Abstract
Phospholipids are essential components of biological membranes and are involved in cell signalization, in several enzymatic reactions, and in energy metabolism. In addition, phospholipids represent an evolutionary and non-negligible step in life emergence. Progress in the past decades has led to a deeper understanding of these unique hydrophobic molecules and their most pertinent functions in cell biology. Today, a growing interest in “prebiotic lipidomics” calls for a new assessment of these relevant biomolecules.
1. Introduction
Here, we present a complete update of the prebiotic synthesis of phospholipids, and for the first time, we compared biochemistry pathways with bench chemistry and synthetic routes. First, we introduce prebiotic systems chemistry (Section 1.1), phospholipids as structural components in cells (Section 1.2), before the phospholipid world (Section 1.3), structure of a phospholipid, sn-glycerol, and the Cahn/Ingold/Prelog nomenclatures (Section 1.4), and geochemical scenario and energy sources (Section 1.5). The synthesis is divided into three parts corresponding to the three main structural components of phospholipids: synthesis of fatty acids under abiotic conditions (Section 2), synthesis of glycerol and glycerol precursors under abiotic conditions (Section 3), and prebiotic synthesis of complete and incomplete phospholipid esters (Section 4). Each section reports on synthesis under abiotic conditions, biosynthesis, and comparisons between synthetic routes and biochemical pathways. Finally, we discuss the philogeny of Archaea, Bacteria, and Eukarya from the last ancestor and Commonote commonote (Section 5).
1.1. Prebiotic systems chemistry
Prebiotic systems chemistry (Ashkenasy et al., 2017; Ruiz-Mirazo et al., 2017; Fiore, 2019), an emerging branch of chemistry, offers a new perspective to analyze the impact of the prebiotic synthesis of such molecules, including amphiphiles, for the emergence of life, a topic that has now been explored for more than 50 years (Oro, 1995; Lane and Martin, 2012). Two complete reviews on the chemical synthesis of natural phospholipids were written in the last 50 years (Eibl, 1984; Paltauf and Hermetter, 1994). From this period, significant progress has been made, especially in the field of prebiotic synthesis of phospholipids and total synthesis of phospholipids. However, no mention on their prebiotic synthesis was present, with some exceptions (Fiore and Strazewski, 2016).
The main purpose of synthetic chemistry is the preparation of pure samples of specific compounds by using well-defined reactions combined in multistep synthetic pathways. It involves the use of orthogonal protecting groups, of yield optimization, and of minimization of reaction steps. The paradigm of the pure compound has hindered the development of complex chemical systems and the investigation of networks of chemical reaction. From such systems, new, unexpected, and unpredicted, emergent properties could arise while never observed in any of the components alone. Systems chemistry and, for the present review, prebiotic systems chemistry are therefore an advanced way to do—and to think and rethink—chemistry.
Current science uses a top-down approach to establish the causes of a phenomenon, or in case of synthetic chemistry, the preparation of a compound, by following well-defined retrosynthetic pathways. Concerning the protocellular systems, the top-down approach consists in simplifying the machinery of contemporary cells to obtain a simple and efficient system that would resemble the primitive cells and could explain how they contributed to life emergence (Lancet et al., 2018; Lopez and Fiore, 2019). Systems chemistry instead emphasizes bottom-up approaches to create complex systems where properties can emerge coming from the combination of simple elements. The bottom-up approach allows describing the properties of living systems as due to the cooperation between simple molecules present in a chemical mixture. Such mixture was likely available in a prebiotic environment, alimented for a part with the meteorite components.
Primitive meteorites contain a variety of organic molecules, delivered to the Earth via a late accretion, thereby providing organic compounds important for the emergence of life or that served as a feedstock for further chemical evolution (Llorca, 2004). Formamide is a classical key example and among the simple molecules found in interstellar space, is emblematic as a monocarbon source of several biotic molecules such as nucleosides, sugars, and aminoacids (Saladino et al., 2015, 2018). Ethanolamine delivered to the Earth could have contributed to the assembling and early evolution of primitive membranes (Rivilla et al., 2021).
Acetonitrile (Belloche et al., 2008) and glycolonitrile (Zeng et al., 2019) from the interstellar medium could have been the starting materials for amino acid synthesis, while interstellar Z-cyanomethanimine (Rivilla et al., 2019), NH2OH (Rivilla et al., 2020), and urea (Jiménez-Serra et al., 2020) could have been a part of the natal materials for the synthesis of ribonucleotides. Thus, a disordered mixture of organic and inorganic molecules can form, under certain abiotic conditions, molecules (amphiphiles), although in limited quantities, that can assemble as an organized and semipermeable barrier from the outer environment (Dalai and Sahai, 2019; Gözen, 2021). This is what is hypothesized for protocell bilayers (Fiore et al., 2018; Lopez and Fiore, 2019; Lopez et al., 2020a, 2020b).
More recently, Albertsen et al. (2014) and Fiore et al. (2017) have demonstrated that prebiotic amphiphile mixtures are out-of-equilibrium systems as complex membrane bilayers are in evolved cells (Bruckner et al., 2009; Mansy and Szostak, 2009; Piedrafita et al., 2017; Altamura et al., 2020). This way of proceeding tends to reproduce the process that led to the outbreak of life and biology from chemistry (Strazewski, 2019a, 2019b). Prebiotic synthesis of phospholipids is an excellent example of a bottom-up approach.
1.2. Phospholipids as structural components in cells
Modern cells are surrounded by a plasma membrane, which contains cytoplasm and creates internal compartments (Saier and Stiles, 1975; Graham and Higgins, 1997). Proteins and lipids are components of the cell membranes (Saier and Stiles, 1975). Cell membranes, composed of a complex mixture of phospholipids, membrane proteins, and many other amphiphiles, are three-dimensional fluid molecular layers that define the boundaries and the substructures (Deamer, 2017). The role of these boundaries is to spatially distinguish, separate and colocate microenvironments while protecting and keeping them at defined concentrations (Fig. 1).
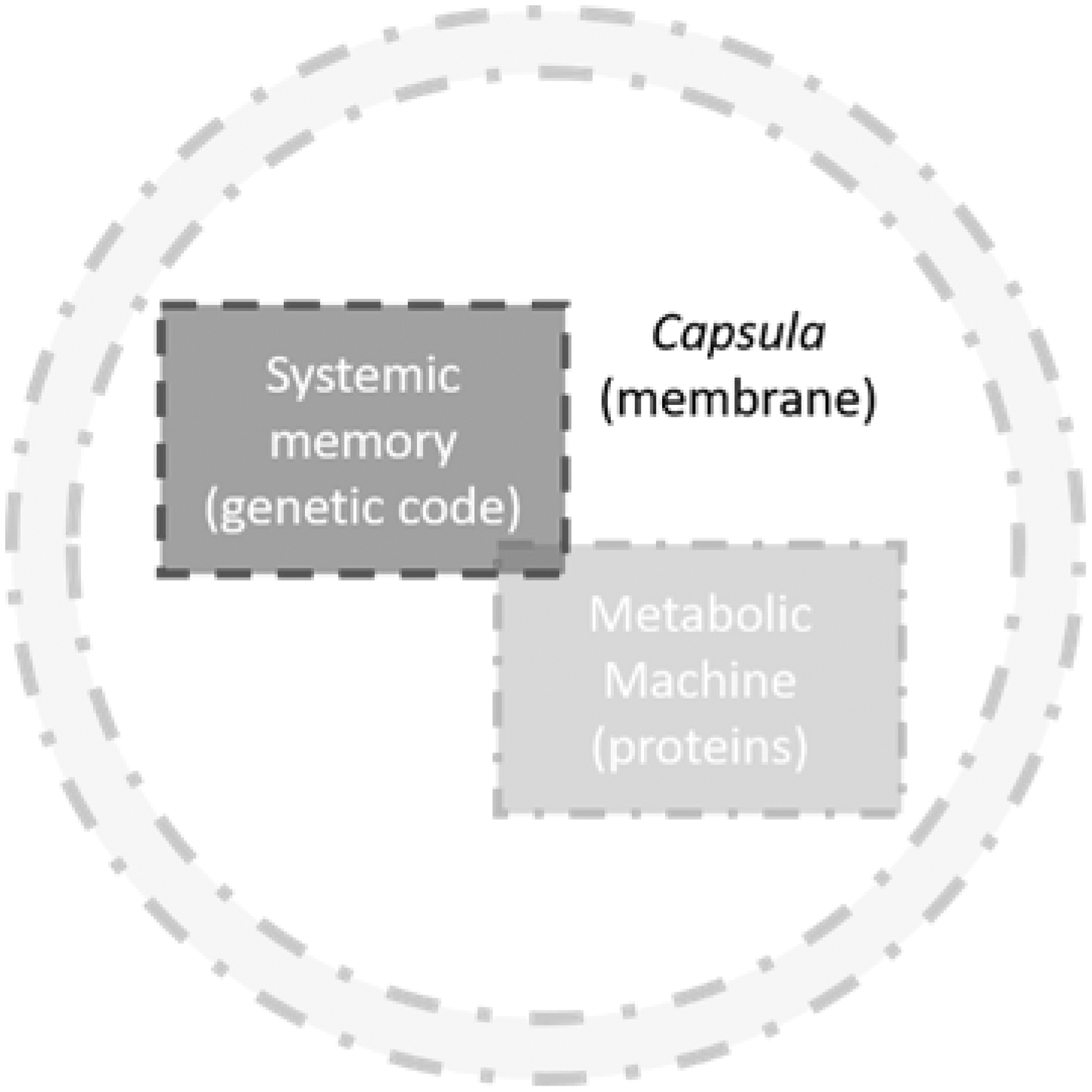
A simplified representation of a cell: the systemic memory (genetic code box) and the metabolic machinery (proteins) encapsulated together into a lipidic membrane (dashed circle).
Lipid boundaries enable a multitude of often competing and interfering biochemical reactions to occur simultaneously (Monnard and Walde, 2015; Deamer, 2017). Examples of biochemical reactions include the systemic memory (genetic code) and the metabolic machinery encapsulated together into a lipidic membrane (Fig. 1). In evolved cells, the genetic code is stocked in DNA, which is packaged by histones to form chromatin, which in turn is condensed to form chromosomes (Voet and Voet, 1995). The nuclei containing chromosomes are surrounded by a nuclear membrane (Voet and Voet, 1995). Mitochondrion, which is the site of several metabolic processes, is an organelle surrounded by two membranes (Voet and Voet, 1995).
Primitive membranes were probably heterogeneous giant vesicles (Stano and Luisi, 2010; Walde et al., 2010) that yielded to the ancestors of cell membranes, and provided a privileged environment to early reaction networks involving simple molecules enabling the formation of the earliest protocells (Ma and Feng, 2015; Lopez and Fiore, 2019). Among the large variety of amphiphiles plausibly present on the early Earth, phospholipids are rather complex. Although fatty acid vesicles have been extensively studied as early membranes, such vesicles are notably fragile and do not withstand high salinity, especially when divalent cations such as Mg2+ are involved (Cornell et al., 2019). Oleic acid vesicles were shown to leak when exposed to Mg2+ concentrations as low as 5 mM (Jin et al., 2018).
Such intolerance to salts that are essential to several (bio)chemical processes and likely present in early aqueous environments makes pure fatty acid vesicles poorly plausible. When vesicles were prepared with increasing fractions of phospholipid, the salt tolerance of the vesicles increased significantly, supporting the transition from early sensitive lipids to more robust ones (Kindt et al., 2020). Phospholipid selection among other vesicle-forming amphiphiles must have been based on specific, desirable physicochemical properties, and potentially derives from the formation of several intermediate lipids of increasing complexity (Budin and Szostak, 2011). Fatty acids form usually micellar structures, while phospholipids, as bilayer structures, yield more stable vesicles than fatty acids (Lopez et al., 2020a, 2020b).
1.3. Before the phospholipids world
Life likely began around 3.3–3.8 billion years ago (Glansdorff et al., 2008), in a ferruginous and anoxic environment that was replaced progressively by hydrogen sulfide. This atmospheric condition was maintained up to around 600 million years ago (Olson, 2019). H2S and SO2 were released into the atmosphere from hydrothermal vents and crust fissures or volcanos (Kasting et al., 1989). Geochemical HS− upon ultraviolet photolysis could contribute to the oxidation of α-hydroxy carboxylate to form α-oxo carboxylate, including oxaloactetate, pyruvate, α-ketoglurate, and glycoxylate, which are constituents of the Krebs cycle (Ritson, 2021).
The formation of large vesicles, precursors of protocells (Lopez and Fiore, 2019), occurred before the rise of full-fledged cells, since vesicles form spontaneously in an aqueous solution from a variety of surfactants (Walde, 2006). Closed membranes exert confinement and protection of an internalized chemical network, including reactions, on their hydrophobic region (Koga et al., 1998; Peretó et al., 2004; Douliez and Gaillard, 2014). According to the current view, early membranes were more likely formed from derivatives of alkanols (Fiore and Strazewski, 2016), fatty acids (Hargreaves and Deamer, 1978), monoalkyl and dialkyl phosphates (Fiore, 2018), monoalkyl sulfates (Hargreaves and Deamer, 1978), and isoprenoids (Ourisson and Nakatani, 1994). Most probably prebiotic protocell membranes were composed of a mixture of components (Jordan et al., 2019), including acyl glycerols that lacked phosphate headgroups (Fig. 2) (Fiore and Strazewski, 2016).
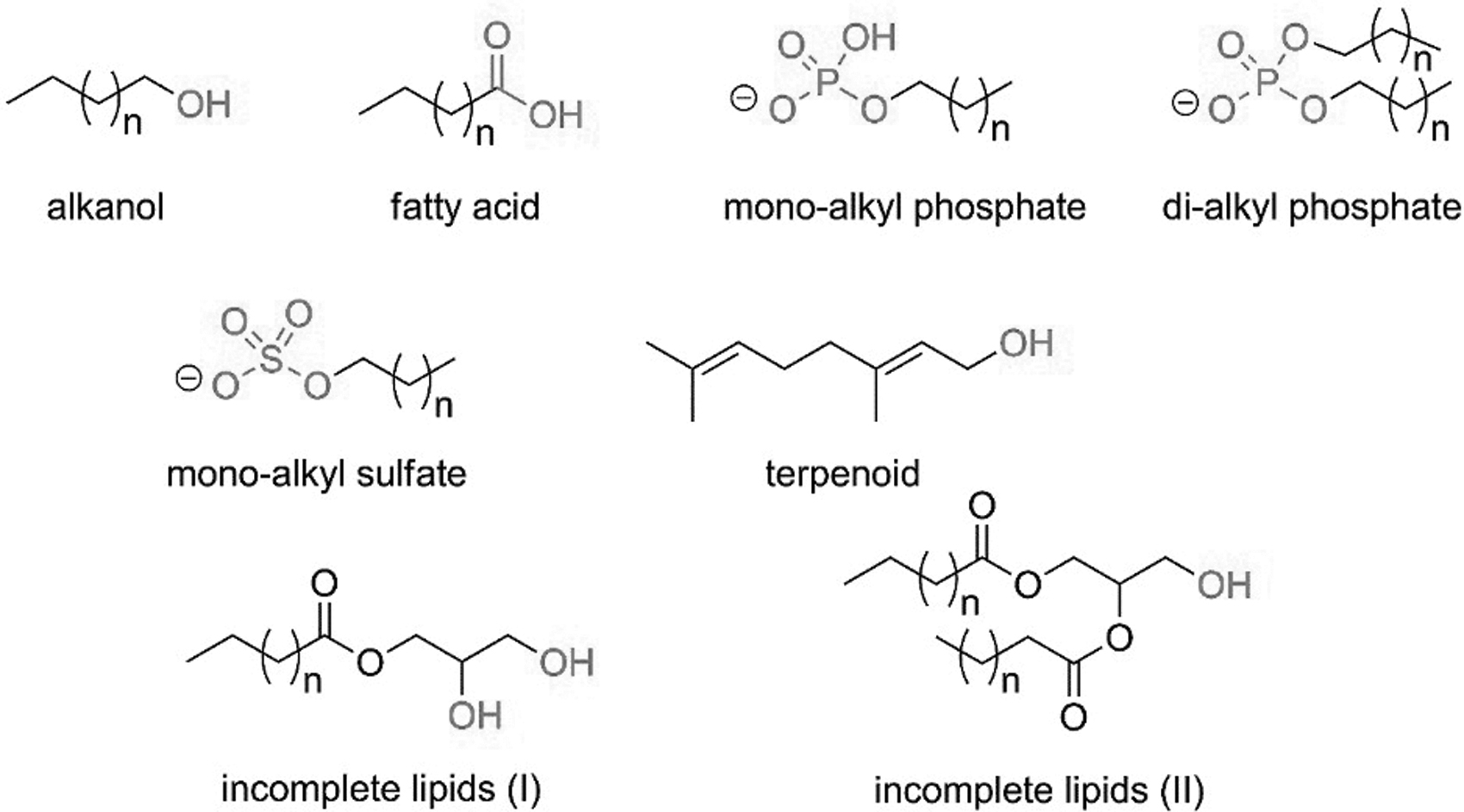
Plausibly prebiotic lipid derivatives.
The progressive transition between early amphiphiles and modern phospholipids lays on the fact that the latter represents rather complex synthetic targets and is unlikely to have been produced spontaneously, but rather elaborated over time by successive chemical processes. In this light, Fiore and Strazewski (2016) suggested to classify lipids as “incomplete” when the phosphate head group is lacking (fatty acids and alcohols, mono- and diacylglycerols, Fig. 2), and “complete” when a phosphate head group is present (cf. Fig. 2).
1.4. Structure of a phospholipid: sn-glycerol and Cahn/Ingold/Prelog nomenclatures
Phospholipids, comprising phosphatidyl derivatives and sphingophospholipids, possess two main domains: a lipid domain constituted of one or more lipidic chains and a polar head. Phosphatidyl derivatives have a glycerol backbone that bears two identical or two different acyl chains linked by an ester or ether bond (Fig. 3).
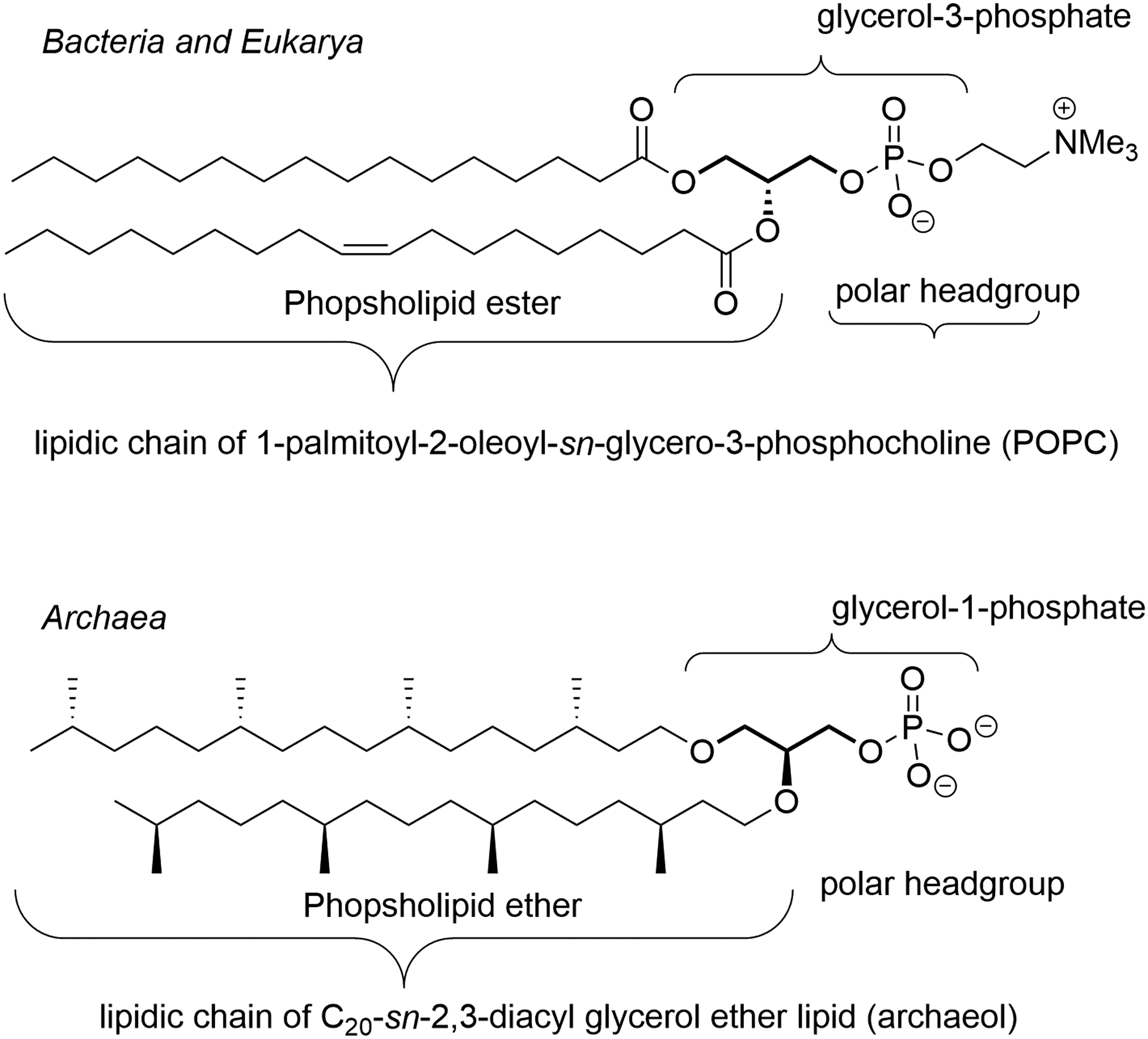
Representative structures of phospholipid ethers and esters. Both complete lipids are naturally occurring molecules that form lipid boundaries in Eukarya, Bacteria, and Archaea.
The glycerol backbone of glycerophospholipids is chiral. To designate the stereoisomers, the sn-glycerol nomenclature is widely used in biological sciences, while Cahn/Ingold/Prelog is the preferred nomenclature for organic chemistry. Solely, on phospholipid esters, the glycerol backbones possess the unique chiral center. The most common natural enantiomer for glycerol-based phospholipids is
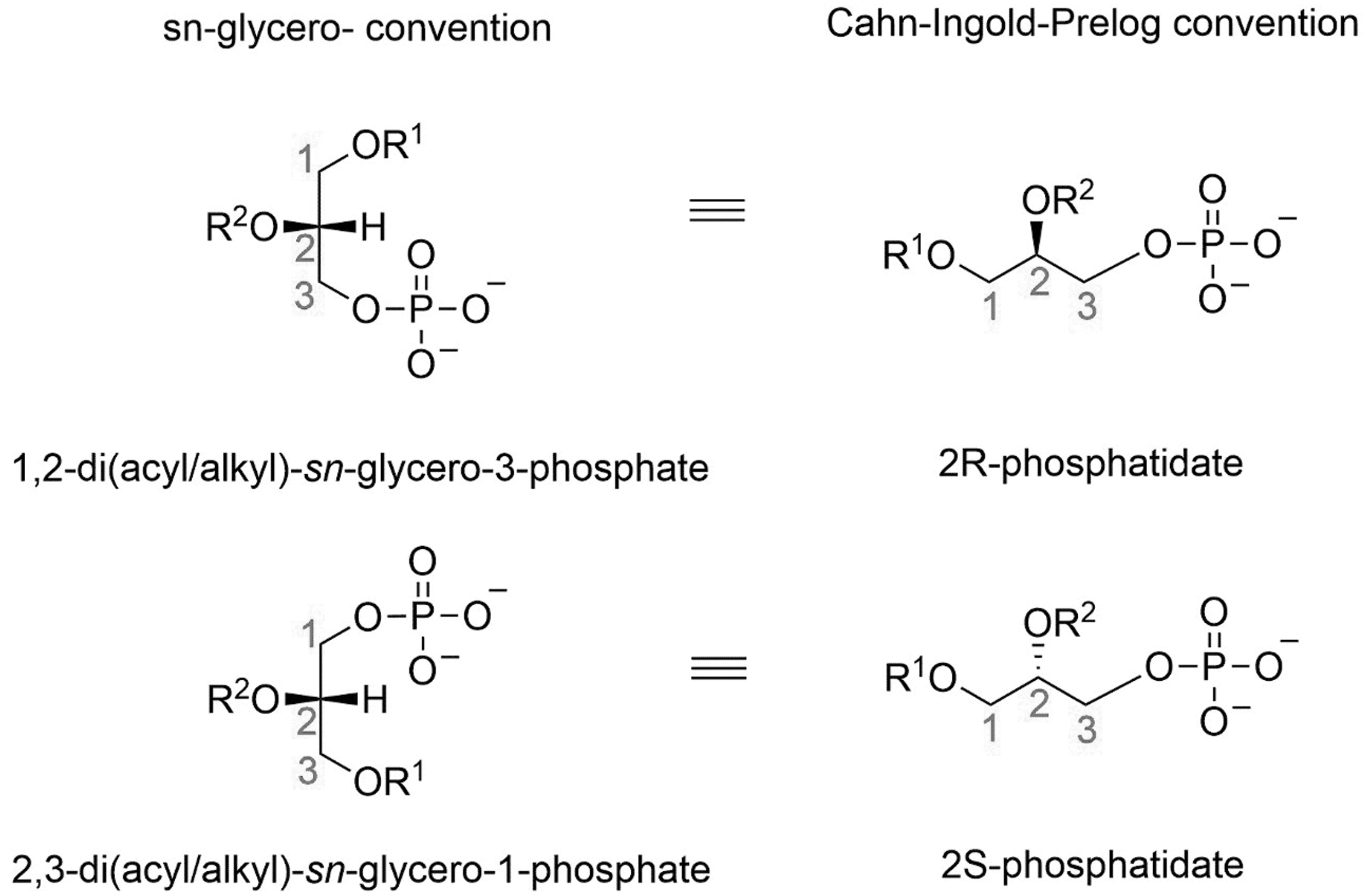
Phosphatidate enantiomers and their nomenclatures according to sn-glycerol and Cahn/Ingold/Prelog conventions. 1,2-diacyl-sn-glycero-3-phosphate is the enantiomer of 2,3-diacyl-sn-1-glycerophosphate: (i) The stereo numbering (sn-glycerol) is based on the position of the second oxygen of the glycerol moiety to the left side in the Fischer representation, with the top carbon numbered as one, second as two, and the bottom carbon numbered as three. (ii) The enantiomer changes the order of numbers of glycerol-moiety due to the opposite position of the second oxygen.
Lysophospholipids are phosphatidyl derivatives containing one single acyl chain. In contrast to phosphatidates, sphingolipids are derivatives of sphingosin, (2S,3R,4E)-2-amino-octadec-4-ene-1,3-diol. Ceramides contain an alkene linked to the 2-amino moiety of the sphingosin. A very common sphingolipid in mammalian is the sphingomyelin, where a phosphatidylcholine is linked to the first alcohol of the ceramide.
1.5. Geochemical scenarios and energy sources
The idea that hot, evaporative, environment concentrating reagents at the surface of clays and sands can promote the condensation of building blocks to produce biomolecules and polymers was applied from the beginning of prebiotic systems chemistry (Szostak et al., 2001; Damer and Deamer, 2015, 2020). These scenarios could take place in two kinds of environments: hydrothermal vents and hydrothermal fields. Hydrothermal vents, also called hydrothermal black smokers or submarine hot springs (Miller and Bada, 1988), are alkaline, far-from equilibrium environments. Since their discovery, they were proposed as sites where chemical reactions could initiate primitive metabolism involving the reduction of CO2 by dissolved H2 (Martin et al., 2008).
The alternative environments are hydrothermal fields or hydrothermal pools. Recently, Damer and Deamer (2015, 2020), pointed out that fluctuating hydrothermal pools could be considered as plausible prebiotic reactors for the synthesis of several key molecules in the development of life, including lipids, nucleic acids, and peptides. Condensation of biopolymers, lipid synthesis, aggregation, and protocell formation occurs into a sort of “bath tub” environment, thanks to the cold/hot and wet/dry cycles (Damer and Deamer, 2015).
2. Synthesis of Fatty Acids Under Abiotic Conditions
Simple long-chain substrates, including fatty acids, aldehydes, alcohols, and similar compounds, were likely formed in relatively harsh but potentially wet conditions by thermal Fischer/Tropsch-type processes from simple gaseous precursors (Fig. 5) (Klein and Pilpel, 1973; Deamer, 1985; Deamer and Pashley, 1989; Mccollom et al., 1999; Dworkin et al., 2001; Rushdi and Simoneit, 2001; Simoneit, 2004; Meierhenrich et al., 2010).
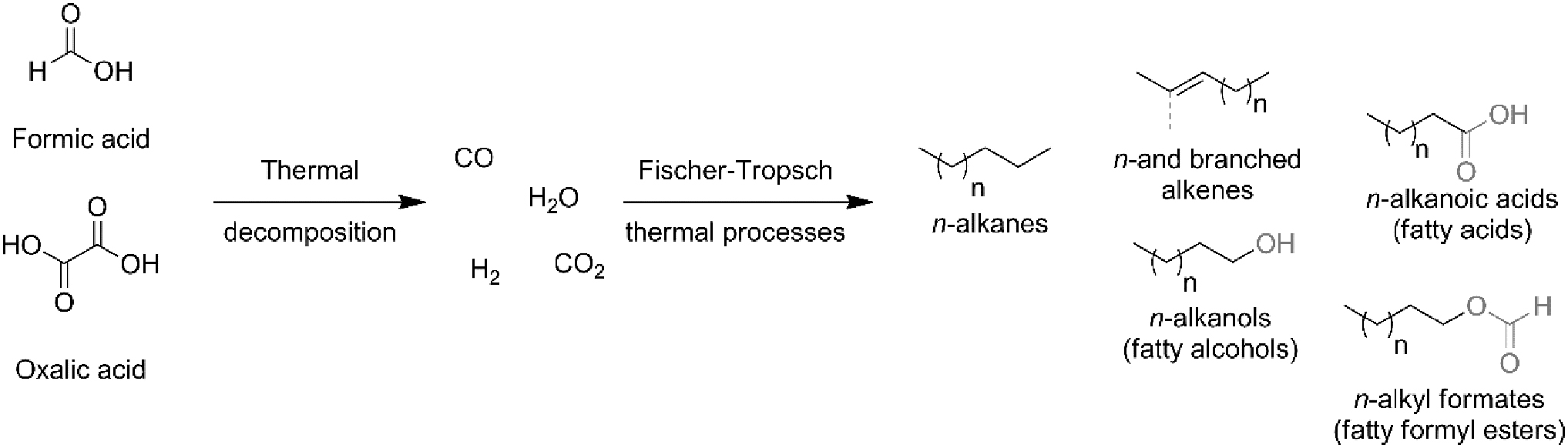
Simple amphiphiles obtained by Simoneit et al. (2007) in hydrothermal Fischer/Tropsch-like conditions. Formic and oxalic acids were used as convenient precursors of simple C0 and C1 gases. Chain lengths going from C6 to C22 (fatty acids), C23 (fatty alcohols), and more than C30 (hydrocarbons) were detected by GC analysis of dichloromethane extracts of the crude mixtures.
Alkanes can be transformed into amphiphilic oxygenated material by photo-oxygenation from fatty alcohols (Klein and Pilpel, 1973). In this context, investigation on the nature of carbon containing compounds of carbonaceous meteorites revealed the presence of small but relevant quantities of lipophilic and amphiphilic materials. Those molecules, once delivered to the early Earth upon meteorite falls (Deamer, 1985; Deamer and Pashley, 1989), have probably played a pivotal role in the formation of very prebiotic membranes. Similarly, cometary ices furnished materials able to encapsulate structures upon hydration (Dworkin et al., 2001). Fluorescent hydrophilic probes were used as model compounds to prove those theories (Lopez et al., 2020a).
2.1. Analogies between prebiotic synthesis and biotic synthesis of fatty acids
The fatty acid synthesis is well described in most biochemistry textbooks (Voet and Voet, 1995). The formation of hydrocarbon chains does not require any thermal process, but an overall endoergonic reduction, with CO2 and H+ as minimum ingredients. A key evolutionary step toward evolved biosystems is the appearance of photosynthesis in living cells as exemplified by the phototrophy evolved phosphorylation in Halobacteria, and photosynthesis in Heliobacteria toward oxygenic photosynthesis in Cyanobacteria (Lake and Sinsheimer, 2013). Autotrophic organisms sustain all ecosystems through the process of fixing CO2 into organic matter (Shih, 2015). In the case of saturated hydrocarbon chain, the minimal requirements may be written in Equation 1 as follows:
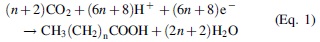
Equation 1. Fixation of carbon dioxide into saturated hydrocarbon chains by autotrophic organisms. In this equation n = 1, 2,3,..
The number of protons (H+) and electrons (e−) required is reduced by two for each insaturation. The overall reaction scheme indicates that during evolution, three key points were solved to yield the in vivo biosynthesis of lipids. It is still unclear which point occurred first during the evolution. Therefore, the key points correspond to a list of problems to be solved. The first point is that CO2 was replaced by acetyl coenzyme A (CH3-CO-S-CoA) (Table 1) and malonyl coenzyme A (CH3-CH2-CO-S-CoA) as starting materials to produce fatty acids. CH3-CH2-CO-S-CoA is obtained from a carboxylation of CH3-CO-S-Co (Equation 2):
Differences or Analogies Between Prebiotic and Biotic Syntheses of Fatty Acids
Equation 2. Carboxylation of CH3-CO-S-CoA yields CH3-CH2-CO-S-CoA. Pi stands for HPO4 2−
The condensation of one CH3-CO-S-CoA (a two-carbon derivative of H-S-CoA) and one CH3-CH2-CO-S-CoA (a three-carbon derivative of HS-CoA) yields one CH3-(CH2)2-CO-S-CoA (a four-carbon derivative of H-S-CoA), one H-S-CoA, and one CO2. This is somewhat the chemical equivalent of the thermal Fischer/Tropsch reaction. However, in contrast to the Fischer/Tropsch reaction, the condensation is performed from activated alcane derivatives containing CoA at room temperature and is catalyzed by enzymes (Table 1).
Due to the two-carbon derivative acetyl-S-CoA, the acyl length can be incremented only by two carbons at each synthetic cycle, yielding, in most cases, a fatty acid with even number of carbons (Voet and Voet, 1995). In rare cases, fatty acids with an uneven number of carbons are synthesized from a three-carbon propionyl moiety instead of a two-carbon acetyl moiety, which is catalyzed by a distinct enzyme (Voet and Voet, 1995). In Archaea, the elongation process occurs by incrementation of five-carbon length isoprenyls (Table 1) activated as pyrophosphates. Isoprenoids derive from a five-carbon long isopentyl pyrophosphate (IPP) or from dimethylallyl pyrophosphate (DMAPP) (Fig. 6) (Wang and Ohnuma, 1999; Vandermoten et al., 2009; Jain, 2014).
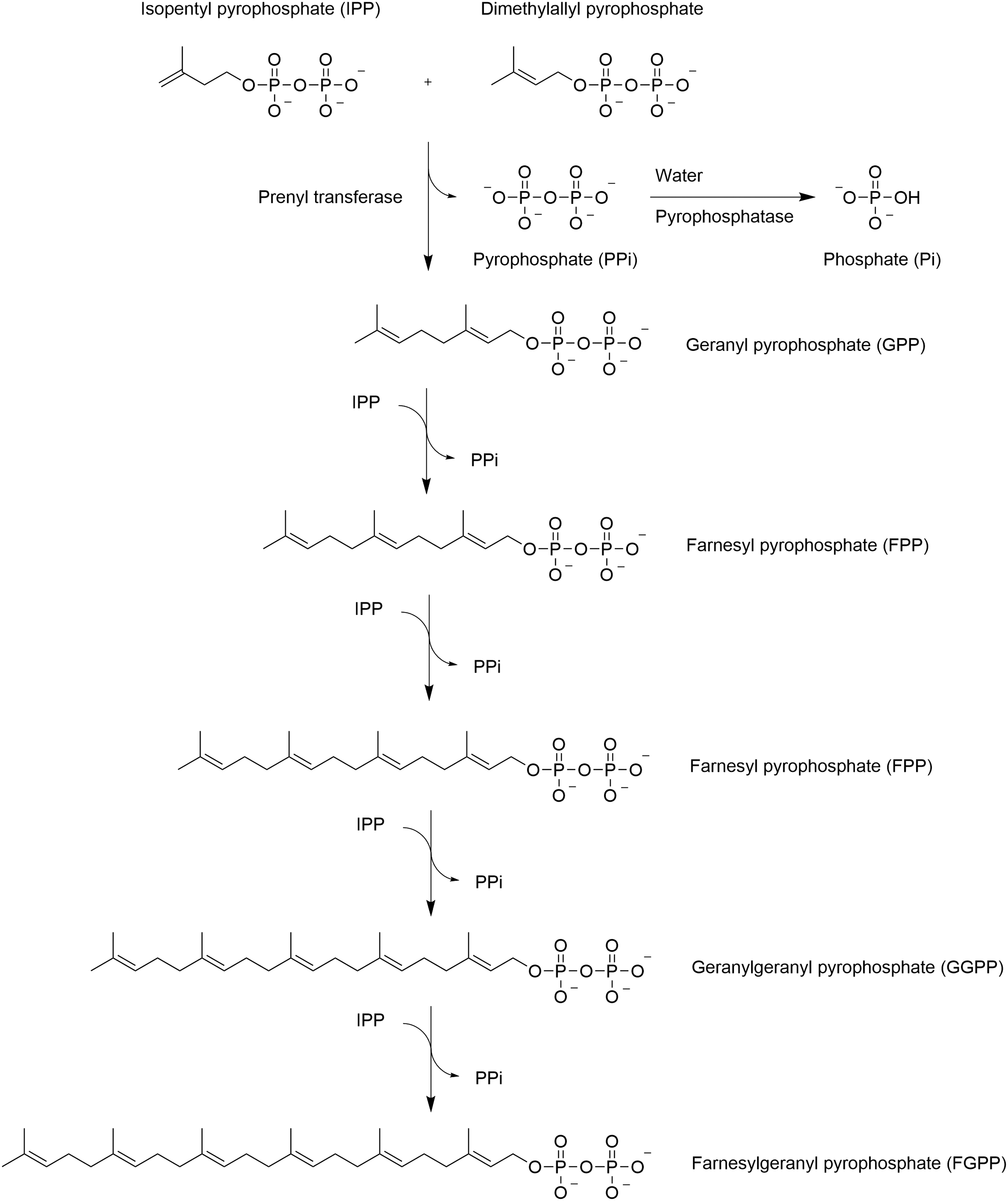
Isoprenyl pyrophosphate synthases, which are soluble enzymes, catalyze the chain elongation of allylic pyrophosphate substrates (DMAPP, GPP, FPP, GGPP, etc.) via consecutive condensation reactions with isopentyl pyrophosphate to yield, respectively, GPP (C10), FPP (C15), GGPP (C20), FGPP (C25), and FFPP (C30). DMAPP = dimethylallyl pyrophosphate; GGPP = geranylgeranyl pyrophosphate.
However, the formation of isoprenoids (chains in Archaea), via the mevalonate pathway, starts with acetyl-S-CoA, which yields sequentially acetoacetyl-S-CoA, hydroxymethyl-glutaryl-S-CoA, mevalonate, mevalonavate-5-phosphate, and then IPP, which can be converted into DMAPP (Jain, 2014). The enzymes that participate in the mevalonate pathway and the isoprenyl pyrophosphate synthases are soluble enzymes, indicating that the reactions occur in the cytoplasm (Jain, 2014).
The second point is that electrons and protons are transferred from the so-called cofactor, NADPH (Table 1), which can be recycled (Equations 3 and 4):
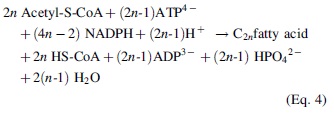
Cofactor NADPH serves to transfer H+ and e− during the synthesis of fatty acid of 2n-carbon length. Acetyl-S-CoA serves as a CO2 supplier. The reaction schemes are valid for an even number of carbons in fatty acids.
The third point is the energy supply. Acetyl-S-CoA and NADPH contribute as energy fuel for the fatty acid biosynthesis (Table 1). Since the overall chain building reaction is endergonic, the energy is supplied by ATP produced during photosynthesis and by electron transfer via NADPH, mostly used for reductive biosynthesis in contrast to NADH, which serves primarily in the production of ATP. It seems that the evolution path favored the use of identical recycling building blocks to solve each point of the chemical synthesis, especially since acetyl-S-CoA, NADPH, and ATP are adenosine derivatives, such as FADH (Fig. 7).
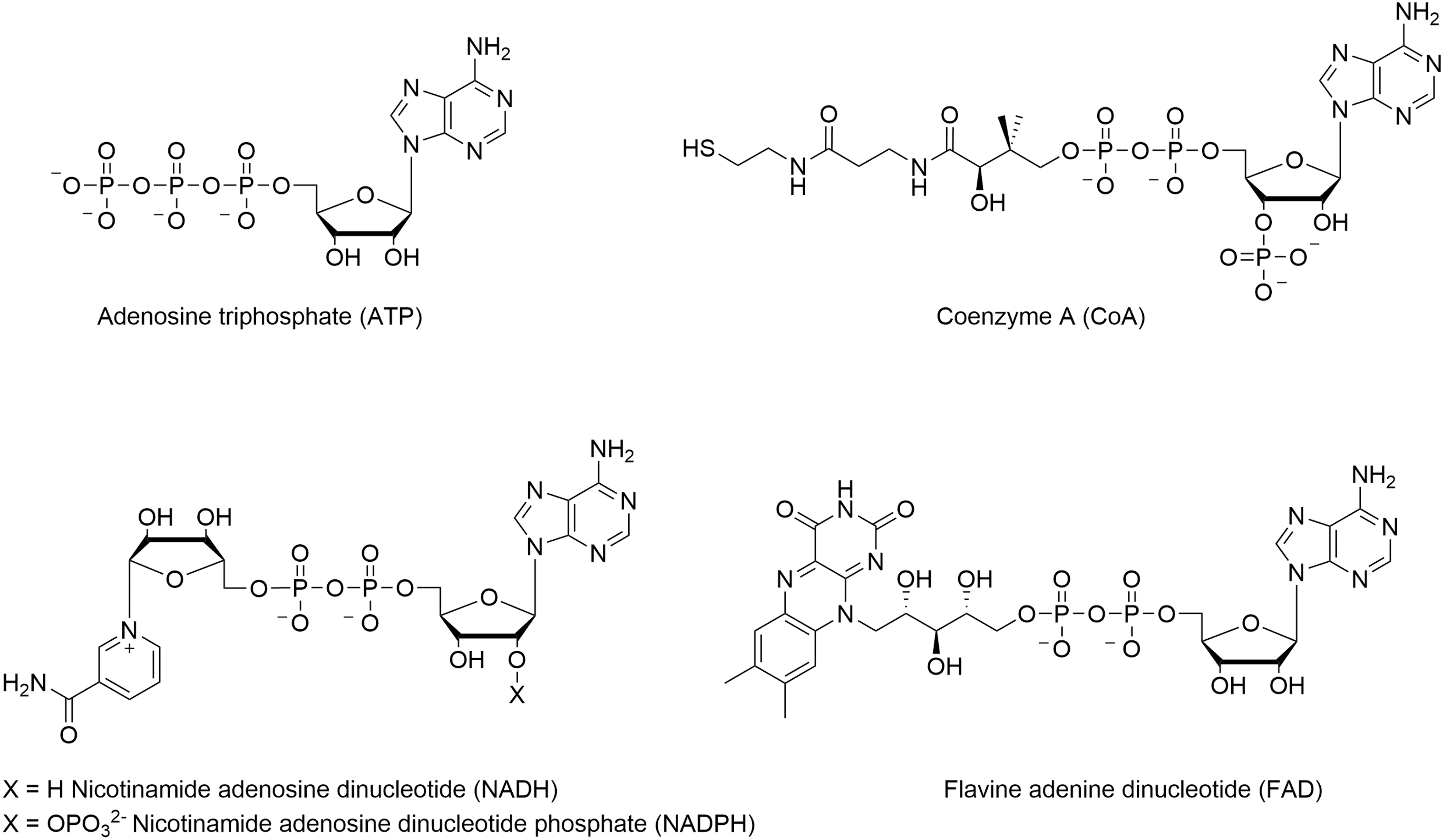
Structures of ATP, CoA, NADH, NADPH, and FAD, all containing adenosine diphosphate residues.
Escherichia coli and other gram-negative bacteria (GN) are enclosed by two membranes. In GN, earlier precursors of fatty acids and polar headgroups are synthesized in the cytoplasm (Raetz, 1986). The biosynthesis and turnover of lipids and their precursors are catalyzed by enzymes, which with few exceptions function on the cytoplasmic side of the inner membrane (Raetz, 1986). In mammals, the synthesis of phospholipids is usually restricted to the cytosolic side of the organelle endoplasmic reticulum (ER) (Vance, 2015). Thus, lipids are transported from the inner membrane to the outer membrane in bacteria or from ER to plasma membranes in mammals.
Two breakthroughs appeared during evolution from protocells toward living cells: first, the emergence of separate compartments where the synthesis of lipids is performed, and second the transport mechanisms toward the outer membranes or plasma membranes. For example, in E. coli, fatty acids are initiated in the cytoplasm from acetyl groups during a condensation reaction involving two acyl carrier proteins (HS-ACP), having the same phosphopantetheine group containing HS as in CoA: one acetyl-S-ACP and one malonyl-S-ACP (Fig. 8) (Volpe and Vagelos, 1976).
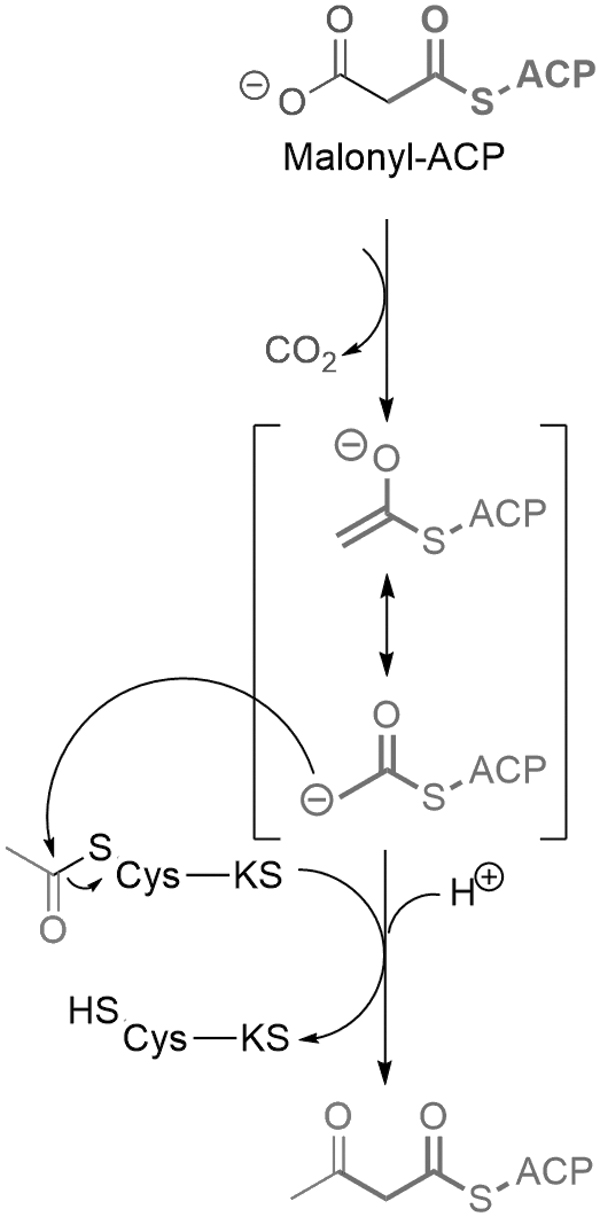
The mechanism of carbon–carbon bond formation in fatty acid biosynthesis. The condensation of an acetyl group on the active site Cys of β-ketoacyl-ACP synthase (KS) with a malonyl group on the phosphopantetheine arm of ACP forms a β-ketoacyl-ACP. The reaction is driven by the exergonic elimination of CO2 from the malonyl group to generate a resonance stabilized acetyl-ACP carbanion intermediate that functions as a good nucleophile. ACP = acyl carrier protein.
This is followed by one reduction, one dehydration, and one last reduction to obtain a butyryl-ACP (Fig. 9) (Voet and Voet, 1995).
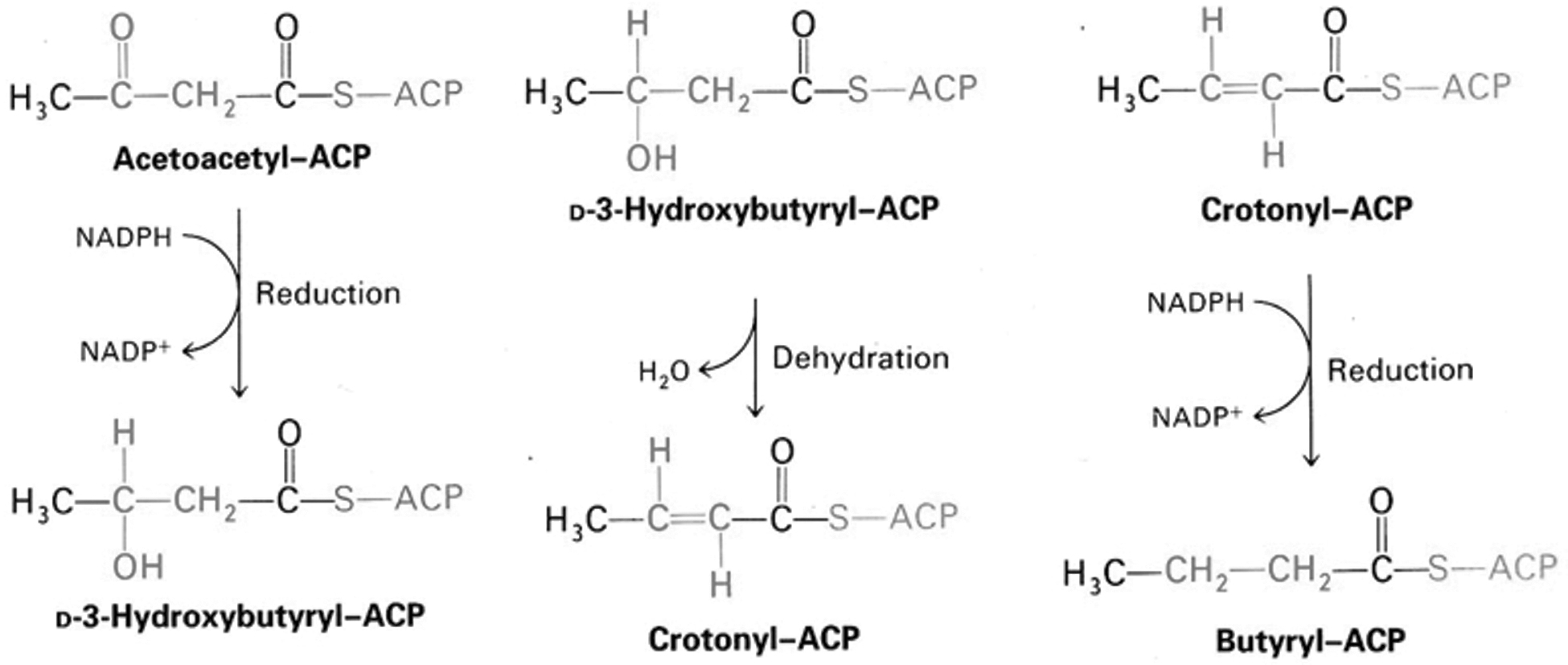
Formation of butyryl-ACP from acetoacetyl-ACP, adapted from Voet and Voet (1995).
The butyryl-ACP can be either hydrolyzed forming butyric acid and ACP or can be used for its elongation. Condensation of one butyryl-ACP with one malonyl-ACP yields one hexanoyl-ACP similarly to the obtention of one butyryl-ACP. The elongation process can continue by incrementating two carbons at each step (Voet and Voet, 1995). Once the elongation step is achieved, hydrolysis of the acyl-ACP generates ACP and the fatty acid containing even number of carbons. Thus, in analogy with the Fischer/Tropsch processes, the biosynthesis of a fatty acid starts from natural carbon sources such as CO2.
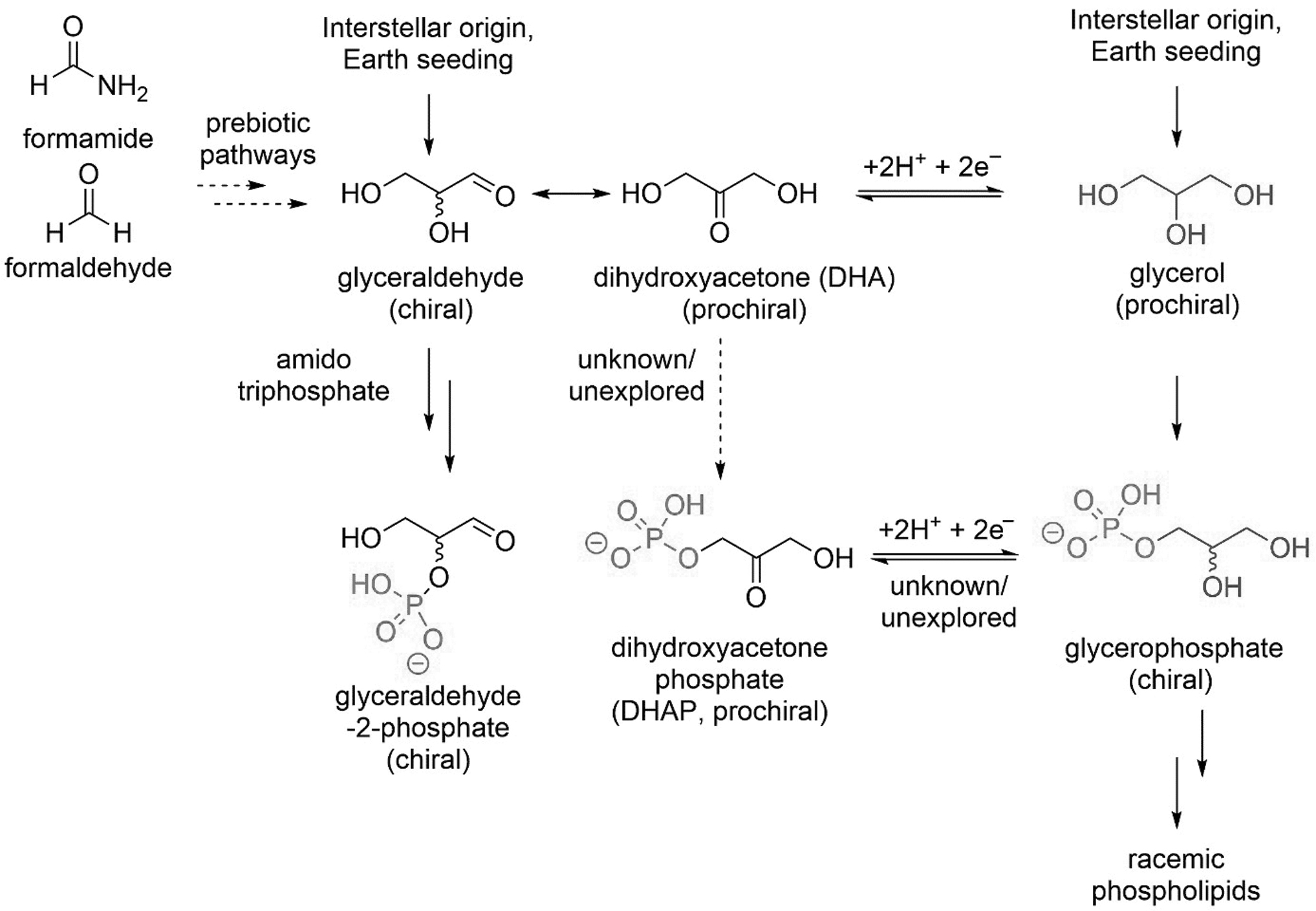
In recent years, Griesser et al. (2017) achieved concomitant formation of cofactors, together with peptide and nucleoside elongation in the same primordial soup. However, the role of these molecules in the presence of phospholipids or their precursors was not investigated (Jauker et al., 2015; Griesser et al., 2017). Tricyanocuprate [Cu(CN)3]2− and tetracyanocuprate [Cu(CN)4]2− are supposed to be a source of electrons for the oxidoreduction of glyceraldehyde (cf. Fig. 6) in enzyme-free conditions (Table 1) (Katagiri et al., 1981; Wachtershauser, 2000; Sutherland, 2016). In this contest, the hydrogen cyanide–cyanocuprate photochemistry has proven to be effective for synthesis in abiotic conditions of glycerophoshate precursors starting from the tautomerization of glyceraldehyde into dihydroxyacetone at physiologic pH and then reduction to glycerol, respectively (cf. Fig. 10).
Recently, iron (III)/sulfur/
3. Synthesis of Glycerol and Glycerol Precursors Under Abiotic Conditions
3.1. Prebiotic origin of glycerol and glycerol precusors
Phosphate-rich lakes may have preferentially formed on the prebiotic earth because of carbonic acid weathering under CO2-rich atmospheres and the absence of microbial phosphate concentration (Toner and Catling, 2019). Glyceraldehyde, dihydroxyacetone and glycerol, together with their phosphate derivatives, are the most plausible chemical precursors of glycerophospholipids such as phospholipid esters and ethers. Several studies have proposed formamide or formaldehyde (Fig. 10) as two of the most simple and plausible precursors of glycerol (Fig. 10) and phospholipids (Patel et al., 2015; Bizzarri et al., 2021).
Glycerophosphate, a plausible phospholipid precursor, is the product of phosphorylation of glyceraldehyde, dihydroxyacetone or glycerol. The prebiotic preparation of dihydroxyacetone phosphate (DHAP) from glyceraldehyde or glycerophosphate remains unexplored (Fiore and Buchet, 2020). Further investigations showed that glyceraldehyde, the first chiral product of the “formose” reaction (Benner et al., 2019), is also present in comets and other space bodies (Garcia et al., 2019). Glycerol was detected in interstellar ices (Kaiser et al., 2015). However, the question is still open how glycerol was generated (Myrgorodska et al., 2015). Notably glyceraldehyde can be phosphorylated in the presence of amidotriphosphate (cf. red box in Fig. 11), followed by pyrophosphate hydrolysis in very good yields (81%) (Krishnamurthy et al., 2000).
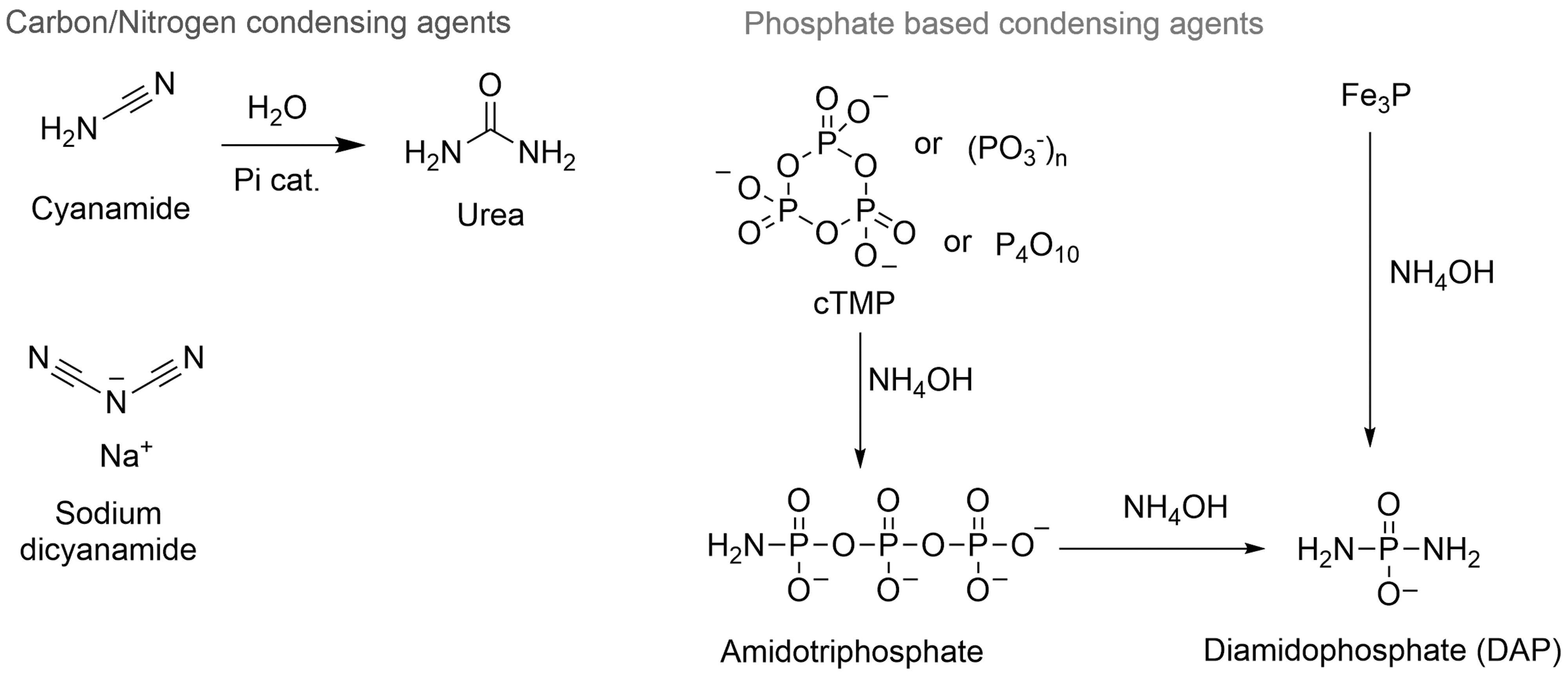
Condensing agents plausibly available on early Earth as reactants for lipid synthesis as well as for other condensations. Cyanamide, urea and sodium dicyanamide. Cyanamide may arise from HCN and is slowly hydrolyzed to urea under acidic conditions such as a phosphate solution at pH 4.5 (Fiore et al., 2017). Polyphosphates and phosphoric anhydrides (as conveniently modeled by (cTMP) and P4O10) may have been formed in volcanic environments. Some of these products are prone to evaporation and could have rained in ammonia-rich environments to provide amidophosphates such as AmTP and DAP (Gibard et al., 2018). AmTP = amidotriphosphate; cTMP = cyclic trimetaphosphate; DAP = diamidophosphate; HCN = hydrogen cyanide; P4O10 = phosphorus pentoxide.
3.2. Glycerol, glycerol phosphates, and the unsolved problem of symmetry breaking of phospholipids
Carbonaceous chondrite meteorites may reach enantiomeric excesses of glycerol and glycerol derivatives up to 50–60%, suggesting that the exogenous delivery of primed prebiotic compounds to early Earth from meteorites could have favored the emergence of asymmetric molecules (Pizzarello, 2016; Pizzarello and Shock, 2017). Speculations about where and how “enantiopure” life emerged from a primordial soup of abiotic mixtures of racemic molecules are extremely well reviewed (Blackmond, 2010; Ruiz-Mirazo et al., 2014; Fiore and Buchet, 2020).
One main conclusion was that all chiral molecules have formed in both enantiomers (racemic), suggesting that a symmetry imbalance between the two possible stereoisomers occurred in a second step (racemic→scalemic→enantiopure) (Altamura et al., 2020; Fiore and Buchet, 2020). Prebiotic symmetry breaking scenarios were mostly depicted by using mathematical models (Subramanian and Gatenby, 2018), and a single chemical example is effective to date: the well-known Soai reaction (Hawbaker and Blackmond, 2019; Soai et al., 2019). No mathematical or experimental models were proposed to explain the symmetry breaking in phospholipid ester synthesis (Fiore and Buchet, 2020). The abiotic synthesis in the prebiotic environment of phospholipid precursors from glycerol (prochiral) and fatty acids (achiral) for the formation of diacylglycerols does not lead to any symmetry breaking (Rushdi and Simoneit, 2006). The symmetry breaking could have emerged thanks to supposedly available chiral and markedly nonracemic catalysts such as peptides (Canavelli et al., 2019), RNA (Gilbert, 1986; Tamura, 2008), or chiral crystals (Hitz and Luisi, 2002; Park, 2011). To date, a few experimental clues support such hypotheses: chiral recognition of
3.3. Prebiotic synthesis of glycerol phosphates
Several plausibly prebiotic syntheses of glycerophosphates were explored (Fig. 13) (Gull et al., 2010; Patel et al., 2015; Pasek et al., 2017; Gibard et al., 2018; Bonfio et al., 2019; Xu et al., 2019).
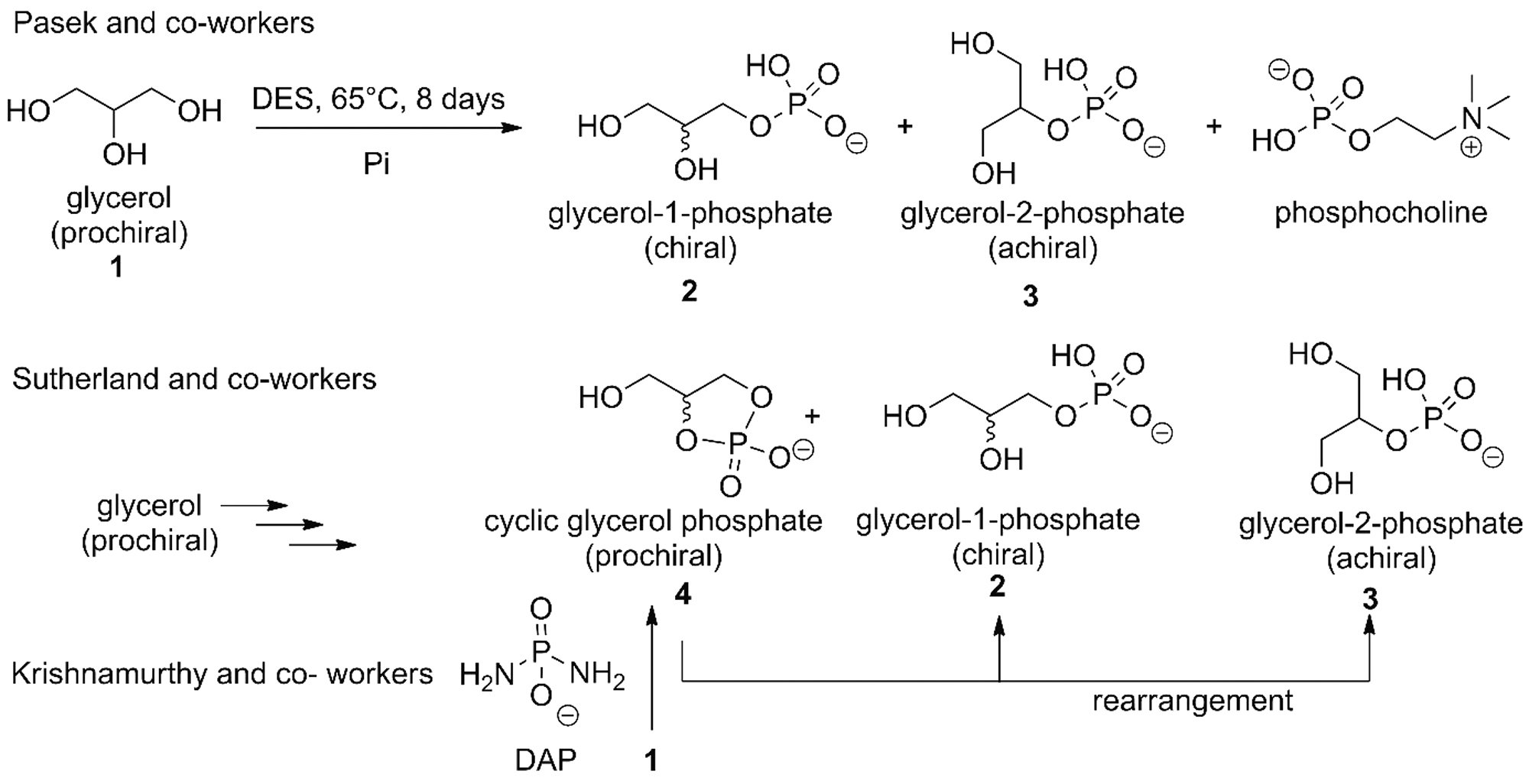
Glycerol phosphates and other organic phosphates obtained under hydrothermal (Gull et al., 2014) and prebiotic conditions (Patel et al., 2015). Cyclic glycerol phosphate was also obtained using DAP (Gibard et al., 2018); DES (urea: choline chloride 2:1); Pi: phosphate salts. DES = deep eutectic solvent.
Glycerol-1-phosphate (G1P) and glycerol-2-phosphate (
3.4. Analogies between prebiotic synthesis and biotic synthesis of glycerol phosphates: analysis on enzymes forming sn-glycerol-1-phosphate or sn-glycerol-3-phosphate
In contrast to the prebiotic synthesis, which yielded racemic glycerol phosphate mixtures, biotic syntheses are catalyzed by specific enzymes, producing either sn-G1P or sn-glycerol-3-phosphate (G3P) (Table 2).
Differences or Analogies Between Prebiotic and Biotic Syntheses of Glycerol Phosphates
G1P = glycerol-1-phosphate; G3P = glycerol-3-phosphate; G1PDH = G1P dehydrogenase; G3PDH = G3P dehydrogenase.
Since all cells contain polar lipids with a glycerol backbone, comparisons of the molecular mechanisms of the synthesis of G1P or G3P provide a detailed view of the evolution of cellular membranes (Yokobori et al., 2016). So far, there were no reports of prebiotic synthesis starting from DHAP (Table 2). There are two biosynthetic pathways to obtain G3P from either DHAP or from glycerol, while there is only one to obtain G1P from DHAP (Fig. 14) (Koga et al., 1998).
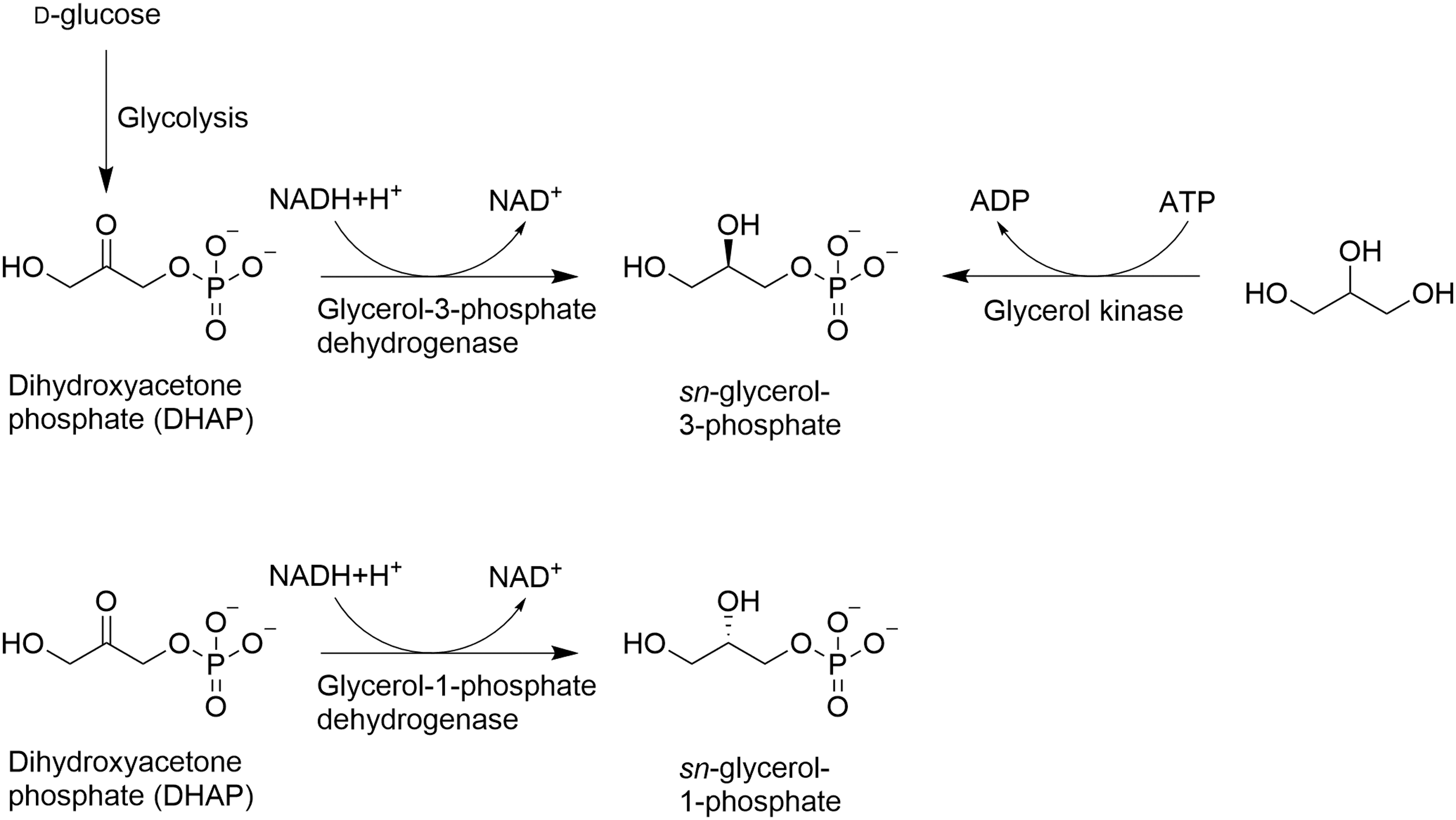
Biosynthetic pathways leading to G3P (top) and G1P (bottom). G1P = glycerol-1-phosphate; G3P = glycerol-3-phosphate.
The energy is provided by NADH + H+ for the reaction starting with DHAP, while ATP is provided for the reaction starting with glycerol, in contrast to the thermal proccess in the case of prebiotic synthesis (Table 2). Bacterial and eukaryotic phospholipids are derived from G3P, being esterified with two fatty acids to form phosphatidates (Voet and Voet, 1995). Archaeal phospholipids are derived from G1P containing two ether-bonds with isoprenoids, typically with a C20 chain length (Kate, 1993; Koga et al., 1993; Wächtershäuser, 2003; Matsumi et al., 2011). Glycerol kinase (GK) catalyzes the synthesis of G3P from glycerol (Fig. 14). G3P is also a metabolic intermediate of the glycolysis, while G1P is not.
This probably suggests that there is no GK catalyzing the formation of G1P. G1P in Archaea originates solely from DHAP and NADH + H+, catalyzed by a G1P dehydrogenase (G1PDH), as depicted in Fig. 14 (Nishihara et al., 1999; Yokobori et al., 2016). G3P is also formed from DHAP and NADH + H+, catalyzed by a G3P dehydrogenase (G3PDH) (Fig. 14 and Table 2) (Larsson et al., 1993). To determine which types of cells containing G3P-lipid membranes or G1P-lipid membranes appeared first, EgsA and AraM, which are genes encoding for the enzyme G1PDH, were searched for in Bacteria and Archaea (Yokobori et al., 2016). EgsA codes for GIPDH in Archaea, while AraM may code for GIPDH in Bacteria (Yokobori et al., 2016).
Similarly, GpsA and GLpA/GlpD, which are genes encoding for the enzyme G3PDH, were searched for in Bacteria and Archaea (Yokobori et al., 2016). GpsA, GlpA/GlpD, and EgsA are different, indicating that G3PDH and G1PDH evolved apart from each other (Koga et al., 1998). G3PDH activity has been reported from few archaeal lineages (Yokobori et al., 2016). Indeed, Archaea species, such as Archaeoglobus fulgidus, have GpsA gene (Yokobori et al., 2016). The presence of genes is not enough to ascertain the expression of G1PDH and G3PDH as well as the formation of their respective G1P-lipids or G3P-lipids. For example, GlpA/GlpB coding for G3PDH has been evidenced in several eukaryotic cells, but this gene is regulating the glycerol shuttle and not the formation of G3P-lipids. GpsA is the most abundant gene expressing G3PDH, responsible for the formation of G3P-lipids in Eukarya (Yokobori et al., 2016).
The phylogenetic analysis on these genes provided an evolutionary tree (see Section 5). It was hypothesized that C. commonote had heterochiral membrane lipids (Kandler, 1995; Wächtershäuser, 2003). The phylogenetic analysis on these genes (Yokobori et al., 2016) is consistent with prebiotic synthesis yielding a racemic lipid mixture at earlier times, which evolved during evolution to produce enantiomeric lipids.
4. Prebiotic Synthesis of Complete and Incomplete Phospholipids
The prebiotic synthesis of phospholipids may therefore be split into two parts: the formation of incomplete lipids (ILs, cf. Fig. 2), the critical step being the acylation of glycerol, and the phosphorylation of such into complete lipids (CLs, cf. Fig. 2). The phosphorylation of long-chain alcohols into the corresponding n-alkyl phosphates has been summarized elsewhere (Powner and Sutherland, 2011; Albertsen et al., 2014; Fiore et al., 2017; Fiore, 2018).
Interestingly, both acylation of glycerol and phosphorylation fall under the category of condensation reactions, which require the elimination of one molecule of water. Plausible condensing agents are urea, cyanamide and related compounds (Fig. 11). Urea at moderate temperatures (Lohrmann and Orgel, 1971; Osterberg and Orgel, 1972; Powner and Sutherland, 2011) and cyanamide and derivatives thereof (Hulshof and Ponnamperuma, 1976; Fiore et al., 2017) are, among others, the most popular dehydrating agents in the context of prebiotic chemistry. This may be achieved under pure thermal conditions or using a dehydrating agent that reacts with water, thus removing it from the medium. Some examples of phosphate-based condensing agents are reported in Fig. 11.
Iron phosphide (Fe3P as the mineral schreibersite) may also be a source of amidophosphates upon corrosion by ammonia (Gibard et al., 2018), and, more generally, a source of inorganic phosphates on the Earth (Pasek, 2020). The elimination of a molecule of water through the condensation of phosphate with an alcohol calls for the use of either thermal dehydration or the capture of water by a dehydrating agent, which is involved in the reaction mechanism (Powner and Sutherland, 2011; Burcar et al., 2019).
4.1. Prebiotic synthesis of IL esters
The thermal formation of glycerol esters under simulated prebiotic conditions was demonstrated as early as 1972. Incubation of palmitic acid (C16:0) and glycerol under wet anaerobic conditions at 85°C in the presence of the clay kaolin (aluminum silicate) yielded small quantities of mono- to triglycerides (Fig. 15). When phosphate was included, the authors were not able to clearly establish the formation of phospholipids for analytical reasons (Harvey et al., 1972).
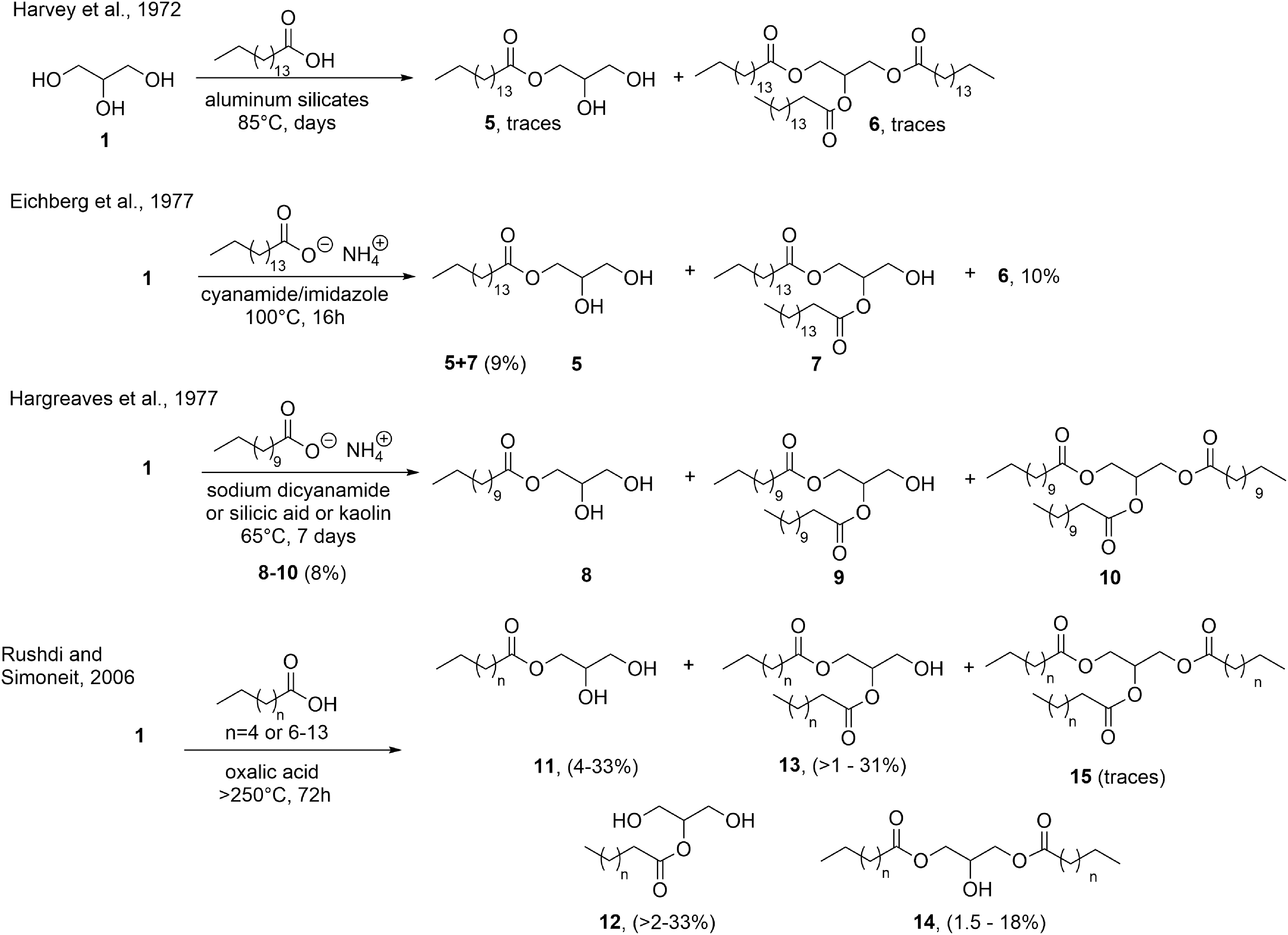
Incomplete lipid synthesis obtained in independent studies and different abiotic conditions.
A few years later, Oró and coworkers obtained mono-, 1,2-di-, 1,3-di-, and tripalmitoyl glycerols (
Deamer and coworkers used for the first time 14C-radiolabeled glycerol, and fatty acids, aldehydes or alcohols, the latter being by far less reactive (Fig. 15, third row), to produce racemic ILs together with CLs (cf. Fig. 16) upon incubation over silicic acid or kaolin (
More recently, Rushdi and Simoneit and coworkers (Rushdie and Simoneit, 2006) reported the thermal formation of racemic ILs bearing different n-acyl residues (
4.2. Prebiotic synthesis of CL esters
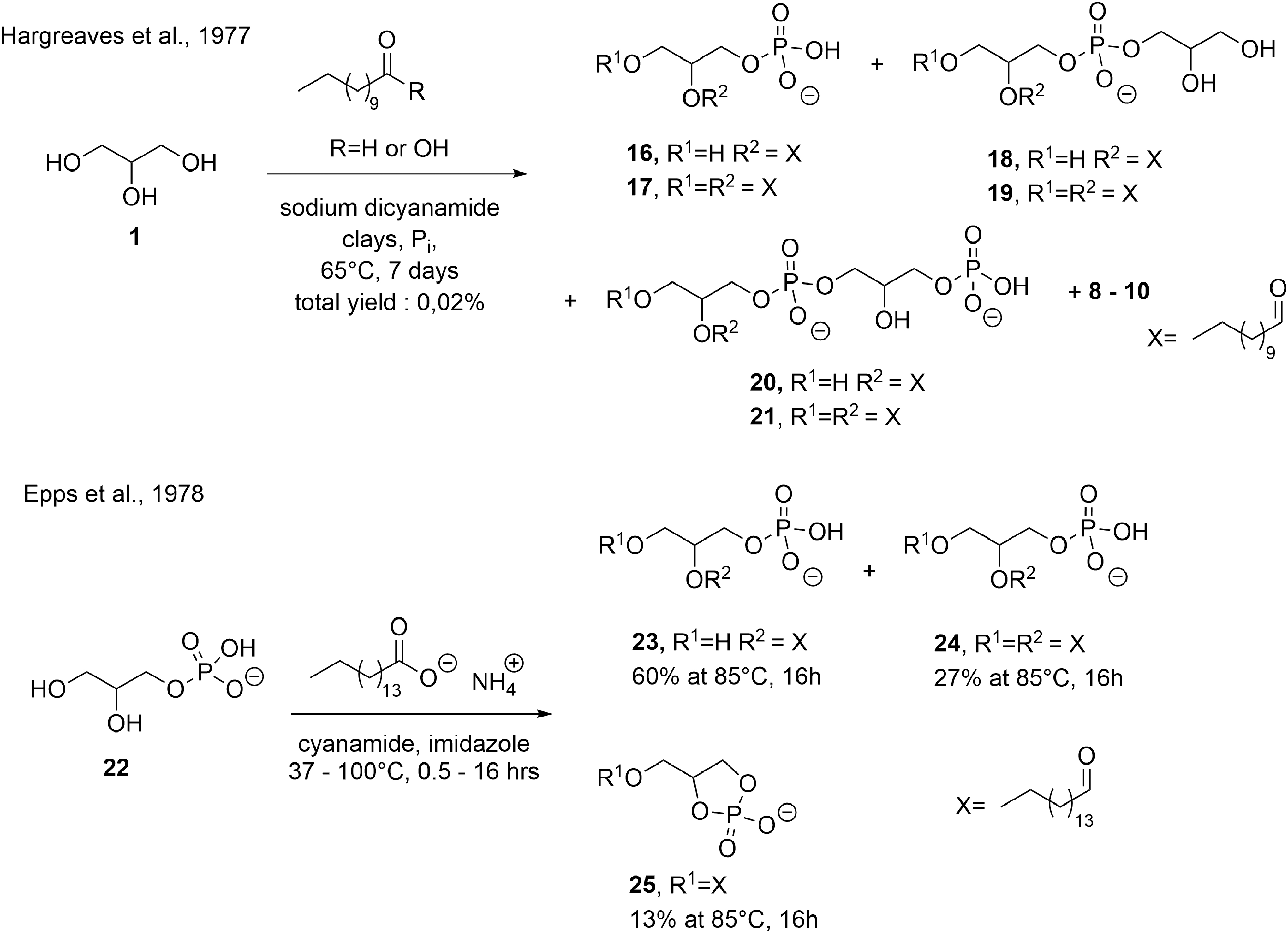
Deamer et al. obtained low amounts (0.2% total yield) of racemic phosphatidic acids (PAs,
Oró and coworkers explored an alternative synthetic pathway (Fig. 16, second row) by acylating a racemic glycerol phosphate, a product of the condensation of glycerol with ammonium dihydrogen phosphate, promoted by either cyanamide or urea (cf. Fig. 11) (Epps et al., 1979). In their experiments, 14C-sn-glycero-
Very recently, Liu et al. (2020) have reported on the preparation of six phosphatidylcholine (PC) via enzyme-free reaction in water from the corresponding lyso-phosphocholine (Fig. 16, third row). This new synthetic pathway shed lights on the origin of membrane forming CLs in the absence of preformed membranes. As expected, all the prepared compounds (PC) form liposomes and multilamellar giant vesicles along their formation in the reaction mixtures. Conversion of the lyso compounds into phosphatidylcholine has extremely interesting yields (55–87%) (Liu et al., 2020). This pathway must be considered barely prebiotic because lyso compounds are “evolved” compounds produced in modern cells via complex prebiotic pathways.
Oró (1995) demonstrated that these PAs could be converted into phosphatidylethanolamine (PE,
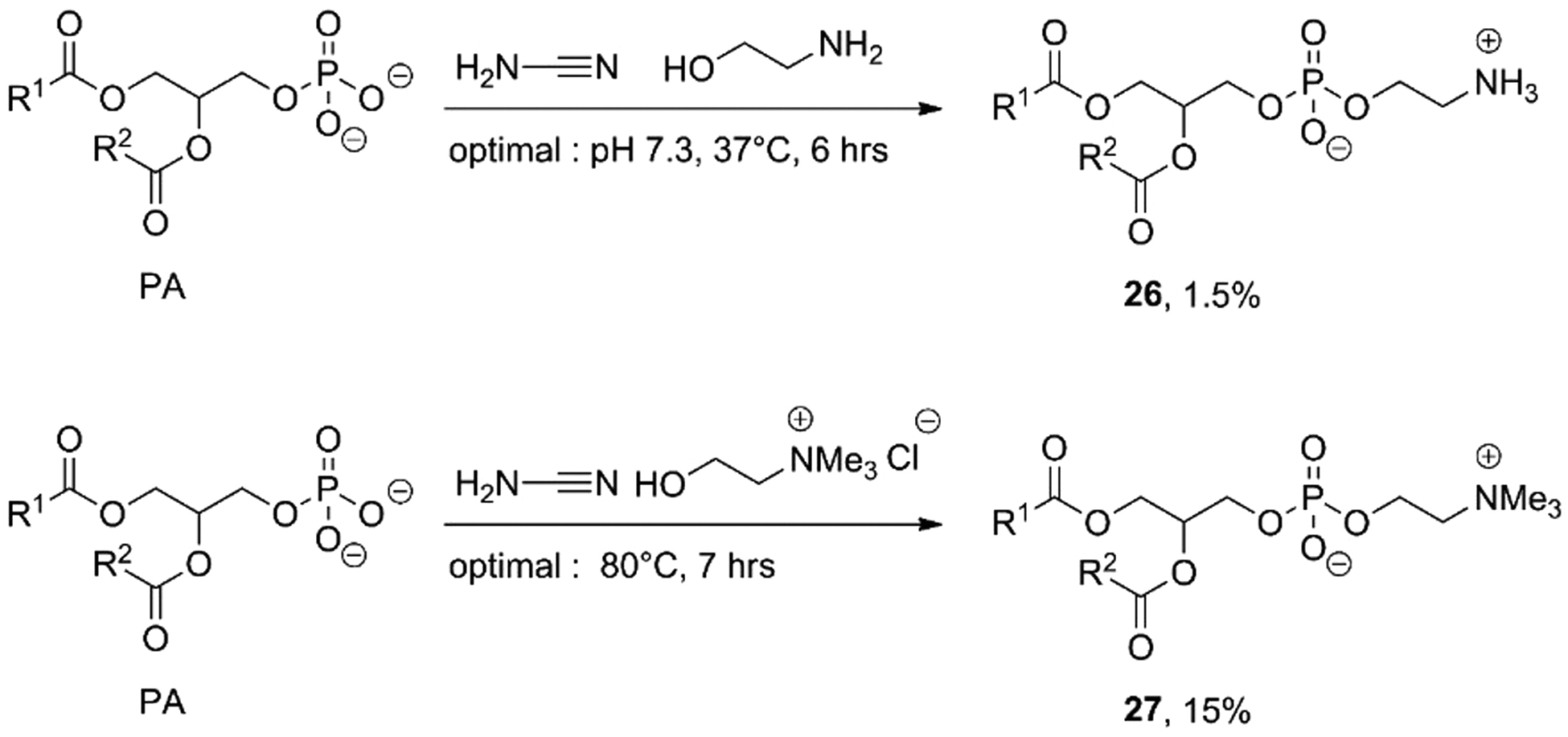
An excess of cyanamide showed a decrease of conversion, while the optimal molar ratio between PA, ethanolamine, and cyanamide was 1:2:10. The optimal molar ratio for the synthesis of PC was PA:choline: cyanamide, 1:20:20. Optimal pH and reaction time were 7.3 for 6 h at 37°C for the synthesis of
These reports outlined several synthetic routes in which the prebiotic synthesis of complex lipids can be carried out in essentially identical conditions, using some prebiotic condensing agent that was reported to promote many other condensations (Steinman et al., 1964) such as peptide bond formation and phosphodiester formation between nucleic acids.
4.3. Alternative prebiotic synthesis of phospholipid esters
The acylation of glycerol phosphates was explored in conditions only sparingly related to prebiotic chemistry but that still provided meaningful insight (Fayolle et al., 2017; Fernández-García and Powner, 2017; Bonfio et al., 2019). Fiore and coworkers (Fayolle et al., 2017) phosphorylated dioleoylglycerol (rac-DOG,
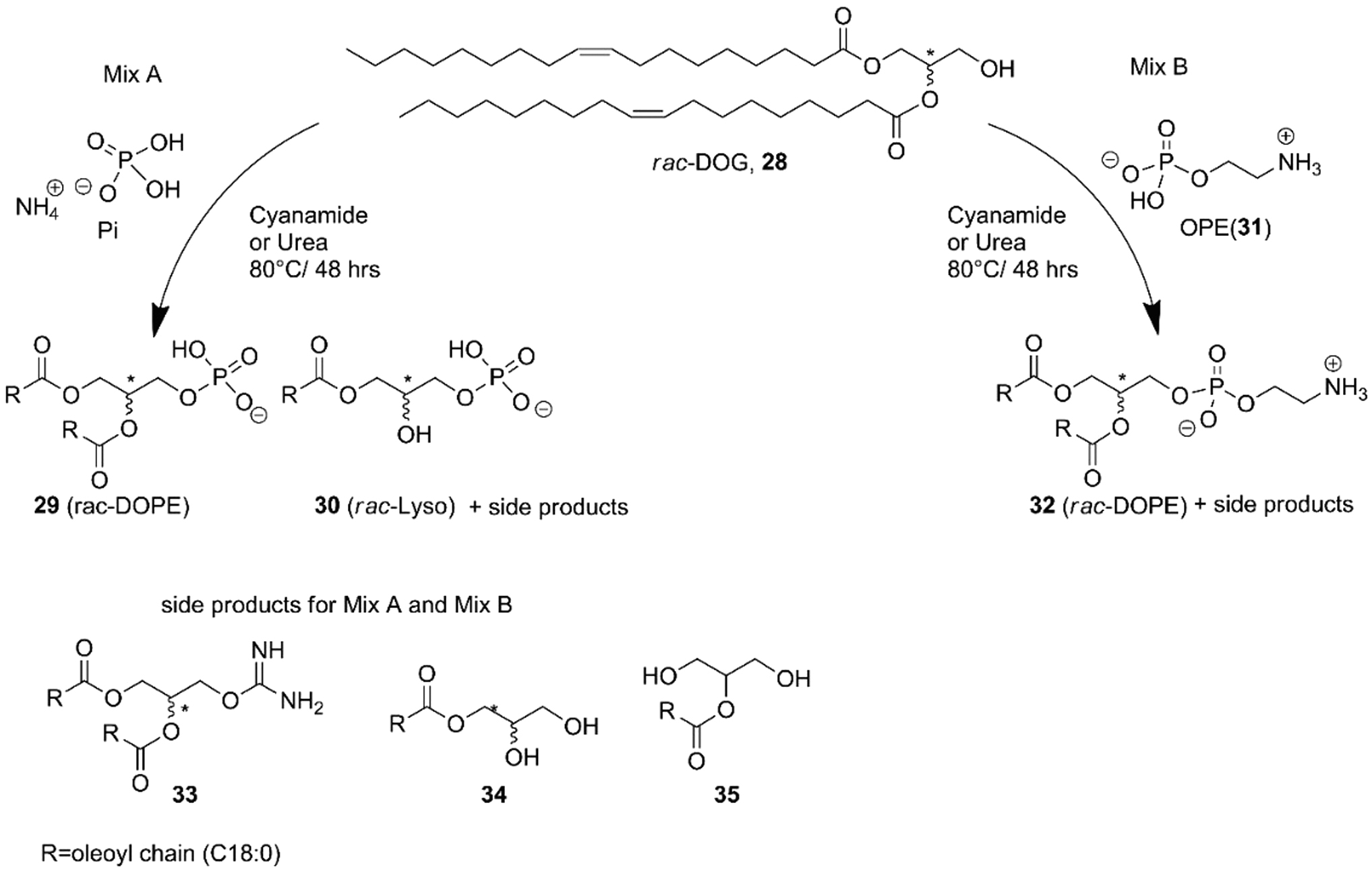
Phosphorylation of acyl glycerols through inorganic ammonium phosphate and phosphoethanol amine (
OPE is considered a plausible another prebiotic compound as proven by Pasek et al. (2017) and Gull et al. (2014). The phosphorylation of
For the first time, a complete analysis of the non-extracted crude mixtures was carried out using a combination of mono- and bidimensional nuclear magnetic resonance (NMR) and infrared spectroscopy, in addition to mass spectrometry. The crude mixtures containing DOPA (
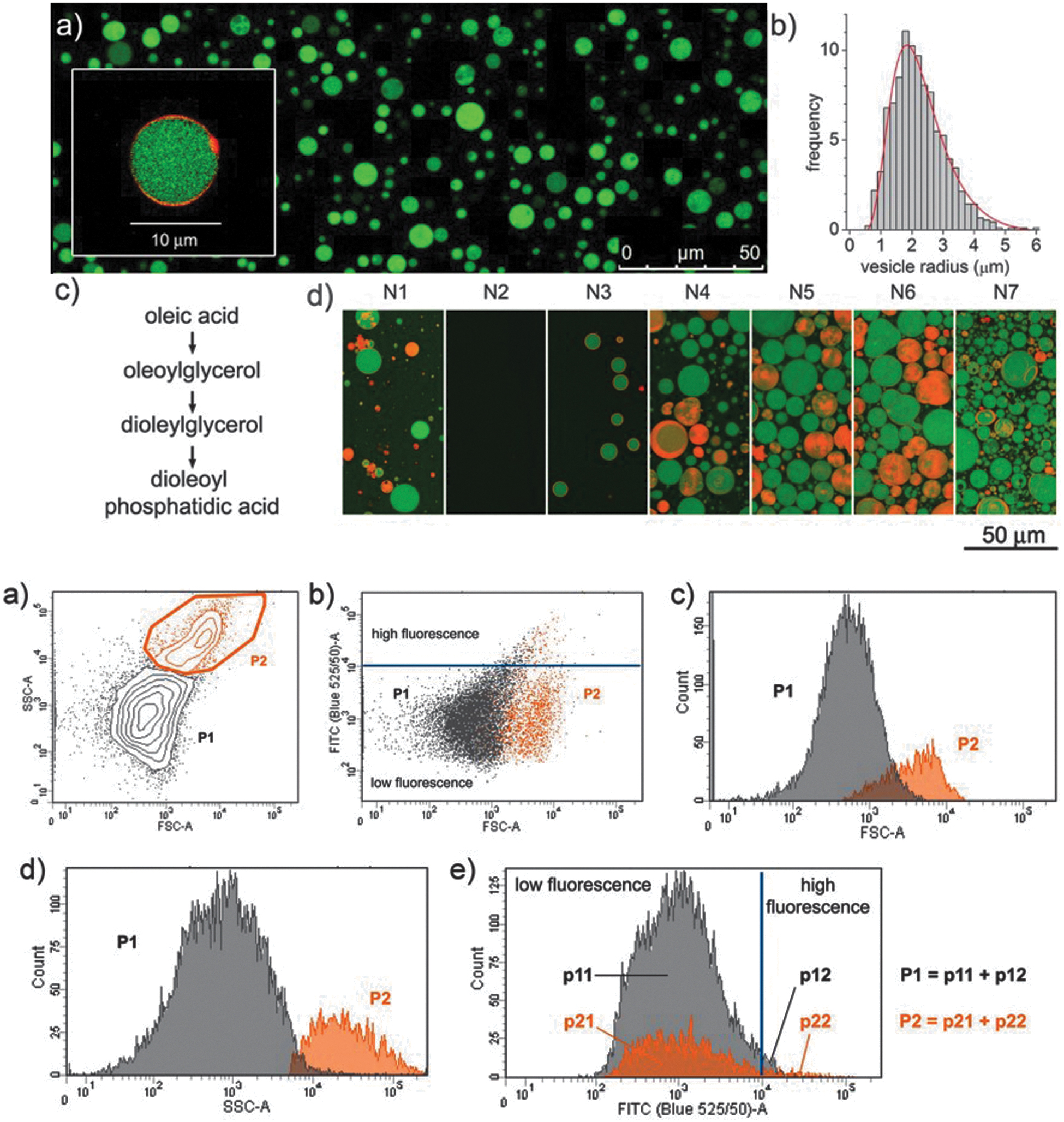
Powner and Sutherland (2011) showed that glycerol phosphates such as glycerol-3-phosphocholine (
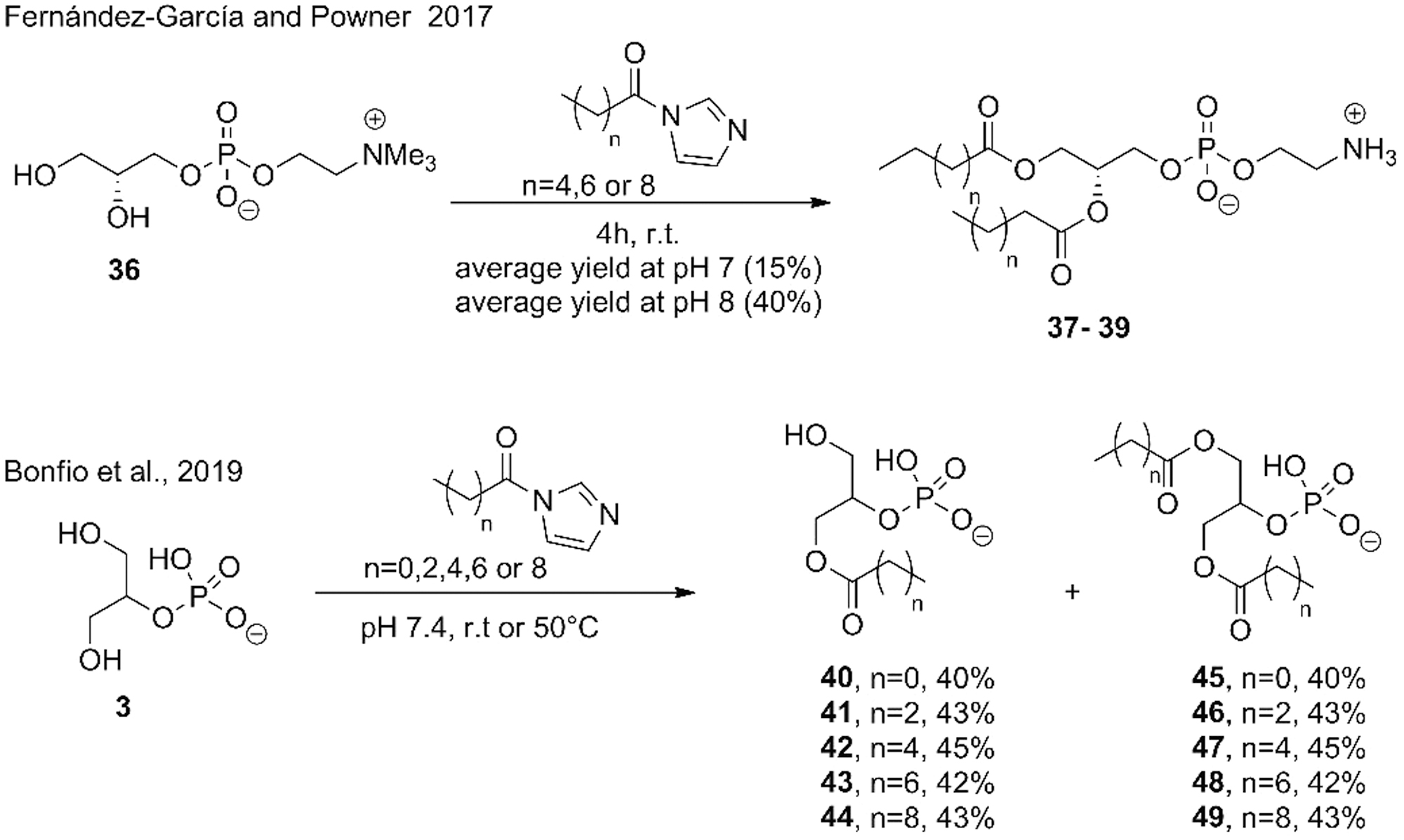
N-acyl imidazoles were used as acylating agents for PC and PA synthesis under conditions promoting long-chain diacylation through reagent colocalization.
Solubility issues due to lipophilic chains were overcome by using a mixture of acetonitrile:water (1:1 v/v) that has the conflict of not to be a prebiotic molecule. Experiments were followed by 1H NMR and showed up to 95% conversion (Fernández-García and Powner, 2017). Sutherland and coworkers (Bonfio et al., 2019) used N-acetyl imidazole and N-acyl imidazoles from tetra- hexa-, octa-, and decanoic acids for the acylation of glycerol-2-phosphate (
The authors proposed an intriguing reaction mechanism as well, where acylation was promoted via a phosphate ester intermediate (Bonfio et al., 2019). Remarkably, the authors observed membranes made of mixed acylglycerol-2-phosphates only from the second acylation-hydrolysis step from a monoacylated compound.
Soon thereafter, the group of Krishnamurty reported the use of DAP for the phosphorylation of glycerol into the corresponding racemic 1,2-cyclic phosphates (
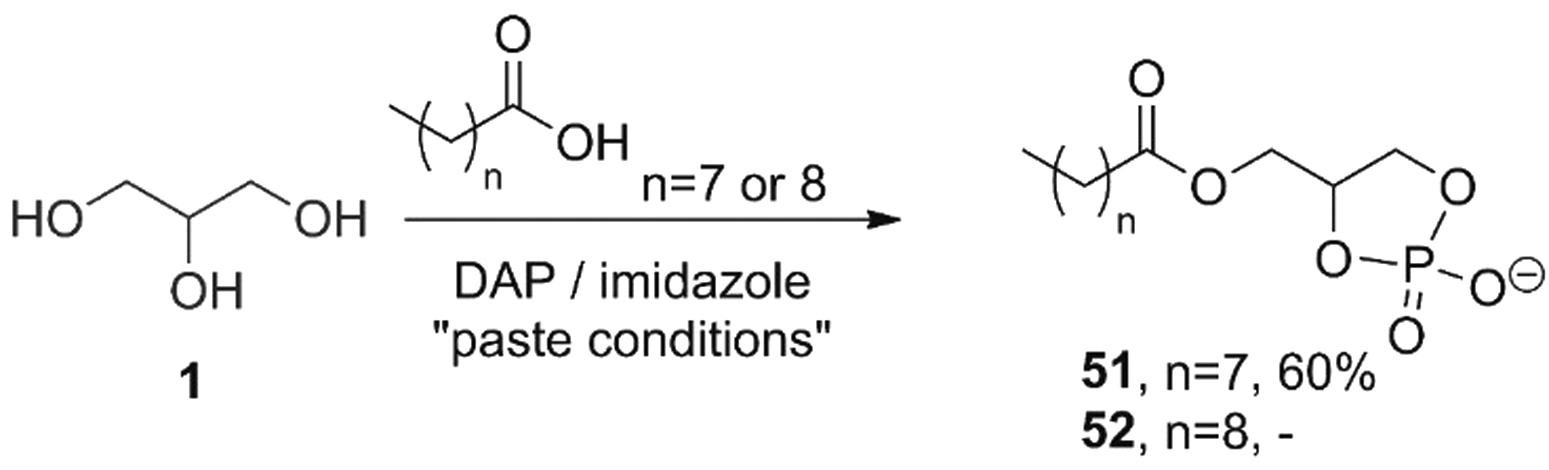
DAP and imidazole promote the acylation of glycerol by fatty acids (n = 7 or 8) and the cyclophosphorylation of resulting monoacylglycerols to yield cyclophospholipids in a one-pot process. Hydration of cyclophospholipids with fatty acids and/or fatty alcohol as cosurfactants gave rise to vesicles that were able to encapsulate dyes and were stable under a wide range of conditions, including saline environments (Gibard et al., 2018; Toparlak et al., 2019).
Such cyclolipids (
4.4. Prebiotic synthesis of phospholipid ethers
A few examples were reported for the preparation of ethers in abiotic conditions. From the bottom-down approach: phospholipid ethers are complex molecules and their synthesis in the laboratory requires several steps, including protections and deprotections of the glycerol backbone and the polar head (Sutter et al., 2015). Chemically speaking, the synthesis of an ether requires two molecules of alcohols that condense at moderate temperature. Deamer and coworkers have reported that glycerol ethers
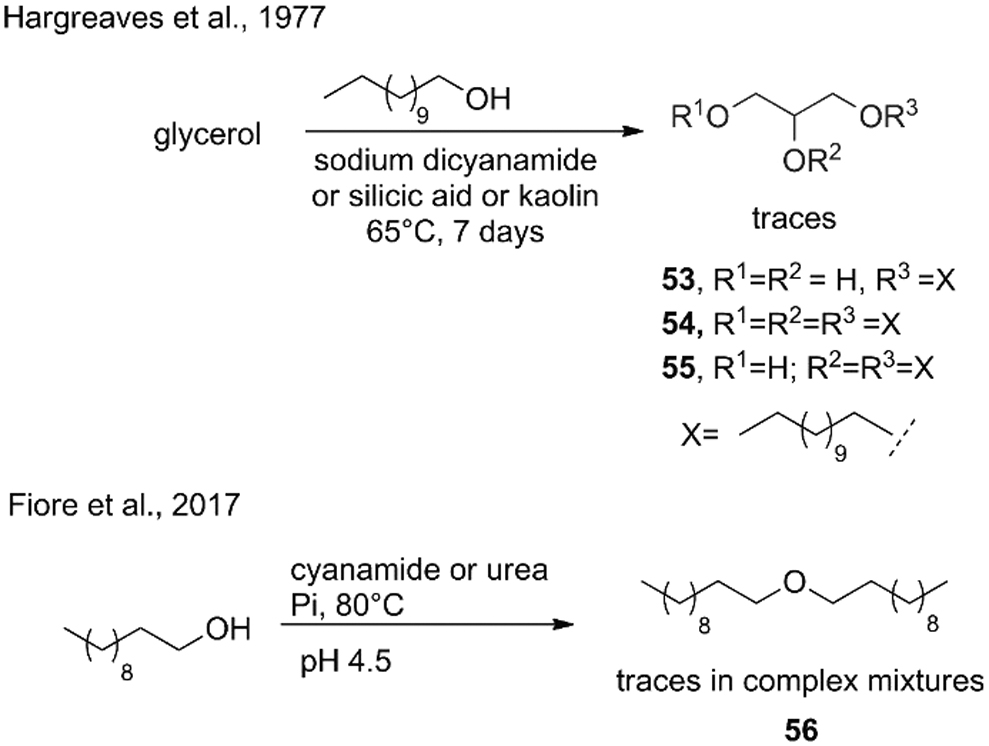
Tentative synthesis of ethers under prebiotic conditions.
Similarly, Fiore et al. (2017) have found traces of 1-undecoxyundecane (
4.5. Comparisons and analogies between prebiotic and biotic syntheses of phospholipids: synthesis of phospholipid esters
The striking difference between the organic synthesis and the biosynthesis of phosphatidates in Archaea is that the first acylation is localized in the cytoplasm of the Archaea, while the second acylation is catalyzed by an enzyme inside the membrane (Table 3).
Differences or Analogies Between Prebiotic and Biotic Syntheses of Phospholipids
CTP = cytidine triphosphate.
For example, the formation of an ether bond between G1P and the 20-carbon geranylgeranyl pyrophosphate (GGPP), yielding 3-O-geranylgeranyl-sn-glycerol-1-phosphate (Fig. 23), is catalyzed inside the cytoplasm by a geranylgeranyl pyrophosphate synthase (GGPP synthase) present in all Archaea except Nanoarchaeota (Jain, 2014). This may result in a better biological synthesis yield, compared with that of organic synthesis, since the lipid formed after its acylation is directly inserted in the membrane in the Archaea. GGPP synthases are regarded as Archaea-specific (Kate, 1993; Koga et al., 1993; Wächtershäuser, 2003; Matsumi et al., 2011), affecting the evolutionnary separation between the distinct domains of life (Peretó et al., 2004; Payandeh and Pai, 2007; Koga, 2011; Lombard et al., 2012b).
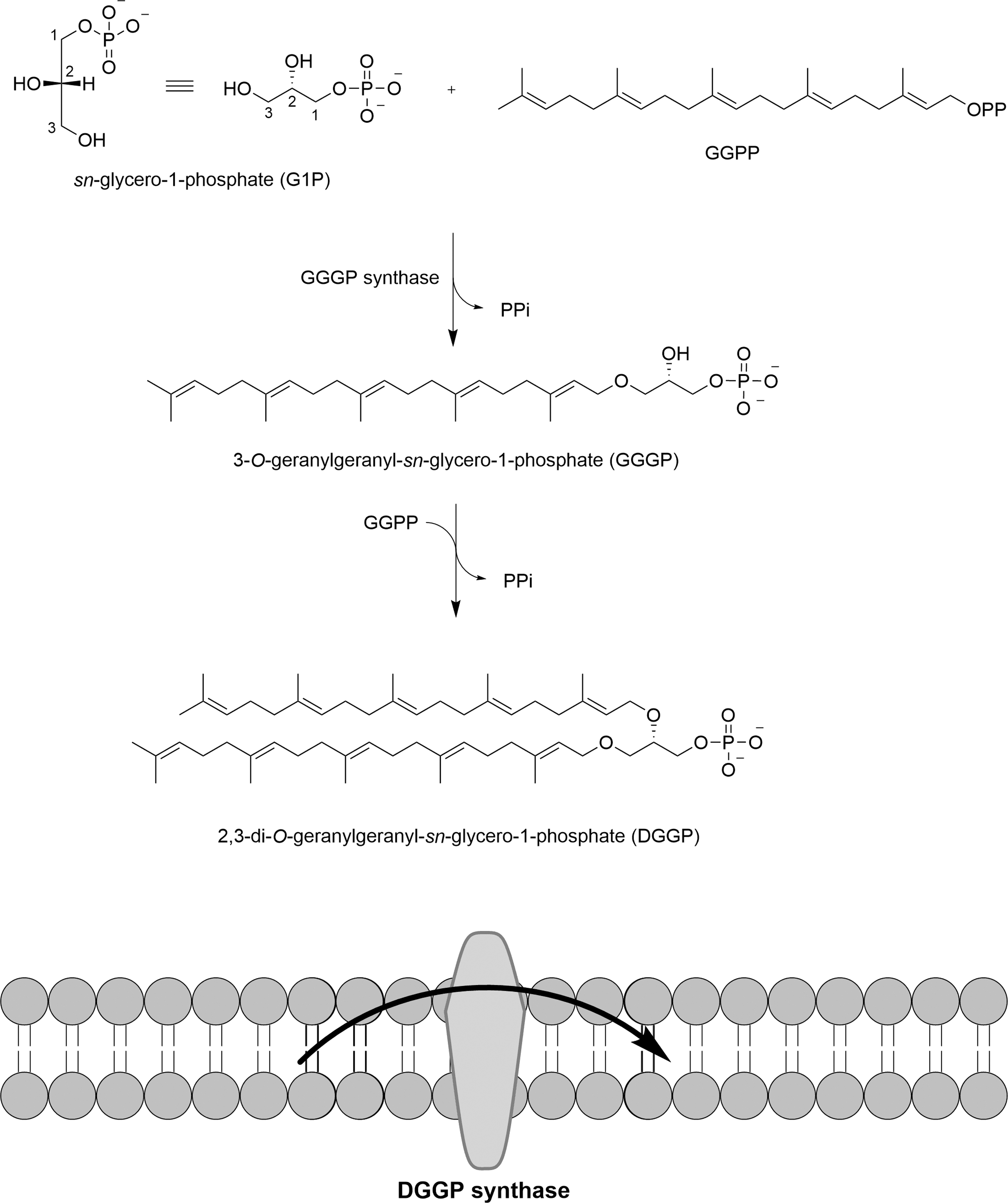
The formation of an ether bond between sn-glycerol-1-phosphate and GGPP in Archaea yields GGGP catalyzed by a GGGP synthase inside the cytoplasm. The formation of a second ether bond between GGGP and GGPP forming a DGGGP is catalyzed by a DGGGP synthase inserted in the membrane. DGGGP = 2,3-bis-O-geranylgeranyl-sn-glycerol-1-phosphate; GGGP = 3-O-geranylgeranyl-sn-glycerol-1-phosphate.
The hydrolysis of a pyrophosphate group (Fig. 23) is the source of energy to obtain GGPP, while for abiotic synthesis, the energy is provided during a thermal process (Table 3). In an attempt to provide a link between abiotic organic synthesis and evolved biochemical pathway, a minimal biochemical route involving a soluble enzyme FadD10 to form fatty acyl adenylates that react with amine-functionalized lysolipids to produce phospholipids was reported (Bhattacharya et al., 2019). The dodecanoyl-AMP was transfered to an amine-functionalized lysolipid to form a phospholipid inserted in a bilayer (Bhattacharya et al., 2019).
Although the biochemical route is far from abiotic conditions due to the presence of enzyme and lysolipid derivatives, this report stimulated further works toward possible evolution paths. This culminated with a model of enzyme-free synthesis of phospholipids in water from charged lysophospholipids and acyl donors (Liu et al., 2020).
In all the three domains of life, serine, ethanolamine, glycerol, and myoinositol as polar group moiety are found in phospholipids (Jain, 2014). To facilitate the transfer of the alcohol moiety of the serine to DGGP, activation of the DGGP from cytidine triphosphate (CTP) (Fig. 24) is necessary in Archaea. CTP is the energy analogue of ATP and is the source of energy to transfer head groups of phospholipids (Table 3).
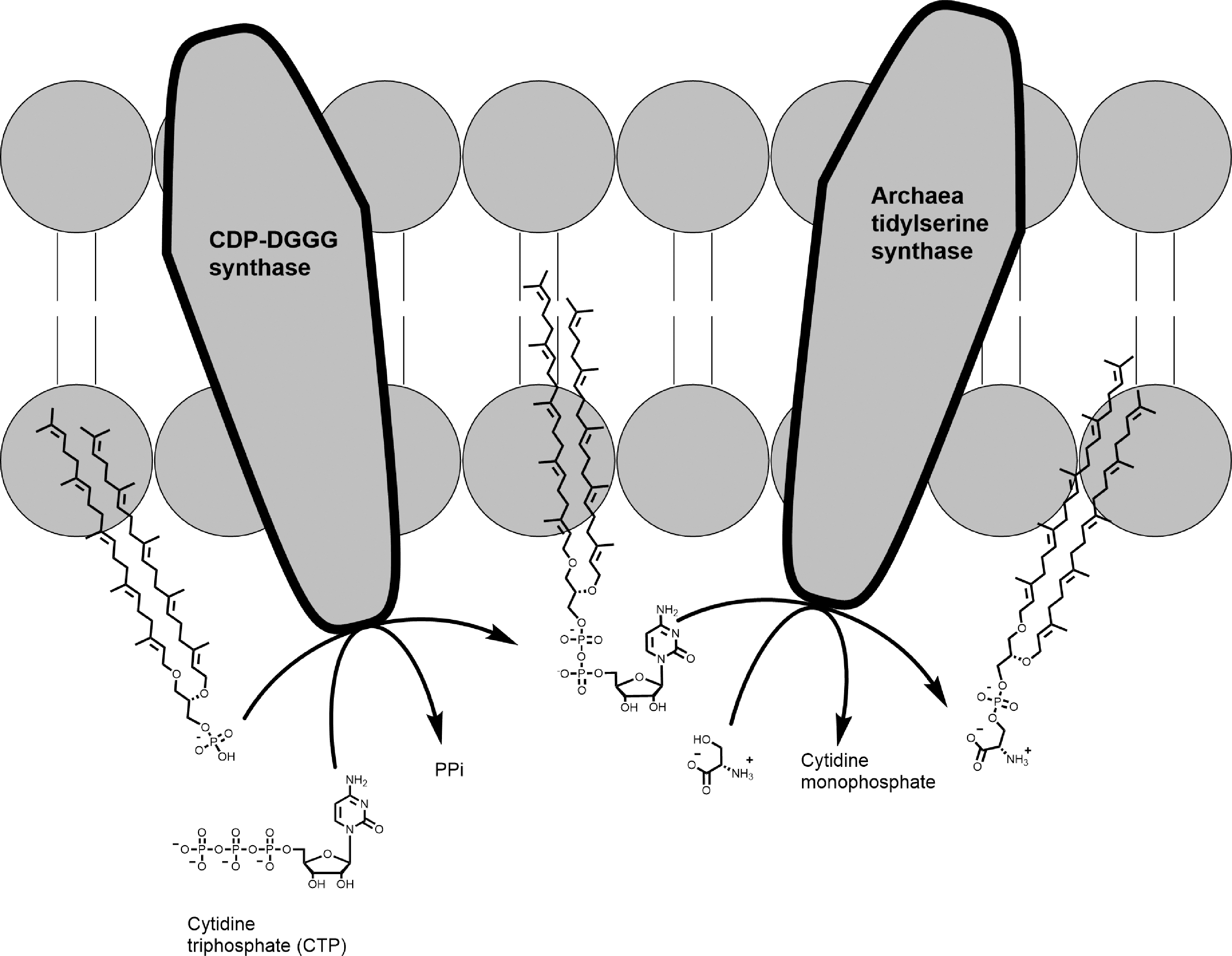
Formation of CDP-2,3-di-O-geranylgeranyl-sn-glycerol from DGGGP from CTP catalyzed by a CDP-DGGG synthase. Formation of DGGGPS from CDP-DGGG and
This reaction in Archaea is catalyzed by a transmembrane CDP-DGGG synthase (Fig. 24) (Morii et al., 2000; Koga and Morii, 2005, 2007; Jain, 2014; Jain et al., 2014; Ren et al., 2017). Hence, the phospholipid is activated, and formation of 2,3-di-O-geranylgeranyl-sn-glycerol-
However, CDP-DGGG synthase is specific to unsaturated CD-DGGG, suggesting that saturated phospholipids that are found in Archaea are formed by hydrogenolysis of unsaturated chains after the synthesis of the phospholipid (Fig. 24) (Jain et al., 2014). Inositol phospholipids are produced via CDP-DGGG and
In mammalians, the active sites of enzymes that catalyze the formation of phospholipids from either diacyl glycerol (DG) or PA are located at the cytosolic face of the ER (Vance, 2015). PE is synthesized in the ER and in mitochondria. PE in mitochondria is made in situ by decarboxylation of phosphatidylserine (PS) (Vance, 2015), probably due to an inefficient transport from the ER to mitochondrial membranes.
The biosynthesis of PS in Archaea, where the base exchange with serine can occur after the activation of phosphatidate by CTP, forming PS and CDP (Fig. 24), is not reported in mammalian cells. PS is synthesized in the ER of mammalian cells by a different base-exchange reaction in which the alcohol group of PC or of PE is replaced by serine (Vance, 2015). In mammalian cells, PS can be transported to mitochondria, where it can be decarboxylated to PE (Vance, 2015). Enzymes homologous to PS decarboxylase have been identified in Archaea (Daiyasu et al., 2005), suggesting that the decarboxylation of PS occurred in primitive cells.
Otherwise, there are two distinct pathways to generate phospholipids. Negatively charged phospholipids such as phosphatidylinositol (PI) and PG are synthesized from CDP-diacylglycerol at both the ER and mitochondria (Fig. 25) (Vance et al., 1977).
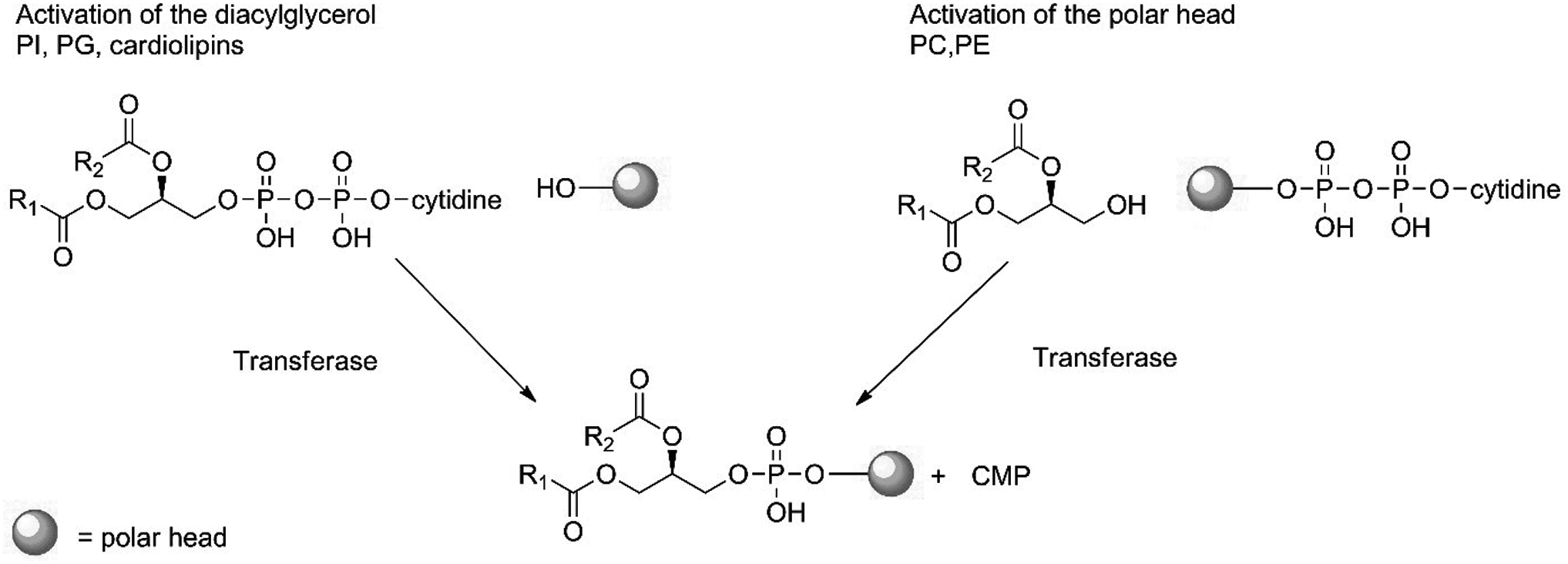
Synthesis of phospholipids via the CDP-diacylglycerol pathway for negatively charged phospholipids or via the CDP-alcohol pathway for neutral phospholipids. The polar head is the alcohol (inositol, glycerol, phosphadidylglycerol for the left side of the panel. Choline and ethanolamine for the right side of the panel).
PG is formed from a CDP-diacylglycerol and glycerol via a PG synthase (or transferase), while PI is formed from a CDP-diacylglycerol and myo-inositol via a PI synthase (or transferase) (Fig. 25). Diphosphatidylglycerol or cardiolipin is obtained from PG and a second CDP-diacylglycerol via a cardiolipin synthase. Neutral phospholipids such as phosphatidylcholine and PE are synthesized via CDP–choline and CDP–ethanolamine Kennedy-pathways (Kennedy and Weiss, 1956).
This starts from the formation of a phosphorylated choline (Fagone and Jackowski, 2013) or phosphorylated ethanolamine (Vance and Tasseva, 2013) via a choline kinase or ethanolamine kinase, respectively, in the cytoplasm. Then a phosphatidylcholine or ethanolamine cytidyl transferase catalyzes the formation of CDP-choline (Fagone and Jackowski, 2013) and CDP-ethanolamine, respectively (Vance and Tasseva, 2013). Alternatively, PE N-methyltransferase, a transmembrane protein in ER, catalyzes the reaction from PE to PC (Vance and Tasseva, 2013; Vance, 2014).
The majority of sphingomyelin synthases (SMs) are catalyzed by an SM synthase-1 in the Golgi apparatus from ceramide and PC, which are transported from the ER to the Golgi apparatus (Tafesse et al., 2006). A small part of SMs is obtained via SM synthase-2 in the plasma membrane. Archaeal phospholipids can be distinguished from bacterial phospholipids by their G1P backbone instead of a G3P backbone, respectively (Koga et al., 1998). CDP-diacylglycerol synthases are integral transmembrane proteins in E. coli, Saccharomyces cerevisiae, mice, and humans (Langley and Kennedy, 1978; Icho et al., 1985; Sparrow and Raetz, 1985; Kelley and Carman, 1987; Wu et al., 1995; Shen et al., 1996).
Instead of etherification in Archaea, esterification of acyl chains occurs in bacteria and higher evolved cells. Acylation shall start from G3P in Bacteria and Eukarya. However, a quite rare formation of monoacylglycerol from acyl CoA and either endogenous or extracellular source glycerol in mammalian was evidenced through glycerol:acyltransferase activity leading to monoacyl glycerol, and subsequently, diacylglycerol and triacylglycerol (Fig. 26) (Lee et al., 2001).
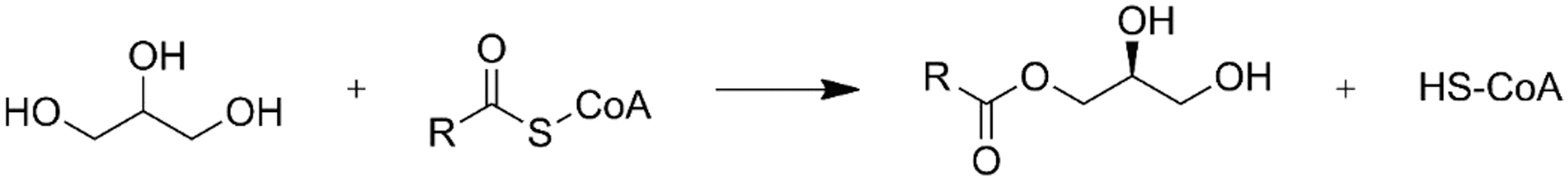
Rare formation of monoacyl glycerol from a direct acylation to glycerol and not from G3P.
The sn-glycerol-3-phosphate is acylated via an acyl CoA-SH forming a lysophosphatidate (Lyso-PA) and HS-CoA (Wilkison and Bell, 1997). Membranes of ER and the outer membrane of mitochondria contain distinct forms of glycerol-3-phosphate acyltransferase (Pellon-Maison et al., 2007). In most phosphatidates, the hydrocarbon chain at sn1 is saturated. A second acylation of Lyso-PA at sn2 by acyl-CoA (generally an unsaturated hydrocarbon chain) catalyzed by a lyso-PA-acyltransferase gives rise to CoA-SH and PA (Fig. 27), a precursor for CDP-diacylglycerol-dependent phospholipid synthesis.
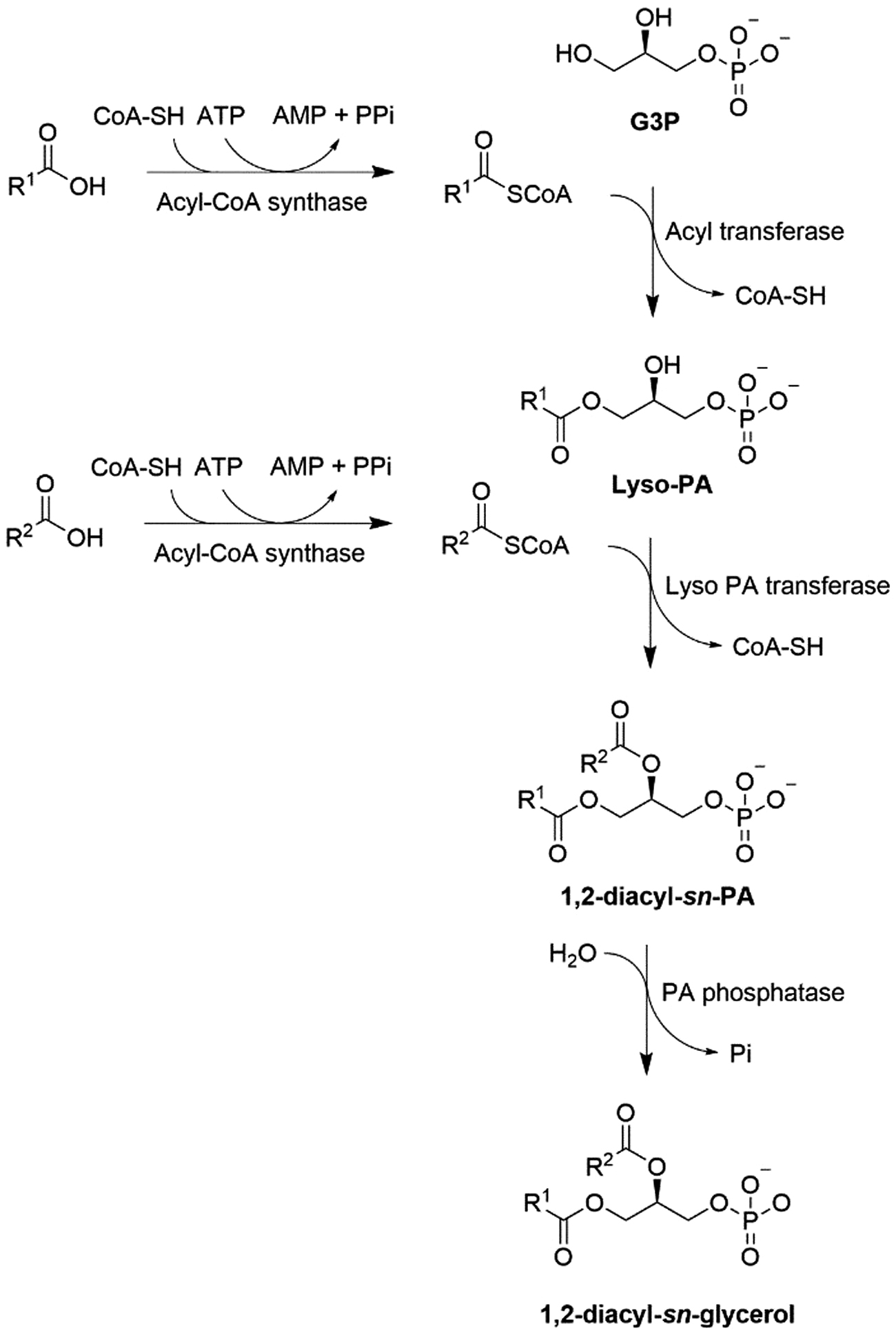
Formation of 1,2-diacyl-sn-glycerol-3-phosphate and 1,2-diacyl-sn-glycerol. R1 is a saturated chain, whereas R2 is unsaturated.
This is also catalyzed at the cytosolic side of ER and at the mitochondrial outer membranes (Vance, 2015). PA is further dephosphorylated by the lipin family of phosphatases to produce diacylglycerol. Before acylation, the fatty acid is activated by CoA-SH and ATP, providing a reactive acyl-CoA. PPi produced from ATP is hydrolyzed, favoring acyl-CoA formation, a substrate for the synthesis of triglycerides by DG acyltransferases and a precursor for phospholipid synthesis via the CDP–choline and CDP–ethanolamine Kennedy pathways (Fig. 25) (Kennedy, 1956; Lee and Ridgway, 2020).
In analogy with what nature does, bottom-up approaches for the synthesis of complete phospholipids suggest that the phosphorylation of glycerol followed by acylation using an “activated” fatty acid is very similar with biochemical processes acted in evolved cells. The main difference is that bottom-up approaches do not give any symmetry imbalance between R and S CLs, whereas evolved enzymes give enantiomeric pure glycerophosphates before their conversion into phospholipid esters (and ethers) (Table 3).
The main reason for the lack of symmetry imbalance observed during organic synthesis is probably due to the lack of selective catalysts that may function as enzymes. This is exemplified in the case of phosphorylation of glycerol, which provides solely either G1P or G3P during enzymatic reaction. Of importance, each reaction step during phospholipid formation is catalyzed by a specific enzyme, in contrast to prebiotic synthesis (Table 3). Again, prebiotic conditions do exclude the advantage of using plausible prebiotic cofactors where heat and condensing agents (dehydrating agents) (Fiore and Strazewski, 2016) promote the condensation between parts.
5. Phylogeny of Archaea, Bacteria, and Eukarya from LCA and C. commonote
The phylogeny tree of selected genes coding for the enzymes G1DPH and G3DPH may contribute to determine the biosynthesis routes of phospholipids. This strategy is based from an analysis of genes in Archaea (reminiscent of primitive cells), Bacteria, and Eukarya (considered more evolved descendants). LCA or C. commonote (Yamagishi, 1998) lived around 3.8–3.5 Ga ago.
There are still controversies about the environment where the LCA lived (Weiss et al., 2016). A sulfur-containing atmosphere (Kasting et al., 1989; Glansdorff et al., 2008; Olson, 2019) together with CO2, H2, N2 and CH4 (Catling and Zahnle, 2020) is the most probable. Contemporary Archaea, comprising methanogens (which currently generate around 85% of the methane in Earth's atmosphere), Halobacteria, and thermoacidophiles live under extreme conditions reminiscent of this primitive environment. These descendants are phylogenetically related to each other, while they share very little phylogenetic characteristics with Bacteria and Eukarya (Koga et al., 1998; Yokobori et al., 2016).
C. commonote had archaeal and bacterial characteristics (Guy and Ettema, 2011; Akanuma et al., 2013; Eme and Doolittle, 2015; Eme et al., 2017), while Eukarya evolved from Archaea (Guy and Ettema, 2011; Akanuma et al., 2013; Eme et al., 2017). There is an open debate between the three domains of life (Archaea, Bacteria and Eukarya) that evolved separately from the LCA versus the Eocyte hypothesis where Eukarya are descendants of Archaea Crenarchaeota (Williams et al., 2013) or other evolution models (Akanuma et al., 2013; Weiss et al., 2016; Yokobori et al., 2016).
A comparative analysis of genes within the different species, as “finger prints,” shall indicate their descendant characteristics when their genes are homologues, while a separate evolution of species is inferred when genes are distinct from each other. The origin of the controversy lies in the inconsistencies of the phylogenetic distributions and in the selection of appropriate genes to build up the phylogenetic tree (Akanuma et al., 2013; Lake and Sinsheimer, 2013; Yokobori et al., 2016).
Here, we focus on the phylogenetic tree based on the genes that encode sn-glycerol-1-phosphate dehydrogenase (G1DPH) or sn-glycerol-3-phosphate dehydrogenase (G3DPH), enzymes that catalyze the formation of, respectively, sn-G1P and sn-G3P from prochiral DHAP. These genes may not represent the best descendant path. The reason to focus on the two genes for encoding G1DPH and G3DPH in this review is that G1P and G3P are key enantiomeric precursors of phospholipids. The choice of these two genes is essential to determine the mechanisms of symmetry breaking associated with the enantiomeric excess of G1P and G3P.
Abiotic synthesis from achiral compounds yields racemic mixtures. How chiral molecules such as G1P and G3P emerged may reveal the plausible evolution of how racemic phospholipids became enantiomeric phospholipids due to the appearance of enzymes G1DPH and G3DPH.
The lipid composition in Archaea is distinct from those in Bacteria and Eukarya (Akanuma et al., 2013; Yokobori et al., 2016). Archaea membranes usually contain phospholipids having G1P moiety and isoprenoid hydrocarbon chains ether-linked to the G1P moiety (Yokobori et al., 2016), whereas membranes in Bacteria and Eukarya are usually composed of phospholipids derived from G3P and alkyl chains ester-linked to the G3P moiety (Koga et al., 1998; Yokobori et al., 2016). The presence of genes coding for these enzymes in Archaea and Bacteria has contributed to build up a phylogenetic tree (Daiyasu et al., 2002; Peretó et al., 2004; Carbone et al., 2015; Yokobori et al., 2016), revealing possible evolutions of synthetic pathways from a common ancestor.
Several models, based on the occurrence of G1P-lipids or G3P-lipids, were inferred from the presence of either G1PDH based from EgsA/AraM genes coding for this enzyme, or G3PDH based from GpsA, GlpA/GlpB genes (Kandler, 1995; Koga et al., 1998; Wächtershäuser, 2003; Yokobori et al., 2016). It was suggested that Commonote bacteria and Commonote archaea could have emerged directly from the LCA (Martin and Russell, 2003) instead of cellular and heteromembranous C. commonote (Peretó et al., 2004) giving rise to a distinct evolutionary tree.
C. bacteria and C. archaea plausibly acquired, respectively, G3PDH and G1PDH from C. commonote (Fig. 28A) or from LCA (Fig. 28B, C). Eventually, both G3PDH and G1PDH coexisted (Fig. 28D), which led to either C. bacteria containing G3P-lipids or C. archaea containing G1P-lipids that were preserved up to Bacteria and Archaea. The phylogenetic analysis of gene product G1PDH inferred that G1PDH was acquired by C. archaea, suggesting that neither the common ancestor of Bacteria nor C. commonote had G1PDH (Yokobori et al., 2016). On the contrary, the phylogenetic analysis of gene product G3PDH is consistent with the presence of genes GlpA/GlpD coding for G3PDH (in C. commonote and in C. archaea) (Fig. 28B), which makes this evolutionary tree the most likely. This suggests that G3PDH occurred before G1PDH.
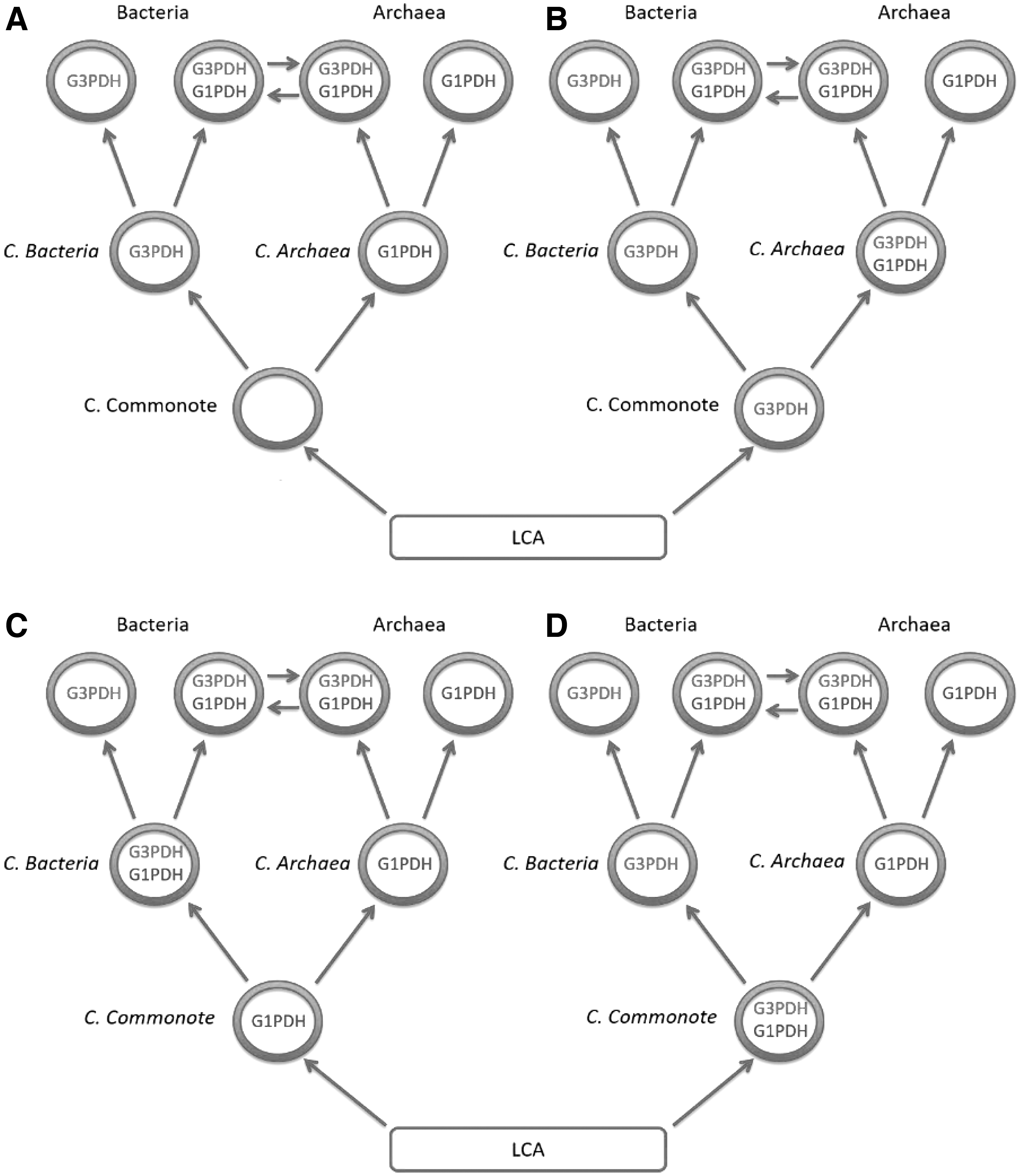
Models of early evolution of membranes based on the phylogenetic analysis of G1PDH and G3PDH genes. Adapted from Yokobori et al. (2016).
Of further interest is that another gene GpsA coding for G3PDH (GpsA) was acquired in C. bacteria, which is the major G3PDH in Bacteria as well as in Eukarya (Yokobori et al., 2016). GlpA/GlpD and EgsA genes are different, indicating that G3PDH and G1PDH evolved apart from each other (Koga et al., 1998).
6. Conclusions
Phospholipids are clearly more complex than simpler amphiphiles such as fatty acids, monoalkyl phosphates, isoprenoids, and most probably participated in the formation of protocellular membranes together with several other components. Phospholipids, in addition to their chemical diversity of headgroups and tails, are chiral molecules. Theoretical predictions and experimental evidence confirm that phospholipids form abiotically as racemic mixtures. This implies that the occurrence of enantiomeric phospholipids should have been initiated from chiral products or from selective catalytic reactions. It is more likely that the reactions catalyzed by the nonenzymatic process appeared before than those that were catalyzed by enzymes.
In this respect, amino acids with L configurations are present in G3PDH and G1PDH enzymes, which points out that the controlled production of enantiomeric phospholipids was probably initiated at a later stage of the evolution path than those catalyzed by the nonenzymatic process. This is also supported by the fact that the chemical energy for the phospholipid biosynthesis originates mostly from ATP, acetyl-S-CoA, and NAD(P)H, which have a ribosyl derivative of configuration D in the adenosine moiety. Therefore, not only enantiomeric amino acids but also enantiomeric carbohydrates contributed to initiate the efficient formation of enantiomeric phospholipids.
In analogy with the Fischer/Tropsch processes, the biosynthesis of a fatty acid starts from natural carbon sources such as CO2. Recent discoveries also showed that the reduction of glyceraldehyde was possible under enzyme-free conditions thanks to cyanide–cyanocuprate photochemistry. This suggests that simple but effective catalytic networks probably existed in protocells before the advent of LCA. A wide range of plausibly prebiotic conditions by condensation of acyl chains, glycerol backbone, and inorganic phosphates in the presence of condensing agents such as cyanamide, sodium dicyanamide, or urea yield phospholipids having phosphoester links. Glycerol phosphate was also used as a phospholipid precursor and its acylation occurred under similar conditions upon chemical activation, in analogy with biological processes driven by coenzymes and ATP as energy sources. Abiotic condensation conditions require mild temperatures and wet/dry cycles in fluctuating hydrothermal pools (Gözen, 2021).
From a systems chemistry point of view, these few analogies suggest that in abiotic conditions, the properties of phospholipids emerged from inanimate mixtures (Dalai and Sahai, 2019; Gözen, 2021). The unique property to form boundaries, upon hydration, permits to colocate microenvironments where Darwinian evolution cycles placed, protected, and kept other biomolecules (nucleic acids and peptides) at defined concentrations, avoiding chemical degradation. This microenvironment enables a multitude of often competing and interfering biochemical reactions to occur simultaneously.
Footnotes
Acknowledgments
M.F. wishes to dedicate this work to his beloved daughter Océane (2015–2017). Collaboration among E.A. and M.F. has been fostered by the European COST Action CM1304 Emergence and Evolution of Complex Chemical Systems. D.F. and M.F. acknowledge cofunding from the Volkswagen Stiftung (Molecular Life, Az. 92 850); Volkswagen Stiftung funded the PhD program of D.F. (2017–2020).
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was provided for this study.
Abbreviations Used
Associate Editor: Lewis Dartnell
