Abstract
Amino acids have been detected in extraterrestrial bodies such as carbonaceous chondrites (CCs), which suggests that extraterrestrial organics could be the source of the first life on Earth, and interplanetary dust particles (IDPs) or micrometeorites (MMs) are promising carriers of extraterrestrial organic carbon. Some amino acids found in CCs are amino acid precursors, but these have not been well characterized. The Tanpopo mission was conducted in Earth orbit from 2015 to 2019, and the stability of glycine (Gly), hydantoin (Hyd), isovaline (Ival), 5-ethyl-5-methylhydantoin (EMHyd), and complex organics formed by proton irradiation from CO, NH3, and H2O (CAW) in space were analyzed by high-performance liquid chromatography and/or gas chromatography/mass spectrometry. The target substances showed a logarithmic decomposition over 1–3 years upon space exposure. Recoveries of Gly and CAW were higher than those of Hyd, Ival, and EMHyd. Ground simulation experiments showed different results: Hyd was more stable than Gly. Solar ultraviolet light was fatal to all organics, and they required protection when carried by IDPs/MMs. Thus, complex amino acid precursors (such as CAW) were possibly more robust than simple precursors during transportation to primitive Earth. The Tanpopo 2 mission is currently being conducted to expose organics to more probable space conditions.
1. Introduction
1.1. Prebiotic formation of organic compounds: Terrestrial versus extraterrestrial
Organic compounds accumulated on primitive Earth prior to the generation of life, and it is not yet clear whether they were formed in primitive terrestrial or extraterrestrial environments or both. Many experiments have been conducted since the 1950s to simulate methods of possible organic compound formation, particularly with respect to bioorganic compounds such as amino acids. In the early stage of simulation experiments, gas mixtures (such as a mixture of CH4, NH3, and H2O) were often reduced (Miller, 1953; Harada and Fox, 1964; Bar-Nun et al., 1970; Sagan and Khare, 1971). However, as primitive Earth's atmosphere was generated during accretion by planetesimal impacts, it was suggested that the atmosphere would not have been strongly reducing (e.g., Matsui and Abe, 1982; Kasting, 1990), and amino acids and other N-containing compounds could not have been easily produced under such a low reducing atmosphere (Schlesinger and Miller, 1983; Kuwahara et al., 2012).
A wide variety of organic compounds have been found in extraterrestrial bodies such as carbonaceous chondrites (CCs) (Kvenvolden et al., 1970; Pizzarello et al., 2006; Schmitt-Kopplin et al., 2010; Elsila et al., 2016) and comets (Kissel and Krueger, 1987; Goesmann et al., 2015), and researchers believe they could be the method of transport delivering organics to primitive Earth. For example, the known candidate organic carriers, interplanetary dust particles (IDPs; mainly collected in the stratosphere) or micrometeorites (MMs; mainly collected in Antarctica), are derived from comets and asteroids (Bradley, 2007). Chyba and Sagan (1992) estimated that more than 10 kt/yr of organic carbon was delivered by IDPs/MMs at 3.8 Gyr ago, which exceeds that delivered by meteorites and comets or that relating to endogenous production.
1.2. Amino acids and their precursors in extraterrestrial bodies
Most of the organics found in CCs and IDPs/MMs are relatively complex molecules with a large molecular weight, and they are insoluble in water and other solvents (Alexander et al., 2017), but CCs also contain soluble organic molecules, including amino acids. For example, in the case of the Murchison meteorite (CM2), approximately 0.1% of the total organic carbon exists in the form of amino acids (Kvenvolden et al., 2000; Sephton, 2002). However, the soluble organic compound content (including amino acids) of IDPs/MMs has not been adequately clarified.
Interplanetary dust particles and micrometeorites have been collected in ocean sediments, Antarctic ice, and air in the stratosphere (Robert, 2015), and very small amounts of bioorganic compounds have been found therein. However, as these were collected in the terrestrial biosphere, it is difficult to determine whether they are pristine to the IDPs/MMs. Matrajt et al. (2004) reported the detection of α-aminoisobutyric acid in micrometeorites recovered from Antarctic ices. As α-aminoisobutyric acid is a nonprotein amino acid that is rarely found in the terrestrial biosphere, it is feasible that IDPs/MMs can deliver certain amino acids. Nevertheless, it is difficult to determine the origin of the amino acids, as contamination of amino acids could easily have occurred when the Antarctic ice was thawed to extract the dust particles. Therefore, to avoid terrestrial contamination of amino acids to IDPs, it would be useful to collect IDPs from outside the terrestrial biosphere. The Tanpopo mission used ultra-low-density silica aerogel to collect dust within low Earth orbit (Yamagishi et al., 2011), and the analysis of amino acids within captured particles is currently in progress.
1.3. Characterization of amino acid precursors formed in simulated extraterrestrial environments
Information about the molecular structure of amino acids delivered to Earth by extraterrestrial bodies is limited. Concentrations of amino acids in water extracted from CCs have been reported to increase after acid hydrolysis (Glavin et al., 2020), which indicates that amino acids are partly present in the form of amino acid precursors in CCs. Therefore, it is necessary to determine what types of amino acid precursors exist in extraterrestrial bodies. Hydantoin (Hyd) (Fig. 1a) and its derivatives are possible candidates, as Hyd has been detected in CCs (Shimoyama and Ogasawara, 2002). Simulation experiments have suggested that alternative amino acid precursor candidates could be complex molecules with large molecular weights, and several scenarios have been proposed to explain the origins of meteoritic amino acids: for example, Greenberg and Li (1997) proposed that cometary organics were formed in molecular clouds prior to the formation of the Solar System. In this respect, most molecules such as H2O, CO, CH3OH, and NH3 were frozen onto the surface of interstellar dust, an ice mantle was formed in molecular clouds at cold temperatures (10–20 K), and it is likely that reactions with galactic cosmic rays (GCRs) and GCR-induced ultraviolet (UV) light formed molecules with greater complexities. The main component of a GCR is a proton whose spectrum is nearly flat from 101 to 108 GeV (Berezhko and Völk, 2007).
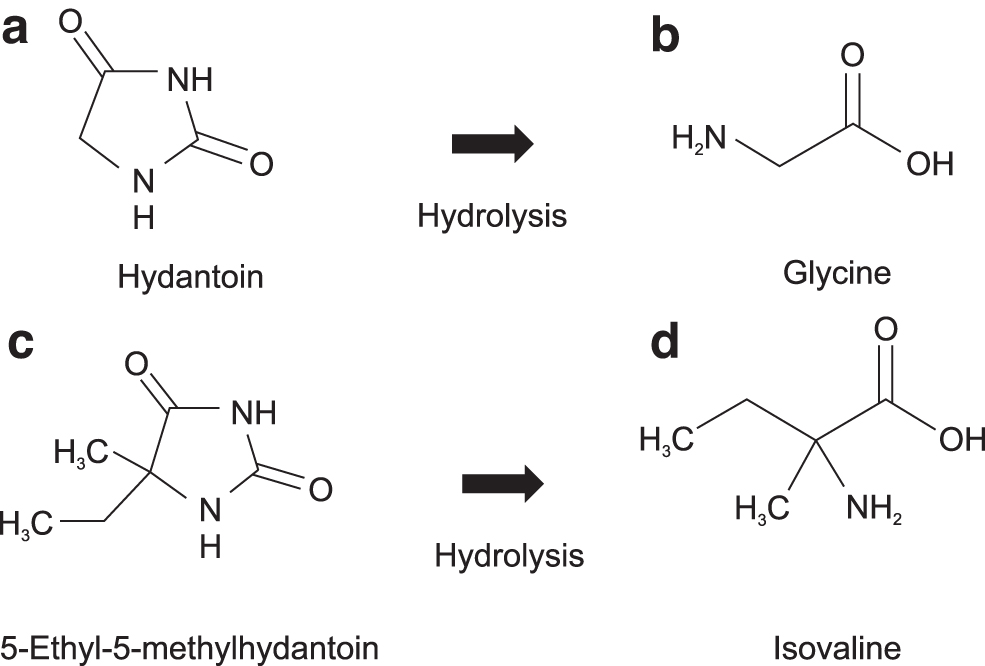
Structures of the target molecules (amino acids and their simple precursors). IUPAC names are (
Complex organics (including amino acid precursors) were then formed when interstellar ice analogs were irradiated with high-energy particles (Kobayashi et al., 1995; Kasamatsu et al., 1997a) or UV light (Bernstein et al., 2002; Munoz Caro et al., 2002). In these experiments, amino acid precursors were formed instead of free amino acids. Furthermore, via proton irradiation of interstellar media analogs, Takano et al. (2004a, 2004b) found that the major products after proton irradiation of a mixture of CO, NH3, and H2O were complex molecules containing large-molecular-weight (800–3000 Da) compounds with complex structures. The product synthesized from a mixture of CO, NH3, and H2O by proton irradiation is hereafter referred to as CAW.
In addition to the molecular cloud scenario, Cody et al. (2011) proposed that complex organics were formed in the interior of small bodies in the early Solar System in the presence of liquid water and showed similarity in molecular structures between insoluble organic matter in CCs and hydrothermal products from an aqueous solution containing formaldehyde and glycolaldehyde (CH2OHCHO). When an aqueous solution containing formaldehyde, glycolaldehyde, and NH3 was reacted under conditions simulating the interior of meteorite parent bodies, amino acid precursors were formed in addition to solid macromolecules, which resembled the insoluble organic matter in CCs (Kebukawa et al., 2013, 2017). The amino acid precursors were mainly complex organics with large molecular weights (Elmasry et al., 2021b), and simulation experiments conducted for both scenarios resulted in the formation of complex organics with large (several hundred daltons or more) molecular weights.
It is thus reasonable to consider that such complex organic compounds whose molecular weights are over several hundred daltons can be added to the list of amino acid precursors that are possibly present in meteorites and IDPs/MMs.
1.4. Stability of amino acids and their precursors in space
Organic molecules (including amino acid-related compounds) formed in dense clouds might have been imported to small bodies accreted in the cold outer region of the protoplanetary disk such as comets (Herbst and van Dishoeck, 2009) and delivered to primitive Earth in meteorites, comets, and/or IDPs/MMs. Organics formed in asteroid interiors would also have been delivered by meteorites and IDPs/MMs.
However, prior to their arrival on Earth, they would have been altered by various energies in space: interstellar dust in molecular clouds would have been exposed to GCRs and induced UV light; dust in protosolar nebula would have been irradiated with harsh X-rays and UV light from the young Sun, and organics in asteroids would have been exposed to γ-rays originated from radioactive nuclides such as 26Al and resulting heating (MacPherson et al., 1995; Iglesias-Groth et al., 2011). Dust ejected from meteorites and comets would have been directly exposed to GCRs, solar energetic particles (SEPs), and the full spectrum of solar radiation in interplanetary space.
We should thus consider the stability of organic compounds against different radiation exposure to discuss whether they could have safely arrived on primitive Earth. Numbers of studies have analyzed the stability of amino acids and other bioorganic compounds in space (e.g., Ehrenfreund et al., 2001). For example, Takano et al. (2004c) examined the stability of free amino acids and bound amino acids (in albumin) against γ-rays (from a 60Co source) and UV light (from a D2 lamp) and found that bound amino acids were considerably more stable than free amino acids after both types of irradiation. Sarker et al. (2012) compared the stability of isovaline (Ival) and 5-ethyl-5-methylhydantoin (EMHyd, cyclic precursor of Ival) against γ-rays and UV light and found that the cyclic precursor was more radiostable than Ival but that Ival was somewhat more photostable than the precursor. The radiochemical stabilities of free amino acids and bound amino acids were also examined against heavy ions, and results showed that complex amino acid precursors made by proton irradiation of a mixture of CO, NH3, and H2O (CAW) and amino acids comprising albumin were more stable than free amino acids (Kobayashi et al., 2004).
1.5. Space experiments to examine stability of organics in space: Objectives of the present research
These results suggest that free amino acids and their precursors have different stabilities in simulated space environments. As there is limited information about the form (free or precursor) of amino acids in IDPs/MMs, it is thus necessary to determine which form of amino acids could have been protected and reached Earth. In space, molecules are exposed to various energetic photons (including solar X-rays, vacuum ultraviolet light, and near-ultraviolet light) as well as GCRs and SEPs. In ground experiments, we can test the stability against each type of radiation exposure, but it is difficult to see their synergetic effects; therefore, space exposure experiments are required to examine real stability in space.
Several space experiments have exposed organic compounds to space environments. Of these, the AMINO experiment and the PROCESS experiment included the space exposure of amino acids: the AMINO experiment was conducted as a part of the EXPOSE-R from 2009 to 2011 by utilizing the Russian exposed facility Zvezda on the International Space Station (ISS; Bertrand et al., 2015), and the PROCESS experiment was performed in the EXPOSE-E from 2008 to 2009 on the European Technology Exposure Facility platform on the ISS (Bertrand et al., 2012). Various free amino acids deposited on MgF2 windows with or without meteorite powders were exposed to space in these experiments, and the superior survival of amino acids with meteorite powders was reported. However, no space exposure experiments have exposed amino acid precursors to enable a comparison with the results of free amino acids.
The first Japanese astrobiology experiments in Earth orbit were named “the Tanpopo mission” and conducted from 2015 to 2019. As a part of this mission, amino acids and their precursors were exposed to space environments to test their stability in space. The targeted amino acids were glycine (Gly) (Fig. 1b), the simplest amino acid, and Ival (Fig. 1d), an asymmetric nonprotein amino acid often found in CCs (Cronin and Pizzarello, 1997). Their small cyclic precursors, Hyd (Fig. 1a) and EMhyd (Fig. 1c), were also studied. In addition, a proton irradiation product of CO, NH3, and H2O (CAW) was used as a complex precursor of amino acids (Takano et al., 2004a).
The objective of the present work was to compare the stability of free amino acids and their precursors (simple precursors vs. complex precursors) in the outer space (low Earth orbit) environment. In addition, we discussed what type of amino acids (free, simple precursors, or complex precursors) are robust after their production in space and delivery to Earth.
2. Material and Methods
2.1. Chemicals
All target organics used in the space exposure experiment, except Hyd, were labeled with 13C. 2-13C-Gly was purchased from Cambridge Isotope Laboratories, Inc.; 4-13C-EMHyd was synthesized from methylethylketone, ammonium carbonate, and 13C-KCN (Cambridge Isotope Laboratories, Inc.) by Bucherer-Bergs Reaction; and 1-13C-Ival was obtained by the alkaline hydrolysis of EMHyd (Pirkle et al. 1992). Hyd (a precursor of Gly, special grade), hexatriacontane (HTC; special grade), HCl (amino acid analysis grade), and ethanol (special grade) were purchased from Fujifilm Wako Co.
The procedure used to synthesize CAW was as follows: a mixture of 13CO (350 Torr, ISOTEC, USA, 13C > 99%) and NH3 (350 Torr, Ultra-Pure Grade, Sumitomo Seika Co.) together with 5.0 mL H2O purified with a Milli-Q system were introduced to a Pyrex glass tube (400 mL; Takano et al., 2004a) with a Havar foil (Nilaco Co.) window. The gas mixture was irradiated with 2.5 MeV protons through the window from a tandem accelerator at Tokyo Institute of Technology (Tokyo, Japan): the total energy deposited to the gas mixture was calculated to be 3.16 kJ, which was equivalent to cosmic ray (>100 MeV) flux in the molecular cloud (1 m2) for 107 years (Morfill et al., 1976; Kasamatsu et al., 1997b). The resulting products were recovered by adding 10 mL of pure H2O.
All glassware and metallic parts of the exposure units were heated in an oven at 500°C for approximately 5 h prior to use for sterilization and removal of organic compounds.
2.2. Sample setup for space experiment
In the Tanpopo mission, Exposure Panels (10 cm × 10 cm × 2.0 cm) were developed and used together with Capture Panels. Each Exposure Panel was divided into 20 rectangular exposure units, and each of these contained an aluminum plate (18 mmφ) (Yamagishi et al., 2018). The aluminum plate for exposing organic compounds contained 19 pits, as shown in Fig. 2. We injected 1.5 μL each of 50 mM aqueous solution of Gly, Hyd, Ival, and EMHyd into each pit; therefore, each pit exposed to space contained 75 nmol of each compound. Three pits in a single plate were used for each compound. CAW was also injected into the pits as an aqueous solution; the amount of CAW injected per pit was controlled to yield 15 nmol of Gly after acid hydrolysis. After air-drying on a clean bench, 1 μL of saturated HTC solution in ethanol was added to each pit, and they were then air-dried to enable a thin membrane to form, which prevented the sublimation of the molecules in space. Usage of HTC was determined at the early stage of the Tanpopo project. Because the highest temperature of the samples was estimated to be higher than 80 °C, we considered the safety regulations and decided to use HTC in order to protect samples from the heat sublimation caused by high temperatures. As mentioned in our results, the highest temperature was 24 ± 5 °C (Yamagishi et al., 2018). Because HTC molecules absorb light with wavelengths shorter than 160 nm, irradiation with UV light may have only limited effect in regions with wavelengths larger than 160 nm.
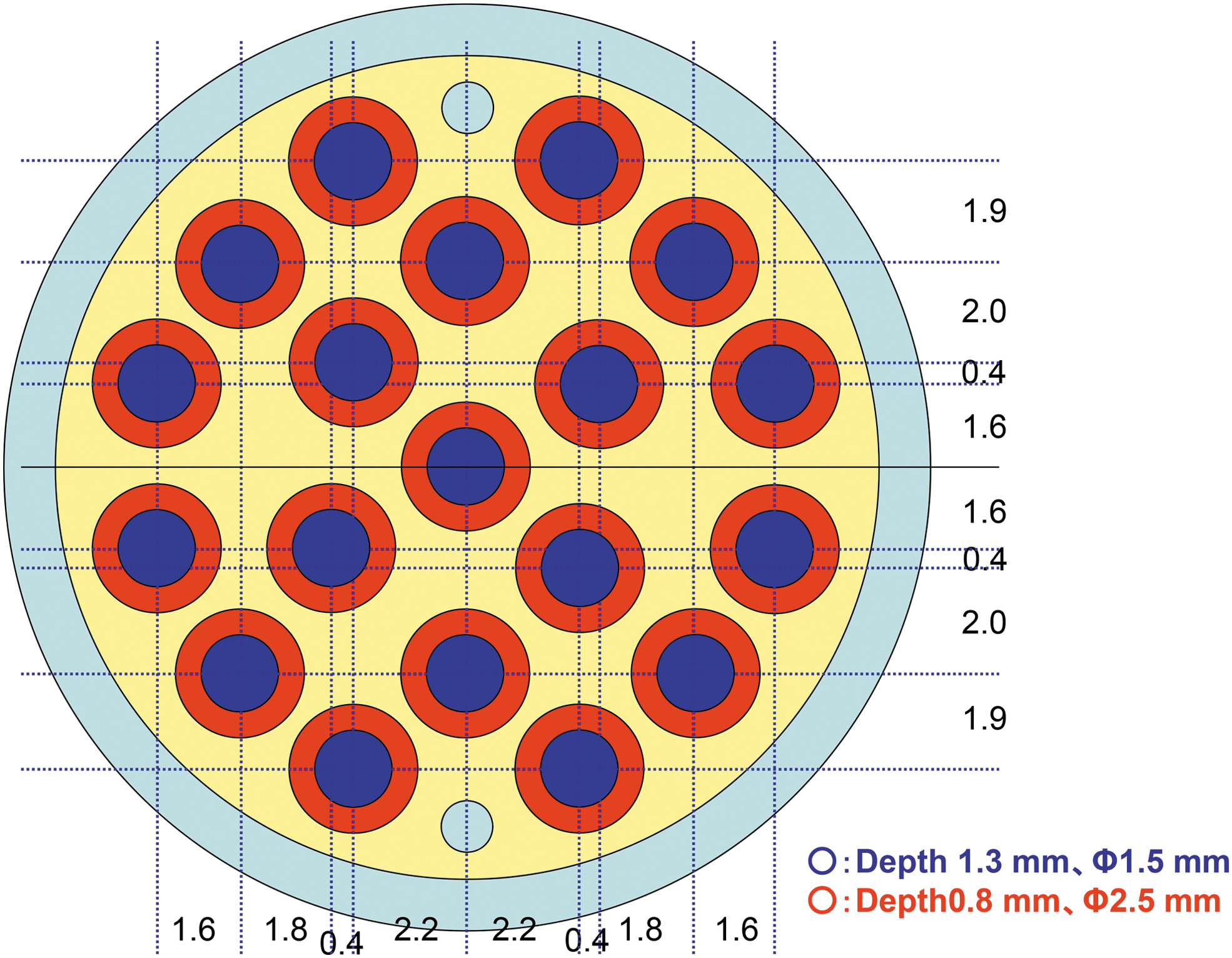
Exposure plate (plain view). The diameter of the plate is 18 mm. Each plate was set to the exposure unit (Fig. 3) with a quartz window or a MgF2 window. Unit of length is millimeters.
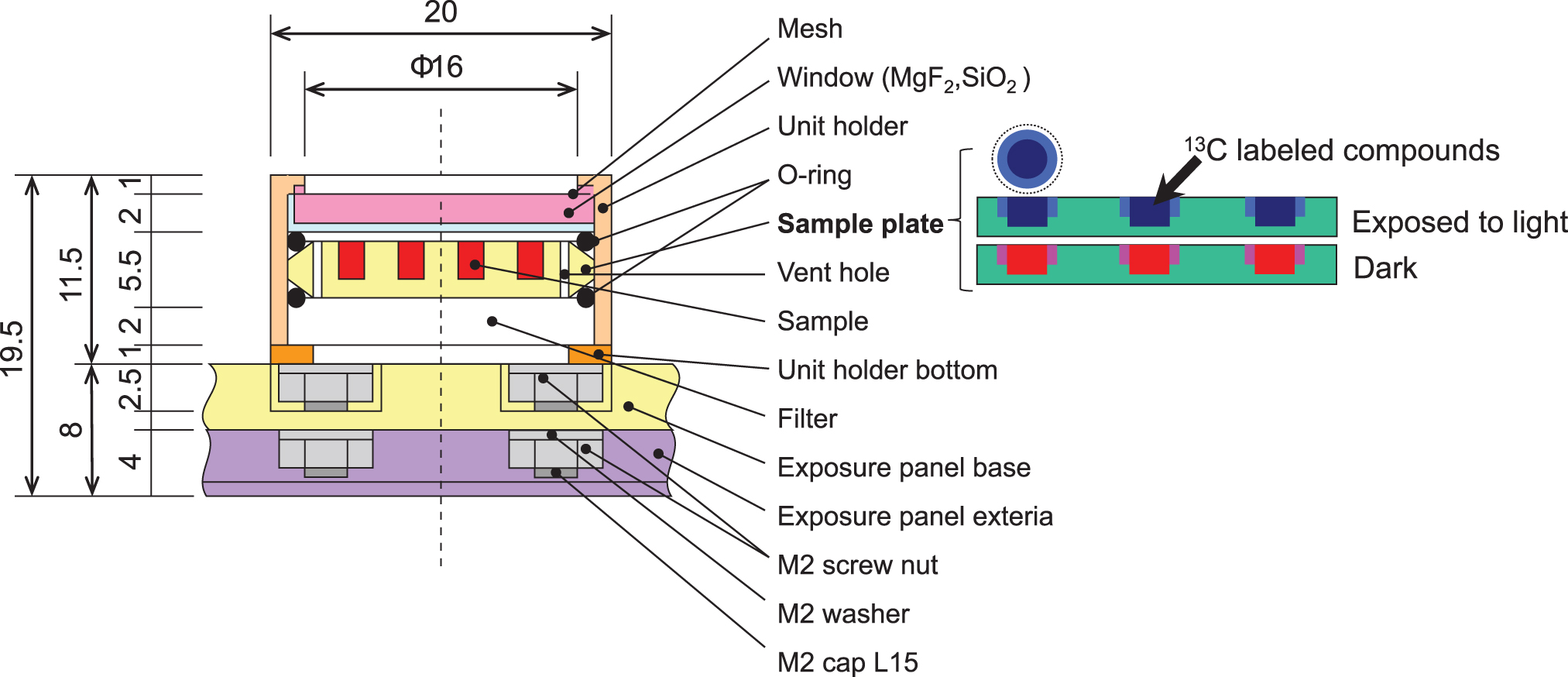
Exposure unit (cross-sectional side view). One unit (20 mm × 25 mm × 20 mm) contained two exposure plates. One plate was attached with a window (MgF2 or quartz) and a metal mesh and exposed to light. The other was set beneath it and was not exposed to light (“dark”). Unit of length is millimeters.
Figure 3 shows a cross-section of the exposure unit. In each of the exposure units, two plates were set horizontally (as shown in the figure): one was set under a window (quartz or MgF2) and exposed to both cosmic radiation and solar light, and the other was set beneath the upper plate and exposed only to cosmic radiation (the Dark control). Each Exposure Panel contained 20 exposure units (Fig. 4): each Panel contained exposure units with quartz or MgF2 windows (16 mmφ) together with microbe exposure units and an alanine UV dosimeter (to be described later). The windows were covered with a metal mesh to protect against scattering from accidentally broken windows following ISS regulations. A #350 stainless steel wire mesh with a diameter of 0.015 and transmittance of 0.29 was used. The exposed samples with quartz windows are hereafter referred to as the quartz samples and those with MgF2 windows as the MgF2 samples.

Exposure panel (plain view photo). The panel was 100 mm × 100 mm × 20 mm. One panel consisted of 20 exposure units.
Three of the Exposure Panels were attached to the Exposure Handrail Attachment Mechanism (ExHAM) on the Exposed Facility (EF) of the Japanese Experimental Module (JEM) for 1, 2, or 3 years from May 26, 2015; another three Panels were stored in the cabin of JEM for 1, 2, or 3 years (the Cabin control); and three additional Panels were stored in an incubator with desiccant blocks at 20°C at Tokyo University of Pharmacy and Life Sciences for 1, 2, or 3 years (the Ground control).
Temperature was monitored with a mechanical thermometer (Hashimoto et al., 2016, 2019; Yamagishi et al., 2018). Aluminum oxide–based optically stimulated luminescence dosimeters and silver-activated phosphate glass-based radiophotoluminescence dosimeters were used to measure radiation dosimetry inside and outside the ISS (Yamagishi et al., 2018). A newly developed alanine vacuum ultraviolet light (VUV) dosimeter was used to monitor the UV/VUV dose: an alanine film coated on a quartz window absorbs VUV wavelengths shorter than 190 nm, and alanine is dissociated with VUV light. The UV/VUV dosage was calculated based on the dissociation quantum efficiency of the alanine film with a 172 nm photon, and by using a 172-nm Xe2* Excimer lamp (Ushio). This was calculated as 0.1 (Izumi and Nakagawa, 2011; Kawaguchi et al., 2020).
The exposure units for organic exposure were assembled with those for microbe exposure, the alanine UV dosimeter, and the cosmic radiation dosimeter in the Exposure Panel. Three sets were assembled for 1, 2, and 3 years of exposure. Three Tanpopo Exposure Panels were attached to the zenith face of the ExHAM in the JEM cabin, and the ExHAM was placed on the JEM-EF of the ISS on May 26, 2015 (Kawaguchi et al., 2016; Yamagishi et al., 2018).
The 1-year space-exposed unit was retrieved on June 13, 2016, after 384 days of exposure and returned to Earth on August 27. The 2-year space-exposed unit was stored in a pressurized area of the ISS after 769 days of exposure and returned to Earth on September 17, 2017. The 3-year space-exposed unit was returned to the ISS cabin after 1126 days of exposure and returned to Earth on August 2, 2018. These periods are hereafter referred to as 1 year, 2 years, and 3 years, respectively.
2.3. Ground simulations
The stability of the target molecules in space environments was estimated by conducting ground simulation experiments. Prior to space experiments, amino acids and their precursors (Gly, Ival, Hyd, EMHyd, and CAW) were exposed to VUV, γ-rays, heavy ions, and heat (100°C) to estimate their survival in low Earth orbit for 1 year (Kobayashi et al., 2014). In these experiments, solid samples were used to compare with the space exposure experiment. We also examined the stability of the compounds (Gly, aminoacetonitrile [AAN], Hyd, and CAW) in such space environments as molecular clouds, protoplanetary disks, and interiors of asteroids. Table 1 summarizes the ground simulation experiments to show the environments in which they were simulated.
Extraterrestrial Energies which would Alter Organic Compounds and Ground Simulations Performed
We performed several ground simulation experiments to estimate recoveries of the target molecules in low Earth orbit for 1 year prior to the Tanpopo space exposure and found that such energies as cosmic rays (including SEPs) and heat (the estimated highest temperature was 100°C) would not affect the stability, but VUV light irradiation would severely have an effect on it (Kobayashi et al., 2014). VUV light irradiation was performed as follows.
The target molecules were irradiated with VUV from an Ushio standard type excimer light emission unit (λ = 172 nm). Solutions of exposure samples were added into the small pits (1 mmφ) on a SUS plate and dried under reduced pressure. The plate was then placed into a vacuum chamber and irradiated for 6.2 h at 4 × 1014 photons s−1 cm−2; the irradiation was equivalent to approximately 4 years of full irradiation around the ISS orbital.
We conducted ground simulation experiments to examine recoveries of the compounds against various energies in space environments as follows.
Heavy ion irradiation: Heavy ion irradiation was performed to study stability in ice mantles of interstellar dust in molecular clouds. Each targeted molecule was dissolved in water, sealed in a glass ampule, frozen in a liquid N2 bath, and irradiated with 290 MeV/u carbon ions obtained from HIMAC, the National Institute of Radiological Sciences, National Institutes for Quantum and Radiological Science and Technology, Japan. The total irradiation dose was 15 kGy.
Soft X-ray irradiation: Soft X-ray irradiation was performed to test the stability of molecules during the protoplanetary disk stage. An aqueous solution of each target molecule was added to the pits of the aluminum plate, dried, and irradiated with soft X- rays from the synchrotron radiation of the Beam Line 06 at the NewSUBARU facility (University of Hyogo, Japan) (Kanda et al., 2003). The amount of each molecule tested was 200 nmol. The source emitted continuous light at wavelengths ranging from X-rays to infrared, with an emission maximum of 1 nm (soft X-ray region). The estimated total energy dose (0.1 < λ < 10 nm) provided to the samples was 100 J cm−2, which was equivalent to 1.7 years' light intensity from the young Sun (4 Ga) (calculated from Ribas et al., 2005). After irradiation, the resulting materials in the pits were dissolved in a small amount of pure water, acid-hydrolyzed with 6 M HCl at 110°C for 24 h.
γ-Ray irradiation: γ-Ray irradiation was performed to examine the stability of the compounds against γ-rays that were radiated from 26Al and thawed water ice in asteroid interiors (Grimm and McSween, 1993). An aqueous solution of each target molecule was sealed in a glass ampule and irradiated with γ-rays from a 60Co source at the Tokyo Institute of Technology at ambient temperature. The total dose of the solution samples was 10 kGy.
Hydrothermal reaction: Each target molecule was dissolved in water, sealed in a glass tube, and heated in an oven at 150°C (Yamato Hi-Temp Oven DR-200) for 6–24 h. This was done to study the stability of the compounds in liquid water in asteroid interiors during aqueous alteration (Brearley, 2006).
Ultraviolet–visible light irradiation: Stability of each target molecule against near-ultraviolet (NUV) and visible light from the Sun was examined. An aqueous solution of each target molecule was dried on an aluminum plate and irradiated with a Hamamatsu Photonics 450 W Xenon arc lamp, which mainly emitted NUV light (mostly λ > 200 nm) and visible light. The estimated UV energy dose for 10 min to the target was 90 mJ cm−2 (200 < λ < 400 nm). The spectra of the xenon lamp resembled those of the Sun in the terrestrial upper atmosphere (Finlayson-Pitts and Pitts, 2000).
2.4. Analysis of products
The space-exposed samples in the pits of the aluminum exposure plates were extracted twice with 4 μL of methanol, followed by extraction with 4 μL of pure water 10 times. All extracts were combined and dried by centrifugal drying. The recovery of samples in the extraction process was calculated by triplicate analysis with the samples prepared by the same procedure.
The dried samples of the ground simulation experiments were recovered with a small amount of pure water from the aluminum plates or test tubes. The aqueous samples were concentrated to dryness by vacuum drying. Both groups of samples were acid hydrolyzed with 6 M HCl at 110°C for 24 h. Determination of amino acids was conducted by reversed-phase high-performance liquid chromatography (HPLC) and/or by cation-exchange HPLC.
Reversed-phase HPLC was conducted by using an automatic pretreatment function of an ultra-high-performance liquid chromatograph (UHPLC, Nexera X2, Shimadzu) system equipped with an autosampler (SIL-30AC). The system included an LC-30AD pump, a RF-20Axs fluorescence detector, a CBM-20A/lite system controller, and a CTO-20AC column oven. In addition, a reversed-phase column (Inertsil-ODS4, 3.0 mm i.d. × 100 mm) was used for separation. Gradient elution was conducted with (A) a solution of 15 mM potassium dihydrogenphosphate and 5 mM potassium hydrogenphosphate and (B) a mixture of 45% CH3CN, 40% CH3OH, and 15% H2O (v/v) at a flow rate of 0.8 mL min−1 at 35°C. Detailed conditions, including the precolumn derivatization method using mercaptopropionic acid and o-phthalaldehyde, are described in the work of Elmasry et al. (2021b).
Cation-exchange HPLC was conducted by using a Shimadzu Amino Acid Analysis System including two LC-10AT pumps, a RF-20Axs fluorescence detector, and a Shimpak ISC-07/S 1504 column (4.0 mm i.d. × 150 mm). Gradient elution was conducted by using (A) 0.2 M sodium citrate aqueous solution with ethanol and perchloric acid (pH 3.20) and (B) 0.6 M sodium citrate in borate buffer (pH 10.0) at a flow rate of 0.3 mL min−1 at 55°C. In the postcolumn derivatization, o-phthalaldehyde and N-acetyl-L-cysteine were used (Takano et al., 2004a).
The samples were also analyzed by gas chromatography/mass spectrometry (GC/MS) without hydrolysis using a Shimadzu GC-MS QP2010 Plus. The GC was equipped with a FFAP bonded FS-WCOT capillary column (30 m × 0.25 mm i.d., TC-FFAP, GL Science). The GC oven temperature was programmed to be 60°C for 1 min, increase from 60°C to 180°C at a rate of 30°C min−1, increase from 180°C to 200°C at a rate of 5°C min−1, and increase from 200°C to 230°C at a rate of 3°C min−1. The final temperature was then held for 30 min. Helium was used as a carrier gas with a flow rate of 1 mL min−1. The electron impact (at 70 eV) mass spectra were obtained by scanning from m/z 50 to 300 every 0.5 s.
Liquid chromatography/mass spectrometry (LC/MS) was used to identify the 13C-labeled products as follows to avoid the selection of contaminants: An Orbitrap Q Exactive high-resolution mass spectrometer with a DIONEX HPLC system (Thermo Fisher Scientific, Massachusetts, USA) was used. A heated electrospray ion source (HESI-II) was used for ionization in a positive ion detection mode at a mass resolving power of 140,000 (FWHM at m/z 200). The mass range of 60–900 was monitored for the qualitative analysis, and ions of 76.8427–77.2427 and 118.8896–119.2896 were acquired for the quantitative analyses of 13C-Gly and 13C-Ival, respectively. The automatic gain control target was set to 106, and the injection time was 50 ms. Formic acid (0.1%) was used as an eluent, and a flow injection mode without column was used.
2.5. UV spectra measurements
The absorption spectra for the vacuum ultraviolet (VUV) and near-ultraviolet (NUV) regions of the free amino acids Gly, alanine, and valine were obtained from Kamohara et al. (2008), and the spectra of Hyd and CAW were obtained in this work. The NUV spectra of the aqueous solution of the target compounds were measured with a JASCO V-550 UV-visible spectrophotometer, and a vacuum monochromator system VM-504 (Acton Research Corporation, MA, USA) was used to measure VUV absorption spectra. A Hyd thin film was prepared on a MgF2 plate by using the vacuum evaporation technique. The optical absorption cross-section σ of the Hyd film was determined on the basis of the value of σ at a peak of 202 nm in the NUV spectra of Hyd aqueous solution (19.7 mg Hyd was dissolved in 300 mL water). The CAW dry film was produced from the aqueous solution (42 mg of CAW was dissolved in 0.3 mL water) on a MgF2 plate. The thickness of the film was not uniform, and the VUV spectrum of the thinner part was measured, the area of which was sufficiently larger than the VUV light spot. Assuming that the density, ρ, of CAW is 1.6 g cm−3, and the thickness of the thinner part was approximately one-third of the average thickness, the absorption spectrum of CAW was expressed in terms of the linear absorption coefficient μ(λ). For Gly, Ala, Val, and Hyd, the values of σ(λ) in the work of Kamohara et al., (2008) were transformed into μ(λ) by the relation μ(λ) = σ(λ)N A ρ/M, where N A is Avogadro's number, ρ is the density, and M is the molecular weight.
The NUV spectra of the aqueous solution of the target compounds were measured with a JASCO V-550 UV-visible spectrophotometer.
3. Results
3.1. Environmental measurements
The highest and lowest temperatures of the exposure units measured by the mechanical space thermometer during 3 years' exposure were 29 ± 5 °C and -42 ± 5 °C, respectively (Hashimoto et al., 2016, 2019). The decomposed temperature of Gly was reported as 233°C (Windholtz et al., 1983); and those of Ival, Hyd, and EMHyd were 237°C, 260°C, and 228°C, respectively, determined by thermal gravimetry (Rigaku, TG8120). In addition, the half-life of Ival at 100°C was obtained as 3100 years by using the Arrhenius equation of the heating experiment of powdered Ival. According to the above data, thermal alteration of the target molecules could be ignored.
The mean absorbed radiation dose with the thinnest cover (0.55 g/cm2) monitored by the dosimeters was 231 ± 5 mGy/yr (SD) for the exposed units, and 82 ± 1 mGy/yr (SD) for the units in the cabin of the Kibo module (where the Cabin control was stored) during the duration of the flight (Kawaguchi et al., 2020). The radiation dose outside the ISS cabin was approximately three times greater than that inside the cabin, but these doses were much lower than the dose required to alter the present target molecules, based on the results of the ground irradiation experiments (Kobayashi et al., 2014).
The UV fluence measured with the alanine dosimeter was 124–177 MJ m2 yr−1 (110–400 nm; applied to MgF2 samples) and 114–163 MJ m2 yr−1 (170–400 nm; applied to Quartz samples), which corresponded to 44–63 equivalent solar days and 41–58 equivalent solar days, respectively (Kawaguchi et al., 2020, Yamagishi et al. 2021). It was suggested that the UV dose was the most fatal parameter affecting the stability of the target molecules (Kobayashi et al., 2014).
3.2. Recovery of target molecules
Glycine was the major compound after acid hydrolysis of the products following irradiation of Gly, Hyd, and CAW in the Ground control experiments and was still the major amino acid after space exposure. The solar UV radiation spectrum is characterized by the presence of blackbody radiation of 5800 K showing typical emission lines, for example, the spectra produced by H and He. Specifically, the Lyman alpha emission line of the hydrogen atom at 121.5 nm is very strong, with an intensity of 68 mW m−2 nm−1 (Lean, 1991), which is comparable with intensity at 250 nm.
Thus, their stabilities were evaluated by using the recoveries of Gly in the flight samples (MgF2, SiO2, Dark control, and Cabin control) after hydrolysis versus those of the Ground control. In the cases of Ival and EMHyd, Ival was the major product after acid hydrolysis, and their stabilities were evaluated by using the recoveries of Ival after acid hydrolysis. The recoveries of hydantoins (Hyd, EMHyd) were also determined by GC/MS without hydrolysis.
Figure 5 shows the recoveries of the target molecules after 1 year's space exposure. However, due to the small numbers of each sample, it was not possible to conduct sufficient analyses to provide a standard deviation, and we thus estimated the uncertainty of the values by using standard Gly deposited on the same type of sample pits as those used in space experiments. The relative standard deviation of Gly determined by the reversed-phase HPLC was 3.2% after the multiplicated experiments using standard Gly. The recovery of Gly from the sample pits of the exposure plate with methanol and water was 75.3 ± 16.1 % (relative standard deviation was 21.4%). Therefore, each value in the graph derived from the analytical procedures has an uncertainty of approximately 25%.
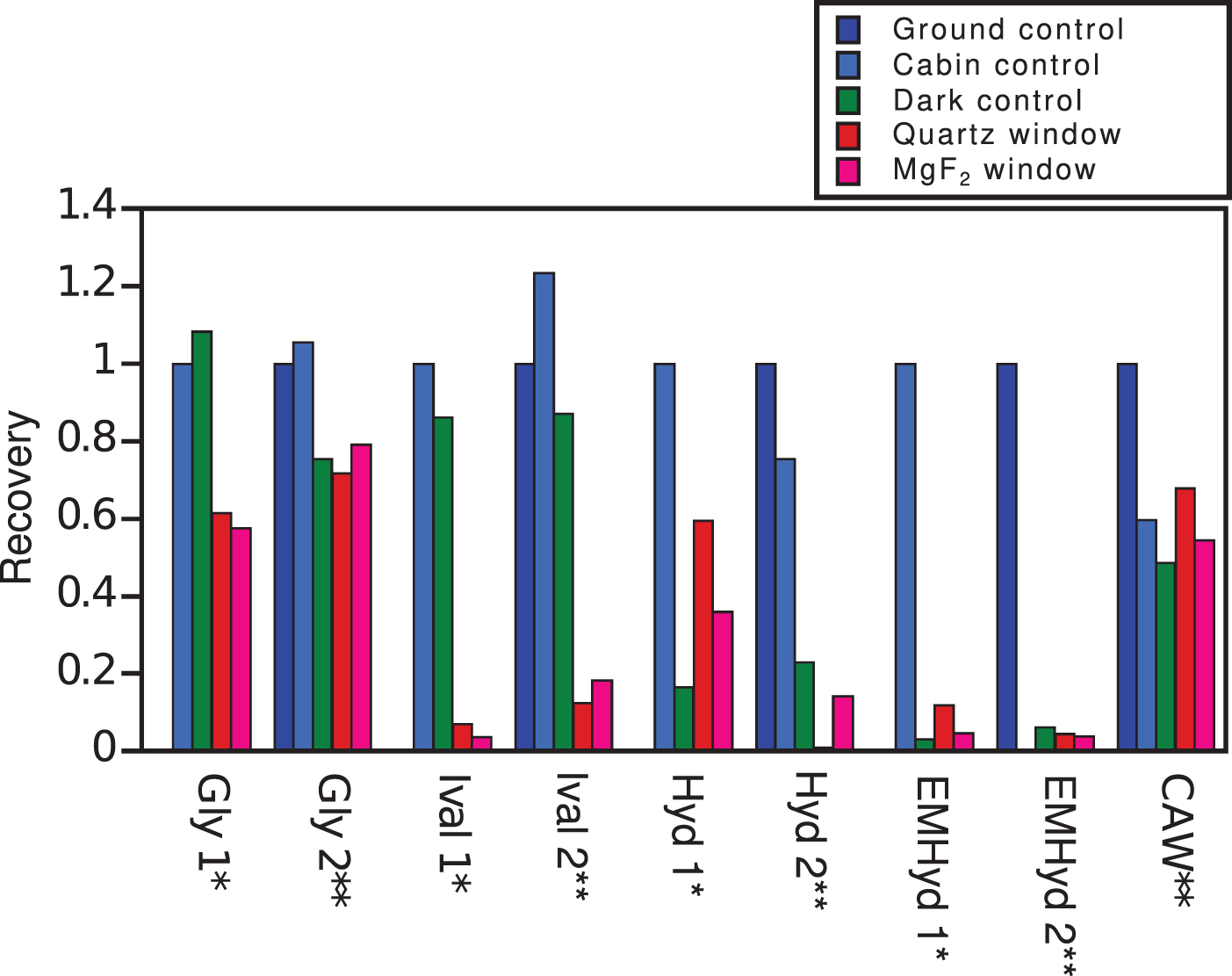
Recoveries of amino acids and their precursors after 1 year's space exposure. *Relative recovery without hydrolysis (Cabin control = 1); **Relative recovery determined as amino acids after hydrolysis (Ground control = 1). Gly 1, Ival 1 determined by LC/MS; Hyd 1, EMHyd 1 determined by GC/MS; others determined by HPLC.
We applied two kinds of analytical techniques (HPLC and GC/MS or LC/MS) to each sample. The results by GC/MS or LC/MS generally agreed with those by HPLC. The 13C glycine with little 12C glycine was detected on the LC/MS spectrum of the space-exposed sample of Gly (Fig. 6). Since Gly is the most likely to be contaminated amino acids, we concluded that there was little contamination during the experiment. Therefore, the results obtained by HPLC were deemed congruent to those obtained by LC/MS and GC/MS. Gly showed a recovery of approximately 60% (or more), even when it was exposed to solar UV through the MgF2 or quartz windows. Although Lyman alpha radiation at 121.5 nm was expected to affect the MgF2 samples and not the SiO2 samples, there were minimal differences between the MgF2 and quartz samples after irradiation. This might have been caused by the strong attenuation of HTC films with cutoff wavelengths around 160 nm.
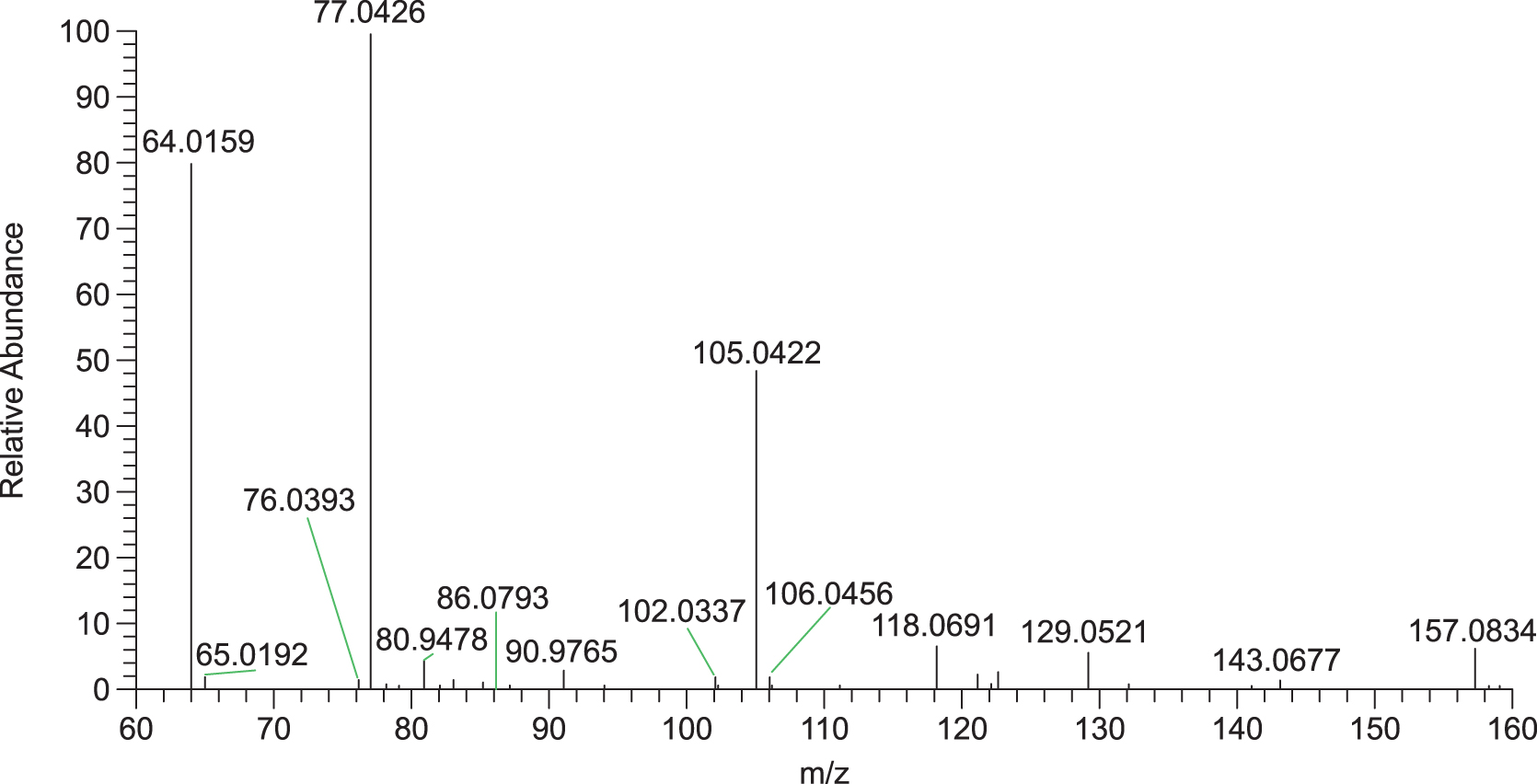
The LC/MS spectrum of the space-exposed sample of Gly. The calculated mass numbers (m/z) of 12C glycine and 13C glycine are 76.0393 and 77.0426, respectively.
However, the recoveries of Ival, Hyd, and EMHyd after exposure to solar UV were lower than Gly, and the recoveries of Hyd and EMHyd decreased even in the dark controls. In contrast, the recovery of Gly in CAW was approximately similar to that of Gly, which will be discussed later (Section 3.3).
Figure 7 shows the major by-products of acid hydrolysis, which were determined by cation-exchange HPLC. When Ival and EMHyd were irradiated with UV light from a deuterium lamp, alanine was the major by-product (Sarker et al., 2012). However, in the case of the space-exposed samples, Gly was the major by-product of Ival and EMHyd, whereas CAW yielded a wide variety of amino acids in addition to Gly (alanine was the second most abundant amino acid followed by β-alanine).
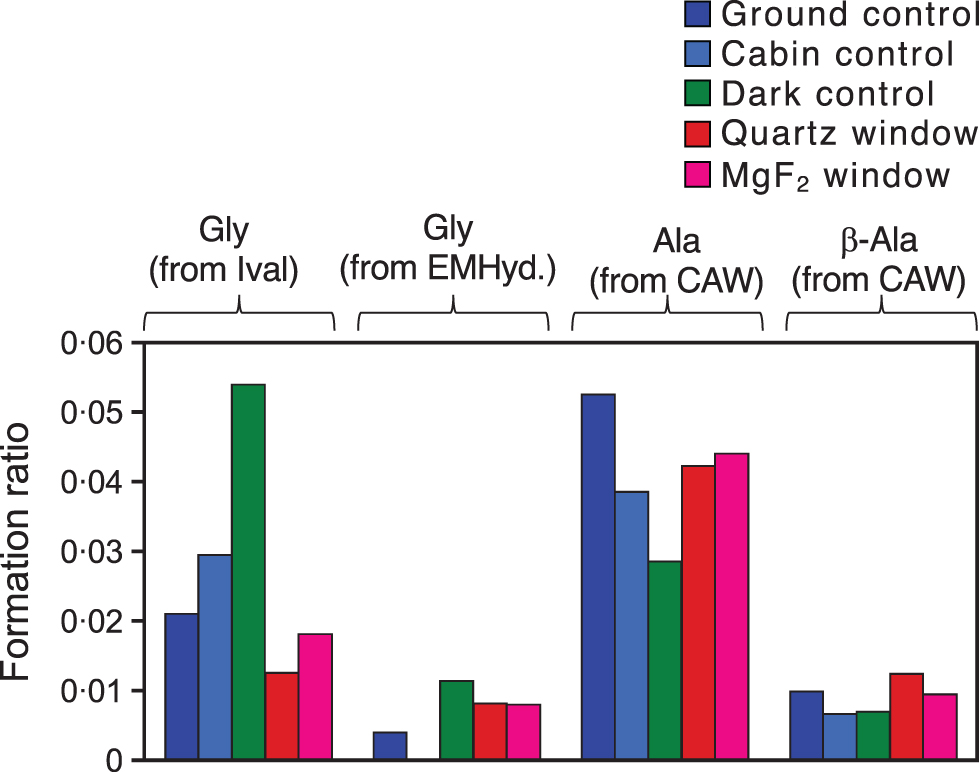
Major by-products of amino acids after 1 year's space exposure of Ival, EMHyd, and CAW (after acid hydrolysis) determined by ion-exchange HPLC. Values were normalized by the amount of the major products Ival (in the case of Ival and EMHyd) or Gly (in the case of CAW).
Figure 8 shows the recovery curves of Gly, Hyd, and CAW after 1, 2, and 3 years' exposure, determined by reversed-phase HPLC after acid hydrolysis. The general tendency of the recovery change obtained by GC/MS agreed well with that obtained by HPLC. Ival and EMHyd were mostly decomposed after 2 or 3 years' exposure, and the amount of Ival recovered after acid hydrolysis was below its detection limit. Thus, the data for Ival and EMHyd are not shown in this figure. The data expressed are the ratios of the recovery of Cabin, Dark, Quartz or MgF2 to that of Ground. All recovery at 0 years (without space exposure) was set to 1. The logarithmic plot of the recovery showed a straight declination, which means that the rate of decomposition was near to that of pseudo-first-order reactions. CAW and Hyd were below the detection limits in the MgF2 sample, and there were low recoveries of all target molecules from the MgF2 and quartz samples after 3 years, which means that they could not survive in space for a number of years without UV shields. These results agree with those of the PROCESS experiments in EXPOSE-E (Bertrand et al. 2012).
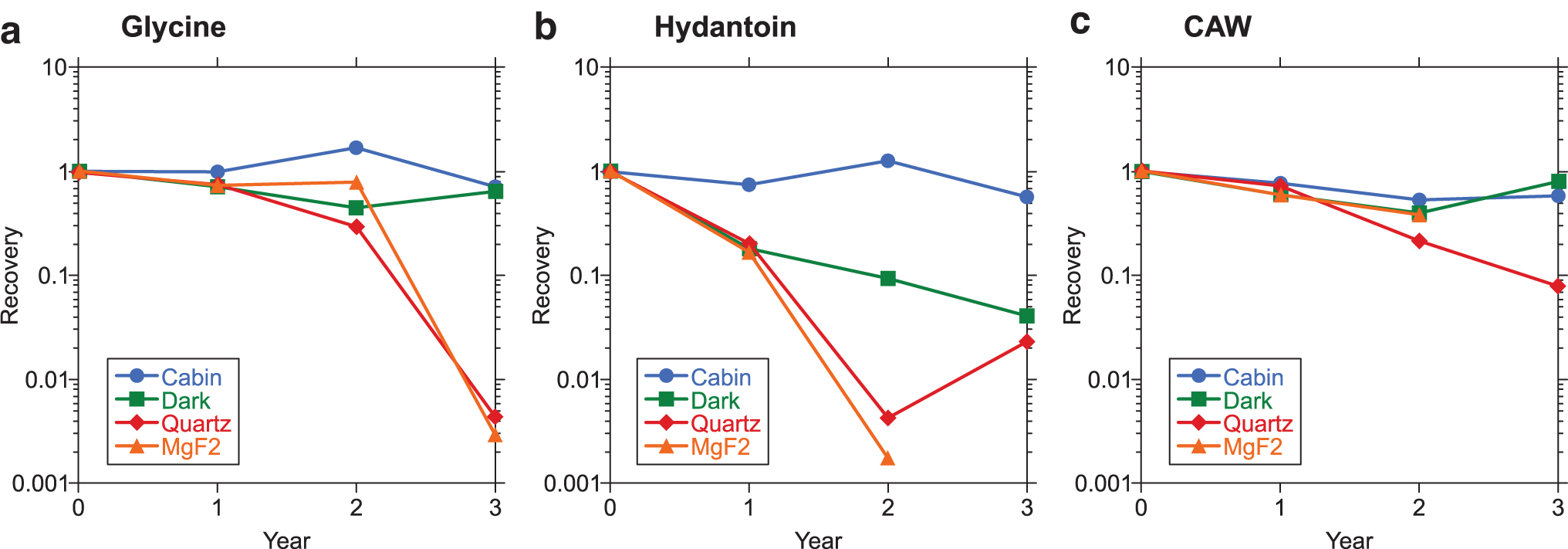
Relative recoveries of Gly, Hyd, and CAW after 1, 2, and 3 years' space exposure determined by reversed-HPLC after acid hydrolysis. All the values were normalized with those of “ground.” The value at 0 years was taken before the space exposure; thus all the plots were defined as 1. Hydantoin (MgF2 , 3 years) and CAW (MgF2 , 3 years) were below the detection limit and thus not plotted.
3.3. Survival rates of molecules and their absorption spectra
The UV/VUV spectra of free amino acids (Gly, alanine, and valine), Hyd, and CAW are shown in Fig. 9. The spectra of free amino acids and Hyd were measured by using vapor-deposited films, and the absolute absorption cross-sections were calculated. During the Tanpopo mission, all exposed samples were covered with HTC to prevent sublimation. Although HTC is transparent in the NUV region, it absorbs VUV (λ < 160 nm); therefore, both the MgF2 and quartz samples were mainly affected by solar UV photons of λ > 160 nm. This means that, in both the cases of MgF2 and quartz samples, the Lyman alpha light (121.5 nm) was strongly attenuated by HTC. A comparison between the spectra of Gly and Hyd shows that Hyd has a higher absorption of approximately 2.5 times that of Gly in the wavelength range 160–210 nm, which appears to be the most fatal wavelength range for these molecules covered with HTC. In the case of CAW, using the vapor-deposited film of CAW was not possible; therefore, it was not possible to obtain the absolute absorption coefficient. Based on the NUV/visible spectra of CAW and Gly, however, CAW absorbed a greater amount of UV/visible photons (CAW is yellow-brownish colored) than the other target molecules in the range of 160–210 nm and showed a continuous absorption spectrum up to the visible light region.
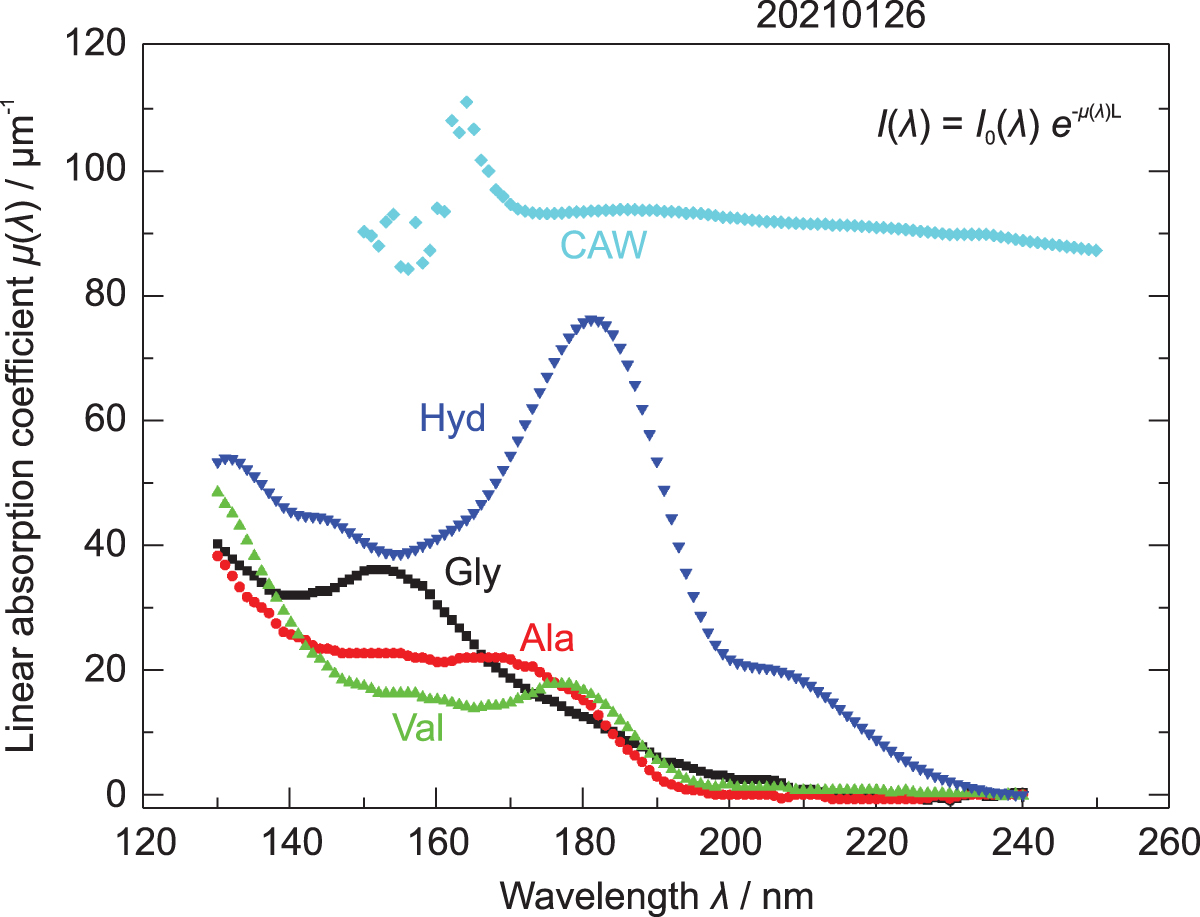
VUV/UV spectra of amino acids, Hyd, and CAW. Spectra of free amino acids were cited from Kamohara et al. (2008), and those of Hyd and CAW were measured in the present work. Details are shown in Section 2.5.
3.4. Ground simulation versus space exposure
Table 2 shows estimated recoveries in low Earth orbit for 1 year. In the case of heavy ions, γ-rays, and soft X-rays, the total dose (or energy deposition) to each sample was much greater than the value provided in the low Earth orbit over a period of a few years. We previously conducted ground experiments using these energies and concluded that they would cause minimal damage to the target molecules during a few years' exposure to the low Earth orbit (Kobayashi et al., 2014). However, it has been reported that ultraviolet light (VUV and NUV) photons have a critical effect on the target molecules in the low Earth orbit and that a greater amount of a molecule is decomposed when larger amounts of UV photons are absorbed by it (e.g., Bertrand et al., 2015). A comparison between Hyd and Gly showed that the former absorbed more UV photons. Hyd's absorbance at 172 nm was approximately 4 times greater than that of Gly; therefore, Hyd showed higher stability than Gly in the preflight ground experiment when using an excimer lamp, and this was probably due to its stable ring structure and absence of the free carboxyl group. Dissociation of the carboxyl group has been reported to be important (Bertrand et al., 2015). In the Tanpopo Mission, solar VUV/NUV photons of λ > 160 nm mainly reached the target molecules via windows and the HTC film. Hyd showed much higher absorption in the NUV region, whereas Gly showed little absorption. In addition, the Sun emits many more NUV photons than VUV photons: solar irradiance in the 200–280 nm region is approximately 60 times greater than that in the 0–200 nm region (Gueymard, 2004). These facts could explain why Gly showed much higher stability than Hyd in the space exposure experiment.
Estimated Recovery (%) of Amino Acids and Their Precursors after 1 Year's Space Exposure
Gly, glycine; Ival, isovaline; Hyd, hydantoin, EMHyd, 5-ethyl-5-methylhydantoin; CAW, complex organics synthesized from carbon monoxide, ammonia, and water (cited from Kobayashi et al. [2014] with modification).
A comparison between CAW (a mixture of complex amino acid precursors) and free Gly showed the same kind of tendency: although CAW was much more stable than Gly when the 172 nm Excimer lamp was used, CAW and Gly showed almost the same recoveries in the Tanpopo Mission. CAW showed much higher absorption in both the VUV and NUV regions than Gly. As Gly showed limited absorption in the λ > 160 nm region, photochemical decomposition in the Tanpopo mission was restricted.
Several photon sources were used in the ground experiments: a 60Co source (gamma rays), a synchrotron (soft X-rays), an excimer lamp (VUV), and a xenon lamp (mostly NUV and visible light). It is evident from Table 3 that CAW showed much greater stability against soft X-rays and VUV, comparatively higher stability than Gly against γ-rays, but slightly lower stability against NUV/visible photons than free Gly. In the present Tanpopo mission, longer-wavelength VUV and NUV/visible photons mainly irradiated the samples, and CAW and Gly thus showed comparable recoveries. These results suggest that determining stability when using a single energy source in a ground experiment cannot be generalized to stability in space. Thus, it would be of interest to observe stability against the full solar spectrum range, including soft X-rays, VUV, and NUV, and prepare for space exposure of organic molecules in the successor mission, Tanpopo 2.
Recovery (%) of Glycine and Its Precursors after Irradiation/Heating
Gly, glycine; Hyd, hydantoin, AAN: aminoacetonitrile, CAW, complex organics synthesized from carbon monoxide, ammonia, and water; r.t., room temperature.
4. Discussion
Table 3 shows the recoveries of Gly and their precursors against various energies, obtained from ground experiments simulating various space environments. The relative recoveries of Gly, Hyd, aminoacetonitrile (AAN), and CAW were determined according to the procedure used for Gly after acid hydrolysis. AAN was not used in the space exposure in the Tanpopo mission since it was quite volatile and could not be used by the present exposure system, but it was used in the ground simulation experiments since AAN is a quite popular Gly precursor formed by the Strecker synthesis (Miller, 1957).
Carbonaceous chondrites contain amino acids or their precursors, and the percentage of free to total amino acids has been reported as 38–64% for CI, CM, and CR chondrites (Glavin et al., 2011). However, the actual percentage of free amino acids in total amino acids is unknown, because amino acids were usually extracted from meteorite powders with hot water; therefore, the original amino acid precursors may have been hydrolyzed to be free amino acids during the extraction process. It has also been reported that short and mild hydrolysis may hydrolyze some amino acid precursors (Kobayashi et al., 1997). Furthermore, the structures of amino acid precursors are currently unknown. The precursors of amino acids have been detected during analyses of meteoritic amino acids, and the yields were found to increase after acid hydrolysis, which suggests that both amino acids and their precursors were present together. Experiments simulating interstellar environments (e.g., Kobayashi et al., 1995) and meteorite parent bodies (e.g., Kebukawa et al., 2017) have shown that most of the amino acids formed were as precursors; therefore, it has been suggested that most amino acids were formed as precursors in space. It was thus considered that if such precursors interacted with liquid water, they might be converted to free amino acids. The possible sites of hydrolysis of amino acid precursors would have been the interior of asteroids (meteorite parent bodies), where liquid water was formed during aqueous alteration by the heat of 26Al decay (MacPherson et al., 1995) and other events. It would thus be safe to say that both free amino acids and amino acid precursors are present in extraterrestrial bodies such as anhydrous IDPs/MMs. However, knowledge about the forms of amino acid precursors in such bodies is limited, with the exception of the presence of Hyd in CCs (Shimoyama and Ogasawara 2002).
The relevance of organic compounds formed in molecular clouds to the generation of life on Earth has been discussed through simulation experiments (e.g., Meierhenrich et al., 2005; Nuevo et al., 2014; Oba et al., 2019). If free amino acids, aminonitriles, hydantoins, and complex amino acid precursors, such as CAW, were formed in interstellar environments, they would have experienced cosmic ray irradiation while in molecular clouds. When the protoplanetary disk was formed, organics contained in silicate dust were exposed to strong radiation, including soft X-rays, from the young Sun. Our experiments show that organics (particularly free amino acids) in the dust located on the outer ridge of the disk are fragile against soft X-rays. They could, however, survive in the dust in the inner part of the disk since soft X-rays do not penetrate deeply into the disk, and complex amino acid precursors would have more chance to survive than free amino acids or simple amino acid precursors.
The aggregates of dust would evolve to planetesimals, and organics in the planetesimals located at a distance from the Sun (e.g., in the asteroid belt) would not undergo such intense heating and would thus survive. Liquid water was formed in the interior of the asteroids, and the formation and decomposition (or alteration) of amino acid-related compounds would have occurred together. Table 1 shows that aminonitriles were the least stable during such processes.
Although IDPs/MMs transported large quantities of organic carbons to primitive Earth, knowledge about the delivery of amino acid-related compounds is limited. The present results suggest that neither amino acids nor amino acid precursors could survive if they were directly exposed to solar UV for years. Thus, they would have to be protected from energetic photons by UV shields, such as minerals (Bertrand et al., 2015). Nevertheless, it is necessary to evaluate the relative stability of free amino acids and their precursors. This study suggests that it is possible that the forms of both Gly and complex amino acid precursors (such as CAW) could have been delivered protected within dust particles if most of the λ < 160 nm photons were shielded. The Tanpopo 2 mission will examine their stability against photons with higher energy in a space environment by exposing them with and without the use of optical windows.
It was recently suggested that the young Sun might have emitted a larger flux of high-energy particles (SEPs) prior to the generation of life on Earth, which could have influenced the composition of primitive Earth's atmosphere (Airapetian et al., 2016). If so, it will be necessary to evaluate the effects of SEPs on the organic compounds in IDPs/MMs. As energetic particles (SEPs and GCRs) are partly shielded by the Van Allen radiation zone, their effect is limited compared with solar photons. It would thus be of interest to examine the stability of organic compounds in relation to the synergetic effects of higher fluxes of SEPs/GCRs with solar radiation and solar UV by utilizing the space environment situated outside the Van Allen radiation zone, for example on the Lunar Orbital Platform-Gateway (Duggan and Moseman, 2018).
5. Conclusion and Future Prospects
Amino acids (Gly and Ival), their possible simple precursors (Hyd and EMHyd), and complex amino acid precursors (CAW) synthesized from a mixture of simple molecules found in interstellar media by proton irradiation were exposed to space in the Tanpopo mission on the JEM-EF of the ISS. All samples were placed in pits within aluminum plates, covered with HTC film, and exposed to the space environment under MgF2 or quartz windows. The exposures began in 2015 and finished after approximately 1, 2, or 3 years.
The decomposition of each followed pseudo-first-order reactions, and the recoveries of Gly and CAW exceeded those of Ival, Hyd, and EMHyd. The behaviors of Gly and Hyd in the space exposure experiments were different from those observed in ground VUV irradiation experiments, which suggests that space experiments are required to enable a discussion about the stability of molecules in space. CAW showed almost the same recovery as that of Gly after exposure, even though CAW showed much higher absorption in the VUV/UV region than Gly. Ground-based simulation experiments suggested that amino acid precursors (not free amino acids) were mainly formed in extraterrestrial environments (see Section 1.3) and that CCs contained a fair percentage of such amino acid precursors. A comparison between simple amino acid precursors (e.g., hydantoins) and complex amino acid precursors (e.g., CAW) in the space exposure experiment showed that the latter had higher stability.
It is necessary to consider the contribution of extraterrestrial complex amino acid precursors to the first life on Earth to discuss chemical evolution pathway scenarios on primitive Earth. Further space experiments, including the direct exposure of amino acids and their precursors to solar photons without windows, is thus required to evaluate the stability of the target molecules in space, and these are now being conducted by the Tanpopo 2 mission.
The Tanpopo 2 mission was designed based on the results of the Tanpopo mission. In the Tanpopo mission, organic compounds were covered with an HTC film, though the behavior of the film in space was unknown. Therefore, in the Tanpopo 2 Mission, we prepared a set of experiments using the same type of exposure panel (the Tanpopo-type exposure panel) and the same types of exposure plates. Samples (amino acids and their precursors) were placed in the pits of the plates and covered with HTC films with different thicknesses (including the use of no film). Another type of exposure panel that had been used in a previous space experiment by Sakon et al. (2018) was also employed and referred to as the “QCC-type exposure panel.” Organic compounds deposited to silicone plates were attached to the panel with or without optical windows, and the organics were exposed to the full spectrum of solar radiation in space. The analysis of these samples is in progress.
Footnotes
Acknowledgments
We are grateful to JAXA and NASA for their excellent support during the space exposure experiment. We would like to thank Mr. Ryohei Aoki, Mr. Taku Harada, Mr. Shusuke Misawa, Mr. Keisuke Naito, Mr. Yukinori Kawamoto (Yokohama National University), Dr. Miki Nakayama (Fukuoka Institute of Technology), and Mr. Kimihiro Ishiyama (Kobe University) for their kind assistance with sample analyses. We also extend our gratitude to members of the Tanpopo research team for contributing to our discussions. A part of this present work was conducted as a research project on heavy ions at NIRS-HIMAC. We would like to thank Editage (
Authorship Confirmation Statement
K.K., H.M., Y.K., and K.N. conducted the experiments, analyses, and interpreted the data, with the help of T.K., Y.O., T.S., T.Y., S.M., H.F., Y.O., I.Y., S.Y., and K.K. The present space exposure experiments were conducted as part of the Tanpopo mission, which was mainly conducted by E.I., H.Y., H.H., S.Y., and A.K. All the authors contributed to and approved the final manuscript.
Funding Statement
The work was financially supported by JSPS KAKENHI Grant-in-Aid for Scientific Research (B) 16H04823, 17H02991, 19K21895, and 20H02014; the Astrobiology Center of National Institutes of Natural Sciences Satellite Program (AB282002, AB292002, AB302005, AB312006, AB022002, AB032001); and the Research Project 2018 of the Environments Research Laboratory, Fukuoka Institute of Technology.
Authors' Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations Used
Associate Editor: Petra Rettberg
