Abstract
Quantum dots (QDs) are usually seen as artificial semiconductor particles exhibiting optical and electronic properties interesting for nanotechnological applications. However, they may also play a role in prebiotic chemistry. Starting from zinc acetate, cadmium acetate, and mercaptosuccinic acid, we demonstrate the formation of ZnCd QDs upon UV irradiation in prebiotic liquid formamide. We show that ZnCd QDs are able to increase the yield of RNA nucleobase synthesis from formamide up to 300 times, suggesting they might have served as universal catalysts in a primordial milieu. Based on the experimentally observed peroxidase-like activity of ZnCd QDs upon irradiation with visible light, we propose that QDs could be relevant to a broad variety of processes relating to the emergence of terrestrial life.
1. Introduction
The idea that minerals and rocks might have played an important role in prebiotic synthesis (Lambert, 2015) was pioneered in the early 1950s when physicist John Desmond Bernal and geochemist Victor Moritz Goldschmidt (Goldschmidt, 1952; Ferris, 2005) realized clay minerals exhibit several attributes essential for the origin of life (see the review in Ferris [2005] and references therein). Chemist Graham Cairns-Smith extended this idea and even proposed that the first life was entirely based on colloidal clay crystallites (Cairns-Smith, 1966). Modern findings indicate that not only clays but the full complex Hadean geochemical inventory played a crucial role in prebiotic evolution (Hazen and Sverjensky, 2010). A living chemical entity very likely did not evolve in an isolated single warm pond. Its origin probably involved the interplay and coincidence of a number of processes and conditions, in which minerals and crystalline surfaces are central for supporting the increase of chemical complexity, providing interfaces for selection and concentration of more dilute solutions, creating physical and chemical gradients, and supporting fluxes and cycles on their surfaces (Hazen and Sverjensky, 2010; Powner and Sutherland, 2011). Plausible geochemical environments might have been central to permitting the robust reactions plausible for prebiotic chemistry (Eschenmoser, 2007). The most influential current theory suggests that life probably began in the unique physical, chemical, and geochemical environment of seawater near hydrothermal vents (Mulkidjanian et al., 2012). From the point of view of a biologist, the seawater is rich in ions important to life. On the other hand, the plausibility of condensation reactions in an aqueous environment is questioned. Damer and Deamer have recently published the hot spring hypothesis introducing another compatible scenario, where unique conditions were provided by fluctuating volcanic hot spring pools on land while mineral surfaces served, for instance, as an accumulating kinetic trap (Damer and Deamer, 2020).
In the current paper, we focus on the potential catalytic role of binary metallic nanoparticles—quantum dots (QDs), in the frame of our attempts to describe the effect of the geological environment on prebiotic synthesis of nucleobases. The wider scientific community understands QDs as artificial nanocrystals (nanoparticles) produced for technological research and applications (Zhou et al., 2015). They are also identified as artificial atomlike entities with respect to their unique physical properties (electronic energy levels leading to specific fluorescent, emission, and absorption spectra, photochemical stability, electrical and semiconductor characteristics) and chemical properties (photoactivity and large surface area [Zhang and Liu, 2020]), that differ from the bulk material they are made of (Schwirn et al., 2014). However, QDs are not only artificial objects but also originate from natural processes (Bundschuh et al., 2018) such as hydrothermal carbonization of organic material (Das et al., 2018 and references therein) and biomass combustion (Maddenc, 2012), and appear as important constituents of the atmosphere, oceans, soils, biota (produced or ingested by organisms [Zhou et al., 2015]) (Hochella et al., 2008). Nanocrystal arrays (resembling QDs) attributed to ferropericlase in amorphous silica matrices have been discovered in the Chelyabinsk meteorite (Pavlov et al., 2014), and our recent study demonstrates a surprisingly simple method for ZnCd nanoparticle synthesis starting from solutions containing Zn(II) and Cd(II) cations in the presence of a thiol capping agent upon UV light irradiation (Nejdl et al., 2020). The prebiotic potential of QDs as a universal catalyst and light-activated nanozyme is a new topic worth exploring. Over the past few decades, researchers have established artificial enzymes (nanozymes) as highly stable and low-cost alternatives to natural enzymes (Wu et al., 2019) and also introduced light to nanozymes (light-activation), especially with respect to photosensitive oxygen activation (Zhang and Liu, 2020). It is important to experimentally examine the formation of QDs in the context of minerals and rocks under prebiotically relevant conditions, compare their role in prebiotic chemistry with other plausible minerals, such as clays, and explore their participation in processes potentially leading to the origin of life according to state-of-the-art theories.
For the purpose of this study, we have selected ZnCd QDs as a light-activatable nanozyme and contextualize their role in the hot spring hypothesis (Damer and Deamer, 2020). We start with formamide as a building block for RNA nucleobases (Costanzo et al., 2007), an important hub in complex chemical networks and a solvent promoting polymerization and phosphorylation (Saladino et al., 2012, 2015, 2019; Pietrucci and Saitta, 2015; Šponer et al., 2016). Formamide-based synthesis represents an alternative scenario for the expected series of steps leading to important prebiotic building blocks (McCollom, 2013; Sutherland, 2017; Saladino et al., 2019) and the emergence of the first living entity prior to 3.8 Ga (Mojzsis et al., 1996), or even earlier (Bell et al., 2015; Dodd et al., 2017; Tashiro et al., 2017). Recent studies have confirmed plausibility of prebiotic thermal formamide-based one-pot synthesis in the presence of meteorite material (Saladino et al., 2013; Ferus et al., 2015a), iron-rich clays (Pastorek et al., 2019), titanium oxide (Saladino et al., 2003), iron sulfide minerals (Saladino et al., 2008), zirconium minerals (Saladino et al., 2010), borate minerals (Saladino et al., 2011), or cosmic dust analogs (Saladino et al., 2005). Other energy sources have also been investigated, such as a proton beam simulating the solar wind (Saladino et al., 2015), asteroid impact plasma (Barks et al., 2010; Ferus et al., 2015b, 2017, 2019), UV irradiation, or electric discharges (Ferus et al., 2018). Another possibility is the existence of lagoons exposed to UV radiation and sunlight on the surface of early Earth (Patel et al., 2015; Botta et al., 2018; Kitadai et al., 2019) and the occurrence of zinc (Mulkidjanian, 2009) and cadmium salts in metal-rich hot springs. Common chondrite meteorites contain both elements embedded within silicates (Nishimura and Sandell, 1964; Rosman and De Laeter, 1974; Kruijer et al., 2013): about 50 ppm zinc and 30 ppb cadmium on the average, with approximate ranges from 30 to 70 ppm for zinc and 1 ppb to 1 ppm for cadmium.
Of course, our pioneering research into ZnCd QDs should be continued with the examination of other types of nanoparticles (nanoclusters) (e.g., Ni, Fe, or carbon-based), and containing other more-abundant elements such as Cu and Al but also Ag, Co, Pb, Mo, and so on (Kumar et al., 2014; Bizzarri et al., 2018). The metal ions such as Fe and Ni particularly should exist in greater quantities in prebiotic hot springs. The chemistry of other molecules with a role in hydrogen cyanide–based chemistry (Hörst et al., 2012; Patel et al., 2015; Pietrucci and Saitta, 2015; Sutherland, 2017; Ferus et al., 2020) must be examined in connection to QDs. QD-based prebiotic synthesis should be compared with other chemical schemes, such as the Zinc World (Mulkidjanian, 2009), Thioester World (Deduve, 1992), Lipid World (Segre et al., 2001), and Polycyclic Aromatic Hydrocarbon World (Groen et al., 2012), that are currently being considered as explanations for the origin of life on Earth (Vasas et al., 2010).
2. Materials and Methods
In this study, we perform three experiments exploring the role of QDs in individual processes relevant to a hot spring environment: The feasibility of ZnCd QDs forming in the presence of a prebiotic parent molecule. We select formamide for this study, but we suggest future experiments exploring HCN chemistry. The effect of ZnCd QDs on the yield of canonical RNA nucleobases from formamide-based thermal synthesis. The role of ZnCd QDs in nanozymatic chemistry and their potential to resemble peroxidase activity.
2.1. Chemicals
Standards and other chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) in ACS purity.
2.2. ZnCd QD precursor preparation
Stock solutions of 16 mM mercaptosuccinic acid (MSA), 6 mM zinc(II) acetate, 6 mM cadmium(II) acetate, 100 mM phosphate buffer pH 7, ≥99% formamide, 15% ammonia (NH3) were prepared in distilled water and stored at 4°C.
2.3. UV irradiation of ZnCd QD precursors in formamide and fluorescence analysis
The samples were pipetted into five neighboring wells of a UV-transparent 96-wellplate with a flat bottom by CoStar (Corning, USA) as 12.5 μL of 6 mM zinc(II) acetate, 12.5 μL of 6 mM cadmium(II) acetate, 12.5 μL of 16 mM MSA, 12.5 μL of 15%, 7.5%, 3.7%, 1.8%, and 0% (v/v) NH3. Subsequently, 50 μL of ≥99% formamide was added to each well. A total volume of the samples was 100 μL each. The plate was then placed into the UV transilluminator (Vilber Lourmat, Marne-la-Vallee, France) and irradiated for 5 min. The sample area of 20 × 20 cm was illuminated by 6 UV tubes with total output power of 90 W and λmax = 254 nm. Fluorescence has been induced by UV light flash lamp with λex = 350 nm. The emission spectral scan was recorded within the range from 380 to 800 nm by a Tecan Infinite 200 M PRO (TECAN, Switzerland).
2.4. Preparation of ZnCd QDs for thermolysis experiment in formamide
One liter of ZnCd QDs precursors containing 0.4 L 6 mM zinc(II) acetate, 0.1 L 6 mM cadmium(II) acetate, 0.25 L 16 mM MSA, and 0.25 L 100 mM phosphate buffer pH 7 was prepared. Stock solution (1 L of ZnCd QDs precursors) was divided into twenty 50 mL plastic tubes. Ten plastic tubes were placed in the UV transilluminator (Vilber Lourmat, Marne-la-Vallee, France) and irradiated for approximately 2 h. Thereafter, all samples were frozen in liquid nitrogen. Finally, 10 (frozen) un-irradiated (Zn:Cd:MSA precursors) and 10 (frozen) irradiated (ZnCd QDs) 50 mL tubes were placed in a lyophilizer (2.5 Liter Benchtop Freeze Dry System, Labsconco, KS, USA) until transformation from the liquid phase to the solid phase was complete. Then the solid phase was collected and marked as a Zn:Cd (un-irradiated sample) and ZnCd QDs (irradiated sample). Their characterization (fluorescence properties, dispersity, zeta potential, and electron microscopy imaging) can be found in our previous work (Nejdl et al., 2020).
2.4.1. Thermolysis of formamide samples with ZnCd QDs and Zn:Cd
Thermolysis of formamide samples with Zn:Cd and ZnCd QDs were carried out as follows. Three different samples were prepared in glass flasks. Each sample represents a closed system consisting of 2.0 mL of pure formamide, optionally about 0.05 g of Zn:Cd or ZnCd QDs and 500 torr of nitrogen (N2) atmosphere. All samples were then placed into a preheated oil bath at 160°C, schematically shown in Fig. 1. The thermolysis performed in this way lasted 24 h. The samples were then removed from the oil bath and allowed to cool to room temperature. Following that, the flasks were opened, and the nonvolatile phase material was divided into two parts and stored in Eppendorf tubes. One part of each sample was immediately subjected to gas chromatography–mass spectrometry (GC-MS) and micellar electrokinetic chromatography (MEKC) analyses.
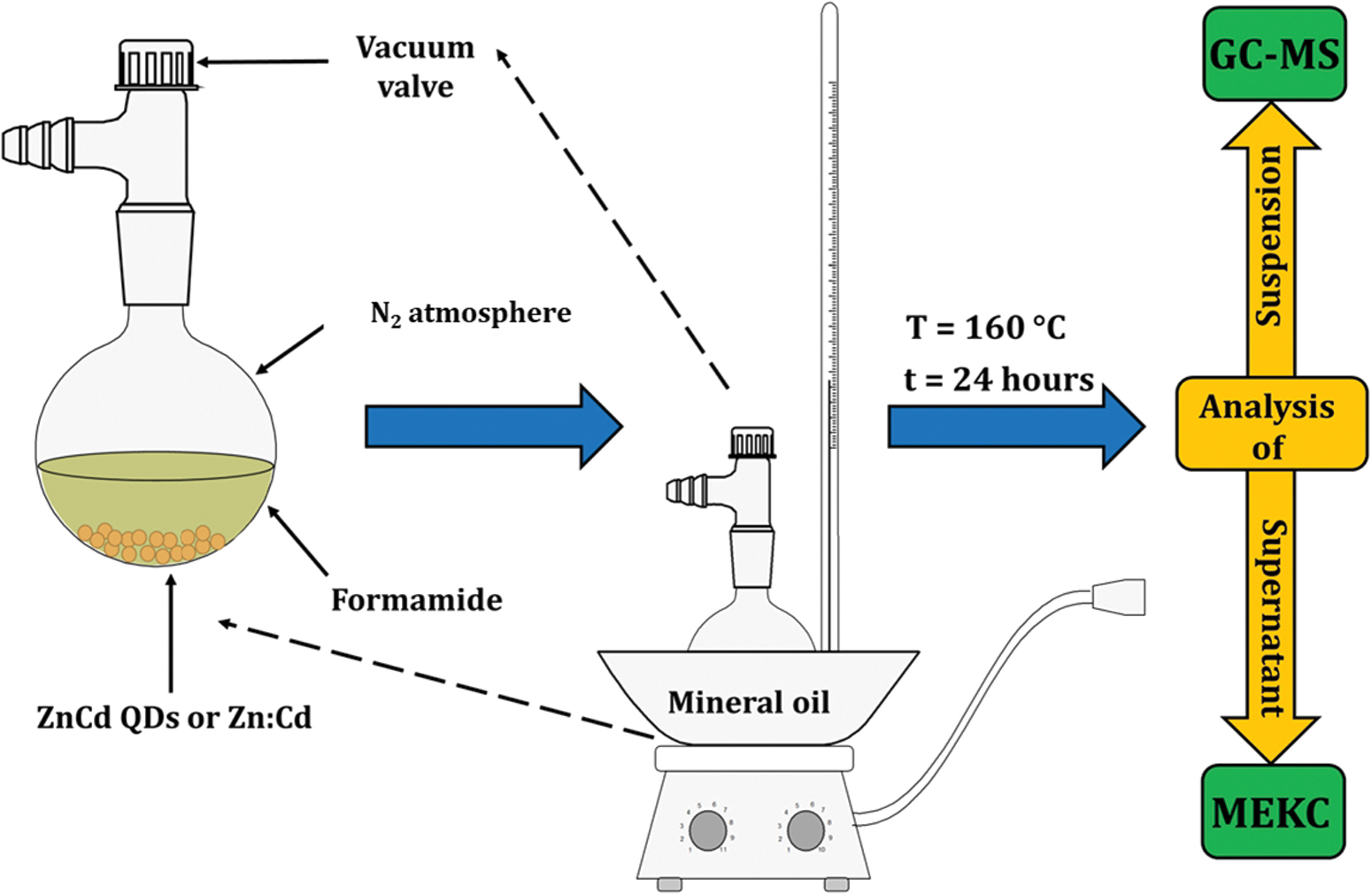
Schematic representation of the apparatus used for thermolytic experiments. Color images are available online.
2.5. Gas chromatography with mass spectrometry (GC-MS)
The nonvolatile phase material was diluted in methanol in 1:1 ratio and the mixture macerated for 2 h with occasional stirring. Then 200 μL of this suspension was transferred into separate vials and dried under vacuum at room temperature. The dried samples were derivatized. Fifty microliters of the derivatization agent N–(tert–butyldimethylsilyl)–N–methyl–trifluoroacetamide (Sigma-Aldrich; 99% (wt/wt) MTBSTFA, CAS 77377 − 52 − 7) and 150 μL of pyridine aprotic solvent (Scharlau; 99.5% (wt/wt) anhydrous) were added to the dried sample. Then the sample was kept at 110°C for 2 h. The measurement was performed using a Trace 1300 gas chromatograph coupled with an ion-trap mass spectrometer 1100 (Thermo Scientific, USA). The chromatograph was equipped with a nonpolar Trace GOLD TG-5 MS capillary column (Thermo Scientific, USA; 95% (wt/wt) dimethylpolysiloxane and 5% (wt/wt) phenylmethylpolysiloxane). One microliter of the derivatized sample was injected into the system. The separation was performed using a 10°C min−1 gradient from 180°C to 280°C. The samples were scanned for the presence of nucleobases, urea, purine, and glycine. The detection of the products was confirmed by comparison of the retention times and mass spectra of the sample with standards of the detected compounds treated in the same manner as the sample, Fig. S1 (MS spectra of silylated purine), Fig. S2 (MS spectra of silylated adenine), Fig. S3 (MS spectra of silylated guanine), and Fig. S4 (MS spectra of silylated cytosine). Blank runs were also performed to rule out any external contamination.
2.6. Micellar electrokinetic capillary chromatography (MEKC)
A quantification of nucleobases was performed with a capillary electrophoresis instrument 7100 (Agilent Technologies, Germany) with absorbance detection at a wavelength of 260 nm. A fused silica capillary with an internal diameter of 75 μm, a total length of 64.5 cm, and an effective length of 56 cm was used. The following parameters were used: the sample was introduced hydrodynamically by 40 mbar for 5 s, and a separation voltage of 15 kV was applied. A background electrolyte (BGE) was composed of 40 mM borate, 60 mM sodium dodecyl sulfate at pH 9.8. Prior to the analysis, the capillary was washed for 100 s using BGE. Before MEKC analysis, the formamide samples were thawed and prepared as follows: samples were centrifuged at 12,000 rpm for 10 min; the supernatant was taken and subsequently diluted 10-fold by distilled water and immediately measured by MEKC. In the MEKC electropherograms the signals corresponding to nucleobases can be clearly distinguished based on their typical migration times (Zemankova et al., 2021). Blank runs were also performed to rule out any external contamination (Fig. S5A), and detection of the products was confirmed by the method of standard additions, Fig. S5B.
2.7. Peroxidase activity exploration
The three samples of ZnCd QDs with red, yellow, and blue emission in 50% formamide (1 mL each) were prepared (according Section 2.3) and precipitated by 2-propanol (in 1:1 ratio, 10 min incubation under laboratory conditions) and centrifuged (10 min, 12,000 rpm). Subsequently, supernatants were removed and 100 μL of distilled water added. Peroxidase-like activity of 40 μL ZnCd QDs with 60 μL peroxidase substrate–tetramethylbenzidine (TMB) in the presence of H2O2 was investigated (under laboratory conditions) after visible light irradiation by table 6800K (6W) LED lamp (EMOS, Přerov, Czech Republic). The absorption maximum of the reaction mixture (ZnCd QDs with TMB) is 652 nm, which originates from the oxidation of TMB (Mu et al., 2012). A UV/vis spectrophotometer Tecan Infinite 200 M PRO (TECAN, Switzerland) and UV-transparent 96-wellplate with a flat bottom by CoStar (Corning, USA) were used.
3. Results
We have explored three important aspects of the potential relevance of ZnCd QDs to a prebiotic hot springs scenario (Damer and Deamer, 2020):
(de novo) synthesis of ZnCd QDs from a solution of zinc, cadmium salts, and MSA in the presence of formamide;
the catalytic effect of ZnCd QDs on the formamide-based synthesis of RNA nucleobases; and
the peroxidase-like activity of ZnCd QDsP.
Based on these results, we tentatively suggest that QDs may have played a role in prebiotic synthesis, and we suggest introducing a new concept of the “Nanoparticle World” (Lukas et al., 2020).
The results of investigations I, II, and III can be summarized as follows.
3.1. Prebiotic synthesis of ZnCd QDs in formamide
De novo QD formation has been observed by detection of their fluorescence in 0.75 mM cadmium and zinc acetate in 50% (v/v) formamide water solution alkalized by NH3 in the presence of 4 mM MSA after 5 min of UV irradiation (λmax = 254 nm). First, the fluorescence of a non-irradiated sample was tested and the same measurement made for a sample with 5 m of irradiation in the UV transilluminator. Figure 2 shows the difference between fluorescence signals measured for non-irradiated (panel A) and irradiated (panel B) samples.
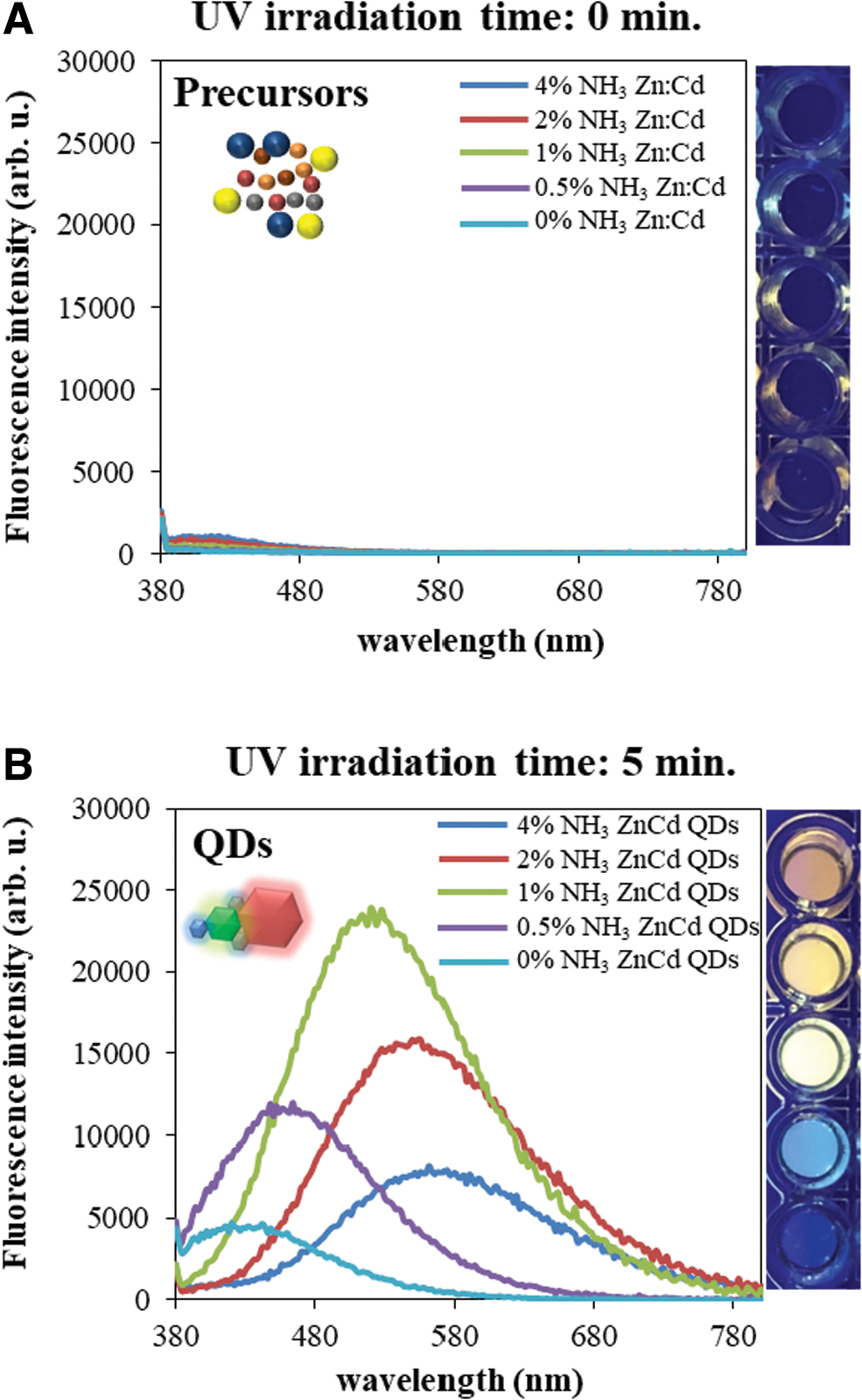
Precursors (zinc acetate, cadmium acetate, mercaptosuccinic acid, and ammonia in 50% formamide) were UV-irradiated for 0 (
The recorded fluorescence spectra confirm the formation of ZnCd QDs upon UV irradiation (Nejdl et al., 2017). The emission of QDs exhibits a typical shape covering the spectral range 380–780 nm (Fig. 2B), with an additional red shift correlated with increasing ammonia concentration (see the Experimental Section). The physical (fluorescence properties, dispersity, zeta potential, and electron micrographs) and chemical properties (xanthine oxidase–like activity) were determined (not shown) and correlated well with our previously published work (Nejdl et al., 2020). Even small differences in particle size result in varied fluorescence color (quantum size effects [Yang, 2021]) and also nanozymatic activity. This is specifically discussed in Section 3.3.
3.2. Formamide-based one-pot nucleobase synthesis in the presence of ZnCd QDs
Both GC-MS and MECK analyses find greater synthesis yields in the presence of ZnCd QDs for P, A, and C. GC-MS detection exhibits at least an order of magnitude higher yield when compared with Zn:Cd precursors, as well as with most materials used in our previous study (Pastorek et al., 2019). The most noticeable difference is for the case of C. Pyrimidine nucleobases usually exhibit lower yields in a formamide-based synthesis than purine nucleobases, while this ordering is reversed in the presence of QDs.
The GC-MS results are close to a representative measurement of nucleobases synthesized both in liquid and adsorbed solid phases, because the liquid formamide is completely evaporated under vacuum and the vessel is flushed with methanol prior to analysis. This is very time-consuming because evaporation of formamide takes about 2 weeks. In contrast, the MECK technique detects nucleobases in their equilibrium concentration in the liquid phase. These methods are complementary and have become standard in our studies (Zemankova et al., 2021). Table 1 compares relative synthesis yields in the presence of iron-rich smectites or ZnCd QDs and their precursors (marked as a Zn:Cd) under the same conditions (160°C for 24 h).
Results of Our Experiments Determined by GC-MS and MEKC Analysis
The yields of products (P, A, G, C) are relative comparison to experiment without ZnCd QDs or Zn:Cd.
U was detected only by MEKC in this study (Current paper).
A = adenine; C = cytosine; G = guanine; GC-MS = gas chromatography–mass spectrometry; MEKC = micellar electrokinetic chromatography; P = purine; U = uracil.
3.3. ZnCd QDs peroxidase-like activity
Our results also demonstrate that ZnCd QDs are able to mimic peroxidase in the presence of H2O2 by oxidation of the 3′,5,5′-tetramethylbenzidine (TMB) substrate, commonly used for enzyme-linked immunosorbent assay for demonstration of peroxidase activity (Chen et al., 2019; Mohamad et al., 2019). The interaction of TMB with ZnCd QDs (red, yellow, and blue emission) in the presence of H2O2 leads to the formation (under visible light condition and about 10 min irradiation) of a deep-blue-colored product with a strong absorption peak at λ = 652 nm shown in Fig. 3A. The time dependence (60 min duration) of the TMB interaction with ZnCd QDs (red, yellow, and blue emission) in mode 10 min irradiation by visible light (VL ON) and/or 10 min in dark (VL OFF) is demonstrated in Fig. 3B. Best results were achieved for the case of ZnCd QDs with red emission, because bigger QDs are able to better absorb (utilize) the VL from a LED lamp. In Fig. S6A is shown the comparison of excitation spectra (λem = 600 nm, λex = 250–570 nm, Fig. S6Aa) of ZnCd QDs with emission spectrum of the LED lamp, Fig. S6Ab. Excitation spectra of ZnCd QDs demonstrate the ability of ZnCd QDs to absorb visible light from a LED lamp (Fig. S6Ac) and lead to TMB oxidation. The decrease of absorption signal at λ = 652 nm (VL OFF mode) is associated with TMB reduction by NH3 or -SH groups from MSA, Fig. 3C. These types of reversible (oxidation/reduction) reactions are crucial for forming the first sustainable proto-metabolic pathways.
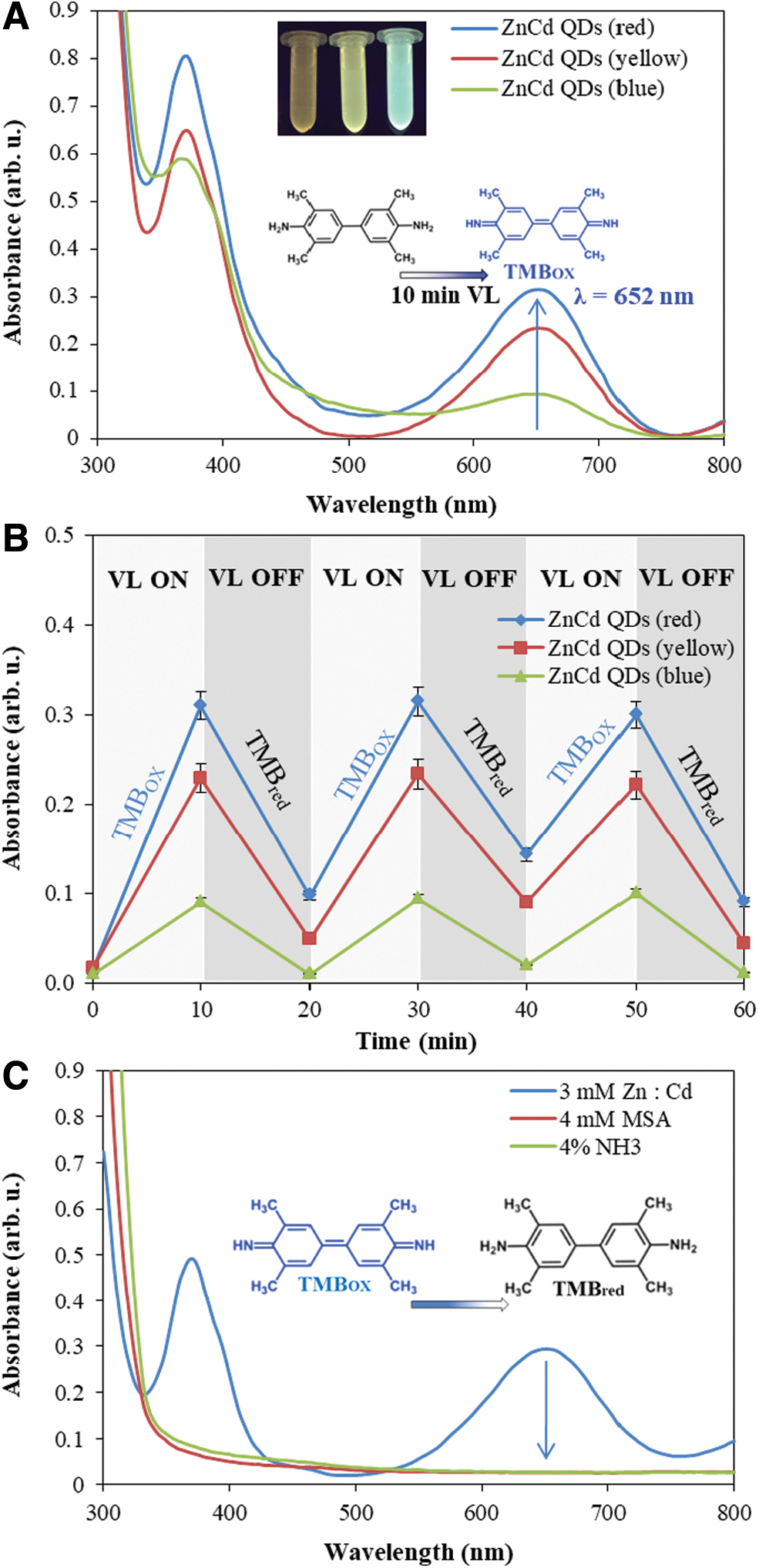
(
The size of ZnCd QDs strictly depends on the ratio of precursors (Zn/Cd/MSA), NH3 concentration, and the duration of UV irradiation (Nejdl et al., 2020). The absorption of visible light leads to the formation of reactive oxygen species, such as the superoxide anion (˙O2 -), hydroxyl radical (˙OH), singlet oxygen (1O2), and also (photogenerated) holes (h+) (Liu et al., 2020). Excited electrons have a strong reduction potential (generate reactive oxygen species) and (h+) are very strong oxidizing agents as we demonstrated in the case of TMB. These photocatalytic nanozymes can be prepared from prebiotically plausible materials like thiols, metals, and ammonia in formamide in one pot and controlled with light. The redox (nanozymatic) versatility (controlled by light) of ZnCd QDs is also crucial for the first sustainable proto-metabolic pathways.
4. Discussion
For our experimental exploration of the aspects summarized by (I) de novo synthesis of QDs, (II) their catalytic effects, and (III) peroxidase-like activity, see Section 3. Results truly implicate potential relevance of ZnCd QDs to the hot spring origin-of-life scenario suggested by Damer and Deamer (2020) or to possible extension of this theory toward chemistry of a hydrothermal environment in post-impact craters (Pastorek et al., 2019). Particularly, QDs might have played a role in prebiotic evolution in hot springs and lagoons exposed to UV radiation (Patel et al., 2015; Damer and Deamer, 2020) and, as further discussed, also in establishing the first proto-metabolic reactions.
4.1. Origin of QDs and their catalytic properties
The results demonstrate that formation of nanoparticles from a solution of corresponding salts of Zn and Cd is a simple process either in water or in the presence of formamide, an important prebiotic feedstock molecule. Alongside natural occurrence (Hochella et al., 2008; Maddenc, 2012; Pavlov et al., 2014; Bundschuh et al., 2018), we show that QDs can form in salt solutions simply upon UV irradiation in the presence of one of the fundamental feedstock prebiotic compounds: formamide. The origin of QDs may therefore be relevant to salty, mineral-rich formamide-containing lagoons that are considered to be a plausible environment for prebiotic synthesis on the surface of early Earth or Mars (Bizzarri et al., 2021).
Our investigations have also shown that emergent QDs can potentially participate in the synthesis of nucleobases in these harbors of prebiotic chemistry. Our experiments indicate that not only can they be synthesized in the presence of formamide but also they do not interfere with ongoing synthesis of prebiotic substances. In comparison to previous experiments with heating formamide in the presence of iron-rich smectites (Pastorek et al., 2019), influence of QDs on nucleobase yield is comparable or even more positive. Particularly, QDs increase the yield of cytosine synthesized by a factor of 300 compared to neat formamide thermolysis, which implicates their potential role in the origin of pyrimidine base worth to be further explored in the future regarding also other types of QDs containing other far more abundant elements in meteorites such as Fe, Cu, Ni, Cr, Ti, and Mn (Jarosewich, 1990).
Another challenge is posed by prebiotic relevance of mercaptosuccinic acid used in the current work for stabilizing ZnCd QDs. Although this fact indirectly implicates its possible role in prebiotic chemistry of QDs, related compound, the succinic acid, has long been considered an important constituent of the prebiotic broth (Negronmendoza and Ponnamperuma, 1982). A recent study illustrates its facile prebiotic synthesis in an Fe-catalyzed chemistry (Muchowska et al., 2019). Nevertheless, our previous study also illustrates that ZnCd QDs may be also alternatively stabilized by cysteine (Assi et al., 2021). Cysteine was not observed among products of the Miller-Urey experiment conducted in the presence of H2S (Parker et al., 2011), but a recent study outlines a prebiotic route to this compound (Foden et al., 2020).
4.2. Proto-enzymatic reactions
In addition, the optoelectronic properties of nanoparticles, which could be activated by photons over a wide range of electromagnetic wavelengths, could prompt a wide range of reactions toward prebiotic evolution. We suggest that in the first step of chemical evolution, nanoparticles probably catalyzed prebiotically relevant reactions in the same way as minerals or smectites but, in a next step, the nanoparticles could behave as nanozymes and form (or improve) a proto-metabolism leading to sustainable metabolic pathways, as described in the next section.
Prebiotic proto-enzymatic reactions are widely discussed (Mamajanov et al., 2020). Rowena Ball and John Brindley assume that early Earth was perfused with a flow of hydrogen peroxide (H2O2) from a sustained external source, which powered and mediated molecular evolution and the proto-cellular RNA world (Ball and Brindley, 2019). This chemistry is potentially closely linked to primitive proto-enzymatic behavior of ZnCd QDs when serving as nanozymes activated by light.
Our experimental results clearly show that ZnCd QDs can serve as a nanozyme agent instead of peroxidase, and it has already been observed that QDs mimic also the restriction endonuclease enzyme (Sun et al., 2018). It should be noted that QDs are also used for improving the product yield and specificity of polymerase chain reactions (Ma et al., 2009; Sang et al., 2015). These findings support our assumption that abiotic artificial materials (nanozymes) could be connected to the RNA world.
Also, QDs have been emerging as ideal light absorbers (Li et al., 2018, 2019), photosynthetic reaction centers (light harvesters) (Nabiev et al., 2010; Werwie et al., 2012, 2018), and photocatalysts (Jensen et al., 2016), and they are able to mimic electron transport chains (Ma et al., 2013; Harvie et al., 2018). QDs are able to keep their properties under a wide range of conditions starting from ambient and ending with considerably harsh environments (Ko et al., 2020). This robustness and stability might have potentially comprised an important advantage provided by QDs (if they truly participated in prebiotic evolution) to the first living chemical system for its survival under harsh conditions and demonstrates that QDs are interesting because of not only their chemical diversity and functionality but also their stability in aqueous and non-aqueous solvents (Hazen and Sverjensky, 2010; Wei and Wang, 2013; Walther et al., 2019; Wu et al., 2019; Mamajanov et al., 2020).
4.3. Toward a Nanoparticle World?
Post-impact hydrothermal sites that might have existed for hundreds of thousands years in craters on early Earth and Mars (Abramov and Kring, 2005; Schwenzer et al., 2012; Osinski et al., 2013), likely also on Venus (Taylor and Grinspoon, 2009; Way and Del Genio, 2020), including the broader boundaries of a volcanic hydrothermal lagoon environment, are anticipated by the hot spring hypothesis to be plausible localities for the origin of life (Damer and Deamer, 2020). In our previous study, we have demonstrated that various iron-rich smectites (Pastorek et al., 2019) are suggested to form in volcanic environments, or by water alteration of impact craters, support the synthesis of adenine (A), guanine (G), cytosine (C), thymine, uracil, purine (P), urea, hypoxanthine, guanidine, and glycine following the thermal treatment of formamide.
Following this evidence, we can also hypothesize that post-impact hydrothermal chemistry on early Earth could have triggered the formation of metal nanoparticles from eroded metal-rich material. This may happen upon UV radiation not long (on a geological time scale) after a deep impact event. The atmospheric dust residence time is in fact about 104 times shorter lived than the post-impact environment: We compare, for example, a 30-year dust residence time following the K/T event (Brugger et al., 2017) and the 300,000-year duration of hydrothermal activity in Gale Crater (Schwenzer et al., 2012). Thiols, in the role of an important agent in QD synthesis, might have been produced by hydrothermal fluids in a nonbiological reduction of hydrothermal inorganic carbon (Reeves et al., 2014). The suggested scenario is depicted in Fig. 4. We suggest that a labyrinth of micropores in a hydrothermal vent environment might create excellent conditions for increasing chemical complexity in the presence of potentially catalytic minerals and rocks such as meteorite residue (Sojo et al., 2016), iron-rich smectites produced by complete erosion (Pastorek et al., 2019), and also QDs (Kitadai et al., 2018). On the other hand, there are several limiting factors: The formation of QDs other than ZnCd has not been explored under prebiotically relevant conditions. The influence of QDs on prebiotic chemistry starting from hydrogen cyanide should be also examined along with their complex nanozymatic activity under prebiotic conditions.
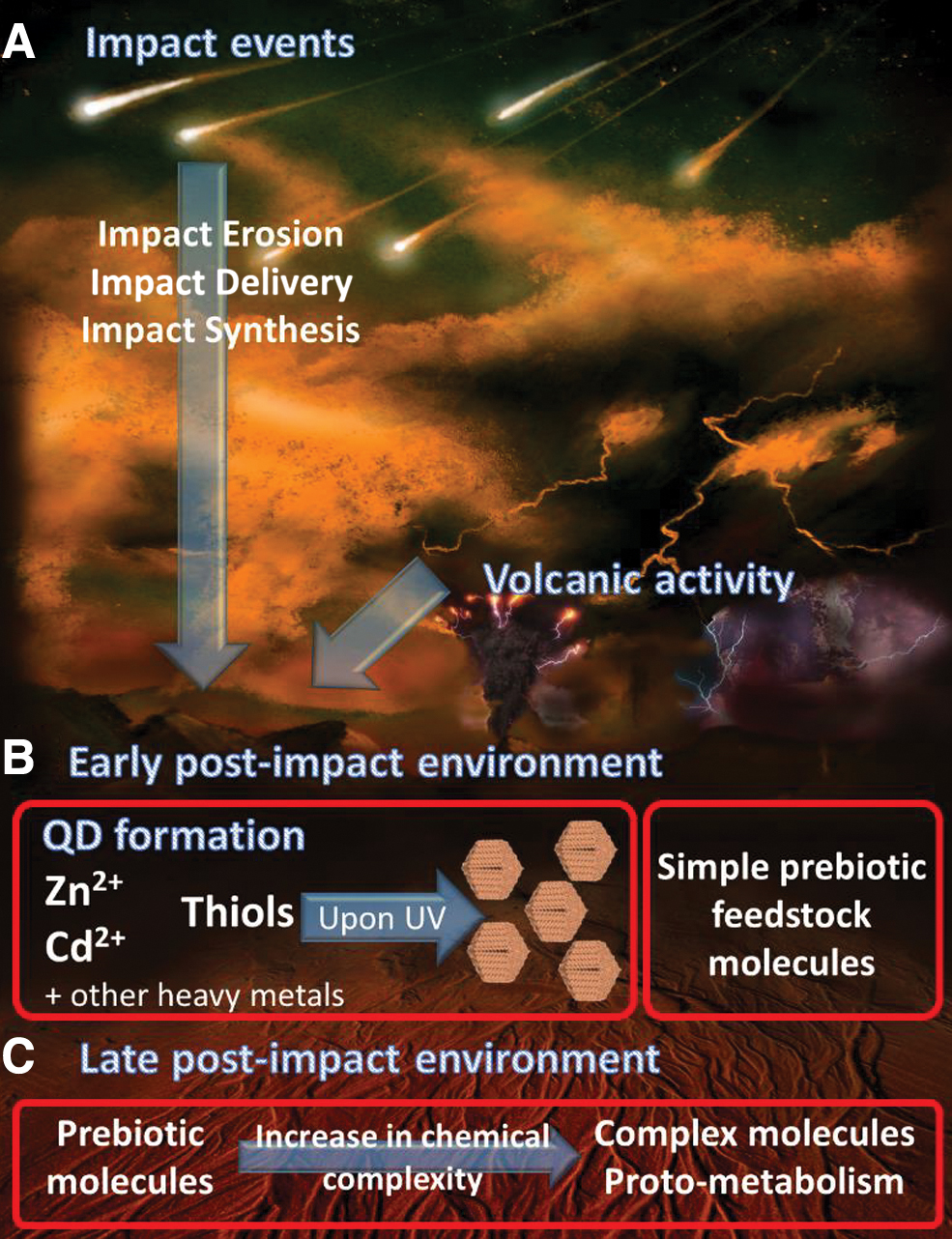
A tentative and simplified story of the Nanoparticle World. (
Based on our findings, we also suggest that the products of prebiotic chemistry may be then adsorbed on the surface of a mineral catalyst and be subjected to different chemistry than when present in the liquid phase. Prebiotic molecules are adsorbed due to the size and properties of the nanoparticles present. Heavier and larger pieces of minerals or clays likely occur at the bottom of prebiotic ponds. In contrast, an advantage of nanoparticles is posed by their possible dispersion evenly throughout the liquid phase. Thus, prebiotic molecules may adsorb on nanoparticles just several centimeters below the surface and be exposed to UV or visible radiation that is not able to penetrate into bottom sediments of the pond or lacustrine environment.
5. Conclusions
The present work demonstrates that the prebiotically relevant parent molecule formamide is a suitable solvent for UV-driven synthesis of ZnCd QDs. ZnCd QDs synthesized in formamide do not interfere with, or actually increase, the yield of formamide-based nucleobase synthesis. These experiments also demonstrate the light-harvesting ability of ZnCd QDs that mimic peroxidase-like activity upon visible-light irradiation. In this way, we confirm their potential role as nanozymes. Our experimental results tentatively support the plausibility of a role for QDs in prebiotic chemistry.
Footnotes
Acknowledgments
This research was founded by the Internal Grant Agency of Mendel University in Brno IGA MENDELU 2019_TP_009 and also by CEITEC 2020 (LQ1601) with financial support from the Ministry of Education, Youth and Sports of the Czech Republic under the National Sustainability Programme II. Martin Ferus acknowledges grant no. 19-03314S of the Czech Science Foundation. Laboratory of high resolution spectroscopy acknowledges ERDF/ESF “Centre of Advanced Applied Sciences” (No. CZ.02.1.01/0.0/0.0/16_019/0000778).
Author Disclosure Statement
No competing financial interests exist.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Abbreviations Used
Associate Editor: Christopher McKay
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
