Abstract
The hyperarid Atacama Desert is a unique Mars-analog environment with a large near-surface soil nitrate reservoir due to the lack of rainfall leaching for millennia. We investigated nitrogen (N) cycling and organic matter dynamics in this nitrate-rich terrestrial environment by analyzing the concentrations and isotopic compositions of nitrate, organic C, and organic N, coupled with microbial pathway-enzyme inferences, across a naturally occurring rainfall gradient. Nitrate deposits in sites with an annual precipitation of <10 mm carry atmospheric δ15N, δ18O, and Δ17O signatures, while these values are overprinted by biological cycling in sites with >15 mm annual precipitation. Metagenomic analyses suggest that the Atacama Desert harbors a unique biological nitrogen cycle driven by nitrifier denitrification, nitric oxide dioxygenase-driven alternative nitrification, and organic N loss pathways. Nitrate assimilation is the only nitrate consumption pathway available in the driest sites, although some hyperarid sites also support organisms with ammonia lyase- and nitric oxide synthase-driven organic N loss. Nitrifier denitrification is enhanced in the “transition zone” desert environments, which are generally hyperarid but see occasional large rainfall events, and shifts to nitric oxide dioxygenase-driven alternative nitrifications in wetter arid sites. Since extremophilic microorganisms tend to exploit all reachable nutrients, both N and O isotope fractionations during N transformations are reduced. These results suggest that N cycling on the more recent dry Mars might be dominated by nitrate assimilation that cycles atmospheric nitrate and exchanges water O during intermittent wetting, resulting stable isotope biosignatures could shift away from martian atmospheric nitrate endmember. Early wetter Mars could nurture putative life that metabolized nitrate with traceable paleoenvironmental isotopic markers similar to microbial denitrification and nitrification stored in deep subsurface.
1. Introduction
The central depression of the Atacama Desert in northern Chile is embedded between the Coastal Range and the Andes mountains, and has been extremely dry for more than 10 million years due to amplification of local climatic effects by the Andean uplift (Rech et al., 2006; Sun et al., 2018). The altitudes of the Coastal Range (Cordillera de la Costa) and the Andes are generally between 2000 and 6000 m, while the altitude of the central Atacama is about 900–2500 m (Sernageomin, 2003). Soil temperatures in the dry areas of the Atacama Desert varied from −4.9°C to 57.4°C between the years 1994 to 1998, and the soil relative humidity can vary from 9% to 64%.
Because of the intense ionizing radiation and ultraviolet (UV)-driven photochemical reactions, the Atacama Desert acts as a perfect model to study natural salt deposits derived from the atmosphere (Melchiorre et al., 2018; Xu et al., 2019). Large reservoirs of multiple oxyanions, including nitrate, sulfate, perchlorate, manganese oxides, and iron oxides, have developed in this region (Michalski et al., 2004; Fernandez-Remolar et al., 2013; Xu et al., 2019). The Atacama Desert is one of the nonpolar deserts that contain a huge nitrate reservoir, and the near-surface presence of nitrate has been exploited for hundreds of years.
Nitrate salts are highly soluble and generally leached from most soil profiles, but the miniscule rainfall in hyperarid regions permits its accumulation in the shallow subsurface (Walvoord et al., 2003), and nitrates become one of the important constituents of Atacama mineralogy. The common nitrate minerals include Chilean saltpeter NaNO3, darapskite Na3(SO4)(NO3)·H2O, and humberstonite Na7K3Mg2(SO4)6(NO3)2·6H2O (Ericksen, 1983). Similarly, nitrate deposits in martian sediments ranging from 70 to 1100 parts per million (ppm), as determined at the Gale Crater and in martian meteorites (Kounaves et al., 2014; Stern et al., 2015; Jaramillo et al., 2019), are assumed to derive from atmospheric photochemistry (Michalski et al., 2004; Catling et al., 2010; Smith et al., 2014). Thus, the hyperarid core of the Atacama Desert has long been considered an analog for Amazonian Mars, due to its large nitrate reservoirs, hyperarid conditions, and high UV intensities (e.g., Navarro-Gonzalez et al., 2003; Shen et al., 2019).
Due to its overwhelming abundance over other nitrogen (N) sources, nitrate likely drives biological N cycling in hyperarid soils (Shen et al., 2019). Stable N and oxygen (O) isotopes of nitrate have long been utilized as useful tools to unravel the environmental N sources and cycling processes. As microorganisms consume or produce nitrate, the isotopes of N and O are fractionated by these biological transformations in accordance with the priority of lighter isotopes (Casciotti et al., 2013). O isotopes are also affected by the O atom sources incorporated into nitrate, such as water and atmospheric O2 (Buchwald and Casciotti, 2010; Casciotti et al., 2010). Worldwide, desert soils contain intriguing correlations between N/O isotopes and indicators of water/microbial activity (Jackson et al., 2015), which motivated some of the work presented here.
With enhanced microbial N cycling in the soils, the N and O isotope ratios of an abiotic atmospheric nitrate source can be overprinted and preserved as a biosignature. N cycling is one of the key components of biogeochemical ecosystem functioning because of the fundamental roles of N in all organisms. Nitrate can be biologically cycled directly by processes such as denitrification, dissimilatory nitrate reduction to ammonium (DNRA or nitrate ammonification), and assimilatory nitrate reduction (or nitrate assimilation). Denitrification and DNRA are the processes that reduce nitrate to N2 and ammonium (NH4 +), respectively, with nitrite (NO2 −) as an intermediate product (Lam and Kuypers, 2011). Assimilatory nitrate reduction is the process of microbial assimilation of nitrate for the incorporation into organonitrogen compounds (Wada and Hattori, 1978; Tischner, 2000). These three processes all require nitrate molecules to pass through cell membranes and enter the outer or inner bacterial membranes or periplasm for reductive catalysis (Granger et al., 2004, 2008). All of these N cycling pathways are catalyzed by enzymes (Montoya, 2008; Casciotti, 2009), which impart distinctive isotopic partitioning.
These biological N cycling processes generally result in kinetic fractionation of N and O isotopes that can be expressed in the residual nitrate or organic N products. The ranges of isotopic enrichment factors of denitrification, DNRA, assimilatory nitrate reduction, and nitrification are illustrated in Fig. 1. In nitrate reduction and assimilation, the enrichment factor of O is usually very close to the factor of N (Sigman et al., 2005; Granger et al., 2010). These fractionation effects can vary significantly, for example, depending on microbial growth rate and nitrate concentrations (Kohl and Shearer, 1980; Mariotti et al., 1980, 1982).
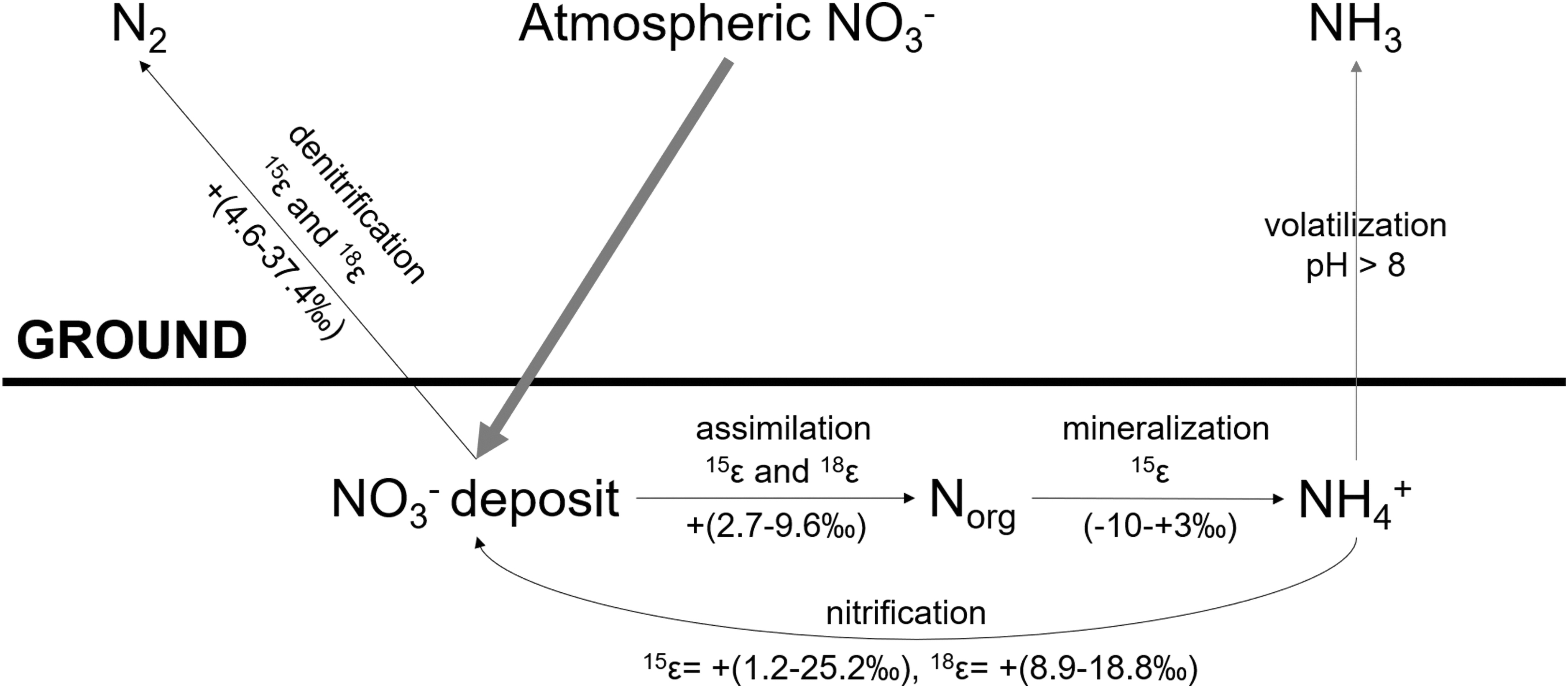
Schematic diagram of a hypothesized N cycle for a hyperarid environment, including deposition of nitrate, denitrification (includes DNRA range), assimilatory nitrate reduction, mineralization, and nitrification, illustrating the ranges of N and O isotopic enrichment factors (Mccready et al., 1983; Bottcher et al., 1990; Casciotti, 2009; Sigman et al., 2009; Vicars and Savarino, 2014; Granger and Wankel, 2016; Stueken et al., 2016; Denk et al., 2017). ɛ is defined by δreactant–δproduct. Black arrows denote biological processes; red arrows denote abiotic processes. DNRA = dissimilatory nitrate reduction to ammonium.
Since Atacama nitrate deposits are derived from the atmosphere, their δ15N, δ18O, and Δ17O values (as defined in the Materials and Methods section) should primarily reflect their sources, which are ultimately dinitrogen gas (N2) and some mixture of ozone (O3), oxygen (O2), and OH derived from H2O, respectively (Reich and Bao, 2018). Atmospheric N2 has a δ15N value of 0‰ by definition, and most (pollution-free) NOx species also feature a δ15N close to zero. Broadly speaking, the O atoms in atmospheric nitrate are a mixture (Michalski et al., 2003, 2014) from O3 (with Δ17O of transferrable O up to 39‰ and δ18O of O3 as high as 115‰—[Vicars and Savarino, 2014]) and from atmospheric water with Δ17O and variable δ18O (Fig. 1).
Previous studies have measured δ15NNO3 values in Atacama nitrates ranging from −5‰ to +8‰ (Bohlke et al., 1997; Michalski et al., 2004; Ewing et al., 2007; Melchiorre et al., 2018). Compared with more biologically active environments (Qin et al., 2012), δ15N values in Atacama nitrate are generally low, implying that the extent of microbial nitrate reduction is insufficient to overprint the atmospheric isotope signatures. Previously measured δ18ONO3 values from Atacama nitrates vary from 31‰ to 64‰ (Michalski et al., 2004; Ewing et al., 2007), similar to other sites that preserve large atmospheric nitrate deposits (Michalski et al., 2005; Qin et al., 2012). The Δ17ONO3 of Atacama nitrate can be as high as 9‰ to 23‰ (Michalski et al., 2004; Ewing et al., 2007). Since biological processes follow mass-dependent fractionation, the biological signal of Δ17ONO3 is approximately zero. Thus, these high Δ17ONO3 values demonstrate the atmospheric origin of nitrate deposits in the Atacama Desert.
Cross-plots of δ15NNO3 versus δ18ONO3 from the Atacama and other more humid deserts suggest a distinct abiotic atmospheric endmember nitrate composition that takes on differential isotopic signatures after biological processing (Jackson et al., 2015). If better understood, N/O isotope systematics in nitrate could function as a geochemical biosignature that could be explored robotically on Mars or on returned samples. During microbial nitrate consumptions, fractionations in δ18ONO3 largely reflect the δ18O values of O in ambient water absorbed by microorganisms (Casciotti 2009). However, the relevant N and O isotope fractionation effects have largely been measured in laboratory enrichment cultures and in water-rich or aqueous environments, which may not express in the extremely dry condition as the Atacama Desert, or indeed, Mars itself.
Here we examine the stable isotope variations in nitrates (δ15N, δ18O, and Δ17O) along an aridity gradient in the Atacama Desert, to determine when and how biological N cycling acts to modify atmospheric N and O isotope signals in a dry Mars analog environment. We combine these analyses with δ15N and carbon isotope (δ13C) analyses, soil metagenome extraction, and microbial pathway-enzyme inferences in associated organic matter to directly match the resulting N isotope biosignatures to the microbial processes they represent. This study contributes to quantifying potential isotopic fractionation effects and resulting biosignatures of N transformation processes in a hyperarid Mars-analogous landscape, with a focus on identifying geochemical biosignatures measurable by robotic exploration.
2. Materials and Methods
2.1. Geochemical characterization and statistical analyses
The detailed descriptions of study sites, sampling, and geochemical analytical methods are reported in the work of Shen et al. (2019). Briefly, samples from ∼15 cm depth were taken in triplicate from seven sites at roughly 1143 ± 392 m elevation in the central depression of the Atacama Desert, along a latitudinal gradient that reflects increasing annual rainfall (Fig. 2). From the driest north to the wettest south, these sites were María Elena South (MES), Point of No Return (PONR), Yungay, Transition Zone (TZ) TZ-0, TZ-4, TZ-5, and TZ-6. Based on hierarchical cluster analysis of measured geophysical and geochemical values, MES, PONR, Yungay, and TZ-0 were designated as hyperarid sites; TZ-4 was designated as a transitional site; TZ-5 and TZ-6 were designated as arid sites (Shen et al., 2019). The annual precipitation of each sampling site was calculated as the mean value of multiple nearest rain gauges between 1951 and 2017 (the sampling year) from Explorador Climático, Center for Climate and Resilience Research (Shen et al., 2019).
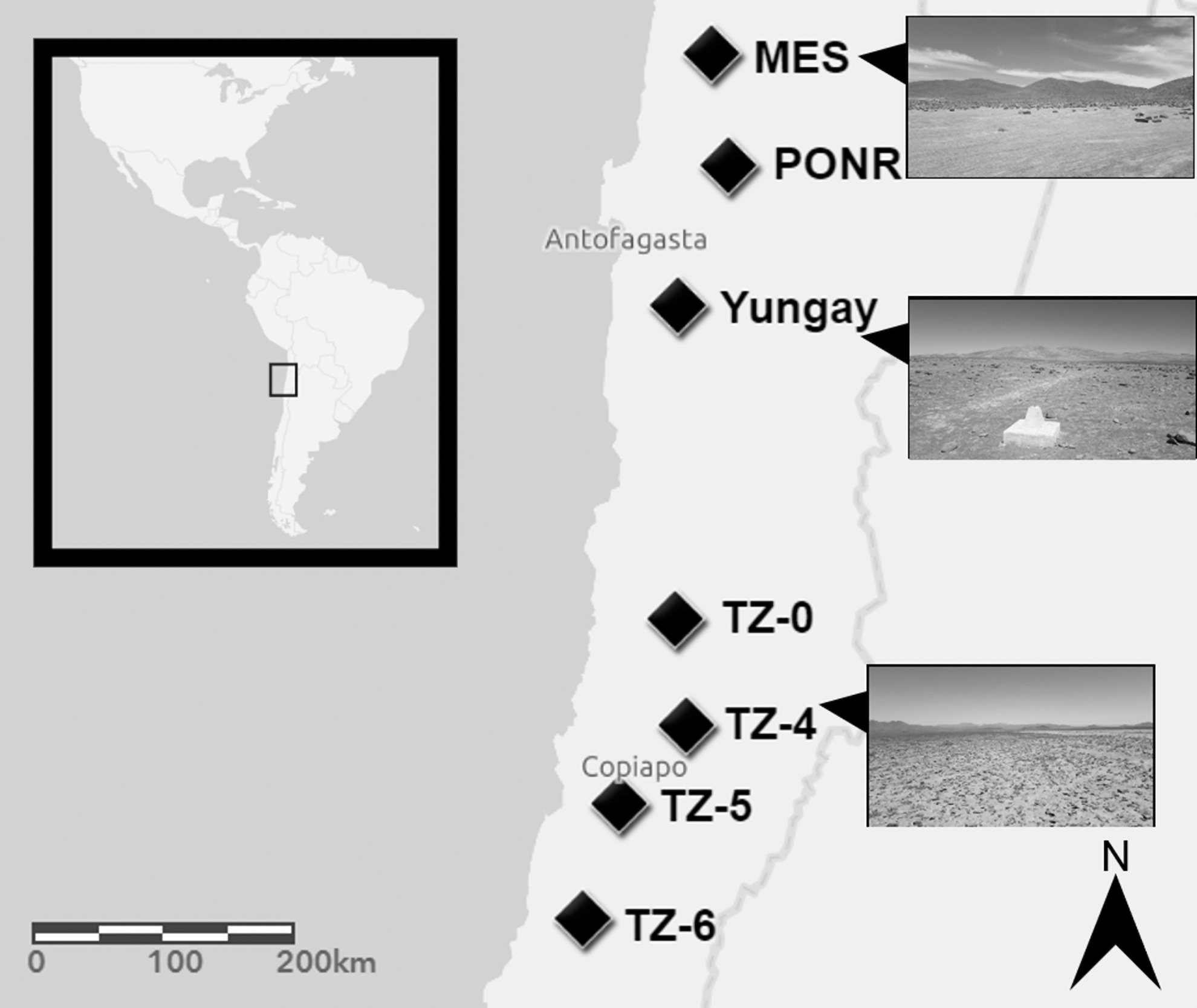
Sampling sites in the Atacama Desert, Northern Chile.
Concentrations of nitrate, NO2 −, and ammonium were measured in triplicate by ion chromatography (IC) at the University of St Andrews and using standard methods detailed in the work of Shen et al. (2019). Soils were pulverized and decarbonated by using dilute (10% HCl) for total organic C (TOC), total N (TN), and C and N isotope ratios, and they were analyzed by elemental analysis/isotope ratio mass spectrometry at the University of St Andrews (Stueken et al., 2017; Shen et al., 2019). Since the concentrations of TN were significantly correlated with those of TOC, the ammonium concentrations were minimal, and TN did not exhibit correlation with the clay-bound potassium (determined by X-ray fluorescence), the TN contents were referred to as the total organic N (TON), and N isotope values were referred to as δ15Norg (as reported in Shen et al., 2019). Carbon and nitrogen isotopes were calibrated with USGS-40, USGS-41, USGS-62, and SGR-1b. USGS-62 (with δ13C of −14.79‰ and δ15N of 20.17‰) was measured as samples for quality control, which returned experimental δ13C values of −14.89 ± 0.19‰ and δ15N values of 19.83 ± 0.36‰.
We additionally performed normality test and bivariate correlation analyses on geochemical data using IBM SPSS Statistics 26 (IBM Corp, 2019). If both variables were normally distributed, Pearson's r correlation was used. Otherwise, Spearman's ρ correlation was used.
2.2. Bacterial denitrifier and thermal decomposition methods
Soil NO3 − (plus trivial NO2 −) used for isotope analyses were re-extracted and quantified by using IC, and they were kept at −20°C. Stable isotopes (δ18O, Δ17O, and δ15N, as defined below) of the extracted NO3 − were measured by the coupled bacterial denitrifier and thermal decomposition method described in the work of Kaiser et al. (2007) at the Science Analytical Facilities of the University of East Anglia. In brief, Pseudomonas chlororaphis spp. aureofaciens, a strain of denitrifying bacteria deficient in nitrous oxide (N2O) reductase, was used to convert 50 nmol NO3 − into N2O (Sigman et al., 2001; Casciotti et al., 2002) that was then pyrolyzed to N2 and O2 by decomposition in a gold furnace at 800°C (Kaiser et al., 2007). N2 and O2 were separated and analyzed on a Geo 20-20 device by gas chromatography-isotope ratio mass spectrometry (Comer-Warner et al., 2020). International nitrate reference standards USGS-34, USGS-35, and IAEA-NO3 were used to calibrate the triple nitrate isotope measurements. The precisions for the δ18O, Δ17O, and δ15N analyses were typically up to ±2.1‰, ± 0.5‰, and ±0.2‰, respectively (1σ based on replicate standard measurements).
We herein define the δ and Δ isotope ratio notations as follows:
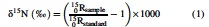
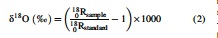
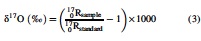
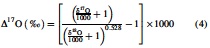
where xxRsample is the 15N/14N, 18O/16O, or 17O/16O ratio in a given sample, and xxRstandard is the corresponding ratio of international standards, that is, atmospheric N2 for N stable isotopes and Vienna Standard Mean Ocean Water for O stable isotopes. We further define the isotopic enrichment factors, 15ɛ and 18ɛ, as follows:
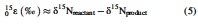
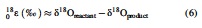
2.3. Enzyme and functional pathway inferences
The soil metagenome extraction, 16S rRNA gene sequencing, and enzyme and functional pathway inferences were described in detail in the works of Shen et al. (2020) and Shen et al. (2021). Briefly, only three sites of MES, two sites of TZ-0, three sites of TZ-4, three sites of TZ-5, and three sites of TZ-6 obtained sufficient yields for Illumina sequencing. 16S rRNA amplicons for Illumina MiSeq System were sequenced following the protocol 16S Metagenomic Sequencing Library Preparation (Illumina® 2013). Potential functions of these sequences were inferred by PICRUSt2 (Douglas et al., 2019) referencing the Enzyme Commission and MetaCyc pathway databases (Shen et al., 2020, 2021). Here, we focus solely on enzymes and pathways that are relevant to N cycling, and these were converted to heatmaps with OriginPro 2019 (OriginLab Corp, 2019).
3. Results
3.1. Nitrate, nitrite, and ammonium
The concentrations of nitrate and TON were previously reported in the work of Shen et al. (2019) (Table 1 and Fig. 3a, b). In summary, nitrate and TON concentrations ranged 1.4–6800 ppm and 18–180 ppm, respectively. Hyperarid sites PONR and Yungay had the highest concentrations of NO2 −, between 0.77 and 0.87 ppm (Table 1 and Fig. 3c). High NO2 − was associated with the rainfall event in 2017. Other sites all had low NO2 − concentrations from 0.001 to 0.11 ppm. Ammonium remained at low levels from 0.02 to 0.3 ppm and did not show a trend with either precipitation data (Fig. 3c).
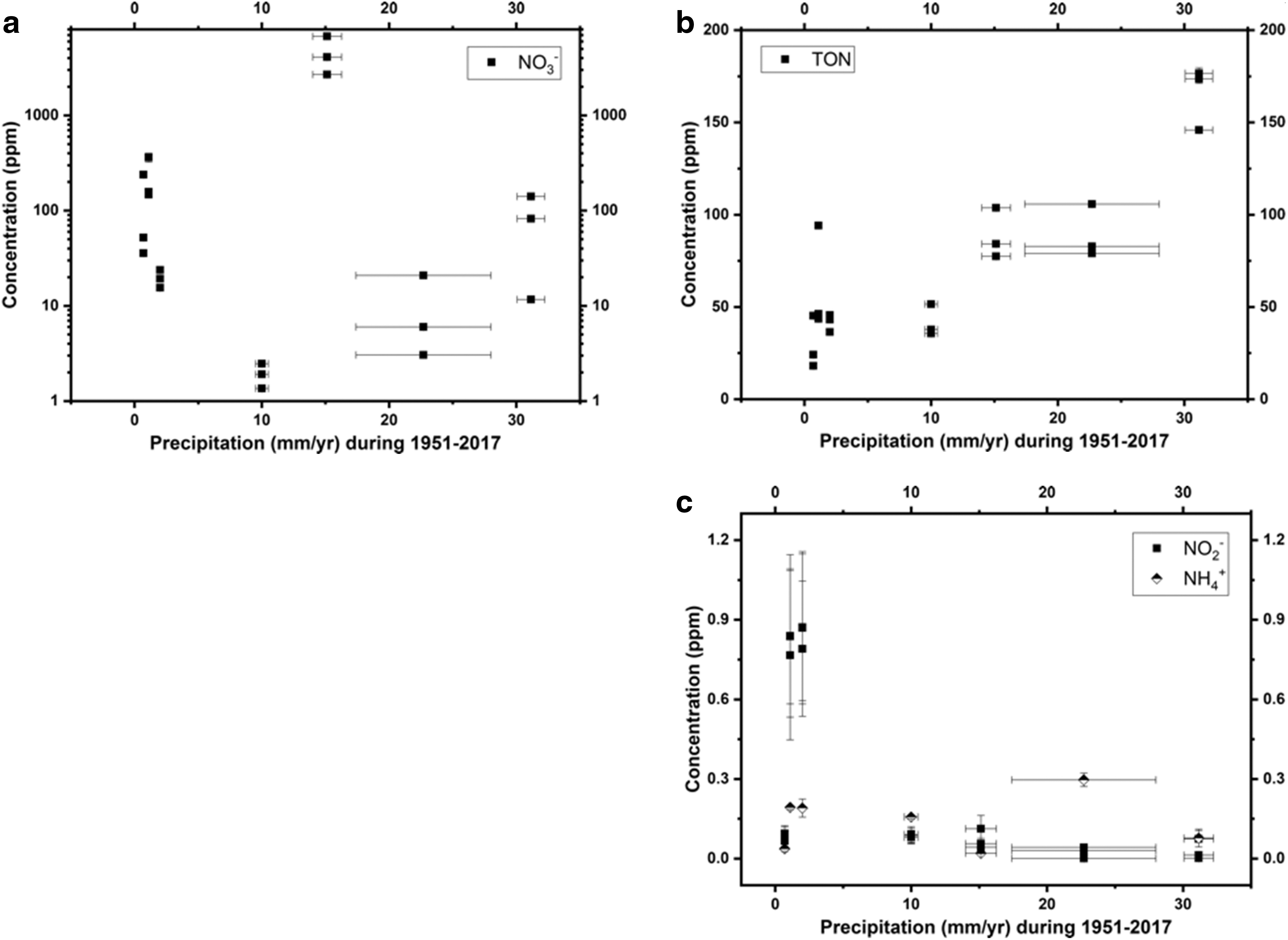
Concentrations of
Concentrations of Nitrate (from Shen et al., 2019), Nitrite, and Ammonium, Molar Ratios of Organic C to N, and Stable Isotope Ratios of Organic C and N (Mean ± Standard Errors on Triplicates When Applicable)
M1–M3, three MES pits; P1-P3, three PONR pits; Y1–Y3, three Yungay pits; T01–T03, three TZ-0 pits; T41–T43, three TZ-4 pits; T51–T53, three TZ-5 pits; T61–T63, three TZ-6 pits.
— = not measured; MES = María Elena South; NH4 + = ammonium; NO2 − = nitrite; PONR = Point of No Return; TZ = Transition Zone.
3.2. C/N molar ratios, δ13Corg, and δ15Norg
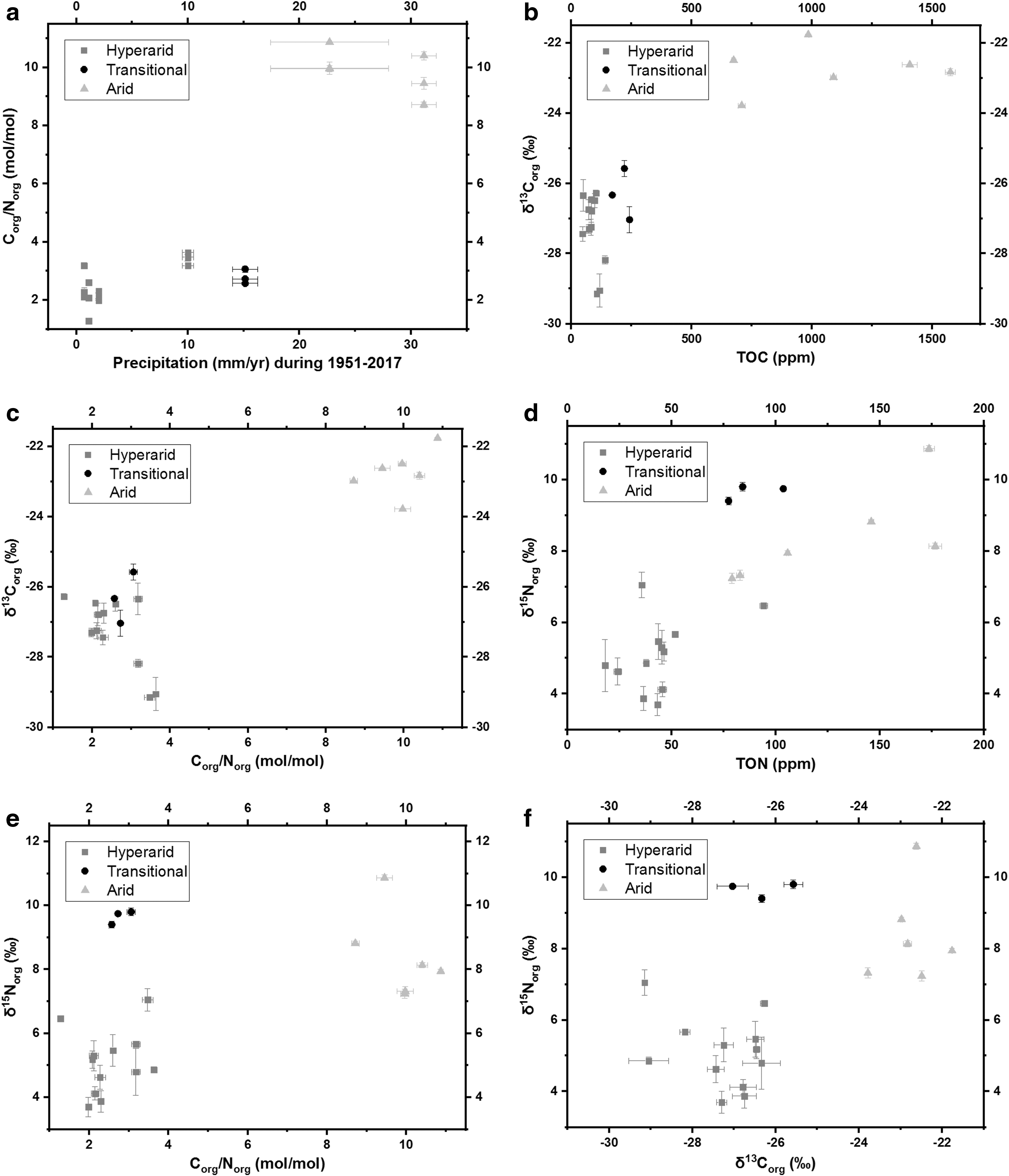
The organic C/N molar ratios in the hyperarid (MES, PONR, Yungay, and TZ-0) and transitional (TZ-4) sites were between 1 and 4, while C/N ratios in the arid sites (TZ-5 and TZ-6) were between 8 and 11 (Table 1 and Fig. 4a). δ13Corg ranged from −30‰ in the hyperarid sites to −21‰ in the arid sites. δ15Norg ranged from +3‰ in the hyperarid sites to +11‰ in the arid sites. Both C and N isotope ratios were positively correlated with the concentrations of organic matter (Fig. 4b, d). Different C/N ratios were grouped with δ13Corg, and high C/N ratios were associated with C sources of high δ13Corg (Fig. 4c). δ15Norg values did not clearly associate with C/N ratios or δ13Corg values but were more closely related to the aridity of sites (Fig. 4e, f).
3.3. Nitrate stable isotopes
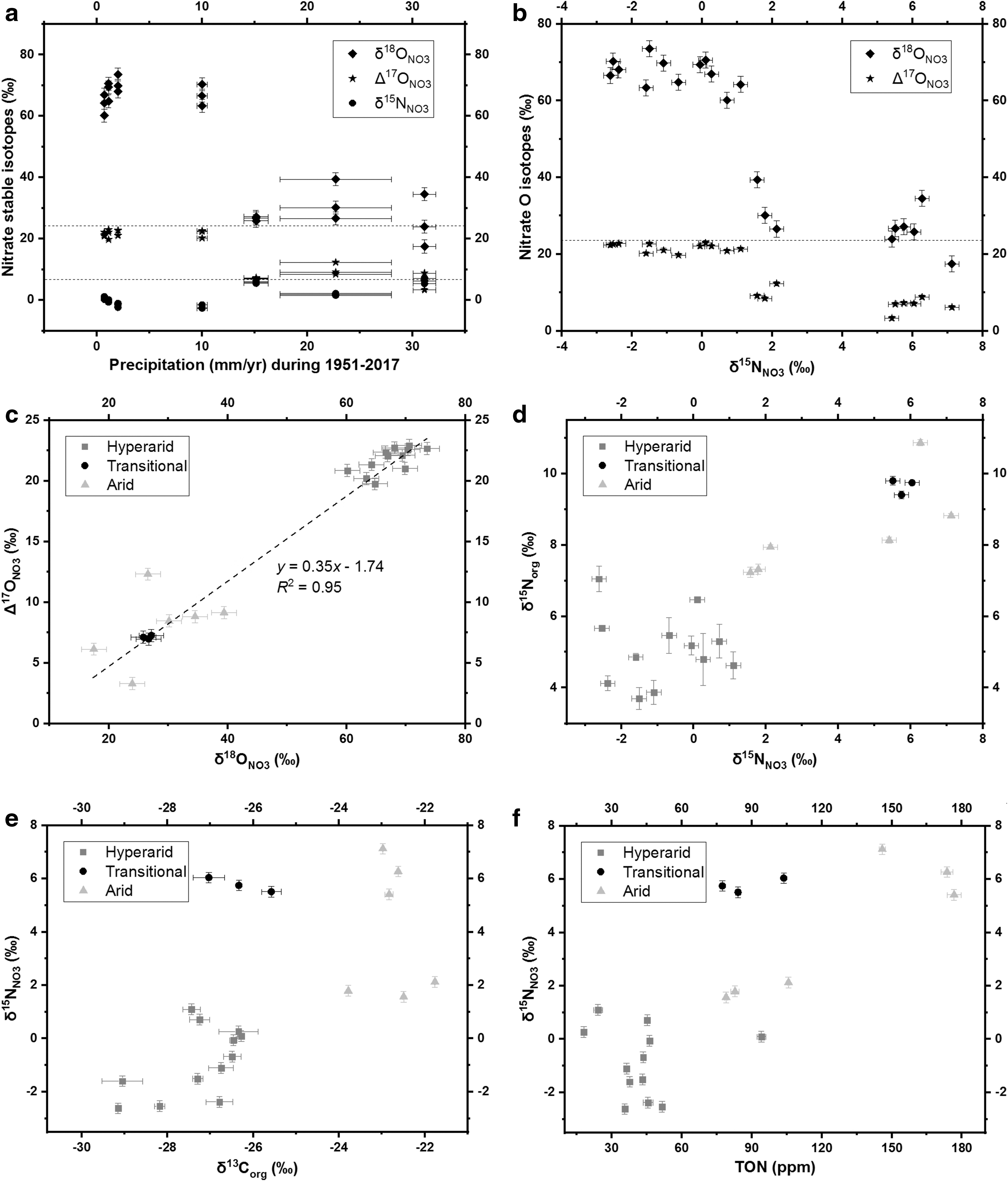
NO2 − constituted from 0.001% to 6% of the Atacama nitrate deposit and contributed minimally to the isotope values (Table 2). In the hyperarid sites, δ18ONO3, Δ17ONO3, and δ15NNO3 ranged from +60‰ to +74‰, from +19‰ to +23‰, and from −3‰ to +2‰, respectively (Fig. 5a). In the transitional and arid sites, δ18ONO3 and Δ17ONO3 ranged from +17‰ to +40‰ and from +3‰ to +13‰, respectively. However, the δ15NNO3 values of the transitional and arid sites were variable within these sites: the δ15NNO3 of TZ-4 and TZ-6 ranged from +5‰ to +8‰, but the δ15NNO3 of TZ-5 ranged from +1‰ to +3‰ (Table 2 and Fig. 5a). As δ15NNO3 increased, δ18ONO3 and Δ17ONO3 dropped from the hyperarid to the transitional and arid sites (Fig. 5b). δ18ONO3 and Δ17ONO3 were positively associated with each other (Fig. 5c). According to correlation analyses, δ15NNO3 was significantly positively correlated with δ15Norg (ρ = 0.75***, p < 0.001), δ13Corg (ρ = 0.67***, p < 0.001), TON (ρ = 0.67***, p < 0.001), and TOC (ρ = 0.57**, p = 0.003) (Fig. 5d–f).
Nitrate Stable Isotope Values, the Percentage of Total Soil Nitrate That Is Atmospheric (Assuming 100% Atmospheric Nitrate = 22.9‰ Δ17ONO3), and the Difference Between δ15Norg and δ15NNO3 in Atacama Soils Examined in This Study
3.4. N cycling enzymes and pathways
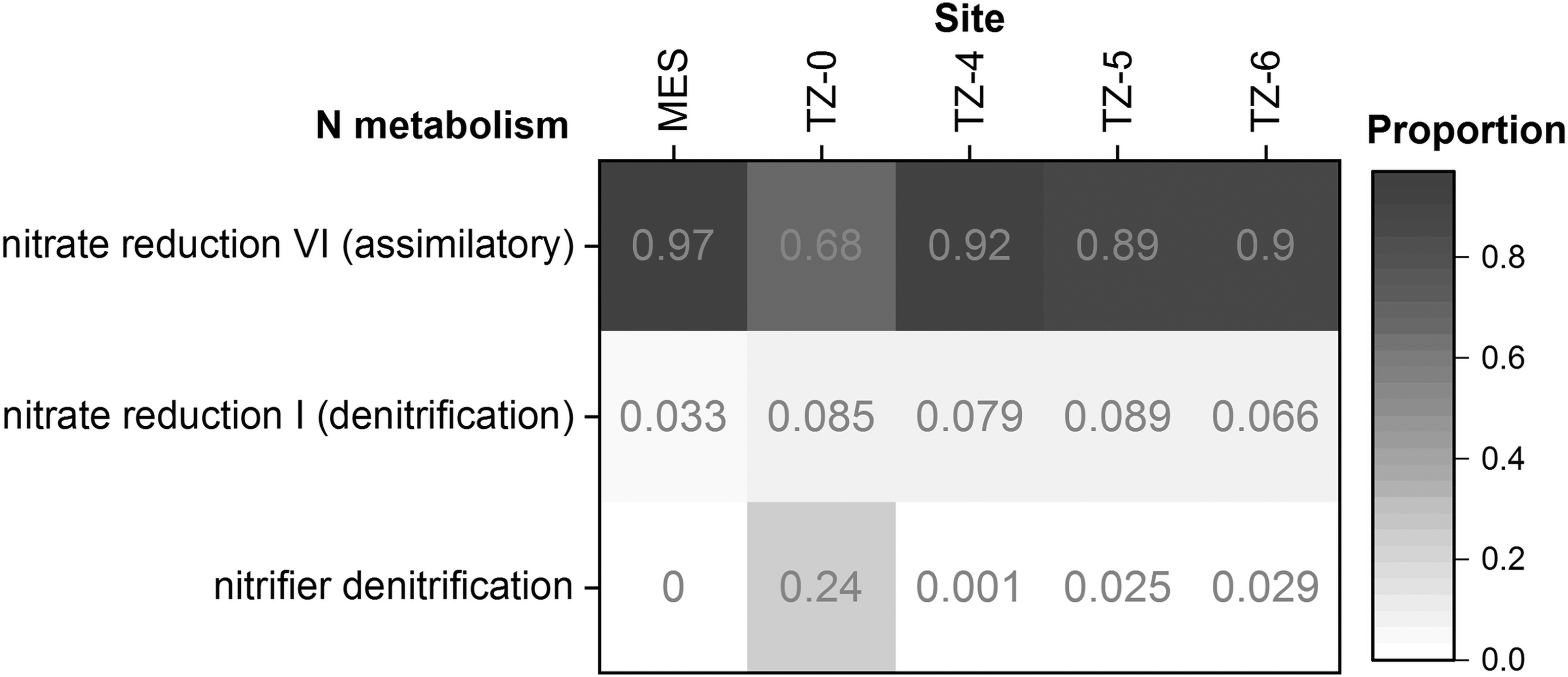
Pathway inference results of 16S rRNA gene sequencing demonstrate that the only full pathways of N cycling-related metabolisms were assimilatory nitrate reduction (nitrate assimilation), denitrification, and nitrifier denitrification (Fig. 6), and that related enzymes included nitrogenase, NO2 − reductases, nitrate reductases, N2O reductase, nitric oxide reductase, dioxygenase, synthase, hydroxylamine reductase and dehydrogenase, and ammonia monooxygenase, lyases, and ligases (Fig. 7 and Table 3).
Proportions of predicted N-processing pathways in microbial N cycling.
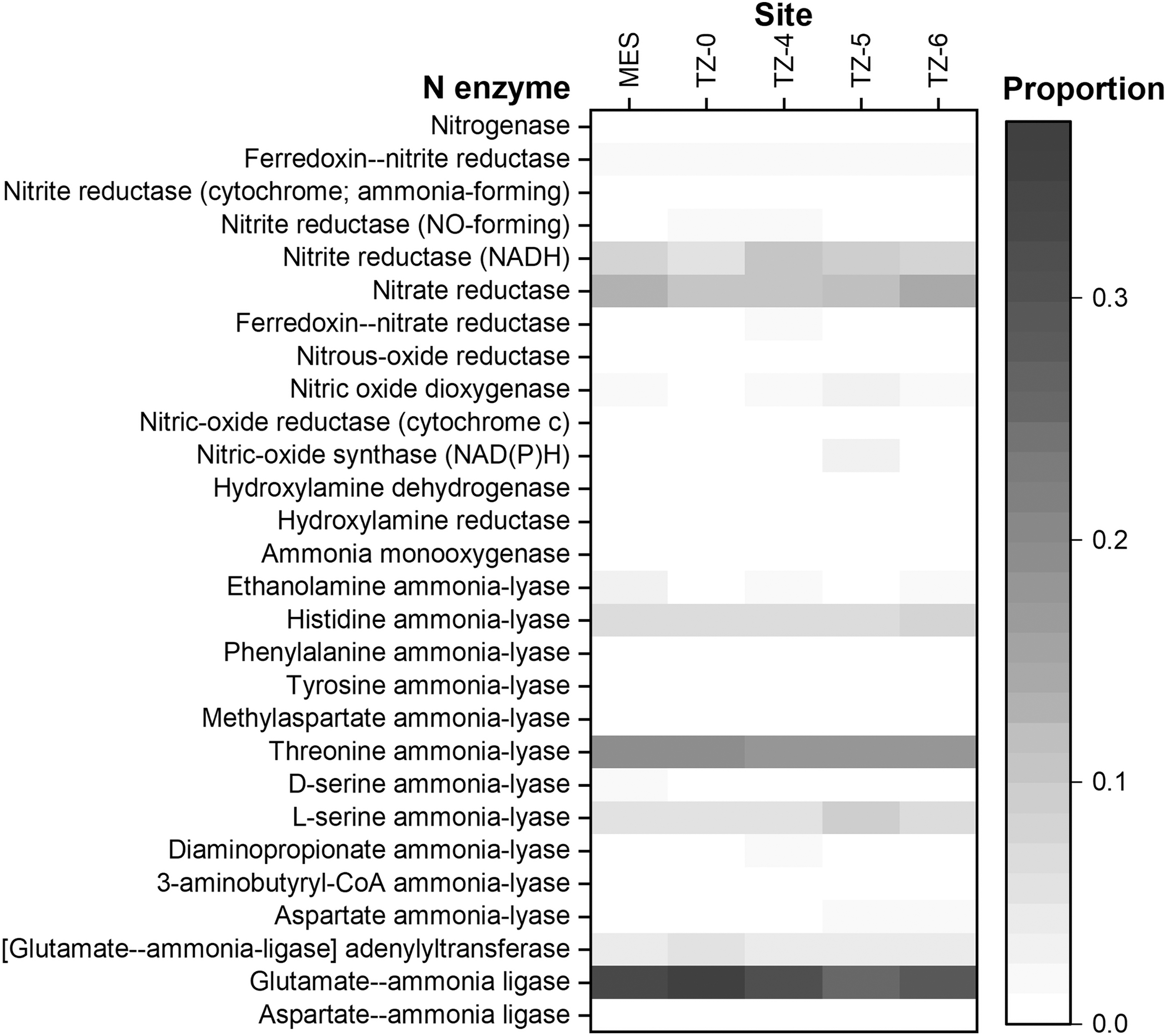
Proportions of predicted N-processing enzymes in microbial N cycling.
Percentages of Each Class of N-Processing Enzyme
n.a. = not applicable.
Assimilatory nitrate reduction was consistently the most abundant N metabolism in all sites (Fig. 6). Denitrification also universally existed in all sites but at slightly lower abundances in the hyperarid site MES. Nitrifier denitrification was significantly more dominant in TZ-0 than other sites, and TZ-0 was the only site that had more abundant nitrifier denitrification pathways than the normal denitrification pathways.
The universally abundant N enzymes were glutamate-ammonia ligase, adenylyltransferase, threonine ammonia-lyase, histidine ammonia-lyase, L-serine ammonia-lyase, nitrate reductase, and NO2 − reductase (Fig. 7). Ammonia monooxygenase and hydroxylamine dehydrogenase were not predicted in MES. The transitional and arid sites had more dominant nitric oxide synthase and dioxygenase than the hyperarid sites (Table 3).
4. Discussion
4.1. Atmospheric and biological signatures in Atacama nitrate deposits
Nitrate in the hyperarid sites had high δ18O and Δ17O and near-0‰ δ15N values (Table 2 and Fig. 5a), suggesting a predominantly atmospheric source (Ewing et al., 2007; Melchiorre and Talyn, 2014; Reich and Bao, 2018). NO2 − composed <6% of total nitrate deposits (Table 2), and transformations between NO2 − and nitrate did not seem to produce a significant fractionation effect, since δ15NNO3 values were not correlated with NO2 − concentrations (Supplementary Fig. S1b). In transitional and arid sites, biological activity mostly overprinted the atmospheric stable isotope signals in the nitrate (Fig. 5b), although an atmospherically derived correlation between δ18ONO3 and Δ17ONO3 still exists (Fig. 5c).
Based on mass balance calculations assuming the Δ17ONO3 value of 22.9‰ for atmospheric nitrate, 95 ± 4% of the total nitrate deposit in hyperarid sites was atmospherically derived, while 34 ± 10% of the nitrate was atmospheric in the transitional and arid sites (Table 2). This suggests that up to 60% of nitrate in the transitional and arid sites was derived from biological exchange (i.e., from microbial nitrification, denitrification, and assimilation) between intermediate nitrogen oxide species and surrounding water molecules (Casciotti, 2009). Microbial activity tends to decrease the values of δ18ONO3, because the local water sources for Atacama microbial communities are mostly 18O-depleted snowmelt and rainwater (Fritz et al., 1981; Schotterer et al., 1996; Aravena et al., 1999; Boschetti et al., 2019; Jordan et al., 2019).
Denitrification and nitrate assimilation likely contributed to the modification of residual nitrate deposits when the intermediate product NO2 − was kinetically converted back to nitrate (Casciotti, 2009; Reich and Bao, 2018). Since denitrification transforms lighter N isotopes from solid to gaseous phase, the δ15N of leftover nitrate pool remains higher. Besides, no nitrification pathway was inferred from metagenomic analyses of these Atacama microbial communities (Fig. 6), but alternative nitrate production pathways could additionally supplement the nitrate pool. According to the predicted enzymes (Fig. 7), NO2 − oxidoreductase that is involved in classical nitrification did not exist, but nitric oxide dioxygenase that oxidizes nitric oxide to nitrate was present (Igamberdiev et al., 2006). Therefore, two alternative nitrification pathways that produce nitric oxide could contribute to the nitrate: (1) nitric oxide synthase could generate nitric oxide by using organic N as substrates (Gusarov et al., 2008); (2) ammonia monooxygenase or hydroxylamine reductase followed by hydroxylamine dehydrogenase could generate nitric oxide from ammonia (Hooper and Terry, 1979) dissociated from organic N by ammonia lyases (Scarlett and Turner, 1976; Kyndt et al., 2002) (Fig. 5f).
Microbial nitrate production follows the mass-dependent fractionation effect on O isotopes, and thus the lower δ18ONO3 and Δ17ONO3 values in transitional and arid sites reflect the biological incorporation of local water sources that are usually δ18O-depleted (Reich and Bao, 2018; Shen et al., 2020). In addition, nitrification regularly overwrites the isotopic signal of increased δ18O by denitrification (Granger and Wankel, 2016) (Fig. 1). However, in dry soils, denitrification might not always increase nitrate δ18O, since at slow denitrification rates, NO2 − can react with ambient water to inversely produce nitrate (Casciotti, 2009; Reich and Bao, 2018). After a prolonged period, O isotopes in nitrate will achieve an equilibrium state with O in the ambient water. The average δ18ONO3 and Δ17ONO3 values measured in the transitional and arid sites of this study were 28.0 ± 5.9‰ and 7.7 ± 2.3‰, respectively, and likely reflect this equilibrium (Table 2).
The δ15N values of nitrate, on the contrary, should generally increase during biological cycling since the lighter N isotope is preferred in biological reactions (Fig. 1), particularly in the absence of other more exchangeable nitrogenous compounds (Reich and Bao 2018). Accordingly, the increase of δ15NNO3 values in the transitional and arid sites (Table 2 and Fig. 5a) reflects biological nitrate consumption and N loss, including denitrification and nitrate assimilation (Schatz et al., 2011). δ15NNO3 values near 0‰ (−0.9 ± 1.2‰) in hyperarid sites indicated that the microbial activity of nitrate consumption was minimal or much reduced (Bohlke et al., 1997; Michalski et al., 2004; Ewing et al., 2007; Melchiorre et al., 2018). Conversely, the δ15NNO3 values of TZ-5 were 3.3–5.6‰ lower than TZ-4 and TZ-6, which can probably be attributed to lower microbial denitrification activity along with higher alternative nitrification activity, as evidenced by the highest proportions of nitric oxide synthase and dioxygenase in TZ-5 (Fig. 7 and Table 3).
In extracellular environments, ammonium salts are mostly volatilized under the alkaline conditions of the Atacama Desert, and only a small background amount of ammonium is left in the shallow subsurface soils (Fig. 3c) (Ehleringer et al., 1992; Ewing et al., 2007; Orlando et al., 2010; Shen et al., 2019). On the contrary, the external NO2 − content increased in the two hyperarid sites (PONR and Yungay) that received 20–30 mm precipitation within 2 days about half a year before sampling (Azua-Bustos et al., 2018; Shen et al., 2019). These NO2 − salts should be primarily generated from the nitrate reductase catalysis, because the NO2 − generated by hydroxylamine dehydrogenase was usually further processed through nitrifier denitrification intracellularly and both ammonia monooxygenase and hydroxylamine reductase were absent in the more northern hyperarid sites (Table 3).
To summarize (Fig. 8), the atmospheric nitrate deposits without biological disturbance have a low/near-zero δ15NNO3 and high δ18ONO3 (60–75‰). As microbial activity increases in wetter sites, the δ15NNO3 values increase due to intensified denitrification, and the δ18ONO3 values shift toward the slightly negative desert rain δ18OH2O values (Aravena et al., 1999; Reich and Bao, 2018; Jordan et al., 2019) due to more frequent nitrate/water O exchange triggered by alternative nitrification, assimilation, and denitrification. Melchiorre et al. (2018) documented similar trends in nitrate stable isotopes with depth in Atacama soils. These authors showed that, on a 10–200-cm-depth scale, deeper nitrate deposits generally demonstrated abiotic isotope signatures, while shallower deposits demonstrated increasing biological activity. Thus, more abiotic endmembers might be observed deeper into Caliche or Coba layers even in the transitional and arid sites of this study, possibly due to limited moisture penetration beyond a certain depth or texture in Atacama nitrate deposits (Melchiorre et al., 2018). As Δ17ONO3 strongly correlates with δ18ONO3 (Fig. 5c), the plot of Δ17ONO3 versus δ15NNO3 was not elucidated. In addition, the slope of Δ17ONO3-δ18ONO3 linear regressive model was <1, and δ18ONO3 could magnify the difference between abiotic and biogenic signatures for possible martian biomarker detection.
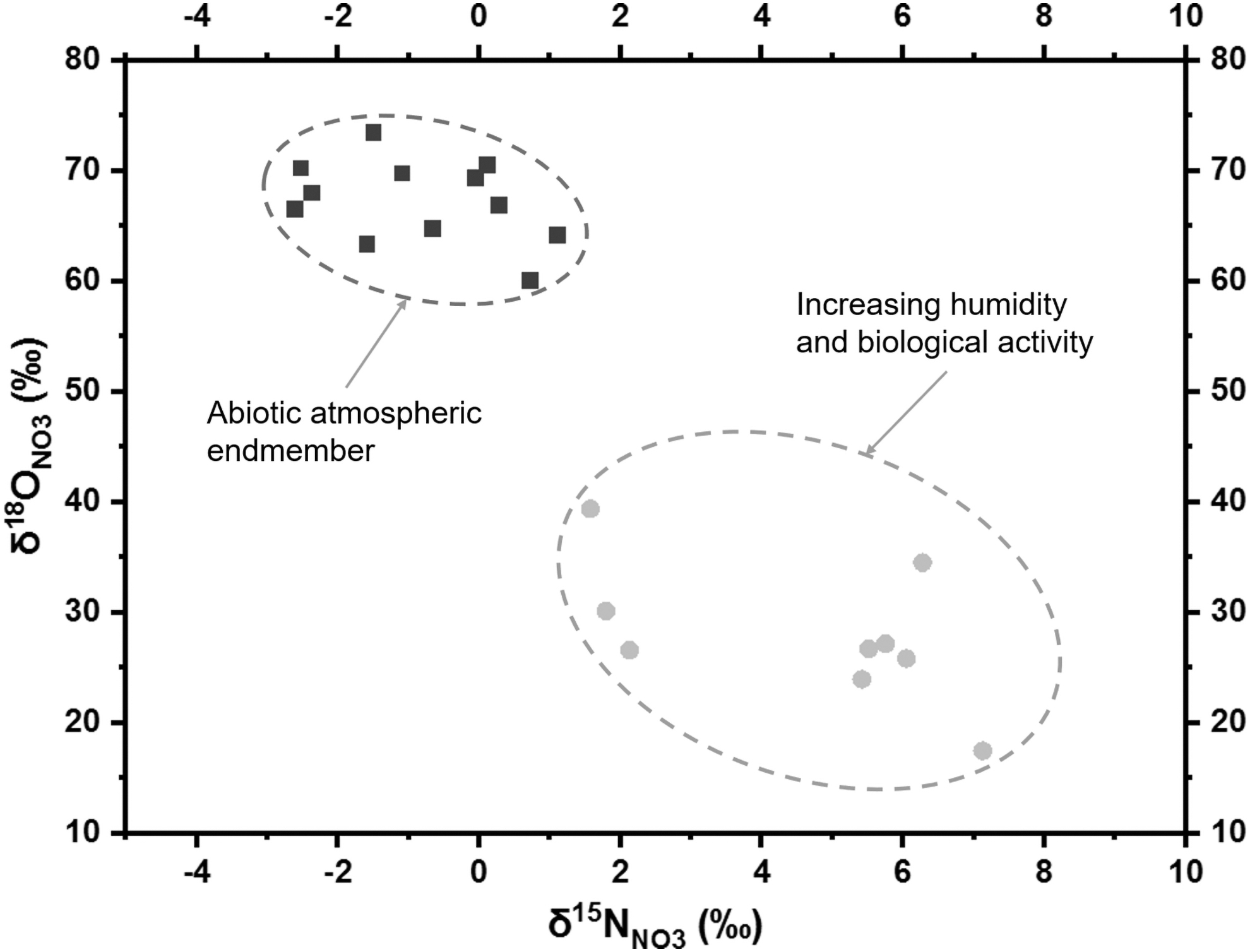
Differentiation between the abiotic signature of atmospheric endmember and the biogenic signature with increasing annual precipitation based on δ18ONO3 versus δ15NNO3 plot.NO3
4.2. Corresponding biosignatures in organic matter
δ13C provides an additional tool in studying microbial carbon and nutrient cycling. δ13C values in organic carbon largely reflect the dominant type of autotroph and associated carbon dioxide (CO2) fixation pathway in the ecosystem, namely photosynthetic bacteria, heterotrophic bacteria, algae, three-carbon (C3) plants, four-carbon (C4) plants, and Crassulacean acid metabolism (CAM) plants. In the Atacama Desert, most plants that inhabit sandy soils in the south are C4 grass plants (Quade et al., 2007). The C4 grass plants have higher δ13Corg values than C3 and CAM plants and various bacteria, with a mean value of −12‰ (Hamilton and Lewis, 1992; Schidlowski, 2001; Zerkle et al., 2005). Besides these plants, autotrophic microorganisms could also have been an important supply of organic matter to the sediments. Accordingly, our δ13C results suggest that organic matter in Atacama soils was largely derived from local microbial sources (Ewing et al., 2008; Perez-Fodich et al., 2014) with a δ13Corg of −27.1 ± 1.0‰ (Table 1 and Fig. 4b). In the arid sites, δ13Corg values of −22.7 ± 0.6‰ suggest a mixture of carbon from terrestrial C4 plants and microbial biomass (Muller, 1977; Prahl et al., 1994; Meyers and Doose, 1999; Melchiorre et al., 2018) (Fig. 4a–c).
Microbial organic carbon can be synthesized or utilized in different N cycling processes. For instance, (1) nitrifying bacteria usually utilize CO2 as the C source for growth (Marsh et al., 2005). Since atmospheric CO2 has much higher δ13C values (−7‰ to −6‰) than local microbial sources (Hare et al., 2018), δ13Corg tends to increase as the nitrification activity increases. (2) Denitrifying bacteria utilize their own organic C as the electron donor (Carlson and Ingraham, 1983). As microorganisms prefer to process lighter organic C isotopes, soil δ13Corg again tends to rise with the increasing microbial abundance and denitrification activity (Knief et al., 2020). Hence, increasing δ13Corg in our samples from the hyperarid to arid sites suggests corresponding increases in the nitrification and denitrification activity (Fig. 4b), which was also supported by the significant correlations between δ15NNO3 and δ13Corg (ρ = 0.71**, p = 0.001, Fig. 5e) as well as TOC (ρ = 0.61**, p = 0.008).
In contrast to the δ13Corg values that largely reflect the original sources of organic carbon, δ15Norg was more sensitive to recent microbiological activity, as shown by trends in δ15Norg versus C/N ratios and δ13Corg in transitional site TZ-4 (Fig. 4e, f). Given the absence of appreciable ammonium in Atacama soils, organic N would be dominantly generated via nitrate assimilation and nitrogen fixation. However, the proportion of nitrogenase was very low in these soils (Fig. 7 and Table 3), and nitrogen fixation only causes ±2‰ fractionations in δ15N (Zerkle et al., 2008; Nishizawa et al., 2014; Stueken et al., 2016), as opposed to the >3.5‰ δ15Norg values we measured. Therefore, most of the organic N in Atacama soils was likely formed via nitrate assimilation (Shen et al., 2019).
Despite the high nitrate concentrations in these soils, lack of rainfall leads to low overall N bioavailability, and thus, fractionation effects produced during nitrate uptake and other N transformations are expected to be much lower, due to complete consumption (Supplementary Fig. S1a, b). In addition, since NO2 − in the hyperarid sites could only be produced as a by-product of assimilatory nitrate reduction, increasing levels of assimilatory nitrate reduction at PONR and Yungay sites that received the most rainfall should result in a corresponding increase in NO2 − concentrations. As NO2 − availability became higher in hyperarid sites, the resulting δ15Norg did not decrease by >1‰ (Table 1 and Supplementary Fig. S1d). Thus, the observed 15ɛ of nitrate assimilation was negligible, although it could decrease when nitrate is solubilized during rainfall.
Intriguingly, despite the small to negligible 15ɛ we infer for nitrate assimilation, the δ15Norg values were consistently significantly higher than δ15NNO3 (standardized Wilcoxon signed rank statistic = 4.02***, p < 0.001) (Tables 1, 2, and Fig. 5d), implying that additional pathways and enzymes were involved in organic N transformations. Metagenomic analyses indicate the presence of ammonia lyases and nitric oxide synthase (Table 3), which transform organic N into ammonia and nitric oxide, respectively. Ammonia could then be oxidized to hydroxylamine that could be further oxidized to nitric oxide or NO2 − by hydroxylamine dehydrogenase. Subsequently, NO2 − would be denitrified to N2 through NO2 − reductase, nitric oxide reductase, and N2O reductase (Fig. 7 and Table 3). These processes assembled form the complete pathway of nitrifier denitrification (Fig. 6). The intermediate product, nitric oxide, could be reduced to N2, oxidized to nitrate by nitric oxide dioxygenase, or diffused to the atmosphere. The metagenomic analyses further suggest that nitrifier denitrification and nitric oxide dioxygenase-driven alternative nitrifications increased when TON and δ15Norg increased (Figs. 4d and 7, and Table 3) (Knief et al., 2020).
Since organic N was mostly produced by nitrate assimilation with minimal 15ɛ, the baseline δ15Norg value should be similar to the residual δ15NNO3 after denitrification. We observed a fairly constant difference of 5.2 ± 1.7‰ between δ15Norg and δ15NNO3 values among all sites (Table 1 and Table 2), which indicated a similar expression level of these alternative microbial organic N transformations. For example, the TZ-0 samples had the largest difference between δ15Norg and δ15NNO3 (Table 2), because site TZ-0 had the most dominant nitrifier denitrification compared with other sites (Fig. 6), indicating that organic N was fractionated in δ15N during conversion to gaseous N2. TZ-4, on the contrary, had the smallest difference between δ15Norg and δ15NNO3, probably due to the inhibition of nitrifier denitrification or alternative nitrification pathways by its extremely high nitrate concentrations (Fig. 3a).
4.3. N cycling models
Based on these coupled geochemical and metagenomic data, we suggest three separate models for nitrogen cycling in Atacama soils, as summarized in Fig. 9. Denitrification was almost absent in all four hyperarid sites (Fig. 9a, b), but organic N alterations catalyzed by ammonia lyases and nitric oxide synthase resulted in an increase in δ15Norg over background nitrate isotope values. The volatilization of ammonia and nitric oxide is a driver for increasingly heavy δ15Norg values, despite the minimal fractionation effect inferred for ammonia lyase-driven mineralization (Supplementary Fig. S1c). Based on the difference between δ15Norg and δ15NNO3 values in the more northern hyperarid sites (MES, PONR, and Yungay), we estimate 15ɛ of 3.5‰ to 6.5‰ for these processes (Table 2). In the southern hyperarid site (TZ-0), nitrifier denitrification is an additional organic N loss pathway. Assuming the intensity of ammonia lyase- and nitric oxide synthase-driven organic N loss in TZ-0 was similar to other hyperarid sites, the inferred 15ɛ of nitrifier denitrification was estimated to range from 2‰ to 6‰ (Fig. 9b).
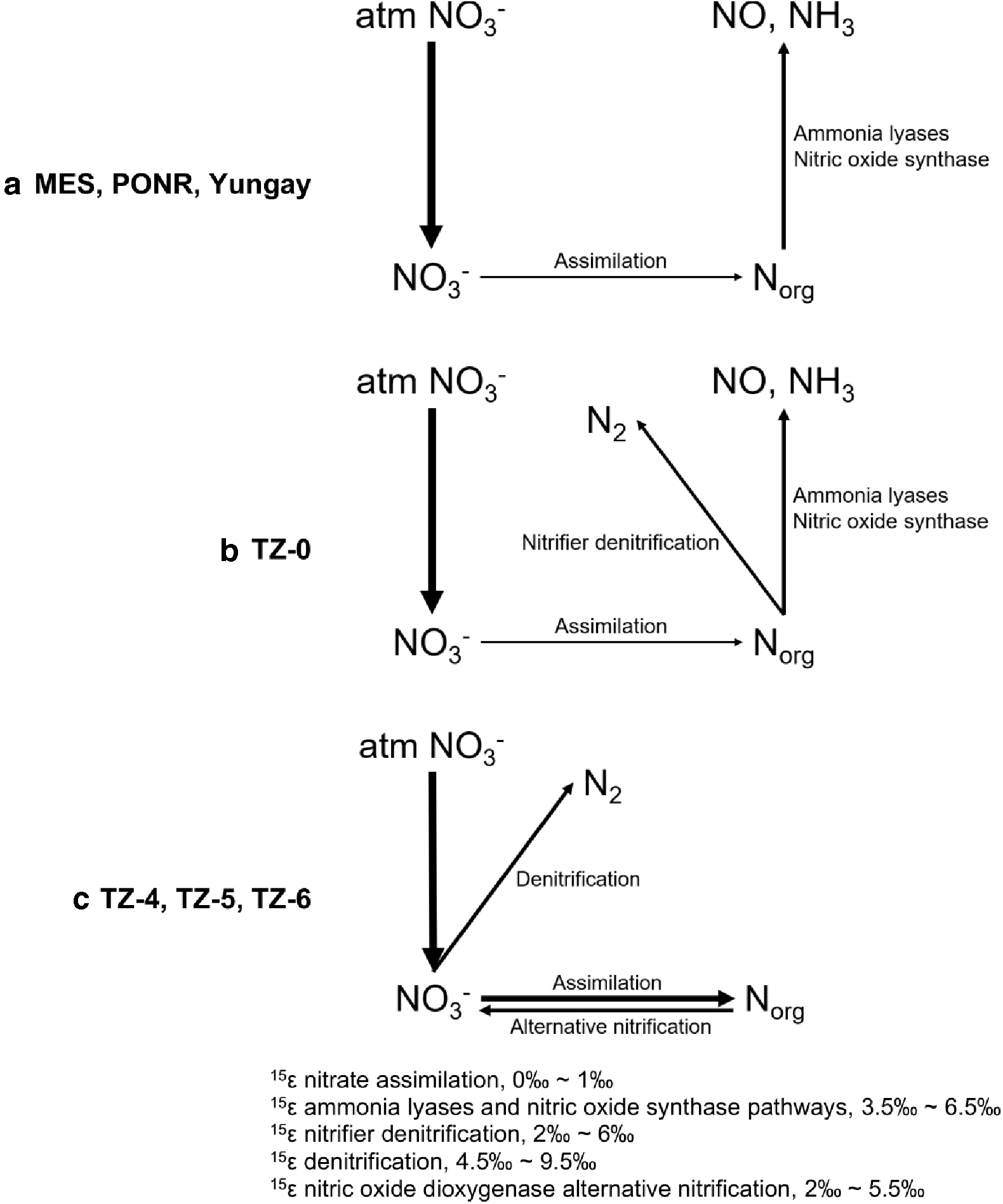
N cycling models and inferred isotopic enrichment values 15ɛ in
Within the transitional and arid sites, nitrate assimilation, denitrification, and alternative nitrification reached a steady state of biological exchange between O atoms of nitrate and ambient water molecules as supported by the equilibrium δ18ONO3 and Δ17ONO3 values (Fig. 5a, b). O isotope fractionations in nitrate δ18O of up to 30–40‰ approach equilibrium values set by that of local water sources (Michalski et al., 2012). The increasing δ15NNO3 in these soils primarily reflected the activity of denitrification. Comparing the δ15NNO3 in the transitional-arid sites and in the hyperarid sites, the inferred 15ɛ of denitrification was from 4.5‰ to 9.5‰.
Nitrifier denitrification by nitrifying bacteria in the transitional and arid sites was shifted to alternative nitrification pathways (Wrage et al., 2001; Zhu et al., 2013) (Fig. 6). The products of ammonia lyases and nitric oxide synthase would also be more actively involved in the intracellular alternative nitrification pathways (Fig. 9c). Therefore, the increase in δ15Norg in these sites can be attributed primarily to alternative nitrification pathways with an inferred 15ɛ of 2‰ to 5.5‰ (Michalski et al., 2012). Furthermore, nitrogen cycling was heterogeneous in each sampling site. For example, assimilatory nitrate reduction was stronger in PONR and Yungay, with higher NO2 − concentrations than in the other hyperarid sites; TZ-4 had slightly lower expression of alternative nitrification pathways than TZ-5 and TZ-6, possibly due to product inhibition; and TZ-5 might have a lower microbial denitrification activity and/or a higher alternative nitrification activity, in comparison.
To move the findings of this study forward, we may apply our determined nitrate biosignatures in the future martian soil analysis interpretation. On early Mars (Noachian and early Hesperian Periods), life may have thrived if liquid water was abundant as evidenced by the trace of outflow channels, ocean basins, deltaic deposits, and fan-shaped aprons (Irwin et al., 2002; Malin and Edgett, 2003; Carr and Head, 2010; Di Achille and Hynek, 2010; Hynek et al., 2010). Microbial life that could cycle nitrate might exist and perform denitrification- and nitrification-like processes, leaving traceable elevated δ15NNO3 and altered δ18ONO3 markers that had been buried in deep subsurface since the Hesperian Period.
As for the more recent hyperarid Mars, the formation of a subsurface thin layer of liquid water can be promoted by diurnal cycles (Anderson and Tice, 1979; Martin-Torres et al., 2015) and can be retained by hydrated minerals (Davila et al., 2010; Meslin et al., 2013). In addition, a recent radar survey has detected an appreciable subglacial liquid water repository on the current Mars (Orosei et al., 2018). Thus, if these water sources could support emergent martian microorganisms that cycled essential elements, they would likely consume the abundant nitrate as nutrient (Shen et al., 2019) and could potentially produce detectable geochemical biomarkers. Although this study suggests that the hyperarid core of the Atacama Desert preserves the abiotic atmospheric nitrate signature, putative microorganisms on Mars could perform nitrate/water O exchange by nitrate assimilation when ambient moisture rises to a suitable level for this metabolism. This O exchange activity could then shift nitrate O isotope ratios toward those of water.
It is noteworthy that enzymes and pathways discussed in this article are identified via the database-based metagenome function inference method. No matter how robust and valid the PICRUSt2 inference algorithm is, further transcriptomic, proteomic, or metabolomic analyses (Haider and Pal, 2013; Seyler et al., 2020) could be conducted in the future and optimize the arguments made in this study.
5. Conclusions
Nitrate and organic matter in Atacama soils are broadly affected by an assortment of microbial N dynamics, abiotic processes, and the intensity of different enzyme expressions, all of which are manifested in their C, N, and O stable isotope records. Although we have documented some microbial activity, nitrates in the hyperarid sites with an annual precipitation of <10 mm have δ15N, δ18O, and Δ17O values that largely reflect their atmospheric source, with little biological influence.
Stable isotope signatures of nitrates from the transitional and arid sites with >15 mm annual precipitation are overprinted by biologically driven enrichment in δ15NNO3 and nitrate/water equilibrium in δ18ONO3 and Δ17ONO3. Increasing δ15NNO3 in transitional and arid sites primarily reflects enhanced denitrification. Nitrate assimilation with seemingly minimal stable isotope effects is the only nitrate consumption pathway in the hyperarid sites, while ammonia lyase- and nitric oxide synthase-driven organic N loss leads to an increase in hyperarid δ15Norg. Nitrifier denitrification further increases the δ15Norg values in the southernmost hyperarid site, but nitric oxide dioxygenase-driven alternative nitrification pathways dominate the transitional and arid sites. Notably, stable isotope fractionations expressed by these N transformations are all at the low end of what has previously been proposed for these processes, due to the low bioavailability of N sources.
We have demonstrated that the effects of water and microbial activity impart measurable differences in proxies (e.g., δ18ONO3 or Δ17ONO3 vs. δ15NNO3, δ13Corg or δ15Norg vs. Corg:Norg), across a gradient of habitability in desert soils. Soils in the driest regions reflect predominantly an abiotic atmospheric endmember, but in themselves do not constitute an antibiosignature. However, we interpret the spread in isotopic values away from the atmospheric endmember as a clear biosignature for enhanced microbial processing in wetter (although still extremely arid) soils, demonstrating that relatively simple geochemical measurements (which could be performed robotically) distinguish biogenic processing in dry soils as they move through various regimens toward full biogeochemical nitrogen cycling. We speculate that extinct or potentially extant martian microorganisms may consume atmospheric nitrate via assimilation, and that isotopic variation in nitrates and organic matter could be biosignatures in martian soils indicating where the moisture content is (or was) intermittently relatively high.
Footnotes
Data Availability
The raw sequencing data for enzyme and functional pathway inferences in this study are stored in the National Center for Biotechnology Information (NCBI) under the BioProject ID PRJNA595740.
Acknowledgments
We thank Adam Wyness from Scottish Oceans Institute, School of Biology, University of St Andrews, St Andrews KY16 8LB, UK, and the Department of Zoology and Entomology, Rhodes University, Grahamstown 6139, South Africa, for assisting the soil DNA extractions. We also thank Jan Kaiser and Sarah Wexler from Science Analytical Facility, University of East Anglia, Norwich NR4 7TJ, UK, for nitrate stable isotope measurements.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was funded by the European Research Council (ERC) under the European Union's Horizon 2020 Research and Innovation Programme (grant agreement 678812) (to M.W.C.). J.S. also acknowledges support from the China Scholarship Council (CSC).
Supplementary Material
Supplementary Figure S1
Abbreviations Used
Associate Editor: Victor Parro
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
