Abstract
Potentially habitable zones have been discovered on Mars today in underground areas containing perchlorate brines. Understanding the low-pressure adaptations of microorganisms is essential in learning more about what life could potentially be found on Mars today or could have existed in the distant past. Many studies have looked at low-pressure adaptations in bacteria; however, studies aimed at understanding these adaptations in archaea are scarcer. Haloferax volcanii is a species of halophilic archaea documented to tolerate high concentrations of oxidizing agents present on Mars (i.e., perchlorates and nitrates). In this study, we expose H. volcanii to a hypobaric (2.4 kPa) and an anoxic CO2-enriched atmosphere in the presence of perchlorate, chlorate, and nitrate. While no growth was observed during incubation in these conditions, survivability was increased in cultures incubated in low-pressure atmospheric conditions compared to ambient Earth atmospheric pressures. Scanning electron microscopy observations showed morphological changes in low-pressure conditions not observed at ambient Earth atmospheric pressures. Results suggest that previously undocumented low-pressure adaptations in H. volcanii increase survivability in simulated subsurface martian conditions. Future experiments to understand the changes in gene expression under these conditions may be valuable to understand more about low-pressure adaptations in archaea.
1. Introduction
In the search for life on Mars, the gold standard has been to follow the water. Wherever liquid water is found on Earth it is almost always accompanied by the presence of life (Landis, 2001). Evidence from the geologic history of Mars points to large amounts of liquid water that existed on the planet's surface several billion years ago when atmospheric conditions were more favorable. The possible Earth-like conditions of Mars' distant past may have given rise to life (Squyres et al., 2004), but today the surface of Mars is not the ideal place for life to exist. For life as we know it to currently exist on Mars, the life-forms would need to overcome multiple “Earthly” biocidal conditions including a hypobaric (∼0.7 kPa avg) anoxic (∼96% CO2) atmosphere; low average surface temperatures (−61°C); regolith with low water activity; presence of oxidizing compounds (e.g., perchlorates); and a UV-irradiated surface, solar particle events, and galactic cosmic radiation (Schwendner and Schuerger, 2020). We often associate Earth's ability to support life with the presence of water. In comparison, on Mars the average surface pressure of 0.7 kPa is below the vapor pressure of pure liquid water, and the average temperature, -63°C, is below the freezing point of water. This makes the existence of pure liquid water on the surface of Mars unlikely and presents a hard barrier for the existence of life (Haberle et al., 2001).
In 2002, data from the Odyssey orbiter confirmed the current existence of subsurface water ice deposits on Mars (Boynton et al. 2002); and in 2008, data from the Phoenix lander confirmed the existence of water ice currently on the surface in the polar regions of Mars (Smith et al., 2009). In 2015, the existence of hydrated salts of perchlorate and chlorate were discovered in the equatorial regions of Mars (Ojha et al., 2015). And in 2018, it was announced that a perchlorate brine lake had been discovered ∼1.5 km underground by satellite radar observations. For this subsurface body of water to be detected with the methods used in these observations, the site of interest needed to be several kilometers wide, indicating limitations in the detection method that could omit smaller bodies of water. These findings leave open the possibility of habitable brine lakes being spread across the entire subsurface of Mars (Orosei et al., 2018). The possibility of subsurface water on Mars has been suggested as a source for the water in the hydrated perchlorate salts present on the surface (Abotalib and Heggy, 2019). In addition to the presence of liquid brine, the subsurface would also provide possible protection from the high levels of UV radiation present on the surface (Moores et al., 2007; Godin et al., 2019).
Halophiles, prokaryotes that thrive in environments of high salinity, are of high astrobiological interest due to their ability to thrive in high salt concentrations, high concentrations of oxidizing agents found on the martian surface (e.g., perchlorates and nitrates), and their high resistance to UV and gamma radiation (Oren, 2014). In the Atacama Desert (South America), an Earth analogue of martian conditions, subterranean halophilic microbial communities benefit from the hygroscopic properties of perchlorate and other salts (e.g., nitrates), which provide a source of water in an otherwise hyperarid environment (Davila et al., 2008; Parro et al., 2011). Pilot Valley Basin in Utah is another Earth analogue of martian conditions due to its naturally high perchlorate content in soil. At this site, research has shown evidence of native microbial communities that are able to break down the perchlorate present in soil and use it as a terminal electron acceptor (Lynch et al., 2019). Laboratory studies of halophiles in the presence of perchlorate salts have also yielded very notable results. For example, Planococcus halocryophilus was observed to have increased survival rates in the presence of perchlorate brines at lower temperatures (range of +25°C to −30°C), and higher salt concentrations were observed to increase survival rates during freeze-thaw cycles (Heinz et al., 2018). Oren et al. (2014) documented the ability of several species of haloarchaea to grow anaerobically on perchlorate and chlorate. In addition, haloarchaea exposed to spaceflight conditions are capable of surviving both high vacuum and UV radiation. In particular, Haloferax volcanii is capable of surviving vacuum UV radiation up to 150 J m−2 (Abrevaya et al., 2011). Furthermore, dormant haloarchaea have been isolated from halite that was determined to be 195–250 million years old (Fendrihan et al., 2006).
While exobiology studies are focused on the implication for microbial life outside of Earth, planetary protection studies focus on assessing the threat to Mars from Earth microorganisms hitching a ride on spacecraft. A study focused on planetary protection showed growth of 20 species of bacteria isolated from soil under hypobaric (0.7 kPa) and anoxic (CO2-enriched) conditions, but no archaea nor fungi were successfully isolated under these conditions (Schuerger and Nicholson, 2016). The highest surface pressure on Mars is thought to be 1.2 kPa in Hellas Basin (Haberle et al., 2001). For potential organisms in a subsurface aquifer, the pressure may vary greatly from those on the surface. The pressure exerted on potential organisms would increase in relation to depth and the density of the overlying material (Schuerger et al., 2013). Haloferax volcanii is a halophilic archaeon capable of denitrification (Hattori et al., 2016), growing anaerobically on chlorate and able to tolerate high concentrations of perchlorate (Oren et al., 2014). The primary objective of these experiments was to further exobiology research by assessing the ability of Haloferax volcanii to grow and survive in conditions that mimic subsurface brines on Mars.
While previously published research has revealed the surface conditions of Mars to be very hostile to terrestrial life, the goal of this research is to test whether subsurface environments may be a suitable habitat for potential extant microbial life on Mars. To test our hypothesis, H. volcanii was incubated in a hypobaric (2.4 kPa) and anoxic (CO2-enriched) atmosphere in the presence of sodium perchlorate, sodium chlorate, and sodium nitrate. This study used scanning electron microscopy (SEM) to observe cell morphology under the different atmospheric conditions tested.
2. Materials and Methods
2.1. Media and electron acceptors
Haloferax volcanii (ATCC 29605) stock cultures were grown in modified ATCC-974 liquid media containing 125 g L−1 NaCl; 50 g L−1 MgCl2 · 6H2O; 5 g L−1 K2SO4; 0.2 g L−1 CaCl2 · 2H2O; 5 g L−1 casamino acids; and 5 g L−1 yeast extract. Trace elements and chelated iron solutions were prepared as in the work of Hattori et al. (2016). Trace elements solution contained 0.1 g L−1 Na2MoO4 · 2H2O; 0.2 g L−1 MnCl2 · 6H2O; 2 mg L−1 CoCl2 · 6H2O; 0.1 g L−1 ZnSO4 · 7H2O; and 0.1 g L−1 CuSO4 · 5H2O. Chelated iron solution contained 0.1 g L−1 FeSO4 · 7H2O and 0.1 g L−1 EDTA. One milliliter of each solution was added to 1 liter of media. The pH was adjusted to 7.0 with 1.0 M NaOH and autoclaved for sterilization.
NaNO3, NaClO3, and NaClO4 solutions were filter-sterilized with Luer lock 0.22 μm hydrophilic polyvinylidene fluoride (PVDF) filters and added to the sterile modified media for a final concentration of 100 mM for the desired electron acceptor.
2.2. Experimental design
Two sets of conditions were used in these experiments. Cultures were either placed in the experimental conditions at 21°C in an anoxic (CO2-enriched) hypobaric atmosphere (2.4 kPa) or the control conditions at 21°C in anaerobic conditions under ambient atmospheric pressure (101.3 kPa). Cultures were incubated in the presence of 100 mM either NO3 -, ClO3 -, ClO4 -, or no-electron acceptor. All cultures were incubated for 113 days. The details of the experimental conditions are described below.
2.2.1. Incubation at 21°C in hypobaric (2.4 kPa) and anoxic (CO2-enriched) atmospheric conditions
Experimental design was heavily inspired by Schuerger et al. (2013). A vacuum desiccator was used to create the hypobaric (2.4 kPa) and anoxic (CO2-enriched) environment (Fig. 1A, 1B). For the purpose of this study, CO2-enriched refers to an environment with gas composition that is primarily carbon dioxide. In comparison, Mars' atmospheric gas composition is primarily (∼96%) carbon dioxide (Schwendner and Schuerger, 2020). To create an anoxic CO2-enriched atmosphere similar in composition to Mars, three BD GasPak EZ Anaerobe packs were placed in the desiccator, and a gas cylinder containing 99.9% CO2 was used to flush the system by evacuating the chamber to 2.4 kPa and pressurizing it with CO2. This was repeated four times, leaving the gas composition of the chamber primarily carbon dioxide. After the final flush, the system was sealed off with the atmospheric pressure at 2.4 kPa. The pressure was monitored every other day, and adjustments were made by applying vacuum pressure when necessary if there was a variation from 2.4 kPa. The CO2 generation packets activate and begin to heat from the presence of oxygen. After the oxygen is removed, the packets stop heating. At the conclusion of the experiment, the chamber was pressurized and opened. After opening the chamber, the packets began heating again, indicating the environment remained anaerobic and any oxygen that had leaked in during the experiment was converted to CO2.

A vacuum desiccator was used to incubate the cells in low-pressure (2.4 kPa) and anoxic (CO2-enriched) conditions. (
Flat-bottom test tubes were sterilized in an autoclave. A total of 5 mL of sterile media was added to each tube followed by 100 μL of “preculture.” Preculture was defined as healthy cells grown at 30°C for 1 week in ATCC-974 liquid media.
Triplicate sets of tubes were prepared containing one of the three electron acceptors. Tubes containing no-electron acceptor were used as controls to measure growth from dissolved oxygen. Potential growth on electron acceptors was measured from growth from dissolved oxygen as a starting value.
To prevent contamination of the culture tubes during the flushing procedure, sterile 0.45 μm mixed cellulose ester membrane (47 mm diameter) filters were used in place of caps. The filters were aseptically placed on top of the tubes and secured in place with parafilm (Fig. 1C). Controls of blank media (in triplicate) were included to ensure this procedure prevented the introduction of contamination. At the conclusion of the experiment, 1 mL was removed from the blank media sample and stained with the nucleic acid dye 4′,6-diamidino-2-phenylindole (DAPI) to confirm no contaminants were present. Briefly, 1 mL of the sample was added to 10 μL of 10−3 M DAPI solution and 25 μL of a 10% formalin solution prepared in a solution of ATCC-974 media salts. These samples were incubated for 30 min and then run through a black 0.22 μm filter (Millipore GTBP02500). The filter pad was mounted on a glass microscope slide and observed by using a Zeiss Axio Scope.A1 epifluorescence microscope. This method was effective, as no contaminants were observed in blank media at the end of the experiment.
Water loss was monitored throughout the experiment because the pressure of the chamber was near the vapor pressure of water at the incubation temperature. After inoculation and filter cap placement, a mass measurement was taken for each individual test tube immediately before being placed into the vacuum desiccator. Final mass measurements were taken at the conclusion of the experiment, and water loss was calculated by the difference in mass. The tubes were adjusted to their initial volumes by using 0.22 μm filter sterilized buffer solutions. Buffer solutions were prepared at the calculated final salt concentration of the media (i.e., ATCC-974 salt solution) in order to minimize the disruption of cells in the tubes by changing the osmotic pressure. To ensure this procedure was an accurate measure of water loss given potential changes of scale calibration over time, empty test tubes with filter caps were included with the experimental set where initial and final mass measurements were taken. Differences in the control tubes were ±0.001 g, indicating reliable scale calibration.
2.2.2. Incubation at 21°C under anaerobic ambient (101.3 kPa) atmospheric conditions
Haloferax volcanii is a facultative anaerobe, and previous research has shown that when grown under anaerobic conditions in the presence of alternative electron acceptors it will first use up all dissolved oxygen in media. After the oxygen in the media is depleted, H. volcanii will switch over to other terminal electron acceptors if present. Therefore, there was no reason to reduce media to remove dissolved oxygen, as the cells will deplete the media of oxygen (Oren, 1991). To provide anaerobic conditions at 101.3 kPa, Hungate anaerobic tubes were utilized. The Hungate tubes were autoclaved to sterilize them; then 15 mL of sterile media was added to completely fill the tubes. Three-hundred microliters of preculture was added to triplicate sets of tubes containing one of the three electron acceptors (Na-nitrate, Na-chlorate, or Na-perchlorate) and tubes containing no electron acceptor. The cultures containing media with no added electron acceptor were used as controls for growth from dissolved oxygen. Potential growth on electron acceptors was measured from growth from dissolved oxygen in tubes that contained no other terminal electron acceptor, as a starting value. These cultures were incubated at 21°C and 101.3 kPa to serve as “Earth conditions” for the study.
2.3. Indirect measurement of growth
Growth was indirectly measured by using increases in optical density of the liquid broth indicating increased cell density as described by Oren et al. (2014). Briefly, 150 μL aliquots were taken in triplicate from each culture sample and placed into a 96-well microtiter plate. Samples were taken initially and at the completion of the experiment to determine the change in optical density (OD) at 630 nm over the course of the experiment. The optical density OD630 was measured spectrophotometrically with a BioTek ELx800 Absorbance Reader.
For any samples that had no change in OD630, final cell viability was determined by incubating the final culture aerobically at the optimum growth temperature of 45°C for 6 days. The OD630 of the culture was measured at the end of the 6-day period. Samples with an increase of OD630 were considered viable.
2.4. Measurement of perchlorate, chlorate, and nitrate concentrations
Initial perchlorate and nitrate concentrations were confirmed, and final concentrations were analyzed by using Perchlorate and Nitrate Ion-Selective Electrodes (Cole-Parmer Combination ISE) and an Oakton 450 pH/ISE meter. Aliquots of 300 μL were taken from each tube and were placed into a 10 mL beaker. The sample was then diluted by a factor of 25 with 7.2 mL deionized water. Then 150 μL of an ionic strength adjuster (2 M ammonium sulfate ISA) was added to the diluted 7.5 mL sample, giving the necessary 1:50 ratio of ISA to sample volume. The observed concentrations were multiplied by the dilution factor to determine the actual values. Theoretically, exactly 0.1 M or 9945 mg L−1 perchlorate in culture would be measured as 397.8 mg L−1. Likewise, exactly 0.1 M or 6200 mg L−1 nitrate in culture would be measured as 248.0 mg L−1.
Chlorate concentrations were measured with the spectrophotometric assay utilizing o-tolidine outlined in Urone and Bonde (1960). As for perchlorate and nitrate samples, dilutions were made to accommodate the different concentrations of chlorate.
2.5. Scanning electron microscopy observations
To examine possible cell morphology changes under test conditions, all cultures were viewed using scanning electron microscopy (SEM).
Cultures were centrifuged down, and pellets were collected and treated for SEM. Cells and biofilm were prepared by using a procedure modified from Liao et al. (2016). All steps involved used 1 mL of described reagents or solutions. First, pellets were rinsed three times with ATCC 974 salt solution that was prepared at the final concentrations of the culture media at the end of the experiment. They were then fixed in a 2% solution of glutaraldehyde in ATCC 974 salt solution at 4°C for 48 h. The pellet was rinsed again three times with ATCC 974 salt solution. Following a dehydration series of 30, 50, 70, 90, 100, 100, 100% ethanol, 2:1 ethanol/hexamethyldsilazane, 1:1 ethanol/hexamethyldsilazane, and 100, 100, 100% hexamethyldsilazane. In each step, the pellet was incubated for 10 min. After the final dehydration step, the pellet was collected on a black 0.22 μm filter (Millipore GTBP02500) and dried in a desiccator overnight. The filter pad was then mounted on an SEM sample stub and sputter-coated with Au/Pd alloy. Samples were observed with a Hitachi S-3500 variable pressure scanning electron microscope.
3. Results
3.1. Indirect measurement of growth
Cultures incubated under anaerobic conditions at 101.3 kPa and those incubated under the CO2-enriched conditions at 2.4 kPa did not grow on either the alternative electron acceptors or dissolved oxygen in the media (Table 1). Water loss was observed over the 113-day experiment. Water loss was highest in tubes with just Na-chloride and no supplemented oxyanions with ∼15.4% loss from the starting media volume. Water loss in the tubes containing oxyanion was lower with Na-chlorate at ∼13.8%, Na-perchlorate at ∼12.9%, and Na-nitrate at ∼12% (Supplementary Fig. S1).
Potential Growth after the 113-Day Incubation Period Was Indirectly Measured Using a Spectrophotometer
Data is reported as ΔOD 630, the change in the initial and final OD values. None of the cultures incubated under anaerobic conditions at 101.3 kPa or those incubated under the CO2-enriched conditions at 2.4 kPa showed a significant increase in OD values.
Since no growth was observed, cell viability after being removed from the chamber was determined by incubating all cultures aerobically at the optimum growth temperature of 45°C. Increases in OD630 were observed in all cultures when incubated at 45°C for 6 days (Fig. 2). The increase in OD630 suggests the viability of the cells remained at the conclusion of the experiment.
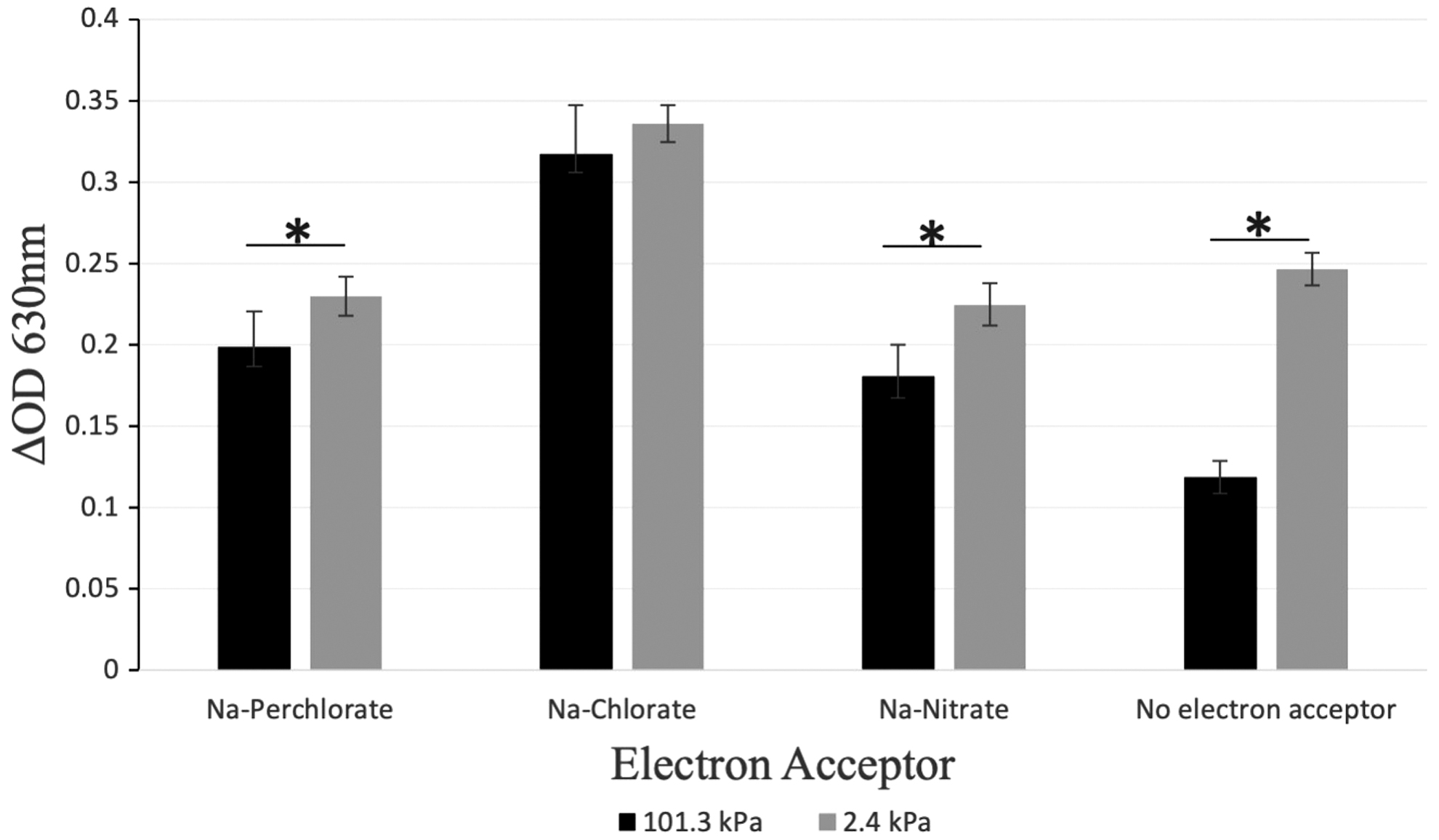
Cell viability was indirectly measured using a spectrophotometer. After 113 days of incubation, all cultures were placed at 45°C for 6 days under aerobic conditions. ΔOD 630 is the change in the initial and final OD values. *p < 0.05.
To ensure our methods were correct after no growth was observed following the 113-day incubation period, we grew H. volcanii in aerobic conditions at 21°C and performed replication experiments of those documented in the literature for anaerobic growth on chlorate (Oren et al., 2014) and nitrate (Hattori et al., 2016). We confirmed growth of H. volcanii in these conditions (Supplementary Fig. S2).
3.2. Measurement of perchlorate, chlorate, and nitrate concentrations
There was no observed change in the concentrations of perchlorate, chlorate, or nitrate at the end of the 113-day incubation under martian conditions. There was also no change after aerobic growth at 45°C.
3.3. Scanning electron microscopy observations
Before SEM sample prep, macroscopic observations after the experiment showed differences in both the consistency and color of the pellets. For the 2.4 kPa samples, the pellets from the Na-perchlorate and no-electron acceptor tubes had no red pigment left and were completely white. The Na-chlorate pellet looked semi-bleached as a white pellet with a pinkish hue. And the nitrate pellet retained its full red pigmentation. Biofilms from 2.4 kPa samples were filamentous and formed a less dense pellet than those from 101.3 kPa samples. All 101.3 kPa samples retained their full red pigmentation, looking similar to healthy stock cultures.
Scanning electron micrographs of all cultures incubated under anaerobic conditions at 101.3 kPa (Fig. 4A, 4C, 4E, 4G) exhibited similar features to those of healthy preculture cells and biofilm (Fig. 3). Standard cell morphology (Fig. 3) is pleomorphic with a mixture of sphere- and rod-shaped cells.
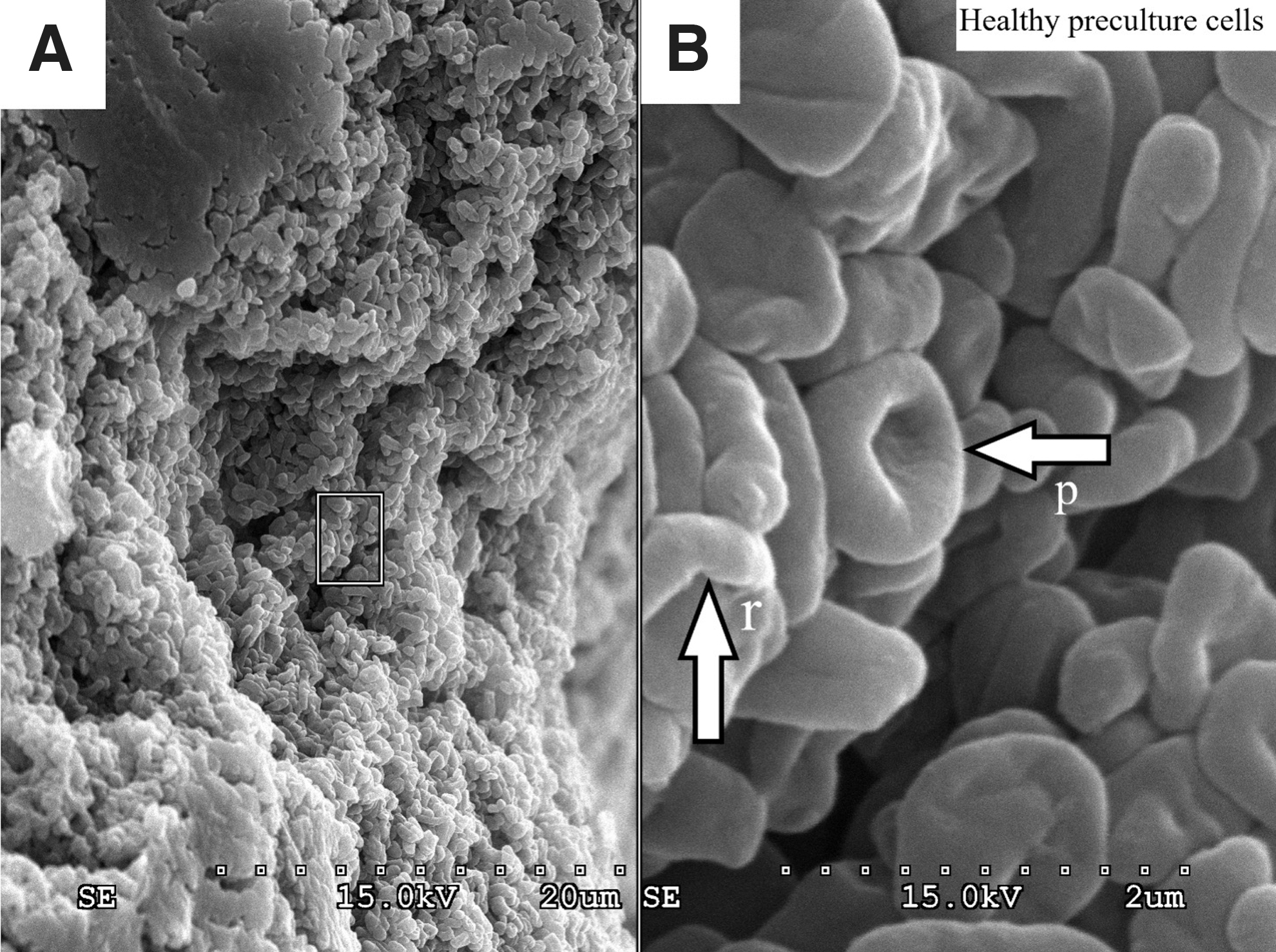
Normal morphology of Haloferax volcanii cells and biofilm. (

Morphology of Haloferax volcanii cells and biofilm under anaerobic conditions after the 113-day incubation period. (
Observations of cellular and biofilm morphology for cultures incubated under the CO2-enriched 2.4 kPa atmospheric conditions showed clear differences from standard morphology. Those incubated in the presence of 100 mM Na-nitrate showed a less dense biofilm with less cell compaction and a filamentous substance throughout the cells and biofilm matrix (Fig. 4B).
Those incubated in the presence of 100 mM Na-perchlorate showed a mixture of normal-sized spherical cells and shrunken spherical cells. The filamentous substance was also observed but not at the frequency of that observed in the nitrate cultures (Fig. 4D).
In both the Na-chlorate and no-electron acceptor cultures, extremely dense biofilm was observed with cells deeply embedded in the biofilm matrix (Fig. 4F, 4H).
4. Discussion
It was not surprising that Haloferax volcanii did not grow anaerobically on Na-perchlorate, as it is not known to be capable of doing so (Oren et al., 2014). It was surprising to observe no growth of H. volcanii because previous research has shown it is capable of denitrification at 37°C (Hattori et al., 2016) and anaerobic growth on Na-chlorate at 35°C (Oren et al., 2014). Our results indicated no growth in the presence of these electron acceptors under ambient atmospheric pressure at 21°C. We hypothesized the lack of growth was due to the lower temperature in our study. To support our assumptions, we performed replication experiments as described in the works of Oren et al. (2014) and Hattori et al.(2016) at 37°C and 35°C. These incubation temperatures yielded H. volcanii growth in Na-nitrate and Na-chlorate containing media (Supplementary Fig. S2). Haloferax volcanii is capable of growing aerobically at 20°C with a growth rate one-quarter of that at the optimum temperature of 45°C (Mullakhanbhai and Larsen, 1975). We also confirmed aerobic growth at lower temperatures (Supplementary Fig. S2). The results of our temperature experiments suggest the lower temperatures are inhibiting anaerobic growth of Haloferax volcanii. Our findings that suggest growth is inhibited in anaerobic conditions at 21°C are an observation that has not been previously documented in the literature.
All initial experimental cultures were inoculated with the same amount of preculture, allowing for more direct measure of the differences in viability between the atmospheric conditions. While no growth was observed after the 113-day incubation, an interesting observation was made when looking at the growth at 45°C during the viability assessment. Cell viability in Na-perchlorate, Na-nitrate, and no-electron acceptor treatments from 2.4 kPa cultures was significantly higher compared to 101.3 kPa cultures (Fig. 2). But there was no significant difference in cell viability between cultures incubated at 2.4 kPa and 101.3 kPa for the Na-chlorate treatment (Fig. 2). We suggest exposure to the 2.4 kPa low-pressure anoxic atmosphere induced a stress response that increased survivability compared to those incubated under ambient atmospheric conditions.
Scanning electron micrographs of cultures incubated under anaerobic conditions at 101.3 kPa (Fig. 4A, 4C, 4E, 4G) all looked very similar to those of healthy preculture stocks (Fig. 3), but differences become apparent in the micrographs of the cultures incubated at 2.4 kPa, as we see two stress responses of haloarchaea that have been previously documented in the literature. Firstly, we suggest the filamentous substance seen in Fig. 4B may be a stress response to the hypobaric conditions resulting in accumulation of excess extracellular DNA (eDNA) or could also be intracellular bridges that are being formed to enable increased genetic transfer. It is possible the observations in Fig. 4B are even a combination of both. For example, eDNA has been documented as a primary component of haloarchaeal biofilm (Fröls et al., 2012). Liao et al. (2016) described the extracellular lattice network between Halorubrum lacusprofundi cells that look remarkably similar to the filamentous substance seen in Fig. 4B. The substance used in the work of Liao et al. (2016) was degraded by DNase I, suggesting DNA as a primary component. Chimileski et al. (2014) documented the ability of H. volcanii to metabolize exogenous double-stranded DNA as a starvation response. Additionally, intracellular cytoplasmic bridges formed by H. volcanii are involved in bidirectional genetic transfer. SEM imaging of these bridges greatly resembles the substance seen in Fig. 4B (Rosenshine et al., 1989). A second stress response documented in haloarchaea is a starvation-survival response where haloarchaea shrink in cell size (Winters et al., 2015). Rod-shaped cells will also become spherical and miniaturize. This miniaturization results in spherical cells that are <1 μm in diameter (Winters et al., 2015). As another example, Halobacterium salinarum cells will shrink down to a size of 0.45 μm in response to low water activity (Fendrihan et al., 2012). This is clearly seen in Fig. 4D, as some of the H. volcanii cells incubated in the presence of Na-perchlorate can be observed as small as ∼0.5 μm. Additionally, the morphology of both the Na-chlorate and no-electron acceptor treatments at 2.4 kPa look similar to each other (Fig. 4F, 4H), where very dense biofilm with cells deeply embedded inside can be seen.
Because differences in morphology were observed exclusively in samples from 2.4 kPa cultures and the atmospheric conditions were the only difference between the two experimental sets, the differences may be attributed to the anoxic and hypobaric conditions. Additionally, the differences in morphology between the treatments at 2.4 kPa suggest a combination of both hypobaric/anoxic conditions and oxidative stress from the different oxyanions. Furthermore, as a stress response H. volcanii is known to form persister cells that are metabolically inactive and go dormant when lethal stress is introduced (Megaw and Gilmore, 2017). We hypothesize the lower temperatures inhibited anaerobic growth of cultures at 101.3 kPa and put cells into a metabolically inactive state. But our data also suggests the low pressure activated additional stress responses within cells, placing them into a metabolically inactive state that increased their survival compared to those at standard atmospheric pressure. Both viability OD data and SEM observations support this hypothesis.
Serratia liquefaciens has become somewhat of a model organism for bacterial growth under simulated martian conditions as it is the most studied organism under these conditions and has its complete genome sequenced (Schwendner and Schuerger, 2020). Initial studies involving Serratia liquefaciens under simulated martian conditions had focused on primarily observing the effects of these conditions and confirming growth (Schuerger et al., 2013). A recent study by Fajardo-Cavazos et al. (2018) characterized up-regulation and down-regulation of transcripts for the bacterium Serratia liquefaciens grown in a low-temperature (0°C), hypobaric (0.7 kPa), and anoxic (CO2-enriched) atmosphere. The study was the first to characterize changes in gene regulation for any microbial species under simulated martian conditions (Fajardo-Cavazos et al., 2018). Since H. volcanii is a model organism for the domain Archaea and its entire genome has been published (Hartman et al., 2010), it may be valuable for future experiments to use genomic and transcriptomic analysis to understand more about the stress responses this organism undergoes in hypobaric atmospheric conditions.
The potential habitability of special regions on Mars considers the probabilities of the presence of liquid water, biologically available energy sources, and biologically essential elements. The detection of water, inorganic carbon, and elements required for life make it essential to investigate the biological limitations of Mars' special regions (Stoker et al., 2010). Many experiments on microbial survival and growth in martian conditions focus on special regions on the surface of Mars. Unfortunately, they attempt to mimic the conditions present there using pressures higher than are possible on the surface of Mars today. Unless these experiments use pressures that are possible on the surface of Mars (0.2–1.2 kPa), they do not give an accurate representation of the environmental conditions they intended to mimic (Schwendner and Schuerger, 2020). Additionally, with the low likelihood that liquid water can exist on the surface (Rummel et al., 2014), it is difficult for surface simulation experiments to appropriately incorporate the use of liquid media. In our case, the experiment presented was designed to mimic hypothesized brines in the subsurface of Mars; therefore, the higher pressure of 2.4 kPa in this case was appropriate. Additionally, if the pressure was any lower than 2.4 kPa at 21°C, the liquid water in the media would begin to evaporate rapidly. This would cause the experiment to end well before the 113-day incubation period. To our knowledge, the study we present is the first endeavor documented to attempt to grow an organism from the domain Archaea in low-pressure conditions similar to those in the subsurface of Mars. The methods and design parameters we present in this paper may provide useful guidance for future Mars astrobiology experiments that seek to mimic potentially habitable subsurface environments on Mars.
Our results suggest Haloferax volcanii exhibits previously undocumented stress responses to low-pressure conditions that are comparable to potentially habitable zones in the subsurface of Mars. Further understanding the response this terrestrial microorganism has to simulated subsurface martian conditions has implications in preparing for the possibility of finding extant life on Mars. The harsh conditions on Mars today may not support life as we know it in a metabolically active state. However, future studies incorporating halophiles could increase the likelihood for finding microbial life lying dormant under the surface of Mars today. Our study further expands the hypothesis that underground aquifers on Mars could be microbial oases on an otherwise desert planet.
Footnotes
Acknowledgments
From St. Petersburg College we would like to thank Thao Nguyen, Chelsea Knapp, Chris Lue, Cliff Stodden, Adam Davis, Mira Janjus, Erin Goergen, Anna Tingley, and Charlie Crapple for all of their help and guidance. From the University of South Florida College of Marine Science we would like to thank Mya Breitbart, Bob Ulrich, John Paul, Makenzie Kerr, Chelsea Bonnain, Y Nguyen, Natalie Sawaya, Karyna Rosario, Natalia Lopez, Kema Malki, and Adrienne Hollister for all of their incredible advice and support. We are extremely grateful to Tony Greco for providing his expertise with SEM. Lastly, from the University of South Florida Tampa we would like to thank Xiaopeng Li, Guangqiang Yin, and Bo Song for their support.
Author Contribution
Adam Robinson and Shannon McQuaig Ulrich took part in all aspects of experimental design and planning. The experiments and data collection were performed by Adam Robinson. Adam Robinson and Shannon McQuaig Ulrich both took part in the manuscript writing process.
Author Disclosure Statement
The authors Adam Robinson and Shannon McQuaig Ulrich declare no conflicts of interest.
Funding
This research was funded in part by St. Petersburg College Foundation Innovation Grant.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Associate Editor: Radu Popa
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
