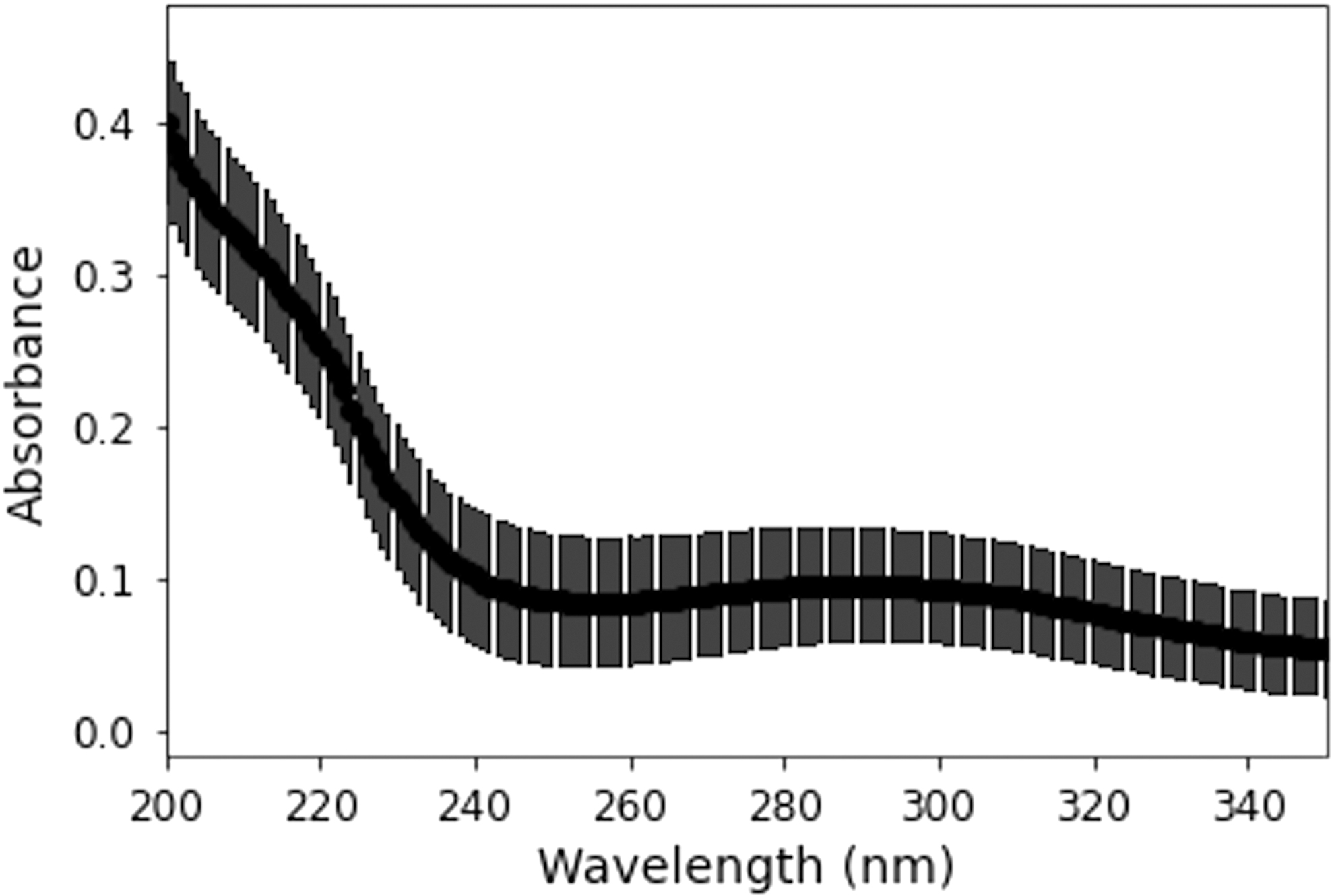
Absorption of 37% formaldehyde solution, with 10% methanol, as a function of wavelength.
The early Earth atmosphere, especially the likely presence of CO2 as a bulk constituent, coupled with the solar output, causes shorter wavelengths of UV light to be less abundant on the surface of early Earth. To understand the wavelength-dependent attenuation due to molecular absorption in surface waters on early Earth, we calculated the scalar irradiance of solutions containing equal concentrations of each of the molecules investigated here. Scalar irradiance,
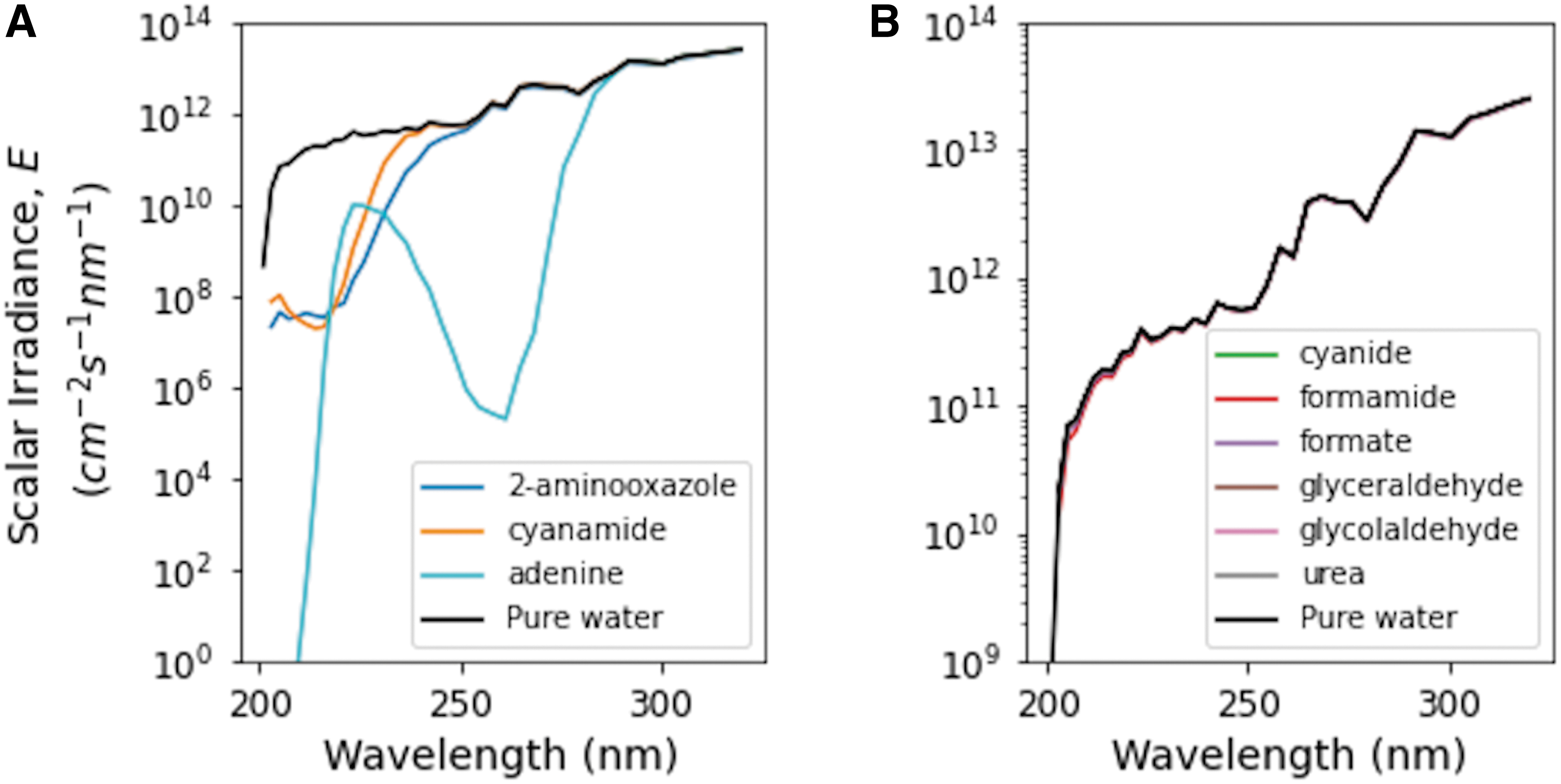
Scalar irradiance just below the surface of 1 mM solutions of each molecule. (
Adenine is by far the most UV-absorptive molecule investigated here; it effectively truncates the scalar irradiance in surface waters due to its strong UV absorption, especially in the ∼200–280 nm range (Fig. 4A). 2-Aminooxazole and cyanamide also significantly decrease the scalar irradiance at shorter (roughly <220 nm) wavelengths, though to a lesser extent than adenine. The remaining molecules do not significantly change the scalar irradiance from the case of pure water, shown in black in Fig. 4B, at the fairly low concentration (1 mM) used in this calculation.
To understand the cumulative effect across all wavelengths, we can integrate the scalar irradiance just below the surface from 200–320 nm (Table 1). The pure water case receives the highest overall scalar irradiance, which we then use as normalization for the other cases, also shown in Table 1. Adenine, the most absorptive case, receives 83.4% of the scalar irradiation in the pure water case.
Wavelength-Integrated Scalar Irradiance for 1 mM Solutions of Each Molecule, Sorted from Most to Least Absorptive
Values normalized to the pure water case are also shown.
This further illustrates that the presence of certain prebiotic species in surface waters may have a wide range of effects on the total UV transmission. In particular, we note that increasing complexity generally corresponds to higher extinction and lower scalar irradiance. We place these findings in the context of various prebiotic chemistry scenarios in Section 4.2.
Prebiotic chemistry scenarios
While the mechanisms by which the relatively simple molecules made available by abiotic processes complexified into biomolecules remains debated, various suggested prebiotic chemistries often make use of similar molecules. Historically, the formose reaction has been proposed for the synthesis of various sugars, including ribose. In the formose reaction, formaldehyde polymerizes to form a variety of products (Butlerov, 1861). Also historically, HCN was recognized as a potentially important molecule for prebiotic chemistry when it was demonstrated that HCN polymerization could make adenine (Oro, 1960; Oro and Kimball, 1961).
In more recent prebiotic reaction schemes, these types of small molecules continue to play important roles. On the path of the formose reaction, formaldehyde polymerizes to form the 2- and 3-carbon sugars glycolaldehyde and glyceraldehyde (Omran et al., 2020). A more targeted approach toward sugar synthesis, involving the use of UV light, also makes glycolaldehyde and glyceraldehyde through the reduction of HCN to formaldehyde (Ritson and Sutherland, 2012). In the presence of borate, formaldehyde condensation can favor ribose over other aldopentoses (Furukawa et al., 2013). Thus, a common approach to synthesizing the sugar components of nucleosides and nucleotides uses glycolaldehyde and glyceraldehyde, typically derived from a source of formaldehyde.
The nitrogenous components of nucleosides are commonly suggested to form through hydrogen cyanide or related compounds, such as formamide, urea, cyanamide, and so on. Various chemistries differ in the order of reactions: for example, Kim and Benner (2017) favor distinct syntheses of sugars derived from formaldehyde and nucleobases derived from HCN, before coupling the two to form ribonucleotides and threofuranosyl nucleotides via activated phosphate intermediates. On the other hand, the approach taken by work from the Sutherland lab (Powner et al., 2009) uses a mixed nitrogenous-oxygenous chemistry, combining glycolaldehyde and cyanamide to form 2-aminooxazole, which then adds to glyceraldehyde to form ribose aminooxazoline and subsequently the pyrimidine ribonucleosides. Becker et al. (2016) reacts glycolaldehyde and glyceraldehyde with formamidopyrimidines to ultimately form purine ribonucleosides. Formamidopyrimidines can be synthesized from compounds derived from ammonium cyanide, ammonium thiocyanate, cyanamide, and so on (Becker et al., 2016, 2019). Interestingly, a formamidopyrimidine is among the degradation products of UV irradiation of guanine (Crespo-Hernandez and Arce, 2004). Condensation of formamide leads to the generation of a wide array of nucleobases, nucleosides, sugars, and amino acids (Saladino et al., 2019). Noncanonical nucleobases, for potential use as pre-RNA, can be synthesized from similar molecules, including urea, guanidine, and malonic acid (Menor-Salvan et al., 2020).
UV transmission under selected prebiotic scenarios
Our measurements of the molar decadic extinction coefficients of these feedstock molecules allow for the UV transmission in certain experimental conditions to be explicitly modeled. These calculations can provide insights and constraints on the overall UV environment under which prebiotic chemistry may have occurred. However, it is important to note that the precise conditions under which prebiotic syntheses take place have not been exhaustively explored; there is a wide range of parameter space possible. Therefore, we caution against our measurements of UV extinction coefficients being used to rule for or against certain scenarios; rather, we suggest that these results can be used to better inform the overall conditions during certain scenarios, and used to assess the overall consistency of scenarios or better define various constraints on the chemistry. In particular, it should be noted that prebiotic syntheses in the laboratory often use ideal conditions (e.g., high concentrations, minimal unnecessary components) for experimental feasibility. In reality on prebiotic Earth, a much wider range of parameters (chemical components, concentrations, etc.) could have been possible.
As examples of how our measurements can be used, we model the UV transmission in specific scenarios by calculating the penetration depth of photons as a function of wavelength. The penetration depth δp is defined as the depth at which the transmitted irradiation falls to 1/e of the incident irradiation (see Supplementary Information Section 4 for details of the calculation). We select the concentrations used in reported syntheses while noting that these concentrations may not be representative of natural prebiotic environments. Many of the high concentrations used in synthetic reactions are selected for experimental ease; in actuality, on prebiotic Earth, it may be possible that much lower concentrations and longer timescales are sufficient for driving prebiotic synthesis (e.g., Todd et al., 2018).
Oro and Kimball (1961) showed that concentrated solutions of HCN heated in the presence of ammonia yielded adenine, the pentamer of HCN. In particular, for an 11.1 M HCN solution, 110 mg of adenine per liter of original solution (or 0.814 mM) was synthesized. The penetration depth of UV photons as a function of wavelength is shown in Fig. 5A for both 11.1 M HCN (blue) and 0.814 mM adenine (orange). At wavelengths <220 nm, UV photons only penetrate roughly 1 mm or less in both concentrated HCN solution and adenine solution. Adenine also efficiently truncates UV photons up through ∼280 nm, unlike concentrated HCN. At longer wavelengths, photons pass through both solutions relatively unimpeded.
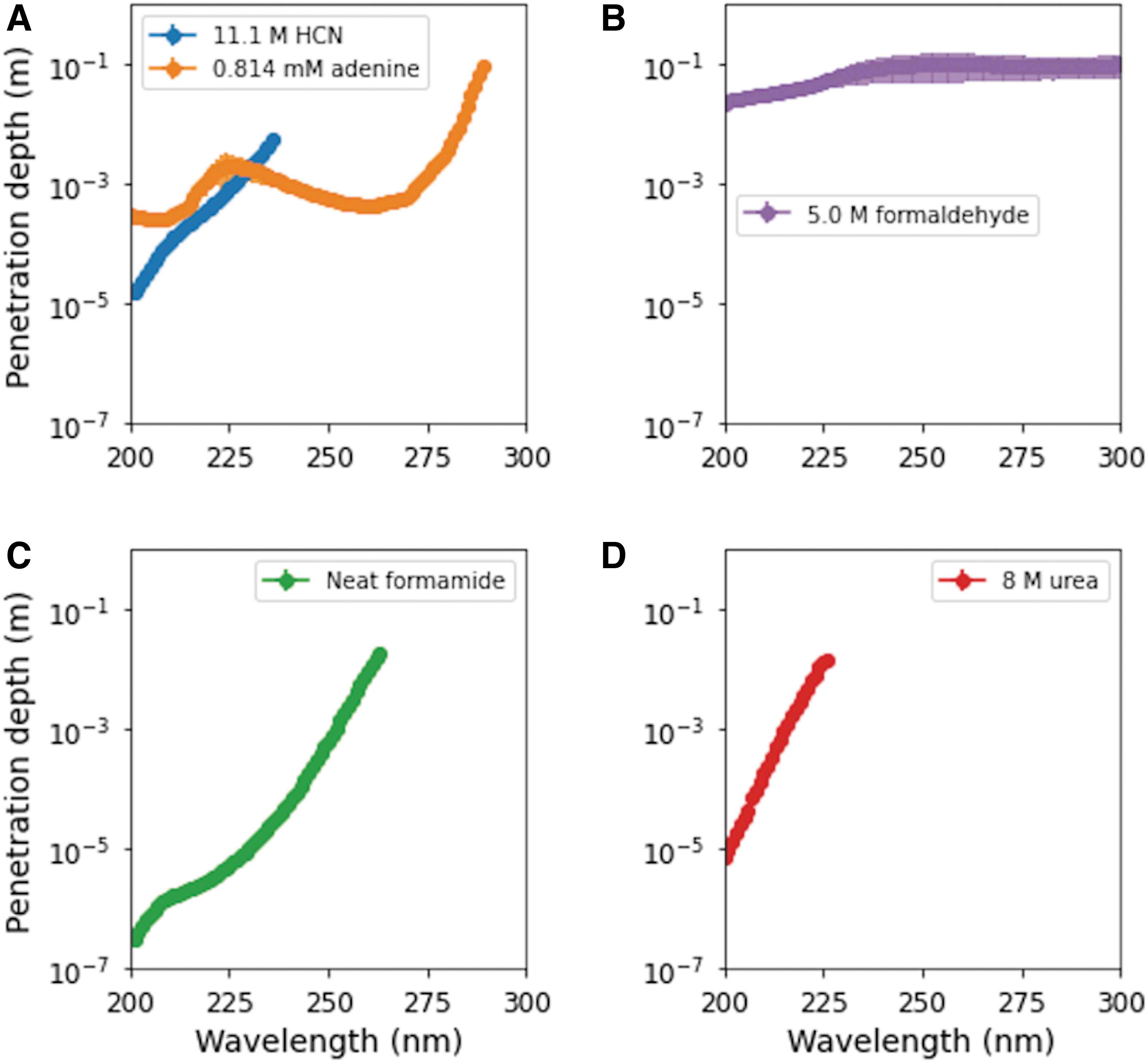
Penetration depth of UV photons under selected prebiotic scenarios used for synthesis: (
The formose reaction relies on concentrated solutions of formaldehyde to synthesize more complex sugars, including glycolaldehyde, glyceraldehyde, and ribose. We determined the penetration depth of UV photons through solutions of 5.0 M formaldehyde, stabilized with methanol (Fig. 5B). Even at the shortest wavelengths where formaldehyde is most absorptive, photons can penetrate on the order of centimeters. At longer wavelengths, photons can pass through tens to hundreds of centimeters in 5 M formaldehyde solution.
Formamide has often been invoked as a source for prebiotic synthesis of multiple types of building blocks (Hudson et al., 2012; Green et al. 2023; Saladino et al., 2019 and references therein). In particular, formamide can be used in the synthesis of nucleobases, nucleosides, and a number of other metabolically relevant compounds (see Saladino et al., 2012 and references therein). These chemical syntheses often heat solutions of pure formamide. We therefore calculated the penetration depth of UV photons in solutions of pure liquid formamide (Fig. 5C). These solutions effectively truncate photons (penetration depth <1 cm) up through wavelengths of ∼250 nm, making these solutions among the most UV-absorptive of the various prebiotic scenarios we calculate here.
Urea, the hydrolysis product of cyanamide, has also been used in prebiotic synthesis of pre-RNA building blocks (Menor-Salvan et al., 2020). In this case, solutions of 8 M urea, mixed with other molecules like guanidine and malonic acid, yielded noncanonical pyrimidines including 2,4,6-triaminopyrimidine (TAP) and melamine upon wet-dry cycling (Menor-Salvan et al., 2020). Figure 5D shows the penetration depth of UV photons through 8 M urea solutions. At the shortest wavelengths, UV photons are efficiently truncated, but penetration depth increases significantly at longer wavelengths, to >1 cm at wavelengths longer than 224 nm.
We also calculate the UV transmission in solutions along various steps in the cyanosulfidic scenario outlined in Patel et al. (2015) and Powner et al. (2009) using the experimentally determined extinction coefficients for the respective individual components. To synthesize nucleosides in this scenario, cyanamide and glycolaldehyde are reacted to form 2-aminooxazole, which then reacts with glyceraldehyde to form ribose aminooxazoline. Experimentally, Powner et al. (2009) used 1 M concentrations of each reactant, though the synthesis may proceed over a much wider range of conditions. Figure 6A shows the UV transmission for 1 M cyanamide +1 M glycolaldehyde, in comparison to 1 M 2-aminooxazole; Fig. 6B shows the UV transmission for 1 M 2-aminooxazole +1 M glyceraldehyde. UV light with wavelength <220 nm is truncated within 10−3 cm in these two cases; longer wavelengths can penetrate deeper but still are truncated in <1 cm. The implications for the chemistry overall in these scenarios remains a question for further study. Some UV light is productive for driving certain steps of the chemistry, but irradiation can also be destructive for some reactants, intermediates, and products. The consistency and plausibility of such chemistry is likely to come down to a question of kinetics of various productive versus destructive steps. We therefore advocate for further studies toward understanding the kinetics of various prebiotic chemical reactions, including both those that make desired products and those that degrade such molecules. Furthermore, such studies should be understood in the context of their environments, which may suggest the planetary conditions that could be most favorable for given reactions.
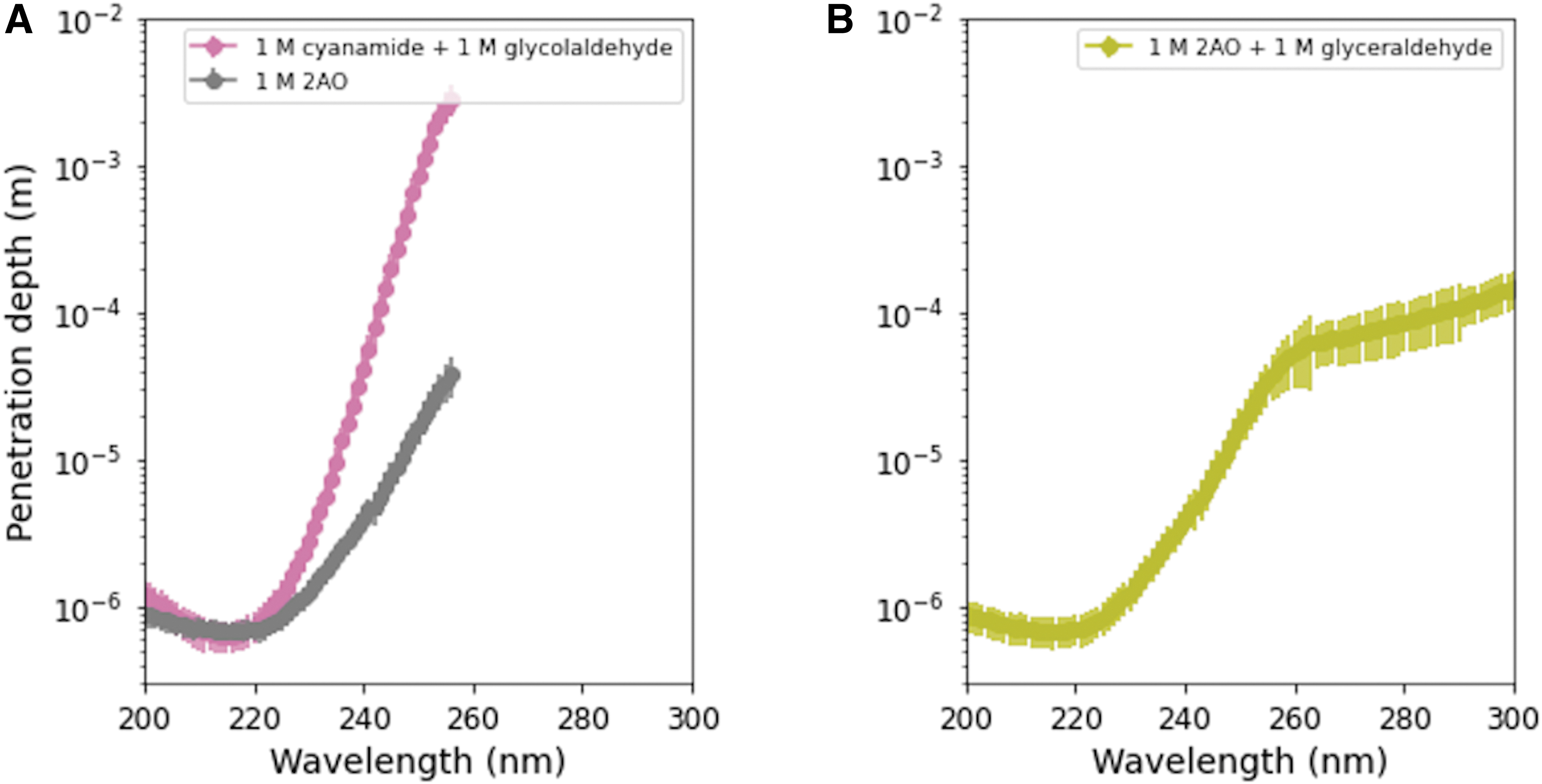
Penetration depth of UV photons at two steps in the cyanosulifidic scenario outlined in, for example, Powner et al. (2009), Patel et al. (2015). (
In the geochemical scenario, all the molecules measured here have plausible sources. HCN is available from a variety of potential sources, including atmospheric photochemistry in a methane-rich atmosphere (Tian et al., 2011), lightning generation (Chameides and Walker, 1981), impact generation (Ferus et al., 2017; Parkos et al., 2018), post-impact photochemistry (Zahnle et al., 2020), and impact delivery (Todd and Öberg, 2020). Over time, hydrolysis of HCN leads first to formamide, followed by formate (Miyakawa et al., 2002). Cyanide can be a source of urea through irradiation (Lohrmann, 1972); urea is also formed from irradiation of cometary model ices (Förstel et al., 2016). Cyanamide can be generated through the thermal decomposition of calcium ferrocyanide to form calcium cyanamide, which then liberates cyanamide upon hydration (Patel et al., 2015). Formaldehyde can be generated through atmospheric photochemistry (Pinto et al., 1980), impacts (Masuda et al., 2021), or photoreductive homologation of HCN (Ritson and Sutherland, 2012).
The types of small molecules investigated in this study overall have a number of potential sources in the planetary environment. It is possible that these molecules could have been localized to similar environments, potentially allowing for prebiotic synthesis to occur. A common theme across many varying prebiotic synthetic pathways is the transition from these simple molecules toward increasing complexity. Regardless of the precise path, many syntheses use the same precursors, indicating that the molecules described here could be useful across a number of theories for the origins of life.
UV light in prebiotic chemistry: Toward self-consistent scenarios
Early Earth would have been subjected to higher amounts of UV light, due to a lack of atmospheric shielding by ozone and other species (Farquhar et al., 2001; Pavlov and Kasting, 2002); some have postulated that this vast amount of energy could have been crucial for driving prebiotic synthesis. Others have suggested that life must have originated in UV-shielded environments, due to the damaging nature of UV light. Our work here is relevant in both these cases; our data allows for the determination of the UV environment in various conditions. In general, the species measured here absorb more strongly at shorter wavelengths. So photochemistry using longer-wavelength photons may be more robust in prebiotic environments involving the molecules we have measured in this study. For example, if UV light is required for a given synthesis, our measured molar decadic extinction coefficients can allow constraints on the concentrations of reactants to be determined. Alternatively, if certain chemistry is particularly UV-sensitive, the work presented here can allow for determination of when solutions of various reactants and concentrations will be optically thick to UV photons. It is also important to consider that surface waters on early Earth may not have been stagnant over time, both on a geological scale and on shorter timescales. For example, wind could drive currents in waters, leading to circulation of water, allowing water from depth to reach the surface and vice versa (e.g., Hui et al., 2019 and references therein). Furthermore, wet-dry cycling, including seasonal evaporation, could significantly change the concentrations of species in surface waters and therefore affect the UV penetration depth. In combination with the results presented here, these influences can help constrain possible scenarios and conditions that may allow for productive prebiotic chemistry to occur. Regardless of whether UV light is invoked as a source of energy, it is important to understand the UV environment that molecules on the surface of a planet will be facing, due to photodestruction potentially limiting the useful lifetimes of these molecules. For example, attenuation of UV light due to dissolved species has been suggested as mechanism to protect liposomes from damaging UV radiation (Subbotin and Fiksel, 2023a, 2023b). In addition, the chemical environment has the potential to strongly influence the lifetime of molecules to UV light: if more UV-absorbing material is present, photosensitive molecules can persist for longer periods under irradiation (Todd et al., 2021).
Caveats and future work
With our compilation of the molar decadic extinction coefficients of a number of commonly invoked prebiotic molecules, we allow for the UV penetration depth for specific scenarios to be calculated. With our current state of understanding of prebiotic chemistry, there are multiple scenarios that could potentially be invoked; we do not attempt to address these all here. It should also be noted that even in a specific scenario, the constraints of the chemistry (e.g., to various reactant concentrations, the presence of other molecules) have not necessarily been completely explored. Therefore, we do not make absolute claims as to the plausibility or success of a given scenario here; rather, we present several test cases to show what information our results can provide. Furthermore, we encourage the use of our data for other specific cases as well as new and emerging prebiotic synthesis suggestions. While our set of molecules is obviously not complete, we hope this provides a basis for many scenarios and offers a springboard for future characterization of other molecules that become of interest as prebiotic chemistry continues to develop.
Conclusions
Here we have measured the molar decadic extinction coefficients of nine prebiotically relevant molecules as a function of wavelength. These measurements aid in estimating the UV transmission in prebiotic waters containing these compounds. Understanding the UV environment present in various waters where prebiotic chemistry may have occurred is important, not only for chemistry that may make use of UV photons as a source of energy but for any prebiotic chemistry, due to the ubiquitous nature of prebiotic UV. Since UV light can degrade biomolecules and small molecules such as those measured here over time, it is important to understand how solutions behave with respect to UV transmission and potential shielding. Our measurements allow for the calculation of photon penetration under various scenarios in order to better constrain both the UV environment and the chemistry. While the molecules investigated here are not a complete set of possible prebiotic molecules, we have focused on molecules commonly invoked as synthetic sources across various prebiotic syntheses. Future work can continue to add to the catalog of molecules to provide better understanding of more complex and complete networks.
Footnotes
Acknowledgments
We would like to thank P.B. Rimmer and J.D. Sutherland for helpful discussions. We would also like to acknowledge the support of the Harvard Origins of Life Initiative and the University of Washington Astrobiology Program. Support for this work was provided by NASA through the NASA Hubble Fellowship (grant #HST-HF2-51471) awarded by the Space Telescope Science Institute, which is operated by the Association of Universities for Research in Astronomy, Inc., for NASA under contract NAS5-26555. We would also like to acknowledge funding from the Simons Foundation (Grant #511570 to D.C.C. and 290360 to D.D.S.).
Supplementary Material
Supplementary Information
Supplementary Figure S1
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
